Abstract
Aims
Serotonergic (5‐HT) modulation of the lateral habenula (LHb) activity is central in normal and pathologic conditions such as mood disorders. Among the multiple 5‐HT receptors (5‐HTRs) involved, the 5‐HT2CR seems to play a pivotal role. Yet, the role of 5‐HT2ARs in the control of the LHb neuronal activity is completely unknown.
Methods
Single‐cell extracellular recording of the LHb neurons was used in rats to study the effect of the general activation and blockade of the 5‐HT2CR and 5‐HT2AR with Ro 60‐0175 and SB242084, TCB‐2 and MDL11939, respectively. The expression of both receptors in the LHb was confirmed using immunohistochemistry.
Results
Cumulative doses (5‐640 μg/kg, iv) of Ro 60‐0175 and TCB‐2 affected the activity of 34% and 63% of the LHb recorded neurons, respectively. LHb neurons were either inhibited at low doses or excited at higher doses of the 5‐HT2A/CR agonists. SB242084 or MDL11939 (both at 200 μg/kg, iv) did not modify neuronal firing when injected alone, but reverted the bidirectional effects of Ro 60‐0175 or TCB‐2, respectively. 5‐HT2CRs and 5‐HT2ARs are expressed in less than the 20% of the LHb neurons, and they neither colocalize nor make heterodimers. Strikingly, only 5‐HT2ARs are expressed by the majority of LHb astrocyte cells.
Conclusions
Peripheral administration of 5‐HT2AR agonist promotes a heterogeneous pattern of neuronal responses in the LHb, and these effects are more prominent than those induced by the 5‐HT2CR activation.
Keywords: addiction, depression, immunohistochemistry, lateral habenula, serotonin
1. INTRODUCTION
The connection between the lateral habenula (LHb) and the serotonergic system has been studied for several years.1, 2 The reciprocal interaction is believed to play an important role in mood disorders1, 2 while serotonin‐2A receptor (5‐HT2AR) and 5‐HT2CR subtypes are thought to play a role in the mechanism of action of antidepressant drugs and drug of abuse.3, 4, 5 Nonetheless, the control exerted by these receptors on the activity of LHb neurons has been poorly investigated.
The LHb receives dense serotonergic innervation from the median and dorsal raphe nuclei (DRN)1 and expresses several 5‐HTR subtypes.1, 2 Early evidence showed that 5‐HT2CR mRNA and protein were densely expressed in the LHb, while 5‐HT2AR mRNA was virtually absent.6, 7 Therefore, successive work has focused on the expression of 5‐HT2CR within the LHb and shown that the distribution of the 5‐HT2CR mRNA exhibits heterogeneous distribution in correlation to the heterogeneous cytoarchitecture of the LHb.8 In agreement with this evidence, a recent microarray study confirmed a preferential enriched HT2C gene expression in the LHb compared to the MHb and also showed an unexpected high expression of HT2A in the LHb.9 Nevertheless, the anatomical distribution of the 5‐HT2AR protein remains obscure. It is therefore likely that the LHb 5‐HT2A/2CRs might play an important role in modulating the activity of this epithalamic nucleus. In addition, 5‐HT2A/2CR modulation of the LHb might be indirect via other brain regions innervating the LHb that are rich in 5‐HT2C and 5‐HT2ARs, such as raphe nuclei, ventral tegmental area (VTA) and medial prefrontal cortex (mPFC).1 LHb 5‐HT2A/2CRs may also be important in the postsynaptic 5‐HT long‐feedback modulating the activity of serotonergic neurons.10
Limited recent data indicate that 5‐HT modulates the activity of the LHb neurons. It is noteworthy that the high‐frequency electrical stimulation of the subthalamic nucleus, presumably via actions on serotonergic neurons, induced heterogeneous responses composed by excitation, inhibition or no effect on the LHb neurons.11 Conversely, mainly excitatory modulations have been described by electrophysiological in vivo and in vitro studies using 5‐HT and different 5‐HT2R ligands. It has been reported in vitro, on slices containing the LHb, that the application ofexogenous 5‐HT depolarizes LHb neurons postsynaptically, an effect mimicked by the non‐selective 5‐HT2CR agonist m‐CPP.12 The pharmacology employed would suggest a 5‐HT2CR‐mediated mechanism. Consistently, the preferential 5‐HT2B/2CR agonist Ro 60‐0175 enhanced the firing rate of several neurons and promoted burst mode of discharge upon its local injection into the LHb in vivo.13 To the best of our knowledge, no studies have specifically looked at the resulting effect of systemic injection of 5‐HT2R agonists on the LHb neuron activity, despite its clear translational significance.
In this study, to fill this gap, we studied the effect of the intravenous (iv) administration of the 5‐HT2CR agonist Ro 60‐017514 and the 5‐HT2AR agonist TCB‐215 on the activity of LHb neurons using extracellular single‐cell recordings in vivo. The use of SB24208416 and MDL11939,17 two selective antagonists at 5‐HT2CR and 5‐HT2AR, respectively, furthered the role of 5‐HT2CR and 5‐HT2AR in the LHb neuronal responses. The electrophysiological approach was extended with an immunohistochemical study and a proximity ligation assay to determine the distribution of 5‐HT2A/2CRs and the presence of 5‐HT2AR protein and eventual heterodimers within the LHb, respectively.
2. METHODS
2.1. Animals
Male Sprague‐Dawley rats, obtained from Charles River Laboratories in Margate, UK, and maintained at the Department of Physiology and Biochemistry at the University of Malta, were housed at 21 ± 1°C, with 60 ± 5% humidity, and a 12‐hour light/dark cycle (lights on at 7 am and off at 7 pm). Food and water were provided ad libitum. Adult rats that weighed 270 g‐320 g on the day of surgery or brain extraction were used. All procedures were carried out in accordance with institutional guidelines (Institutional Animal Use and Care Committee (IAUCC) of the University of Malta), the ARRIVE guidelines and the EU Directive 2010/63/EU for animal experiments. Utmost care was taken to limit the number of rats used and their suffering.
2.2. Extracellular single‐unit recordings
Standard extracellular recording in vivo was performed. After the first injection of chloral hydrate (400 mg/kg, ip, Sigma‐Aldrich, UK), rats, placed on a homoeothermic blanket (37 ± 0.5°C) received an iv infusion of chloral hydrate (8% w/v; 8 mL/h). Borosilicate glass micropipettes (4‐7 MΩ resistance), filled with 2% pontamine sky blue solution in 0.5 mol/L sodium acetate, were positioned in the LHb (3.4‐3.8 mm AP from bregma; 1.4‐1.8 mm ML from midline; 4‐5 mm DV from surface of cortex; electrode set at a 10°angle18 using a one‐axis hydraulic micromanipulator (MO‐10, Narishige, Japan). Signal acquisition was performed with a micro1401 CED laboratory interface connected to Spike2 v7.4 (Cambridge Electronic Design, Cambridge, UK) and a Neurolog amplifier and filtering system (Digitimer Ltd. UK; 10k amplification, band‐pass filter set at 0.5‐5 kHz). A postmortem histological analysis was performed to locate the recorded area (Figure 2, inset)19, 20 (see Data S1 for signal analysis).
Figure 2.

Effect of the systemic administration of the 5‐HT2C R agonist Ro 60‐0175 and 5‐HT2CR antagonist SB242084 on LHb neuronal firing. (A) Dose–response curve of Ro 60‐0175 (5‐640 μg/kg, iv) showing the mean % change in firing rate ± SEM. About 10% of recorded neurons responded with an increase in firing rate, 24% with a decrease, while 66% showed no overall change in their firing frequency (not shown). One‐way ANOVA for repeated‐measures followed by Tukey's post hoc test, *P < 0.05 vs Vehicle. (B) Representative rate histograms obtained from single neurons showing 2 of the 3 different neuronal responses observed: excitation (top), inhibition (bottom), and a control neuron (middle). (C) Dose–response curve of SB242084 (5‐640 μg/kg, iv) showing the mean % change in firing rate ± SEM. Neurons showed no overall change compared to their basal firing activity (shown in green). A representative rate histogram of a recorded neuron is shown in the inset. (D) The involvement of the 5‐HT2CRs in mediating Ro 60‐0175‐induced changes in LHb neuronal activity was confirmed through a single administration of SB242084 (200 μg/kg, iv) 2 min after the last dose of Ro 60‐0175. SB242084, a selective 5‐HT2CR antagonist, was capable of reversing Ro 60‐0175‐induced changes in firing rate. iv: intravenous; predrug: the 2‐min period before the administration of the first dose of Ro 60‐0175; Ro 60‐0175: the 2‐min period following the administration of the last dose of Ro 60‐0175; SB242084: the 2‐min period following SB242084 administration. (E) Representative rate histograms showing SB242084‐induced reversal of both excitation (top) and inhibition (bottom) responses in firing rate induced by Ro 60‐0175. Paired t‐test, § P < 0.05 predrug vs Ro 60‐0175, P < 0.05 Ro 60‐0175 vs SB242084. The inset shows a microphotograph of a rat brain coronal section containing the neuron recorded on the right and the corresponding section taken from the atlas of Paxinos and Watson (2017). FR: fasciculus retroflexus; LHbL: lateral portion of the lateral habenula; LHbM: medial portion of the lateral habenula; MHb: medial habenula; SM: stria medularis
2.3. Drugs and pharmacological treatments
TCB‐2, Ro 60‐0175, MDL11939, and SB242084 were purchased from Tocris Biosciences, UK and dissolved according to our previous study20 (see Data S1). The doses have been chosen on the basis of previous experiments reporting their efficacy and selectivity.20, 21, 22, 23 All laboratory reagents were purchased from Sigma‐Aldrich, UK.
Once a stable neuron was detected using the procedure described previously, the rat was given, through the lateral tail vein, cumulative doses (640 μg/kg, iv) of 1 drug or its own vehicle. A total of 8 doses (5, 5, 10, 20, 40, 80, 160, and 320 μg/kg), each dissolved in 100 μL of vehicle, were given at 2‐minute intervals. Combined injections of antagonists and agonists and appropriate vehicle were performed. Post‐treatment of SB242084 (200 μg/kg, iv) was given 2 min after Ro 60‐0175 (5‐640 μg/kg). A post‐treatment was used instead of a pretreatment as less than 50% of neurons were responsive to Ro 60‐0175 cumulative treatment. Pretreatment with MDL11939 (200 μg/kg, iv) was given 5 min before TCB‐2 (5‐640 μg/kg).
2.4. Statistical analysis
Individual responses of neurons to Ro 60‐0175, TCB‐2, SB242084, MDL11939 (all at cumulative doses of 5‐640 μg/kg) and their respective vehicles were expressed in % change in basal firing rate. The data report the mean ± SEM for each group, if any. One‐way ANOVA with repeated‐measures was performed separately for each treatment group compared to their respective controls. It was followed by Tukey's post hoc test (if required).
The interaction of Ro 60‐0175 (5‐640 μg/kg) with SB242084 (200 μg/kg) was analyzed using a paired t‐test which compared the firing rate at 3 different periods: (i) predrug (the 2‐min period before Ro 60‐0175 administration); (ii) max‐dose (the 2 min following Ro 60‐0175 administration); and (iii) postblock (the 2‐min period following SB242084 administration). This was performed separately for the different neuronal response groups.
In the case of the interaction of the factors for TCB‐2 (5‐640 μg/kg) and MDL11939 (200 μg/kg) pretreatment, a one‐way ANOVA with repeated‐measures was performed including the TCB‐2 excitation group, and the TCB‐2 inhibition group. A post‐Tukey test was performed for each time point. Significant difference was considered when P < 0.05.
2.5. Histological procedures and Immunocytochemistry
Eight rats were used for the immunocytochemistry. Procedures of anesthesia, fixation, and slicing have been already reported19 (see Data S1 for Immunoperoxidase experiments, Double immunofluorescence experiments, Specificity of antibodies, Thionin staining, and Analysis of sections details).
The following antibodies were used: (i) Rabbit anti‐5‐HT2AR polyclonal antibody (diluted 1:300; code 24288; ImmunoStar, WI, USA; (ii) mouse anti‐5‐HT2CR monoclonal antibody (diluted 1:100; code sc‐17797; Santa Cruz, CA, USA). The following secondary antibodies were used for immunoperoxidase experiments: (i) goat biotinylated anti‐rabbit (diluted 1:200, Vector, Burlingame, CA, BA‐1000); (ii) goat biotinylated anti‐mouse (diluted 1:200, Vector, Burlingame, CA, BA‐9200).
The following primary antibodies were used for colocalization studies: (i) rabbit anti‐5‐HT2AR polyclonal antibody (diluted 1:300; code 24288; ImmunoStar, WI, USA) together with mouse anti‐HuC/Dmonoclonal antibody (diluted 1:200; code A21271; Molecular Probes, Leiden, the Netherlands); (ii) mouse anti‐5‐HT2CR monoclonal antibody (diluted 1:100; code sc‐17797; Santa Cruz, CA, USA) together with rabbit anti‐Protein Gene Product 9.5 (PGP 9.5) polyclonal antibody (diluted 1:100, AB1761, Millipore, Temecula, CA, USA); (iii) rabbit anti‐5‐HT2AR polyclonal antibody (diluted 1:300; code 24288; ImmunoStar, WI, USA) together with chicken anti‐GFAP (diluted 1:1000; code Ab4674; Abcam, UK); (iv) mouse anti‐5‐HT2CR monoclonal antibody (diluted 1:100; code sc‐17797; Santa Cruz, CA, USA) together with rabbit anti‐GFAP (diluted 1:500; code 20334; DAKO, Denmark). The following secondary antibodies were used for double immunofluorescence experiments: Alexa 488‐conjugated goat anti‐mouse IgG (1:400, #A11029, Molecular Probes, Leiden, the Netherlands) and Alexa 594‐conjugated goat anti‐rabbit IgG (1:400, #A11012, Molecular Probes, Leiden, the Netherlands); Alexa 488‐conjugated goat anti‐mouse IgG (1:400, #A11029, Molecular Probes, Leiden, the Netherlands); and TRITC‐conjugated donkey anti‐chicken (1:200; code 703‐025‐155, Jackson, PA, USA).
2.6. Proximity ligation assay experiments
Proximity ligation assay (PLA) experiments were carried out using Duolink kits supplied by Sigma‐Aldrich. The following antibodies were used: (i) primaries goat anti‐5‐HT2CR (diluted 1:1000; code ab32887; Abcam, Cambridge, UK) and (ii) rabbit anti‐5‐HT2AR (diluted 1:1000; code ab66049; Abcam, Cambridge, UK).
Briefly, 4% paraformaldehyde fixed coronal brain sections containing the LHb were exposed to antigen retrieval with citrate buffer, pH 6, and microwave heating for a total of 10 min. After an overnight exposure to primary antibodies chicken anti‐MAP2 (diluted 1:2000; code AB15452; Millipore, Massachusetts, USA) and mouse anti‐GAD67 (diluted 1:500, code ab26116; Abcam, Cambridge, UK), as neuronal markers, the immunofluorescence assay was completed with fluorescently labeled secondary antibodies MAP2 and GAD67. The samples were successively incubated overnight with 5‐HT2CR and 5‐HT2AR primary antibodies for PLA‐based detection of 5‐HT2C:5‐HT2AR interaction. PLA was performed as previously described24 according to the manufacturer's instructions.
3. RESULTS
3.1. Electrophysiological characteristics of spontaneously active LHb neurons
We recorded 210 spontaneously active neurons in the LHb extracellularly. In accordance with previous evidence,13, 25 the recorded neurons fired at 11.9 ± 0.67 Hz with a waveform duration of 1.1 ± 0.01 ms. The LHb neurons recorded had a predominantly (82.9%) biphasic waveform and 3 distinct firing patterns; the majority were irregular (82.9%), while regular and bursty neurons contributed to less than 10% of total (Figure 1). The average coefficient of variation (CV) was 0.74 ± 0.03.
Figure 1.
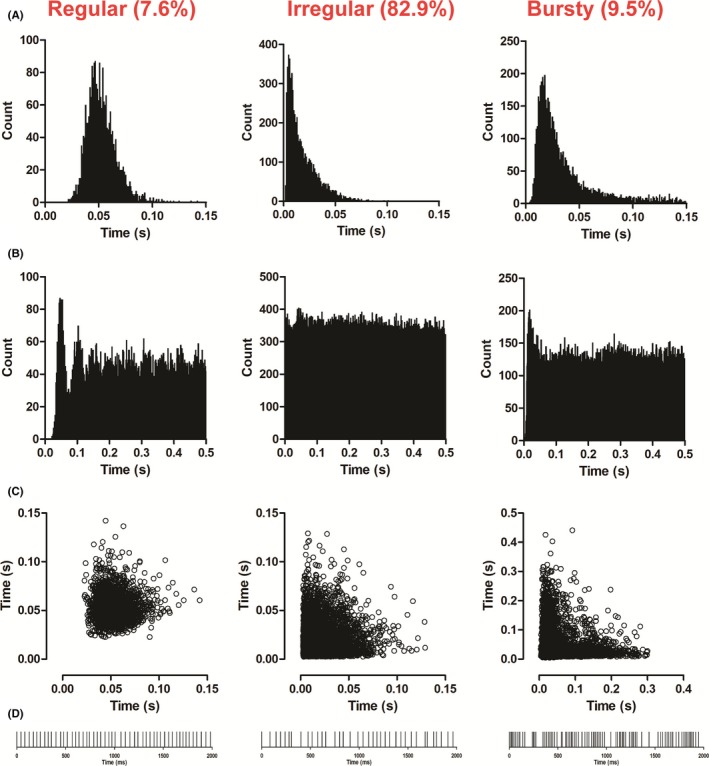
Firing pattern of recorded LHb neurons. The firing pattern of LHb neurons was classified as either regular, irregular or bursty, according to their interspike interval histograms (A), autocorrelorgrams (B) and scattergrams (C). Regular firing neurons were characterized by a narrow bell‐shaped distribution of the interspike intervals histogram, multiple initial peaks in the autocorrelogram, and a well‐defined cluster in the scattergrams. Neurons displaying an irregular firing pattern typically showed a skewed Poisson‐like distribution in their interspike interval histograms, a flat distribution with no peaks in the autocorrelograms and a dispersed cluster in the scattergrams. Finally, bursty firing neurons showed interspike interval histograms with a bimodal or very skewed distribution with a long tail, autocorrelograms with an initial narrow peak and a scattergram with a “L”‐shaped distribution along the 2 main axis. The raster plots (D) show the differences between the 3 patterns of firing, in which the interspike intervals were more constant in regular firing neurons, more erratic in irregular firing neurons, and clumped with periods of silence in bursty firing neurons. In the figure, interspike interval histograms, autocorrelograms, and scattergrams of representative neurons of each firing patterns are shown
3.1.1. Effect of systemic administration of 5‐HT2CR agonist Ro 60‐0175 and 5‐HT2CR antagonist SB242084 on the firing rate and pattern of LHb neurons
The intravenous administration of Ro 60‐0175 (5‐640 μg/kg, iv) induced a mixed response in the LHb neuronal firing (Figure 2A). The majority of the neurons (66%; 33 of 50) were not affected by Ro 60‐0175 administration. The most represented effect (24%; n = 12) was a dose‐dependent decrease in firing rate, which took effect from 160 μg/kg (1‐way ANOVA, F (3,56) = 20.922, P < 0.01) and reached the maximum inhibition (50% below baseline) at the highest dose (Figure 2 A, B). The dose‐dependent increase in firing rate was observed only in 10% (n = 5) of recorded neurons, starting at 40 μg/kg Ro 60‐0175 (one‐way ANOVA, F (3,56) = 7.855, P < 0.01) and reaching a maximal increase (45% over baseline) at 320 μg/kg, iv. (Figure 2 A, B). Qualitatively, Ro 60‐0175 was unable to alter the pattern of firing, as the CV remained unchanged in all the neurons recorded (n = 50) (paired t‐test, t (56) = −2.181, P > 0.05).
Due to the low proportion of neurons responding to Ro 60‐0175 (34%) with a change of their neuronal discharge, the 5‐HT2CR antagonist SB242084 was administered at the end of the cumulative injection of Ro 60‐0175 as performed by Queree et al26 only to some responsive neurons. As shown in Figure 2 D, both Ro 60‐0175‐induced excitation (n = 4; paired t‐test: max dose vs postblock, t(3) = 3.650, P = 0.035); and inhibition (n = 6; paired t‐test: max dose vs postblock, t(5) = −4119, P = 0.009) were reversed by SB242084 (200 μg/kg, iv). Administration of SB242084 (5‐640 μg/kg, iv) on its own modified neither basal firing rate of LHb neurons (one‐way ANOVA, F (1,14) = 2.055, P > 0.05) nor the CV (paired sample t‐test, t (8) = −1.707, P > 0.05) (Figure 2C). Histological reconstruction of electrode placement revealed that Ro 60‐0175 effects on LHb neuronal discharges were not limited to one LHb subregion but were randomly distributed and intermixed throughout the nucleus (not shown).
3.1.2. Effect of systemic administration of 5‐HT2AR agonist TCB‐2 and 5‐HT2AR antagonist MDL11939 on the firing rate and pattern of LHb neurons
Similar to the activation of 5‐HT2CRs, the administration of TCB‐2 induced a mixed neuronal response in terms of LHb neuronal firing rate. In 37% of recorded neurons (11 of 30), TCB‐2 (5‐640 μg/kg, iv) did not induce any changes in the firing rate. Conversely, administration of TCB‐2 induced a significant dose‐dependent decrease in firing rate in 37% (n = 11) of recorded neurons from the dose of 20 μg/kg (one‐way ANOVA, F (3,36) = 19.949, P < 0.01) reaching the maximum inhibition (60% below baseline) at the highest dose (Figure 3A, B). On the other hand, TCB‐2 induced a significant dose‐dependent increase in firing rate in 26% (n = 8) of the neurons from the dose of 10 μg/kg (one‐way ANOVA, F (3,36) = 5.011, P < 0.01). A sharp increase was observed from 10 to 40 μg/kg TCB‐2 followed by a more progressive increase reaching a maximum of 125 ± 25.6% of baseline at the highest dose (Figure 3A, B). TCB‐2 was unable to alter the pattern of firing in all the neurons recorded (n = 30), as the CV remained unchanged at the end of TCB‐2, 640 μg/kg, iv (paired sample t‐test, t (29) = −1.108, P > 0.05). Moreover, no correlation was found between the localization of the recorded neurons within the LHb and their response to TCB‐2 treatment (not shown).
Figure 3.
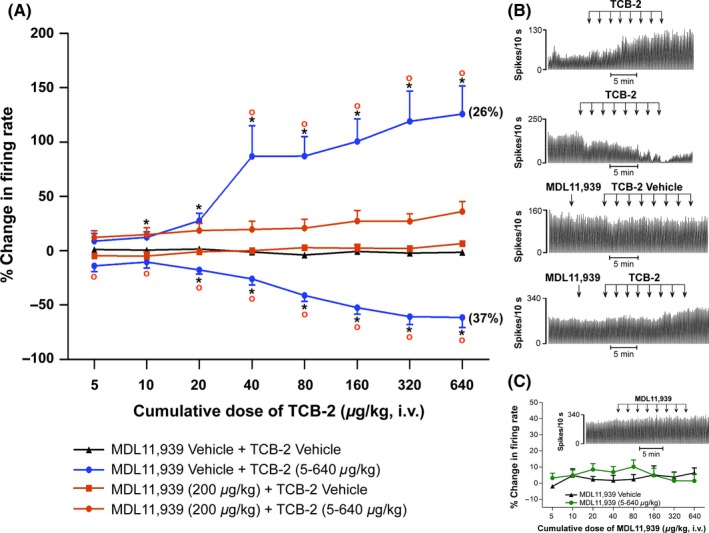
Effect of the systemic administration of the 5‐HT2AR agonist TCB‐2 and 5‐HT2AR antagonist MDL11939 on LHb neuronal firing. (A) Dose–response curves of TCB‐2 (5‐640 μg/kg, iv) showing the mean % change in firing rate ± SEM (in blue). About 26% of recorded neurons increased, 37% decreased and 37% did not change (not shown) their baseline firing rates in response to TCB‐2 administration. The role of the 5‐HT2AR in mediating TCB‐2 induced changes in LHb neuronal activity was confirmed through the pretreatment with MDL11939 (200 μg/kg, iv, in orange). MDL11939 pretreatment blocked TCB‐2 induced changes in firing rate, which accounted for 63% of neurons when TCB‐2 was administered on its own. One‐way ANOVA for repeated measures followed by Tukey's post hoc test, * P < 0.05 vs Vehicle, ° P < 0.05 vs MDL11939+ TCB‐2. (B) Representative rate histograms obtained from single LHb neurons, showing TCB‐2 induced excitation/inhibition (top 2 traces, respectively) of neuronal activity, the lack of effect of MDL11939 pretreatment (third trace) and the effect of MDL11939 pretreatment on TCB‐2 administration (fourth trace). (C) Dose–response curve of MDL11939 (5‐640 μg/kg, iv) showing the mean % change in firing rate ± SEM. The neurons showed no overall significant change in firing rate (in green) when compared to its vehicle (in black). The inset shows a rate histogram illustrating the neuronal response of a single LHb cell to MDL11939 (5‐640 μg/kg, iv) administration
With a pre‐treatment of MDL11939 (200 μg/kg, iv), TCB‐2 (5‐640 μg/kg) no longer induced inhibition or excitation of firing rate in all the neurons tested, even at the highest cumulative dose (n = 16; one‐way ANOVA, vehicle vs TCB‐2 excitation vs TCB‐2 inhibition vs MDL11939+ vehicle vs MDL11939+ TCB‐2, followed by Tukey post‐hoc test, MDL11939+ TCB‐2 vs MDL11939+ vehicle at 640 μg/kg, P > 0.05) (Figure 3A).
Similar to the 5‐HT2CR antagonist, we found that MDL11939 did not alter basal firing rate (one‐way ANOVA, F (1,14) = 0.785, P > 0.05), CV and firing pattern (paired sample t‐test, t (8) = 0.878, P > 0.05) of LHb neurons over a full cumulative dose response (5‐640 μg/kg, iv) (Figure 3C).
3.2. Distribution of the 5‐HT2AR‐ and 5‐HT2CR‐immunoreactivity in the LHb
In the LHb, the 5‐HT2AR‐IR was found to be associated with somatodendritic profiles and neuropil (Figure 4A, B). The immunolabeling for the 5‐HT2CR was found in somata and neuropil (Figure 4C, D) similarly to other structures.19 Immunoreactive neurons for both receptors had spherical, fusiform, or polygonal cell bodies of different sizes. Interestingly, 5‐HT2AR‐IR neurons appeared larger than those immunopositive for the 5‐HT2CR. The LHb had a low/medium density of 5‐HT2AR‐ and 5‐HT2CR‐immunopositive neurons. The density of immunostained somata and the average percentage of the image covered by immunostaining are reported in Figure 4E. The intensity of neuropil staining was similar for both receptors (Figure 4A‐D,F). In the medial habenula, the intensity of the 5‐HT2AR immunoreactivity decreased markedly, especially at neuropilar level (Figure 4A). On the contrary, the immunostaining obtained for the 5‐HT2CR appeared similar when comparing lateral with medial habenula (Figure 4C).
Figure 4.

Brightfield photomicrographs of 5‐HT2AR‐ and 5‐HT2CR‐ immunoreactivity in the rat LHb. Brightfield photomicrographs of coronal sections (A‐D) and histogram (E, F) showing the distribution of 5‐HT2ARs (A, B) and 5‐HT2CRs (C, D) immunoreactivity in the rat LHb. The density of immunostained neurons, as well as the intensity of neuropilar immunoreactivity, was similar compared to the immunostaining obtained for these 2 receptors. Scale bar = 200 μm in C (applies to A and C); 20 μm in D (applies to B and D)
The morphology as well as the distribution of 5‐HT2AR‐ and 5‐HT2CR‐IR neurons observed in immunofluorescence experiments were identical to those observed employing immunoperoxidase. The double immunofluorescence analysis consisted of the colocalization of the 5‐HT2AR with HuC/D (panneuronal marker) (Figure 5A1‐A3), the 5‐HT2CR with PGP 9.5 (panneuronal marker) (Figure 5B1‐B3), the 5‐HT2AR with 5‐HT2CR (Figure 5C1‐C3), the 5‐HT2AR with GFAP (astrocyte cell marker) (Figure 5D1‐D3), and 5‐HT2CR with GFAP (Figure 5E1‐E3). As panneuronal markers stained every neuron, we estimated the percentage of 5‐HT2AR‐IR and 5‐HT2CR‐IR somata distributed in the LHb. As reported in Table 1, the proportion of 5‐HT2AR‐IR and 5‐HT2CR‐IR neurons to the total neurons was low. Interestingly, in the LHb only the 5‐HT2AR‐IR was also found to be associated with astrocyte cells (Figure 5D1‐D3). 5‐HT2AR and 5‐HT2CR do not colocalize as the percentage of neurons that expressed both receptors was very low (1.5%; Table 1).
Figure 5.

Photomicrographs of the colocalization of 5‐HT2AR/HuC/D, 5‐HT2CR/PGP 9.5, and 5‐HT2AR/5‐HT2CR in the rat lateral habenula. Photomicrographs of coronal sections showing the colocalization of the 5‐HT2AR with HuC/D (A1‐A3), the 5‐HT2CR with PGP 9.5 (B1‐B3), and the 5‐HT2AR with the 5‐HT2CR (C1‐C3) in the rat LHb. Arrowheads indicate double‐immunolabeled neurons. The percentage of neurons that expressed 5‐HT2ARs (A1‐A3) and 5‐HT2CRs (B1‐B3) was very low. In addition, few neurons expressed both receptors (C1‐C3). Arrows in C1‐C3 indicate single‐labeled neurons. (D‐E) Photomicrographs of the colocalization of 5‐HT2AR/GFAP and 5‐HT2CR/GFAP in the rat LHb. Photomicrographs of coronal sections showing the colocalization of the 5‐HT2AR with GFAP (D1‐D3) and the 5‐HT2CR with GFAP (E1‐E3) in the rat LHb. Colocalization is indicated by yellow in merged. Note the strong 5‐HT2AR immunoreactivity in astrocytes (D1‐D3; see also boxed inset). Virtually, none of astrocyte cells expressed the 5‐HT2CR (E1‐E3; see also boxed inset). Arrows in D1 and D3 indicate a 5‐HT2AR‐immunoreactive neurons. Arrows in E1 and E3 indicate a 5‐HT2CR‐immunoreactive neurons. 5‐HT2AR: serotonin‐2A receptor; 5‐HT2CR: serotonin‐2C receptor. Scale bar = 50 μm in E3 (applies to A1‐E3)
Table 1.
Colocalization of 5‐HT2ARs and 5‐HT2CRs in the rat LHb
| HuC/D‐IR neurons single‐labeled neurons | HuC/D/5‐HT2AR‐IR double‐labeled neurons | % of 5‐HT2AR‐IR double‐labeled neurons | ||
|---|---|---|---|---|
| 600 | 64 | 9.6% (64/664) |
| PGP 9.5‐IR neurons single‐labeled neurons | PGP 9.5/5‐HT2CR‐IR double‐labeled neurons | % of 5‐HT2CR ‐IR double labeled neurons | ||
|---|---|---|---|---|
| 624 | 64 | 9.3% (64/688) |
| 5‐HT2AR‐IR neurons single‐labeled neurons | 5‐HT2CR‐IR neurons single‐labeled neurons | 5‐HT2AR/5‐HT2CR‐IR double‐labeled neurons | % of 5‐HT2AR ‐IR double‐labeled neurons | % of 5‐HT2CR ‐IR double‐labeled neurons |
|---|---|---|---|---|
| 64 | 64 | 1 | 1.5% (1/65) | 1.5% (1/65) |
The table shows the colocalization of (i) HuC/D with 5‐HT2AR, (ii) PGP 9.5 with 5‐HT2CR, and (iii) 5‐HT2AR with 5‐HT2CR in the rat LHb. HuC/D: pan‐neuronal marker; IR: immunoreactive; PGP 9.5: Protein Gene Product 9.5, highly specific to neurons.
3.3. Proximity ligation assay of 5‐HT2A and 5‐HT2CRs in the LHb
The lack of localization was confirmed by a proximity ligation assay which showed no 5‐HT2CR:5‐HT2AR interaction in the LHb (Figure 6). We also performed PLA for NMDA subunits NR1:NR2A as positive control (Figure 6B) to assess the accuracy of the method. As showed, PLA for NR1:NR2A provided a strong signal of interaction between the 2 NMDA receptor subunits that, in CNS, are widely represented in the same NMDARs tetramers. The result obtained therefore provides evidence of validation of the method.27, 28
Figure 6.
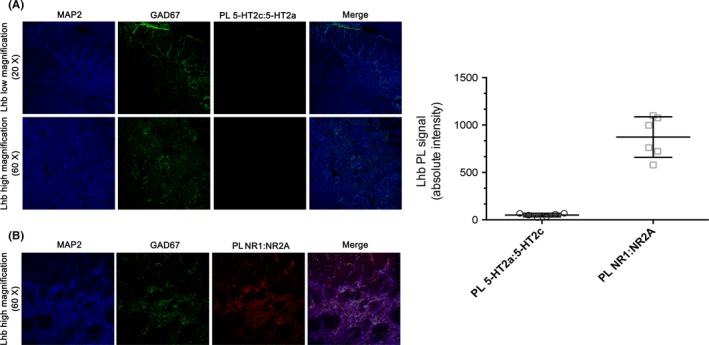
Assessing Proximity ligation assay (PLA)‐based 5‐HT2AR:5‐HT2CR interaction. (A) PL for 5‐HT2A:5‐HT2CR interaction did not reveal signal in the LHb, suggesting that the 2 serotonin receptor isoforms are not represented in the same complex. (B) Positive PL control reporting signal of interaction between the 2 NMDA receptor subunits NR1:NR2A. (C) Quantification of absolute PL signal intensity for 5‐HT2A:5‐HT2C and NR1:NR2A. Each dot represents the average of the analysis performed in a single animal (n = 6 for each experimental group). 5‐HT2A: serotonin‐2A; 5‐HT2C: serotonin‐2C; PL: proximity ligation; NMDA: N‐methyl‐d‐aspartate
4. DISCUSSION
We are the first to report that the peripheral administration of (i) 5‐HT2AR agonist TCB‐2 induces stronger bidirectional effects on the firing rate of LHb when compared to (ii) 5‐HT2CR agonist Ro 60‐0175. Our immunohistochemical study reveals that (iii) 5‐HT2AR and 5‐HT2CR proteins are similarly expressed in the LHb, although by distinct LHb cell populations. Consistently (iv) we did not reveal any 5‐HT2A‐5‐HT2CR heterodimers in this nucleus. Moreover, (v) only 5‐HT2ARs are strongly expressed by astrocytes in the LHb. Our data revealed a more complex control than that shown so far due to the clear heterogeneous responses associated with 5‐HT2AR and 5‐HT2CR stimulation. Therefore, the present findings present a strong case for a reappraisal of the influence of 5‐HT2AR and 5‐HT2CR in the control of the LHb neuronal activity.
An increase in neuronal firing rate and burst firing has been previously reported upon local application of Ro 60‐0175 into the LHb in vivo.13 This excitation would result from a simultaneous post‐ and presynaptic effect in the LHb. Indeed, in vitro application of 5‐HT induced postsynaptic depolarization and an increase in the firing rate of the majority of the LHb neurons,12 blocked by ritanserin and SB200646,29 antagonists of 5‐HT2A/CRs and 5‐HT2B/CRs, respectively, and mimicked by the nonselective 5‐HT2CR agonist m‐CPP. Moreover, the application of the nonselective 5‐HT2R agonist α‐methyl‐5‐HT enhanced presynaptic depolarization of the LHb neurons at low concentrations, facilitating glutamate release.30 In our in vivo anesthetized conditions instead, the iv administration of Ro 60‐0175 did not induce an appreciable change in the neuronal discharge in the majority of the neurons recorded, while only 10% of them were excited and 24% inhibited. Possible explanations for the differences between our results and those shown by Han et al13 are (i) principally the different routes of administration (general vs local administration), therefore (ii) the involvement of other brain areas targeting the LHb and expressing the 5‐HT2CRs, and (iii) the selection of the neurons recorded (they focused on those expressing the excitatory amino acid transporter‐1 positive neurons,13 while we did not make any such distinction in our recordings). Nevertheless, the study by Han et al13 must be interpreted with caution because, for example, the effects of local administration of Ro 60‐0175 were apparent only 10 min following its local injection and no vehicle control group was shown.
In further contrast to its local applications in vivo, 13 Ro 60‐0175 had no effect on the majority of the LHb neurons recorded. The remaining neurons were principally inhibited at slightly higher doses and excited at lower doses instead. The excitatory and the inhibitory effects induced by Ro 60‐0175 seem to be 5‐HT2CR‐dependent as they were both reversed by the selective antagonist SB 242084. Indeed, pharmacological responses of Ro 60‐0175 that are not blocked by 5‐HT2CR antagonists have been described at relatively higher regimens.31, 32 From our antagonism experiments, we can rule out the involvement of other receptors in the inhibition induced by Ro 60‐0175, that is, the 5‐HT1AR that was capable of hyperpolarizing some LHb neurons in vitro.12 Peripheral administration of the antagonist SB 242084 did not alter the LHb neuron basal activity as its local application did,13 suggesting that 5‐HT2CRs do not exert tonic control over this nucleus. It is concluded that central 5‐HT2CR activation exerts phasic inhibitory and excitatory responses in distinct small population of neurons of the LHb.
Considering the relative lack of interest in the 5‐HT2AR in the LHb,2 one of the main and surprising findings of our study is that the 5‐HT2AR agonist TCB‐2 induced a strong dose‐dependent excitation or inhibition of the LHb neurons. The proportion of neurons globally affected by TCB‐2 was higher compared to Ro 60‐0175 (63% vs 34%) as was the magnitude of its maximum excitatory effect (125% vs 45%). On the other hand, like Ro 60‐0175, the pattern of discharge was not modified by TCB‐2. The pretreatment by the selective 5‐HT2AR antagonist MDL11939 reversed the excitatory and inhibitory effects of TCB‐2. Moreover, MDL11939 post‐treatment blocked both TCB‐2‐induced inhibitory and excitatory effects (G. Di Giovanni, unpublished observations). Although the pharmacological profile of TCB‐2 is poorly known beyond its high affinity and efficacy toward 5‐HT2AR,15 most pharmacological responses are blocked by 5‐HT2AR antagonists at low to moderate dosage,22 as in the present study. MDL11939 did not alter basal activity suggesting that 5‐HT2AR‐dependent controls, as for the 5‐HT2CRs, are also phasic.
These data suggest that peripheral administration of the 5‐HT2CR and 5‐HT2AR agonists induces mixed excitatory/inhibitory effects on LHb neurons. 5‐HT would preferentially excite LHb neurons via 5‐HT2ARs (higher magnitude compared to the 5‐HT2CRs and affecting a larger population of LHb neurons) and equally inhibit them via 5‐HT2CRs and/or 5‐HT2ARs. The pharmacological responses to both the 5‐HT2A/2CR agonists are quite similar: higher sensitivity to the dose of the excitatory response, no modification of the pattern of discharge, and no biphasic dose‐dependent effects for a neuron. These unexpected results prompted us to re‐evaluate the expression of these 5‐HT2R subtypes in the LHb and notably 5‐HT2AR, using standard immunohistochemical approaches.19 Interestingly, we reported moderate 5‐HT2AR‐like immunolabeling in the LHb, a finding that was not expected based on the lack of 5‐HT2AR mRNA reported in a previous study6 but now in agreement with recent microarray evidence.9 The 5‐HT2AR‐labeling concerned only 9% of LHb neuronal cells and also it was highly associated with GFAP‐labeled astrocytes and neuropil. Accompanying these results, we confirm previous in situ hybridization, immunohistochemical, and quantitative autoradiography data stating the presence of 5‐HT2CR in the LHb.6, 7, 8 The 5‐HT2CR‐labeling was located at the level of cell bodies in few neurons (9% also) and mainly in the neuropil. Interestingly, 5‐HT2CR‐like immunostaining was never associated with astrocyte cells.
Therefore, the stronger excitatory effects elicited by TCB‐2 compared to Ro 60‐0175 might be dependent on a concomitant activation of astrocytic 5‐HT2ARs. Indeed, 5‐HT2AR activation is capable of transcriptionally activating astrocytes in the rat cortex33 and increasing glial glutamate release via a calcium‐dependent mechanism in both C6 glioma cells34 and primary culture from newborn rat cerebral cortex.35 Strikingly, reactive astrocytes in different pathological states present an upregulation of 5‐HT2ARs in postmortem brain tissue36 including depression.37 Therefore, as a defective astrocytic uptake activity of extracellular glutamate has been shown to cause LHb hyperactivity and inducing depressive‐like behaviors,38 it would be very important to understand whether a concurrent upregulation of the 5‐HT2ARs occurs in the LHb of depressed patients.
Moreover, the different effects of 5‐HT2ARs and 5‐HT2CRs in LHb firing could be related to distinct cellular circuits in the LHb. Nevertheless, the protein of both receptors was not clustered in any specific LHb subnuclei, differently from their mRNA.8, 9 Moreover, in addition to the distinct distribution of 5‐HT2AR‐IR and 5‐HT2CR‐IR, we provide evidence that 5‐HT2CR and 5‐HT2AR antibodies were not co‐localized. In agreement, we did not find any 5‐HT2C‐5‐HT2AR heterodimers in the LHb, which recently have been shown to be expressed instead in the locus coeruleus.39 The part of the local mechanisms relative to the whole effects of the agonists is presently unknown. It is tempting to speculate that the excitatory effects, which have been reported in vitro and in vivo upon local administration of Ro 60‐0175,12, 13, 40 would occur locally. It would match the small proportion of neurons expressing 5‐HT2AR or 5‐HT2CR, the small proportion of neurons exhibiting excitatory responses and the high sensitivity to the dose of either agonist. Nonetheless, 5‐HT2AR and 5‐HT2CR are diffusely expressed in the brain6, 7, 41, 42, 43, 44; ectopic and indirect effects can be numerous including projections from the basal ganglia and notably the substantia nigra pars reticulata, the VTA, the entopeduncular nucleus, the mPFC, the lateral hypothalamus, or the DRN, just to cite a few.1
Some overlapping responses triggered by Ro 60‐0175 and TCB‐2 could also be envisioned, notably via their indirect inhibitory effects on DRN serotonergic neurons.26, 45, 46, 47 Indeed, Sharp et al48 have shown that the LHb inhibits DRN activity, through LHb neurons expressing 5‐HT2CRs (T. Sharp, personal communication), and the DRN also promotes a heterogeneous pattern of neuronal responses in the LHb,11 similar to the effects that we recorded here after 5‐HT2A/2CR stimulation. Additional data are warranted to further the role of 5‐HT system in the LHb effects of 5‐HT2A/2CR agonists.
5. CONCLUSION
The data presented here are important in the context of 5‐HT control of the LHb function in normal and pathological conditions. It can be suggested that the responses to 5‐HT2 agonists are heterogeneous as they recruit distinct neuronal populations and possibly astrocytic cells too. This result is not surprising if we consider the complexity of the LHb, characterized by a heterogeneous expression of neuropeptides and proteins, delimitating numerous and distinct habenular neuronal subpopulations which could serve different biological functions.8, 9
Our new findings on 5‐HT2AR and 5‐HT2CR are very interesting. Firstly, in consideration of the fact that these receptors play important roles in the mechanisms of action of antidepressant drugs and drug of abuse.3, 4, 21, 49, 50, 51 Secondly, most of the published data on 5‐HT2Rs in the LHb have been interpreted in light of 5‐HT2CR subtypes, while our data would suggest a main role for 5‐HT2ARs. Finally, our results suggest that blocking 5‐HT2ARs within the LHb might play an important role in the action of antidepressant drugs by normalizing the hyperexcitability of the LHb observed in depressive states.38, 52
Although no final conclusions can be drawn on the basis of the present findings due to some limitations, that is, the use of one agonist/antagonist ligand for each receptor and the route of drug administration, it appears clear that activation of either 5‐HT2AR or 5‐HT2CR induces heterogeneous neuronal responses of LHb neurons, with 5‐HT2ARs playing a more pivotal role in modulating this nucleus. Additional studies are warranted for a better understanding of the contribution of this heterogeneity to the central effects of 5‐HT2AR and 5‐HT2CR in normal and pathological conditions.
CONFLICT OF INTEREST
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supporting information
ACKNOWLEDGMENTS
We thank The University of Malta (PHBRP08‐17 to GDG) for supporting our research reported here. FD was supported by a Research Officer Fellowship from the MCST R&I‐2013‐014. MP was supported by a Research Officer Fellowship from the Faculty of Medicine and Surgery, University of Malta.
Delicata F, Bombardi C, Pierucci M, Di Maio R, De Deurwaerdère P, Di Giovanni G. Preferential modulation of the lateral habenula activity by serotonin‐2A rather than ‐2C receptors: Electrophysiological and neuroanatomical evidence. CNS Neurosci Ther. 2018;24:721–733. 10.1111/cns.12830
REFERENCES
- 1. Metzger M, Bueno D, Lima LB. The lateral habenula and the serotonergic system. Pharmacol Biochem Behav. 2017;162:22‐28. [DOI] [PubMed] [Google Scholar]
- 2. Tchenio A, Valentinova K, Mameli M. Can the Lateral Habenula Crack the Serotonin Code? Front Synaptic Neurosci. 2016;8:34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. De Deurwaerdere P, Di Giovanni G. Serotonergic modulation of the activity of mesencephalic dopaminergic systems: therapeutic implications. Prog Neurobiol. 2017;151:175‐236. [DOI] [PubMed] [Google Scholar]
- 4. Howell LL, Cunningham KA. Serotonin 5‐HT2 receptor interactions with dopamine function: implications for therapeutics in cocaine use disorder. Pharmacol Rev. 2015;67:176‐197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Millan MJ. Serotonin 5‐HT2C receptors as a target for the treatment of depressive and anxious states: focus on novel therapeutic strategies. Therapie. 2005;60:441‐460. [DOI] [PubMed] [Google Scholar]
- 6. Pompeiano M, Palacios JM, Mengod G. Distribution of the serotonin 5‐HT2 receptor family mRNAs: comparison between 5‐HT2A and 5‐HT2C receptors. Brain Res Mol Brain Res. 1994;23:163‐178. [DOI] [PubMed] [Google Scholar]
- 7. Clemett DA, Punhani T, Duxon MS, Blackburn TP, Fone KC. Immunohistochemical localisation of the 5‐HT2C receptor protein in the rat CNS. Neuropharmacology. 2000;39:123‐132. [DOI] [PubMed] [Google Scholar]
- 8. Aizawa H, Kobayashi M, Tanaka S, Fukai T, Okamoto H. Molecular characterization of the subnuclei in rat habenula. J Comp Neurol. 2012;520:4051‐4066. [DOI] [PubMed] [Google Scholar]
- 9. Wagner F, Bernard R, Derst C, French L, Veh RW. Microarray analysis of transcripts with elevated expressions in the rat medial or lateral habenula suggest fast GABAergic excitation in the medial habenula and habenular involvement in the regulation of feeding and energy balance. Brain Struct Funct. 2016;221:4663‐4689. [DOI] [PubMed] [Google Scholar]
- 10. Sharp T, Boothman L, Raley J, Quérée P. Important messages in the ‘post’: recent discoveries in 5‐HT neurone feedback control. Trends Pharmacol Sci. 2007;28:629‐636. [DOI] [PubMed] [Google Scholar]
- 11. Hartung H, Tan SK, Temel Y, Sharp T. High‐frequency stimulation of the subthalamic nucleus modulates neuronal activity in the lateral habenula nucleus. Eur J Neurosci. 2016;44:2698‐2707. [DOI] [PubMed] [Google Scholar]
- 12. Zuo W, Zhang Y, Xie G, Gregor D, Bekker A, Ye JH. Serotonin stimulates lateral habenula via activation of the post‐synaptic serotonin 2/3 receptors and transient receptor potential channels. Neuropharmacology. 2016;101:449‐459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Han LN, Zhang L, Li LB, et al. Activation of serotonin(2C) receptors in the lateral habenular nucleus increases the expression of depression‐related behaviors in the hemiparkinsonian rat. Neuropharmacology. 2015;93:68‐79. [DOI] [PubMed] [Google Scholar]
- 14. Martin JR, Bös M, Jenck F, et al. 5‐HT2C receptor agonists: pharmacological characteristics and therapeutic potential. J Pharmacol Exp Ther. 1998;286:913‐924. [PubMed] [Google Scholar]
- 15. McLean TH, Parrish JC, Braden MR, Marona‐Lewicka D, Gallardo‐Godoy A, Nichols DE. 1‐Aminomethylbenzocycloalkanes: conformationally restricted hallucinogenic phenethylamine analogues as functionally selective 5‐HT2A receptor agonists. J Med Chem. 2006;49:5794‐5803. [DOI] [PubMed] [Google Scholar]
- 16. Kennett GA, Wood MD, Bright F, et al. SB 242084, a selective and brain penetrant 5‐HT2C receptor antagonist. Neuropharmacology. 1997;36:609‐620. [DOI] [PubMed] [Google Scholar]
- 17. Dudley MW, Wiech NL, Miller FP, et al. Pharmacological effects of MDL 11,939: A selective, centrally acting antagonist of 5‐HT2 receptors. Drug Dev Res. 1988;13:29‐43. [Google Scholar]
- 18. Paxinos G, Watson C. The rat brain in stereotaxic coordinates. New York: Academic Press; 2007. [Google Scholar]
- 19. Orban G, Bombardi C, Marino Gammazza A, et al. Role(s) of the 5‐HT2C receptor in the development of maximal dentate activation in the hippocampus of anesthetized rats. CNS Neurosci Ther. 2014;20:651‐661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Venzi M, David F, Bellet J, et al. Role for serotonin2A (5‐HT2A) and 2C (5‐HT2C) receptors in experimental absence seizures. Neuropharmacology. 2016;108:292‐304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Di Giovanni G, De Deurwaerdere P. New therapeutic opportunities for 5‐HT2C receptor ligands in neuropsychiatric disorders. Pharmacol Ther. 2016;157:125‐162. [DOI] [PubMed] [Google Scholar]
- 22. Di Giovanni G, De Deurwaerdere P. TCB‐2 [(7R)‐3‐bromo‐2, 5‐dimethoxy‐bicyclo[4.2.0]octa‐1,3,5‐trien‐7‐yl]methanamine]: a hallucinogenic drug, a selective 5‐HT2A receptor pharmacological tool, or none of the above? Neuropharmacology. 2018, 10.1016/j.neuropharm.2017.10.004 [DOI] [PubMed] [Google Scholar]
- 23. Invernizzi RW, Pierucci M, Calcagno E, et al. Selective activation of 5‐HT(2C) receptors stimulates GABA‐ergic function in the rat substantia nigra pars reticulata: a combined in vivo electrophysiological and neurochemical study. Neuroscience. 2007;144:1523‐1535. [DOI] [PubMed] [Google Scholar]
- 24. Söderberg O, Gullberg M, Jarvius M, et al. Direct observation of individual endogenous protein complexes in situ by proximity ligation. Nat Methods. 2006;3:995‐1000. [DOI] [PubMed] [Google Scholar]
- 25. Kowski AB, Veh RW, Weiss T. Dopaminergic activation excites rat lateral habenular neurons in vivo. Neuroscience. 2009;161:1154‐1165. [DOI] [PubMed] [Google Scholar]
- 26. Quérée P, Peters S, Sharp T. Further pharmacological characterization of 5‐HT(2C) receptor agonist‐induced inhibition of 5‐HT neuronal activity in the dorsal raphe nucleus in vivo. Br J Pharmacol. 2009;158:1477‐1485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Paoletti P, Bellone C, Zhou Q. NMDA receptor subunit diversity: impact on receptor properties, synaptic plasticity and disease. Nat Rev Neurosci. 2013;14:383‐400. [DOI] [PubMed] [Google Scholar]
- 28. Traynelis SF, Wollmuth LP, McBain CJ, et al. Glutamate receptor ion channels: structure, regulation, and function. Pharmacol Rev. 2010;62:405‐496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Xie G, Zuo W, Wu L, et al. Serotonin modulates glutamatergic transmission to neurons in the lateral habenula. Sci Rep. 2016;6:23798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Hwang EK, Chung JM. 5HT(1B) receptor‐mediated pre‐synaptic depression of excitatory inputs to the rat lateral habenula. Neuropharmacology. 2014;81:153‐165. [DOI] [PubMed] [Google Scholar]
- 31. Fletcher PJ, Sinyard J, Higgins GA. The effects of the 5‐HT(2C) receptor antagonist SB242084 on locomotor activity induced by selective, or mixed, indirect serotonergic and dopaminergic agonists. Psychopharmacology. 2006;187:515‐525. [DOI] [PubMed] [Google Scholar]
- 32. Navailles S, Lagière M, Le Moine C, De Deurwaerdère P. Role of 5‐HT2C receptors in the enhancement of c‐Fos expression induced by a 5‐HT2B/2C inverse agonist and 5‐HT 2 agonists in the rat basal ganglia. Exp Brain Res. 2013;230:525‐535. [DOI] [PubMed] [Google Scholar]
- 33. Martin DA, Nichols CD. Psychedelics Recruit Multiple Cellular Types and Produce Complex Transcriptional Responses Within the Brain. EBioMedicine. 2016;11:262‐277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Meller R, Harrison PJ, Elliot JM, Sharp T. In vitro evidence that 5‐hydroxytryptamine increases efflux of glial glutamate via 5‐HT(2A) receptor activation. J Neurosci Res. 2002;67:399‐405. [DOI] [PubMed] [Google Scholar]
- 35. Hagberg GB, Blomstrand F, Nilsson M, Tamir H, Hansson E. Stimulation of 5‐HT2A receptors on astrocytes in primary culture opens voltage‐independent Ca2+ channels. Neurochem Int. 1998;32:153‐162. [DOI] [PubMed] [Google Scholar]
- 36. Wu C, Singh SK, Dias P, Kumar S, Mann DM. Activated astrocytes display increased 5‐HT2a receptor expression in pathological states. Exp Neurol. 1999;158:529‐533. [DOI] [PubMed] [Google Scholar]
- 37. Shelton RC, Sanders‐Bush E, Manier DH, Lewis DA. Elevated 5‐HT 2A receptors in postmortem prefrontal cortex in major depression is associated with reduced activity of protein kinase A. Neuroscience. 2009;158:1406‐1415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Cui W, Mizukami H, Yanagisawa M, et al. Glial dysfunction in the mouse habenula causes depressive‐like behaviors and sleep disturbance. J Neurosci. 2014;34:16273‐16285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Moutkine I, Quentin E, Guiard BP, Maroteaux L, Doly S. Heterodimers of serotonin receptor subtypes 2 are driven by 5‐HT2C protomers. J Biol Chem. 2017;292:6352‐6368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Hwang CK, D'Souza UM, Eisch AJ, et al. Dopamine receptor regulating factor, DRRF: a zinc finger transcription factor. Proc Natl Acad Sci U S A. 2001;98:7558‐7563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. López‐Giménez JF, Mengod G, Palacios JM, Vilaró MT. Selective visualization of rat brain 5‐HT2A receptors by autoradiography with [3H]MDL 100,907. Naunyn Schmiedebergs Arch Pharmacol. 1997;356:446‐454. [DOI] [PubMed] [Google Scholar]
- 42. López‐Giménez JF, Vilaró MT, Palacios JM, Mengod G. [3H]MDL 100,907 labels 5‐HT2A serotonin receptors selectively in primate brain. Neuropharmacology. 1998;37:1147‐1158. [DOI] [PubMed] [Google Scholar]
- 43. Mengod G, Nguyen H, Le H, Waeber C, Lübbert H, Palacios JM. The distribution and cellular localization of the serotonin 1C receptor mRNA in the rodent brain examined by in situ hybridization histochemistry. Comparison with receptor binding distribution. Neuroscience. 1990;35:577‐591. [DOI] [PubMed] [Google Scholar]
- 44. Wright DE, Seroogy KB, Lundgren KH, Davis BM, Jennes L. Comparative localization of serotonin1A, 1C, and 2 receptor subtype mRNAs in rat brain. J Comp Neurol. 1995;351:357‐373. [DOI] [PubMed] [Google Scholar]
- 45. Boothman L, Raley J, Denk F, Hirani E, Sharp T. In vivo evidence that 5‐HT(2C) receptors inhibit 5‐HT neuronal activity via a GABAergic mechanism. Br J Pharmacol. 2006;149:861‐869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Boothman LJ, Allers KA, Rasmussen K, Sharp T. Evidence that central 5‐HT2A and 5‐HT2B/C receptors regulate 5‐HT cell firing in the dorsal raphe nucleus of the anaesthetised rat. Br J Pharmacol. 2003;139:998‐1004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Spoida K, Masseck OA, Deneris ES, Herlitze S. Gq/5‐HT2c receptor signals activate a local GABAergic inhibitory feedback circuit to modulate serotonergic firing and anxiety in mice. Proc Natl Acad Sci U S A. 2014;111:6479‐6484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Varga V, Kocsis B, Sharp T. Electrophysiological evidence for convergence of inputs from the medial prefrontal cortex and lateral habenula on single neurons in the dorsal raphe nucleus. Eur J Neurosci. 2003;17:280‐286. [DOI] [PubMed] [Google Scholar]
- 49. Millan MJ. The neurobiology and control of anxious states. Prog Neurobiol. 2003;70:83‐244. [DOI] [PubMed] [Google Scholar]
- 50. Millan MJ, Marin P, Kamal M, et al. The melatonergic agonist and clinically active antidepressant, agomelatine, is a neutral antagonist at 5‐HT2C receptors. Int J Neuropsychopharmacol. 2010;14:768‐783. [DOI] [PubMed] [Google Scholar]
- 51. Di Matteo V, Di Giovanni G, Di Mascio M, Esposito E. Effect of acute administration of hypericum perforatum‐CO2 extract on dopamine and serotonin release in the rat central nervous system. Pharmacopsychiatry. 2000;33:14‐18. [DOI] [PubMed] [Google Scholar]
- 52. Li Y, Wang Y, Xuan C, et al. Role of the Lateral Habenula in Pain‐Associated Depression. Front Behav Neurosci. 2017;11:31. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials


