Abstract
Background
Colorectal cancer (CRC) is the third leading cause of cancer-related deaths in the United States. African Americans are disproportionately affected by CRC. Our hypothesis is that driver genes with known and novel mutations have an impact on CRC outcome in this population. Therefore, we investigated the variants’ profiles in a panel of 15 CRC genes.
Patients & Methods
Colorectal specimens (n=140) were analyzed by targeted exome sequencing using an Ion Torrent platform. Detected variants were validated in 36 samples by Illumina sequencing. The novel status of the validated variants was determined by comparison to publicly available databases. Annotated using ANNOVAR and in-silico functional analysis of these variants were performed to determine likely pathogenic variants.
Results
Overall, 121 known and novel variants were validated: APC (27%), AMER1 (3%), ARID1 (7%), MSH3 (12%), MSH6 (10%), BRAF (4%), KRAS (6%), FBXW7 (4%), PIK3CA (6%), SMAD4 (5%), SOX9 (2%), TCF7L2 (2%), TGFBR2 (5%), TP53 (7%). From these validated variants, 12% were novel in 8 genes (AMER1, APC, ARID1A, BRAF, MSH6, PIK3CA, SMAD4, and TCF7L2). Of the validated variants, 23% were non-synonymous, 14% were stopgains, 24% were synonymous and 39% were intronic variants.
Conclusion
We here report the specifics of variants’ profiles of African Americans with colorectal lesions. Validated variants showed that Tumor Suppressor Genes (TSGs) APC and ARID1 and DNA Mismatch repair (MMR) genes MSH3 and MSH6 are the genes with the highest numbers of validated variants. Oncogenes KRAS and PIK3CA are also altered and likely participate in the increased proliferative potential of the mutated colonic epithelial cells in this population.
Keywords: colon, targeted sequencing, African Americans, actionable, druggable
INTRODUCTION
Colorectal Cancer (CRC) is the third leading cause of cancer-related deaths in the US. Its incidence and mortality are higher in African Americans (AAs) than other ethnic groups in the US. The reasons for this disparity are not yet well established [1]. It is thought that the underlying factors behind this disparity are multiple (diet, lifestyle, microbiome, socioeconomic, healthcare access and genetic predispositions). Cancer driver mutations play an important role in the carcinogenic process [2, 3]. There are limited next generation sequencing studies in African Americans with cancer in general and with CRC in particular [4–6]. However, none of these studies investigated cancer specimens along with pre-neoplastic lesions.
Molecularly, CRCs are categorized into those with microsatellite instability (MSI) which are located primarily in the right colon and frequently associate with the CpG island methylator phenotype (CIMP) and those that are microsatellite stable (MSS) but are chromosomally unstable [7–9]. MSI characterizes 10–15% of sporadic CRCs and has been related to a better prognosis compared with MSS colorectal cancer patients [7, 10]. Most MSI CRCs have been assigned previously to defects in MLH1 and MSH2 genes within the DNA Mismatch Repair (MMR) genes machinery. However recent attention has been given to other genes such as MSH3 and MSH6 that were found to be altered and associated with a different category of microsatellite instability within the genome, primarily at tetranucleotide repeats [11].
Several pathogenic gene panels that are frequently mutated in CRC have been designed for targeted sequencing. We examined 15 genes associated with CRC using a Personal Genome Machine (PGM; Ion Torrent-ThermoFisher Scientific; Waltham, MA) sequencing platform for variant discovery, and a MiSeq (Illumina; San Diego, CA) sequencing platform for validation. The 15 genes correspond to two DNA MMR genes, 6 oncogenes and 7 tumor suppressor genes (TSGs). The DNA MMR gene MSH3 have acquired more attention recently in CRC patients as variants in this gene were found to be prevalent in African Americans (50 to 60%) and associate with poor prognosis [11, 12] in contrast with MSI-H phenotype that is driven by altered MLH1 and MSH2 alterations. The 15 genes panel also consisted of 6 oncogenes namely: BRAF, NRAS, KRAS, PIK3CA, SMAD4 and SOX9. KRAS is involved in the pathogenesis of many different malignant tumors, including lung, pancreatic, and colon cancers [13]. Around 30 to 40% of CRCs have KRAS variants [14]. NRAS is a member of a family of oncoproteins that are commonly mutated in cancer. Activating variants in NRAS occur in a subset of CRC but little is known about how the mutant protein contributes to the onset and progression of the disease [14]. BRAF is mutated in 4 to 12% of unselected CRC, particularly those with high microsatellite instability [15]. BRAF mutations in CRC are associated with distinct clinical characteristics and a worse prognosis [16]. PIK3CA encodes for the catalytic p110-alpha subunit of Phosphatidylinositol 3-Kinase (PI3K) alpha, which orchestrates cell responses including cell proliferation, survival, migration and morphology [17]. Activating mutations in PIK3CA were reported in 10 to 15% of colorectal carcinomas [18]. SOX9 has been widely studied in the context of development and cell lineage determination in various tissues. Recent studies have indicated tissue- and context-specific roles of this gene [19].
The gene panel contained 7 tumor suppressor genes (AMER1, APC, ARID1, FBXW7, TCF7L2, TGFBR2, and TP53). APC is one of the key genes in the initiation of polyp formation [20] in both familial adenomatous polyposis (FAP) and FAP-like sporadic CRCs [21]. Current studies have shown mutations of APC in many cancers including CRC. Several studies have suggested that chromosome 18q loss is a critical event during CRC progression and that the SMAD4 tumor suppressor is the primary target for inactivation [22]. Clinical studies have shown that patients retaining heterozygosity at the 18q locus benefit significantly better from treatment with 5-Fluorouracil than patients with loss of heterozygosity (LOH) at this site [23]. AT-Rich Interactive Domain 1A (ARID1A) has recently been identified as a novel tumor suppressor in various tumor types. Loss of ARID1A expression is uncommon and not associated with oncologic outcome but may be related to less invasive pathologic features in CRC [24]. Most CRCs with microsatellite instability (MSI-H) have mutations in a microsatellite sequence encoding Transforming Growth Factor β Receptor II (TGFBR2). Therefore, it is understood that TGFBR2 is defective in these tumors, even though CRC cells with TGFBR2 variants have been described to remain sensitive to TGFβ [25]. Resistance to growth inhibition by TGFβ is standard in a variety of human cancers, emphasizing the importance of intracellular pathways mediated by this polypeptide to the neoplastic process [26]. FBXW7 variants occur in a variety of human cancers including CRC [27]. FBXW7 and TP53 are tumor suppressors intensively implicated in colorectal carcinogenesis [28]. FBXW7 constitutes one of the four subunits of SCF (SKP1-cullin-F-box)-E3 ubiquitin-protein ligase complex, which functions in phosphorylation-dependent ubiquitination [27]. About half of all CRCs show TP53 gene variants, with lower frequencies in proximal tumors and higher frequencies in distal colon and rectal tumors and in those with the microsatellite instability or methylator phenotypes [28]. AMER1 regulates the distribution of the tumor suppressor APC between microtubules and the plasma membrane [29]. It is frequently mutated in colorectal cancer tumors [30]. TCF7L2 is a transcription factor of which polymorphisms have been associated with cancers including colon and prostate [31]. However, this gene’s polymorphisms have been intensively studied in the context of diabetes-associated disorders [32].
In the present study, we examined and validated variants in a panel of cancer genes and assessed their association with disease characteristics in African Americans with colorectal neoplastic lesions.
RESULTS
Clinical and pathological characteristics of patients
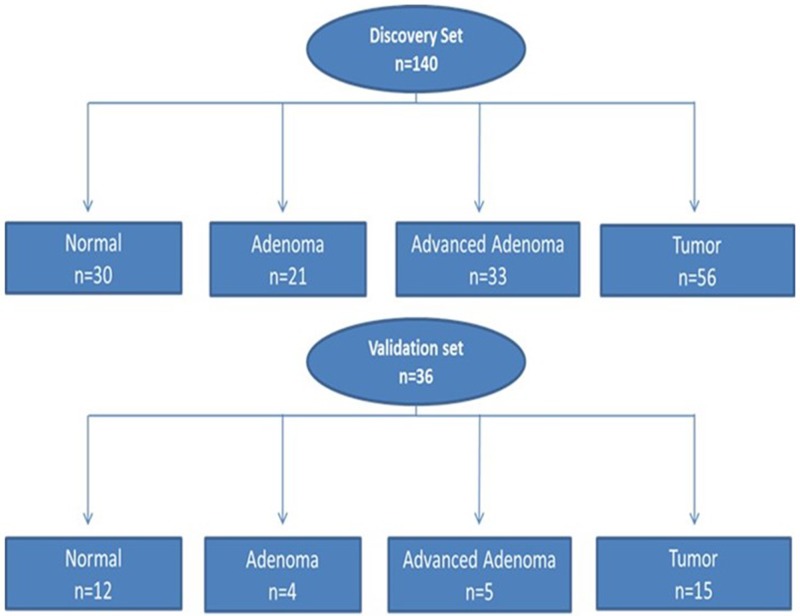
A: Discovery set: The characteristics of the 123 patients from whom the 140 samples were collected are reported in Figure 1. The patients consisted of 69 (57%) males. The age range at the time of diagnosis was 24 to 95 years, with a median of 61 years. With regard to cancer stage, 11% (6/56) were stage I, 34% (19/56) were stage II, 29% (16/56) were stage III, and 4% (2/56) were stage IV (23% (13/56) had no staging data).
B: Validation set: Contained a subset of the discovery set and was made of 36 samples from 26 patients. There were 10 (38%) females and 16 (62%) males. With regard to cancer stage, 35% (9/26) were stage II, 15% (4/26) were stage III and 8% (2/26) stage IV, and 42% (11/26) had no staging data. The age range was from 41 to 88 with median age of 61 years (Table 1).
Figure 1. Flow chart of patient selection for both discovery and validation sets for somatic variants analysis.
Discovery set: 140 samples (n = 123 patients) and validation set: 36 samples (n = 26 patients).
Table 1. Clinico-pathological characteristics of the validation set (26 Patients & 36 Samples).
| Sample ID | Age | Sex | Type of Tissue | Location | TNM | STAGING |
|---|---|---|---|---|---|---|
| CC1018N | 84 | F | N | RIGHT | N | NA |
| CC1018 | 84 | F | T | LEFT | T3N1b | II |
| CC1024 | 73 | F | T | RIGHT | T3N1MX | III |
| CC1028 | 42 | M | T | LEFT | T3N0MX | II |
| CC1028N | 42 | M | N | RIGHT | N | NA |
| CC1029 | 51 | F | T | RIGHT | T2N0MX | II |
| CC1029N | 51 | F | N | LEFT | N | NA |
| CC1036 | 63 | M | T | LEFT | T3N2M1 | IV |
| CC1036N | 63 | M | N | RIGHT | N | NA |
| CC1038 | 54 | M | T | LEFT | T3N0Mx | II |
| CC1038N | 54 | M | N | RIGHT | N | NA |
| CC1053 | 50 | F | T | RIGHT | T3N0M0 | II |
| CC1053N | 50 | F | N | LEFT | N | NA |
| CC1054 | 53 | M | T | RIGHT | T3N0M0 | II |
| CC1054N | 53 | M | N | LEFT | N | NA |
| CC1055 | 79 | F | AA | RIGHT | AA | NA |
| CC1056 | 66 | M | T | LEFT | T1N1MX | III |
| CC1056N | 66 | M | N | RIGHT | N | NA |
| CC1057 | 88 | M | T | LEFT | T3N2M0 | III |
| CC1057N | 88 | M | N | RIGHT | N | NA |
| CC1059 | 60 | F | T | RIGHT | T3N1MX | II |
| CC1060 | 53 | F | T | LEFT | T3N2M1 | IV |
| CC1060N | 53 | F | N | RIGHT | N | NA |
| CC1061 | 63 | M | N | LEFT | N | NA |
| CC1065 | 41 | M | T | LEFT | T3N0M0 | II |
| CC1109 | 62 | F | A | RIGHT | A | NA |
| CC1258 | 52 | M | T | RIGHT | T3N1bMx | III |
| CC1386 | 70 | M | T | LEFT | T3N0Mx | II |
| CC1621N | 49 | M | N | RIGHT | N | NA |
| CC1680 | 75 | F | AA | RIGHT | AA | NA |
| CC1681 | 75 | M | AA | LEFT | AA | NA |
| CC1682 | 71 | M | AA | RIGHT | AA | NA |
| CC1683 | 45 | M | A | RIGHT | AA | NA |
| CC1698 | 54 | M | A | RIGHT | A | NA |
| CC1720 | 70 | F | A | RIGHT | A | NA |
| CC1721 | 54 | M | A | Missing | A | NA |
N = Normal, T = CRC, A = Adenoma, AA = Advanced Adenoma
Tumor suppressor genes
APC variants
We detected 944 variants in the discovery set, of which 822 are novel. The detailed information of these variants was recently published [5]. The Illumina sequencing led to the validation of 33 variants of which 4 were novel. Variant at locus chr5:112176918 with a G to A change had a frequency 0.03 (1/33, heterozygous) in advanced adenoma (Amino acid change from R to T). Variant at locus chr5: 112174763 with an A to T change had a frequency of 0.02 (1/56, heterozygous) in CRCs (Amino acid change from R to X). Variant at locus chr5:112154980 with a T to A change had a frequency 0.05 (1/21, heterozygous) in adenoma (Amino acid change from C to X). Variant at locus 112157658 with a G to T change had a frequency of 0.05 (1/21, heterozygous) in adenoma (Amino acid change from E to X). One variant was mapped in the 5’ UTR, 9 prior to ARM, 4 on the ARM, 3 prior to B-Catenin binding region, 12 in the B-Catenin binding region, 3 in the basic region, and 1 in the EB1 binding domain [5] (Table 2 and Figure 3A).
Table 2. Number of samples per validated variants in the targeted gene panel.
| Locus | Ref | Var | Gene | Variant type | Status (1=novel, 0=known) | AA-Normal Het (30) | AA-Normal Hom (30) | AA-Adenoma Het (21) | AA-Adenoma Hom (21) | AA-Ad. Adenoma Het (33) | AA-Ad. Adenoma Hom (33) | AA-CRC Het (56) | AA-CRC Hom (56) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 63409685 | C | T | AMER1 | intronic | 1 | 2 | 0 | 1 | 0 | 1 | 0 | 1 | 0 |
| 63410110 | T | C | AMER1 | synonymous SNV | 0 | 8 | 16 | 10 | 9 | 9 | 13 | 8 | 33 |
| 63412291 | C | G | AMER1 | non-synonymous SNV | 0 | 2 | 1 | 3 | 0 | 0 | 0 | 4 | 1 |
| 63412690 | A | C | AMER1 | non-synonymous SNV | 0 | 13 | 6 | 9 | 2 | 13 | 2 | 27 | 10 |
| 112043384 | T | G | APC | intronic | 0 | 6 | 0 | 3 | 0 | 7 | 1 | 12 | 0 |
| 112103015 | C | A | APC | stopgain | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 |
| 112116592 | C | T | APC | stopgain | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2 | 0 |
| 112128191 | C | T | APC | stopgain | 0 | 0 | 0 | 1 | 0 | 2 | 0 | 2 | 0 |
| 112136947 | A | T | APC | intronic | 0 | 3 | 0 | 2 | 0 | 3 | 0 | 3 | 0 |
| 112151261 | C | T | APC | stopgain | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 1 | 0 |
| 112154942 | C | T | APC | stopgain | 0 | 0 | 0 | 1 | 0 | 1 | 0 | 1 | 0 |
| 112154980 | T | A | APC | stopgain | 1 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 |
| 112157658 | G | T | APC | stopgain | 1 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 |
| 112162854 | T | C | APC | synonymous SNV | 0 | 11 | 1 | 7 | 2 | 14 | 2 | 17 | 6 |
| 112164561 | G | A | APC | synonymous SNV | 0 | 16 | 5 | 14 | 6 | 20 | 7 | 27 | 16 |
| 112173553 | T | G | APC | synonymous SNV | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 |
| 112173899 | C | T | APC | non-synonymous SNV | 0 | 1 | 0 | 1 | 0 | 2 | 0 | 3 | 0 |
| 112174096 | C | A | APC | stopgain | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 |
| 112174763 | A | T | APC | stopgain | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 |
| 112175023 | A | G | APC | synonymous SNV | 0 | 3 | 0 | 2 | 0 | 4 | 0 | 3 | 0 |
| 112175030 | G | A | APC | non-synonymous SNV | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 2 | 0 |
| 112175069 | C | T | APC | stopgain | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 2 | 0 |
| 112175207 | G | T | APC | stopgain | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2 | 0 |
| 112175399 | A | T | APC | stopgain | 0 | 0 | 0 | 0 | 0 | 2 | 0 | 1 | 0 |
| 112175576 | C | T | APC | stopgain | 0 | 0 | 0 | 0 | 0 | 3 | 0 | 0 | 0 |
| 112175639 | C | T | APC | stopgain | 0 | 0 | 0 | 2 | 0 | 4 | 0 | 2 | 0 |
| 112175770 | G | A | APC | synonymous SNV | 0 | 17 | 5 | 13 | 7 | 20 | 8 | 26 | 14 |
| 112176325 | G | A | APC | synonymous SNV | 0 | 19 | 4 | 14 | 6 | 21 | 6 | 26 | 17 |
| 112176541 | C | G | APC | synonymous SNV | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 2 | 0 |
| 112176559 | T | G | APC | synonymous SNV | 0 | 18 | 4 | 13 | 7 | 19 | 8 | 26 | 17 |
| 112176756 | T | A | APC | non-synonymous SNV | 0 | 3 | 26 | 4 | 17 | 8 | 24 | 4 | 50 |
| 112176918 | G | C | APC | non-synonymous SNV | 1 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 |
| 112177171 | G | A | APC | synonymous SNV | 0 | 16 | 5 | 14 | 6 | 21 | 7 | 31 | 15 |
| 112178492 | C | T | APC | synonymous SNV | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 1 | 0 |
| 112178795 | G | A | APC | non-synonymous SNV | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 2 | 0 |
| 112178995 | A | G | APC | synonymous SNV | 0 | 7 | 0 | 3 | 0 | 6 | 0 | 6 | 2 |
| 112179909 | C | A | APC | intronic | 0 | 9 | 1 | 9 | 1 | 12 | 0 | 20 | 3 |
| 27057621 | A | C | ARID1A | intronic | 0 | 3 | 0 | 0 | 0 | 8 | 0 | 0 | 0 |
| 27089446 | G | C | ARID1A | intronic | 0 | 2 | 0 | 0 | 0 | 5 | 1 | 1 | 0 |
| 27089585 | C | A | ARID1A | synonymous SNV | 0 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 0 |
| 27101278 | C | T | ARID1A | synonymous SNV | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 1 | 0 |
| 27102075 | G | A | ARID1A | synonymous SNV | 0 | 2 | 0 | 2 | 0 | 0 | 0 | 2 | 0 |
| 27102188 | A | G | ARID1A | non-synonymous SNV | 0 | 1 | 0 | 4 | 0 | 7 | 0 | 7 | 0 |
| 27105676 | G | T | ARID1A | stopgain | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 |
| 27106323 | C | T | ARID1A | synonymous SNV | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 4 | 0 |
| 140434597 | G | A | BRAF | intronic | 0 | 7 | 0 | 4 | 0 | 6 | 0 | 5 | 0 |
| 140449071 | C | G | BRAF | intronic | 0 | 7 | 15 | 9 | 7 | 20 | 8 | 28 | 18 |
| 140449150 | T | C | BRAF | synonymous SNV | 0 | 10 | 13 | 11 | 5 | 20 | 8 | 30 | 18 |
| 140481511 | A | C | BRAF | intronic | 0 | 2 | 0 | 2 | 0 | 0 | 0 | 3 | 0 |
| 140487360 | G | A | BRAF | non-synonymous SNV | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 |
| 79950497 | C | T | DHFR,MSH3 | intronic | 0 | 6 | 4 | 6 | 2 | 3 | 2 | 14 | 5 |
| 79950508 | C | T | DHFR,MSH3 | intronic | 0 | 4 | 0 | 1 | 0 | 3 | 0 | 5 | 0 |
| 79950512 | A | G | DHFR,MSH3 | intronic | 0 | 5 | 14 | 1 | 8 | 3 | 8 | 13 | 26 |
| 153245446 | G | A | FBXW7 | non-synonymous SNV | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 |
| 153247138 | G | T | FBXW7 | intronic | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 1 | 0 |
| 153247289 | G | A | FBXW7 | non-synonymous SNV | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 2 | 0 |
| 153247366 | C | T | FBXW7 | non-synonymous SNV | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 |
| 153303509 | C | T | FBXW7 | intronic | 0 | 1 | 0 | 0 | 0 | 1 | 0 | 1 | 0 |
| 25362854 | C | T | KRAS | intronic | 0 | 11 | 3 | 12 | 0 | 10 | 0 | 24 | 5 |
| 25368462 | C | T | KRAS | synonymous SNV | 0 | 0 | 29 | 0 | 21 | 1 | 32 | 1 | 53 |
| 25398255 | G | T | KRAS | non-synonymous SNV | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 |
| 25398281 | C | T | KRAS | non-synonymous SNV | 0 | 0 | 0 | 3 | 0 | 5 | 0 | 9 | 0 |
| 25398284 | C | T | KRAS | non-synonymous SNV | 0 | 0 | 0 | 3 | 0 | 2 | 0 | 12 | 0 |
| 25398285 | C | A | KRAS | non-synonymous SNV | 0 | 0 | 0 | 0 | 0 | 2 | 0 | 1 | 0 |
| 25398285 | C | T | KRAS | non-synonymous SNV | 0 | 0 | 0 | 0 | 0 | 3 | 0 | 2 | 0 |
| 79960955 | G | A | MSH3 | intronic | 0 | 11 | 5 | 10 | 4 | 16 | 1 | 24 | 7 |
| 79966029 | G | A | MSH3 | synonymous SNV | 0 | 6 | 1 | 11 | 0 | 5 | 2 | 16 | 0 |
| 79966197 | G | A | MSH3 | intronic | 0 | 11 | 5 | 11 | 4 | 19 | 1 | 26 | 5 |
| 79968496 | C | T | MSH3 | intronic | 0 | 5 | 3 | 3 | 3 | 11 | 1 | 13 | 3 |
| 80024685 | C | A | MSH3 | non-synonymous SNV | 0 | 0 | 0 | 2 | 0 | 0 | 0 | 0 | 0 |
| 80024738 | A | G | MSH3 | non-synonymous SNV | 0 | 0 | 0 | 0 | 0 | 2 | 0 | 0 | 0 |
| 80024783 | G | A | MSH3 | non-synonymous SNV | 0 | 1 | 0 | 0 | 0 | 5 | 0 | 2 | 0 |
| 80083459 | G | A | MSH3 | synonymous SNV | 0 | 1 | 0 | 0 | 0 | 2 | 0 | 1 | 0 |
| 80149981 | A | G | MSH3 | non-synonymous SNV | 0 | 2 | 27 | 4 | 16 | 7 | 23 | 5 | 48 |
| 80160610 | T | A | MSH3 | intronic | 0 | 5 | 2 | 4 | 0 | 4 | 1 | 12 | 1 |
| 80168937 | G | A | MSH3 | non-synonymous SNV | 0 | 11 | 15 | 10 | 8 | 19 | 10 | 22 | 30 |
| 48022981 | G | T | MSH6 | intronic | 0 | 10 | 3 | 12 | 1 | 11 | 1 | 23 | 4 |
| 48023115 | T | C | MSH6 | synonymous SNV | 0 | 9 | 1 | 5 | 1 | 7 | 1 | 14 | 1 |
| 48026286 | C | T | MSH6 | synonymous SNV | 0 | 4 | 0 | 2 | 0 | 4 | 0 | 7 | 0 |
| 48027375 | T | C | MSH6 | synonymous SNV | 0 | 4 | 0 | 2 | 0 | 7 | 0 | 5 | 1 |
| 48030692 | T | A | MSH6 | synonymous SNV | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 |
| 48030838 | A | T | MSH6 | intronic | 0 | 10 | 2 | 12 | 0 | 8 | 1 | 17 | 2 |
| 48032908 | A | G | MSH6 | intronic | 1 | 1 | 0 | 1 | 0 | 0 | 0 | 2 | 0 |
| 48032937 | T | C | MSH6 | intronic | 0 | 4 | 25 | 4 | 17 | 9 | 21 | 12 | 40 |
| 48033514 | T | C | MSH6 | intronic | 0 | 0 | 0 | 2 | 0 | 2 | 0 | 1 | 0 |
| 48033545 | A | C | MSH6 | intronic | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 |
| 48033551 | C | G | MSH6 | intronic | 0 | 4 | 20 | 3 | 11 | 8 | 9 | 13 | 34 |
| 48033700 | G | A | MSH6 | non-synonymous SNV | 0 | 1 | 0 | 2 | 0 | 0 | 0 | 2 | 0 |
| 178917005 | A | G | PIK3CA | intronic | 0 | 8 | 7 | 11 | 2 | 16 | 3 | 28 | 7 |
| 178921639 | C | A | PIK3CA | intronic | 0 | 12 | 16 | 11 | 9 | 15 | 14 | 24 | 25 |
| 178922274 | C | A | PIK3CA | intronic | 0 | 6 | 17 | 11 | 9 | 8 | 14 | 14 | 26 |
| 178927345 | T | C | PIK3CA | intronic | 0 | 5 | 1 | 5 | 0 | 5 | 0 | 9 | 1 |
| 178927410 | A | G | PIK3CA | non-synonymous SNV | 0 | 4 | 2 | 7 | 0 | 12 | 2 | 23 | 1 |
| 178947985 | C | G | PIK3CA | intronic | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 |
| 178948196 | A | G | PIK3CA | intronic | 0 | 6 | 0 | 4 | 1 | 4 | 0 | 6 | 0 |
| 48575700 | T | C | SMAD4 | intronic | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 |
| 48584420 | G | A | SMAD4 | intronic | 0 | 3 | 0 | 1 | 0 | 1 | 0 | 3 | 0 |
| 48584624 | T | C | SMAD4 | intronic | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 |
| 48591762 | G | A | SMAD4 | intronic | 0 | 1 | 0 | 9 | 1 | 7 | 0 | 9 | 0 |
| 48592020 | T | C | SMAD4 | intronic | 0 | 2 | 0 | 1 | 0 | 1 | 0 | 3 | 0 |
| 48602941 | G | C | SMAD4 | intronic | 0 | 1 | 0 | 4 | 0 | 0 | 0 | 1 | 0 |
| 70117450 | G | A | SOX9 | intronic | 0 | 3 | 0 | 1 | 0 | 0 | 1 | 3 | 1 |
| 70118935 | C | T | SOX9 | synonymous SNV | 0 | 10 | 0 | 3 | 0 | 3 | 0 | 16 | 1 |
| 70120551 | A | C | SOX9 | intronic | 0 | 13 | 1 | 8 | 0 | 11 | 3 | 19 | 0 |
| 114912081 | G | A | TCF7L2 | intronic | 0 | 1 | 0 | 2 | 0 | 2 | 0 | 4 | 0 |
| 114920452 | T | C | TCF7L2 | intronic | 1 | 0 | 0 | 0 | 0 | 2 | 0 | 0 | 0 |
| 30686414 | A | G | TGFBR2 | intronic | 0 | 11 | 4 | 10 | 3 | 16 | 4 | 23 | 5 |
| 30713126 | T | A | TGFBR2 | intronic | 0 | 6 | 0 | 10 | 0 | 12 | 0 | 13 | 1 |
| 30713619 | C | T | TGFBR2 | non-synonymous SNV | 0 | 0 | 0 | 0 | 0 | 2 | 0 | 0 | 0 |
| 30713674 | A | G | TGFBR2 | synonymous SNV | 0 | 6 | 0 | 3 | 0 | 3 | 0 | 10 | 0 |
| 30713842 | C | T | TGFBR2 | synonymous SNV | 0 | 2 | 0 | 1 | 0 | 2 | 0 | 2 | 0 |
| 30729931 | C | T | TGFBR2 | synonymous SNV | 0 | 2 | 0 | 0 | 0 | 0 | 0 | 2 | 0 |
| 7574003 | G | A | TP53 | stopgain | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 1 | 0 |
| 7576501 | G | A | TP53 | intronic | 0 | 1 | 0 | 0 | 0 | 2 | 0 | 5 | 0 |
| 7577120 | C | T | TP53 | non-synonymous SNV | 0 | 0 | 0 | 0 | 0 | 2 | 0 | 2 | 0 |
| 7578210 | T | C | TP53 | synonymous SNV | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 1 | 0 |
| 7578212 | G | A | TP53 | stopgain | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 4 | 0 |
| 7578406 | C | T | TP53 | non-synonymous SNV | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 3 | 0 |
| 7579311 | C | A | TP53 | intronic | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 |
| 7579472 | G | C | TP53 | non-synonymous SNV | 0 | 21 | 0 | 14 | 0 | 22 | 0 | 34 | 0 |
| 7579801 | G | C | TP53 | intronic | 0 | 23 | 0 | 12 | 1 | 20 | 0 | 36 | 0 |
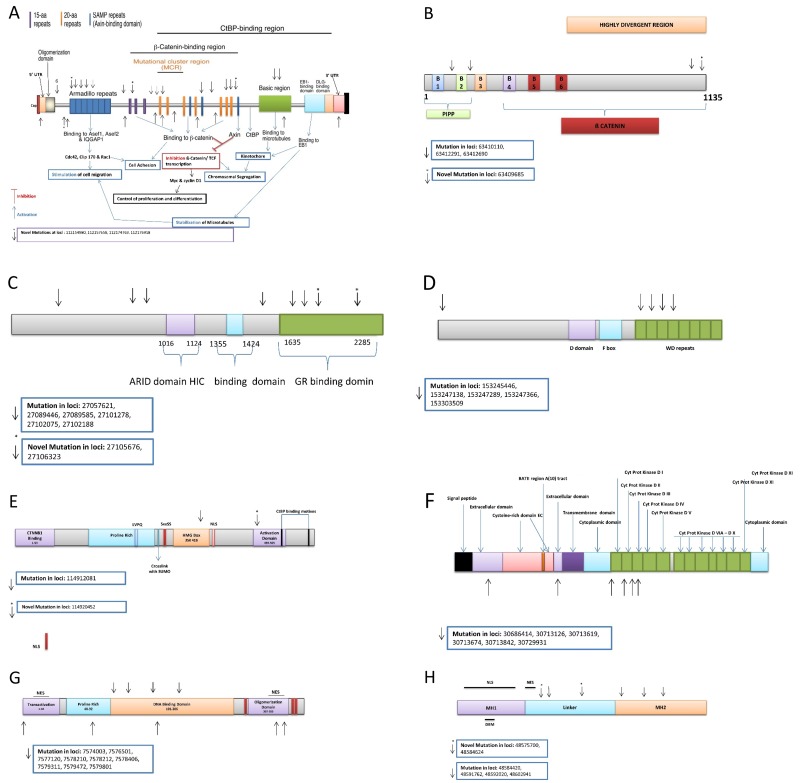
Figure 3.
Tumor suppressor genes validated variants: (A) APC, (B) AMER1, (C) ARID1, (D) FBXW7, (E) TCF7L2, (F) TGFBR2, (G) TP53, (H) SMAD4.
AMER1 variants
We found 461 variants in the discovery set of which 433 were novel. From these, 402 were non-synonymous, 25 were stopgains, and 6 were frameshift substitutions. All of these variants were in exon 2 of the AMER1 gene. Four variants in the AMER1 gene were validated on Illumina HiSeq of which 1 was novel. This variant at locus chrX: 63409685, a C to T change, had a frequency of 0.07 (2/30, both heterozygous) in normal tissue, 0.05 (1/21, heterozygous) in adenoma, 0.03 (1/33, heterozygous) in advanced adenoma and 0.02 (1/56, heterozygous) in CRC. (Table 2 and Figure 3B).
ARID1A variants
We found 742 variants in the discovery set, of which 695 were novel. From these, 653 were non-synonymous, 35 were stopgains, 6 were frameshift and 1 was a non-frameshift variant. Eight variants in the ARID1A gene were validated on the HiSeq platform of which 2 were novel (Table 2). Variant at locus chr2: 27106323, was synonymous, a C to T change, with a frequency of 0.03 (1/30, heterozygous) in normal and 0.07 (4/56, all heterozygous) in CRCs. Variant at locus chr2: 27105676 was a stopgain, a G to T change, with a frequency of 0.02 (1/56, heterozygous) in CRCs (Amino acid change from E to X). These variants were on exon 19 and 20 of the ARID1A, respectively. Three variants were mapped to N-terminus sequences before the ARID domain, 1 prior to the GR [the C-terminus of the protein can stimulate Glucocorticoid Receptor-dependent transcriptional activation] binding domain, and 4 in the GR binding domain (Table 2 and Figure 3C).
FBXW7 variants
We found 262 variants in the discovery set, of which 230 were novel. From these, there were 210 non-synonymous and 20 stopgains. We validated five known variants. From these five, 2 were flanking intronic, and 3 were non-synonymous. Variant at locus Chr4: 153247289, exon 9, codon change of CGC to CAC had a frequency of 0.03 (1/33, heterozygous) in advanced adenoma and 0.04 (2/56, heterozygous) in CRCs, this variant was non-synonymous (Table 2). Non-synonymous variant at locus Chr4: 153247366, exon 9, a C to T change, had a frequency of 0.03 (1/33, heterozygous) in advanced adenoma. Variant at locus Chr4: 153245446, exon 10, codon change CAG to CAA, had a frequency of 0.03 (1/33, heterozygous) in advanced adenoma (Amino acid change from S to L), this variant was non-synonymous (Table 2). Four variants were mapped to the WD repeat domain while 1 was prior to the D domain (Figure 3D).
TCF7L2 variants
We found 194 variants in the discovery set, of which 186 were novel. From these, 178 were non-synonymous, 5 were stopgains, 2 were frameshift substitutions, and 1 was stoploss. We validated two of these variants: 1 was novel flanking intronic at locus Chr10: 114920452, a T to C change, with a frequency of 0.06 (2/33, both heterozygous) in advanced adenoma. The other variant at locus Chr10: 114912081 was known, a G to A change, had a frequency of 0.03 (1/30, heterozygous) in normal tissue, 0.10 (2/21, both heterozygous) in adenoma, 0.06 (2/33, both heterozygous) in advanced adenoma, and 0.07 (4/56, all heterozygous) in CRCs (Table 2). One variant was mapped to the HMG box while 1 was to the activation domain (Figure 3E).
TGFβR2 variants
We found 169 variants in the discovery set, of which 157 were novel. From these, 142 were non-synonymous and 15 were stopgains. We validated 6 known variants (Table 2). Three were synonymous at locus Chr3: 30713674 exon 4 with codon change of CTA to CTG with a frequency of 0.20 (6/30, all heterozygous) in normal, 0.14 (3/21, all heterozygous) in adenoma (No Amino acid change), 0.09 (3/33, all heterozygous) in advanced adenoma, and 0.18 (10/56, all heterozygous) in CRC; variant at locus Chr 3: 30713842 exon 4 with codon change C to T with a frequency of 0.07 (2/30, both heterozygous) in normal, 0.05 (1/21, heterozygous) in adenoma, 0.06 (2/33, both heterozygous) in advanced adenoma, and 0.04 (2/56, both heterozygous) in CRC; variant at locus Chr: 30729931 exon 6 with codon change of GTC to GTT with a frequency of 0.07 (2/30, both heterozygous) in normal, and 0.04 (2/56, both heterozygous) in CRC; and 1 was non-synonymous at locus chr: 30713619 exon 4 with codon change of ACG to ATG with a frequency of 0.10 (2/33, both heterozygous) in advanced adenoma (Table 2) (Amino acid change from T to M). The other 2 were flanking intronic. Four variants were mapped to the Cyt ProtKinase I, II and III domains, and 2 on the extracellular domain (Figure 3F).
TP53 variants
We found 162 variants in the discovery set, of which 51 were novel. From these, 49 were non-synonymous, 1 was stopgain, and 1 was frameshift variant. We validated 9 variants that were all known. There were 3 non-synonymous variants at locus Chr17: 7579472 exon 3 with nucleotide change G to C and a frequency of 0.70 (21/30, all heterozygous) in normal, 0.67 (14/21, all heterozygous) in adenoma, 0.67 (22/33, all heterozygous) in advanced adenoma, and 0.61 (34/56, all heterozygous) in CRC (Amino acid change from P to R); locus Chr17: 7578406 exon 1 with nucleotide change C to T with a frequency of 0.03 (1/30, heterozygous) in normal, and 0.05 (3/56, all heterozygous) in CRC (Amino acid change from R to H); and locus Chr17: 7577120 exon 4 with nucleotide change C to T with a frequency of 0.04 (2/56, both heterozygous) in CRC (Amino acid change from R to H). There was one synonymous variant at locus Chr17: 7578210 exon 2 with codon change of CGA to CGG with a frequency 0.05 (1/21, all heterozygous) in adenoma, and 0.02 (1/56, all heterozygous) in CRC. There were two stopgain variants at locus Chr17: 7578212 exon 2 with codon change GCT to ACT with a frequency of 0.07 (4/56, all heterozygous) in CRC (Amino acid change from R to X); and locus Chr17: 7574003 exon 6 with nucleotide change G to A with a frequency 0.03 (1/33, all heterozygous) in advanced adenoma, and 0.02 (1/56, all heterozygous) in CRC (Amino acid change from R to X). The remaining 3 variants were intronic (Table 2). We mapped 1 variant to the transactivation domain, 1 in the proline rich domain, 5 in the DNA binding domain, and 2 in the oligomerization domain (Figure 3G).
SMAD4 variants
We found 196 variants in the discovery set, of which 159 were novel. From these, 153 were non-synonymous, 4 were stopgains, and 2 were frameshift variants. We validated six intronic variants of which 2 were novel (Table 2). Variant at locus chr8: 48584624 with a T to C change had a frequency 0.02 (1/56, heterozygous) in CRCs. Variant at locus chr8: 48575700 with a T to C change had a frequency 0.02 (1/56, heterozygous) in CRCs. Of the 6 validated variants, three were found in the linker region and 3 in the MH2 region (Figure 3H).
Oncogenes
KRAS variants
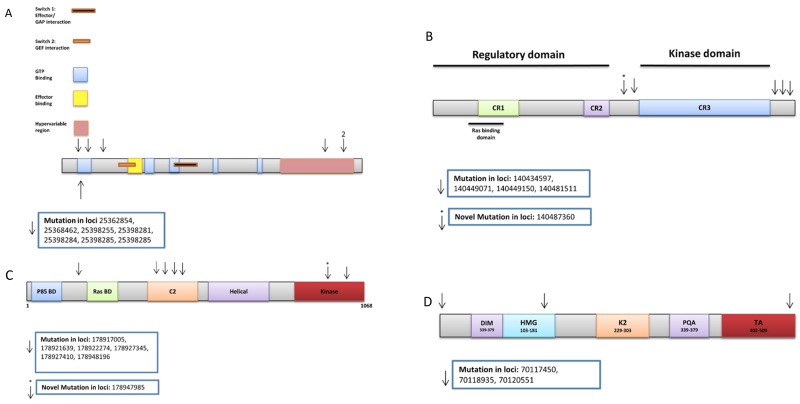
We found 54 variants in the discovery set, of which 44 were novel. From these, 41 were non-synonymous and 3 were stopgains. All variants were in the RAS domain. We validated seven known variants, 5 of which were non-synonymous, 1 synonymous, and 1 flanking intronic. The non-synonymous variants are as follows: locus 25398281 exon 2, a C to T change, with a frequency of 0.14 (3/21, all heterozygous) in adenoma, 0.15 (5/33, all heterozygous) in advanced adenoma, and 0.16 (9/56, all heterozygous) in CRC (Amino acid change from G to D); locus 25398284 exon 2, a C to T change, with a frequency of 0.14 (3/21, all heterozygous) in adenoma, 0.06 (2/33, both heterozygous) in advanced adenoma, and 0.21 (12/56, all heterozygous) in CRC (Amino acid change from G to D); locus 25398285 exon 2, a C to A change, with a frequency of 0.06 (2/33, both heterozygous) in advanced adenoma, and 0.02 (1/56, heterozygous) in CRC (Amino acid change from G to C); locus 25398285 exon 2, a C to T change, with a frequency of 0.09 (3/33, all heterozygous) in advanced adenoma, and 0.04 (2/56, both heterozygous) in CRC (Amino acid change from G to S); locus 25398255 exon 2 with nucleotide change G to T, and a frequency of 0.03 (1/33, heterozygous) in advanced adenoma (Table 2). Three variants were mapped in the GTP binding region, 1 C-terminal to the GTP binding region, (Amino acid change from Q to K) and 3 in the hypervariable region (Figure 4A).
Figure 4.
Oncogenes’ validated variants: (A) KRAS, (B) BRAF, (C) PIK3CA, (D) SOX9.
NRAS variants
We found 57 variants in the discovery set, of which 50 were novel. From these, 44 were non-synonymous, 2 frameshift substitutions, and 4 stopgains. No NRAS variants were validated on the Illumina platform.
BRAF variants
We found 221 variants in the discovery set, of which 200 were novel. From these, 184 were non-synonymous and 16 were stopgain variants. We validated 5 variants of which 1 was novel. One variant was non-synonymous, 1 synonymous, and 3 flanking intronic. Non-synonymous novel variant Chr7: 140487360 exon 9 has a frequency of 0.02 (1/56, heterozygous) in CRC. Synonymous variant Chr7: 140449150 exon 16 has a frequency of 0.77 (23/30, 13 homozygous and 10 heterozygous) in normal, 0.76 (16/21, 5 homozygous and 11 heterozygous) in adenoma, 0.85 (28/33, 8 homozygous and 20 heterozygous) in advanced adenoma, and 0.86 (48/56, 18 homozygous and 30 heterozygous) in CRC (Table 2). The variants were mapped prior and after CR3 (Figure 4B).
PIK3CA variants
We found 251 variants in the discovery set, of which 214 were novel. From these, 191 are non-synonymous and 23 were stopgain variants. We validated seven variants of which 1 was novel (Table 2). One variant was non-synonymous while the other 6 were flanking intronic. Non-synonymous variant at locus Chr3: 178927410 exon 7, an A to G change, had a frequency of 0.27 (6/30, 2 homozygous and 4 heterozygous) in normal, (7/21, all heterozygous), 0.33 in adenoma, 0.42 (14/33, 2 homozygous and 12 heterozygous) in advanced adenoma and 0.41 (23/56, 1 homozygous and 22 heterozygous) in CRC. One variant was mapped prior to Ras BD region, 4 in the C2 domain, and 2 in the kinase domain (Table 2 and Figure 4C).
SOX9 variants
We found 189 variants in the discovery set, of which 183 were novel. From these, 162 were non-synonymous, 13 were stopgains, and 8 were frameshift variants. We validated three known variants; 1 synonymous and 2 flanking intronic (Table 2). Synonymous variant at locus Chr17: 70118935 had a frequency of 0.33 (10/30, all heterozygous) in normal, 0.14 (3/21, all heterozygous) in adenoma, 0.09 (3/33, all heterozygous) in advanced adenoma and 0.32 (17/56, 1 homozygous and 16 heterozygous) in CRCs (Table 2). One variant was mapped prior to the DIM region, 1 in the HMG region, and 1 in the TA region (Figure 4D).
MMR genes
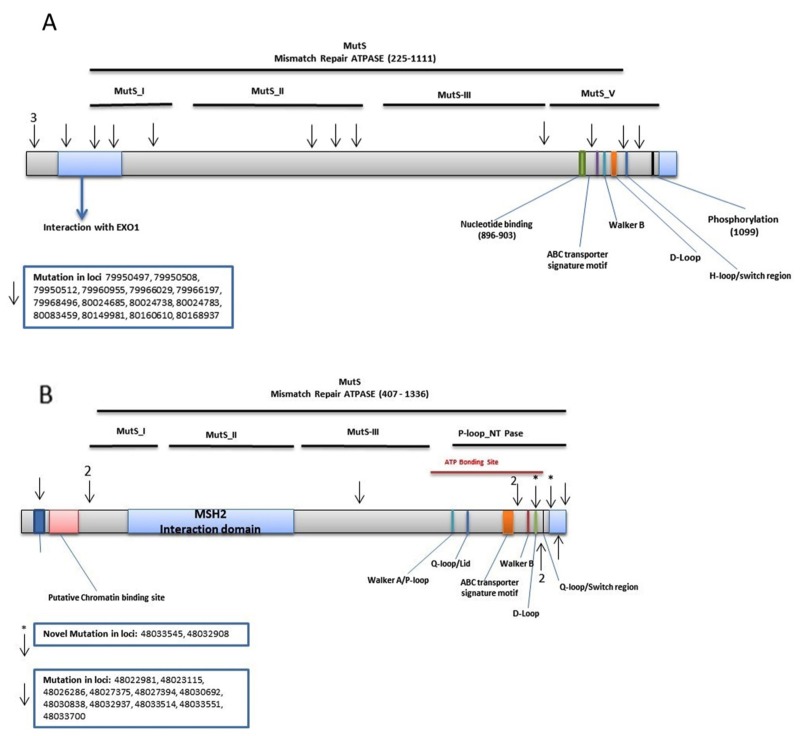
MSH3: We found 315 MSH3 variants in the discovery set, of which 298 were novel. We validated 14 known variants. Five of these variants were non-synonymous and altered exons 10, 21, and 23. Two were synonymous with changes in exon 4 and 18. Variant at locus 79966029 with a G to A change, with a frequency of 0.23 (7/30, 1 homozygous and 6 heterozygous) in normal, 0.52 (11/21, all heterozygous) in adenoma, 0.21 (7/33, 2 homozygous and 5 heterozygous) in advanced adenoma, and 0.29 (16/56, all heterozygous) in CRCs. Variant at locus 80083459 with a G to A change, with a frequency of 0.03 (1/30, heterozygous) in normal, 0.06 (2/33, both heterozygous) in advanced adenoma, and 0.02 (1/56, heterozygous) in CRCs. The rest were flanking intronic (Table 2). The variants were mapped to the MSH3-MSH2-MSH6 region with 3 variants prior to EXO1, 3 in EXO1, 1 in MutS_I, 3 in MutS_II, 1 in MutS_III, and 3 in MutS_V [5] (Figure 2A).
Figure 2. Distribution of validated variants per targeted MMR genes.
(A) MSH3 (B) MSH6.
MSH6: We detected 434 variants in the discovery set, of which 396 were novel. The Illumina sequencing led to the validation of 12 variants. Two of these were novel. The A>G and A>C variants are flanking the region coding for the MutS-V domain and the MSH2 binding site, respectively. An A>C intronic variant (IVS8-45) at locus chr2:48033545 was observed in 1 CRC sample with a frequency of 0.02 (1/56, heterozygous). The A>G variant at locus chr2:48032908 was intronic (IVS7-50) and was observed in 1 sample with a frequency of 0.03 (1/30, heterozygous) in normal, 0.05 (1/21, heterozygous) in adenoma, and 0.04 (2/56, both heterozygous) in CRC (Table 2). One variant was mapped in PWWP, 2 prior to the MutS_I, 1 in MutS_III, and 8 in P-loop_NTPase [5] (Figure 2B).
Summary of validated variants
In total, there were 12 validated variants in the 14 genes: APC (33/121 [27%]), AMER1 (4/121) [3%]), ARID1 (8/121 [7%]), MSH3 (14/121 [12%]), MSH6 (12/121 [10%]), BRAF (5/121 [4%]), KRAS (7/121 [6%]), FBXW7 (5/121 [4%]), PIK3CA (7/121 [6%]), SMAD4 (6/121 [5%]), SOX9 (3/121 [2%]), TCF7L2 (2/121 [2%]), TGFBR2 (6/121 [5%]), TP53 (9/121 [7%]). From these validated variants, 14/121 (12%) were novel variants in 8 genes (AMER1, APC, ARID1A, BRAF, MSH6, PIK3CA, SMAD4, and TCF7L2). Of the validated variants, 23% (28/121) were non-synonymous, 14% (17/121) were stopgains, 24% (29/121) were synonymous and 39% (47/121) were intronic variants.
DISCUSSION
We performed a targeted exome sequencing in African Americans with colorectal lesions with the goal of detecting particular variant profiles that can partially explain the observed CRC disparity in this population through the identification of highly pathogenic and frequent variants and mutations. While most genes displayed a high number of variants (novel and known) on one platform (Ion Torrent), not all were validated on the second sequencing platform (HiSeq, Illumina). We previously reported such discrepancies on our recent study [33] thus mandating a necessary validation step that we performed on the HiSeq platform. We validated 121 variants in 14 mechanistically known driver genes including Tumor Suppressor Genes (TSGs): APC (33/121 [27%]), AMER1 (4/121) [3%]), ARID1 (8/121 [7%]), FBXW7 (5/121 [4%]), SMAD4 (6/121 [5%]), TCF7L2 (2/121 [2%]), TGFBR2 (6/121 [5%]), TP53 (9/121 [7%]), oncogenes: PIK3CA (7/121 [6%]), BRAF (5/121 [4%]), KRAS (7/121 [6%]), SOX9 (3/121 [2%]), and MMR genes, MSH3 (14/121 [12%]), MSH6 (12/121 [10%]). From these validated variants, 14/121 (12%) were novel variants in 8 genes (AMER1, APC, ARID1A, BRAF, MSH6, PIK3CA, SMAD4, and TCF7L2). Of the validated variants, 23% (28/121) were non-synonymous, 14% (17/121) were stopgains, 24% (29/121) were synonymous and 39% (47/121) were intronic variants. A sizable portion of the validated variants (39%) were intronic. While it is difficult to assess the potential effects of such variants on the protein product and function, many studies have shown that such variants lead to aberrant splicing with different levels of impact on the protein function in many pathological diseases [34].
Here, we report the profile of variants in African American colorectal lesions using targeted exome sequencing. Validated variants showed that the tumor suppressor genes APC and ARID1 and the DNA MMR genes MSH3 and MSH6 are the genes with the highest numbers of validated variants. Oncogenes KRAS and PIK3CA were also primary variant targets that likely participated in increased proliferative potential of the mutated colonic epithelial cells in this population. Many of these recurrent and frequent variants were novel and not previously reported in any of the known databases.
The 8 TSGs (AMER1, APC, ARID, FBXW7, TCF7L2, TGFBR2, SMAD4, and TP53) in our gene panel accounted for 59% of the validated variants. If added to the 20% of validated DNA MMR genes’ variants, this will give a 73% rate of validated variants within TSGs. This finding is in line with the importance of TSGs in the initiation of the carcinogenic process and with the fact that such genes need double-hits for complete inactivation, unlike oncogenes. More than 2/3 TSGs validated variants in our study reflects perfectly the roles and rates at which such genes are targeted in the neoplastic transformation process.
APC is part of WNT pathway in CRC and was the top target gene with 27% (33/121) of the validated variants (4 novel variants, Table 3). Its role for downstream signaling with B-catenin, GSK and AXIN has been well established in previous studies [35]. Wnt/β-catenin pathway plays multiple and diverse roles in development by regulating gene expression via T-cell factor/Lymphoid enhancer-binding factor (Tcf/Lef) DNA binding factors [36]. Angus-Hill et al. showed that Tcf4 (Tcf7L2) functions as a tumor suppressor gene in colon carcinogenesis [36].
Table 3. Distribution of validated variants in signaling pathways.
| Gene | Signaling Pathway | TSG vs. Oncogene | Function | Total # of Variants (n=121) | Total Variant FRQ | Novel Variant n, % |
|---|---|---|---|---|---|---|
| AMER1 | Wnt/β-catenin | TSG | Proliferation, subcellular distribution of APC | 4 | 3% | 1, 1 |
| APC | Wnt/β-catenin | TSG | Proliferation | 33 | 27% | 4, 3 |
| ARID1A | Wnt/β-catenin | TSG | Proliferation | 8 | 7% | 2, 2 |
| BRAF | RTK-RAS | Oncogene | Cell survival, translation, proliferation | 5 | 4% | 1, 1 |
| FBXW7 | Wnt/β-catenin | TSG | Proliferation | 5 | 4% | No |
| KRAS | RTK-RAS | Oncogene | Cell survival, translation, proliferation | 7 | 6% | No |
| MSH3 | Mismatch repair | Suppression of Tumor | Mismatch repair | 14 | 12% | No |
| MSH6 | Mismatch repair | Suppression of Tumor | Mismatch repair | 13 | 11% | 2, 2 |
| MMR (MSH3*, MSH6, MSH2, PMS2, MLH1 | Mismatch repair | Suppression of Tumor | Mismatch repair | 27 | 23% | 2, 2 |
| PIK3CA | PI3K-Akt | Oncogene | Cell survival, translation, proliferation | 7 | 6% | 1, 1 |
| SMAD4 | Wnt/β-catenin | Oncogene | Proliferation | 6 | 5% | 2, 2 |
| SOX9 | Wnt/β-catenin | Oncogene | Proliferation, Self-Renewal of Oncogene Targeted Cells |
3 | 2% | No |
| TCF7L2 | Wnt/β-catenin | Both (TSG/ Oncogene) | Proliferation | 2 | 2% | 1, 1 |
| βR2 | TGF-β (growth factor) | TSG | Proliferation | 6 | 5% | No |
| TP53 | P53 | TSG | Proliferation, Cell survival |
9 | 7% | No |
Exome Sequencing has revealed AMER1 as a frequent target in CRC [37]. In our study, we report one novel variant with a frequency of 0.07 in normal, 0.05 in adenoma, 0.03 in advanced adenoma and 0.02 in CRC samples. Overexpression of AMER1 increases the expression of APC and causes subcellular re-localization of APC from the microtubule ends to the plasma membrane of epithelial cells [37]. As such, variants within this gene may downregulate the APC gene and downstream signaling pathways. SMAD4 is a well-established tumor suppressor gene that displayed a high number of novel variants (n=112), however none were validated on the Illumina platform. Only 4 known variants were validated and they were all mapped to the linker region (Figure 3H).
ARID1A has been identified as a novel tumor suppressor gene in ovarian cancer and subsequently in various other tumor types. ARID1A encodes a protein that belongs to the ARID domain containing family, which consists of 15 genes encoding proteins involved in transcriptional regulation, proliferation and chromatin remodeling [38]. There were 7% (8/121) validated variants highlighting a potential bigger role of this TSG in CRC in African Americans (Table 2, Figure 3C).
TGFBR2 signaling is involved in cell-cell communication, cell adhesion and cell migration. The role of this pathway in the glycosylation pattern of cell surface proteins is largely unexplored. Experimental evidence suggested a possible link between mutated MSI target genes and the glycosylation pattern at the cell surface [39]. We detected 5% (6/121) of the variants in TGFBR2 gene (Table 3), 1 non-synonymous variant at locus Chr: 30713619 exon 4 with codon change of ACG to ATG that mapped to the Cyt ProtKinase I, II and III domain (Figure 3F). This variant could have an effect on tumor invasive phenotype.
FBXW7 has been identified as a transcriptional target of TP53 and lower expression levels of FBXW7 in correlation with TP53-variants have been reported [40]. FBXW7 loss leads to induction of p53-phosphorylation at Serine-15, p53 stabilizes in the nucleus to act as a transcriptional activator for tumor suppression, implicating phospho-p53 (Ser15) as a marker of FBXW7-associated carcinogenesis [27]. We found intronic and non-synonymous variants both in advanced adenoma and in CRCs (Table 2). All four variants were mapped to the WD repeat domain of the protein (Figure 3D). The mutations in this gene may contribute to the decreased efficacy of therapy in FBXW7-mutated CRC [27].
TP53 is frequently inactivated by variant or deletion in most human tumors [41]. A tremendous effort has been made to restore TP53 activity in cancer therapies. However, no effective p53-based therapy has been successfully translated into clinical cancer treatment owing to the complexity of p53 signaling [41]. For the TP53 gene, there were 7% (9/121) validated that were mapped (Figure 3G). The potential deleterious effect of these variants needs experimental validation.
The oncogenes within the sequenced gene panel had a total of 22 out of 121 validated variants (PIK3CA (7/121 [6%]), BRAF (5/121 [4%]), KRAS (7/121 [6%]), SOX9 (3/121 [2%])). We previously reported on variants targeting BRAF and KRAS in African Americans with colorectal cancer, however the present data seems to give weight to other oncogenes such as PIK3CA as major targets of variants.
Establishing KRAS variants’ status in each patient is indeed important to determine the appropriate therapy, patients with wild-type KRAS could receive monoclonal antibodies against EGFR [42] while KRAS mutated patients have been associated with no-response to targeted therapies and poor prognosis in different studies [43]. In our study, we found 1 non-synonymous known variant with a 0.03 frequency in patients with advanced adenoma (Table 2). There were no detected variants in the notable hotspots of KRAS (codons 12/13/61/144) [44].
Variants in the BRAF oncogene is a key step in malignant transformation within a subset of CRCs, generally MSI-H and with the CpG methylator phenotype [16]. It is unclear to what extent the lack of response in KRAS wild-type CRCs is due to BRAF variants, but data suggest that mutated BRAF confers resistance to anti-EGFR therapy [45, 46]. According to Palmirotta et al., a KRAS variant (CAG>TAG) determining a premature stop signal at codon 22 (Gln22Stop) has been previously found in a patient with metastatic colorectal cancer [47]. Whether or not the validated stopgain variants in KRAS associate with an activating effect remains to be explored and investigated.
The prevalence of PIK3CA variants increases continuously from rectal to cecum cancers, supporting the ‘colorectal continuum’ paradigm, and an important interplay of gut microbiota and host immune/inflammatory reaction [48]. PIK3CA variants also contribute to significantly decreased survival for patients with wild-type BRAF tumors [49]. Numerous studies have shown that variants are concentrated in 2 hotspots of the PIK3CA gene: the helical domain in codons 542 and 545 of exon 9 and the kinase domain in codon 1047 of exon 20 [49]. In our study, PIK3CA variants were 6% of the validated variants (Table 2). The novel variant was intronic with a CRC frequency of 0.02 (Table 2). This variant was mapped to N-terminus region before the Kinase region of the PIK3CA. The known variant was also intronic with a variants’ frequency of 0.2 in normal, 0.29 in adenoma, 0.12 in advanced adenoma, and 0.11 in CRCs (Table 2). Recently Kim et al, showed that PIK3CB rare point variant is associated with tumorigenesis [50]. This somatic variant event in the cancer, supports our results that low frequency of the variants may have tumorigenic function that needs verification with in vivo and in vitro analysis.
In the intestinal epithelium, SOX9 is expressed in the stem/progenitor cells, as well as in the nuclei of terminally differentiated Paneth cells of the small intestinal crypts and tuft cells in the villi. It plays a crucial role in Paneth cell differentiation [19, 51]. From our panel, 2% (3/124) of validated variants were in this gene. This corresponds to an intronic variant with a frequency of 0.1 in normal, 0.05 in adenoma, 0.06 in advanced adenoma and 0.09 in CRC samples (Table 2). This variant was mapped to the DIM region of SOX9 (Figure 4D).
Among MMR genes, 26 of the validated variants were in MSH3 and MSH6 genes accounting for 22% of the validated variants [5]. This finding highlights the important role that these genes play in the setting of genome-wide instability aside from the already well described roles of MLH1 and MSH2 genes in this process. Indeed, variants within these genes, and specifically within MSH3, have been linked to the EMAST phenotype characterized by instability within tetranucleotide repeats. Patients with such phenotype have a poor prognosis. This might help partly understand colorectal cancer disparity in African Americans. Indeed, EMAST is reported to be highly prevalent (50 to 60%) in this population when compared to others [11].
The lack of matching normal for many of the analyzed the samples, and the absence of MSH2, POLE, MLH1 and SMAD2 on our targeted exome panel constitute limitations to our study. However, our study is a good example for other more comprehensive studies that will use larger sample size and more inclusive gene panels particularly for African American patients which is under-represented.
In this study, we examined and validated variants in 15 driver cancer genes in African Americans with colorectal neoplastic lesions. Many of these driver genes are involved and different pathways are affected by these variants which are all part of the carcinogenic process. Among oncogenes and TSGs, PIK3CA and APC genes were the most frequently altered genes, respectively. DNA MMR genes MSH3 and MSH6 also displayed a high level of variants that probably affect overall genetic stability. The distinct variant profiles with novel variants can help to predict, diagnose and establish new targeted therapeutic modalities for optimal CRC patients’ care.
MATERIALS AND METHODS
Discovery set
A total of 140 colorectal samples including 30 normals, 21 adenomas, 33 advanced adenomas (> 1 cm and/or with villous histology), and 56 cancers were used to establish variant profiles by targeted exome sequencing, using a PGM sequencing platform (Figure 1). These samples were collected from 123 patients at different stages of the disease. The Howard University Institutional board approved (06-MED-39) the collection of archival unidentified human samples.
Validation set
A total of 36 samples from 26 patients including 12 normals, 5 adenomas, 4 advanced adenomas and 15 cancers, were used to validate the Ion Torrent detected variants on a second sequencing platform (HiSeq, Illumina). The validation set is a subset from the discovery set samples (Figure 1).
Proton (Ion Torrent) NGS
A targeted, multiplex PCR panel was designed using the custom Ion AmpliSeq Designer v1.2 (ThermoFisher Scientific, Grand Island, NY). The panel amplified 56.9 kb and included the coding regions of 20 genes, with an average coverage of 96.9% of the protein coding regions and splice junctions (+5 intronic bases). In this study, we only report data from the 15 genes that are common to the Illumina gene panel that were sequenced in the validation set. The panel was designed to amplify PCR products with an average amplicon size of 150 base pairs (bp). Sample DNA (20 ng/primer pool) was amplified using the PCR panel, and libraries were prepared using the AmpliSeq Library Preparation kit following the manufacturer’s protocol (Thermo-Fisher Scientific, Grand Island, NY). Individual samples were barcoded, pooled, and sequenced on a Proton Sequencer using the Ion PI Template OT2 200v3 and Ion PI Sequencing 200v2 kits per the manufacturer’s instructions. Raw sequencing reads were filtered for high quality reads and the adaptors were removed using the Ion Torrent Suite 4.0.4, then reads were aligned to the hg19 reference sequence by TMAP (https://github.com/iontorrent/TS/tree/master/Analysis/TMAP) using default parameters. Resulting BAM files were processed through an in-house quality control (QC) filter and coverage analysis pipeline. BAM files were aligned using the GATK LeftAlignIndels module. Amplicon primers were trimmed from aligned reads using the Torrent Suite. Variant calls were made by the Torrent Variant Caller 4.0 (www.edgebio.com/variant-calling-ion-torrent-data).
HiSeq (Illumina) NGS
DNA quantification and quality assessment for the validation set, NGS using a HiSeq platform (Illumina, San Diego, CA), SNV and indel detection, and assessment of copy number alterations were performed as previously described [5, 6].
Bioinformatics and comparison of African Americans' variants to available databases
We used R software (version 2.15.2, http://www.r-project.org) to compare the variants in the normal and tumor samples with publicly available databases. Variants were annotated using ANNOVAR (56) and the 1000Genomes database, which represents a nominally noncancerous population. TCGA and GENIE databases were used to compute the frequency of mutations in another colorectal cancer database (TCGA) and a nominally non-cancerous population (GENIE). All samples displayed more or less an equal number of SNVs in their tumors compared with their matched normal samples. We would like to make it clear that variants identified using both platforms allowed us to identify weaknesses in bioinformatics associated with each platform that were addressed by rigorous manual review that improved variant detection on each individual platform. Subsequent to incorporating the more rigorous review, we identified additional variants with high confidence using a single platform.
Footnotes
Author contributions
Conception and design: HA, HB
Development of methodology: MN, SV, HAz
Acquisition of data (acquired and managed patients and samples): HAz, EL, AOL,
Analysis and interpretation of data (e.g., statistical analysis, biostatistics, computational analysis): SV, HA, HB, MN
Writing, review, and/or revision of the manuscript: HA, HB
Administrative, technical, or material support (i.e., reporting, organizing data): HA, HB
Study supervision: HA
CONFLICTS OF INTEREST
No potential conflicts of interest were disclosed.
GRANT SUPPORT
This project was supported (in part) by the National Institute on Minority Health and Health Disparities of the National Institutes of Health under Award Number G12MD007597.
REFERENCES
- 1.DeSantis CE, Siegel RL, Sauer AG, Miller KD, Fedewa SA, Alcaraz KI, Jemal A. Cancer statistics for African Americans, 2016: progress and opportunities in reducing racial disparities. CA Cancer J Clin. 2016;66:290–308. doi: 10.3322/caac.21340. [DOI] [PubMed] [Google Scholar]
- 2.Carethers JM. Racial and ethnic factors in the genetic pathogenesis of colorectal cancer. J Assoc Acad Minor Phys. 1999;10:59–67. [PubMed] [Google Scholar]
- 3.Satia JA, Keku T, Galanko JA, Martin C, Doctolero RT, Tajima A, Sandler RS, Carethers JM. Diet, lifestyle, and genomic instability in the North Carolina Colon Cancer Study. Cancer Epidemiol Biomarkers Prev. 2005;14:429–36. doi: 10.1158/1055-9965.EPI-04-0486. [DOI] [PubMed] [Google Scholar]
- 4.Guda K, Veigl ML, Varadan V, Nosrati A, Ravi L, Lutterbaugh J, Beard L, Willson JK, Sedwick WD, Wang ZJ, Molyneaux N, Miron A, Adams MD, et al. Novel recurrently mutated genes in African American colon cancers. Proceedings of the National Academy of Sciences of the United States of America. 2015;112:1149–54. doi: 10.1073/pnas.1417064112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ashktorab H, Azimi H, Varma S, Tavakoli P, Nickerson ML, Brim H. Distinctive DNA mismatch repair and APC rare variants in African Americans with colorectal neoplasia. Oncotarget. 2017;8:99966–77. doi: 10.18632/oncotarget.21557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ashktorab H, Daremipouran M, Devaney J, Varma S, Rahi H, Lee E, Shokrani B, Schwartz R, Nickerson ML, Brim H. Identification of novel mutations by exome sequencing in African American colorectal cancer patients. Cancer. 2015;121:34–42. doi: 10.1002/cncr.28922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ashktorab H, Ahuja S, Kannan L, Llor X, Nathan E, Xicola RM, Adeyinka LO, Carethers JM, Brim H, Nouraie M. A meta-analysis of MSI frequency and race in colorectal cancer. Oncotarget. 2016;7:34546–57. doi: 10.18632/oncotarget.8945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ashktorab H, Daremipouran M, Goel A, Varma S, Leavitt R, Sun X, Brim H. DNA methylome profiling identifies novel methylated genes in African American patients with colorectal neoplasia. Epigenetics. 2014;9:503–12. doi: 10.4161/epi.27644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ashktorab H, Rahi H, Wansley D, Varma S, Shokrani B, Lee E, Daremipouran M, Laiyemo A, Goel A, Carethers JM, Brim H. Toward a comprehensive and systematic methylome signature in colorectal cancers. Epigenetics. 2013;8:807–15. doi: 10.4161/epi.25497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Haugen AC, Goel A, Yamada K, Marra G, Nguyen TP, Nagasaka T, Kanazawa S, Koike J, Kikuchi Y, Zhong X, Arita M, Shibuya K, Oshimura M, et al. Genetic instability caused by loss of MutS homologue 3 in human colorectal cancer. Cancer Res. 2008;68:8465–72. doi: 10.1158/0008-5472.CAN-08-0002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Carethers JM, Koi M, Tseng-Rogenski SS. EMAST is a form of microsatellite instability that is initiated by inflammation and modulates colorectal cancer progression. Genes (Basel) 2015;6:185–205. doi: 10.3390/genes6020185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Venderbosch S, van Lent-van Vliet S, de Haan AF, Ligtenberg MJ, Goossens M, Punt CJ, Koopman M, Nagtegaal ID. EMAST is associated with a poor prognosis in microsatellite instable metastatic colorectal cancer. PLoS One. 2015;10:e0124538. doi: 10.1371/journal.pone.0124538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zocche DM, Ramirez C, Fontao FM, Costa LD, Redal MA. Global impact of KRAS mutation patterns in FOLFOX treated metastatic colorectal cancer. Front Genet. 2015;6:116. doi: 10.3389/fgene.2015.00116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Barresi V, Bonetti LR, Bettelli S. KRAS, NRAS, BRAF mutations and high counts of poorly differentiated clusters of neoplastic cells in colorectal cancer: observational analysis of 175 cases. Pathology. 2015;47:551–6. doi: 10.1097/PAT.0000000000000300. [DOI] [PubMed] [Google Scholar]
- 15.Vandrovcova J, Lagerstedt-Robinsson K, Pahlman L, Lindblom A. Somatic BRAF-V600E mutations in familial colorectal cancer. Cancer Epidemiol Biomarkers Prev. 2006;15:2270–3. doi: 10.1158/1055-9965.EPI-06-0359. [DOI] [PubMed] [Google Scholar]
- 16.Kalady MF, Dejulius KL, Sanchez JA, Jarrar A, Liu X, Manilich E, Skacel M, Church JM. BRAF mutations in colorectal cancer are associated with distinct clinical characteristics and worse prognosis. Dis Colon Rectum. 2012;55:128–33. doi: 10.1097/DCR.0b013e31823c08b3. [DOI] [PubMed] [Google Scholar]
- 17.Miled N, Yan Y, Hon WC, Perisic O, Zvelebil M, Inbar Y, Schneidman-Duhovny D, Wolfson HJ, Backer JM, Williams RL. Mechanism of two classes of cancer mutations in the phosphoinositide 3-kinase catalytic subunit. Science. 2007;317:239–42. doi: 10.1126/science.1135394. [DOI] [PubMed] [Google Scholar]
- 18.Muzny DM, Bainbridge MN, Chang K, Dinh HH, Drummond JA, Fowler G, Kovar CL, Lewis LR, Morgan MB, Newsham IF, Reid JG, Santibanez J, Shinbrot E, et al. Comprehensive molecular characterization of human colon and rectal cancer. Nature. 2012;487:330–7. doi: 10.1038/Nature11252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Shi Z, Chiang CI, Labhart P, Zhao Y, Yang J, Mistretta TA, Henning SJ, Maity SN, Mori-Akiyama Y. Context-specific role of SOX9 in NF-Y mediated gene regulation in colorectal cancer cells. Nucleic Acids Res. 2015;43:6257–69. doi: 10.1093/nar/gkv568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Segditsas S, Rowan AJ, Howarth K, Jones A, Leedham S, Wright NA, Gorman P, Chambers W, Domingo E, Roylance RR, Sawyer EJ, Sieber OM, Tomlinson IP. APC and the three-hit hypothesis. Oncogene. 2009;28:146–55. doi: 10.1038/onc.2008.361. [DOI] [PubMed] [Google Scholar]
- 21.Schneikert J, Brauburger K, Behrens J. APC mutations in colorectal tumours from FAP patients are selected for CtBP-mediated oligomerization of truncated APC. Hum Mol Genet. 2011;20:3554–64. doi: 10.1093/hmg/ddr273. [DOI] [PubMed] [Google Scholar]
- 22.Thiagalingam S, Lengauer C, Leach FS, Schutte M, Hahn SA, Overhauser J, Willson JK, Markowitz S, Hamilton SR, Kern SE, Kinzler KW, Vogelstein B. Evaluation of candidate tumour suppressor genes on chromosome 18 in colorectal cancers. Nat Genet. 1996;13:343–6. doi: 10.1038/ng0796-343. [DOI] [PubMed] [Google Scholar]
- 23.Shin YK, Yoo BC, Hong YS, Chang HJ, Jung KH, Jeong SY, Park JG. Upregulation of glycolytic enzymes in proteins secreted from human colon cancer cells with 5-fluorouracil resistance. Electrophoresis. 2009;30:2182–92. doi: 10.1002/elps.200800806. [DOI] [PubMed] [Google Scholar]
- 24.Lee SY, Kim DW, Lee HS, Ihn MH, Oh HK, Park DJ, Kim HH, Kang SB. Loss of AT-rich interactive domain 1A expression in gastrointestinal malignancies. Oncology. 2015;88:234–40. doi: 10.1159/000369140. [DOI] [PubMed] [Google Scholar]
- 25.de Miranda NF, van Dinther M, van den Akker BE, van Wezel T, ten Dijke P, Morreau H. Transforming Growth Factor β Signaling in Colorectal Cancer Cells With Microsatellite Instability Despite Biallelic Mutations in TGFBR2. Gastroenterology. 2015;148:1427–37.e8. doi: 10.1053/j.gastro.2015.02.052. [DOI] [PubMed] [Google Scholar]
- 26.Papageorgis P. Complex interplay between aging and cancer: role of TGF-beta signaling. Crit Rev Oncog. 2017;22:313–21. doi: 10.1615/CritRevOncog.2017025134. [DOI] [PubMed] [Google Scholar]
- 27.Li N, Lorenzi F, Kalakouti E, Normatova M, Babaei-Jadidi R, Tomlinson I, Nateri AS. FBXW7-mutated colorectal cancer cells exhibit aberrant expression of phosphorylated-p53 at Serine-15. Oncotarget. 2015;6:9240–56. doi: 10.18632/oncotarget.3284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Markowitz SD, Bertagnolli MM. Molecular origins of cancer: molecular basis of colorectal cancer. N Engl J Med. 2009;361:2449–60. doi: 10.1056/NEJMra0804588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Grohmann A, Tanneberger K, Alzner A, Schneikert J, Behrens J. AMER1 regulates the distribution of the tumor suppressor APC between microtubules and the plasma membrane. J Cell Sci. 2007;120:3738–47. doi: 10.1242/jcs.011320. [DOI] [PubMed] [Google Scholar]
- 30.Mur P, Aiza G, Sanz-Pamplona R, Gonzalez S, Navarro M, Moreno V, Capella G, Valle L. AMER1 is a frequently mutated gene in colorectal cancer--letter. Clin Cancer Res. 2015;21:4985. doi: 10.1158/1078-0432.CCR-15-1756. [DOI] [PubMed] [Google Scholar]
- 31.Folsom AR, Pankow JS, Peacock JM, Bielinski SJ, Heiss G, Boerwinkle E. Variation in TCF7L2 and increased risk of colon cancer: the atherosclerosis risk in communities (ARIC) study. Diabetes Care. 2008;31:905–9. doi: 10.2337/dc07-2131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Shen J, Fang Y, Ge W. Polymorphism in the transcription factor 7-like 2 (TCF7L2) gene is associated with impaired proinsulin conversion--A meta-analysis. Diabetes Res Clin Pract. 2015;109:117–23. doi: 10.1016/j.diabres.2015.04.020. [DOI] [PubMed] [Google Scholar]
- 33.Ashktorab H, Azimi H, Nickerson ML, Bass S, Varma S, Brim H. Targeted Exome Sequencing Outcome variations of colorectal tumors within and across two sequencing platforms. Next Gener Seq Appl. 2016;3:123. doi: 10.4172/2469-9853.1000123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ward AJ, Cooper TA. The pathobiology of splicing. J Pathol. 2010;220:152–63. doi: 10.1002/path.2649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Li L, Sheng Y, Li W, Hu C, Mittal N, Tohyama K, Seba A, Zhao YY, Ozer H, Zhu T, Qian Z. Beta-catenin is a candidate therapeutic target for myeloid neoplasms with del(5q) Cancer Res. 2017;77:4116–26. doi: 10.1158/0008-5472.CAN-17-0202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Angus-Hill ML, Elbert KM, Hidalgo J, Capecchi MR. T-cell factor 4 functions as a tumor suppressor whose disruption modulates colon cell proliferation and tumorigenesis. Proc Natl Acad Sci U S A. 2011;108:4914–9. doi: 10.1073/pnas.1102300108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sanz-Pamplona R, Lopez-Doriga A, Pare-Brunet L, Lazaro K, Bellido F, Alonso MH, Ausso S, Guino E, Beltran S, Castro-Giner F, Gut M, Sanjuan X, Closa A, et al. Exome sequencing reveals AMER1 as a frequently mutated gene in colorectal cancer. Clin Cancer Res. 2015;21:4709–18. doi: 10.1158/1078-0432.CCR-15-0159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Cajuso T, Hanninen UA, Kondelin J, Gylfe AE, Tanskanen T, Katainen R, Pitkanen E, Ristolainen H, Kaasinen E, Taipale M, Taipale J, Bohm J, Renkonen-Sinisalo L, et al. Exome sequencing reveals frequent inactivating mutations in ARID1A, ARID1B, ARID2 and ARID4A in microsatellite unstable colorectal cancer. Int J Cancer. 2014;135:611–23. doi: 10.1002/ijc.28705. [DOI] [PubMed] [Google Scholar]
- 39.Patsos G, Andre S, Roeckel N, Gromes R, Gebert J, Kopitz J, Gabius HJ. Compensation of loss of protein function in microsatellite-unstable colon cancer cells (HCT116): a gene-dependent effect on the cell surface glycan profile. Glycobiology. 2009;19:726–34. doi: 10.1093/glycob/cwp040. [DOI] [PubMed] [Google Scholar]
- 40.Matsumoto A, Onoyama I, Nakayama KI. Expression of mouse Fbxw7 isoforms is regulated in a cell cycle- or p53-dependent manner. Biochem Biophys Res Commun. 2006;350:114–9. doi: 10.1016/j.bbrc.2006.09.003. [DOI] [PubMed] [Google Scholar]
- 41.Liu Y, Zhang X, Han C, Wan G, Huang X, Ivan C, Jiang D, Rodriguez-Aguayo C, Lopez-Berestein G, Rao PH, Maru DM, Pahl A, He X, et al. TP53 loss creates therapeutic vulnerability in colorectal cancer. Nature. 2015;520:697–701. doi: 10.1038/nature14418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Schubbert S, Shannon K, Bollag G. Hyperactive ras in developmental disorders and cancer. Nat Rev Cancer. 2007;7:295–308. doi: 10.1038/nrc2109. [DOI] [PubMed] [Google Scholar]
- 43.Liu Y, Luan L, Wang X. A randomized Phase II clinical study of combining panitumumab and bevacizumab, plus irinotecan, 5-fluorouracil, and leucovorin (FOLFIRI) compared with FOLFIRI alone as second-line treatment for patients with metastatic colorectal cancer and KRAS mutation. Onco Targets Ther. 2015;8:1061–8. doi: 10.2147/OTT.S81442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Normanno N, Rachiglio AM, Lambiase M, Martinelli E, Fenizia F, Esposito C, Roma C, Troiani T, Rizzi D, Tatangelo F, Botti G, Maiello E, Colucci G, et al. Heterogeneity of KRAS, NRAS, BRAF and PIK3CA mutations in metastatic colorectal cancer and potential effects on therapy in the CAPRI GOIM trial. Ann Oncol. 2015;26:1710–4. doi: 10.1093/annonc/mdv176. [DOI] [PubMed] [Google Scholar]
- 45.Di Nicolantonio F, Martini M, Molinari F, Sartore-Bianchi A, Arena S, Saletti P, De Dosso S, Mazzucchelli L, Frattini M, Siena S, Bardelli A. Wild-type BRAF is required for response to panitumumab or cetuximab in metastatic colorectal cancer. J Clin Oncol. 2008;26:5705–12. doi: 10.1200/JCO.2008.18.0786. [DOI] [PubMed] [Google Scholar]
- 46.Prahallad A, Sun C, Huang S, Di Nicolantonio F, Salazar R, Zecchin D, Beijersbergen RL, Bardelli A, Bernards R. Unresponsiveness of colon cancer to BRAF(V600E) inhibition through feedback activation of EGFR. Nature. 2012;483:100–3. doi: 10.1038/nature10868. [DOI] [PubMed] [Google Scholar]
- 47.Palmirotta R, Ludovici G, De Marchis ML, Leone B, Formica V, Ettorre GM, Cavaliere F, Della-Morte D, Ferroni P, Roselli M, Guadagni F. A comprehensive procedural approach to genotyping KRAS and BRAF from paraffin embedded tissues for diagnostic purposes. In Vivo. 2012;26:537–47. [PubMed] [Google Scholar]
- 48.Ogino S, Lochhead P, Giovannucci E, Meyerhardt JA, Fuchs CS, Chan AT. Discovery of colorectal cancer PIK3CA mutation as potential predictive biomarker: power and promise of molecular pathological epidemiology. Oncogene. 2014;33:2949–55. doi: 10.1038/onc.2013.244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Rosty C, Young JP, Walsh MD, Clendenning M, Sanderson K, Walters RJ, Parry S, Jenkins MA, Win AK, Southey MC, Hopper JL, Giles GG, Williamson EJ, et al. PIK3CA activating mutation in colorectal carcinoma: associations with molecular features and survival. PLoS One. 2013;8:e65479. doi: 10.1371/journal.pone.0065479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Kim E, Ilic N, Shrestha Y, Zou L, Kamburov A, Zhu C, Yang X, Lubonja R, Tran N, Nguyen C, Lawrence MS, Piccioni F, Bagul M, et al. Systematic functional interrogation of rare cancer variants identifies oncogenic alleles. Cancer Discov. 2016;6:714–26. doi: 10.1158/2159-8290.CD-16-0160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Bastide P, Darido C, Pannequin J, Kist R, Robine S, Marty-Double C, Bibeau F, Scherer G, Joubert D, Hollande F, Blache P, Jay P. Sox9 regulates cell proliferation and is required for Paneth cell differentiation in the intestinal epithelium. J Cell Biol. 2007;178:635–48. doi: 10.1083/jcb.200704152. [DOI] [PMC free article] [PubMed] [Google Scholar]