Abstract
Fatty liver is a common finding in clinical practice and at autopsy. It is most commonly seen associated with alcohol abuse and in non-alcoholic fatty liver disease (NAFLD). It may also be seen in many other conditions in both adults and children. It is now recognized that NAFLD, like alcoholic liver disease, may lead to end stage liver disease. Nonalcoholic fatty liver disease is associated with increased mortality from other disorders, particularly cardiovascular diseases. Fatty liver may be seen in many conditions that concern autopsy pathologists, including drug toxicity, anorexia, hepatic ischemia, and heatstroke. In infants, steatosis is common in sudden unexpected deaths. Fatty liver has been associated with sudden death and this review examines the pathology and role of fatty liver in sudden death. Acad Forensic Pathol. 2018 8(2): 296-310
Keywords: Forensic pathology, Fatty, Steatosis, Liver, NAFLD, Alcohol
Introduction
Fatty liver is common and can occur in many conditions in both adults and children (1). It is frequently found at autopsy, especially if microscopy is conducted. Fatty liver can be considered as a marker of other disorders and has been used as a cause of death in the past. While fatty liver is important in the pediatric age group, this paper concentrates on fatty liver in adults at autopsy and discusses its role in causing sudden death.
Discussion
Histopathology of Fatty Liver
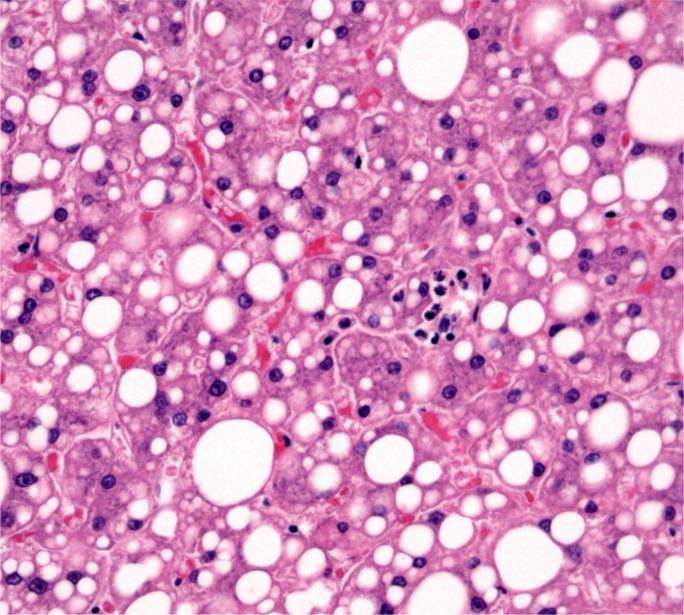
Fatty liver, also known as steatosis, occurs in two main patterns, macrovesicular steatosis, the most common pattern, and microvesicular steatosis (2). The two main patterns often overlap. Macrovesicular steatosis is identified by a single fat vacuole, with the nucleus displaced to the edge (Image 1), while microvesicular steatosis has multiple small cytoplasmic vacuoles with a typically centrally placed nucleus (Image 2). Macrovesicular steatosis can be seen in a wide range of insults to the liver including alcohol, diabetes mellitus and metabolic syndrome, obesity, malnutrition, various metabolic disorders, drugs and toxins, and viral hepatitis. Microvesicular steatosis can be seen in a number of conditions such as acute fatty liver of pregnancy, Reyes syndrome, inherited disorders of fatty acid metabolism, infection, and drug toxicity, including salicylate, valproate, and tetracycline (1). Conditions associated with macrovesicular and microvesicular steatosis are listed in Table 1.
Image 1:
Predominantly macrosvesicular steatosis (H&E, x 200).
Image 2:
Microvesicular steatosis (H&E, x 400).
Table 1:
Causes of Liver Steatosis
| Macrovesicular Steatosis | Microvesicular Steatosis |
|---|---|
| Alcoholic fatty liver | Alcoholic foamy degeneration |
| Nonalcoholic fatty liver | Reyes syndrome |
| Obesity | Drugs |
| Diabetes mellitus | Acute fatty liver of pregnancy |
| Viral hepatitis | Inherited disorders of fatty acid metabolism |
| Drugs | Inherited urea cycle disorders |
| Total parenteral nutrition | Wolman disease |
| Malnutrition | Mitochondrial cytopathies |
| Wilson disease | Infection |
| Polycystic ovarian syndrome | Cholesterol ester storage disease |
| Mandibuloacral dysplasia | |
| Familial partial lipodystrophies | |
| Acquired lipodystrophies | |
| Hepatic ischemia |
Steatohepatitis
Steatohepatitis, also known as steatonecrosis, refers to the pattern of cell injury in fatty liver disease. The mechanisms of injury overlap (3), and the morphology is similar between alcoholic and nonalcoholic etiologies, such that the diagnosis of alcoholic steatohepatitis cannot be made on histology alone (4). Furthermore, alcoholic liver disease may exist with other comorbidities including NAFLD and hepatitis C infection (5).
Alcoholic Liver Disease
Alcoholic steatosis is seen in up to 90% of patients presenting for treatment of chronic alcoholism (6). Fatty change is mainly seen in the perivenular zone, but as the fatty change progresses it can be seen in all zones. Abstinence of alcohol results in fat disappearing in two to four weeks. Lipogranulomata may been seen at this stage, otherwise inflammation is absent in simple steatosis (1).
Alcoholic foamy degeneration is a term used to describe microvesicular change seen with alcohol use (7). It is not associated with inflammation and Mallory-Denk bodies are usually absent. The condition subsides with abstinence.
Alcoholic hepatitis, also called alcoholic steatonecrosis, is characterized by steatosis, liver cell necrosis, and neutrophil inflammation (Image 3) (2, 5). In one study, it was present in 17 out of 100 liver biopsies in patients presenting for treatment of alcoholism (8). Sclerosing hyaline necrosis occurs when there is extensive perivenular hepatocyte necrosis with fibrosis (9). This may result in portal hypertension without cirrhosis. Venous lesions seen in alcoholic liver disease include lymphocytic phlebitis, phlebosclerosis, and veno-occlusive disease (10, 11). Phlebosclerosis, compression of hepatic vein radicles by perivenular fibrosis, is a universal finding in alcoholic hepatitis and cirrhosis, whereas veno-occlusive lesions, intimal proliferation, and fibrosis were seen in 25% of biopsies and lymphocytic phlebitis, a predominantly lymphocytic inflammatory infiltrate in the hepatic vein branches, in 4%.
Image 3:
Steatohepatitis (H&E, x 200).
Perivenular fibrosis is defined as fibrosis surrounding two-thirds of a terminal hepatic venule, with the fibrosis measuring over 4 μm (Image 4). Fibrosis is a marker for more progressive disease if alcohol consumption continues (Image 5). In males drinking 40-80 grams of alcohol a day, nearly 40% had perivenular fibrosis.
Image 4:
Perivenular fibrosis (Masson trichrome, x100).
Image 5:
Fibrosis with fatty change (Masson trichrome, x100).
Perivenular fibrosis is believed to be the precursor of cirrhosis (12, 13); however, only 20% of heavy drinkers develop cirrhosis (1). Cirrhosis is most typically micronodular, with nodules less than 3 mm. Macro-nodular cirrhosis may develop especially following abstinence, when regeneration of nodules takes place (14).
Nonalcoholic Fatty Liver Disease
Nonalcoholic fatty liver disease (NAFLD) is a term used for a range of changes seen in the liver and encompasses various terms including nonalcoholic steatohepatitis (NASH) and non-NASH fatty liver (NNFL), which is a term used to describe fatty liver disease that carries a better prognosis than NASH and typically presents with steatosis but with minimal, if any, inflammation or fibrosis (15).
Steatosis is defined as abnormal when exceeding 5% in microscopic sections (16). Nonalcoholic steatohepatitis involves inflammation, ballooned hepatocytes, and evidence of cell death; fibrosis may be present. Nonalcoholic steatohepatitis may progress to cirrhosis, typically in people who are over 60 years of age, are obese, and have diabetes. This cirrhosis is some-times referred to as cryptogenic cirrhosis, though it has been questioned whether cirrhosis related to NASH is the same entity (17).
For a diagnosis of NASH, alcohol consumption should be less than 20 grams per day (18). Although significant ethanol consumption is an exclusionary criterion for the diagnosis of NASH, patients classified as having NASH were found to have had a significant lifetime exposure to alcohol (19). As there is overlap between NASH and alcoholic steatohepatitis (ASH) histologically, with the exception of an increased incidence of glycogenated nuclei (Image 6) in NASH (20), it can be impossible to tell the two disorders apart, especially in a medicolegal investigation when medical and social history may be limited.
Image 6:
Glycogenated nuclei (H&E, x200).
Non-NASH-fatty liver (NNFL) is a term used for fatty liver without significant fibrosis or inflammation. It is more stable than NASH, though progress to NASH may occur.
The frequency of NAFLD has been reported to be high in the general population (21). While a wide range of conditions may be associated with NAFLD, diabetes mellitus, obesity, and hyperlipidemia are the commonest associations (22). Nonalcoholic fatty liver disease may have a worldwide prevalence of 25%, and as high as 38% depending on criteria and region studied. In obese patients undergoing bariatric surgery, typically with a body mass index (BMI) over 35 kg/m2, the prevalence of steatosis was 85-90%, with the prevalence of NASH of 25-20%, and unexpected cirrhosis of 1-2% (23).
Histological features of NASH include macrovesicular and microsvesicular steatois with portal and/or lobular inflammation (1). Fibrosis is present, typically in zone 3, around the peri-central veins. The hepatocytes may contain Mallory-Denk bodies (Mallory hyaline) and there may be lipogranulomas and glycogenated nuclei. The disease may progress to cirrhosis. Scoring systems for severity of NAFLD have been devised (24).
The risk of NAFLD is increased in obesity, which carries a 4.6-fold increase over nonobese people, as assessed by ultrasound (25). However, people with a normal BMI may still have fatty liver (26, 27). These patients tend to have visceral adiposity along with hyperinsulinemia and poor physical conditioning. Diabetes mellitus increases the risk of fatty liver and also increases the risk of cirrhosis (28). Patients with NASH cirrhosis are at risk of developing hepatocellular carcinoma (HCC) (29). Hepatocellular carcinoma has been associated with NNFL, suggesting steatosis alone is a risk factor for the development of HCC (30, 31).
Nonalcoholic fatty liver disease may coexist with other liver disorders. Patients with hepatitis C infection (HCV) have a poorer response to interferon therapy if they have steatosis, obesity, and hyperinsulinism and probably the combination accelerates progression (32). Steatosis also affects progression of primary biliary cholangitis (33).
There are genetic, gender, and familial variations in NAFLD, which is more common among certain ethnic groups (34 –36). Men have a higher rate of NAFLD than women. In the US, Latinos have a higher rate and African Americans a lower rate, when compared to rates of obesity in the population. There are differences in the distribution of body fat in these groups and in lipoprotein metabolism (37 –39). First degree relatives have higher rates of NASH and genetic studies have linked NAFLD to certain genes including for patatin-like phospholipase 3 (PNPLA3), which may have a role in transferring fatty acids between lipids (40 –43). Another is transmembrane 6 superfamily member 2 (TM6SF2), which is a regulator of hepatic lipid droplet content. These two genes are related to disease severity and related morbidities including vascular disease.
Clinical Features of Nonalcoholic Fatty Liver Disease
Patients with NAFLD are typically asymptomatic (44). They may have a history of fatigue and right upper quadrant pain. They often have obstructive sleep apnea and women may have polycystic ovaries. Neurological deficits have been reported occasionally, believed to be related to lipotoxicity. On external examination, there may be acanthosis nigrans, palmar erthyema, and spider angiomata (spider nevi) (15). Biochemical abnormalities include elevated liver enzymes, iron, uric acid, and immunoglobulin A (15). Antinuclear antibodies may be positive. There is hepatomegaly.
Other Causes of Fatty Liver
Fatty liver is found in many conditions other than ALD and NAFLD. Macrovesicular and microvesicular steatosis can be seen with drug toxicity (Image 7), including nifedipine, diltiazem, tamoxifen, estrogen, and corticosteroids (1, 15). Solvents and petrochemicals can also cause fatty liver. Hepatitis C, B, and D infection may cause steatosis, as may disorders of lipoprotein and nutritional disorders, including anorexia nervosa and total parenteral nutrition, bypass surgery for obesity and small bowel resection, hepatic ischemia and systemic disorders such as cachexia, heat stroke, cystic fibrosis, and inflammatory bowel disease (Image 8).
Image 7:
Steatosis in colchicine toxicity (H&E, x200).
Image 8A:
Steatosis in anorexia nervosa (Masson trichrome, x40).
Image 8B:
Steatosis in anorexia nervosa (Masson trichrome, x100).
Mortality and Nonalcoholic Fatty Liver Disease
Patients diagnosed with NAFLD have a mortality of 10-12% over a 10 to 15 year period (45 –49). Fibrosis is a predictor of mortality (50). The main causes of death are reported to be coronary artery disease, extrahepatic malignancies, and cirrhosis of the liver. One long-term study indicated that deaths from cirrhosis exceeded cardiac causes (47).
Fatty Liver at Autopsy
There have been few systematic studies of fatty liver at autopsy. A study of 207 obese patients was compared with 207 control patients examined at autopsy at the Western General Hospital in Toronto, Canada between 1960 and 1987 (51). Sixty-three patients had a history of alcohol use and were excluded from further analysis, leaving 351 in the analysis. Fatty liver was present in both groups, though more prevalent in the obese patients, and with greater severity than in controls. Steatohepatitis was seen in 22 of the 351 cases, including 18.5% of obese patients and only 2.7% of nonobese patients. Weight loss before death was also associated with steatosis. Fibrosis was seen more frequently in obese patients and was associated with diabetes mellitus.
An autopsy study from Northwestern Greece revealed frequent steatosis and steatohepatitis (52). The authors examined 498 medicolegal cases with a mean age of 64.51 years. The most common causes of death were ischemic heart disease (47.59%), traffic accidents (13.45%), and pulmonary embolism (8.23%). Other causes of death were each less than 5%. Of the 498 cases, 28.9% of cases had normal livers, steatosis was present in 31.3%, and steatohepatitis in 39.8% (52).
An autopsy study of livers in people dying of diabetic ketoacidosis with no prior history of diabetes mellitus revealed significant steatosis and steatohepatitis (53). The BMIs of these people were typically within the normal (nonobese) range.
Is Fatty Liver a Cause of Death?
Fatty liver has been reported as a cause of sudden death (54). In a study of alcohol-related deaths from Baltimore examined between 1957 and 1966, fatty liver and cirrhosis were noted to have increased as a cause of death (55). The authors noted that several hypotheses had been suggested for the proximate cause of death, including fatty emboli to the lungs and the brain, hypoglycemia secondary to alcoholism, and alcoholic cardiomyopathy. They noted that a cardiac arrhythmia has been suspected as the mechanism of death, a cause they noted would be impossible to determine from postmortem examination.
Clark, in a review of 500 alcohol deaths from Glasgow, Scotland, classified 51 deaths as being “obscure,” with 32 cases having fatty liver (56). Both the papers from Baltimore and Glasgow were published before alcoholic ketoacidosis was reported in postmortem cases (57). This condition certainly provides an explanation for some deaths in cases of fatty liver. Another mechanism now recognized to be associated with alcoholics is cardiac arrhythmias associated with cirrhosis and alcoholic liver disease more severe than simple steatosis.
Nonalcoholic fatty liver disease has been associated with cardiac arrhythmias (58 –61), the most common of which is atrial fibrillation. There is less data on ventricular arrhythmias. A small study of patients with NAFLD revealed they had evidence of autonomic dysfunction, and another study of diabetics revealed that those with more severe NAFLD had prolonged QTc intervals on electrocardiogram examination (58). Thus, there are potential mechanisms for sudden death in patients with NAFLD, though further work is required to determine if this is so and what the risks are. Nonalcoholic fatty liver disease can be considered a multisystem disease (62)
Fatty Liver in the Pediatric Age Group
Fatty liver is not uncommon in the pediatric age group. An autopsy study in 2006 suggested an prevalence of around 10% (63), and the prevalence in a Japanese population was found to be 2.4% in another study (64). Nonalcoholic fatty liver disease in the pediatric age group is associated with greater severity of steatosis, less lobular inflammation, and portal fibrosis and inflammation without perivenular fibrosis (65). Nonalcoholic fatty liver disease in children may progress to end stage liver disease (66).
Steatosis may be seen in metabolic disorders such as medium chain fatty acid disorders (67). Reyes syndrome and metabolic disorders should be excluded; however, steatosis has been reported to be common in sudden unexpected infant deaths and is not diagnostic of a metabolic disorder or Reyes syndrome (68, 69).
Conclusion
Fatty liver is common at autopsy in all age groups. In adults, it is most commonly seen in alcoholic and nonalcoholic fatty liver disease. The most common pattern is macrovesicular steatosis. As the disease progresses, fibrosis increases and cirrhosis may result. The pathologic features of these conditions typically overlap and histology alone is insufficient to distinguish them; history and other investigative findings may aid in diagnostic discernment. Fatty liver disease may be seen with some drugs and in some metabolic disorders. Microvesicular steatosis may be seen, including acute fatty liver of pregnancy and some drug toxicities.
Fatty liver is not an adequate stand alone cause of death. The presence of fatty liver at autopsy should not be considered as alcohol-related unless there is a clear history of alcohol use. Fatty liver is an indication of an underlying disorder, which may be the cause of death. The presence of fatty liver disease should prompt a search for an underlying cause of death such as alcoholic or diabetic ketoacidosis. While fatty liver as a cause of death has been challenged, there is now some evidence to support alcoholic and nonalcoholic fatty liver disease being related to cardiac arrhythmias and thus to sudden death.
Biography
Christopher M. Milroy MBChB MD LLB BA LLM FRCPath FFFLM FRCPC DMJ, The Ottawa Hospital - Anatomical Pathology
Roles: Project conception and/or design, data acquisition, analysis and/or interpretation, manuscript creation and/or revision, approved final version for publication, accountable for all aspects of the work.
Footnotes
Ethical Approval: As per Journal Policies, ethical approval was not required for this manuscript
Statement of Human and Animal Rights: This article does not contain any studies conducted with animals or on living human subjects
Statement of Informed Consent: No identifiable personal data were presented in this manuscript
Disclosures & Declaration of Conflicts of Interest: Christopher M. Milroy is the Editor-In-Chief of Academic Forensic Pathology: The Official Publication of the National Association of Medical Examiners. The author, reviewers, editors, and publication staff do not report any other relevant conflicts of interest
Financial Disclosure: The author has indicated that he does not have financial relationships to disclose that are relevant to this manuscript
References
- 1). Tiniakos DG, Anstee QM, Burt AD. MacSween’s pathology of the liver. 7th ed Philadephia: Elsevier; c2018. Chapter 5, Fatty liver disease; p. 308–71. [Google Scholar]
- 2). MacSween RN, Burt AD. Histologic spectrum of alcoholic liver disease. Semin Liver Dis. 1986. August; 6(3):221–32. PMID: 3022386 10.1055/s-2008-1040605. [DOI] [PubMed] [Google Scholar]
- 3). Syn WK, Teaberry V, Choi SS, Diehl AM, Similarities and differences in the pathogenesis of alcoholic and nonalcoholic steatohepatitis. Semin Liver Dis. 2009. August; 29(2):200–10. PMID: 19387919. PMCID: PMC3644873 10.1055/s-0029-1214375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4). Tannapfel A, Denk H, Dienes HP, et al. Histopathological diagnosis of non-alcoholic and alcoholic fatty liver disease. Virchows Arch. 2011. August; 458(5):511–23. PMID: 21442288 10.1007/s00428-011-1066-1. [DOI] [PubMed] [Google Scholar]
- 5). Yip WW, Burt AD. Alcoholic liver disease. Semin Diagn Pathol. 2006. Aug-Nov; 23(3-4):149–60. PMID: 17355088 10.1053/j.semdp.2006.11.002. [DOI] [PubMed] [Google Scholar]
- 6). Edmondson HA, Peters RL, Frankel HH, Borowsky S. The early stage of liver injury in the alcoholic. Medicine (Baltimore). 1967. March; 46(2):119–29. PMID: 6027458 10.1097/00005792-196703000-00006. [DOI] [PubMed] [Google Scholar]
- 7). Uchida T, Kao H, Quispe-Sjogren M, Peters RL. Alcoholic foamy degeneration--a pattern of acute alcoholic injury of the liver. Gastroenterology. 1983. April; 84(4):683–92. PMID: 6825980. [PubMed] [Google Scholar]
- 8). Bhathal PS, Wilkinson P, Clifton S, et al. The spectrum of liver diseases in alcoholism. Aust N Z J Med. 1975. February; 5(1):49–57. PMID: 1057913 10.1111/j.1445-5994.1975.tb03255.x. [DOI] [PubMed] [Google Scholar]
- 9). Edmondson HA, Peters RL, Reynolds TB, Kuzma OT. Sclerosing hyaline necrosis of the liver in the chronic alcoholic. A recognizable clinical syndrome. Ann Intern Med. 1963. November; 59:646–73. PMID: 14082718 10.7326/0003-4819-59-5-646. [DOI] [PubMed] [Google Scholar]
- 10). Goodman ZD, Ishak KG. Occlusive venous lesions in alcoholic liver disease. A study of 200 cases. Gastroenterology. 1982. October; 83(4): 786–96. PMID: 7106509. [PubMed] [Google Scholar]
- 11). Burt AD, MacSween RN. Hepatic vein lesions in alcoholic liver disease: retrospective biopsy and necropsy study. J Clin Pathol. 1986. January; 39(1):63–7. PMID: 3950032. PMCID: PMC499614 10.1136/jcp.39.1.63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12). Nakano M, Worner TM, Lieber CS. Perivenular fibrosis in alcoholic liver injury: ultrastructure and histologic progression. Gastroenterology. 1982. October;83(4):777–85. PMID: 7106508. [PubMed] [Google Scholar]
- 13). Worner TM, Lieber CS. Perivenular fibrosis as precursor lesion of cirrhosis. JAMA. 1985. August 2; 254(5):627–30. PMID: 4009897 10.1001/jama.254.5.627. [DOI] [PubMed] [Google Scholar]
- 14). Fauerholdt L, Schlichting P, Christensen E, et al. Conversion of micronodular cirrhosis into macronodular cirrhosis. Hepatology. 1983 Nov-Dec; 3(6):928–31. PMID: 6629323 10.1002/hep.1840030607. [DOI] [PubMed] [Google Scholar]
- 15). Argo CK, Henry ZH, Caldwell SH. Schiff’s diseases of the liver. 12th ed Hoboken (NJ): John Wiley and Sons; c2018. Chapter 32, Nonalcoholic fatty liver disease; p. 867–910. [Google Scholar]
- 16). Brunt EM. Pathology of nonalcoholic fatty liver disease. Nat Rev Gastroenterol Hepatol. 2010. April; 7(4):195–203. PMID: 20195271 10.1038/nrgastro.2010.21. [DOI] [PubMed] [Google Scholar]
- 17). Thuluvath PJ, Kantsevoy S, Thuluvath AJ, Savva Y. Is cryptogenic cirrhosis different from NASH cirrhosis? J Hepatol. 2018. March; 68(3):519–525. PMID: 29162389 10.1016/j.jhep.2017.11.018. [DOI] [PubMed] [Google Scholar]
- 18). Becker U, Deis A, Sørensen TI, et al. Prediction of risk of liver disease by alcohol intake, sex, and age: a prospective population study. Hepatology. 1996. August; 23(5):1025–9. PMID: 8621128 10.1053/jhep.1996.v23.pm0008621128. [DOI] [PubMed] [Google Scholar]
- 19). Hayashi PH, Harrison SA, Torgerson S, et al. Cognitive lifetime drinking history in nonalcoholic fatty liver disease: some cases may be alcohol related. Am J Gastroenterol. 2004. January; 99(1):76–81. PMID: 14687145 10.1046/j.1572-0241.2003.04013.x. [DOI] [PubMed] [Google Scholar]
- 20). Pinto HC, Baptista A, Camilo ME, et al. Nonalcoholic steatohepatitis. Clinicopathological comparison with alcoholic hepatitis in ambulatory and hospitalized patients. Dig Dis Sci. 1996. January; 41(1):172–9. PMID: 8565753 10.1007/bf02208601. [DOI] [PubMed] [Google Scholar]
- 21). Younossi ZM, Koenig AB, Abdelatif D, et al. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology. 2016. July; 64(1):73–84. PMID: 27607365 10.1002/hep.28431. [DOI] [PubMed] [Google Scholar]
- 22). Angulo P, Keach JC, Batts KP, Lindor KD. Independent predictors of liver fibrosis in patients with nonalcoholic steatohepatitis. Hepatology. 1999. December; 30(6):1356–62. PMID: 10573511 10.1002/hep.510300604. [DOI] [PubMed] [Google Scholar]
- 23). Machado M, Marques-Vidal P, Cortez-Pinto H. Hepatic histology in obese patients undergoing bariatric surgery. J Hepatol. 2006. October; 45(4):600–6. PMID: 16899321 10.1016/j.jhep.2006.06.013. [DOI] [PubMed] [Google Scholar]
- 24). Kleiner DE, Brunt EM, Van Natta M, et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology. 2005. June; 41(6):1313–21. PMID: 15915461 10.1002/hep.20701. [DOI] [PubMed] [Google Scholar]
- 25). Bellentani S, Saccoccio G, Masutti F, et al. Prevalence of and risk factors for hepatic steatosis in Northern Italy. Ann Intern Med. 2000. January 18; 132(2):112–7. PMID: 10644271 10.7326/0003-4819-132-2-200001180-00004. [DOI] [PubMed] [Google Scholar]
- 26). Park SH, Kim BI, Yun JW, et al. Insulin resistance and C-reactive protein as independent risk factors for non-alcoholic fatty liver disease in non-obese Asian men. J Gastroenterol Hepatol. 2004. June; 19(6):694–8. PMID: 15151626 10.1111/j.1440-1746.2004.03362.x. [DOI] [PubMed] [Google Scholar]
- 27). Omagari K, Kadokawa Y, Masuda J, et al. Fatty Liver in non-alcoholic non-overweight Japanese adults: incidence and clinical characteristics. J Gastroenterol Hepatol. 2002. October; 17(10):1098–105. PMID 12201871 10.1046/j.1440-1746.2002.02846.x. [DOI] [PubMed] [Google Scholar]
- 28). Silverman JF, O’Brien KF, Long S, et al. Liver pathology in morbidly obese patients with and without diabetes. Am J Gastroenterol. 1990. October; 85(10):1349–55. PMID: 2220728. [PubMed] [Google Scholar]
- 29). Younossi ZM, Otgonsuren M, Henry L, et al. Association of nonalcoholic fatty liver disease (NAFLD) with hepatocellular carcinoma (HCC) in the United States from 2004 to 2009. Hepatology. 2015. December; 62(6):1723–30. PMID: 26274335 10.1002/hep.28123. [DOI] [PubMed] [Google Scholar]
- 30). Ertle J, Dechêne A, Sowa JP, et al. Non-alcoholic fatty liver disease progresses to hepatocellular carcinoma in the absence of apparent cirrhosis. Int J Cancer. 2011. May 15; 128(10):2436–43. PMID: 21128245 10.1002/ijc.25797. [DOI] [PubMed] [Google Scholar]
- 31). Baffy G, Brunt EM, Caldwell SH. Hepatocellular carcinoma in non-alcoholic fatty liver disease: an emerging menace. J Hepatol. 2012. June; 56(6):1384–91. PMID: 22326465 10.1016/j.jhep.2011.10.027. [DOI] [PubMed] [Google Scholar]
- 32). Solis-Herruzo JA, Pérez-Carreras M, Rivas E, et al. Factors associated with the presence of nonalcoholic steatohepatitis in patients with chronic hepatitis C. Am J Gastroenterol. 2005. August; 100(5): 1091–8. PMID: 15842583 10.1111/j.1572-0241.2005.41059.x. [DOI] [PubMed] [Google Scholar]
- 33). Sorrentino P, Terracciano L, D’Angelo S, et al. Oxidative stress and steatosis are cofactors of liver injury in primary biliary cirrhosis. J Gastroenterol. 2010. October; 45(10):1053–62. PMID: 20393861 10.1007/s00535-010-0249-x. [DOI] [PubMed] [Google Scholar]
- 34). Weston SR, Leyden W, Murphy R, et al. Racial and ethnic distribution of nonalcoholic fatty liver in persons with newly diagnosed chronic liver disease. Hepatology. 2005. February; 41(2):372–9. PMID: 15723436 10.1002/hep.20554. [DOI] [PubMed] [Google Scholar]
- 35). Browning JD, Szczepaniak LS, Dobbins R, et al. Prevalence of hepatic steatosis in an urban population in the United States: impact of ethnicity. Hepatology. 2004. December; 40(6):1387–95. PMID: 15565570 10.1002/hep.20466. [DOI] [PubMed] [Google Scholar]
- 36). Schwimmer JB, McGreal N, Deutsch R, et al. Influence of gender, race, and ethnicity on suspected fatty liver in obese adolescents. Pediatrics. 2005. August; 115(5): e561–5. PMID: 15867021 10.1542/peds.2004-1832. [DOI] [PubMed] [Google Scholar]
- 37). Lear SA, Humphries KH, Kohli S, Birmingham CL. The use of BMI and waist circumference as surrogates of body fat differs by ethnicity. Obesity (Silver Spring). 2007. November; 15(11):2817–24. PMID: 18070773 10.1038/oby.2007.334. [DOI] [PubMed] [Google Scholar]
- 38). Wang J, Thornton JC, Russell M, et al. Asians have lower body mass index (BMI) but higher percent body fat than do whites: comparisons of anthropometric measurements. Am J Clin Nutr. 1994. July; 60(1): 23–8. PMID: 8017333 10.1093/ajcn/60.1.23. [DOI] [PubMed] [Google Scholar]
- 39). Heo M, Faith MS, Pietrobelli A, Heymsfield SB. Percentage of body fat cutoffs by sex, age, and race-ethnicity in the US adult population from NHANES 1999–2004. Am J Clin Nutr. 2012. March; 95(3):594–602. PMID: 22301924 10.3945/ajcn.111.025171. [DOI] [PubMed] [Google Scholar]
- 40). Anstee QM, Day CP. The genetics of NAFLD. Nat Rev Gastroenterol Hepatol. 2013. November; 10(11):645–55. PMID: 24061205 10.1038/nrgastro.2013.182. [DOI] [PubMed] [Google Scholar]
- 41). Rinella ME, Sanyal AJ. NAFLD in 2014: genetics, diagnostics and therapeutic advances in NAFLD. Nat Rev Gastroenterol Hepatol. 2015. February; 12(2):65–6. PMID: 25560844. PMCID: PMC4984668 10.1038/nrgastro.2014.232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42). Anstee QM, Day CP. The genetics of nonalcoholic fatty liver disease: spotlight on PNPLA3 and TM6SF2. Semin Liver Dis. 2015. August; 35(3):270–90. PMID: 26378644 10.1055/s-0035-1562947. [DOI] [PubMed] [Google Scholar]
- 43). Dongiovanni P, Valenti L. Genetics of nonalcoholic fatty liver disease. Metabolism. 2016. August; 65(8):1026–37. PMID: 26409295 10.1016/j.metabol.2015.08.018. [DOI] [PubMed] [Google Scholar]
- 44). Reid AE. Nonalcoholic steatohepatitis. Gastroenterology. 2001. September; 121(3):710–23. PMID: 11522755 10.1053/gast.2001.27126. [DOI] [PubMed] [Google Scholar]
- 45). Adams LA, Lymp JF, Sauver JS, et al. The natural history of nonalcoholic fatty liver disease: a population-based cohort study. Gastroenterology. 2005. July; 129(1):113–21. PMID: 16012941 10.1053/j.gastro.2005.04.014. [DOI] [PubMed] [Google Scholar]
- 46). Ekstedt M, Franzén LE, Mathiesen UL, et al. Long-term follow-up of patients with NAFLD and elevated liver enzymes. Hepatology. 2006. October; 44(4):865–73. PMID: 17006923 10.1002/hep.21327. [DOI] [PubMed] [Google Scholar]
- 47). Ong JP, Pitts A, Younossi ZM. Increased overall mortality and liver-related mortality in non-alcoholic fatty liver disease. J Hepatol. 2008. October; 49(4):608–12. PMID: 18682312 10.1016/j.jhep.2008.06.018. [DOI] [PubMed] [Google Scholar]
- 48). Younossi ZM, Stepanova M, Rafiq N, et al. Pathologic criteria for nonalcoholic steatohepatitis: interprotocol agreement and ability to predict liver-related mortality. Hepatology. 2011. June; 53(6):1874–82. PMID: 21360720 10.1002/hep.24268. [DOI] [PubMed] [Google Scholar]
- 49). Stepanova M, Rafiq N, Makhlouf H, et al. Predictors of all-cause mortality and liver-related mortality in patients with non-alcoholic fatty liver disease (NAFLD). Dig Dis Sci. 2013. October; 58(10): 3017–23. PMID: 23775317 10.1007/s10620-013-2743-5. [DOI] [PubMed] [Google Scholar]
- 50). Ekstedt M, Hagström H, Nasr P, et al. Fibrosis stage is the strongest predictor for disease-specific mortality in NAFLD after up to 33 years of follow-up. Hepatology. 2015. August; 61(5):1547–54. PMID: 25125077 10.1002/hep.27368. [DOI] [PubMed] [Google Scholar]
- 51). Wanless IR, Lentz JS. Fatty liver hepatitis (steatohepatitis) and obesity: an autopsy study with analysis of risk factors. Hepatology. 1990. November; 12(5):1106–10. PMID: 2227807 10.1002/hep.1840120505. [DOI] [PubMed] [Google Scholar]
- 52). Zois CD, Baltayiannis GH, Bekiari A, et al. Steatosis and steatohepatitis in postmortem material from Northwestern Greece. World J Gastroenterol. 2010. August 21; 16(31):3944–9. PMID: 20712056. PMCID: PMC2923769 10.3748/wjg.v16.i31.3944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53). Lal A, Parai JL, Milroy CM. Liver pathology in first presentation diabetic ketoacidosis at autopsy. Acad Forensic Pathol. 2016. June; 6(2): 271–80. 10.23907/2016.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54). Randall B. Fatty liver and sudden death. A review. Hum Pathol. 1980. March; 11(2):147–53. PMID: 6105125 10.1016/s0046-8177(80)80133-x. [DOI] [PubMed] [Google Scholar]
- 55). Kramer K, Kuller L, Fisher R. The increasing mortality attributed to cirrhosis and fatty liver, in Baltimore (1957-1966). Ann Intern Med. 1968. August; 69(2):273–82. PMID: 5667768 10.7326/0003-4819-69-2-273. [DOI] [PubMed] [Google Scholar]
- 56). Clark JC. Sudden death in the chronic alcoholic. Forensic Sci Int. 1988. January; 36(1-2):105–11. PMID: 3338681 10.1016/0379-0738(88)90222-8. [DOI] [PubMed] [Google Scholar]
- 57). Milroy C. Sudden death and chronic alcoholism. Acad Forensic Pathol. 2014. June, 4(2):168–71. 10.23907/2014.027. [DOI] [Google Scholar]
- 58). Targher G, Valbusa F, Bonapace S, et al. Association of nonalcoholic fatty liver disease with QTc interval in patients with type 2 diabetes. Nutr Metab Cardiovasc Dis. 2014. June; 24(6):663–9. PMID: 24594085 10.1016/j.numecd.2014.01.005. [DOI] [PubMed] [Google Scholar]
- 59). Ballestri S, Lonardo A, Bonapace S, et al. Risk of cardiovascular, cardiac and arrhythmic complications in patients with non-alcoholic fatty liver disease. World J Gastroenterol. 2014. February 21; 20(7):1724–45. PMID: 24587651. PMCID: PMC3930972 10.3748/wjg.v20.i7.1724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60). Mantovani A, Rigamonti A, Bonapace S, et al. Nonalcoholic fatty liver disease is associated with ventricular arrhythmias in patients with type 2 diabetes referred for clinically indicated 24-hour Holter monitoring. Diabetes Care. 2016. August; 39(8):1416–23. PMID: 27222503 10.2337/dc16-0091. [DOI] [PubMed] [Google Scholar]
- 61). Mantovani A, Ballestri S, Lonardo A, Targher G. Cardiovascular disease and myocardial abnormalities in nonalcoholic fatty liver disease. Dig Dis Sci. 2016. August; 61(5):1246–67. PMID: 26809873 10.1007/s10620-016-4040-6. [DOI] [PubMed] [Google Scholar]
- 62). Byrne CD., Targher G. NAFLD: a multisystem disease. J Hepatol. 2015. April; 62(1 Suppl): S47–64. PMID: 25920090 10.1016/j.jhep.2014.12.012. [DOI] [PubMed] [Google Scholar]
- 63). Schwimmer JB, Deutsch R, Kahen T, et al. Prevalence of fatty liver in children and adolescents. Pediatrics. 2006. October; 118(4):1388–93. PMID: 17015527 10.1542/peds.2006-1212. [DOI] [PubMed] [Google Scholar]
- 64). Tominaga K, Kurata JH, Chen YK, et al. Prevalence of fatty liver in Japanese children and relationship to obesity. An epidemiological ultrasonographic survey. Dig Dis Sci. 1995. September; 40(9):2002–9. PMID: 7555456 10.1007/bf02208670. [DOI] [PubMed] [Google Scholar]
- 65). Schwimmer JB, Behling C, Newbury R, et al. Histopathology of pediatric nonalcoholic fatty liver disease. Hepatology. 2005. September; 42(3):641–9. PMID: 16116629 10.1002/hep.20842. [DOI] [PubMed] [Google Scholar]
- 66). Feldstein AE, Charatcharoenwitthaya P, Treeprasertsuk S, et al. The natural history of nonalcoholic fatty liver disease in children. a follow-up study for up to 20 years. Gut. 2009. November; 58(11):1538–44. PMID: 19625277. PMCID: PMC2792743 10.1136/gut.2008.171280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67). Howat AJ, Bennett MJ, Variend S, Shaw L. Deficiency of medium chain fatty acylcoenzyme A dehydrogenase presenting as the sudden infant death syndrome. Br Med J (Clin Res Ed). 1984. March 31; 288(6422):976 PMID: 6423169. PMCID: PMC1442514 10.1136/bmj.288.6422.976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68). Bonnell HJ, Beckwith JB. Fatty liver in sudden childhood death. Implications for Reye’s Syndrome? Am J Dis Child. 1986. January; 140(1):30–3. PMID: 3942104 10.1001/archpedi.1986.02140150032027. [DOI] [PubMed] [Google Scholar]
- 69). Berry PJ. Pathological findings in SIDS. J Clin Pathol. 1992. November; 45(11 Suppl):11–6. PMID: 1474151. [PubMed] [Google Scholar]