Abstract
Objective:
The aim of this study was to determine the clinical meaningfulness of TX-001HR in reducing moderate to severe vasomotor symptoms (VMS) in menopausal women with a uterus.
Methods:
In the REPLENISH study (NCT01942668), women with moderate to severe hot flushes (≥7/d or ≥50/wk) were enrolled in a VMS substudy and randomized to four doses of daily TX-001HR (17β-estradiol/progesterone) or placebo. Participants assessed improvement of their VMS by the Clinical Global Impression and the Menopause-Specific Quality of Life (MENQOL) questionnaire, which were used to define clinical responders, clinically important differences (CIDs) or minimal CID (MCID) in VMS frequency. Response thresholds were determined by nonparametric discriminant analyses utilizing bootstrapping methods.
Results:
In the modified intent-to-treat VMS substudy population (n = 726), statistically significantly more Clinical Global Impression–based clinical responders were observed with TX-001HR than placebo for MCID (weekly reduction of ≥25 moderate to severe VMS: 82-88% vs 69%; all, P < 0.05) and CID (weekly reduction of ≥39 VMS: 68%-73% vs 52%; all, P < 0.05) at week 12. Week 4 results were similar. For Menopause Quality of Life–based analysis, significantly more clinical responders were observed with TX-001HR than placebo for MCID (weekly reduction of ≥34 VMS: 74%-81% vs 55%; all, P < 0.01) and CID (weekly reduction of ≥44 VMS: 61%-69% vs 42%; all, P < 0.01) at week 12.
Conclusions:
TX-001HR provided clinically meaningful improvements (as measured by 2 different methods), in addition to statistically significant reductions, in menopausal VMS frequency. TX-001HR may provide a new option, as a single oral capsule of estradiol and progesterone (identical to the hormones naturally occurring in women) for the treatment of moderate to severe VMS in menopausal women with a uterus.
Keywords: Estradiol, Hot flushes, Menopause, Progesterone, Vasomotor
Vasomotor symptoms (VMS) in menopausal women are effectively treated with hormone therapy (HT) reducing hot flush frequency and severity. Although statistically significant reductions are reported with various doses and regimens of HT, the clinical relevance to women is typically not determined. Reductions in VMS frequency of 50% to 86% from baseline, or 5.5 to 9.0 hot flushes per day, have been reported in some randomized controlled trials1-3; however, the relevance of these findings is often unknown.1,4-6 The REPLENISH trial, evaluated TX-001HR (TherapeuticsMD, Boca Raton, FL), a once-daily, oral capsule containing 17β-estradiol (E2) and progesterone (P4) for the treatment of moderate to severe VMS in menopausal women with an intact uterus.7 In this study, women took one of four doses of TX-001HR (E2/P4 [mg/mg] 1/100, 0.5/100, 0.5/50, 0.25/50) or placebo for up to 52 weeks. All doses of TX-001HR, compared with placebo, showed statistically significant reductions in hot flush frequency from baseline to weeks 4 and 12, except for the 0.5 mg E2/50 mg P4 dose at week 4.7 The two highest doses of TX-001HR also had statistically significant improvements in hot flush severity at weeks 4 and 12, as did the 0.5 mg E2/50 mg P4 at week 12.7
The objectives of this analysis were to determine what reduction in VMS frequency was meaningful to women, using data from the REPLENISH trial, and to determine the clinical importance (meaningfulness) of TX-001HR versus placebo effects when treating moderate to severe VMS in menopausal women.
METHODS
Study design
REPLENISH (NCT01942668) was a phase 3, randomized, double-blind, placebo-controlled, multicenter trial that evaluated TX-001HR in menopausal women (age 40-65 y; body mass index ≤34 kg/m2) with an intact uterus. Study details have been published elsewhere.7 Briefly, women with moderate to severe hot flushes (≥7/d or ≥50/wk) were included in a VMS substudy and were randomized 1:1:1:1:1 to daily E2/P4 (mg/mg) of 1/100, 0.5/100, 0.5/50, 0.25/50, or placebo. Women who did not qualify for the VMS substudy were randomized 1:1:1:1 to active E2/P4 doses as part of the primary safety endpoint analysis of endometrial hyperplasia (reported elsewhere).7 All women took two capsules to maintain blinding as different doses were two different sizes.
The safety population included all women who took at least one capsule of the study drug. Women were included in the modified intent-to-treat (MITT)-VMS population (primary efficacy population) if they were randomized to the VMS substudy, took at least one dose (two capsules) of study medication, had ≥5 days of VMS diary data at baseline, and had ≥4 days of VMS diary data for one on-treatment week. All efficacy analyses were performed using this MITT-VMS population.
Four coprimary efficacy endpoints of the study included mean changes in frequency and severity of moderate to severe VMS from baseline to weeks 4 and 12 with active treatment versus placebo in the MITT-VMS population. Secondary endpoints included changes from baseline in the Clinical Global Impression (CGI) scale and the Menopause-Specific Quality of Life (MENQOL) questionnaire. Participants completed a daily VMS diary and recorded the number and severity of hot flushes up to week 12. Weekly hot flush frequency was the total number of moderate and severe hot flushes in the previous 7 days. Severity of hot flushes was rated as mild, moderate, or severe.
Evaluation of clinical meaningfulness
The clinical meaningfulness of treatment was assessed using two separate patient-reported outcome (PRO) measures: the CGI scale and the MENQOL questionnaire. In this study, CGI and MENQOL were used as anchor measures to determine clinically important differences (CIDs) and minimal clinically important differences (MCIDs) in VMS frequency. Evaluating TX-001HR with two PRO tools was intended to assess the consistency of the TX-001HR effect. Analyses were performed in the MITT-VMS population.
Clinical global impression
Participants in the VMS substudy completed CGI at weeks 4, 8, and 12, by answering the following question: “Rate the total improvement, whether or not in your judgment it is due entirely to drug treatment. Compared to your condition at admission to the study, how much has it changed?” Potential responses were rated using a 7-point Likert scale and included “very much improved,” “much improved,” “minimally improved,” “no change,” “minimally worse,” “much worse,” or “very much worse.” Each E2/P4 dose was compared with placebo at weeks 4 and 12.
To evaluate clinical meaningfulness, participants with CGI ratings of much improved or very much improved were categorized as having a “clinically meaningful” response, whereas those with a rating of minimally improved were categorized as having a “minimally improved” response, and those with ratings of no change to very much worse were categorized as having “no change or worse response.” Nonparametric discriminant analyses utilizing bootstrapping methods were used to determine a clinically meaningful threshold (ie, CID) for a decrease in moderate to severe VMS at weeks 4 and 12, regardless of treatment.8,9 Thresholds for an MCID were also determined. Proportions of women who had a clinically important response who took TX-001HR were compared with those taking placebo using the Fisher's exact test.
Menopause-Specific Quality of Life questionnaire
Participants in the VMS substudy self-administered the MENQOL questionnaire at baseline, week 12, and months 6 and 12. The MENQOL questionnaire, a validated research tool, assessed menopause-specific quality of life changes in study participants with 29 questions on symptoms distributed across 4 domains: vasomotor, psychosocial, physical, and sexual.10 The vasomotor domain consisted of three questions: How much have you been bothered by (1) hot flushes or flashes, (2) night sweats, and (3) sweating. Changes in quality of life were evaluated over the previous 1-month period. If women experienced symptoms, the symptoms were rated using a 7-item Likert scale ranging from “not at all bothered” (score of 0) to “extremely bothered” (score of 6). Scores were converted to an analysis score with a range of 1 (no symptoms) to 8 (extremely bothered). Changes from baseline in the MENQOL vasomotor domain scores were analyzed using analysis of covariance at week 12.
Changes in the MENQOL vasomotor domain were also used to calculate clinically meaningful responses. CID was defined as a participant with a change of −2.0 (improvement by 2 categories) from baseline to week 12, whereas those with a change of −1.0 were categorized as having an MCID. Those with ratings of 0 to +3 were categorized has having no change or a worse response. Clinical threshold levels were calculated by averaging the VMS reduction of women with an improvement score of 1 category (MCID) or 2 categories (CID), regardless of treatment. Proportions of women who had a clinically important response who took TX-001HR were compared with those taking placebo using the Fisher's exact test.
RESULTS
Disposition and demographics
A total of 1,835 women were randomized to the endometrial and general safety study and took at least one capsule of study drug. Of these, 766 were enrolled in the VMS substudy and 726 were eligible for the MITT-VMS population. Eighty-nine percent of the women completed the 12-week VMS efficacy substudy. Overall discontinuation rates from the study at 12 months were 28.3% with TX-001HR and 31.1% with placebo. Discontinuations due to lack of efficacy occurred in 1.2% of the women treated with TX-001HR and in 8.9% of the women treated with placebo.
Women in the MITT-VMS population had a mean age of 55 years (40-65) and a mean body mass index of 27 kg/m2 at study entry (Table 1). Approximately two thirds of the women were White (67%) and one third were African American (31%). At baseline, the frequency of moderate to severe VMS ranged from 72.1 to 77.0 per week or 10.3 to 11.0 per day. Statistical comparisons between groups for baseline demographics were not performed.
TABLE 1.
Participant demographics and baseline characteristics of the modified intent-to-treat (MITT)-VMS population
| Estradiol/progesterone | |||||
| Characteristic | 1 mg/100 mg | 0.5 mg/100 mg | 0.5 mg/50 mg | 0.25 mg/50 mg | Placebo |
| n | 141 | 149 | 147 | 154 | 135 |
| Age, y | 54.7 ± 4.8 | 54.9 ± 4.5 | 54.8 ± 4.6 | 54.5 ± 3.8 | 54.3 ± 4.3 |
| Race, n (%) | |||||
| White | 95 (67.4) | 99 (66.4) | 99 (67.3) | 102 (66.2) | 91 (67.4) |
| African American | 45 (31.9) | 48 (32.2) | 43 (29.3) | 48 (31.2) | 41 (30.4) |
| Othera | 1 (0.7) | 2 (1.3) | 5 (3.4) | 4 (2.6) | 3 (2.2) |
| BMI, kg/m2 | 26.5 ± 3.9 | 27.1 ± 4.3 | 26.6 ± 3.9 | 26.4 ± 4.0 | 26.6 ± 3.8 |
| Time since menopause, y | 6.1 ± 5.5 | 6.5 ± 5.4 | 6.0 ± 4.8 | 5.2 ± 4.8 | 5.7 ± 4.9 |
| Bilateral oophorectomy | 3 (2.1) | 3 (2.0) | 1 (0.7) | 1 (0.6) | 0 |
| Baseline VMS parameters | |||||
| Weekly frequency | 74.4 ± 35.3 | 72.1 ± 27.8 | 75.9 ± 28.0 | 77.0 ± 30.4 | 72.4 ± 23.3 |
| Weekly severity | 2.54 ± 0.32 | 2.51 ± 0.25 | 2.50 ± 0.23 | 2.51 ± 0.26 | 2.52 ± 0.25 |
Data presented as mean ± SD, unless stated otherwise.
SD, standard deviation; BMI, body mass index; VMS, vasomotor symptoms.
aOther includes other (n = 10), American Indian or Alaska Native (n = 2), Native Hawaiian or Pacific Islander (n = 2), and unknown (n = 1).
Clinical global impression
Response
On the CGI, significantly more women responded that they felt their symptoms had at least “much improved” with TX-001HR compared with placebo at week 4 (50%-63% vs 33%; all, P < 0.01), as well as at week 12 (73%-82% vs 53%; all, P < 0.01; Fig. 1).
FIG. 1.
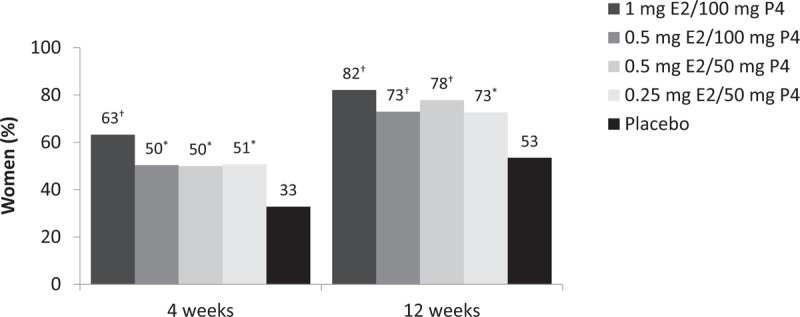
Proportion of women who rated their condition as very much or much improved (Clinical Global Impression [CGI] response rate) at weeks 4 and 12. ∗P < 0.01; †P < 0.001 versus placebo, calculated with Fisher exact test.
Anchor-based minimal clinically important difference and clinically important difference calculations
Calculated clinical meaningfulness thresholds for week 4 were weekly reductions in moderate to severe VMS frequency of ≥15 for MCID and ≥36 for CID (Fig. 2A). Based on these thresholds, significantly more clinical responders were found with all four doses of TX-001HR than with placebo at week 4 for MCID (75%-85% vs 64%; all, P < 0.05) and for CID (46%-59% vs 33%; all, P < 0.05; Fig. 2B). Similar results were observed at week 12. Calculated clinical meaningfulness thresholds at week 12 were weekly reductions in moderate to severe VMS frequency of ≥25 for MCID and ≥39 for CID (Fig. 2C). Significantly more clinical responders were found with all four doses of TX-001HR than with placebo at week 12 for MCID (82%-88% vs 69%; all, P < 0.05) and for CID (68%-73% vs 52%; all, P < 0.05; Fig. 2D).
FIG. 2.

(A) Clinical meaningfulness threshold analysis at week 4. (B) Clinical Global Impression (CGI)-based CID and MCID analysis at week 4. (C) Clinical meaningfulness threshold analysis at week 12. (D) CGI-based CID and MCID analysis at week 12. ∗P < 0.05; †P < 0.01; ‡P < 0.001 versus placebo, calculated with Fisher's exact test. CID, clinically important difference; MCID, minimal clinically important difference; VMS, vasomotor symptoms.
Menopause-Specific Quality of Life questionnaire
Response
Participants treated with TX-001HR compared with placebo had significantly greater improvements in the MENQOL vasomotor domain score from baseline to week 12 (−3.2 to −3.8 vs −2.2 points; all, P < 0.001; Fig. 3). Vasomotor domain scores ranged from 6.9 to 7.2 at baseline and improved to 3.3 to 3.9 with TX-001HR and 5.0 with placebo at week 12.
FIG. 3.

Change from baseline in Menopause-Specific Quality of Life questionnaire (MENQOL) vasomotor domain at week 12. ∗P < 0.001 versus placebo in least squares mean, derived from the analysis of covariance (ANCOVA) model with treatment as factors and baseline as covariate.
Anchor-based minimal clinically important difference and clinically important difference calculations
Women with MCID, a change of one category on the MENQOL vasomotor domain, had a weekly reduction in VMS frequency of ≥34, whereas those with CID (change of 2 categories) had a weekly reduction in VMS frequency of ≥44 (Fig. 4A). Based on these clinical response thresholds significantly more TX-001HR users (61%-69%) had CIDs at week 12 than placebo users (42%; all, P < 0.01; Fig. 4B). Similarly, significantly more women at week 12 had MCID with TX-001HR doses (74%-81%) than with placebo (55%; all, P < 0.01).
FIG. 4.

A, Threshold response analysis for MENQOL at week 12. B, MENQOL-based clinical meaningfulness analysis at week 12 for MCID and CID. ∗P < 0.01; †P ≤ 0.001 versus placebo, calculated with Fisher's exact test. CID, clinically important difference; MCID, minimal clinically important difference; MENQOL, Menopause-Specific Quality of Life questionnaire; VMS, vasomotor symptoms.
DISCUSSION
In the REPLENISH trial, TX-001HR was associated with clinically meaningful improvements in moderate to severe VMS frequency in menopausal women. These clinically important changes were observed with two separate PROs: CGI and the MENQOL vasomotor domain. Both PROs consistently showed that a significantly larger proportion of women who took TX-001HR had clinically meaningful symptom improvements compared with those who took placebo.
Statistically significant reductions in the frequency and severity of moderate to severe hot flushes between active treatment and placebo are required for demonstrating efficacy of potential treatments for moderate to severe VMS in menopausal women. These co-primary endpoints were met with most TX-001HR doses versus placebo.7 It is also, however, important to determine whether statistically significant differences in hot flush frequency and severity between active treatment and placebo are clinically meaningful. In our study, we determined clinically important changes in VMS frequency by evaluating the proportion of women who have CID or MCID with treatment versus placebo. We determined that women who had clinically meaningful responses or CIDs had weekly reductions in VMS frequency at 12 weeks of ≥39 hot flushes (by CGI), or ≥44 hot flushes (by the MENQOL vasomotor domain), whereas MCIDs were associated with weekly reductions in VMS frequency of ≥25 hot flushes (CGI-based), or ≥34 hot flushes (MENQOL-based).
The CGI anchor-based method was empirically used in a study evaluating the effects of estradiol/drospirenone and estradiol monotherapy for the treatment of moderate to severe VMS in menopausal women with ≥50 moderate to severe hot flushes per week.8 In that study, the MCID thresholds were defined as being a weekly reduction of at least 19.1 hot flushes at week 4 and 40.3 hot flushes at week 12.8 The CGI thresholds in our study were higher or comparable, also distinguishing the effect of TX-001HR from placebo for symptom improvement. Clinically meaningful effects on moderate to severe VMS were also reported with desvenlafaxine using a different outcome.11
PROs are commonly used as primary or secondary endpoints to evaluate the efficacy of treatments. FDA guidance on the use of PROs for new treatments is to compare the proportion of clinically meaningful responders by treatment group.11 Guyatt et al12,13 defined an MCID as the smallest change in PRO endpoint scores that are considered beneficial and important to patients and their clinicians. The MCID represents the smallest improvement that patients (and their clinicians) perceive as beneficial and important, CID therefore offers a threshold above which the treatment outcome is experienced as relevant by the patient. This is the first study to use two PROs to test the effect of treatment and assess clinical meaningfulness. The results demonstrated the consistency of TX-001HR treatment, showing that 68% to 73% and 61% to 69% of women who took TX-001HR for 12 weeks had clinically meaningful improvements defined by thresholds of the CGI (52% with placebo) and MENQOL vasomotor domain (42% with placebo), respectively.
After the publication of the Women's Health Initiative findings in 2002,14 many women discontinued FDA-approved HT,15 with many starting to use non-FDA−approved compounded hormone therapy (CHT). Recent annual estimates of CHT use have shown that 1 to 2.5 million women filled 21 to 39 million prescriptions in the United States.16,17 CHT products have, however, not been rigorously tested for efficacy and safety.18 Although many women prefer taking “natural” products and believe they are safer options than FDA-approved HT products,19-22 they may be unaware of risks associated with CHT, including insufficient endometrial protection resulting in endometrial cancer or hyperplasia.23-25 The PRO data reported here, which show that women taking TX-001HR have clinically meaningful responses to treatment, might help women and healthcare providers better understand the benefits of FDA-approved products. If approved, the formulation of TX-001HR combining E2/P4, which has been rigorously evaluated in the randomized, placebo-controlled REPLENISH trial, may become a new option for women taking poorly regulated CHT.
Some of the limitations of the study include a short duration of treatment, studying women that are healthier than the general population and evaluating only women residing in the United States. Strengths of the study include that it was a well-designed, double-blind, randomized controlled trial planned with the guidance of the FDA. The study design, health of the population, and discontinuation rates are similar to other studies that have evaluated menopausal drugs.
CONCLUSIONS
TX-001HR significantly reduced the frequency and/or severity of VMS in the REPLENISH study. Significantly more women had a clinically meaningful improvement in the number of hot flushes with TX-001HR versus placebo using two PROs, the CGI, and the MENQOL vasomotor domain. The results of this analysis extend the primary efficacy results of the REPLENISH trial, which showed significant improvements in the frequency and severity of moderate to severe VMS with TX-001HR compared with placebo at weeks 4 and 12.7 TX-001HR, if approved, may provide a new option for treating VMS in menopausal women with an intact uterus using E2 and P4, similar to the hormones naturally occurring in women, in a single, oral capsule.
Acknowledgments
The authors would also like to acknowledge the medical writing assistance of Dominique Verlaan, PhD, CMPP of Precise Publications, LLC, which was supported by TherapeuticsMD.
Footnotes
Trial Registration: https://clinicaltrials.gov/ct2/show/NCT01942668
Funding/support: TherapeuticsMD sponsored the study, and provided support for the medical writing assistance of Dominique Verlaan, PhD, CMPP (Precise Publications, LLC).
Financial disclosure/conflicts of interest: Dr Constantine consults to multiple pharmaceutical companies including but not limited to TherapeuticsMD, and has stock options from TherapeuticsMD. Dr Revicki is a consultant for TherapeuticsMD. Dr Kagan is a consultant to Allergan, AMAG, Pfizer, P&G, Duchesnay, Cooper Surgical; Speakers Bureau for Pfizer, Valeant, AMAG; and has received research grants and support from TherapeuticsMD, but paid to Sutter Health. Dr Simon has served (within the past year, or current) as a consultant/advisor to AbbVie, Allergan, AMAG, Amgen, Ascend Therapeutics, Azure Biotech, Bayer Healthcare, CEEK Enterprises, Covance, Millendo Therapeutics, Mitsubishi Tanage, ObsEva SA, Radius Health, Sanofi SA, Sebela, Sermonix, Shionogi, Symbiotec Pharmalab, TherapeuticsMD, and Valeant; and has received (within the past year, or current) grant/research support from AbbVie, Agile Therapeutics, Allergan, Bayer Healthcare, New England Research Institute, ObsEva SA, Palatin Technologies, Symbio Research, and TherapeuticsMD; and has also served (within the past year, or current) on the speaker's bureaus of Duchesnay, Novo Nordisk, Shionogi, and Valeant; and is a stockholder (direct purchase) in Sermonix Pharmaceuticals. Dr Bernick is a board member and an employee of TherapeuticsMD with stock/stock options. Dr Graham and Dr Mirkin are employees of TherapeuticsMD with stock/stock options.
REFERENCES
- 1.Lobo RA, Pinkerton JV, Gass MLS, et al. Evaluation of bazedoxifene/conjugated estrogens for the treatment of menopausal symptoms and effects on metabolic bone parameters and overall safety profile. Fertil Steril 2009; 92:1025–1038. [DOI] [PubMed] [Google Scholar]
- 2.Archer DF, Pickar JH, MacAllister DC, Warren MP. Transdermal estradiol gel for the treatment of symptomatic postmenopausal women. Menopause 2012; 19:622–629. [DOI] [PubMed] [Google Scholar]
- 3.Archer DF, Schmelter T, Schaefers M, Gerlinger C, Gude K. A randomized, double-blind, placebo-controlled study of the lowest effective dose of drospirenone with 17beta-estradiol for moderate to severe vasomotor symptoms in postmenopausal women. Menopause 2014; 21:227–235. [DOI] [PubMed] [Google Scholar]
- 4.MacLennan AH, Broadbent JL, Lester S, Moore V. Oral oestrogen and combined oestrogen/progestogen therapy versus placebo for hot flushes. Cochrane Database Syst Rev 2004; CD002978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Loprinzi CL, Sloan J, Stearns V, et al. Newer antidepressants and gabapentin for hot flashes: an individual patient pooled analysis. J Clin Oncol 2009; 27:2831–2837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Public Citizen. Testimony Before the FDA's Advisory Committee on Reproductive Health Drugs on New Drug Application (NDA) 22-506, Gabapentin Tablets (Proposed Trade Name SEFELSA) for Treatment of Vasomotor Symptoms due to Menopause. Michael A. Carome MD and Sidney M. Wolfe MD Public Citizen's Health Research Group. March 4, 2013. Available at: https://www.citizen.org/sites/default/files/2099.pdf Accessed September 20, 2017. [Google Scholar]
- 7.Lobo R, Archer DF, Kagan R, et al. A 17β-estradiol-progesterone oral capsule for postmenopausal women: a randomized controlled trial. Obstet Gynecol 2018; 132:161–170. [DOI] [PubMed] [Google Scholar]
- 8.Gerlinger C, Gude K, Hiemeyer F, Schmelter T, Schafers M. An empirically validated responder definition for the reduction of moderate to severe hot flushes in postmenopausal women. Menopause 2012; 19:799–803. [DOI] [PubMed] [Google Scholar]
- 9.Revicki D, Hays RD, Cella D, Sloan J. Recommended methods for determining responsiveness and minimally important differences for patient-reported outcomes. J Clin Epidemiol 2008; 61:102–109. [DOI] [PubMed] [Google Scholar]
- 10.Hilditch JR, Lewis J, Peter A, et al. A menopause-specific quality of life questionnaire: development and psychometric properties. Maturitas 1996; 24:161–175. [DOI] [PubMed] [Google Scholar]
- 11.Food and Drug Administration. Guidance for Industry: Patient-reported Outcome Measures—Use in Medical Product Development to Support Labeling Claims. Available at: http://www.fda.gov/downloads/Drugs/Guidances/UCM193282.pdf Accessed December 2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Guyatt G, Walter S, Norman G. Measuring change over time: assessing the usefulness of evaluative instruments. J Chronic Dis 1987; 40:171–178. [DOI] [PubMed] [Google Scholar]
- 13.Jaeschke R, Singer J, Guyatt GH. Measurement of health status. Ascertaining the minimal clinically important difference. Control Clin Trials 1989; 10:407–415. [DOI] [PubMed] [Google Scholar]
- 14.Rossouw JE, Anderson GL, Prentice RL, et al. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women's Health Initiative randomized controlled trial. JAMA 2002; 288:321–333. [DOI] [PubMed] [Google Scholar]
- 15.Steinkellner AR, Denison SE, Eldridge SL, Lenzi LL, Chen W, Bowlin SJ. A decade of postmenopausal hormone therapy prescribing in the United States: long-term effects of the Women's Health Initiative. Menopause 2012; 19:616–621. [DOI] [PubMed] [Google Scholar]
- 16.Pinkerton J, Santoro N. Compounded bioidentical hormone therapy: identifying use trends and knowledge gaps among US women. Menopause 2015; 22:926–936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Pinkerton J, Constantine G. Compounded non-FDA-approved menopausal hormone therapy prescriptions have increased: results of a pharmacy survey. Menopause 2016; 23:359–367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Pinkerton JV, Pickar JH. Update on medical and regulatory issues pertaining to compounded and FDA-approved drugs, including hormone therapy. Menopause 2016; 23:215–223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Fishman JR, Flatt MA, Settersten RA., Jr Bioidentical hormones, menopausal women, and the lure of the “natural” in U.S. anti-aging medicine. Soc Sci Med 2015; 132:79–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Iftikhar S, Shuster LT, Johnson RE, Jenkins SM, Wahner-Roedler DL. Use of bioidentical compounded hormones for menopausal concerns: cross-sectional survey in an academic menopause center. J Womens Health (Larchmt) 2011; 20:559–565. [DOI] [PubMed] [Google Scholar]
- 21.Adams C, Cannell S. Women's beliefs about “natural” hormones and natural hormone replacement therapy. Menopause 2001; 8:433–440. [DOI] [PubMed] [Google Scholar]
- 22.Gass ML, Stuenkel CA, Utian WH, et al. Use of compounded hormone therapy in the United States: report of the North American Menopause Society Survey. Menopause 2015; 22:1276–1284. [DOI] [PubMed] [Google Scholar]
- 23.Eden JA, Hacker NF, Fortune M. Three cases of endometrial cancer associated with “bioidentical” hormone replacement therapy. Med J Aust 2007; 187:244–245. [DOI] [PubMed] [Google Scholar]
- 24.Davis R, Batur P, Thacker HL. Risks and effectiveness of compounded bioidentical hormone therapy: a case series. J Womens Health (Larchmt) 2014; 23:642–648. [DOI] [PubMed] [Google Scholar]
- 25.Dezman VL, Gersak MZ, Gersak K. Two case of atypical endometrial hyperplasia associated with “bioidentical” hormone replacement therapy: IGCS-0084 Uterine Cancer, including Sarcoma. Int J Gynecol Cancer 2015; 25 suppl 1:71. [DOI] [PubMed] [Google Scholar]


