Abstract
Diagnosis of prostate cancer (PCa) and adequate staging play a fundamental role for clinical and patient care. Despite major advances in biology and imaging, rectal examination and prostate-specific antigen (PSA) blood test remain the cornerstone for screening, and multiparametricmagnetic resonance imaging (mpMRI) for local staging. Recent advances in mpMRI lead to standardised interpretation and increased prescription by clinicians in order to improve detection of clinically significant PCa and select patients requiring targeted biopsies. However its indication remains controversial in biopsy-naïve patients. Nuclear medicine is also in a continuous evolution and utilisation of new radiopharmaceutical agent like choline or 68gallium with computed tomography or magnetic resonance imaging has led to the improvement in the detection of lymph nodes, distant metastases and prostate recurrence. Considering this very heterogneneous disease, combined utilisation of these tools will help clinicians and patients in choosing the most appropriate and personalised treatment.
Keywords: Prostate cancer, Staging, Prostate-specific antigen, Multiparametric magnetic resonance imaging, Markers
1. Introduction
Prostate cancer (PCa) has traditionally been diagnosed by digital rectal examination (DRE) and prostate-specific antigen (PSA) blood test, followed by transrectal ultrasound (TRUS) guided biopsy. However, considering the wide prescription of PSA tests for PCa, and the development of screening programs, more than 60% of PCas are diagnosed in asymptomatic patients, with normal DRE and elevated PSA.
As PCa is a heterogeneous disease, ranging from small, indolent, low-grade tumors, to large, agressive, life-threatening tumors, the primary goal for urologists during baseline evaluation of PCa is, after establishing the presence of the disease, to evaluate local and distant cancer extension, and its aggressiveness by staging. This staging will be important for estimating prognosis, guiding treatment discussed during multi-disciplinary boards and organising patients' follow-up.
Today, TRUS prostate biopsy remains the gold standard examination to confirm diagnosis in all these clinical situations.
2. TRUS prostate biopsy: The key for PCa diagnosis
TRUS-guided systematic biopsy has been the standard diagnostic test for PCa since a landmark study in 1989 which showed that it was superior to digitally directed biopsy sampling of the prostate [1]. However, because needle positioning relative to tumor location is essentially random, TRUS biopsy has a false negative rate of 15%–46% and a tumor undergrading rate of up to 38% when compared with the final Gleason score at radical prostatectomy [2]. It has been also shown that random TRUS biopsy detects low grade indolent cancer and this may lead to overtreatment.
Thus urologists need to improve the selection of patients requiring a biopsy and evaluate their technique to identify and hit “potential aggressive” lesions.
3. Indications of prostate biopsy
3.1. DRE
While rectal examination is carried out in a systematic way to evaluate voiding dysfunction in male, its performance for initial detection of cancer is limited: Most patients detected with PCa during screening PSA program have normal DRE. However, palpation of irregularity or nodule during DRE still remains an indication for prostate biopsy regardless of the level of PSA [3].
3.2. PSA
Total PSA (tPSA) remains the cornerstone of biological test in this evolving landscape of tumor markers, as reported in a recent exhaustive review [4]. High PSA value is clearly associated with an increased risk of PCa. Unfortunately, PSA is organ-specific and not prostate cancer-specific, and this explains the overlap in PSA levels between benign pathologies (BPH) prostatitis and PCa [5], [6]. It is therefore relevant for an urologist sharing with his patient a biopsy indication, to analyse the PSA level in the context of the patient's clinical status, including co-morbidities, age, familial history and patient's preferences.
At the same time, the PCa prevention trial (PCPT) study demonstrated that PCa can be detected even if PSA is below 4 ng/mL, pointing out the fact that there is no PSA cut-off threshold below which the risk of detecting a PCa on biopsy is zero. The choice of a PSA threshold at which a clinician might recommend a biopsy remains controversial. This requires from the urologist a thorough explanation regarding the respective risks and benefits of the procedures and the possible utilisation of other biological markers [7].
3.3. PSA isoforms, PSA density and velocity
To improve screening and management of PCa, several methods to optimize PSA or PSA derivatives have been used.
3.3.1. PSA aged adjusted
The tPSA value does not account for age-related PSA changes due to prostate volume changes related to the development of BPH. Oesterling et al. [8] were the first to suggest considering age-related reference ranges in order to improve cancer detection rates in young men. They recommended thresholds of 2.5, 3.5, 4.5 and 6.5 ng/mL in men in their 40s, 50s, 60s and 70s, respectively, to improve specificity [8].
Loeb et al. [9] examined the usefulness of baseline PSA testing in 13 943 men younger than 60 years from a large prospective PCa screening trial in the United States, in which biopsy was performed for a PSA >2.5 ng/mL or suspicious findings on DRE.
Vickers et al. [10] found that the tPSA value at age 60 years predicts not only a lifetime risk of clinically detected PCa, but also metastases and death from the disease.
Clearly in the discussion with patients about screening, taking into account the results of the Prostate Cancer Intervention versus Observation Trial PIVOT and prostate testing for cancer and treatment (PROTECT) trial, the threshold for indicating biopsy should integrate age and life expectancy [11].
3.3.2. Free PSA (fPSA)
The ratio fPSA to PSA is used in men with normal DRE and PSA levels between 4 and 10 ng/mL. A lower percentage of this ratio (<15%) has been found in men with cancer compared with men without PCa. A meta-analysis determined that the fPSA-to-tPSA ratio is generally only clinically helpful at extreme values of the ratio [12].
In a recent publication of 2310 consecutive Chinese men with PSA value from 4.0 to 10.0 ng/mL undergoing initial prostate biopsy, adding percentage fPSA to PSA was shown to moderately improve the diagnostic accuracy for any PCa and high grade PCa (HGPC), compared with PSA alone in patients ≥ 60 years old but not in patients aged from 40 to 59 years [13].
3.3.3. PSA density (PSAd)
PSAd is calculated as tPSA divided by total prostate volume or transitional prostate volume (TZ PSAd) measured on TRUS to avoid unnecessary biopsies.
To evalulate the efficiencies in the diagnosis of PCa for different PSA and TZ PSAd cut-offs in men with PSA 4.0–10.0 ng/mL but also 10.1–20.0 ng/mL, Ping et al. [13] reviewed a total of 189 men included in the study. They showed that the best cut-off of TZ PSAd in predicting PCa in men with a PSA of 4.0–10.0 ng/mL was 0.37 ng/mL, the sensitivity of which equaled 68.8%, the specificity 72.6%, and for men with a PSA of 10.1–20.0 ng/mL the best cut-off was 0.50 ng/mL with a sensitivity of 70.8% and a specificity of 70.1%.
In a more recent paper reporting on a prospective cohort of 1290 men undergoing extended template biopsy of the prostate, Jue et al. [14] re-examined the role of PSAd (cut-off of ≥0.15 ng/mL) and compared it with PSA for the detection of PCa. There were 34% of men having PSA <4 ng/mL, 56% ranged between 4 and 10 ng/mL and 10% >10 ng/mL. In the PSA range of 4–10 ng/mL, the area under the curve (AUC) of PSAd was significantly greater than that of PSA for any PCa (AUC: 0.70 vs. 0.53, p < 0.0001) and significant PCa (AUC: 0.72 vs. 0.57, p < 0.0001). PSAd performed better than PSA for detecting PCa in men who either had a previous negative biopsy (AUC: 0.69 vs. 0.56, p = 0.0001) or not (AUC: 0.72 vs. 0.67, p = 0.0001). To reduce an unacceptable number of insignificant cancers (41%–58%), the author recommend the use of a continuous score (from 0.05 to 0.20 ng/mL) that is tailored to the individual goals of each patient [14].
After having analysed the paper of Verma et al. [15], Roobol [16] suggested that, as long as the PSA test is the mainstay in the decision to perform a prostate biopsy, information on prostate volume must be taken into account, preferably on the basis of an objective individual risk assessment using an risk calculator (RC) or a nomogram.
3.3.4. PSA velocity (PSAV): PSA doubling time (PSADT)
PSA kinetics have emerged and may have greater specificity for clinically significant PCa.
Specifically, a PSAV greater than 0.75 ng/mL per year was significantly associated with PCa rather than BPH and PSAV could be most useful for risk stratification many years before diagnosis at a time when the tPSA is low [17].
Loeb et al. [9] suggested that the 0.4 ng/mL per year threshold may be more clinically relevant for men with lower PSA levels and highlighted the importance of the tPSA range for interpreting PSAV. However, in a systematic review, Loughlin [18] underlined the heterogeneity of definition used in the literature regarding the number of PSA values and the interval of time over which they are obtained. He concluded that, for screening, when PSAV definitions are rigourously applied, its calculation adds little to the measurement of PSA level alone.
Simultaneously other studies have examined the implications of PSAV on PCa aggressiveness. D'Amico et al. [19] after reviewing 1095 men undergoing radical prostatectomy demonstrated that PSAV >2 ng/mL per year and PSADT measurements were associated with shorter time to death, higher Gleason score, and an advanced pathology.
3.4. Other blood test
3.4.1. Prostate health index (Phi)
A novel approach to improve the clinical performance of PSA is to combine the results of three automated blood tests (tPSA, fPSA, and [-2] proPSA) using a mathematical formula termed the Phi. The Phi test specifically uses the formula ([-2] proPSA/fPSA × tPSA) to calculate and report a Phi result, which can improve the rate of PCa detection compared with either tPSA or fPSA/tPSA alone [20].
Catalona et al. [20] conducted a large prospective multicenter study initiated in the United States from 2003 to 2009 looking at Phi as a predictor of biospy outcome, including 892 men undergoing 6 to 12 or greater core prostate biopsy with a pre-study of tPSA levels of 1.0 to 1.5 ng/mL and non suspicious DRE.
The study showed that Phi had significantly greater specificity at 95% sensitivity compared to %fPSA (16.0% vs. 8.4%; p = 0.015), and it was also more specific than tPSA [20].
The Phi score has an AUC of 0.703 for discrimination of high-grade cancer from low-grade cancer or negative biopsies [20]. De la Calle et al. [21], after analysing two independent prospective cohorts of 561 and 395 subjects, respectively, with no prior prostate biopsy, concluded that Phi had an AUC to detect aggressive PCa of 0.815 and they proposed a threshold of 24 for the Phi value to eliminate 36% of unnecessary biopsies, while missing only 2.5% of high-grade cancers.
In a large review, Lepor et al. [22], after analysing numerous prospective studies from geographically diverse regions concluded that Phi is more specific for PCa detection than existing standard reference tests of tPSA and fPSA. They also pointed out the Phi value to predict a greater risk of clinically significant disease on biopsy and an adverse outcome after prostatectomy and suggested that this test could help monitoring patients on active surveillance.
3.4.2. 4K score
4K score is a four-kallikrein panel including kallikrein-related peptidase 2 (hK2), intact PSA, fPSA and tPSA.
This marker was evaluated retrospectively on the population screened by the European Randomised Study of Screening for Prostate Cancer (ERSPC) study. The review of different studies done on the ERSPC population showed that 4K score increases detection of high grade cancer and the AUC between 0.03 and 0.11. According to the cohorts, 2.5%–12% of high grade cancers were missed [23], [24].
This test was then validated in a prospective multi-institutional study conducted in United States on 1370 men. Twenty-six investigators compared the 4K score with the PCPT RC 2.0 risk calculator and showed that 4K score was superior to predict Gleason score 7 or more with an AUC of 0.82 vs. 0.74 (p < 0.0001). With a cut-off of 9%, this test could reduce the number of prostate biopsies performed for indolent cancers up to 41% while the diagnosis of Gleason score ≥7 could be missed in 24 men (2.4%) including two patients with Gleason score 4 + 4 or higher. With a cut-off of 15%, this test could reduce the number of prostate biopsies performed for indolent cancer up to 58% while the diagnosis of Gleason score ≥7 could be missed in 48 men (4.7%) [25].
Comparison between 4K score and Phi was performed in a study on 531 men in Stockholm county who had a PSA between 3 ng/mL and 15 ng/mL. Both tests reduced the number of unnecessary biopsies and improved discrimination when predicting high grade cancer. Head to head evaluation of 4K score and Phi was similar in term of reduced number of biopsies and missed cancer. The authors could not explain the poorer results of 4K score compared to previously reported results, raising questions about calibration of these biochemical analyses [26].
3.5. Urinary test
PCa gene 3 (PCA3) mRNA, which is over-expressed in men with a PCa has been evaluated for guiding biopsy decisions for men with previously negative biopsies and PSA levels persistently >4 ng/mL. Up to now, this remains the main indication for this test [27]. Different thresholds have been proposed to improve the sensibility/specificity ratio of this test. Usually a threshold of 35 is reported. In a large study of 3073 men undergoing initial prostate sampling of 12–14 areas, Chevli et al. [28] have also demonstrated that PCA3 was a useful tool in identifying patients at risk for PCa.
3.6. Nomograms
It is challenging to accurately assess the clinical significance of PCa at the time of diagnosis and to predict the risk of disease recurrence after definitive treatment. Multiple individual variables have been shown to be useful in this setting.
In this landscape, different risk calculators and nomograms that agregate data were developed in an attempt to better estimate tumor stage and aggressiveness (https://www.mskcc.org/nomograms/prostate). For example, the clinician can use well known tables (https://www.hopkinsmedicine.org/brady-urology-institute/specialties/conditions-and-treatments/postate-cancer/fighting-prostate-cancer/partin-table.htm). Some of them are accessible online and may be useful in helping to determine the potential risk of cancer on an individual basis [29].
4. How to improve TRUS prostate biopsy technique
The gold standard to diagnose PCa with an elevated PSA is the 12 core extended sextant TRUS, the TRUS-guided prostate needle biopsy. However, as show in the figure below, the TRUS non-targeted technique carries a risk of sampling error (Fig. 1A), of missing an aggressive cancer (Fig. 1B) and of overdiagnosing an “indolent” cancer (Fig. 1C). As most lesion are now small and sometime located in regions that are not identifiable by DRE and TRUS, different protocols or strategies to improve the detection of clinically significant cancer were evaluated and published.
Figure 1.
The TRUS non-targeted technique carries a risk of sampling error. (A) Missed anterior and posterior cancer; (B) Under evaluation of TRUS for aggressive cancer indicated in red colour; (C) Over detection of indolent cancer. TRUS, transrectal ultrasound.
4.1. Number of cores, saturation biopsies, perineal approach
Extended prostate biopsy with a scheme of 14, 18, 24 or saturation biopsies up to 50 cores were performed mostly via a trans-perineal approach during repeated biopsy to increase PCa detection. Although the trans-perineal approach has not resulted in a higher detection rate than transrectal biopsies, according to several authors, the complication rate is lower, especially infectious complications with a 0–1% report of fever in the different series [30], [31].
This procedure remains more invasive, and usually requires a general anesthesia which is clearly a limitation. Recently different devices were developed to obtain magnetic resonance imaging (MRI)/ultrasound image fusion using a trans-perineal protocol. This approach could improve the diagnosis of anterior lesions that are always difficult to sample with a transrectal approach. In several papers, authors have reported a positive predictive value (PPV) of 0.43 and 0.63 for Lickert MRI lesions scaled respectively 4 and 5, and a negative predictive value (NPV) of 0.97 for Gleason score ≥4+3 cancer [30], [31], [32].
4.2. Targeted MRI-TRUS fusion biopsy
Due to advances in MRI technology, its use with multiple sequences (T2-Weighted Imaging + diffusion-weighted imaging [DWI] +/− contrast injection dynamic contrasted-enhanced [DCE] MRI) has improved the ability to localize PCa in vivo within the prostate gland.
Simultaneously, standardized interpretation guidelines of MRI, such as the Prostate Imaging Reporting and Data System (PI-RADS version 2) [33], have improved the concordance of radiologists' reports and interpretation of images, leading to an increase in the prescription of this examination and to the development of targeted TRUS/MRI fusion biopsies.
Today, in the 2016 the Committee of Cancerology of the French Association of Urology (CCAFU) guidelines, the primary clinical indication for mpMRI is a persistently elevated PSA, despite previous negative prostate biopsies [34].
4.2.1. mpMRI guided biopsy
Three different methods of incorporating mpMRI into targeted prostate biopsies have been described: In-bore magnetic resonance (MR)-guided fusion, cognitive MR-ultrasound (MR-US) fusion, and device-mediated MR-US fusion.
Although in-bore biopsies under real-time MRI guidance are quite accurate for targeting areas of interest within the prostate, this technique is time-consuming, costly, and limited to expert centers. Cognitive MR-US fusion is expert-dependant. Despite this limit, the tumor detection rate of cognitive fusion seems similar to that obtained by device-mediated fusion.
Compared to random systematic sampling alone, both cognitive and device-mediated fusion appear to improve diagnosis of significant cancer with a sensitivity of MR-US fusion ranging between 80% and 95% [35]. Three platforms using different modes of image fusions are available for transrectal biopsy: Uronav-(http://www.invivocorp.com), Koelis (http://www.koelis.com), and Artemis (http://www.eigen.com).
A recent meta-analysis demonstrated that MRI-targeted biopsy significantly increases the detection rate of clinically significant cancers and decreases the detection on “indolent cancers” [36].
Indeed the quality of MRI study and its radiological interpretation will impact significant cancer detection as shown in the prostate MRI imaging study (PROMIS) study, which reported a sensibility of MRI from 87% to 93%, according to the aggressiveness of the cancer [37].
As most of anterior and apical cancers are missed with systematic random transrectal biopsy, MRI must be discussed before a second biopsy set, and urologist must choose the best approach to reach the MRI lesion [38].
5. Clinical workup after confirmation of diagnosis on biopsy
Primary treatment including active surveillance depends on rectal examination, PSA, biopsy results and Gleason grading as described during the 2014 International Society of Urological Pathology (ISUP) consensus conference, but radiological local and distant staging, surgical risk, performance status and age have also to be performed.
5.1. Local staging
Being able to accurately diagnose the extraprostatic extension and the limits of the tumor are essential to adapt surgical decisions or to select another treatment.
Rectal examination for local staging: The literature has suggested that errors in clinical staging are common, and intra observer variability of DRE may lead to misinterpretation of real staging but can detect more aggressive cancer more selectively [39].
5.2. Radiological evaluation
For local staging, mpMRI is indicated at least 6 weeks after biopsies to minimize artefacts induced by post punctures hemorrhages.
A meta-analysis reported by de Rooij et al. [40] analysed the role of MRI for staging. For extra capsular extension (ECE) (45 studies, 5681 patients), seminal vesicle Invasion (SVI) (34 studies, 5677 patients), and overall stage T3 detection (38 studies, 4001 patients), the results showed a sensitivity and a specificity of 0.57 (95% confidence interval [CI]: 0.49–0.64) and 0.91 (95% CI: 0.88–0.93), 0.58 (95% CI: 0.47–0.68) and 0.96 (95% CI: 0.95–0.97), and 0.61 (95% CI: 0.54–0.67) and 0.88 (95% CI: 0.85–0.91), respectively [38]. Accuracy of mpMRI remains very poor for detection of microscopic capsular extension. Sensitivity for ECE detection increases with the importance of capsular extension and 3T mpMRI could be added to nomograms to improve the prediction of pathological T3a disease while for low-risk patients MRI is not very helpful (Fig. 2).
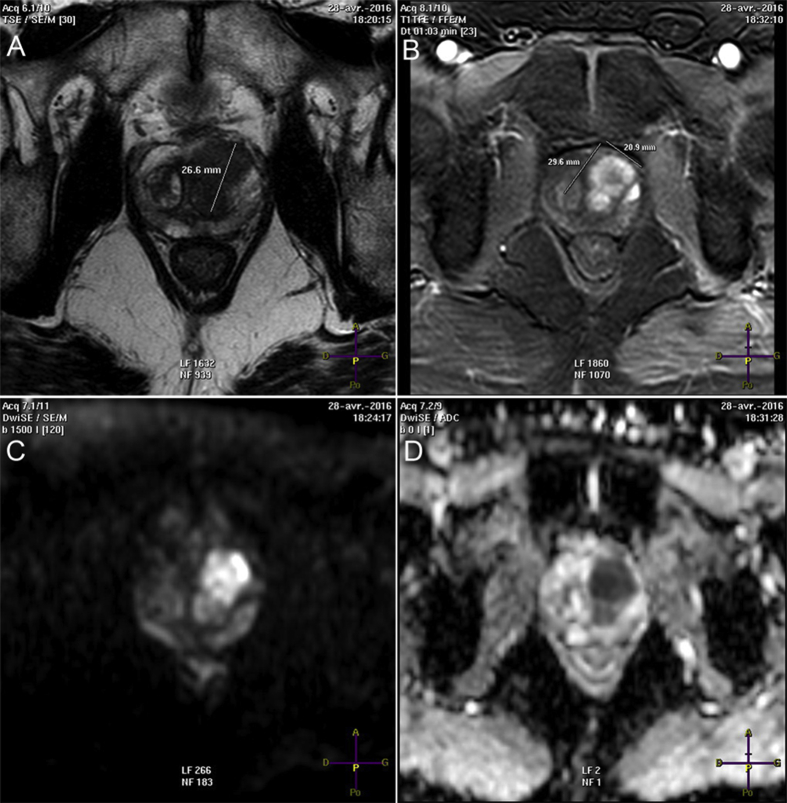
Figure 2.
mpMRI demonstrate anterior T3 cancer, Gleason 4 + 3 on biopsy core, with suspected capsular extension to seminal vesicle. (A) Large hypointense signal on T2 axial-weighted imaging; (B) Tumor enhancement on DCE-Imaging; (C) Diffusion-weighted imaging; (D) Apparent diffusion coefficient map (used as predictor of tumor aggressiveness). mpMRI, multiparametricmagnetic resonance imaging; DCE, dynamic contrasted-enhanced.
Consequently, given its low sensitivity for focal microscopic extraprostatic extension, the National Comprehensive Cancer Network (NCCN) and the European Association of Urology (EAU) guidelines state that “mpMRI is not recommended for local staging in low-risk patients. However, mpMRI can still be useful for treatment planning in selected low-risk patients”. Nevertheless, clinical indications for the use of MRI in the diagnosis and management of PCa are rapidly evolving [41].
5.3. Lymph node staging
Although nomograms can be helpful in predicting lymph node metastases risk, computed tomography (CT) or MRI can be considered as standard imaging modalities for the assessment of lymph node extension for patients with intermediate or high-risk PCa.
Number, shape and size of lymph nodes (diameter 8–12 mm in short axis) are analyzed. But, according to the diameter threshold, sensitivity is ranging from 0.26 to 0.56 (median 0.42) with a specificity around 0.82 [42]. As micro metastases are often undetected today, the ability of CT scan to detect nodal metastases is clearly poor. According to initial risk factors and nomograms, extended lymphadenectomy remains the standard of care for lymph node assessment. The NCCN guidelines recommend CT if the clinical stage is T3/T4 or if there is a probability of nodal involvement of 10% as indicated by a nomogram for T1/T2 lesions.
More recently, choline positon emission tomography (PET)/CT which combine anatomical and functional imaging was introduced. 18F choline and 11C choline were evaluated via a meta-analysis for initial lymph node staging [43]. Many of the authors underlined a high false-negative rate inrelation to the small dimension of a lymph node. However, some investigators have reported a similar PPV for 18F-choline PET/CT in lymph nodes both <5 mm and >5 mm in diameter (PPVs = 82%), suggesting that the false positive (FP) rate can be reduced by acquiring delayed images rather than early images (Fig. 3) [44].
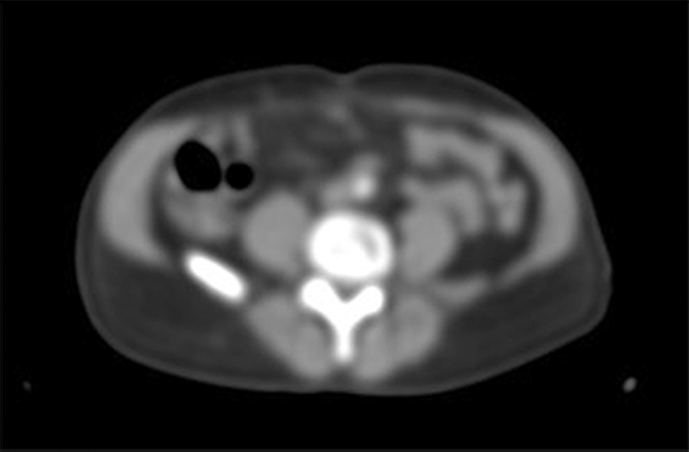
Figure 3.
Suspected Lymph Node on 18F-choline positon emission tomography (confirmed during lymphadenectomy).
In a recent review, the authors report an improvement of staging with Choline PET/CT compared to anatomical imaging with a sensitivity for lymph node detection up to 0.69 (95% Cl: 0.40–0.84) and a good specificity of 0.95 (95% CI: 0.92–0.97), but these studies were limited by the insufficient number of analyzed patients [45].
Today, as medicoeconomic studies are missing for initial staging, choline PET/CT has a limited place and is clearly more useful after local treatment to evaluate PSA recurrence, looking for lymph nodes extension or recurrence outside the pelvic field.
Other innovative PET tracers like Ga68 prostate specific membrane antigen (PSMA) represent a new emerging and promising challenge for PCa lymph node imaging. Different types of radiotracers have been developed because a large number of ligand are available. Budäus et al. [46], on a retrospective cohort of 30 patients which were stratified after prostatectomy by nodal status, have reported with Ga68 PSMA a PPV of 100% and a NPV of 69.2%. These promising results were influenced by lymph node metastases size [46].
5.4. Distant bone metastasis
With a sensibility of 0.79 (95% CI: 0.73–0.83) and a specificity of 0.82 (95% CI: 0.78–0.85) technetium bone scintigraphy remains the gold standard nuclear examination for high risk disease or symptomatic patients to identify bone metastase.
Technetium scintigraphy, despite a poor performance with PSA values <20 ng/mL, still remains the gold standard for detection of bone metastasis with classical uptake of technetium at metastatic sites [47].
The NCCN guidelines suggest that bone scan should be performed in men who meet any of the following criteria: Clinical T1 disease and PSA >20 ng/mL, clinical T2 disease and PSA >10 ng/mL, Gleason score 8, clinical T3 or T4 disease, or symptoms suggestive of metastases [48].
When metastasis are suspected, MRI optimizes morphologic evaluation of helping to determine bone fracture and neurological risk.
5.5. Practical considerations for radiological and nuclear evaluation
In a recent publication, a multidisciplinary expert panel from the American College of Radiology reviewed the guidelines and literature to rate the appropriateness of imaging for different clinical situations. A selection of major results was summarized in this publication and gave an overview of radiological exams indications according to their clinical relevance and could help the clinician organizing his prescription [49].
6. Conclusion
After a complete clinical workup, clinicians should classify patients according to different major classifications (D'Amico, EAU, NCCN). All these classifications use clinical stage, PSA, and Gleason score on biopsy and estimate cancer risk. Even if more precise scores like California San Francisco-Centre of the Prostate Risk Assessment tool (UCSF-CAPRA) are now validated to predict the pathologic stage, and assess disease risk, the great progress in biology, genetics, and imaging will, in a short future, help the clinician to personalize treatment taking into account patient risk factors on a multidisciplinary approach.
Conflicts of interest
The author declares no conflict of interest.
Footnotes
Peer review under responsibility of Second Military Medical University.
References
- 1.Hodge K.K., McNeal J.E., Terris M.K., Stamey T.A. Random systematic versus directed ultrasound guided transrectal core biopsies of the prostate. J Urol. 1989;142:71–75. doi: 10.1016/s0022-5347(17)38664-0. [DOI] [PubMed] [Google Scholar]
- 2.Kvale R., Moller B., Wahlqvist R., Fosså S.D., Berner A., Busch C. Concordance between Gleason scores of needle biopsies and radical prostatectomy specimens: a population-based study. BJU Int. 2009;103:1647–1654. doi: 10.1111/j.1464-410X.2008.08255.x. [DOI] [PubMed] [Google Scholar]
- 3.Loeb S., Catalona W.J. What is the role of digital rectal examination in men undergoing serial screening of serum PSA levels? Nat Clin Pract Urol. 2009;6:68–69. doi: 10.1038/ncpuro1294. [DOI] [PubMed] [Google Scholar]
- 4.Lamy P.J., Allory Y., Gauchez A.S., Asselain B., Beuzeboc P., de Cremoux P. Pronostic biomarkers used for localised prostate cancer management: asystematic review. Eur Urol Focus. 2017;7:30065–30072. doi: 10.1016/j.euf.2017.02.017. [DOI] [PubMed] [Google Scholar]
- 5.Oesterling J.E., Rice D.C., Glenski W.J., Bergstralh E.J. Effect of cystoscopy, prostate biopsy, and transurethral resection of prostate on serum prostate-specific antigen concentration. Urology. 1993;42:276–282. doi: 10.1016/0090-4295(93)90616-i. [DOI] [PubMed] [Google Scholar]
- 6.Nadler R.B., Humphrey P.A., Smith D.S., Catalona W.J., Ratliff T.L. Effect of inflammation and benign prostatic hyperplasia on elevated serum prostate specific antigen levels. J Urol. 1995;154:407–413. doi: 10.1097/00005392-199508000-00023. [DOI] [PubMed] [Google Scholar]
- 7.Thompsen I.M., Goodman P.J., Tangen C.M., Parnes H.L., Minasian L.M., Godley P.A. Long-term survival of participants in prostate cancer prevention trial. N Engl J Med. 2013;369:603–610. doi: 10.1056/NEJMoa1215932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Oesterling J.E., Jacobsen S.J., Cooner W.H. The use of age-specific reference ranges for serum prostate specific antigen in men 60 years old or older. J Urol. 1995;153:1160–1163. [PubMed] [Google Scholar]
- 9.Loeb S., Roehl K.A., Antenor J.A.V., Catalona W.J., Suarez B.K., Nadler R.B. Baseline prostate-specific antigen compared with median prostate-specific antigen for age group as predictor of prostate cancer risk in men younger than 60 years old. Urology. 2006;67:316–320. doi: 10.1016/j.urology.2005.08.040. [DOI] [PubMed] [Google Scholar]
- 10.Vickers A., Cronin A.M., Björk T., Manjer J., Nilsson P.M., Dahlin A. Prostate-specific antigen concentration at age 60 and death or metastasis from prostate cancer: case-control study. BMJ. 2010;341:c4521. doi: 10.1136/bmj.c4521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wilt T.J., Brawer M.K., Jones K.M., Barry M.J., Aronson W.J., Fox S. Radical prostatectomy versus observation for localized prostate cancer. N Engl J Med. 2012;367:203–213. doi: 10.1056/NEJMoa1113162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lee R., Localio A.R., Armstrong K., Malkowicz S.B., Schwartz J.S., Free PSA Study Group A meta-analysis of the performance characteristics of the free prostate-specific antigen test. Urology. 2006;67:762–768. doi: 10.1016/j.urology.2005.10.052. [DOI] [PubMed] [Google Scholar]
- 13.Ping T., Wei D., Keji X. Transition zone PSA density improves the prostate cancer detection rate both in PSA 4.0–10.0 and 10.1–20.0 ng/ml in Chinese men. Urol Oncol. 2013;31:744–748. doi: 10.1016/j.urolonc.2011.06.012. [DOI] [PubMed] [Google Scholar]
- 14.Jue J.S., Barboza M.P., Prakash N.S., Venkatramani V., Sinha V.R., Pavan N. Re-examining prostate-specific antigen (PSA) density: defining the optimal psa range and patients for using psa density to predict prostate cancer using extended template biopsie. Urology. 2017;105:124–128. doi: 10.1016/j.urology.2017.04.015. [DOI] [PubMed] [Google Scholar]
- 15.Verma A., St. Onge J., Dhillon K., Chorneyko A. PSA density improves prediction of prostate cancer. Can J Urol. 2014;21:7312–7321. [PubMed] [Google Scholar]
- 16.Roobol M.J. Words of wisdom. Eur Urol. 2014;66:964–969. doi: 10.1016/j.eururo.2014.08.027. [DOI] [PubMed] [Google Scholar]
- 17.Carter H.B., Ferrucci L., Kettermann A., Landis P., Wright E.J., Epstein J.I. Detection of life-threatening prostate cancer with prostate-specific antigen velocity during a window of curability. J Natl Cancer Inst. 2006;98:1521–1527. doi: 10.1093/jnci/djj410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Loughlin K.R. PSA velocity: a systematic review of clinical applications. Urol Oncol. 2014;32:1116–1125. doi: 10.1016/j.urolonc.2014.04.010. [DOI] [PubMed] [Google Scholar]
- 19.D'Amico A.V., Chen M.H., Roehl K.A., Catalona W.J. Preoperative PSA velocity and the risk of death from prostate cancer after radical prostatectomy. N Engl J Med. 2004;351:125–135. doi: 10.1056/NEJMoa032975. [DOI] [PubMed] [Google Scholar]
- 20.Catalona W.J., Partin A.W., Sanda M.G., Wei J.T., Klee G.G., Bangma C.H. A multicenter study of [-2] pro-prostate specific antigen combined with prostate specific antigen and free prostate specific antigen for prostate cancer detection in the 2.0 to 10.0 ng/ml prostate specific antigen range. J Urol. 2011;185:1650–1655. doi: 10.1016/j.juro.2010.12.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.De la Calle C., Patil D., Wei J.T., Scherr D.S., Sokoll L., Chan D.W. Multicenter evaluation of the prostate health index to detect aggressive prostate cancer in biopsy naive men. J Urol. 2015;194:65–72. doi: 10.1016/j.juro.2015.01.091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lepor A., Catalona W.J., Loeb S. The prostate health index its utility in prostate cancer detection. Urol Clin N Am. 2016;43:1–6. doi: 10.1016/j.ucl.2015.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Vedder M.M., de Bekker-Grob E.W., Lilja H.G., Vickers A.J., van Leenders G.J., Steyerberg E.W. The added value of percentage of free to total prostate-specific antigen, PCA3, and a kallikrein panel to the ERSPC risk calculator for prostate cancer in prescreened men. Eur Urol. 2014;66:1109–1115. doi: 10.1016/j.eururo.2014.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Russo G.I., Regis F., Castelli T., Favilla V., Privitera S., Giardina R. A systematic review and meta-analysis of the diagnostic accuracy of prostate health index and 4-kallikrein panel score in predicting overall and high-grade prostate cancer. Clin Genitourin Cancer. 2017;15:429–439. doi: 10.1016/j.clgc.2016.12.022. [DOI] [PubMed] [Google Scholar]
- 25.Parekh D.J., Punnen S., Sjoberg D.D., Asroff S.W.4, Bailen J.L.5, Cochran J.S.6. A multi-institutional prospective trial in the USA confirms that the 4k score accurately identifies men with high-grade prostate cancer. Eur Urol. 2015;68:464–470. doi: 10.1016/j.eururo.2014.10.021. [DOI] [PubMed] [Google Scholar]
- 26.Nordström T., Vickers A., Assel M., Lilja H., Grönberg H., Eklund M. Comparison between the four-kallikrein panel and prostate health index for predicting prostate cancer. Eur Urol. 2015;68:139–146. doi: 10.1016/j.eururo.2014.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wei J.T., Feng Z., Partin A.W., Brown E., Thompson I., Sokoll L. Can urinary PCA3 supplement PSA in the early detection of prostate cancer? J Clin Oncol. 2014;32:4066–4072. doi: 10.1200/JCO.2013.52.8505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Chevli K.K., Duff M., Walter P., Yu C., Capuder B., Elshafei A. Urinary PCA3 as a predictor of prostate cancer in a cohort of 3073 men undergoing initial prostate biopsy. J Urol. 2014;191:1743–1748. doi: 10.1016/j.juro.2013.12.005. [DOI] [PubMed] [Google Scholar]
- 29.Gandaglia G., Ploussard G., Valerio M., Mattei A., Fiori C., Fossati N. A novel nomogram to identify candidates for extended pelvic lymph node dissection among patients with clinically localized prostate cancer diagnosed with magnetic resonance imaging-targeted and systematic biopsies. Eur Urol. 2018 doi: 10.1016/j.eururo.2018.10.012. [DOI] [PubMed] [Google Scholar]
- 30.Abdollah F., Novara G., Briganti A., Scattoni V., Raber M., Roscigno M. Transrectal versus trans perineal saturation re biopsy of the prostate: is there a difference in cancer detection rate. Urology. 2011;77:921–925. doi: 10.1016/j.urology.2010.08.048. [DOI] [PubMed] [Google Scholar]
- 31.Galfano A. Editorail comment: the European association of Uroogyguidelines methodology: a critical evaluation. Eur Urol. 2009;58:864. doi: 10.1016/j.eururo.2008.07.013. [DOI] [PubMed] [Google Scholar]
- 32.Hansen, Patruno G., Wadhwa K., Gaziev G., Miano R., Barrett T. Magnetic resonance and ultrasound image fusion supported transperineal prostate biopsy using the ginsburg protocol: technique, learning points, and biopsy results. Eur Urol. 2016;70:332–340. doi: 10.1016/j.eururo.2016.02.064. [DOI] [PubMed] [Google Scholar]
- 33.Weinreb J.C., Barentsz J.O., Choyke P.L., Cornud F., Haider M.A., Macura K.J. PI-RADS prostate imaging—reporting and data system: 2015, version 2. Eur Urol. 2016;69:16–40. doi: 10.1016/j.eururo.2015.08.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Rozet F., Hennequin C., Beauval J.B., Beuzeboc P., Cormier L., Fromont G. CCAFU Guidelines on prostate cancer. Prog Urol. 2016;(Suppl 2):S51–S98. [Google Scholar]
- 35.Tyson M.D., Arora S.S., Scarpato K.R., Barocas D. Magnetic resonance-ultrasound fusion prostate biopsy in the diagnosis of prostate cancer. Urol Oncol. 2016;34:326–332. doi: 10.1016/j.urolonc.2016.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Schoots I.G., Roobol M.J., Nieboer D., Bangma C.H., Steyerberg E.W., Hunink M.G. Magnetic resonance imaging-targeted biopsy may enhance the diagnostic accuracy of significant prostate cancer detection compared to standard transrectal ultrasound-guided biopsy: a systematic review and meta-analysis. Eur Urol. 2015;68:438–450. doi: 10.1016/j.eururo.2014.11.037. [DOI] [PubMed] [Google Scholar]
- 37.Ahmed H.U., El-Shater Bosaily A., Brown L.C., Gabe R., Kaplan R., Parmar M.K. Diagnostic accuracy of multi-parametric MRI and TRUS biopsy in prostate cancer (PROMIS): apaired validating confirmatory study. Lancet. 2017;389:815–822. doi: 10.1016/S0140-6736(16)32401-1. [DOI] [PubMed] [Google Scholar]
- 38.Mottet N., Bellmunt J., Bolla M., Briers E., Cumberbatch M.G., De Santis M. EAU-ESTRO-SIOG guidelines on prostate cancer. Part 1: screening, diagnosis, and local treatment with curative intent. Eur Urol. 2017;71:618–629. doi: 10.1016/j.eururo.2016.08.003. [DOI] [PubMed] [Google Scholar]
- 39.Gosselaar C., Kranse R., Roobol M.J., Roemeling S., Schröder F.H. The inter observer variability of digital rectal examination in a large randomized trial for the screening of prostate cancer. Prostate. 2008;68:985–993. doi: 10.1002/pros.20759. [DOI] [PubMed] [Google Scholar]
- 40.de Rooij M., Hamoen E.H., Witjes J.A., Barentsz J.O., Rovers M.M. Accuracy of magnetic resonance imaging for local staging of prostate cancer: adiagnostic metaanalyse. Eur Urol. 2016;70:233–245. doi: 10.1016/j.eururo.2015.07.029. [DOI] [PubMed] [Google Scholar]
- 41.Fulgham P.F., Rukstalis D.B., Turkbey I.B., Rubenstein J.N., Taneja S., Carroll P.R. AUA policy statement on the use of multiparametric magnetic resonance imaging in the diagnosis, staging and management of prostate cancer. J Urol. 2017;198:832–838. doi: 10.1016/j.juro.2017.04.101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Hövels A.M., Heesakkers R.A., Adang E.M., Jager G.J., Strum S., Hoogeveen Y.L. The diagnostic accuracy of CT and MRI in the staging of pelvic lymph nodes in patients with prostate cancer: a meta-analysis. Clin Radiol. 2008;63:387–395. doi: 10.1016/j.crad.2007.05.022. [DOI] [PubMed] [Google Scholar]
- 43.Evangelista L., Guttilla A., Zattoni F., Muzzio P.C., Zattoni F. Utility of choline positron emission tomography/computed tomography for lymph node involvement identification in intermediate- to high-risk prostate cancer: a systematic literature review and meta-analysis. Eur Urol. 2013;63:1040–1048. doi: 10.1016/j.eururo.2012.09.039. [DOI] [PubMed] [Google Scholar]
- 44.Beheshti M., Imamovic L., Broinger G., Vali R., Waldenberger P., Stoiber F. 18F Choline PET/CT in the preoperative staging of prostate cancer patients with intermediate and high risk of extra-capsular disease: a prospective study in 130 patients. Radiology. 2010;254:925–933. doi: 10.1148/radiol.09090413. [DOI] [PubMed] [Google Scholar]
- 45.Evangelista L., Briganti A., Fanti S., Joniau S., Reske S., Schiavina R. New clinical indications for18 f/11c-choline, new tracers for positron emission tomography and a promising hybrid device for prostate cancer staging: a systematic review. Eur Urol. 2016;70:161–175. doi: 10.1016/j.eururo.2016.01.029. [DOI] [PubMed] [Google Scholar]
- 46.Budäus L., Leyh-Bannurah S.R., Salomon G., Michl U., Heinzer H., Huland H. Initial experience of (68)Ga-PSMA PET/CT imaging in high-risk prostate cancer patients prior to radical prostatectomy. Eur Urol. 2016;69:393–396. doi: 10.1016/j.eururo.2015.06.010. [DOI] [PubMed] [Google Scholar]
- 47.Langsteger W., Haim S., Knauer M., Waldenberger P., Emmanuel K., Loidl W. Imaging of bone metastasis in prostate cancer: an update. Q J Nuclear Med Mol Imag. 2012;56:447–458. [PubMed] [Google Scholar]
- 48.Carroll P.R., Parsons J.K., Andriole G., Bahnson R.R., Castle E.P., Catalona W.J. NCCN guidelines insights: prostate cancer early detection, version 2.2016. J Natl Compr Canc Netw. 2016;14:509–519. doi: 10.6004/jnccn.2016.0060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Expert Panel on Urologic Imaging, Coakley F.V., Oto A., Alexander L.F., Allen B.C., Davis B.J. ACR Appropriateness criteria prostate cancer—pretreatment detection, surveillance, and staging. J Am Coll Radiol. 2017;14:S245–S257. doi: 10.1016/j.jacr.2017.02.026. [DOI] [PubMed] [Google Scholar]