Abstract
Abstract. In the industrialized world, cardiovascular disease alone is responsible for almost half of all deaths. Many of the conditions can be treated successfully with surgery, often using transplantation techniques; however, autologous vessels or human‐donated organs are in short supply. Tissue engineering aims to create specific, matching grafts by growing cells on appropriate matrices, but there are many steps between the research laboratory and the operating theatre. Neo‐tissues must be effective, durable, non‐thrombogenic and non‐immunogenic. Scaffolds should be bio‐compatible, porous (to allow cell/cell communication) and amenable to surgery. In the early days of cardiovascular tissue engineering, autologous or allogenic cells were grown on inert matrices, but patency and thrombogenicity of grafts were disappointing. The current ethos is toward appropriate cell types grown in (most often) a polymeric matrix that degrades at a rate compatible with the cells’ production of their own extracellular matrical proteins, thus gradually replacing the graft with a living counterpart. The geometry is crucial. Computer models have been made of valves, and these are used as three‐dimensional patterns for mass‐production of implant scaffolds. Vessel walls have integral connective tissue architecture, and application of physiological level mechanical forces conditions bio‐engineered components to align in precise orientation. This article reviews the concepts involved and successes achieved to date.
INTRODUCTION
In the ageing populations of the industrialized world, cardiovascular disease (CVD) is a predominant killer. In Europe as a whole CVD causes 4 million deaths per year (48% of all deaths), 1.5 million of which are within the states of the European Union. It is the main cause of death in women in all European countries, and the main cause of death in men in all countries except France. In central and eastern Europe incidence of CVD is on the rise (Rayner & Petersen 2000). Changes in lifestyle practices can improve these statistics over time, but at present large numbers of CVD patients rely on treatment, often surgery with transplantation techniques, to alleviate their suffering and improve their quality of life. Demands on replacement cardiovascular components greatly outstrip availability of human‐donated organs, so it is attractive to seek synthetic counterparts, whether non‐biological, natural biological or engineered constructs (which combine the appropriate cell types grown with suitable supportive matrices, often elastic, porous, polymers). Tissue engineering in medicine integrates the natural history of tissue development, its biology and its physiology, cognizant of mechanical demands on body structures. Normal cardiovascular tissues, particularly, have evolved with their geometry and architecture determined by conditions of perpetual and changing operational forces. Consideration of composition of the natural tissues is essential before attempting to engineer an equivalent construct.
HEART VALVE AND ARTERIAL HISTOLOGY
Heart valves
Although embryologically derived from the proximal heart chamber/great vessel, valves have no true muscular component. In life, hydrolic forces alone, of blood streaming under pulsating pressure, close the appropriate valves and prevent retrograde flow. Valve leaflets are three‐layered sheets, with endothelium on each face, supported by an integral dense collagenous, elastic‐containing lamina fibrosa, whose cells express alpha‐smooth muscle actin, and are thus classified as myofibroblasts (Taylor et al. 2000).
Arteries
Walls of arteries have three layers, the tunicae intima, media and adventitia. The luminal surface is the intima, composed of a single layer of endothelium which is non‐thrombogenic. The intima and the media are separated by a fine, mesh‐like internal elastic lamina. Interspersed are occasional fibroblasts and smooth muscle‐like cells. Exact phenotypes within this population are of interest, as the cells’ contractile components are not those of arterial smooth muscle cells, but resemble a myofibroblast population by expressing alpha‐smooth muscle actin (Becker et al. 2000). Yet, these cells can contribute to production of extracellular matrix; thus, there is some plasticity of phenotype between smooth muscle cells, myofibroblasts and fibroblasts. Fibroblasts express the myofibroblast phenotype under the influence of transforming growth factor β (Grinnell & Ho 2002).
Composition of the media varies progressively away from the heart. In the aorta, the media is thick with concentric fenestrated elastic sheets separated by fibroblast‐produced collagen; smooth muscle cells are present, but progressively, the further away from the heart the greater the proportion of smooth muscle to elastin. In arterioles, the media may be as little as six concentric smooth muscle cells or less in depth. The three‐dimensional tracks of smooth muscle cells, fibroblasts and collagen fibres with respect to the long axis of vessels, is in alternating left‐ and right‐handed spirals, they are not annular. Due to the resultant extensibility, large increments of luminal force produce little increase in vessel length (Williams et al. 1973). This is a crucial point for tissue engineers to remember as it has profound implications in attempts to synthesize a strong, flexible bio‐engineered artery. The outer circumference of an artery is bounded by the adventitia, separated from the media by an external elastic lamina. This is robust collagenous connective tissue, defining the integrity of the vessel and able to withstand its rhythmic contractions and expansions.
Heart valve, aortic and arterial engraftment
Narrowing of heart valve orifices and valve defects can be congenital, degenerative, associated with inflammatory conditions or can be idiopathic. Between 1990 and 2000 Gudi & Sheppard (2003) reported common categories of valve disease (Table 1). Calcific valvular disease is common, rheumatic valvular disease less so. Valve replacement is a customary technique for end‐stage valvular heart disease (Herzog et al. 2002), and autologous tissue grafting has been used in valve repair for many years (Williams et al. 1973). In the Gudi & Sheppard (2003) study, 11.8% of cases concerned replacement valves.
Table 1.
Aortic valve and mitral valve pathology (reproduced with the kind permission of Dr M. N. Sheppard and M. A. Gudi, Imperial College London)
| % | |
|---|---|
| Aortic valve pathology | |
| Degenerative calcification | 43.7 |
| Aortic regurgitation | 16.1 |
| Rheumatic aortic stenosis | 8.3 |
| Bacterial endocarditis | 3.9 |
| Congential conditions | |
| Bicuspid aortic valve | 22.2 |
| Aortic stenosis | 2.9 |
| Subvalvular aortic stenosis | 2.7 |
| Supravalvular aortic stenosis | rare |
| Mitral valve pathology | |
| Mitral stenosis | 35.9 |
| Mitral regurgitation | 64.1 |
Pulmonary and tricuspid valve pathologies are less common.
Most common disorders of the arterial system are coronary artery atheroma with occlusion, aneurysm of the ascending aorta and coronary artery occlusion. Coronary artery bypass grafting using the autologous mammary artery or saphenous vein is common (Mann & West 2001; Yaginuma et al. 2001). Complete aortic root replacement can be performed in cases of narrow aortic annulus (Urbanski 2002).
Tissue engineered constructs are necessary due to lack of sufficient natural tissue, mismatch of exact anatomy or poor patency. The potential clinical application of synthetic components determines the choice of support matrix and cell population; valves, in particular, have intricate three‐dimensional shapes that must be duplicated. The major decision is to select a type of implant that will provide the most advantageous result for the patient. It should be biocompatible, amenable to surgery, effective, reliable, long lasting, have low thromboembolic potential and be non‐immunogenic. The giant step between research laboratory and hospital shelf must be considered. World‐wide demand is such that biomaterial production techniques must be reproducible on a mass‐market scale. Implants need to be readily available and able to withstand packaging, shipping and storage. Stereolithic 3‐D computer models of valves have molten thermoplastic deposited under computer control (Stratasys Inc., Berlin, Germany) to the exact shape (Morsi 2002). For vessels, patches can be made (McCulloch et al. 2003), or tubes, either grown in culture around a cylindrical mandrel (L’Heureux et al. 1998), or the biomaterial grown flat, then wrapped (Huynh et al. 1999). In the cardiovascular system, the enduring changes in mechanical force are central to the requirements of design and manufacture of potential prostheses. Ideally, the scaffolds should be of resorbable fabric and provide physical support until local cells produce their own extracellular matrix proteins.
Non‐biological prostheses
Although a surrogate of composition as close as possible to the natural tissue would seem preferable, non‐biological prostheses have been clinically effective to a degree. Mechanical heart valves have been used for many years (Feindel & David 1993) and, currently, success has been reported using both bio‐stable and novel durable but non‐bio‐stable polyurethanes (Butterfield et al. 2001) where there has been improved hydrodynamic function and lower energy loss in pulsatile flow. Wholly acellular synthetic materials have been used in vascular grafts, but small diameter plastic polymer arterial implants are subject to occlusion (Huynh et al. 1999).
Wholly biological surrogates
In total contrast to inorganic implants, a variety of cellular complexes in the absence of artificial or modified‐biological extracellular matrix have been tested in animals. Working in a lamb model, Shinoka et al. (1997) constructed heart valves using cells from femoral arteries, which developed into acceptable leaflets. The group found that arterial myofibroblasts were preferable from dermal fibroblasts for this purpose.
Fresh pericardial tissue has been fashioned into small diameter arterial grafts in a dog model (Kim et al. 2002). In a technically ingenious experiment in vitro L’Heureux et al. (1998) used an acellular collagenous membrane as temporary tubular substructure for human vascular smooth muscle cells. Human fibroblasts were in turn wrapped over this ‘media’. The neo‐vessel was turned inside‐out, endothelial cells cultured on the free surface of the smooth muscle cells and the structure re‐inverted generating a three‐layered vessel. The fibroblasts produced extracellular matrix proteins; von Willebrand factor and desmin, were made by the endothelium and smooth muscle cells, respectively. A burst strength of 2000 mmHg was reported – comparable with that of human vessels.
BIO‐ENGINEERED TISSUE OF CELLS ON A SCAFFOLD
In true tissue engineering, biological matrix fabric is seeded with cells that would be found naturally in the cardiovascular system; they must proliferate, be functional, and self‐assemble their extracellular matrix. In 1973, Williams et al. (1973) used tissue from the vena cava arranged on a woven silicone rubber frame to produce valve cusp replacements. They used a pulse duplicator to determine the optimal geometry of the valve, and their constructs were an improvement on contemporary mechanical prostheses.
An early attempt to construct a cells‐plus‐scaffold blood vessel was in 1986 (Weinberg & Bell 1986). Bovine smooth muscle cells in a collagen culture medium were cast around an annular mould. A non‐absorbable plastic Dacron® (Wilmington, DE, USA) mesh sleeve was rolled over for mechanical support, followed by adventitial cells. Constructs were lined with endothelium, but the study had not incorporated elastin in the matrix. Models made with only one Dacron® mesh ruptured at less than 10 mmHg and further layers of Dacron® made the neo‐vessel stronger but too stiff. The Dacron® mesh, like Williams’ group silicone rubber frame, was a physical support for the neo‐tissue only, with no pretension toward contributing either biochemically or physiologically to tissue synthesis. L’Heureux's wholly biological model more than 10 years later improved on this.
In subsequent years, advances have been made in the principle of using native cells with specifically created polymer lattices (Table 2); the cells proliferate and populate the fabric. Matrices need an interconnected honeycomb geometry or pore structure for expansion of the cell population. Common techniques for production of polymer scaffolds are weaving, meshing, porogen leaching, emulsion freeze‐drying, expansion in high pressure gas, 3D‐printing and phase separation (Shi et al. 2002). There are hydrolytic degradable substrates (Shinoka et al. 1996; Nkere et al. 1998; Zund et al. 1998; Hoerstrup et al. 2000; Sodian et al. 2000), biologically modified native tissues (Huynh et al. 1999; McCulloch et al. 2003) and collagen sponges non‐crosslinked or crosslinked with stabilizing molecules (Chevallay et al. 2000; Roche et al. 2001; Harris & Eastwood 2003).
Table 2.
Examples of synthetic polymers used as scaffolds in tissue engineering techniques
| • Poly beta‐hydroxybutrate (Nkere et al. 1998) |
| • Poly(carbonate‐urea)urethane (Rashid 2003) |
| • Polydioxanone (Greisler et al. 1987) |
| • Polyglycolic acid (Shinoka et al. 1996; Zund et al. 1998) |
| • Polyglycolic acid/poly(l‐lactic acid) copolymer (Mahoney & Saltzman 2001) |
| • Polyglycolic acid/polyhydroxyalkanoate (Shum‐Tim et al. 1999) |
| • Polyethylene oxide/polypropaline oxide (Bots et al. 1986) |
| • Polyhydroxyalkanoate (Sodian et al. 2000) |
| • Poly(l‐lactic acid) (Shi et al. 2002) |
| • Poly(l‐lactic‐coglycolic acid) (Shi et al. 2002) |
| • Polyurethane (Butterfield et al. 2001) |
Ideally, polymer matrices require a woven, meshed or honeycomb‐like structure with interconnecting pores to house the proliferating cells and their matrix.
Hydrolytic degradable scaffolds
Polyglycolic acid (PGA) is biocompatible and biodegradable and it has been used as substrate in an ovine model of autologous heart valve leaflet replacement (Shinoka et al. 1996). Complexes were tissue‐engineered by first seeding fibroblasts then endothelial cells on to the polymer. The synthetic valves were grafted into lambs, where they expressed Factor VIII, elastic filaments and collagen. The ethos for ultimate biodegradability was that, upon implantation, such a framework would gradually deteriorate, provoke little or no immune response in the host and minimize any potential chronic inflammatory reaction. Such a tissue‐engineered PGA/cell complex has the advantage of being able to grow and potentially to remodel and repair.
In 1998, PGA mesh sheets were used as a substrate for seeding human fibroblasts and human endothelial cells (Zund et al. 1998). The fibroblasts spread through the mesh, became attached to the polymeric fibres, proliferated and formed intercellular bridges. Endothelial cells configured into a monolayer over the fibroblasts and did not invade the matrix as capillary‐like structures. Subsequently, Sodian et al. (2000) produced trileaflet heart valves from sheep carotid artery myofibroblasts grown on polyhydroxyalkanoate, which in a pulsatile flow bioreactor opened and closed synchronously. Hoerstrup et al. (2000) also manufactured valves with non‐woven PGA mesh coated with a thin layer of poly 4‐hydroxybutrate. Again they used pulsatile forces, here to improve valve architecture.
In calf, poly beta‐hydroxybutrate has been used to close the pericardium after cardiopulmonary bypass. At investigatory reoperation, the patch material contained pronounced macrophage infiltration, but no regenerative mesothelium (Nkere et al. 1998).
Biologically modified native tissues
For heart valves, a technique for implanting a complex composed of combined fresh non‐vascular tissue and cells was pioneered by Manothaya et al. (1980). Dura mater was employed for frame‐supported valves, and short‐term results were reported as being satisfactory at that time.
A porcine cell‐free collagen layer has been used to engineer a narrow diameter, non‐thrombogenic artery for implantation (Huynh et al. 1999). To reduce thrombogenicity, luminal surfaces were treated with heparin‐benzalkonium chloride crosslinked with 1‐ethyl‐3‐(3‐dimethylaminopropyl) carbodiimide hydrochloride, then bovine type I collagen. These acellular grafts were implanted into rabbits, and even though they consisted of porcine and bovine collagen components, antigenicity was minimal. There was extensive rabbit cell infiltration into the grafts, the implants were reactive to experimental vascular agonists, had a non‐thrombogenic surface, and exhibited burst resistance up to 1000 mmHg.
Collagen sponge matrices
Collagen sponges non‐crosslinked or crosslinked with diphenylphosphorylazide (Chevallay et al. 2000) seeded with bovine foetal chrondrocytes have been used to reconstruct cartilaginous tissues (Roche et al. 2001), and non‐crosslinked or crosslinked with chitosan and chondroitin sulphate (Harris & Eastwood 2003) have been used with porcine aortic wall cells. Sponges are made from solutions of extracted calf skin collagen and freeze dried, resulting in a porous matrix with a mean pore size 58 ± 24 µm (Perouse SARL, Lyon, France). When similar sponges were seeded with porcine aortic myofibroblasts, cells were infiltrative and proliferative (Sarraf et al. 2002a), and porosity of the sponge provided excellent conduits for cell migration. After 4 weeks in culture, myofibroblasts had progressed to the interior, while those at the surface were in stratified layers, four to six cells deep (Sarraf et al. 2002b). Surface myofibroblasts generated sparing amounts of matrical proteins (Fig. 1), whereas central sponge zones had dense aggregates of banded collagen and elastic fibres (Fig. 2), emanating from well‐spaced cells. This is in accord with their normal behaviour in a native tissue, where cells are separated by considerable extracellular components and myofibroblasts readily change direction of metabolic activity (Sarraf et al. 1998; Guma et al. 2001). In addition, a variety of non‐synthetic polymer carrier materials of natural origin can be used as bio‐engineering matrices (Table 3).
Figure 1.

Transmission electron micrograph of porcine aortic wall myofibroblasts at the surface (arrow) of a collagen sponge matrix. The cells are spindle‐shaped with their long axes parallel to the surface of the construct. M is a mitotic figure. Bar = 4 µm.
Figure 2.

Transmission electron micrograph of extracellular matrix fibres produced by cells at the centre of a collagen matrix (collagen, solid arrow; elastin fine arrow). Bar = 0.6 µm.
Table 3.
Examples of carrier materials of natural origin used as scaffolds in tissue engineering techniques
| • Decellularised small intestine submucosa |
| • Collagen sponges |
| • Collagen gel |
| • Alginate |
| • Fibrin |
| • Hyaluronic acid |
| • Gelatin |
| • Collagen‐glycosaminoglycan matrices |
BIOPOLYMERS AND THE FUTURE
Inert biomedical grade materials for implantation have been defined as ‘first generation’ and those of the second generation as having controlled chemical self‐breakdown and resorption (Hench & Polak 2002). Extrapolation of properties of these materials lies in development of tissue substitutes that are auxiliary to, and able to integrate with, the host body. Stimulatory proteins can be bound to polymer surfaces; for example, polyglycolic acid/poly(l‐lactic acid) co‐polymer has been molecularly modified to enhance local levels of nerve growth factor (Mahoney & Saltzman 2001). Subcellular localization of gene products can be demonstrated by techniques such as electron microscopy, immunocytochemistry (Sarraf 2000) and in situ hybridization. Care needs to be exercised, however, with potential exogenous growth‐factor stimulated cell proliferation. In normal wound healing there is limited and reversible hyperplasia. Excessive stimulation of cell proliferation could all too easily result in uncontrolled neoplasia.
MECHANICAL CONDITIONING AND CARDIOVASCULAR NEO‐TISSUE
In vivo, architecture, development and function of the cardiovascular tissues are intimately related to their physical environment. Fibroblasts are sensitive to forces applied to them and integrate both mechanical and biochemical signals for growth, maintenance and repair. Pulsatile blood flow gives rise to a radial and also to a longitudinal strain; a steady state flow with no pulsatile component causes only shear stress on luminal wall surfaces. This results in endothelial cells aligning and bearing in the direction pertinent to the shear. Axial loading causes pure longitudinal strain.
Bioreactors have been used in cardiovascular tissue engineering since prototype valves and blood vessels were constructed, but initially the role of the bioreactor was as a culture vessel rather than for mechanical conditioning. Recently, however, forces derived by the cells have been measured (Eastwood et al. 1994; Brown et al. 1998; 1998a, 1998b), and external forces have been applied to structures, resulting in orientated tissues (McCulloch et al. 2003).
Culture force monitor and tensioning‐culture force monitor
Extensile cells create forces and also respond to stimuli applied to them from their surroundings. Such forces in non‐tubular constructs have been accurately measured on a culture force monitor (CFM) and have been applied and measured by a tensioning‐culture force monitor (t‐CFM) (Eastwood et al. 1998a).
Culture force monitor
A CFM (Fig. 3) was developed in 1994 (Eastwood et al. 1998a) with a force transducer coupled directly to a computer via an analogue‐to‐digital converter. It is able to measure mechanical force in different cell types in a three‐dimensional environment on a wide range of scaffold materials. It is sensitive enough to digitally record and measure distances (down to 10−6 m) covered and forces produced (average of 10−5 Newtons) by individual fibroblasts suspended in a collagen matrix. The majority of force is generated by changes in cell shape and cytoskeletal rearrangement (Brown et al. 1996). Forces are derived from fibroblast attachment and movement in the lattice (Eastwood et al. 1996). In recessive dystrophic epidermolysis bullosa (RDEB), forces of normal fibroblasts and RDEB fibroblasts have been quantified and compared. RDEB fibroblasts are hypercontractile, thus responsible for the symptoms of the disease (Talas et al. 1997); similarly, the CFM has shown that burn fluid stimulates fibroblasts to contract (Wilson et al. 1997). An adaptation of the CFM measuring forces in tissue cohesion after injury, demonstrated levels of fibroblast‐mediated adhesion in vitro (Cacou et al. 1996). Formation of post‐surgical adhesions are also important in human primary and re‐operative pericardium (Nkere et al. 1995). Also, the CFM can measure cells’ responses to molecules such as therapeutic drugs and/or growth factors.
Figure 3.

Plan of the structure of the culture force monitor indicating relationships of the culture well, force transducer and computer.
Tensioning‐culture force monitor
The t‐CFM (Fig. 4) is a derivative of the CFM for application of precise, physiological level mechanical loads, to be applied to three‐dimensional fibroblast populated lattices (Eastwood et al. 1998a). For the t‐CFM, a microstepping motor was added to the existing monitoring devices of the CFM so that cells could be loaded in matrices, forces of predetermined magnitude engaged, and cellular responses in terms of both matrix tension and biochemical constitution measured.
Figure 4.

Plan of the structure of the tensioning‐culture force monitor showing the addition of a microstepping motor.
When cyclical loads are applied to produce unidirectional strain, fibroblasts that had been spherical at seeding adopt the characteristic spindle shape and align parallel to the loading axis of the neo‐tissue. Minute unidirectional loads with specific loading patterns cause striking, predictable and repeatable cell alignment (Eastwood et al. 1998a). Mechanical conditioning occurs, fibroblasts rank themselves along lines of maximum principle isostrain (Eastwood et al. 1998b) and orientate in consistent responsive patterns. This has been shown in dermal fibroblasts (Brown et al. 1998); across restrained gels fibroblasts migrate more energetically than across untethered gels (Porter et al. 1998), modulated by the geometry of culture and strain contours as well as magnitude and direction of loading.
Biochemical changes occur in fibroblasts in response to application of force. Of the matrix metalloproteases (MMP)s, MMP‐9 synthesis is more sensitive to tension than MMP‐2, and stromelysin activity decreases with tension (Prajapati et al. 2000a). Quantitative reverse transcription polymerase chain reaction (RT‐PCR) for MMP‐1, MMP‐2 and MMP‐3 has revealed 2–4‐fold increases in non‐aligned strain zones relative to aligned strain zones during contraction in wound healing (Mudera et al. 2000).
Pulsatile bioreactors
For tissue engineering of valves, mechanical conditioning has been applied in the form of pulsatile flow through the centre of cell‐seeded constructs (Williams et al. 1973; Hoerstrup et al. 2000; Zeltinger et al. 2001). These bioreactors have three common components, a bioreaction chamber, a means of generating pulsatile flow and a culture media exchange mechanism.
For long‐term culture of vascular tissue surrogates, a tubular pulsatile bioreactor is required. The tissue‐engineered construct is not a planar 3‐D sheet, but has either been grown on an annular mould or has been rolled, sutured, and/or glued with an effective fibrin sealant. The neo‐vessel now has a luminal surface and a circular exterior surface, and cell types are seeded appropriately – endothelium on the inside, fibroblasts/smooth muscle cells centripetal to this. Force transducers and microstepping motors are attached and multiple computer‐controlled loading regimes can be used to condition the cell/scaffold tubular complex.
Multi‐cue bioreactor
The multi‐cue bioreactor (M‐CB) (McCulloch et al. 2003) has a range of prompts or cues that can be delivered – pulsatile flow, non‐pulsatile flow, induced longitudinal strain, via a hermetically sealed electric motor linked to a rack and pinion straining system (Fig. 5). The main pulses are driven by computer controlled microstepper motor units, programmed through X‐Ware® software (Parker Hannifin, Poole, UK); parameters include acceleration, velocity and distance of rotation. There is two‐way perfusion ensuring sufficient circulation of cell culture media. Mechanical stimuli are applied and cells and matrix orientate, changing from a mechanically unstructured culture into a viable, biologically sufficient entity (Brown et al. 1998; Eastwood et al. 1994; 2000a, 2000b). Histology reminiscent of conditions in vivo has been achieved with porcine aortic wall cells (Fig. 6) grown on a small intestinal submucosa matrix® (Cook Biotech Inc., IN, USA). Alternatively, the M‐CB can be employed to determine varying mechanical properties of tissue by performing in situ non‐destructive tensile testing at key intervals during the conditioning process.
Figure 5.

Plan of the structure of the multicue bioreactor. The tissue construct cassette holds the tubular neo‐vascular specimen and the pulsatile pump delivers culture medium at physiological forces comparable with those produced by the heart.
Figure 6.
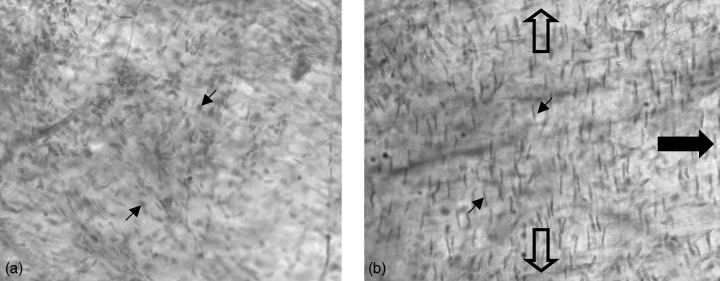
En face preparation of porcine aortic wall cells on a small intestine submucosa matrix in static culture for 7 days (a) and bioreacted in the M‐CB for 7 days under pulmonary artery conditions (b). Direction of culture media flow, large closed arrow; direction of circumferential strain due to applied pressure, open arrows; cell nuclei, small arrows. (Light micrograph, haematoxylin and eosin stained, original magnification × 40.)
CELL ORIGIN AND GRAFT SUCCESS
To manufacture an effective, durable bio‐engineered composite graft for human implantation, the biological source of the seeded cells is of the highest importance. To avoid immune system complications, ideally cells would be autologous. Pragmatically, this might not be possible (the time required for cells to proliferate into a sufficient culture is substantial) thus, preformed grafts of cells from other sources might be usable.
Autologous cells have many advantages, not least that ethical considerations are not incurred. Allogenic cell sources are used experimentally for cardiovascular implants (Shinoka et al. 1996; Hoerstrup et al. 2000), but for humans even use of human cells from a stock for in vitro investigation poses ethical constraint. Allogenic dermal fibroblasts have been shown to be acceptable immunologically, and a source is, to some extent, available from human foreskin (Harding et al. 2002), but dermal fibroblasts in potential vessel prostheses have proven inferior to vascular fibroblasts (Shinoka et al. 1997). Similarly, human umbilical cord endothelium is a source of suitable human cells (Barker & Wagner 2002), but neonatal fibroblasts and embryonic fibroblast stem cells for allogenic surgical purposes, although useful animal experimental or veterinary tools, cannot be considered for the purpose of making human grafts.
Populating a bio‐engineered tissue scaffold with adult human stem cells (autologous or allogenic) might be possible. The potential for stimulating maturation of adult stem‐cell progeny along predetermined lines has been demonstrated (Alison et al. 2000), and it has been shown (in mice at least) that myofibroblasts in the colonic lamina propria have been derived from bone marrow stem cells (Brittan et al. 2002). Harvesting and transplanting human bone marrow are commonly performed, and selectively directing differentiation in an engrafted, bio‐engineered scaffold could solve problems of effectiveness and durability of synthetic connective tissue surrogates.
CONCLUSION
Ideal tissue‐engineered cardiovascular replacements must possess all properties of the natural tissue. Scaffolds, whether synthetic or biological, must satisfy anatomical, biochemical and mechanical demands in addition to being non‐immunogenic.
Appropriate mechanical stresses and strains can be applied to valve‐ and blood vessel‐constructs in vitro to condition them prior to surgery. This science has progressed rapidly in the last 30 years with new polymer technology and computerized systems at the centre of modern developments. The possibility of incorporating cellular growth factors into a potential implant matrix is attractive, though this concept should be pursued with caution. When a chosen scaffold slowly disintegrates in a controlled fashion after implantation, while being replaced by totally sufficient, natural structural cells and fibres, the outcome will be a replaced tissue rather than a replacement graft.
ACKNOWLEDGEMENTS
We would like to thank the Engineering and Physical Science Research Council, the Phoenix Trust and Pearl Assurance for financial support for this work.
REFERENCES
- Alison MR, Poulsom R, Jeffery R, Dhillon AP, Quaglia A, Jacob J, Novelli M, Prentice G, Williamson J, Wright NA (2000) Hepatocytes from non‐hepatic adult stem cells. Nature 406, 257. [DOI] [PubMed] [Google Scholar]
- Barker JN, Wagner JE (2002) Umbilical cord blood transplantation: current state of the art. Curr. Opin. Oncol. 14, 160. [DOI] [PubMed] [Google Scholar]
- Becker NA, Kelm RJ, Vrana JA, Getz MJ, Maher LJ (2000) Altered sensitivity to single‐strand‐specific reagents associated with the genomic vascular smooth muscle alpha‐actin promoter during myofibroblast differentiation. J. Bio. Chem. 275, 15384. [DOI] [PubMed] [Google Scholar]
- Bots JGF, Van Der Does L, Bantjes A (1986) Small diameter blood vessel prostheses from blends of polyethylene oxide and polypropaline oxide. Biomaterials 7, 393. [DOI] [PubMed] [Google Scholar]
- Brittan M, Hunt T, Jeffery R, Poulsom R, Forbes SJ, Hodivala‐Dilke K, Goldman J, Alison MR, Wright NA (2002) Bone marrow derivation of pericryptal myofibroblasts in the mouse and human small intestine and colon. Gut 50, 752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown RA, Talas G, Porter RA, McGrouther DA, Eastwood M (1996) Balanced mechanical forces and microtubule contribution to fibroblast contraction. J. Cell. Physiol. 196, 439. [DOI] [PubMed] [Google Scholar]
- Brown RA, Prajapati R, McGrouther DA, Yannas IV, Eastwood M (1998) Tensional homeostasis in dermal fibroblasts: mechanical responses to mechanical in three‐dimensional substrates. J. Cell. Physiol. 175, 323. [DOI] [PubMed] [Google Scholar]
- Butterfield M, Wheatley DJ, Williams DF, Fisher J (2001) A new design for polyurethane heart valves. J. Heart Valve Dis. 10, 105. [PubMed] [Google Scholar]
- Cacou C, Eastwood M, McGrouther DA, Brown RA (1996) Culture force monitor for investigating the formation of adhesions between tissue interfaces in vitro . Cell. Eng. 1, 109. [Google Scholar]
- Chevallay B, Abdul‐Malak N, Herbage D (2000) Mouse fibroblasts in long‐term culture within collagen three‐dimensional scaffolds: influence of crosslinking with diphenylphosphorylazide on matrix reorganization, growth, and biosynthetic and proteolytic activities. J. Biomed. Mater. Res. 49, 448. [DOI] [PubMed] [Google Scholar]
- Eastwood M, McGrouther DA, Brown RA (1994) A culture force monitor for measurement of contraction forces generated in human dermal fibroblast cultures: evidence for cell‐matrix mechanical signalling. Biochimica Biophysica Acta 1201, 186. [DOI] [PubMed] [Google Scholar]
- Eastwood M, Porter R, Khan U, McGrouther G, Brown R (1996) Quantitative analysis of collagen gel contractile forces generated by dermal fibroblasts and the relationship to cell morphology. J. Cell Physiol. 166, 33–42. [DOI] [PubMed] [Google Scholar]
- Eastwood M, McGrouther DA, Brown RA (1998a) Fibroblast responses to mechanical forces. Proc. Inst. Mech. Eng. 212, 85. [DOI] [PubMed] [Google Scholar]
- Eastwood M, Mudera VC, McGrouther DA, Brown RA (1998b) Effect of precise mechanical loading on fibroblast populated collagen lattices: morphological changes. Cell Mot. Cyto. Skel. 40, 13. [DOI] [PubMed] [Google Scholar]
- Feindel CM, David TE (1993) Heart valve surgery. Curr. Opin. Cardiol. 82, 47–53. [DOI] [PubMed] [Google Scholar]
- Greisler HP, Ellinger J, Schwarcz TH, Golan J, Raymond RM, Kim DU (1987) Arterial regeneration over polydioxanone prostheses in the rabbit. Arch. Surg. 122, 715. [DOI] [PubMed] [Google Scholar]
- Grinnell F, Ho C‐H (2002) Transforming growth factor β stimulates fibroblast‐collagen matrix contraction by different mechanisms in mechanically loaded and unloaded matrices. Exp. Cell Res. 273, 248. [DOI] [PubMed] [Google Scholar]
- Gudi MA, Sheppard MN (2003) Pathology of surgically removed cardiac valves at a specialist center. Abstract of Pathological Society of Great Britain and Ireland, Bristol Meeting 34, 35. [Google Scholar]
- Guma FCR, Mello TG, Mermelstein CS, Fortuna VA, Wofchuk ST, Gottfried C, Guaragna RM, Costa ML, Borojevic R (2001) Intermediate filaments modulation in an in vitro model of the hepatic stellate cell activation or conversion into the lipocyte phenotype. Biochem. Cell Biol. 79, 409. [PubMed] [Google Scholar]
- Harding KG, Morris HL, Patel GK (2002) Healing chronic wounds. BMJ 324, 160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harris AB, Eastwood M (2003) Parameters for a 3‐D cardiovascular tissue construct. Cell Prolif. 36, 235. [Google Scholar]
- Hench LL, Polak JM (2002) Third generation biomedical materials. Science 295, 1015. [DOI] [PubMed] [Google Scholar]
- Herzog CA, Ma JZ, Collins AJ (2002) Long‐term survival of dialysis patients in the United States with prosthetic heart valves. Circulation 105, 1336. [DOI] [PubMed] [Google Scholar]
- Hoerstrup SP, Sodian R, Daebritz S, Wang J, Bacha EA, Martin DP, Moran AM, Guleserian KJ, Sperling JS, Kaushal S, Vacanti JP, Schoen FJ, Mayer JE (2000) Functional living trileaflet heart valves grown in vitro . Circulation 102, 44. [DOI] [PubMed] [Google Scholar]
- Huynh T, Abraham G, Murray J, Brockbank K, Hagen P‐O, Sullivan S (1999) Remodelling of an acellular collagen graft into a physiologically responsive neovessel. Nature Biotechnol. 17, 1083. [DOI] [PubMed] [Google Scholar]
- Kim WG, Yang JH, Sung SH (2002) Light and electron microscope analyses of autologous pericardial tissue used as a small‐diameter arterial graft in dogs. Artif. Organs 26, 58. [DOI] [PubMed] [Google Scholar]
- L’Heureux N, Paquet S, Labbe R, Germain L, Auger FA (1998) A completely biological tissue‐engineered human blood vessel. FASEB J. 12, 47. [DOI] [PubMed] [Google Scholar]
- Mahoney MJ, Saltzman WM (2001) Transplantation of brain cells assembled around a programmable synthetic microenvironment. Nature Biotechnol. 19, 934. [DOI] [PubMed] [Google Scholar]
- Mann BK, West JL (2001) Tissue engineering in the cardiovascular system: progress toward a tissue engineered heart. Anat. Rec. 263, 367. [DOI] [PubMed] [Google Scholar]
- Manothaya C, Vattanapat S, Somabutr C (1980) New technique for construction of tissue heart valves. Thorax 35, 611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mcculloch AD, Harris AB, Sarraf CE, Eastwood M (2003) A new multi‐cue bioreactor (M‐CB) for tissue engineering of tubular cardiovascular samples under physiological conditions. Tissue Eng. in press. [DOI] [PubMed]
- Morsi Y (2002) Tissue engineering of biological cardiovascular system surrogates: reviewer's commentary. Heart Lung Circ. 11, 151. [DOI] [PubMed] [Google Scholar]
- Mudera VC, Pleass R, Eastwood M, Tarnuzzer R, Schultz G, Khaw P, McGrouther DA, Brown RA (2000) Molecular responses of human dermal fibroblasts to dual cues: contact guidance and mechanical load. Cell Motil. Cytoskel. 45, 1. [DOI] [PubMed] [Google Scholar]
- Nkere UU, Whawell SA, Sarraf CE, Schofield JB, Thompson JN, Taylor K (1995) Pericardial trauma and adhesions in relation to reoperative cardiac surgery. Thorac. Cardiovasc. Surg. 43, 338. [DOI] [PubMed] [Google Scholar]
- Nkere UU, Whawell SA, Sarraf CE, Schofield JB, O'Keefe PA (1998) Pericardial substitution after cardiopulmonary bypass surgery: a trial of an absorbable patch. Thorac. Cardiovasc. Surg. 46, 77. [DOI] [PubMed] [Google Scholar]
- Porter R, Brown RA, Eastwood M, Occleston NL, Khaw PT (1998) Ultrastructural changes during contraction of collagen lattices by ocular fibroblasts. Wound Report Regen. 6, 157. [DOI] [PubMed] [Google Scholar]
- Prajapati RT, Chavally B, Herbage D, Eastwood M, Brown RA (2000a) Mechanical loading regulates protease production by fibroblasts in three dimensional collagen substrates. Wound Report Regen. 8, 227. [DOI] [PubMed] [Google Scholar]
- Prajapati R, Eastwood M, Brown R (2000b) Duration and orientation of mechanical loads determine fibroblast cyto‐mechanical activation: monitored by protease release. Wound Report Regen. 8, 239. [DOI] [PubMed] [Google Scholar]
- Rashid ST (2003) Cellular engineering of conduits for coronary and lower limb bypass surgery: role of cell attachment peptides and preconditioning in optimizing smooth muscle cells (SMC) adherence to polymer scaffolds. Cell Prolif. 36, 238. [DOI] [PubMed] [Google Scholar]
- Rayner M, Petersen S (2000) The European Heart Health Initiative Executive Summary. 2. The burden of cardiovascular disease. European cardiovascular disease statistics. Available from: www.ehnheart.org/ehhi_summary2asp.
- Roche S, Ronziere M‐C, Herbage D, Freyria A‐M (2001) Native and DPPA cross‐linked collagen sponges seeded with fetal bovine epiphyseal chondrocytes used for cartilage tissue engineering. Biomaterials 22, 9. [DOI] [PubMed] [Google Scholar]
- Sarraf CE (2000) Immunolabelling for electron microscopy In: George AJT, Urch CE, eds. Diagnostic and Therapeutic Antibodies. Methods in Molecular Medicine Series, p. 439 Vol. 40 Totowa NJ: Humana Press. [DOI] [PubMed] [Google Scholar]
- Sarraf CE, Mahmood MS, Aitken E (1998) Hepatic extramedullary granulopoiesis after treatment with liver‐stimulatory xenobiotics in lpr mice. J. Ultrastruct. Pathol. 22, 443. [DOI] [PubMed] [Google Scholar]
- Sarraf CE, Harris AB, Mcculloch AD, Eastwood M (2002a) Engineered tissue for cardiovascular implantation. Tissue Eng. 8, 1250. [Google Scholar]
- Sarraf CE, Harris AB, Mcculloch AD, Eastwood M (2002b) Tissue engineering of biological cardiovascular system surrogates. Heart, Lung Circ. 11, 142. [DOI] [PubMed] [Google Scholar]
- Shi G, Cai Q, Wang C, Lu N, Wang S, Bei J (2002) Fabrication and biocompatibility of cell scaffolds of poly (l‐lactic acid) and poly (l‐lactic‐co‐glycolic acid). Polm. Adv. Technol. 13, 227. [Google Scholar]
- Shinoka T, Ma PX, Shum‐Tim D, Breuer CK, Cusick RA, Zund G, Langer R, Vacanti JP, Mayer JE (1996) Tissue engineered heart valves: autologous valve leaflet replacement, study in a lamb model. Circulation 94, 164. [PubMed] [Google Scholar]
- Shinoka T, Shum‐Tim D, Ma PX, Tanel RE, Langer R, Vacanti JP, Mayer JE (1997) Tissue‐engineered heart valve leaflets: does cell origin affect outcome? Circulation 96, 102. [PubMed] [Google Scholar]
- Shum‐Tim D, Stock U, Hrkach J, Shinoka T, Lien J, Moses MA, Stamp A, Taylor G, Moran AM, Landis W, Langer R, Vacanti JP (1999) Tissue engineering of autologous aorta using a new biodegradable polymer. Ann. Thorac. Surg. 68, 2298. [DOI] [PubMed] [Google Scholar]
- Sodian R, Hoerstrup SP, Sperling JS, Daebritz SH, Martin DP, Schoen FJ, Vacanti JP, Mayer JE (2000) Tissue engineering of heart valves: in vitro experiences. Soc. Thorac. Surg. 70, 140. [DOI] [PubMed] [Google Scholar]
- Talas G, Adams TST, Eastwood M, Rubio G, Brown RA (1997) Phenytoin reduces the contraction of recessive dystrophic epidermolysis bullosa fibroblast populated collagen gels. Int. J. Biochem. Cell Biol. 29, 261. [DOI] [PubMed] [Google Scholar]
- Taylor PM, Allen SP, Yacoub MH (2000) Phenotypic and functional characterization of interstitial cells from human heart valves, pericardium and skin. J. Heart Valve Dis. 9, 150. [PubMed] [Google Scholar]
- Urbanski PP (2002) Complete aortic root replacement in patients with small aortic annulus. Ann. Thorac. Surg. 73, 725. [DOI] [PubMed] [Google Scholar]
- Weinberg CB, Bell E (1986) A blood vessel model constructed from collagen and cultured vascular cells. Science 231, 397. [DOI] [PubMed] [Google Scholar]
- Williams BT, Bellhouse BJ, Ashton F (1973) Autologous superior vena cava as a material for valve replacement. J. Thorac. Cardiovasc. Surg. 66, 952. [PubMed] [Google Scholar]
- Wilson AM, McGrouther DA, Eastwood M, Brown RA (1997) The effect of burn blister fluid on fibroblast contraction. Burns 23, 306. [DOI] [PubMed] [Google Scholar]
- Yaginuma G, Sakurai M, Ota K (2001) Thoracodorsal artery as a free arterial graft for myocardial revascularization. Ann. Thorac. Surg. 72, 915. [DOI] [PubMed] [Google Scholar]
- Zeltinger J, Landeen LK, Alexander HG, Sibanda B (2001) Development and characterization of tissue‐engineered aortic valves. Tissue Eng. 7, 9. [DOI] [PubMed] [Google Scholar]
- Zund G, Hoerstrup SP, Schoeberlein AS, Lachat M, Uhlschmid G, Vogt PR, Turina M (1998) Tissue engineering: a new approach in cardiovascular surgery; seeding of human fibroblasts followed by human endothelial cells on resorbable mesh. Cardio-Thoracic Surg. 13, 160. [DOI] [PubMed] [Google Scholar]


