Abstract
Aims/Introduction
Diabetes mellitus is an established risk factor for stroke and maybe associated with poorer outcomes after stroke. The aims of the present literature review were to determine: (i) the prevalence of diabetes in acute stroke patients through a meta‐analysis; (ii) the association between diabetes and outcomes after ischemic and hemorrhagic stroke; and (iii) to review the value of glycated hemoglobin and admission glucose‐based tests in predicting stroke outcomes.
Materials and Methods
Ovid MEDLINE and EMBASE searches were carried out to find studies relating to diabetes and inpatient stroke populations published between January 2004 and April 2017. A meta‐analysis of the prevalence of diabetes from included studies was undertaken. A narrative review on the associations of diabetes and different diagnostic methods on stroke outcomes was carried out.
Results
A total of 66 eligible articles met inclusion criteria. A meta‐analysis of 39 studies (n = 359,783) estimated the prevalence of diabetes to be 28% (95% confidence interval 26–31). The rate was higher in ischemic (33%, 95% confidence interval 28–38) compared with hemorrhagic stroke (26%, 95% confidence interval 19–33) inpatients. Most, but not all, studies found that acute hyperglycemia and diabetes were associated with poorer outcomes after ischemic or hemorrhagic strokes: including higher mortality, poorer neurological and functional outcomes, longer hospital stay, higher readmission rates, and stroke recurrence. Diagnostic methods for establishing diagnosis were heterogeneous between the reviewed studies.
Conclusions
Approximately one‐third of all stroke patients have diabetes. Uniform methods to screen for diabetes after stroke are required to identify individuals with diabetes to design interventions aimed at reducing poor outcomes in this high‐risk population.
Keywords: Diabetes, Outcomes, Stroke
Introduction
Diabetes mellitus is a highly prevalent and growing chronic disease affecting an estimated 415 million people globally in 2015, and is predicted to affect 642 million people by 20401. Given that diabetes is a well‐recognized risk factor for neurovascular disease2, 3, 4, it is postulated that a significant proportion of stroke inpatients will have comorbid diabetes mellitus. A large, international, multicenter case–control study across 32 countries (n = 26,919) showed that diabetes, defined using a threshold of glycated hemoglobin (HbA1c) of ≥6.5% (48 mmol/mol), was found in 26% of acute stroke inpatients compared with 22% of non‐stroke controls. Those with diabetes had higher odds (odds ratio [OR] 1.33, 95% confidence interval [CI] 1.18–1.5) of ischemic stroke compared to those with hemorrhagic stroke (OR 0.72, 95% CI 0.6–0.87)3. Additionally, studies have estimated that approximately 20–33% of acute stroke inpatients may have diabetes3, 5, 6. A problem in the literature remains the lack of consistency in the definition of the diagnosis of diabetes.
A number of studies have shown an association between comorbid diabetes and increased mortality7, 8, length of hospital stay, readmission rates, and poorer functional and rehabilitation outcomes after stroke9, 10, 11, 12. In contrast, other studies have reported no significant differences in post‐stroke outcomes between people with or without diabetes13, 14.
The best measure of dysglycemia that predicts adverse stroke outcomes is unknown. Some researchers argue that it is the acute or ‘stress’ hyperglycemia; that is, peaks of blood glucose levels during an acute stroke admission, which confers poorer outcomes,15, 16 whereas others propose that it is chronic dysglycemia that drives the pathological processes in stroke patients8, 17. The uncertainty in the literature offers an opportunity for further research to inform best clinical practice.
Compared with fasting blood glucose (FBG), testing of HbA1c has the advantage of providing an average measure of glycaemia over the past 120 days, thereby reducing the potential for misdiagnosis as a result of stress hyperglycemia18. Testing requires only one blood draw and does not require the patient to be fasted, and as such, has the potential to be utilized in the hospital setting for routine screening of diabetes mellitus19. A limitation of the use of HbA1c relates to conditions that affect red blood cell count and the survival time of red blood cells, such as anemia or other hemoglobinopathies18. Previous studies have shown that HbA1c can predict the risk of incident stroke20. With the increasing burden of diabetes, there might be a role for routine HbA1c testing in all people admitted with stroke21 in order to identify and improve glycemic management22, 23.
The aims of the present literature review were: (i) to estimate the prevalence of recognized and unrecognized diabetes in stroke populations from the included studies through a meta‐analysis; (ii) to examine the associations of acute hyperglycemia and diabetes and outcomes after ischemic or hemorrhagic stroke; and (iii) to review the value of HbA1c compared with admission serum glucose‐based tests in predicting stroke outcomes.
Methods
Two reviewers carried out a literature search using both Ovid Medline and Ovid Embase databases to include articles published in English between 1 January 2004 and 1 April 2017. The following medical subject headings (MeSH) were combined: ‘stroke,’ ‘stroke, lacunar,’ ‘cerebral hemorrhage,’ ‘cerebral infarction,’ ‘cerebrovascular accident,’ ‘diabetes mellitus,’ ‘hyperglycemia,’ ‘stress, physiological,’ ‘length of stay,’ ‘readmission,’ ‘patient readmission,’ ‘hemoglobin A, glycosylated,’ ‘morbidity,’ ‘mortality’ and ‘prevalence’ and their variants. Articles published in English that described the prevalence of diabetes mellitus or acute hyperglycemia in a stroke population and/or its effects on mortality, neurological recovery, rehabilitation outcomes, inpatient length of stay, readmission rates and stroke recurrence were included in the present review. Additional articles were obtained by manually reviewing the reference lists of included studies.
A meta‐analysis of the prevalence of diabetes in stroke cohorts was carried out of 39 studies that included a clear definition of diagnosis for diabetes based on either patient history, use of antidiabetic medications or biochemical diagnostic methods. Thrombolysis studies were excluded in the meta‐analysis of diabetes prevalence in stroke patients, as these studies would have selected for a subgroup of ischemic stroke patients receiving thrombolysis, but were included in the outcome analysis. Given that stress hyperglycemia might overestimate the prevalence of diabetes in studies using glucose‐based criteria to diagnose diabetes, a further meta‐analysis of diabetes prevalence was carried out on studies using HbA1c as diagnostic criteria for diabetes. Variables included in the analysis were the number of participants, type of stroke and the prevalence of study participants with known diabetes. The meta‐analysis was carried out in Stata 12.1 using the metaprop procedure (StataCorp, College Station, TX, USA; https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4373114). No ethics approval was required for this literature review.
Results
A total of 2,242 articles were identified during the literature search. After excluding duplicates and reviewing titles and abstracts to meet our inclusion criteria, a total of 66 articles containing data from 522,645 patients were included. Details regarding type and year of study, number of participants, study setting, diagnostic criteria for diabetes, type of stroke and outcomes measured are displayed in Tables 1, 2, 3, 4.
Table 1.
Relationship between diabetes status and neurological function and rehabilitation outcome in stroke inpatients
| Study | Sample size | Diabetes diagnosis | Patient population | Outcome measure | Poorer outcomes observed in patients with: | ||
|---|---|---|---|---|---|---|---|
| No diabetes | No significant difference | Diabetes | |||||
| Tuttolomondo et al.14 | 306 | Known diabetes treated by diet, oral hypoglycemic agents or insulin | AIS | mRS | ✓ | ||
| Scandinavian stroke scale | ✓ | ||||||
| Tanaka et al.26 | 242 | OGTT or HbA1c ≥6.5% or random BGL ≥11.1 mmol/L | AIS | mRS | ✓ | ||
| NIHSS (increased in ≥2 points in first 14 days) | ✓ | ||||||
| Roquer et al.55 | 1,088 | Previous diagnosis or HbA1c ≥6.5% | AIS | mRS | ✓ | ||
| Lei et al.8 | 1,877 | History or FBG or random BGL | AIS | mRS | ✓ | ||
| Tang et al.56 | 419 | FBG or OGTT or HbA1c | AIS (Thrombolysis only) | NIHSS and mRS | ✓ | ||
| Wang et al.38 | 1,438 | FBG or 2‐h post glucose | AHS | mRS | ✓ | ||
| Saxena et al.57 | 2,653 | Medical history | AHS | mRS | ✓ | ||
| Ripley et al.13 | 367 | Medical history | Both | FIM | ✓ | ||
| Koennecke et al.50 | 16,518 | Random FBG level or a history of diabetes | Both | mRS | ✓ | ||
| Piernik‐Yoder et al.12 | 35,243 | ICD‐9 coding | Both | FIM | ✓ | ||
| FIM gain | ✓ | ||||||
Studies are ordered in priority of stroke type and year of study. AHS, acute hemorrhagic stroke; AIS, acute ischemic stroke; BGL, blood glucose level; FBG, fasting blood glucose; FIM, Functional Independence Measure; HbA1c, glycated hemoglobin; mRS, modified Rankin Score; NIHSS, National Institute of Health Stroke Scale; OGTT, oral glucose tolerance test.
Table 2.
Relationship between diabetes status and length of stay, readmission rates or stroke recurrence in stroke inpatients
| Study | Sample size | Diabetes diagnosis | Patient population | Outcome of interest | Poorer outcomes observed in: | ||
|---|---|---|---|---|---|---|---|
| No diabetes | No significant difference | Diabetes | |||||
| Sun et al.39 | 9,766 | History or FBG | AIS | Stroke recurrence (1 year) | ✓ | ||
| AHS | Stroke recurrence (1 year) | ✓ | |||||
| Tseng et al.42 | 468 | ICD‐9‐CM coding | Both | Readmission (1 year) | ✓ | ||
| Li et al.11 | 1,194 | ICD‐9‐CM coding | AIS | Readmission (1 month) | ✓ | ||
| Readmission (6 months) | ✓ | ||||||
| Readmission (1 year) | ✓ | ||||||
| Wu et al.58 | 2,186 | Medical history | AIS | Stroke recurrence (3 months and 1 year) | ✓ | ||
| Sun et al.39 | 9,766 | History or FBG | AIS | Length of stay | ✓ | ||
| AHS | Length of stay | ✓ | |||||
| Delbari et al.54 | 953 | History or FBG | AIS | Length of stay | ✓ | ||
| Piernik‐Yoder et al.12 | 35,243 | ICD‐9 coding | Both | Length of stay | ✓ | ||
| Nakagawa et al.47 | 740 | Unspecified | AIS | Length of stay | ✓ | ||
Studies were arranged in order of stroke type and year of publication. AHS, acute hemorrhagic stroke; AIS, acute ischemic stroke; FBG, fasting blood glucose; ICD‐9‐CM, International Classification of Diseases Ninth Revision Clinical Modification.
Table 3.
Relationship between diabetes status and mortality in stroke inpatients
| Study | Sample size | Diabetes diagnosis | Patient population | Outcome of interest | Poorer outcomes observed in: | ||
|---|---|---|---|---|---|---|---|
| No diabetes | No significant difference | Diabetes | |||||
| Kamalesh et al.45 | 48,733 | Medical records | AIS | Mortality (60 days and 1 year post‐discharge) | ✓ | ||
| Lei et al.8 | 1,877 | Medication use or FBG ≥7.0 mmol/L or random blood glucose ≥11.0 mmol/L or self‐reported | AIS | Mortality (3 months and 1 year post‐discharge) | ✓ | ||
| Saxena et al.57 | 2,653 | Medical records | AHS | Death and major disability (mRS 3–6) | ✓ | ||
| Wang et al.38 | 1,438 | FBG or 2‐h post glucose | AHS | Mortality (≥1 month) | ✓ | ||
| Sun et al.39 | 9,766 | ICD‐9CM codes | Both | In‐hospital mortality | ✓ | ||
| Koennecke et al.50 | 16,518 | FBG ≥7 mmol/L or a history of diabetes | Both | In‐hospital mortality | ✓ | ||
| Eriksson et al.7 | 12,375 | Medical records | Both | Median survival | ✓ | ||
| Braun et al.51 | 537 | History or prescribed anti‐diabetic treatment or blood glucose ≥11.1mmol/L | Both | In‐hospital mortality | ✓ | ||
Studies were arranged in order of stroke type and year of publication. AHS, acute hemorrhagic stroke; AIS, acute ischemic stroke.
Table 4.
Glycated hemoglobin as a continuous measure and its relationship to post‐stroke outcomes
| Study | Sample size | Patient population | Outcome of interest | HbA1c association with outcome of interest |
|---|---|---|---|---|
| Wu et al.58 | 1,540 | AIS | Stroke recurrence (1 year) | Positive association |
| Lei et al.8 | 1,877 | AIS | Mortality | Positive association |
| Hjalmarsson et al.17 | 501 | AIS | Mortality (30 days and 12 months) | Positive association |
| Stroke severity (NIHSS ≥7) | Positive association | |||
| Masrur et al.71 | 58,265 | AIS (thrombolysis only) | Mortality | Positive association |
| Xu et al.59 | 2,137 | General medical inpatients | Cardiovascular mortality | U‐shaped association |
Studies were arranged in order of stroke type and year of publication. AIS, acute ischemic stroke; NIHSS, National Institute of Health Stroke Scale.
Prevalence of diabetes
The present meta‐analysis of 39 studies totaling 359,783 patients estimated that the prevalence of diabetes in all stroke inpatients was 28% (95% CI 26–31), with significant heterogeneity in the proportion of diabetes between the studies (Figure 1; P = 0.017). The prevalence of diabetes was higher in people with ischemic stroke (33%, 95% CI 28–38) compared with hemorrhagic stroke (26%, 95% CI 19–33) and in studies that included both stroke types (24%, 95% CI 20–28). A meta‐analysis of studies using HbA1c alone to diagnose diabetes estimated the prevalence of diabetes in all stroke types to be 37% (95% CI 27–47; Figure 2).
Figure 1.
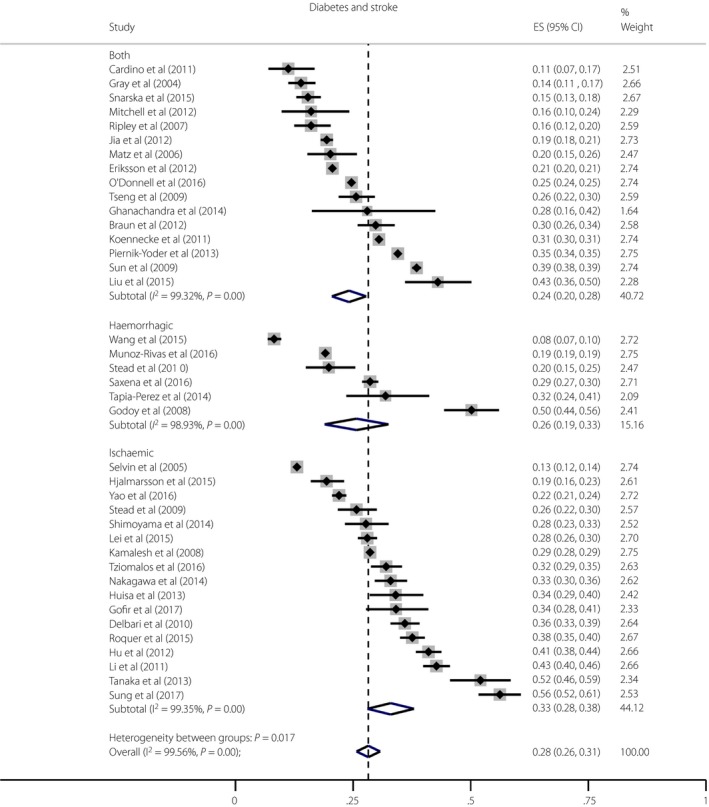
Meta‐analysis of the prevalence of diabetes in studies of ischemic and/or hemorrhagic stroke patients. A total of 39 studies in total were meta‐analyzed: 17 studies included only ischemic stroke, six studies included only hemorrhagic stroke, and 16 studies included both ischemic and hemorrhagic stroke. Heterogeneity testing (I 2) was carried out. Only full articled studies with a clear definition of diagnosis for diabetes based on either history or antidiabetic medications including insulin or biochemical diagnostic measures were included. Thrombolysis studies were excluded. CI, confidence interval; ES, effect size.
Figure 2.
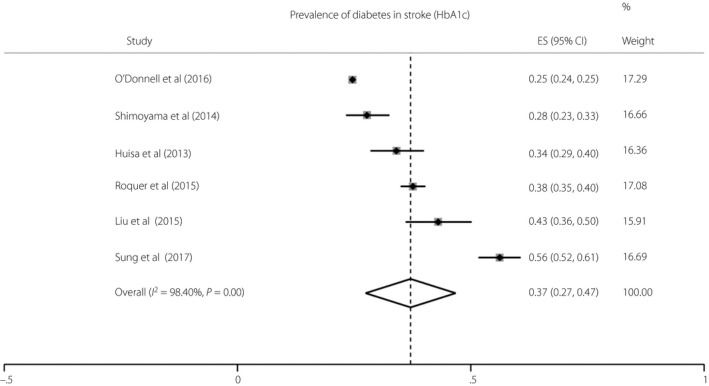
Meta‐analysis of the prevalence of diabetes in studies of ischemic and/or hemorrhagic stroke patients using glycated hemoglobin (HbA1c) alone as the diagnostic criteria for diabetes mellitus. A total of six studies were meta‐analyzed: four studies included only ischemic stroke, and two studies included both ischemic and hemorrhagic stroke. Heterogeneity testing (I 2) was carried out. Thrombolysis studies were excluded. CI, confidence interval; ES, effect size.
The general prevalence of known diabetes in the included studies ranged between 8.2 and 56.2%, and the prevalence of previously unrecognized diabetes mellitus ranged between 5 and 33% (Table S1)5, 6, 21, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33. After excluding studies that used glucose‐based criteria to diagnose diabetes, the prevalence of known diabetes ranged between 16 and 56.2%, and undiagnosed diabetes between 5 and 30.9%. In studies using HbA1c and a history of diabetes as a diagnostic criteria, an estimated 24.7–56.2% of stroke patients had known diabetes.
There were five studies34, 35, 36, 37, 38 that included purely hemorrhagic stroke, with a prevalence of diabetes ranging between 8.2 and 50.2%.
Geographically, the mean prevalence of diabetes in stroke inpatients was highest in Southeast Asian (Singapore39, India32, Indonesia40) and East Asian countries (China8, 29, 38, 41, Japan16, 26, Taiwan11, 42, 43, 44), which were 33.5 and 32.3%, respectively, and mostly in Taiwan11, 43, 44. The mean prevalence in studies from North America12, 13, 20, 21, 30, 37, 45, 46, 47 and Europe5, 7, 17, 34, 35, 36, 48, 49, 50, 51, 52, 53 was 27.5 and 26.4%, respectively. Only one study in the Middle East estimated a prevalence of 36%54. There have been no studies in Australasia examining the effect of acute hyperglycemia or diabetes on post‐stroke outcomes in our defined search period.
Diabetes and outcomes after stroke
The relationship between diabetes and neurological outcome is summarized in Table 1. Six studies12, 26, 50, 55, 56, 57 showed an association between diabetes and poorer neurological outcome, whereas four studies8, 13, 14, 38 did not show a difference. Between these studies, the outcome measures used were the modified Rankin scale (mRS)8, 14, 26, 38, 50, 55, 56, 57, Functional Independence Measure12, 13 and the Scandinavian stroke scale14. The diagnosis of diabetes differed between the studies, with some using medical history alone or medical coding12, 13, 14, 57, whereas others carried out either FBG8, 38, 50, HbA1c55 or both26, 56.
Most studies12, 39, 47 showed an association between diabetes and a longer in‐hospital length of stay among stroke inpatients (Table 2)54. In three studies11, 39, 58, diabetes was associated with increased hospital readmission and stroke recurrence at 3 months58, 6 months11 and 1 year11, 39 after index stroke in people who had ischemic stroke, but not hemorrhagic stroke (Table 2)39, 42. Two out of seven studies showed that diabetes was associated with increased mortality post‐discharge8 and reduced median survival7 compared with people without diabetes (Table 3). In all studies that analyzed HbA1c as a continuous measure, increasing HbA1c was associated with worse stroke outcomes; increased stroke severity, mortality rates and stroke recurrence8, 17, 58, 59 (Table 4).
Differences in diagnostic methods for diabetes
All studies utilized various methods of diagnosing diabetes: 32 used medical history or International Classification of Diseases Ninth Revision coding alone, 15 studies carried out glucose‐based methods (i.e., random or FBG, or the oral glucose tolerance test [OGTT]), 11 studies used HbA1c alone, and six studies combined HbA1c and glucose‐based methods. Just two studies used all three methods. Given the variability in the use of different diagnostic criteria, Figure 3 outlines the clinical advantages and disadvantages of HbA1c compared with traditional glucose‐based testing (i.e., FBG and OGTT).
Figure 3.
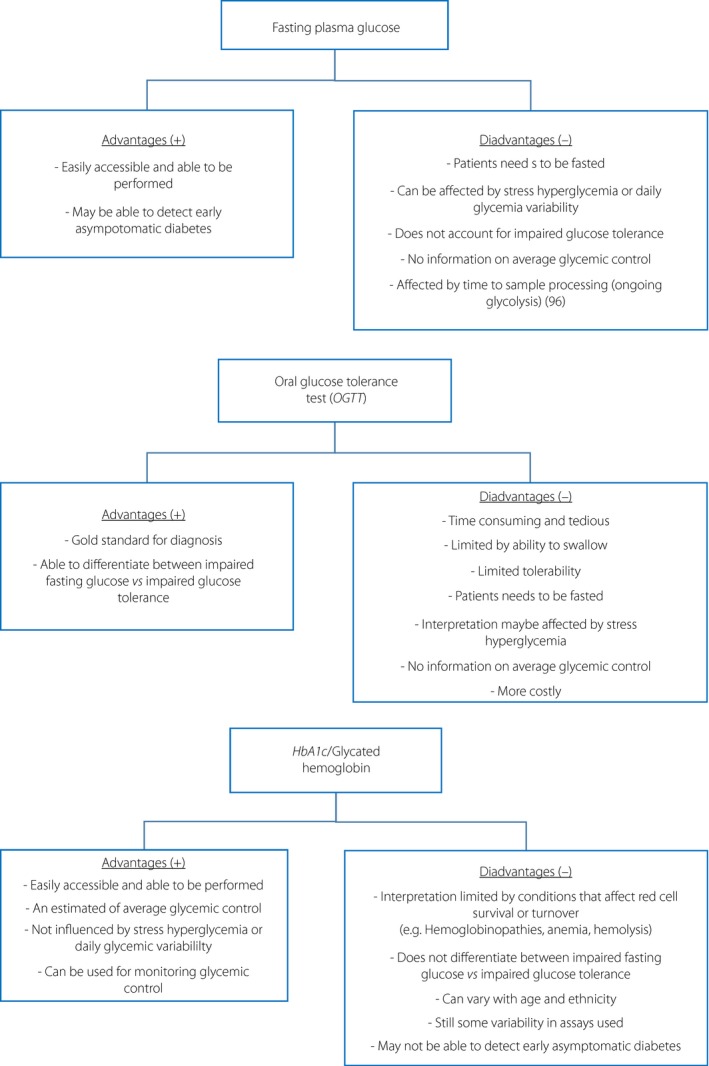
This figure outlines the clinical advantages and disadvantages of glucose‐based diagnostic methods vs glycated hemoglobin.
Discussion
By undertaking a meta‐analysis, we showed that the combined mean prevalence of diabetes in ischemic and/or hemorrhagic stroke studies was 28%. The present review showed that the presence of acute hyperglycemia and diabetes was associated with poorer stroke outcomes. We also showed that significant heterogeneity exists in the diagnostic methods for diabetes within the current literature.
The present meta‐analysis showed that the mean prevalence of diabetes was higher in stroke patients compared with the general inpatient population, which ranges between 20 and 33%60, 61, 62, 63, and higher in ischemic compared with hemorrhagic stroke patients. The higher prevalence of diabetes in ischemic compared with hemorrhagic stroke is consistent with the findings of a recent systematic review and meta‐analysis of risk factors between ischemic and hemorrhagic stroke64.
An important observation in the present review of the literature is the heterogeneity in the method of diagnosing diabetes. The phenomenon of stress hyperglycemia65 is a significant confounder that might overestimate the prevalence of diabetes in studies using glucose‐based testing (i.e., FBG or OGTT). Despite this, studies using HbA1c alone still estimated the prevalence of known and undiagnosed diabetes to range between 16–56.2% and 5–30.9%, respectively.
Stress hyperglycemia is a well‐recognized phenomenon that occurs after any acute illness, and is usually detected during a hospital admission. It is defined as ‘hyperglycemia resolving spontaneously after dissipation of an acute illness,’ which can also occur in people with pre‐existing diabetes66. Three prospective observational studies33, 67, 68 showed that 50–70% of people with hyperglycemia on admission had a normal OGTT at 3 or 6 months post‐stroke, suggesting that stress hyperglycemia might be transient in many acute stroke inpatients. Post‐stroke hyperglycemia, defined as an elevated random or FBG, in people with and without diabetes, is common within hours of stroke49, 65, and has been independently associated with poorer outcomes after stroke, particularly in those without known diabetes. Identification and correct classification of post‐stroke hyperglycemia are clearly required to better clarify the pathophysiological relationships between glycemic control and stroke. Although FBG remains a commonly measured glycemic parameter to diagnose pre‐diabetes and diabetes69, its accuracy in acute stroke might be affected by stress hyperglycemia49, 67, 68. In a majority of the studies included in the present review, an admission random glucose or a FBG was carried out and included in the analysis of the outcomes.
Stress hyperglycemia has been associated with poorer functional outcomes15, 17, 40, 70 and mortality71, irrespective of diabetes status after an acute ischemic stroke. However, more studies have found that stress hyperglycemia has been associated with infarct volume growth, early neurological deterioration (increase of ≥4 points on National Institute of Health Stroke Scale)16, poorer functional outcome12, 40, 41, 71, 72, higher mortality32, 43, 46, 53, 73 and longer hospital stay40 in people without a history of diabetes, including patients who underwent thrombolysis71, 74, 75, 76 and mechanical thrombectomy77. This association remained even after adjusting for stroke severity and stroke subtype. The definition of diabetes in these studies was, however, heterogeneous.
In contrast, two prospective observational studies52, 78 did not find a direct relationship between stress hyperglycemia and poorer stroke outcomes, but found that stress hyperglycemia was associated with more severe stroke and rather the severity of the stroke that led to poorer outcomes.
On a physiological and radiological level, stress hyperglycemia has been associated with a larger infarct volume on presentation32, 79, and might contribute to poorer stroke recovery through impairment of the fibrinolytic process80, 81 and delayed reperfusion of the ischemic penumbra82. In animal studies, stress hyperglycemia appears to induce further cerebral damage by inducing a pro‐inflammatory response83, exacerbating brain edema surrounding intracranial hemorrhage84, disrupting the blood–brain barrier79 and increasing the risk of hemorrhagic transformation85, 86.
In acute hemorrhagic stroke, stress hyperglycemia has also been found to be associated with early mortality36, 57, 87 and worse functional outcome34, 36, 37, 48, 57, irrespective of prior diabetes status34 and after adjusting for stroke severity and subtype34, 36, 57, 87. In a study that included all stroke patients, Snarska et al. observed that hyperglycemia as a continuous variable on admission was associated with increased in‐hospital mortality, with a higher BGL threshold in those with diabetes (≥12.2 mmol/L) compared with those without diabetes (≥6.3 mmol/L). This risk is likely to be an underestimate, given that diabetic status was recorded from medical records, therefore people with unrecognized diabetes were included in the ‘non‐diabetic’ group48. A separate study34 also found that every increase in 1.0 mmol/L in admission plasma glucose >5.0 mmol/L was associated with a 33% increased likelihood of 30‐day mortality in people with hemorrhagic stroke, after adjusting for diabetic status.
Overall, these studies highlight the importance of detecting hyperglycemia post‐stroke and recognizing the potential for poorer outcomes. However, the variability in the findings of stress hyperglycemia and stroke outcomes between people with and without diabetes raises the question of whether chronic dysglycemia (i.e., existing diabetes) carries some degree of neuroprotection in the setting of post‐stroke stress hyperglycemia. The mechanisms are not clearly understood and warrant further research.
Diabetes has been associated with an increased all‐cause in‐hospital mortality in general hospital inpatients, with a higher contribution from a cerebrovascular cause4, 10, 88, 89, 90. Diabetes is associated with atherothrombotic strokes, whereas cardioembolic strokes are more common in patients without diabetes8, 14, 26, 46.
Although there is some conflicting evidence in studies of people with ischemic13, 14, 51 and hemorrhagic stroke38, diabetes is typically associated with poorer functional outcome8, 12, 26, 27, 50, 55, especially in poorly controlled diabetes (HbA1c >7%)91. Diabetes has also been associated with worse neurological deterioration (decrease of National Institute of Health Stroke Scale points <4 at 24 h, or <8 at day 7), poorer recovery (mRS >1 at 3 months) and increased risk of hemorrhagic development72 in people treated with thrombolysis for ischemic stroke56. An explanation for this might be incomplete recanalization after thrombolysis, as suggested in one study92. Diabetes has been associated with a reduced amount of recovery after rehabilitation12, higher risk of mortality in some7, 8, 71 but not all studies45, 51, 93 and a risk factor for recurrent ischemic stroke11, 39, 58. The effect of diabetes on length of hospital stay has been inconsistent12, 39, 47, 54. The relationship between diabetes and poorer stroke outcomes still remained after adjusting for stroke severity and subtypes.
A larger study by Kamalesh et al.45 who followed 48,733 people from Veteran Hospitals after an acute ischemic stroke found no significant difference in mortality between those with and without diabetes at 60 days and at 1 year. A major limitation of that study was the confounding effect of having a strong male bias (98% men), limiting its generalizability, as women with diabetes might have a higher risk of cardiovascular mortality compared with men94, 95. Furthermore, the diagnosis of diabetes was based solely on discharge reports.
Overall, diabetes has been associated with poorer clinical outcomes, except for mortality, after ischemic stroke, and there appears to be a relationship between increasing HbA1c and poorer clinical outcomes after stroke. The lack of a strong association between diabetes and increased mortality after stroke is an interesting point, and would warrant further larger, prospective studies using a combination of HbA1c as a measure of chronic glycemia and glucose‐based testing to evaluate this association. The exact mechanism to explain the difference between the effects of acute vs chronic dysglycemia on poorer stroke outcomes remains unclear and could have implications in its management.
Only a few studies have examined a hemorrhagic‐stroke‐only population, raising the question of the importance of diabetes and its management in hemorrhagic strokes.
In the limited number of small retrospective studies that recruited only people with hemorrhagic stroke, acute hyperglycemia36, 57, 87 and a history of diabetes48, 57 were both independent predictors of poor outcomes at 14 days (National Institute of Health Stroke Scale >15)36 and 3 months (mRS 3–6)57, and increased mortality36, 57, 87 in people with intracerebral hemorrhage. This might be explained by larger initial intracerebral hematomas in people who present with acute hyperglycemia57.
In a study of 237 people who presented to the emergency department with hemorrhagic stroke37, admission hyperglycemia (random plasma glucose >7.8 mmol/L) predicted early mortality (death within 7 days) and poor functional outcome at discharge (mRS ≥3) only in the non‐diabetic subgroup, again suggesting a difference in pathophysiological effects of acute and chronic hyperglycemia.
Other studies also did not find an association between diabetes and increased in‐hospital mortality35, 38 or hospital length of stay39 in hemorrhagic stroke patients after adjusting for common cardiovascular risk factors including age, hypertension, atrial fibrillation and other comorbidities.
In the present review, the evidence between diabetes and stroke outcomes in hemorrhagic stroke was heterogeneous. The fact that acute hyperglycemia was more strongly associated with poorer stroke outcomes compared with existing diabetes probably reflects the higher severity of stroke in patients with hyperglycemia rather than the effects chronic dysglycemia.
The studies in the present review varied in both the method and timing of diagnosing diabetes. Studies using the OGTT might underestimate the prevalence of diabetes due to the inability to complete the test, but might also overestimate the prevalence of diabetes as a result of stress hyperglycemia.
Two studies that used a combination of FBG, OGTT or HbA1c estimated the prevalence of undiagnosed diabetes in stroke inpatients to be between 27 and 32.7%28, 31. Despite heterogeneous study methods, the consistently high proportion of people with undiagnosed diabetes suggests an urgent need for wider diabetes screening of people in the acute stroke setting.
Studies that compared three glycemic measures – FBG vs random admission glucose vs HbA1c – consistently found that elevated FBG (≥7 mmol/L)96, random admission glucose or blood glucose 2‐h post OGTT (≥11.1 mmol/L)44, 53, 96 were independent predictors of poor neurological outcome44, 96, higher rates of mortality53, 96 and stroke recurrence96 post‐stroke compared with a single HbA1c ≥6.5%. Receiver operating characteristic analysis44 showed a different threshold of FBG in individuals with diabetes (FBG >7.8 mmol/L) compared with individuals without diabetes (FBG >6.05 mmol/L). In one study96, an elevated random glucose ≥11.1 mmol/L was a poor prognostic marker in non‐diabetics or individuals with diabetes with good control (HbA1c <6.5%). Nevertheless, an elevated HbA1c has been shown to be associated with poorer stroke outcomes8, 17, 91 and stroke recurrence58, even after accounting for mean plasma glucose and stroke severity. HbA1c >6.5% has also been found to be a predictor of symptomatic intracerebral hemorrhage after thrombolysis for ischemic stroke72.
Interestingly, a Chinese study carried out by Xu et al.59 that followed 2,137 people with diabetes aged >65 for 10 years showed a U‐shaped relationship between HbA1c and stroke mortality, with a HbA1c inflection point of 6.5%. The significance and reason for this finding are uncertain.
Given these studies show the utility of both traditional glucose‐based criteria and HbA1c in both the diagnosis of diabetes and predictors of stroke outcome, there might be a role in the combined use of these diagnostic tools as a screen for diabetes and a predictor for stroke outcomes. More prospective studies incorporating all three glycemic measures would be useful in confirming its utility.
The present review incorporated a large number of studies over the past 10 years, giving a current overview and understanding of the association between diabetes and stroke outcomes. It is important to note that clinical factors, such as age, sex, body mass index, antihyperglycemic medication and the presence of diabetes complications, could confound the effects of diabetes and stroke outcomes, and might not have been accounted for in some of the studies given their retrospective nature. Hypoglycemia is associated with increased morbidity and mortality97, 98; however, only one study34 recorded hypoglycemia as a covariable to assess its effects on morbidity or mortality in stroke patients.
As the burden of diabetes rises, stroke as a major complication of diabetes is expected to rise. More efficient and accurate ways of screening for diabetes are required to minimize the progressive burden this will have on the global healthcare system. Whether tight control of diabetes or acute hyperglycemia after stroke is beneficial is not within the scope of the present review and remains an important question.
Currently, the literature examining the relationship between diabetes and stroke outcomes has been inconsistent and variable, raising the need for larger prospective studies, with consistent definitions of diabetes and stroke outcome measures. The use of HbA1c as a diagnostic tool for diabetes, and implementation of new recommendations for functional outcome measurement in stroke research99 would provide consistency in future studies to develop clinically robust evidence to guide management.
In conclusion, diabetes is a highly prevalent comorbidity in acute stroke patients, and is associated with poorer stroke outcomes compared with people without diabetes. Acute hyperglycemia is strongly associated with poorer stroke outcomes in people with or without diabetes. Until now, there is significant heterogeneity in the diagnostic methods of diabetes in studies examining the association between diabetes and post‐stroke outcomes. A combination of HbA1c with glucose‐based testing might serve as a solution to this.
Disclosure
VT and EIE are on the Steering Committee of the TEXAIS trial. VT has received travel support/honoraria for speaking/consulting for Medtronic and Daiichi‐Sankyo. EIE was supported by a Viertel Clinical Investigator Award, RACP Fellowship, Sir Edward ‘Weary’ Dunlop Medical Research Foundation research grant and Diabetes Australia Research Program grant. The other authors declare no conflict of interest.
Supporting information
Table S1 Prevalence of previously unrecognized diabetes, known diabetes and pre‐diabetes in stroke inpatients.
J Diabetes Investig 2019; 10: 780–792
References
- 1. International Diabetes Federation . IDF Diabetes Atlas, 7th edn Brussels: International Diabetes Federation, 2015. [Google Scholar]
- 2. Cavender MA, Scirica BM, Raz I, et al Cardiovascular outcomes of patients in SAVOR‐TIMI 53 by baseline hemoglobin A1c. Am J Med 2016; 129: 340.e341–348.e341. [DOI] [PubMed] [Google Scholar]
- 3. O'Donnell MJ, Chin SL, Rangarajan S, et al Global and regional effects of potentially modifiable risk factors associated with acute stroke in 32 countries (INTERSTROKE): a case‐control study. Lancet 2016; 388: 761–775. [DOI] [PubMed] [Google Scholar]
- 4. Hu G, Jousilahti P, Sarti C, et al The effect of diabetes and stroke at baseline and during follow‐up on stroke mortality. Diabetologia 2006; 49: 2309–2316. [DOI] [PubMed] [Google Scholar]
- 5. Gray CS, Scott JF, French JM, et al Prevalence and prediction of unrecognised diabetes mellitus and impaired glucose tolerance following acute stroke. Age Ageing 2004; 33: 71–77. [DOI] [PubMed] [Google Scholar]
- 6. Zahra F, Kidwai SS, Siddiqi SA, et al Frequency of newly diagnosed diabetes mellitus in acute ischaemic stroke patients. J Coll Physicians Surg Pak 2012; 22: 226–229. [PubMed] [Google Scholar]
- 7. Eriksson M, Carlberg B, Eliasson M. The disparity in long‐term survival after a first stroke in patients with and without diabetes persists: the Northern Sweden MONICA study. Cerebrovasc Dis 2012; 34: 153–160. [DOI] [PubMed] [Google Scholar]
- 8. Lei C, Wu B, Liu M, et al Association between hemoglobin A1C levels and clinical outcome in ischemic stroke patients with or without diabetes. J Clin Neurosci 2015; 22: 498–503. [DOI] [PubMed] [Google Scholar]
- 9. Wu S, Wang C, Jia Q, et al HbA1c is associated with increased all‐cause mortality in the first year after acute ischemic stroke. Neurol Res 2014; 36: 444–452. [DOI] [PubMed] [Google Scholar]
- 10. Papazafiropoulou A, Tentolouris N, Bousboulas S, et al In‐hospital mortality in a tertiary referral hospital: causes of death and comparison between patients with and without diabetes. Exp Clin Endocrinol Diabetes 2010; 118: 315–319. [DOI] [PubMed] [Google Scholar]
- 11. Li HW, Yang MC, Chung KP. Predictors for readmission of acute ischemic stroke in Taiwan. J Formos Med Assoc 2011; 110: 627–633. [DOI] [PubMed] [Google Scholar]
- 12. Piernik‐Yoder B, Ketchum N. Rehabilitation outcomes of stroke patients with and without diabetes. Arch Phys Med Rehabil 2013; 94: 1508–1512. [DOI] [PubMed] [Google Scholar]
- 13. Ripley DL, Seel RT, Macciocchi SN, et al The impact of diabetes mellitus on stroke acute rehabilitation outcomes. Am J Phys Med Rehabil 2007; 86: 754–761. [DOI] [PubMed] [Google Scholar]
- 14. Tuttolomondo A, Pinto A, Salemi G, et al Diabetic and non‐diabetic subjects with ischemic stroke: differences, subtype distribution and outcome. Nutr Metab Cardiovasc Dis 2008; 18: 152–157. [DOI] [PubMed] [Google Scholar]
- 15. Cao W, Ling Y, Wu F, et al Higher fasting glucose next day after intravenous thrombolysis is independently associated with poor outcome in acute ischemic stroke. J Stroke Cerebrovasc Dis 2015; 24: 100–103. [DOI] [PubMed] [Google Scholar]
- 16. Shimoyama T, Kimura K, Uemura J, et al Elevated glucose level adversely affects infarct volume growth and neurological deterioration in non‐diabetic stroke patients, but not diabetic stroke patients. Eur J Neurol 2014; 21: 402–410. [DOI] [PubMed] [Google Scholar]
- 17. Hjalmarsson C, Manhem K, Bokemark L, et al The role of prestroke glycemic control on severity and outcome of acute ischemic stroke. Stroke Res Treat 2015; 2014: 1–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. d'Emden MC, Shaw JE, Colman PG, et al The role of HbA1c in the diagnosis of diabetes mellitus in Australia. Med J Aust 2012; 197: 220–221. [DOI] [PubMed] [Google Scholar]
- 19. Mackey PA, Whitaker MD. Diabetes mellitus and hyperglycemia management in the hospitalized patient. J Nurse Pract 2015; 11: 531. [Google Scholar]
- 20. Selvin E, Coresh J, Shahar E, et al Glycaemia (haemoglobin A1c) and incident ischaemic stroke: the Atherosclerosis Risk in Communities (ARIC) Study. Lancet Neurol 2005; 4: 821–826. [DOI] [PubMed] [Google Scholar]
- 21. Huisa BN, Roy G, Kawano J, et al Original article: glycosylated hemoglobin for diagnosis of prediabetes in acute ischemic stroke patients. J Stroke Cerebrovasc Dis 2013; 22: e564–e567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Norrving B, Kissela B. The global burden of stroke and need for a continuum of care. Neurology 2013; 80: S5–S12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Feigin VL, Forouzanfar MH, Krishnamurthi R, et al Global and regional burden of stroke during 1990‐2010: findings from the Global Burden of Disease Study 2010. Lancet 2014; 383: 245–254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Urabe T, Watada H, Okuma Y, et al Prevalence of abnormal glucose metabolism and insulin resistance among subtypes of ischemic stroke in Japanese patients. Stroke 2009; 40: 1287p. [DOI] [PubMed] [Google Scholar]
- 25. Sulehria SB, Memon MM, Bashir A. To determine the frequency of newly diagnosed diabetes mellitus in patients of acute stroke presenting in tertiary care hospital. Pak J Med Health Sci 2015; 9: 34–36. [Google Scholar]
- 26. Tanaka R, Ueno Y, Miyamoto N, et al Impact of diabetes and prediabetes on the short‐term prognosis in patients with acute ischemic stroke. J Neurol Sci 2013; 332: 45–50. [DOI] [PubMed] [Google Scholar]
- 27. Matz K, Keresztes K, Tatschl C, et al Disorders of glucose metabolism in acute stroke patients: an underrecognized problem. Diabetes Care 2006; 29: 796p. [DOI] [PubMed] [Google Scholar]
- 28. Cardino MJT, Josol CV, Guillermo IM, et al Prevalence and outcomes of unrecognized diabetes mellitus and prediabetes among acute stroke patients with admission Hyperglycemia at the Philippine general hospital: DASH study. Philippine J Int Med 2011; 49: 79–87. [Google Scholar]
- 29. Jia Q, Zheng H, Zhao X, et al Abnormal glucose regulation in patients with acute stroke across China: prevalence and baseline patient characteristics. Stroke 2012; 43: 650–657.658p. [DOI] [PubMed] [Google Scholar]
- 30. Liu A, Carmichael KA, Schallom ME, et al Detecting and managing diabetes mellitus and prediabetes in patients with acute stroke. Diabetes Educ 2015; 41: 592–598. [DOI] [PubMed] [Google Scholar]
- 31. Fonville S, Zandbergen AA, Vermeer SE, et al Prevalence of prediabetes and newly diagnosed diabetes in patients with a transient ischemic attack or stroke. Cerebrovasc Dis 2013; 36: 283–289. [DOI] [PubMed] [Google Scholar]
- 32. Ghanachandra Singh K, Singh SD, Bijoychandra K, et al A study on the clinical profile of stroke in relation to glycaemic status of patients. J Ind Acad Clin Med 2014; 15: 177–181. [Google Scholar]
- 33. Borschmann KN, Ekinci EI, Iuliano S, et al Reducing sedentary time and fat mass may improve glucose tolerance and insulin sensitivity in adults surviving 6 months after stroke: a phase I pilot study. Eur Stroke J 2017; 2: 144–153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Godoy DA, Pinero GR, Svampa S, et al Hyperglycemia and short‐term outcome in patients with spontaneous intracerebral hemorrhage. Neurocrit Care 2008; 9: 217–229. [DOI] [PubMed] [Google Scholar]
- 35. Munoz‐Rivas N, Mendez‐Bailon M, Hernandez‐Barrera V, et al Type 2 diabetes and hemorrhagic stroke: a population‐based study in Spain from 2003 to 2012. J Stroke Cerebrovasc Dis 2016; 25: 1431–1443. [DOI] [PubMed] [Google Scholar]
- 36. Tapia‐Perez JH, Gehring S, Zilke R, et al Effect of increased glucose levels on short‐term outcome in hypertensive spontaneous intracerebral hemorrhage. Clin Neurol Neurosurg 2014; 118: 37–43. [DOI] [PubMed] [Google Scholar]
- 37. Stead LG, Jain A, Bellolio MF, et al Emergency department hyperglycemia as a predictor of early mortality and worse functional outcome after intracerebral hemorrhage. Neurocrit Care 2010; 13: 67–74. [DOI] [PubMed] [Google Scholar]
- 38. Wang Q, Wang D, Liu M, et al Is diabetes a predictor of worse outcome for spontaneous intracerebral hemorrhage? Clin Neurol Neurosurg 2015; 134: 67–71. [DOI] [PubMed] [Google Scholar]
- 39. Sun Y, Toh MP. Impact of diabetes mellitus (DM) on the health‐care utilization and clinical outcomes of patients with stroke in Singapore. Value Health 2009; 12(Suppl 3): S101–S105. [DOI] [PubMed] [Google Scholar]
- 40. Gofir A, Mulyono B, Sutarni S. Hyperglycemia as a prognosis predictor of length of stay and functional outcomes in patients with acute ischemic stroke. Int J Neurosci 2017; 127: 923–929. [DOI] [PubMed] [Google Scholar]
- 41. Yao M, Ni J, Zhou L, et al Elevated fasting blood glucose is predictive of poor outcome in non‐diabetic stroke patients: a sub‐group analysis of SMART. PLoS ONE 2016; 11: e0160674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Tseng MC, Lin HJ. Readmission after hospitalization for stroke in Taiwan: results from a national sample. J Neurol Sci 2009; 284: 52–55. [DOI] [PubMed] [Google Scholar]
- 43. Hu GC, Hsieh SF, Chen YM, et al Relationship of initial glucose level and all‐cause death in patients with ischaemic stroke: the roles of diabetes mellitus and glycated hemoglobin level. Eur J Neurol 2012; 19: 884–891. [DOI] [PubMed] [Google Scholar]
- 44. Sung JY, Chen CI, Hsieh YC, et al Comparison of admission random glucose, fasting glucose, and glycated hemoglobin in predicting the neurological outcome of acute ischemic stroke: a retrospective study. PeerJ 2017; 5: e2948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Kamalesh M, Shen J, Eckert GJ. Long term postischemic stroke mortality in diabetes: a veteran cohort analysis. Stroke 2008; 39: 2727–2731.2725p. [DOI] [PubMed] [Google Scholar]
- 46. Stead LG, Gilmore RM, Bellolio MF, et al Hyperglycemia as an independent predictor of worse outcome in non‐diabetic patients presenting with acute ischemic stroke. Neurocrit Care 2009; 10: 181–186. [DOI] [PubMed] [Google Scholar]
- 47. Nakagawa K, Vento MA, Ing MM, et al Impact of diabetes on prolonged hospital stay among Native Hawaiians and other Pacific Islanders with ischemic stroke. Hawaii J Med Public Health 2014; 73: 4–7. [PMC free article] [PubMed] [Google Scholar]
- 48. Snarska KK, Bachórzewska‐Gajewska H, Kapica‐Topczewska K, et al Hyperglycemia and diabetes have different impacts on outcome of ischemic and hemorrhagic stroke. Arch Med Sci 2017; 13: 100–108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Mitchell EA, Coates VE, Ryan AA, et al Hyperglycaemia monitoring and management in stroke care: policy vs. practice. Diabet Med 2012; 29: 1108–1114. [DOI] [PubMed] [Google Scholar]
- 50. Koennecke HC, Belz W, Berfelde D, et al Factors influencing in‐hospital mortality and morbidity in patients treated on a stroke unit. Neurology 2011; 77: 965–972. [DOI] [PubMed] [Google Scholar]
- 51. Braun KF, Otter W, Sandor SM, et al All‐cause in‐hospital mortality and comorbidity in diabetic and non‐diabetic patients with stroke. Diabetes Res Clin Pract 2012; 98: 164–168. [DOI] [PubMed] [Google Scholar]
- 52. Tziomalos K, Dimitriou P, Bouziana SD, et al Stress hyperglycemia and acute ischemic stroke in‐hospital outcome. Metabolism 2017; 67: 99–105. [DOI] [PubMed] [Google Scholar]
- 53. Roquer J, Giralt‐Steinhauer E, Cerda G, et al Glycated hemoglobin value combined with initial glucose levels for evaluating mortality risk in patients with ischemic stroke. Cerebrovasc Dis 2015; 40: 244–250. [DOI] [PubMed] [Google Scholar]
- 54. Delbari A, Salman Roghani R, Tabatabaei SS, et al A stroke study of an urban area of Iran: risk factors, length of stay, case fatality, and discharge destination. J Stroke Cerebrovasc Dis 2010; 19: 104–109. [DOI] [PubMed] [Google Scholar]
- 55. Roquer J, Rodriguez‐Campello A, Cuadrado‐Godia E, et al Ischemic stroke in prediabetic patients. J Neurol 2014; 261: 1866–1870. [DOI] [PubMed] [Google Scholar]
- 56. Tang H, Zhang S, Yan S, et al Unfavorable neurological outcome in diabetic patients with acute ischemic stroke is associated with incomplete recanalization after intravenous thrombolysis. J Neurointerv Surg 2016; 8: 342–346. [DOI] [PubMed] [Google Scholar]
- 57. Saxena A, Anderson CS, Wang X, et al Prognostic significance of hyperglycemia in acute intracerebral hemorrhage: the INTERACT2 Study. Stroke 2016; 47: 682–688. [DOI] [PubMed] [Google Scholar]
- 58. Wu S, Shi Y, Wang C, et al Glycated hemoglobin independently predicts stroke recurrence within one year after acute first‐ever non‐cardioembolic strokes onset in A Chinese cohort study. PLoS ONE 2013; 8: e80690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Xu L, Chan WM, Hui YF, et al Association between HbA1c and cardiovascular disease mortality in older Hong Kong Chinese with diabetes. Diabet Med 2012; 29: 393–398. [DOI] [PubMed] [Google Scholar]
- 60. Baker ST, Chiang CY, Zajac JD, et al Outcomes for general medical inpatients with diabetes mellitus and new hyperglycaemia. Med J Aust 2008; 188: 340–343. [DOI] [PubMed] [Google Scholar]
- 61. Nanayakkara N, Nguyen H, Churilov L, et al Inpatient HbA1c testing: a prospective observational study. BMJ Open Diabetes Res Care 2015; 3: e000113–e000113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Silverman RA, Thakker U, Ellman T, et al Hemoglobin A1c as a screen for previously undiagnosed prediabetes and diabetes in an acute‐care setting. Diabetes Care 2011; 34: 1908–1912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Wexler DJ, Nathan DM, Grant RW, et al Prevalence of elevated hemoglobin A1c among patients admitted to the hospital without a diagnosis of diabetes. J Clin Endocrinol Metab 2008; 93: 4238–4244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Tsai CF, Anderson N, Thomas B, et al Comparing risk factor profiles between intracerebral hemorrhage and ischemic stroke in chinese and white populations: systematic review and meta‐analysis. PLoS ONE 2016; 11: e0151743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Capes SE, Hunt D, Malmberg K, et al Stress hyperglycemia and prognosis of stroke in nondiabetic and diabetic patients: a systematic overview. Stroke 2001; 32: 2426–2432. [DOI] [PubMed] [Google Scholar]
- 66. Dungan KM, Braithwaite SS, Preiser JC. Stress hyperglycaemia. Lancet 2009; 373: 1798–1807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Dave JA, Engel ME, Freercks R, et al Abnormal glucose metabolism in non‐diabetic patients presenting with an acute stroke: prospective study and systematic review. QJM 2010; 103: 495–503. [DOI] [PubMed] [Google Scholar]
- 68. Vancheri F, Curcio M, Burgio A, et al Impaired glucose metabolism in patients with acute stroke and no previous diagnosis of diabetes mellitus. QJM 2005; 98: 871–878. [DOI] [PubMed] [Google Scholar]
- 69. Furie KL, Kasner SE, Adams RJ, et al Guidelines for the prevention of stroke in patients with stroke or transient ischemic attack: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2011; 42: 227–276. [DOI] [PubMed] [Google Scholar]
- 70. Mandava P, Martini SR, Munoz M, et al Hyperglycemia worsens outcome after rt‐PA primarily in the large‐vessel occlusive stroke subtype. Transl Stroke Res 2014; 5: 519–525. [DOI] [PubMed] [Google Scholar]
- 71. Masrur S, Cox M, Bhatt DL, et al Association of acute and chronic hyperglycemia with acute ischemic stroke outcomes post‐thrombolysis: findings from get with the guidelines‐stroke. J Am Heart Assoc 2015; 4: e002193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Rocco A, Heuschmann PU, Schellinger PD, et al Glycosylated hemoglobin A1 predicts risk for symptomatic hemorrhage after thrombolysis for acute stroke. Stroke 2013; 44: 2134–2138. [DOI] [PubMed] [Google Scholar]
- 73. Tuna M, Manuel DG, Bennett C, et al One‐ and five‐year risk of death and cardiovascular complications for hospitalized patients with hyperglycemia without diagnosed diabetes: an observational study. J Hosp Med 2014; 9: 365–371. [DOI] [PubMed] [Google Scholar]
- 74. Yaghi S, Hinduja A, Bianchi N. The effect of admission hyperglycemia in stroke patients treated with thrombolysis. Int J Neurosci 2012; 122: 637–640. [DOI] [PubMed] [Google Scholar]
- 75. Poppe AY, Majumdar SR, Jeerakathil T, et al Admission hyperglycemia predicts a worse outcome in stroke patients treated with intravenous thrombolysis. Diabetes Care 2009; 32: 617–622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Ahmed N, Davalos A, Eriksson N, et al Association of admission blood glucose and outcome in patients treated with intravenous thrombolysis: results from the Safe Implementation of Treatments in Stroke International Stroke Thrombolysis Register (SITS‐ISTR). Arch Neurol 2010; 67: 1123–1130. [DOI] [PubMed] [Google Scholar]
- 77. Kim JT, Jahan R, Saver JL. Impact of glucose on outcomes in patients treated with mechanical thrombectomy: a post hoc analysis of the solitaire flow restoration with the intention for thrombectomy study. Stroke 2016; 47: 120–127. [DOI] [PubMed] [Google Scholar]
- 78. Meurer WJ, Scott PA, Caveney AF, et al Lack of association between hyperglycaemia at arrival and clinical outcomes in acute stroke patients treated with tissue plasminogen activator. Int J Stroke 2010; 5: 163–166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Huang J, Liu B, Yang C, et al Acute hyperglycemia worsens ischemic stroke‐induced brain damage via high mobility group box‐1 in rats. Brain Res 2013; 1535: 148–155. [DOI] [PubMed] [Google Scholar]
- 80. Gentile NT, Vaidyula VR, Kanamalla U, et al Factor VIIa and tissue factor procoagulant activity in diabetes mellitus after acute ischemic stroke: impact of hyperglycemia. Thromb Haemost 2007; 98: 1007–1013. [DOI] [PubMed] [Google Scholar]
- 81. Luitse MJA, van Seeters T, Horsch AD, et al Admission hyperglycaemia and cerebral perfusion deficits in acute ischaemic stroke. Cerebrovasc Dis 2013; 35: 163–167. [DOI] [PubMed] [Google Scholar]
- 82. Ribo M, Molina C, Montaner J, et al Acute hyperglycemia state is associated with lower tPA‐induced recanalization rates in stroke patients. Stroke 2005; 36: 1705–1709. [DOI] [PubMed] [Google Scholar]
- 83. Khan MA, Schultz S, Othman A, et al Hyperglycemia in stroke impairs polarization of monocytes/macrophages to a protective noninflammatory cell type. J Neurosci 2016; 36: 9313–9325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Song EC, Chu K, Jeong SW, et al Hyperglycemia exacerbates brain edema and perihematomal cell death after intracerebral hemorrhage. Stroke 2003; 34: 2215–2220. [DOI] [PubMed] [Google Scholar]
- 85. Hafez S, Abdelsaid M, El‐Shafey S, et al Matrix metalloprotease 3 exacerbates hemorrhagic transformation and worsens functional outcomes in hyperglycemic stroke. Stroke 2016; 47: 843–851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Gliem M, Klotz L, van Rooijen N, et al Hyperglycemia and PPARgamma antagonistically influence macrophage polarization and infarct healing after ischemic stroke. Stroke 2015; 46: 2935–2942. [DOI] [PubMed] [Google Scholar]
- 87. Kimura K, Iguchi Y, Inoue T, et al Hyperglycemia independently increases the risk of early death in acute spontaneous intracerebral hemorrhage. J Neurol Sci 2007; 255: 90–94. [DOI] [PubMed] [Google Scholar]
- 88. Hyvärinen M, Qiao Q, Tuomilehto J, et al Hyperglycemia and stroke mortality: comparison between fasting and 2‐h glucose criteria. Diabetes Care 2009; 32: 347p. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Zelada H, Bernabe‐Ortiz A, Manrique H. Inhospital mortality in patients with type 2 diabetes mellitus: a prospective cohort study in Lima, Peru. J Diabetes Res 2016; 2016: 7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Sakurai M, Saitoh S, Miura K, et al HbA1c and the risks for all‐cause and cardiovascular mortality in the general Japanese population: NIPPON DATA90. Diabetes Care 2013; 36: 3759–3765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Lattanzi S, Bartolini M, Provinciali L, et al Glycosylated hemoglobin and functional outcome after acute ischemic stroke. J Stroke Cerebrovasc Dis 2016; 25: 1786–1791. [DOI] [PubMed] [Google Scholar]
- 92. Saqqur M, Shuaib A, Alexandrov AV, et al The correlation between admission blood glucose and intravenous rt‐PA‐induced arterial recanalization in acute ischemic stroke: a multi‐centre TCD study. Int J Stroke 2015; 10: 1087–1092. [DOI] [PubMed] [Google Scholar]
- 93. Chonchol M, Katz R, Fried LF, et al Glycosylated hemoglobin and the risk of death and cardiovascular mortality in the elderly. Nutr Metab Cardiovasc Dis 2010; 20: 15–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94. Ballotari P, Venturelli F, Greci M, et al Sex differences in the effect of type 2 diabetes on major cardiovascular diseases: results from a population‐based study in Italy. Int J Endocrinol 2017; 2017: 1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Lin GM, Li YH, Lin CL, et al Gender differences in the impact of diabetes on mortality in patients with established coronary artery disease: a report from the eastern Taiwan integrated health care delivery system of Coronary Heart Disease (ET‐CHD) registry, 1997‐2006. J Cardiol 2013; 61: 393–398. [DOI] [PubMed] [Google Scholar]
- 96. Jing J, Pan Y, Zhao X, et al Prognosis of ischemic stroke with newly diagnosed diabetes mellitus according to hemoglobin A1c Criteria in Chinese Population. Stroke 2016; 47: 2038–2044. [DOI] [PubMed] [Google Scholar]
- 97. McCoy RG, Van Houten HK, Ziegenfuss JY, et al Increased mortality of patients with diabetes reporting severe hypoglycemia. Diabetes Care 2012; 35: 1897–1901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Akirov A, Grossman A, Shochat T, et al Mortality among hospitalized patients with hypoglycemia: insulin related and noninsulin related. J Clin Endocrinol Metab 2017; 102: 416–424. [DOI] [PubMed] [Google Scholar]
- 99. Kwakkel G, Lannin NA, Borschmann K, et al Standardized measurement of sensorimotor recovery in stroke trials: consensus‐based core recommendations from the Stroke Recovery and Rehabilitation Roundtable. Int J Stroke 2017; 12: 451–461. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1 Prevalence of previously unrecognized diabetes, known diabetes and pre‐diabetes in stroke inpatients.


