Abstract
Oxidative stress (OS) influences vascular function and structure in spontaneously hypertensive rats (SHRs). It is also responsible for the decreased nitric oxide (NO) bioavailability that influences endothelial vasodilation. The effects of high-intensity exercise on endothelial function and ultrastructure in hypertension remain unknown. Thus, this study investigated the effects of moderate- and high-intensity exercise on hypertension-associated endothelial dysfunction and ultrastructural remodeling. Moderate-intensity (SHR-M) and high-intensity (SHR-H) aerobic exercise training groups were compared in age-matched sedentary SHRs (SHR-C) and normotensive Wistar–Kyoto rats (WKY-C). The results showed that the endothelial ultrastructure was impaired in the SHR-H and SHR-C groups. Glutathione peroxidase levels were significantly increased in the SHR-M group compared to the SHR-C group. MDA content was higher in the SHR-H group than in the SHR-C group, but the levels of antioxidant enzymes did not increase accordingly. Apocynin scavenging reactive oxygen species (ROS) ameliorated endothelium-dependent vasodilator function in the SHR-H group. However, the SHR-M and WKY-C groups abolished the increased vasodilation induced by apocynin. L-NAME, a NO synthase inhibitor, was applied to isolated mesenteric arteries (MAs) to evaluate NO contribution. Moderate-intensity exercise reversed the decreased NO contribution to MAs in hypertension, and high-intensity exercise aggravated this change. These data suggest that moderate-intensity exercise ameliorated adverse remodeling of the endothelial ultrastructure and function in hypertension by decreasing oxidative stress and increasing NO contribution. However, high-intensity exercise exacerbated all of these changes by increasing OS and ROS contribution, and decreasing NO contribution.
Keywords: exercise intensity, hypertension, endothelium, mesenteric arteries
INTRODUCTION
Hypertension, a prominent risk factor and leading cause of death from cardiovascular and cerebrovascular diseases, accounts for 9.4 million deaths annually.1 Considerable research has demonstrated that altered vascular structure and function, such as increased vascular stiffness, decreased endothelium-dependent vasodilation, and enhanced contractile response, are closely related to hypertension.2 Hypertension leads to inward eutrophic remodeling with corresponding greater media thickness, reduced lumen and external diameter, and increased media-to-lumen ratio.3 Endothelial cells modulate many physiological activities of the cardiovascular system and respond quickly to chronic pathophysiological stimuli, which can lead to injured intercellular junctions of endothelial cells, some protrusions, and bridging of the endothelium into lumen accompanied by luminal narrowing.4
Nitric oxide (NO) plays an important role in the regulation of vascular tone and inhibition of vascular smooth muscle cell migration, white blood cell adhesion, and platelet aggregation.5 Endothelial nitric oxide synthase (eNOS), the NO synthase (NOS) isoform expressed in endothelial cells, synthesizes NO from L-arginine in endothelial cells. As the production of NO and/or effects to the arteries decrease, this type of pathological process called NO bioavailability decreases. The decreased NO bioavailability leads to impairment of NO-dependent dilation in resistant vessels, and influences the endothelial vasodilation. Many studies have demonstrated that oxidative stress (OS), a critically pathophysiological process in hypertension, is responsible for the decreased NO bioavailability.6 Erbs et al.7 observed that as vitamin C was injected into spontaneously hypertensive rats (SHRs), the function of the coronary artery improved by increasing NO bioavailability and decreasing the levels of reactive oxygen species (ROS). ROS plays an important role in various cellular processes, such as cell growth and gene expression. Moderate levels of ROS stimulate activation of the Nrf2 transcription factor and trigger an increase in antioxidant enzymes such as catalase, glutathione peroxidase/glutathione disulfide-reductase (GSH-Px/GSR), and glutathione S-transferase.8 However, excess ROS activates the overexpression of pro-inflammatory and adhesion factors.9 Therefore, ROS are crucial factors in the modulation of the cardiovascular system.
Antioxidant therapy can control the development of hypertension in animal models, but it remains controversial in humans. Thus, selecting a non-pharmacological therapy to prevent the development of hypertension has become a hot topic in recent years. Exercise training is a well-recommended non-pharmacological measure that can effectively treat patients with hypertension.10 To date, most research has used low- and moderate-intensity aerobic exercise as exercise prescriptions. Moreover, most studies have used large arteries to investigate the effects of exercise on vascular function and structure in hypertensive model; however, minimal attention has been given to the vital role that resistance arteries play in regulating blood pressure during exercise.
METHODS
Animals and experimental protocols
Three-month-old male SHRs (n = 24) and the same-aged Wistar Kyoto Rats (WKYs, n = 8) were purchased from Vital River Laboratory Animal Technology Co. Ltd. (Beijing, China). The rats were randomly assigned to four groups: the sedentary WKY control group (WKY-C, n = 8), the sedentary SHR control group (SHR-C, n = 8), the moderate-intensity exercise group (SHR-M, n = 8), and the high-intensity exercise group (SHR-H, n = 8). All of the rats were housed in a temperature-controlled room with a 12:12 h light-dark cycle. All of the experimental protocols were approved by the Ethics Committee of Beijing Sport University and they were performed in accordance with the Chinese Animal Protection Laws and Institutional Guidelines.
All of the rats, including the sedentary animals, were habituated to treadmill exercise. Each rat walked on a motor-driven treadmill at 15 m/min (0° slope) for 5 min/day for 5 days. After one-week acclimation period, the animals in the SHR-M and SHR-H groups ran on a motor treadmill (0° slope) at 18–20 m/min (~55–65% of the maximal aerobic velocity) and 26–28 m/min (~75–85% of the maximal aerobic velocity), respectively. Each exercise-trained group ran 60 min per day for 5 days each week for 8 weeks. To determine the maximal exercise capacity, the rats underwent a progressive exercise test using an incremental speed protocol of 5 m/min every 3 min and no grade until exhaustion. The rats were considered to be exhausted when they could no longer run at the treadmill speed. The treadmill exercise test was repeated after 4 weeks to adjust the training intensity.
Blood pressure (BP) was measured using an indirect tail–cuff method (BP-2010A, Softron Biotechnology, Japan). Before the measurements, experimental rats were restrained for about 10 min to keep calm and feel comfortable at the room temperature. Temperature-controlled (37°C) warming holder was equipped with the rat’s tail. The sampling of data was usually repeated three times at intervals of about 1 min, and the average of three recordings was used as the data of individual rats.11
Assessment of vascular function
At the end of the 8-week exercise training, the rats were anesthetized with sodium pentobarbitone (60 mg/kg, intraperitoneal) and decapitated using a guillotine. The mesenteric arteries (MAs) were rapidly extracted and removed in cold Krebs’ solution composed of (mM): 131.5 NaCl, 5 KCl, 1.2 NaH2PO4, 1.2 MgCl2, 2.5 CaCl2, 11.2 glucose, 13.5 NaHCO3, and 0.025 EDTA, and they were continuously gassed with 95% O2 and 5% CO2 (pH 7.4). Short segments of third-order branches (A3) were used for contractile studies with a multi-myograph system (620 M, DMT, Denmark), and pEC50 (−logEC50), the negative logarithm of the half-maximal inhibitory concentration, a measure of the effectiveness of a substance in inhibiting a specific biological or biochemical function, was calculated.
To determine the ROS and NO contributions of vasodilation function, the following experiments were conducted. (1) KCl (60 mM) was first applied to induce the maximal contraction. Krebs’ solution was washed three times after the tension reached the platform. (2) Endothelium-dependent relaxation response to acetylcholine (ACh) was measured as MAs pre-constricted with norepinephrine (NE). (3) As pre-incubated with apocynin (15 min), an important antioxidant to scavenge ROS, we observed the changes of relaxant responses to ACh. (4) With L-NAME incubation (20 min), we measured the NO-dependent vasodilation to evaluate the NO bioavailability. (5) Endothelium-independent vasodilatation response to sodium nitroprusside (SNP) was measured.
Oxidative stress biomarkers in serum
Blood samples were immediately collected from the postcava and centrifuged at 3,500 rpm for 15 min. Then, the serum was frozen and stored in −80°C until OS markers’ determination next time. The concentrations of malondialdehyde (MDA) was measured using thiobarbituric-acid-reacting substance (TBARS) production at 532 nm.12 The activity of superoxide dismutase (SOD) was measured using the method of Sun et al at 560 nm by detecting the exhibition degree of this reaction.13 The activity of glutathione peroxidase (GSH-Px) was measured using the method of Paglia and Valentine.14 All reagents and samples were equilibrated to room temperature before the assay was performed.
Endothelial ultrastructure
The MAs were rapidly dissected from unnecessary tissues to prevent autolysis and they were quickly removed in cold (4°C) fresh 2.5% glutaraldehyde. The PBS was washed three times (every washing lasting for a minimum of 15 min) in the next day. Tissues were dehydrated in acetone (70%, 80%, 90%, and 100%). The vessels were embedded vertically in paraffin blocks using standard histological steps. Sections were cut and stained to detect the endothelial ultrastructure by transmission electron microscope (JEOL JEM-1230, Japan).
Arterial morphology
Vascular structure was measured using histological method. The segments of MAs were fixed in 4% paraformaldehyde. Haematoxylin and eosin (HE) staining was applied in sections of MA with paraffin. Sections cut into 5-μm and underwent series of steps for histological analysis. These steps were consistent with our former studies.15
Western blotting
Total proteins were isolated and pooled for use in Western blots as previously described.16 Briefly, fresh segments of MAs were frozen in liquid nitrogen and stored at −80°C until assay. After pulverization, frozen MAs were homogenized in ice cold Tris-EDTA buffer. Protein concentration was determined using a protein assay (Bio-Rad Laboratories; Hercules, USA). Equivalent amounts (60 μg) of total protein from the MAs were added to the adjacent lanes. The related primary antibody was mouse monoclonal antibodies for eNOS (1:1000, Transduction Laboratories, Lexington, UK). The secondary antibody was anti-mouse IgG-HRP (1:8000, Proteintech Group Inc., Chicago, Illinois, USA). The immunoreactive bands were visualized with enhanced chemiluminescence, and the signals were recorded with Bio-Rad ChemiDOC XRS+ (Bio-Rad Laboratories, Hercules, CA, USA). GAPDH was detected and used to correct for the equal loading of all of the samples. The eNOS protein content was expressed relative to the GAPDH content.
Data analysis
Data are expressed as the mean ± SEM. pEC50 (−logEC50), the negative logarithm of the half-maximal inhibitory concentration, a measure of the effectiveness of a substance in inhibiting a specific biological or biochemical function, was calculated from the concentration–response curve by nonlinear regression curve calculation using Graph Pad Prism 5.0. The data were analyzed statistically with Student’s t-test or one-way ANOVA followed by Bonferroni adjusted t-test. The Student’s t-test was used for comparisons between two groups. A value of P < 0.05 was considered to be statistically significant.
RESULTS
Blood pressure
As shown in Table 1, before 8-week exercise training, systolic blood pressure (SBP) was significantly higher in the SHRs than in the WKYs. After long-term exercise training, the SBP was significantly lower in the moderate-intensity SHR group (SHR-M group) than in the age-matched sedentary SHR group (SHR-C group) (P < 0.05), whereas it was significantly higher in the high-intensity SHR group (SHR-H group) than in the SHR-M and SHR-C groups (P < 0.05). Similar observations were made with regard to diastolic blood pressure (DBP). The DBP was significantly lower in the SHR-M group than in the SHR-C and SHR-H groups (P < 0.05)
Table 1.
Effects of different-intensity exercise on blood pressure in rats
| WKY-C (n=8) | SHR-C (n=8) | SHR-M (n=8) | SHR-H (n=8) | ||
|---|---|---|---|---|---|
| SBP (mmHg) | Initial | 134.9±1.6 | 191.6±2.3* | 192.1±1.9 | 189.7±3.4 |
| Final | 137.6±1.8 | 197.8±2.0*+ | 182.9±1.6#+ | 212.1±5.6#$+ | |
| DBP (mmHg) | Initial | 93.7±4.6 | 145.6±4.2* | 145.4±2.4 | 147.7±4.7 |
| Final | 98.2±2.8 | 148.4±2.7* | 139.6±2.7#+ | 159.8±5.2#$+ |
SBP, systolic blood pressure; DBP, diastolic blood pressure. Initial indicated before 8 weeks of exercise training; final indicated after 8 weeks of exercise training.
P < 0.05, versus the WKY-C group
P < 0.05, versus the SHR-C group
P < 0.05, versus the SHR-M group; and
P < 0.05, versus Initial.
Vascular ultrastructure
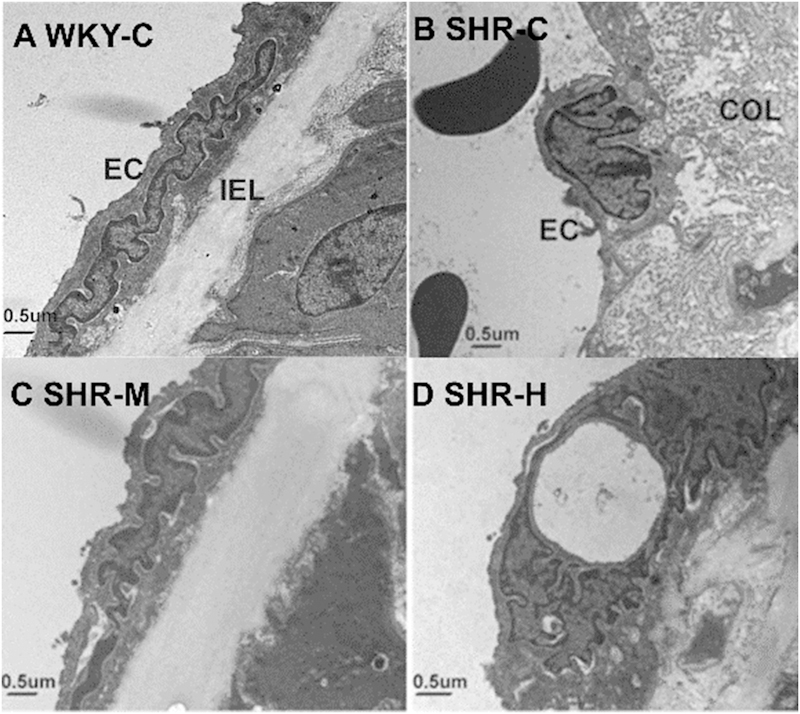
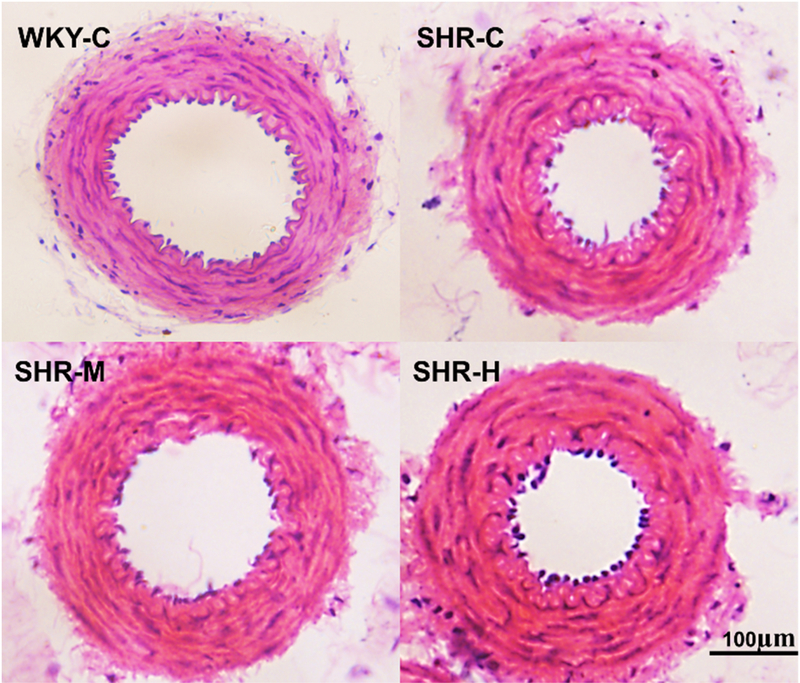
The endothelial ultrastructure of the WKY-C group showed flat shapes and microvilli on the cell surface (Fig. 1). In addition, there was good inner membrane continuity, a flat inner elastic membrane, and smooth muscle cells that were neatly arranged. Images of the SHR-C group showed that the endothelium in the mesenteric arteries (MAs) lacked a normal ultrastructure. Endothelial cells with swelling of the nucleus and disappearance of the microvilli appeared as finger-like projections oriented to the inner cavity. There was an irregular internal elastic membrane, and a large number of collagen fibers proliferated in the endothelium. However, moderate-intensity exercise inhibited endothelial cell changes in SHR rats, maintaining normal ultrastructural integrity. High-intensity exercise in SHRs demonstrated severe damage to the endothelium with swelling of the nucleus, and collagen fibers had excessive proliferative activity. As shown in Figure 2, the SHR-C group had greater media thickness and narrowed lumen compared with the WKY-C group. Moderate-intensity exercise improved hypertension-associated adverse structural remodeling. However, high-intensity exercise exacerbated these changes and increased media thickness compared with the SHR-C group.
Fig. 1.
Exercise training of different-intensity in SHRs affect the endothelial ultrastructure in mesenteric arteries. EC, endothelial cell; IEL, internal elastic lamina; and COL, collagen.
Fig. 2.
Effect of different-intensity exercise on arterial morphology in rat mesenteric arteries.
Biomarkers of oxidative stress
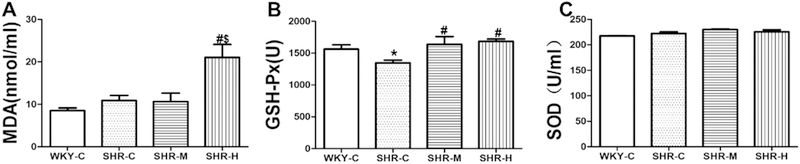
As shown in Figure 3A, MDA concentrations did not significantly differ between the WKY-C group (8.5 ± 0.6 μM) and SHR-C group (10.9 ± 1.2 μM, P ˃ 0.05). MDA serum levels were significantly increased in the SHR-H group (21.0 ± 3.1 μM) compared with the SHR-C and SHR-M groups (10.7 ± 2.0 μM; P < 0.05). As shown in Figure 3B, GSH-Px activity was much lower in the SHR-C group than in the WKY-C group (1348.1 ± 42.5 U vs. 1562.6 ± 68.9 U, respectively; P < 0.05). Compared with the SHR-C group, GSH-Px activity was significantly increased in the SHR-M (1639.2 ± 119.5 U; P < 0.05) and SHR-H groups (1685.1 ± 35.00 U, P < 0.05). However, there was no apparent difference in GSH-Px activity between the SHR-M and SHR-H groups. As shown in Figure 3C, there was no marked difference in superoxide dismutase activity among the WKY-C (217.68 ± 0.58), SHR-C (222.32 ± 3.14), SHR-M (230.07 ± 1.18), and SHR-H groups (225.70 ± 3.81).
Fig. 3.
Effects of different-intensity exercise on biomarkers of oxide stress. (A) Serum MDA content; (B) serum GSH-Px activity; and (C) serum SOD activity. *P < 0.05 versus WKY-C; #P < 0.05 versus the SHR-C group; and +P < 0.05 versus the SHR-M group. n=8 in each group.
Vascular function
Contractile response
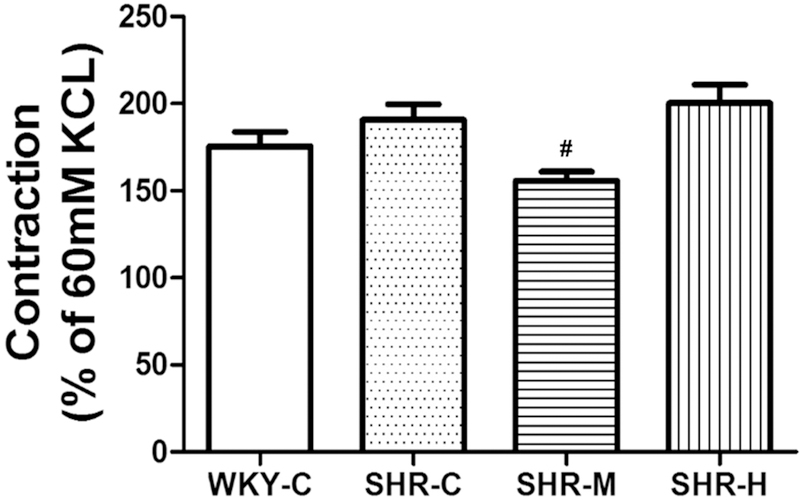
The contractile response was evaluated with potassium chloride (KCl) followed by norepinephrine (NE) (10−5 M). In each MA ring, KCl (60 mM) was first applied to induce maximal contractions (Kmax). The response induced by KCl (60 mM) was not significantly different between WKYs and SHRs. Exercise training with different-intensity levels did not modify this response. Then NE was added to the bath to induce vessel constriction, and the different groups had distinct reactions to NE (10−5 M). The contractile response was defined as the maximal response induced by the NE to Kmax ratio (Fig. 4). The NE-induced contractile response in the SHR-C group (190.6 ± 9.1% Kmax) was not markedly different than that in the WKY-C group (175.5 ± 8.3% Kmax; P > 0.05), but it tended to worsen the maximal response to NE. However, the force was markedly decreased in the SHR-M group (155.8 ± 5.1% Kmax), although there were no apparent differences between the SHR-H group (200.2 ± 10.8% Kmax) and SHR-C group.
Fig. 4.
NE-induced tension as %KCl (60 mM) in the small MAs. #P < 0.05 versus the SHR-C group, n=8 in each group.
Endothelium-dependent vasodilation response
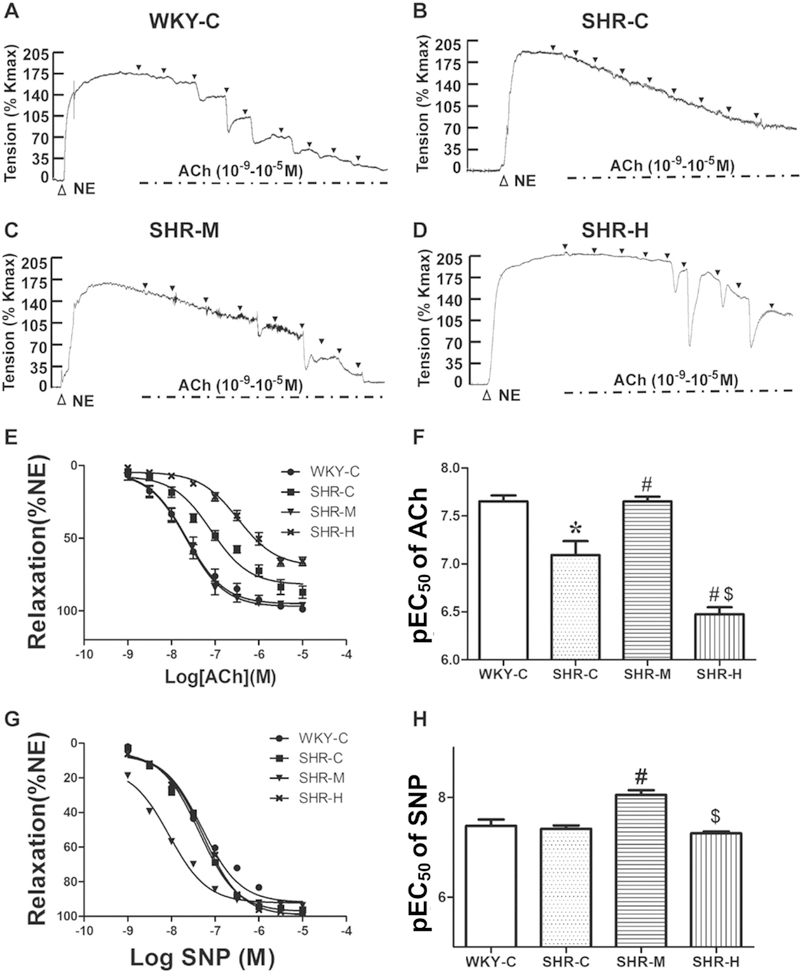
To determine the effects of exercise training on endothelium-dependent vasodilation in MAs, we investigated the concentration-dependent response in MAs induced by acetylcholine (ACh). As shown in Figure 5A–F, after the tension equilibrium of the NE-mediated vasoconstriction, ACh (10−9–10−5 M) was added in half-log increments. As expected, ACh induced concentration-dependent vasorelaxation, with a pEC50 of 7.10 ± 0.15 in the SHR-C group, which was significantly lower than that in the WKY-C group (7.65 ± 0.06; P < 0.05). However, the pEC50 was higher in the SHR-M group (7.65 ± 0.05; P < 0.05) than in the SHR-C group. The pEC50 in the SHR-H group (6.47 ± 0.07) was significantly than that in the SHR-C group, and even lower than that in the SHR-M group (both P < 0.05; Fig. 5F). Of note, moderate-intensity exercise promoted hypertension-associated lower vessel sensitivity to ACh, while high-intensity exercise worsened this sensitivity. The relaxant responses to ACh were calculated as the percentage reduction of NE-induced maximum contraction, which was much lower in the SHR-C group (87.2 ± 4.8% NE) than in the WKY-C group (97.9 ± 1.2% NE; P < 0.05). There was a significant difference in maximum relaxation induced by ACh between the SHR-C and different intensity exercise SHR groups. The SHR-M group (95.7 ± 1.2% NE) improved hypertension-associated with the decline of endothelium-dependent relaxation (Fig. 5E). However, endothelium-dependent relaxation was significantly decreased after high-intensity exercise training. These results suggest that compared with the WKY group, there was a profound decrease in ACh-mediated endothelium-dependent vasodilation in the SHR groups, which was reversed by moderate-intensity exercise training but exacerbated with high-intensity exercise training.
Fig. 5.
Concentration-response curve to ACh and SNP in small MAs. (A–D) Example of real-time recording of ACh-induced vasorelaxation in MA rings. Black triangles mark the time of addition of ACh (10−9, 3 × 10−9, 10−8, 3 × 10−8, 10−7, 3 × 10−7, 10−6, 3 × 10−6, and 10−5 M). (E) Concentration-response curves to ACh in MAs. (F) Sensitivity (pEC50) to Ach. (G) Concentration-response curves to SNP in MAs. (H) Sensitivity (pEC50) to SNP. *P < 0.05 versus the WKY-C group; #P < 0.05 versus the SHR-C group; and $P < 0.05 versus the SHR-M group; n = 8 in each group.
Endothelium-independent vasodilation response
Sodium nitroprusside (SNP) is an exogenous NO donor. When SNP (10−9–10−5 M) contributes to vascular smooth cell, it results in vasodilation. As shown in Figure 5G, the maximum relaxation induced by SNP was not different across all of the groups, which indicated that relaxation could be achieved in each group. Thus, exercise training had no effect on endothelium-independent vasodilation in SHRs. As shown in Figure 5H, the pEC50 values did not differ in SNP-induced vasodilatation between the WKY-C (7.43 ± 0.13) and SHR-C groups (7.37 ± 0.07). However, the pEC50 values were higher in the SHR-M group (8.05 ± 0.09, P < 0.05) than in the SHR-C group.
Effects of ROS scavenging on endothelium-dependent relaxation
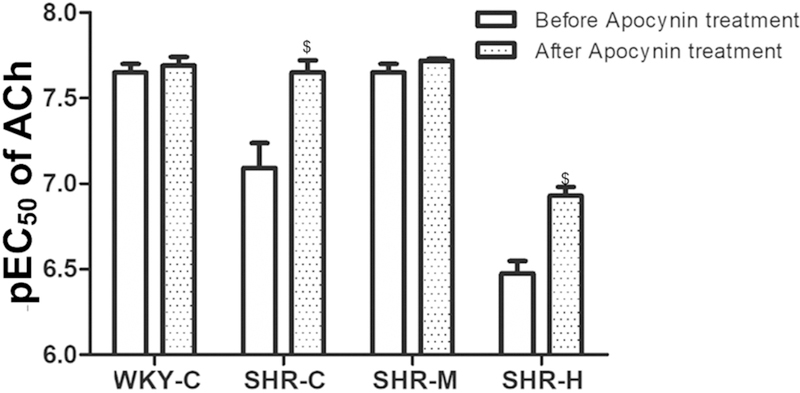
To assess the contribution of ROS to the endothelial function of MAs, after incubation for 15 min, apocynin improved the relaxation induced by ACh in each group. The results showed that apocynin failed to modify maximum relaxation in response to ACh in the WKY-C, SHR-C, and SHR-M groups. However, apocynin pre-incubation improved the impaired maximal relaxation induced by ACh in MAs in the SHR-H group (P < 0.05, Table 2). Apocynin also produced significant potentiation of MA sensitivity to ACh on NE pre-constricted rings in the SHR-C and SHR-H groups (P < 0.05; Fig. 6).
Table 2.
Impact of apocynin on the maximumendothelial-dependent vasodilation
| Tension (% NE) | WKY-C (n = 8) | SHR-C (n = 8) | SHR-M (n = 8) | SHR-H (n = 8) |
|---|---|---|---|---|
| Before treatment | 98.9 ± 1.2 | 87.2 ± 4.8 | 95.7 ± 1.2 | 66.0 ± 3.0 |
| After treatment | 102.0 ± 1.1 | 95.8 ± 3.2 | 98.9 ± 0.9 | 97.9 ± 0.9* |
Apocynin (300 μM) contribution to the relaxation in response to Acetylcholine (ACh) in small MAs.
P < 0.05 versus before treatment.
Fig. 6.
Comparison of sensitivity (pEC50) to Ach between before and after apocynin treatment. $P < 0.05 versus before apocynin treatment in each group.
Effects of NOS antagonism on endothelium-dependent relaxation
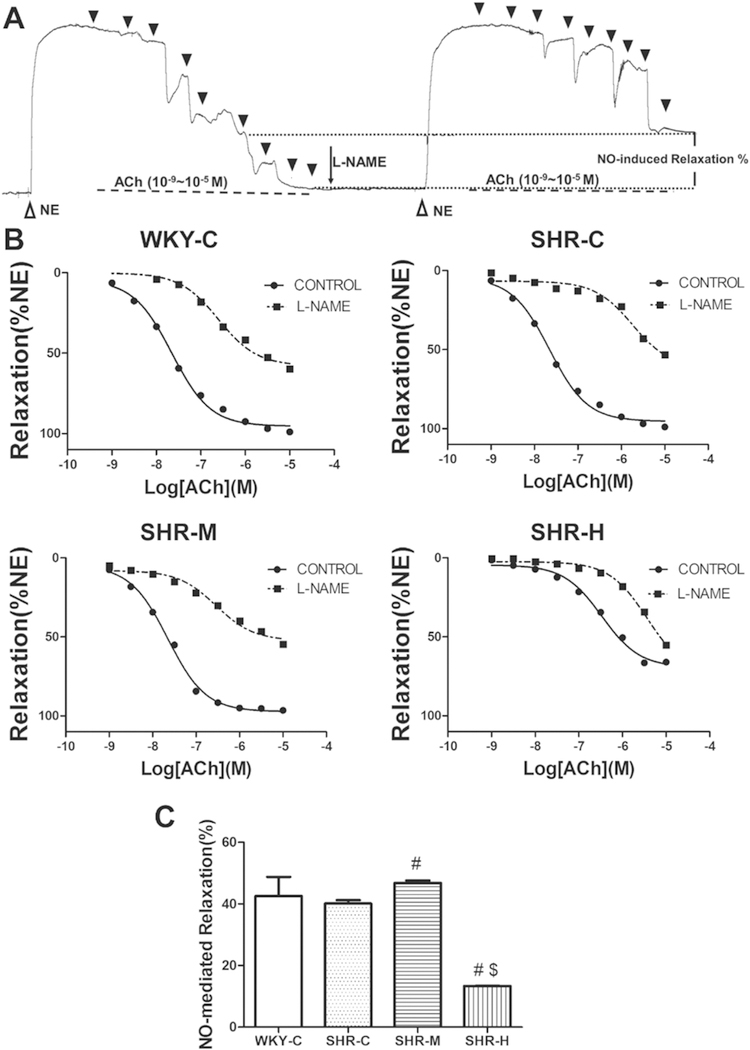
L-NAME incubation (20 min) inhibited ACh-mediated relaxation in MAs from all of the experimental groups. As shown in Figure 7A and 7B, the relaxation response to ACh in each group showed different degrees of attenuation with L-NAME application.NO-dependent dilation was determined from the maximal endothelium-dependent dilation in the absence or presence of L-NAME according to the following formula:
Fig. 7.
NO-dependent relaxation in small MAs. (A) Example of real-time recording of ACh-induced vasorelaxation in the absence and presence of L-NAME (0.1 mM). Black triangles mark the time of addition of ACh (10−9, 3×10−9, 10−8, 3×10−8, 10−7, 3×10−7, 10−6, 3×10−6, and 10−5 M). (B) Concentration-response curves of ACh in small MAs from the WKY-C, SHR-C, SHR-M, and SHR-H groups. (C) Summary of NO-dependent relaxation in the four groups. #P < 0.05 versus the SHR-C group; and $P < 0.05 versus the SHR-M group; n = 8 in each group.
NO-dependent relaxation (% NE) = maximum relaxation (relaxation induced by ACh) (% NE) - maximum relaxation (L-NAME incubation and relaxation induced by ACh) (% NE).
As shown in 7C, there was a significant difference in NO-dependent relaxation (% NE) between the SHR-C and SHR-M groups. It was significantly higher in the SHR-M group (46.8 ± 0.8% NE) than in the SHR-C group (40.1 ± 1.1% NE; P < 0.05). However, it was much lower in the SHR-H group (13.4 ± 0.1% NE) than in the SHR-C and SHR-M groups (P < 0.05).
eNOS protein expression
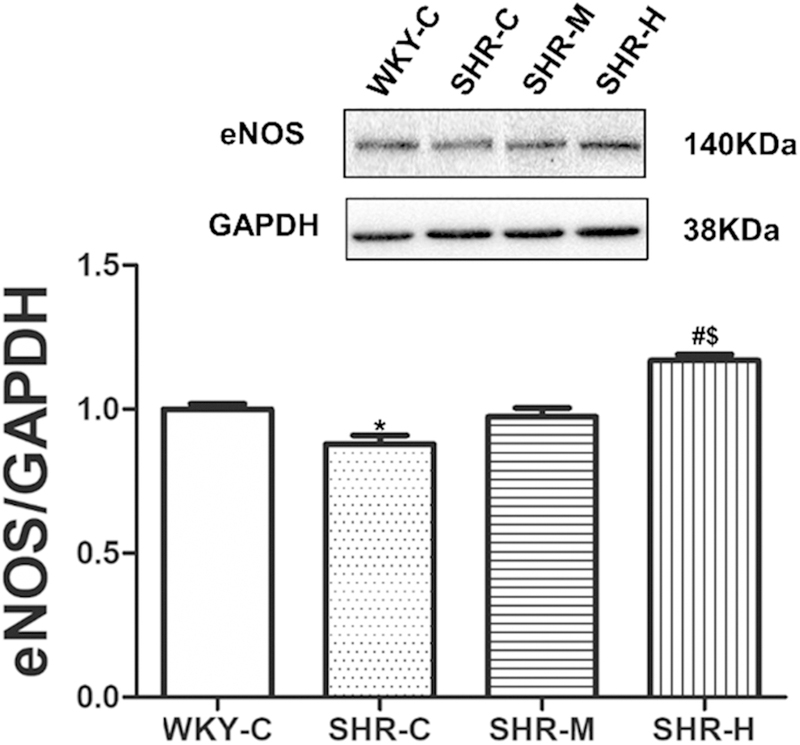
Next, eNOS protein expression in the MAs was determined. As shown in Figure 8, there was no apparent difference in eNOS protein expression between the SHR-C and SHR-M groups. However, eNOS protein expression was significantly higher in the SHR-H group than in the SHR-C group (P < 0.05), but it was significantly lower in the SHR-C group than in the WKY-C group (P < 0.05).
Fig. 8.
Densitometric analysis and eNOS protein expression in homogenates of small MAs. GAPDH expression is shown as the loading control. * P < 0.05 versus WKY-C; #P < 0.05 versus the SHR-C group, $P < 0.05 versus the SHRM group.
DISCUSSION
The novel finding of this study was that high-intensity exercise exacerbated hypertension-associated endothelial dysfunction and ultrastructural remodeling in MAs by increasing oxidative stress and ROS contribution to vasodilation, and decreasing NO contribution. These results suggested that different levels of exercise intensity play distinct roles in the endothelial function and ultrastructural remodeling in hypertension. This evidence suggests that exercises of reasonable intensity should be chosen for hypertensive patients.
Previous studies have suggested that low-intensity swimming training in SHRs of different ages decreased the arterial blood pressure.17 Long-term moderate-intensity exercise could influence hypertension patients’ blood pressure, as the SBP decreased by about 6–10 mmHg, while the DBP decreased by about 4–8 mmHg.18 However, little information has been obtained on the effects of SHRs with high-intensity exercise on blood pressure. This experiment demonstrated the dose-effect relationship between exercise intensity and blood pressure. Our results demonstrated that the SBP and DBP were significantly lower in the SHR-M group than in the SHR-C group, but were significantly higher in the SHR-H group than in the SHR-C group. Thus, hypertension with different intensities exercise could exert the opposite influence on modulating blood pressure.
In hypertension, a series of unfavorable responses are triggered, such as adverse remodeling of endothelial ultrastructure and function.19 Previous studies have shown that in hypertension, endothelial cells with swelling of the nucleus and a lack of microvilli have loss of the normal ultrastructure, which was consistent with our results.20 This study also found that moderate-intensity exercise had a positive influence on the integrity of the endothelial cells in SHRs, while high-intensity exercise aggravated the deformity and apoptosis of endothelial cells, and caused swelling of the nucleus, disappearance of the microvilli, and increased collagen fibers in endothelial cells. A previous study21 demonstrated that inward eutrophic remodeling in SHRs is characterized by greater media thickness, and a reduced lumen and external diameter with increased media-to-lumen ratio, which is consistent with the results of our study. Moderate-intensity exercise reversed this inward eutrophic remodeling in SHRs, whereas high-intensity exercise increased media thickness.
A growing number of research studies have shown that hypertension is accompanied by decreased endothelium-dependent vasodilation, which is consistent with our results.22 The structural changes of small arteries in response to various agents is associated with functional changes, such as different vasoconstriction and vasodilation. These results reflect that moderate-intensity exercise decreased the maximal response to NE and ameliorated the hypertension-associated endothelium-dependent response to ACh. These results are in agreement with a study by Roque,23 which showed that aerobic exercise (55–65% of maximal running speed) normalizes altered vascular function by reducing contractile responses and increasing endothelium-dependent relaxation.23 However, high-intensity exercise enhanced the contractile response to NE and decreased endothelium-dependent relaxation, which exacerbated endothelial dysfunction in hypertension. NE constriction is mediated by both α1- and α2-adrenergic receptors (ARs).24 Consistent with the findings in this study, it was previously reported that chronic low- or moderate-intensity exercise could reduce vascular contraction to α-adrenergic stimuli in hypertensive rats, and α2-AR responsiveness was attenuated by mild-intensity exercise.25,26 During moderate-exercise intervention, the most likely mechanisms underlying the decreased contraction elicited by NE in hypertension with moderate-intensity exercise are changes in the sympathetic nerve system,27 calcium sensitivity, calcium influx,28,29 and α-AR expression.30 Of note, that differences in NE-induced contraction might influence the vasorelaxation results which is a limitation of this study. To determine whether changes in smooth muscle dilation were due to different exercise intensities in hypertension, vasodilation induced by the endothelium-independent agonist SNP was measured. The results showed that maximum response was not altered by exercise.
OS is the common pathophysiological process of hypertension and other cardiovascular diseases, and improved OS in the vasculature is proposed to be a vital mechanism by supplementing antioxidant products to improve cardiovascular function in rats.31 Considerable evidence has suggested the excess production of ROS in SHRs,32 which plays a detrimental role in vascular endothelial function in SHRs,33 while an appropriate amount of ROS triggers some adaptive changes such as improvements in antioxidant capacity. Antioxidant therapy can control the development of hypertension in animal models, but remains controversial in humans because of possible confounding factors in patients, such as age, sex, diet, smoking, and degree of OS.34 Thus, selecting a non-pharmacological therapy to prevent hypertension is imperative. There is an evidence that exercise increases the amount of ROS produced by the mitochondria, and increases ROS by 10–20 fold than normal respiration.35 In exercise training, the production of ROS acts as a signal for the production of more related antioxidant enzymes to maintain homeostasis.36 We hypothesized that the change of OS in exercises of different intensities may induce changes in endothelial function and ultrastructure. The most common OS indicators are malondialdehyde (MDA, a biomarker of oxidative products), GSH-Px, and SOD (biomarkers of antioxidant enzymes).37 A previous study showed that the MDA content in serum did not change because exercise led to adaptive changes in the rats, which inhibited the temporary OS state resulting from a single movement cycle, which was consistent with our results.6 This study showed that the MDA content in the serum of the SHR-C group had a tendency to increase compared with that in the WKY-C group, but the difference was not significant. Serum levels of MDA and GSH-Px were increased in the SHR-H group by 1-fold and 0.3-fold, respectively. These data showed that the oxidant capacity exceeded the antioxidant capacity in the SHR-H group. Compared with the SHR-C group, the MDA content of the SHR-M group was unchanged. However, GSH-Px levels were significantly increased, indicating that OS decreased in the SHR-M group upon an increase in antioxidant enzymes. In our experiment, SHRs with different intensity exercise showed a significant increase in GSH-Px content, which may be due to adaptive changes in the production of ROS during exercise. However, previous reports about changes in GSH-Px following exercise have been inconsistent. Although some studies have reported that GSH-Px activity increases following exercise, consistent with our results,38 a reduction of GSH-Px activity or no change in activity in hypertension after exercise have also been shown.39,40 A study by Azizbeigi41 showed that different antioxidant enzymes change in accordance with the mode of exercise. Thus, changes in antioxidant enzymes appear to correlate with the mode and intensity of exercise.
Antioxidant enzymes could not scavenge the serum MDA content, which triggered serious OS. To further assess the influence of ROS on endothelial function, maximum endothelium-dependent response to ACh was compared in the absence and presence of apocynin treatment. Apocynin, which scavenges ROS, is an effective way to evaluate the effects of OS scavenging on endothelial-dependent vasodilation and ROS contribution. Our research provided evidence that apocynin improved the maximal response and sensitivity to ACh in the SHR-H group but not in the WKY-C and SHR-M groups. In addition, ROS contribution was greater in the SHR-H group than in the SHR-C group. The increased ROS contribution induced by high-intensity exercise promoted the impairment of endothelial dysfunction; and the reduction of ROS contribution induced by moderate-intensity exercise led to improvements in endothelium-dependent vasodilation in hypertension.
eNOS coupling can produce NO, while eNOS uncoupling is responsible for the switch from NO to O2− production. eNOS function plays an important role in NO bioavailability and ROS production. It is also well known that OS is responsible for the decreased NO bioavailability.42 Thus, our research evaluated the contribution of NO to endothelial-dependent vasodilation, and we found that NO-mediated vasodilation and eNOS protein expression were lower in the SHR-C group than in the WKY-C group. In addition, NO contribution was greater in the SHR-M group than in the SHR-C group, and the the SHR-H group had increased eNOS expression but decreased NO contribution. We focused on the correlation between the contribution of ROS and NO to explain the probable effects on endothelial function in SHRs with different-intensity exercise. This research showed that moderate-intensity exercise ameliorated endothelial dysfunction in SHRs, which could be attributed to the improvement in NO contribution. The reduction of NO contribution and increased ROS contribution in SHRs with high-intensity exercise was responsible for exacerbated endothelial dysfunction. eNOS expression enhanced with declined NO bioavailability in the SHR-H group does not mean “eNOS uncoupling,” but rather, might be a compensatory mechanism for decreased NO bioavailability and increased OS. However, our hypothesis was that “eNOS uncoupling” is a probable mechanism by which high-intensity exercise worsens hypertension-associated endothelial dysfunction and adverse structural remodeling in MAs. Additional studies are needed to confirm the “eNOS uncoupling” mechanism.
Considering that eNOS inhibition with L-NAME failed to completely abolish ACh-mediated endothelial-dependent vasodilation, which is consistent with a previous study,43 We analyzed the reason that with the exception of NO as a dominant endothelium-derived relaxing factor, other endothelium-derived relaxing factors and endothelium-derived hyperpolarizing factors may play crucial roles in regulating vascular response.44 Previous research has shown that there is mutual compensation function between NO and hydrogen peroxide; as NO is inhibited, hydrogen peroxide acts as the dominant endothelium-derived relaxing factor; and as hydrogen peroxide is inhibited, NO acts as the dominant endothelium-derived relaxing factor.45 Hydrogen peroxide can activate the calcium-sensitive potassium channels of smooth muscle cells, or depend upon the cyclophosphate mechanism, which leads to hyperpolarization of vascular smooth muscle cells.46
CONCLUSION
The results of this study may contribute to optimizing exercise prescriptions in patients with hypertension who perform aerobic exercise. The data indicated that moderate-intensity exercise prevented hypertension-associated endothelial ultrastructural remodeling and endothelial dysfunction by decreasing OS and elevating NO-mediated relaxation in MAs. Moreover, high-intensity exercise exacerbated these changes in hypertension by augmenting OS and ROS contribution, and decreasing NO contribution. Thus, exercises need to be carefully chosen by intensity for hypertensive patients.
PERSPECTIVES.
Further studies on the endothelium-derived hyperpolarizing factor mediating the cardiovascular system will provide more insights into the molecular mechanisms underlying the amelioration endothelial dysfunction in hypertension with moderate-intensity exercise. In addition, our hypothesis that “eNOS uncoupling” is likely responsible for the effects of high intensity exercise on exacerbating hypertension-associated adverse remodeling of endothelial ultrastructure and function in MAs needs further investigation.
Acknowledgment of grant support
This work was supported by the National Natural Science Foundation of China [31771312], the Beijing Natural Science Foundation [5172023], the Chinese Universities Scientific Fund [2018GJ010], the National Institutes of Health Grants R01HL135623 (D.X.), R01HD088039 (D.X.), and R03DA041492 (D.X.).
Footnotes
CONFLICT OF INTEREST
The authors declare no conflict of interest.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
REFERENCES
- 1.Asayama K, Thijs L, Brguljan-Hitij J, Niiranen TJ, Hozawa A, Boggia J, Aparicio LS, Hara A, Johansson JK, Ohkubo T, Tzourio C, Stergiou GS, Sandoya E, Tsuji I, Jula AM, Imai Y, Staessen JA; International Database of Home Blood Pressure in Relation to Cardiovascular Outcome (IDHOCO) investigators. Risk stratification by self-measured home blood pressure across categories of conventional blood pressure: a participant-level meta-analysis. PLoS Med 2014; 11: e1001591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Montezano AC, Dulak-Lis M, Tsiropoulou S, Harvey A, Briones AM, Touyz RM. Oxidative stress and human hypertension: vascular mechanisms, biomarkers, and novel therapies. Can J Cardiol 2015; 31: 631–641. [DOI] [PubMed] [Google Scholar]
- 3.Laurent S, Boutouyrie P. The structural factor of hypertension: large and small artery alterations. Circ Res. 2015;116:1007–1021. [DOI] [PubMed] [Google Scholar]
- 4.Okruhlicova L, Tribulova N, Weismann P, Sotnikova R. Ultrastructure and histochemistry of rat myocardial capillary endothelial cells in response to diabetes and hypertension. Cell Res. 2005;15(7):532–538. [DOI] [PubMed] [Google Scholar]
- 5.Stefek M, Sotnikova R, Okruhlicova L, Volkovova K, Kucharska J, Gajdosik A, Gajdosikova A, Mihalova D, Hozova R, Tribulova N, Gvozdjakova A. Effect of dietary supplementation with the pyridoindole antioxidant stobadine on antioxidant state and ultrastructure of diabetic rat myocardium. Acta Diabetol 2000; 37:1171–1177. [DOI] [PubMed] [Google Scholar]
- 6.Yu TT, Guo K, Chen HC, Lan CZ, Wang J, Huang LL, Wang XH, Zhang Z, Gao S. Effects of traditional Chinese medicine Xin-Ji-Er-Kang formula on 2k1c hypertensive rats: role of oxidative stress and endothelial dysfunction. BMC Complement Altern Med 2013; 13: 173–180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Erbs S, Gielen S, Linke A, Möbius-Winkler S, Adams V, Baither Y, Schuler G, Hambrecht R. Improvement of peripheral endothelial dysfunction by acute Vitamin C application: different effects in patients with coronary artery disease, ischemic, and dilated cardiomyopathy. Am Heart J 2003; 146: 280–285. [DOI] [PubMed] [Google Scholar]
- 8.Sasaki S, Tozawa T, Suqamoto K, et al. A novel di terpene para-hydroquinone compound derived from cryptoquinone protects neuronal cells against oxidative stress and activates the Nrf2/ARE pathway. Neurosci Lett 2013; 548: 132–136. [DOI] [PubMed] [Google Scholar]
- 9.Sirotkin AV, Alexa R, Kišová G, Matsushita Y, Satoh T. MicroRNAs control transcription factor NF-kB (p65) expression in human ovarian cells. Funct Inteqr Genomics 2015; 15: 271–275. [DOI] [PubMed] [Google Scholar]
- 10.Sousa LE, Maqalhaes WG, Bezerra FS, Santos RA, Campagnole-Santos MJ, Isoldi MC, Alzamora AC. Exercise training restores oxidative stress and nitric oxide synthases in the rostral ventrolateral medulla of renovascular hypertensive rats. Free Radic Res 2015; 49: 1335–1343. [DOI] [PubMed] [Google Scholar]
- 11.Sakata S, Hanaoka T, Ishizawa R, Keiko I, Yoshihiro T, Hidetaka I, Akira M, Hidefumi W, Akira N. Poor Wheel-Running Exercise Can Decrease Blood Pressure through Hormonal Control and Increase Endurance Exercise Capacity in Middle-Aged Normal Rats. J Biosci Med 2015, 3, 10–24. [Google Scholar]
- 12.Chang D, Zhang X, Rong S, Sha Q, Liu P, Han T, Pan H. Serum antioxidative enzymes levels and oxidative stress products in age-related cataract patients. Oxid Med Cell Longev. 2013;2013:587826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sun Y, Oberley LW, Li Y. A simple method for clinical assay of superoxide dismutase, Clin Chem 1988, 34: 497–500. [PubMed] [Google Scholar]
- 14.Paglia DE, Valentine WN. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med. 1967;70:158–169. [PubMed] [Google Scholar]
- 15.Zhang L, Xu Z, Wu Y, Liao J, Zeng F, Shi L. Akt/eNOS and MAPK signaling pathways mediated the phenotypic switching of thoracic aorta vascular smooth muscle cells in aging/hypertensive rats. Physiol Res. 2018;67:543–553. [DOI] [PubMed] [Google Scholar]
- 16.Qiu F, Liu X, Zhang Y, Wu Y, Xiao D, Shi L. Aerobic exercise enhanced endothelium-dependent vasorelaxation in mesenteric arteries in spontaneously hypertensive rats: the role of melatonin. Hypertens Res. 2018;41:718–729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zamo FS, Barauna VG, Chiavegatto S, Irigoyen MC, Oliveira EM. The renin–angiotensin system is modulated by swimming training depending on the age of spontaneously hypertension rats. Life Sci 2011; 89: 93–99. [DOI] [PubMed] [Google Scholar]
- 18.Ciampone S, Borqes R, de Lima IP, Mesquita FF, Cambiucci EC, Gontijo JA. Long-term exercise attenuates blood pressure responsiveness and modulates kidney Angiotensin II signalling and urinary sodium excretion in SHR. J Renin Anqiotensin Aldosterone Syst 2011; 12: 394–403. [DOI] [PubMed] [Google Scholar]
- 19.Ceci R, Beltran Valls MR, Duranti G, Dimauro I, Quaranta F, Pittaluga M, Sabatini S, Caserotti P, Parisi P, Parisi A, Caporossi D. Oxidative stress responses to a graded maximal exercise test in older adults following explosive-type resistance training. Redox Biol 2014; 43: 65–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhu L, Yu J, Jia B, Zhao F, Tang M, Hu L, Lin F. Effect of losartan with folic acid on plasma homocysteine and vascular ultrastructural changes in spontaneously hypertensive rats. Int J Exp Pathol 2015; 8: 12908–12914. [PMC free article] [PubMed] [Google Scholar]
- 21.Rizzoni D, Agabiti-Rosei E. Structural abnormalities of small resistance arteries in essential hypertension. Intern Emerg Med. 2012;7:205–212. [DOI] [PubMed] [Google Scholar]
- 22.Sibal L, Aqarwal SA, Home PD, Boger RH. The role of asymmetric dimethylarginine (ADMA) in endothelial dysfunction and cardiovascular disease. Curr Cardiol Rev 2010; 6: 82–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Roque FR, Briones AM, García-Redondo AB, Galán M, Martínez-Revelles S, Avendaño MS, Cachofeiro V, Fernandes T, Vassallo DV, Oliveira EM, Salaices M. Aerobic exercise reduces oxidative stress and improves vascular changes of small mesenteric and coronary arteries in hypertension. Br J Pharmacol 2013;168:686–703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Polonia JJ, Guerreiro M, Guimarães S, Garrett J. Postsynaptic alpha-adrenoceptor subtypes in the internal carotid, mesenteric, splenic, renal and femoral vascular beds of the dog. Arch Int Pharmacodyn Ther. 1986.) [PubMed] [Google Scholar]
- 25.Sun MW, Qian FL, Wang J, Tao T, Guo J, Wang L, Lu AY, Chen H. Low-intensity voluntary running lowers blood pressure with simultaneous improvement in endothelium-dependent vasodilatation and insulin sensitivity in aged spontaneously hypertensive rats. Hypertens Res. 2008;31(3):543–52.) [DOI] [PubMed] [Google Scholar]
- 26.Buckwalter JB, Naik JS, Valic Z, Clifford PS. Exercise attenuates alpha-adrenergic-receptor responsiveness in skeletal muscle vasculature. J Appl Physiol (1985). 2001;90(1):172–8.) [DOI] [PubMed] [Google Scholar]
- 27.Insulin secretion and glucose kinetics during exercise with and without pharmacological alpha(1)- and alpha(2)-receptor blockade. [DOI] [PubMed]
- 28.Carter RW, Kanagy NL. Mechanism of enhanced calcium sensitivity and alpha 2-AR vasoreactivity in chronic NOS inhibition hypertension. [DOI] [PubMed]
- 29.Mukundan H, Kanagy NL. Ca2+ influx mediates enhanced alpha2-adrenergic contraction in aortas from rats treated with NOS inhibitor. Am J Physiol Heart Circ Physiol. 2001;281(5):H2233–40. [DOI] [PubMed] [Google Scholar]
- 30.Carter RW, Kanagy NL. Mechanism of enhanced calcium sensitivity and alpha 2-AR vasoreactivity in chronic NOS inhibition hypertension. Am J Physiol Heart Circ Physiol. 2003;284(1):H309–16. [DOI] [PubMed] [Google Scholar]
- 31.Ceriello A, Novials, Orteqa E, Canivell S, La Sala L, Pujadas G, Bucciarelli L, Rondinelli M, Genovese S. Vitamin C further improves the protective effect of glucagon-like peptide-1 on acute hypoglycemia-induced oxidative stress, inflammation, and endothelial dysfunction in Type 1 diabetes. Diabetes Care 2013; 36: 4104–4108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kim YW, Byzova TV. Oxidative stress in angiogenesis and vascular disease. Blood 2014; 123: 625–631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Miyagawa K, Ohashi M, Yamashita S, Kojima M, Sato K, Ueda K, Dohi Y. Increased oxidative stre ss impairs endothelial modulation of contractions in arteries from spontaneously hypertensive rats. J Hypertens 2007; 25: 415–421. [DOI] [PubMed] [Google Scholar]
- 34.Sinha N, Dabla PK. Oxidative stress and antioxidants in hypertension-a current review. Curr Hypertens Rev 2015; 11: 132–142. [DOI] [PubMed] [Google Scholar]
- 35.Parker L, McGuckin TA, Leicht AS. Influence of exercise intensity on systemic oxidative stress and antioxidant capacity. Clin Physiol Funct Imaqing 2014; 34: 377–383. [DOI] [PubMed] [Google Scholar]
- 36.Gomez-Cabrera MC, Domenech E, Viña J. Moderate exercise is an antioxidant: upregulation of antioxidant genes by training. Free Radic Biol Med 2008; 44: 126–131. [DOI] [PubMed] [Google Scholar]
- 37.Rodrigo R, Libuy M, Feliu F. Oxidative stress-related biomarkers in essential hypertension and ischemia-reperfusion myocardial damage. Dis Marker 2013, 35(6): 773–790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Tung BT, Rodríguez-Bies E, Ballesteros-Simarro M, Motilva V, Navas P, López-Lluch G. Modulation of endogenous antioxidant activity by resveratrol and exercise in mouse liver is age dependent. J Gerontol A Biol Sci Med Sci 2014; 69: 398–409. [DOI] [PubMed] [Google Scholar]
- 39.Hong H, Johnson P. Antioxidant enzyme activities and lipid peroxidation levels in exercised and hypertensive rat tissues. Int J Biochem Cell Biol 2005; 27: 923–931. [DOI] [PubMed] [Google Scholar]
- 40.Camiletti-Moirón D, Aparicio VA, Nebot E, Medina G, Martínez R, Kapravelou G, Andrade A, Porres JM, López-Jurado M, Aranda P. High-protein diet induces oxidative stress in rat brain: protective action of high-intensity exercise against lipid peroxidation. Nutr Hosp 2014; 31: 866–874. [DOI] [PubMed] [Google Scholar]
- 41.Azizbeigi K, Stannard SR, Atashak S, Haghighi MM. Antioxidant enzymes and oxidative stress adaptation to exercise training: comparison of endurance, resistance, and concurrent training in untrained males. J Exerc Sci & Fit 2014; 12: 1–6. [Google Scholar]
- 42.Cho JS, Oh YJ, Kim OS, Na S. The effects of arginase inhibitor on lung oxidative stress and inflammation caused by pneumoperitoneum in rats. Anesthesiol. 2015;15:129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Shimokawa H, Matoba T. Hydrogen peroxide as an endothelium-derived hyperpolarizing factor. Pharmacol Res 2004; 49: 543–549. [DOI] [PubMed] [Google Scholar]
- 44.Shimokawa H Hydrogen peroxide as an endothelium-derived hyperpolarizing factor. Pflugers Arch 2010; 459: 915–922. [DOI] [PubMed] [Google Scholar]
- 45.Shimokawa H, Morikawa K. Hydrogen Peroxide is an endothelium-derived hyperpolarizing factor in animals and humans. J Mol Cell Cardiol 2005; 39: 725–732. [DOI] [PubMed] [Google Scholar]
- 46.Fujimoto S, Mori M, Tsushima H. Mechanisms underlying the hydrogen peroxide-induced, endothelium-independent relaxation of the norepinephrine-contraction in guinea-pig aorta. Eur J Pharmacol 2003; 459: 659–73. [DOI] [PubMed] [Google Scholar]