Abstract
Prior studies have shown that beginning hemodialysis (HD) with a hemodialysis catheter (HC) is associated with worse mortality than with an arteriovenous fistula (AVF) or arteriovenous graft (AVG). We hypothesized that transplant waitlisting would modify the effect of HD access on mortality, given waitlist candidates’ more robust health status. Using the US Renal Data System, we studied patients with incident ESRD who initiated HD between 2010–2015 with an AVF, AVG, or HC. We used Cox regression including an interaction term for HD access and waitlist status. There were 587,607 patients that initiated HD, of whom 82,379 (14.0%) were waitlisted for transplantation. Only 26,264 (4.5%) were transplanted. Among patients not listed, those with an AVF had a 34% lower mortality compared to HC (adjusted hazard ratio [aHR] 0.66, 95% confidence interval [CI] 0.65–0.67) while those with an AVG had a 21% lower mortality compared to HC (aHR 0.79, 95% CI 0.77–0.81). Transplant waitlisting attenuated the association between hemodialysis access type and mortality (interaction p<0.001 for both AVF and AVG vs HC). Among patients on the waitlist, those with an AVF had a 12% lower mortality compared to HC (aHR 0.88, 95% CI 0.84–0.93), while those with an AVG had no difference in mortality (aHR 0.95, 95% CI 0.84–1.08). While all patients benefit from AVF or AVG over HC, the benefit was attenuated in waitlisted patients. Efforts to improve health status and access to healthcare for non-waitlisted ESRD patients might decrease HD-associated mortality and improve rates of AVF and AVG placement.
Keywords: hemodialysis access, end-stage renal disease, kidney transplantation, transplant waitlisting
INTRODUCTION
Nearly 100,000 people in the US initiate hemodialysis (HD) each year, with 30 million adults additionally at risk for developing end-stage renal disease (ESRD) [1]. Prior studies have shown that beginning HD with a hemodialysis catheter (HC) is associated with significantly higher mortality than with an arteriovenous fistula (AVF) or arteriovenous graft (AVG) [2–5]. The association between worse mortality and HD access type has been demonstrated even in the elderly [3]. Whether the association between mortality and HD access type holds for ESRD patients who are waitlisted for kidney transplantation, and thus might not have a need for long term HD access, is not clear.
Historically, healthier ESRD patients were placed on the waitlist for kidney transplantation because they had the most survival benefit from transplantation [6]. However, socioeconomic and geographic factors also impact whether a patient becomes waitlisted, so in many ways transplant waitlisting might be considered a proxy for access to care as well [7–12]. In this sense, ESRD patients who are waitlisted for transplantation might be considered the healthiest subset of ESRD patients who have the most ability to access healthcare when needed. This subset of ESRD patients might therefore benefit less from AVF or AVG as compared to HC.
The goal of this study was to determine whether the association between HD access and mortality would vary by transplant waitlist status, given transplant candidates’ shorter need for HD access and more robust health status. We used data from the United States Renal Data System (USRDS) to study this interaction. We hypothesized that transplant waitlisting would modify the effect of HD access on mortality, given waitlist candidates’ more robust health status.
METHODS
Data source
We studied patients with incident ESRD 2010–2015 in the United States Renal Data System (USRDS). The USRDS includes demographic, clinical, treatment, and survival data for nearly all patients with ESRD in the US [13]. The USRDS is linked with claims data from the Centers for Medicare and Medicaid Services (CMS) and with transplantation data from the Organ Procurement and Transplantation Network/United Network for Organ Sharing (OPTN/UNOS). This study was acknowledged as exempt non-human subjects research by the Johns Hopkins Institutional Review Board.
Study population
We included all patients with incident ESRD, who initiated HD between 1/1/2010–12/31/2015 with an AVF, AVG, or HC. Demographic characteristics, clinical comorbidities, and initial HD access type were taken from CMS Form 2728. We excluded patients who had been on HD prior to 1/1/2010 and patients who did not have CMS Form 2728 data available. Transplant waitlisting was ascertained through linkage to OPTN/UNOS data. Patients who were waitlisted at any time during the study period were considered to have been waitlisted. Descriptive characteristics of patients who were waitlisted were compared to those who were not waitlisted using t-tests or chi-square tests, as appropriate.
Mortality
Mortality was ascertained through linkage to CMS data. We used Cox proportional hazards models to study the association between HD access and mortality, including an interaction term to examine effect modification of HD access on mortality by waitlist status. We censored at transplantation or on 12/31/2015. We adjusted for subject age, gender, African American race, Hispanic ethnicity, insurance prior to Medicare enrollment, body mass index (BMI), etiology of ESRD, being in the care of a nephrologist, and comorbid conditions of congestive heart failure (CHF), atherosclerotic heart disease (ASHD), cerebrovascular disease (CVD), peripheral vascular disease (PVD), diabetes mellitus (DM), chronic obstructive pulmonary disease (COPD), current tobacco smoking, history of cancer, alcohol dependence, drug dependence, and inability to ambulate.
Sensitivity analyses
As a sensitivity analysis, we used doubly robust Cox proportional hazards models with matching by propensity to be waitlisted to examine effect modification of HD access type on mortality by waitlist status, separately for AVG vs. HC and AVF vs. HC. The sensitivity analysis was limited to patients with no missing data. The MatchIt package for R was used to create propensity scores and match using 1:1 nearest-neighbor matching [14]. Covariates included in creation of propensity scores were the same as those included in the primary analysis regression model: age, gender, African American race, Hispanic ethnicity, insurance prior to Medicare enrollment, BMI, cause of ESRD, being in the care of a nephrologist, and comorbid conditions of CHF, ASHD, CVD, PVD, DM, COPD, current tobacco smoking, history of cancer, alcohol dependence, drug dependence, and inability to ambulate. Covariate balance was assessed through the standardized differences between the means of waitlisted and not waitlisted matched subcohorts, with good balance considered standardized differences less than 0.25 [15,16]. Data from the matched subcohorts do not generalize to all patients with ESRD, as they represent a non-random minority subcohort selected for their high likelihood to be waitlisted for transplantation.
As an additional sensitivity analysis, we used a competing risk regression in the method of Fine and Gray to study a 10% random sample of the study population. This analysis confirmed effect modification of the association between mortality and HD access by transplant waitlisting (interaction p<0.001) and direction of the associations between HD access and mortality within waitlisted and not waitlisted groups, thus full results of the model are not included.
Statistical analysis
An α of 0.05 was considered significant. Analyses were performed using Stata 14.2/SE for Windows (College Station, Texas) and R (Vienna, Austria).
RESULTS
Study population
Of 587,607 subjects, 82,379 (14.0%) were ever waitlisted for kidney transplantation. Only 26,264 (4.5%) were transplanted. Subjects who were not waitlisted had a median of 1.6 years with maximum 6 years follow-up, while subjects who were waitlisted had a median of 2.6 years with maximum 6 years follow-up. Although the majority of subjects had an HC for HD access (80.7%), transplant waitlisted subjects were more likely to have an AVF at ESRD registration than those who were never waitlisted (26.6% vs. 14.7%, p<0.001) (Table 1). Subjects who were waitlisted were younger, less frequently female, more frequently African American or Hispanic, and more frequently under the care of a nephrologist than those who were not waitlisted (Table 1). Waitlisted subjects more frequently had private insurance prior to Medicare coverage (38.8% vs 15.2%, p<0.001). Waitlisted subjects were less likely to have comorbid conditions including CHF, ASHD, CVD, PVD, DM, COPD, current tobacco smoking, a history of cancer, alcohol or drug dependence, and inability to ambulate compared to those who were not waitlisted (Table 1).
Table 1.
Characteristics of study population, stratified by transplant waitlisting.
| Waitlisted n=82,379 |
Not Waitlisted n=505,228 |
p-value | |
|---|---|---|---|
| Hemodialysis access | <0.001 | ||
| AVF | 26.6% | 14.7% | |
| AVG | 2.8% | 2.9% | |
| HC | 70.6% | 82.4% | |
| Age, median (IQR) years | 53 (42–62) | 67 (57–76) | <0.001 |
| Female sex | 35.9% | 43.5% | <0.001 |
| BMI, median (IQR) | 28 (24–33) | 28 (24–34) | <0.001 |
| African American race | 28.9% | 27.3% | <0.001 |
| Hispanic ethnicity | 20.0% | 14.5% | <0.001 |
| Cause of ESRD | <0.001 | ||
| Diabetes | 42.0% | 47.6% | |
| Hypertension | 24.9% | 30.9% | |
| Glomerulonephritis | 12.4% | 4.0% | |
| Secondary glomerulonephritis | 3.8% | 1.6% | |
| Interstitial nephritis | 2.7% | 2.6% | |
| Cystic/hereditary/congenital | 7.0% | 1.3% | |
| Neoplasms | 1.4% | 2.5% | |
| Other | 6.0% | 9.4% | |
| Under the care of a nephrologist | 74.8% | 65.7% | <0.001 |
| Insurance prior to ESRD | <0.001 | ||
| Medicare | 14.0% | 32.0% | |
| Medicaid | 21.5% | 26.7% | |
| Private | 38.8% | 15.2% | |
| Other | 15.9% | 20.0% | |
| Uninsured | 9.8% | 5.9% | |
| Comorbid disease | |||
| CHF | 14.2% | 34.4% | <0.001 |
| ASHD | 9.2% | 19.2% | <0.001 |
| CVD | 4.2% | 10.0% | <0.001 |
| PVD | 5.4% | 13.5% | <0.001 |
| DM | <0.001 | ||
| On no medication | 4.8% | 5.9% | |
| On oral medication(s) | 9.7% | 11.0% | |
| On insulin | 33.8% | 41.4% | |
| COPD | 2.4% | 11.2% | <0.001 |
| Current tobacco smoker | 3.9% | 6.7% | <0.001 |
| History of cancer | 2.8% | 8.3% | <0.001 |
| Alcohol dependence | 1.1% | 1.8% | <0.001 |
| Drug dependence | 0.6% | 1.4% | <0.001 |
| Inability to ambulate | 0.9% | 8.7% | <0.001 |
AVF vs. HC
Among those with an AVF at ESRD registration, 5-year mortality for waitlisted subjects was 25.6% compared to 58.2% in those not waitlisted. Transplant waitlisting attenuated the association between access with an AVF vs. HC and mortality (interaction p<0.001). Among patients who were on the waitlist, those with an AVF had a 12% lower risk of mortality compared to HC (adjusted hazard ratio [aHR] 0.88, 95% confidence interval [CI] 0.84–0.93, p<0.001) (Table 2, Figure 1). Among patients not on the waitlist, those with an AVF had a 34% lower risk of mortality compared to HC (aHR 0.66, 95% CI 0.65–0.67, p<0.001).
Table 2.
Mortality risk of transplant waitlisted vs. non-waitlisted patients.
| Waitlisted* | Not Waitlisted | |
|---|---|---|
| Hemodialysis access | ||
| HC | Ref | Ref |
| AVG | 0.95 (0.84–1.08), p=0.5 | 0.79 (0.77–0.81), p<0.001 |
| AVF | 0.88 (0.84–0.93), p<0.001 | 0.66 (0.65–0.67), p<0.001 |
| Age, per 5 years | 1.14 (1.13–1.16), p<0.001 | 1.14 (1.14–1.14), p<0.001 |
| Female sex | 0.98 (0.94–1.03), p=0.5 | 0.96 (0.95–0.97), p<0.001 |
| BMI, per 5 units | 0.91 (0.90–0.93), p<0.001 | 0.94 (0.94–0.94), p<0.001 |
| African American race | 0.57 (0.54–0.60), p<0.001 | 0.74 (0.73–0.75), p<0.001 |
| Hispanic ethnicity | 1.74 (1.63–1.85), p<0.001 | 1.40 (1.38–1.42), p<0.001 |
| Cause of ESRD | ||
| Diabetes | Ref | Ref |
| Hypertension | 0.90 (0.84–0.96), p=0.003 | 0.99 (0.98–1.01), p=0.3 |
| Glomerulonephritis | 0.70 (0.63–0.78), p<0.001 | 0.84 (0.81–0.86), p<0.001 |
| Secondary glomerulonephritis | 0.83 (0.71–0.97), p=0.02 | 1.03 (0.99–1.07), p=0.2 |
| Interstitial nephritis | 0.87 (0.75–1.01), p=0.06 | 0.91 (0.89–0.94), p<0.001 |
| Cystic/hereditary/congenital | 0.76 (0.66–0.86), p<0.001 | 0.72 (0.69–0.76), p<0.001 |
| Neoplasms | 1.63 (1.41–1.88), p<0.001 | 1.53 (1.49–1.57), p<0.001 |
| Other | 1.55 (1.41–1.70), p<0.001 | 1.16 (1.14–1.18), p<0.001 |
| Under the care of a nephrologist | 1.14 (1.08–1.21), p<0.001 | 0.93 (0.92–0.94), p<0.001 |
| Insurance prior to ESRD | ||
| Medicare | Ref | Ref |
| Medicaid | 0.93 (0.86–1.00), p=0.06 | 1.00 (0.99–1.02), p=0.6 |
| Private | 0.91 (0.86–0.97), p=0.005 | 0.89 (0.88–0.91), p<0.001 |
| Other | 0.97 (0.90–1.04), p=0.4 | 0.98 (0.97–0.99), p<0.001 |
| Uninsured | 0.65 (0.59–0.72), p<0.001 | 0.71 (0.69–0.73), p<0.001 |
| Comorbid disease | ||
| CHF | 1.21 (1.14–1.28), p<0.001 | 1.31 (1.30–1.33), p<0.001 |
| ASHD | 1.12 (1.05–1.20), p<0.001 | 1.05 (1.03–1.06), p<0.001 |
| CVD | 1.04 (0.94–1.14), p=0.5 | 1.05 (1.04–1.07), p<0.001 |
| PVD | 1.19 (1.10–1.29), p<0.001 | 1.11 (1.10–1.12), p<0.001 |
| DM | ||
| None | Ref | Ref |
| On no medication | 1.00 (0.90–1.12), p=0.9 | 0.95 (0.93–0.97), p<0.001 |
| On oral medication(s) | 1.04 (0.96–1.14), p=0.3 | 0.96 (0.94–0.97), p<0.001 |
| On insulin | 1.40 (1.31–1.50), p<0.001 | 1.09 (1.08–1.11), p<0.001 |
| COPD | 1.21 (1.09–1.35), p<0.001 | 1.24 (1.22–1.26), p<0.001 |
| Current tobacco smoker | 1.12 (1.01–1.24), p=0.04 | 1.07 (1.05–1.09), p<0.001 |
| History of cancer | 1.11 (1.00–1.24), p=0.04 | 1.27 (1.25–1.29), p<0.001 |
| Alcohol dependence | 1.94 (1.64–2.29), p<0.001 | 1.27 (1.23–1.32), p<0.001 |
| Drug dependence | 1.10 (0.84–1.44), p=0.5 | 1.12 (1.08–1.17), p<0.001 |
| Inability to ambulate | 1.44 (1.23–1.69), p<0.001 | 1.64 (1.62–1.66), p<0.001 |
Adjusted hazard ratios with 95% confidence intervals shown.
Figure 1. Mortality by hemodialysis access, stratified by waitlist status.
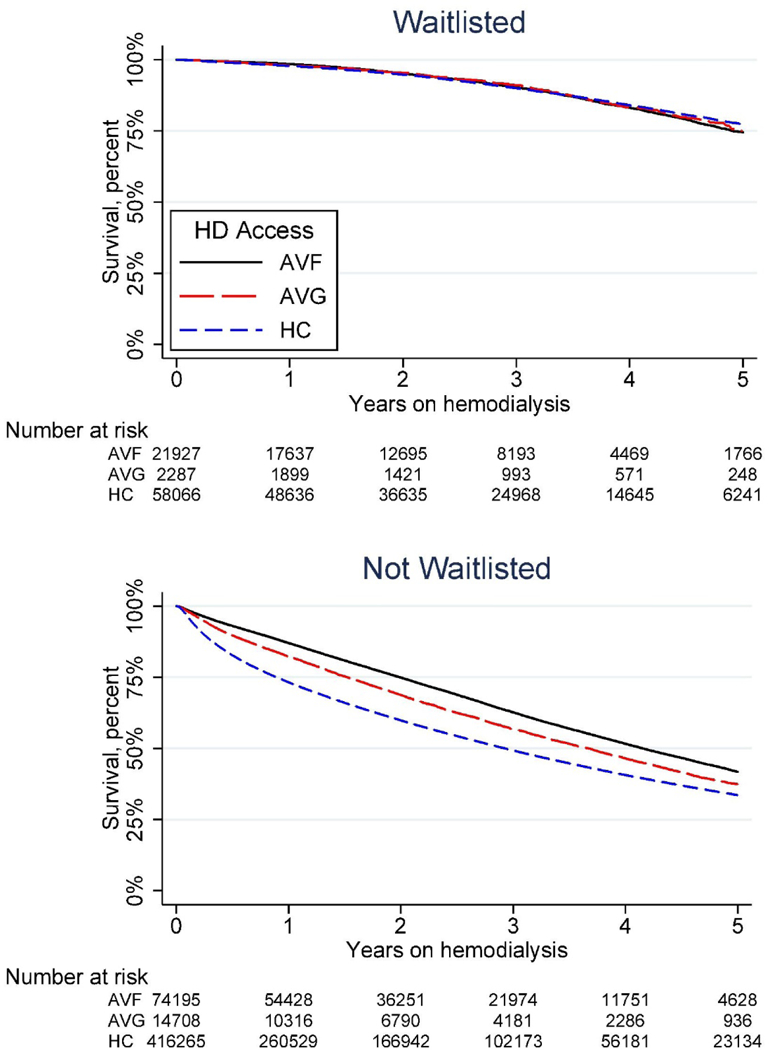
Among patients who were on the waitlist, those with an AVF had a 12% lower risk of mortality (aHR 0.88, 95% CI 0.84–0.93, p<0.001) and those with an AVG had no difference in mortality (aHR 0.95, 95% CI 0.84–1.08, p=0.5) compared to HC. Among patients not on the waitlist, those with an AVF had a 34% lower risk of mortality (aHR 0.66, 95% CI 0.65–0.67, p<0.001) and those with an AVG had a 21% lower risk of mortality (aHR 0.79, 95% CI 0.77–0.81, p<0.001) compared to HC.
AVG vs. HC
Among those with an AVG at ESRD registration, 5-year mortality for waitlisted subjects was 25.2% compared to 62.6% in those not waitlisted. Transplant waitlisting attenuated the association between access with an AVG vs. HC and mortality (interaction p<0.001). Among patients who were on the waitlist, those with an AVG had no difference in mortality (aHR 0.95, 95% CI 0.84–1.08, p=0.5) (Table 2, Figure 1). Among patients not on the waitlist, those with an AVG had a 21% lower risk of mortality compared to HC (aHR 0.79, 95% CI 0.77–0.81, p<0.001).
Sensitivity analyses
AVF vs. HC sensitivity analysis
The matched subset comparing AVF to HC (n=142,860) demonstrated good balance in matched covariates in the standardized difference in means between the waitlisted and not waitlisted groups (Table 3). The modification of the effect of AVF on mortality by transplant waitlisting was confirmed (p<0.001). For those not transplant waitlisted, AVF was associated with a 32% lower risk of mortality (aHR 0.68, 95% CI 0.66–0.71), while for waitlisted patients AVF was associated with an 11% lower risk of mortality (aHR 0.89, 95% CI 0.85–0.93).
Table 3.
Characteristics of population with AVF versus HC for HD access, before and after propensity score matching by propensity to be waitlisted.
| Unmatched | Matched | |||||
|---|---|---|---|---|---|---|
| Wait- listed n= 71,430 |
Not listed n= 412,744 |
Std. mean diff.* |
Wait- listed n= 71,430 |
Not listed n= 71,430 |
Std. mean diff. |
|
| AVF, % | 28.8 | 16.5 | 0.2718 | 28.8 | 27.7 | 0.0241 |
| Age, mean years | 51 | 65 | −1.0233 | 51 | 52 | −0.0647 |
| Female, % | 35.3 | 42.9 | −0.1593 | 35.3 | 35.0 | 0.0063 |
| BMI, mean units | 29 | 30 | −0.0782 | 29 | 30 | −0.0635 |
| African American, % | 28.2 | 26.4 | 0.0401 | 28.2 | 30.1 | −0.0436 |
| Hispanic, % | 19.5 | 14.2 | 0.1329 | 19.5 | 19.5 | −0.0018 |
| Cause of ESRD, % | ||||||
| Diabetes | 41.9 | 47.7 | −0.1169 | 41.9 | 46.0 | −0.0838 |
| Hypertension | 24.0 | 30.1 | −0.1421 | 24.0 | 25.4 | −0.0321 |
| Glomerulonephritis | 13.0 | 4.3 | 0.2574 | 13.0 | 10.0 | 0.0892 |
| Secondary glomerulonephritis | 3.8 | 1.7 | 0.1130 | 3.8 | 3.5 | 0.0158 |
| Interstitial nephritis | 2.7 | 2.8 | −0.0005 | 2.7 | 2.8 | −0.0040 |
| Cystic/hereditary/ congenital |
7.3 | 1.4 | 0.2268 | 7.3 | 4.5 | 0.1071 |
| Neoplasms | 1.4 | 2.7 | −0.1019 | 1.4 | 1.6 | −0.0095 |
| Other | 5.8 | 9.4 | −0.1553 | 5.8 | 6.2 | −0.0167 |
| Under the care of a nephrologist, % | 74.4 | 65.1 | 0.2127 | 74.4 | 73.2 | 0.0287 |
| Insurance prior to ESRD registration, % | ||||||
| Medicare | 14.0 | 32.2 | −0.5243 | 14.0 | 14.1 | −0.0039 |
| Medicaid | 20.7 | 25.8 | −0.1263 | 20.7 | 21.9 | −0.0296 |
| Private insurance | 39.8 | 16.0 | 0.4870 | 39.8 | 37.1 | 0.0562 |
| Other insurance | 15.8 | 20.2 | −0.1207 | 15.8 | 15.7 | 0.0025 |
| Uninsured | 9.7 | 5.8 | 0.1305 | 9.7 | 11.2 | −0.0511 |
| Comorbid disease, % | ||||||
| CHF | 14.4 | 35.0 | −0.5886 | 14.4 | 14.5 | −0.0044 |
| ASHD | 9.6 | 20.5 | −0.3711 | 9.6 | 9.6 | −0.0010 |
| CVD | 4.2 | 10.1 | −0.2950 | 4.2 | 4.3 | −0.0068 |
| PVD | 5.6 | 14.1 | −0.3726 | 5.6 | 5.7 | −0.0052 |
| DM | 48.4 | 58.5 | −0.2024 | 48.4 | 52.8 | −0.0878 |
| COPD | 2.5 | 11.4 | −0.5746 | 2.5 | 2.2 | 0.0197 |
| Current tobacco smoker | 4.1 | 7.0 | −0.1497 | 4.1 | 4.4 | −0.0181 |
| History of cancer | 2.9 | 8.7 | −0.3394 | 2.9 | 3.0 | −0.0015 |
| Alcohol dependence | 1.0 | 1.8 | −0.0797 | 1.0 | 1.1 | −0.0108 |
| Drug dependence | 0.6 | 1.4 | −0.1199 | 0.6 | 0.6 | −0.0023 |
| Inability to ambulate | 0.9 | 8.4 | −0.8068 | 0.9 | 0.6 | 0.0243 |
Std. mean diff.: standardized mean difference between treated and untreated.
AVG vs. HC sensitivity analysis
The matched subset comparing AVG to HC (n=105,998) demonstrated good balance in matched covariates in the standardized difference in means between the waitlisted and not waitlisted groups (Table 4). There was not significant modification of the effect of AVG on mortality by transplant waitlisting (p=0.09). For those not transplant waitlisted, AVG was associated with 15% lower risk of mortality (aHR 0.85, 95% CI 0.78–0.92), while for waitlisted patients AVG was not statistically significantly associated with risk of mortality (aHR 1.06, 95% CI 0.95–1.19) (Figure 2).
Table 4.
Characteristics of population with AVG versus HC for HD access, before and after propensity score matching by propensity to be waitlisted.
| Unmatched | Matched | |||||
|---|---|---|---|---|---|---|
| Wait- listed n= 52,999 |
Not listed n= 357,969 |
Std. mean diff.* |
Wait- listed n= 52,999 |
Not listed n= 52,999 |
Std. mean diff. |
|
| AVG, % | 4.0 | 3.7 | 0.0160 | 4.0 | 4.2 | −0.0105 |
| Age, mean years | 50 | 65 | −1.0850 | 50 | 50 | −0.0582 |
| Female, % | 37.6 | 44.4 | −0.1413 | 37.6 | 37.0 | 0.0117 |
| BMI, mean units | 29 | 30 | −0.1021 | 29 | 29 | −0.0674 |
| African American, % | 29.8 | 27.2 | 0.0547 | 29.8 | 31.5 | −0.0376 |
| Hispanic, % | 20.6 | 14.5 | 0.1516 | 20.6 | 20.6 | 0.0021 |
| Cause of ESRD, % | ||||||
| Diabetes | 41.6 | 47.1 | −0.1127 | 41.6 | 45.5 | −0.0809 |
| Hypertension | 24.1 | 29.8 | −0.1317 | 24.1 | 25.0 | −0.0210 |
| Glomerulonephritis | 12.9 | 4.1 | 0.2625 | 12.9 | 10.0 | 0.0861 |
| Secondary glomerulonephritis | 4.6 | 1.8 | 0.1322 | 4.6 | 4.1 | 0.0207 |
| Interstitial nephritis | 2.7 | 2.8 | −0.0069 | 2.7 | 2.6 | 0.0011 |
| Cystic/hereditary/ congenital |
5.7 | 1.1 | 0.1964 | 5.7 | 3.8 | 0.0835 |
| Neoplasms | 1.6 | 2.9 | −0.1050 | 1.6 | 1.7 | −0.0114 |
| Other | 6.9 | 10.4 | −0.1385 | 6.9 | 7.2 | −0.0097 |
| Under the care of a nephrologist, % | 66.4 | 60.2 | 0.1315 | 66.4 | 65.8 | 0.0133 |
| Insurance prior to ESRD registration, % | ||||||
| Medicare | 12.7 | 31.7 | −0.5718 | 12.7 | 12.4 | 0.0067 |
| Medicaid | 21.5 | 26.6 | −0.1233 | 21.5 | 22.4 | −0.0214 |
| Private insurance | 39.0 | 15.7 | 0.4777 | 39.0 | 37.2 | 0.0385 |
| Other insurance | 14.9 | 19.7 | −0.1351 | 14.9 | 14.7 | 0.0064 |
| Uninsured | 11.9 | 6.3 | 0.1725 | 11.9 | 13.4 | −0.0447 |
| Comorbid disease, % | ||||||
| CHF | 14.9 | 36.1 | −0.5948 | 14.9 | 14.7 | 0.0045 |
| ASHD | 9.0 | 20.4 | −0.3970 | 9.0 | 8.9 | 0.0037 |
| CVD | 4.2 | 10.3 | −0.3062 | 4.2 | 4.2 | −0.0019 |
| PVD | 5.4 | 14.3 | −0.3895 | 5.4 | 5.5 | −0.0028 |
| DM | 48.3 | 58.5 | −0.2025 | 48.3 | 52.8 | −0.0893 |
| COPD | 2.4 | 11.7 | −0.6098 | 2.4 | 2.0 | 0.0233 |
| Current tobacco smoker | 4.1 | 7.1 | −0.1509 | 4.1 | 4.4 | −0.0127 |
| History of cancer | 2.6 | 8.7 | −0.3770 | 2.6 | 2.6 | 0.0038 |
| Alcohol dependence | 1.2 | 2.0 | −0.0682 | 1.2 | 1.4 | −0.0114 |
| Drug dependence | 0.6 | 1.6 | −0.1199 | 0.6 | 0.6 | 0.0024 |
| Inability to ambulate | 1.0 | 9.2 | −0.8034 | 1.0 | 0.8 | 0.0257 |
Std. mean diff.: standardized mean difference between treated and untreated.
Figure 2. Adjusted hazard ratios and 95% confidence intervals for the association of HD access type with mortality.

Unmatched and matched cohorts demonstrated similar findings.
DISCUSSION
In this national study of patients registering for ESRD benefits, over 80% of patients were initiating HD with an HC. Only 14.0% were waitlisted for kidney transplantation, and only 4.5% received a kidney transplant during the study period. Although the majority of waitlisted patients did not receive a kidney transplant, they had a significantly lower burden of comorbid disease and were more likely to have private insurance, a marker of socioeconomic status. Among waitlisted patients, those with an AVF had a 12% lower risk of mortality compared to those with an HC, and there was no difference between AVG and HC in mortality. Among patients not waitlisted, those with an AVF had a 34% lower risk of mortality compared to those with an HC, and those with an AVG had a 21% lower risk of mortality compared to those with an HC.
Our finding that over 80% of patients initiated HD with an HC at a decade after Kidney Disease Outcomes Quality Initiative (KDOQI) recommendations to begin HD with an AVF is concerning, but reaffirms the findings of others. Slinin et al. found that survival at one year after HD initiation was associated with the number of evidence-based KDOQI guidelines met, and also found that 81% of incident HD patients began HD with an HC, despite more than 57% being in the care of a nephrologist prior to initiation of HD [17]. Indeed, Zarkowsky et al. found that there is marked regional variation in AVF placement [18] and that a third of ESRD patients initiating HD with an HC had been in the care of a nephrologist for over six months [19]. This has led to the creation of simulation models to help nephrologists predict when to refer for AVF placement [20], however we found that initiating HD with an HC remained common.
Interestingly, despite the low likelihood of receiving a kidney transplant, waitlisted patients do not have meaningfully worse mortality with an HC as compared to an AVF or AVG. This suggests that the differences in outcomes with these HD access types might be due to socioeconomic differences and differences in ability to access healthcare. Our propensity score matched analysis demonstrated this concept: these matched subcohorts are all highly likely to have become transplant waitlisted. In these subcohorts, despite having similar age, gender, race, ethnicity, insurance, and comorbid conditions, those who were waitlisted had lower mortality. Many groups have reported similar disparities in HD access type [2,3], access to the transplant waitlist [21,7–9], and more broadly, disparities in access to healthcare [22–24,11,25]. Importantly, this suggests that in order to impact the HD-associated mortality rate, we must improve rates of AVF placement, improve access to the transplant waitlist, and address and modify comorbid conditions in ESRD patients. A counterintuitive finding is that for waitlisted patients, being in the care of a nephrologist was associated with a 14% higher risk of mortality; this may be because patients who were more physiologically ill were more likely to be in the care of a nephrologist prior to HD initiation.
Our study has several limitations worth discussion. First, we are limited by our use of administrative data. This introduces the potential for unmeasured confounders not captured in USRDS data, which cannot be accounted for through regression analysis or propensity score matching. Still, USRDS is a near complete sample of the population on HD in the US [13], and limitations in granularity of data are balanced by the generalizability of our conclusions to the US ESRD population. Second, our comparison of transplant waitlisted to not waitlisted subjects is limited by overlap of characteristics of these populations. That is, patients on HD who become waitlisted are very different than those who are not waitlisted, and regression analysis using the full population might extrapolate the effect of HD access type across waitlisted and not waitlisted groups. However, our sensitivity analyses which used a subcohort matched by propensity to be waitlisted confirmed our findings. Finally, our inferences might not be generalizable beyond the US population we studied.
In conclusion, in this national study of how the association between HD access type and mortality varies by transplant waitlisting, we found that for ESRD patients who are waitlisted for kidney transplant, the benefit of AVF is attenuated compared to those patients not waitlisted, but still associated with a lower mortality risk than initiating HD with an HC. For ESRD patients who are not waitlisted, initiating HD with an HC is associated with markedly higher mortality than AVF or AVG. To address the impact of HD access type on mortality, efforts must focus on those patients who are least linked in to the healthcare system, and thus at highest risk for poor outcomes. Still, even for patients with the most access to healthcare, AVF is associated with lower mortality.
ACKNOWLEDGEMENTS
This work was supported by grants number F32DK109662 and K24DK101828 from the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK), F32AG053025 from the National Institute on Aging (NIA), and an American College of Surgeons Resident Research Scholarship. The analyses described here are the responsibility of the authors alone and do not necessarily reflect the views or policies of the Department of Health and Human Services, nor does mention of trade names, commercial products or organizations imply endorsement by the U.S. Government.
Footnotes
COMPLIANCE WITH ETHICAL STANDARDS
The authors of this manuscript have no conflicts of interest to disclose. This study was considered exempt non-human subject research by the Johns Hopkins Medicine Institutional Review Board.
This study was presented as a poster at the Society for Vascular Surgery 2018 Vascular Annual Meeting.
REFERENCES
- 1.Saran R, Robinson B, Abbott KC, Agodoa LYC, Bhave N, Bragg-Gresham J, Balkrishnan R, Dietrich X, Eckard A, Eggers PW, Gaipov A, Gillen D, Gipson D, Hailpern SM, Hall YN, Han Y, He K, Herman W, Heung M, Hirth RA, Hutton D, Jacobsen SJ, Jin Y, Kalantar-Zadeh K, Kapke A, Kovesdy CP, Lavallee D, Leslie J, McCullough K, Modi Z, Molnar MZ, Montez-Rath M, Moradi H, Morgenstern H, Mukhopadhyay P, Nallamothu B, Nguyen DV, Norris KC, O’Hare AM, Obi Y, Park C, Pearson J, Pisoni R, Potukuchi PK, Rao P, Repeck K, Rhee CM, Schrager J, Schaubel DE, Selewski DT, Shaw SF, Shi JM, Shieu M, Sim JJ, Soohoo M, Steffick D, Streja E, Sumida K, Tamura MK, Tilea A, Tong L, Wang D, Wang M, Woodside KJ, Xin X, Yin M, You AS, Zhou H, Shahinian V (2018) US Renal Data System 2017 Annual Data Report: Epidemiology of Kidney Disease in the United States. American journal of kidney diseases : the official journal of the National Kidney Foundation 71 (3s1):A7. doi: 10.1053/j.ajkd.2018.01.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Zarkowsky DS, Arhuidese IJ, Hicks CW, Canner JK, Qazi U, Obeid T, Schneider E, Abularrage CJ, Freischlag JA, Malas MB (2015) Racial/Ethnic Disparities Associated With Initial Hemodialysis Access. JAMA surgery 150 (6):529-536. doi: 10.1001/jamasurg.2015.0287 [DOI] [PubMed] [Google Scholar]
- 3.Hicks CW, Canner JK, Arhuidese I, Zarkowsky DS, Qazi U, Reifsnyder T, Black JH 3rd, Malas MB (2015) Mortality benefits of different hemodialysis access types are age dependent. Journal of vascular surgery 61 (2):449–456. doi: 10.1016/j.jvs.2014.07.091 [DOI] [PubMed] [Google Scholar]
- 4.Malas MB, Canner JK, Hicks CW, Arhuidese IJ, Zarkowsky DS, Qazi U, Schneider EB, Black JH 3rd, Segev DL, Freischlag JA (2015) Trends in incident hemodialysis access and mortality. JAMA surgery 150 (5):441–448. doi: 10.1001/jamasurg.2014.3484 [DOI] [PubMed] [Google Scholar]
- 5.Arhuidese IJ, Obeid T, Hicks C, Qazi U, Botchey I, Zarkowsky DS, Reifsnyder T, Malas MB (2015) Vascular access modifies the protective effect of obesity on survival in hemodialysis patients. Surgery 158 (6):1628–1634. doi: 10.1016/j.surg.2015.04.036 [DOI] [PubMed] [Google Scholar]
- 6.Wolfe RA, Ashby VB, Milford EL, Ojo AO, Ettenger RE, Agodoa LY, Held PJ, Port FK (1999) Comparison of mortality in all patients on dialysis, patients on dialysis awaiting transplantation, and recipients of a first cadaveric transplant. The New England journal of medicine 341 (23):1725–1730. doi: 10.1056/nejm199912023412303 [DOI] [PubMed] [Google Scholar]
- 7.Ashby VB, Kalbfleisch JD, Wolfe RA, Lin MJ, Port FK, Leichtman AB (2007) Geographic variability in access to primary kidney transplantation in the United States, 1996–2005. American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons 7 (5 Pt 2):1412–1423. doi: 10.1111/j.1600-6143.2007.01785.x [DOI] [PubMed] [Google Scholar]
- 8.Tonelli M, Klarenbach S, Rose C, Wiebe N, Gill J (2009) Access to kidney transplantation among remote- and rural-dwelling patients with kidney failure in the United States. Jama 301 (16):1681–1690. doi: 10.1001/jama.2009.545 [DOI] [PubMed] [Google Scholar]
- 9.Mathur AK, Ashby VB, Sands RL, Wolfe RA (2010) Geographic variation in end-stage renal disease incidence and access to deceased donor kidney transplantation. American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons 10 (4 Pt 2):1069–1080. doi: 10.1111/j.1600-6143.2010.03043.x [DOI] [PubMed] [Google Scholar]
- 10.Schold JD, Sehgal AR, Srinivas TR, Poggio ED, Navaneethan SD, Kaplan B (2010) Marked variation of the association of ESRD duration before and after wait listing on kidney transplant outcomes. American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons 10 (9):2008–2016. doi: 10.1111/j.1600-6143.2010.03213.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Schold JD, Gregg JA, Harman JS, Hall AG, Patton PR, Meier-Kriesche HU (2011) Barriers to evaluation and wait listing for kidney transplantation. Clinical journal of the American Society of Nephrology : CJASN 6 (7):1760–1767. doi: 10.2215/cjn.08620910 [DOI] [PubMed] [Google Scholar]
- 12.Fissell RB, Srinivas T, Fatica R, Nally J, Navaneethan S, Poggio E, Goldfarb D, Schold J (2012) Preemptive renal transplant candidate survival, access to care, and renal function at listing. Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association - European Renal Association 27 (8):3321–3329. doi: 10.1093/ndt/gfs012 [DOI] [PubMed] [Google Scholar]
- 13.Foley RN, Collins AJ (2013) The USRDS: what you need to know about what it can and can’t tell us about ESRD. Clinical journal of the American Society of Nephrology : CJASN 8 (5):845–851. doi: 10.2215/cjn.06840712 [DOI] [PubMed] [Google Scholar]
- 14.Stuart EA, King G, Imai K, Ho D (2011) MatchIt: nonparametric preprocessing for parametric causal inference. Journal of Statistical Software 42 (8) [Google Scholar]
- 15.Rubin DB (2001) Using propensity scores to help design observational studies: application to the tobacco litigation. Health Services and Outcomes Research Methodology 2 (3–4):169–188 [Google Scholar]
- 16.Harder VS, Stuart EA, Anthony JC (2010) Propensity score techniques and the assessment of measured covariate balance to test causal associations in psychological research. Psychological methods 15 (3):234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Slinin Y, Guo H, Gilbertson DT, Mau LW, Ensrud K, Rector T, Collins AJ, Ishani A (2010) Meeting KDOQI guideline goals at hemodialysis initiation and survival during the first year. Clinical journal of the American Society of Nephrology : CJASN 5 (9):1574–1581. doi: 10.2215/cjn.01320210 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zarkowsky DS, Hicks CW, Arhuidese I, Canner JK, Obeid T, Qazi U, Schneider E, Abularrage CJ, Black JH 3rd, Freischlag JA, Malas MB (2015) Quality Improvement Targets for Regional Variation in Surgical End-Stage Renal Disease Care. JAMA surgery 150 (8):764–770. doi: 10.1001/jamasurg.2015.1126 [DOI] [PubMed] [Google Scholar]
- 19.Zarkowsky DS, Hicks CW, Malas MB (2016) One-Third of Patients in a National Cohort Initiating Hemodialysis With a Catheter Despite 6 Months of Nephrology Care. JAMA surgery 151 (7):687. doi: 10.1001/jamasurg.2015.5559 [DOI] [PubMed] [Google Scholar]
- 20.Shechter SM, Skandari MR, Zalunardo N (2014) Timing of arteriovenous fistula creation in patients With CKD: a decision analysis. American journal of kidney diseases : the official journal of the National Kidney Foundation 63 (1):95–103. doi: 10.1053/j.ajkd.2013.06.021 [DOI] [PubMed] [Google Scholar]
- 21.Axelrod DA, Lentine KL, Xiao H, Bubolz T, Goodman D, Freeman R, Tuttle-Newhall JE, Schnitzler MA (2014) Accountability for end-stage organ care: implications of geographic variation in access to kidney transplantation. Surgery 155 (5):734–742. doi: 10.1016/j.surg.2013.12.010 [DOI] [PubMed] [Google Scholar]
- 22.Kurella-Tamura M, Goldstein BA, Hall YN, Mitani AA, Winkelmayer WC (2014) State medicaid coverage, ESRD incidence, and access to care. Journal of the American Society of Nephrology : JASN 25 (6):1321–1329. doi: 10.1681/asn.2013060658 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hod T, Goldfarb-Rumyantzev AS (2014) The role of disparities and socioeconomic factors in access to kidney transplantation and its outcome. Renal failure 36 (8):1193–1199. doi: 10.3109/0886022x.2014.934179 [DOI] [PubMed] [Google Scholar]
- 24.Kucirka LM, Grams ME, Balhara KS, Jaar BG, Segev DL (2012) Disparities in provision of transplant information affect access to kidney transplantation. Am J Transplant 12 (2):351–357. doi: 10.1111/j.1600-6143.2011.03865.x [DOI] [PubMed] [Google Scholar]
- 25.Udayaraj U, Ben-Shlomo Y, Roderick P, Casula A, Dudley C, Johnson R, Collett D, Ansell D, Tomson C, Caskey F (2010) Social deprivation, ethnicity, and access to the deceased donor kidney transplant waiting list in England and Wales. Transplantation 90 (3):279–285. doi: 10.1097/TP.0b013e3181e346e3 [DOI] [PubMed] [Google Scholar]


