Abstract
Besides the well-known hepatobiliary pathway of cholesterol excretion into the feces, transintestinal cholesterol excretion (TICE) is a second major pathway through which cholesterol is disposed from the body. In the process of TICE, cholesterol is taken up from lipoprotein particles at the basolateral side of the enterocyte and translocates towards the apical side of the enterocyte. At the apical side, the ATP-binding cassette transporters G5 and G8 form a heterodimer that transports cholesterol into the intestinal lumen. A substantial amount of the secreted cholesterol is likely reabsorbed by the cholesterol influx transporter Niemann-Pick C1-Like 1 (NPC1L1) since recent data indicate that inhibition of NPC1L1 increases the efficacy of TICE for disposal of cholesterol via the feces. The pathways and proteins involved in intracellular cholesterol trafficking in the enterocyte have not yet been identified. Therefore, in addition to discussing known mediators of TICE, this review will also examine potential candidates involved in cholesterol translocation in the enterocyte. Both the cholesterol reuptake and efflux pathways can be influenced by pharmaceutical means; thus, the TICE pathway is a very attractive target to increase cholesterol excretion from the body and prevent or mitigate atherosclerotic cardiovascular disease.
Keywords: transintestinal cholesterol excretion, TICE, NPC1L1, lipid-transfer proteins, GRAMD1, OSBP, ORP1L, STARD3, TEX2
INTRODUCTION
Cholesterol is a crucial molecule for animal life: it modulates the integrity and fluidity of cell membranes, is involved in multiple signal transduction pathways, and is the source molecule for bile acids and steroid hormones.1,2 Excessive amounts of cholesterol can result in diseases such as atherosclerosis, caused by accumulation of cholesterol in the arteries.3 To prevent this accumulation, at least two pathways exist to remove cholesterol from the body via the feces—the hepatobiliary pathway, the better-known of the two, and transintestinal cholesterol excretion (TICE). In the hepatobiliary pathway, high-density lipoprotein (HDL) mediates the transport of cholesterol from peripheral tissues to the liver, where it is secreted via bile.1 In an attempt to increase HDL-mediated cholesterol excretion, various therapies have been tested that inhibit the cholesteryl ester transfer protein. Although they did raise HDL cholesterol, these inhibitors did not increase fecal sterol excretion in humans.4 This suggests that inducing HDL concentrations alone might be insufficient to stimulate cholesterol excretion, and that stimulation of the TICE pathway may prove to be a more effective strategy. This review provides a brief overview of the classic hepatobiliary pathway and discusses how stimulation of the TICE pathway may enhance cholesterol efflux in humans.
CHOLESTEROL EFFLUX BY TICE
Classically, cholesterol is excreted out of the body in a pathway that depends on HDL particles, which transport cholesterol from peripheral tissues to the liver. This process includes the assembly of HDL, the transport of cholesterol into the HDL particle mediated primarily by the ATP-binding cassette A1 (ABCA1) transporter, and hepatic HDL cholesterol uptake by the scavenger receptor class B type I (SR-B1). In the liver, cholesterol can be converted into bile acids that are secreted into the bile along with phospholipids and cholesterol.5 Normally, about 95% of the secreted bile acids and up to 80% of intestinal cholesterol are reabsorbed into the intestinal lumen by apical sodium bile acid transporter (ASBT) and Niemann-Pick C1-Like 1 (NPC1L1), respectively.6,7 The exclusive biliary origin of fecal neutral sterol was originally challenged almost a century ago by Sperry, who suggested that there must be another, nonbiliary source of fecal cholesterol.8 In 1967, Simmonds et al. perfused humans and reported that cholesterol was not only absorbed but also secreted by the intestine.9
With the emergence of knockout mice, it became possible to study this intestinal cholesterol secretion we now call TICE in more detail. For instance, mice deficient for the phosphatidylcholine transporter ATP-binding cassette B4 (ABCB4) had an almost completely abolished biliary cholesterol flow, but their fecal cholesterol loss was hardly affected.10 Similar results were obtained with mice deficient for ATP-binding cassettes G5 and G8 (ABCG5 and ABCG8) that together constitute a sterol cotransporter heterodimer located in the apical membrane of the intestine. These mice still had more than half of the fecal sterol excretion of their wild-type littermates despite a 90% reduction in biliary cholesterol excretion.11 Similarly, Temel et al. found that mass fecal cholesterol loss was not affected in mice upon external biliary diversion.12 In all these studies, TICE could be calculated as cholesterol excreted in the feces that was not from dietary or biliary origin. Van der Velde et al. determined TICE directly with isolated murine intestines, in which they observed cholesterol secretion predominantly located in the proximal part of the small intestine.13 Altogether, these studies validated that the intestine can indeed secrete cholesterol into the intestinal lumen.
In addition to mouse studies, TICE was also examined by Jakulj et al. using a stable isotope-based method adapted to humans. In their study of 15 mildly hypercholesterolemic but healthy males, approximately 100 mg of the 900-mg total daily amount of neutral sterols excreted by the feces was derived from the diet, whereas biliary cholesterol secretion and TICE contributed about 600 mg and 200 mg, respectively.14 Thus, almost one-fourth of the fecal cholesterol excretion in humans is TICE, at least in mildly hypercholesterolemic but healthy males.
Although the molecular mechanisms of TICE remain largely elusive, it is evident that four subsequent steps are involved: (1) cholesterol uptake by the basolateral membrane of the enterocyte; (2) translocation of cholesterol from the basolateral to the apical side of the enterocyte; (3) excretion over the apical membrane into the intestinal lumen; and (4) partial reabsorption of the secreted cholesterol by NPC1L1 followed by translocation (Figure 1). The roles of the first and third step have been addressed by various studies, and cholesterol absorption by NPC1L1 has been known for many years, as reviewed before.15 However, cholesterol translocation in the enterocyte has been largely neglected. Therefore, much of this review will discuss likely mediators of this process as well as methods to modulate the steps.
Figure 1.
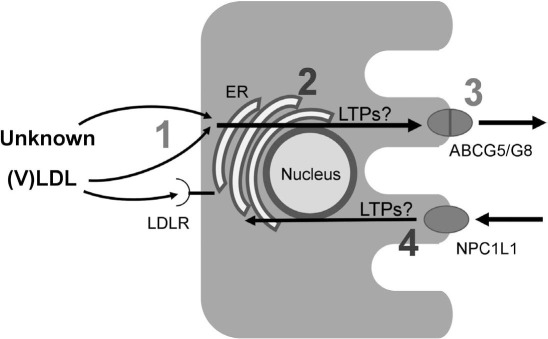
Schematic representation of the pathways in transintestinal cholesterol excretion (TICE) in the enterocyte. (1) Cholesterol uptake by the basolateral membrane of the enterocyte. Cholesterol from very low-density lipoprotein (VLDL) and/or a yet unknown source is taken up by the low-density lipoprotein receptor (LDLR) and/or another pathway. (2) Translocation of cholesterol from the basolateral to the apical side of the enterocyte. The mechanisms behind this translocation are not known; however, we hypothesize that lipid-transfer proteins (LTPs) form tethers between different membranes, such as that of the endoplasmic reticulum (ER), allowing transfer of cholesterol between these membranes. (3) Excretion over the apical membrane into the intestinal lumen, a process that is mainly mediated by the ATP-binding cassettes G5/G8 (ABCG5/G8) transporter. (4) Absorption of cholesterol by the cholesterol influx transporter NPC1L1 followed by translocation, a process presumably also involving the actions of LTPs. NPC1L1: Niemann-Pick C1-Like 1, a protein that mediates extracellular sterol transport
BASOLATERAL CHOLESTEROL UPTAKE AND APICAL CHOLESTEROL SECRETION
The specific source of cholesterol needed for TICE is not currently known. Mice deficient for both ABCA1 and SR-B1 lack HDL and have a defective hepatic HDL-cholesterol clearance.16 In these mice, TICE of radiolabeled cholesterol from HDL was similar to that of their wild-type littermates.17 These observations indicate that HDL is not essential as a cholesterol source or transport moiety for TICE. However, it cannot be excluded that HDL does exert this function under physiological circumstances and that a backup pathway with high affinity takes over HDL's functions in the absence of this class of lipoproteins.
The cholesterol delivery or transport roles of very low-density lipoproteins (VLDL) or low-density lipoprotein (LDL) particles in TICE are not completely clear. Experiments with mice deficient for the LDL receptor (LDLR) do not show an effect on TICE, but interestingly, proprotein convertase subtilisin/kexin type 9 (PCSK9) knockout mice have an increased TICE while PCSK9 injections reduced TICE.18 This indicates that either the LDLR knockout mice upregulated an alternative pathway, or PCSK9 targets another receptor. In any case, these data suggest that VLDL/LDL cholesterol is a source for TICE.
It is evident that the ABCG5/ABCG8 sterol cotransporters, which are located in the apical membrane, play an important role in cholesterol excretion by the enterocyte into the intestinal lumen and are thus involved in TICE.19 Several studies have shown that TICE is strongly decreased in mice deficient for ABCG5 or ABCG8.20–23 The residual TICE in both knockout mouse models could be due to other apical cholesterol transporters, such as ABCB1.18
PHARMACEUTICAL INTERVENTIONS THAT MODULATE BASOLATERAL OR APICAL CHOLESTEROL TRANSPORTERS
In accordance with the likely role of LDLR-mediated LDL uptake in TICE, pharmaceutical interventions (i.e., statins and PCSK9 inhibitors) that induce the LDLR protein have been shown to stimulate TICE in mice and ex vivo.18 Treatments that stimulate the expression of ABCG5/G8, such as pharmacological activation of the liver X receptor (LXR), have also been shown to stimulate TICE.10,20 Due to their lipogenic effect resulting in hepatic steatosis, the present LXR agonists have not yet been approved for use in humans.24 Intestinal LXR agonists that will not be absorbed beyond the enterocyte might prove to stimulate TICE in humans. Bile acids are ligands of the nuclear farnesoid X receptor (FXR), and they and other FXR ligands have been shown to stimulate TICE, likely by inducing the transcription of ABCG5 and ABCG8.21,25 Finally, the anticholesterolemic drug ezetimibe has also been reported to stimulate fecal cholesterol excretion.14,23,26 Ezetimibe lowers intestinal cholesterol absorption via inhibition of the cholesterol influx transporter NPC1L1 located in the apical membrane. These studies suggest that part of the cholesterol excreted by the enterocyte is reabsorbed by enterocytes further down the intestinal tract.27
CHOLESTEROL ABSORPTION IN THE TICE PATHWAY
Assuming that ezetimibe does exclusively inhibit NPC1L1, the magnitude of the drug's effect on fecal cholesterol secretion provides information about the flux of cholesterol as it recycles through the enterocyte under normal conditions. In a study of mice and humans treated with ezetimibe, the amount of cholesterol reabsorbed by the enterocytes was at least twice the amount that was finally secreted via the feces.14 Thus, the amount of cholesterol secreted by TICE is apparently regulated at two sites: the excretion of cholesterol into the intestinal lumen counterbalanced by the reabsorption of cholesterol. This dual regulation of TICE could enable a more rapid regulation of the net TICE effect. Note that the estimated rate of cholesterol recycling in the enterocyte is probably too low because the enhanced secretion at the apical side of the enterocyte must be compensated by increased uptake of cholesterol at the basolateral side, which could become rate controlling. The high capacity of the intracellular cholesterol trafficking machinery may explain why changes in the rate of TICE have not been linked to changes in gene expression.
TRANSLOCATION OF CHOLESTEROL THROUGH THE ENTEROCYTE
Little is currently known about the molecular mechanism that enables cholesterol translocation between the basolateral and apical membranes of the enterocyte. When the transport follows a receptor-mediated pathway, cholesterol will be released from late endosomes/lysosomes via the coordinated action of Niemann-Pick type C1 and C2 proteins.28 However, it is unknown how cholesterol is subsequently released from the lysosomal membrane and which proteins/vesicles are involved in the intracellular transport. Lipid-transfer proteins (LTPs) have been shown to play a role in these processes, as they have the ability to bind lipids within a hydrophobic fold to mediate their transport.29 Among the LTPs, the likely sterol transporters are GRAM domain-containing protein 1 (GRAMD1) isoforms, oxysterol-binding protein (OSBP), OSBP-related protein 1 long form (ORP1L), steroidogenic acute regulatory protein (StAR), StAR-related lipid transfer (START) domain containing 3 (STARD3), and testis-expressed protein 2 (TEX2).30 It is clear from publicly accessible databases containing the immunohistochemical staining patterns of 76 human cell types, or the microarray profiles of 79 human or 61 mouse tissues, that GRAMD1 isoforms, OSBP, ORP1L, STARD3, and TEX2 might be involved in TICE since their protein and/or mRNA is found in the small intestine of mice and humans (Table 1).31–33
Table 1.
Expression of lipid-transfer proteins in the small intestine of mice and humans. The higher the expression, the higher the value for messenger RNA expression.31,32 ND: not detected; GRAMD1: GRAM domain-containing protein 1; OSBP: oxysterol-binding protein; ORP1L: OSBP-related protein 1 long form; StAR: steroidogenic acute regulatory protein; STARD3: StAR-related lipid transfer (START) domain containing 3; TEX2: testis-expressed protein 2
| LIPID-TRANSFER PROTEIN | MOUSE | HUMAN | |
|---|---|---|---|
| MRNA | MRNA | PROTEIN | |
| GRAMD1a | 92 | 6 | Medium |
| GRAMD1b | 68/89* | 88 | Medium |
| GRAMD1c | 514 | 8 | ND |
| OSBP | 3493 | 14/146 | High |
| ORP1L | 30/94 | 36/176 | Medium |
| StAR | 5/5/11 | 5 | ND |
| STARD3 | 228 | 7 | High |
| TEX2 | 5/595 | 36 | Medium |
* More than one value is due to multiple primers for the gene.
LIPID-TRANSFER PROTEINS IMPLICATED IN TICE
GRAMD1 Proteins
Multiple functions of the endoplasmic reticulum (ER) depend on its contact with other organelles via membrane contact sites (MCSs). Within these MCSs, the distance between the organelles is less than 30 nm, allowing localization of lipids and proteins involved in organelle functioning. This localization requires protein or protein complex tethers in the MCS that bind the two membranes.34,35 GRAMD1a is part of a tether between the ER and the plasma membrane (PM).36 The GRAMD1a ER-PM contact site contains a domain that has been found to be involved in the intermembrane transfer of sterols in yeast.37,38 The mammalian GRAMD1 isoforms also transfer sterols, but it is unclear if they are involved in TICE.39 Because of its high intestinal expression in both mice and humans, the GRAMD1b isoform could be a good candidate for involvement in TICE sterol flux (Table 1). Unfortunately, no pharmaceutical compounds are known to increase GRAMD1 activity. However, any future GRAMD1 activator designed for inducing TICE should be intestinal specific since GRAMD1 proteins are ubiquitously expressed, and inducing their activity outside the intestinal tract might have deleterious effects.
OSBP and ORP1L
OSBP is a cytosolic protein that can bind cholesterol, oxysterols, and phospholipids. It is best known for its cholesterol-transporting properties between the ER and the Golgi apparatus; it localizes to the apposed membranes of these two organelles and exchanges cholesterol for phosphatidylinositol 4-phosphate (PI4P), subsequently enriching the Golgi membrane.40 To form the ER-Golgi tether, OSBP must interact with the vesicle-associated membrane protein-associated protein (VAP) anchored in the ER.41 OSBP and OSBP-related proteins (ORPs) such as ORP1L share a C-terminal oxysterols-binding domain and an N-terminal pleckstrin-homology domain important for the membrane interaction.40 Interestingly, a loss-of-function of ORP1L in humans has been associated with low HDL-mediated cholesterol efflux, while ORP1L overexpression in macrophages reduced cholesterol flux to HDL in LDLR-deficient mice.42,43 Whether and how enterocyte OSBP and/or ORP proteins are critically involved in TICE needs to be investigated. A few compounds have been reported to inhibit OSBP and ORP proteins, and it would be of interest to test whether they affect TICE.44,45
START Proteins: STARD3
STARD3 is part of a family of proteins that contains a START domain, named after the StAR-related lipid transfer. StAR, alias STARD1, regulates cholesterol transfer within mitochondria, a rate-limiting step in steroid hormones synthesis.46 Currently, 15 members of the START family have been identified in mammals.47 STARD3 is anchored at membranes of late endosomes and forms a tether with the ER-anchored VAP to enable cholesterol transport within the MCS.48 As such, STARD3 mediates cholesterol transport from the ER to late endosomes to maintain low cholesterol concentrations in the ER. Although members of the START family have not been shown to play a role in TICE, they have been implicated in cholesterol and lipoprotein metabolism. In hepatocytes, for example, phosphatidylcholine transfer protein, alias STARD2, transfers phospholipids to ABCB4 to facilitate their excretion via bile.49 Moreover, it is also suggested that cholesterol delivery to the hepatic ABCG5/ABCG8 transporter may depend on a yet unidentified START protein.47 Most of the cholesterol excreted over the apical membrane of the enterocyte is also transported by ABCG5/ABCG8, thus a role for START proteins in TICE is certainly not beyond imagination, with STARD3 as a good candidate.19–22
TEX2
A fourth type of lipid-transfer protein contains a synaptotagmin-like mitochondrial-lipid-binding (SMP) domain. A well-known member of this family is cholesteryl ester transfer protein, which facilitates transfer of cholesteryl esters and other lipid species between the different circulating lipoproteins.50 TEX2, another member of the SMP family, might be a cytosolic sterol transporter, but it is mainly recognized as a transporter of ceramides.51,52 Hardly anything is known about the function of TEX2, especially in mammals. In yeast, TEX2 is part of a tether between the membranes of the Golgi and ER and between the nuclear envelop and vacuoles.51,52 As is evident for this obscure protein, more work is required to decipher its role in mammals before determining its role in TICE.
CONCLUSION
TICE is a nonbiliary route of excreting excess cholesterol out of the body via feces. Proteins involved in cholesterol uptake and efflux from enterocytes have been identified. It is probable that LDL and/or VLDL deliver cholesterol to the basolateral membrane of the enterocyte, while ABCG5/ABCG8 in the apical membrane transfer cholesterol into the intestinal lumen. Existing pharmaceutical compounds that modulate LDL uptake or ABCG5/ABCG8 will very likely affect TICE in humans. However, the machinery involved in intracellular trafficking of cholesterol remains enigmatic. Lipid-transfer proteins such as GRAMD1 isoforms, OSBP, ORP1L, STARD3, and TEX2 might be involved in this process. More dedicated research is eagerly awaited to test whether these lipid-transfer proteins indeed mediate TICE.
KEY POINTS
Transintestinal cholesterol excretion (TICE) is a nonbiliary route to excrete excess cholesterol out of the body via feces.
The cholesterol in TICE is delivered by low-density lipoproteins and/or very low-density lipoproteins to the basolateral membrane of the enterocyte, while ATP-binding cassettes G5 and G8 (ABCG5/ABCG8) in the apical membrane transfer cholesterol into the intestinal lumen.
Stimulation of cholesterol efflux by ABCG5/ABCG8 will very likely stimulate TICE in humans.
The machinery involved in intracellular trafficking of cholesterol over the enterocyte is unknown, but various lipid transfer proteins might be involved.
Footnotes
Conflict of Interest Disclosure: The authors have completed and submitted the Methodist DeBakey Cardiovascular Journal Conflict of Interest Statement and none were reported.
REFERENCES
- 1.Dietschy JM, Turley SD, Spady DK. Role of liver in the maintenance of cholesterol and low density lipoprotein homeostasis in different animal species, including humans. J Lipid Res. 1993 Oct;34(10):1637–59. [PubMed] [Google Scholar]
- 2.Ikonen E. Cellular cholesterol trafficking and compartmentalization. Nat Rev Mol Cell Biol. 2008 Feb;9(2):125–38. doi: 10.1038/nrm2336. [DOI] [PubMed] [Google Scholar]
- 3.Tabas I, García-Cardeña G, Owens GK. Recent insights into the cellular biology of atherosclerosis. J Cell Biol. 2015 Apr 13;209(1):13–22. doi: 10.1083/jcb.201412052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Brousseau ME, Diffenderfer MR, Millar JS et al. Effects of Cholesteryl Ester Transfer Protein Inhibition on High-Density Lipoprotein Subspecies, Apolipoprotein A-I Metabolism, and Fecal Sterol Excretion. Arterioscler Thromb Vasc Biol. 2005 Feb 24;25(5):1057–64. doi: 10.1161/01.ATV.0000161928.16334.dd. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Groen AK, Oude Elferink RP. Lipid transport into bile and role in bile formation. Curr Drug Targets Immune Endocr Metabol Disord. 2005 Jun;5(2):131–5. doi: 10.2174/1568008054064887. [DOI] [PubMed] [Google Scholar]
- 6.Dawson PA. Role of the intestinal bile acid transporters in bile acid and drug disposition. Handb Exp Pharmacol. 2011;201:169–203. doi: 10.1007/978-3-642-14541-4_4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bosner MS, Lange LG, Stenson WF, Ostlund RE. Percent cholesterol absorption in normal women and men quantified with dual stable isotopic tracers and negative ion mass spectrometry. J Lipid Res. 1999 Feb;40(2):302–8. [PubMed] [Google Scholar]
- 8.Sperry WM. Lipid Excretion: IV. A Study Of The Relationship Of The Bile To The Fecal Lipids With Special Reference To Certain Problems Of Sterol Metabolism. J Biol Chem. 1927;71:351–78. [Google Scholar]
- 9.Simmonds WJ, Hofmann AF, Theodor E. Absorption of Cholesterol from a Micellar Solution: Intestinal Perfusion Studies in Man. J Clin Invest. 1967 May;46(5):874–90. doi: 10.1172/JCI105587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kruit JK, Plosch T, Havinga R et al. Increased fecal neutral sterol loss upon liver X receptor activation is independent of biliary sterol secretion in mice. Gastroenterology. 2005 Jan;128(1):147–56. doi: 10.1053/j.gastro.2004.10.006. [DOI] [PubMed] [Google Scholar]
- 11.Yu L, Hammer RE, Li-Hawkins J et al. Disruption of Abcg5 and Abcg8 in mice reveals their crucial role in biliary cholesterol secretion. Proc Natl Acad Sci U S A. 2002 Dec 10;99(25):16237–42. doi: 10.1073/pnas.252582399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Temel RE, Sawyer JK, Yu L et al. Biliary sterol secretion is not required for macrophage reverse cholesterol transport. Cell Metab. 2010 Jul 7;12(1):96–102. doi: 10.1016/j.cmet.2010.05.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.van der Velde AE, Vrins CLJ, van den Oever K et al. Direct intestinal cholesterol secretion contributes significantly to total fecal neutral sterol excretion in mice. Gastroenterology. 2007 Sep;133(3):967–75. doi: 10.1053/j.gastro.2007.06.019. [DOI] [PubMed] [Google Scholar]
- 14.Jakulj L, van Dijk TH, de Boer JF et al. Transintestinal Cholesterol Transport Is Active in Mice and Humans and Controls Ezetimibe-Induced Fecal Neutral Sterol Excretion. Cell Metab. 2016 Dec;24(6):783–94. doi: 10.1016/j.cmet.2016.10.001. [DOI] [PubMed] [Google Scholar]
- 15.de Boer JF, Kuipers F, Groen AK. Cholesterol Transport Revisited: A New Turbo Mechanism to Drive Cholesterol Excretion. Trends Endocrinol Metab. 2018 Feb;29(2):123–33. doi: 10.1016/j.tem.2017.11.006. [DOI] [PubMed] [Google Scholar]
- 16.Zhao Y, Pennings M, Vrins CLJ et al. Hypocholesterolemia, foam cell accumulation, but no atherosclerosis in mice lacking ABC-transporter A1 and scavenger receptor BI. Atherosclerosis. 2011 Oct;218(2):314–22. doi: 10.1016/j.atherosclerosis.2011.07.096. [DOI] [PubMed] [Google Scholar]
- 17.Vrins CLJ, Ottenhoff R, van den Oever K et al. Trans-intestinal cholesterol efflux is not mediated through high density lipoprotein. J Lipid Res. 2012 Oct;53(10):2017–23. doi: 10.1194/jlr.M022194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Le May C, Berger JM, Lespine A et al. Transintestinal cholesterol excretion is an active metabolic process modulated by PCSK9 and statin involving ABCB1. Arterioscler Thromb Vasc Biol. 2013 Jul;33(7):1484–93. doi: 10.1161/ATVBAHA.112.300263. [DOI] [PubMed] [Google Scholar]
- 19.Repa JJ, Berge KE, Pomajzl C, Richardson JA, Hobbs H, Mangelsdorf DJ. Regulation of ATP-binding Cassette Sterol Transporters ABCG5 and ABCG8 by the Liver X Receptors α and β. J Biol Chem. 2002 May;277(21):18793–800. doi: 10.1074/jbc.M109927200. [DOI] [PubMed] [Google Scholar]
- 20.van der Veen JN, van Dijk TH, Vrins CL et al. Activation of the liver X receptor stimulates trans-intestinal excretion of plasma cholesterol. J Biol Chem. 2009 Jul 17;284(29):19211–9. doi: 10.1074/jbc.M109.014860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.de Boer JF, Schonewille M, Boesjes M et al. Intestinal Farnesoid X Receptor Controls Transintestinal Cholesterol Excretion in Mice. Gastroenterology. 2017 Apr;152(5):1126–1138.e6. doi: 10.1053/j.gastro.2016.12.037. [DOI] [PubMed] [Google Scholar]
- 22.Brufau G, Kuipers F, Lin Y, Trautwein EA, Groen AK. A reappraisal of the mechanism by which plant sterols promote neutral sterol loss in mice. PLoS One. 2011 Jun;6(6) doi: 10.1371/journal.pone.0021576. e21576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Jakulj L, Vissers MN, van Roomen CP et al. Ezetimibe stimulates faecal neutral sterol excretion depending on abcg8 function in mice. FEBS Lett. 2010 Aug;584(16):3625–8. doi: 10.1016/j.febslet.2010.07.035. [DOI] [PubMed] [Google Scholar]
- 24.Oosterveer MH, Grefhorst A, Groen AK, Kuipers F. The liver X receptor: control of cellular lipid homeostasis and beyond. Implications for drug design. Prog Lipid Res. 2010 Oct;49(4):343–52. doi: 10.1016/j.plipres.2010.03.002. [DOI] [PubMed] [Google Scholar]
- 25.Lefebvre P, Cariou B, Lien F, Kuipers F, Staels B. Role of bile acids and bile acid receptors in metabolic regulation. Physiol Rev. 2009 Jan 1;89(1):147–91. doi: 10.1152/physrev.00010.2008. [DOI] [PubMed] [Google Scholar]
- 26.Nakano T, Inoue I, Takenaka Y et al. Ezetimibe Promotes Brush Border Membrane-to-Lumen Cholesterol Efflux in the Small Intestine. PLoS One. 2016 Mar;11(3) doi: 10.1371/journal.pone.0152207. e0152207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Clader JW. The discovery of ezetimibe: a view from outside the receptor. J Med Chem. 2004 Jan 1;47(1):1–9. doi: 10.1021/jm030283g. [DOI] [PubMed] [Google Scholar]
- 28.Infante RE, Wang ML, Radhakrishnan A, Kwon HJ, Brown MS, Goldstein JL. NPC2 facilitates bidirectional transfer of cholesterol between NPC1 and lipid bilayers, a step in cholesterol egress from lysosomes. Proc Natl Acad Sci U S A. 2008 Oct 7;105(40):15287–92. doi: 10.1073/pnas.0807328105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wong LH, Čopič A, Levine TP. Advances on the Transfer of Lipids by Lipid Transfer Proteins. Trends Biochem Sci. 2017 Jul;42(7):516–30. doi: 10.1016/j.tibs.2017.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hanada K. Lipid-transfer proteins rectify inter-organelle flux and accurately deliver lipids at membrane contact sites. J Lipid Res. 2018 Aug;59(8):1341–66. doi: 10.1194/jlr.R085324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.BioGPS [Internet] La Jolla, CA: The Scripps Research Institute; c2018. Dataset: GeneAtlas MOE430; 2018 [cited 2018 Oct 31]. Available from: www.biogps.org. [Google Scholar]
- 32.BioGPS [Internet] La Jolla, CA: The Scripps Research Institute; c2018. Dataset: GeneAtlas U133A, gcrma; 2018 [cited 2018 Oct 31]. Available from: www.biogps.org. [Google Scholar]
- 33.The Human Protein Atlas [Internet] Stockholm: The Knut & Alice Wallenberg Foundation; 2017. Dec 1, [cited 2018 Oct 31]. Available from: www.protein-atlas.org. [Google Scholar]
- 34.Prinz WA. Bridging the gap: Membrane contact sites in signaling, metabolism, and organelle dynamics. J Cell Biol. 2014 Jun 23;205(6):759–69. doi: 10.1083/jcb.201401126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Helle SCJ, Kanfer G, Kolar K, Lang A, Michel AH, Kornmann B. Organization and function of membrane contact sites. Biochim Biophys Acta - Mol Cell Res. 2013 Nov;1833(11):2526–41. doi: 10.1016/j.bbamcr.2013.01.028. [DOI] [PubMed] [Google Scholar]
- 36.Besprozvannaya M, Dickson E, Li H et al. GRAM domain proteins specialize functionally distinct ER-PM contact sites in human cells. Elife. 2018 Feb 22;7 doi: 10.7554/eLife.31019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Murley A, Sarsam RD, Toulmay A, Yamada J, Prinz WA, Nunnari J. Ltc1 is an ER-localized sterol transporter and a component of ER-mitochondria and ER-vacuole contacts. J Cell Biol. 2015 May 25;209(4):539–48. doi: 10.1083/jcb.201502033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Gatta AT, Wong LH, Sere YY et al. A new family of StART domain proteins at membrane contact sites has a role in ER-PM sterol transport. Elife. 2015 May 22;4 doi: 10.7554/eLife.07253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Horenkamp FA, Valverde DP, Nunnari J, Reinisch KM. Molecular basis for sterol transport by StART-like lipid transfer domains. EMBO J. 2018 Mar 15;37(6) doi: 10.15252/embj.201798002. e98002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Pietrangelo A, Ridgway ND. Bridging the molecular and biological functions of the oxysterol-binding protein family. Cell Mol Life Sci. 2018 Sep;75(17):3079–98. doi: 10.1007/s00018-018-2795-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Mesmin B, Bigay J, Moser von Filseck J, Lacas-Gervais S, Drin G, Antonny B. A four-step cycle driven by PI(4)P hydrolysis directs sterol/PI(4)P exchange by the ER-Golgi tether OSBP. Cell. 2013 Nov 7;155(4):830–43. doi: 10.1016/j.cell.2013.09.056. [DOI] [PubMed] [Google Scholar]
- 42.Motazacker MM, Pirhonen J, van Capelleveen JC et al. A loss-of-function variant in OSBPL1A predisposes to low plasma HDL cholesterol levels and impaired cholesterol efflux capacity. Atherosclerosis. 2016 Jun;249:140–7. doi: 10.1016/j.atherosclerosis.2016.04.005. [DOI] [PubMed] [Google Scholar]
- 43.Yan D, Jauhiainen M, Hildebrand RB et al. Expression of human OSBP-related protein 1L in macrophages enhances atherosclerotic lesion development in LDL receptor-deficient mice. Arter Thromb Vasc Biol. 2007;27(7):1618–24. doi: 10.1161/ATVBAHA.107.144121. [DOI] [PubMed] [Google Scholar]
- 44.Arita M, Kojima H, Nagano T, Okabe T, Wakita T, Shimizu H. Oxysterol-Binding Protein Family I Is the Target of Minor Enviroxime-Like Compounds. J Virol. 2013 Apr 15;87(8):4252–60. doi: 10.1128/JVI.03546-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Burgett AWG, Poulsen TB, Wangkanont K et al. Natural products reveal cancer cell dependence on oxysterol-binding proteins. Nat Chem Biol. 2011 Aug 7;7(9):639–47. doi: 10.1038/nchembio.625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kallen CB, Billheimer JT, Summers SA, Stayrook SE, Lewis M, Strauss JF. Steroidogenic acute regulatory protein (StAR) is a sterol transfer protein. J Biol Chem. 1998 Oct 9;273(41):26285–8. doi: 10.1074/jbc.273.41.26285. [DOI] [PubMed] [Google Scholar]
- 47.Soccio RE, Breslow JL. StAR-related lipid transfer (START) proteins: mediators of intracellular lipid metabolism. J Biol Chem. 2003 Jun 20;278(25):22183–6. doi: 10.1074/jbc.R300003200. [DOI] [PubMed] [Google Scholar]
- 48.Wilhelm LP, Wendling C, Védie B et al. STARD3 mediates endoplasmic reticulum-to-endosome cholesterol transport at membrane contact sites. EMBO J. 2017 May 15;36(10):1412–33. doi: 10.15252/embj.201695917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kanno K, Wu MK, Scapa EF, Roderick SL, Cohen DE. Structure and function of phosphatidylcholine transfer protein (PC-TP)/StarD2. Biochim Biophys Acta - Mol Cell Biol Lipids. 2007 Jun;1771(6):654–62. doi: 10.1016/j.bbalip.2007.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Barter PJ. Hugh sinclair lecture: the regulation and remodelling of HDL by plasma factors. Atheroscler Suppl. 2002 Dec;3(4):39–47. doi: 10.1016/s1567-5688(02)00041-7. [DOI] [PubMed] [Google Scholar]
- 51.Toulmay A, Prinz WA. A conserved membrane-binding domain targets proteins to organelle contact sites. J Cell Sci. 2012 Jan 1;125(Pt 1):49–58. doi: 10.1242/jcs.085118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Liu L-K, Choudhary V, Toulmay A, Prinz WA. An inducible ER-Golgi tether facilitates ceramide transport to alleviate lipotoxicity. J Cell Biol. 2017 Jan 2;216(1):131–47. doi: 10.1083/jcb.201606059. [DOI] [PMC free article] [PubMed] [Google Scholar]


