Abstract
Abstract. Pluripotent stem cells, such as embryonic stem cells (ESCs), proliferate rapidly while maintaining pluripotency, namely, the ability to differentiate into various types of cells. Embryonic stem cells are promising donor sources for cell transplantation therapies. However, human ESCs are also associated with ethical issues regarding the use of human embryos and rejection reactions after allogenic transplantation. It may be possible to overcome these issues by generating pluripotent stem cells directly from a patient's somatic cells. That somatic cell nuclei acquire an embryonic stem‐like status by fusion with ESCs suggests the existence of ‘pluripotency‐inducing’ factors. Previous studies have recently shown that retrovirus‐mediated transfection with four transcription factors (Oct‐3/4, Sox2, KLF4 and c‐Myc), which are highly expressed in ESCs, into mouse fibroblasts has resulted in generation of induced pluripotent stem (iPS) cells. iPS cells are similar to ESCs in morphology, proliferation, and pluripotency, judged by teratoma formation and chimaera contribution. If iPS cells can be derived from human somatic cells, then such cells may thus lead to important drug discoveries and advances in regenerative medicine.
INTRODUCTION
Embryonic stem cells (ESCs) are derived from the inner cell mass of blastocyst and proliferate extensively while maintaining pluripotency. The isolation of the first ESCs from mouse embryos (Evans & Kaufman 1981; Martin 1981) resulted in the development of ‘knockout mouse’ technology (Doetschman et al. 1987; Hooper et al. 1987). Because ESCs, theoretically, have the capacity to develop into any type of cell, generation of ESC lines from human blastocyst stage embryos (Thomson et al. 1998) has opened up the possibility of using them as a donor source for cell transplantation therapies. Potential clinical applications include the treatment of juvenile diabetes, Parkinson's disease, heart failure and spinal cord injury. However, the use of human embryos and tissue rejection remain concerns for ESC transplantation. One strategy to avoid such issues is to re‐programme the nuclei of differentiated cells to an ESC‐like, pluripotent state, and then use these cells to generate the appropriate donor cells for transplantation.
RE‐PROGRAMMING BY FUSION WITH EMBRYONIC STEM CELLS
In 1976, Miller and Ruddle demonstrated that thymocytes acquired pluripotency upon fusion with embryonal carcinoma cells (ECCs) (1976), and similar results were later obtained by electrofusion with embryonic germ cells (EGCs) (Tada et al. 1997) and mouse ESCs (Tada et al. 2001). Transplantation of these cells into nude mice results in the formation of teratomas consisting of various tissues from all three germ layers, confirming the pluripotency of these cells. Finally, re‐programming by fusion with human ESCs was reported in 2005 (Cowan et al. 2005; Yu et al. 2006). These results suggest that ESCs contain factors that induce pluripotency in somatic cells. However, this technology would result in tetraploid cells containing the genome derived from ESCs. The risk of tetraploid cells becoming malignant after transplantation remains to be determined. In addition, the fused cells are not free from immune rejection after transplantation.
GENERATION OF INDUCED PLURIPOTENT STEM CELLS
Successful re‐programming by fusion indicates that ESCs contain factors that induce pluripotency in somatic cells. It is likely that these pluripotency‐inducing factors also play important roles in the maintenance of pluripotency in ES cells. Based on this hypothesis, three groups of potential pluripotency‐inducing factors have been selected (Takahashi & Yamanaka 2006). The first group consists of transcription factors specifically expressed in ESCs, including Nanog, Oct‐3/4, Sox2, UTF1, Sall4, Sox15 and Rex1. The second group consists of growth‐ and tumour‐related gene products that have been shown to play important roles in ESCs, including c‐Myc, Stat3, β‐catenin, Grb2, KLF4, TCL1 and ERas. The third group consists of factors that are also specifically expressed in ESCs, but with, as yet, less defined functions. They include ECAT1, ESG1, Fbx15, DNMT3L, ECAT8, GDF3, ECAT15‐1, ECAT15‐2, Fthl17 and Stella (Fig. 1).
Figure 1.

Candidates for pluripotency inducing factors. Three groups including 24 factors have been selected as candidates.
To test these candidates, a system has been developed in which the induction of pluripotency can be detected by marker gene expression (Fig. 2). This system utilized Fbx15 that is specifically expressed in ESCs and early embryos, but is dispensable for self‐renewal of ESCs and development (Tokuzawa et al. 2003). The βgeo cassette (a fusion of β‐galactocidase and the neomycin resistant gene) has been inserted into mouse Fbx15 genes by homologous recombination. Embryonic stem cells homozygous for βgeo knock‐in (Fbx15 βgeo/βgeo) were found to be resistant to an extremely high concentration of G418 (up to 12 mg/mL); whereas somatic cells derived from Fbx15βgeo/βgeo mice were sensitive to the selection. Even partial induction of pluripotency would be expected to make somatic cells resistant to normal concentration of G418 (0.3 mg/mL).
Figure 2.
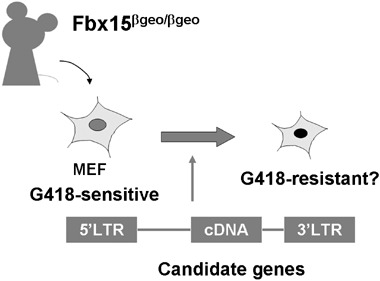
Evaluation system for the candidate factors. To prove pluripotency is a time‐ and labour‐consuming process. Therefore, a simple method to evaluate the candidate factors is essential to identify pluripotency‐inducing factor(s).
The candidate genes were introduced into Fbx15βgeo/βgeo mouse embryonic fibroblasts (MEF) by retrovirus‐mediated transfection and these cells were cultured in ESC medium containing G418. No G418‐resitant colonies were obtained with any single factor. However, by combining four factors (Oct3/4, Sox2, c‐Myc and Klf4), multiple G418‐resitant colonies were obtained (Fig. 3). These cells showed morphological and proliferation characteristics similar to ESCs. Furthermore, when transplanted into nude mice, these embryonic stem‐like cells produced teratomas containing various tissues of the three germ layers. These cells were designated iPS, induced pluripotent stem cells. These data demonstrated that pluripotent cells can be generated from fibroblast cultures with a few defined factors (Takahashi & Yamanaka 2006). The introduction of the three factors excluding Sox2 results in cells somewhat similar to ESCs in morphology and proliferation, but lacking pluripotency (Fig. 3). Combination of the four factors (Oct‐3/4, Sox2, c‐Myc and KLF4) thus appears to be the minimum requirement for induction of pluripotent iPS cells from mouse fibroblast cultures. Fbx15‐selected iPS cells, however, are not able to contribute to adult chimaeric mice. DNA microarray analyses have shown significant difference in global gene expression patterns between Fbx15‐selected iPS cells and ESCs. In addition, the promoter regions of Oct‐3/4 and Nanog remained methylated in Fbx15‐selected iPS cells. These data collectively showed that only partial re‐programming occurs in Fbx15‐selected iPS cells. To overcome this issue, Nanog was utilized as a selection maker to generate iPS cells (Maherali et al. 2007; Okita et al. 2007; Wernig et al. 2007). Nanog might serve as a better marker as it is more tightly associated with pluripotency in expression and function than Fbx15 (Pan & Thomson 2007). All the three groups showed that Nanog‐selected iPS cells were nearly indistinguishable from ESCs in gene expression and epigenetic status. Furthermore, Nanog‐selected iPS cells can produce adult and germline‐competent chimaeras.
Figure 3.
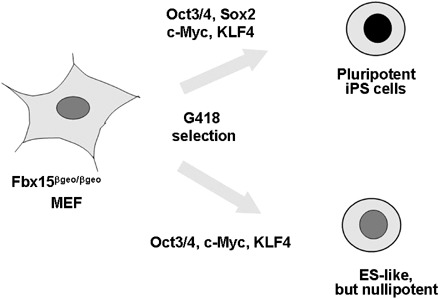
The induction of induced pluripotent stem (iPS) cells by Oct‐3/4, Sox2, c‐Myc and KLF4. Retroviral transfection of Oct‐3/4, Sox2, c‐Myc and KLF4 into mouse fibroblasts results in the induction of pluripotent iPS cells. The introduction of three factors devoid of Sox2 results in cells with embryonic stem‐like morphology, but they are nulipotent. Nanog is dispensable for induced pluripotent stem (iPS) cell induction.
HOW DO OCT3/4, SOX2, C‐MYC, AND KLF4 INDUCE PLURIPOTENCY IN FIBROBLAST CULTURES?
Embryonic stem cells are similar to tumour cells in that they are immortal and proliferate rapidly. In addition, ESCs can form tumours (teratomas) when transplanted into immune‐deficient mice. Embryonic stem cells, in this sense, behave like ‘transformed’ cells. This transformed phenotype, however, can be reversed when ESCs are injected into blastocysts in the course of forming apparently normal chimaeric mice. This reversible transformation takes place during the course of in vitro culture of inner cell mass. Some genes, such as E‐Ras, are activated in this process (Takahashi et al. 2003).
Taking this into account, induction of iPS cells from somatic cells should also require transformation by the two tumour‐related gene products, c‐Myc and KLF4. The Myc protein can elicit various aspects of transformation (Adhikary & Eilers 2005). However, it also elicits p53‐dependent apoptosis. KLF4 might be required to suppress p53 and c‐Myc‐induced apoptosis (Rowland & Peeper 2006). KLF4, in contrast, activates p21 and suppresses proliferation. The Myc protein can alleviate this cytostatic effect of KLF4 by suppressing p21. The balance between c‐Myc and KLF4 might be a critical for transformation in iPS cells.
Pluripotent stem cells have open and active chromatin structures (Meshorer et al. 2006). Myc proteins probably loosen the chromatin structure of somatic cells by binding to numerous sites throughout the genome and by recruiting multiple histone acetylase complexes (Knoepfler et al. 2006). Consistent with this model, iPS cells show acetylated histone in the promoter regions of several ESC‐specific genes (Takahashi & Yamanaka 2006). The forced expression of c‐Myc and KLF4 alone would result in generation of tumour cells, but not pluripotent stem cells. It is probably Oct‐3/4 and Sox2 that would direct cell fate from tumour cells to embryonic stem‐like cells. Effects of c‐Myc on the chromatin structure should enable Oct‐3/4 and Sox2 to bind to their target genes. KLF4 may also function as a co‐factor of Oct‐3/4 and Sox2 (Nakatake et al. 2006). It has been shown that retroviral vectors are silenced favourably in iPS cell induction by preventing continuous overexpression of c‐Myc and KLF4 in pluripotent stem cells (Yao et al. 2004); the more cells become similar to ESCs, the more the transgene expression of the four factors should be silenced. This repression should function favourably in iPS cell induction by preventing the continuous overexpression of c‐Myc and KLF4.
CONCLUSION
This review has provided an overview of the induction of mouse iPS cells by Oct‐3/4, Sox2, c‐Myc and KLF4. The four factors, however, can not explain every aspect of iPS cell induction. Among fibroblasts that express all of the four transgenes, only a small portion (less than 1%) can become iPS cells (Fig. 4). In addition, currently, the use of a retroviral transfection system is indispensable for iPS cell induction, which represents a severe disadvantage of iPS cells in terms of safety issue; around 20% of iPS cell‐derived mice developed tumours (Okita et al. 2007). By adding other factor(s), it may be able to induce iPS cells, with higher efficiency and without need for retroviruses. Identification of such factor(s) may therefore eventually lead to the generation of human iPS cells, which may thus lead to important advances in regenerative medicine and new drug discoveries.
Figure 4.
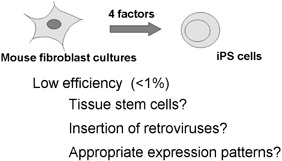
Low efficiency of induced pluripotent stem (iPS) cell induction. Among fibroblasts expressing all four transgenes, less than 1% becomes iPS cells. The low efficiency may suggest that tissue stem cells co‐existing in fibroblast culture are the origin of iPS cells. Alternatively, the activation of additional genes by retroviral integration may be required. Another possible mechanism is that induction of iPS cells requires specific amounts and patterns of expression of the four factors.
REFERENCES
- Adhikary S, Eilers M (2005) Transcriptional regulation and transformation by Myc proteins. Nat. Rev. Mol. Cell Biol. 6, 635–645. [DOI] [PubMed] [Google Scholar]
- Cowan CA, Atienza J, Melton DA, Eggan K (2005) Nuclear reprogramming of somatic cells after fusion with human embryonic stem cells. Science 309, 1369–1373. [DOI] [PubMed] [Google Scholar]
- Doetschman T, Gregg RG, Maeda N, Hooper ML, Melton DW, Thompson S, Smithies O (1987) Targetted correction of a mutant HPRT gene in mouse embryonic stem cells. Nature 330, 576–578. [DOI] [PubMed] [Google Scholar]
- Evans MJ, Kaufman MH (1981) Establishment in culture of pluripotential cells from mouse embryos. Nature 292, 154–156. [DOI] [PubMed] [Google Scholar]
- Hooper M, Hardy K, Handyside A, Hunter S, Monk M (1987) HPRT‐deficient (Lesch‐Nyhan) mouse embryos derived from germline colonization by cultured cells. Nature 326, 292–295. [DOI] [PubMed] [Google Scholar]
- Knoepfler PS, Zhang XY, Cheng PF, Gafken PR, McMahon SB, Eisenman RN (2006) Myc influences global chromatin structure. EMBO J. 25, 2723–2734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maherali N, Sridharan R, Xie W, Utikal J, Eminli S, Arnold K, Stadtfeld M, Yachechko RJT, Jaenisch R, Plath K, Hochedlinger K (2007) Directly reprogrammed fibroblasts show global epigenetic remodelling and widespread tissue contribution. Cell Stem Cell 1, 55–70. [DOI] [PubMed] [Google Scholar]
- Martin GR (1981) Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proc. Natl Acad. Sci. USA 78, 7634–7638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meshorer E, Yellajoshula D, George E, Scambler PJ, Brown DT, Misteli T (2006) Hyperdynamic plasticity of chromatin proteins in pluripotent embryonic stem cells. Dev. Cell 10, 105–116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller RA, Ruddle FH (1976) Pluripotent teratocarcinoma‐thymus somatic cell hybrids. Cell 9, 45–55. [DOI] [PubMed] [Google Scholar]
- Nakatake Y, Fukui N, Iwamatsu Y, Masui S, Takahashi K, Yagi R, Yagi K, Miyazaki J, Matoba R, Ko MS, Niwa H (2006) Klf4 cooperates with Oct3/4 and Sox2 to activate the Lefty1 core promoter in embryonic stem cells. Mol. Cell. Biol. 26, 7772–7782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okita K, Ichisaka T, Yamanaka S (2007) Generation of germline‐competent induced pluripotent stem cells. Nature 448, 313–317. [DOI] [PubMed] [Google Scholar]
- Pan G, Thomson JA (2007) Nanog and transcriptional networks in embryonic stem cell pluripotency. Cell Res. 17, 42–49. [DOI] [PubMed] [Google Scholar]
- Rowland BD, Peeper DS (2006) KLF4, p21 and context‐dependent opposing forces in cancer. Nat. Rev. Cancer 6, 11–23. [DOI] [PubMed] [Google Scholar]
- Tada M, Tada T, Lefebvre L, Barton SC, Surani MA (1997) Embryonic germ cells induce epigenetic reprogramming of somatic nucleus in hybrid cells. EMBO J. 16, 6510–6520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tada M, Takahama Y, Abe K, Nakatsuji N, Tada T (2001) Nuclear reprogramming of somatic cells by in vitro hybridization with ES cells. Curr. Biol. 11, 1553–1558. [DOI] [PubMed] [Google Scholar]
- Takahashi K, Mitsui K, Yamanaka S (2003) Role of ERas in promoting tumour‐like properties in mouse embryonic stem cells. Nature 423, 541–545. [DOI] [PubMed] [Google Scholar]
- Takahashi K, Yamanaka S (2006) Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676. [DOI] [PubMed] [Google Scholar]
- Thomson JA, Itskovitz‐Eldor J, Shapiro SS, Waknitz MA, Swiergiel JJ, Marshall VS, Jones JM (1998) Embryonic stem cell lines derived from human blastocysts. Science 282, 1145–1147. [DOI] [PubMed] [Google Scholar]
- Tokuzawa Y, Kaiho E, Maruyama M, Takahashi K, Mitsui K, Maeda M, Niwa H, Yamanaka S (2003) Fbx15 is a novel target of Oct3/4 but is dispensable for embryonic stem cell self‐renewal and mouse development. Mol. Cell. Biol. 23, 2699–2708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wernig M, Meissner A, Foreman R, Brambrink T, Ku M, Hochedlinger K, Bernstein BE, Jaenisch R (2007) In vitro reprogramming of fibroblasts into a pluripotent ES cell‐like state. Nature 318, 318–324. [DOI] [PubMed] [Google Scholar]
- Yao S, Sukonnik T, Kean T, Bharadwaj RR, Pasceri P, Ellis J (2004) Retrovirus silencing, variegation, extinction, and memory are controlled by a dynamic interplay of multiple epigenetic modifications. Mol. Ther. 10, 27–36. [DOI] [PubMed] [Google Scholar]
- Yu J, Vodyanik MA, He P, Slukvin II, Thomson JA (2006) Human embryonic stem cells reprogram myeloid precursors following cell–cell fusion. Stem Cells 24, 168–176. [DOI] [PubMed] [Google Scholar]


