Abstract
Reduced expression of caveolin-1 (Cav-1) is an important pathogenic factor in hypertrophic scarring (HTS). Such a reduction can be found in connection with the main known risk factors for HTS, including dark skin, female gender, young age, burn site and severity of the injury. The degree of overexpression of Cav-1 associated with different therapeutic options for HTS correlates with clinical improvements in HTS. This makes endo- or exogenous induction of Cav-1 not only an important therapeutic target for HTS, but also highlights its use as a preventive target to reduce or avoid HTS formation.
Subject terms: Diabetes complications, Type 2 diabetes
Introduction
Fibrosis is a tissue response to injury generally preceded by a long-term inflammatory reaction and connected with excessive deposition of extracellular matrix, especially of collagen. In the skin, fibrosis can lead to production of hypertrophic scars (HTS) which are typical for the full thickness skin injury1 and appear in 30–72% of patients following thermal injury.2 The main risk factors in HTS include dark skin, female gender, and young age.
Different types of non-surgical treatments have been applied to the treatment of HTS, among them various kinds of static and dynamic mechanical forces, light-based therapies, and application of different injectable and topical drugs, including corticosteroids, chemotherapeutics, and immune-modulators.3,4 Recently it was reported that intralesional injections of hyaluronidase can also effectively reduce HTS.5 None of these treatments was confirmed to be effective in the prevention of initial HTS formation.
It is widely accepted that transforming growth factor beta (TGF-β) is a master regulator of fibroblast activation and fibrosis.6 However, it is not clear how the TGF-β pathway is connected with the main risk factors. Neither is it known how it affects the clinical effectiveness of the various non-invasive treatment strategies applied for the reduction of HTS.
TGF-β signaling is tightly connected with plasma membrane structures known as caveolae. Caveolae are characteristic Ω-shaped plasma membrane invaginations forming the nanodomains with typical sizes of 50–100 nm which are present in different cells but especially highly abundant in mechanically stressed cells, such as endothelial cells, fibroblasts, adipocytes, and muscle cells, where they constitute up to 50% of the total surface area and can exist as single invaginations or clusters.7 These nanodomains play an important role in rapid regulation of cellular volumes, cell adhesion, different signal transduction processes, as well as in the processes of endo- and exocytosis.8 Depending on the cell type, caveolae contain different types of caveolins (Cav-1 to 3), all of which are known to be involved in the processes of local proliferation and inflammation in various tissues. Importantly, Cav-1 is involved in the regulation of TGF-β signaling by means of a physical interaction with TGF-β membrane receptors9 and participates in the internalization of these receptors.10 Internalized TGF-β receptors undergo rapid degradation, thereby leading to an effective reduction of TGF-β signaling. Such interactions explain the negative correlation between Cav-1 expression and TGF-β activity observed in pulmonary and dermal fibrosis.11,12 Accordingly, induction of Cav-1 expression leads to suppression of TGF-β signaling and an improvement of fibrosis.10
Recently, the regression of HTS was connected with an induction of endothelial dysfunction causing atrophy of endothelial cells.13 This is consistent with previous reports that the administration of angiogenesis inhibitors can reduce HTS formation. At the same time, angiogenesis inhibitors can form a complex with Cav-1,14 and Cav-1 inhibits endothelial cell proliferation by inducing a cell cycle arrest in the G0/G1 phase.15 These results additionally support the interrelation between Cav-1 expression and HTS formation and regression, and demonstrate that low local Cav-1 expression may be an important pathophysiological factor in HTS.
Here, we connect the risk factors in HTS and structural modification of these scars observed after application of different physical and pharmacological agents with increased expression of Cav-1 in the skin and subcutaneous white adipose tissue (WAT) underneath the lesion-containing skin and discuss whether Cav-1 has the potential to be a therapeutic target in HTS treatment. For this, we will first consider the possible involvement of Cav-1 in some important epipathogenic factors in HTS formation.
Caveolin in inflammation and collagen synthesis
Local tissue inflammation and overexpression of collagens are the typical hallmarks of HTS. Caveolins are significantly involved in both processes.
Cav-1 in inflammation
There are a number of different observations that implicate Cav-1 and Cav-2 in the regulation of local tissue inflammation.16 Cav-1−/− mice are characterized by a low-grade systemic proinflammatory status.17 The elimination of Cav-1 promotes the polarization of M2 macrophages in mice.18 Whereas macrophages of the M1 subtype secrete mediators promoting inflammation, M2 macrophages are known to suppress inflammation and promote fibrosis. Both of these processes must be substantially involved in HTS formation, since systemic macrophage depletion in the subacute phase of wound healing caused by intraperitoneal injections of clodronate significantly reduced HTS formation.19 The dermis of Cav-1−/− mice is indeed strongly infiltrated with macrophages and autophagic cells,20 and it was reported that the ratio of M1 vs. M2 macrophages in the dermis of keloids is significantly shifted to the M2 subtype.21 Moreover, Cav-1−/− mice display an almost complete absence of the dermal WAT (dWAT) layer22 which is involved in inflammatory skin reactions.23–25 Additionally, Cav-1 deficiency provided reduced trafficking of dendritic cells to lymph nodes.26 Glucocorticoid receptors are co-localized with caveolae and downregulation or ablation of Cav-1 lead to impaired functioning of glucocorticoids.27
These observations underline that Cav-1 is not only a marker, but also the target for inflammation.
Cav-1 in collagen expression
We appreciate that the local expression of Cav-1 and collagen 1 (Col1) are negatively correlated. This correlation is especially pronounced in scleroderma,20 keloids,28 and HTS.29 Moreover, Cav-1−/− mice demonstrate significant shift in the synthesis/degradation balance of collagens towards an increase in net synthesis, which correlates with increased local density of myofibroblasts within the skin and the enhanced cell death and fibrosis observed in WAT.30
Myofibroblasts, which are strongly involved in excessive collagen production in HTS, have a high-level expression of the contractile marker α-SMA. These cells typically demonstrate significantly lower levels of Cav-1 expression compared to fibroblasts, which led to the suggestion that there is a negative correlation between Cav-1 and TGF-β expression.11,12 Upon comparing normal fibroblasts and fibroblasts from fibrotic lesions in scleroderma subjects (myofibroblast-rich population of cells), these cells react very differently to the modulation of Cav-1 expression.31 A reduction of Cav-1 expression induces increased α-SMA expression only in normal fibroblasts expressing relatively low levels of α-SMA, but not in myofibroblasts which already express the high levels of this marker. On the other hand, an increase in Cav-1 expression decreases the α-SMA expression in scleroderma fibroblasts, but not in normal fibroblasts.31
These results support the idea that Cav-1 is not merely involved in the regulation of collagen production by myofibroblasts, but may also play a part in the differentiation process of these cells and thus may be directly involved in the pathogenesis of HTS.
Cav-1 in the regulation of heat shock proteins
Fibro-proliferative diseases are characterized by overexpression of some heat shock proteins (HSPs) which are involved in the inflammatory response of the tissue as well as in collagen synthesis.32,33 For example, Hsp27 (up to 10-fold), Hsp47 (up to 16-fold) and Hsp70 (up to 3-fold) were found to be strongly expressed in keloid tissue.32 Hsp27 is a cellular differentiation marker, which can affect the formation of actin microfilaments and regulate endothelial cell migration. Hsp47 is a collagen-specific molecular chaperone, playing a critical role in the biosynthesis and secretion of procollagens. Its overexpression in keloid fibroblasts can induce excessive collagen accumulation through increased collagen synthesis, as demonstrated both in vitro34 and in vivo.35 Hsp70 is a multifunctional chaperone responsible for regulating refolding of misfolded proteins and degradation of unstable proteins, and the various Hsp70 isoforms are distributed in different intracellular, plasma membrane and extracellular compartments. A potent suppression of HSP-production in some diseases (e.g., diabetes) highly correlates with delayed wound healing36 and HSPs may, therefore, be targets to enhance this process.37 At least some HSPs were shown to be co-localized with caveolae in the plasma membrane of different cells,38,39 and HSP activity correlates with expression of Cav-1.39
Recent observations suggest that Hsp27 is critically involved in bleomycin-induced pulmonary fibrosis, thereby influencing the differentiation of lung fibroblasts into myofibroblasts and the overproduction of Col1.40 An induced siRNA knockdown of Hsp27 provided by these authors demonstrated effective suppression of bleomycin-induced pulmonary fibrosis. At the same time, Cav-1 was reported to be a negative regulator of ERK1/2-Hsp27 signaling, thereby influencing the uptake of exosomes and modifying the exosomal exchange between the cells.41
Strong negative correlations between Cav-1 and Hsp47 were very recently reported in myocardial fibrosis.42 Injections of a peptide containing the Cav-1 scaffolding domain led to reversing of Cav-1 deficiency in the tissue and to a significant reduction of expression levels of Col1 and the collagen chaperone Hsp47. Moreover, the anti-fibrotic and anti-inflammatory effects of pirfenidone (used in the treatment of idiopathic pulmonary fibrosis) were not only connected with an inhibition of Col1 expression, but also with a suppression of Hsp47 expression in lung fibroblasts.43 In fact, pirfenidone significantly increases the protein expression levels of Cav-1 in lung tissue subjected to bleomycin-induced pulmonary fibrosis, and this Cav-1 induction strongly correlates with improvements in the lung fibrosis score.44
Similar interactions were reported for Cav-1 and Hsp70. A knockdown of Hsp70 by siRNA strongly reduces mRNA and protein levels of Col1 and Col3, and also induces the MMP-2 expression in keloid-derived fibroblasts.45 At the same time, there is a negative correlation between protein Cav-1 and Hsp70 expressions in tubulointerstitial fibrosis.46 Hsp70 is also functionally involved in the cell surface localization of glycolytic enzyme alpha-enolase,47 which, in turn, is connected with Cav-1 and Annexin 2.48 A knockdown of these caveolae-associated proteins provides markedly decreased expression levels of alpha-enolase, which mediates an increased roughness of the plasma membrane and impairs the ability of cells to adhere to Col1 and Col4.49
These results demonstrate strong negative correlations between Cav-1 expression and functional HSPs, thereby explaining the remarkable overexpression of these proteins in fibro-proliferative disorders.
Cav-1 involvement in the regulation of matrix metalloproteinases
Cav-1 is involved in remodeling of the extracellular matrix through interactions with different matrix metalloproteinases (MMPs).50 Suppression of Cav-1 was shown to activate expression of gelatinases MMP-2 and MMP-9, whereas the induction of Cav-1 causes suppression of these MMPs.51 This effect may be connected to the fact that MMP-2 and MT1-MMP are colocalized with Cav-1 on the cell surface.52
Whereas MMP-2 in mature HTS is strongly increased compared to normal tissue, the activity of MMP-9 in such scars is similar to normal skin.53 The highest activity of MMP-2 was found in keloids, followed by hypertrophic scars, normal skin, and atrophic scars.54 Myofibroblasts suppress the expression of the MMP-2 gene; moreover, expression of MMP-2 is inversely related to the level of α-SMA in these cells.55 This means that a potent increase in the activity of MMP-2 in HTS cannot simply be connected with the appearance of myofibroblasts in the wound. Interestingly, a long-lasting application of mechanical compression to HTS leading to clinical improvements in scar reduction correlates with an almost complete depletion of MMP-2 and a simultaneous increase of MMP-9 activity in HTS.56
Of note, MMP-2 is present but MMP-9 is absent in non-differentiated mesenchymal stem cells; on the other hand, MMP-2 expression is significantly reduced, whereas MMP-9 expression is highly increased in differentiating adipocytes.57 Thus, the observed expression of MMPs in HTS may be connected with the presence of non-differentiated mesenchymal stem cells in mature HTS and with the disappearance of these cells from HTS during the process of scar reduction. This means that dermis-WAT interactions may be an important feature in HTS formation.
Taken together, low expression of Cav-1 in the skin can promote local inflammation and induce fibro-proliferative conditions, leading to the production of HTS. This supports the recently proposed hypothesis that Cav-1 may be a potential therapeutic target for fibrosis.58
Cav-1 expression and risk factors in HTS
Established and widely accepted risk factors for HTS include dark skin, female gender, young age, and burn site and severity.2 If we implicate a reduction of Cav-1 expression in enhanced HTS formation, these risk factors should be associated with a reduction of Cav-1 expression under conditions prone to HTS.
Ethnic differences in Cav-1 expression
Indeed, cells obtained from a healthy African-American population demonstrate much lower Cav-1 content than the cells obtained from a Caucasians population, which correlates with higher rates of systemic sclerosis-related interstitial lung diseases in the African-American population.59,60 Moreover, enhanced expression of chemokine-receptors observed in lung monocytes isolated from African-Americans was driven by low Cav-1 expression.61 Recently, the same authors have reported that Cav-1 cooperates with the master adipogenic factor peroxisome proliferator-activated receptor gamma in maintaining a balance between fibrinogenic and adipogenic differentiation of precursors. This balance is biased towards a fibrogenic path both in subjects with systemic sclerosis and in healthy African-Americans.62 This lends further (though only correlational) support regarding involvement of the WAT in development of systemic sclerosis and dermal fibrosis.23,63
Cav-1 expression in young and old cells
The expression of caveolins is significantly upregulated in chronological aging, and it was even suggested that Cav-1 reduction could serve as a potential anti-aging target.64 Substantial upregulation of Cav-1 in aging was observed in different tissues from 26-month-old rats as well as in human diploid fibroblasts.65 Lack of Cav-1 expression in lung fibroblasts dramatically inhibited premature senescence of these cells.66 Additionally, oxidative stress was shown to upregulate Cav-1 expression, which was connected with premature cellular senescence.67 This means that an increase of Cav-1 overexpression may be typical not only in chronological, but also in photo-induced aging. Indeed, UV-C irradiation (with fluence of 10 J/cm2) of mouse embryonic fibroblasts dramatically increased Cav-1 expression in these cells.68
Sexual dimorphism in Cav-1 expression
Some authors reported that females compared to males demonstrate higher rates of HTS formation in the same body areas (odds ratios of 1.2–1.3),69 whereas others failed to attribute any sexual dimorphism for this process.70 These seemingly contradictory results can at least partly be explained by the fact that expression of Cav-1 is strongly dependent on the level of steroid hormones.71 Whereas expression of Cav-1 in abdominal WAT was shown to be slightly higher in control females than in males, application of steroid hormones reversed this relationship.72 Strong sexual dimorphism was reported also for Cav-1 expression in pulmonary hypertension73 and in osteoclastogenesis.74
Taken together, expression of Cav-1 is gender dependent, whereas its specific level is dependent on the body area and some additional parameters, such as the local levels of sexual hormones in the tissue.
Burn site and severity
Burn site and its severity are known to significantly influence the probability of HTS development. Skin histology reveals a correlation between the presence of cone-like invaginations of the superficial WAT layer into the dermis and body areas that are typically susceptible to HTS formation,75,76 thereby explaining the anatomical site-dependent HTS occurrence as a function whether adipose tissue can undergo fibrosis or directly interact with the dermis.
Adipocytes can indeed locally interact with epithelial, endothelial and mesenchymal cells23 and at least some of these interactions involve Cav-1. For example, perivascular adipose tissue can induce enhanced Cav-1 expression in endothelial cells.77 Interfacial WAT is also directly involved in dermal fibrosis through induction of adipocyte-myofibroblast transition.63,78 In fact, the direct interactions between the dermis and dWAT leading to the substitution of adipose tissue by fibrotic structures is connected with a dysfunctional adiponectin pathway:79 in Cav-1−/− mice, the secretion of adiponectin is reduced and the transmembrane signaling of adiponectin in endothelial cells is significantly blocked. Vice versa, the overexpression of adiponectin correlates with increased expression of Cav-1 in adipocytes.80 Moreover, in human adipocytes, adiponectin receptor 1 (AdipoR-1) interacts with Cav-1, producing an “AdipoR-1/Cav-1 signalsome”.81 In light of these observations, it is not surprising that the silencing of both adiponectin and Cav-1 leads to a severe inflammatory lung injury.82
To provide such interactions between the dermis and subcutis, skin injury must be deep enough to reach the dWAT layer. This can at least partly explain the dependence of the HTS formation on the depth of the skin burn.
Figure 1 summarizes the possible role of Cav-1 in risk factors affecting HTS formation.
Fig. 1.
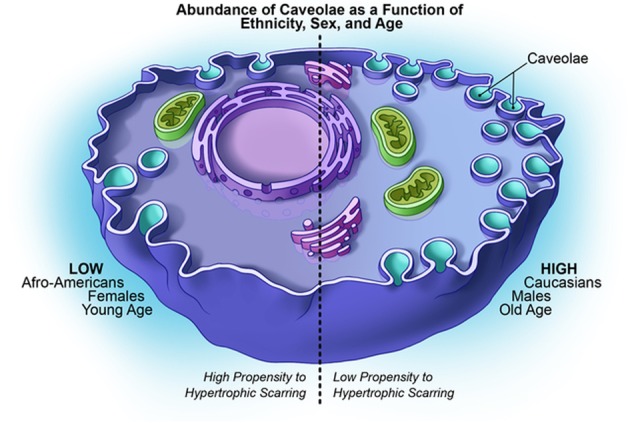
Possible role of Cav-1 in risk factors affecting HTS formation
Cav-1 expression in therapeutic approaches to HTS treatment
Since deficient Cav-1 expression can both enhance the differentiation of myofibroblasts and induce the overexpression of extracellular proteins which can lead to the development of fibro-proliferative conditions in the skin, it seems obvious that endo- or exogenous induction of Cav-1 expression can improve HTS or even serve as preventive target to avoid its formation in the first place. Different treatment methods, including the application of static and dynamic mechanical forces, light-based technologies, various chemotherapeutics, corticosteroids, immunomodulators, anti-allergic drugs, and even hyaluronidase were reported to demonstrate some effectiveness in HTS treatment.4,5 Comparable effectiveness of such very distinct therapeutic options for the treatment of the same skin condition obviously suggests that these approaches must have similar targets which can be reached directly or indirectly. Further, the well-established therapeutic options in HTS have to be discussed from the viewpoint of their involvement in Cav-1 expression.
Therapy options based on application of supra-physiological temperatures
Various light sources have been applied for the treatment of HTS, among them CO2, Er:YAG, Nd:YAG and PDL lasers, as well as intense pulsed light.4,83,84 Different authors have reported high levels of clinical improvements, including a significant height reduction in HTS. Since the wavelengths of these light sources broadly vary between 500 nm and 10,000 nm, the main impact of their application must be the heat transfer to the HTS. Indeed, thermography revealed that application of Nd:YAG laser causes a temperature rise in HTS up to 43–46 °C at a skin depth of about 0.5–1.0 mm.83
On the other hand, supra-physiological temperatures were shown to stimulate expression and re-localization of Cav-1. Mild hyperthermia can significantly increase expression of Cav-1 in different cells.85,86 Moreover, Cav-1 is internalized to the perinuclear region in NIH-3T3 cells at a temperature of 43 °C, but re-localizes to the plasma membrane after return to 37 °C.87
These results clearly demonstrate that application of supra-physiological temperatures enhances Cav-1 expression.
Therapeutic options based on the application of drugs
For further discussion, it should be taken into account that the induced overexpression of Cav-1 can cause cell arrest of fibroblasts in the G0/G1 phase.15,88 Examination of fibroblasts obtained from normal skin as well as from 3-, 6-, 12- and 24-month old HTS reveals significant differences in the cell cycle distribution: whereas fibroblasts from normal skin and from old HTS (12- and 24-month) were predominantly in G0/G1 phase, fibroblasts from the 3- and 6-month old HTS were concentrated in the S and G2/M phases, respectively.89 Such a redistribution of cell cycles for fibroblasts during HTS maturation can significantly influence the cell cycle-specific effects of cytostatic drugs and thus modify clinical outcomes.
It is widely accepted that injections of corticosteroids can effectively improve HTS. Corticosteroids such as dexamethasone and triamcinolone acetonide were applied for the treatment of HTS and keloids. Their effects were explained by suppression of fibroblast proliferation through the TGF-β1 pathway.90,91 Since TGF-β1 is a negative regulator of Cav-1,11,12 we can expect that the application of these corticosteroids enhances Cav-1 expression. Indeed, dexamethasone in physiologically relevant concentrations induces Cav-1 expression in endothelial cells both at the mRNA and protein levels.92 Cav-1 is also essential for non-genomic actions of glucocorticoid receptors, since the dexamethasone effects were not observed in the Cav-1−/− model.93
Bleomycin is a cytotoxic antibiotic which acts as a strong TGF-β suppressor. Cultured human dermal fibroblasts treated with bleomycin demonstrate reduced collagen synthesis even upon exogenous application of TGF-β1.94 Bleomycin is known to induce dermal and lung fibrosis accompanied with a dramatic reduction of Cav-1 in affected tissue.95 This pro-fibrotic effect of bleomycin is actually considered to be connected with the recruitment of new myofibroblasts from adipogenic progenitors63,78 and thus does not contradict its antifibrotic activity by superficial injections in HTS where such progenitors should be absent. On the other hand, bleomycin induces cell senescence and strongly increases expression of Cav-1 and -2 expression in epithelial lung cancer cells.96 Importantly, bleomycin induces significant changes in cell cycle distribution, shifting the Cav-1 positive cells from the G0/G1 into G2/M phase and producing irreversible cell cycle arrest. However, a knockdown of Cav-1 before the bleomycin treatment was able to prevent this effect.
Fluorouracil (5-FU) is a chemotherapeutic drug (inhibitor of thymidine synthase) which was reported to be effective in the therapy of keloids and HTS.97 In contrast to bleomycin, treatment with 5-FU demonstrated no effect on collagen synthesis in cultured human dermal fibroblasts.94 At the same time, 5-FU was able to inhibit fibroblast proliferation. 5-FU can also modulate Cav-1 expression: Cav-1 was strongly upregulated after breast cancer therapy with 5-FU both in vitro and in vivo.98 Very recently, it was reported that the downregulation of Cav-1 expression increases the cell sensitivity to 5-FU,99 which explains the high efficiency of this drug in HTS characterized by low Cav-1 levels.
Some other drugs which were successfully applied for the treatment of HTS demonstrate an interaction with TGF-β. Tranilast inhibits collagen production in cultured fibroblasts100 through suppression of TGF-β1 receptors.101 Imiquimod is an immune-modulator and agonist of toll-like receptor 7 (TLR7). TLR4 and TLR7 participate in fibrosis through the TLR-TGF-β-SMAD signal pathway, increasing the expression of TGF-β.102 Botulinum toxin A is also effective in HTS reduction103 and strongly suppresses the expression of TGF-β1 in fibroblasts derived from HTS.104 Taken together, suppression of the TGF-β1 pathway induced by these drugs generally leads to an increase in the expression of Cav-112 which can significantly modify the structure of the HTS tissue.
Successful application of hyaluronidases (Hyal) for HTS treatment which was reported in ref. 5 can be connected with involvement of hyaluronan in linking the TGF-β receptors to caveolae9 and by the fact that endogenous hyaluronidases are known to counteract the TGF-β activity.105,106 Whereas TGF-β1 was shown to promote growth of mouse fibroblasts L929, this effect was almost completely suppressed in presence of Hyal-1 or -2.105 Stimulation of cells with TGF-β1 resulted in formation of the TGF-β1/Hyal-2 complex on the cell membrane followed by its internalization via endosomes.106 Correspondingly, it can be strongly assumed that exogenous hyaluronidase can also counteract TGF-β1, increasing its internalization and thus demonstrating anti-fibrotic activity.
Whereas no significant cancer risk was reported for the treatment methods discussed above, it should be noted that some primary tumors and metastasis exhibit high Cav-1 expression, which correlates with tumor progression and invasion. Theoretically, application of methods providing a strong local modulation of Cav-1 expression have the potential to be connected with some risk if pre-cancerous lesions are present in the treated area.
Figure 2 summarizes the possible role of Cav-1 as a target in different therapeutic approaches.
Fig. 2.
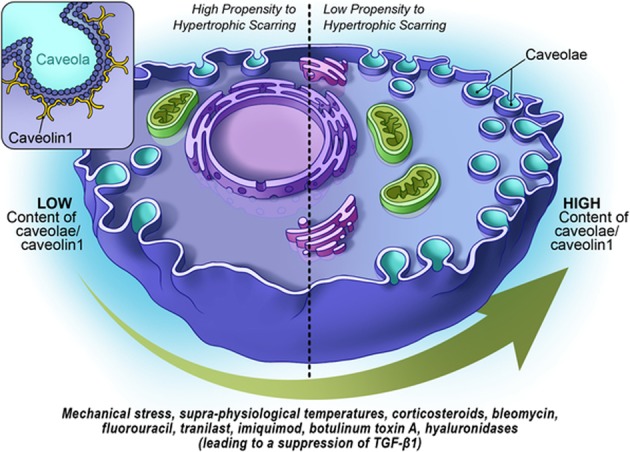
Cav-1 as a target in different therapeutic approaches to HTS treatment
Cav-1 expression as a target in ultrasound treatment of HTS
Ultrasound is not considered as a modern therapy option in HTS because of mixed clinical outcomes and limited evidence for effectiveness. However, recent reports results demonstrate that this treatment option should be revisited.
Caveolae are linked to the actin cytoskeleton,7 and modification of the intracellular network strongly affects the surface density of these caveolar membrane structures. Application of a transient stretch to the cells can cause not only a stiffening of the cytoskeletal structures, but counterintuitively can also provide their softening and fluidization.107,108 Fluidization of the cytoskeleton is strongly dependent on the mechanical strain (relative deformation) amplitude, and the temporal behavior of such a system is also strain-rate dependent.109 Further, low-frequency (about 1 Hz) mechanical forces can effectively fluidize the cytoskeleton and modify the microdomain structure of the plasma membrane at strains of about 10%; at the same time, application of mechanical forces at frequencies of 1 MHz reduces the strain needed for fluidization of the cytoskeleton to about 10−5.110 Such behavior is typical for ultrasound waves where the amplitudes of the particles' displacement in the medium inversely depends on the frequency. Additionally, more recently, there was a report indicating that the application of higher ultrasound intensity and higher ultrasound frequencies induces higher levels of strain in cells, causing stronger fluidization of their cytoskeleton structure.111 It is however noteworthy that high-frequency ultrasound can induce expression of HSPs and that this expression is strongly ultrasound frequency-dependent.112
Recent findings make high-frequency ultrasound waves to an interesting modality for the modulation of the Cav-1 content in target tissues. Indeed, the application of ultrasound with a frequency of about 1 MHz and intensity of up to 2.5 W/cm2 significantly increases the expression of Cav-1 in HEp-2 cells in a time- and dose-dependent manner.113 The application of ultrasound with a frequency of 1.875 MHz and intensity of 0.25 W/cm2 also demonstrated the involvement of Cav-1 in the endothelial tissue response.114 Importantly, the modification of endothelial tissue observed in wildtype animals disappeared in Cav-1−/− mice, which supports a direct involvement of Cav-1 in this process.
Additional effects of high-frequency ultrasound should be the induction of supra-physiological temperatures in HTS, which can also stimulate the expression and re-localization of Cav-1 in this tissue. Spatiotemporal distribution of temperatures and temperature gradients produced by ultrasound waves with frequencies of 3 MHz, 10 MHz, and 19 MHz in the skin and sWAT were recently investigated in ref. 115 Application of ultrasound of 3 MHz, 10 MHz, and 19 MHz with intensities of 1 W/cm2 for 10 s produced temperature rises in different depths of the skin of approximately 1–1.5 °C, 5–9 °C, and 8–16 °C, respectively. At the same time, ultrasound with a frequency of 19 MHz was able to produce high-temperature gradients of up to 14 °C/mm on the interface between skin and WAT. These thermo-mechanical properties produced by high frequency ultrasound build a theoretical foundation for applications of these waves for the treatment of fibro-proliferative diseases, especially of HTS.
Conclusion and future directions
Reduced Cav-1 expression in the skin causes amplification of TGF-β signaling and enhanced differentiation of myofibroblasts leading to the overexpression of extracellular proteins and the development of fibro-proliferative conditions. This highlights Cav-1 prominently as an important factor in HTS pathogenesis. Reduced Cav-1 expression levels are a characteristic feature for the main known risk factors in HTS, such as dark skin, female gender, young age, burn site and its severity. Moreover, many therapeutic avenues for HTS are associated directly or indirectly with an increase in Cav-1 levels. This makes the endo- or exogenous induction of Cav-1 not only an important therapeutic goal for HTS treatment, but also highlights its potential as a preventive target to reduce or avoid HTS formation altogether. Altogether, we argue that more attention should be paid to different pharmacological and physical interventions that lead to an effective modulation of Cav-1 expression in HTS.
Acknowledgements
Graphics created by R. Howdy (Visually Medical, Allen, TX, USA). P.E.S. is supported by NIH grants R01-DK55758, R01-DK099110, P01-DK088761 and P01-AG051459. P.E.S. was also supported by an unrestricted grant from the Novo Nordisk Research Foundation.
Author contributions
I.L.K. and P.E.S. wrote this review article
Competing interests
I.L.K. is the managing partner of Wellcomet GmbH. Wellcomet GmbH provided support in the form of salaries for I.L.K., but did not have any additional role in the decision to publish or the preparation of this manuscript. The commercial affiliation of I.L.K. with Wellcomet GmbH does not alter the adherence to all journal policies on sharing data and materials. P.E.S. declares no competing interests.
Footnotes
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Chiang RS, et al. Current concepts related to hypertrophic scarring in burn injuries. Wound Rep. Regen. 2016;2:466–77. doi: 10.1111/wrr.12432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lawrence JW, et al. Epidemiology and impact of scarring after burn injury: a systematic review of the literature. J. Burn Care Res. 2012;33:136–46. doi: 10.1097/BCR.0b013e3182374452. [DOI] [PubMed] [Google Scholar]
- 3.Viera MH, et al. Innovative therapies in the treatment of keloids and hypertrophic scars. J. Clin. Aesthet. Dermatol. 2010;3:20–6. [PMC free article] [PubMed] [Google Scholar]
- 4.Arno AI, et al. Up-to-date approach to manage keloids and hypertrophic scars: a useful guide. Burns. 2014;40:1255–66. doi: 10.1016/j.burns.2014.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wollina U. Narbenkorrektur mit Hyaluronidase-Injektionen. J. Ästhet. Chir. 2017;10:111–3. doi: 10.1007/s12631-016-0068-x. [DOI] [Google Scholar]
- 6.Meng XM, et al. TGF-[beta]: the master regulator of fibrosis. Nat. Rev. Nephrol. 2016;12:325–38. doi: 10.1038/nrneph.2016.48. [DOI] [PubMed] [Google Scholar]
- 7.Echarri A, Del Pozo MA. Caveolae–mechanosensitive membrane invaginations linked to actin filaments. J. Cell Sci. 2015;128:2747–58. doi: 10.1242/jcs.153940. [DOI] [PubMed] [Google Scholar]
- 8.Sinha B, et al. Cells respond to mechanical stress by rapid disassembly of caveolae. Cell. 2011;144:402–13. doi: 10.1016/j.cell.2010.12.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ito I, et al. Hyaluronan regulates transforming growth factor-β1 receptor compartmentalization. J. Biol. Chem. 2004;279:25326–32. doi: 10.1074/jbc.M403135200. [DOI] [PubMed] [Google Scholar]
- 10.del Galdo F, et al. Decreased expression of caveolin 1 in patients with systemic sclerosis: crucial role in the pathogenesis of tissue fibrosis. Arthritis Rheum. 2008;58:2854–65. doi: 10.1002/art.23791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Xia H, et al. Pathologic caveolin-1 regulation of PTEN in idiopathic pulmonary fibrosis. Am. J. Pathol. 2010;176:2626–37. doi: 10.2353/ajpath.2010.091117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sanders YY, et al. SMAD-independent down-regulation of caveolin-1 by TGF-β: effects on proliferation and survival of myofibroblasts. PloS ONE. 2015;10:e0116995. doi: 10.1371/journal.pone.0116995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wang XQ, Song F, Liu YK. Hypertrophic scar regression is linked to the occurrence of endothelial dysfunction. PloS ONE. 2017;12:e0176681. doi: 10.1371/journal.pone.0176681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wickström SA, Alitalo K, Keski-Oja J. Endostatin associates with integrin α5β1 and caveolin-1, and activates Src via a tyrosyl phosphatase-dependent pathway in human endothelial cells. Cancer Res. 2002;62:5580–9. [PubMed] [Google Scholar]
- 15.Fang K, et al. Overexpression of caveolin-1 inhibits endothelial cell proliferation by arresting the cell cycle at G0/G1 phase. Cell Cycle. 2007;6:199–204. doi: 10.4161/cc.6.2.3740. [DOI] [PubMed] [Google Scholar]
- 16.de Almeida CJG. Caveolin-1 and caveolin-2 can be antagonistic partners in inflammation and beyond. Front. Immunol. 2017;8:1530. doi: 10.3389/fimmu.2017.01530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Codrici E, et al. Caveolin-1-knockout mouse as a model of inflammatory diseases. J. Immunol. Res. 2018;2018:2498576. doi: 10.1155/2018/2498576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Shivshankar P, et al. Caveolin-1 deletion exacerbates cardiac interstitial fibrosis by promoting M2 macrophage activation in mice after myocardial infarction. J. Mol. Cell Cardiol. 2014;76:84–93. doi: 10.1016/j.yjmcc.2014.07.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zhu Z, et al. Systemic depletion of macrophages in the subacute phase of wound healing reduces hypertrophic scar formation. Wound Rep. Regen. 2016;24:644–56. doi: 10.1111/wrr.12442. [DOI] [PubMed] [Google Scholar]
- 20.Castello-Cros R, et al. Scleroderma-like properties of skin from caveolin-1-deficient mice: implications for new treatment strategies in patients with fibrosis and systemic sclerosis. Cell Cycle. 2011;10:2140–50. doi: 10.4161/cc.10.13.16227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Li X, et al. Status of M1 and M2 type macrophages in keloid. Int. J. Clin. Exp. Pathol. 2017;10:11098–105. [PMC free article] [PubMed] [Google Scholar]
- 22.Razani B, et al. Caveolin-1-deficient mice are lean, resistant to diet-induced obesity, and show hypertriglyceridemia with adipocyte abnormalities. J. Biol. Chem. 2002;277:8635–47. doi: 10.1074/jbc.M110970200. [DOI] [PubMed] [Google Scholar]
- 23.Zhang LJ, et al. Dermal adipocytes protect against invasive Staphylococcus aureus skin infection. Science. 2015;347:67–71. doi: 10.1126/science.1260972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kruglikov IL, Scherer PE. Dermal adipocytes: from irrelevance to metabolic targets? Trend Endocrinol. Metab. 2016;27:1–10. doi: 10.1016/j.tem.2015.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kruglikov IL. Interfacial adipose tissue in systemic sclerosis. Curr. Rheum. Rep. 2017;19:4. doi: 10.1007/s11926-017-0627-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Oyarce C, et al. Caveolin-1 expression increases upon maturation in dendritic cells and promotes their migration to lymph nodes thereby favoring the induction of CD8+ T cell responses. Front. Immunol. 2017;8:1794. doi: 10.3389/fimmu.2017.01794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Matthews L, et al. Caveolin mediates rapid glucocorticoid effects and couples glucocorticoid action to the antiproliferative program. Mol. Endocrin. 2008;22:1320–30. doi: 10.1210/me.2007-0154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zhang GY, et al. Role of caveolin‐1 in the pathogenesis of tissue fibrosis by keloid‐derived fibroblasts in vitro. Br. J. Dermatol. 2011;164:623–7. doi: 10.1111/j.1365-2133.2010.10130.x. [DOI] [PubMed] [Google Scholar]
- 29.Zhang GY, et al. Caveolin 1 inhibits transforming growth factor-β1 activity via inhibition of Smad signaling by hypertrophic scar derived fibroblasts in vitro. J. Dermatol. Sci. 2011;62:128–31. doi: 10.1016/j.jdermsci.2010.10.018. [DOI] [PubMed] [Google Scholar]
- 30.Martin S, et al. Caveolin-1 deficiency leads to increased susceptibility to cell death and fibrosis in white adipose tissue: characterization of a lipodystrophic model. PloS ONE. 2012;7:e46242. doi: 10.1371/journal.pone.0046242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Tourkina E, et al. Antifibrotic properties of caveolin-1 scaffolding domain in vitro and in vivo. Am. J. Physiol. Lung. Cell. Mol. Physiol. 2008;294:L843–61. doi: 10.1152/ajplung.00295.2007. [DOI] [PubMed] [Google Scholar]
- 32.Totan S, Echo A, Yuksel E. Heat shock proteins modulate keloid formation. Eplasti. 2011;11:190–202. [PMC free article] [PubMed] [Google Scholar]
- 33.Suarez E, Syed F, Alonso-Rasgado T, Bayat A. Identification of biomarkers involved in differential profiling of hypertrophic and keloid scars versus normal skin. Arch. Dermatol. Res. 2015;307:115–33. doi: 10.1007/s00403-014-1512-4. [DOI] [PubMed] [Google Scholar]
- 34.Chen JJ, et al. Effect of heat shock protein 47 on collagen accumulation in keloid fibroblast cells. Br. J. Dermatol. 2007;156:1188–95. doi: 10.1111/j.1365-2133.2007.07898.x. [DOI] [PubMed] [Google Scholar]
- 35.Chen JJ, et al. Effect of heat shock protein 47 on collagen synthesis of keloid in vivo. ANZ J. Surg. 2011;81:425–30. doi: 10.1111/j.1445-2197.2010.05534.x. [DOI] [PubMed] [Google Scholar]
- 36.Atalay M, et al. Heat shock proteins in diabetes and wound healing. Curr. Protein Pept. Sci. 2009;10:85–95. doi: 10.2174/138920309787315202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kruglikov I, Kruglikova E. Dual treatment strategy by venous ulcers: Pilot study to dual-frequency ultrasound application. J. Cos. Dermatol. Sci. Appl. 2011;1:157–63. [Google Scholar]
- 38.Broquet AH, et al. Expression of the molecular chaperone Hsp70 in detergent-resistant microdomains correlates with its membrane delivery and release. J. Biol. Chem. 2003;278:21601–6. doi: 10.1074/jbc.M302326200. [DOI] [PubMed] [Google Scholar]
- 39.Black AT, et al. Regulation of Hsp27 and Hsp70 expression in human and mouse skin construct models by caveolae following exposure to the model sulfur mustard vesicant, 2-chloroethyl ethyl sulfide. Toxicol. Appl. Pharmacol. 2011;253:112–20. doi: 10.1016/j.taap.2011.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Park AM, et al. Heat shock protein 27 plays a pivotal role in myofibroblast differentiation and in the development of bleomycin-induced pulmonary fibrosis. PloS ONE. 2016;11:e0148998. doi: 10.1371/journal.pone.0148998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Svensson KJ, et al. Exosome uptake depends on ERK1/2-heat shock protein 27 signaling and lipid Raft-mediated endocytosis negatively regulated by caveolin-1. J. Biol. Chem. 2013;288:17713–24. doi: 10.1074/jbc.M112.445403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Pleasant-Jenkins D, et al. Reversal of maladaptive fibrosis and compromised ventricular function in the pressure overloaded heart by a caveolin-1 surrogate peptide. Lab. Invest. 2017;97:370–82. doi: 10.1038/labinvest.2016.153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Nakayama S, et al. Pirfenidone inhibits the expression of HSP47 in TGF-β1-stimulated human lung fibroblasts. Life Sci. 2008;82:210–7. doi: 10.1016/j.lfs.2007.11.003. [DOI] [PubMed] [Google Scholar]
- 44.Yu W, Guo F, Song X. Effects and mechanisms of pirfenidone, prednisone and acetylcysteine on pulmonary fibrosis in rat idiopathic pulmonary fibrosis models. Pharm. Biol. 2017;55:450–5. doi: 10.1080/13880209.2016.1247879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Shin JU, et al. Hsp70 knockdown by siRNA decreased collagen production in keloid fibroblasts. Yonsei Med. J. 2015;56:1619–26. doi: 10.3349/ymj.2015.56.6.1619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.García IM, et al. Caveolin-1–eNOS/Hsp70 interactions mediate rosuvastatin antifibrotic effects in neonatal obstructive nephropathy. Nitric Oxide. 2012;2:95–105. doi: 10.1016/j.niox.2012.05.006. [DOI] [PubMed] [Google Scholar]
- 47.Perconti G, et al. Pro-invasive stimuli and the interacting protein Hsp70 favour the route of alpha-enolase to the cell surface. Sci. Rep. 2017;7:3841. doi: 10.1038/s41598-017-04185-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Zakrzewicz D, et al. The interaction of enolase-1 with caveolae-associated proteins regulates its subcellular localization. Biochem. J. 2014;460:295–307. doi: 10.1042/BJ20130945. [DOI] [PubMed] [Google Scholar]
- 49.Principe M, et al. Alpha-enolase (ENO1) controls alpha v/beta 3 integrin expression and regulates pancreatic cancer adhesion, invasion, and metastasis. J. Hematol. Oncol. 2017;10:16. doi: 10.1186/s13045-016-0385-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Senetta R, et al. Caveolin‐1 as a promoter of tumour spreading: when, how, where and why. J. Cell Mol. Med. 2013;17:325–36. doi: 10.1111/jcmm.12030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Fu P, et al. The different functions and clinical significances of caveolin-1 in human adenocarcinoma and squamous cell carcinoma. Onco Target Ther. 2017;10:819–35. doi: 10.2147/OTT.S123912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Puyraimond A, et al. MMP-2 colocalizes with caveolae on the surface of endothelial cells. Exp. Cell Res. 2001;262:28–36. doi: 10.1006/excr.2000.5069. [DOI] [PubMed] [Google Scholar]
- 53.Ulrich D, et al. Matrix metalloproteinases and tissue inhibitors of metalloproteinases in patients with different types of scars and keloids. J. Plast. Reconstr. Aesthet. Surg. 2010;63:1015–21. doi: 10.1016/j.bjps.2009.04.021. [DOI] [PubMed] [Google Scholar]
- 54.Tanriverdi‐Akhisaroglu S, Menderes A, Oktay G. Matrix metalloproteinase‐2 and‐9 activities in human keloids, hypertrophic and atrophic scars: a pilot study. Cell Biochem. Funct. 2009;27:81–7. doi: 10.1002/cbf.1537. [DOI] [PubMed] [Google Scholar]
- 55.Howard EW, et al. MMP-2 expression by fibroblasts is suppressed by the myofibroblast phenotype. Exp. Cell. Res. 2012;318:1542–53. doi: 10.1016/j.yexcr.2012.03.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Renò F, et al. Release and activation of matrix metalloproteinase-9 during in vitro mechanical compression in hypertrophic scars. Arch. Dermatol. 2002;138:475–8. doi: 10.1001/archderm.138.4.475. [DOI] [PubMed] [Google Scholar]
- 57.Sillat T, et al. Basement membrane collagen type IV expression by human mesenchymal stem cells during adipogenetic differentiation. J. Cell Mol. Med. 2012;16:1485–95. doi: 10.1111/j.1582-4934.2011.01442.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Shihata WA, Putra MR, Chin-Dusting JP. Is there a potential therapeutic role for caveolin-1 in fibrosis? Front. Pharmacol. 2017;8:567. doi: 10.3389/fphar.2017.00567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Silver RM, et al. Racial differences between blacks and whites with systemic sclerosis. Curr. Opin. Rheumatol. 2012;24:642–8. doi: 10.1097/BOR.0b013e328356d9dc. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Reese C, et al. Caveolin‐1 deficiency may predispose African Americans to systemic sclerosis–related interstitial lung disease. Arth. Rheumatol. 2014;66:1909–19. doi: 10.1002/art.38572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Lee R, et al. Enhanced chemokine-receptor expression, function, and signaling in healthy African American and scleroderma-patient monocytes are regulated by caveolin-1. Fibrogenes. Tissue Rep. 2015;8:11. doi: 10.1186/s13069-015-0028-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Lee R, et al. Deficient adipogenesis of scleroderma patient and healthy african american monocytes. Front. Pharmacol. 2017;8:174. doi: 10.3389/fphar.2017.00174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Marangoni RG, et al. Myofibroblasts in murine cutaneous fibrosis originate from adiponectin‐positive intradermal progenitors. Arth. Rheumatol. 2015;67:1062–73. doi: 10.1002/art.38990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Lee JA, et al. Methyl-β-cyclodextrin up-regulates collagen I expression in chronologically-aged skin via its anti-caveolin-1 activity. Oncotarget. 2015;6:1942–53. doi: 10.18632/oncotarget.3039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Park WY, et al. Up-regulation of caveolin attenuates epidermal growth factor signaling in senescent cells. J. Biol. Chem. 2000;275:20847–52. doi: 10.1074/jbc.M908162199. [DOI] [PubMed] [Google Scholar]
- 66.Volonte D, Galbiati F. Caveolin-1, cellular senescence and pulmonary emphysema. Aging (Albany NY) 2009;1:831–5. doi: 10.18632/aging.100079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Zou H, Stoppani E, Volonte D, Galbiati F. Caveolin-1, cellular senescence and age-related diseases. Mech. Ageing Devel. 2011;132:533–42. doi: 10.1016/j.mad.2011.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Volonte D, Zhang K, Lisanti MP, Galbiati F. Expression of caveolin-1 induces premature cellular senescence in primary cultures of murine fibroblasts stress-induced premature senescence upregulates the expression of endogenous caveolin-1. Mol. Biol. Cell. 2002;13:2502–17. doi: 10.1091/mbc.01-11-0529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Gangemi EN, et al. Epidemiology and risk factors for pathologic scarring after burn wounds. Arch. Facial Plast. Surg. 2008;10:93–102. doi: 10.1001/archfaci.10.2.93. [DOI] [PubMed] [Google Scholar]
- 70.Li-Tsang CW, Lau JC, Chan CC. Prevalence of hypertrophic scar formation and its characteristics among the Chinese population. Burns. 2005;31:610–6. doi: 10.1016/j.burns.2005.01.022. [DOI] [PubMed] [Google Scholar]
- 71.Oh YS, et al. Modulation of insulin sensitivity and caveolin-1 expression by orchidectomy in a nonobese type 2 diabetes animal model. Mol. Med. 2011;17:4–11. doi: 10.2119/molmed.2009.00105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Mukherjee R, Kim SW, Choi MS, Yun JW. Sex-dependent expression of caveolin 1 in response to sex steroid hormones is closely associated with development of obesity in rats. PloS ONE. 2014;9:e90918. doi: 10.1371/journal.pone.0090918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Rafikova O, et al. The sexual dimorphism associated with pulmonary hypertension corresponds to a fibrotic phenotype. Pulm. Circ. 2015;5:184–97. doi: 10.1086/679724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Lee YD, et al. Caveolin-1 regulates osteoclastogenesis and bone metabolism in a sex-dependent manner. J. Biol. Chem. 2015;290:6522–30. doi: 10.1074/jbc.M114.598581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Matsumura H, et al. Cones of skin occur where hypertrophic scar occurs. Wound Rep. Regen. 2001;9:269–77. doi: 10.1046/j.1524-475X.2001.00269.x. [DOI] [PubMed] [Google Scholar]
- 76.Engrav LH, et al. Functional genomics unique to week 20 post wounding in the deep cone/fat dome of the Duroc/Yorkshire porcine model of fibroproliferative scarring. PloS ONE. 2011;6:e19024. doi: 10.1371/journal.pone.0019024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Lee MHH, Chen SJ, Tsao CM, Wu CC. Perivascular adipose tissue inhibits endothelial function of rat aortas via caveolin-1. PLoS ONE. 2014;8:e99947. doi: 10.1371/journal.pone.0099947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Varga J, Marangoni RG. Systemic sclerosis in 2016: Dermal white adipose tissue implicated in SSc pathogenesis. Nat. Rev. Rheumatol. 2017;13:71–2. doi: 10.1038/nrrheum.2016.223. [DOI] [PubMed] [Google Scholar]
- 79.Marangoni RG, et al. Adiponectin is an endogenous anti-fibrotic mediator and therapeutic target. Sci. Rep. 2017;7:4397. doi: 10.1038/s41598-017-04162-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Asterholm IW, Scherer PE. Enhanced metabolic flexibility associated with elevated adiponectin levels. Am. J. Pathol. 2010;176:1364–76. doi: 10.2353/ajpath.2010.090647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Du Y, et al. Adiponectin at physiologically relevant concentrations enhances the vasorelaxative effect of acetylcholine via Cav-1/AdipoR-1 signaling. PloS ONE. 2016;11:e0152247. doi: 10.1371/journal.pone.0152247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Cai L, et al. Loss of caveolin-1 and adiponectin induces severe inflammatory lung injury following LPS challenge through excessive oxidative/nitrative stress. Am. J. Physiol. Lung Cell Mol. Physiol. 2014;306:L566–73. doi: 10.1152/ajplung.00182.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Akaishi S, et al. Nd: YAG laser treatment of keloids and hypertrophic scars. Eplasty. 2012;12:e1. [PMC free article] [PubMed] [Google Scholar]
- 84.Kim DY, Park HS, Yoon HS, Cho S. Efficacy of IPL device combined with intralesional corticosteroid injection for the treatment of keloids and hypertrophic scars with regards to the recovery of skin barrier function: A pilot study. J. Dermatol. Treat. 2015;26:481–4. doi: 10.3109/09546634.2015.1024598. [DOI] [PubMed] [Google Scholar]
- 85.Jung BK, et al. Mild hyperthermia induced by gold nanorod-mediated plasmonic photothermal therapy enhances transduction and replication of oncolytic adenoviral gene delivery. ACS Nano. 2016;10:10533–43. doi: 10.1021/acsnano.6b06530. [DOI] [PubMed] [Google Scholar]
- 86.de Andrade Mello P, et al. Hyperthermia and associated changes in membrane fluidity potentiate P2X7 activation to promote tumor cell death. Oncotarget. 2017;8:67254. doi: 10.18632/oncotarget.18595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Kang YS, Ko YG, Seo JS. Caveolin internalization by heat shock or hyperosmotic shock. Exp. Cell Res. 2000;25:221–8. doi: 10.1006/excr.1999.4792. [DOI] [PubMed] [Google Scholar]
- 88.Galbiati F, et al. Caveolin-1 expression negatively regulates cell cycle progression by inducing G0/G1 arrest via a p53/p21WAF1/Cip1-dependent mechanism. Mol. Biol. Cell. 2001;12:2229–44. doi: 10.1091/mbc.12.8.2229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Jin WH, et al. The expression of Cyclin A and p21cip1 in fibroblasts from hypertrophic scars. Zhonghua Zheng Xing Wai Ke Za Zhi. 2010;26:295–8. [PubMed] [Google Scholar]
- 90.Slavin J, Unemori E, Hunt TK, Amento E. Transforming growth factor beta (TGF-β) and dexamethasone have direct opposing effects on collagen metabolism in low passage human dermal fibroblasts in vitro. Growth Factors. 1994;11:205–13. doi: 10.3109/08977199409046918. [DOI] [PubMed] [Google Scholar]
- 91.Carroll LA, et al. Triamcinolone stimulates bFGF production and inhibits TGF‐β1 production by human dermal fibroblasts. Dermatol. Surg. 2002;28:704–9. doi: 10.1046/j.1524-4725.2002.02012.x. [DOI] [PubMed] [Google Scholar]
- 92.Igarashi J, et al. Dexamethasone induces caveolin-1 in vascular endothelial cells: implications for attenuated responses to VEGF. Am. J. Physiol. Cell Physiol. 2013;304:C790–C800. doi: 10.1152/ajpcell.00268.2012. [DOI] [PubMed] [Google Scholar]
- 93.Samarasinghe RA, et al. Nongenomic glucocorticoid receptor action regulates gap junction intercellular communication and neural progenitor cell proliferation. Proc. Nat. Acad. Sci. USA. 2011;108:16657–62. doi: 10.1073/pnas.1102821108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Hendriks T, Martens MFMW, Huyben CMLC, Wobbes T. Inhibition of basal and TGF β-induced fibroblast collagen synthesis by antineoplastic agents. Implications for wound healing. Br. J. Cancer. 1993;67:545–50. doi: 10.1038/bjc.1993.100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Wang XM, et al. Caveolin-1: a critical regulator of lung fibrosis in idiopathic pulmonary fibrosis. J. Exp. Med. 2006;203:2895–906. doi: 10.1084/jem.20061536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Linge A, et al. Downregulation of caveolin-1 affects bleomycin-induced growth arrest and cellular senescence in A549 cells. Int. J. Biochem. Cell Biol. 2007;39:1964–74. doi: 10.1016/j.biocel.2007.05.018. [DOI] [PubMed] [Google Scholar]
- 97.Shah VV, et al. 5-fluorouracil in the treatment of keloids and hypertrophic scars: a comprehensive review of the literature. Dermatol. Ther. 2016;6:169–83. doi: 10.1007/s13555-016-0118-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Wang Z, et al. Caveolin-1 mediates chemoresistance in breast cancer stem cells via β-catenin/ABCG2 signaling pathway. Carcinogenesis. 2014;35:2346–56. doi: 10.1093/carcin/bgu155. [DOI] [PubMed] [Google Scholar]
- 99.Li Z, et al. Downregulation of caveolin-1 increases the sensitivity of drug-resistant colorectal cancer HCT116 cells to 5-fluorouracil. Oncol. Lett. 2017;13:483–7. doi: 10.3892/ol.2016.5390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Yamada H, Tajima S, Nishikawa T. Tranilast inhibits collagen synthesis in normal, scleroderma and keloid fibroblasts at a late passage culture but not at an early passage culture. J. Dermatol. Sci. 1995;9:45–7. doi: 10.1016/0923-1811(94)00355-I. [DOI] [PubMed] [Google Scholar]
- 101.Platten M, et al. N‐[3, 4‐dimethoxycinnamoyl]‐anthranilic acid (tranilast) inhibits transforming growth factor‐β release and reduces migration and invasiveness of human malignant glioma cells. Int. J. Cancer. 2001;93:53–61. doi: 10.1002/ijc.1289. [DOI] [PubMed] [Google Scholar]
- 102.Chen J, Zeng B, Yao H, Xu J. The effect of TLR4/7 on the TGF-β-induced Smad signal transduction pathway in human keloid. Burns. 2013;39:465–72. doi: 10.1016/j.burns.2012.07.019. [DOI] [PubMed] [Google Scholar]
- 103.Xiao Z, Zhang F, Cui Z. Treatment of hypertrophic scars with intralesional botulinum toxin type A injections: a preliminary report. Aesth. Plast. Surg. 2009;33:409–12. doi: 10.1007/s00266-009-9334-z. [DOI] [PubMed] [Google Scholar]
- 104.Xiao Z, Zhang M, Liu Y, Ren L. Botulinum toxin type A inhibits connective tissue growth factor expression in fibroblasts derived from hypertrophic scar. Aesth. Plast. Surg. 2011;35:802–7. doi: 10.1007/s00266-011-9690-3. [DOI] [PubMed] [Google Scholar]
- 105.Chang NS. Transforming growth factor-beta protection of cancer cells against tumor necrosis factor cytotoxicity is counteracted by hyaluronidase. Int. J. Mol. Med. 1998;2:653–62. [PubMed] [Google Scholar]
- 106.Hsu LJ, et al. Transforming growth factor β1 signaling via interaction with cell surface Hyal-2 and recruitment of WWOX/WOX1. J. Biol. Chem. 2009;284:16049–59. doi: 10.1074/jbc.M806688200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Trepat X, et al. Universal physical responses to stretch in the living cell. Nature. 2007;447:592–5. doi: 10.1038/nature05824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Krishnan R, et al. Reinforcement versus fluidization in cytoskeletal mechanoresponsiveness. PloS ONE. 2009;4:e5486. doi: 10.1371/journal.pone.0005486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Oliver M, et al. Remodeling of integrated contractile tissues and its dependence on strain-rate amplitude. Phys. Rev. Lett. 2010;105:158102. doi: 10.1103/PhysRevLett.105.158102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Mizrahi N, et al. Low intensity ultrasound perturbs cytoskeleton dynamics. Soft Matter. 2012;8:2438–43. doi: 10.1039/c2sm07246g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Samandari M, Abrinia K, Mokhtari-Dizaji M, Tamayol A. Ultrasound induced strain cytoskeleton rearrangement: An experimental and simulation study. J. Biomech. 2017;60:39–47. doi: 10.1016/j.jbiomech.2017.06.003. [DOI] [PubMed] [Google Scholar]
- 112.Sontag W, Kruglikov IL. Expression of heat shock proteins after ultrasound exposure in HL-60 cells. Ultrasound Med. Biol. 2009;35:1032–41. doi: 10.1016/j.ultrasmedbio.2008.12.011. [DOI] [PubMed] [Google Scholar]
- 113.Ye Q, et al. Caveolin-1 mediates low-intensity ultrasound-induced apoptosis via downregulation of signal transducer and activator of transcription 3 phosphorylation in laryngeal carcinoma cells. Ultrasound Med. Biol. 2016;42:2253–60. doi: 10.1016/j.ultrasmedbio.2016.04.017. [DOI] [PubMed] [Google Scholar]
- 114.Shindo T, et al. Low-intensity pulsed ultrasound enhances angiogenesis and ameliorates left ventricular dysfunction in a mouse model of acute myocardial infarction. Arterioscler. Thromb. Vasc. Biol. 2016;36:1220–9. doi: 10.1161/ATVBAHA.115.306477. [DOI] [PubMed] [Google Scholar]
- 115.Kruglikov IL. Modeling of the spatiotemporal distribution of temperature fields in skin and subcutaneous adipose tissue after exposure to ultrasound waves of different frequencies. AIP Adv. 2017;7:105317. doi: 10.1063/1.4997833. [DOI] [Google Scholar]


