Summary
Aims
Our study aimed to figure out brain functional reorganization evidence after repetitive transcranial magnetic stimulation (rTMS) using the resting‐state functional magnetic resonance imaging (rsfMRI).
Methods
Twelve patients with unilateral subcortex lesion in the middle cerebral artery territory were recruited. Seven of them received a 10‐day rTMS treatment beginning at about 5 days after stroke onset. The remaining five received sham treatment. RsfMRI and motor functional scores were obtained before and after rTMS or sham rTMS.
Results
The rTMS group showed motor recovery according to the behavioral testing scores, while there was no significant difference of motor functional scores in the sham group before and after the sham rTMS. It proved that rTMS facilitates motor recovery of early ischemic stroke patients. Compared with the sham, the rTMS treatment group achieved increased functional connectivity (FC) between ipsilesional M1 and contralesional M1, supplementary motor area, bilateral thalamus, and contralesional postcentral gyrus. And decreased FC was found between ipsilesional M1 and ipsilesional M1, postcentral gyrus and inferior and middle frontal gyrus.
Conclusion
Increased or decreased FC detected by rsfMRI is an important finding to understand the mechanism of brain functional reorganization. The rTMS treatment is a promising therapeutic approach to facilitate motor rehabilitation for early stroke patients.
Keywords: Functional magnetic resonance imaging (fMRI), Ischemic stroke, Motor recovery, Repetitive transcranial magnetic stimulation (rTMS), Resting‐state fMRI (rsfMRI)
Introduction
Stroke, triggered by ischemia (lack of blood flow), blockage (thrombosis, arterial embolism), or hemorrhage, causes long‐term neurological impairment and functional disability 1, 2. It is also the second most common cause of death which follows behind the ischemic heart disease, causing 4.38 million of a total 50.5 million death each year 3. One‐half of the survivors of acute stroke are incompletely recovered, of whom twenty percent require assistance in at least one activity, making stroke the most important single cause of severe disability in people living in their own homes 4, 5. However, there are no known drug therapies to improve recovery after stroke 1. So it is desirable to develop available neurorehabilitative treatments.
Repetitive transcranial magnetic stimulation (rTMS), serving as a novel neuro‐modulated technique, is widely being used in treating depression, schizophrenia, and posttraumatic stress disorder 6, 7, 8. Its treating mechanism is to excite or inhibit brain cortex. As rTMS can modulate motor cortex, we hypothesized that it may play a positive role in facilitating motor recovery after ischemic stroke. However, to date, there are few published studies on this issue. So our study aimed to figure out the effectiveness of rTMS treatment in stroke patients with motor dysfunction.
Along with the development of multimodality imaging technique, functional magnetic resonance imaging (fMRI) such as blood oxygenation level dependent fMRI (BOLD‐fMRI), diffusion tensor imaging (DTI), perfusion, and magnetic resonance spectroscopy (MRS) are widely used to investigate brain functional changes of stroke patients 9, 10, 11, 12. Our study used the resting‐state fMRI (rsfMRI). It is based on the quantification of BOLD contrast. As we all know, oxygenated hemoglobin is diamagnetic whereas deoxygenated hemoglobin is paramagnetic. The MRI signal will change when the type of the hemoglobin alters, and this changed signal is detectable as the BOLD contrast. When some cerebral area is motivated, the increasing neural activity leads to more oxygen demand, as a result the deoxygenated hemoglobin increases first. Then, the cerebral blood flow rises to compensate for the increased oxygen consumption and ultimately delivers an over‐supply of oxygenated blood, leading to an increase in the BOLD response, which is the so‐called activation in fMRI studies 13. In this way, fMRI can reflect the hemodynamic changes within the brain. Therefore, it can provide us with a direct visualization of the rTMS‐induced cortical activation. The fMRI includes task‐fMRI and rsfMRI, and our study used the rsfMRI because it is much easier to carry out on patients with motor dysfunction after stroke.
In this study, we aimed to investigate cerebral functional changes induced by high‐frequency rTMS in ischemic stroke patients with motor dysfunction and therefore can figure out the effectiveness of rTMS treatment in motor rehabilitation after stroke.
Materials and methods
Subjects
All patients were scanned by routine MR sequences. Only stroke patients within 1 week after onset with unilateral cerebral subcortex lesion in the middle cerebral artery (MCA) territory detected by diffusion weighted image (DWI) were enrolled in our study. All the patients were right‐handed without memory loss or intelligence disorder, and none of them have ever suffered stroke before. The exclusion criteria included are as follows: (1) direct damage to the cerebral cortex, (2) a history of cerebral vessel disease, (3) tendency to hemorrhage or existed brain hemorrhage, (4) epilepsy or other mental disorders, and (5) any MRI contraindications.
Patients were divided into two groups randomly. They were scanned with rsfMRI prior to and after rTMS or sham rTMS. For behavioral testing, motor functional scores of the patients were assessed by an experienced neurologist. The scores included (1) National Institutes of Health Stroke Scale (NIHSS), (2) Barthel Index (BI), and (3) Fugl‐Meyer assessment (FMA) (Tables 1 and 2).
Table 1.
Motor functional assessment peri‐rTMS treatment
| Patient | Assessment time | NIHSS(0–15) | UE‐FMA(0–66) | LE‐FMA(0–34) | BI(0–100) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Pre‐ | Post‐ | Pre‐ | Post‐ | Pre‐ | Post‐ | Pre‐ | Post‐ | Pre‐ | Post‐ | |
| rTMS 1 | 5 | 33 | 6 | 2 | 53 | 60 | 30 | 34 | 60 | 95 |
| rTMS 2 | 1 | 30 | 6 | 2 | 33 | 50 | 26 | 34 | 70 | 90 |
| rTMS 3 | 3 | 28 | 6 | 4 | 52 | 60 | 28 | 34 | 60 | 80 |
| rTMS 4 | 3 | 34 | 6 | 3 | 40 | 50 | 26 | 32 | 60 | 85 |
| rTMS 5 | 2 | 31 | 6 | 4 | 34 | 45 | 28 | 30 | 65 | 85 |
| rTMS 6 | 5 | 33 | 9 | 5 | 38 | 48 | 24 | 28 | 75 | 90 |
| rTMS 7 | 5 | 32 | 11 | 3 | 34 | 48 | 20 | 26 | 55 | 85 |
| Mean ± SD | 3.4 ± 1.6 | 31.6 ± 2.1 | 7.1 ± 2.0 | 3.3 ± 1.1 | 40.6 ± 8.5 | 51.6 ± 6.0 | 26.0 ± 3.3 | 31.1 ± 3.2 | 63.6 ± 6.9 | 87.1 ± 4.9 |
Assessment time, the number of days from stroke onset; Pre‐, before the treatments; Post‐, after the treatments; UE‐FMA, Fugl‐Meyer assessment of upper extremity; LE‐FMA, Fugl‐Meyer assessment of lower extremity; NIHSS, National Institutes of Health Stroke Scale; BI, Barthel Index.
Table 2.
Motor functional assessment peri‐sham rTMS
| Patient | Assessment time | NIHSS(0–15) | UE‐FMA(0–66) | LE‐FMA(0–34) | BI(0–100) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Pre‐ | Post‐ | Pre‐ | Post‐ | Pre‐ | Post‐ | Pre‐ | Post‐ | Pre‐ | Post‐ | |
| Sham 1 | 3 | 31 | 7 | 3 | 50 | 54 | 25 | 30 | 60 | 90 |
| Sham 2 | 1 | 32 | 7 | 2 | 48 | 55 | 28 | 32 | 70 | 90 |
| Sham 3 | 2 | 31 | 9 | 5 | 32 | 48 | 20 | 28 | 50 | 70 |
| Sham 4 | 7 | 34 | 4 | 2 | 44 | 56 | 24 | 30 | 70 | 95 |
| Sham 5 | 1 | 30 | 6 | 4 | 50 | 60 | 26 | 30 | 65 | 80 |
| Mean ± SD | 2.8 ± 2.5 | 31.6 ± 1.5 | 6.6 ± 1.8 | 3.2 ± 1.3 | 44.8 ± 7.6 | 54.6 ± 4.3 | 24.6 ± 3.0 | 30.0 ± 1.4 | 63.0 ± 8.4 | 85.0 ± 10.0 |
Assessment time, the number of days from stroke onset; Pre‐, before the sham rTMS; Post‐, after the sham rTMS; UE‐FMA, Fugl‐Meyer assessment of upper extremity; LE‐FMA, Fugl‐Meyer assessment of lower extremity; NIHSS, National Institutes of Health Stroke Scale; BI, Barthel Index.
The study was approved by the ethics committee of the hospital, and full written consent was obtained from all participants.
rTMS
The rTMS treatments were performed using a Medtronic MagPro type magnetic stimulation device (Medtronic, Minneapolis, MN, USA) and a figure‐of‐eight coil (MC‐B70, Medtronic). The stimulation protocol involved 50 trains of 20 pulses applied over the ipsilesional M1 at a frequency of 5 HZ, with the stimulus intensity set at 120% of the resting motor threshold of the unaffected extremity. The rTMS began at averagely 5 days after stroke onset and lasted for 10 consecutive days. The sham group received sham rTMS.
Data Acquisition
MRI data including rsfMRI and structural images were performed on a 3.0 T MRI scanner (MAGNETOM Skyra; Siemens, Erlangen, Germany) using a twenty‐channel phased‐array head coil. RsfMRI were obtained via a gradient echo‐planar imaging sequence (EPI) with the following parameters: repetition time (TR) = 2510 ms, echo time (TE) = 30 ms, flip angle = 90°, slice thickness = 3 mm (no slice gap), matrix size = 80 × 80, field of view (FOV) = 240 × 240 mm2, and voxel size of 3 × 3×3 mm3. High‐resolution T1‐weighted structural images were also acquired for all participants using the following parameters: TR = 2300 ms, TE = 2.8 ms, time of inversion (TI) = 900 ms, flip angle = 8°, slice thickness = 1.0 mm, slice gap 0.5 mm, matrix size = 256 × 256, and field of view (FOV) = 256 mm × 256 mm. During the resting‐state scan, the participants were explicitly instructed to stay motionless, to close their eyes, and not to think of anything in particular nor to fall asleep. In addition, earplugs and earphones were used to reduce scan noises, and head motion was minimized by stabilizing the head with cushions.
Data Preprocessing
Image preprocessing was carried out using Data Processing Assistant for Resting‐State fMRI (DPARSF) 14. The first 10 time points of the fMRI time series were discarded for initial signal instability and participants’ adaption to the scan process. Slice timing and head motion correction were performed. T1‐weighted structural images were registered to their corresponding functional images and then segmented. Functional images were then normalized to the standard Montreal Neurological Institute (MNI) space, resampled to 3 × 3 × 3 mm3, and smoothed using a 4 mm full width at half maximum (FWMH) Gaussian kernel. The time series for each voxel was filtered (band pass 0.01–0.08 HZ) to remove the effects of very low‐frequency drift and high‐frequency noise. At last, the removal of nuisance covariates was performed.
Functional Connectivity (FC) Analysis
Functional Connectivity was examined using a seed‐based voxel‐wise correlation approach. The ipsilesional primary motor cortex (M1) was defined as the region of interest (ROI) (Figure 1) and was derived from the WFU PickAtlas in SPM8 (http://www.fil.ion.ucl.ac.uk/spm). Pearson's correlation analysis between the time course of the ipsilesional M1 and that of every voxel in the whole brain was computed for a map of correlation coefficients, which were Fisher's z‐transformed and called as z‐FC maps.
Figure 1.

The ipsilesional M1 was defined as the ROI in our study. (A) Axial view, (B) coronal view, and (C) sagittal view.
Statistical Analysis
Statistical analysis was performed using the Resting‐State fMRI Data Analysis Toolkit 15. The contrast between the z‐FC maps post‐ and pre‐rTMS treatment was calculated for each patient. The achieved differential contrast images (∆z‐FC maps) were then used for a two‐sample t‐test to compare the connectivity changes between the rTMS treatment group and the sham group. A cluster connectivity criterion of 5 mm (edge connected), spatial smoothness of 4 mm, and a threshold of P < 0.05 were achieved for the results of the two‐sample t‐tests, with a mask of the whole brain. Statistical parametric maps were overlaid on the MNI template. Increased FC and decreased FC of brain regions were projected onto a three‐dimensional ICBM152 smoothed‐brain using the BrainNet Viewer software 16. Clinical statistical analysis was calculated using the GraphPad Prism software. Wilcoxon signed‐rank tests were performed to compare the motor functional scores before and after the rTMS or sham rTMS in each group.
Results
Lesion Location
Twelve patients (10 males, two females; mean age: 55.3 years old; range: 30–76 years old) with unilateral subcortex lesion in the MCA territory detected by DWI (Figure 2) were enrolled in our study. Eight of the 12 patients had right hemispheric lesion and the other four patients’ activation maps were therefore flipped along the midsagittal plane so that the affected hemisphere corresponded to the right side of the brain images for all patients.
Figure 2.
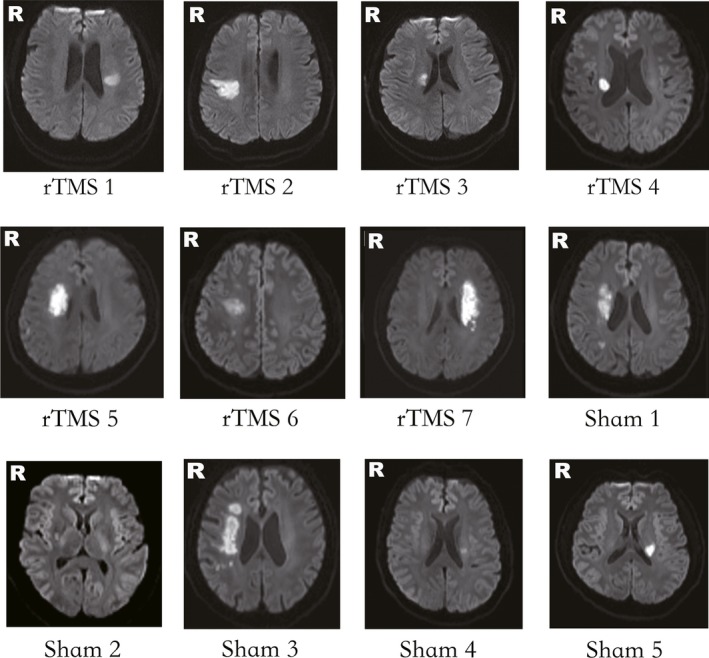
Lesion locations of stroke patients on DWI. rTMS 1–7 are patients of the rTMS treatment group. Sham 1–5 are patients of the sham group.
rTMS Treatment
Patients were assigned to the rTMS treatment group and the sham group randomly. Seven patients were included in the treatment group and received a 10‐day rTMS beginning at about 5 days after stroke onset. The rest five patients were provided with sham rTMS. There were no other significant differences between the two groups, such as the routine drug therapies and the rehabilitation training. None of the participants reported any adverse side effect during the course of the rTMS.
Motor Functional Scores
The NIHSS scores of the treatment group were significantly decreased (P < 0.05, Wilcoxon signed‐rank test) after the rTMS, while the BI and FMA scores remarkably increased (P < 0.05) (Table 1, Figure 3). However, for the sham group, there is no significant difference between the motor functional scores before and after the sham rTMS (P > 0.05, Wilcoxon signed‐rank test) (Table 2, Figure 4). The results demonstrated that rTMS treatment facilitated motor recovery after stroke.
Figure 3.

Comparison of the rTMS group's motor functional scores post‐ and pre‐rTMS treatment (Wilcoxon signed‐rank test, P < 0.05). Pre‐rTMS, before the rTMS treatment; Post‐rTMS, after the rTMS treatment; NIHSS, National Institute of Health Stroke Scale; UE‐FMA, Fugl‐Meyer assessment of upper extremity; LE‐FMA, Fugl‐Meyer assessment of lower extremity; BI, Barthel Index.
Figure 4.
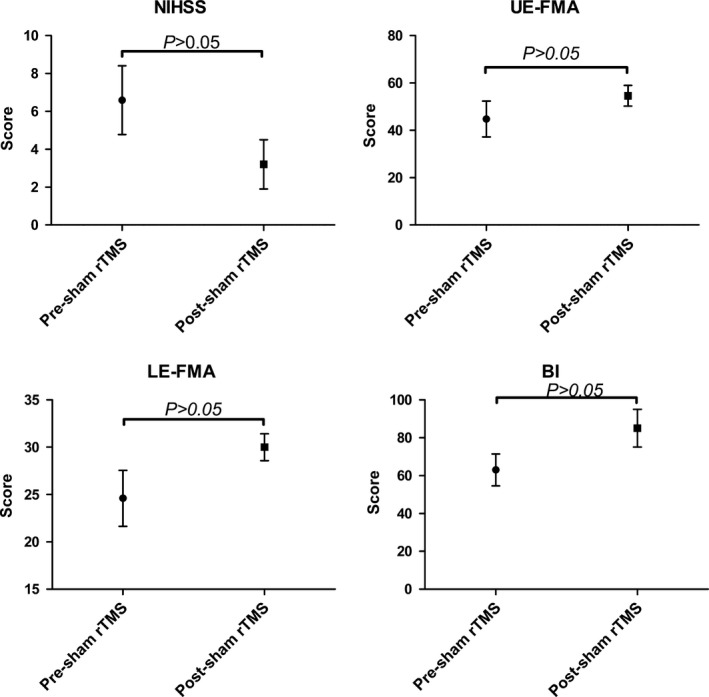
Comparison of the sham group's motor functional scores post‐ and pre‐ sham rTMS (Wilcoxon signed‐rank test, P < 0.05). Pre‐sham rTMS, before the sham rTMS; Post‐sham rTMS, after the sham rTMS; NIHSS, National Institute of Health Stroke Scale; UE‐FMA, Fugl‐Meyer assessment of upper extremity; LE‐FMA, Fugl‐Meyer assessment of lower extremity; BI, Barthel Index.
FC Analysis
Compared with the sham group, the rTMS treatment group showed significantly increased FC between the ipsilesional M1 and the contralesional M1, supplementary motor area (SMA), bilateral thalamus, contralesional postcentral gyrus, and superior temporal gyrus (two‐sample t‐test, P < 0.05, AlphaSim corrected, cluster size ≥ 85 voxels; Figure 5). Decreased FC of the ipsilesional M1 was demonstrated in the ipsilesional postcentral gyrus, M1, middle frontal gyrus, and superior parietal gyrus (Figure 5). These changed brain regions induced by the rTMS treatment were also projected onto a three‐dimensional ICBM152 smoothed‐brain using the BrainNet Viewer software (Figure 6).
Figure 5.
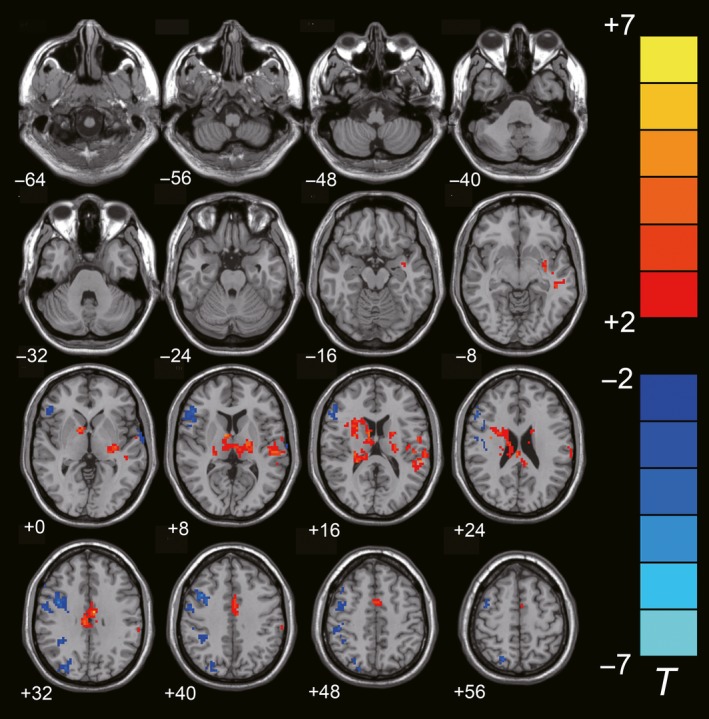
Differences of FC with the ipsilesional M1 between the rTMS treatment group and the sham group (two‐sample t‐test, P < 0.05, AlphaSim Corrected, cluster size ≥ 85 voxels). The warm‐toned areas represent the regions which have increased FC with the ipsilesional M1, while the cool‐toned ones represent the regions which have decreased FC with the ipsilesional M1. The left side of the images refers to the ipsilesional hemisphere.
Figure 6.
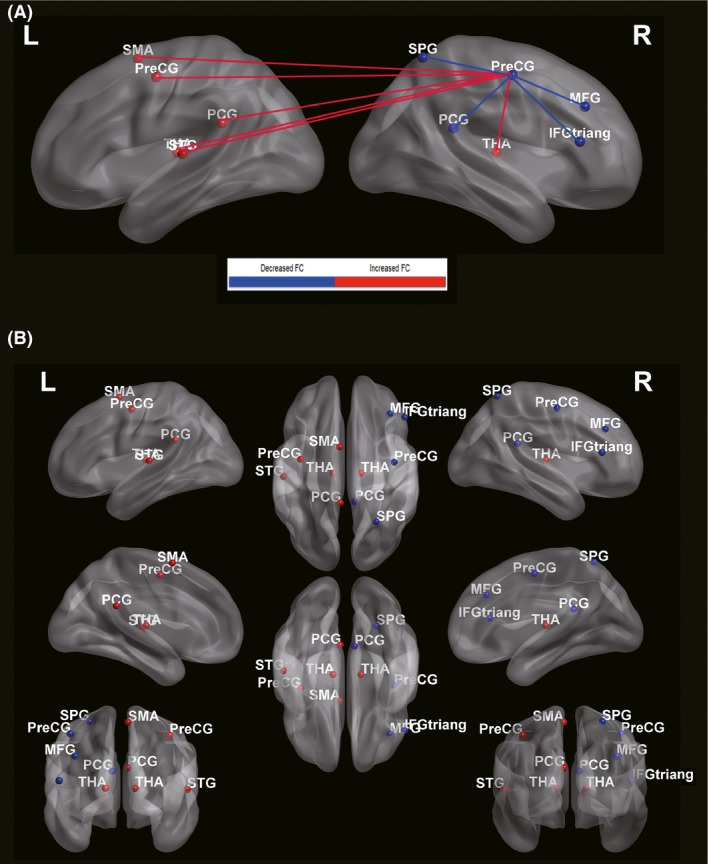
The increased and decreased FC of brain regions projected onto a three‐dimensional ICBM152 smoothed‐brain using the BrainNet Viewer software. (A): The red lines represent the increased FC, while the blue lines show the decreased ones. (B): The red spheres represent the cerebral regions that have increased FC with the ipsilesional M1, while the blue spheres show the decreased ones. The right hemisphere is corresponded to the ipsilesional hemisphere. Pre‐CG, Precentral gyrus; MFG, Middle frontal gyrus; IFGtriang, Inferior frontal gyrus, triangular part; SMA, Supplementary motor area; PCG, Postcentral gyrus; THA, Thalamus; STG, Superior temporal gyrus; SPG, Superior parietal gyrus; L, left hemisphere; R, right hemisphere.
Discussion
To the best of our knowledge, this is the first study using the method of rsfMRI to evaluate cerebral functional changes after rTMS in ischemic stroke patients. Our research is a longitudinal study instead of a cross‐sectional one. As we all know, stroke develops over a certain time course and motor recovery in stroke patients is a dynamic process 17, 18, 19. From this aspect, the longitudinal study is a better approach. Second, our research includes stroke patients in the acute stage only, which can minimize the bias caused by the course of the disease. Last but not the least, former study demonstrated that high‐frequency rTMS applied over ipsilesional M1 improved movement kinematics in most of the patients with subcortical stroke, but not in patients with additional cortical stroke 20. Our research only recruited subcortical stroke patients, which on one hand improved the effectivity of the rTMS treatment and on the other hand excluded the probable influence of the cerebral cortex to motor function.
The frequency of the rTMS Treatment in our Study
It is generally believed that low‐frequency (<1 HZ) rTMS tends to decrease the corticospinal excitability, while the high‐frequency (≥5 HZ) one is more likely to provoke a lasting facilitation 21, 22. For stroke patients, using high‐frequency rTMS to stimulate the ipsilesional hemisphere can enhance its function, thus resulting in motor improvements. Applying low‐frequency rTMS over the contralesional hemisphere will also work. Because the decreased activity of the contralesional hemisphere leads to the decreased transcallosal inhibition (TCI), which in consequence improves the ipsilesional hemispheric excitability. In this regard, both kinds of rTMS are suitable for our study. However, some previous studies showed that low‐frequency rTMS may deteriorate some motor function because of the reduced TCI, which plays a role in the bimanual movement 23. For this reason, we used high‐frequency rTMS in our study.
Motor Recovery of Stroke Patients after rTMS Treatment
According to the clinical scores in our research, the rTMS group's motor function improved after the treatment while the sham group did not show any significant improvement after the sham rTMS. The result indicates that the rTMS treatment can facilitate motor recovery of stroke patients. This result is also consistent with those of previous studies using either electromyography or movement assessment to evaluate corticomotor changes after rTMS for stroke patients 20, 21, 24. As we mentioned before, high‐frequency rTMS can facilitate corticospinal excitability, which could be approved by the increase of the mean peak amplitude of motor evoked potential. In our study, we applied 5 HZ rTMS over the ipsilesional M1 to promote its activity in motor function, thus facilitating motor recovery of stroke patients. Although the neural mechanism of the rTMS treatment is still unclear, it suggests that the rTMS‐induced inhibitory postsynapses on principal neurons may play a role 25. Our study observed the cerebral functional changes after rTMS using the rsfMRI method, which is noninvasive and convenient to carry out on stroke patients with motor dysfunction. The rsfMRI FC result in our study showed a few discrepant brain areas induced by the rTMS treatment. Some of these regions are closely related to the motor functional reorganization. From this perspective, the rTMS treatment is helpful in motor recovery of ischemic stroke patients.
Roles of the Activated Areas in Motor Recovery
In comparison with the sham group, the rTMS treatment group showed increased FC between bilateral M1. Former researches demonstrated that the activation of contralesional M1 was correlated with good motor recovery in stroke patients 26, 27. It reflects the compensatory reorganization of the contralesional homologous motor area after stroke. The rTMS in our study induced increased activation of the contralesional M1 compared with the sham, thus improving motor rehabilitation of patients in the rTMS treatment group.
The FC between the ipsilesional M1 and the contralesional SMA was increased in our study. SMA is a crucial compensatory area of motor function. It provides an anatomical substrate for motor recovery following the interruption of M1 output 28. SMA is divided into two parts: The rostral SMA plays a role in the movement preparation while the caudal SMA is more likely to be a motor executive area 29. Previous studies also revealed that the activation of SMA in the fMRI result is associated with the attention to intention, which is central to voluntary motor action 30, 31, 32. Compared with the sham group, the rTMS treatment group showed increased FC between the ipsilesional M1 and the contralesional SMA, which suggested that the SMA was constantly activated after rTMS. The increased connectivity between the ipsilesional M1 and the contralesional SMA in our study reflected that our patients in the rTMS treatment group were experiencing the procedure of motor recovery, which in some aspects proves the positive role of rTMS in rehabilitation after stroke.
An increased FC of the ipsilesional M1 and the bilateral thalamus was found in the rTMS treatment group compared with the sham. Thalamus is conceived to be the relay station of the sensorimotor pathway. The ventrolateral nucleus of thalamus receives fibers from the cerebellar dentate nucleus and the cerebellar fastigial nucleus and projects fibers to the cerebral premotor cortex. As an important part of the extrapyramidal system, it is closely related to motor coordination. That the bilateral thalamus was activated in our study may be interpreted as a potential compensatory mechanism of the extrapyramidal system after the stroke lesion interrupted the corticospinal tract. What is more, action observation is suggested to be a possible way of retraining motor function after stroke. In a longitudinal fMRI study, Dr. Brunner and his colleagues found that the thalamus's neural response in the action observation task was increased along with the recovery after stroke. This means the thalamus plays a possible role in action observation, which is helpful in retraining motor function, thus facilitating motor recovery 33.
We found an increased FC between the ipsilesional M1 and the contralesional postcentral gyrus in our research. This result was very enlightening and worth great attention. So we hypothesized that the postcentral gyrus plays a crucial role in the cerebral functional reorganization. It is well known that postcentral gyrus is the primary somatosensory cortical center and has neurons that receives messages of the fibers from spinal and bulbar motor neurons 34. Yin and his colleagues divided the stroke patients into two groups, namely the completely paralyzed hands (CPH) and partially paralyzed hands (PPH). They found reduced FC of the ipsilesional M1 with the contralesional postcentral gyrus in the CPH group compared with the PPH group, which demonstrated that the preservation of connectivity between the ipsilesional M1 and the contralesional postcentral gyrus implies good motor function 35. So the activation of the contralesional postcentral gyrus in our study is of great importance in motor functional reorganization. It may reflect a compensatory recruitment of similar cortical motor neurons, therefore facilitating motor recovery.
Our study investigated cerebral FC changes with the ipsilesional M1 of stroke patients. Areas of increased FC with the ROI are mainly in the contralesional hemisphere while those of decreased FC are in the ipsilesional cerebrum. It is generally believed that brain reorganization after stroke includes recruitment of perilesional tissue and compensatory involvement of mirror regions 36. The increased FC of ipsilesional M1 with contralesional motor‐related areas and decreased ones in the ipsilesional hemisphere showed that rTMS treatment facilitates motor recovery mainly through strengthening the positive role of contralesional mirror regions.
As to the decreased FC of the ipsilesional M1 with the frontal and parietal cortex in our study, it may be caused by the initial language and sensory difference between the rTMS treatment group and the sham group, which should be confirmed by other functional scores in further research.
Conclusion
According to the behavioral testing scores, rTMS can facilitate motor recovery after stroke. Our study also used the noninvasive rsfMRI to observe the cerebral functional changes after the rTMS treatment and found some significant brain regions which are closely related to motor functional reorganization. In conclusion, rTMS may be a crucial and safe rehabilitation method for stroke patients and has an important prospect of clinical application.
Conflict of Interest
The authors declare no conflict of interest.
Supporting information
Appendix S1. Our previous studies about stroke.
Acknowledgments
This work was supported by funds from the National Natural Science Foundation of China (81271545), The Scientific Research Foundation for the Returned Overseas Chinese Scholars, The Science and Technology Foundation for the Selected Returned Overseas Chinese Scholars, and The Youth Foundation of Peking Union Medical College Hospital (2010104).
The first three authors contributed equally to this work.
Contributor Information
Yu‐Zhou Guan, Email: guanyz001@163.com.
Wei‐Hong Zhang, Email: zhangweihong@pumch.cn.
References
- 1. Robert AA, Zamzami MM. Stroke in Saudi Arabia: A review of the recent literature. Pan Afr Med J 2014;17:14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Sims NR, Muyderman H. Mitochondria, oxidative metabolism and cell death in stroke. Biochim Biophys Acta 2010;1802:80–91. [DOI] [PubMed] [Google Scholar]
- 3. Murray CJ, Lopez AD. Mortality by cause for eight regions of the world: Global Burden of Disease Study. Lancet 1997;349:1269–1276. [DOI] [PubMed] [Google Scholar]
- 4. Bonita R, Solomon N, Broad JB. Prevalence of stroke and stroke‐related disability. Estimates from the Auckland stroke studies. Stroke 1997;28:1898–1902. [DOI] [PubMed] [Google Scholar]
- 5. Hankey GJ. Stroke: How large a public health problem, and how can the neurologist help? Arch Neurol 1999;56:748–754. [DOI] [PubMed] [Google Scholar]
- 6. Berlim MT, Van Den Eynde F. Repetitive transcranial magnetic stimulation over the dorsolateral prefrontal cortex for treating posttraumatic stress disorder: An exploratory meta‐analysis of randomized, double‐blind and sham‐controlled trials. Can J Psychiatry 2014;59:487–496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. McGirr A, Van den Eynde F, Tovar‐Perdomo S, Fleck MP, Berlim MT. Effectiveness and acceptability of accelerated repetitive transcranial magnetic stimulation (rTMS) for treatment‐resistant major depressive disorder: An open label trial. J Affect Disord 2015;173:216–220. [DOI] [PubMed] [Google Scholar]
- 8. Hasan A, Guse B, Cordes J, et al. Cognitive effects of high‐frequency rTMS in schizophrenia patients with predominant negative symptoms: Results from a multicenter randomized sham‐controlled trial. Schizophr Bull 2016;42:608–618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Zhang WH, Li X, Zhang JY, et al. Landmark‐referenced voxel‐based analysis of diffusion tensor images of the brainstem white matter tracts: Application in patients with middle cerebral artery stroke. NeuroImage 2009;44:906–913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Globas C, Lam JM, Zhang WH, et al. Mesencephalic corticospinal atrophy predicts baseline deficit but not response to unilateral or bilateral arm training in chronic stroke. Neurorehabil Neural Repair 2011;25:81–87. [DOI] [PubMed] [Google Scholar]
- 11. Zhang S, Tang H, Yu YN, Yan SQ, Parsons MW, Lou M. Optimal magnetic resonance perfusion thresholds identifying ischemic penumbra and infarct core: A Chinese population‐based study. CNS Neurosci Ther 2015;21:289–295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Craciunas SC, Brooks WM, Nudo RJ, et al. Motor and premotor cortices in subcortical stroke: Proton magnetic resonance spectroscopy measures and arm motor impairment. Neurorehabil Neural Repair 2013;27:411–420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Stagg CJ, O'Shea J, Johansen‐Berg H. Imaging the effects of rTMS‐induced cortical plasticity. Restor Neurol Neurosci 2010;28:425–436. [DOI] [PubMed] [Google Scholar]
- 14. Yan CG, Zang YF. DPARSF: A MATLAB toolbox for “Pipeline” data analysis of resting‐state fMRI. Front Syst Neurosci 2010;4:13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Song XW, Dong ZY, Long XY, et al. REST: A toolkit for resting‐state functional magnetic resonance imaging data processing. PLoS ONE 2011;6:e25031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Xia MR, Wang J, He Y. BrainNet Viewer: A network visualization tool for human brain connectomics. PLoS ONE 2013;8:e68910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Park CH, Chang WH, Ohn SH, et al. Longitudinal changes of resting‐state functional connectivity during motor recovery after stroke. Stroke 2011;42:1357–1362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Wang L, Yu CS, Chen H, et al. Dynamic functional reorganization of the motor execution network after stroke. Brain 2010;133:1224–1238. [DOI] [PubMed] [Google Scholar]
- 19. Feydy A, Carlier R, Roby‐Brami A, et al. Longitudinal study of motor recovery after stroke: Recruitment and focusing of brain activation. Stroke 2002;33:1610–1617. [DOI] [PubMed] [Google Scholar]
- 20. Ameli M, Grefkes C, Kemper F, et al. Differential effects of high‐frequency repetitive transcranial magnetic stimulation over ipsilesional primary motor cortex in cortical and subcortical middle cerebral artery stroke. Ann Neurol 2009;66:298–309. [DOI] [PubMed] [Google Scholar]
- 21. Kim YH, You SH, Ko MH, et al. Repetitive transcranial magnetic stimulation‐induced corticomotor excitability and associated motor skill acquisition in chronic stroke. Stroke 2006;37:1471–1476. [DOI] [PubMed] [Google Scholar]
- 22. Peinemann A, Reimer B, Loer C, et al. Long‐lasting increase in corticospinal excitability after 1800 pulses of subthreshold 5 Hz repetitive TMS to the primary motor cortex. Clin Neurophysiol 2004;115:1519–1526. [DOI] [PubMed] [Google Scholar]
- 23. Stanćák A, Cohen ER, Seidler RD, Duong TQ, Kim SG. The size of corpus callosum correlates with functional activation of medial motor cortical areas in bimanual and unimanual movements. Cereb Cortex 2003;13:475–485. [DOI] [PubMed] [Google Scholar]
- 24. Khedr EM, Ahmed MA, Fathy N, Rothwell JC. Therapeutic trial of repetitive transcranial magnetic stimulation after acute ischemic stroke. Neurology 2005;65:466–468. [DOI] [PubMed] [Google Scholar]
- 25. Lenz M, Galanis C, Müller‐Dahlhaus F, et al. Repetitive magnetic stimulation induces plasticity of inhibitory synapses. Nat Commun 2016;7:10020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Bütefisch CM, Kleiser R, Körber B, et al. Recruitment of contralesional motor cortex in stroke patients with recovery of hand function. Neurology 2005;64:1067–1069. [DOI] [PubMed] [Google Scholar]
- 27. Schaechter JD, Perdue KL. Enhanced cortical activation in the contralesional hemisphere of chronic stroke patients in response to motor skill challenge. Cereb Cortex 2008;18:638–647. [DOI] [PubMed] [Google Scholar]
- 28. Tombari D, Loubinoux I, Pariente J, et al. A longitudinal fMRI study: In recovering and then in clinically stable sub‐cortical stroke patients. NeuroImage 2004;23:827–839. [DOI] [PubMed] [Google Scholar]
- 29. Jenkins IH, Jahanshahi M, Jueptner M, Passingham RE, Brooks DJ. Self‐initiated versus externally triggered movements II. The effect of movement predictability on regional cerebral blood flow. Brain 2000;123:1216–1288. [DOI] [PubMed] [Google Scholar]
- 30. Fried I, Katz A, McCarthy G, et al. Functional organization of human supplementary motor cortex studied by electrical stimulation. J Neurosci 1991;11:3656–3666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Desmurget M, Sirigu A. A parietal‐premotor network for movement intention and motor awareness. Trends Cogn Sci 2009;13:411–419. [DOI] [PubMed] [Google Scholar]
- 32. Haggard P. Neuroscience. The sources of human volition. Science 2009;324:731–733. [DOI] [PubMed] [Google Scholar]
- 33. Brunner IC, Skouen JS, Ersland L, Grüner R. Plasticity and response to action observation: A longitudinal FMRI study of potential mirror neurons in patients with subacute stroke. Neurorehabil Neural Repair 2014;28:874–884. [DOI] [PubMed] [Google Scholar]
- 34. Li XG, Florence SL, Kaas JH. Areal distributions of cortical neurons projecting to different levels of the caudal brain stem and spinal cord in rats. Somatosens Mot Res 1990;7:315–335. [DOI] [PubMed] [Google Scholar]
- 35. Yin D, Song F, Xu D, et al. Patterns in cortical connectivity for determining outcomes in hand function after subcortical stroke. PLoS ONE 2012;7:e52727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Saur D, Lange R, Baumgaertner A, et al. Dynamics of language reorganization after stroke. Brain 2006;129:1371–1384. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Appendix S1. Our previous studies about stroke.


