Summary
Background
Recent studies revealed that baicalin, a flavonoid compound derived from the root of Scutellaria baicalensis Georgi, could promote neuron differentiation of NSPCs after commencing the differentiation process in vitro. However, this may not be the most efficacious strategy to determinate cell fate. Here, we have investigated whether baicalin can influence early events of neuron generation and stimulate adult neurogenesis.
Results
Transient exposure of NSPCs to baicalin during proliferation could activate Mash1 to alter the differential fate and increase the proportion of cells expressing neuronal markers. Seven days after, rats were exposed to transient cerebral ischemia, they were treated for 3 weeks with baicalin, BrdU labeling study showed that exposure to baicalin increased the number of newly generated cells in hippocampus, BrdU/NeuN double staining analysis indicated that baicalin could promote new neuron production after cerebral ischemia. Additionally, Morris water maze test showed that delayed postischemic treatment with baicalin improved cognitive impairment.
Conclusions
These results identify the existence of a single molecule, baicalin, which can specify the neuronal fate of multipotent NSPCs and stimulate neurogenesis, making it a promising candidate for developing clinically relevant strategies to manipulate neuronal fate of NSPCs for brain repair.
Keywords: Balcalin, Cognitive functions., Ischemic stroke, Neural stem/progenitor cells, Neurogenesis, Neuronal fate
Introduction
Neural stem/progenitor cells (NSPCs) are excellent candidates for developing therapeutic strategies to repair the injured central nervous system (CNS) 1. The neurons in the hippocampus would be injured following with the proliferation of NSPCs after cerebral ischemia. However, a current limitation in this strategy is that the majority of NSPCs tend to differentiate into astrocytes, which are thought to be undesirable as they can inhibit neurite outgrowth and contribute to regenerative failure when a glial scar is formed 2. The ability to predetermine the neuronal fate of NSPCs would be beneficial to NSPCs therapy. Inducing a stable and predictable program of neural cell fate in NSPCs is an important goal for utilizing these cells for the treatment of neurological disorder. Recent studies have demonstrated that local signaling factors specify cell fate 3, 4, 5. And Åkerblom et al. 6 reported that miR‐124 is a neuronal fate determinant in the subventricular zone. These findings indicated that the ability to direct NSPCs cell fate may have therapeutic potential.
NSPCs fate can be determined by intrinsic signals and affected by small molecules 7, 8, 9, 10. The main mechanisms of the neuronal fate specification would be regulating the proneural genes. Genetic studies have provided evidence that proneural genes are necessary to restrict the neuronal cell fate of NSPCs 11. Neurogenin and Mash are bHLH transcription factors, which initiate neurogenesis 12, 13, 14. These findings indicate that bHLH transcription factors are the key points in the fate specification of NSPCs. Small molecules have been shown to be useful chemical materials in promoting neurogenesis and manipulating cell fate 9, 15. A variety of priming agents used to direct neuronal fate in NSPCs, such as lithium chloride 16, fibroblast growth factor‐2 17, and insulin‐like growth factor 1 18 had been identified.
In the search for new therapeutics of brain injury or neurodegenerative disease, the herbs being used in traditional medicines for neurogenesis are promising candidates. Baicalin (Figure 1A) is one of the predominant flavonoid derivatives isolated from the dry roots of Scutellaria baicalensis Georgi, which has protective effects against cerebral ischemia, brain inflammation, and other brain injuries 19, 20, 21. Our previous studies have showed that baicalin treatment could induce neuron differentiation of NSPCs after its commencing the differentiation process 22, 23, 24, which has been confirmed by a latest study 25. However, this may not be the most efficacious strategy to determinate cell fate. As emphasized by Vazey and Connor 16, alternative avenue for directing fate may be to pretreat NSPCs while they maintain their undifferentiated phenotype. Although a variety of active ingredients of herbs, such as ginsenosides 26, 27, 28, curcumin 29, ferulic acid 30, salvianolic acids 31, 32, could affect the biological properties of NSPCs. However, due to the presence of blood–brain barrier, some of the effective drugs in vitro cannot produce efficacy in vivo. Interestingly, some researchers had demonstrated that baicalin could pass through the blood–brain barrier 33, making it more likely to promote neurogenesis in the central nervous system.
Figure 1.
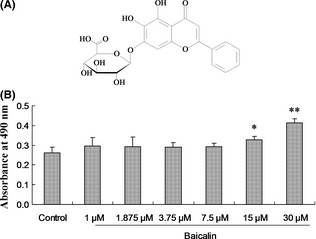
Effect of baicalin on the cell viability of NSPCs. Dissociated NSC/NPCs were cultured on a 96‐well plate with the indicated baicalin (A) concentration (1, 1.875, 3.75, 7.5, 15, 30 μM) for 2 days. (B) Cell proliferation was measured using a MTS assay kit. Values were expressed as means ± SD of three individual experiments. n = 6, *P < 0.05, **P < 0.01 compared with control.
In the present study, we demonstrated that after transient exposure to baicalin during proliferation could increase the new neurons production and improve the cognitive functions in a rat model of stroke. The neuronal fate determination and neurogenesis stimulating effects of baicalin makes it a promising candidate for developing clinically relevant strategies to stimulate NSPCs for brain repair.
Materials and Methods
Animals
Pregnant rats and Adult male Wistar rats (Certificate No SCXX 2006‐0009; Vital River Lab Animal Technology Co., Ltd., Beijing, China) were used in this study. The rats were housed in temperature‐controlled conditions with a 12‐h light/dark cycle following surgery. They had access to food and water ad libitum. All experimental procedures were evaluated and approved by the animal ethics committee of Tianjin University of Traditional Chinese Medicine (TCM‐2009‐034‐E02), and the animal care was according to the NIH Guide for the Care and Use of Laboratory Animals.
Neurosphere Culture and Cell Viability Assay
NSPCs were isolated from the cerebral cortex of 13.5‐day‐embryonic Wistar rats as previously described 34. NSPCs were cultured in neurosphere proliferation media consisting of DMEM/F12 supplemented with 2% (v/v) B27 Supplement (Invitrogen, Carlsbad, CA, USA), 20 ng/mL EGF (Millipore, Temecula, CA, USA), 20 ng/mL FGF2 (Millipore), and 100 μg/mL heparin (Sigma, St. Louis, MO, USA). For the in vitro cell viability assay, Cell Titer 96® AQueous One Solution Cell Proliferation Assay kit (Promega, Charbonnières‐les‐Bains, France) was used in this study. NSPCs were plated at 30,000 cells/well in a 96‐well plate in the presence and absence of baicalin (1, 1.875, 3.75, 7.5, 15, 30 μM). Cell proliferation was assessed at day 2 of cell culture. According to the manufacturer's recommendations, 40‐μL MTS solution was then added into each of the wells, and data were obtained at a wavelength of 490 nm using a microplate reader (FlexStation 3; Molecular Devices, Sunnyvale, CA, USA). The same volume of medium without cells was used as blank.
RT‐PCR Analysis
NSPCs were cultured with or without baicalin (7.5, 15, 30 μM) in proliferation medium for 3 days. Cells were harvested and total RNA was isolated from treated and untreated NSPCs with TRIzol (Roche, Mannheim, Germany). The first strand of cDNA was synthesized from 1 μg of total RNA using reverse transcriptase (TIANGEN) and random primer as described in the manufacture's instructions. After synthesis, 1 μL of cDNA were used in PCR reaction with gene‐specific primers. The sequences of the PCR primer pairs (5′ to 3′) that were used for each gene are as follows: β‐tubulin III, 5′‐GGACCTCAACCACCTTGTGT‐3′ (forward), and 5′‐AACATGGCCGTAAACTGCTC‐3′ (reverse); GFAP, 5′‐ATTCCGCGCCTCTCCCTGTCTC‐3′ (forward), 5′‐GCTTCATCCGCCTCCTGTCTGT‐3′ (reverse); GAPDH, 5′‐CCAAGGTCATCCATGACAA‐3′ (forward), and 5′‐TGTCATACCAGGAAATGAGC‐3′ (reverse); RT‐ve control were seted for RT‐PCRs analysis (RT‐ve control = reaction with H2O instead of RNA as control). Products were analyzed on 1.5% agarose gel and visualized by ethidium bromide staining. The relative quantitative expressions of tissue‐specific markers were calculated after normalization with GAPDH as a housekeeping gene.
Real‐time RT‐PCR Analysis
Real‐time RT‐PCR was performed in a reaction mixture containing Power SYBR green PCR master mix (Applied Biosystems, Carlsbad, CA, USA), gene‐specific primers (Mash1, 5′‐CTCGTCCTCTCCGGAACTGATG‐3′ (forward), and 5′‐ATGCTCCCGGAGGGTGGCAAAA‐3′ (reverse); Ngn1, 5′‐ATGCCTGCCCCTTTGGAGACCT‐3′ (forward), and 5′‐TGTAGCCTGGCACAGTCCTCCT‐3′ (reverse); Ngn2, 5′‐CGGGTCAGACGTGGACTACT‐3′ (forward), and 5′‐GGCGGGAGAAGGATGGGAAGA‐3′ (reverse). Quantitative real‐time PCR reactions were run in a 7500 Fast real‐time PCR system (Applied Biosystems) with 7500 Fast System Sequencing detection software version 1.3.1 and the thermal profile of 95°C for 10 min followed by 40 cycles of 95°C for 15 second and 60°C for 1 min. The expression level of each gene was enumerated in three independent treatments. The relative gene expression between baicalin treatment and control was calculated by the ΔΔCt method with GAPDH as the internal control.
In Vitro Differentiation Assay
For the in vitro differentiation assay, NSPCs were cultured with or without baicalin (7.5, 15, 30 μM; Baicalin > 98.5% was purchased from Tianjin Zhongxin Pharmaceutical Group Co., Ltd., Tianjin, China) in proliferation medium for 3 days. Then, neurospheres were collected and plated into poly‐l‐lysine‐coated 12‐well plates in differentiation culture medium without growth factors or baicalin. Neurospheres were allowed to differentiate for 3 days before being fixed for immunocytochemistry. For fluorescence immunostaining, cells were fixed with 4% paraformaldehyde for 30 min. After fixing, cells were incubated overnight at 4°C with the following primary antibody: Rabbit anti‐MAP‐2 (1:500; Millipore), Mouse anti‐GFAP (1:500; Millipore), followed by FITC‐conjugated goat anti‐mouse IgG (1:100; Boster, Wuhan, China), and Cy3‐conjugated goat anti‐rabbit IgG (1:100; Boster). 4′,6‐Diamino‐2‐phenylindole (DAPI; Sigma) was used as a fluorescent nuclear counterstain. Immunostained cells were visualized by indirect fluorescence under the fluorescent microscope (Leica, Wetzlar, Germany). MAP‐2 or GFAP‐positive cells were counted in 10 randomly selected fields from three different chambers.
Western Blotting Assay
Protein extracts were prepared and subjected to Western blot analysis. The protein concentrations of the lysates were determined with a Bradford protein assay kit (Bio‐Rad, Hercules, CA, USA) according to the manufacturer's instructions. An equal amount of protein was fractionated by SDS‐polyacrylamide gel electrophoresis (PAGE) and transferred onto polyvinylidine difluoride membranes. After blocking with 5% skim milk in TBS‐T, the membrane was probed with goat anti‐actin (1:2000; Santa Cruz, CA, USA); mouse anti‐mash1 (1:1000; Millipore) primary antibody in a blocking solution of nonfat milk (5%). Secondary horseradish peroxidase‐conjugated goat anti‐mouse, goat anti‐rabbit (Boster) antibody in nonfat milk blocking solution (5%) was then applied. The immunoreactivity was visualized with ECL Western blotting detection reagents (Millipore). The total protein content was normalized using mouse anti‐actin antibodies.
Induction of the Transient Global Ischemia
Adult male Wistar rats weighing 200–250 g were subjected to transient forebrain ischemia by a method combining those described previously 35, 36, 37. In brief, 12 h before the induction of ischemia, rats were anesthetized with sodium pentobarbital, the bilateral carotid arteries were exposed to facilitate the occluding on the following day, and then the vertebral arteries were irreversibly occluded by electrocoagulation. The next day, ischemia was induced by bilaterally occluding carotid arteries with aneurysm clips, and carotid arteries were clamped for 6 min exactly. Rats lost their righting reflex during ischemic, the clips were removed to restore cerebral blood flow. The rats that remain their righting reflex in 1 min after occluding of the both carotid arteries were considered to be the failure of ischemia and eliminated. Sham‐operated rats were anesthetized, the carotid arteries were isolated, but they were not clamped.
Drug Administration
The study was carried out on rats divided into three groups. (1) Sham group + vehicle (sham), (2) Ischemic group + vehicle (vehicle), and (3) Ischemic group + baicalin (Baicalin). Baicalin was diluted in saline at the concentration of 100 mg/mL (Stock solution) and incubated under agitation for 1 h at 37°C. The solution was then filtered with 0.22‐mm filter. Baicalin were administered intraperitoneally (i.p.) at a dose of 50 mg/kg body weight, once daily for 3 weeks. All controls received an amount of vehicle equivalent to drug treatment conditions.
BrdU Labeling and Analysis of In Vivo Neurogenesis
BrdU (50 mg/kg; Sigma) was injected i.p. to the rats once every 2 h over a period of 8 h at the 20th day after baicalin treatment to label the proliferating cells after ischemia. The next day (21th), rats were anesthetized with ether and perfused with PBS, followed by a cold 4% paraformaldehyde solution. Brains were collected and postfixed overnight in a 4% paraformaldehyde solution at 4°C. Coronal sections (20 μm thickness) were obtained throughout the hippocampus. For analysis concerning BrdU or BrdU/NeuN double stain immunohistochemistry, sections were incubated in 50% formamide/2 × saline sodium citrate for 2 h at 65°C, followed by a rinse with PBS. Sections were then incubated in 2 N HCl for 30 min at 37°C to denature double‐stranded DNA, and rinsed in 0.1 M borate buffer (pH 8.5). After blocking for 2 h with 1% BSA in PBS, sections were incubated overnight at 4°C with mouse anti‐BrdU monoclonal antibody (1:100; senta), or rabbit anti‐NeuN polyclonal antibody (1:100; Millipore). Followed by rinsing in PBS, sections were incubated for 2 h at RT with FITC‐conjugated goat anti‐mouse IgG (1:100; Boster) and Cy3‐conjugated goat anti‐rabbit IgG (1:100; Boster). BrdU or BrdU/NeuN double‐positive cells were visualized under the fluorescent microscope (Lica, Germany).
Morris Water Maze Task
The cognitive functions were examined using the Morris water maze. A black circular pool 1.5 m in diameter was filled with water (22 ± 1°C), and a hidden platform 10 cm in diameter was placed at a constant position in the center of one of the four quadrants within the tank. Rats in each groups (n = 10) were allowed to swim freely for 2 min to become habituated to the apparatus. From the next day, in the hidden platform trials, acquisition trials were carried out two times per day for 5 days. In each trial, rats were placed into the water at a fixed starting position, and the time taken to escape onto the hidden platform was measured. Rats were given 90 second to find the hidden platform during each acquisition trial. If it failed to locate the platform within 90 second, it was guided there, and the rat was allowed to stay on the platform for 20 second. The latency to finding the escape platform was analyzed as an index of spatial learning. Performance was tested 24 h after the final training day in a probe trial during which the platform was removed, the rat was placed in the start and its behavior was monitored for 90 second.
Data Analysis
Data were expressed as mean ± SD. Statistical comparison between different treatments was carried out by one‐way analysis of variance, and then post hoc least significant difference (LSD) test was employed for comparisons between control and other groups. To analyze water maze place‐navigation performance, the average escape latency was calculated and evaluated by repeated‐measures ANCOVA. Differences were considered statistically significant for P < 0.05.
Results
Baicalin Upregulated Neuronal Genes Expression
To investigate the effect of baicalin on the viability of NSPCs, NSPCs were exposed to baicalin for 2 days. The effect of baicalin treatments on the growth rates of the NSPCs was compared using a cell viability measurement. As demonstrated in Figure 1B, baicalin had a promoting activity for NSPCs proliferation and exhibited a dose‐dependent effect. Based on the cell viability study, the doses of 7.5, 15, 30 μM were chosed in the following study. Mash1 and Ngn1 are thought to positively regulate neuronal development 38, 39. The expression of proneural transcription factors (Mash1, Ngn1 and Ngn2) in the proliferating NSPCs in the presence and absence of baicalin was measured. Changes in gene expression in the NSPCs treated and untreated with baicalin were evaluate by real‐time RT‐PCR technique. In our study, the results showed that Mash1 mRNA increased after baicalin treatment (Control 0.50 ± 0.10% vs. 15 μM baicalin 1.22 ± 0.38%, 30 μM baicalin 1.54 ± 0.09, n = 6, P < 0.05, P < 0.01, respectively; Figure 2A). To provide further evidence that baicalin upregulated mash1 expression, Mash1 protein was detected by Western blotting assay with anti‐mash1 antibody. As shown in Figure 2B, baicalin significantly increased mash1 expression compared with untreated control (Control 0.34 ± 0.08 vs. 7.5 μM baicalin 0.83 ± 0.11, 15 μM baicalin 0.82 ± 0.13, 30 μM baicalin 0.87 ± 0.10, n = 6, P < 0.01). These results indicated that baicalin stimulates neuronal differentiation by activating bHLH transcription factors.
Figure 2.
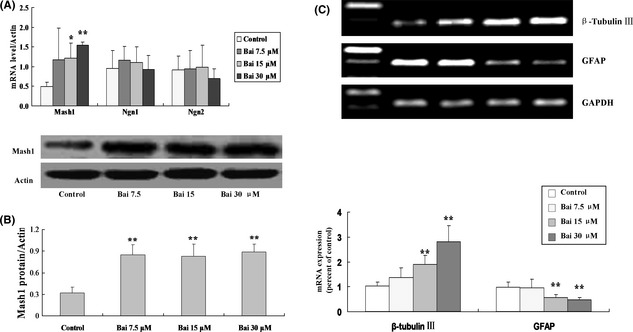
Effect of baicalin on the bHLH transcription factors expression and the neural fate of NSPCs. The mRNA or protein levels of Mash1 treated with designated concentrations (7.5, 15, 30 μM) of baicalin analyzed by real‐time RT‐PCR or Western blotting. (A) The mRNA levels quantified were calculated by the ΔΔCt method, normalized with GAPDH internal control. Mash1 expression were significantly upregulated in baicalin‐treated NSPCs compared with control (n = 6); (B) Semiquantified scores of Mash1 expression were significantly upregulated in baicalin‐treated NSPCs compared with control, normalized with Actin internal control (n = 6); (C) The mRNA levels of β‐tubulin III, GFAP treated with designated concentrations (7.5, 15, 30 μM) of baicalin for 3 days were analyzed by RT‐PCR. The mRNA levels were semiquantified by densitometric measurements, normalized with GAPDH internal control (n = 6), and expressed as means ± SD of three individual experiments. Values were expressed as means ± SD of three individual experiments. *P < 0.05, **P < 0.01 compared with control.
Baicalin Specified the Neural Fate of NSPCs
Given the induction of bHLH factors by baicalin, in the following research we investigated whether baicalin treatment during proliferation would increase neuronal fate determination. Cells were subjected to baicalin treatment for 3 days under proliferative conditions and the ability of their neuronal fate commitment was compared by RT‐PCR technique, using specific neuronal markers. The expressions of β‐tubulin III (neuron marker) and GFAP (astroglia marker) were measured by RT‐PCR technique. Results showed that no bands were detected in RT‐ve control samples, and after 3 days of baicalin treatment, NSPCs significantly decreased the mRNA expressions of GFAP (Control 0.98 ± 0.21 vs. 15 μM baicalin 0.57 ± 0.12, 30 μM baicalin 0.48 ± 0.09, n = 6, P < 0.01), and baicalin increased the expression of β‐tubulin III (Control 1.02 ± 0.16 vs. 15 μM baicalin 1.89 ± 0.37, 30 μM baicalin 2.80 ± 0.64, n = 6, P < 0.01; Figure 2C).
Immunostaining was also employed to evaluate the ability of neuronal fate determination of baicalin. NSPCs were subjected to baicalin treatment under proliferative conditions for 3 days, after that we withdrew the baicalin, and made the NSPCs adherent to differentiate without baicalin for 3 days. Based on the differentiation assay, we also found that, along with elevated proneural gene expression, the fraction of MAP‐2‐(neuron marker) positive cells was also highly upregulated compared with control (Figure 3A). Our immunocytochemistry approach identified the percentage of MAP‐2‐positive cells with more than twofold changes between baicalin (30 μM) exposure and control (Control 11.8 ± 3.4% vs. baicalin 24.9 ± 4.3%, n = 8, P < 0.01; Figure 3B). There was no significant difference in the fraction of GFAP‐positive cells (Control 71.8 ± 10.5% vs. 7.5 μM baicalin 75.6 ± 8.5%, 15 μM baicalin 73.3 ± 14.6%, 30 μM baicalin 79.0 ± 10.6%, n = 8, P > 0.05; Figure 3C). These results suggested that exposed to baicalin during proliferation was sufficient to specify the neuronal fate of NSPCs.
Figure 3.
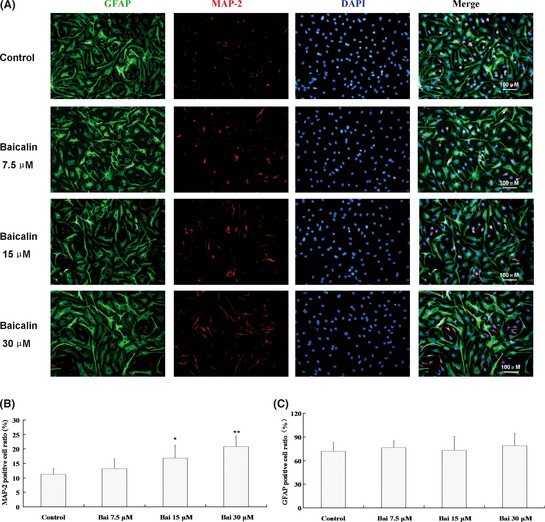
Baicalin primed toward a neuronal fate of NSPCs during their subsequent differentiation. (A) Fluorescence microscopic photos showing the neural differention of NSPCs treated with designated concentrations (7.5, 15, 30 μM) of baicalin for 3 days in proliferation medium. Red, MAP‐2; Green, GFAP; blue, DAPI. Scale bar, 100 μm. (B) Baicalin significantly increased the fraction of MAP‐2‐positive cells. (C) There was no significant difference in the fraction of GFAP‐positive cells. The data expressed as means ± SD of three individual experiments. n = 8, *P < 0.05, **P < 0.01 compared with control.
Baicalin Increased the Number of Newly Generated Cells in the Dentate Gyrus
To elucidate the possible effects of baicalin on adult NSPCs of the hippocampus, in the following study, we employed experimental cerebral ischemic rats. Newly generated cells in hippocampus dentate gyrus after transient cerebral ischemia were evaluated in the absence or presence of baicalin. The dividing cells labeled with BrdU were visualized by BrdU immunohistochemistry. As showed in Figure 4, a few BrdU‐positive cells were observed in the SGZ of hippocampal dentate gyrus in the sham, and BrdU immunoreactivity did not significantly differ between vehicle and sham‐operated animals (Sham 10.4 ± 3.0 vs. Vehicle 12.9 ± 6.3, n = 3, P > 0.05). The numbers of BrdU‐positive cells in the dentate gyrus were significantly greater in baicalin‐treated rats compared with the rats in vehicle (Vehicle 12.9 ± 6.3 vs. Baicalin 23.8 ± 4.3, n = 3, P < 0.05). The results suggested that baicalin increased the number of newly generated cells in the hippocampus dentate gyrus in adult rats after cerebral ischemic injury.
Figure 4.
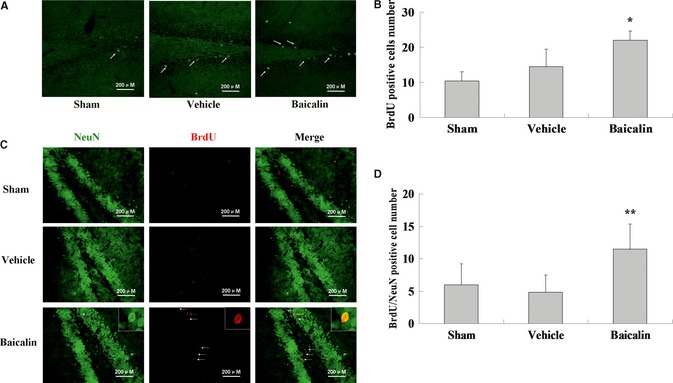
Baicalin increased the number of BrdU and BrdU/NeuN double staining‐positive cells in vivo. (A) Representative photomicrographs showing BrdU‐positive cells in the hippocampus of each group. BrdU‐labeled cells (Green) were indicated by arrows. (B) Quantification of BrdU‐positive cells in the hippocampus. (C) Representative photomicrographs showing BrdU/NeuN double staining‐positive cells (newly generated neurons) in the hippocampus of each group. New generated neurons were indicated by arrows. (D) Quantification of new generated neurons in the hippocampus. Scale bar: 200 μm. Each column represents the represent the mean ± SD (n = 3). *P < 0.05, **P < 0.01. Significant difference from the vehicle.
Baicalin Enhanced Adult Hippocampal Neurogenesis
To determine neurons from the newly generated cells, we performed double‐label fluorescence immunohistochemistry on the hippocampus using antibodies against the neuron‐specific protein (NeuN) in combination with the BrdU antibody. BrdU/NeuN double staining‐positive cells were seemed to be new neurons. As shown in Figure 4, BrdU/NeuN double staining‐positive cells were seldom found in sham group in the dentate gyrus of the hippocampus. A few BrdU/NeuN double staining‐positive cells were observed in the SGZ of hippocampal dentate gyrus in the vehicle‐treated rats. We observed a significant increase in the numbers of BrdU/NeuN double staining‐positive cells in the SGZ of hippocampal dentate gyrus in the baicalin‐treated group (Vehicle 4.8 ± 2.6 vs. Baicalin 11.5 ± 3.8, n = 3, P < 0.05), which was consistent with our in vitro findings that baicalin could prime proliferating NSPCs to be a neuronal fate during their subsequent differentiation. The results suggested that baicalin could promote new neurons production after cerebral ischemic injury.
Baicalin Improved the Learning and Memory Functions
Next, we used the Morris water maze task to examine the effect of baicalin on the cognitive functions after cerebral ischemic injury. Figure 5A showed the escape latencies of the three groups of animals during acquisition training (5 days). As expected, in all the groups, escape latencies became shorter with the training time prolonged. The decreased escape latency during the training process in the three groups clearly reflected memory for the escape platform place. However, differences were observed between the three groups, sham‐operated animals memorized the position of the hidden platform very quickly, thereby reached it in a shorter latency with the training time prolonged. In contrast, the vehicle‐treated ischemic animals showed a deficiency compared with the sham group. Although they were gradually trained and showed better performance, their escape latencies were still much longer than those of the sham group animals, and there were a significantly difference in the day 4 (Sham 18.37 ± 11.60 vs. Vehicle 44.38 ± 30.82, n = 10, P < 0.05) and day 5 (Sham 14.63 ± 5.03 vs. Vehicle 38.23 ± 24.29, n = 10, P < 0.01). Administration of baicalin significantly ameliorated such deficiencies. Post hoc analysis revealed that baicalin‐injected rats showed significantly faster learning rates compared with vehicle‐treated animals in the day 5 (Vehicle 38.23 ± 24.29 vs. Baicalin 20.05 ± 10.35, n = 10, P < 0.05). Figure 5B presents representative performances by the rat when the platform was removed on day 6. Clearly, the swim pattern of sham‐operated rats revealed a direct route to the platform, and continued to search in the original platform location. While the vehicle‐treated animals showed a complex search pattern in the pool. The search pattern in baicalin treatment group was more similar to the sham. The results indicated that baicalin treatment facilitated performance in the water maze task.
Figure 5.
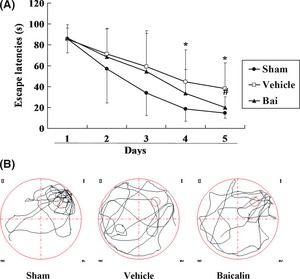
Effects of baicalin on spatial learning in Morris water maze. (A) The ordinate indicated escape latency to the hidden platform, and the abscissa represented the training time. There was a decrease in escape latencies with training in all three groups. Baicalin‐injected rats showed improved performance in the Morris water maze task compared with vehicle on day 5. Data are expressed as means ± SD of 10 rats in each group. *Significant difference from the Sham at P < 0.05, #Significant difference from the vehicle at P < 0.05. (B) Representative examples of search patterns in the pool of each group rats on day 6 after the platform removed.
Discussion
Previously studies had demonstrated that baicalin treatment could promote neuronal differentiation of C17.2 neural stem cells or primary cultured NSPCs after its commencing the differentiation process in vitro 22, 23, 24, 25. However, an alternative avenue for directing fate may be to pretreat NSPCs while they maintain their undifferentiated phenotype. In the present study, we demonstrated that a short‐time exposure of baicalin during proliferation was sufficient to induce neuronal fate commitment of NSPCs and stimulate neurogenesis in vivo.
While baicalin‐induced neuronal differentiation is prominent in our and others in vitro system 22, 23, 24, 25; in the present study, we found that baicalin could not only promote the neuronal differentiation, but also specify a neuronal fate of NSPCs during the proliferation process and then facilitate the neuron differentiation. From our results on the fate commitment of NSPCs, it is clear that baicalin activates the expression of proneural genes such as Mash1. Reports in various researches suggest that bHLH transcription factors direct neural differentiation 12, 13, 40. In line with this notion, our results indicate that increased expression levels of Mash1 point to a shift toward neuronal fate determination targeted by baicalin. Cell fate determination of NSPCs and subsequent successive steps of differentiation should be under strict control at the molecular and cellular levels. To provide more clear mechanisms that baicalin could prime the neuronal fate of the NSPCs, further study will be carried out.
There was evidence to suggest that proneural genes are involved in lineage restriction of cortical progenitors 41. The neuronal gene β‐tubulin III is a terminal differentiation gene 42. Considering the hierarchic order of these genes and preneural genes, it was not surprising that upregulation of mash1 leads to a similar effect on β‐tubulin III expression. To allow the dissection of the events that occur during neural cell‐fate determination, NSPCs were first subjected to baicalin treatment under proliferative conditions for 3 days, after that we withdrew the baicalin. In our differentiation system, after 3 days, the fraction of MAP‐2‐positive cells was also highly increased, as indicated by upregulation of the expression of the proneural genes. However, the fraction of GFAP‐positive cells showed no difference between baicalin treatment and control. Although this was in contradiction to the RT‐PCR data, this might be due to the proliferation of astroglia. Taken together, these data suggested that baicalin could efficiently induce neural specification of NSPCs during proliferation.
Baicalin could induce the NSPCs differentiate into neurons, and specify a neuronal fate of NSPCs in vitro; however, its role on promoting neurogenesis in vivo was still unclear. Because cerebral ischemia can cause prolonged spatial memory disturbance in rats 43. In order to examine the in vivo effects of baicalin on NSPCs, experimental cerebral ischemic rats were employed in the present study. Forebrain ischemia was performed by four‐vessel occlusion in this work. We demonstrated that baicalin significantly increased the numbers of newly generated cells in the dentate gyrus of the hippocampus after transient forebrain ischemia. To determine the neurons from the newly generated cells in the baicalin‐treated animals, we assessed the differentiated cells by colocalization of NeuN, a neuron phenotypic marker, in BrdU‐labeled cells. Animals treated with baicalin displayed an increase in the fraction of BrdU/NeuN double‐positive cells compared with vehicle‐treated animals (Figure 5). The in vivo BrdU/NeuN double staining analyses support our conclusion that chronic baicalin treatment enhanced the neurogenesis. In parallel, our in vitro data showed that baicalin could prime the NSPCs toward a neuronal fate during their proliferation and then facilitate the subsequent differentiation to the neurons.
To test baicalin that enhanced hippocampal memory function, Morris water maze had to be employed. The major finding of the present study was that baicalin facilitated spatial learning in cerebral ischemic rats. Interestingly, baicalin increased the newly generated neurons in the hippocampal compared with the vehicle‐treated animals. Considering that the hippocampus is an essential structure for spatial learning, it seems reasonable to assume that increased hippocampal neurogenesis induced by baicalin injection contributed at least partly to improving spatial learning. Thus, our data indicated that baicalin enhanced the neurogenesis and improved the cognitive functions after cerebral ischemic injury.
In conclusion, the present study demonstrated that baicalin could prime the NSPCs toward a neuronal fate and then facilitate the subsequent differentiation to the neurons and stimulate adult hippocampal neurogenesis after cerebral ischemic injury. Baicalin might be developed into a promising candidate for developing clinically relevant strategies to stimulate NSPCs for brain repair.
Conflict of Interest
The authors declare no conflict of interest.
Acknowledgments
This study was supported by National Natural Science Foundation of China (30873395 and 30472177), Key Technologies R&D Program of China (NO. 2011BAI07B08) and Program for Changjiang Scholars and Innovative Research Team in University (PCSIRT, No. IRT0973). We would thank Dr. Hui Wang and Prof. Yan Zhu for their helpful discussion and critical reading of our manuscript. We would thank Mr. Huaien Bu for technical assistant of the Statistical analysis.
The first two authors contributed equally to this work.
References
- 1. Emsley JG, Mitchell BD, Kempermann G, Macklis JD. Adult neurogenesis and repair of the adult CNS with neural progenitors, precursors, and stem cells. Prog Neurobiol 2005;75:321–341. [DOI] [PubMed] [Google Scholar]
- 2. McKeon RJ, Schreiber RC, Rudge JS, Silver J. Reduction of neurite outgrowth in a model of glial scarring following CNS injury is correlated with the expression of inhibitory molecules on reactive astrocytes. J Neurosci 1991;11:3398–3411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Jang ES, Goldman JE. Pax6 expression is sufficient to induce a neurogenic fate in glial progenitors of the neonatal subventricular zone. PLoS ONE 2011;6:e20894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Wang H, Ge G, Uchida Y, Luu B, Ahn S. Gli3 is required for maintenance and fate specification of cortical progenitors. J Neurosci 2011;31:6440–6448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Bond AM, Bhalala OG, Kessler JA. The dynamic role of bone morphogenetic proteins in neural stem cell fate and maturation. Dev Neurobiol 2012;72:1068–1084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Åkerblom M, Sachdeva R, Barde I, et al. MicroRNA‐124 is a subventricular zone neuronal fate determinant. J Neurosci 2012;32:8879–8889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Rogers CD, Moody SA, Casey ES. Neural induction and factors that stabilize a neural fate. Birth Defects Res C Embryo Today 2009;87:249–262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Johnson MA, Ables JL, Eisch AJ. Cell‐intrinsic signals that regulate adult neurogenesis in vivo: Insights from inducible approaches. BMB Rep 2009;42:245–259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Schneider JW, Gao Z, Li S, et al. Small‐molecule activation of neuronal cell fate. Nat Chem Biol 2008;4:408–510. [DOI] [PubMed] [Google Scholar]
- 10. Diamandis P, Wildenhain J, Clarke ID, et al. Chemical genetics reveals a complex functional ground state of neural stem cells. Nat Chem Biol 2007;3:268–273. [DOI] [PubMed] [Google Scholar]
- 11. Bertrand N, Castro DS, Guillemot F. Proneural genes and the specification of neural cell types. Nat Rev Neurosci 2002;3:517–530. [DOI] [PubMed] [Google Scholar]
- 12. Sommer L, Ma Q, Anderson DJ. Neurogenins, a novel family of atonal related bHLH transcription factors, are putative mammalian neuronal determination genes that reveal progenitor cell heterogeneity in the developing CNS and PNS. Mol Cell Neurosci 1996;8:221–241. [DOI] [PubMed] [Google Scholar]
- 13. Farah MH, Olson JM, Sucic HB, Hume RI, Tapscott SJ, Turner DL. Generation of neurons by transient expression of neural bHLH proteins in mammalian cells. Development 2001;27:693–702. [DOI] [PubMed] [Google Scholar]
- 14. Ma Q, Kintner C, Anderson DJ. Identification of neurogenin, a vertebrate neuronal determination gene. Cell 1996;87:43–52. [DOI] [PubMed] [Google Scholar]
- 15. Longo FM, Yang T, Xie Y, Massa SM. Small molecule approaches for promoting neurogenesis. Curr Alzheimer Res 2006;3:5–10. [DOI] [PubMed] [Google Scholar]
- 16. Vazey EM, Connor B. In vitro priming to direct neuronal fate in adult neural progenitor cells. Exp Neurol 2009;216:520–524. [DOI] [PubMed] [Google Scholar]
- 17. Whittemore SR, Morassutti DJ, Walters WM, Liu RH, Magnuson DS. Mitogen and substrate differentially affect the lineage restriction of adult rat subventricular zone neural precursor cell populations. Exp Cell Res 1999;252:75–95. [DOI] [PubMed] [Google Scholar]
- 18. Arsenijevic Y, Weiss S. Insulin‐like growth factor‐I is a differentiation factor for postmitotic CNS stem cell‐derived neuronal precursors: distinct actions from those of brain‐derived neurotrophic factor. J Neurosci 1998;18:2118–2128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Zhang Z, Wu R, Li P, et al. Baicalin administration is effective in positive regulation of twenty‐four ischemia/reperfusion‐related proteins identified by a proteomic study. Neurochem Int 2009;54:488–496. [DOI] [PubMed] [Google Scholar]
- 20. Chang CP, Huang WT, Cheng BC, Hsu CC, Lin MT. The flavonoid baicalin protects against cerebrovascular dysfunction and brain inflammation in experimental heatstroke. Neuropharmacology 2007;52:1024–1033. [DOI] [PubMed] [Google Scholar]
- 21. Tu XK, Yang WZ, Shi SS, Wang CH, Chen CM. Neuroprotective effect of baicalin in a rat model of permanent focal cerebral ischemia. Neurochem Res 2009;34:1626–1634. [DOI] [PubMed] [Google Scholar]
- 22. Zhuang PW, Zhang YJ, Pang T. Effects of brain microvascular endothelial cells and astrocytes following treatment with baicalin on neural stem cell differentiation. J Clin Rehabil Tissue Eng Res 2009;13:43–47. [Google Scholar]
- 23. Zhang MX, Li Y, Du R, Zhang YJ. Effect of baicalin on differentiation of neural stem cells in vitro. J Tianjin Univ Traditional Chinese Med 2007;26:199–201. [Google Scholar]
- 24. Li Y, Zhuang PW, Shen BG, Zhang YJ, Shen JG. Baicalin promotes neuronal differentiation of neural stem/progenitor cells through modulating p‐stat3 and bHLH family protein expression. Brain Res 2012;1429:36–42. [DOI] [PubMed] [Google Scholar]
- 25. Li M, Tsang KS, Choi ST, Li K, Shaw PC, Lau KF. Neuronal differentiation of c17.2 neural stem cells induced by a natural flavonoid, baicalin. ChemBioChem 2011;12:449–456. [DOI] [PubMed] [Google Scholar]
- 26. Mook‐Jung I, Hong HS, Boo JH, et al. Ginsenoside Rb1 and Rg1 improve spatial learning and increase hippocampal synaptophysin level in mice. J Neurosci Res 2004;63:509–515. [DOI] [PubMed] [Google Scholar]
- 27. Shen LH, Zhang JT. Ginsenoside Rg1 promotes proliferation of hippocampal progenitor cells. Neurol Res 2004;26:422–428. [DOI] [PubMed] [Google Scholar]
- 28. Liu JW, Tian SJ, de Barry J, Luu B. Panaxadiol glycosides that induce neuronal differentiation in neurosphere stem cells. J Nat Prod 2007;70:1329–1334. [DOI] [PubMed] [Google Scholar]
- 29. Kim SJ, Son TG, Park HR, et al. Curcumin stimulates proliferation of embryonic neural progenitor cells and neurogenesis in the adult hippocampus. J Biol Chem 2008;283:14497–14505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Yabe T, Hirahara H, Harada N, et al. Ferulic acid induces neural progenitor cell proliferation in vitro and in vivo. Neuroscience 2010;165:515–524. [DOI] [PubMed] [Google Scholar]
- 31. Guo G, Li B, Wang Y, et al. Effects of salvianolic acid B on proliferation, neurite outgrowth and differentiation of neural stem cells derived from the cerebral cortex of embryonic mice. Sci China Life Sci 2010;53:653–662. [DOI] [PubMed] [Google Scholar]
- 32. Zhuang P, Zhang Y, Cui G, et al. Direct stimulation of adult neural stem/progenitor cells in vitro and neurogenesis in vivo by salvianolic Acid B. PLoS ONE 2012;7:e35636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Huang H, Zhang Y, Yang R, Tang X. Determination of baicalin in rat cerebrospinal fluid and blood using microdialysis coupled with ultra‐performance liquid chromatography‐tandem mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 2008;874:77–83. [DOI] [PubMed] [Google Scholar]
- 34. Davis AA, Temple S. A self‐renewing multipotential stem cell in embryonic rat cerebral cortex. Nature 1994;372:263–266. [DOI] [PubMed] [Google Scholar]
- 35. Nitatori T, Sato N, Waguri S, et al. Delayed neuronal death in the CA1 pyramidal cell layer of the gerbil hippocampus following transient ischemia is apoptosis. J Neurosci 1995;15:1001–1011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Pulsinelli WA, Brierley JB, Plum F. Temporal profile of neuronal damage in a model of transient forebrain ischemia. Ann Neurol 1982;11:491–498. [DOI] [PubMed] [Google Scholar]
- 37. Quintard H, Borsotto M, Veyssiere J, et al. MLC901, a traditional Chinese medicine protects the brain against global ischemia. Neuropharmacology 2011;61:622–631. [DOI] [PubMed] [Google Scholar]
- 38. Casarosa S, Fode C, Guillemot F. Mash1 regulates neurogenesis in the ventral telencephalon. Development 1999;126:525–534. [DOI] [PubMed] [Google Scholar]
- 39. Schuurmans C, Armant O, Nieto M, et al. Sequential phases of cortical specification involve neurogenin‐dependent andindependent pathways. EMBO J 2004;23:2892–2902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Perron M, Harris WA. Determination of vertebrate retinal progenitor cell fate by the Notch pathway and basic helix‐loop‐helix transcription factors. Cell Mol Life Sci 2000;57:215–223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Nieto M, Schuurmans C, Britz O, Guillemot F. Neural bHLH genes control the neuronal versus glial fate decision in cortical progenitors. Neuron 2001;29:401–413. [DOI] [PubMed] [Google Scholar]
- 42. Gangemi RM, Perera M, Corte G. Regulatory genes controlling cell fate choice in embryonic and adult neural stem cells. J Neurochem 2004;89:286–306. [DOI] [PubMed] [Google Scholar]
- 43. Yonemori F, Yamada H, Yamaguchi T, Uemura A, Tamura A. Spatial memory disturbance after focal cerebral ischemia in rats. J Cereb Blood Flow Metab 1996;16:973–980. [DOI] [PubMed] [Google Scholar]


