Abstract
Background
Falls are a common occurrence and the most effective quality improvement (QI) strategies remain unclear.
Methods
We conducted a systematic review and network meta-analysis (NMA) to elucidate effective quality improvement (QI) strategies for falls prevention. Multiple databases were searched (inception−April 2017). We included randomised controlled trials (RCTs) of falls prevention QI strategies for participants aged ≥65 years. Two investigators screened titles and abstracts, full-text articles, conducted data abstraction and appraised risk of bias independently.
Results
A total of 126 RCTs including 84,307 participants were included after screening 10,650 titles and abstracts and 1210 full-text articles. NMA including 29 RCTs and 26,326 patients found that team changes was statistically superior in reducing the risk of injurious falls relative to usual care (odds ratio [OR] 0.57 [0.33 to 0.99]; absolute risk difference [ARD] −0.11 [95% CI, −0.18 to −0.002]). NMA for the outcome of number of fallers including 61 RCTs and 40 128 patients found that combined case management, patient reminders and staff education (OR 0.18 [0.07 to 0.47]; ARD −0.27 [95% CI, −0.33 to −0.15]) and combined case management and patient reminders (OR, 0.36 [0.13 to 0.97]; ARD −0.19 [95% CI, −0.30 to −0.01]) were both statistically superior compared to usual care.
Conclusions
Team changes may reduce risk of injurious falls and a combination of case management, patient reminders, and staff education, as well as case management and patient reminders may reduce risk of falls. Our results can be tailored to decision-maker preferences and availability of resources.
Systematic review registration
PROSPERO (CRD42013004151)
Keywords: falls, falling, quality improvement, older people, fall prevention, systematic review
Key points
Numerous randomised controlled trials (RCTs) and systematic reviews have examined falls prevention quality improvement (QI) strategies, but the key effective elements remain unclear.
Network meta-analysis (NMA) allows the ranking of all available falls prevention and combinations of falls prevention QI strategies for each outcome.
Our systematic review and NMA of all available falls prevention QI strategies for older people encompassing 126 RCTs and 84,307 participants, is the first to identify effective QI strategies for preventing falls using NMA.
We found that effective falls prevention QI strategies are multi-faceted, and including components targeting patients (such as education and reminders), as well as components targeting clinicians (such as team changes, case management and staff education) will increase likelihood of effectiveness.
Depending on the outcome desired, our results can be tailored according to decision-maker preferences and availability of resources.
Introduction
Despite high-quality evidence of effective interventions for prevention [1–5], falls remain a common occurrence among those aged 65 years and older, representing an enormous burden to patients, their caregivers and the health care system [6, 7]. This represents a quality of care gap because high-quality evidence is not being implemented to prevent falls and injuries. In order to reduce this gap, quality improvement (QI) strategies can be considered.
QI strategies can help with the implementation of evidence-based clinical interventions, such as falls prevention programmes. QI strategies can target patients (e.g. patient education), health care providers (e.g. clinician education), and/or the health care system (e.g. financial incentives). As the population continues to age [8], QI interventions will have the potential to prevent numerous falls in the future. In a previous study of falls prevention interventions, QI strategies were effective at reducing injurious falls [9], but specific types of QI strategies and their combinations were not explored. The effectiveness of specific QI strategies alone or in combination for preventing falls amongst older people was explored through a systematic review and network meta-analysis (NMA).
Methods
Protocol
A protocol was prepared using the Preferred Reporting Items for Systematic Review and Meta-analysis (PRISMA) Protocols Statement [10], registered with PROSPERO (CRD42013004151), and published [11]. An integrated knowledge translation approach was used, whereby the systematic review team included knowledge users who posed the research question, and were invited to participate in all steps of the review. The PRISMA-NMA extension [12] was followed for the reporting of results (see Appendix P in the supplementary data, available at Age and Ageing online), which are related to another paper examining all clinical interventions for falls prevention in addition to QI strategies [9].
Eligibility criteria
Eligible studies were randomised controlled trials (RCTs). Quasi-randomised trials were excluded. The RCTs had to include patients aged 65 years or older and any type of setting (e.g. living independently in the community, long-term care) was eligible for inclusion. The RCT had to examine one of 13 QI strategies that were adapted from the US Agency for Healthcare Research and Quality, which are outlined in Box 1 [13, 14]. The comparators examined in the RCTs were usual care or other falls prevention QI strategies.
Box 1.
Falls prevention quality improvement strategies
| Strategy | Description | Examples |
|---|---|---|
| QI strategies targeting health care providers (clinic or care team) | ||
| 1) Case management | Any system for coordinating diagnosis, treatment, or routine management of patients (e.g. arrangement for referrals, follow-up of test results) by a person or multidisciplinary team in collaboration with, or supplementary to, the primary-care clinician. If the study called the intervention ‘case management’ we classified it as such | Nurse functioning as case management role, which included assessment of patient status, planning, evaluation of adherence to dietary/medication recommendation, advocacy, home visits, and care coordination |
| 2) Team changes | Changes to the structure or organisation of the primary health care team (adding team member, multidisciplinary teams, expansion or revision of professional roles) | Modifying a geriatric outpatient clinic to include assessment by a nurse and physiotherapist, in addition to a geriatrician |
| 3) Facilitated relay of info to clinicians | Clinical information collected from patients and transmitted to clinicians by means other than the existing medical record (excluding conventional means of correspondence between clinicians.) | Communicating a patient’s medication information to care providers in the long-term care facilities including the family physician, nursing staff, and community physician |
| 4) Continuous QI | Interventions explicitly identified as involving the techniques of continuous QI, total quality management, or plan-do-study-act, or any iterative process for assessing quality problems, developing solutions to those problems, testing their effects, and then reassessing the need for further action | Weekly continuous quality improvement meetings to discuss progress of individual participants and to help maximise their success in the intervention |
| 5) Audit & feedback | Summary of clinical performance of health care delivered by an individual clinician or clinic over a specified period, which was then transmitted back to the clinician. This strategy was strictly based on clinical data and excluded clinical skills | Providing data on the number of patients with missing tests to the clinician |
| 6) Staff education | Interventions for staff designed to promote increased understanding of principles guiding clinical care or awareness of specific recommendations for a target disorder or population of patients | Includes conferences or workshops, distribution of educational materials (written, video or other), and educational outreach visits |
| 7) Clinician reminders | Paper-based or electronic systems intended to prompt a health professional to recall patient-specific information (e.g. most recent HbA1c value) or to do a specific task (e.g. foot examination) | Development of a Fall Prevention Tool Kit software (decision support system) to prompt physicians when evaluating a patient |
| QI strategies targeting health system | ||
| 8) Financial incentives | Interventions with positive or negative financial incentives directed at providers (e.g. linked to adherence to some process of care or achievement of some target outcome). This strategy also includes positive or negative financial incentives directed at patients or system-wide changes in reimbursement | Examples are gym memberships, drug assistance programs, free medications. Rides to the intervention or parking are not included |
| QI strategies targeting patients | ||
| 9) Promotion of self-management | Provision of equipment or access to resources to promote self-management. If the study called the intervention promotion of self-management, personalised goal-setting, or action-planning, we included it here. (We generally thought this a more active strategy than education of patients) | Problem-solving skills, tracking number of steps, fit bit, self-help groups, working with patient to develop strategies to reduce risk of falling |
| 10) Patient Reminders | Any effort to remind patients about upcoming appointments or important aspects of self-care. If the intervention included case management, reminders to patients needed to be explicit | Reminders via postcards or telephone calls |
| 11) Patient education-written materials, videos, lectures, other |
|
Pamphlets, booklets, brochures on fall prevention, videos, classes, lectures, workshops, unspecified instructions/advice to prevent falls |
| 12) Motivational interviewing | ‘a directive and client-centred counselling style that relies upon identifying and mobilising the client’s intrinsic values and goals to stimulate behaviour change (Miller & Rollnick, 2002), thus encouraging client and family involvement in all aspects of care.’ | Restructuring misconceptions to promote a view of falls risk and fear of falling as controllable, setting realistic goals for increasing activity in a safe manner (taking personal capabilities into account) |
The knowledge users identified primary (number of injurious falls and number of fall-related hospitalisations) [15] and secondary (number of fallers, fall-related emergency department visits, fall-related physician visits, fractures, intervention-related harms, and quality of life and health utility [measured by 36-Item or 12-item Short Form Survey (SF-36 or SF-12), SF-36 physical and mental summary component measures and health utility measured by EuroQol-5D (EQ-5D)] outcomes of interest [16, 17].
Data sources
A search strategy was developed and peer-reviewed using PRESS [18] by experienced librarians, and published [11]. Subsequently, the following databases were searched from inception until April 2017: MEDLINE, EMBASE, Cochrane Central Register of Controlled Trials and Ageline. In addition, trial registries, conference abstracts, reference lists of included RCTs, and references of related systematic reviews were searched. Authors of relevant RCTs were emailed to obtain additional or unpublished data.
Study selection
The eligibility criteria and screening explanation document were calibrated on a random sample of 50 citations. After 75% agreement was achieved, pairs of reviewers screened all citations and potentially relevant full-text articles independently. Conflicts were resolved by a third reviewer.
Data abstraction
A data abstraction form was developed in Excel with an accompanying guidance document, and calibrated using a 5% random sample of included studies. After calibration, all data were independently abstracted by pairs of reviewers. To increase consistency, all discrepancies were resolved by a third reviewer. As falls prevention QI strategies are complex interventions, which are difficult to classify [19], all studies were coded independently by a knowledge user and a methodologist, using the pre-established coding guide for QI strategies.
Risk-of-bias appraisal
The Cochrane Effective Practice and Organisation of Care (EPOC) Group’s risk-of-bias tool was used to appraise the internal validity of included RCTs [20]. The tool was calibrated using a random sample of five included studies. Subsequently, each included RCT was appraised by independent pairs of reviewers. Conflicts were resolved by a third reviewer.
Data synthesis and analysis
All analyses were conducted by an experienced statistician who used the metafor and netmeta packages [21–23] in R statistical version 3.3.3 [24] software and the netfunnel command in STATA 13.0 [25–28]. Pooled odds ratios (ORs) for dichotomous data, which were converted to the absolute risk differences (ARDs) using standard formulae [29] and mean differences (MD) for continuous data were estimated using pairwise random-effects meta-analysis. RCTs reporting dichotomous outcomes with zero events across all arms were excluded from the analysis. RCTs reporting continuous outcomes with missing measures of variance (e.g. standard deviation) were included in the analysis, with standard errors imputed using established methods, if feasible [30,31]. Cluster-RCTs were adjusted using an effective sample size and intracluster correlation coefficient (ICC) for each cluster-RCT [13, 14]. The between-study variance in both meta-analysis and NMA was estimated using the DerSimonian and Laird method [32,33]. In each meta-analysis, heterogeneity was quantified using the I2- measure [34]. The mean control event rate across included studies was calculated for each outcome when possible.
For outcomes forming a connected network of falls prevention interventions, random-effects NMA was conducted using a common within-network between-study variance (τ2) across treatment comparisons. To surmount small-study effects [35], RCTs with 100 or fewer patients across all treatment arms were excluded from analysis. A comparison-adjusted funnel plot was drawn for each outcome with at least 10 studies to examine small-study effects [27] by estimating the overall treatment effect for each comparison under the fixed-effect meta-analysis model and ordering all interventions based on the potential for effectiveness and target.
In order to predict the interval within which a new RCT treatment effect would lie, a 95% predictive interval was calculated [36]. The NMA assumptions were tested a priori, first by assessing transitivity and comparing all potential effect modifiers across treatment comparisons, and then evaluating consistency between different sources of evidence in the network using the design-by-treatment model [37]. Several effect modifiers were considered (see Appendix A in the supplementary data, available at Age and Ageing online). If results suggested global inconsistency, then local inconsistency was examined within each loop in a network separately using the loop-specific method [38,39]. If results suggested local inconsistency or important heterogeneity, or over 10 RCTs were available, and the number of RCTs was larger than the number of the included interventions, a subgroup NMA was conducted considering the aforementioned effect modifiers. Sensitivity analyses were conducted on the risk of bias results for the primary outcomes. All interventions were ranked using P-scores measuring the mean extent of certainty that a treatment is better when compared to the remaining treatments in the network [40]. P-scores were depicted across all outcomes in a rank-heat plot (http://rh.ktss.ca/) [41]. Both meta-analysis and NMA treatment effect estimates were presented along with 95% confidence intervals (CI).
Results
Study selection
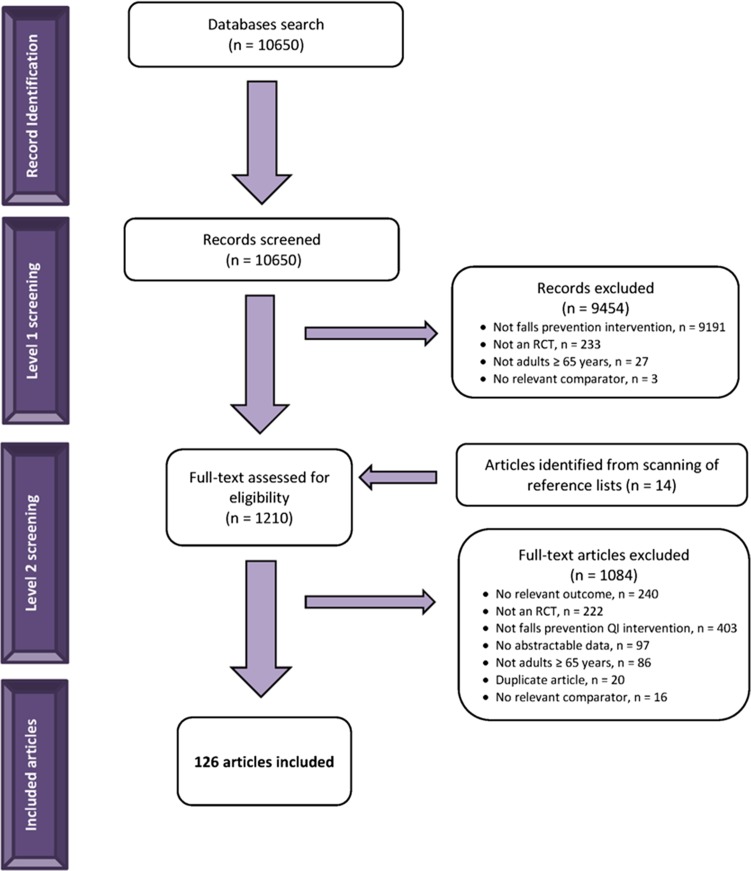
A total of 10,650 citations and 1,210 full-text articles were screened (Figure 1). Of these, 126 RCTs (84,307 participants) involved a relevant QI strategy with a relevant comparator were included (Figure 1, see Appendix B in the supplementary data, available at Age and Ageing online). None of the included studies were written in languages other than English; one paper was a dissertation [42].
Figure 1.
Study flow.
Study and participant characteristics
The mean of ages of participants across studies ranged between 65 and 88 years across the 126 included RCTs (Table 1, see Appendix C in the supplementary data, available at Age and Ageing online). Most RCTs included a high proportion (over 50%) of female participants (89%) (Table 1, see Appendix C in the supplementary data, available at Age and Ageing online), and included a mixture of participants with and without a previous history of falling (66%). The number of medications used by participants was not reported in most RCTs (59%).
Table 1.
Summary of patient and study characteristics
| Patient characteristics | ||
|---|---|---|
| Number of RCTs | % of RCTs | |
| (n = 126) | ||
| Age (mean) | ||
| 65–74.99 years | 30 | 23.8 |
| 75–84.99 years | 73 | 57.9 |
| ≥85 years | 9 | 7.1 |
| NR | 14 | 11.1 |
| Female (%) | ||
| 0–49.99% | 9 | 7.1 |
| 50–69.99% | 42 | 33.3 |
| 70–89.99% | 54 | 42.9 |
| 90–100% | 16 | 12.7 |
| NR | 5 | 4.0 |
| History of falls | ||
| All have history of falls | 30 | 23.8 |
| Mixed history of falls | 83 | 65.9 |
| NR | 13 | 10.3 |
| Medications takena | ||
| 0–4 medications | 25 | 19.8 |
| ≥5 medications | 27 | 21.4 |
| NR | 74 | 58.7 |
| Study characteristics | ||
| Year of publication | ||
| 1992–96 | 4 | 3.2 |
| 1997–2001 | 9 | 7.1 |
| 2002–6 | 23 | 18.3 |
| 2007–11 | 43 | 34.1 |
| 2012–17 | 47 | 37.3 |
| Continent | ||
| Europe | 49 | 38.9 |
| Australia/New Zealand | 41 | 32.5 |
| North America | 19 | 15.1 |
| Asia | 16 | 12.7 |
| Multi | 1 | 0.8 |
| Study design | ||
| Parallel RCT | 103 | 81.7 |
| Cluster RCT | 23 | 18.3 |
| Site | ||
| Multi-centre | 83 | 65.9 |
| Single centre | 43 | 34.1 |
| Settingsb | ||
| Home | 72 | 57.1 |
| Clinic | 35 | 27.8 |
| Community | 34 | 27.0 |
| Hospital | 19 | 15.1 |
| Chronic care | 20 | 15.9 |
| Retirement home | 10 | 7.9 |
| NR | 2 | 1.6 |
| Sample Size | ||
| Patient RCTs | ||
| 22–99 | 20 | 19.4 |
| 100–199 | 25 | 24.3 |
| 200–299 | 15 | 14.6 |
| 300–999 | 37 | 35.9 |
| 1,000–5,310 | 6 | 5.8 |
| Cluster RCTs | ||
| 80–499 | 8 | 34.8 |
| 500–999 | 7 | 30.4 |
| 1,000–3,999 | 5 | 21.7 |
| 4,000–10,558 | 3 | 13.0 |
| Study duration | ||
| <1–24 weeks | 53 | 42.1 |
| 25–50 weeks | 22 | 17.5 |
| 51–75 weeks | 31 | 24.6 |
| 76–208 weeks | 9 | 7.1 |
| NR | 11 | 8.7 |
NR, not reported; RCT, randomised controlled trial.
aMean or median.
bTotal numbers are >110 and >100% due to studies reporting multiple settings.
The majority of the RCTs were published in 2004 or later (77%), and conducted mainly in Europe (39%) and Australia/New Zealand (33%) (Table 1, see Appendix D in the supplementary data, available at Age and Ageing online). The studies were most frequently conducted in a home setting (57%), clinic (28%), or community (27%), with a lower proportion occurring in a hospital (15%), chronic care (16%), or retirement home (8%) setting. The number of included participants ranged from 22 to 5,310 for patient RCTs, and 80 to 10,558 for cluster-RCTs (3–842 clusters).
Risk of bias
The majority of the included RCTs were assessed as having a low risk of bias for random sequence generation (69% low; 30% unclear; 1% high), allocation concealment (58% low; 40% unclear; 2% high), similar baseline outcome measures (82% low; 13% unclear; 6% high), similar baseline characteristics (80% low; 10% unclear; 10% high), incomplete outcome data (77% low; 8% unclear; 15% high), blinding (93% low; 3% unclear; 4% high), and other bias (69% low; 22% unclear; 9% high) (see Appendices E and F in the supplementary data, available at Age and Ageing online). However, the majority of the RCTs were assessed as having an unclear risk of bias for contamination (58% unclear; 40% low; 2% high) and selective outcome reporting (56% unclear; 41% low; 2% high) (see Appendices E and F in the supplementary data, available at Age and Ageing online). The comparison-adjusted funnel plots suggested no evidence of publication bias or small-study effects (see Appendix G in the supplementary data, available at Age and Ageing online).
Outcome results
Twenty-two studies included less than 100 patients and were excluded from the analysis [43–64]. All pairwise meta-analysis results can be found in Appendix H in the supplementary data, available at Age and Ageing online. Across all NMAs, no evidence of significant inconsistency was indicated (see Appendix I in the supplementary data, available at Age and Ageing online). The overall NMA results for each outcome, with a focus on the QI strategy effects relative to usual care are presented below. The results from all significant treatment comparisons can be found in Appendix I in the supplementary data, available at Age and Ageing online. Subgroup and sensitivity analyses are summarised in Table 2, and all the P-scores from the overall NMAs, subgroup, and sensitivity analyses can be found in Appendices J and K in the supplementary data, available at Age and Ageing online. The rank-heat plot is presented in Appendix L in the supplementary data, available at Age and Ageing online.
Table 2.
Statistically significant additional analysis results for the outcome of number of fallers
| Intervention (versus usual care) | OR (95% CI) | Direction of result |
|---|---|---|
| Subgroup: follow-up <12 months (24 studies, 14,135 patients) | ||
| Staff education | 2.33 (1.06–5.26) | Increased risk |
| Subgroup: follow-up >12 months (36 studies, 22,692 patients) | ||
| Patient education | 0.73 (0.54–0.98) | Reduced risk |
| Patient education + self-management | 0.50 (0.26–0.99) | Reduced risk |
| Patient education + staff education | 0.59 (0.40–0.88) | Reduced risk |
| Patient education + team changes | 0.59 (0.40–0.86) | Reduced risk |
| Case management + patient reminders + staff education | 0.18 (0.07–0.45) | Reduced risk |
| Case management + motivational interviewing + patient education + team changes | 0.33 (0.12–0.88) | Reduced risk |
| Subgroup: age <80 years (40 studies, 26,785 patients) | ||
| Staff education | 3.23 (1.03–10.00) | Increased risk |
| Subgroup: age ≥80 years (19 studies, 9,368 patients) | ||
| Patient education + self-management | 0.57 (0.33–0.96) | Reduced risk |
| Patient education + staff education | 0.61 (0.38–0.99) | Reduced risk |
| Patient education + team changes | 0.48 (0.24–0.96) | Reduced risk |
| Patient education + self-management + staff education | 0.44 (0.21–0.95) | Reduced risk |
| Case management + patient reminders + staff education | 0.18 (0.07–0.44) | Reduced risk |
| Subgroup: female <75% (37 studies, 24,658 patients) | ||
| Case management + patient reminders | 0.36 (0.14–0.92) | Reduced risk |
| Subgroup: female >75% (21 studies, 11,375 patients) | ||
| Patient education + team changes | 0.24 (0.07–0.87) | Reduced risk |
| Case management + patient reminders + staff education | 0.18 (0.06–0.56) | Reduced risk |
| Subgroup: Medication use ≥5 (15 studies, 8,634 patients) | ||
| Patient education | 0.18 (0.05–0.69) | Reduced risk |
| Patient education + team changes | 0.24 (0.07–0.81) | Reduced risk |
| Subgroup: medication use <5 (15 studies, 7,950 patients) | ||
| Patient education | 2.00 (1.03–4.00) | Increased risk |
| Case management + patient reminders + staff education | 0.18 (0.07–0.48) | Reduced risk |
| Subgroup: history of falling − all fallers (12 studies, 3,709 patients) | ||
| No significant comparisons | – | – |
| Subgroup: mixed history of falling (40 studies, 33,959 patients) | ||
| Case management + patient reminders + staff education | 0.18 (0.07–0.43) | Reduced risk |
| Subgroup: home setting (24 studies, 15,418 patients) | ||
| Case management + patient reminders + staff education | 0.18 (0.08–0.43) | Reduced risk |
| Sensitivity analysis: low risk of contamination (29 studies, 24,967 patients) | ||
| Case management + patient reminders + staff education | 0.18 (0.06–0.53) | Reduced risk |
| Case management + motivational interviewing + patient education + team changes | 0.28 (0.08–0.97) | Reduced risk |
| Sensitivity analysis: low risk of selective outcome reporting (24 studies, 14,564 patients) | ||
| Case management + patient reminders | 0.36 (0.14–0.91) | Reduced risk |
Injurious falls
NMA for number of injurious falls included 29 RCTs (26,326 patients), with a total of 18 QI strategies examined (see Appendix M Panel A in the supplementary data, available at Age and Ageing online). The average event rate for the usual care group was 31.79%. Across all 171 comparisons, 3 (1.75%) were statistically significant (see Appendix I in the supplementary data, available at Age and Ageing online). The QI intervention of team changes was statistically superior in reducing the risk of injurious falls relative to usual care (OR, 0.57 [95% CI, 0.33–0.99]; ARD −0.11 [95% CI, −0.18 to −0.002]). All studies of team changes were in mixed settings. An additional 12 single or multifactorial interventions were also superior to usual care, but the differences did not reach statistical significance. While five interventions were inferior (i.e. associated with increased falls) to usual care, none of these differences were statistically significant (see Appendix I in the supplementary data, available at Age and Ageing online). Subgroup and sensitivity analyses can be found in Appendices A and J in the supplementary data, available at Age and Ageing online.
Fall-related hospitalisations
For falls-related hospitalisations, eight RCTs (4,887 patients) with 10 treatments were included. NMA was not possible because of network disconnectivity. No pairwise meta-analysis was possible across all pairwise comparisons (see Appendix H in the supplementary data, available at Age and Ageing online).
Number of fallers
NMA for the number of fallers included 61 RCTs (40 128 patients), examining 35 QI interventions (see Appendix M Panel B in the supplementary data, available at Age and Ageing online). The total event rate for the usual care group was 39.50%. Across all 595 comparisons, 27 (4.5%) were statistically significant (see Appendix I in the supplementary data, available at Age and Ageing online). Combined case management, patient reminders and staff education were statistically superior in reducing the number of fallers relative to usual care (OR, 0.18 [95% CI, 0.07–0.47]; ARD −0.29 [95% CI, −0.35 to −0.16]). Combined case management and patient reminders were also statistically superior to usual care in reducing the number of fallers (OR, 0.36 [95% CI, 0.13–0.97]; ARD −0.20 [95% CI, −0.32 to −0.007]). An additional 28 single or multifactorial interventions were superior to usual care, but the differences were not statistically significant. While four interventions were inferior to usual care, none were statistically significant. Subgroup and sensitivity analyses are summarised in Table 2 and Appendices A and J in the supplementary data, available at Age and Ageing online.
Emergency department visits
For number of emergency department visits, four RCTs (1,280 patients) with five treatments were included. Pairwise meta-analysis was not possible across any comparisons (see Appendix H in the supplementary data, available at Age and Ageing online).
Outpatient physician visits
For outpatient physician visits, nine RCTs (6,856 patients) with 11 treatments were included. NMA was not possible due to network disconnectivity. Only one pairwise meta-analysis was possible (see Appendix H in the supplementary data, available at Age and Ageing online). There was no statistically significant difference between combined patient education and team changes compared to patient education alone (OR, 1.14 [95% CI, 0.82–1.59]).
Fractures
NMA for the number of fractures included 23 RCTs (38 497 patients), which examined 17 different QI strategies (see Appendix M Panel C in the supplementary data, available at Age and Ageing online). The total event rate for the usual care group was 6.84%. Across all 153 comparisons, one was statistically significant (see Appendix I in the supplementary data, available at Age and Ageing online). Patient education was statistically superior compared to usual care in reducing the number of fractures (OR, 0.81 [95% CI, 0.68–0.97]; ARD −0.012 [95% CI, −0.021 to −0.0019]). Eleven interventions appeared superior to usual care and five were inferior to usual care, but these differences were not statistically significant.
Hip fractures
Eleven RCTs (15 883 participants) with 11 treatments were included for the outcome of hip fractures. Across all comparisons, pairwise meta-analyses were not possible (see Appendix H in the supplementary data, available at Age and Ageing online).
Harms
Twenty of the included RCTs reported no intervention-related harmful events in all study arms, and 13 RCTs reported at least one harm event (see Appendix N in the supplementary data, available at Age and Ageing online). Pairwise meta-analysis was not possible across any comparison (see Appendix H in the supplementary data, available at Age and Ageing online).
Quality of life
Ten RCTs reported on quality of life using the SF-12 or SF-36 physical and mental summary component measures or EuroQol-5D. One pairwise meta-analysis was possible across all comparisons (see Appendix H in the supplementary data, available at Age and Ageing online). There was no statistically significant difference between team changes compared to usual care (OR, −4.12 [95% CI, −8.87 to 0.63]).
Discussion
Team changes, which refers to modifying the structure of the primary health care team (see Box 1), may reduce risk of injurious falls, and patient education, which includes provision of health-related educational materials to patients, may reduce risk of fractures. Furthermore, a combination of case management (any system for coordinating diagnosis, treatment, or routine management of patients by a person or multidisciplinary team in collaboration with, or supplementary to, the primary-care clinician), patient reminders, and staff education, as well as a combination of case management and patient reminders may reduce falls. These results can be used to tailor treatment to decision-maker preferences for outcome and availability of resources. For example, if a clinic has many patients over 80 years of age and available resources, clinicians can consider the following interventions for reducing falls: combined patient education and self-management, combined patient education and staff education, combined patient education and team changes, combined patient education, self-management, staff education, and combined case management, patient reminders, and staff education (Table 2). The longest term studies (i.e. follow-up ≥12 months) suggested that patient education, combined patient education and self-management, combined patient education and staff education, combined patient education and team changes, combined case management, patient reminders, staff education, as well as combined case management and motivational interviewing and patient education and team changes are likely effective for reducing the risk of falling. While staff education appeared to increase the number of falls in patients below the age of 80 years, and those with shorter follow-up (<12 months), this finding is not unexpected (Table 2). Educational interventions about fall prevention targeted at staff typically result in greater awareness, and can subsequently lead to increased reporting of fall-related events. Overall, QI interventions should target patients and the team (rather than clinicians alone) to have impact on falls, injurious falls, and fractures, and simple interventions are unlikely to be effective.
There are numerous strengths to this systematic review. The methods conform to the Cochrane Handbook for Systematic Reviews [65] and the International Society for Pharmacoeconomics and Outcomes Research (ISPOR) guidance on NMA validity [66]. In addition, studies written in languages other than English were sought (although none were identified), as well as unpublished studies. The use of RCTs to evaluate the effectiveness of QI interventions produces generalisable knowledge which can be applied to a variety of settings, rather than the localised knowledge produced by a PDSA cycle. However, there are limitations. The results are only as valid as the included RCTs, and although most contained a low risk of bias, contamination and outcome reporting bias were frequently unclear. In addition, several outcomes of interest lacked sufficient data to conduct analysis. Furthermore, an intervention coding scheme adapted from the Agency for Healthcare Research and Quality was used, however other taxonomies [67,68] may have led to different results. Coding the QI strategies was complex, and definitions of these varied across studies. For example, case management may include different personnel with varying levels of skills across studies, contributing to heterogeneity. As well, some analyses (subgroup and sensitivity) were not possible due to limited data. Some additional analyses for the number of fallers found that the QI strategies may increase falls. Further exploration of the effects of combinations of falls prevention interventions and different QI interventions should be explored to identify optimal combinations of these complex interventions. This issue highlights a challenge in clinical practice whereby patients require multiple interventions and little is known about how these interventions interact.
Future RCTs can be designed to examine outcomes, such as outpatient physician visits, harms and quality of life. In addition, the results can be extended to a cost-effectiveness analysis, which is currently being planned by members of the team, and will provide important information to decision-makers considering implementation of these approaches. Finally, a different model can be used to explore our results further, such as the additive model [69], which is currently being conducted by the coauthors.
Conclusions
Team changes may reduce risk of injurious falls, patient education may reduce the risk of fractures, and a combination of case management, patient reminders, and staff education, as well as case management and patient reminders combined may reduce risk of falls. These results can be tailored according to decision-maker preferences and availability of resources.
Supplementary Material
Supplementary data mentioned in the text are available to subscribers in Age and Ageing online.
Ethics Approval: Not required.
Consent for Publication: Not applicable.
Availability of Data and Materials: The full dataset is available from the corresponding author upon reasonable request.
Declaration of Conflicts of Interests: RR owns stock in GlaxoSmithKline and provides consulting services to a contract research organisation that provides services to various pharmaceutical companies, which are not funding this project. The methodology used in this study, such as screening, selection and abstraction conducted independently by two separate reviewers, was designed to manage any potential conflict of interest.
Declaration of Sources of Funding: The research is funded by a Canadian Institutes of Health research and Knowledge Synthesis Grant (KRS289648).
References
- Note: The first 30 cited references in this manuscript are listed below. For the remaining references, please refer to Supplement 2: Appendix O in the supplementary data, available at Age and Ageing online.
- 1. Parry SW, Deary V, Finch T et al. The STRIDE (Strategies to Increase confidence, InDependence and Energy) study: cognitive behavioural therapy-based intervention to reduce fear of falling in older fallers living in the community—study protocol for a randomised controlled trial. Trials 2014; 15: 210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Ray WA, Taylor JA, Brown AK et al. Prevention of fall-related injuries in long-term care: a randomized controlled trial of staff education. Arch Intern Med 2005; 165: 2293–8. [DOI] [PubMed] [Google Scholar]
- 3. Dykes PC, Carroll DL, Hurley A et al. Fall prevention in acute care hospitals: a randomized trial. JAMA 2010; 304: 1912–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Cumming RG, Sherrington C, Lord SR et al. Cluster randomised trial of a targeted multifactorial intervention to prevent falls among older people in hospital. BMJ 2008; 336: 758–60. (Clinical research ed). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Hornbrook MC, Stevens VJ, Wingfield DJ et al. Preventing falls among community-dwelling older persons: results from a randomized trial. Gerontologist 1994; 34: 16–23. [DOI] [PubMed] [Google Scholar]
- 6. Centers for Disease Control and Prevention Important Facts about Falls. 2016. http://www.cdc.gov/homeandrecreationalsafety/falls/adultfalls.html (10 October 2018, date last accessed).
- 7.Age UK. Stop Falling: Start Saving Lives and Money. 2009. http://www.ageuk.org.uk/documents/en-gb/campaigns/stop_falling_report_web.pdf?dtrk=true (10 October 2018, date last accessed).
- 8. Parachute The Cost of Injury in Canada Report. 2015. http://www.parachutecanada.org/costofinjury (10 October 2018, date last accessed).
- 9. Tricco AC, Thomas SM, Veroniki AA et al. Comparisons of interventions for preventing falls in older adults: a systematic review and meta-analysis. JAMA 2017; 318: 1687–99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Moher D, Shamseer L, Clarke M et al. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst Rev 2015; 4: 1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Tricco AC, Cogo E, Holroyd-Leduc J et al. Efficacy of falls prevention interventions: protocol for a systematic review and network meta-analysis. Syst Rev 2013; 2: 38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Hutton B, Salanti G, Caldwell DM et al. The PRISMA extension statement for reporting of systematic reviews incorporating network meta-analyses of health care interventions: checklist and explanations. Ann Intern Med 2015; 162: 777–84. [DOI] [PubMed] [Google Scholar]
- 13. Tricco AC, Ivers NM, Grimshaw JM et al. Effectiveness of quality improvement strategies on the management of diabetes: a systematic review and meta-analysis. Lancet 2012; 379: 2252–61. [DOI] [PubMed] [Google Scholar]
- 14. Shojania KG, Ranji SR, McDonald KM et al. Effects of quality improvement strategies for type 2 diabetes on glycemic control: a meta-regression analysis. JAMA 2006; 296: 427–40. [DOI] [PubMed] [Google Scholar]
- 15. Lamb SE, Jorstad-Stein EC, Hauer K, Becker C. Development of a common outcome data set for fall injury prevention trials: the Prevention of Falls Network Europe consensus. J Am Geriatr Soc 2005; 53: 1618–22. [DOI] [PubMed] [Google Scholar]
- 16. Ware J Jr, Kosinski M, Keller SD. A 12-Item Short-Form Health Survey: construction of scales and preliminary tests of reliability and validity. Med Care 1996; 34: 220–33. [DOI] [PubMed] [Google Scholar]
- 17. Ware J, Kosinski M, Dewey J How to Score Version Two of the SF-36 Health Survey. Lincoln, RI: QualityMetric; 2000. EuroQol Research Foundation. EQ-5D health questionnaire. http://www.euroqol.org/ (10 October 2018, date last accessed).
- 18. McGowan J, Sampson M, Salzwedel DM et al. PRESS Peer Review of Electronic Search Strategies: 2015 Guideline Statement. J Clin Epidemiol 2016; 75: 40–6. [DOI] [PubMed] [Google Scholar]
- 19. Shepperd S, Lewin S, Straus S et al. Can we systematically review studies that evaluate complex interventions? PLoS Med 2009; 6: e1000086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Suggested risk of bias criteria for EPOC reviews. https://epoc.cochrane.org/sites/epoc.cochrane.org/files/public/uploads/Resources-for-authors2017/suggested_risk_of_bias_criteria_for_epoc_reviews.pdf (10 October 2018, date last accessed).
- 21. Rücker G, Schwarzer G, Krahn U, König J netmeta: Network Meta-Analysis using Frequentist Methods. 2017. https://github.com/guido-s/netmeta (10 October 2018, date last accessed).
- 22. Schwarzer G, Carpenter J, Rücker G Meta-Analysis with R. 2018; http://www.imbi.uni-freiburg.de/lehre/lehrbuecher/meta-analysis-with-r (10 October 2018, date last accessed).
- 23. Viechtbauer W. Conducting Meta-Analyses in R with the metafor Package. 2010. 2010 2010-08-05;36(3):48.
- 24. Team RC R: A Language and Environment for Statistical Computing. Vienna, Austria: R Foundation for Statistical Computing, 2017. [Google Scholar]
- 25. R Core Team R: A Language and Environment for Statistical Computing. Vienna, Austria: R Foundation for Statistical Computing, 2016. [Google Scholar]
- 26. Lunn D, Spiegelhalter D, Thomas A, Best N. The BUGS project: evolution, critique and future directions. Stat Med 2009; 28: 3049–67. [DOI] [PubMed] [Google Scholar]
- 27. Chaimani A, Higgins JP, Mavridis D, Spyridonos P, Salanti G. Graphical tools for network meta-analysis in STATA. PLoS One 2013; 8: e76654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Stata Statistical Software : Release 13. College Station, TX: StataCorp LP.; 2013.
- 29. Wang Z. Converting odds ratio to relative risk in cohort studies with partial data information. 2013. 2013 2013-10-22;55(5):11.
- 30. Furukawa TA, Barbui C, Cipriani A, Brambilla P, Watanabe N. Imputing missing standard deviations in meta-analyses can provide accurate results. J Clin Epidemiol 2006; 59: 7–10. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.