Abstract
Background
Among subfertile couples undergoing assisted reproductive technology (ART), pregnancy rates following frozen‐thawed embryo transfer (FET) treatment cycles have historically been found to be lower than following embryo transfer undertaken two to five days following oocyte retrieval. Nevertheless, FET increases the cumulative pregnancy rate, reduces cost, is relatively simple to undertake and can be accomplished in a shorter time period than repeated in vitro fertilisation (IVF) or intracytoplasmic sperm injection (ICSI) cycles with fresh embryo transfer. FET is performed using different cycle regimens: spontaneous ovulatory (natural) cycles; cycles in which the endometrium is artificially prepared by oestrogen and progesterone hormones, commonly known as hormone therapy (HT) FET cycles; and cycles in which ovulation is induced by drugs (ovulation induction FET cycles). HT can be used with or without a gonadotrophin releasing hormone agonist (GnRHa). This is an update of a Cochrane review; the first version was published in 2008.
Objectives
To compare the effectiveness and safety of natural cycle FET, HT cycle FET and ovulation induction cycle FET, and compare subtypes of these regimens.
Search methods
On 13 December 2016 we searched databases including Cochrane Gynaecology and Fertility's Specialised Register, CENTRAL, MEDLINE, Embase, PsycINFO and CINAHL. Other search sources were trials registers and reference lists of included studies.
Selection criteria
We included randomized controlled trials (RCTs) comparing the various cycle regimens and different methods used to prepare the endometrium during FET.
Data collection and analysis
We used standard methodological procedures recommended by Cochrane. Our primary outcomes were live birth rates and miscarriage.
Main results
We included 18 RCTs comparing different cycle regimens for FET in 3815 women. The quality of the evidence was low or very low. The main limitations were failure to report important clinical outcomes, poor reporting of study methods and imprecision due to low event rates. We found no data specific to non‐ovulatory women.
1. Natural cycle FET comparisons
Natural cycle FET versus HT FET
No study reported live birth rates, miscarriage or ongoing pregnancy.
There was no evidence of a difference in multiple pregnancy rates between women in natural cycles and those in HT FET cycle (odds ratio (OR) 2.48, 95% confidence interval (CI) 0.09 to 68.14, 1 RCT, n = 21, very low‐quality evidence).
Natural cycle FET versus HT plus GnRHa suppression
There was no evidence of a difference in rates of live birth (OR 0.77, 95% CI 0.39 to 1.53, 1 RCT, n = 159, low‐quality evidence) or multiple pregnancy (OR 0.58, 95% CI 0.13 to 2.50, 1 RCT, n = 159, low‐quality evidence) between women who had natural cycle FET and those who had HT FET cycles with GnRHa suppression. No study reported miscarriage or ongoing pregnancy.
Natural cycle FET versus modified natural cycle FET (human chorionic gonadotrophin (HCG) trigger)
There was no evidence of a difference in rates of live birth (OR 0.55, 95% CI 0.16 to 1.93, 1 RCT, n = 60, very low‐quality evidence) or miscarriage (OR 0.20, 95% CI 0.01 to 4.13, 1 RCT, n = 168, very low‐quality evidence) between women in natural cycles and women in natural cycles with HCG trigger. However, very low‐quality evidence suggested that women in natural cycles (without HCG trigger) may have higher ongoing pregnancy rates (OR 2.44, 95% CI 1.03 to 5.76, 1 RCT, n = 168). There were no data on multiple pregnancy.
2. Modified natural cycle FET comparisons
Modified natural cycle FET (HCG trigger) versus HT FET
There was no evidence of a difference in rates of live birth (OR 1.34, 95% CI 0.88 to 2.05, 1 RCT, n = 959, low‐quality evidence) or ongoing pregnancy (OR 1.21, 95% CI 0.80 to 1.83, 1 RCT, n = 959, low‐quality evidence) between women in modified natural cycles and those who received HT. There were no data on miscarriage or multiple pregnancy.
Modified natural cycle FET (HCG trigger) versus HT plus GnRHa suppression
There was no evidence of a difference between the two groups in rates of live birth (OR 1.11, 95% CI 0.66 to 1.87, 1 RCT, n = 236, low‐quality evidence) or miscarriage (OR 0.74, 95% CI 0.25 to 2.19, 1 RCT, n = 236, low‐quality evidence) rates. There were no data on ongoing pregnancy or multiple pregnancy.
3. HT FET comparisons
HT FET versus HT plus GnRHa suppression
HT alone was associated with a lower live birth rate than HT with GnRHa suppression (OR 0.10, 95% CI 0.04 to 0.30, 1 RCT, n = 75, low‐quality evidence). There was no evidence of a difference between the groups in either miscarriage (OR 0.64, 95% CI 0.37 to 1.12, 6 RCTs, n = 991, I2 = 0%, low‐quality evidence) or ongoing pregnancy (OR 1.72, 95% CI 0.61 to 4.85, 1 RCT, n = 106, very low‐quality evidence).
There were no data on multiple pregnancy.
4. Comparison of subtypes of ovulation induction FET
Human menopausal gonadotrophin(HMG) versus clomiphene plus HMG
HMG alone was associated with a higher live birth rate than clomiphene combined with HMG (OR 2.49, 95% CI 1.07 to 5.80, 1 RCT, n = 209, very low‐quality evidence). There was no evidence of a difference between the groups in either miscarriage (OR 1.33, 95% CI 0.35 to 5.09,1 RCT, n = 209, very low‐quality evidence) or multiple pregnancy (OR 1.41, 95% CI 0.31 to 6.48, 1 RCT, n = 209, very low‐quality evidence).
There were no data on ongoing pregnancy.
Authors' conclusions
This review did not find sufficient evidence to support the use of one cycle regimen in preference to another in preparation for FET in subfertile women with regular ovulatory cycles. The most common modalities for FET are natural cycle with or without HCG trigger or endometrial preparation with HT, with or without GnRHa suppression. We identified only four direct comparisons of these two modalities and there was insufficient evidence to support the use of either one in preference to the other.
Plain language summary
Cycle regimens for frozen‐thawed embryo transfer
Review question: Cochrane review authors investigated different regimens used for preparing the uterus (womb) for transferring frozen‐thawed embryos to the uterus in women undergoing assisted reproductive technology (ART) to become pregnant.
Background
In subfertile women undergoing ART, eggs are collected from the ovaries and fertilised by sperm in a laboratory (in vitro fertilisation or IVF). Some or all embryos may be frozen, to be thawed and transferred to the womb at a later stage. This is called frozen‐thawed embryo transfer (or FET).
Women with regular spontaneous periods (menstrual cycles) may be offered a range of cycle regimens to prepare the womb lining for FET. Alternatively, FET can be carried out after spontaneous ovulation (release of an egg) in a natural cycle. This is called natural cycle FET.
Women with irregular cycles are either not ovulating or are ovulating randomly. Therefore, natural cycle FET is not suitable for them. These women can be offered either ovulation induction with fertility drugs or hormone therapy (HT) to prepare them for FET.
The most common regimens for FET are natural cycle with or without HCG (human chorionic gonadrotophin) trigger, or endometrial preparation with HT with or without a gonadotrophin‐releasing hormone agonist (GnRHa) to temporarily suppress ovarian function.
We conducted this review to find out if a particular FET regimen is more effective or safer than others. Our main outcomes were live birth rates and miscarriage rates per woman.
Study characteristics
We included 18 randomized controlled trials with 3815 women. The trials were conducted in Belgium, France, Israel, Italy, Iran, Singapore, the Netherlands and the UK. The evidence is current to 2 December 2016.
Key results
This review did not find sufficient evidence to support the use of one cycle regimen in preference to another in preparation for FET in subfertile women with regular ovulatory cycles. The most common modalities for FET are natural cycle with or without HCG trigger or endometrial preparation with HT, with or without GnRHa suppression. We identified only four direct comparisons of these two modalities and there was insufficient evidence to support the use of either one in preference to the other. We found no evidence specific to non‐ovulatory women.
Quality of the evidence
The evidence was of low or very low quality. The main limitations were failure to report important clinical outcomes, poor reporting of study methods and unclear findings due to lack of data.
Summary of findings
Summary of findings for the main comparison. Natural cycle FET versus HT FET.
| Natural cycle FET versus HT FET | ||||||
| Population: subfertile women Settings: assisted reproductive technology clinics Intervention: natural cycle FET Comparison: HT FET | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Assumed risk | Corresponding risk | |||||
| HT FET | Natural cycle FET | |||||
| Live birth rate per woman | No data available | Not estimable | ‐ | |||
| Miscarriage rate per woman | No data available | Not estimable | ‐ | |||
| Ongoing pregnancy rate per woman | No data available | Not estimable | ‐ | |||
| Multiple pregnancy rate per woman | See comment | OR 2.48 (0.09 to 68.14) | 21 (1 study) | ⊕⊝⊝⊝ very low1,2 | No events in the control group | |
| *The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; FET: frozen‐thawed embryo transfer; HT: hormone therapy; OR: odds ratio | ||||||
| GRADE Working Group grades of evidence High quality: We are very confident that the true effect lies close to that of the estimate of the effect Moderate quality: We are moderately confident in the effect estimate: The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different Low quality: Our confidence in the effect estimate is limited: The true effect may be substantially different from the estimate of the effect Very low quality: We have very little confidence in the effect estimate: The true effect is likely to be substantially different from the estimate of effect | ||||||
1Downgraded one level for serious risk of bias: study at unclear risk of bias in all domains. 2Downgraded two levels due to very serious imprecision: single study, very few events. Confidence intervals compatible with benefit in either group or with no effect.
Summary of findings 2. Natural cycle FET versus HT plus GnRHa suppression FET.
| Natural cycle FET versus HT + GnRHa suppression FET | ||||||
| Population: subfertile women Settings: assisted reproductive technology clinics Comparison: HT + GnRHa FET | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Assumed risk | Corresponding risk | |||||
| HT + GnRHa FET | Natural cycle FET | |||||
| Live birth rate per woman | 316 per 1000 | 262 per 1000 (153 to 414) | OR 0.77 (0.39 to 1.53) | 159 (1 study) | ⊕⊕⊝⊝ low1 | Only 46 events |
| Miscarriage rate per woman | No data available | Not estimable | ‐ | |||
| Ongoing pregnancy rate per woman | No data available | Not estimable | ‐ | |||
| Multiple pregnancy rate per woman | 63 per 1000 | 38 per 1000 (9 to 144) | OR 0.58 (0.13 to 2.50) | 159 (1 study) | ⊕⊕⊝⊝ low1 | Only 8 events |
| *The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; FET: frozen‐thawed embryo transfer; GnRHA: gonadotrophin‐releasing hormone agonist; HT: hormone therapy; OR: odds ratio | ||||||
| GRADE Working Group grades of evidence High quality: We are very confident that the true effect lies close to that of the estimate of the effect Moderate quality: We are moderately confident in the effect estimate: The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different Low quality: Our confidence in the effect estimate is limited: The true effect may be substantially different from the estimate of the effect Very low quality: We have very little confidence in the effect estimate: The true effect is likely to be substantially different from the estimate of effect | ||||||
1Downgraded two levels due to very serious imprecision: single study, few events, confidence interval compatible with benefit in either group or with no effect.
Summary of findings 3. Natural cycle FET versus modified natural cycle FET (HCG trigger).
| Natural cycle FET versus other regimens for primary or secondary subfertility | ||||||
| Population: subfertile women Settings: assisted reproductive technology clinics Intervention: natural cycle FET Comparison: natural cycle plus HCG trigger FET1 | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Assumed risk | Corresponding risk | |||||
| Modified natural cycle FET (HCG trigger) | Natural cycle FET | |||||
| Live birth rate per woman | 267 per 1000 | 167 per 1000 (55 to 413) | OR 0.55 (0.16 to 1.93) | 60 (1 study) | ⊕⊝⊝⊝ Very low2,3 | Only 13 events |
| Miscarriage rate per woman | 24 per 1000 | 5 per 1000 (0 to 92) | OR 0.20 (0.01 to 4.13) | 168 (1 study) | ⊕⊝⊝⊝ Very low2,4 | Only 2 events |
| Ongoing pregnancy rate per woman | 107 per 1000 | 226 per 1000 (110 to 408) | OR 2.44 (1.03 to 5.76) | 168 (1 study) | ⊕⊝⊝⊝ Very low2,4 | Only 28 events |
| Multiple pregnancy per woman | No data available | |||||
| *The basis for the assumed risk is the mean control group risk across studies. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; FET: frozen‐thawed embryo transfer; GnRHa: gonadotrophin releasing hormone agonist;HCG: human chorionic gonadotrophin; HT: hormone therapy; OR: odds ratio. | ||||||
| GRADE Working Group grades of evidence High quality: We are very confident that the true effect lies close to that of the estimate of the effect Moderate quality: We are moderately confident in the effect estimate: The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different Low quality: Our confidence in the effect estimate is limited: The true effect may be substantially different from the estimate of the effect Very low quality: We have very little confidence in the effect estimate: The true effect is likely to be substantially different from the estimate of effect | ||||||
1One other study compared natural cycle FET versus natural cycle plus human menopausal gonadotrophin, but did not report any per‐woman data. 2Downgraded two levels due to very serious imprecision: single study, few events, confidence interval compatible with benefit in the modified natural cycle only or with no effect. 3Downgraded one level due to serious risk of bias: high attrition rate, baseline characteristics unequal. 4Downgraded one level due to serious risk of bias: no allocation concealment.
Summary of findings 4. Modified natural cycle FET (HCG trigger) versus HT FET.
| Modified natural cycle FET (HCG trigger) versus HT FET | ||||||
| Population: subfertile women Settings: assisted reproductive technology clinics Intervention: modified natural cycle FET (HCG trigger) Comparison: HT FET | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Assumed risk | Corresponding risk | |||||
| HT FET | Modified natural cycle FET (HCG trigger) | |||||
| Live birth rate per woman | 88 per 1000 | 114 per 1000 (78 to 165) | OR 1.34 (0.88 to 2.05) | 959 (1 study) | ⊕⊕⊝⊝ low1,2 | |
| Miscarriage rate per woman | No data available | Not estimable | ‐ | |||
| Ongoing pregnancy rate per woman | 97 per 1000 | 115 per 1000 (79 to 164) | OR 1.21 (0.80 to 1.83) | 959 (1 study) | ⊕⊕⊝⊝ low1,2 | |
| Multiple pregnancy rate per woman | No data available | Not estimable | ‐ | |||
| *The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; FET: frozen‐thawed embryo transfer;HCG: human chorionic gonadotrophin; HT: hormone therapy; OR: odds ratio. | ||||||
| GRADE Working Group grades of evidence High quality: We are very confident that the true effect lies close to that of the estimate of the effect Moderate quality: We are moderately confident in the effect estimate: The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different Low quality: Our confidence in the effect estimate is limited: The true effect may be substantially different from the estimate of the effect Very low quality: We have very little confidence in the effect estimate: The true effect is likely to be substantially different from the estimate of effect | ||||||
1Downgraded one level due to serious risk of bias: high attrition rate, unclear risk of allocation concealment 2Downgraded one level due to serious imprecision: confidence intervals compatible with benefit in either group or with no effect
Summary of findings 5. Modified natural cycle FET (HCG trigger) versus HT + GnRHa suppression FET.
| Modified natural cycle FET (HCG trigger) versus HT + GnRHa FET | ||||||
| Population: subfertile women Settings: assisted reproductive technology clinics Intervention: modified natural cycle FET (HCG trigger) Comparison: HT + GnRHa FET | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Assumed risk | Corresponding risk | |||||
| HT + GnRHa FET | Modified natural cycle FET (HCG trigger) | |||||
| Live birth rate per woman | 398 per 1000 | 423 per 1000 (304 to 553) | OR 1.11 (0.66 to 1.87) | 236 (1 study) | ⊕⊕⊝⊝ low1,2 | |
| Miscarriage rate per woman | 68 per 1000 | 51 per 1000 (18 to 138) | OR 0.74 (0.25 to 2.19) | 236 (1 study) | ⊕⊕⊝⊝ low1,2 | |
| Ongoing pregnancy rate | No data available | Not estimable | ‐ | |||
| Multiple pregnancy rate per woman | No data available | Not estimable | ‐ | |||
| *The basis for the assumed risk is the mean control group risk across studies. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; FET: frozen‐thawed embryo transfer; GnRHa: gonadotrophin releasing hormone agonist;HCG: human chorionic gonadotrophin; HT: hormone therapy; OR: odds ratio. | ||||||
| GRADE Working Group grades of evidence High quality: We are very confident that the true effect lies close to that of the estimate of the effect Moderate quality: We are moderately confident in the effect estimate: The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different Low quality: Our confidence in the effect estimate is limited: The true effect may be substantially different from the estimate of the effect Very low quality: We have very little confidence in the effect estimate: The true effect is likely to be substantially different from the estimate of effect | ||||||
1Downgraded one level due to serious risk of bias: study at unclear risk of in most domains of bias (allocation concealment, blinding, selective reporting and other sources of bias). 2Downgraded one level due to serious imprecision: confidence intervals compatible with benefit in either group or with no effect.
Summary of findings 6. HT FET versus HT + GnRHa FET.
| HT FET versus other regimens for primary or secondary subfertility | ||||||
| Population: women with primary or secondary subfertility Settings: assisted reproductive technology clinics Intervention: HT FET Comparison: HT plus GnRHa trigger | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Assumed risk | Corresponding risk | |||||
| HT + GnRHa FET | HT FET | |||||
| Live birth rate per woman | 742 per 1000 | 223 per 1000 (103 to 463) | OR 0.10 (0.04 to 0.30) | 75 (1 study) | ⊕⊕⊝⊝ Low1,2 | Only 33 events |
| Miscarriage rate per woman | 48 per 1000 | 31 per 1000 (18 to 53) | OR 0.64 (0.37 to 1.12) | 991 (6 studies) | ⊕⊕⊝⊝ Low3,4 | ‐ |
| Ongoing pregnancy rate per woman | 132 per 1000 | 207 per 1000 (85 to 424) | OR 1.72 (0.61 to 4.85) | 106 (1 study) | ⊕⊝⊝⊝ Very low4,5 | Only 18 events |
| Multiple pregnancy rate per woman | No data available | |||||
| *The basis for the assumed risk is the mean control group risk across studies. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; FET: frozen‐thawed embryo transfer; GnRHa: gonadotrophin releasing hormone agonist;HT: hormone therapy; OR: odds ratio | ||||||
| GRADE Working Group grades of evidence High quality: We are very confident that the true effect lies close to that of the estimate of the effect Moderate quality: We are moderately confident in the effect estimate: The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different Low quality: Our confidence in the effect estimate is limited: The true effect may be substantially different from the estimate of the effect Very low quality: We have very little confidence in the effect estimate: The true effect is likely to be substantially different from the estimate of effect | ||||||
1Downgraded one level due to serious imprecision: single study, few events. 2Downgraded one level due to serious inconsistency: clinical pregnancy rate in this study was higher than in six other studies in the same analysis (none of which reported live birth) and this study accounted for all inconsistency in the analysis for clinical pregnancy (I2 = 46%). 3Downgraded one level due to serious imprecision: confidence intervals compatible with benefit in HT‐only arm or with no effect. 4Downgraded one level due to serious risk of bias: method of allocation concealment unclear in all studies/only study. 5Downgraded two levels due to very serious imprecision: single study, few events.
Summary of findings 7. HMG FET versus clomiphene + HMG FET.
| HMG FET versus clomiphene + HMG FET | ||||||
| Population: subfertile women Settings: assisted reproductive technology clinics Intervention: HMG FET Comparison: clomiphene + HMG FET | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Assumed risk | Corresponding risk | |||||
| Clomiphene+ HMG FET | HMG FET | |||||
| Live birth rate per woman | 84 per 1000 | 186 per 1000 (89 to 347) | OR 2.49 (1.07 to 5.80) | 209 (1 study) | ⊕⊝⊝⊝ very low1,2 | Only 26 events |
| Miscarriage rate per woman | 37 per 1000 | 49 per 1000 (13 to 164) | OR 1.33 (0.35 to 5.09) | 209 (1 study) | ⊕⊝⊝⊝ very low1,3 | Only 9 events |
| Ongoing pregnancy rate per woman | No data available | Not estimable | ‐ | |||
| Multiple pregnancy rate per woman | 28 per 1000 | 39 per 1000 (9 to 157) | OR 1.41 (0.31 to 6.48) | 209 (1 study) | ⊕⊝⊝⊝ very low1,3 | Only 7 events |
| *The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; FET: frozen‐thawed embryo transfer; HMG: human menopausal gonadotrophin; HT: hormone therapy; OR: odds ratio | ||||||
| GRADE Working Group grades of evidence High quality: We are very confident that the true effect lies close to that of the estimate of the effect Moderate quality: We are moderately confident in the effect estimate: The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different Low quality: Our confidence in the effect estimate is limited: The true effect may be substantially different from the estimate of the effect Very low quality: We have very little confidence in the effect estimate: The true effect is likely to be substantially different from the estimate of effect | ||||||
1Downgraded one level for serious risk of bias: study at unclear risk of bias in all domains. 2Downgraded two levels due to very serious imprecision: single study, few events. Confidence intervals compatible with benefit in the HMG‐only group or with no clinically meaningful effect. 3Downgraded two levels due to very serious imprecision: single study, very few events. Confidence intervals compatible with benefit in either group or with no effect.
Background
Description of the condition
For subfertile couples undergoing assisted reproductive technology (ART), embryo transfer can be performed with either fresh or frozen‐thawed embryos. Pregnancy rates following frozen‐thawed embryo transfer (FET) treatment cycles have historically been found to be lower than following fresh embryo transfer. Nevertheless, FET increases the cumulative pregnancy rate, reduces cost and is relatively simple to undertake. Moreover, it can be accomplished in a shorter time period than repeated in vitro fertilisation (IVF) or intracytoplasmic sperm injection (ICSI) cycles accompanied by fresh embryo transfer.
Description of the intervention
FET has been carried out using a variety of cycle regimens; spontaneous ovulatory cycles (natural cycle), cycles in which the endometrium is artificially prepared by oestrogen and progesterone hormones and is called hormone therapy (HT) cycle, and cycles in which ovulation is induced by drugs (ovulation induction cycle).
For FET to be successful the age of the embryos after thawing has to be synchronised with the age of the endometrium on the day of embryo transfer. Thus, endometrial receptivity and synchronisation between embryonic and endometrial development are important factors in the process of embryo implantation.
Natural cycle
Performing FET in a monitored natural cycle has the advantage that no medications are used, making such cycles preferable to many women. It is only feasible for women with regular ovulatory cycles, however, even in women with regular menstrual cycles, ovulation may not always occur and the timing of FET can also be problematic. Monitoring of the cycle requires several pelvic ultrasound scans to confirm follicular development and to time the commencement of urine testing for detection of the luteinising hormone (LH) surge. In addition, a further scan for ultrasonic evidence of ovulation may be required. Spontaneous endometrial development in the follicular phase is affected by age, and this may be a contributing factor in the lower pregnancy rate in older women (Sher 1991). To avoid some of these problems, ovulation induction agents, such as tamoxifen, clomiphene citrate, gonadotrophins or a combination of clomiphene and gonadotrophins have been employed (Mandelbaum 1987). However, even with their use, cycle cancellation rates can be high.
Hormone therapy cycle (with or without gonadotrophin‐releasing hormone agonist)
An alternative method for establishing an endometrium receptive to implantation is an artificial, hormonally‐controlled cycle (HT) using sequentially administered exogenous oestrogen and progesterone. Such a regimen was first used in women without ovarian function receiving embryos derived from donor oocytes (Lutjen 1984). In women with remaining ovarian function, a gonadotrophin‐releasing hormone agonist (GnRHa) was used to temporarily suppress ovarian function and render the woman functionally agonadal prior to inducing an artificial cycle with oestrogen and progesterone. Using such a regimen, implantation and pregnancy rates in donor oocyte recipients with retained ovarian function were similar to those in recipients without ovarian function (Borini 1995; Flamigni 1993; Pados 1992). However, these cycles are more expensive, and the GnRHa can have adverse effects and delay the resumption of spontaneous ovulation if FET fails. A simplified regimen, retaining the benefits but reducing cost and adverse effects and consisting of exogenous oestrogen and progesterone only (without a GnRHa), has also been used in women with remaining ovarian function (Jaroudi 1991; Lelaidier 1992). With this approach, the initiation of orally administered exogenous oestrogen on day one of the cycle prevents follicular recruitment by suppressing follicle‐stimulating hormone (FSH) and consequently spontaneous ovulation is avoided. Estradiol implants have also been shown to suppress the hypothalamic pituitary ovarian axis resulting in adequate endometrial preparation in women with retained ovarian function (Ben‐Nun 1997).
The advantage of hormone regimens is a high level of control and flexibility in the timing of transfer. The length of the follicular phase can be varied without detriment to implantation or pregnancy rates (Leeton 1991; Navot 1989), and the cycle cancellation rate is low. However, cost is greater, particularly if a GnRHa is used. Fixed regimens of oestrogen and progesterone may be inadequate for proper endometrial development in certain circumstances. In women with functioning ovaries, the possibility has been expressed that stimulatory factors of embryonic origin (such as human chorionic gonadotrophins) could lead to the production of ovarian substances (e.g. androgens or certain peptides) with an effect on endometrial quality. If a pregnancy occurs, oestrogen and progesterone must be continued until placental autonomy is established to replace the absent corpus luteum.
Most artificial cycle protocols mimic the natural cycle, but the active substances used and routes of delivery of oestrogen and progesterone vary. Oestrogen may be administered in the form of oral tablets, transdermal patches, subcutaneous implants and vaginal rings or tablets. Progesterone may be given in the form of oral tablets, intramuscular injections and intravaginal suppositories or rings (Devroey 1998).
Ovulation induction cycle
Theoretically, ovulation induction can be offered to women with irregular or anovulatory cycles undergoing FET. The possible advantage is to induce ovulation with natural LH surge and semi‐natural endometrial development. The development of a corpus luteum with production of natural oestrogen and progesterone helps support the luteal phase and early pregnancy without the need of exogenous HT. The use of clomiphene can be associated with an anti‐oestrogenic effect on the endometrium. Other ovulation induction agents include oral anti‐oestrogens such as tamoxifen and aromatase inhibitors such as letrozole. Drawbacks of ovulation induction cycles include drug exposure and its possible adverse effects, and risks including high cancellation rate due to under or over response, the cost of gonadotrophins, the need for intense monitoring and the limited flexibility in connection with FET timing. For these reasons and in the absence of evidence supporting any benefit compared to other cycle regimens, most centres do not use ovulation induction.
How the intervention might work
All methods of endometrial preparation aim to achieve a state of the endometrium that will be receptive to replaced frozen‐thawed embryos. The natural ovulatory cycle is the most physiological method. Using oestrogen and progesterone preparations as HT aims to prepare the endometrium in two stages. The first phase is using oestrogen preparations mimicking the follicular phase of a natural cycle. Following that progesterone preparations are added to oestrogen to mimic the luteal phase. Another method is the use of clomiphene tablets or human menopausal gonadotrophins (HMG) to induce ovulation and build up the endometrium.
Why it is important to do this review
Early studies reported pregnancy rates in artificial cycles to be equivalent to those obtained after FET in spontaneous ovulatory cycles, with low cancellation rates (de Ziegler 1990; Frydman 1988; Meldrum 1989; Schmidt 1989; Troup 1991). Some subsequent studies (often containing small numbers) reported better outcomes in artificial than in natural cycles (Davies 1991; Mausher 1991), while others found no difference (Irianni 1992; Sathanandan 1991). However, concern about the adequacy of the various replacement regimens and the possibility of higher rates of early pregnancy loss has also been expressed (Lelaidier 1992). Therefore, uncertainty remains as to which type of cycle regimen is superior and many fertility clinics use a mixture of protocols for FET. The clinical effectiveness of the different approaches can only be determined by randomized controlled trials (RCTs) comparing the three cycle regimens (natural, HT and ovulation induction).
This review aimed to compare the outcome of FET in an artificial HT cycle or an ovulation‐induction cycle with that of a monitored, natural cycle. The findings may be of interest to subfertile couples embarking on ART treatment, as well as practitioners and healthcare providers offering ART. This is the first update of a review first published in 2008, which included seven RCTs (1120 women) and concluded that there was insufficient evidence to support the use of one intervention in preference to another (Ghobara 2008).
Objectives
To compare the effectiveness and safety of natural cycle FET, HT cycle FET and ovulation induction cycle FET, and compare subtypes of these regimens.
Methods
Criteria for considering studies for this review
Types of studies
We considered published and unpublished RCTs for inclusion in this review. We excluded non‐randomised studies and quasi‐randomised trials (e.g. studies with evidence of inadequate sequence generation such as alternate days, participant numbers, etc.) as they are associated with a high risk of bias.
Types of participants
Couples/women undergoing FET as treatment for primary or secondary subfertility, irrespective of the indication for embryo freezing (supernumerary embryos obtained following fresh embryo transfer, elective embryo freezing as a result of ovarian hyperstimulation syndrome or for other reasons).
The participants may be of two main groups:
women with spontaneous ovulatory cycles;
women with anovulatory cycles
Types of interventions
Trials comparing any of the following cycle regimens regardless of the route of administration or drug‐containing regimen were eligible for inclusion:
natural ovulatory FET cycle versus HT FET cycle or other FET regimens
modified natural cycle FET (e.g. natural cycle with the use of HCG trigger) versus HT FET or other FET regimens
HT FET cycle versus HT plus GnRHa suppression or other FET cycle regimen
comparison of subtypes of ovulation induction FET using clomiphene, HMG, FSH or any combination of these drugs.
We excluded trials comparing FET cycle regimens with placebo only; however, trials comparing at least two FET cycle regimens with each other and with a placebo were eligible for inclusion.
Types of outcome measures
Primary outcomes
1. Live birth rate per woman, defined as delivery of a live fetus after 24 completed weeks of gestational age.
2. Miscarriage rate per woman.
Secondary outcomes
3. Ongoing pregnancy rate per woman, defined as evidence of a gestational sac with fetal heart motion at 12 weeks, confirmed with ultrasound.
4. Clinical pregnancy rate per woman, defined as evidence of a gestational sac, confirmed by ultrasound.
5. Other adverse events (adverse events were not grouped as composite measure but were rather analyzed separately):
a. ectopic pregnancy rate per woman;
b. multiple pregnancy rate per woman.
6. Cycle cancellation rate per woman.
7. Endometrial thickness during FET cycle.
8. Number of centre visits to monitor FET cycle.
Additional outcomes not appropriate for statistical pooling
Data per cycle or per transfer are not appropriate for pooling, because of what statisticians refer to as 'unit of analysis errors'. Simple group comparison tests for categorical data require that observations are statistically independent. The use of multiple observations per participant leads to unpredictable bias in the estimate of treatment difference (Vail 2003). Where we were unable to obtain per‐woman data for any of our outcomes of interest with multiple cycles or transfers per woman, we reported data in an additional table.
Search methods for identification of studies
We searched for all published and unpublished RCTs comparing the various cycle regimens without language restriction and in consultation with Cochrane Gynaecology and Fertility's Information Specialist.
The search was performed on titles, abstracts and keywords of the listed articles. When required, we contacted the study authors to obtain additional information.
Electronic searches
We searched the following electronic databases.
Cochrane Gynaecology and Fertility (CGF) Specialised Register of Controlled Trials from inception to 13 December 2016 (Appendix 1)
The Cochrane Central Register of Sudies (CENTRAL CRSO) searched 13 December 2016 (Appendix 2)
MEDLINE from inception to 13 December 2016 (Appendix 3)
Embase from inception to 13 December 2016 (Appendix 4)
PsycINFO from inception to 13 December 2016 (Appendix 5)
CINAHL from inception to 13 December 2016 (Appendix 6)
We combined the MEDLINE search with the Cochrane Highly Sensitive Search Strategy for identifying RCTs which appears in the Cochrane Handbook of Systematic Reviews of Interventions (Chapter 6; Lefebvre 2011). The Embase, PsycINFO and CINAHL searches were combined with trial filters developed by the Scottish Intercollegiate Guidelines Network (SIGN).
We searched the following electronic sources.
-
Trials registers for ongoing and registered trials:
the World Health Organization (WHO) International Clinical Trials Registry Platform (ICTRP), searched to 13th December 2016;
ClinicalTrials.gov, searched to 13th December 2016.
DARE (Database of Abstracts of Review of Effects) in the Cochrane Library (for reference lists from relevant non‐Cochrane reviews), date of last search 13th December 2016.
Web of Science, searched to 13th December 2016.
LILACS, for trials from the Portuguese and Spanish‐speaking world, date of last search 13th December 2016.
OpenGrey for unpublished literature from Europe, searched to 13th December 2016.
PubMed and Google Scholar (for recent trials not yet indexed in MEDLINE), searched 13th December 2016.
Searching other resources
We handsearched reference lists of articles retrieved by the search and contacted experts in the field to obtain additional data. We also handsearched relevant journals and conference abstracts that are not covered in the CGF specialized register, in liaison with the Information Specialist.
The searches were conducted using the Cochrane Highly Sensitive Search Strategy as outlined in the Cochrane Handbook for Systematic Reviews of Interventions (Lefebvre 2011), and the following MeSH terms in combination:
cryopreservation
freezing
embryo
pre‐embryo
frozen or cryopreserved embryo transfer
embryo replacement
frozen or cryopreserved embryo transfer
frozen or cryopreserved embryo replacement.
We searched citation lists of relevant publications, reviews and included studies.
We searched relevant conference abstract books including the European Society of Human Reproduction and Embryology, the American Society for Reproductive Medicine, the British Fertility Society, the British Congress of Obstetrics and Gynaecology, the World Congress of In Vitro Fertilisation and Human Reproductive Genetics, and the Fertility Society of Australia in liaison with the CGF Information Specialist, and contacted we experts in the field seeking additional data.
Data collection and analysis
Selection of studies
After an initial screen of titles and abstracts retrieved by the searches, we retrieved the full texts of all potentially eligible studies. Two review authors (TG and RA) independently examined these full‐text articles for compliance with the inclusion criteria and selected studies eligible for inclusion in the review. We contacted study investigators as required, to clarify study eligibility. We resolved disagreements on study eligibility by discussion or by involving a third review author (TAG). We documented the selection process with a PRISMA flow chart (Figure 1; Moher 2009).
1.
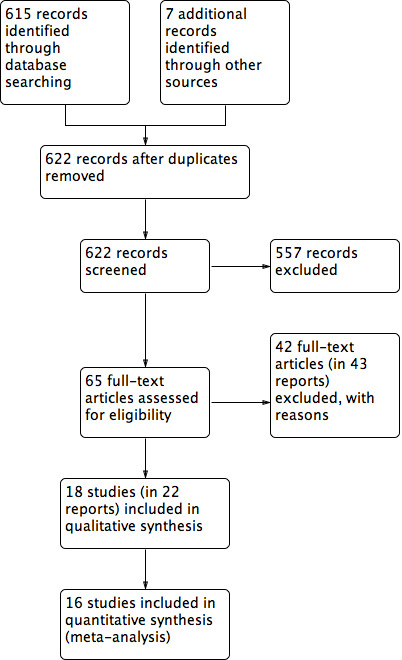
Study flow diagram
Data extraction and management
Two review authors (TG and RA) independently extracted data from eligible studies using a data extraction form designed and pilot‐tested by the review authors. We resolved any disagreements by discussion or by involving a third review author (TAG). Data extracted included study characteristics and outcome data. The review authors collated multiple reports of the same study, so that each study rather than each report was the unit of interest, and such studies were given a single study ID with multiple references.
We sent correspondence to study investigators to request further data on methods or results, or both, as required.
Assessment of risk of bias in included studies
Two review authors independently assessed the included studies for risk of bias using the Cochrane 'Risk of bias' assessment tool to assess: selection (random sequence generation and allocation concealment); performance (blinding of participants and personnel); detection (blinding of outcome assessors); attrition (incomplete outcome data); reporting (selective reporting); and other bias such as difference in participants' baseline characteristics, embryo quality, participants' number of visits and number of days until LH‐HCG surge (Higgins 2011a). We resolved disagreements by discussion or by involving a third review author. We described all judgements fully and present the conclusions, which were incorporated into the interpretation of review findings, in the 'Risk of bias' tables, and Figure 2 and Figure 3.
2.
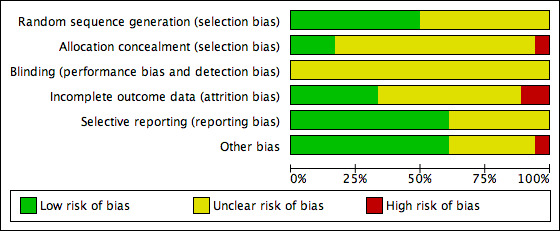
Risk of bias graph: review authors' judgements about each risk of bias item presented as percentages across all included studies
3.
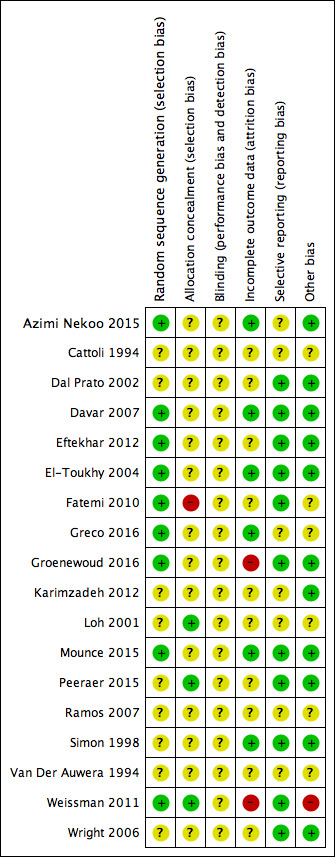
Risk of bias summary: review authors' judgements about each risk of bias item for each included study
We took care to search for within‐trial selective reporting, such as trials failing to report obvious outcomes, or reporting them in insufficient detail to allow inclusion. We sought published protocols where possible and compared the outcomes between the protocol and the final published study. Where identified studies failed to report any of the primary outcomes (e.g. live birth), but did report interim outcomes, we undertook informal assessment by comparing the interim values to those reported in studies that also reported the primary outcomes to detect whether there were any similarities between them.
Measures of treatment effect
For dichotomous data (e.g. live birth rates), we used the numbers of events in the control and intervention groups of each study to calculate Mantel‐Haenszel odds ratios (OR). For continuous data (e.g. endometrial thickness), where studies reported exactly the same outcomes we calculated mean differences (MDs) between treatment groups. We presented 95% confidence intervals (CI) for all outcomes. We compared the magnitude and direction of effect reported by studies with how they are presented in the review, taking account of legitimate differences.
Unit of analysis issues
The primary analysis was per woman randomized. We briefly summarized data that did not allow valid analysis (e.g. 'per cycle' data), entered them in additional tables and did not use them in meta‐analyses. We counted multiple live births (e.g. twins or triplets) as one live birth event. We planned to include only first‐phase data from cross‐over trials. However, we included no cross‐over trial in this review.
Where studies reported only 'per cycle' or 'per embryo transfer' data and there were multiple cycles or transfers per woman, we contacted authors of such studies and requested data 'per woman'. Where 'per woman' data could not be obtained following correspondence with the study authors, we summarized findings in additional tables as noted above.
Dealing with missing data
We analyzed the data on an intention‐to‐treat basis as far as possible and attempted to obtain missing data from the trial authors. The primary outcomes (e.g. live birth) were assumed not to have occurred in participants without a reported outcome. For other outcomes, we analyzed only the available data.
If studies reported sufficient detail to calculate MDs but no information on associated standard deviation (SD), we assumed the outcome to have an SD equal to the highest SD from other studies within the same analysis.
Assessment of heterogeneity
We assessed whether the clinical and methodological characteristics of the included studies were sufficiently similar for meta‐analysis to provide a clinically meaningful summary. We assessed statistical heterogeneity using the I2 statistic (Higgins 2003). An I2 greater than 50% indicated substantial heterogeneity (Deeks 2011).
Assessment of reporting biases
In view of the difficulty of detecting and correcting for publication bias and other reporting biases, the review authors minimised the potential impact of these biases by ensuring a comprehensive search for eligible studies and avoiding duplication of data. Where there were 10 or more studies in an analysis, we planned to use funnel plots to explore the possibility of small‐study effects (a tendency for estimates of the intervention effect to be more beneficial in smaller studies). However, we did not generate a funnel plot for any of the analyses as none of them included 10 or more studies.
Data synthesis
For studies that were sufficiently similar, we planned to combine the data using a fixed‐effect model in the following comparisons:
Natural cycle FET comparisons
Natural cycle versus HT
Natural cycle versus HT + GnRHa
Natural cycle versus HMG
Natural cycle versus modified natural cycle (HCG‐induced)
Modified natural cycle FET comparisons
Modified natural cycle (HCG induced) versus HT
Modified natural cycle versus HT + GnRHa
HT FET comparisons
HT versus HT + GnRHa
HT versus FSH
HT versus HT + HCG
Comparisons of subtypes of ovulation‐induction FET.
HMG versus clomiphene + HMG
HT plus GnRHa versus clomiphene
An increase in the odds of a particular outcome, which may be beneficial (e.g. live birth) or detrimental (e.g. adverse effects) was displayed graphically in the meta‐analyses to the right of the centre line and a decrease in the odds of an outcome to the left of the centre line.
Subgroup analysis and investigation of heterogeneity
We planned no subgroup analyses.
We took any statistical heterogeneity into account when interpreting the results, especially where there was any variation in the direction of effect.
Sensitivity analysis
We conducted sensitivity analyses for the primary outcomes to determine whether the conclusions were robust to arbitrary decisions made regarding eligibility and analysis. These analyses included consideration of whether the review conclusions would have differed if:
we had used a random‐effects (RE) model;
the summary effect measure had been risk ratio (RR) rather than OR.
Overall quality of the body of evidence: 'Summary of findings' table
We prepared 'Summary of findings' tables for the review's main comparisons:
natural cycle FET versus HT FET;
natural cycle FET versus HT plus GnRHa suppression FET;
modified natural cycle FET versus HT FET; and
modified natural cycle FET versus HT plus GnRHa suppression.
We prepared additional 'Summary of findings' tables for three other comparisons:
natural cycle FET versus modified natural cycle FET (HCG trigger);
HT FET versus HT plus GnRHa suppression; and
HMG FET versus clomiphene plus HMG FET.
We used GRADEPro GDT (GRADEPro GDT 2014) and Cochrane methods (Schünemann 2011). These tables evaluated the overall quality of the body of evidence for the main review outcomes (live birth, miscarriage, ongoing pregnancy and multiple pregnancy), using GRADE criteria (study limitations (i.e. risk of bias), consistency of effect, imprecision, indirectness and publication bias). Judgements about evidence quality (high, moderate, low or very low) were made by two review authors working independently, with disagreements resolved by discussion. Our judgements were justified, documented, and incorporated into reporting of results for each outcome.
Results
Description of studies
Results of the search
The search retrieved 622 articles, of which 65 studies were potentially eligible and we retrieved the full texts. Eighteen studies (22 reports) met our inclusion criteria. We excluded 42 studies (43 reports). See Characteristics of included studies and Characteristics of excluded studies tables. The PRISMA flow chart in Figure 2 illustrates the flow of literature through the search and assessment process.
We added 11 new studies in this update (Azimi Nekoo 2015; Davar 2007; Eftekhar 2012; Fatemi 2010; Greco 2016; Groenewoud 2016; Karimzadeh 2012; Mounce 2015; Peeraer 2015; Ramos 2007; Weissman 2011).
Included studies
Design and setting
All 18 included studies were parallel‐design RCTs (Azimi Nekoo 2015; Cattoli 1994; Dal Prato 2002; Davar 2007; Eftekhar 2012; El‐Toukhy 2004; Fatemi 2010; Greco 2016; Groenewoud 2016; Karimzadeh 2012; Loh 2001; Mounce 2015; Peeraer 2015; Ramos 2007; Simon 1998; Van Der Auwera 1994; Weissman 2011; Wright 2006). One of the included studies was a three‐arm trial comparing three different cycle regimens: HT plus GnRHa, HT FET and clomiphene FET (Loh 2001). Sixteen of the trials were conducted in a single centre; the remaining two trials were conducted in two or more ART centres (Groenewoud 2016; Azimi Nekoo 2015). The included RCTs were undertaken in Belgium (Fatemi 2010; Peeraer 2015; Van Der Auwera 1994), France (Wright 2006), Iran (Azimi Nekoo 2015; Davar 2007; Eftekhar 2012; Karimzadeh 2012), Israel (Simon 1998; Weissman 2011), Italy (Cattoli 1994; Dal Prato 2002; Greco 2016), the Netherlands (Groenewoud 2016), Singapore (Loh 2001), Spain (Ramos 2007) and the UK (El‐Toukhy 2004; Mounce 2015). The studies were published between 1994 and 2016.
Participants
The studies included 3815 subfertile women undergoing FET. Their ages ranged across studies from 19 to 45 years. Thirteen of the studies included women with functional ovaries or regular ovulatory cycles; in five studies, the ovulatory status of the women was not reported.
All the RCTs assessed women using embryos originating from their own oocytes. Participants used embryos resulting from either IVF or ICSI treatment.
Interventions
Natural cycle FET comparisons
Natural cycle FET versus HT FET (2 RCTs)(Cattoli 1994; Karimzadeh 2012)
Natural cycle FET versus HT plus GnRHa suppression FET (1 RCT) (Mounce 2015)
Natural cycle FET versus HMG FET (1 RCT) (Peeraer 2015)
Natural cycle FET versus modified natural cycle FET (HCG trigger) (2 RCTs) (Fatemi 2010; Weissman 2011)
Modified natural cycle FET comparisons
Modified natural cycle FET (HCG trigger) versus HT FET (1 RCT) (Groenewoud 2016)
Modified natural cycle FET versus HT plus GnRHa suppression FET (1 RCT) (Greco 2016)
HT FET comparisons
HT FET versus HT plus GnRHa suppression FET (7 RCTs) (Azimi Nekoo 2015; Dal Prato 2002; Davar 2007; El‐Toukhy 2004; Loh 2001; Ramos 2007; Simon 1998)
HT FET versus FSH ovulation induction (Wright 2006)
HT FET versus HT plus HCG supplement FET (Eftekhar 2012)
Comparisons of subtypes of ovulation‐induction FET.
HMG FET versus clomiphene plus HMG FET (Van Der Auwera 1994)
HT plus GnRHa versus clomiphene (Loh 2001),
We found no RCTs of other ovulation induction agents such as oral anti‐oestrogens (e.g. tamoxifen) or aromatase inhibitors (e.g. letrozole).
Outcomes
The trials reported the following outcomes:
live birth rate per woman (El‐Toukhy 2004; Greco 2016; Groenewoud 2016; Mounce 2015; Van Der Auwera 1994; Weissman 2011);
miscarriage rate per woman (Azimi Nekoo 2015; Dal Prato 2002; Davar 2007; El‐Toukhy 2004; Fatemi 2010; Greco 2016; Ramos 2007; Simon 1998; Van Der Auwera 1994);
ongoing pregnancy rate per woman (Fatemi 2010; Groenewoud 2016; Simon 1998);
clinical pregnancy rate per woman (Azimi Nekoo 2015; Cattoli 1994; Dal Prato 2002; Davar 2007; El‐Toukhy 2004; Fatemi 2010; Greco 2016; Groenewoud 2016; Mounce 2015; Simon 1998; Weissman 2011; Wright 2006);
multiple pregnancy rate per woman (Cattoli 1994; Mounce 2015; Van Der Auwera 1994);
cycle cancellation rate per woman (Dal Prato 2002; El‐Toukhy 2004; Groenewoud 2016; Simon 1998; Wright 2006);
endometrial thickness (Dal Prato 2002; El‐Toukhy 2004; Greco 2016; Groenewoud 2016; Simon 1998; Wright 2006);
number of centre visits to monitor FET cycle (Fatemi 2010; Weissman 2011);
clinical pregnancy rate per cycle (Loh 2001);
miscarriage rate per clinical pregnancy (El‐Toukhy 2004).
None of the studies reported ectopic pregnancy.
In four studies, outcome data were either not reported (Eftekhar 2012) or reported in forms that could not be analyzed (per embryo transfer: Karimzadeh 2012; Peeraer 2015 or per cycle: Loh 2001, and the numbers of transfers and cycles were not equivalent to the numbers of women randomized). Thus, three studies reported their outcome data in a form that could not be included in meta‐analyses (Karimzadeh 2012; Loh 2001; Peeraer 2015) (see Table 8; Table 9; Table 10; Table 11; Table 12; Table 13).
1. Live birth rate: per embryo transfer data.
| Study | Intervention (number of embryo transfer) | Control (number of embryo transfer) | Live birth rate | P value |
| Peeraer 2015 | Natural cycle FET (n = 332) | HMG FET (n = 340) | 32/332 vs 45/340 | n/s |
FET: frozen‐thawed embryo transfer; HMG: human menopausal gonadotrophin; n/s: not significant.
2. Miscarriage rate: per embryo transfer data.
| Study | Intervention (number of embryo transfer) | Control (number of embryo transfer) | Miscarriage rate | P value |
| Karimzadeh 2012 | Natural cycle FET | HT FET | 41.7% vs 22.2% | n/s |
FET: frozen‐thawed embryo transfer; HT: hormone therapy; n/s: not significant.
3. Ongoing pregnancy rate: per embryo transfer data.
| Study | Intervention (number of embryo transfer) | Control (number of embryo transfer) | Ongoing pregnancy rate | P value |
| Karimzadeh 2012 | Natural cycle FET | HT FET | 24.1% vs 21.9% | n/s |
FET: frozen‐thawed embryo transfer; HT: hormone therapy; n/s: not significant.
4. Clinical pregnancy rate: per cycle data.
| Study | Intervention (number of cycles) | Control (number of cycles) | Clinical pregnancy rate | P value |
| Loh 2001 | Clomiphene‐induced ovulation (n = 35) | HT (n = 52) | 3/35 vs 5/52 | n/s |
| Clomiphene‐induced ovulation (n = 32) | HT plus GnRHa trigger (n = 37) | 2/32 vs 6/37 | n/s |
GnRHa: gonadotrophin releasing hormone agonist; HT: hormone therapy; n/s: not significant.
5. Clinical pregnancy rate: per embryo transfer data.
| Study | Intervention (number of embryo transfer) | Control (number of embryo transfer) | Clinical pregnancy rate | P value |
| Karimzadeh 2012 | Natural cycle FET | HT FET | 27.6% vs 25% | n/s |
FET: frozen‐thawed embryo transfer; HT: hormone therapy; n/s: not significant.
6. Endometrial thickness: data not suitable for analysis.
| Study | Intervention (number of cycles/embryo transfer) | Control (number of cycles/embryo transfer) | Endometrial thickness | P value |
| Loh 2001 | Clomiphene‐induced ovulation (n = 67) | HT alone or HT plus GnRHa suppression (n = 37) | 9.7 vs 9.8 | n/s |
| Peeraer 2015 | Natural cycle FET (n = 332) | HMG FET (n = 340) | 8.9 vs 8.9 | n/s |
FET: frozen‐thawed embryo transfer; GnRHa: gonadotrophin releasing hormone agonist; HMG: human menopausal gonadotrophin; HT: hormone therapy; n/s: not significant.
Excluded studies
We excluded 42 studies for the following reasons:
thirty‐four were not true RCTs (Al‐Shawaf 1993; Awonuga 1996; Bals‐Pratsch 1999; Belaisch‐Allart 1994; Benfer 1994; Chen 2007; de Ziegler 1990; Dolan 1991; Dor 1991; Gelbaya 2006; Givens 2007; Gonzalez 1992; Imthurn 1996; Jaroudi 1991; Kawamura 2007; Lelaidier 1992; Lelaidier 1995; Loh 1999; Lornage 1990; Mausher 1991; Morozov 2007; Oehninger 2000; Pattinson 1992; Queenan 1994; Queenan 1997; Sathanandan 1991; Schmidt 1989; Shiotani 2006; Simon 1999; Spandorfer 2004; Tanos 1996; Wada 1992; Yee 1995; Yishai 2001);
seven studies did not use interventions relevant to the review (Davar 2015; Elhelw 2008; Garrisi 1991; Groenewoud 2015; Page 2005; Taskin 2002; Yu 2015);
one study did not report the number of participants randomized to, or analyzed in, each treatment group (Lee 2008).
Risk of bias in included studies
Details of the quality criteria of the relevant studies can be found in the Characteristics of included studies table.
Allocation
Sequence generation
Nine studies reported using computer‐generated randomization and we judged them to be at low risk of bias (Azimi Nekoo 2015; Davar 2007; Eftekhar 2012; El‐Toukhy 2004; Fatemi 2010; Greco 2016; Groenewoud 2016; Mounce 2015; Weissman 2011). Nine studies did not report the randomization method and we judged them to be at unclear risk of bias (Cattoli 1994; Dal Prato 2002; Karimzadeh 2012; Loh 2001; Peeraer 2015; Ramos 2007; Simon 1998; Van Der Auwera 1994; Wright 2006).
Allocation concealment
Concealment of allocation was adequate in three studies using sealed envelopes ; Loh 2001; Peeraer 2015; Weissman 2011). The allocation concealment was unclear in 14 studies (Azimi Nekoo 2015; Cattoli 1994; Davar 2007; Dal Prato 2002; Eftekhar 2012; El‐Toukhy 2004; Greco 2016; Groenewoud 2016; Karimzadeh 2012; Mounce 2015; Ramos 2007; Simon 1998; Van Der Auwera 1994; Wright 2006). Fatemi 2010 stated that no concealment took place and was rated as at high risk of bias.
Blinding
Blinding of the woman or the clinician was not practically possible in any of the included studies. Moreover, non‐blinding may not affect some of the outcome measures as they were objectively assessed. Therefore, we rated all the included studies as unclear with respect to blinding and detection bias.
Incomplete outcome data
Five studies used an intention‐to‐treat analysis, that is, all women randomized at baseline were included in the analysis (Azimi Nekoo 2015; Davar 2007; El‐Toukhy 2004; Mounce 2015; Simon 1998); in another study, the proportions of withdrawals and reasons for withdrawals were fairly balanced between the two groups (Greco 2016). We therefore assessed these six studies as at low risk of bias with respect to attrition. There was insufficient information in 10 studies to make a conclusive judgement on attrition bias and we rated them as unclear in this domain (Cattoli 1994; Dal Prato 2002; Eftekhar 2012; Fatemi 2010; Karimzadeh 2012; Loh 2001; Peeraer 2015; Ramos 2007; Van Der Auwera 1994; Wright 2006). Two studies were at high risk of attrition bias because the reasons for withdrawals and the proportions of withdrawals were substantially different between the two treatment groups (Groenewoud 2016; Weissman 2011).
Selective reporting
Eleven studies were at low risk of selective reporting bias (Dal Prato 2002; Davar 2007; Eftekhar 2012; El‐Toukhy 2004; Fatemi 2010; Groenewoud 2016; Mounce 2015; Peeraer 2015; Simon 1998; Weissman 2011; Wright 2006). Reporting bias was assessed as unclear in the remaining seven studies as there was insufficient information (Azimi Nekoo 2015; Cattoli 1994; Greco 2016; Karimzadeh 2012; Loh 2001; Ramos 2007; Van Der Auwera 1994).
Other potential sources of bias
In one study participants in the groups differed significantly in age so we assessed it at high risk of bias with respect to demographic characteristics (Weissman 2011). We found no potential sources of within‐study bias in 11 included studies (Azimi Nekoo 2015; Dal Prato 2002; Davar 2007; Eftekhar 2012; El‐Toukhy 2004; Groenewoud 2016; Karimzadeh 2012; Mounce 2015; Peeraer 2015; Simon 1998; Wright 2006). Information was insufficient to form a clear judgement in six studies (Cattoli 1994; Fatemi 2010; Greco 2016; Loh 2001; Ramos 2007; Van Der Auwera 1994).
Effects of interventions
See: Table 1; Table 2; Table 3; Table 4; Table 5; Table 6; Table 7
The included studies compared a wide range of FET cycle regimens and outcome measures. We could not combine data for most of the comparisons as they were reported in single trials.
NATURAL CYCLE FET COMPARISONS
1 Natural cycle FET versus HT FET
Two studies made this comparison (Cattoli 1994; Karimzadeh 2012). For Karimzadeh 2012 no per‐woman outcome data were available. See Table 9; Table 10; Table 12.
Primary outcomes
1.1 Live birth rate per woman
No data were available.
1.2 Miscarriage rate per woman
No data were available.
Secondary outcomes
1.3 Ongoing pregnancy rate per woman
No data were available.
1.4 Clinical pregnancy rate per woman
There was no evidence of a difference between the groups in the clinical pregnancy rate (OR 1.06, 95% CI 0.40 to 2.80, 1 RCT, n = 100) (Analysis 1.1).
1.1. Analysis.
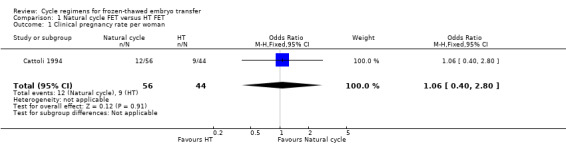
Comparison 1 Natural cycle FET versus HT FET, Outcome 1 Clinical pregnancy rate per woman.
1.5 Multiple pregnancy rate per woman
There was no evidence of a difference between the groups in the multiple pregnancy rate (OR 2.48, 95% CI 0.09 to 68.14, 1 RCT, n = 21, very low‐quality evidence) (Analysis 1.2).
1.2. Analysis.
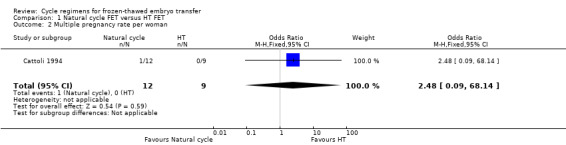
Comparison 1 Natural cycle FET versus HT FET, Outcome 2 Multiple pregnancy rate per woman.
1.6 Cycle cancellation rate per woman
No data were available.
1.1.7 Endometrial thickness during FET cycle
No data were available.
1.8 Number of centre visits to monitor FET cycle
No data were available.
2 Natural cycle FET versus HT plus GnRHa suppression FET
Primary outcomes
2.1 Live birth rate per woman
There was no evidence of a difference between the groups (OR 0.77, 95% CI 0.39 to 1.53, 1 RCT, n = 159, low‐quality evidence). The evidence suggests that if the chance of live birth in an HT plus GnRHa suppression cycle is assumed to be 32%, the chance in a natural cycle would be between 15% and 41% (Analysis 2.1).
2.1. Analysis.
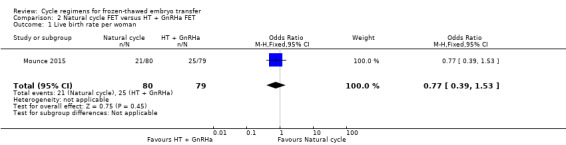
Comparison 2 Natural cycle FET versus HT + GnRHa FET, Outcome 1 Live birth rate per woman.
Sensitivity analysis using a random‐effects model (OR 0.77, 95% CI 0.39 to 1.53) or RR as a measure of treatment effect (RR 0.83, 95% CI 0.51 to 1.35) did not substantially change our finding.
2.2 Miscarriage rate per woman
No data were available.
Secondary outcomes
2.3 Ongoing pregnancy rate per woman
No data were available.
2.4 Clinical pregnancy rate per woman
There was no evidence of a difference between the groups in the clinical pregnancy rate (OR 0.87, 95% CI 0.45 to 1.71, 1 RCT, n = 159) (Analysis 2.2).
2.2. Analysis.
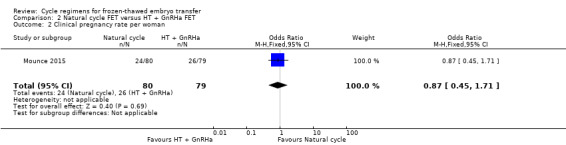
Comparison 2 Natural cycle FET versus HT + GnRHa FET, Outcome 2 Clinical pregnancy rate per woman.
2.5 Multiple pregnancy rate
There was no evidence of a difference between the groups in multiple pregnancy rates (OR 0.58, 95% CI 0.13 to 2.50, 1 RCT, n = 159, low‐quality evidence). The evidence suggests that if the risk of multiple pregnancy following an HT with GnRHa suppression cycle is assumed to be 6%, the risk following a natural cycle would be between 1% and 14% (Analysis 2.3).
2.3. Analysis.
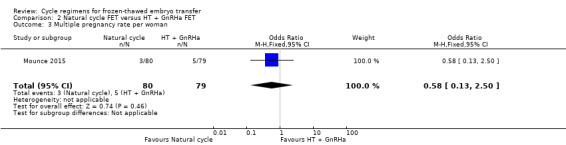
Comparison 2 Natural cycle FET versus HT + GnRHa FET, Outcome 3 Multiple pregnancy rate per woman.
2.6 Cycle cancellation rate per woman.
No data were available.
2.7 Endometrial thickness during FET cycle
No data were available.
2.8 Number of centre visits to monitor FET cycle
No data were available.
3. Natural cycle FET versus HMG FET
One study reported this comparison (Peeraer 2015), but no per‐woman outcome data were available. See Table 8, Table 13.
4 Natural cycle FET versus modified natural cycle FET (HCG trigger)
Two studies made this comparison (Fatemi 2010; Weissman 2011).
Primary outcomes
4.1 Live birth rate per woman
One study reported on live birth rate. There was no evidence of a difference between the groups (OR 0.55, 95% CI 0.16 to 1.93, 1 RCT, n = 60, very low‐quality evidence). This evidence suggests that if the chance of live birth in a natural cycle with HCG trigger is assumed to be 27%, the chance in a natural cycle with FET only would be between 6% and 41% (Analysis 3.1).
3.1. Analysis.
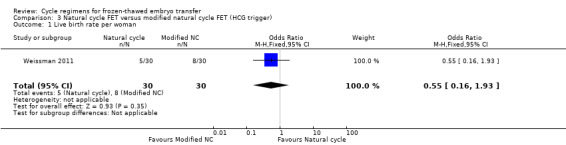
Comparison 3 Natural cycle FET versus modified natural cycle FET (HCG trigger), Outcome 1 Live birth rate per woman.
Sensitivity analysis using a random‐effects model (OR 0.55, 95% CI 0.16 to 1.93) or RR as a measure of treatment effect (RR 0.63, 95% CI 0.23 to 1.69) did not substantially change our finding.
4.2 Miscarriage rate per woman
One study reported on miscarriage. There was no evidence of a difference between the groups (OR 0.20, 95% CI 0.01 to 4.13, 1 RCT, n = 168, very low‐quality evidence). The evidence suggests that if the risk of miscarriage following a natural cycle with HCG trigger is assumed to be 2%, the risk following a natural cycle only would be between 0% and 9%.
Sensitivity analysis using a random‐effects model (OR 0.20, 95% CI 0.01 to 4.13) or RR as a measure of treatment effect (RR 0.20, 95% CI 0.01 to 4.10) did not substantially change our finding (Analysis 3.2).
3.2. Analysis.
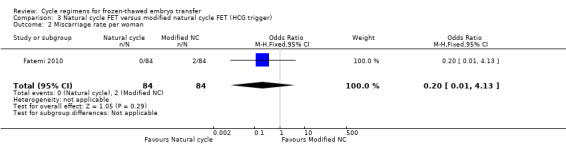
Comparison 3 Natural cycle FET versus modified natural cycle FET (HCG trigger), Outcome 2 Miscarriage rate per woman.
Secondary outcomes
4.3 Ongoing pregnancy per woman
One study reported ongoing pregnancy. The evidence marginally favoured a natural cycle FET (OR 2.44, 95% CI 1.03 to 5.76, 1 RCT, n = 168, very low‐quality evidence). The evidence suggests that if the chance of ongoing pregnancy following a natural cycle with HCG trigger is assumed to be 11%, the chance following a natural cycle only would be between 11% and 41% (Analysis 3.3).
3.3. Analysis.
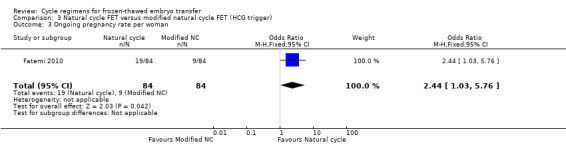
Comparison 3 Natural cycle FET versus modified natural cycle FET (HCG trigger), Outcome 3 Ongoing pregnancy rate per woman.
4.4 Clinical pregnancy per woman
One study reported on clinical pregnancy. There was no evidence of a difference between the groups (OR 1.00, 95% CI 0.32 to 3.14, 1 RCT, n = 60) (Analysis 3.4).
3.4. Analysis.
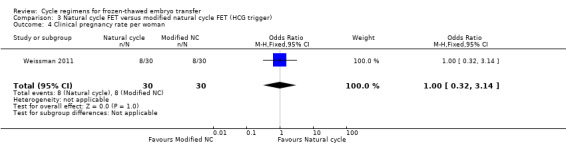
Comparison 3 Natural cycle FET versus modified natural cycle FET (HCG trigger), Outcome 4 Clinical pregnancy rate per woman.
4.5 Multiple pregnancy
No data were available.
4.6 Cycle cancellation rate per woman
No data were available.
4.7 Endometrial thickness during FET cycle
No data were available.
4.8 Number of centre visits to monitor FET cycle
No data were available.
MODIFIED NATURAL CYCLE FET COMPARISONS
5. Modified natural cycle FET (HCG trigger) versus HT FET
Only one study reported data on this comparison (Groenewoud 2016).
Primary outcomes
5.1 Live birth rate per woman
There was no evidence of a difference between the two groups in the rate of live births (OR 1.34, 95% CI 0.88 to 2.05, 1 RCT, n = 959, low‐quality evidence). The evidence suggests that if the chance of live birth in HT FET cycle is assumed to be 9%, the chance in modified natural cycle FET would be between 8% and 17%. (Analysis 4.1).
4.1. Analysis.
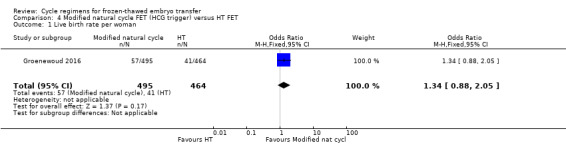
Comparison 4 Modified natural cycle FET (HCG trigger) versus HT FET, Outcome 1 Live birth rate per woman.
5.2 Miscarriage rate per woman
No data were available.
Secondary outcomes
5.3 Ongoing pregnancy rate per woman
There was no evidence of a difference between the two groups in the ongoing pregnancy rate (OR 1.21, 95% CI 0.80 to 1.83, 1 RCT, n = 959, low‐quality evidence). The evidence suggests that if the chance of ongoing pregnancy in HT FET cycle is assumed to be 10%, the chance in modified natural cycle FET would be between 8% and 16%. (Analysis 4.2).
4.2. Analysis.
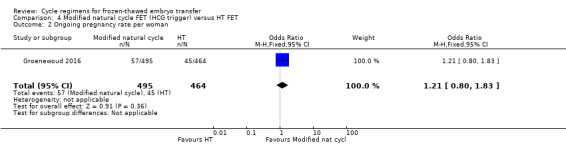
Comparison 4 Modified natural cycle FET (HCG trigger) versus HT FET, Outcome 2 Ongoing pregnancy rate per woman.
5.4 Clinical pregnancy rate per woman
There was no evidence of a difference in clinical pregnancy rate between the two groups (OR 1.22, 95% CI 0.87 to 1.70, 1 RCT, n = 959) (Analysis 4.3).
4.3. Analysis.
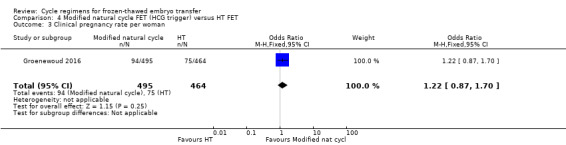
Comparison 4 Modified natural cycle FET (HCG trigger) versus HT FET, Outcome 3 Clinical pregnancy rate per woman.
5.5 Multiple pregnancy rate per woman
No data were available.
5.6 Cycle cancellation rate per woman
There was evidence of a difference in the rate of cycle cancellation between the two groups with more cycles cancelled in the HT group compared to the modified natural cycle group (OR 0.70, 95% CI 0.52 to 0.95, 1 RCT, n = 959) (Analysis 4.4).
4.4. Analysis.
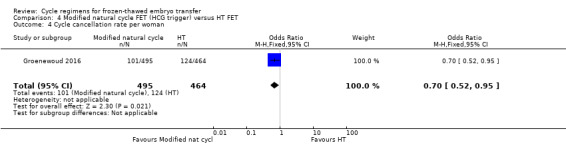
Comparison 4 Modified natural cycle FET (HCG trigger) versus HT FET, Outcome 4 Cycle cancellation rate per woman.
5.7 Endometrial thickness
There was no evidence of a difference between the two groups in endometrial thickness (MD 0.10 mm, 95% CI ‐0.13 to 0.33, 1 RCT, n = 959) (Analysis 4.5).
4.5. Analysis.
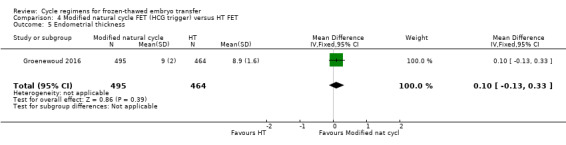
Comparison 4 Modified natural cycle FET (HCG trigger) versus HT FET, Outcome 5 Endometrial thickness.
5.8 Number of centre visits to monitor FET cycle
No data were available.
6. Modified natural cycle FET (HCG trigger) versus HT + GnRHa suppression FET
One trial reported data on this comparison (Greco 2016).
Primary outcomes
6.1 Live birth rate per woman
There was no evidence of a difference between the two groups in live birth rate (OR 1.11, 95% CI 0.66 to 1.87, 1 RCT, n = 236, low‐quality evidence). The evidence suggests that if the chance of live birth in HT plus GnRHa FET cycle is assumed to be 40%, the chance in a modified natural cycle would be between 30% and 55% (Analysis 5.1).
5.1. Analysis.
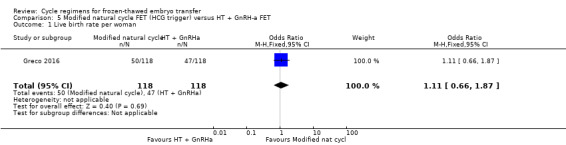
Comparison 5 Modified natural cycle FET (HCG trigger) versus HT + GnRH‐a FET, Outcome 1 Live birth rate per woman.
6.2 Miscarriage rate per woman
There was no evidence of a difference between the two groups in the rate of miscarriage (OR 0.74, 95% CI 0.25 to 2.19, 1 RCT, n = 236, low‐quality evidence). The evidence suggests that if the risk of miscarriage in HT plus GnRHa FET cycle is assumed to be 7%, the risk in a modified natural cycle would be between 2% and 14% (Analysis 5.2).
5.2. Analysis.
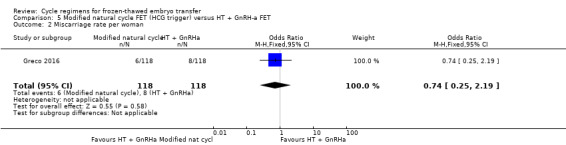
Comparison 5 Modified natural cycle FET (HCG trigger) versus HT + GnRH‐a FET, Outcome 2 Miscarriage rate per woman.
Secondary outcomes
6.3 Ongoing pregnancy rate per woman
No data were available.
6.4 Clinical pregnancy rate per woman
There was no evidence of a difference between the two groups in clinical pregnancy rate (OR 1.07, 95% CI 0.64 to 1.78, 1 RCT, n = 236) (Analysis 5.3).
5.3. Analysis.
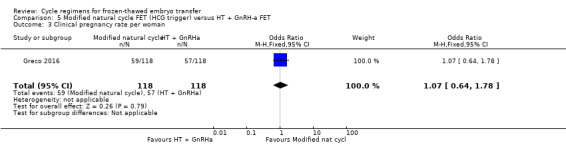
Comparison 5 Modified natural cycle FET (HCG trigger) versus HT + GnRH‐a FET, Outcome 3 Clinical pregnancy rate per woman.
6.5 Multiple pregnancy rate per woman
No data were available.
6.6 Cycle cancellation rate per woman
No data were available.
6.7 Endometrial thickness
There was no evidence of a difference between the two groups in endometrial thickness (MD ‐0.20 mm, 95% CI ‐0.54 to 0.14, 1 RCT, n = 236) (Analysis 5.4).
5.4. Analysis.
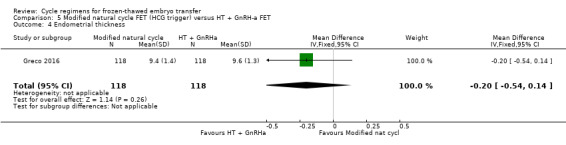
Comparison 5 Modified natural cycle FET (HCG trigger) versus HT + GnRH‐a FET, Outcome 4 Endometrial thickness.
6.8 Number of centre visits to monitor FET cycles
No data were available.
HT FET COMPARISONS
7. HT FET versus HT plus GnRHa suppression FET
Seven studies made this comparison (Azimi Nekoo 2015; Dal Prato 2002; Davar 2007; El‐Toukhy 2004; Loh 2001; Ramos 2007; Simon 1998). For Loh 2001 no per‐woman outcome data were available. See Table 11, Table 13.
Primary outcomes
7.1 Live birth rate per woman
There was evidence of a difference between the two groups in live birth rate, with a higher proportion of women who were treated with HT plus GnRHa suppression having live births compared to women treated with HT alone (OR 0.10, 95% CI 0.04 to 0.30, 1 RCT, n = 75, low‐quality evidence). The evidence suggests that if the chance of a live birth following HT plus GnRHa suppression is assumed to be 74%, the chance following treatment with HT alone would be between 10% and 46% (Analysis 6.1).
6.1. Analysis.
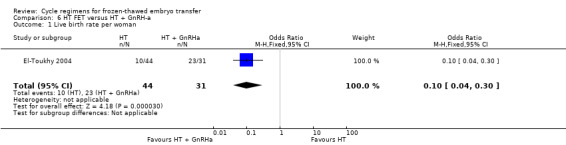
Comparison 6 HT FET versus HT + GnRH‐a, Outcome 1 Live birth rate per woman.
Sensitivity analysis using a random‐effects model (OR 0.10, 95% CI 0.04 to 0.30) or RR as a measure of treatment effect (RR 0.31, 95% CI 0.17 to 0.55) did not substantially change our finding.
7.2 Miscarriage rate per woman
There was no evidence of a difference between the two groups in miscarriage rate (OR 0.64, 95% CI 0.37 to 1.12, 6 RCTs, n = 991, I2 = 0%, low‐quality evidence). The evidence suggests that if the risk of miscarriage in an HT plus GnRHa suppression cycle is assumed to be 5%, the risk following treatment with HT alone would be between 2% and 5% (Analysis 6.2).
6.2. Analysis.
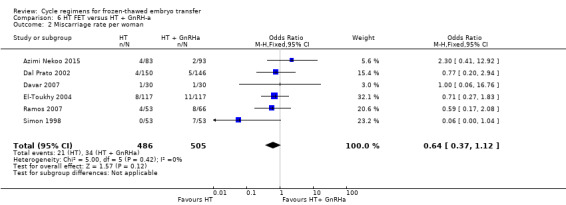
Comparison 6 HT FET versus HT + GnRH‐a, Outcome 2 Miscarriage rate per woman.
Sensitivity analysis using a random‐effects model (OR 0.73, 95% CI 0.40 to 1.31) or RR as a measure of treatment effect (RR 0.66, 95% CI 0.39 to 1.11) did not substantially change our finding.
Secondary outcomes
7.3 Ongoing pregnancy rate per woman
There was no evidence of a difference between the two groups in ongoing pregnancy rates (OR 1.72, 95% CI 0.61 to 4.85, 1 RCT, n = 106, very low‐quality evidence). The evidence suggests that if the chance of an ongoing pregnancy following treatment with HT plus GnRHa suppression is assumed to be 13%, the chance following treatment with HT alone would be between 9% and 42% (Analysis 6.3).
6.3. Analysis.
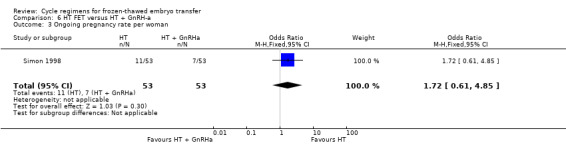
Comparison 6 HT FET versus HT + GnRH‐a, Outcome 3 Ongoing pregnancy rate per woman.
7.4 Clinical pregnancy rate per woman
There was no evidence of a difference between the two groups in clinical pregnancy rate (OR 0.90, 95% CI 0.65 to 1.25, 5 RCTs, n = 872, I2 = 54%) (Analysis 6.4).
6.4. Analysis.
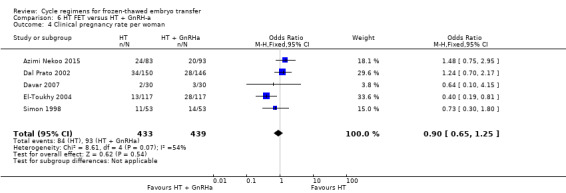
Comparison 6 HT FET versus HT + GnRH‐a, Outcome 4 Clinical pregnancy rate per woman.
7.5 Multiple pregnancy rate per woman
No data were available.
7.6 Cycle cancellation rate per woman
There was no evidence of a difference between the two groups in cycle cancellation rate (OR 2.73, 95% CI 0.79 to 9.38, 3 RCTs, n = 636, I2 = 0%).(Analysis 6.5).
6.5. Analysis.
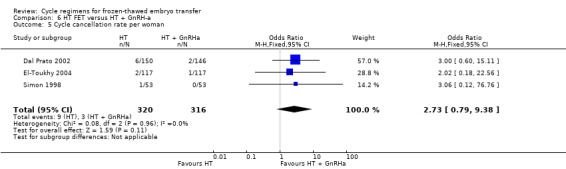
Comparison 6 HT FET versus HT + GnRH‐a, Outcome 5 Cycle cancellation rate per woman.
7.7 Endometrial thickness during FET cycle
There was no evidence of a difference between the two groups in endometrial thickness (MD ‐0.16 mm, 95% CI ‐0.41 to 0.09, 3 RCTs, n = 625, I2 = 79%).( Analysis 6.6).
6.6. Analysis.
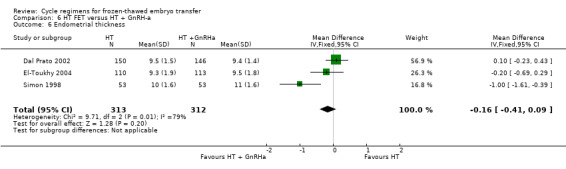
Comparison 6 HT FET versus HT + GnRH‐a, Outcome 6 Endometrial thickness.
On sensitivity analysis using a random‐effects estimate, there was a similar pattern of result (MD ‐0.32, 95% CI ‐0.92 to 0.27).
7.8 Number of centre visits to monitor FET cycles
No data were available.
8. HT FET versus FSH ovulation induction
One study made this comparison (Wright 2006)
Primary outcomes
8.1 Live birth rate per woman
No data were available.
8.2 Miscarriage rate per woman
No data were available.
Secondary outcomes
8.3 Ongoing pregnancy rate per woman
No data were available.
8.4 Clinical pregnancy rate per woman
There was no evidence of a difference between the two groups in clinical pregnancy rate (OR 1.09, 95% CI 0.45 to 2.62, 1 RCT, n = 175) (Analysis 7.1).
7.1. Analysis.
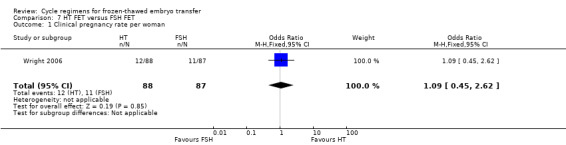
Comparison 7 HT FET versus FSH FET, Outcome 1 Clinical pregnancy rate per woman.
8.5 Multiple pregnancy rate per woman
No data were available.
8.6 Cycle cancellation rate per woman
There was no evidence of a difference between the two groups in cycle cancellation rates (OR 0.99, 95% CI 0.49 to 2.00, 1 RCT, n = 75) (Analysis 7.2).
7.2. Analysis.
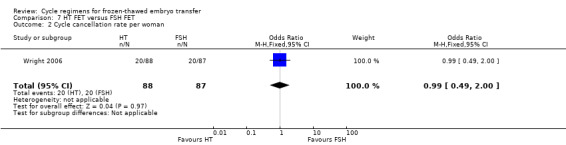
Comparison 7 HT FET versus FSH FET, Outcome 2 Cycle cancellation rate per woman.
3.1.7 Endometrial thickness during FET cycles
There was no evidence of a difference between the two groups in endometrial thickness (MD 0.00 mm, 95% CI ‐0.31 to 0.31, 1 RCT, n = 175) (Analysis 7.3).
7.3. Analysis.
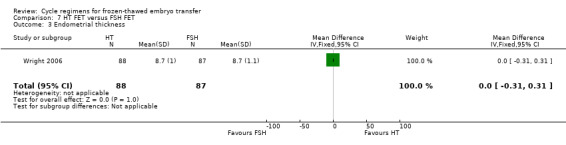
Comparison 7 HT FET versus FSH FET, Outcome 3 Endometrial thickness.
8.8 Number of centre visits to monitor FET cycles
No data were reported.
9. HT FET versus HT plus HCG supplement FET
One study made this comparison (Eftekhar 2012), but no outcome data were reported.
COMPARISONS OF SUBTYPES OF OVULATION‐INDUCTION FET
10. HMG FET versus clomiphene + HMG FET
One study made this comparison (Van Der Auwera 1994).
Primary outcomes
10.1 Live birth rate per woman
There were lower live birth rates in women treated with clomiphene plus HMG than in women treated with HMG alone (OR 2.49, 95% CI 1.07 to 5.80, 1 RCT, n = 209, very low‐quality evidence). The evidence suggests that if the chance of a live birth following treatment with clomiphene combined with HMG is assumed to be 8%, the chance following treatment with HMG alone would be between 9% and 35% (Analysis 8.1).
8.1. Analysis.
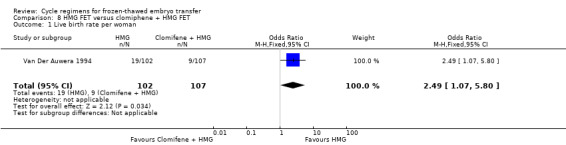
Comparison 8 HMG FET versus clomiphene + HMG FET, Outcome 1 Live birth rate per woman.
Sensitivity analysis using a random‐effects model (OR 2.49, 95% CI 1.07 to 5.80) or RR as a measure of treatment effect (RR 2.21, 95% CI 1.05 to 4.67) did not substantially change our finding.
10.2 Miscarriage rate per woman
There was no evidence of a difference between the two groups in miscarriage rates (OR 1.33, 95% CI 0.35 to 5.09, 1 RCT, n = 209, very low‐quality evidence). The evidence suggests that if the risk of miscarriage following treatment with clomiphene combined with HMG is assumed to be 4%, the risk following treatment with HMG alone would be between 1% and 16% (Analysis 8.2).
8.2. Analysis.
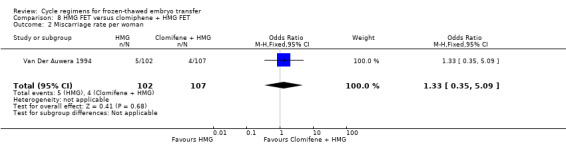
Comparison 8 HMG FET versus clomiphene + HMG FET, Outcome 2 Miscarriage rate per woman.
Sensitivity analysis using a random‐effects model (OR 1.33, 95% CI 0.35 to 5.09) or RR as a measure of treatment effect (RR 1.31, 95% CI 0.36 to 4.75) did not substantially change our finding.
Secondary outcomes
10.3 Ongoing pregnancy rate per woman
No data were available.
10.4 Clinical pregnancy rate per woman
No data were available
10.5 Multiple pregnancy rate per woman
There was no evidence of a difference between the two groups in multiple pregnancy rates (OR 1.41, 95% CI 0.31 to 6.48, 1 RCT, n = 209, very low‐quality evidence). The evidence suggests that if the risk of a multiple pregnancy following treatment with clomiphene combined with HMG is assumed to be 3%, the risk following HMG alone would be between 1% and 16% (Analysis 8.3).
8.3. Analysis.
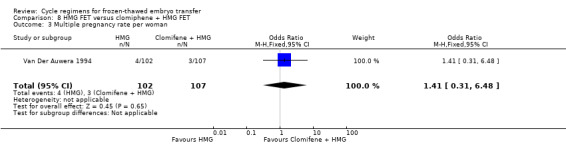
Comparison 8 HMG FET versus clomiphene + HMG FET, Outcome 3 Multiple pregnancy rate per woman.
10.6 Cycle cancellation rate per woman.
No data were available.
10.7 Endometrial thickness
No data were available.
10.8 Number of centre visits to monitor FET cycles
No data were available.
11. HT plus GnRHa FET versus clomiphene FET
One study made this comparison (Loh 2001), but reported no per‐woman data.
Discussion
Summary of main results
We found 18 RCTs with 3815 participating women. The largest study included 959 women. The mean number of women per trial was 164 (range 60 to 959). Therefore, findings from this review should be interpreted with caution due to the limited number and small size of available RCTs.
Natural cycle FET comparisons
There was no evidence of a difference in live birth, miscarriage or multiple pregnancy rates between women in natural cycle and women undergoing other FET regimens (HT with GnRHa suppression or natural cycle plus HCG trigger). However, women in natural cycle had a marginally higher ongoing pregnancy rate than women who had natural cycle with HCG trigger. This might be attributed to administration of HCG in late follicular phase, which induces a cascade of events in the endometrium and a rise in endogenous luteinising hormone (LH). These events might have a negative impact on implantation and ultimately on ongoing pregnancy rates.
Modified natural cycle FET comparisons
There was no evidence of a difference in the rates of live birth, miscarriage, or ongoing pregnancy between women in modified natural cycle and those undergoing HT FET or HT with GnRHa suppression.
HT FET comparisons
Hormonally prepared (HT) FET cycles combined with GnRHa suppression resulted in higher live birth rates compared with FET cycles prepared with HT alone, but there was no evidence of a difference in miscarriage or ongoing pregnancy rates. The difference in live births may be attributed to a premature rise in LH levels that may occur in women with functioning ovaries not suppressed by a GnRHa. This rise in LH was reported even with no observed follicular recruitment (de Ziegler 1991; Speroff 1994). LH is known to affect endometrial development through its effect on oestrogen and progesterone production but there has also been evidence to suggest that LH promotes localised synthesis of oestrogen and progesterone in the endometrium (Ku 2002; Shemesh 2001). This premature LH rise may interfere with endometrial development and may hinder embryo implantation or it may cause suboptimal embryo implantation.
The review authors acknowledge the difficulty in combining studies in meta‐analyses in the comparison HT versus HT plus GnRHa suppression. One study used a fixed‐dose oestrogen preparation for both groups (El‐Toukhy 2004), while in two studies the starting dose of oestrogen was lower in the HT‐plus‐GnRHa group compared to the HT‐alone group (Dal Prato 2002; Simon 1998). The El‐Toukhy 2004 study may have accounted for the presence of significant heterogeneity observed in one of the outcome measures, that is, endometrial thickness (MD ‐0.16 mm, 95% CI ‐0.41 to 0.09, 3 RCTs, n = 625, I2 = 79%) (Analysis 18.6).
Comparisons of subtypes of ovulation‐induction FET
We identified two subtypes of FET comparisons: HMG versus clomiphene plus HMG; and HT plus GnRHa versus clomiphene. However, there were no per‐woman clinical outcomes reported for HT plus GnRHa versus clomiphene. Women treated with HMG alone reported higher live birth rates compared with those who received clomiphene combined with HMG (Van Der Auwera 1994). There was no evidence of a difference in either miscarriage rates or multiple pregnancy rates
In current practice, most centres use natural cycle or use GnRHa suppression in conjunction with HT for FET. Clomiphene has an anti‐oestrogenic effect on the endometrium leading to suboptimal endometrial development, therefore few centres, if any, are using it for ovulation induction cycle FET. It is highly unlikely that there will be future studies comparing clomiphene with HT only cycles.
Overall completeness and applicability of evidence
We included only 18 RCTs and 3815 women. The included studies did not investigate some of our comparisons in full. In some of those that were investigated, data were either not available on some important outcome measures or were reported in non‐usable forms. The majority of the investigated comparisons were in single, small trials. The largest number of participants that could be combined in this review related to the comparison of HT versus HT plus GnRHa (six RCTs, n = 991). Thus, in most of the comparisons, we could not combine data in meta‐analyses for some outcome measures, resulting in effect estimates with wide CIs. In addition, most of the included studies were not adequately powered to detect any significant difference in treatment effects between the various treatment groups. In addition, the majority of the trials included women with regular ovulatory cycles with no information on women with anovulatory cycles In all, the body of current identified evidence does not allow a robust conclusion regarding the superiority of one cycle regimen over another in preparation for FET.
Quality of the evidence
The overall quality of the evidence was low or very low. The main limitations were failure to report important clinical outcomes, poor reporting of study methods and lack of precision due to low event rates.
The quality of fertility trials has been criticised repeatedly. One of the areas of particular concern is what statisticians refer to as the 'unit of analysis' error (Vail 2003). It is methodologically incorrect to report data per cycle when it is women or couples who are randomized because many of the women will have undergone more than one treatment cycle (Dias 2008; Johnson 2003; Vail 2003). However, pregnancy rate per cycle is a commonly reported outcome in fertility trials and reviews.
Potential biases in the review process
To prevent selection bias, two review authors independently assessed the included studies. The review was undertaken using its protocol as a guide to minimise bias as much as possible (Ghobara 2002). For meta‐analysis, the unit of analysis was 'per woman randomized' resulting in the exclusion of outcomes reported in 'per‐cycle'. As a result, the analyses are statistically less biased although a selection bias may have occurred.
We conducted a comprehensive search for eligible studies so as to minimise the effect of publication bias. However, we could not construct funnel plots for any comparisons thereby making it difficult to detect the presence of publication bias.
Agreements and disagreements with other studies or reviews
Findings from the current and previous versions of the review were essentially similar and substantially in agreement with each other (see Ghobara 2008 for previous version of the review).
In nine non‐randomised controlled studies, there was no evidence of a difference in either clinical or multiple pregnancy rates between women who had natural cycle FET and women who had HT FET (Belaisch‐Allart 1994; Dolan 1991; Dor 1991; Givens 2007; Kawamura 2007; Oehninger 2000; Pattinson 1992; Schmidt 1989; Yishai 2001). These findings are in agreement with those of this review. In contrast, however, results of three retrospective studies comparing these two regimens showed higher pregnancy rates in the natural‐cycle group (Gonzalez 1992; Loh 1999; Morozov 2007).
We found one quasi‐randomised study involving 162 cycles (Sathanandan 1991) and five non‐randomised controlled studies assessing natural‐cycle FET versus HT plus GnRHa suppression (149 cycles (Al‐Shawaf 1993), 419 cycles (Chen 2007), 417 cycles (Gelbaya 2006), 512 cycles (Queenan 1994), 125 cycles (Wada 1992)). There were no differences in live birth or clinical pregnancy rates in any of these studies. Again, these findings are in agreement with those of this review.
Authors' conclusions
Implications for practice.
This review did not find evidence to support the use of one cycle regimen in preference to another in preparation for frozen‐thawed embryo transfer (FET) in women with regular ovulatory cycles. The most common modalities for FET are natural cycle with or without HCG trigger or endometrial preparation with hormone therapy (HT), with or without gonadotrophin‐releasing hormone agonist (GnRHa) suppression. We identified only four direct comparisons of these two modalities and there was insufficient evidence in this review to support the use of either of these interventions in preference to the other. One small study compared clomiphene plus human menopausal gonadotrophin (HMG) with HMG; however, the evidence was of very low quality and should be interpreted with caution. We found no data specific to non‐ovulatory women.
Implications for research.
There are few and insufficient randomized controlled trials (RCTs) assessing the different methods of endometrial preparation prior to FET. There is a continuing need to design and conduct adequately powered RCTs. Researchers should ensure adherence to a clear randomization method and sound allocation concealment measures. An intention‐to‐treat analysis should be employed. The outcome measures should be expressed 'per woman' rather than per cycle. In the reporting of studies' results, the absolute figures should be included in addition to percentages. The follow‐up period should be of sufficient duration to enable reporting on live birth rates. Because of the relative complexity and cost of ovulation induction cycles, future RCTs should focus on further evaluation of the simpler or cheaper (or both) natural‐cycle FET and the HT cycle regimens. Future research should address the possible use of GnRHa to suppress luteinising hormone release in FET cycles and to compare its use with GnRHa HT FET cycles. The role of the relatively newer oral ovulation induction agents, such as aromatase inhibitors in FET cycles, needs to be evaluated in well‐conducted RCTs.
What's new
| Date | Event | Description |
|---|---|---|
| 13 December 2016 | New search has been performed | This review has been updated, and 11 new studies have been included: Davar 2007; Eftekhar 2012; Fatemi 2010; Greco 2016; Groenewoud 2016; Karimzadeh 2012; Mounce 2015; Azimi Nekoo 2015; Peeraer 2015; Ramos 2007; Weissman 2011 |
| 13 December 2016 | New citation required but conclusions have not changed | There was no change to our conclusions |
History
Protocol first published: Issue 1, 2002 Review first published: Issue 1, 2008
| Date | Event | Description |
|---|---|---|
| 10 November 2008 | Amended | Converted to new review format. |
| 11 October 2007 | New citation required and conclusions have changed | Substantive amendment |
Acknowledgements
The authors would like to thank the Cochrane Gynaecology and Fertility editorial staff for their advice and support through the review process. The authors of the 2017 update thank Patrick Vanderkerchove for his contributions to previous versions of this review.
Appendices
Appendix 1. Cochrane Gynaecology and Fertility Specialised Register search strategy
Gynaecology and Fertility database search from inception to 13 December 2016
PROCITE platform
Keywords CONTAINS "cryopreservation" or "frozen embryo transfer" or "frozen embryos" or "frozen‐thawed cycle" or "frozen‐thawed embryo transfer" or "frozen‐thawed embryos" or "FET" or "cryopreserved embryos" or "cryopreserved‐thawed embryos" or "embryo vitrification" or "vitrification" or "vitrified" or "vitrified‐warmed embryos" or "frozen‐thawed" or "embryo vitrification" or Title CONTAINS "cryopreservation" or "frozen embryo transfer" or "frozen embryos" or "frozen‐thawed cycle" or "frozen‐thawed embryo transfer" or "frozen‐thawed embryos" or "FET" or "cryopreserved embryos" or "cryopreserved‐thawed embryos" or "embryo vitrification" or "vitrification" or "vitrified" or "vitrified‐warmed embryos" or "embryo vilification"
AND
Keywords CONTAINS "ovulation induction" or "endometrial preparation" or "*Clomiphene" or "clomiphene citrate" or "menotrophin" or "menotropin" or "HMG" or "human menopausal gonadotrophin" or "gonadotropin‐releasing hormone" or "gonadotropin releasing hormone agonist" or "gonadotrophin stimulation" or "Gonadotrophin releasing hormones" or "Gonadorelin" or "GnRh" or "GnRHa" or "GnRH a" or "GnRH agonist"or "GnRH agonists" or "GnRHa‐gonadotropin" or "rFSH" or "Fsh" or "FSH HMG" or "follicle stimulating hormone" or "follitropin" or "natural cycle" or "natural cycles" or "artificial cycle" or" modified natural cycle" or" estrogen" or "Estrogens" or "Progesterone" or "Estradiol" or "hormone therapy" or "hormone therapy estrogen" or "hormone replacement therapy" or "letrozole" or "tamoxifen" or "stimulated cycle" or "stimulation of endometrium embryo transfer" or "stimulation protocol" or Title CONTAINS "natural cycle"or "natural cycles" or "artificial cycle" or "endometrial preparation" or "*Clomiphene" or "modified natural cycle" (155 hits)
Appendix 2. CENTRAL Register of Studies Online (CRSO) search strategy
Searched 13th December 2016
Web platform
1 Cryopreservation/ (253) 2 (Cryopreserv$ adj7 embryo$).tw. (176) 3 (Cryopreserv$ adj7 blastocyst$).tw. (32) 4 freezing/ or vitrification/ (95) 5 (vitrification adj7 embryo$).tw. (45) 6 (vitrification adj7 blastocyst$).tw. (34) 7 (frozen adj5 embryo$).tw. (196) 8 (freez$ adj5 embryo$).tw. (55) 9 (freez$ adj5 blastocyst$).tw. (7) 10 (frozen adj5 blastocyst$).tw. (25) 11 FET.tw. (94) 12 (Cryo‐preserv$ adj7 embryo$).tw. (0) 13 (Cryo‐preserv$ adj7 blastocyst$).tw. (0) 14 or/1‐13 (728) 15 exp Ovulation Induction/ (1030) 16 ((ovar$ adj5 stimula$) or (ovulat$ adj5 induct$)).tw. (1683) 17 (endometri$ adj2 prepar$).tw. (89) 18 Clomiphene.tw. or Clomiphene/ (875) 19 clomid.tw. (26) 20 exp Menotropins/ (358) 21 (Menotropin$ or menopausal gonadotrop$ or HMG).tw. (1421) 22 exp Follicle Stimulating Hormone/ (1639) 23 (Follicle Stimulating Hormone or FSH).tw. (2873) 24 Gonadotropin‐Releasing Hormone/ad, ag, aa, de, pd, tu [Administration & Dosage, Agonists, Analogs & Derivatives, Drug Effects, Pharmacology, Therapeutic Use] (137) 25 Gonadotrop?in‐Releasing Hormone$.tw. (1194) 26 GnRH$.tw. (2051) 27 exp Estrogens/ (5949) 28 (?estrogen$ or ?estradiol).tw. (8709) 29 exp Progesterone/ or progesterone.tw. (4154) 30 (natural$ adj4 cycle$).tw. (157) 31 (artificial$ adj2 cycle$).tw. (34) 32 (cycle$ adj2 regimen$).tw. (266) 33 pituitary suppression.tw. (103) 34 human menopausal.tw. (401) 35 spontaneous ovulation.tw. (24) 36 HCG trigger$.tw. (52) 37 (stimulat$ adj3 cycle$).tw. (438) 38 (hormone$ adj2 replacement).tw. (2088) 39 (endometri$ adj2 stimulat$).tw. (87) 40 HRT.tw. (1234) 41 or/15‐40 (18545) 42 14 and 41 (208)
Appendix 3. MEDLINE search strategy
From inception to 13th December 2016
Ovid platform
1 Cryopreservation/ (21360) 2 (Cryopreserv$ adj7 embryo$).tw. (3130) 3 (Cryopreserv$ adj7 blastocyst$).tw. (445) 4 freezing/ or vitrification/ (23937) 5 (vitrifi$ adj5 embryo$).tw. (957) 6 (vitrifi$ adj5 blastocyst$).tw. (641) 7 (frozen adj5 embryo$).tw. (2517) 8 (freez$ adj5 embryo$).tw. (1110) 9 (freez$ adj5 blastocyst$).tw. (175) 10 (frozen adj5 blastocyst$).tw. (335) 11 FET.tw. (2142) 12 (Cryo‐preserv$ adj7 embryo$).tw. (10) 13 or/1‐12 (47595) 14 exp Ovulation Induction/ (11949) 15 ((ovar$ adj5 stimula$) or (ovulat$ adj5 induc$)).tw. (16099) 16 (endometri$ adj2 prepar$).tw. (460) 17 hormon$ regimen$.tw. (279) 18 Clomiphene.tw. or Clomiphene/ (6564) 19 clomid.tw. (175) 20 (Tamoxifen or Letrozole).tw. (23668) 21 aromatase inhibitor$.tw. (6705) 22 anti‐?estrogen$.tw. (2427) 23 exp Menotropins/ (3127) 24 (Menotropin$ or menopausal gonadotrop$ or HMG).tw. (15378) 25 exp Follicle Stimulating Hormone/ (37398) 26 (Follicle Stimulating Hormone or FSH or rFSH or rhFSH).tw. (39284) 27 Gonadotropin‐Releasing Hormone/ad, ag, aa, de, pd, tu [Administration & Dosage, Agonists, Analogs & Derivatives, Drug Effects, Pharmacology, Therapeutic Use] (15972) 28 Gonadotrop?in‐Releasing Hormone$.tw. (16126) 29 GnRH$.tw. (21800) 30 exp Estrogens/ (162202) 31 (?estrogen$ or ?estradiol).tw. (177832) 32 exp Progesterone/ or progesterone.tw. (108635) 33 exogenous steroid$.tw. (506) 34 (natural$ adj4 cycle$).tw. (2642) 35 (artificial$ adj3 cycle$).tw. (482) 36 (cycle$ adj2 regimen$).tw. (278) 37 pituitary suppression.tw. (320) 38 human menopausal.tw. (2109) 39 spontaneous ovulation.tw. (408) 40 (HCG adj3 trigger$).tw. (263) 41 hormone therapy.tw. (12827) 42 (stimulat$ adj3 cycle$).tw. (3805) 43 (hormone$ adj2 replacement).tw. (15866) 44 (endometri$ adj2 stimulat$).tw. (547) 45 (HRT or HT).tw. (70350) 46 or/14‐45 (464462) 47 randomized controlled trial.pt. (469833) 48 controlled clinical trial.pt. (95075) 49 randomized.ab. (405868) 50 randomised.ab. (81587) 51 placebo.tw. (197475) 52 clinical trials as topic.sh. (189503) 53 randomly.ab. (286433) 54 trial.ti. (179694) 55 (crossover or cross‐over or cross over).tw. (76212) 56 or/47‐55 (1211177) 57 exp animals/ not humans.sh. (4669484) 58 56 not 57 (1117076) 59 13 and 46 and 58 (262)
Appendix 4. Embase search strategy
From inception to 13th December 2016
Ovid platform
1 cryopreservation/ (33757) 2 (Cryopreserv$ adj7 embryo$).tw. (4445) 3 (Cryo‐preserv$ adj7 embryo$).tw. (33) 4 (Cryopreserv$ adj7 blastocyst$).tw. (826) 5 freezing/ or vitrification/ (35415) 6 (vitrifi$ adj5 embryo$).tw. (1716) 7 (vitrifi$ adj5 blastocyst$).tw. (1242) 8 (frozen adj5 embryo$).tw. (3976) 9 (freez$ adj5 embryo$).tw. (1588) 10 (freez$ adj5 blastocyst$).tw. (283) 11 (frozen adj5 blastocyst$).tw. (688) 12 FET.tw. (2754) 13 freeze thawing/ or freezing/ (36470) 14 vitrification/ (4402) 15 or/1‐14 (71558) 16 exp ovulation induction/ (13019) 17 ((ovar$ adj5 stimula$) or (ovulat$ adj5 induc$)).tw. (20432) 18 (endometri$ adj2 prepar$).tw. (701) 19 hormon$ regimen$.tw. (316) 20 Clomiphene.tw. or Clomiphene/ (9591) 21 clomid.tw. (1068) 22 (Tamoxifen or Letrozole).tw. (30044) 23 aromatase inhibitor$.tw. (9118) 24 exp human menopausal gonadotropin/ (9848) 25 (Menotropin$ or menopausal gonadotrop$ or HMG).tw. (17974) 26 exp follitropin/ (55231) 27 (Follicle Stimulating Hormone or FSH or rFSH or rhFSH).tw. (46559) 28 gonadorelin/ (35623) 29 Gonadotrop?in‐Releasing Hormone$.tw. (16598) 30 GnRH$.tw. (25145) 31 exp estrogen/ (255742) 32 (?estrogen$ or ?estradiol).tw. (194821) 33 exp progesterone/ (85120) 34 exp Progesterone/ or progesterone.tw. (117583) 35 (natural$ adj2 cycle$).tw. (2548) 36 (artificial$ adj2 cycle$).tw. (425) 37 (cycle$ adj2 regimen$).tw. (523) 38 pituitary suppression.tw. (409) 39 human menopausal.tw. (2323) 40 spontaneous ovulation.tw. (478) 41 (HCG adj3 trigger$).tw. (693) 42 (stimulat$ adj3 cycle$).tw. (4793) 43 exogenous steroid$.tw. (560) 44 exogenous steroid$.tw. (560) 45 (hormone adj2 therap$).tw. (33347) 46 (endometri$ adj2 stimulat$).tw. (682) 47 or/16‐46 (491845) 48 15 and 47 (5220) 49 Clinical Trial/ (1004097) 50 Randomized Controlled Trial/ (465768) 51 exp randomization/ (83937) 52 Single Blind Procedure/ (27791) 53 Double Blind Procedure/ (137638) 54 Crossover Procedure/ (54096) 55 Placebo/ (323380) 56 Randomi?ed controlled trial$.tw. (150645) 57 Rct.tw. (22599) 58 random allocation.tw. (1637) 59 randomly allocated.tw. (26733) 60 allocated randomly.tw. (2210) 61 (allocated adj2 random).tw. (844) 62 Single blind$.tw. (18743) 63 Double blind$.tw. (173480) 64 ((treble or triple) adj blind$).tw. (657) 65 placebo$.tw. (248420) 66 prospective study/ (389403) 67 or/49‐66 (1790257) 68 case study/ (93504) 69 case report.tw. (324719) 70 abstract report/ or letter/ (989413) 71 or/68‐70 (1398438) 72 67 not 71 (1739458) 73 (exp animal/ or animal.hw. or nonhuman/) not (exp human/ or human cell/ or (human or humans).ti.) (5736041) 74 72 not 73 (1678022) 75 48 and 74 (720)
Appendix 5. PsycINFO search strategy
From inception to 13th December 2016
Ovid platform
1 exp reproductive technology/ (1610) 2 (frozen adj5 embryo$).tw. (27) 3 ((frozen‐thawed or cryopreserv$) adj5 embryo$).tw. (28) 4 exp Embryo/ (1632) 5 FET.tw. (56) 6 ((embry$ adj5 transf$) or embryo replacement or embryo deposition).tw. (272) 7 or/1‐6 (3386) 8 exp Ovulation/ (346) 9 ((ovar$ adj5 stimula$) or (ovulat$ adj5 induct$)).tw. (172) 10 (endometri$ adj2 prepar$).tw. (1) 11 Clomiphene.tw. (46) 12 clomid.tw. (1) 13 (Menotropin$ or menopausal gonadotrop$ or HMG).tw. (205) 14 (Follicle Stimulating Hormone or FSH).tw. (659) 15 exp Gonadotropic Hormones/ (4043) 16 Gonadotropin‐Releasing Hormone$.tw. (689) 17 GnRH$.tw. (887) 18 exp Estrogens/ (6065) 19 Estrogen$.tw. (7196) 20 exp Progesterone/ (2010) 21 Progesterone.tw. (3831) 22 oestrogen$.tw. (690) 23 (natural$ adj2 cycle$).tw. (127) 24 (artificial$ adj2 cycle$).tw. (28) 25 (cycle$ adj2 regimen$).tw. (5) 26 or/8‐25 (15793) 27 7 and 26 (80) 28 random.tw. (48495) 29 control.tw. (375857) 30 double‐blind.tw. (20313) 31 clinical trials/ (10039) 32 placebo/ (4746) 33 exp Treatment/ (669890) 34 or/28‐33 (1034673) 35 27 and 34 (36)
Appendix 6. CINAHL search strategy
From inception to 13th December 2016
EBSCO platform
| # | Query | Results |
| S48 | S35 AND S47 | 30 |
| S47 | S36 OR S37 OR S38 OR S39 OR S40 OR S41 OR S42 OR S43 OR S44 OR S45 OR S46 | 1,097,934 |
| S46 | TX allocat* random* | 5,898 |
| S45 | (MH "Quantitative Studies") | 15,110 |
| S44 | (MH "Placebos") | 9,934 |
| S43 | TX placebo* | 42,609 |
| S42 | TX random* allocat* | 5,898 |
| S41 | (MH "Random Assignment") | 42,016 |
| S40 | TX randomi* control* trial* | 115,809 |
| S39 | TX ( (singl* n1 blind*) or (singl* n1 mask*) ) or TX ( (doubl* n1 blind*) or (doubl* n1 mask*) ) or TX ( (tripl* n1 blind*) or (tripl* n1 mask*) ) or TX ( (trebl* n1 blind*) or (trebl* n1 mask*) ) | 866,076 |
| S38 | TX clinic* n1 trial* | 196,374 |
| S37 | PT Clinical trial | 79,958 |
| S36 | (MH "Clinical Trials+") | 207,314 |
| S35 | S12 AND S34 | 107 |
| S34 | S13 OR S14 OR S15 OR S16 OR S17 OR S18 OR S19 OR S20 OR S21 OR S22 OR S23 OR S24 OR S25 OR S26 OR S27 OR S28 OR S29 OR S30 OR S31 OR S32 OR S33 | 20,839 |
| S33 | TX(stimulat* N3 cycle*) | 229 |
| S32 | TX spontaneous* ovulat* | 23 |
| S31 | TX pituitary suppression | 79 |
| S30 | TX(cycle* N2 regimen*) | 91 |
| S29 | TX(artificial* N2 cycle*) | 8 |
| S28 | TX(natural* N2 cycle*) | 120 |
| S27 | TX Progesterone | 4,593 |
| S26 | (MM "Progesterone") | 1,105 |
| S25 | TX estrogen* or TX oestrogen* | 14,913 |
| S24 | (MM "Estrogens") | 2,993 |
| S23 | TX GnRH* | 499 |
| S22 | TX Gonadotrop?in‐Releasing Hormone* | 90 |
| S21 | (MM "Gonadorelin") OR (MM "Pituitary Hormone Release Inhibiting Hormones") | 479 |
| S20 | TX(Follicle Stimulating Hormone or FSH) | 1,763 |
| S19 | (MM "Follicle‐Stimulating Hormone") | 262 |
| S18 | TX(Menotropin* or menopausal gonadotrop* or HMG) | 722 |
| S17 | TX Clomiphene or TX clomid | 360 |
| S16 | (MM "Clomiphene") | 121 |
| S15 | TX(endometri* N2 prepar*) | 18 |
| S14 | TX((ovar* N5 stimula*) or (ovulat* N5 induct*)) | 877 |
| S13 | (MM "Ovulation Induction") | 260 |
| S12 | S1 OR S2 OR S3 OR S4 OR S5 OR S6 OR S7 OR S8 OR S9 OR S10 OR S11 | 1,100 |
| S11 | TX FET | 101 |
| S10 | TX(frozen N5 blastocyst*) | 5 |
| S9 | TX(freez* N5 blastocyst*) | 0 |
| S8 | TX(freez* N5 embryo*) | 51 |
| S7 | TX(frozen N5 embryo*) | 129 |
| S6 | TX(vitrification N7 blastocyst*) | 8 |
| S5 | TX(vitrification N7 embryo*) | 18 |
| S4 | (MM "Freezing") | 136 |
| S3 | TX(Cryopreserv* N7 blastocyst*) | 12 |
| S2 | TX(Cryopreserv* N7 embryo*) | 170 |
| S1 | (MM "Cryopreservation+") | 668 |
Data and analyses
Comparison 1. Natural cycle FET versus HT FET.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Clinical pregnancy rate per woman | 1 | 100 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.06 [0.40, 2.80] |
| 2 Multiple pregnancy rate per woman | 1 | 21 | Odds Ratio (M‐H, Fixed, 95% CI) | 2.48 [0.09, 68.14] |
Comparison 2. Natural cycle FET versus HT + GnRHa FET.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Live birth rate per woman | 1 | 159 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.77 [0.39, 1.53] |
| 2 Clinical pregnancy rate per woman | 1 | 159 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.87 [0.45, 1.71] |
| 3 Multiple pregnancy rate per woman | 1 | 159 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.58 [0.13, 2.50] |
Comparison 3. Natural cycle FET versus modified natural cycle FET (HCG trigger).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Live birth rate per woman | 1 | 60 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.55 [0.16, 1.93] |
| 2 Miscarriage rate per woman | 1 | 168 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.20 [0.01, 4.13] |
| 3 Ongoing pregnancy rate per woman | 1 | 168 | Odds Ratio (M‐H, Fixed, 95% CI) | 2.44 [1.03, 5.76] |
| 4 Clinical pregnancy rate per woman | 1 | 60 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.0 [0.32, 3.14] |
Comparison 4. Modified natural cycle FET (HCG trigger) versus HT FET.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Live birth rate per woman | 1 | 959 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.34 [0.88, 2.05] |
| 2 Ongoing pregnancy rate per woman | 1 | 959 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.21 [0.80, 1.83] |
| 3 Clinical pregnancy rate per woman | 1 | 959 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.22 [0.87, 1.70] |
| 4 Cycle cancellation rate per woman | 1 | 959 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.70 [0.52, 0.95] |
| 5 Endometrial thickness | 1 | 959 | Mean Difference (IV, Fixed, 95% CI) | 0.10 [‐0.13, 0.33] |
Comparison 5. Modified natural cycle FET (HCG trigger) versus HT + GnRH‐a FET.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Live birth rate per woman | 1 | 236 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.11 [0.66, 1.87] |
| 2 Miscarriage rate per woman | 1 | 236 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.74 [0.25, 2.19] |
| 3 Clinical pregnancy rate per woman | 1 | 236 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.07 [0.64, 1.78] |
| 4 Endometrial thickness | 1 | 236 | Mean Difference (IV, Fixed, 95% CI) | ‐0.20 [‐0.54, 0.14] |
Comparison 6. HT FET versus HT + GnRH‐a.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Live birth rate per woman | 1 | 75 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.10 [0.04, 0.30] |
| 2 Miscarriage rate per woman | 6 | 991 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.64 [0.37, 1.12] |
| 3 Ongoing pregnancy rate per woman | 1 | 106 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.72 [0.61, 4.85] |
| 4 Clinical pregnancy rate per woman | 5 | 872 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.90 [0.65, 1.25] |
| 5 Cycle cancellation rate per woman | 3 | 636 | Odds Ratio (M‐H, Fixed, 95% CI) | 2.73 [0.79, 9.38] |
| 6 Endometrial thickness | 3 | 625 | Mean Difference (IV, Fixed, 95% CI) | ‐0.16 [‐0.41, 0.09] |
Comparison 7. HT FET versus FSH FET.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Clinical pregnancy rate per woman | 1 | 175 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.09 [0.45, 2.62] |
| 2 Cycle cancellation rate per woman | 1 | 175 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.99 [0.49, 2.00] |
| 3 Endometrial thickness | 1 | 175 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [‐0.31, 0.31] |
Comparison 8. HMG FET versus clomiphene + HMG FET.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Live birth rate per woman | 1 | 209 | Odds Ratio (M‐H, Fixed, 95% CI) | 2.49 [1.07, 5.80] |
| 2 Miscarriage rate per woman | 1 | 209 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.33 [0.35, 5.09] |
| 3 Multiple pregnancy rate per woman | 1 | 209 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.41 [0.31, 6.48] |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Azimi Nekoo 2015.
| Methods | 2‐arm, parallel RCT 2 ART centres |
|
| Participants | 176 women undergoing FET Inclusion criteria: infertile women (male factor) aged 20‐37 years who had regular menstrual cycles and had previously undergone IVF or ICSI with the same induction protocol with embryo cryopreservation. Exclusion criteria: not reported Baseline characteristics were similar in the 2 groups |
|
| Interventions |
HT (83 women) Women received oral estradiol valerate 4 mg/d from day 2‐day 5 and 6 mg/d from day 6 to the day of the pregnancy test. In day 13 of cycle, an US examination was performed. After US confirmation of endometrial thickness (8 mm) and no ovarian activity, progesterone suppository 800 mg/d was added. The dose of estradiol was increased to 8 mg/d if endometrial thickness was < 8 mm. 2 or 3 embryos were transferred via transcervical route 48 h after the beginning of progesterone administration. In addition to HT, steroid supplementation was commenced without prior pituitary suppression. HT plus GnRHa (93 women) In addition to HT, triptorelin 3.75 mg IM, as a depot GnRHa was administered in the mid‐luteal phase (day 21) of previous cycle. |
|
| Outcomes | Miscarriage rate per woman Clinical pregnancy rate per woman |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated random sequence |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported but non‐blinding of outcome assessors may not have affected some of the outcome measures as they were objectively assessed |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | All women randomized were included in data analysis |
| Selective reporting (reporting bias) | Unclear risk | Insufficient information to make a conclusive judgement |
| Other bias | Low risk | Baseline characteristics were similar in the 2 groups |
Cattoli 1994.
| Methods | Single‐centre parallel RCT | |
| Participants | 100 women Inclusion criteria: women who had cryopreservation of embryos following IVF treatment Exclusion criteria: not stated Mean number of transferred embryos per woman: similar, HT group 2.7 ± 0.0 (1‐4 embryos), natural cycle group 2.8 ± 0.8 (1‐5 embryos) |
|
| Interventions |
FET HT cycle (56 women (64 cycles)) Natural cycle FET (44 women (50 cycles)) |
|
| Outcomes | Clinical pregnancy rate per woman Multiple pregnancy rate per pregnancy |
|
| Notes | This was an abstract. The review authors contacted the first author requesting more data but as this study was not published and was presented in 1994, the study author could not supply any more data. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Process involved in random sequence generation not reported |
| Allocation concealment (selection bias) | Unclear risk | Methods used in allocation concealment not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | No information provided on the blinding of participants and personnel |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | No reports on number of missing outcome data and reasons for drop out or exclusion from study |
| Selective reporting (reporting bias) | Unclear risk | Insufficient information reported to arrive at a judgement |
| Other bias | Unclear risk | Insufficient information reported to arrive at a judgement |
Dal Prato 2002.
| Methods | Single‐centre parallel RCT | |
| Participants | 296 women Inclusion criteria: women with functioning ovaries who had surplus embryos frozen following fresh ET in a cycle of IVF or ICSI Exclusion criteria: women with only 1 frozen embryo or women who had all their embryos cryopreserved because of OHSS Number of transferred embryos per woman: similar, 2.1 ± 0.7 in HT group and 2.1 ± 0.6 in HT plus GnRHa group % embryos that survived freezing‐thawing: similar, 76.6% in HT group and 77.1% in HT plus GnRHa Baseline characteristics were similar in the 2 groups Infertility aetiology: similar (tubal, idiopathic and male factors) |
|
| Interventions |
HT plus GnRHa group (146 women) Long‐acting triptorelin, 3.75 mg IM starting on mid‐luteal phase of the cycle. E2 transdermal patches 100 μg was started from day 1‐day 7 of menstrual cycle, 200 μg from day 8‐day 10, and 300 μg from day 11 onwards. Patches were replaced every 84 h. In women with endometrium of < 8 mm thickness, E2 dose was increased to 400 μg. If endometrial thickness was ≥ 8 mm with no evidence of preovulatory follicles, corpus luteum or hyperechoic endometrium, progesterone in oil was administered IM at a dose of 100 mg E2 and progesterone treatment was at least continued until pregnancy test was done at 15 days after ET HT group (150 women) E2 patches were started from day 1 of cycle as 200 μg then increased to 300 μg on day 8 Progesterone: as in HT plus GnRHa group Cycle monitoring: by US aiming for endometrial thickness of ≥ 8 mm prior to FET |
|
| Outcomes | Clinical pregnancy rate per woman Miscarriage rate per pregnancy Cycle cancellation rate per woman Endometrial thickness on day of starting progesterone |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Stated that randomization performed using sealed envelopes but the process involved in generating the sequence not reported |
| Allocation concealment (selection bias) | Unclear risk | Allocation was said to have been concealed in sealed envelopes which were sequentially numbered. Not stated whether the envelopes were opaque |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Stated that contents of envelopes were known only to medical staff who were not involved with the trial. Not reported whether the outcome assessor was blinded; however non‐blinding was likely to influence some of the outcome measures |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Stated that statistical analysis performed on an ITT basis which included all randomized women who started progesterone therapy, but this was not the case from the results tables. Nevertheless, the review authors applied ITT to study data |
| Selective reporting (reporting bias) | Low risk | Although the trial protocol was not available for review, data were reported on the prespecified outcomes in the methods section. |
| Other bias | Low risk | No other potential source of within‐study bias found. |
Davar 2007.
| Methods | Single‐centre, 2‐arm, parallel RCT | |
| Participants | 60 women undergoing FET cycle Inclusion: women < 30 years, had previously undergone IVF or ICSI with embryo cryopreservation, had regular menstrual cycles Exclusion criteria: not explicitly stated Baseline characteristics were similar in both groups. |
|
| Interventions |
HT (30 women) Estradiol valerate orally at 2 mg/d from day 1‐day 4; 4 mg/d from day 5‐day 9; and 6 mg/d from day 10 onwards up to the day of pregnancy test. If the endometrial thickness was > 8 mm, progesterone 100 mg/d IM in oil and transfer of 2 frozen‐thawed embryos was performed on day 2 after progesterone administration HT plus GnRHa (30 women) As for HT alone. In addition to HT, buserelin acetate (a GnRHa) 0.5 mg SC was administered in the mid‐luteal phase (day 21) of the menstrual cycle and was continued until day 11 of cycle |
|
| Outcomes | Clinical pregnancy rate per woman randomized Miscarriage rate per woman randomized |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated random sequence |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported but non‐blinding of outcome assessors may not have affected some of the outcome measures as they were objectively assessed. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Number of participants analyzed was same as the number of participants randomized |
| Selective reporting (reporting bias) | Low risk | Outcomes measured were prespecified in methods section |
| Other bias | Low risk | Baseline characteristics similar in both groups |
Eftekhar 2012.
| Methods | Single‐centre, 2‐arm, parallel RCT | |
| Participants | 130 infertile women who were candidates for FET Inclusion criteria: women who had undergone IVF or ICSI with cryopreservation of excess embryos and fresh cycles with implantation failure. Exclusion criteria: aged > 38 years, BMI > 30 kg/m2, history of endocrine disorders and severe endometriosis Baseline characteristics similar in both groups |
|
| Interventions |
HT (65 women) All women received estradiol valerate 6 mg/d orally from day 2 of menstrual cycle and progesterone in oil 100 mg IM when the endometrial thickness reached 8 mm ET was performed 3 days after the beginning of progesterone administration. Estradiol and progesterone were continued until the 10th week of gestation HT plus HCG (65 women) As for HT and in addition, women received an HCG 5000 IU injection on the first day of progesterone administration and the day of ET. Embryo thawing was performed 2 days after the first progesterone injection. Embryos were transferred 1 day after thawing using a Labotect catheter (Labotect, Gotting, Germany). |
|
| Outcomes | Miscarriage Ongoing pregnancy Clinical pregnancy |
|
| Notes | No outcome data available | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated random sequence |
| Allocation concealment (selection bias) | Unclear risk | No information reported on allocation concealment |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | No information reported but non‐blinding of outcome assessors may not have affected some of the outcome measures as they were objectively assessed. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | No information reported on withdrawals or losses to follow‐up, or both |
| Selective reporting (reporting bias) | Low risk | Outcome measures were prespecified in the methods section |
| Other bias | Low risk | Baseline characteristics similar in both group |
El‐Toukhy 2004.
| Methods | Single‐centre parallel RCT | |
| Participants | 234 women Inclusion criteria: women with functioning ovaries and regular menstrual cycles who had surplus embryos frozen following fresh ET in a cycle of IVF or ICSI Exclusion criteria: FET originating from donated oocytes and women with irregular cycles. Number of transferred embryos per woman: HT group 2.2 ± 0.6, HT plus GnRHa group 2.3 ± 0.6 Baseline characteristics were similar in the 2 groups Infertility cause: similar (variety of causes) |
|
| Interventions |
HT group (117 women) Estradiol valerate 6 mg/d/tablet started on day 1 of menstruation HT plus GnRHa group (117 women) Buserelin nasal spray was started in mid‐luteal phase (day 21) of cycle. On day 1 of subsequent cycle, oral E2 initiated as 6 mg/d in 2 divided doses In both groups, E2 dose continued for 12‐14 days then endometrial thickness was assessed by US. If endometrial thickness was < 8 mm, E2 dose was increased to 8 mg/d for further 7‐12 days Once 8 mm endometrial thickness had been confirmed, micronised progesterone pessaries 400 mg twice daily were commenced. GnRHa was stopped at this stage. There was no endocrine or US monitoring of ovulation. Embryos were transferred on day 3 of progesterone initiation. Progesterone use was for 2 weeks following FET. Pregnant women were advised to continue E2 and progesterone supplement up to 12th gestational week. |
|
| Outcomes | Clinical pregnancy rate per woman Miscarriage rate per clinical pregnancy Live birth rate per woman Cycle cancellation rate per woman Endometrial thickness prior to FET |
|
| Notes | Miscarriage rate per clinical pregnancy was used to calculate miscarriage rate per woman. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated random list |
| Allocation concealment (selection bias) | Unclear risk | Methods used in concealing the allocation not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | No reports on blinding of participants and personnel, although blinding of outcome assessor could not have influenced some of the reported outcome measures |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Analysis reported to have been performed on an ITT basis. ITT was not fully defined but it was apparent from 1 of the result tables that all participants randomized were included in data analysis (other aspects of ITT could not be verified) |
| Selective reporting (reporting bias) | Low risk | Data reported on all the outcomes prespecified in the methods section. |
| Other bias | Low risk | No other potential source of bias found. |
Fatemi 2010.
| Methods | Single‐centre, parallel RCT Computer‐generated, not concealed randomization Undertaken 1 October 2007‐30 November 2008 Power calculation performed |
|
| Participants | 168 women Inclusion criteria: maternal age ≤ 36 years, regular menstrual cycles (25‐34 days), previous conventional IVF or ICSI Exclusion criteria: use of testicular sperm for ICSI, early (day 3) FSH levels ≤ 12 IU/L, American Society for Reproductive Medicine grades ≥ 3 for endometriosis and BMI ≥ 29 kg/m2 Women allowed to participate in the study only once |
|
| Interventions |
HCG‐induced natural cycle FET (63 women) Spontaneous LH surge natural cycle FET (61 women) |
|
| Outcomes | Ongoing pregnancy rate per woman Biochemical pregnancy rate per woman Miscarriage rate per woman Number of visits to clinic |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated random list |
| Allocation concealment (selection bias) | High risk | Reported that allocation was not concealed |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Some of the personnel were blinded (pre‐ET US examination); no information on blinding of either participants or outcome assessor and non‐binding of outcome assessor could influence some of the outcome measures |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Reason for attrition stated and similar between groups; however, number of missing outcome data differed between groups |
| Selective reporting (reporting bias) | Low risk | Outcome prespecified in the methods section was reported |
| Other bias | Unclear risk | Although it was reported that participants' characteristics and number of embryos transferred were similar in both groups, the groups differed in terms of embryo quality, participants' number of visits and number of days until LH‐HCG surge |
Greco 2016.
| Methods | Single‐centre, 2‐arm, parallel RCT Computer‐generated random sequence, unclear method of allocation concealment |
|
| Participants | 236 women Inclusion criteria: maternal age < 42 years, regular menstrual cycle, normal intrauterine cavity, the presence of at least one vitrified euploid blastocyst obtained after ICSI followed by preimplantation genetic diagnosis by aCGH, and a consent to undergo a frozen‐thawed single transfer in a modified‐natural cycle (NC) or after hormonal endometrium preparation (AC_FET) Exclusion criteria: ovulation disorders, BMI > 29 kg/m2, endometriosis grade ≥ III according to the American Fertility Society criteria, and the use of testicular sperm for ICSI |
|
| Interventions |
Modified natural cycle FET (natural cycle with HCG trigger) Modified natural cycle: natural cycle was modified by triggering of ovulation using HCG injection then supporting luteal phase by progesterone injections Artificial cycle FET (HT + GnRHa suppression) GnRH agonist injection was started in preceding cycle to prevent follicular development Then estrogen tablets accompanied by progesterone injections were used |
|
| Outcomes | Live birth rate per woman Miscarriage rate per woman Clinical pregnancy rate per woman Endometrial thickness |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Described as, "Computer‐generated, not cancelled simple randomization" |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Blinding was not used due to the nature of the interventions; no information on outcome assessment; however, non‐blinding of outcome assessors may not have affected some of the outcome measures as they were objectively assessed. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Proportions of withdrawals and reasons for withdrawals or exclusion were fairly balanced between the two groups. |
| Selective reporting (reporting bias) | Unclear risk | There were discrepancies in the outcomes between pre‐specified and reported outcomes |
| Other bias | Unclear risk | Insuficient information to make a conclusive judgement |
Groenewoud 2016.
| Methods | Multi‐centre, 2‐arm, parallel RCT (17 fertility clinics) Computer‐generated random sequence, unclear method of allocation concealment |
|
| Participants | 959 women Inclusion criteria: women aged 18‐40 years, ovulatory cycle of 26‐35 days' duration and transferred frozen embryos originated from participant’s first three IVF or ICSI cycles Exclusion criteria: contraindications to estrogen or progesterone supplementation and anatomical uterine anomalies. Participants undergoing a gamete donor procedure (except those with a genetic disease) |
|
| Interventions |
Modified natural FET cycle (natural cycle, HCG triggered) Serial US scan from day 10‐12 of cycle. Once dominant follicle reached 16‐20 mm, HCG injection was given SC then FET day was decided Artificial FET cycle (HT FET) 2 mg oral estrogen was used from day 1 or 2 of cycle, scan was done after 12‐14 days Once endometrial thickness reached ≥ 8 mm and in the absence of a dominant follicle, FET was decided |
|
| Outcomes | Live birth rate per woman Ongoing pregnancy rate per woman Clinical pregnancy rate per woman Cycle cancellation rate per woman Endometrial thickness |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Authors described randomization as, "a web‐based randomization module using a computerized list was used" |
| Allocation concealment (selection bias) | Unclear risk | Not explicitly reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | It was reported that "The nature of the treatment interventions precluded blinding of patients and treating physicians." No information on outcome assessment. However, non‐blinding of outcome assessors may not have affected some of the outcome measures as they were objectively assessed. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Proportions of withdrawals were high and not balanced between the two groups (20% vs 27%). Data were not analyzed on true ITT basis for all outcomes |
| Selective reporting (reporting bias) | Low risk | All outcome measures were pre‐specified in the methods section |
| Other bias | Low risk | No other potential source of bias found |
Karimzadeh 2012.
| Methods | Single‐centre, 2‐arm, parallel RCT | |
| Participants | 70 women undergoing FET Inclusion criteria: not stated Exclusion criteria: not stated Baseline demographic and infertility characteristics similar in both groups |
|
| Interventions |
Natural cycle (36 women) Women did not receive any HT. When mature follicle reached a mean diameter of 18 mm and endometrial thickness > 8 mm, HCG 10,000 IU administrated and FET was done 4 days after HCG injection HT (34 women) Women received oral estradiol valerate 6 mg/d from day 2 of menstrual cycle and progesterone 100 mg IM in oil. When endometrial thickness reached > 8 mm, FET was done 72 hours after beginning of progesterone administration, and estradiol and progesterone were continued until the 12th gestational week |
|
| Outcomes | Miscarriage per ET Ongoing pregnancy per ET Clinical pregnancy per ET |
|
| Notes | Data reported as per 'embryo transfer' and number of embryos transferred was not equivalent to the number of women randomized | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Not reported |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported but non‐blinding of outcome assessors may not have affected some of the outcome measures as they were objectively assessed. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Proportions of withdrawals/losses to follow‐up and reasons for withdrawal not reported; analysis was per ET |
| Selective reporting (reporting bias) | Unclear risk | Insufficient information to make a conclusive judgement |
| Other bias | Low risk | Baseline demographic and infertility characteristics similar in both groups |
Loh 2001.
| Methods | Single‐centre, 3‐arm, parallel RCT | |
| Participants | 130 women (156 FET cycles) Baseline characteristics were similar in the 2 groups Infertility cause: variety of causes |
|
| Interventions |
HT group (44 women (52 cycles)) E2 was used in graduated dose (2 mg‐8 mg) until endometrial thickness was ≥ 8 mm Progesterone pessaries for 2 days before FET. E2 and progesterone continued until day 17 post FET Clomiphene group (55 women (67 cycles)) Low‐dose clomiphene HT plus GnRHa group (31 women (37 cycles)) GnRHa then E2 and progesterone (as per HT group) |
|
| Outcomes | Clinical pregnancy rate per cycle Endometrial thickness |
|
| Notes | The study was an abstract; however, the first author provided some study details but it was still not possible to obtain the pregnancy rate per woman; there were multiple cycles per woman and total number of cycles were not equivalent to number of women randomized. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Method used in generating sequence not reported |
| Allocation concealment (selection bias) | Low risk | Allocation was said to have been concealed using sequentially‐numbered, sealed, opaque envelopes |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Reported that intervention providers were blinded to endometrial preparation; however, participants and outcome assessors were not blinded. Non‐blinding of outcome assessors could affect some of the outcome measures |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | There were no reports on attrition or reasons for withdrawal |
| Selective reporting (reporting bias) | Unclear risk | There was insufficient information to determine whether outcomes were selectively reported |
| Other bias | Unclear risk | Insufficient information provided to assess possible sources of other bias |
Mounce 2015.
| Methods | Single‐centre, 2‐arm, parallel, open‐label RCT | |
| Participants | 159 women undergoing FET cycle Inclusion criteria: women were eligible to participate if they were aged < 40 years at the time their embryos were frozen, had at least 1 blastocyst or 2 cleavage‐stage embryos in storage, had regular ovulatory cycles and ≤ 2 previous FET cycles Exclusion criteria: not reported Baseline demographic and infertility characteristics similar between the 2 groups |
|
| Interventions |
Natural cycle (80 women) Women had a US assessment between day 10 and day 13 of their cycle to confirm follicular growth and endometrial thickness, followed by additional US monitoring in subsequent days if necessary. On detection of LH surge, unit was informed and ET scheduled for up to 1 week later, depending on the stage of embryo development at freezing (i.e. day‐2 cleavage embryos, day‐3 cleavage embryos or day‐5 blastocysts). HT plus GnRHa (79 women) Women commenced daily nasal administration of the GnRHa nafarelin 400 mg twice daily on day 21 of their menstrual cycle until advised to stop, depending on stage of embryo, before the ET procedure. Once down‐regulation was confirmed, women started oral administration of E2 2 mg/d for endometrial preparation, which was increased by a step‐up protocol to 6 mg/d. Women commenced luteal support via vaginal administration of progesterone pessaries 400 mg twice daily according to the proposed day of FET; women with embryos cryopreserved at the cleavage day‐2 stage started pessaries 2 days before the transfer day; women with cryopreserved day‐3 embryos started pessaries 3 days before; and women with cryopreserved blastocysts started their pessaries 5 days before. ET was correspondingly scheduled for up to 1 week after the scan, depending on embryo stage |
|
| Outcomes | Live birth per woman Clinical pregnancy rate per woman Multiple pregnancy rate per woman |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Randomisation carried out using a minimisation algorithm |
| Allocation concealment (selection bias) | Unclear risk | No information reported on allocation concealment |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Open‐label RCT but non‐blinding of outcome assessors may not have affected some of the outcome measures as they were objectively assessed. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Data were analyzed on the basis of ITT, i.e. all women randomized were included in data analysis. |
| Selective reporting (reporting bias) | Low risk | Outcome measures were prespecified in the methods section. |
| Other bias | Low risk | Baseline demographic and infertility characteristics were similar between the 2 groups. |
Peeraer 2015.
| Methods | Single‐centre, 2‐arm parallel, open‐label RCT | |
| Participants | 472 women undergoing FET Inclusion criteria: women undergoing FET were eligible for the study when they had a regular cycle (between 21 and 35 days) and were aged 21‐45 years Exclusion criterion: FET after PGD Participants were similar in demographic and infertility characteristics at baseline |
|
| Interventions |
Natural cycle (235 women) Women underwent a first pelvic US and blood analysis around day 10‐day 12 of the menstrual cycle. HCG administered when the leading follicle had a mean diameter of ≥ 17 mm and endometrial thickness ≥ 7 mm with serum estradiol levels preferably 150 ng/L. HMG cycle (237 women) Women started SC injections of gonadotrophins (follitropin plus LH) on day 2 of the menstrual cycle. Starting dose of gonadotrophins (37 or 75 IU) determined by the treating clinician, based on woman's age, BMI, basal serum FSH (days 2‐5) and (if applicable) response to previous ovarian stimulation. On day 6 or 7 of the menstrual cycle, a first US and serum hormonal analysis (E2, progesterone, LH, FSH) performed. Based on these results, dose of gonadotrophins could be adjusted if needed. In both natural cycle FET and HMG FET cycle groups, the follicular response was monitored by regular vaginal US and serum hormonal analysis. ET was performed the same way in both groups. |
|
| Outcomes | Live birth per ET Ongoing pregnancy per ET Clinical pregnancy per ET Endometrial thickness |
|
| Notes | Outcome data reported as per 'embryo transfer cycle' (dichotomous data) or not clearly stated and there were multiple transfers per woman | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Process of randomization not sufficiently explained. |
| Allocation concealment (selection bias) | Low risk | Allocations concealed in opaque sealed envelope. |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Described as an open‐label trial, thus both participants and personnel were aware of the treatment protocols. However, non‐blinding of outcome assessors may not have affected some of the outcome measures as they were objectively assessed. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Analysis was based on per cycle transferred. |
| Selective reporting (reporting bias) | Low risk | Outcome measures were prespecified in the methods section. |
| Other bias | Low risk | Participants similar in demographic and infertility characteristics at baseline. |
Ramos 2007.
| Methods | 2‐arm RCT Undertaken January 2006‐March 2007 IVI Sevilla in Seville, Spain |
|
| Participants | 119 women with functioning ovaries who were having FET in artificially prepared cycles | |
| Interventions |
HT cycle (53 women) Endometrial preparation achieved using estradiol transdermal patches started on 2nd day of menstruation and used every other day with an initial dose of 100 mg/d and after 2 days increased to 200 mg/d. Progesterone 800 mg/d vaginally, starting after at least 11 days of transdermal estradiol HT plus GnRHa (66 women) In addition to HT, women received triptorelin depot 3.75 mg IM, 1 ampoule |
|
| Outcomes | Miscarriage per woman | |
| Notes | Published as an abstract in the abstract book of the annual meeting of the American Society for Reproductive Medicine Emailed and wrote to the authors requesting study data. However, no reply received |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Process used in random sequence generation not reported |
| Allocation concealment (selection bias) | Unclear risk | Not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | No information reported on withdrawals/losses to follow‐up as well as reasons for withdrawals; not clear whether data were analyzed on the basis of ITT |
| Selective reporting (reporting bias) | Unclear risk | Insufficient information to make a conclusive judgement |
| Other bias | Unclear risk | Insufficient information to make a conclusive judgement |
Simon 1998.
| Methods | Single‐centre parallel RCT | |
| Participants | 106 women Inclusion criteria: women with functioning ovaries who had embryos originating from IVF or ICSI using their own oocytes Indication to freeze the embryos: not stated Number of transferred embryos per woman: HT group 3.3 ± 1.0, HT plus GnRHa group 3.4 ± 1.2 Baseline comparison: only women's ages were compared and were similar |
|
| Interventions |
HT plus GnRHa group (53 women) Long‐acting, triptorelin 3.75 mg IM (starting on cycle day 21 in regularly menstruating or starting on day 1 in oligomenorrhoeic women) E2: started ≥ 14 days of GnRHa once E2 level was < 100 pmol/L. Oral micronised E2 4 mg/d in 2 doses for about 17 days. On the day of E2 intake: if serum E2 was < 800 pmol/L or endometrial thickness < 8 mm, E2 dose was increased to 6 mg/d or 8 mg/d for 5‐10 days Progesterone: started once endometrial thickness was ≥ 8 mm. Vaginal micronised progesterone tablets, 300 mg, 3 times a day until 12th gestational week Cycle monitoring: by US scan aiming for endometrial thickness of ≥ 8 mm prior to FET HT group (53 women) E2: started in 1st day of menstrual cycle. Oral micronised E2 6 mg/d for 7 days then E2 dose was adjusted as in HT plus GnRHa group Day of FET: for 2‐ to 4‐cell embryos: 48 h after progesterone initiation; for 6‐ to 8‐cell embryos: 72 h after progesterone initiation |
|
| Outcomes | Clinical pregnancy rate per woman Ongoing pregnancy rate per woman Miscarriage rate per woman Cycle cancellation rate per woman Endometrial thickness on day of progesterone initiation |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Procedure used for generating sequence was not reported |
| Allocation concealment (selection bias) | Unclear risk | Not reported whether allocation was concealed |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | No information as to whether participants or personnel (or both) were blinded; non‐blinding of outcome assessor could have affected the validity of some of the outcomes measured. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Reason for attrition stated and was similar between the 2 groups, number of missing outcome data fairly balanced between the 2 groups (1 versus 0) |
| Selective reporting (reporting bias) | Low risk | Data reported on all the outcomes prespecified in the methods section |
| Other bias | Low risk | No other potential source of bias found |
Van Der Auwera 1994.
| Methods | Single‐centre parallel RCT | |
| Participants | 209 women Baseline comparison: similar Infertility cause: endometriosis, tubal, male factor, mixed or unexplained Inclusion criteria: women with functioning ovaries who had embryos originating from IVF using their own oocytes Exclusion criteria: none Number of transferred embryos: in clomiphene group 1.8 ± 0.1, in HMG group 2.0 ± 0.1 |
|
| Interventions |
Clomiphene plus HMG (107 women) Oral clomiphene 100 mg started in days 2‐6 and HMG 150 μg/d IM from day 6 HMG (102 women) HMG 150 μg/d IM from day 2 In both groups from day 7 onwards, OI was done on individual basis Monitoring: by US and plasma E2 measurements Once leading follicle was ≥ 18 mm and E2 > 500 pg/mL, ovulation was induced using HCG 10,000 IU Day of HCG administration: in clomiphene plus HMG group 11.7 ± 0.2, in HMG group 10.8 ± 0.2 (P < 0.01) FET was performed 64 h post HCG administration or 48 h after LH surge. Luteal phase support: HCG 1500 IU IM on days 4, 7 and 10 post FET and progesterone vaginal suppositories 100 mg/d |
|
| Outcomes | Live birth rate per woman Miscarriage rate per woman Multiple pregnancy rate per woman |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Procedure used in random sequence generation not reported |
| Allocation concealment (selection bias) | Unclear risk | No report on allocation concealment |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported whether participants or personnel (or both) were blinded |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Insufficient information on attrition although tabular data were presented in a way that suggested possible inclusion of all randomized women in data analysis |
| Selective reporting (reporting bias) | Unclear risk | Data reported on outcomes prespecified in the methods section |
| Other bias | Unclear risk | Both groups were similar with respect to participants' characteristics, and number and quality of embryos transferred but HCG was administered on different days, although this was said not to have affected the outcome of the trial |
Weissman 2011.
| Methods | Single‐centre, non‐blinded, open‐label RCT Undertaken April 2006‐December 2008 Power calculation: not stated |
|
| Participants | 60 women undertaken FET following IVF or ICSI Inclusion criteria: women aged ≤ 38 years at embryo freezing time Exclusion criteria: use of testicular spermatozoa for ICSI, basal FSH ≥ 12 IU/L Women could participate in the study only once |
|
| Interventions |
HCG‐induced natural cycle FET (30 women) Spontaneous LH surge natural cycle FET (30 women) |
|
| Outcomes | Number of monitoring visits at the clinic per cycle Implantation rate Clinical pregnancy rate Live birth rate |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Patients were randomized into two groups before entering the treatment cycle according to a computer‐generated list by using opaque sealed envelopes." |
| Allocation concealment (selection bias) | Low risk | Allocation was said to have been concealed in sealed, opaque envelopes. |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Reported that study was a non‐blinded open trial and this could have affected the validity of the entire processes leading to outcome measures; however, non‐blinding may not have affected some of the outcome measures as they were objectively assessed. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Reasons for attrition stated and were found to be different between the 2 groups. Numbers of missing outcome data were not balanced between the groups. |
| Selective reporting (reporting bias) | Low risk | Data were reported on all outcome measures prespecified in the methods section. |
| Other bias | High risk | Women in both groups differed significantly in terms of age. |
Wright 2006.
| Methods | Single‐centre parallel RCT | |
| Participants | 175 women (194 FET cycles) Inclusion criteria: women with functioning ovaries either normo‐ovulatory or oligo‐ovulatory who had surplus embryos frozen following IVF or ICSI and women who had elective freezing of all embryos following OHSS Exclusion criteria: oocytes recipients Number of transferred embryos: HT group 1.77 ± 0.57, FSH group 1.66 ± 0.56 Baseline comparison: similar including age, day 3 FSH and % of ICSI cycles |
|
| Interventions |
HT group (88 women (94 cycles)) Oral E2 2 mg twice daily from day 1 of cycle. On day 9 or 10 of cycle started US and hormonal assay. Once endometrial thickness was > 7 mm started vaginal micronised progesterone (100 mg in morning and 200 mg in evening) and continued oral E2. If endometrial thickness was < 7 mm switched to vaginal E2 2 mg/d then as above. E2 and progesterone continued on same dose after FET and in pregnant women it was stopped at 8th gestational week FSH group (87 women (100 cycles)) Recombinant FSH 150 U on days 6, 8 and 10 of cycle. US and hormonal assay started on day 9 or 10 and until the endometrium was > 7 mm with a follicle of 16 mm‐20 mm then recombinant HCG was given Vaginal progesterone 100 mg in morning and 200 mg in evening was started in the day following HCG. FET was performed 48 h post progesterone initiation in embryos frozen on day 2 and 72 h in embryos frozen in day 3. Progesterone was continued till 8th gestational week |
|
| Outcomes | Clinical pregnancy per cycle Cycle cancellation rate per woman Endometrial thickness |
|
| Notes | Contacted first author but she was unable to provide more data, particularly the pregnancy rate per woman | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Process involved in random sequence generation not reported |
| Allocation concealment (selection bias) | Unclear risk | Methods used in concealing the allocation not reported |
| Blinding (performance bias and detection bias) All outcomes | Unclear risk | Not reported whether participants or personnel (or both) were blinded; non‐blinding of outcome assessors could have influenced some of the outcome measures. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Although reasons for missing data were the same in both groups, numbers of missing outcome data were not balanced between groups. |
| Selective reporting (reporting bias) | Low risk | All the prespecified outcome measures in the methods section were reported. |
| Other bias | Low risk | No other potential source of within‐study bias found |
ART: assisted reproductive technology; BMI: body mass index; E2: 17 β‐estradiol; ET: embryo transfer; FET: frozen‐thawed embryo transfer; FSH: follicle‐stimulating hormone; GnRHa: gonadotrophin releasing hormone agonist; HCG: human chorionic gonadotrophin; HT: hormone therapy; ICSI: intracytoplasmic sperm injection; IM: intramuscular; ITT: intention to treat; IU: international unit; IVF: in vitro fertilisation; LH: luteinising hormone; OHSS: ovarian hyperstimulation syndrome; OI: ovulation induction; PGD: preimplantation genetic diagnosis; RCT: randomized controlled trial; SC: subcutaneous; US: ultrasound.
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Al‐Shawaf 1993 | Retrospective study Comparing natural cycle FET versus GnRHa plus E2 plus progesterone No difference between the 2 interventions |
| Awonuga 1996 | Allocation to each intervention was not random but based on woman's choice. Comparing FET results following elective embryo cryopreservation in OHSS. FET done in natural cycle or in GnRHa plus oestrogen plus progesterone cycles. |
| Bals‐Pratsch 1999 | Uncontrolled study Case series of FET following E2 plus progesterone cycles |
| Belaisch‐Allart 1994 | Retrospective study Comparison of natural cycle FET, HMG ovulation induction FET and E2 plus progesterone FET |
| Benfer 1994 | Allocation to each intervention was not random Comparing results of natural cycle FET with GnRHa plus E2 plus progesterone FET No difference in outcomes between the 2 interventions |
| Chen 2007 | Retrospective study Comparison of natural cycle FET, GnRHa plus oestrogen plus progesterone FET, HMG FET and natural cycle plus HCG FET No difference in outcomes between the 4 interventions |
| Davar 2015 | Interventions not relevant: luteal phase support |
| de Ziegler 1990 | Allocation to each intervention was not random Comparing FET results in women who had IVF with women having FET using donated oocytes. IVF women with regular cycles were randomly assigned to natural cycle FET or GnRHa plus E2 plus progesterone FET cycles. IVF women with oligo‐ovulation were arbitrarily attributed to GnRHa plus E2 plus progesterone FET cycles. Women using embryos originating from donated oocytes had E2 plus progesterone FET cycles. |
| Dolan 1991 | Retrospective study Comparing natural cycle FET versus oestrogen plus progesterone FET No difference in outcomes between the 2 interventions |
| Dor 1991 | Allocation to each intervention was not random Comparing natural cycle FET (in the first 6 months of the study) versus HMG FET (in the second 6 months) versus oestrogen plus progesterone FET (in the last 7 months) No difference in outcomes between the 3 interventions |
| Elhelw 2008 | Interventions not relevant |
| Garrisi 1991 | Interventions did not meet inclusion criteria: compared success rate of fresh IVF cycle with success rate of thawed‐frozen embryos both in a natural cycle regimen |
| Gelbaya 2006 | Retrospective study Assessing natural cycle FET versus GnRHa plus oestrogen plus progesterone FET No difference in outcomes between the 2 interventions |
| Givens 2007 | A retrospective study Comparison of natural cycle FET versus oestrogen plus progesterone FET No difference in outcomes between the 2 interventions |
| Gonzalez 1992 | Retrospective study. Comparison of natural cycle FET versus oestrogen plus progesterone FET A trend of higher pregnancy rate in natural cycle FET was observed |
| Groenewoud 2015 | Interventions not reported |
| Imthurn 1996 | A quasi‐randomised study: allocation of intervention was based on presence or absence of spontaneous ovulation Natural cycle FET was allocated to women with a history of regular cycles while ovulation induction FET was allocated to women with anovulation history. Ovulation induction method was GnRHa followed by HMG. A trend towards fewer cancelled cycles was seen in the HMG FET group. |
| Jaroudi 1991 | Uncontrolled study Case series of FET following E2 plus progesterone cycles |
| Kawamura 2007 | Retrospective study Comparison of oestrogen plus progesterone FET versus natural cycle FET No difference in outcomes between the 2 interventions |
| Lee 2008 | Number of women randomized at baseline to each treatment group or number of women analyzed in each treatment group not reported |
| Lelaidier 1992 | Uncontrolled study Case series of FET following E2 plus progesterone cycles |
| Lelaidier 1995 | Uncontrolled study Case series of FET following E2 plus progesterone cycles |
| Loh 1999 | Non‐randomised study as allocation of intervention was based on presence or absence of spontaneous ovulation Ovulatory women had natural cycle FET while anovulatory women had oestrogen plus progesterone FET A significant higher live birth rate and a trend for higher clinical pregnancy rate in the natural cycle FET |
| Lornage 1990 | Retrospective study Comparison of natural cycle FET versus HCG‐induced ovulation cycle FET No difference in outcomes between the 2 interventions |
| Mausher 1991 | Uncontrolled study Case series of FET following GnRHa plus E2 plus progesterone cycles |
| Morozov 2007 | Retrospective study Comparison of natural cycle FET with oestrogen plus progesterone FET Significantly higher pregnancy rates in natural cycle FET |
| Oehninger 2000 | Non‐randomised study as allocation of intervention was based on presence or absence of spontaneous ovulation Ovulatory women had natural cycle FET while anovulatory women had E2 plus progesterone FET No difference in outcomes between the 2 interventions |
| Page 2005 | Interventions not relevant. Natural cycle versus FSH/HCG/progesterone |
| Pattinson 1992 | Non‐randomised study as women were given the choice of which type of FET cycle regimen to have Comparison of natural cycles FET with E2 plus progesterone FET No difference in outcomes between the 2 interventions |
| Queenan 1994 | Retrospective study Comparing natural cycle FET versus GnRHa plus E2 plus progesterone FET No difference in outcomes between the 2 interventions |
| Queenan 1997 | Uncontrolled retrospective study Analysing results of E2 plus progesterone FET |
| Sathanandan 1991 | Semi‐randomised study (quasi‐randomised) as women with irregular cycles, who had inadequate luteal function, women with amenorrhoea or oligo‐menorrhoea and women who were not pregnant in previous natural cycle FET were allocated to the GnRHa plus E2 plus progesterone intervention without randomization. Women having FET for first time and who had regular cycles were alternately allocated to either of the 2 interventions. Comparing GnRHa plus E2 plus progesterone FET with natural cycle FET No difference in outcomes between the 2 interventions except in women with oligomenorrhoea |
| Schmidt 1989 | Non‐randomised study as allocation of intervention was based on past history of ovulation disorder. Prospective comparison of oestrogen plus progesterone FET versus natural cycle FET A trend towards higher pregnancy rate was noted in oestrogen plus progesterone FET. |
| Shiotani 2006 | Not an RCT |
| Simon 1999 | Case series FET following E2 plus progesterone preparation |
| Spandorfer 2004 | Not an RCT |
| Tanos 1996 | Non‐randomised study as allocation of each type of intervention was based on presence or absence of regular ovulation. Women experiencing oligo‐ovulation were alternately offered ovulation induction or E2 plus progesterone endometrial preparation cycle. Prospectively comparing natural cycle FET versus GnRHa plus E2 plus progesterone FET versus GnRHa plus HMG FET No difference in outcomes among the 3 interventions |
| Taskin 2002 | Interventions not relevant |
| Wada 1992 | Non‐randomised controlled study as intervention allocation was based on couple's choice Comparison of natural cycle FET with GnRHa plus oestrogen plus progesterone FET No difference in outcomes between the 2 interventions |
| Yee 1995 | Retrospective study Comparing of GnRHa plus transdermal oestrogen plus progesterone FET versus GnRHa plus oral oestrogen plus progesterone FET versus oral oestrogen plus progesterone FET |
| Yishai 2001 | Retrospective controlled study Comparison of natural cycle FET with E2 plus progesterone FET No difference in outcomes between the 2 interventions |
| Yu 2015 | Interventions not relevant: administered different interventions within each treatment groups |
E2: 17 β‐estradiol; FET: frozen‐thawed embryo transfer; GnRHa: gonadotrophin releasing hormone agonist; HCH: human chorionic gonadotrophin; HMG: human menopausal gonadotrophin; IVF: in vitro fertilisation; OHSS: ovarian hyperstimulation syndrome; RCT: randomized controlled trial
Characteristics of ongoing studies [ordered by study ID]
NCT01780610.
| Trial name or title | The effects of two endometrium preparation protocols in frozen‐thawed embryo transfer in women with irregular cycles |
| Methods | Randomised Parallel assignment Single‐blind (participant) |
| Participants | 670 women 18‐40 years with irregular menstruation and > 3 frozen embryos |
| Interventions | Letrozole and HCG vs estradiol and progesterone |
| Outcomes | Primary: ongoing pregnancy rate Secondary: pregnancy rate; clinical pregnancy rate |
| Starting date | January 2012, last data collection date January 2015 |
| Contact information | Zhang Qingxue, Doctor: tel 13602797433, Memorial Hospital of Sun Yat‐Sen University |
| Notes |
NCT02197208.
| Trial name or title | A randomized controlled comparison of spontaneous natural cycles and human chorionic gonadotrophin‐induced natural cycles in frozen‐thawed embryos transfer |
| Methods | Randomised Parallel assignment Single‐blind (outcomes assessor) |
| Participants | 300 women aged 19‐43 years Regular menstrual cycles ranging from 21‐35 days with not more than 4 days variation between cycles Undergoing FET in natural cycles Normal uterine cavity as shown on saline sonogram performed before the IVF cycle or normal uterine cavity shown on pelvic scanning during the stimulated IVF cycle Endometrial thickness ≥ 8 mm in both stimulated IVF and FET cycles |
| Interventions | Daily monitoring of LH and E2 vs HCG‐induced natural cycle |
| Outcomes | Primary: ongoing pregnancy rate (defined as the number of viable pregnancies beyond 10‐12 weeks' gestation per transfer cycle) Secondary: days of monitoring for timing FET; endometrial thickness on day of HCG or the next day after LH surge; implantation rate; pregnancy rate; clinical pregnancy rate; miscarriage rate; multiple pregnancy rate |
| Starting date | October 2014, last data collection date December 2015 |
| Contact information | Vivian CY Lee, University of Hong Kong |
| Notes | No email address provided |
NCT02251925.
| Trial name or title | Frozen embryo transfer in natural and hormonal replacement cycles |
| Methods | Randomised, open‐label |
| Participants | 460 women 20‐37 years with regular menstruation cycles; undergoing long protocol; BMI ≤ 30 kg/m2; undergoing frozen embryo transfer for the first time |
| Interventions | Natural cycle (with or without HCG for ovulation induction) or hormonal cycle (with or without administration of GnRHa) or injection of GnRHa (Superfact) at a SC daily dose of 0.5 mg or hormonal group without GnRHa, endometrial preparation with daily administration of 6 mg estradiol valerate |
| Outcomes | Primary: clinical pregnancy rate Secondary: implantation rate; chemical pregnancy rates; ongoing pregnancy; miscarriage rate |
| Starting date | September 2012, completion date November 2015 |
| Contact information | Dr Nasser Aghdami nasser.aghdami@royaninstitute.org |
| Notes |
NCT02825108.
| Trial name or title | Evaluation the effect of intrauterine injection of human chorionic gonadotrophin injection (HCG) before frozen embryo transfer on implantation and clinical pregnancy rates per cycle, phase 3 randomized double blinded clinical trial |
| Methods | Randomised, participant and investigator blinded |
| Participants | 150 women 19‐39 years, with history of one fresh embryo transfer failure, primary infertility, and at least 1 embryo with excellent quality |
| Interventions | ET ET + intra uterine injection of tissue culture medium containing HCG ET + intra uterine injection of tissue culture medium without HCG |
| Outcomes | Primary: implantation rate Secondary: pregnancy loss; early miscarriage rate; late miscarriage rate |
| Starting date | January 2015, completion date July 2017 |
| Contact information | Dr Nasser Aghdami, nasser.aghdami@royaninstitute.org |
| Notes |
NCT02834117.
| Trial name or title | Comparison of the number of visits and the quality of life versus natural cycle in stimulated cycle before frozen embryo transfer |
| Methods | Randomised, open‐label |
| Participants | 124 women with regular cycles 26‐35 days, support in IVF or ICSI |
| Interventions | Natural cycle vs moderate ovarian stimulation |
| Outcomes | Primary: number of visits for clinical examination, ultrasound and hormonal dosage required to monitor ovulation in both groups Secondary: QoL; defrost cancellation rate; transfer on weekends and holidays; HCG levels > 100 U / L; pregnancy; birth; gestational age at delivery; implantation; miscarriage; cost |
| Starting date | May 2015, completion date March 2018 |
| Contact information | Maxime Brussieux, m.brussieux.chic@gmail.com |
| Notes |
E2: 17 β‐estradio; ET: embryo transfer; FET: frozen‐thawed embryo transfer; GnRHa: gonadotrophin releasing hormone agonist; HCG: human chorionic gonadotrophin; ICSI: intracytoplasmic sperm injection; IVF: in vitro fertilisation; LH: luteinising hormone;
Differences between protocol and review
We edited Objectives and 'Types of interventions' section to clarify that comparisons between types of modality are eligible: this was also the case in the previous version of the review but was not stated very clearly.
We rearranged outcomes, with 'Miscarriage rate per woman' now being the second primary outcome in the review and 'Clinical pregnancy rate' becoming a secondary outcome. We added one secondary outcome, 'Number of centre visits to monitor FET cycle'.
We amended the definition of the primary outcome 'Live birth' to be "delivery of a live fetus after 24 completed weeks of gestational age".
Contributions of authors
For the 2017 update:
TG: performed the updated search, selection of included studies, data extraction, and contributed to the writing and updating of the review.
TAG: contributed to the writing and updating of the review.
ROA: performed the updated search, selection of included studies and data extraction, created the 'Summary of findings' tables, and contributed to the writing and updating of the review.
For the previous update:
TG and Patrick Vanderkerchove (PV) shared the writing of the protocol, searching for and assessing the relevant studies and the writing up of the review. TG and PV shared the update of the review including search for relevant randomized controlled trials (RCTs), selection of included RCTs and writing up the updated review.
Sources of support
Internal sources
Gynaecology and Fertility Cochrane Subgroup, New Zealand.
External sources
None, Other.
Declarations of interest
Tarek Ghobara: no known conflict of interest Tarek A Gelbaya: no known conflict of interest Reuben Olugbenga Ayeleke: no known conflict of interest
New search for studies and content updated (no change to conclusions)
References
References to studies included in this review
Azimi Nekoo 2015 {published data only}
- Azimi Nekoo E, Chamani M, Shahrokh Tehrani E, Hossein Rashidi B, Davari Tanha F, Kalantari V. Artificial endometrial preparation for frozen‐thawed embryo transfer with or without pretreatment with depot gonadotropin releasing hormone agonist in women with regular menses. Journal of Family and Reproductive Health 2015;9(1):1‐4. [PMC free article] [PubMed] [Google Scholar]
Cattoli 1994 {published data only}
- Cattoli M, Ciotti PM, Seracchioli R, Casadio V, Bianchi L, Preti S, et al. A randomized prospective study on cryopreserved‐thawed embryo transfer: natural versus hormone replacement cycles. 10th Annual Meeting of the ESHRE, Brussels. 1994; Vol. 356:139.
Dal Prato 2002 {published data only}
- Dal Prato L, Borini A, Cattoli M, Bonu M, Sciajno R, Flamigni C. Endometrial preparation for frozen‐thawed embryo transfer with or without pretreatment with gonadotrophin‐releasing hormone agonist. Fertility and Sterility 2002;77(5):956‐60. [DOI] [PubMed] [Google Scholar]
Davar 2007 {published data only}
- Davar R, Eftekhar M, Tayebi N. Transfer of cryopreserved‐thawed embryos in a cycle using exogenous steroids with or without prior gonadotrophin‐releasing hormone agonist. Journal of Medical Science 2007;7(5):880‐3. [Google Scholar]
Eftekhar 2012 {published data only}
- Eftekhar M, Rahmani E, Eftekhar T. Effect of adding human chorionic gonadotropin to the endometrial preparation protocol in frozen embryo transfer cycles. Fertility and Sterility 2012;6(3):175‐8. [PMC free article] [PubMed] [Google Scholar]
El‐Toukhy 2004 {published data only}
- El‐Toukhy T, Taylor A, Khalaf Y, Al‐Darazi K, Rowell P, Seed P, et al. Pituitary suppression in ultrasound‐monitored frozen embryo replacement cycles. A randomised study. Human Reproduction 2004;19(4):874‐9. [DOI] [PubMed] [Google Scholar]
Fatemi 2010 {published and unpublished data}
- Fatemi HM, Kyrou D, Bourgain C, Abbeel E, Griesinger G, Devroey P. Cryopreserved‐thawed human embryo transfer: spontaneous natural cycle is superior to human chorionic gonadotropin‐induced natural cycle. Fertility and Sterility 2010;94(6):2054‐8. [DOI] [PubMed] [Google Scholar]
- Kyrou D, Fatemi HM, Stoop D, Tournaye H, Devroey P. Is spontaneous natural cycle the ideal method for planning vitrified/thawed blastocyst transfer in normovulatory patients?. Fertility and Sterility 2012;98(3):S125. [Google Scholar]
Greco 2016 {published data only}
- Greco E, Litwicka K, Arrivi C, Varricchio MT, Caragia A, Greco A, et al. The endometrial preparation for frozen‐thawed euploidblastocyst transfer: a prospective randomized trial comparing clinical results from natural modified cycle and exogenous hormone stimulation with GnRH agonist. Journal of Assisted Reproduction and Genetics 2016;33:873‐84. [DOI: 10.1007/s10815-016-0736-y] [DOI] [PMC free article] [PubMed] [Google Scholar]
Groenewoud 2016 {published data only}
- Groenewoud ER, Cohlen BJ, Al‐Oraiby A, Brinkhuis EA, Broekmans FJM, Bruin JP, et al. A randomized controlled,non‐inferiority trial of modified natural versus artificial cycle for cryo‐thawed embryo transfer. Human Reproduction 2016;31(7):1483–92. [DOI: 10.1093/humrep/dew120] [DOI] [PMC free article] [PubMed] [Google Scholar]
Karimzadeh 2012 {published data only}
- Karimzadeh MA, Mohammadian F, Mashayekhy M. Comparison of frozen‐thawed embryo transfer outcome in natural cycle and hormone replacement cycle. Human Reproduction 2012;27 Suppl 2:P‐284. [Google Scholar]
Loh 2001 {published and unpublished data}
- Loh SKE, Ganesan G, Leong N. Clomid versus hormone endometrial preparation in FET cycles. 17th World Congress on Fertility and Sterility (IFFS), 2001 Nov 25‐30; Melbourne, Australia. 2001.
Mounce 2015 {published data only}
- Child T, McVeigh E, Turner K, Mounce G. A randomized controlled trial of natural versus GnRH‐agonist/HRT regimes for frozen embryo replacement. Fertility and Sterility 2013;100 Suppl(3):S146. [DOI] [PubMed] [Google Scholar]
- Mounce G, Birks J, Bradley C, Child T. Patient satisfaction in a randomized trial comparing natural versus hormone replacement therapy (HRT) frozen embryo replacement (FER) treatment. Human Reproduction 2013;28:260‐1. [Google Scholar]
- Mounce G, McVeigh E, Turner K, Child TJ. Randomized, controlled pilot trial of natural versus hormone replacement therapy cycles in frozen embryo replacement in vitro fertilization. Fertility and Sterility 2015;104(4):915‐20. [DOI] [PubMed] [Google Scholar]
Peeraer 2015 {published data only}
- Peeraer K, Couck I, Debrock S, Neubourg D, Loecker P, Tomassetti C, et al. Frozen‐thawed embryo transfer in a natural or mildly hormonally stimulated cycle in women with regular ovulatory cycles: a RCT. Human Reproduction 2015;30(11):2552‐62. [DOI: 10.1093/humrep/dev224] [DOI] [PubMed] [Google Scholar]
Ramos 2007 {published data only}
- Ramos J, Caligara C, Tocino A, Rodriguez I, Carranza F, Fernandez‐Sanchez M. Prospective randomized study to compare frozen‐thawed embryo transfer cycles outcome in women with function ovaries and HRT for endometrium preparation with or without prior GnRHa suppression. Fertility and Sterility 2007;88 Suppl 1:S114‐5. [Google Scholar]
Simon 1998 {published data only}
- Simon A, Hurwitz A, Zentner B, Bdolah Y, Laufer N. Transfer of frozen‐thawed embryos in artificially prepared cycles with and without prior gonadotrophin‐releasing hormone agonist suppression: a prospective randomised study. Human Reproduction 1998;13(10):2712‐7. [DOI] [PubMed] [Google Scholar]
Van Der Auwera 1994 {published data only}
- Auwera I, Meuleman C, Koninckx P. Human menopausal gonadotrophin increases pregnancy rate in comparison with clomiphene citrate during replacement cycles of frozen/thawed pronucleate ova. Human Reproduction 1994;9(8):1556‐60. [DOI] [PubMed] [Google Scholar]
Weissman 2011 {published and unpublished data}
- Weissman A, Horowitz E, Ravhon A, Steinfeld Z, Golan A, Levranet D. Timing natural cycle frozen‐thawed embryo transfer by HCG triggering: a randomized prospective trial. Fertility and Sterility 2009;92 Suppl 1(3):S24. [Google Scholar]
- Weissman A, Horowitz E, Ravhon A, Steinfeld Z, Mutzafi R, Golan A, et al. Spontaneous ovulation versus HCG triggering for timing natural‐cycle frozen‐thawed embryo transfer: a randomised study. Reproductive BioMedicine Online 2011;23:484‐9. [DOI] [PubMed] [Google Scholar]
Wright 2006 {published data only}
- Wright K, Guibert J, Weitzen S, Davy C, Fauque P, Olivennes F. Artificial versus stimulated cycles for endometrial preparation prior to frozen‐thawed embryo transfer. Reproductive BioMedicine Online 2006;13(3):321‐5. [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
Al‐Shawaf 1993 {published data only}
- Al‐Shawaf T, Dunsong Y, Al‐Magid Y, Seaton A, Iketubosin F, Craft I. Ultrasonic monitoring during replacement of frozen/thawed embryos in natural and hormone replacement cycles. Human Reproduction 1993;8(12):2068‐74. [DOI] [PubMed] [Google Scholar]
Awonuga 1996 {published data only}
- Awonuga A, Dean N, Zaid J, Pittrof R, Bekir J, Tan S. Outcome of frozen embryo replacement cycles following elective cryopreservation of all embryos in women at risk of developing ovarian hyperstimulation syndrome. Journal of Assisted Reproduction and Genetics 1996;13(4):293‐6. [DOI] [PubMed] [Google Scholar]
Bals‐Pratsch 1999 {published data only}
- Bals‐Pratsch M, Al‐Hassani S, Schopper B, Diedrich C, Hoepfner A, Weiss J, et al. A simple, inexpensive and effective artificial cycle without exogenous transdermal oestradiol and vaginal progesterone for the transfer of cryopreserved pronucleated human oocytes in women with normal cycles. Human Reproduction 1999;14:222‐30. [DOI] [PubMed] [Google Scholar]
Belaisch‐Allart 1994 {published data only}
- Belaisch‐Allart J, Mandelbaum J, Cohen J, Plachot M, Chouraqui A, Mayenga JM, et al. Clinical management of a frozen‐thawed embryo transfer cycle. 10th annual meeting of the ESHRE, Brussels. 1994:138‐9.
Benfer 1994 {published data only}
- Benfer K, Check JH, Carlson J, Baker A, Nazari A. Comparison of pregnancy rates following transfer in natural versus hormone replacement cycles of thawed embryos cryopreserved at the pronuclear stage using a modified technique. 10th annual meeting of the ESHRE, Brussels. 1994:138.
Chen 2007 {published data only}
- Chen SL, He JX, Song HD, Li SZ, Liu XN, Li H, et al. Comparison of clinical outcomes of four protocols for frozen‐thawed embryo transfer cycle (in Chinese). Nan Fang Yi Ke Da Xue Bao 2007;27(3):303‐6. [PIMD 17425978] [PubMed] [Google Scholar]
Davar 2015 {published data only}
- Davar R, Mojtahedi MF, Miraj, S. Effects of single dose GnRH agonist as luteal support on pregnancy outcome in frozen‐thawed embryo transfer cycles: an RCT. Iranian Journal of Reproductive Medicine 2015;13(8):483‐8. [PMC free article] [PubMed] [Google Scholar]
de Ziegler 1990 {published data only}
- Ziegler D, Frydman R. Different implantation rates after transfers of cryopreserved embryos originating from donated oocytes or from regular in vitro fertilization. Fertility and Sterility 1990;54(4):682‐8. [DOI] [PubMed] [Google Scholar]
Dolan 1991 {published data only}
- Dolan P, Guzman I, Drews M, Williams M, Bergh PA, Grunfeld L, et al. Natural cycles and estrogen/progesterone induced cycles produce an equally receptive endometrium for implantation of cryopreserved embryos. 47th meeting of the American Fertility Society. 1991:S16.
Dor 1991 {published data only}
- Dor J, Rudak E, Davidson A, Levran D, Ben‐Rafael Z, Mashiach S. Endocrine and biological factors influencing implantation of human embryos following cryopreservation. Gynecological Endocrinology 1991;5:203‐11. [DOI] [PubMed] [Google Scholar]
Elhelw 2008 {published data only}
- Elhelw B, Sadek M, Nomrosy K. Aromatase inhibitor for the transfer of frozen‐thawed embryos: a prospective randomized comparative study. Human Reproduction 2008;23(Suppl 1):i42‐3. [Google Scholar]
Garrisi 1991 {published data only}
- Garrisi G. Prospective randomised study of single cohort embryos transferred during hyperstimulated versus natural endometrial cycles: the rate of endometrial receptivity. Fertility and Sterility 1991;56:s168. [Google Scholar]
Gelbaya 2006 {published data only}
- Gelbaya T, Nardo L, Hunter H, Fitzgerald C, Horne G, Pease E, et al. Cryopreserved‐thawed embryo transfer in natural or down‐regulated hormonally controlled cycles: a retrospective study. Fertility and Sterility 2006;85:603‐9. [DOI] [PubMed] [Google Scholar]
Givens 2007 {published data only}
- Givens C, Ryan I, Chenette P, Herbert C, Schriock E. Outcomes of natural cycles versus programmed cycles for 1390 frozen embryo transfers. Fertility and Sterility 2007;87(4 (2)):S11. [DOI] [PubMed] [Google Scholar]
Gonzalez 1992 {published data only}
- Gonzalez J, Ord T, Marello E, Asch RH, Frederick J, Stone S, et al. Natural cycle and hormonal replacement in FET: implantation and pregnancy rates. 48th Meeting of the American Fertility Society. 1992:S42.
Groenewoud 2015 {published data only}
- Groenewoud ER, Macklon NS, Cohlen BJ. A randomized controlled trial of natural versus artificial cycle for frozen thawed embryo transfer. Human Reproduction 2015;30:i374. [Google Scholar]
Imthurn 1996 {published data only}
- Imthurn B, Macas E, Rosselli M, Keller P. Effect of a programmed short‐term stimulation protocol on the regulation of cryopreserved embryos. Journal of Assisted Reproduction and Genetics 1996;13(9):709‐12. [DOI] [PubMed] [Google Scholar]
Jaroudi 1991 {published data only}
- Jaroudi K, Sieck U, Hamilton C, Roca G, Willemsen W. Artificial endometrial stimulation for frozen embryo replacement. Fertility and Sterility 1991;55:835‐7. [PubMed] [Google Scholar]
Kawamura 2007 {published data only}
- Kawamura T, Motoyama H, Yanaihara A, Yorimitsu T, Arichi A, Karasawa Y, et al. Clinical outcomes of two different endometrial preparation methods for cryopreserved‐thawed embryo transfer in patients with a normal menstrual cycle. Reproductive Medicine and Biology 2007;6:53‐7. [DOI] [PMC free article] [PubMed] [Google Scholar]
Lee 2008 {published data only}
- Lee SJ, Kwon HC, Kim JW, Lee J, Jung Y, Jung J, et al. Comparison of clinical outcome of frozen‐thawed embryo transfer cycles between natural and artificial (hormone‐treated) cycles. Human Reproduction 2008;23(Suppl 1):i127. [Google Scholar]
Lelaidier 1992 {published data only}
- Lelaidier C, Ziegler D, Gaetano J, Hazout A, Fernandez H, Frydman R. Controlled preparation of the endometrium with exogenous oestradiol and progesterone: a novel regimen not using a gonadotrophin‐releasing hormone agonist. Human Reproduction 1992;7:1353‐6. [DOI] [PubMed] [Google Scholar]
Lelaidier 1995 {published data only}
- Lelaidier C, Olivennes F, Ziegler D, Hazout A, Freitas S, Frydman R. Endometrium preparation with exogenous estradiol and progesterone for the transfer of cryopreserved blastocysts. Fertility and Sterility 1995;63(4):919‐21. [DOI] [PubMed] [Google Scholar]
Loh 1999 {published data only}
- Loh SKE, Leong NKY. Factors affecting success in an embryo cryopreservation programme. Annals Academy of Medicine 1999;28(2):260‐5. [PubMed] [Google Scholar]
Lornage 1990 {published data only}
- Lornage J, Boulieu D, Mathieu C, Guerin JF, Pinatel MC, James R, et al. Transfer of frozen‐thawed human embryos in cycles stimulated by HMG. Human Reproduction 1990;5(1):60‐5. [DOI] [PubMed] [Google Scholar]
Mausher 1991 {published data only}
- Mausher S, Kruithoff C, Simonetti S, Oehninger S, Acosta A, Jones G. Controlled preparation of the endometrium with exogenous steroids for the transfer of frozen‐thawed pre‐embryos in patients with anovulatory or irregular cycles. Human Reproduction 1991;6:443‐5. [DOI] [PubMed] [Google Scholar]
Morozov 2007 {published data only}
- Morozov V, Ruman J, Kenigsberg D, Moodie G, Brenner S. Natural cycle cryo‐thaw transfer may improve pregnancy outcome. Journal of Assisted Reproduction and Genetics 2007;24:119‐23. [DOI] [PMC free article] [PubMed] [Google Scholar]
Oehninger 2000 {published data only}
- Oehninger S, Mayer J, Muasher S. Impact of different clinical variables on pregnancy outcome following embryo cryopreservation. Molecular and Cellular Endocrinology 2000;169:73‐7. [DOI] [PubMed] [Google Scholar]
Page 2005 {published data only}
- Page KL, Guibert J, Weitzen S, Davy C, Fauque P, Olivennes F. A prospective randomized trial evaluating endometrial preparation for implantation of frozen/thawed embryos using an artificial cycle versus a stimulated cycle. Fertility and Sterility 2005;84:S171‐2. [Google Scholar]
Pattinson 1992 {published data only}
- Pattinson H, Greene C, Fleetham J, Anderson‐Sykes S. Exogenous control of the cycle simplifies thawed embryo transfer and results in a pregnancy rate similar to that for natural cycles. Fertility and Sterility 1992;58(3):627‐9. [DOI] [PubMed] [Google Scholar]
Queenan 1994 {published data only}
- Queenan J, Veek L, Seltman H, Muasher S. Transfer of cryopreserved‐thawed pre‐embryos in natural cycle or a programmed cycle with exogenous hormonal replacement yields similar pregnancy results. Fertility and Sterility 1994;62(3):545‐50. [DOI] [PubMed] [Google Scholar]
Queenan 1997 {published data only}
- Queenan J, Ramey J, Seltman H, Eure L, Veeck L, Muasher S. Transfer of cryopreserved‐thawed pre‐embryo in a cycle of exogenous steroid without prior gonadotrophin‐releasing hormone agonist suppression yields favourable pregnancy results. Human Reproduction 1997;12(6):1176‐80. [DOI] [PubMed] [Google Scholar]
Sathanandan 1991 {published data only}
- Sathanandan M, Macnamee M, Rainsbury P, Wick K, Brinsden P, Edwards R. Replacement of frozen‐thawed embryos in artificial and natural cycles: a prospective semi‐randomised study. Human Reproduction 1991;6(5):685‐7. [DOI] [PubMed] [Google Scholar]
Schmidt 1989 {published data only}
- Schmidt C, Ziegler D, Gagliardi C, Mellon R, Taney F, Kuhar M, et al. Transfer of cryo‐preserved‐thawed embryos: the natural cycle versus controlled preparation of the endometrium with gonadotrophin‐releasing hormone agonist and exogenous estradiol and progesterone (GEEP). Fertility and Sterility 1989;52:609‐16. [DOI] [PubMed] [Google Scholar]
Shiotani 2006 {published data only}
- Shiotani M, Goto S, Kokeguchi S, Matsunaga M, Watanabe J, Hashimoto H, et al. Is hCG supplementation beneficial for cryopreserved‐thawed embryo transfer in estrogen/progesterone replacement cycles?. Human Reproduction 2006;21:i82. [Google Scholar]
Simon 1999 {published data only}
- Simon A, Hurwitz A, Pharhat M, Revel A, Zentner B, Laufer N. A flexible protocol for artificial preparation of the endometrium without prior gonadotrophin‐releasing hormone agonist suppression in women with functioning ovaries undergoing frozen‐thawed embryo transfer cycles. Fertility and Sterility 1999;71(4):609‐13. [DOI] [PubMed] [Google Scholar]
- Simon A, Hurwitz A, Zentner B, Bdolah Y, Laufer N. Transfer of frozen‐thawed embryos in artificially prepared cycle with and without prior gonadotropin‐releasing hormone agonist suppression ‐ a prospective randomised study. Fertility and Sterility 1998;70(3):S59‐60. [DOI] [PubMed] [Google Scholar]
Spandorfer 2004 {published data only}
- Spandorfer SD, Fasouliotis SJ, Cimmino C, Shpizner M, Veeck L, Rosenwaks Z. Blastocyst frozen embryo transfer (FET): comparison of outcome with replacement in natural or programmed/medicated cycle. Fertility and Sterility 2004;82(Suppl 2):s154. [Google Scholar]
Tanos 1996 {published data only}
- Tanos V, Friedler S, Zajicek G, Neiger M, Lewin A, Schenker JG. The impact of endometrial preparation on implantation following cryopreserved‐thawed‐embryo transfer. Gynecologic and Obstetric Investigation 1996;41(4):227‐31. [DOI] [PubMed] [Google Scholar]
Taskin 2002 {published data only}
- Taskin O, Akkoyunlu G, Simsek M, Demir R, Onoglu A, Sadik S. Comparing the effects of GnRH‐a on endometrial receptivity in patients undergoing ART and prepared frozen embryo transfer cycles. Fertility and Sterility 2002;78(3 Suppl 1):s232. [Google Scholar]
Wada 1992 {published data only}
- Wada I, Matson P, Troup S, Hughes S, Buck P, Lieberman B. Outcome of treatment subsequent to elective cryopreservation of all embryos from women at risk of the ovarian hyperstimulation syndrome. Human Reproduction 1992;7(7):962‐6. [DOI] [PubMed] [Google Scholar]
Yee 1995 {published data only}
- Yee B, Lin Y, Chacon R, Soubra S, Rosen G, Cassidenti D. A simplified method of timing frozen embryo transfers. American Journal of Obstetrics and Gynecology 1995;172(6):1844‐50. [DOI] [PubMed] [Google Scholar]
Yishai 2001 {published data only}
- Yishai D, Rothschild E, Abramovici H, Dirnfeld M. Do we need to artificially prepare the endometrium for frozen embryo transfer in normal cycling women?. Fertility and Sterility 2001;76(3):S122. [Google Scholar]
Yu 2015 {published data only}
- Yu J, Ma Y, Wu Z. Endometrial preparation protocol of the frozen‐thawed embryo transfer in patients with polycystic ovary syndrome. Archives of Gynecological Obstetrics 2015;291:201‐11. [DOI: 10.1007/s00404-014-3396-0] [DOI] [PubMed] [Google Scholar]
References to ongoing studies
NCT01780610 {published data only}
- NCT01780610. The effects of two endometrium preparation protocols in frozen‐thawed embryo transfer in women with irregular cycles. clinicaltrials.gov/ct2/show/NCT01780610 (first received 24 January 2013).
NCT02197208 {published data only}
- NCT02197208. A randomized controlled comparison of spontaneous natural cycles and human chorionic gonadotrophin‐induced natural cycles in frozen‐thawed embryos transfer. clinicaltrials.gov/ct2/show/NCT02197208 (first received 20 July 2014).
NCT02251925 {published data only}
- NCT02251925. Frozen embryo transfer in natural and hormonal replacement cycles. clinicaltrials.gov/ct2/show/NCT02251925 (first received 23 September 2014).
NCT02825108 {published data only}
- NCT02355925. Intrauterine injection of human chorionic gonadotropin injection (HCG) before frozen embryo transfer on cycle outcomes. clinicaltrials.gov/ct2/show/NCT02355925 (received 27 January 2015).
- NCT02825108. Evaluation the effect of intrauterine injection of human chorionic gonadotropin injection (HCG) before frozen embryo transfer on implantation and clinical pregnancy rates per cycle, phase 3 randomized double blinded clinical trial. clinicaltrials.gov/ct2/show/NCT02825108 (received 4 July 2016).
NCT02834117 {published data only}
- NCT02834117. Comparison of the number of visits and the quality of life versus natural cycle in stimulated cycle before frozen embryo transfer. clinicaltrials.gov/ct2/show/NCT02834117 (first received 3 September 2015).
Additional references
Ben‐Nun 1997
- Ben‐Nun I, Shulman A. Induction of artificial endometrial cycles with s.c. oestrogen implants and injectable progesterone in in‐vitro fertilization treatment with donated oocytes: a preliminary report. Human Reproduction 1997;12(10):2267‐70. [DOI] [PubMed] [Google Scholar]
Borini 1995
- Borini A, Violini F, Bianchi L, Bafaro M, Trevisi M, Flamigni C. Improvement of pregnancy and implantation rates in cyclic women undergoing oocyte donation after long‐term down‐regulation. Human Reproduction 1995;10(11):3018‐21. [DOI] [PubMed] [Google Scholar]
Davies 1991
- Davies D, Jenkins J, Anthony F, Gadd S, Watson R, Sakhrani L, et al. Biochemical monitoring during hormonal replacement therapy cycles for transfer of cryopreserved embryos in patients with functional ovaries. Human Reproduction 1991;6(7):934‐8. [DOI] [PubMed] [Google Scholar]
de Ziegler 1991
- Ziegler D, Cornel C, Bergeron C, Hazout A, Bouchard P, Frydman R. Controlled preparation of the endometrium with exogenous estradiol and progesterone in women having functioning ovaries. Fertility and Sterility 1991;56:851‐5. [DOI] [PubMed] [Google Scholar]
Deeks 2011
- Deeks JJ, Higgins JPT, Altman DG (editors). Chapter 9: Analysing data and undertaking meta‐analyses. In: Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Devroey 1998
- Devroey P, Pados G. Preparation of endometrium for egg donation. Human Reproduction Update 1998;4(6):856‐61. [DOI] [PubMed] [Google Scholar]
Dias 2008
- Dias S, McNamee R, Vail A. Bias in frequently reported analyses of subfertility trials. Statistics in Medicine 2008;27(27):5605‐19. [DOI] [PubMed] [Google Scholar]
Flamigni 1993
- Flamigni C, Borini A, Violini F, Bianchi L, Serrao L. Oocyte donation: comparison between recipients from different age groups. Human Reproduction 1993;8(12):2088‐92. [DOI] [PubMed] [Google Scholar]
Frydman 1988
- Frydman R, Bouchard P, Parneix I. LHRH agonists do they have a role in frozen embryo transfer cycle [Les agonistes de la LHRH ont‐ils un role dans le cycle de transfert des embryons congeles]. Contraception Fertilite Sexuality 1988;16:29. [Google Scholar]
GRADEPro GDT 2014 [Computer program]
- GRADE Working Group, McMaster University. GRADEpro GDT. Version accessed prior to November 2016. Hamilton (ON): GRADE Working Group, McMaster University, 2014.
Higgins 2003
- Higgins JPT, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta‐analyses. BMJ 2003;327:557‐60. [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2011a
- Higgins JPT, Altman DG, Sterne JAC (editors). Chapter 8: Assessing risk of bias in included studies. In: Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Irianni 1992
- Irianni F, Veeck L, Toner J, Muasher S. Influence of number of pre‐embryos transferred, progesterone level and oestradiol/progesterone ratio at the thaw on pregnancy results during replacement of cryo‐preserved pre‐embryos in natural cycles. Human Reproduction 1992;7:797‐800. [DOI] [PubMed] [Google Scholar]
Johnson 2003
- Johnson NP, Proctor M, Farquhar CM. Gaps in the evidence for fertility treatment ‐ an analysis of the Cochrane Menstrual Disorders and Subfertility Group database. Human Reproduction 2003;18(5):947‐54. [PubMed] [Google Scholar]
Ku 2002
- Ku SY, Choi YM, Suh CS, Kim SH, Kim JG, Moon SY, et al. Effect of gonadotrophins on human endometrial cell proliferation in vitro. Archive Gynecological and Obstetrical 2002;266:223‐8. [DOI] [PubMed] [Google Scholar]
Leeton 1991
- Leeton J, Rogers P, Healy D. A comparison of pregnancy rates for 131 donor oocyte transfers using either a sequential or fixed regime of steroid replacement therapy. Human Reproduction 1991;6:299‐301. [DOI] [PubMed] [Google Scholar]
Lefebvre 2011
- Lefebvre C, Manheimer E, Glanville J. Chapter 6: Searching for studies. In: Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Lutjen 1984
- Lutjen P, Trounson A, Leeton J, Findlay J, Wood C, Renou P. The establishment and maintenance of pregnancy using in vitro fertilization and embryo donation in a patient with primary ovarian failure. Nature 1984;12(307(5947)):174‐5. [DOI] [PubMed] [Google Scholar]
Mandelbaum 1987
- Mandelbaum J, Junca A, Placchot M, Alvarez S, Debache C, Salat‐Baroux J, et al. Human embryo cryopreservation, extrinsic and intrinsic parameters of success. Human Reproduction 1987;2:709‐14. [DOI] [PubMed] [Google Scholar]
Meldrum 1989
- Meldrum D, Wisot A, Hamilton F, Gutlay‐Yeo A, Marr B, Huynh D. Artificial agonadism and hormone replacement for oocyte donation. Fertility and Sterility 1989;52:509. [DOI] [PubMed] [Google Scholar]
Moher 2009
- Moher D, Liberati A, Tetzlaff J, Altman DG, The PRISMA Group (2009). Preferred Reporting Items for Systematic Reviews and Meta‐Analyses: the PRISMA Statement. PLoS Medicine 2009;6(7):e1000097. [DOI: 10.1371/journal.pmed1000097] [DOI] [PMC free article] [PubMed] [Google Scholar]
Navot 1989
- Navot D, Anderson T, Droesch K, Scott R, Kreiner D, Rosenwaks Z. Hormonal manipulation of endometrial maturation. Journal of Clinical Endocrinology and Metabolism 1989;68(4):801‐7. [DOI] [PubMed] [Google Scholar]
Pados 1992
- Pados G, Camus M, Waesberghe L, Liebaers I, Steirteghem A, Devroey P. Oocyte and embryo donation: evaluation of 412 consecutive trials. Human Reproduction 1992;7(8):1111‐7. [DOI] [PubMed] [Google Scholar]
Schünemann 2011
- Schünemann HJ, Oxman AD, Higgins JPT, Vist GE, Glasziou P, Guyatt GH. Chapter 11: Presenting results and ‘Summary of findings' tables. In: Higgins JPT, Green S (editors), Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Shemesh 2001
- Shemesh M. Actions of gonadotrophins on the uterus. Reproduction 2001;121:835‐42. [DOI] [PubMed] [Google Scholar]
Sher 1991
- Sher G, Herbert C, Maassarani G, Jacobs M. Assessment of the late proliferative phase endometrium by ultrasonography in patients undergoing in‐vitro fertilization and embryo transfer (IVF/ET). Human Reproduction 1991;6:232‐7. [DOI] [PubMed] [Google Scholar]
Speroff 1994
- Speroff L, Glass RH, Kase NG. Regulation of the menstrual cycle. In: Speroff L, Glass RH, Kase NG editor(s). Clinical Gynecologic Endocrinology and Infertility. 5th Edition. Baltimore: Williams and Wilkins, 1994:183‐230. [Google Scholar]
Troup 1991
- Troup S, Matson P, Critchlow J, Morroll D, Lieberman B, Burslem R. Cryopreservation of human embryos at the pronucleate, early cleavage or expanded blastocyst stages. European Journal of Obstetrics, Gynaecology and Reproductive Biology 1991;38(2):133‐9. [DOI] [PubMed] [Google Scholar]
Vail 2003
- Vail A, Gardner E. Common statistical errors in the design and analysis of subfertility trials. Human Reproduction 2003;18:1000‐4. [DOI] [PubMed] [Google Scholar]
References to other published versions of this review
Ghobara 2002
- Ghobara T, Vandekerckhove P. Cycle regimes for frozen‐thawed embryo transfer. Cochrane Database of Systematic Reviews 2002, Issue 1. [DOI: 10.1002/14651858.CD003414] [DOI] [PubMed] [Google Scholar]
Ghobara 2008
- Ghobara T, Vandekerckhove P. Cycle regimens for frozen‐thawed embryo transfer. Cochrane Database of Systematic Reviews 2008, Issue 1. [DOI: 10.1002/14651858.CD003414.pub2] [DOI] [PubMed] [Google Scholar]


