Abstract
Objective
To assess long-term survival and late cardiovascular events in patients with atrial myxoma after surgical intervention.
Methods
Retrospective analysis of 403 patients undergoing resection of atrial myxoma from January 2002 to December 2016 was conducted with a median follow-up period of 4.5 (range: 0.5–15) years.
Results
The cross-clamp time and cardiopulmonary bypass times were 41.1 ± 21.4 and 65.2 ± 27.3 min, respectively. A diagnosis of myxoma was histopathologically confirmed in all cases. The early in-hospital mortality rate was 0.7% (n = 3). During the follow-up period, tumor recurrence occurred in six patients and cerebral infarction in nine. There were 48 (11.9%) patients with late onset atrial fibrillation (AF). By multivariate analysis, age (HR = 1.05, 95% CI: 1.02–1.09, P < 0.001), left atrial diameter (HR = 1.23, 95% CI: 1.08–1.36, P = 0.012), and mitral valve surgery (HR = 1.17, 95% CI: 1.05–1.29, P = 0.027) were independent predictors of late onset AF. Twenty-one (5.2%) patients died during the follow-up period. Advanced age (HR = 1.07, 95% CI: 1.04–1.10, P = 0.003) and multiple surgical procedures (HR = 1.18, 95% CI: 1.06–1.29, P = 0.012) were significantly associated with overall mortality.
Conclusions
Atrial myxoma can be resected with good long-term survival. Late onset AF is common after surgery in patients with atrial myxoma. Advanced age, left atrial diameter, and mitral valve surgery were independent predictors of outcomes.
Keywords: Atrial fibrillation, Atrial myxoma, Embolism, Survival
1. Introduction
Cardiac myxoma is the most common primary heart neoplasm[1] and can arise in any of the cardiac cavities, although most occur in the left atrium.[2] Some patients are asymptomatic, while others present with constitutional symptoms, which include dyspnea, palpitation, fatigue, and chest pain.[3]–[5] In some cases, a cardiac myxoma may be lethal because of valve or outflow tract obstruction, which results in embolic complications, atrial fibrillation (AF), and heart failure.[3]–[5] Surgical resection is the primary treatment for these tumors.
Previous studies on cardiac myxoma are mostly case reports on clinical manifestations, diagnosis, and surgical treatments. However, there is limited data on long-term survival and the incidences of AF, embolic events, and tumor recurrence after surgical resection. The aim of the present study was to assess long-term survival and late cardiovascular events after surgical intervention in a large series of patients with atrial myxoma.
2. Methods
2.1. Study population
The medical records of 448 consecutive patients with cardiac myxoma who underwent surgical resection in our hospital from January 2002 to December 2016 were retrospectively reviewed. After excluding five patients with left ventricle myxoma, four with right ventricle myxoma, and 36 with missing data, 403 patients with atrial myxoma were finally included for analysis. The study protocol was approved by the Institutional Ethics Review Committee of Beijing Anzhen Hospital (China) and was conducted in accordance with the tenets of the Declaration of Helsinki. All patients provided written informed consent prior to enrollment.
2.2. Ultrasound evaluation
All patients routinely underwent transthoracic echocardiography after admission with the use of a Vivid 9 cardiovascular ultrasound system (GE Healthcare, Chicago, IL, USA) equipped with a 5-MHz Omni-Plane TEE Probe Ultrasound Transducer (Koninklijke Philips N.V., Eindhoven, Netherlands). The left atrial diameter (LAD), left ventricular end-diastolic dimension (LVEDD), left ventricular end-systolic dimension (LVESD), left ventricular ejection fraction (LVEF), and other parameters were measured from the parasternal M-mode or two-dimensional images. Transthoracic echocardiography was used to confirm the diagnosis of cardiac myxoma in all cases. The morphologic features of cardiac myxoma were carefully examined [i.e., tumor location, tumor morphology (irregular/smooth), tumor dimension, and pedicle width]. According to the guidelines, planimetry of the mitral orifice was used to assess mitral stenosis and color flow Doppler mapping was used to quantify the regurgitant jet area of the mitral valve.[6],[7] These measurements were evaluated in multiple views. All recordings were performed and interpreted by experienced physicians who were blinded to the study population.
2.3. Surgical procedure, pathological examination, and follow-up
Myxomas were excised through a right atrial approach during total cardiopulmonary bypass support under moderate hypothermia (30°C) with bicaval cannulation for venous drainage. The full thickness of the atrial septum was excised if the lesion was in the region of the fossa ovalis. When the tumor was attached to the left atrial wall or the mitral valve, the extent of resection of normal tissue was evaluated on an individual basis. The atrial septal defect created by resection was closed with a Dacron patch or, less commonly, by primary closure.
Surgical AF ablation was conducted with a bipolar Cardioblate™ Surgical Ablation System (Medtronic, Minneapolis, MN, USA) or an AtriCure clamp (AtriCure, Inc., West Chester, OH, USA). After the aorta was cross-clamped, the heart was arrested and the left atrium was accessed through a left atriotomy. Left atrial ablation lines included isolation of the pulmonary veins and a connecting line between both islands of pulmonary veins on the roof, and a line from the left pulmonary veins to the posterior mitral annulus. The left atrial appendage was excised or excluded. Right atrial ablation lines included the superior vena cava to the inferior vena cava, the lateral free-wall lesion complete to the anterior-medial tricuspid valve annulus, and the medial free-wall lesion complete to the anterior-medial tricuspid valve annulus and cavotricuspid isthmus ablation. Ablation of the right appendage completed the procedure.
The tumor tissues obtained during surgery were sent to the Pathology Department and routinely stained with hematoxylin and eosin. Clinical and follow-up data were obtained from electronic medical records and standardized telephone interviews. Patients were followed-up by trained physicians who were not involved in the surgical procedure or study design. The median follow-up period was 4.5 (range: 0.5–15) years. The follow-up cardiovascular events included tumor recurrence, late onset AF, embolic events, cerebrovascular diseases, and all-cause death postoperatively. Electrocardiograms and Holter recordings were documented when the patients were symptomatic. Late onset AF was defined as any atrial tachyarrhythmia lasting > 30 s and occurring after the first three months.[8]
2.4. Statistical analysis
All statistical analyses were performed using IBM SPSS Statistics for Windows, version 19.0 (IBM Corporation, Armonk, NY, USA). Values are presented as mean ± SD for normally distributed continuous variables and as proportions for categorical variables. The differences between normally distributed continuous variables were assessed using the unpaired two-tailed t-test or the Mann–Whitney U test for skewed variables. Categorical variables were compared using the chi-squared test or Fisher exact test, when appropriate. Cox regression analysis was performed to identify predictors of late onset AF and overall mortality. All hazard ratios (HRs) are given with the 95% confidence interval (CI). The Kaplan–Meier method was used to construct a survival curve. A two-sided probability P value of < 0.05 was considered statistically significant.
3. Results
3.1. Baseline characteristics of patients
The baseline clinical characteristics of the patients are summarized in Table 1. The mean age of the study population was 54.7 ± 8.2 years and there were twice as many females than males (275 vs. 128, respectively). One hundred (24.8%) patients had hypertension, 38 (9.4%) had coronary artery disease, 35 (8.7%) had diabetes mellitus, 35 (8.7%) had a history of AF, and 49 (12.2%) had a history of congestive heart failure. Notably, 60 (14.9%) patients had previous embolic events with cerebral embolism accounting for the majority.
Table 1. Baseline clinical characteristics of the study population.
| Characteristics | All patients |
| Age, yrs | 54.7 ± 8.2 |
| Male | 128 (31.8%) |
| BMI, kg/m2 | 23.9 ± 3.4 |
| Hypertension | 100 (24.8%) |
| Coronary artery disease | 38 (9.4%) |
| Diabetes mellitus | 35 (8.7%) |
| History of AF | 35 (8.7%) |
| Congestive heart failure | 49 (12.2%) |
| Embolic events | 60 (14.9%) |
| Cerebral embolism | 53 (88.3%) |
| Lower limb arterial embolism | 6 (10%) |
| Pulmonary embolism | 1 (1.7%) |
| Myxoma location | |
| Left atrium | 374 (92.8%) |
| Right atrium | 29 (7.2%) |
| Myxoma dimension | |
| Dimension 1, mm | 40.3 ± 18.1 |
| Dimension 2, mm | 30.2 ± 12.1 |
| LAD, mm | 37.2 ± 6.4 |
| LVEDD, mm | 46.9 ± 5.4 |
| LVESD, mm | 30.3 ± 4.5 |
| LVEF | 64.4% ± 6.0% |
Data are presented as means ± SD or n (%). AF: atrial fibrillation; BMI: body mass index; LAD: left atrial diameter; LVEDD: left ventricular end-diastolic dimension; LVEF: left ventricular ejection fraction; LVESD: left ventricular end-systolic dimension.
3.2. Intraoperative events
The cross-clamp time and cardiopulmonary bypass time were 41.1 ± 21.4 and 65.2 ± 27.3 min, respectively. Besides tumor resection, 29 patients underwent coronary artery bypass grafting, 25 mitral valve surgery, two aortic valve surgery, 21 tricuspid valve plasty, and 18 surgical radiofrequency ablation for AF.
All myxoma diagnoses were histopathologically confirmed. The early in-hospital mortality rate was 0.7% (n = 3). Two patients succumbed to postoperative hemorrhage and one due to multiple organ failure.
3.3. Assessment of long-term outcomes
During the follow-up period, tumor recurrence occurred in six (1.5%) patients. The details of patients with recurrence are summarized in Table 2. Nine (2.2%) patients experienced cerebral infarction.
Table 2. Details of patients with tumor recurrence.
| Patient | Gender | Tumor | Age, yrs | Location | Pathology |
| 1 | Male | First | 24 | LA, LV | Myxoma |
| Second | 25 | LA | Myxoma | ||
| Third | 27 | LV | Myxoma | ||
| Fourth | 30 | LV | Myxoma | ||
| 2 | Female | First | 26 | LA | Myxoma |
| Second | 31 | LA | Myxoma | ||
| Third | 38 | RV | Myxoma | ||
| Fourth | 40 | RV | Myxoma | ||
| Fifth | 44 | RV | Myxoma | ||
| 3 | Male | First | 26 | LA | Myxoma |
| Second | 32 | LA | Myxoma | ||
| 4 | Male | First | 59 | LA | Myxoma |
| Second | 67 | LA | Myxoma | ||
| 5 | Female | First | 13 | LA | Myxoma |
| Second | 20 | LA | Myxoma | ||
| 6 | Female | First | 39 | LA | Myxoma |
| Second | 53 | LA | Myxoma |
LA: left atrium; LV: left ventricle; RV: right ventricle.
Late onset AF was noted in 48 (11.9%) patients (Figure 1). Several clinical factors were assessed to identify predictors of late onset AF. As shown in Table 3, age (P < 0.001), LAD (P = 0.012), and mitral valve surgery (P = 0.027) were significantly associated with late onset AF, while male gender, myxoma dimension, myxoma location, history of AF, and surgical ablation had no correlation. In multivariate analysis, age (HR = 1.05, 95% CI: 1.02–1.09, P < 0.001), LAD (HR = 1.23, 95% CI: 1.08–1.36, P = 0.012), and mitral valve surgery (HR = 1.17, 95% CI: 1.05–1.29, P = 0.027) remained independent predictors of late onset AF.
Figure 1. Late onset AF rates in patients with atrial myxoma.
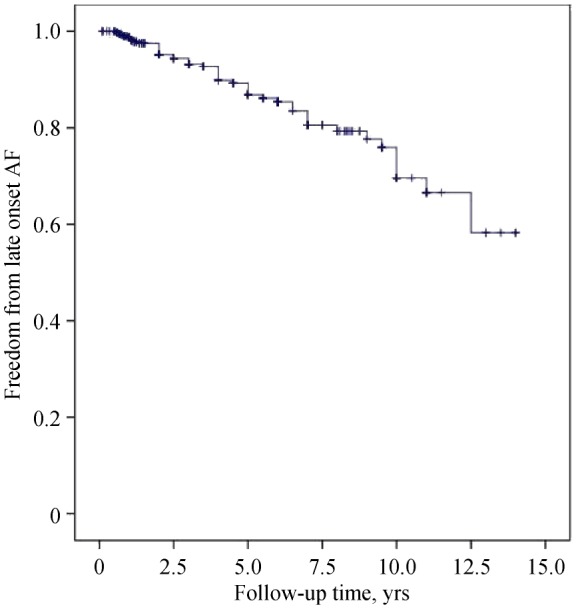
AF: atrial fibrillation.
Table 3. Cox regression model to predict late onset AF.
| Variable | Univariate analysis |
Multivariate analysis |
||||
| HR | 95% CI | P-value | HR | 95% CI | P-value | |
| Age | 1.04 | 1.03–1.06 | < 0.001 | 1.05 | 1.02–1.09 | < 0.001 |
| Male gender | 0.83 | 0.44–1.56 | 0.563 | |||
| LAD | 1.15 | 1.05–1.29 | 0.012 | 1.23 | 1.08–1.36 | 0.005 |
| Myxoma dimension | 1.22 | 0.83–1.61 | 0.465 | |||
| Myxoma location | 1.43 | 0.76–2.05 | 0.679 | |||
| History of AF | 1.18 | 0.92–1.42 | 0.247 | |||
| Surgical ablation | 0.95 | 0.82–1.06 | 0.086 | |||
| Mitral valve surgery* | 1.28 | 1.12–1.41 | 0.027 | 1.17 | 1.05–1.29 | 0.032 |
*Mitral valve surgery refers to the mitral valve repair/replacement. AF: atrial fibrillation; LAD: left atrial diameter.
A total of 21 (5.2%) patients died during the follow-up period. Advanced age (HR = 1.07, 95% CI: 1.04–1.10, P = 0.003) and multiple surgical procedures (HR = 1.18, 95% CI: 1.06–1.29, P = 0.012) were significantly associated with overall mortality (Table 4). There was no significant difference in survival when the patients were stratified by gender (P = 0.138), hypertension (P = 0.561), history of congestive heart failure (P = 0.379), or left ventricular ejection fraction (P = 0.379).
Table 4. Univariate Cox regression model to predict overall mortality.
| Variable | HR | 95% CI | P-value |
| Age | 1.07 | 1.04–1.10 | 0.003 |
| Male gender | 1.13 | 0.85–1.40 | 0.138 |
| Hypertension | 1.36 | 0.77–1.93 | 0.561 |
| Congestive heart failure | 1.22 | 0.91–1.51 | 0.379 |
| LVEF | 0.94 | 0.82–1.05 | 0.254 |
| Multiple surgical procedures* | 1.18 | 1.06–1.29 | 0.012 |
*Mitral surgical procedures refers to the tumor resection plus (at least one) coronary artery bypass grafting, mitral valve surgery, aortic valve surgery, or tricuspid valve plasty. LVEF: left ventricular ejection fraction.
4. Discussion
The focus of this single-center, retrospective study was the long-term outcome in a series of 403 patients with atrial myxoma. The present study aimed to provide some insight to long-term survival and late cardiovascular events, as at present there are insufficient data. To the best of our knowledge, this is the largest series of patients with atrial myxoma to date.
We found that atrial myxoma can be resected with good long-term survival. Advanced age and multiple surgical procedures were significantly associated with overall mortality. Age, left atrial diameter, and mitral valve surgery were independent predictors of late onset AF, which was common in patients with atrial myxoma after surgery. Tumor recurrence and postoperative embolic events were relatively rare.
AF is frequently a concomitant complication of atrial myxoma.[9],[10] In this series, 8.7% of patients with atrial myxoma had a history of AF and 11.9% developed late onset AF postoperatively. Notably, a history of AF was not associated with late onset AF by regression analysis. It is reasonable that advanced age and left atrial diameter are classical predictors of AF.[11] Atrial myxoma is frequently accompanied with mitral valve diseases and surgery is indicated for moderate to severe mitral valve disease. Mitral valve diseases is a predisposing factor to changes in left atrial pressure and remodeling of the left atrium, which facilitate the development of AF.[12] Therefore, both age and functioning of the left atrium contribute to late onset AF among patients with atrial myxoma. Sahin, et al.[13] observed that 27.8% of patients developed postoperative AF within three months after surgery and that age and P-wave dispersion were independent predictors of postoperative AF. In contrast to this previous study on short-term postoperative AF, we were concerned with late onset (> 3 months) AF.
Late mortality ranged from 0 to 10% in previous studies.[14]–[16] Shah, et al.[17] reported 10-, 20-, and 30-year survival rates of 77.0%, 51.8%, and 33.6% in cardiac myxoma, respectively, similar to those of an age- and sex-matched local population, and that advanced age at the time of surgery was the only significant predictor of mortality, as there was no association with sex, tumor location, largest tumor dimension, and surgical technique. In the present series, the overall survival rate was 94.8% during a mean follow-up period of 4.5 years, with the longest period of 15 years. Similar to the results of prior studies, the risk factors for overall mortality were advanced age, multiple surgical procedures, and concomitant diseases. Advanced age, baseline cardiovascular disease, and a longer follow-up period might result in a low late survival rate.[18],[19]
Tumor recurrence is a potential concern due to malignant biological properties.[20] The frequency of recurrence varied between 0–3% for sporadic cases to > 20% for cases of Carney complex,[21]–[23] while the rate in the present series was 1.5%. This cohort included a 30-year-old male patient with four instances of recurrence in the left atrium and left ventricle.[24] He had a heterozygous base pair mutation C > T at chr17:68522867 in the protein kinase A regulatory subunit 1a (PRKAR1A) gene, the causative gene for Carney complex and familial cardiac myxoma. It seems that younger age, smaller size of the initial tumor, and tumor location in the ventricle were predictors of tumor recurrence.[17]
Postoperative neurological complications were rare[25],[26] and only 2.2% of the patients experienced cerebral infarction after surgery. Myxoma-related cerebral infarction differs from atherosclerotic embolism and AF-related venous embolism, as the former might be due to the irregular fragments of the myxoma entering into the bloodstream.[27] Hence, the source of an embolus from a myxoma was addressed perioperatively.
This series of patients were followed-up for a relatively long period (up to 15 years). Although there have been no important changes to surgical interventions during this period, recent improvements in perioperative management might have improved the overall outcome.[28]
There were several limitations to this retrospective study that should be addressed. Firstly, the postoperative in-hospital AF rate was not precisely assessed. Secondly, electrocardiograms and Holter recordings were documented when the patients were symptomatic, so the incidence of late onset AF may have been underestimated. Last but not least, postoperative drug treatment was not evaluated in detail for every case.
In conclusion, atrial myxoma can be resected with good long-term survival. Late onset AF is common in patients with atrial myxoma after surgical treatment, and age, left atrial diameter, and mitral valve surgery were identified are independent prognostic factors. Since there exists a risk for tumor recurrence, regular postoperative examinations are recommended for patients with atrial myxoma.
Acknowledgments
This study was supported by grants from the National Natural Science Foundation of China (No.81670294 & No.81200141 & No.81470430 & No.81770320 & No. 81570291) and the Beijing Novel Program (No.2011081 & Z131103000413116). The authors had no conflicts of interest to disclose.
References
- 1.Di Vito A, Mignogna C, Donato G. The mysterious pathways of cardiac myxomas: a review of histogenesis, pathogenesis and pathology. Histopathology. 2015;66:321–332. doi: 10.1111/his.12531. [DOI] [PubMed] [Google Scholar]
- 2.O'Rourke F, Dean N, Mouradian MS, et al. Atrial myxoma as a cause of stroke: case report and discussion. CMAJ. 2003;169:1049–1051. [PMC free article] [PubMed] [Google Scholar]
- 3.Li Q, Shang H, Zhou D, et al. Repeated embolism and multiple aneurysms: central nervous system manifestations of cardiac myxoma. Eur J Neurol. 2008;15:e112–e113. doi: 10.1111/j.1468-1331.2008.02295.x. [DOI] [PubMed] [Google Scholar]
- 4.Akhtar J, Wasay M, Rauf J. Atrial myxoma: a rare cause of cardioembolic stroke. BMJ Case Rep. 2012;2012:bcr2012006176–bcr2012006176. doi: 10.1136/bcr.2012.006176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Habertheuer A, Laufer G, Wiedemann D, et al. Primary cardiac tumors on the verge of oblivion: a European experience over 15 years. J Cardiothorac Surg. 2015;10:56–56. doi: 10.1186/s13019-015-0255-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Baumgartner H, Hung J, Bermejo J, et al. Echocardiographic assessment of valve stenosis: EAE/ASE recommendations for clinical practice. J Am Soc Echocardiogr. 2009;22:1–23. doi: 10.1016/j.echo.2008.11.029. [DOI] [PubMed] [Google Scholar]
- 7.Zoghbi WA, Enriquez-Sarano M, Foster E, et al. Recommendations for evaluation of the severity of native valvular regurgitation with two-dimensional and Doppler echocardiography. J Am Soc Echocardiogr. 2003;16:777–802. doi: 10.1016/S0894-7317(03)00335-3. [DOI] [PubMed] [Google Scholar]
- 8.Kirchhof P, Benussi S, Kotecha D, et al. 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Eur Heart J. 2016;37:2893–2962. doi: 10.1093/eurheartj/ehw210. [DOI] [PubMed] [Google Scholar]
- 9.Haroun MJ, Nair V, Salehian O. Now you see it, now you don't? Circulation. 2015;131:1872–1879. doi: 10.1161/CIRCULATIONAHA.115.015652. [DOI] [PubMed] [Google Scholar]
- 10.Keeling IM, Oberwalder P, Anelli-Monti M, Schuchlenz H, et al. Cardiac myxomas: 24 years of experience in 49 patients. Eur J Cardiothorac Surg. 2002;22:971–977. doi: 10.1016/s1010-7940(02)00592-4. [DOI] [PubMed] [Google Scholar]
- 11.January CT, Wann LS, Calkins H, et al. 2019 AHA/ACC/HRS focused update of the 2014 AHA/ACC/HRS Guideline for the management of patients with atrial fibrillation: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2019;pii:S0735–1097(19)30209–8. [Google Scholar]
- 12.Gillinov AM, Gelijns AC, Parides MK, et al. Surgical ablation of atrial fibrillation during mitral-valve surgery. N Engl J Med. 2015;372:1399–1409. doi: 10.1056/NEJMoa1500528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sahin M, Tigen K, Dundar C, et al. Postoperative atrial fibrillation in patients with left atrial myxoma. Cardiovasc J Afr. 2015;26:120–124. doi: 10.5830/CVJA-2014-069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Samanidis G, Perreas K, Kalogris P, et al. Surgical treatment of primary intracardiac myxoma: 19 years of experience. Interact CardioVasc Thorac Surg. 2011;13:597–600. doi: 10.1510/icvts.2011.278705. [DOI] [PubMed] [Google Scholar]
- 15.Tasoglu I, Tutun U, Lafci G, et al. Primary cardiac myxomas: clinical experience and surgical results in 67 patients. J Card Surg. 2009;24:256–259. doi: 10.1111/j.1540-8191.2008.00797.x. [DOI] [PubMed] [Google Scholar]
- 16.Garatti A, Nano G, Canziani A, et al. Surgical excision of cardiac myxomas: twenty years experience at a single institution. Ann Thorac Surg. 2012;93:825–831. doi: 10.1016/j.athoracsur.2011.11.009. [DOI] [PubMed] [Google Scholar]
- 17.Shah IK, Dearani JA, Daly RC, et al. Cardiac Myxomas: a 50-year experience with resection and analysis of risk factors for Recurrence. Ann Thorac Surg. 2015;100:495–500. doi: 10.1016/j.athoracsur.2015.03.007. [DOI] [PubMed] [Google Scholar]
- 18.Vroomen M, Houthuizen P, Khamooshian A, et al. Long-term follow-up of 82 patients after surgical excision of atrial myxomas. Interact Cardiovasc Thorac Surg. 2015;21:183–188. doi: 10.1093/icvts/ivv125. [DOI] [PubMed] [Google Scholar]
- 19.D'Alfonso A, Catania S, Pierri MD, et al. Atrial myxoma: a 25-year single-institutional follow-up study. J Cardiovasc Med (Hagerstown) 2008;9:178–181. doi: 10.2459/JCM.0b013e3281ac22cb. [DOI] [PubMed] [Google Scholar]
- 20.Wang JG, Li YJ, Liu H, et al. Clinicopathologic analysis of cardiac myxomas: Seven years' experience with 61 patients. J Thorac Dis. 2012;4:272–283. doi: 10.3978/j.issn.2072-1439.2012.05.07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Briassoulis G, Kuburovic V, Xekouki P, et al. Recurrent left atrial myxomas in Carney complex: a genetic cause of multiple strokes that can be prevented. J Stroke Cerebrovasc Dis. 2012;21:914.e1–914.e8. doi: 10.1016/j.jstrokecerebrovasdis.2012.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Acebo E, Val-Bernal JF, Gómez-Román JJ, et al. Clinicopathologic study and DNA analysis of 37 cardiac myxomas: a 28-year experience. Chest. 2003;123:1379–1385. doi: 10.1378/chest.123.5.1379. [DOI] [PubMed] [Google Scholar]
- 23.Wang Z, Chen S, Zhu M, et al. Risk prediction for emboli and recurrence of primary cardiac myxomas after resection. J Cardiothorac Surg. 2016;11:22–22. doi: 10.1186/s13019-016-0420-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kong X, Zhou M, Tu X, et al. Multiple recurrent cardiac myxomas with protein kinase a regulatory subunit 1α gene mutation. Ann Thorac Surg. 2019;107:e83–e85. doi: 10.1016/j.athoracsur.2018.06.069. [DOI] [PubMed] [Google Scholar]
- 25.Lee SJ, Kim JH, Na CY, et al. Eleven years' experience with Korean cardiac myxoma patients: focus on embolic complications. Cerebrovasc Dis. 2012;33:471–479. doi: 10.1159/000335830. [DOI] [PubMed] [Google Scholar]
- 26.He DK, Zhang YF, Liang Y, et al. Risk factors for embolism in cardiac myxoma: a retrospective analysis. Med Sci Monit. 2015;21:1146–1154. doi: 10.12659/MSM.893855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Dias RR, Fernandes F, Ramires FJ, et al. Mortality and embolic potential of cardiac tumors. Arq Bras Cardiol. 2014;103:13–18. doi: 10.5935/abc.20140096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kuroczyński W, Peivandi AA, Ewald P, et al. Cardiac myxomas: short- and long-term follow-up. Cardiol J. 2009;16:447–454. [PubMed] [Google Scholar]


