Abstract
Objective(s):
To examine the change in physical functional status among persons living with HIV (PLWH) in nursing homes (NHs) and how change varies with age and dementia.
Design:
Retrospective cohort study.
Setting:
NHs in 14 states in the United States.
Participants:
PLWH who were admitted to NHs between 2001 and 2010 and had stays of ≥90 days (N = 3550).
Measurements:
We linked Medicaid Analytic eXtract (MAX) and Minimum Data Set (MDS) data for NH residents in the sampled states and years and used them to determine HIV infection. The main outcome was improvement in physical functional status, defined as a decrease of at least 4 points in the activities of daily living (ADL) score within 90 days of NH admission. Independent variables of interest were age and dementia (Alzheimer’s disease or other dementia). Multivariate logistic regression was used, adjusting for individual-level covariates.
Results:
The average age on NH admission of PLWH was 58. Dementia prevalence ranged from 14.5% in the youngest age group (age <40 years) to 38.9% in the oldest group (age ≥70 years). Overall, 44% of the PLWH experienced ADL improvement in NHs. Controlling for covariates, dementia was related to a significantly lower likelihood of ADL improvement among PLWH in the oldest age group only: the adjusted probability of improvement was 40.6% among those without dementia and 29.3% among those with dementia (P < .01).
Conclusions/relevance:
PLWH, especially younger persons, may be able to improve their ADL function after being admitted into NHs. However, with older age, PLWH with dementia are more physically dependent and vulnerable to deterioration of physical functioning in NHs. More and/or specialized care may be needed to maintain physical functioning among this population. Findings from this study provide NHs with information on care needs of PLWH and inform future research on developing interventions to improve care for PLWH in NHs.
Keywords: HIV, dementia, nursing home, physical functional status
The effectiveness of antiretroviral treatment has increased the life expectancy of persons living with HIV (PLWH). It was estimated that 26% of the persons with HIV infection were 55 years and older in the United States in 2013.1 This increased life expectancy is accompanied with increased prevalence of age-related chronic conditions, such as dementia, diabetes, cardiovascular, liver disease, and cancer among PLWH. These conditions occur at an earlier age among PLWH compared with HIV-negative persons, partially because of the side effects of the antiretroviral medication as well as the persistent impaired immune system and inflammation despite the antiretroviral treatment.2 HIV infection, comorbidities, and the aging process increase the frailty level and the potential need for nursing home (NH) care among PLWH.3
The needs of NH care among PLWH may be accelerated by the presence of dementia, which has been identified as one of the most important predictors for NH placement.4,5 Although the prevalence of dementia increases with advancing age, PLWH may be more vulnerable to the development of dementia compared with HIV-negative persons at similar ages.6–9 It was estimated that 25% of PLWH aged 50 years and older had HIV-associated dementia, based on a study in 2001.10 Furthermore, dementia is an important contributor to the decline of physical functioning among older adults.11–14 One crosssectional study compared physical functioning of PLWH with and without dementia at NH admission, and found that those with dementia were more likely to have functional impairment than those with no dementia.15 Independency of physical functioning significantly influences the quality of life among PLWH. However, there is little empirical evidence on the trajectory of physical functional status among PLWH in NHs. Findings from HIV-negative NH residents may not apply to PLWH as there are significant differences between these 2 populations,16 and PLWH may be at higher risks of functional decline due to HIV infection.3 Furthermore, no studies, to our knowledge, have explored the relationship between the trajectory of physical functional status and dementia among PLWH, as well as the potential variations of this relationship by age. Age is closely related to individual frailty level, and thus the trajectory of physical functioning may well vary with age among PLWH. Understanding the trajectories of physical functioning among PLWH may help NHs understand the needs of this population and to be better prepared to provide care that meets its needs.
To answer these questions, we used data from 2000 to 2010 in the 14 states with the highest HIV prevalence (accounting for 75% of PLWH in the United States).17 The study population included PLWH who were admitted to NHs and stayed for at least 90 days, and who therefore had an admission and follow-up assessment. This study provides information for NH providers as well as policy makers. As PLWH are aging and the demand for NH care among this population is increasing, it is important to understand the special care needs of PLWH and to maintain their physical independence as feasible. Only by doing so can we hope to achieve the goal of “successful aging” and to maintain quality of life among older adults with HIV.
Methods
Data
Medicaid claims data (Medicaid Analytic eXtract [MAX] data) and the Minimum Data Set (MDS) 2.0 were linked for NH residents in the selected 14 states (New York, California, Florida, Texas, Maryland, New Jersey, Pennsylvania, Illinois, Georgia, North Carolina, Virginia, Louisiana, Ohio, and Massachusetts) between 2000 and 2010. MAX data contain information on medical services covered by Medicaid, such as date of services and diagnoses associated with inpatient or outpatient events, as well as information on the use of prescription drugs. Dually eligible patients (those with both Medicaid and Medicare coverage) are included in the MAX data. The MDS 2.0 is a federally mandated assessment tool for all residents in Medicare and/or Medicaid certified nursing facilities.18 Federal regulations require the MDS to be administered at admission, and thereafter at least at 3-month intervals (for those who remain in residence). Additional assessments may be needed if a resident receives Medicare-covered post-acute care services or when there are significant changes in individual health conditions. The MDS contains information about individuals’ demographic characteristics (eg, age, gender, race) and detailed health conditions, such as physical functional status, cognitive status, and comorbidities.
Study Cohort
We identified residents with HIV admitted to NHs in the sampled states who stayed in NHs for at least 90 days, and who had therefore had at least 2 MDS assessments, between 2001 and 2010 (2000 data were used as a look-back period to obtain individual chronic conditions). We used both the MDS and MAX data to identify PLWH in NHs. The MDS contains a check box for a list of medical conditions, including HIV infection, as well as ICD-9 diagnoses. We used both ICD- 9 diagnoses and the diagnosis check boxes in the MDS to identify HIV status. In addition, the MAX data were linked with the MDS data to supplement the identification of PLWH. The approach that we used to identify PLWH in the MAX data has been described previously.19 A person was considered to have HIV if she or he was identified as HIV positive by either the MDS or the MAX data. During the study period, 9 of the 14 states that we studied (FL, GA, LA, NC, NY, OH, PA, TX, and VA) included data on HIV infection in the MDS data that are sent to the Centers for Medicare & Medicaid Services, and 5 states (CA, TX, MD, NJ, and IL) elected to not send data on HIV to the Centers for Medicare & Medicaid Services. Thus, for the states where the HIV diagnosis was included in the MDS, the identified PLWH included both patients who were Medicaid eligible and those who were not. For states that did not include HIV status in the MDS, we were only able to capture NH residents who were Medicaid eligible. However, the majority of PLWH admitted to NHs are covered by Medicaid for medical services,15 which allows us to identify HIV diagnoses for most. We excluded residents with activities of daily living (ADL) score less than 4 points at NH admission (ie, little or no impairment) as there would be no room for ADL improvement (as discussed below). Lastly, we excluded individuals with terminal conditions (a documented prognosis of 6 months or less) and/or those enrolled in hospice (based on the MDS assessments used in our analysis). In total, 3550 unique NH residents (ie, PLWH) were included in the analytical sample.
Variables
The outcome variable was improvement in ADL between the admission MDS assessment and a subsequent MDS that was approximately 90 days later. The ADL scale constructed from the MDS ranged from 0 (no limitation in physical functioning) to 28 (maximal ADL impairment).20 Consistent with previous studies, we considered a 4- point change in the ADL score as a “real” change as ADL scores may fluctuate, and individuals with a 4-point change in ADL score were less likely to return to their original scores in the following assessments.14,20,21 Also, like others, we dichotomized the outcome variable such that a decrease of 4 or more points in the ADL score was considered “improvement” (score of 1). Patients with 3 points or less of improvement, or those with declines, were given a score of zero.
The main independent variables of interest were age (categorized as <40,40-49, 50-59, 60-69, and ≥70 years) and a diagnosis of dementia (Alzheimer’s disease or other dementia). We used both the diagnosis checkbox as well as the ICD-9 codes in the MDS to identify patients with dementia.22 Other individual-level covariates were identified using the MDS and included sociodemographic characteristics (ie, race, gender, marital status), whether the resident was living alone prior to NH admission (we used this variable and marital status as potential indicators of likelihood of home discharge), whether the resident was admitted from hospital (as a proxy for a likely post-acute care admission), the presence of a do-not-resuscitate order, ADL score at admission, comorbid conditions (cardiovascular disease, pulmonary disease, diabetes, anxiety, depression, bipolar, depression, schizophrenia, and cancer), and cognitive status [measured by Cognitive Performance Scale (CPS)]. The CPS was constructed using the MDS data and ranges from 0 (intact cognitive status) to 6 (very severe cognitive impairment).23 Based on the distribution of the CPS scale and previous studies, we categorized individuals as having no or mild cognitive impairment (0-2), moderate cognitive impairment (3-4), and severe impairment (5-6). Lastly, we included dichotomous variables indicating the year of a resident’s NH admission to control for any time trend.
Statistical Analysis
We first compared the characteristics of PLWH in NHs by age and the presence ofdementia. Next we fit a logistic regression model to examine the relationship between ADL improvement, age, and dementia, accounting for the individual covariates as described above. We examined the relationship between age and dementia by introducing an age-by-dementia interaction term into the model. Because interaction terms in a logistic models are difficult to interpret,24 we calculated average adjusted probabilities of ADL improvement for PLWH with and without dementia for each age group, accounting for covariates. For each age group, we tested the differences in the average adjusted probabilities between PLWH with and without dementia. Lastly, sensitivity analysis was performed by including additional interaction effects between age groups and the categories of cognitive impairment.
The study was approved by the institutional review board IRB at Brown University. All the analyses were performed by SAS 9.4 and Stata 15.
Results
The average age of PLWH on NH admission was 58.1, with 10.7% younger than 40 years and 24.5% older than 70 years. The prevalence of dementia was 23.0%, and ranged from 14.5% for those younger than 40 years to 38.9% among those older than 70 years. Forty-four percent of PLWH experienced ADL improvement (within 90 days) subsequent to NH admission.
As presented in Table 1, the likelihood of ADL improvement differed between PLWH with and without dementia, and the difference varied with age. For example, among PLWH whose age was between 40 and 49 years, ADL improvement occurred in 52.1% of residents without dementia compared to 41.4% of those with dementia. For those older than 70 years, the percentage with ADL improvement was 39.7% and 22.7%, respectively. Table 1 also shows the distribution of other comorbidities by age group and the presence of dementia. Although the prevalence of some comorbidities, such as cardiovascular disease, pulmonary disease, and diabetes, increased with age, there was no clear relationship between the prevalence of some psychiatric conditions (eg, anxiety and depression) and age. The distribution of these conditions varied with the presence of dementia as well.
Table 1.
Individual Characteristics of PLWH in NHs, by Age Group and Dementia Status
| Variables | Age Groups, y |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| <40 |
40-49 |
50-59 |
60-69 |
≥70 |
||||||
| Without Dementia (n = 325) |
With Dementia (n = 55) |
Without Dementia (n = 676) |
With Dementia (n = 145) |
Without Dementia (n = 665) |
With Dementia (n = 128) |
Without Dementia (n = 536) |
With Dementia (n = 149) |
Without Dementia (n = 532) |
With Dementia (n = 339) |
|
| ADL improvement (yes) | 48.3 | 52.7 | 52.1 | 41.4 | 51.4 | 42.2 | 43.8 | 36.9 | 39.7 | 22.7 |
| Race | ||||||||||
| White | 29.90 | 29.10 | 26.20 | 35.10 | 29.00 | 32.80 | 31.20 | 18.80 | 44.90 | 46.60 |
| Black | 60.6 | 54.5 | 63.6 | 49.0 | 60.3 | 57.0 | 56.3 | 61.7 | 36.1 | 37.2 |
| Other race | 9.5 | 16.4 | 10.2 | 15.9 | 10.7 | 10.2 | 12.5 | 19.5 | 19.0 | 16.2 |
| Female | 28.6 | 23.6 | 25.0 | 17.2 | 28.7 | 28.9 | 32.3 | 32.2 | 53.9 | 60.8 |
| Married | 6.8 | 10.9 | 8.9 | 6.2 | 12.8 | 12.5 | 15.5 | 11.4 | 18.8 | 20.4 |
| Living status prior to admission | ||||||||||
| Living with others | 59.1 | 65.5 | 54.1 | 58.6 | 47.5 | 54.7 | 44.6 | 53.0 | 46.2 | 49.0 |
| Living alone | 22.8 | 20.0 | 20.7 | 24.8 | 24.4 | 29.7 | 26.7 | 24.2 | 19.2 | 17.1 |
| Living in other facility | 5.5 | 9.1 | 6.1 | 9.7 | 4.8 | 4.7 | 3.5 | 11.4 | 6.2 | 15.0 |
| Missing | 12.6 | 5.5 | 19.1 | 6.9 | 23.3 | 10.9 | 25.2 | 11.4 | 28.4 | 18.9 |
| Admitted from hospital | 75.4 | 76.4 | 71.7 | 73.8 | 69.2 | 75.0 | 64.4 | 77.2 | 59.0 | 61.9 |
| ADL score (0-28) at admission | 16.76(7.26) | 16.49(6.97) | 15.72(6.82) | 15.50(7.03) | 16.41(6.72) | 15.87(6.79) | 17.79(6.34) | 16.75(7.14) | 19.58(6.38) | 20.21(6.66) |
| Cognitive Performance Scale (CPS) score | ||||||||||
| 0-2 | 67.4 | 41.8 | 73.7 | 29.7 | 71.0 | 37.5 | 67.2 | 35.6 | 56.6 | 23.6 |
| 3-4 | 22.2 | 43.6 | 20.7 | 54.5 | 22.1 | 53.1 | 26.1 | 43.0 | 32.5 | 54.3 |
| 5-6 | 10.5 | 14.5 | 5.6 | 15.9 | 6.9 | 9.4 | 6.7 | 21.5 | 10.9 | 22.1 |
| Cardiovascular disease | 22.8 | 25.5 | 36.5 | 35.2 | 48.7 | 43.0 | 68.8 | 70.5 | 72.9 | 80.5 |
| Pulmonary disease | 6.8 | 5.5 | 10.7 | 7.6 | 16.8 | 12.5 | 22.9 | 13.4 | 22.2 | 20.4 |
| Diabetes | 8.6 | 9.1 | 17.6 | 14.5 | 28.3 | 21.1 | 42.2 | 37.6 | 35.0 | 32.7 |
| Cancer | 1.5 | 3.6 | 3.4 | 2.8 | 5.0 | 1.6 | 5.8 | 4.0 | 7.7 | 8.6 |
| Anxiety | 5.8 | 9.1 | 8.0 | 9.7 | 7.7 | 10.9 | 6.3 | 8.7 | 8.3 | 12.1 |
| Bipolar | 7.4 | 3.6 | 6.7 | 7.6 | 4.7 | 5.5 | 3.5 | 2.0 | 1.3 | 2.4 |
| Depression | 34.8 | 38.2 | 33.1 | 31.0 | 32.5 | 39.8 | 29.5 | 24.2 | 26.5 | 30.4 |
| Schizophrenia | 5.8 | 5.5 | 8.1 | 9.7 | 7.5 | 10.2 | 6.2 | 7.4 | 3.4 | 2.9 |
| Do-not-resuscitate order | 10.2 | 20.0 | 10.7 | 14.5 | 11.1 | 14.1 | 11.0 | 16.1 | 20.9 | 31.3 |
ADL improvement defined as a decrease of 4 or more points in the ADL scores between the admission assessment and a subsequent assessment that was approximately 90 days later.
Dementia refers to both Alzheimer’s and other dementia (non-Alzheimer’s).
Numbers in the cell indicate prevalence in percentages (for categorical variables) and mean (standard deviation) for ADL.
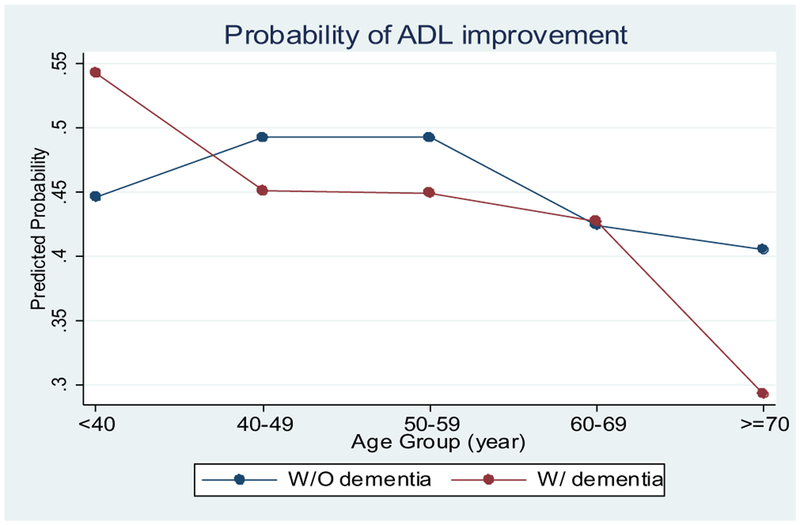
The results from the logistic regression on the likelihood of ADL improvement within 90 days of NH admission, adjusting for other covariates, are shown in Table 2. To more easily interpret the interaction effect between dementia and age, we calculated the average adjusted probabilities of ADL improvement for PLWH with and without dementia for each age group. As presented by Figure 1, the average probabilities of ADL improvement for these 2 groups of residents varied with age. Specifically, the average adjusted probabilities of ADL improvement were 44.7 (age <40 years), 49.3 (age 40-49 years), 49.3 (age 50-59 years), 42.5 (age 60-69 years), and 40.6 (age ≥70 years) percentage points among PLWH without dementia, and were 54.3, 45.1, 45.0, 42.8, and 29.3 percentage points (respectively) for corresponding age groups with dementia. Statistical test results of the differences in these probabilities between PLWH with dementia and without dementia by each age group are shown in Table 3. The relationship between dementia and ADL improvement was only statistically significant among PLWH older than 70 years; the probability of having improved functional status within 90 days of NH admission was 11.2 percentage points lower for PLWH without dementia compared to those with dementia (P < .01).
Table 2.
The Relationship Between ADL Improvement and Individual Characteristics Among PLWH in NHs: Results From Logistic Regression
| Odds Ratio | 95% Confidence Interval |
|
|---|---|---|
| Age groups, y | ||
| <40 | Reference | |
| 40-49 | 1.22 | 0.92, 1.61 |
| 50-59 | 1.22 | 0.92, 1.62 |
| 60-69 | 0.91 | 0.67, 1.23 |
| ≥70 | 0.84 | 0.61, 1.14 |
| Dementia | 1.52 | 0.84, 2.76 |
| Interaction between dementia and age | ||
| Dementia × age 40-49 years | 0.55 | 0.27, 1.12 |
| Dementia × age 50-59 years | 0.55 | 0.27, 1.12 |
| Dementia × age 60-69 years | 0.67 | 0.33, 1.36 |
| Dementia × age ≥70 years | 0.39*** | 0.20, 0.76 |
| Race | ||
| White | Reference | |
| Black | 1.06 | 0.90, 1.26 |
| Other race | 1.19 | 0.95, 1.51 |
| Female | 0.81*** | 0.69, 0.94 |
| Living status prior to admission | ||
| Living with others | Reference | |
| Living alone | 1.60*** | 1.34, 1.92 |
| Living in other facility | 1.05 | 0.77, 1.43 |
| Missing | 2.79*** | 2.10, 3.70 |
| Married | 0.94 | 0.76, 1.16 |
| Admitted from hospital | 2.14*** | 1.68, 2.72 |
| Cardiovascular disease | 0.92 | 0.79, 1.07 |
| Pulmonary disease | 1.04 | 0.85, 1.26 |
| Diabetes | 0.81** | 0.68, 0.95 |
| ADL score (0-28) at admission | 1.03*** | 1.02, 1.04 |
| Do-not-resuscitate order | 0.97 | 0.79, 1.19 |
| Anxiety | 0.95 | 0.73, 1.23 |
| Bipolar | 1.46** | 1.03, 2.06 |
| Depression | 1.03 | 0.88, 1.20 |
| Schizophrenia | 1.15 | 0.86, 1.53 |
| Cancer | 0.97 | 0.70, 1.35 |
| Cognitive Performance Scale (CPS) score | ||
| 0-2 | Reference | |
| 3-4 | 0.69*** | 0.58, 0.81 |
| 5-6 | 0.28*** | 0.21, 0.38 |
| Year | ||
| 2001 | Reference | |
| 2002 | 1.29 | 0.82, 2.03 |
| 2003 | 0.91 | 0.58, 1.41 |
| 2004 | 1.26 | 0.83, 1.92 |
| 2005 | 0.95 | 0.63, 1.44 |
| 2006 | 0.74 | 0.49, 1.11 |
| 2007 | 0.71 | 0.48, 1.05 |
| 2008 | 0.70 | 0.47, 1.03 |
| 2009 | 0.72 | 0.49, 1.07 |
| 2010 | 0.76 | 0.49, 1.16 |
P < .01,
P < .05.
Fig. 1.
The average adjusted probabilities of ADL improvement for PLWH in NHs, by dementia status and age groups.
Table 3.
The Average Effect of Dementia on the Probability of ADL Improvement by Age Group, Accounting for Individual Characteristics (Average Marginal Effect of Dementia)
| Age, y | Average Differences in Probability of ADL Improvement Between PLWH With Dementia and PLWH Without Dementia |
Standard Error | P > z |
|---|---|---|---|
| <40 | 0.10 | 0.07 | .17 |
| 40-49 | −0.04 | 0.05 | .36 |
| 50-59 | −0.04 | 0.05 | .36 |
| 60-69 | 0.00 | 0.05 | .95 |
| ≥70 | −0.11 | 0.03 | .00 |
The findings from sensitivity analysis (ie, also including interaction terms between CPS and age groups) were similar to the main analyses.
Discussion
We identified an admission cohort of PLWH in NHs in 14 states between 2001 and 2010 and followed them for 90 days to explore the trajectory of physical functioning and how this trajectory was modified by age and dementia. We focused on ADL improvement rather than ADL decline in this study, as PLWH were generally younger and were expected to maintain their physical functioning. Indeed, we found that almost half (44%) of the PLWH experienced improvement in physical functioning within 90 days of NH admission. However, the likelihood of ADL improvement varied by age and the presence of dementia. Specifically, older PLWH were more physically dependent at admission and were less likely to improve their physical functioning during their NH stay as compared with younger persons, but the reduction in the likelihood of physical functioning improvement was mainly among older adults with dementia.
Prior studies have suggested that at similar ages PLWH have higher care needs than persons without HIV infection because PLWH are at higher risk for age-related comorbidities, and the prevalence of these comorbidities increase greatly with age for PLWH.2,25 In this study, we also found great variations in the prevalence of dementia among PLWH at different ages. Such variation also modified the trajectory of physical functioning among PLWH. These findings suggest that PLWH are able to improve physical function status after their admission into NHs, and dementia does not seem to affect the likelihood of ADL improvement among younger PLWH. Therefore, it may be important to properly incentivize NHs to provide optimal level of care to this population to maximize the improvement of physical functioning and thus quality of life. On the other hand, older PLWH with dementia are at higher risks of physical decline (even when considering the degree of cognitive impairment), and thus may require a higher level of care to maintain their physical functioning. However, the provision of high-quality care to PLWH could be challenging to NHs, and it is unclear whether NHs are prepared to meet this challenge. NH staff generally provides care to older non-HIV infected residents and may lack knowledge about HIV and have had little training on care provision to PLWH.26 In addition, stigma and negative attitude towards older PLWH may further increase the barriers to providing high-quality care to PLWH.27,28These barriers may potentially accelerate the deterioration of health conditions, the loss of functional independency, and worsening of quality of life among aging PLWH. To achieve the goal of high-quality care and maintenance of physical independence for PLWH in NHs, staff may need to be educated on HIV infection and its care, and trained on caring for aging PLWH.26
The level of care delivered to PLWH may also be affected by the reimbursement for PLWH. Provision of optimal NH care to PLWHs can be costly and require NH investment in staff education and training.29 Although Medicare recognizes the high care needs of PLWH and has adopted an add-on approach to adjust for the additional resources required to care for post-acute residents with HIV infection,30 it is unclear whether the add-on payment sufficiently captures the additional costs of care for PLWH, considering the multiplicative effect between age and comorbidities among PLWH as suggested by our findings. Moreover, Medicare only reimburses NH residents who are Medicare eligible and receive post-acute services in NHs. Many PLWH are reimbursed by Medicaid for their NH stay. States vary significantly in Medicaid NH reimbursement methods, such as the payment rate and case-mix adjustment algorithm. Although some states’ Medicaid programs, such as New York’s, adjust for the potential additional costs among PLWH by adopting an add-on approach, other states do not (eg, CA).31 NHs may not be incentivized to invest in improving the care for PLWH because of insufficient funding for this specialized care. Furthermore, the financial disincentives may create barriers for PLWH to access high-quality NH care. Future research is necessary to examine whether and how consideration of HIV status in payment algorithms affects the quality of care delivered to aging PLWH, including their trajectories of functional decline.
The study has some limitations. First, we focused on PLWH who were admitted to NHs and stayed for at least 90 days. Thus, our findings may not be generalized to shorter-stay PLWHs, who may have different care needs. Second, although we focused on PLWH who stayed in NHs for at least 90 days (and adjusted for admission source), some residents may have been receiving physical or occupational therapy when admitted. These residents may have had different trajectories in functional status than those who received custodial care in NHs. Third, this study did not compare the trajectory of ADL among PLWH vs HIV-negative residents in NHs. A prior study found that about 25% to 27% of NH residents with cognitive impairment (generally) experienced ADL improvement over a 90-day observational window.14 The differences in the likelihood of ADL improvement between PLWH vs residents generally suggest that the differences in profiles and needs of care exist between these 2 populations. Future studies to compare the trajectory of ADL between PLWH and HIVnegative residents can provide a better understanding of the differences that exist.
In spite of these limitations, our study contributes to the literature. The study was based on a population of PLWH who were admitted to NHs in 14 US states across numerous years, and thus it represents the largest such study to date that focuses on an NH admission cohort of PLWH. It is also the first study, to our knowledge, to explore changes in physical functioning among PLWH who are admitted to NHs, and this exploration provided new knowledge about how age and comorbidities appear to affect changes in physical functioning among PLWH.
Conclusions and Relevance
In conclusion, the success of antiretroviral treatment has increased the life expectancy for PLWH but with this increase the prevalence of age-related comorbidities, such as dementia, has also increased, resulting in a greater need for NH care among PLWH. Using a population-based NH admission cohort of PLWH, we found that the combination of older age and the presence of dementia was significantly associated with a lower probability of functional improvement. Findings from this study provide NHs with information on care needs of PLWH and inform future comparative and intervention research aimed at ensuring high-quality NH care, including the maintenance of functional independence, for this vulnerable population.
Acknowledgments
The study is funded by National Institute of Mental Health (NIMH) grant 1R01MH102202. The National Institute of Mental Health had no role in the design and conduct of the study; in the collection, management, analysis, and interpretation of the data; or in the preparation, review, or approval of the manuscript.
The authors declare no conflicts of interest. Dr. Wilson is partially supported by the Providence/Boston Center for AIDS Research (P30AI042853) and by Institutional Development Award Number U54GM115677 from the National Institute of General Medical Sciences of the National Institutes of Health, which funds Advance Clinical and Translational Research (Advance-CTR) from the Rhode Island IDeA-CTR award (U54GM115677).
References
- 1.Centers for Disease Control and Prevention. HIV among people aged 50 and over. Available at: https://www.cdc.gov/hiv/group/age/olderamericans/index.html. Accessed December 18, 2017.
- 2.Deeks SG, Phillips AN. HIV infection, antiretroviral treatment, ageing, and non-AIDS related morbidity. BMJ 2009;338:a3172. [DOI] [PubMed] [Google Scholar]
- 3.Althoff KN, Jacobson LP, Cranston RD, et al. Age, comorbidities, and AIDS predict a frailty phenotype in men who have sex with men. J Gerontol A Biol Sci Med Sci 2014;69:189–198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Gaugler JE, Duval S, Anderson KA, Kane RL. Predicting nursing home admission in the U.S: A meta-analysis. BMC Geriatr 2007;7:13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Miller EA, Weissert WG. Predicting elderly people’s risk for nursing home placement, hospitalization, functional impairment, and mortality: A synthesis. Med Care Res Rev 2000;57:259–297. [DOI] [PubMed] [Google Scholar]
- 6.Harris-Kojetin L, Sengupta M, Park-Lee E, Valverde R. Long-term care services in the United States: 2013 overview. Vital Health Stat 2013;37:1–107. [PubMed] [Google Scholar]
- 7.McArthur JC, Hoover DR, Bacellar H, et al. Dementia in AIDS patients: Incidence and risk factors. Multicenter AIDS Cohort Study. Neurology 1993;43: 2245–2252. [DOI] [PubMed] [Google Scholar]
- 8.Becker JT, Kingsley LA, Molsberry S, et al. Cohort profile: Recruitment cohorts in the neuropsychological substudy of the Multicenter AIDS Cohort Study. Int J Epidemiol 2015;44:1506–1516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gelman BB. Neuropathology of HAND with suppressive antiretroviral therapy: Encephalitis and neurodegeneration reconsidered. Curr HIV/AIDS Rep 2015;12: 272–279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Valcour V, Shikuma C, Shiramizu B, et al. Higher frequency of dementia in older HIV-1 individuals: The Hawaii Aging with HIV-1 Cohort. Neurology 2004;63: 822–827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Aguero-Torres H, Fratiglioni L, Guo Z, et al. Dementia is the major cause of functional dependence in the elderly: 3-year follow-up data from a population-based study. Am J Public Health 1998;88:1452–1456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Black SA, Rush RD. Cognitive and functional decline in adults aged 75 and older. J Am Geriatr Soc 2002;50:1978–1986. [DOI] [PubMed] [Google Scholar]
- 13.Mlinac ME, Feng MC. Assessment of activities of daily living, self-care, and independence. Arch Clin Neuropsychol 2016;31:506–516. [DOI] [PubMed] [Google Scholar]
- 14.Carpenter GI, Hastie CL, Morris JN, et al. Measuring change in activities of daily living in nursing home residents with moderate to severe cognitive impairment. BMC Geriatr 2006;6:7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Buchanan RJ, Wang S, Huang C. Analyses of nursing home residents with HIV and dementia using the minimum data set. J Acquir Immun Defic Syndr 2001; 26:246–255. [DOI] [PubMed] [Google Scholar]
- 16.Buchanan RJ, Wang S, Huang C. Profiles of nursing home residents with HIV. J Health Care Poor Underserved 2002;13:379–391. [DOI] [PubMed] [Google Scholar]
- 17.Centers for Disease Control and Prevention. HIV surveillance report, 2010, vol 22 Atlanta, GA: Division of HIV/AIDS Prevention, National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention Centers for Disease Control and Prevention (CDC), U.S. Department of Health and Human Services; 2012. [Google Scholar]
- 18.Centers for Medicare & Medicaid Services. MDS 2.0 for NHs: Centers for Medicare & Medicaid Services. Available at: http://www.cms.gov/Medicare/Quality-Initiatives-Patient-Assessment-Instruments/NursingHomeQualityInits/NHQIMDS20.html. Accessed December 18, 2017.
- 19.Youn B, Shireman TI, Lee Y, et al. Ten-year trends in antiretroviral therapy persistence among US Medicaid beneficiaries. AIDS 2017;31:1697–1707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Morris JN, Fries BE, Morris SA. Scaling ADLs within the MDS. J Gerontol A Biol Sci Med Sci 1999;54:M546–M553. [DOI] [PubMed] [Google Scholar]
- 21.Mor V, Gruneir A, Feng Z, et al. The effect of state policies on nursing home resident outcomes. J Am Geriatr Soc 2011;59:3–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mor V, Intrator O, Unruh MA, Cai S. Temporal and geographic variation in the validity and internal consistency of the Nursing Home Resident Assessment Minimum Data Set 2.0. BMC Health Serv Res 2011;11:78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hartmaier SL, Sloane PD, Guess HA, et al. Validation of the Minimum Data Set Cognitive Performance Scale: Agreement with the Mini-Mental State Examination. J Gerontol A Biol Sci Med Sci 1995;50:M128–M133. [DOI] [PubMed] [Google Scholar]
- 24.Ai C, Norton EC. Interaction terms in logit and probit models. Econ Lett 2003; 80:123–129. [Google Scholar]
- 25.Oursler KK, Goulet JL, Crystal S, et al. Association of age and comorbidity with physical function in HIV-infected and uninfected patients: Results from the Veterans Aging Cohort Study. AIDS Patient Care STDs 2011;25: 13–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Siou K, Mahan M, Cartagena R. Chan Carusone S. A growing need—HIV education in long-term care. Geriatr Nurs 2017;38:199–206. [DOI] [PubMed] [Google Scholar]
- 27.Cahill S, Valadez R. Growing older with HIV/AIDS: New public health challenges. Am J Public Health 2013;103:e7–e15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Emlet CA. “You’re awfully old to have this disease”: Experiences of stigma and ageism in adults 50 years and older living with HIV/AIDS. Gerontologist 2006; 46:781–790. [DOI] [PubMed] [Google Scholar]
- 29.Swan JH, Benjamin AE, Brown A. Skilled nursing facility care for persons with AIDS: Comparison with other patients. Am J Public Health 1992;82:453–455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Department of Health & Human Services (DHHS), Centers for Medicare & Medicaid Services (CMS). CMS Manual System. Pub. 100–04 Medicare Claims Processing. 2004. Available at: www.cms.gov/transmittals/downloads/r160cp.pdf. Accessed December 18, 2017.
- 31.MACPAC. States’ Medicaid Fee-for-Service Nursing Facility Payment Policies. 2014. Available at: https://www.macpac.gov/wp-content/uploads/2015/01/Nursing-Facilty-Payment-Policies.xlsx. Accessed December 18, 2017.