Abstract
Gulf War Illness (GWI) is a chronic debilitating disease of unknown etiology that affects the brain and has afflicted many veterans of the 1990–91 Gulf War (GW). We showed recently1 that blood serum from patients suffering from GWI exerts detrimental effects on neural cultures, including reduced growth, increased apoptosis, and disruption of neural network function. Remarkably, these adverse effects were prevented by the concomitant addition to the culture of serum from healthy Gulf War (GW) era veterans. We interpreted those findings1 in the context of our hypothesis that GWI is, at least partly, due to circulating pathogenic persistent antigens2, probably coming from vaccines administered to GW veterans who lacked crucial Human Leukocyte Antigen (HLA) class 2 alleles3 and, therefore, could not make antibodies against those antigens; by contrast, healthy GW veterans who received the same vaccines and possessed HLA protection3 made antibodies that neutralized the various antigens. Thus, we hypothesized that the beneficial effect of the healthy serum on preventing the adverse GWI serum effects was due to the presence of antibodies against the persistent antigens. Here we tested this hypothesis by assessing the effect of pooled human immunoglobulin G (IgG) on ameliorating the GWI adverse effects on neural growth and apoptosis in neuroblastoma N2A cultures. We tested this effect in 14 GWI patients and found that IgG exerted a potent ameliorating effect by inhibiting the reduction in growth and increased apoptosis of GWI serum. These results lend support to our persistent antigen hypothesis1,2 and suggest an immunotherapy approach for treating GWI. This approach is further strengthened by our finding that the severity of GWI neurocognitive/mood (NCM) symptoms was positively correlated with the degree of apoptosis caused by GWI serum on the neural culture, thus validating the relevance of the apoptotic effect to NCM symptomatology. Finally, we used this relation to predict NCM scores based on the reduced apoptosis effected by IgG addition and found a predicted reduction in NCM symptom severity by ~60%. Altogether, these findings point to the possible beneficial use of IgG in treating GWI.
Keywords: Gulf War Illness, IgG, Neural cultures, Blood serum, Neural growth, Apoptosis
Introduction
Shortly following the Persian Gulf War of 1990–91, many veterans complained of a variety of chronic physical and neurocognitive symptoms4–7, now referred to as Gulf War Illness (GWI). We described a number of functional and structural brain abnormalities in GWI, notably anomalies in synchronous neural communication patterns7–11 and the presence of subcortical brain atrophy12. Remarkably, we found that this atrophy was absent in veterans carrying the Human Leukocyte Antigen (HLA) allele DRB1*13:022, one of six HLA class 2 alleles that we had reported previously as protective for GWI3. Moreover, the fewer of those alleles carried, the more severe the symptomatology3. The function of HLA class 2 alleles is specific immunity, namely to match to external antigens and present them to CD4+ lymphocytes leading ultimately to the production of specific antibodies by B cells to neutralize the offending antigen. Given these considerations, we hypothesized that the lack of HLA class 2 protection observed in GWI would have allowed offending antigens to persist, hence our “persistent antigen” hypothesis2. We further hypothesized that such persisting antigens could have come from vaccines to which GW veterans were exposed7, ultimately leading to cell damage, low level inflammation13, and chronic disease. Our “persistence antigen” hypothesis above2 predicts that persistent, pathogenic antigens are present in the blood of GWI veterans. It also predicts that healthy GW veterans carrying protective alleles would have specific antibodies in their blood, which could neutralize the hypothesized persistent antigens in GWI serum. As a first step in testing this prediction, (a) we assessed the effect of GWI serum on function and morphology of neural cultures in vitro, and (b) we tested whether GWI-induced neurotoxicity could be neutralized by serum from healthy control GW-era veterans. Indeed, we found1 that (a) GWI serum exerted harmful effects on neural cultures, and (b) those effects were prevented by the addition of serum from healthy GW-era veterans. These findings suggest that healthy serum may contain antibodies against the GWI harmful antigens; if so, such antibodies may hold promise for a successful intervention in treating GWI. Here we tested the possible beneficial effect of pooled human antibodies by adding to the culture pooled human immunoglobulin G (IgG). Although this does not come exclusively from GW-era veterans, it should contain antibodies against a broad range of pathogens partially overlapping with those contained in the vaccines administered to GW veterans. Indeed, we found a highly significant and substantial beneficial effect.
Materials and Methods
Human serum
Serum from 14 veterans with GWI (A-N) and one healthy (control) GW-era veteran was used. The GWI participants were men (age 54.3 ± 2.22 y old, mean ± SEM, N = 14); the control was a man 66.4 y old. GWI participants met both Fukuda et al. (1998)4 and Kansas6 criteria. Each participant completed a standard GWI symptom questionnaire to indicate if they have had a persistent problem over the last 6 months with various symptoms from the following six domains: fatigue, pain, neurological-cognitive-mood (NCM), skin, respiratory, and gastrointestinal. For each symptom rated as present, participants are asked to rate the severity of the symptom as mild, moderate, or severe, and to indicate whether the symptom first became problematic before, during or after deployment to the Gulf. Only symptoms that began during or after Gulf War service are counted toward diagnosis. There were six symptom domains: fatigue, pain, neurological-cognitive-mood, skin, gastrointestinal, and respiratory6. Individual symptom severity was reported in a scale from 0 to 3. For each participant, an average score per domain was calculated; since we used neural cultures for testing, only the NCM severity score was used in the analysis below.
Participants provided informed consent and were financially compensated for their time. Study protocols were approved by the appropriate Institutional Review Boards. All participants were in good health; medications were unremarkable. Participants provided a blood sample that was used for the culture experiments below and HLA genetic testing.
HLA genotyping
DNA isolation was carried out from 3 ml of whole blood drawn in EDTA tubes, using a commercially available kit (ArchivePure cat. 2300730) from 5Prime (distributed by Fisher Scientific or VWR) with an expected yield of 50–15μg of DNA. The purified DNA samples were sent to Histogenetics (http://www.histogenetics.com/) for high-resolution HLA Sequence-based Typing (SBT; details are given in https://bioinformatics.bethematchclinical.org/HLA-Resources/HLA-Typing/High-Resolution-Typing-Procedures/ and https://bioinformatics.bethematchclinical.org/WorkArea/DownloadAsset.aspx?id=6482). Their sequencing DNA templates are produced by locus- and group-specific amplifications that include exon 2 and 3 for class I (A, B, C) and exon 2 for class II (DRB1, DRB¾/5, DQB1, and DPB1) and reported as Antigen Recognition Site (ARS) alleles as per ASHI recommendation14. GWI participants had neurocognitive/mood symptoms and lacked any of the six HLA alleles protective for GWI (DRB1*01:01, DRB1*08:11, DRB1*13:02, DQB1*02:02, DPB1*01:01, DRB1*06:01)3. The control participant was homozygote for the DRB1*01:01 protective allele.
Cell cultures
Neuro-2A neuroblastoma (N2A) cells were cultured in Eagle’s minimal essential medium (EMEM, ATCC, VA, USA) containing 10% fetal bovine serum (ThermoFisher Scientific, Waltham, MA) in 24-well plates at a concentration of 50,000–100,000 /well for 48 h. In sequence, N2A cells were transferred in Neurobasal (NB) medium containing N2 supplement and L-glutamine (ThermoFisher Scientific, Waltham, MA), in the absence (medium control) or presence of human serum. Altogether, there were 3 treatments (Table 1): addition of control serum (Control), addition of GWI serum (GWI), and addition of GWI serum plus 10 μg/ml IgG (GWI + IgG). All treatments were run in 5 replicates concurrently. In a separate experiment, IgG was added alone, without serum.
Table 1.
Experimental design of neuroblastoma N2A cultures experiments.
| Source | Serum (ml) | Media (ml) | Total (ml) | |
|---|---|---|---|---|
| Control | GWI | |||
| Control | 0.1 | 0.0 | 0.9 | 1.0 |
| GWI | 0.0 | 0.1 | 0.9 | 1.0 |
| GWI+IgG | 0.1 | 0.1 | 0.8 | 1.0 |
Cell morphology: cell spreading
The effect of pooled human IgG (Cat. No. I4506, Sigma-Aldrich, St. Louis, MO) was examined as follows: The N2A cells were cultured with a sample GWI serum, randomly selected, which was first pre-incubated in the presence or 7 serial concentrations of IgG in PBS (100-, 50-, 25– 12.5-, 6.25– 2.125- and 1.6 μg/ml) for 1h at 37 deg C, and then added to the N2A cultures in a total of one ml NB medium containing N2 supplement and L-glutamine, for 2 more days; the cells were photographed by obtaining images from 5–12 different fields per sample, with a Motic AE2000-Trinocular inverted microscope (Ted Pella, Redding, Ca), with a Zeiss Axiocam 105 color digital camera (Carl Zeiss Microscopy, LLC, Thornwood, NY). Percent cell spreading was then calculated with ImageJ software by measuring the number of cells with processes relative to the total cell number. Based on this experiment, the optimal concentration of IgG was selected to be 10 μg/ml, since it was the lowest concentration exerting maximal preservation of cell spreading. In sequence, 100 μl of each, from 15 samples of sera (14 GWI, 1 control) were incubated each with 10 μg/ml IgG in PBS at 37deg C for 1h and were then added to cultured N2A cells in a total of 1ml NB medium containing N2 supplement and G-glutamine, and incubated for 2 days as mentioned above. The cells were then photographed and evaluated for percent spreading as described above.
Cell Apoptosis
Terminal deoxynucleotidyl transferase mediated dUTP Nick End Labeling assay (TUNEL) assay. The extent of cell apoptosis of Neuro-2A cells was assessed at 2 days post-exposure to serum treatment (Table 1) using 8-chamber glass slides (ThermoFisher Scientific, Waltham, MA) coated with poly-D-lysine at 50 μg/ml. N2A cells were seeded at a concentration of 50,000–70,000 cells per chamber, in 500 μl of Neurobasal/N2/ L-glutamine medium for 2 days. In sequence, 50 μl of each serum pre-incubated for 60 min at 37deg C in the presence or absence of 10 μg/ml of IgG was added in a total of 500 μl of medium for 2 more days. At the end of the incubation period, the cells were examined for apoptosis. Apoptotic cells were detected using the In Situ Cell Death Detection Kit, TMR red (Terminal deoxynucleotidyl transferase (TdT) enzyme and fluorochrome labeling solution), according to the manufacturer’s protocol. Briefly, the cells were fixed in 4% Paraformaldehyde (PFA)/Phosphate Buffer Saline (PBS) for 60 min at room temperature, rinsed with PBS and permeabilized with 0.1% Triton X-100 in PBS for 3 min on ice and 1 min at room temperature. The cells were then incubated with 80 μl of TUNEL reaction mixture for 60 min at 37deg C in the dark (In situ Cell Death Detection Kit, TMR red, ThermoFisher scientific, Waltham, MA). The cells were then washed 3X with PBS and Diamond AntiFade mounting medium with 4’,6-diamidino-2-phenylindole (DAPI) stain (ThermoFisher Scientific, Waltham, MA) was used for visualization of nuclei, using the EVOS FL Cell Imaging System (ThermoFisher Scientific, Waltham, MA). Eight-10 images were obtained from different fields with each different experimental condition. Apoptosis was then calculated with ImageJ software by measuring the number of TUNEL-labeled cells (red nuclei) relative to the total cell number (DAPI-stained nuclei).
Data analysis
Standard statistical methods15 were used to analyze the data, including analysis of variance (ANOVA), linear regression, t-test, etc. All analyses were done using the IBM-SPSS statistical package (version 25).
Results
Cell spreading
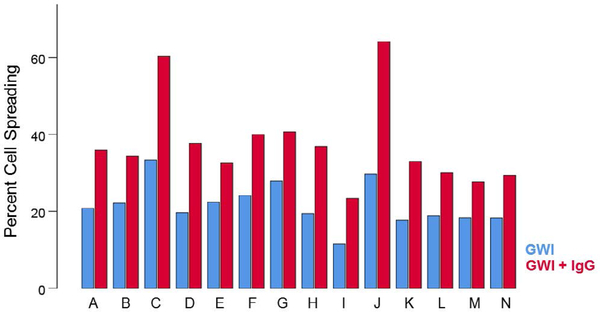
The percentage of cells spreading differed significantly among treatments (Figure 1). GWI spreading was significantly lower than control spreading (P = 4.4 × 10−34, pairwise planned comparison, Least Significant Difference [LSD] test, ANOVA) and also lower than GWI + IgG spreading (P = 3.62 ×10−21); GWI + IgG spreading was lower than control spreading (P = 0.000447). Percent cell spreading in GWI + IgG was consistently higher for each one of the 14 GWI subjects tested (Figure 2).
Figure 1.
Means ± SEM of percent cell spreading in the stated conditions. See text for details.
Figure 2.
Percent cell spreading in the absence (no treatment; blue) and presence (maroon) of pooled human IgG for each GWI patient (A-N). Notice that an improvement in spreading was observed for each one of the 14 patients. See text for details.
Cell apoptosis
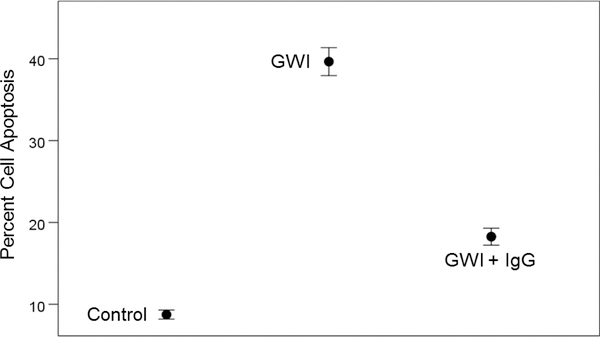
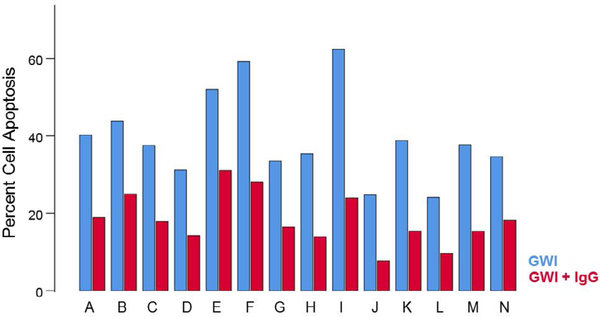
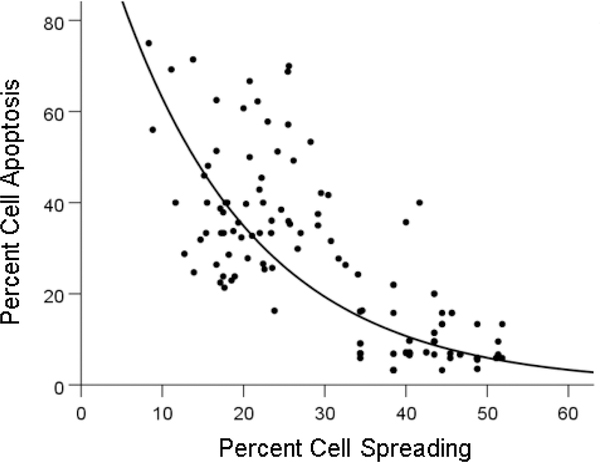
The percentage of apoptotic cells differed significantly among treatments (Figure 3). GWI apoptosis was higher than control apoptosis (P = 6.91 × 10−24) and also higher than GWI + IgG apoptosis (P = 7.2 × 10−22); GWI + IgG apoptosis was higher than control apoptosis (P = 8.98 × 10−11). Percent cell apoptosis in GWI + IgG was consistently lower for each one of the 14 GWI subjects tested (Figure 4). Cell apoptosis decreased with cell spreading in an exponential fashion (Figure 5; P = 7.84 × 10−32, R2 = 0.633, N = 70 GWI + 70 matched control measurements = 140).
Figure 3.
Means ± SEM of percent cell apoptosis in the stated conditions. See text for details.
Figure 4.
Percent cell apoptosis in the absence (no treatment) and presence of pooled human IgG for each GWI patient (A-N). Notice that a reduction in apoptosis was observed for each one of the 14 patients. See text for details.
Figure 5.
Percent cell apoptosis is plotted against percent cell spreading. See text for details.
Association of NCM severity score with cell spreading and apoptosis
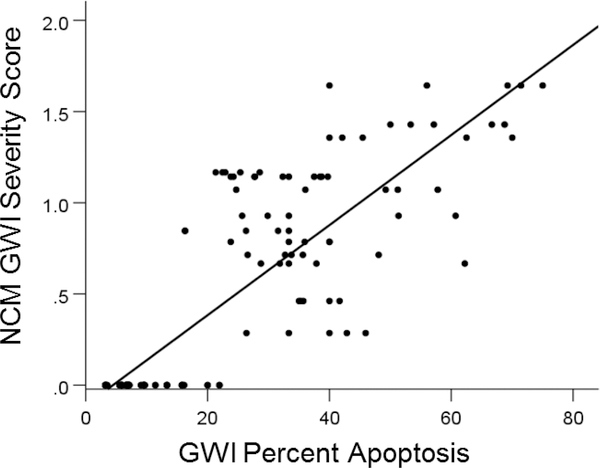
NCM score was higher with lower percentages of cell spreading; specifically, NCM score was negatively correlated with percent cell spreading (r= − 0.808, P = 1.42 × 10−33) and positively correlated with percent apoptosis (r= 0.837, P = 5.84 × 10−38). We quantified further this latter relation because the association was stronger and also because cell apoptosis can happen in the adult brain, whereas cell spreading occurs mostly during development.
The regression equation of NCM vs. apoptosis (Figure 6) was:
| (1) |
Figure 6.
GWI NCM severity score is plotted against percent apoptosis. The values at zero NCM score are those of the control. See text for detail.
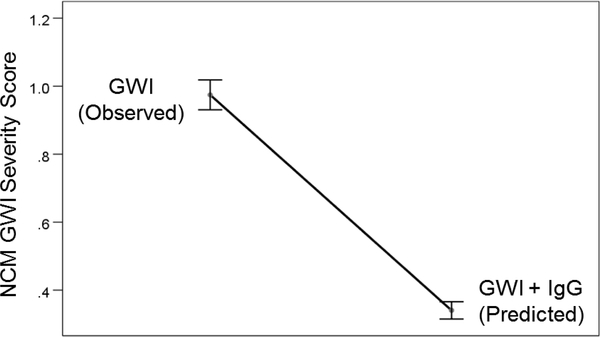
Using this equation, we estimated the predicted NCM score by the IgG intervention. The prediction equation was:
| (2) |
As expected, the predicted NCM scores were significantly lower (by approximately 60%) than the observed NCM scores (Figure 7) (P = 7.27 × 10−24, paired t-test).
Figure 7.
Predicted reduction in GWI NCM symptom severity effected by pooled human IgG. See text for details.
Discussion
Beneficial effect of pooled human IgG
These results document, for the first time, the beneficial effect of pooled human IgG on reducing the negative effect of GWI serum on cell spreading and apoptosis in neural cultures and are in keeping with our previous findings of a beneficial effect of serum from healthy GW veterans to prevent the harmful effect of GWI serum1. Given the positive association between cell apoptosis and GWI NCM severity scores (Figure 6), it is remarkable that addition of pooled human IgG predicts a reduction of the NCM severity scores by ~60% (Figure 7). This finding paves the way to immunotherapy for GWI by administering pooled human IgG, an approved treatment for various conditions, including autoimmunity, pediatric infections and antibody deficiencies16.
Pooled human IgG
Pooled human IgG contains a wide diversity of antibody repertoire with the whole array of variable (Fab/antigen-binding) regions of antibodies in human serum17. Antibodies present in pooled human IgG include antibodies to polio-, Coxsackie B-, and influenza-viruses18, along with a number of other antiviral and antibacterial antibodies19, such as antibodies to tetanus, diphtheria, measles and varicella20, vaccines against all of which were administered to GW veterans7. Additionally, pooled human IgG contains a number of natural antibodies belonging to isotypes IgM, IgG3, and IgA. Recent research about the role of these polyreactive natural antibodies in pathogen elimination provided evidence on the binding of IgG to microbial and viral lectins (present in GW-era vaccine components such as influenza, N. meningitis, S. Typhi, etc.), thus effectively killing invading pathogens, and contributing to innate immunity21.
Pooled human IgG and persistent antigen hypothesis for GWI
According to our persistent antigen/pathogen hypothesis for GWI2, GWI patients lack the ability to produce antibodies to bacterial and/or viral pathogens contained in the administered vaccines. It is likely that the pooled human IgG we used contained a number of antibodies against common pathogens, as well as polyreactive natural antibodies, recognizing viral and microbial vaccine lectins. Thus when added to the neural cells in the presence of GWI serum, the antibodies available bound to pathogens present in this serum with their variable region. In vivo, this binding would be followed by interaction of the Fc IgG portion in order to interact with and signal through the Fcγ receptors on B- and phagocytic-cells to achieve activation of complement and clearance of the microorganisms17. In the cell culture system, the complexes could have been taken up by the scavengers receptors, which bind to a broad spectrum of ligands, including modified and unmodified host-derived molecules or microbial components and pathogens22,23.These receptors, which are also present on neural and neuroblastoma cells24,25, could internalize and degrade the antigen-antibody complexes.
Concluding remarks
Given that pooled human IgG contains pooled antibodies from many individuals, its beneficial effect can be attributed to the presence of such antibodies; this, in turn, indicates that the substances that cause the harmful effects in the neural cultures are neuropathogenic antigens which are eliminated by the antibodies contained in the pooled human IgG. Altogether, our findings provide direct support to our “persistent antigens” hypothesis2 for GWI, namely that GWI is partly due to the harmful effects of circulating pathogenic antigens in GWI serum. We further hypothesized that these antigens may come from vaccines administered7 to GW veterans who lacked the capacity to make antibodies against them, due to the lack of relevant class 2 (specific immunity) HLA alleles3. Given the pooled nature of the IgG used, our study does not provide information concerning the nature of the effective antibodies, and, hence, of the corresponding antigens. However, our results hint at a reasonable possibility regarding this matter, as follows.
Although IgG had a substantial ameliorating effect, it did not totally eliminate the GWI harmful effect, when compared to the effect of the control serum (Figs. 1 and 3). We hypothesize that this is due to the fact that the pooled IgG we used contains antibodies to antigens of common pathogens (e.g. influenza) but not to others that were administered to GW veterans (e.g. anthrax, cholera, yellow fever, Japanese encephalitis, etc.) but should be practically absent from pooled human IgG, since typically people are not exposed to those pathogens. This hypothesis remains to be tested using, in addition to pooled IgG, antibodies specific to those uncommon antigens.
Acknowledgments
Grants
Funded by the University of Minnesota (the American Legion Brain Sciences Chair) and the U.S. Department of Veterans Affairs. The sponsors had no role in the current study design, analysis or interpretation, or in the writing of this paper. The contents do not represent the views of the U.S. Department of Veterans Affairs or the United States Government.
References
- 1.Georgopoulos AP, Tsilibary EP, Souto EP, et al. Adverse effects of Gulf War Illness (GWI) serum on neural cultures and their prevention by healthy serum J Neurol Neuromed. 2018; 3: 19–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.James LM, Christova P, Engdahl BE, et al. Human leukocyte antigen (HLA) and Gulf War Illness (GWI): HLA-DRB1*13:02 spares subcortical atrophy in Gulf War veterans. EBioMedicine. 2017; 26: 126–131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Georgopoulos AP, James LM, Mahan MY, et al. Reduced Human Leukocyte Antigen (HLA) protection in Gulf War Illness (GWI). EBioMedicine 2016; 3: 79–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Fukuda K, Nisenbaum R, Stewart G, et al. Chronic multisymptom illness affecting Air Force veterans of the Gulf War. JAMA. 1998; 280: 981–8. [DOI] [PubMed] [Google Scholar]
- 5.Kang HK, Li B, Mahan CM, et al. Health of US veterans of 1991 Gulf War: a follow-up survey in 10 years. J Occup Environ Med. 2009; 51: 401–10. [DOI] [PubMed] [Google Scholar]
- 6.Steele L Prevalence and patterns of Gulf War illness in Kansas veterans: association of symptoms with characteristics of person, place, and time of military service. Am J Epidemiol. 2000; 152: 992–1002. [DOI] [PubMed] [Google Scholar]
- 7.Institute of Medicine National Research Council. Gulf War and Health: Volume 1. Depleted Uranium, Pyridostigmine Bromide, Sarin, and Vaccines. Washington, DC: National Academies Press, 2000. [PubMed] [Google Scholar]
- 8.Engdahl BE, James LM, Miller RD, et al. A Magnetoencephalographic (MEG) study of Gulf War Illness (GWI). EBioMedicine. 2016; 12: 127–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Georgopoulos AP, James LM, Carpenter AF, et al. Gulf War illness (GWI) as a neuroimmune disease. Exp Brain Res. 2017; 235(10): 3217–3225. [DOI] [PubMed] [Google Scholar]
- 10.James LM, Engdahl BE, Leuthold AC, et al. Brain correlates of Human Leukocyte Antigen (HLA) protection in Gulf War Illness (GWI). EBioMedicine. 2016; 13: 72–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Engdahl BE, James LM, Miller RD, et al. Brain function in Gulf War Illness (GWI) and associated mental health comorbidities. J Neurol Neuromed. 2018; 3: 24–34. [PMC free article] [PubMed] [Google Scholar]
- 12.Christova P, James LM, Engdahl BE, et al. Subcortical brain atrophy in Gulf War Illness. Exp Brain Res. 2017; 235: 2777–2786. [DOI] [PubMed] [Google Scholar]
- 13.Johnson GJ, Slater BC, Leis LA, et al. Blood biomarkers of chronic inflammation in Gulf War Illness. PLoS One. 2016; 11(6): e0157855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cano P, Klitz W, Mack SJ, et al. Common and well-documented HLA alleles: report of the Ad-Hoc committee of the American society for histocompatibility and immunogenetics. Hum Immunol. 2007; 68: 392–417. [DOI] [PubMed] [Google Scholar]
- 15.Zar JH. Biostatistical analysis. 2nd edition. 1974; Prentice-Hall, NJ. [Google Scholar]
- 16.Schwab I, Nimmerjahn F. Intravenous immunoglobulin therapy: how does IgG modulate the immune system. Nat Rev Immunol. 2013; 13: 176–189. [DOI] [PubMed] [Google Scholar]
- 17.Fernández-Cruz E, Alecsandru D, Ramón SS. Mechanisms of action of immune globulin. Clin Exp Immunol. 2009; 157(Suppl 1): 1–2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Funakoshi S, Doi T, Nakajima T, et al. Antimicrobial Effect of Human Serum IgA. Microbiol. Immunol 1982; 22: 227–239. [DOI] [PubMed] [Google Scholar]
- 19.Human Immunglobulins; Transfus. Med. Hemother S. Karger; Gmb H (ed). Freiburg. 2009; 36: 449–459. [PMC free article] [PubMed] [Google Scholar]
- 20.Nobre FA, Garrido da Silva Gonzales I, Simão RM, et al. Antibody levels to tetanus, diphtheria, measles and varicella in patients with primary immunodeficiency undergoing intravenous immunoglobulin therapy: a prospective study. BMC Immunology 2014; 15: 1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Saswati P, Ding JL. Natural Antibodies Bridge Innate and Adaptive Immunity. J. Immunol 2015; 194: 13–20. [DOI] [PubMed] [Google Scholar]
- 22.Yu X, Ch PB, et al. Scavenger Receptors: Emerging Roles in Cancer Biology and Immunology. Adv Cancer Res. 2015; 128: 309–364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zani IA, Stephen SL, Mughal NA, et al. Scavenger Receptor Structure and Function in Health and Disease. Cells. 2015; 4: 178–201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yamauchi K, Tozuka M, Hidaka H, et al. Isoform-specific effect of apolipoprotein E on endocytosis of beta-amyloid in cultures of neuroblastoma cells. Ann Clin Lab Sci. 2002; 32: 65–74. [PubMed] [Google Scholar]
- 25.Panchoo M, Lacko A. Scavenger receptor class B type 1 regulates neuroblastoma cell proliferation, migration and invasion. Biochem Biophys Res Commun. 2018; 495: 614–620. [DOI] [PubMed] [Google Scholar]