Abstract
Background:
The SEER program recently released data on brain metastases (BM) diagnosed during primary cancer staging workup (“synchronous” BM, or SBM); this study examines the incidence of SBM compared to that of lifetime BM (LBM) identified using Medicare claims for patients diagnosed with lung cancer, breast cancer, or melanoma.
Methods:
Incidence proportions (IP) and age-adjusted rates for each of SEER SBM and Medicare LBM are presented along with measures of concordance between the two sources of data, where Medicare LBM were defined by several combinations of diagnosis and putative diagnostic imaging procedure codes.
Results:
The SBM IP in lung, breast, and melanoma cancers were 9.6%, 0.3%, and 1.1%, respectively; the corresponding LBM IP were 13.5%, 1.8%, 3.6%. The greatest SBM IP among lung cancer patients was 13.4% for non-small cell lung cancer, and among breast cancer patients was 0.7% for triple-negative breast cancer. The greatest LBM IP among lung cancers was 23.1% in small-cell lung cancer, and among breast cancers was 4.2% for cases of the Triple Negative subtype.
Conclusions:
Using a large dataset that is representative of the elderly population in the US, these analyses estimate synchronous and lifetime incidence of BM in lung cancers, breast cancers, and melanomas.
Impact:
These and other population-based estimates may be used to guide development of BM screening policy and evaluation of real-world data sources.
Keywords: breast cancer, lung cancer, melanoma, epidemiology, computational methods
Introduction
Brain metastases (BM) are associated with significant morbidity and may impart a median survival of two to fourteen months following simultaneous diagnosis of BM and various primary cancers (1). Estimates of its frequency in the US vary by orders of magnitude depending on study-specific characteristics, but some of this variation in epidemiologic measures of BM may also reflect medical advances in primary cancer control or metastasis detection. This, combined with an aging population and subsequent increase in absolute numbers of patients with brain metastases, suggests that detailed population-level evidence of BM occurrence in the elderly is more and more relevant to clinical practice and the decision to screen for cranial disease (2, 3).
In 2016, the Surveillance, Epidemiology, and End-Results (SEER) program helped address the need for such studies by releasing data on BM diagnosed during staging workup for primary cancer, called “synchronous brain metastases” (SBM) (4). Synchronous metastasis evaluation is helpful to many treatment decisions, and SEER SBM studies performed with these recently-available data contribute valuable information to the pressing need for such work (5, 6).
Metastasis following primary cancer staging may also be studied using Medicare claims, which offer long-term evidence of patient characteristics and may be linked to SEER data. The SEER-Medicare linkage further allows SEER data to serve as a referent to compare with Medicare claims, the classification performance of which may inform Medicare claims-based estimates of lifetime brain metastases (LBM).
The present work examines cases of lung cancer, breast cancer, and melanoma due to their increased incidence of BM (7). Motivated by the availability of new data and a need for estimates over the patient’s lifetime, our aim was twofold: to (1) evaluate Medicare claims BM identification algorithms against SEER SBM data, and (2) report incidences of SEER SBM and Medicare claims-identified LBM for lung cancers, breast cancers, and melanoma.
Materials and Methods
This study was approved as exempt of review by the University Hospitals Cleveland Medical Center Institutional Review Board, and assigned the study number “EM-17–05.”
Data description
The National Cancer Institute SEER program offers cancer registry data covering approximately 28% of the US population; these data may be linked to Medicare claims data to further investigate patient characteristics. Because Medicare is the primary insurer for the vast majority of patients age 65 years or older, the results of SEER-Medicare studies are highly generalizable to the elderly population (8).
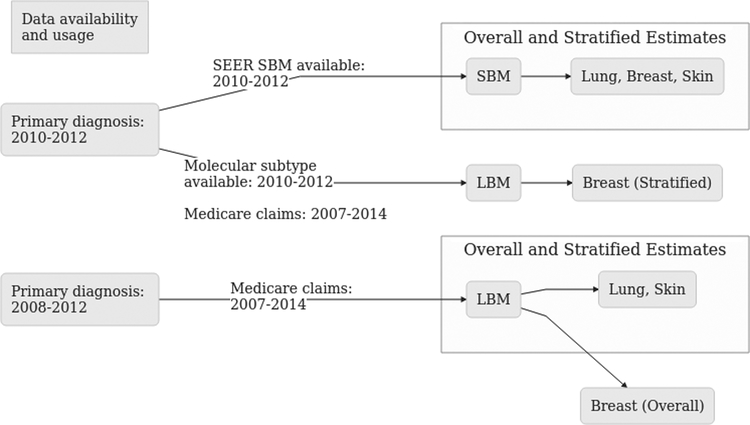
Data for primary cancers diagnosed in the years 2008 through 2012 were linked to Medicare data from 2007 through 2014. SEER SBM data are only available for the year 2010 and onwards, therefore only those years were used to compare Medicare claims identification algorithms. This yielded a total of three diagnosis years for SEER SBM comparison to Medicare identification algorithms and five diagnosis years for Medicare LBM estimates (Figure 1).
Figure 1: Data sources and definitions.
Figure 1 shows the years available for each of synchronous and lifetime brain metastasis data sources. Lifetime brain metastasis includes both synchronous brain metastasis diagnosis and cases diagnosed thereafter, whereas synchronous brain metastasis are those diagnosed during staging workup. Data source restrictions led to the use of cases diagnosed in 2010–2012 for estimates of synchronous brain metastasis, but 2008 – 2012 for lifetime estimates.
Four types of claims files offered as part of SEER-Medicare were used: Part A inpatient, carrier, outpatient, and durable medical equipment files. Each record in these files contains a date of service, International Classification of Diseases, ninth revision, Clinical Modification (ICD-9-CM) diagnosis codes, and Current Procedural Terminology (CPT) procedure codes.
Identifying patients with brain metastases
The 2010 – 2013 SEER Collaborative Staging brain metastases variable uses clinical or pathologic evidence from staging workup (but not thereafter) to identify “distant metastatic involvement of the brain at the time of diagnosis…This includes only the brain, not spinal cord or other parts of the central nervous system (CNS) (9).” In contrast, the closest ICD-9-CM code (198.3X) refers to metastases to any part of the CNS and is not specific to the brain. Therefore, Medicare LBM was defined as the presence of diagnosis codes for secondary cancer of the CNS (ICD-9 code: 198.3X) and procedure codes for a brain or head imaging study (CPT codes 70450 – 70470, 70551 – 70553, 78607 – 78608) each within 60 days of the other, at any time throughout Medicare claims data.
To inform the reliability of Medicare LBM estimates, classification performance metrics (Cohen’s Kappa, predicted and true counts, sensitivity, specificity, and positive predictive value) are presented for both the Medicare LBM algorithm and a Medicare SBM algorithm based on the same codes with the additional requirement that codes occurred within 60 days of primary cancer diagnosis. This duration of 60 days was chosen as an approximation of the time required to clinically establish extent of disease, after which registry data regarding metastases are not updated (4). Classification performance measures for a modified Medicare LBM algorithm that relies only on diagnosis codes are also provided for comparison.
Study Population
The use of Medicare data has several implications for population selection. Exclusion criteria included age less than 65 years, Medicare qualification not due to age, lack of insurance or unknown insurance status, any record of Healthcare Management Organization (HMO) use, or presence of other primary cancer diagnosis in a different site. Before exclusion, there were 198,730 lung, 208,909 breast, and 98,299 melanoma cases. After exclusion, 120,405 lung, 110,983 breast, and 35,268 melanoma cases remained (Supplementary Figure 1). In the cohort of patients diagnosed in 2010 through 2012, there were 70,974 lung, 67,096 breast, and 21,860 melanoma cases.
Study Variables
Age and race were described across BM status. Age was summarized both as a continuous and categorical variable, where categories were age groups 65 – 69, 70 – 79, 80 – 89, and 90+ years. Race was categorized as Asian/Pacific Islander, African American, Native American, White, or other. “White” was further categorized as “White Non-Hispanic” (WNH) or “White Hispanic” (WH) using the North American Association of Central Cancer Registries’ Hispanic Identification Algorithm. For breast cancers, Native American and Asian/Pacific Islander race were collapsed into an “other” category; for melanoma, Native American/American Indian, Black/African American, and Asian race categories were collapsed.
Histology was categorized according to the International Classification of Diseases, Oncology, 3, (ICD-O-3) (10) (https://seer.cancer.gov/icd-o-3/sitetype.icdo3.20180117.pdf). The five most frequent histology labels among each cancer site were identified, and less frequent labels were considered “other” categories. For lung cancer, the most frequent histology was adenocarcinoma (8140–8141, 8144), followed by squamous cell carcinoma (8070–8075, 8078), small cell carcinoma (SCLC, 8041–8045), and non-small cell carcinoma (NSCLC, 8046), and carcinoma (8010–8014). Among breast cancers, the most frequent histology was duct carcinoma (8500–8504, 8507), followed by lobular and other ductal carcinoma (8520–8525), mucinous adenocarcinoma (8480–8481), and adenoid cystic & cribriform carcinoma (8200–8201). Melanoma histology was categorized as nevi & melanomas (8720–8723) and malignant melanoma in junctional nevus (8740–8745) due to a lack of observations of other histology groups.
The derived SEER Summary Stage 2000 data element was used to classify extent of disease at primary diagnosis as in situ, localized, regional with direct extension, regional with extension to lymph nodes only, regional with direct and lymph node extension, regional not otherwise specified (NOS), distant, or unknown.
Breast cancer subtypes were categorized using the 2010+ breast subtype SEER data element, which is a combination of estrogen receptor (ER) status, progesterone receptor (PR) status, and HER2 (also called CD340 or neu) values. It has six categories: Her2+/HR+, Her2+/HR-, Her2-/HR+, Triple Negative, unknown, or not 2010+ Breast, where HR represents ER and PR status. For measures of incidence, categories were consolidated into Her2+, Her2-/HR+, Triple Negative, and unknown.
For privacy and confidentiality purposes, statistics are suppressed or levels collapsed for categorical values representing data from 1 to 11 subjects (11).
Analysis
Concordance was measured using Cohen’s Kappa. Sensitivity, specificity, and positive predictive value (PPV) are presented for further detail, along with predicted and true counts of BM cases.
Incidence proportion (IP) was defined as the ratio of brain metastases case counts to the total number of cases, presented for each primary cancer and its associated strata. Strata were histological for lung cancer and melanoma, and based on molecular subtype for breast cancers. Because molecular subtype was only available from cases in 2010 onwards, stratified estimates of Medicare LBM were restricted to the 2010–2012 diagnosis years (Figure 1). Additionally, analysis included estimates of the IP of primary cancer later associated with Medicare LBM diagnosed in 2008 through 2012, stratified by SEER Summary Stage 2000. Descriptive statistics of demographic and clinical characteristics are presented for the overall population, patients diagnosed with SEER SBM, and patients classified as having Medicare LBM.
The average annual age-adjusted incidence rate (AAIR) was calculated as an age-weighted sum over each year using census values from 2010 for each five-year-interval of ages starting at sixty-five and combining ages 90 or greater into a single interval (12). Crude incidence rates are presented for breast cancer strata where fewer than 11 cases per annum were observed.
Reproducibility
This study was conducted with the goal of providing open, reproducible, and replicable results (13). Analyses reported here were performed using R version 3.4.3 (2017–11–30) and managed using GNU make (14, 15); reproducibility materials are available online at http://doi.org/10.5281/zenodo.1336604 (16), and two authors independently used these materials to arrive at the same conclusions presented herein.
Results
There were 6,789 cases of SBM among lung cancer subjects, 203 among breast cancers, and 230 melanoma SBM cases using the SEER gold standard data, while 5,100, 196, and 152 subjects were found to have Medicare SBM, respectively.
Using the Medicare claims algorithm intended to identify SBM based on both diagnosis and cranial imaging codes, Kappa concordance with SEER-reported SBM was found to be 0.63 (95% CI: 0.62 – 0.64) for lung cancer, 0.46 (95% CI: 0.40 – 0.53) for breast cancers, and 0.66 (95% CI: 0.60 – 0.71) for melanomas (Table 1).
Table 1:
Medicare claims brain metastasis classification accuracy
| Algorithm: Synchronous | |||||||
| Lung | Overall | 5,100 | 6,789 | 0.58 | 0.77 | 0.98 | 0.63 (0.62 – 0.64) |
| Adenocarcinoma | 2,103 | 2,806 | 0.59 | 0.79 | 0.98 | 0.63 (0.62 – 0.65) | |
| Carcinoma | 403 | 571 | 0.57 | 0.80 | 0.98 | 0.62 (0.59 – 0.66) | |
| NSCLC | 506 | 745 | 0.56 | 0.82 | 0.98 | 0.62 (0.58 – 0.65) | |
| Other | 572 | 784 | 0.58 | 0.80 | 0.99 | 0.65 (0.62 – 0.68) | |
| SCLC | 872 | 1,144 | 0.59 | 0.78 | 0.97 | 0.62 (0.60 – 0.65) | |
| Squamous | 644 | 739 | 0.57 | 0.66 | 0.99 | 0.60 (0.56 – 0.63) | |
| Breast | Overall | 196 | 203 | 0.46 | 0.47 | 1.00 | 0.46 (0.40 – 0.53) |
| Her2−/Hr+ | 103 | 86 | 0.41 | 0.34 | 1.00 | 0.37 (0.28 – 0.46) | |
| Her2+/ Hr(+/−) | 27 | 31 | 0.48 | 0.56 | 1.00 | 0.51 (0.36 – 0.67) | |
| Other | 38 | 50 | 0.44 | 0.58 | 1.00 | 0.50 (0.37 – 0.63) | |
| Triple (−) | 28 | 36 | 0.58 | 0.75 | 1.00 | 0.65 (0.52 – 0.79) | |
| Skin | Overall | 152 | 230 | 0.55 | 0.83 | 1.00 | 0.66 (0.60 – 0.71) |
| MMJN | * | * | 0.43 | 0.60 | 1.00 | 0.50 (0.15 – 0.85) | |
| Nevi & Melanomas | 134 | 206 | 0.54 | 0.83 | 1.00 | 0.65 (0.59 – 0.71) | |
| Other | 13 | * | 0.71 | 0.92 | 1.00 | 0.79 (0.64 – 0.95) | |
| Algorithm: Lifetime | |||||||
| Lung | Overall | 9,138 | 6,789 | 0.65 | 0.48 | 0.92 | 0.49 (0.48 – 0.50) |
| Adenocarcinoma | 3,630 | 2,806 | 0.65 | 0.50 | 0.91 | 0.50 (0.48 – 0.51) | |
| Carcinoma | 575 | 571 | 0.63 | 0.62 | 0.95 | 0.57 (0.53 – 0.61) | |
| NSCLC | 804 | 745 | 0.61 | 0.57 | 0.92 | 0.52 (0.49 – 0.55) | |
| Other | 1,028 | 784 | 0.65 | 0.50 | 0.95 | 0.52 (0.49 – 0.55) | |
| SCLC | 1,863 | 1,144 | 0.67 | 0.41 | 0.84 | 0.40 (0.38 – 0.43) | |
| Squamous | 1,238 | 739 | 0.64 | 0.38 | 0.95 | 0.45 (0.42 – 0.48) | |
| Breast | Overall | 967 | 203 | 0.57 | 0.12 | 0.99 | 0.19 (0.16 – 0.22) |
| Her2−/Hr+ | 430 | 86 | 0.53 | 0.11 | 0.99 | 0.18 (0.13 – 0.22) | |
| Her2+/ Hr(+/−) | 181 | 31 | 0.68 | 0.12 | 0.97 | 0.19 (0.12 – 0.26) | |
| Other | 144 | 50 | 0.54 | 0.19 | 0.99 | 0.27 (0.19 – 0.36) | |
| Triple (−) | 212 | 36 | 0.58 | 0.10 | 0.96 | 0.16 (0.10 – 0.22) | |
| Skin | Overall | 658 | 230 | 0.66 | 0.23 | 0.98 | 0.33 (0.29 – 0.37) |
| MMJN | 114 | * | 0.71 | 0.04 | 0.99 | 0.08 (0.01 – 0.15) | |
| Nevi & Melanomas | 498 | 206 | 0.65 | 0.27 | 0.97 | 0.36 (0.32 – 0.41) | |
| Other | 46 | * | 0.82 | 0.30 | 0.95 | 0.42 (0.27 – 0.57) |
For Medicare claims data algorithms identifying synchronous and lifetime brain metastases based on the presence of a diagnosis code for secondary cancer of the CNS and a procedure code indicating intracranial imaging within sixty days of the other, this table shows classification accuracy compared to a SEER SBM gold standard. The rows under “Algorithm: Synchronous” represent such an algorithm with the additional criteria that codes must have occurred within 60 days of primary cancer diagnosis, whereas “Algorithm: Lifetime” indicates that codes may have occurred at anytime in Medicare claims. “Sens” refers to sensitivity; “spec”, specificity; PPV, positive predictive value. An asterisk (“*”) represents values that are suppressed to avoid reporting data from 1 through 11 cases.
The least restrictive algorithm, relying on the presence of a diagnosis code for central nervous system metastases at any time throughout the patients’ Medicare claims history, yielded concordance of 0.73 (95% CI: 0.72–0.74) for lung cancers, 0.49 (95% CI: 0.43 – 0.54) for breast cancers, and 0.78 (95% CI: 0.73 – 0.82) for melanomas (Table 2).
Table 2:
Diagnosis code-only algorithms performance
| Algorithm: Synchronous | |||||||
| Lung | Overall | 6,445 | 6,789 | 0.74 | 0.78 | 0.98 | 0.73 (0.72 – 0.74) |
| Adenocarcinoma | 2,643 | 2,806 | 0.75 | 0.79 | 0.97 | 0.74 (0.72 – 0.75) | |
| Carcinoma | 539 | 571 | 0.75 | 0.79 | 0.97 | 0.74 (0.70 – 0.77) | |
| NSCLC | 664 | 745 | 0.73 | 0.82 | 0.97 | 0.74 (0.71 – 0.77) | |
| Other | 723 | 784 | 0.74 | 0.80 | 0.99 | 0.75 (0.73 – 0.78) | |
| SCLC | 1,079 | 1,144 | 0.74 | 0.78 | 0.97 | 0.72 (0.69 – 0.74) | |
| Squamous | 797 | 739 | 0.72 | 0.67 | 0.98 | 0.68 (0.65 – 0.71) | |
| Breast | Overall | 294 | 203 | 0.60 | 0.41 | 1.00 | 0.49 (0.43 – 0.54) |
| Her2−/Hr+ | 167 | 86 | 0.57 | 0.29 | 1.00 | 0.39 (0.31 – 0.46) | |
| Her2+/ Hr(+/−) | 34 | 31 | 0.55 | 0.50 | 1.00 | 0.52 (0.37 – 0.67) | |
| Other | 55 | 50 | 0.60 | 0.55 | 1.00 | 0.57 (0.46 – 0.68) | |
| Triple (−) | 38 | 36 | 0.69 | 0.66 | 1.00 | 0.67 (0.55 – 0.80) | |
| Skin | Overall | 197 | 230 | 0.72 | 0.84 | 1.00 | 0.78 (0.73 – 0.82) |
| MMJN | * | * | 0.43 | 0.60 | 1.00 | 0.50 (0.15 – 0.85) | |
| Nevi & Melanomas | 177 | 206 | 0.72 | 0.84 | 1.00 | 0.77 (0.73 – 0.82) | |
| Other | 15 | 17 | 0.82 | 0.93 | 1.00 | 0.87 (0.75 – 1.00) | |
| Algorithm: Lifetime | |||||||
| Lung | Overall | 12,126 | 6,789 | 0.85 | 0.48 | 0.89 | 0.55 (0.54 – 0.56) |
| Adenocarcinoma | 4,807 | 2,806 | 0.86 | 0.50 | 0.88 | 0.56 (0.55 – 0.58) | |
| Carcinoma | 780 | 571 | 0.83 | 0.61 | 0.92 | 0.65 (0.62 – 0.68) | |
| NSCLC | 1,102 | 745 | 0.84 | 0.57 | 0.89 | 0.61 (0.58 – 0.64) | |
| Other | 1,359 | 784 | 0.84 | 0.49 | 0.93 | 0.58 (0.55 – 0.60) | |
| SCLC | 2,411 | 1,144 | 0.86 | 0.41 | 0.79 | 0.45 (0.42 – 0.47) | |
| Squamous | 1,667 | 739 | 0.85 | 0.38 | 0.93 | 0.49 (0.46 – 0.51) | |
| Breast | Overall | 1,467 | 203 | 0.76 | 0.11 | 0.98 | 0.18 (0.16 – 0.21) |
| Her2−/Hr+ | 701 | 86 | 0.74 | 0.09 | 0.98 | 0.16 (0.12 – 0.19) | |
| Her2+/ Hr(+/−) | 255 | 31 | 0.81 | 0.10 | 0.96 | 0.17 (0.11 – 0.22) | |
| Other | 217 | 50 | 0.72 | 0.17 | 0.99 | 0.27 (0.20 – 0.34) | |
| Triple (−) | 294 | 36 | 0.83 | 0.10 | 0.95 | 0.17 (0.12 – 0.23) | |
| Skin | Overall | 892 | 230 | 0.87 | 0.22 | 0.97 | 0.35 (0.31 – 0.38) |
| MMJN | 150 | * | 0.86 | 0.04 | 0.98 | 0.07 (0.02 – 0.13) | |
| Nevi & Melanomas | 689 | 206 | 0.87 | 0.26 | 0.96 | 0.39 (0.35 – 0.43) | |
| Other | 53 | * | 0.88 | 0.28 | 0.93 | 0.40 (0.26 – 0.55) |
For Medicare claims data algorithms identifying synchronous and lifetime brain metastases based only on the presence of a diagnosis code for secondary cancer of the CNS, this table shows classification accuracy compared to a SEER SBM gold standard. The rows under “Algorithm: Synchronous” represent such an algorithm with the additional criteria that codes must have occurred within 60 days of primary cancer diagnosis, whereas “Algorithm: Lifetime” indicates that codes may have occurred at any time in Medicare claims. “Sens” refers to sensitivity; “spec”, specificity; PPV, positive predictive value. An asterisk (“*”) represents values that are suppressed to avoid reporting data from 1 through 11 cases. NSCLC stands for non-small-cell lung cancer; SCLC, small-cell lung cancer; MMJN, malignant melanoma in junctional nevi.
120,405 lung cancer cases diagnosed between 2008 and 2012 were selected for inclusion (Supplementary Figure 1), 70,974 of which were diagnosed in 2010 through 2012.
For cases of primary lung cancer diagnosed in 2010 through 2012, the AAIR of SBM was 9,422 per 100,000 cases (95% CI: 9,034 – 9,825) with an IP of 9.6% (Table 3). Medicare LBM was calculated for the 120,405 patients with primary diagnosis from 2008 through 2012. the LBM AAIR was 13,255 per 100,000 cases (95% CI: 12,798 – 13,727) and the IP of Medicare LBM in lung cancer overall was 13.5%.
Table 3:
Incidence of primary cancer associated with brain metastases
| AAIR | % | % | % | AAIR | % | % | ||||
| Lung | Overall | 9422 | 70974 | 9.6 | 84.5 | 5.9 | 13255 | 120405 | 13.5 | 86.5 |
| Adenocarcinoma | 11449 | 23801 | 11.8 | 83.3 | 4.9 | 15059 | 38102 | 15.5 | 84.5 | |
| Carcinoma | 12028 | 5127 | 11.1 | 77.8 | 11.0 | 12829 | 9339 | 11.9 | 88.1 | |
| NSCLC | 13336 | 5547 | 13.4 | 80.5 | 6.1 | 15011 | 11990 | 15.3 | 84.7 | |
| Other | 6704 | 11819 | 6.6 | 84.7 | 8.7 | 9610 | 19911 | 9.4 | 90.6 | |
| SCLC | 12607 | 8467 | 13.5 | 81.0 | 5.5 | 21539 | 14476 | 23.1 | 76.9 | |
| Squamous | 4427 | 16213 | 4.6 | 91.7 | 3.8 | 7920 | 26587 | 8.1 | 91.9 | |
| Breast | Overall | 309 | 67362 | 0.3 | 98.0 | 1.7 | 1790 | 110983 | 1.8 | 98.2 |
| Her2−/Hr+ | * 20.9 | 41135 | 0.2 | 98.7 | 1.1 | 1065 | 41135 | 1.1 | 98.9 | |
| Her2+/ Hr(+/−) | * 52.5 | 5900 | 0.5 | 97.8 | 1.7 | 3017 | 5900 | 3.1 | 96.9 | |
| Other | * 36.7 | 15250 | 0.4 | 96.1 | 3.6 | 1996 | 20406 | 1.7 | 98.3 | |
| Triple Negative | * 70.9 | 5077 | 0.7 | 98.0 | 1.3 | 4103 | 5077 | 4.2 | 95.8 | |
| Skin | Overall | 1049 | 21860 | 1.1 | 97.2 | 1.8 | 3583 | 35268 | 3.6 | 96.4 |
| Mal. Mel. In Junct. Nevus | ** | 7382 | ** | ** | ** | 1758 | 12053 | 1.8 | 98.2 | |
| Nevi & Melanomas | 1473 | 13862 | 1.5 | 96.3 | 2.2 | 4380 | 22041 | 4.3 | 95.7 | |
| Other | 2903 | 616 | 2.8 | 94.6 | 2.6 | 7530 | 1174 | 7.7 | 92.3 |
The first set of AAIR, total at-risk, and present/absent/missing values in this table reflects Surveillance, Epidemiology, and End-Results program synchronous brain metastases data, while the second set reflects Medicare lifetime brain metastases data. The “Present” columns reflect the incidence proportion of brain metastases associated with the cancer described in that row; “Absent”, absence of brain metastases; and N/A, missing. * indicates use of crude values rather than annual or average annual age-adjusted values, and values contained in parentheses reflect estimated 95% confidence intervals. “Her2” stands for Human Epidermal Growth Factor Receptor 2, “Hr” stands for hormone receptors, reflecting either progesterone receptor or estrogen receptor expression status. SCLC stands for small-cell lung cancer; NSCLC, non-small-cell lung cancer.
The IP of SEER SBM among adenocarcinoma lung cancer patients was 11.8%, while the corresponding Medicare LBM value was 15.5%. For patients with carcinoma histology lung cancer, the SEER SBM IP was 11.1%, compared to Medicare LBM at 11.9%. The proportions of SEER SBM and Medicare LBM among squamous cell lung cancer patients were 4.6% and 8.1%, respectively.
Patients with NSCLC had a SEER SBM IP of 13.4%, while the corresponding LBM proportion was 15.3%. Lung cancer cases with SCLC histology had an SBM IP of 13.5%, with the IP of LBM in these cases being 23.1%.
When stratified by stage at primary cancer diagnosis, the IP of Medicare LBM was 18.8% among lung cancer cases with distant metastasis; 10.5% among cases with metastasis to lymph nodes only; and 4.8% among cases that were restricted to tissue local to the disease (Supplementary Table 4).
110,983 subjects with breast cancer were selected for inclusion (Supplementary Figure 1), of whom 67,362 were diagnosed in 2010 through 2012.
The AAIR of SEER SBM was 309 per 100,000 cases (95% CI: 239– 397), and the IP of cases with SBM diagnosis was 0.3% (Table 3).
A total of 110,983 patients were diagnosed with primary breast cancer from 2008 through 2012. The Medicare LBM AAIR for primary breast cancer diagnoses in this population was 1,790 per 100,000 cases (95% CI: 1,618– 1,979), while the overall IP of primary cancer diagnosis that is associated with Medicare LBM is 1.8%.
The IP of SEER SBM was 0.2% among subjects with Her2-/HR+ breast cancer, compared to 0.5% for Her2+/HR(+/−) and 0.7% for Triple Negative cancers. The IPs of Medicare LBM were greater, at 1.1%, 3.1%, and 4.2% for each of Her2-/HR+, Her2+/HR(+/−), and Triple Negative breast cancer, respectively.
The incidence proportion of Medicare LBM among patients with distant disease at primary diagnosis was 13.6%, compared to 4.0% in patients with lymph node or regional involvement; 2.0%, with only regional tissue involvement; 2.4% with only lymph node involvement; and 0.9% with localized disease (Supplementary Table 4).
35,268 subjects diagnosed with melanoma in 2008 through 2012 met inclusion criteria (Supplementary Figure 1), 21,860 of these subjects were diagnosed in 2010 through 2012 (Supplementary Table 3).
The AAIR of SBM in the 2010–2012 cohort was 1,049 per 100,000 cases (95% CI: 826– 1,318), and the IP of cases with SBM diagnosis was 1.1% (Table 3). The Medicare LBM AAIR in the 2008–2012 melanoma cohort was 3,583 per 100,000 cases (95% CI: 3,154– 4,059), while the IP of primary cancer diagnosis later associated with Medicare LBM was 3.6%.
When stratified by stage at primary diagnosis, 30.4% of cases with distant disease at primary diagnosis were later associated with Medicare LBM; 15.2%, regional and lymph node involvement; 13.2%, lymph node involvement only; 7.8%, regional tissue involvement; and 2.5% among cases with localized disease at primary diagnosis (Supplementary Table 4).
Discussion
This study offers population-based estimates of BM incidence at two intervals during the course of disease, while also describing the reliability of the estimates and providing full reproducibility thereof. Given continued advances in primary site control coupled with the use of therapies that may affect metastatic behavior, such a snapshot may prove a valuable reference (17, 18).
In addition, population-based estimates of BM occurrence among Medicare recipients are useful because the 9.7% increase in the US population 65 years of age or older between 2000 and 2010 may continue with concomitant increases in absolute measures of cancer and therefore BM incidence (2). The convergence of biomedical advances with an aging population underscores the need for studies of secondary cancer.
Using the SEER population whose primary cancer diagnosis was made in the years 2010 through 2013, Cagney et al report that the IP of SEER SBM in SCLC was 15.8%; adenocarcinoma, 14.4%; and NSCLC, 12.8% (1). Similarly, the corresponding SEER SBM IP for this study’s 2010 – 2012 Medicare population were 13.5%, 11.8%, and 13.4%, for SCLC, adenocarcinoma, and NSCLC, respectively. Though these differences in IP may be attributable to population selection procedure, the most notable of which is our restriction to the elderly population due to the use of Medicare claims, their approximate similarity lends credence to each estimate.
The greatest difference between synchronous and lifetime BM incidence proportions was observed in SCLC, for which the Medicare LBM IP was 21.7%. In contrast, the Medicare LBM IP among NSCLC patients was found to be 15.3%; relative to each other, these proportions are consistent with literature describing the occurrence of SCLC and NSCLC metastases (19). Further, these increased proportions support National Comprehensive Cancer Network (NCCN) guidelines that suggest brain magnetic resonance imaging (MRI) with contrast should be a part of pretreatment evaluation for people diagnosed with Stage II-IV and high-risk Stage 1B NSCLC (20). Moreover, 4.8% of cases considered localized and 10.5% of cases with lymph node involvement were found to be associated with Medicare LBM, supporting the use of MRI to evaluate patients prior to treatment (Supplementary Table 4).
For patients not diagnosed with SBM, Nussbaum et al (1996) report that the median durations from NSCLC and SCLC diagnosis to presentation with BM were 3 and 6 months, respectively, with a median 10 months from BM presentation to death for each histology (22). This progression is not untreatable, however, and prophylactic cranial irradiation following lung cancer diagnosis has been shown to reduce later risk of BM (23, 24).
Still, at present, deferred occurrence of BM does not address concern regarding the benefit to survival of PCI versus close monitoring and quick response (25–27). Ideally, improved detection of potentially cancerous tissue combined with precision non-surgical treatment such as stereotactic radiosurgery will lower the threshold to benefit from prophylactic cranial irradiation.
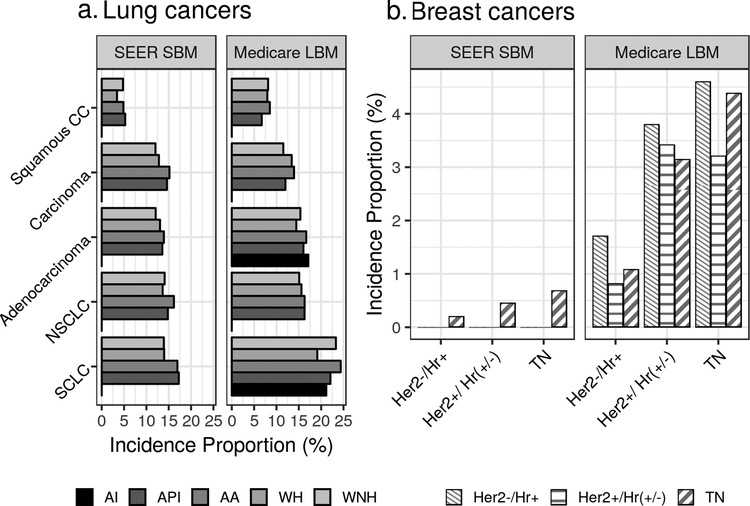
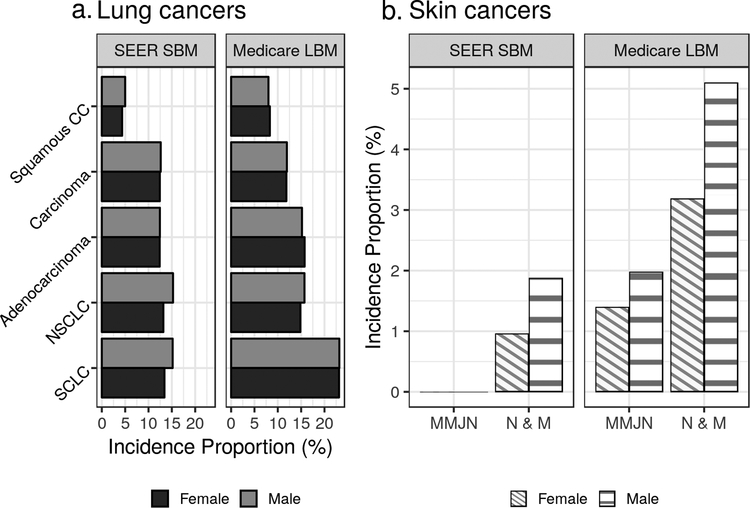
Previous work has also shown that ethnic minorities have been more likely to present with late-stage lung cancer (28, 29). In NSCLC and SCLC, Black/African American cases had greater IPs of SEER SBM compared to WNH or WH, though this did not extend to Medicare LBM (Figure 2a). Males had a greater IP of SEER SBM in both NSCLC and SCLC (Figure 3a), which is notable given the median survival following brain metastasis diagnosis has been reported to be decreased among males (30).
Figure 2: Lung and breast cancer incidence proportions of brain metastases by race.
This figure illustrates incidence proportions of synchronous brain metastasis as found in SEER data (SEER SBM) and lifetime brain metastases as found in Medicare data (Medicare LBM). In addition to stratification by race, lung cancer incidence proportions are stratified by histology and breast cancers stratified by molecular subtype. “AI” stands for American Indian/Native American; “API”, Asian/Pacific Islander; “WH”, White Hispanic; and “WNH”, White Non-Hispanic. Incidence proportions are not shown where fewer than 11 subjects were available for that proportion. “Her2” stands for Human Epidermal Growth Factor Receptor 2, “Hr” stands for hormone receptors, reflecting either progesterone receptor or estrogen receptor expression status.
Figure 3: Lung and melanoma incidence proportions by sex.
This figure illustrates incidence proportions of synchronous brain metastasis as found in SEER data (SEER SBM) and lifetime brain metastases as found in Medicare data (Medicare LBM). Each of lung cancers and melanomas are presented, stratified by histology and sex. Incidence proportions are not shown where fewer than 11 subjects were available for that proportion. “MMJN” stands for malignant melanoma in junctional nevi; “N & M”, nevi and melanomas.
Previous work shows that IP of SEER SBM in the overall population of breast cancer patients is estimated to be 0.41%, which is higher than the observed IP of SEER SBM in the Medicare breast cancer population here (0.3%) (5, 6). This difference is likely multifactorial, however one explanation could be that it reflects differences in study populations. Excluding patients younger than 65 years of age removes a population that is at greater risk of BM, as Barnholtz-Sloan et al note: “the highest IP% for brain metastases in individuals with primary breast cancer occurred in the youngest age category (IP%, 10%; age at diagnosis, 20 to 39 years) (31).”
Though not restricted to the Medicare population, Martin et al report that 361 out of 162,078 (0.22%) Her2-negative breast cancer cases had SBM (6), which is very similar to the corresponding proportion observed here, 0.2% (Table 3). Combining their reported Her2+/HR+ and Her2+/HR- categories, Martin et al report 242 out of 32,095 (0.75%) Her2-positive subjects developed brain metastases in the overall SEER population, which is slightly greater than the corresponding 31 out of 5,900 (0.5%) found in the SEER-Medicare Her2+ population examined here (6) (Supplementary Table 2).
Such associations with hormonal receptor expression status are well-documented (32, 33). In our elderly population, 13.4% of cases were ER negative; in contrast, ER-negative breast cancers comprised 28.4% of cases in the SEER SBM and 34.6% in the Medicare LBM populations. Extending this effect seen in cancers lacking hormone receptor expression, the greatest LBM rates were observed among patients with the Triple Negative (Her2-/ER-/PR-) molecular subtype: the SEER SBM IP among Triple Negative patients was 0.7%, while the Medicare LBM IP was about 5 times greater at 4.2% (Figure 2b). Multiple sources report Triple Negative cancer patients diagnosed with SBM have median survival of about 6 months (95% CI: 2.0 – 20.0), in contrast to median survival following HER2-/HR+ and HER2+/HR+ SEER SBM at 14.0 and 21.0 months, respectively (6, 34, 35).
Previous studies have demonstrated that male sex is associated with a greater IP of melanoma BM, which was the case for IP of both SEER SBM and Medicare LBM in the population studied here (36) (Figure 3b). Kromer et al showed that the IP of SEER SBM in melanoma cases was 1.2% for the general population, which is slightly greater than the IP observed in the elderly population here (1.1%) (5).
Whereas NCCN guidelines recommend cranial MRI for stage III (regional) and IV (metastatic) melanoma (37), our results suggest cranial evaluation may be warranted even for localized disease. Of Medicare LBM cases, 34.6% were considered localized at the time of primary cancer diagnosis (Supplementary Table 3). Considering the broader population, 2.6% of localized melanoma cases later had evidence of Medicare LBM, a proportion that increases to 30.4% among patients with distant disease (Supplementary Table 4).
Because the ICD code 198.3 describes metastasis to the CNS rather than the brain, we used this CNS metastasis code coupled with an intracranial imaging procedure to help ensure localization to the brain. The use of claims data is also problematic because BM is not itself tied to reimbursement, though some investigators report excellent classification performance compared to retrospective chart review (38). The present work reports several BM identification algorithms that yield either sensitivity or PPV exceeding 0.8, but none with both sensitivity and PPV that simultaneously exceeding 0.8 (Table 1, Table 2). Though insufficient for precise, individual-level prediction, estimates of frequency appeared consistent or comparable across data sources, suggesting claims data may reasonably be used to evaluate incidence of metastatic disease.
Two significant limitations to this study are (1) generalizability of the Medicare population to the population at large and (2) accuracy of CNS metastasis diagnosis and imaging procedure codes to detecting BM.
Because BM IP peaks for cancers diagnosed at around 60 years of age, our estimates in the population that was age-eligible for Medicare must be cautiously applied to future work (36). Further, the occurrence of BM arising from breast cancers can greatly exceed the average four years of Medicare claims follow-up available here, though the interval from lung cancer primary diagnosis to BM is well-covered by available claims follow-up. Despite these potential issues, confirmation of the decreased IP of SBM in elderly patients is itself a generalizable conclusion because Medicare covers approximately 97% of the population 65 years or older (8, 40).
Careful consideration must be given to the use of claims data for metastasis research (40–42). The present work addresses this need by purposefully omitting statistical tests of difference, and by providing concordance estimates of Medicare algorithms identifying BM. This is the first study to examine Medicare concordance with a SEER gold standard for BM, thus providing context for many future Medicare claims-driven studies of cranial disease following cancer diagnosis, but such highly-reliable data as SEER are not always available. Studies of Medicare claims (part of what is now widely-agreed to be ‘real-world’ data (43)) may benefit from similar evaluation of codes used to derive patient characteristics.
One significant strength of this work is the availability of documented analysis code, ideally enabling study replication with a single command (“make”) (15). To the best of our knowledge, this is the first such open and reproducible SEER-Medicare work.
The results of this study approximately agree with previous population-based estimates of BM incidence, and rigorously shed light on which disease may warrant closer monitoring. Despite increases in the incidence of brain metastases, the majority of primary cancer diagnoses have no standard of care for brain metastasis screening (4). The development of screening guidelines based on such population-level evidence could help to identify many patients’ cranial disease sooner in its progression, ultimately allowing earlier treatment and improved patient outcomes.
Supplementary Material
Acknowledgements
This work was partially supported by the Central Brain Tumor Registry of the United States (CBTRUS). Funding for CBTRUS was provided by the Centers for Disease Control and Prevention (CDC) under Contract No. 2016-M-9030, the American Brain Tumor Association, The Sontag Foundation, Novocure, AbbVie, the Musella Foundation, as well as private and in kind donations.
This work made use of the High Performance Computing Resource in the Core Facility for Advanced Research Computing at Case Western Reserve University.
This study used the linked SEER-Medicare database. The interpretation and reporting of these data are the sole responsibility of the authors. The authors acknowledge the efforts of the National Cancer Institute; the Office of Research, Development and Information, CMS; Information Management Services (IMS), Inc.; and the Surveillance, Epidemiology, and End Results (SEER) Program tumor registries in the creation of the SEER-Medicare database.
Financial Support
A. E. Sloan is supported by NIH CA217956, the Peter D Cristal Chair & the Center of Excellence for Translational Neuro-Oncology, and the Gerald Kaufman Fund for Glioma Research at University Hospitals of Cleveland, the Kimble Family Foundation, and the Ferry Family Foundation.
Footnotes
Conflicts of Interest
The author Mustafa S Ascha has no potential conflicts of interest to disclose.
The author Quinn T Ostrom has no potential conflicts of interest to disclose.
The author James Wright has no potential conflicts of interest to disclose.
The author Priya Kumthekar has no potential conflicts of interest to disclose.
The author Jeremy S Bordeaux has no potential conflicts of interest to disclose.
The author Andrew E Sloan has no potential conflicts of interest to disclose.
The author Fredrick R Schumacher has no potential conflicts of interest to disclose.
The author Carol Kruchko has no potential conflicts of interest to disclose.
The author Jill S Barnholtz-Sloan has no potential conflicts of interest to disclose.
References
- 1.Cagney DN, Martin AM, Catalano PJ, et al. Incidence and prognosis of patients with brain metastases at diagnosis of systemic malignancy: A population-based study. Neuro-Oncology. 2017:nox077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Werner CA The older population: 2010. 2010 census briefs. document c2010br-09. 2011. [Google Scholar]
- 3.Loeffler JS, Wen PY. Epidemiology, Clinical Manifestations, and Diagnosis of Brain Metastases. (DeAngelis LM, Eichler AF, eds.). Wolters Kluwer Health, UpToDate; 2018. [Google Scholar]
- 4.Collaborative Stage Data Collection System User Documentation and Coding Instructions Version 02.03.02. Chicago, Illinois, United States of America: Collaborative Stage Work Group of the American Joint Committee on Cancer; American Joint Committee on Cancer; 2017. [Google Scholar]
- 5.Kromer C, Xu J, Ostrom QT, et al. Estimating the annual frequency of synchronous brain metastasis in the united states 2010–2013: A population-based study. Journal of Neuro-Oncology. 2017:1–10. [DOI] [PubMed] [Google Scholar]
- 6.Martin AM, Cagney DN, Catalano PJ, et al. Brain metastases in newly diagnosed breast cancer: A population-based study. JAMA oncology. 2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Fox BD, Cheung VJ, Patel AJ, Suki D, Rao G. Epidemiology of metastatic brain tumors. Neurosurgery Clinics of North America. 2011;22(1):1–6. [DOI] [PubMed] [Google Scholar]
- 8.Warren JL, Klabunde CN, Schrag D, Bach PB, Riley GF. Overview of the seer-medicare data: Content, research applications, and generalizability to the united states elderly population. Medical care. 2002;40(8):IV–3. [DOI] [PubMed] [Google Scholar]
- 9.Surveillance E-RP Epidemiology SEER Research Data Record Description: Cases Diagnosed in 1973–2014. Chicago, Illinois, United States of America: National Cancer Institute, National Institutes of Health; 2017. [Google Scholar]
- 10.National Cancer Institute. ICD-O-3 SEER Site/Histology validation list. 2018. [Google Scholar]
- 11.National Cancer Institute. SEER-Medicare: Requirements of investigators following receipt of data. 2018. [Google Scholar]
- 12.Keyfitz N Sampling variance of standardized mortality rates. Human Biology. 1966;38(3):309–317. [PubMed] [Google Scholar]
- 13.Stodden V, Borwein J, Bailey DH. Setting the default to reproducible. computational science research SIAM News. 2013;46(5):4–6. [Google Scholar]
- 14.R Core Team. R: A Language and Environment for Statistical Computing. Vienna, Austria: R Foundation for Statistical Computing; 2017. [Google Scholar]
- 15.Stallman RM, McGrath R, Smith PD. GNU Make: A Program for Directing Recompilation, for Version 3.81. Free Software Foundation; 2004. [Google Scholar]
- 16.Ascha Mustafa. (2018, August 5). mustafaascha/brain-mets-seer: Reproducibility repository for a study of brain metastases in SEER-Medicare (Version v1.0.2). Zenodo. [Google Scholar]
- 17.Shao Y-Y, Lu L-C, Cheng A-L, Hsu C-H. Increasing incidence of brain metastasis in patients with advanced hepatocellular carcinoma in the era of antiangiogenic targeted therapy. The oncologist. 2011;16(1):82–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Pàez-Ribes M, Allen E, Hudock J, et al. Antiangiogenic therapy elicits malignant progression of tumors to increased local invasion and distant metastasis. Cancer cell. 2009;15(3):220–231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Little AG, Gay EG, Gaspar LE, Stewart AK. National survey of non-small cell lung cancer in the united states: Epidemiology, pathology and patterns of care. Lung cancer. 2007;57(3):253–260. [DOI] [PubMed] [Google Scholar]
- 20.National Comprehensive Cancer Network (NCCN). NCCN Framework for Resource Stratification of NCCN Guidelines. Version 3.2018: Non-Small Cell Lung Cancer, Core. 2018. MS-49. [Google Scholar]
- 21.Jacot W, Quantin X, Boher J, et al. Brain metastases at the time of presentation of non-small cell lung cancer: A multi-centric aerio* analysis of prognostic factors. British journal of cancer. 2001;84(7):903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nussbaum ES, Djalilian HR, Cho KH and Hall WA, 1996. Brain metastases: histology, multiplicity, surgery, and survival. Cancer: Interdisciplinary International Journal of the American Cancer Society, 78(8), pp.1781–1788. [PubMed] [Google Scholar]
- 23.Gore EM, Bae K, Wong SJ, Sun A, Bonner JA, Schild SE, Gaspar LE, Bogart JA, Werner-Wasik M and Choy H, 2011. Phase III comparison of prophylactic cranial irradiation versus observation in patients with locally advanced non–small-cell lung cancer: Primary analysis of Radiation Therapy Oncology Group Study RTOG 0214. Journal of Clinical Oncology, 29(3), p.272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Slotman B, Faivre-Finn C, Kramer G, Rankin E, Snee M, Hatton M, Postmus P, Collette L, Musat E and Senan S, 2007. Prophylactic cranial irradiation in extensive small-cell lung cancer. New England Journal of Medicine, 357(7), pp.664–672. [DOI] [PubMed] [Google Scholar]
- 25.Hochstenbag MMH, Twijnstra A, Wilmink JT, Wouters EFM and Ten Velde GPM, 2000. Asymptomatic brain metastases (BM) in small cell lung cancer (SCLC): MR-imaging is useful at initial diagnosis. Journal of neuro-oncology, 48(3), pp.243–248. [DOI] [PubMed] [Google Scholar]
- 26.Gregor A, Cull A, Stephens RJ, Kirkpatrick JA, Yarnold JR, Girling DJ, Macbeth FR, Stout R, Machin D and for Cancer UKCC, 1997. Prophylactic cranial irradiation is indicated following complete response to induction therapy in small cell lung cancer: results of a multicentre randomised trial. European Journal of Cancer, 33(11), pp.1752–1758. [DOI] [PubMed] [Google Scholar]
- 27.Takahashi T, Yamanaka T, Seto T, Harada H, Nokihara H, Saka H, Nishio M, Kaneda H, Takayama K, Ishimoto O and Takeda K, 2017. Prophylactic cranial irradiation versus observation in patients with extensive-disease small-cell lung cancer: a multicentre, randomised, open-label, phase 3 trial. The Lancet Oncology, 18(5), pp.663–671. [DOI] [PubMed] [Google Scholar]
- 28.Halpern MT, Ward EM, Pavluck AL, Schrag NM, Bian J, Chen AY. Association of insurance status and ethnicity with cancer stage at diagnosis for 12 cancer sites: A retrospective analysis. The lancet oncology. 2008;9(3):222–231. [DOI] [PubMed] [Google Scholar]
- 29.Haiman CA, Stram DO, Wilkens LR, et al. Ethnic and racial differences in the smoking-related risk of lung cancer. New England Journal of Medicine. 2006;354(4):333–342. [DOI] [PubMed] [Google Scholar]
- 30.Videtic GM, Reddy CA, Chao ST, et al. Gender, race, and survival: A study in non–Small-cell lung cancer brain metastases patients utilizing the radiation therapy oncology group recursive partitioning analysis classification. International Journal of Radiation Oncology• Biology• Physics. 2009;75(4):1141–1147. [DOI] [PubMed] [Google Scholar]
- 31.Barnholtz-Sloan JS, Sloan AE, Davis FG, Vigneau FD, Lai P, Sawaya RE. Incidence proportions of brain metastases in patients diagnosed (1973 to 2001) in the metropolitan detroit cancer surveillance system. Journal of clinical oncology. 2004;22(14):2865–2872. [DOI] [PubMed] [Google Scholar]
- 32.Hicks DG, Short SM, Prescott NL, Tarr SM, Coleman KA, Yoder BJ, Crowe JP, Choueiri TK, Dawson AE, Budd GT and Tubbs RR, 2006. Breast cancers with brain metastases are more likely to be estrogen receptor negative, express the basal cytokeratin CK5/6, and overexpress HER2 or EGFR. The American journal of surgical pathology, 30(9), pp.1097–1104. [DOI] [PubMed] [Google Scholar]
- 33.Garcia M, Derocq D, FREiss GILLES and Rochefort H, 1992. Activation of estrogen receptor transfected into a receptor-negative breast cancer cell line decreases the metastatic and invasive potential of the cells. Proceedings of the National Academy of Sciences, 89(23), pp.11538–11542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lin NU, Claus E, Sohl J, Razzak AR, Arnaout A, Winer EP. Sites of distant recurrence and clinical outcomes in patients with metastatic triple-negative breast cancer. Cancer. 2008;113(10):2638–2645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Dawood S, Broglio K, Esteva F, et al. Survival among women with triple receptor-negative breast cancer and brain metastases. Annals of Oncology. 2009;20(4):621–627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Bedikian AY, Wei C, Detry M, et al. Predictive factors for the development of brain metastasis in advanced unresectable metastatic melanoma. American journal of clinical oncology. 2011;34(6):603–610. [DOI] [PubMed] [Google Scholar]
- 37.National Comprehensive Cancer Network (NCCN). NCCN Clinical Practice Guidelines Version 2.2018: Melanoma. 2018. [Google Scholar]
- 38.Eichler AF, Lamont EB. Utility of administrative claims data for the study of brain metastases: A validation study. Journal of neuro-oncology. 2009;95(3):427–431. [DOI] [PubMed] [Google Scholar]
- 39.Ostrom QT, Wright CH, Barnholtz-Sloan JS. Brain metastases: Epidemiology. Handbook of clinical neurology. 2018;149:27–42. [DOI] [PubMed] [Google Scholar]
- 40.Cooper GS, Yuan Z, Stange KC, Amini SB, Dennis LK, Rimm AA. The utility of medicare claims data for measuring cancer stage. Medical care. 1999;37(7):706–711. [DOI] [PubMed] [Google Scholar]
- 41.Whyte JL, Engel-Nitz NM, Teitelbaum A, Rey GG, Kallich JD. An evaluation of algorithms for identifying metastatic breast, lung, or colorectal cancer in administrative claims data. Medical care. 2015;53(7):e49–e57. [DOI] [PubMed] [Google Scholar]
- 42.Nordstrom BL, Whyte JL, Stolar M, Mercaldi C, Kallich JD. Identification of metastatic cancer in claims data. Pharmacoepidemiology and drug safety. 2012;21(S2):21–28. [DOI] [PubMed] [Google Scholar]
- 43.Sherman RE, Anderson SA, Dal Pan GJ, Gray GW, Gross T, Hunter NL, LaVange L, Marinac-Dabic D, Marks PW, Robb MA and Shuren J, 2016. Real-world evidence—what is it and what can it tell us. N Engl J Med, 375(23), pp.2293–2297. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.