Abstract
Diabetes mellitus is a common condition affecting both adults and children. Long-standing diabetes is associated with cardiovascular abnormalities such as coronary artery atherosclerosis, microvascular changes, hypertension, kidney disease, and heart failure. Its association with heart failure in the absence of coronary artery disease and hypertension was termed diabetic cardiomyopathy in the 1970s and is believed to account for some of the cardiac mortality in diabetic patients. This entity may be implicated as the cause of sudden cardiac death in the small percentage of diabetic patients in which the autopsy fails to demonstrate evidence of nonketotic hyperosmolar coma, diabetic ketoacidosis, or atherosclerotic and hypertensive cardiovascular disease. Molecular and metabolic alterations have been studied to explain the pathophysiology of this disease.
Keywords: Forensic pathology, Diabetes mellitus, Cardiomyopathy, Cardiovascular disease, Cardiac hypertrophy
Introduction
Diabetes mellitus (DM) is a metabolic disorder characterized by elevated blood glucose concentrations and may be juvenile or adult onset. In DM, hyperglycemia results either from ineffective insulin secretion by the endocrine pancreas or a dulled response to insulin by body tissues (1). In either instance, the diagnosis is classically preceded by a history of weight loss, polydipsia, and polyuria, and its onset also may be preceded by a recent infectious illness (2). In the forensic setting, the diagnosis is made by detection of elevated vitreous glucose and glucosuria. Diabetic ketoacidosis additionally requires the detection of ketones such as acetone or β-hydroxybutyrate in blood, urine, or vitreous fluid and is more commonly observed in juvenile-onset DM (3).
Discussion
Cardiovascular Complications of Diabetes Mellitus
Mortality due to cardiovascular causes is common in diabetic patients. The vast majority of these fatal events are due to coronary artery atherosclerosis, which occurs earlier and with greater severity in diabetics (4, 5). Diabetes mellitus often coincides with obesity, hypertension, and hypercholesterolemia, each of which provides its own contribution to the development of cardiovascular disease. Heart failure also is prevalent among diabetics with an incidence directly proportional to age and a strong association with fatal outcomes (6).
For forensic pathologists, DM is often considered a contributory factor in death related to cardiovascular disease caused by atherosclerosis or hypertension. Additionally, DM can also be implicated as the proximate cause of death in individuals with evidence of diabetic ketoacidosis or neurologic sequelae of hyperosmolar nonketotic diabetic coma.
Diabetic Cardiomyopathy
Diabetic cardiomyopathy was first described more than 40 years ago as a microangiopathic disorder characterized clinically by cardiomegaly and advanced cardiac failure leading to death (7). In that initial study, postmortem examinations of four patients aged 49 to 76 years without a history of valvular, congenital, or hypertensive heart disease demonstrated cardiac hypertrophy, diffuse myocardial fibrosis, and intimal and subendothelial arteriolar thickening. Patients with a history of alcoholism or whose autopsy showed any coronary artery with greater than 40% atherosclerotic stenosis were excluded. Periodic-acid Schiff staining in one patient highlighted the deposition of mucopolysaccharides as the pathophysiologic mechanism of the small vessel disease (7). Two years after the entity was first described, Kannel et al. observed that diabetic men in the Framingham Heart study had double the rates of heart failure compared to nondiabetic men and in diabetic women; the rate of heart failure was five times higher than in nondiabetic women (8). Another study showed that patients with idiopathic cardiomyopathy were more likely to be diabetics and in those who died and had autopsies performed, large coronary artery disease was absent but myocardial microvascular abnormalities were seen (9). Finally, the Strong Heart study also demonstrated that diabetes mellitus is an independent risk factor of heart failure (34).
Numerous subsequent studies have confirmed and/or further characterized the clinical and pathologic features of this entity (6, 10-18). An echocardiographic study showed increased left ventricular mass, left ventricular wall thickness, and abnormal left ventricular end systolic and end diastolic dimensions in male and female subjects with glucose intolerance and in diabetic women (19). Additionally, an increase in heart rate in diabetic patients was observed and attributed to neuropathy-induced changes in vagal tone (20). While cardiac complications are generally thought to occur in longstanding DM, studies have demonstrated abnormalities in cardiac function in diabetics and juvenile diabetics, but also in individuals with prediabetes or impaired glucose tolerance (5).
Antemortem detection of left ventricular hypertrophy, left ventricular diastolic dysfunction, and autonomic neuropathy requires invasive and/or costly testing. Therefore, these changes are often asymptomatic prior to a significant cardiac event and/or sudden cardiac death (21). Such a cardiac event may be preceded by a history of recent exercise intolerance or fluid retention (22).
As diabetic cardiomyopathy has been defined as ventricular dysfunction that is independent of coronary artery and hypertensive disease, it is characterized by diastolic dysfunction. Three stages of diabetic cardiomyopathy have been described (11). The early stage includes no overt functional or structural changes and is characterized by depletion of GLUT4 myocardial glucose transporters, increased free fatty acids (FFA), carnitine deficiency, calcium changes, and insulin resistance. High cellular FFA concentrations may result in increased oxygen demands during FFA metabolism, which may lead to impaired myocardial performance and morphological changes. The second stage demonstrates diastolic dysfunction and slightly increased left ventricular mass. Characteristics include apoptosis/necrosis, changes in angiotensin II and transforming growth factor β (TGF-β1), and early cardiac autonomic neuropathy. The late stage has diastolic dysfunction and a decreased ejection fraction with marked left ventricular hypertrophy and is characterized by microvascular changes and marked autonomic neuropathy.
Forensic Issues
Sudden cardiac death is well-known and frequently observed by the forensic pathologist. Autopsies performed in such instances generally show morphologic cardiac abnormalities and evidence of functional cardiac deficiency. Cardiomyopathies are complex conditions due to a variety of diseases, drugs, and conditions, are often multifactorial, and can cause sudden cardiac death.
Diabetic cardiomyopathy may be considered in the following circumstances: 1) an individual of any age group with a known history of diabetes mellitus and no history of congenital heart disease, hypertension, or alcohol abuse is found dead of an apparent sudden cardiac death; 2) an autopsy is performed and shows cardiomegaly, cardiac myocyte hypertrophy, myocardial fibrosis, microvascular changes, no more than slight coronary artery atherosclerosis (<50% stenosis), and no valvular heart disease, (Image 1); 3) additional autopsy findings may include lower extremity edema, body cavity effusions, cardiac dilatation, and nephrosclerosis; 4) nephrosclerosis is accompanied by characteristic changes of diabetic nephropathy (Kimmelstiel-Wilson nodules, Image 2); 5) nonketotic hyperosmolar coma and diabetic ketoacidosis are excluded by postmortem laboratory studies. The changes of hypertensive versus diabetic nephrosclerosis may be grossly indistinguishable. Since hypertension is common, asymptomatic, and often not formally diagnosed before death, nephrosclerosis without Kimmelstiel-Wilson nodules may be presumed to be due to hypertension.
Image 1:
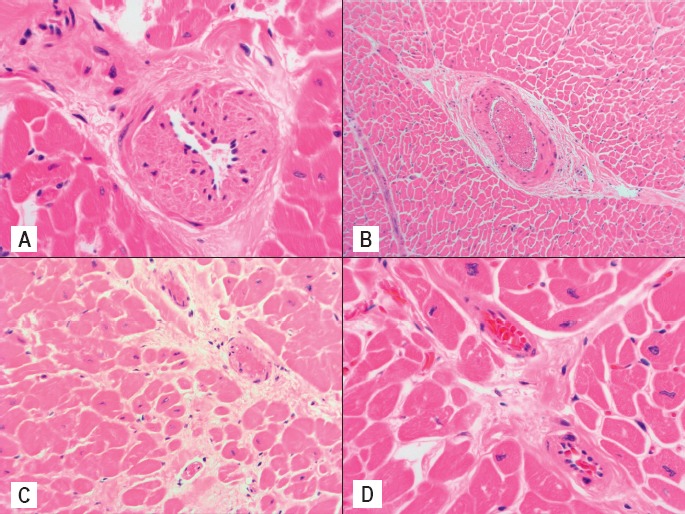
A) Microvascular intimal thickening (H&E, x400), B) Perivascular fibrosis (H&E, x100), C) Interstitial fibrosis (H&E, x200), and D) Cardiac myocyte hypertrophy (H&E, x400) in a 15-year-old male with no history of hypertension, congenital or valvular disease, and no coronary artery atherosclerosis at autopsy. The 380 g heart also showed biventricular dilatation. The diagnosis of diabetic cardiomyopathy was withheld because findings of diabetic ketoacidosis were present.
Image 2:
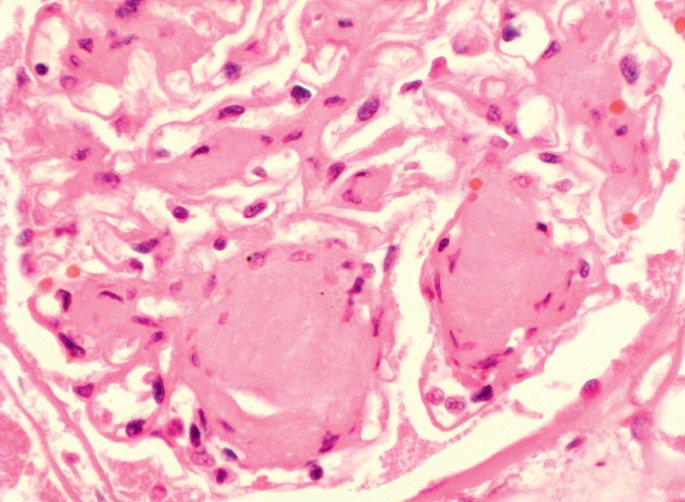
Characteristic changes of diabetic nephropathy including nodular globules of homogeneous eosinophilic hyaline material located at the periphery of the glomerulus (Kimmelstiel-Wilson nodules, H&E, x400).
Metabolic and Molecular Considerations
The exact underlying molecular and metabolic alterations in diabetic cardiomyopathy remain unknown. Numerous studies have uncovered many associated pathways including hyperglycemia-induced oxidative stress leading to cardiac myocyte apoptosis, alterations in cardiac myocyte lipid metabolism, and changes in gene expression through microRNA (12, 13, 16, 18, 23-27).
Liu et al. recently investigated the role of Mitsugumin 53 (MG53) overexpression on cardiac function in transgenic mice (28). MG53 is an E3 ligase targeting both insulin receptor and insulin receptor substrate 1 for ubiquitin-dependent degradation, resulting in decreased protein levels of insulin receptor and insulin receptor substrate 1. MG53 is expressed in heart and skeletal muscle and is involved in cardiac conditioning related to ischemic events. Transgenic mice overexpressing MG53 (MG53 hTG) become obese, insulin resistant, hyperglycemic, and demonstrate cardiac-specific alterations including myocardial insulin resistance, decreased use of insulin substrate, myocardial fibrosis, ventricular hypertrophy, and cardiac dysfunction. MG53 hTG showed cardiac enlargement with ventricular dilatation and dysfunction as evidenced by increased ventricular diameter by 22% and cardiac weight by 15% compared to wild type mice, and decreased cardiac ejection fraction by echocardiography—changes that preceded increases in body weight. These morphologic and physiologic changes coincided with changes in expression of α- and β-myosin heavy chains and upregulation of brain and atrial natriuretic peptides. The observed cardiac changes resulted from two different mechanisms of diabetic influence on the heart. The first was upregulation of peroxisome proliferation-activated receptor α (PPARα). MG53 hTG showed increased expression levels of PPARα while levels in MG53 downregulated mice were decreased, confirming that MG53 is an upstream regulator of PPARα. The second mechanism was impaired insulin signaling as evidenced by decreased protein expression of insulin receptor and insulin receptor substrate 1. MG53 overexpression also resulted in attenuated insulin-stimulated myocardial glucose uptake and increased myocardial uptake of free fatty acids, including a three-fold increased uptake of palmitic acid. Finally, genome-wide expression profiling showed activation of signaling pathways associated with hypertrophic cardiomyopathy, dilated cardiomyopathy, and the PPARα pathway in MG53 hTG.
Interestingly, Rosiglitazone, a pharmacologic PPARαγ agonist, was associated with increased risk of heart failure in diabetics treated with the drug (29), providing further evidence for the role of PPARα in the pathogenesis of diabetic cardiomyopathy.
Controversy
Patients with diabetes mellitus are at increased risk for cardiovascular disease and diabetes can affect multiple components of the heart: coronary macrovascular and microvascular system, autonomic system, and myocardium. Microangiopathy results in retinopathy and renal failure in addition to cardiac vascular effects. While disparities in study design and patient selection criteria have generated controversy regarding the existence of a cardiomyopathy due solely to DM, the aforementioned studies have confirmed that DM does play a role in the development of these cardiac abnormalities (10, 11). Some have questioned if diabetic cardiomyopathy is a distinct clinical entity as its pathogenesis appears to be multifactorial and results from both metabolic and structural alterations (11, 30-32). Additionally, the reported frequent association of late stage diabetic cardiomyopathy with hypertension and coronary artery disease obscures the distinction (11). Proposed putative mechanisms include metabolic disturbances, myocardial fibrosis, small vessel disease, autonomic dysfunction, and insulin resistance (11).
An extensive review by Fang et al. synthesized the existing literature (284 references, both pro and con) and discussed the mechanisms and therapeutic implications of diabetic cardiomyopathy (11). They reviewed data that showed that diabetes mellitus results in cardiac functional and structural changes independent of hypertension and coronary artery disease. Metabolic disturbances include alterations in supply and use of glucose in myocytes, resulting in myocyte injury. Myocardial fibrosis may be related to diabetes-related defects in cellular calcium transport, defects in contractile proteins, and an increase in collagen formation (33). Fang et al. ultimately concluded that evidence strongly supports the existence of diabetic cardiomyopathy as a distinct clinical entity.
Part of the diagnosis of diabetic cardiomyopathy depends upon the absence of hypertension and more than slight coronary artery disease. Exclusion of coronary artery disease may be done by angiography or autopsy examination. The exclusion of primary hypertension may not be as easy since high blood pressure can be unrecognized for years, causing no symptoms but causing progressive damage to the heart and other organs. At autopsy, one may infer the diagnosis of hypertensive cardiovascular disease based on cardiac hypertrophy and small vessel renal disease. In a patient with long standing diabetes mellitus, it may be difficult to discern the etiology of these cardiac and kidney findings (i.e., primarily diabetic versus hypertensive).
Conclusion
Diabetes mellitus is one of the most commonly diagnosed and treated diseases in modern medicine. It is often associated with other chronic diseases and has deleterious effects on many organs and tissues. In most circumstances encountered by the forensic pathologist, the decedent will have a medical history that includes any combination of hypertension, coronary artery disease, diabetes mellitus, obesity, and possibly alcoholism and cardiac findings that are consistent with heart failure and causative of death but may be due to any one or more of the above. Since there is now clear evidence of the role of diabetes mellitus in the formation of both structural and functional cardiac changes leading to heart failure, the disease process contributing most significantly, either by disease duration or severity, to the cardiac changes should be assigned as the proximate cause, with the other associated diseases or conditions listed as contributory causes. Only when other confounding disorders are excluded, is diabetic cardiomyopathy implicated as the proximate cause of death. This translates to a small but important subset of forensic death investigations.
Footnotes
Ethical Approval: As per Journal Policies, ethical approval was not required for this manuscript
Statement of Human and Animal Rights: This article does not contain any studies conducted with animals or on living human subjects
Statement of Informed Consent: No identifiable personal data were presented in this manuscript
Disclosures & Declaration of Conflicts of Interest: The authors, reviewers, editors, and publication staff do not report any relevant conflicts of interest
Financial Disclosure: The authors have indicated that they do not have financial relationships to disclose that are relevant to this manuscript
References
- 1.Kumar V., Abbas A.K., Fausto N., editors. Robbins and Cotran pathologic basis of disease. 7th ed. Philadelphia: Elsevier Saunders; 2005. 1525 p. [Google Scholar]
- 2.van der Werf N1, Kroese F.G., Rozing J., Hillebrands J.L. Viral infections as potential triggers of type 1 diabetes. Diabetes Metab Res Rev. 2007. Mar; 23(3): 169–83. PMID: 17103489. 10.1002/dmrr.695. [DOI] [PubMed] [Google Scholar]
- 3.Laffel L. Ketone bodies: a review of physiology, pathophysiology and application of monitoring to diabetes. Diabetes Metab Res Rev. 1999. Nov-Dec; 15(6): 412–26. PMID: 10634967. . [DOI] [PubMed] [Google Scholar]
- 4.Kengne A.P., Turnbull F., MacMahon S. The Framingham Study, diabetes mellitus and cardiovascular disease: turning back the clock. Prog Cardiovasc Dis. 2010. Jul-Aug; 53(1): 45–51. PMID: 20620425. 10.1016/j.pcad.2010.02.010. [DOI] [PubMed] [Google Scholar]
- 5.Celentano A., Vaccaro O., Tammaro P. et al. Early abnormalities of cardiac function in non-insulin-dependent diabetes mellitus and impaired glucose tolerance. Am J Cardiol. 1995. Dec 1; 76(16): 1173–6. PMID: 7484905. 10.1016/s0002-9149(99)80330-0. [DOI] [PubMed] [Google Scholar]
- 6.Bertoni A.G., Tsai A., Kasper E.K., Brancati F.L. Diabetes and idiopathic cardiomyopathy: a nationwide case-control study. Diabetes Care. 2003. Oct; 26(10): 2791–5. PMID: 14514581. 10.2337/diacare.26.10.2791. [DOI] [PubMed] [Google Scholar]
- 7.Rubler S., Dlugash J., Yuceoglu Y.Z. et al. New type of cardiomyopathy associated with diabetic glomerulosclerosis. Am J Cardiol. 1972. Nov 8; 30(6): 595–602. PMID: 4263660. 10.1016/0002-9149(72)90595-4. [DOI] [PubMed] [Google Scholar]
- 8.Kannel W.B., Hjortland M., Castelli W.P. Role of diabetes in congestive heart failure: the Framingham study. Am J Cardiol. 1974. Jul; 34(1): 29–34. PMID: 4835750. 10.1016/0002-9149(74)90089-7. [DOI] [PubMed] [Google Scholar]
- 9.Hamby R.I., Zoneraich S., Sherman L. Diabetic cardiomyopathy. JAMA. 1974. Sep 23; 229(13): 1749–54. PMID: 4278055. 10.1001/jama.1974.03230510023016. [DOI] [PubMed] [Google Scholar]
- 10.Boudina S., Abel E.D. Diabetic cardiomyopathy revisited. Circulation. 2007. Jun 26; 115(25): 3213–23. PMID: 17592090. 10.1161/circulationaha.106.679597. [DOI] [PubMed] [Google Scholar]
- 11.Fang Z.Y., Prins J.B., Marwick T.H. Diabetic cardiomyopathy: evidence, mechanisms, and therapeutic implications. Endocr Rev. 2004. Aug; 25(4): 543–67. PMID: 15294881. 10.1210/er.2003-0012. [DOI] [PubMed] [Google Scholar]
- 12.Bai T., Wang F., Mellen N. et al. Diabetic cardiomyopathy: role of the E3 ubiquitin ligase. Am J Physiol Endocrinol Metab. 2016. Apr 1; 310(7): E473–83. PMID: 26732687. 10.1152/ajpendo.00467.2015. [DOI] [PubMed] [Google Scholar]
- 13.Bayeva M., Sawicki K.T., Ardehali H. Taking diabetes to heart–deregulation of myocardial lipid metabolism in diabetic cardiomyopathy. J Am Heart Assoc. 2013. Nov 25; 2(6): e000433 PMID: 24275630. PMCID: PMC3886738. 10.1161/jaha.113.000433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Asghar O., Al-Sunni A., Khavandi K., Khavandi A. et al. Diabetic cardiomyopathy. Clin Sci (Lond). 2009. May; 116(10): 741–60. PMID: 19364331. PMCID: PMC2782307. 10.1042/cs20080500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Boudina S., Abel E.D. Diabetic cardiomyopathy, causes and effects. Rev Endocr Metab Disord. 2010. Mar; 11(1): 31–9. PMID: 20180026. PMCID: PMC2914514. 10.1007/s11154-010-9131-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Falcao-Pires I., Leite-Moreira A.F. Diabetic cardiomyopathy: understanding the molecular and cellular basis to progress in diagnosis and treatment. Heart Fail Rev. 2012. May; 17(3): 325–44. PMID: 21626163. 10.1007/s10741-011-9257-z. [DOI] [PubMed] [Google Scholar]
- 17.Pappachan J.M., Varughese G.I., Sriraman R., Arunagirinathan G. Diabetic cardiomyopathy: Pathophysiology, diagnostic evaluation and management. World J Diabetes. 2013. Oct 15; 4(5): 177–89. PMID: 24147202. PMCID: PMC3797883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bugger H., Abel E.D. Molecular mechanisms of diabetic cardiomyopathy. Diabetologia. 2014. Apr; 57(4): 660–71. PMID: 24477973. PMCID: PMC3969857. 10.1007/s00125-014-3171-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Galderisi M., Anderson K.M., Wilson P.W., Levy D. Echocardiographic evidence for the existence of a distinct diabetic cardiomyopathy (the Framingham Heart Study). Am J Cardiol. 1991. Jul 1; 68(1): 85–9. PMID: 2058564. 10.1016/0002-9149(91)90716-x. [DOI] [PubMed] [Google Scholar]
- 20.Page M.M., Watkins P.J. The heart in diabetes: autonomic neuropathy and cardiomyopathy. Clin Endocrinol Metab. 1977. Jul; 6(2): 377–88. PMID: 330039. [DOI] [PubMed] [Google Scholar]
- 21.Fang Z.Y., Yuda S., Anderson V. et al. Echocardiographic detection of early diabetic myocardial disease. J Am Coll Cardiol. 2003. Feb 19; 41(4): 611–7. PMID: 12598073. 10.1016/s0735-1097(02)02869-3. [DOI] [PubMed] [Google Scholar]
- 22.Hunt S.A., Baker D.W., Chin M.H. et al. ACC/AHA guidelines for the evaluation and management of chronic heart failure in the adult: executive summary. A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to revise the 1995 Guidelines for the Evaluation and Management of Heart Failure). J Am Coll Cardiol. 2001. Dec; 38(7): 2101–13. PMID: 11738322. 10.1016/s0735-1097(01)01683-7. [DOI] [PubMed] [Google Scholar]
- 23.Zhang M., Gu H., Chen J., Zhou X. Involvement of long noncoding RNA MALAT1 in the pathogenesis of diabetic cardiomyopathy. Int J Cardiol. 2016. Jan 1; 202: 753–5. PMID: 26476026. 10.1016/j.ijcard.2015.10.019. [DOI] [PubMed] [Google Scholar]
- 24.Adeghate E. Molecular and cellular basis of the aetiology and management of diabetic cardiomyopathy: a short review. Mol Cell Biochem. 2004. Jun; 261(1-2): 187–91. PMID: 15362503. 10.1023/b:mcbi.0000028755.86521.11. [DOI] [PubMed] [Google Scholar]
- 25.Goyal B.R., Mehta A.A. Diabetic cardiomyopathy: pathophysiological mechanisms and cardiac dysfuntion. Hum Exp Toxicol. 2013. Jun; 32(6): 571–90. PMID: 23174745. 10.1177/0960327112450885. [DOI] [PubMed] [Google Scholar]
- 26.Avogaro A., Vigili de Kreutzenberg S., Negut C. et al. Diabetic cardiomyopathy: a metabolic perspective. Am J Cardiol. 2004. Apr 22; 93(8A): 13A–16A. PMID: 15094099. 10.1016/s0002-9149(03)01509-1. [DOI] [PubMed] [Google Scholar]
- 27.Asrih M., Steffens S. Emerging role of epigenetics and miRNA in diabetic cardiomyopathy. Cardiovasc Pathol. 2013. Mar-Apr; 22(2): 117–25. PMID: 22951386. 10.1016/j.carpath.2012.07.004. [DOI] [PubMed] [Google Scholar]
- 28.Liu F., Song R., Feng Y. et al. Upregulation of MG53 induces diabetic cardiomyopathy through transcriptional activation of peroxisome proliferation-activated receptor α. Circulation. 2015. Mar 3; 131(9): 795–804. PMID: 25637627. 10.1161/circulationaha.114.012285. [DOI] [PubMed] [Google Scholar]
- 29.Home P.D., Pocock S.J., Beck-Nielsen H. et al. Rosiglitazone evaluated for cardiovascular outcomes in oral agent combination therapy for type 2 diabetes (RECORD): a multicentre, randomised, open-label trial. Lancet. 2009. Jun 20; 373(9681): 2125–35. PMID: 19501900. 10.1016/s0140-6736(09)60953-3. [DOI] [PubMed] [Google Scholar]
- 30.Letonja M., Petrovič D. Is diabetic cardiomyopathy a specific entity? World J Cardiol. 2014. Jan 26; 6(1): 8–13. PMID: 24527183. PMCID: PMC3920165. 10.4330/wjc.v6.i1.8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Stratmann B., Gawlowski T., Tschoepe D. Diabetic cardiomyopathy–to take a long story serious. Herz. 2010. May; 35(3): 161–8. PMID: 20467928. 10.1007/s00059-010-3336-0. [DOI] [PubMed] [Google Scholar]
- 32.Ernande L., Derumeaux G. Diabetic cardiomyopathy: myth or reality? Arch Cardiovasc Dis. 2012. Apr; 105(4): 218–25. PMID: 22633296. 10.1016/j.acvd.2011.11.007. [DOI] [PubMed] [Google Scholar]
- 33.Regan T.J., Wu C.F., Yeh C.K. et al. Myocardial composition and function in diabetes. The effects of chronic insulin use. Circ Res. 1981. Dec; 49(6): 1268–77. PMID: 7030514. 10.1161/01.res.49.6.1268. [DOI] [PubMed] [Google Scholar]
- 34.de Simone G., Devereux R.B., Chinali M. et al. Diabetes and incident heart failure in hypertensive and normotensive participants of the Strong Heart Study. J Hypertens. 2010. Feb; 28(2): 353–60. PMID: 19844184. PMCID: PMC3005764. 10.1097/hjh.0b013e3283331169. [DOI] [PMC free article] [PubMed] [Google Scholar]


