Abstract
High-density lipoprotein cholesterol (HDL-c) has long been referred to as ‘good cholesterol’ due to its apparent inverse relationship with future CVD risk. More recent research has questioned a causal role for HDL-c in this relationship, however, as both genetic studies and numerous large-scale randomised controlled trials have found no evidence of a cardiovascular protective effect when HDL-c levels are raised. Instead, focus has switched to the functional properties of the HDL particle. Evidence suggests that both the composition and function of HDL may be significantly altered in the context of an inflammatory milieu, transforming the particle from a vasoprotective anti-atherogenic particle to a noxious pro-atherogenic equivalent. This review will summarise evidence relating HDL to CVD risk, explore recent evidence characterising changes in the composition and function of HDL that may occur in chronic inflammatory diseases, and discuss the potential for future HDL-modifying therapeutic interventions.
Keywords: HDL dysfunction, Inflammation, Atherosclerosis, Cardiovascular disease
Introduction
High-density lipoproteins (HDL) participate in the transfer of excess cholesterol from peripheral sites to the liver, and the cholesterol carried within these lipoproteins (HDL-c) is therefore often referred to as ‘good cholesterol’. Over the past half-century, HDL-c has repeatedly been shown to be inversely associated with risk of future cardiovascular events, and has therefore become one of the most well-known and widely studied risk factors for cardiovascular disease (CVD). Recent research, however, has called the causal nature of this relationship into question, with genetic studies finding little evidence of an association between elevated HDL-c levels and reduced CVD risk [1, 2], and a series of major clinical trials failing to demonstrate any clinical benefit when HDL-c levels were pharmacologically raised [3–7]. Recent insights into the complex structural and functional properties of HDL particles—and their vulnerability to modification when exposed to a range of CV risk factors, particularly inflammation—have shed light on the potential reasons for these negative findings, and have posed the question as to whether focusing on the ‘quality’ rather than quantity of HDL may be a more relevant target for drug development. This review will summarise previous research attempting to reduce CVD risk through the modification of HDL-c levels, explore recent evidence characterising changes in the composition and function of HDL that may occur in chronic inflammatory diseases, and discuss the potential for future HDL-modifying therapeutic interventions.
HDL-C and Cardiovascular Risk: When Correlation Does Not Equal Causation
The concept of HDL-c as a beneficial lipid was first proposed in the Framingham Heart Study in the latter half of the twentieth century, where low levels of HDL-c were found to be an independent risk factor for future incidence of coronary heart disease (CHD) in 2815 men and women over 12 years of follow-up [8, 9]. The replication of these findings in further large-scale cohorts suggested a protective role for HDL-c in the atherosclerotic process [10, 11], leading to the adoption of HDL-c’s well-known description as a form of“good cholesterol”. Despite robust relationships in epidemiological studies, however, the observational nature of these findings long meant that the question of whether HDL-c played a direct role in the disease process—or was simply a biomarker of other underlying complications—remained uncertain.
Disappointingly, a number of findings from genetic studies and numerous large-scale randomised clinical trials in the last 10–15 years have suggested the latter. Firstly, the development of genome-wide association studies (GWAS) and Mendelian Randomization studies in the early 2000s allowed the causal nature of this relationship to be investigated for the first time using HDL-raising genetic variants in large observational studies as proxy measures for cumulative HDL-c exposure. Findings from two separate Mendelian Randomization studies containing over 165,000 participants found no association between genetically determined HDL-c and risk of myocardial infarction, despite traditional observational relationships from the same cohorts displaying the expected inverse relationship with plasma levels of HDL-c [1, 2]. These findings challenged the concept that elevating HDL-c would translate into clinical benefit, and these concerns were later confirmed by negative findings from numerous randomised clinical trials investigating a number of different HDL-raising drugs. Perhaps the most well-known of these failures was that of the ILLUMINATE trial, a randomised clinical trial investigating the use of torcetrapib in>15,000 individuals at high risk of CVD, and the first major trial involving a recently developed class of drugs known as cholesterol ester transfer protein (CETP)-inhibitors. As the name suggests, this drug class—which included the agents torcetrapib, dalcetrapib, evacetrapib, anacetrapib, and obicetrapib—was designed to block CETP; the enzyme responsible for transferring cholesterol esters away from HDL and on to VLDL/LDL. Despite raising HDL-c levels by over 70%, ILLUMINATE was forced to terminate prematurely due to an increased risk of CV events and mortality in patients taking the drug, most likely due to off-target effects on aldosterone production and blood pressure [3]. The development of different forms of this drug that lacked these off-target effects maintained hope for CETP-inhibitors as a therapeutic target, but results from later trials proved once again disappointing, with both the dal-OUTCOMES (dalcetrapib) and ACCELERATE (evacetrapib) trials showing no benefit of treatment despite significant increases in HDL-c [4, 5]. Although a rare positive outcome was reported for anacetrapib—the final CETP-inhibitor to be tested in the recent HPS3/TIMI55-REVEAL trial—this was concluded to have likely resulted from a further lowering of LDL-c rather than the observed elevation in HDL-c [12]. Alongside CETP-inhibitors, the early 2000s also saw the design and completion of a number of trials investigating extended release niacin, an agent shown to consistently elevate HDL-c via numerous distinct pathways—including (but not limited to) an upregulation of ApoA-I production, a reduction in CETP activity, and a reduction in hepatic uptake of HDL [13]. In the first of these studies (AIM-HIGH), the addition of niacin to statin treatment in >3400 patients showed no evidence of clinical benefit, leading the trial to be terminated early due to lack of efficacy [6], whereas in the later HPS2-THRIVE trial, extended release niacin not only had no clinical benefit, but was in fact found to increase risk of serious adverse events [7]. Taken together, findings from these trials appear to have ended the concept that elevated levels of HDL-c act as a protective factor against atherosclerotic disease, and efforts to reduce CVD risk through pharmacological manipulation of HDL-c are therefore no longer being pursued.
HDL Function and Cardiovascular Risk: a Shift of Focus from ‘Quantity’ to ‘Quality’
Given the wide range of anti-atherogenic properties attributed to the HDL particle in decades of animal research and preclinical studies, the discovery that HDL-c lacked a causal role in CVD risk was initially unexpected. However, the majority of functional properties exerted by HDL are not dependent on the cholesterol content of the particle per se, but rather on numerous other structural components contained within the complex and highly heterogenous HDL particle. Therefore, with the end of the HDL-c hypothesis in the early 2000s came the start of the HDL function hypothesis—characterised by a focus on the functional capacity of HDL particles, rather than their absolute cholesterol levels alone.
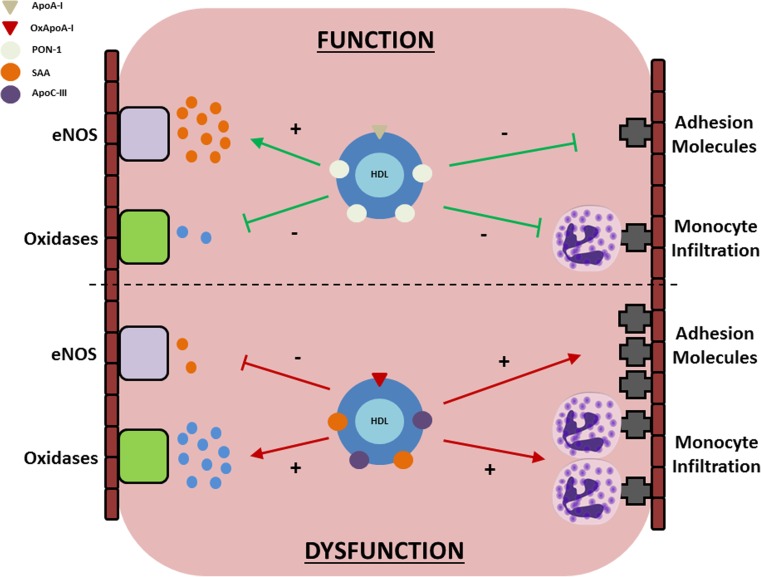
In the basal state, HDL’s functional properties are predominantly anti-atherogenic; the most well-studied of which is its ability to facilitate the removal of excess cholesterol from atherosclerotic plaques in a process known as reverse cholesterol transport (RCT). In addition to RCT, however, HDL has also been shown to have multiple other vascular effects which may be expected to provide additional protection; such as an ability to increase endothelial nitric oxide (NO) bioavailability, a capacity to reduce oxidative stress and inflammation, and an ability to reduce the expression of endothelial adhesion markers and transendothelial monocyte migration [14]. More recent evidence, however, has highlighted the presence of numerous structural and functional changes in the HDL particle that may result in its transformation from an anti-atherogenic vasoprotective particle to a pro-inflammatory noxious equivalent (Fig. 1). These alterations are most commonly observed in the presence of systemic inflammation, and suggest an evolutionary role for HDL in the innate immune response, where the greatest threat to human longevity has long come from viral or bacterial infections [15]. With long-term chronic inflammatory conditions such as CHD or type 2 diabetes (T2D) now prevalent in the population, however, the possibility exists that this previously ‘protective’ response may now paradoxically accelerate vascular damage and increase the risk of mortality via atherosclerotic complications.
Fig. 1.
Potential mechanisms linking dysfunctional HDL to vascular damage. In the absence of inflammation (top), HDL exerts vascular protective effects through its ability to increase endothelial NO bioavailability, decrease oxidative stress, and inhibit the expression of adhesion molecules on the vascular wall. In inflammatory states (bottom), however, numerous structural and functional changes may occur and compromise endothelial function. Beneficial proteins contained within the particle (e.g. ApoA-I, PON-1) are liable for oxidation or replacement, resulting in a noxious particle which impairs NO generation, amplifies oxidative stress, and promotes adhesion molecule expression and monocyte infiltration across the vascular wall
The remainder of this review will therefore seek to address this topic, beginning with (1) evidence surrounding the anti-atherogenic role for HDL, (2) changes in its structure and function that may accompany chronic inflammation, (3) the implications these changes have for CVD risk in different clinical populations and (4) what this may mean for HDL as a risk marker or therapeutic target.
HDL Structure and Function in Healthy Individuals: an Anti-atherogenic Particle Exerting Multiple Vasoprotective Effects
HDL Structure
The term HDL in fact refers to a highly heterogeneous range of particles within human plasma, ranging in size from 5 to 17 nm and density from 1.063–1.210 kg/l. Alongside cholesterol, lipidomic and proteomic techniques have identified over 200 additional lipids and more than 85 proteins which may reside within the HDL particle [16, 17], as well as multiple enzymes and even genetic material in the form of micro RNAs (Table 1). Significant variation in the relative quantities of these components results in a wide spectrum of circulating HDL particles that may differ in size, density, structure, and function. For ease of assessment, however, these are commonly separated into five broad subclasses, ranging from the largest and least dense (termed HDL2b) to the smallest and most dense (termed HDL3b). The major structural protein component of HDL is apolipoprotein A-I (apoA-I), formed in both the liver (~80%) and intestine (~20%) and secreted in a lipid-free state. Efflux of phospholipids and free cholesterol on to at least 2 molecules of apoA-I stimulate the biogenesis of HDL, resulting in the generation of a discoidal lipid-poor ‘nascent’ HDL particle. Subsequent incorporation of numerous enzymes such as lecithin-cholesterol acyltransferase (LCAT), cholesterol ester transfer protein (CETP) and phospholipid transfer protein (PLTP) into this nascent particle further alter the lipid and phospholipid composition—resulting in the generation of mature spherical particles of differing sizes and densities. These particles may be further modified through the incorporation of numerous other structural proteins and enzymes (shown in Table 1) all of which interact to determine the eventual functional capabilities of the HDL particle [18].
Table 1.
Structural components contributing to heterogeneity of HDL molecule
| Shape | Subclasses (size/density) | Apolipoproteins | Lipids | Enzymes | Acute phase proteins | miRNAs |
|---|---|---|---|---|---|---|
| Discoidal (Nascent) | HDL2b (Largest / Least Dense) | ApoA (I, II, IV, V) | Cholesterol | LCAT | SAA | miR-92a |
| Spherical (Mature) | HDL2a | ApoC (I, II, III, IV) | Cholesterol esters | PLTP | LBP | miR-126 |
| HDL3a | ApoD | Triglycerides | CETP | Fibrinogen | miR-150 | |
| HDL3b | ApoE | Glycerophosphlipids | Lp-PLA2 | C3 | miR-223 | |
| HDL3b (Smallest / Most Dense) | ApoF | Sphingolipids | MPO | A1AT | ||
| ApoH | PON-1 | |||||
| ApoJ | ||||||
| ApoM |
HDL, high-density lipoprotein; Apo, apolipoprotein; LCAT, lecithin cholesterol acyl-transferase; PLTP, phospholipid transfer protein; CETP, cholesterol ester transfer protein; Lp-PLA2, lipoprotein-associated phospholipid A2; MPO, myeloperoxidase; PON, paraoxonase; SAA, serum amyloid A; LBP, lipopolysaccharide binding protein; C3, complement C3; A1AT, alpha-1-anti-trypsin
Reverse Cholesterol Transport
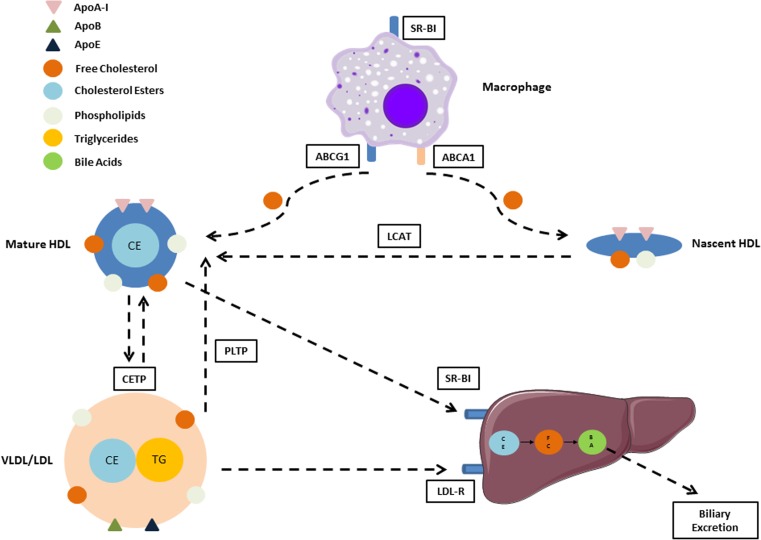
The concept of HDL as a shuttle for removing excess cholesterol from lipid-laden macrophages within peripheral tissues was first proposed over 50 years ago [19], and since then has become one of the most well-recognised pathways underlying HDL’s anti-atherogenic nature (Fig. 2). When cell cholesterol content is normal, this process is dominated via the passive aqueous diffusion of free cholesterol between cell membranes and HDL [20]. Although this pathway is bidirectional, esterification of free cholesterol within HDL by the enzyme LCAT maintains a concentration gradient promoting efflux out of the cells, thereby preventing its accumulation in cell membranes and maintaining homeostasis. In addition to passive diffusion, non-aqueous efflux may also occur through the binding of HDL to scavenger receptor class B1 (SR-BI), with this pathway shown to be enhanced when absolute HDL levels are low, or HDL particle size is large [21]. When cell cholesterol content is elevated, however—as is the case in atherogenic lipid-laden macrophages—a shift to active efflux pathways mediated through the upregulation of the ATP-binding cassette transporters ABCA1 and ABCG1 begin to predominate. The most important of these pathways involves the efflux of phospholipids and free cholesterol on to lipid-free or lipid-poor apolipoproteins via ABCA1, as demonstrated by the inability of ABCA1-knockout mice [20] or patients with Tangier’s disease (who lack ABCA1) to generate HDL [22]. Although all HDL-associated apolipoproteins are capable of accepting cholesterol from ABCA1, interactions with ApoA-I account for the majority of the response [23]. As is the case for passive diffusion, LCAT-mediated esterification of cholesterol on these newly formed nascent HDL particles reduces free extracellular free cholesterol levels by trapping them as esters within the HDL core; with this process both maintaining the concentration gradient and resulting in the development of mature spherical HDL particles capable of mediating further efflux through ABCG1 receptors [24]. Once transferred to HDL, cholesterol esters are then returned to the liver for biliary excretion in one of two ways—they may be exchanged for triglycerides in VLDLs and other remnant lipoproteins by CETP, thereby enabling their uptake and excretion via hepatic LDL receptors, or they may be selectively removed from HDL directly via its binding to hepatic SR-BI [25].
Fig. 2.
Mechanistic pathways underlying reverse cholesterol transport from macrophages to liver. HDL-mediated RCT involves the removal of cholesterol from lipid-laden macrophages, where it may then be transported back to the liver for biliary excretion. In healthy individuals, nascent and mature HDL particles accept cholesterol from lipid-laden macrophages within the vascular wall via an interaction of ApoA-I with ABCA1 and ABCG1 receptors. Following their esterification by the enzyme LCAT, these cholesterol particles may undergo selective uptake to the liver via the interaction of HDL and hepatic SR-BI, or may be transferred by CETP to VLDL/LDL for hepatic uptake via LDL-R. In conditions of chronic disease or inflammation, numerous changes in this pathway have been observed that may render it dysfunctional and potentially increase atherosclerotic risk. These changes include the oxidation or displacement of ApoA-I, alterations in the activity of enzymes such as LCAT or CETP, and reduced binding of HDL to receptors such as ABCG1 or SR-BI
Endothelial Nitric Oxide Bioavailability
Diminished nitric oxide (NO) bioavailability is a hallmark of the vascular disease process, and is thought to underpin every stage of atherosclerosis [26]. The first evidence of HDL’s ability to directly promote endothelial NO production came in the early 2000s, where the addition of HDL to cultured endothelial cells was shown to directly stimulate endothelial NO synthase (eNOS) activation in vitro [27]. In this ground-breaking study, the authors demonstrated an essential role for both ApoA-I and SR-BI in NO generation, as removal of the former using antibodies, or the latter using SR-BI knockout mice, resulted in a loss of effect. However, the use of purified ApoA-I in place of HDL also failed to elicit a response, suggesting a necessary involvement of other components within the HDL particle for eNOS phosphorylation. Later research identified a number of HDL-bound bioactive phospholipids—namely sphingosylphosphorylcholine (SPC), sphingosine-1-phosphate (S1P), and lysosulfatide (LSF)—as at least partially responsible [28], with the binding of these to sphingosine-1-phosphate receptor 3 (S1P3) demonstrated to stimulate vasorelaxation via the activation of phosphatidylinositol 3-kinase(PI3K)/Akt and mitogen-activated protein kinase (MAPK) pathways [29]. In addition to ApoA-I and SR-BI, additional ABCG1 pathways are also likely contribute to this response. HDL-mediated efflux of 7-ketosterol via ABCG1 has also been shown to improve NO release by preventing the generation of NO-scavenging reactive oxygen species within the endothelium, while cholesterol efflux via the same receptor has been shown to elicit similar responses via a reduction in eNOS/caveolin interactions [30, 31].
Anti-oxidant and Anti-inflammatory Capacity
An accumulation of foam cells within the vascular wall is another key process of atherosclerotic disease, initiated via the uptake of circulating LDL-c and its subsequent oxidation by reactive oxygen species (ROS) and pro-inflammatory enzymes such as MPO within the sub-endothelial space. This oxLDL is subsequently taken up by scavenger receptors on macrophages, triggering a cascade of further oxidative and inflammatory responses which encourage the promotion of endothelial adhesion molecule expression and increased monocyte infiltration within the vessel wall. HDL has been shown to exert multiple antioxidant and anti-inflammatory effects that may reduce this vicious circle of oxidation/monocyte infiltration, such as an ability to protect endothelial and smooth muscle cells from the cytotoxic effects of oxLDL [32–34] in vitro. This effect is likely to be attributed to a variety of mechanisms. For example, increases in NO bioavailability (as described in the previous section) likely contribute at least in part to this response, as do the presence of multiple antioxidant enzymes carried within HDL’s protein cargo. The most studied of these enzymes is the antioxidant enzyme paraoxonase-1 (PON-1), although roles for other enzymes such as lipoprotein-associated phospholipid A2 (Lp-PLA2) [35] and LCAT [36] have also been demonstrated. The presence of PON-1 has been shown to protect both HDL and LDL from oxidation in vitro [33, 37], while its absence (using PON-1 knockout mice) has been demonstrated to have the opposite effect [38]. Interactions with ApoA-I appear to be crucial for its activity, as demonstrated by the significantly increased capacity for PON-1 to prevent LDL oxidation and promote RCT in HDL particles containing ApoA-I as opposed to those containing ApoA-II or IV [39]. Additional antioxidant effects of ApoA-I also likely contribute to HDL’s antioxidant properties via its ability to directly bind and remove oxidised lipids from LDL particles within the vascular wall, as treatment of arterial cell walls with ApoA-I or an Apo-AI mimetic peptide in vitro prevents the oxidation of LDL, as does injection of ApoA-I into both mice and humans [40, 41]. HDL has also been shown in a number of studies to reduce superoxide production in endothelial cells treated with tumour necrosis factor alpha (TNF-α) [42–44], possibly through inhibitory effects on nicotinamide adenine dinucleotide phosphate (NADPH)-oxidases mediated through HDL-associated lysosphingolipids and their interaction with S1P3 and SR-BI receptors [45]. Both this pathway and others have also been shown to have downstream effects on the production of numerous inflammatory-mediated adhesion molecules such as vascular and intercellular adhesion molecules (VCAM-1 and ICAM-1) [46], E-selectin [28], and monocyte chemoattractant protein-1 (MCP-1) [45, 47], reducing their expression and limiting monocyte transmigration across the vascular wall. Furthermore, ABCA1-mediated cholesterol efflux to ApoA-I may also provide additional suppression through the activation of anti-inflammatory signalling molecules during reverse cholesterol transport [48].
HDL Structure and Dysfunction in Chronic Inflammation: When “Good Cholesterol” Turns Bad
Inflammation Alters HDL Structure
The concept that individuals with chronic disease may have structurally modified and potentially dysfunctional HDL was initially suggested in the mid-1990s, where evidence was produced for the first time showing the replacement of ApoA-I and paraoxonase-1 (PON-1) during an acute inflammatory response with acute phase proteins such as ceruloplasmin and serum-amyloid A (SAA) [47]. In this seminal study, the authors further noted that the antioxidant and anti-inflammatory vasoprotective properties of these modified HDL particles were also lost—or in certain cases—even completely reversed, suggesting that conformational changes in the HDL particle may have negatively affected its function. Since then, wide-ranging structural changes have been reported in a variety of inflammatory disease states, many of which have been implicated in the generation of a dysfunctional phenotype which may act to increase atherosclerotic risk. The most well-studied of these is the incorporation of acute phase proteins such as SAA, symmetric dimethylarginine (SDMA), lipopolysaccharide binding protein (LBP), alpha-1-antitrypsin (A1AT), or fibrinogen into HDL’s protein cargo [49]. These changes in turn result in reciprocal and detrimental reductions in ApoA-I, a decrease in the activity of HDL-associated antioxidant enzymes such as PON-1 and Lp-PLA2, and an increased presence of inflammatory enzymes and lipid peroxidation products such as myeloperoxidase (MPO) and malondialdehyde (MDA) [49]. Furthermore, compositional changes in HDL’s lipid cargo—such as the enrichment with triglycerides commonly observed in hypertriglyceridaemic states—may further affect particle size and density, and therefore functional ability.
Inflammation Reduces Reverse Cholesterol Transport (RCT)
While a reduction in cholesterol efflux is a well-established aspect of the innate immune system during acute infections, it may have long-term negative implications under conditions of chronic inflammatory stress [15]. Numerous studies have shown that this vital homeostatic process may become impaired by a number of structural and conformational changes within HDL particles exposed to acute or chronic inflammatory conditions. The incorporation of SAA has been shown to impair cholesterol efflux in some [50–53], but not all [42, 54], studies. These changes may arise through the displacement of atheroprotective components such as ApoA-I or PON-1 within the HDL particle itself [50], or by interactions with cell membrane-bound receptors responsible for binding HDL during the RCT process. In mice overexpressing SAA, reduced binding of HDL to SR-BI and a reduction in selective hepatic cholesterol ester uptake from HDL has been observed in conjunction with elevated levels of lipid-free SAA, suggesting a reduced capacity for the liver to remove cholesterol esters for excretion [53]. Further upstream in the RCT pathway, genetic ablation of SAA has been observed to preserve cholesterol efflux in mice following an endotoxin challenge, whereas the same challenge in humans resulted in progressive decreases in efflux capacity in line with elevations in the content of HDL-incorporated SAA [51]. Furthermore, an increased affinity for HDL to bind to proteoglycans at peripheral sites has also been demonstrated following SAA elevation in endotoxin-injected mice [55]. This effect is again virtually abolished following SAA knockout, suggesting that the incorporation of SAA into HDL may further impair RCT through the entrapment of HDL within vascular lesions, where its contents may subsequently be liable to oxidation or enzymatic modification that render it more atherogenic [55]. In support of this, a number of studies have demonstrated extensive structural and functional changes within HDL particles located within the vessel wall in comparison to those circulating in the bloodstream [56–58], resulting in a substantially dysfunctional HDL phenotype which is unable to promote cholesterol efflux. For example, ApoA-I isolated from human atherosclerotic plaques has been shown to be lipid-poor, heavily oxidised by the inflammatory enzyme myeloperoxidase (MPO), and virtually devoid of any cholesterol acceptor activity [59]. This dysfunctional form of ApoA-I—which may also be caused by chlorination, nitration, or sulfoxidation of other amino acid residues—appears to exert its detrimental effects via multiple sites of action, including impairment of ABCA1 cholesterol efflux [58, 60, 61] and decreased ApoA-I-induced activation of LCAT [62]. Further inhibition of ABCA1 via an accumulation of the lipid peroxidation product MDA (a reactive carbonyl that has been shown to increase in HDL in chronic disease states) has also been demonstrated [63], while HDL-enrichment with triglycerides has been shown to impact RCT in some, but not all, studies. These latter findings may perhaps seem counter-intuitive given the finding that triglyceride-enriched HDL may in fact increase macrophage cholesterol efflux [64–66]. However, they are likely explained by further downstream effects that may affect overall RCT, including a reduced ability for LCAT to convert free cholesterol to cholesterol esters [64], and a diminished capacity to deliver these cholesterol esters to hepatic cells via SR-BI [64, 67]. HDL remodelling due to elevated PLTP activity may also adversely affect cholesterol acceptor ability, and has been shown to result in increased atherosclerotic plaque formation in mice [68].
Inflammation Reduces Endothelial Nitric Oxide Bioavailability
The ability of HDL to promote endothelial nitric oxide bioavailability has repeatedly been shown to be impaired in multiple chronic inflammatory and chronic disease states [42–44, 69–71]. HDL isolated from patients with either established coronary heart disease or acute coronary syndrome has a reduced capacity for NO generation that appears mediated, at least in part, by activation of endothelial lectin-like oxidised LDL receptor 1 (LOX-1). This upregulation of LOX-1 has been hypothesised to occur in response to an excess accumulation of the highly reactive lipid peroxidation compound MDA, possibly due to the displacement of antioxidant enzyme PON-1 from within the HDL particle. These changes in turn result in an increased activation of the eNOS-inhibitor endothelial PKCβII, ultimately leading to a reduced capacity for endothelial NO generation [71]. Inactivation of PON-1 in healthy subjects in vitro has been shown to result in decreased eNOS phosphorylation at serine residue 1177 (an eNOS activating site), increased phosphorylation at threonine residue 495 (a deactivating site), and a subsequent decrease in NO bioavailability. This relationship may be mediated at least in part by the oxidative transformation of PON-1 by increased levels of pro-inflammatory enzymes such as MPO [72], as they have been shown to display reciprocal relationships. Furthermore, MPO and other oxidative products within HDL may directly bind to and activate endothelial cells, thereby further reducing NO production via eNOS coupling [73, 74].
Inflammation Impairs HDL’s Antioxidant and Anti-inflammatory Capacity
The first evidence underpinning the concept of HDL ‘dysfunction’ came from the finding that HDL isolated from rabbits undergoing an acute phase response was unable to prevent lipid peroxidation or MCP-1 expression [47]. Since then, multiple other pro-inflammatory and pro-oxidant properties have been attributed to dysfunctional HDL particles; including an increased capacity to stimulate NADPH oxidase activity [44], an increased capacity for endothelial superoxide production [42, 69], an increased expression of cell adhesion markers such as VCAM-1 [43], and increased eNOS uncoupling [73]. Much of these functional changes are likely closely linked to those previously described when addressing reductions in NO bioavailability, with the replacement of protective proteins such as ApoA-I and PON-1 with atypical components such as MDA and MPO of particular importance. For example, while in vitro work has highlighted the ability of ApoA-I to remove seeding molecules responsible for LDL oxidation [40], this ability is lost in HDL isolated from patients with established CHD [75]. PON-1 is frequently observed to be reduced in inflammatory conditions and chronic disease [42, 47, 69], and genetic-knockout mice lacking PON-1 have been shown to have both impaired antioxidant effects and increased risk of atherosclerosis [38].
HDL Function in Clinical Populations: Implications for CVD Risk
Cardiovascular Disease
The cardiovascular disease process has been shown to exert widespread changes in HDL structure and function which may accelerate, rather than reduce, future risk of major adverse events. In a series of studies involving the LURIC, 4S, and KORA cohorts, the association between HDL-c and CVD mortality was assessed in almost 9000 patients undergoing coronary angiography who were stratified by median levels of serum SAA and SDMA [76, 77]. Remarkably, while patients with below median levels of both acute phase proteins showed the traditional inverse relationship between HDL-c and CVD outcomes, those with above median levels were observed to have the complete opposite response—i.e. a significantly increased risk of CVD mortality as HDL-c levels increased. Similar findings have also been reported when stratifying risk by levels of C-reactive protein (CRP) in the PREVEND study [78], and ApoC-III in two prospective case-control studies analysed within the Nurses Health and Health Professionals Follow-Up Studies [79], suggesting that stratification by inflammatory burden in CVD may help to identify the independent effects of ‘biologically effective’ and ‘biologically ineffective’ HDL particles. When assessing HDL function directly, cholesterol efflux capacity has been shown to be decreased in patients with CHD [80], while numerous endothelial protective functions appear to be impaired. Reduced NO bioavailability is evident in HDL isolated from patients with both established coronary heart disease and acute coronary syndrome [71]. Furthermore, and in contrast to its usual antioxidant and anti-inflammatory functions, HDL isolated from patients with CHD has also been shown to promote rather than protect against LDL oxidation [47, 75], as well as increasing the expression of adhesion markers such as MCP-1 [47], and promoting endothelial superoxide production in the presence of TNF-α [71].
Type 2 Diabetes
HDL isolated from patients with type 2 diabetes demonstrates many of the structural and functional alterations previously described for CVD. Structural changes include increased SAA levels due to underlying low-levels of chronic inflammation [81], increased triglyceride content due to insulin resistance and elevated CETP activity [82], decreased ApoA-I and S1P due to displacement by other proteins [83], and increased oxidised lipids and advanced glycation end products (AGE) due to increased levels of systemic oxidative and glycaemic stress [84]. Cholesterol efflux is reduced [85]—possibly due to an impairment of both ABCA1 and SR-BI mediated pathways through oxidation and AGE—and LCAT activity is impaired [86]. Widespread impairments in the endothelial protective effects of HDL are also evident, as demonstrated by a recent study in which HDL isolated from patient with type 2 diabetes demonstrated impaired NO bioavailabilty, increased MPO and NADPH oxidase activity, increased superoxide production, and increased lipid peroxidation [44].
Chronic Kidney Disease
The presence of reduced renal function in chronic kidney disease (CKD) leads to an accumulation of acute phase proteins within a circulating uremic milieu, many of which are capable of negatively altering HDL structure and function. HDL isolated from patients on haemodialysis is enriched with numerous components detrimental to HDL function (e.g. SAA, ApoC-III, A1AT, and triglycerides), and has been observed to have a reduced capacity to initiate RCT from macrophages [87]. Of particular importance in CKD is a significant increase in the acute phase protein symmetric dimethylarginine (SDMA), whose incorporation into the HDL particle is associated with impairments in HDL-mediated RCT and endothelial NO production in a dose-dependent manner [43, 70, 77]. This effect becomes progressively pronounced as kidney function declines, as demonstrated by a 40–60% decline in HDL-mediated endothelial NO bioavailability in patients with stage 5 CKD as opposed to a 20% increase in healthy controls [43]. Interestingly, these changes are largely reversed following kidney transplantation in children, but not adults—suggesting that HDL’s vascular protective functions may be at least partially restored in the young [43]. Using genetic knockout mice models to investigate the pathways underlying this relationship, Speer and colleagues identified toll-like receptor 2 (TLR2) as the likely signalling receptor responsible, suggesting that structural modifications to HDL caused by CKD may result in damage-associated molecular patterns within the HDL particle that trigger a chronic innate immune response [74]. Further post-translational modifications to other circulating proteins may also affect function. Albumin isolated from haemodialysis patients is heavily oxidised and markedly impairs cholesterol efflux in vitro; likely due to its high-affinity for SR-BI reducing binding sites for HDL-mediated RCT [88].
Autoimmune Diseases
HDL dysfunction has recently been demonstrated in a range of autoimmune diseases, including type 1 diabetes [89], rheumatoid arthritis [90, 91], systemic lupus erythematosus [92], and primary antiphospholipid syndrome [69]. Despite being at increased risk of dyslipidaemia, patients with type 1 diabetes commonly demonstrate elevated—rather than reduced—levels of HDL-c. Multiple studies have reported the presence of multiple dysfunctional properties to these particles, however, including reduced cholesterol efflux capacity [93, 94], reduced PON-1 activity [95], increased MPO activity [96], and decreased levels of S1P [97]. These modifications appear to be independent of HbA1c [93], and may at least partially explain the increased risk of CVD in patients with the highest HDL-c levels [98]. HDL isolated from patients with rheumatoid arthritis, systemic lupus erythematosus, and antiphospholipid syndrome also display widespread structural changes, resulting in a pro-inflammatory phenotype that worsens as systemic inflammatory burden increases [69, 91]. Treatment with anti-inflammatory medications in these patients may partially reverse these structural and functional modifications, as evidenced by decreases in SAA and LBP content, increased NO bioavailability and PON-1 activity, and decreased SO production in individuals with rheumatoid arthritis treated with the monoclonal antibodies infliximab and adalimumab [99, 100].
Periodontitis
Periodontal disease represents one of the most prevalent chronic inflammatory conditions in the world today, affecting approximately 20–50% of the global population [101]. Numerous vasoprotective functions of HDL have been shown to be impaired in periodontal patients compared to age-matched controls, including impaired endothelial NO bioavailability, increased superoxide production, and reduced PON-1 activity [42]. When using an intensive periodontal treatment intervention as an experimental model of acute inflammation, further transient decreases in both endothelial NO availability and PON-1 activity—but not RCT—are apparent in the following 24 h, highlighting the vulnerability of HDL to rapidly remodel when exposed to an inflammatory response. These changes were accompanied by substantial increases in acute phase reactants such as SAA, C3, and prothrombin, and returned to baseline levels following resolution of the inflammatory response.
Where Next for HDL as a Therapeutic Target?
Given recent advances in our understanding of HDL’s myriad functions, a limited number of population studies in recent years have attempted to link individual markers of HDL function, rather than HDL-c levels, to future risk of CVD. The largest and most rigorous of these have used cholesterol efflux capacity as the functional measure of interest, and have found increased efflux to be associated with a reduced risk for CVD in some [102–104], but not all [23], of these studies. In the largest of these to date, cholesterol efflux capacity was found to be inversely related to incidence of major CV events in nearly 3000 adults free from CVD at baseline and followed-up for over 9 years as part of the Dallas Heart Study (hazard ratio 0.33 [0.19–0.55] for top quartile of efflux compared to bottom) [102]. Interestingly, while HDL-c levels were found to be inversely associated with well-established CV risk factors such as obesity or insulin resistance, cholesterol efflux capacity was not, suggesting that changes in RCT may occur through distinct mechanisms to those affecting overall HDL-c levels. These findings were supported in a more recent nested case-control analysis in 3500 middle-aged to elderly individuals participating in the EPIC-Norfolk study, where increased cholesterol efflux was once again associated with a substantially reduced risk of incident CV events (fully adjusted odds ratio per SD increase in efflux capacity=0.80 [0.70–0.90] for those in the top tertile of cholesterol efflux capacity compared to those in the bottom) [103].
Alongside hard clinical endpoints, further encouraging findings relating changes in function to surrogate measures of CVD risk have also been reported in a number of other studies. Using carotid intima-media thickness (cIMT) and angiographically confirmed presence of CHD as surrogate markers of atherosclerotic disease progression, Khera and colleagues showed a substantially decreased presence of coronary disease in individuals within the top quartile for cholesterol efflux (OR=0.74 [0.61–0.89]) [105]. In children with CKD, Shroff et al. reported an inverse relationship between HDL-mediated NO bioavailability and multiple surrogate markers of arterial health, including cIMT and pulse-wave velocity (a measure of arterial stiffness) [43]. Likewise, in a case-control study of women with and without the autoimmune condition primary antiphospholipid syndrome, these same surrogate measures were found to be inversely related to PON-1 activity, and were accompanied by additional detrimental changes in HDL functionality such as decreased NO bioavailability and increased SO production [69]. These results suggest that interventions to modify HDL function, rather than simply increasing HDL-c levels—may still hold therapeutic potential for reducing atherosclerotic risk.
Efforts to translate these into clinical benefits in randomised trials, however, have once again proven to be challenging. For example, although 3 months of niacin therapy in patients with T2D was shown to significantly improve both HDL-c levels and numerous measures of HDL function [44], the use of this same drug in individuals at high risk for CVD displayed no clinical benefit in either the AIM-HIGH and HPS2-THRIVE trials [6, 7]. Similar issues were encountered in studies assessing the efficacy of recombinant ApoA-I infusions as a therapeutic target, which have been proposed to reduce atherosclerotic risk via their promotion of ABCA1-mediated reverse cholesterol transport. While findings from both animal studies [106] and a small-scale clinical trial in humans [107] suggested evidence of atherosclerotic plaque regression, the recent publication of two large-scale randomised controlled trials utilising both the wild-type and mutated versions of ApoA-I (the latter termed ApoA-I Milano and considered to be particularly beneficial) have once again found no clinical benefit, despite increases in cholesterol efflux capacity of up to 90% [108, 109].
It is clear that if modification of HDL function is to be pursued as a therapeutic target, further experimental studies are required to identify the most relevant pathways for intervention, and genetic studies are required to provide a causal link between these pathways and CVD. Once appropriate pathways are identified, standardisation of high-throughput functional assays for use in routine clinical practice will also be essential, as this will permit relationships between HDL function and CVD risk to be tested in large-scale cohorts and trials, and will allow comparisons of results between studies. Of note, all major studies investigating cholesterol efflux to-date have used different techniques to quantify efflux capacity, an observation which may potentially explain discrepancies in findings reported so far. Most importantly though, lessons must be learned from previous failings involving HDL-c, where the presence of observational correlations were long-considered to represent a causal relationship between HDL levels and CVD risk. Despite some promising results relating a number of markers of HDL function to future risk of disease, successful completion of large-scale randomised clinical trials altering specific functional properties of HDL are needed to support the use of HDL function as a therapeutic target.
Conclusions
Despite HDL-c’s well-established inverse relationship with CVD risk, it does not play a causal role in CVD risk reduction, and is therefore an unsuitable target for therapeutic intervention. While our understanding of the mechanisms underlying HDL function and dysfunction have been significantly enhanced in recent years, the wide range of different functions elicited by HDL, their vulnerability to modification by external inflammatory responses, and a lack of standardised assays for the measurement in routine clinical practice makes the translation of these findings challenging. While research is continuing to tease out the complexities of this highly heterogenous particle, potential diagnostic and therapeutic strategies involving the modification of HDL remain elusive.
Abbreviations
- A1AT
Alpha-1-antitrypsin
- ABCA1
ATP binding cassette transporter A1
- ABCG1
ATP binding cassette transporter G1
- AGE
Advanced glycation end products
- ApoA-I
Apolipoprotein A1
- ApoA-II
Apolipoprotein A2
- ApoC-III
Apolipoprotein C3
- C3
Complement component 3
- CETP
Cholesterol ester transfer protein
- CHD
Coronary heart disease
- cIMT
Carotid intima-media thickness
- CKD
Chronic kidney disease
- CRP
C-reactive protein
- CVD
Cardiovascular disease
- cIMT
Carotid intima-media thickness
- CVD
Cardiovascular disease
- eNOS
Endothelial nitric oxide synthase
- GWAS
Genome-wide association study
- HbA1c
Glycated haemoglobin
- HDL
High-density lipoprotein
- HDL-c
High-density lipoprotein cholesterol
- ICAM-1
Intercellular adhesion molecule 1
- LBP
Lipopolysaccharide binding protein
- LDL
Low-density lipoprotein
- LCAT
Lecithin-cholesterol acyltransferase
- LOX-1
Lectin-like oxidised LDL receptor 1
- Lp-PLA2
Lipoprotein-associated phospholipid A2
- LSF
Lysosulfatide
- MAPK
Mitogen-activated protein kinase
- MCP-1
Monocyte chemoattract protein 1
- MDA
Malondialdehyde
- MPO
Myeloperoxidase
- NADPH
Nicotinamide adenine dinucleotide phosphate
- NO
Nitric oxide
- oxLDL
Oxidised LDL
- PI3K
Phosphatidylinositol 3-kinase
- PLTP
Phospholipid transfer protein
- PON-1
Paraoxonase 1
- RCT
Reverse cholesterol transport
- RNA
Ribonucleic acid
- ROS
Reactive oxygen species
- S1P
Sphingosine-1-phosphate
- S1P3
Sphingosine-1-phosphate receptor 3
- SAA
Serum amyloid A
- SDMA
Symmetric dimethylarginine
- SPC
Sphingosylphosphorylcholine
- SR-BI
Scavenger receptor class B type I
- SO
Superoxide
- T2D
Type 2 diabetes
- TLR2
Toll-like receptor 2
- TNFα
Tumour necrosis factor alpha
- VCAM-1
Vascular cellular adhesion molecule 1
- VLDL
Very low density lipoprotein
Compliance with Ethical Standards
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of Interest
The authors declare that they have no conflicts of interest.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Haase CL, Tybjærg-Hansen A, Ali Qayyum A, Schou J, Nordestgaard BG, Frikke-Schmidt R. LCAT, HDL cholesterol and ischemic cardiovascular disease: a Mendelian randomization study of HDL cholesterol in 54,500 individuals. J Clin Endocrinol Metab. 2012;97:E248–E256. doi: 10.1210/jc.2011-1846. [DOI] [PubMed] [Google Scholar]
- 2.Voight BF, Peloso GM, Orho-Melander M, Frikke-Schmidt R, Barbalic M, Jensen MK, Hindy G, Hólm H, Ding EL, Johnson T, Schunkert H, Samani NJ, Clarke R, Hopewell JC, Thompson JF, Li M, Thorleifsson G, Newton-Cheh C, Musunuru K, Pirruccello JP, Saleheen D, Chen L, Stewart AFR, Schillert A, Thorsteinsdottir U, Thorgeirsson G, Anand S, Engert JC, Morgan T, Spertus J, Stoll M, Berger K, Martinelli N, Girelli D, McKeown PP, Patterson CC, Epstein SE, Devaney J, Burnett MS, Mooser V, Ripatti S, Surakka I, Nieminen MS, Sinisalo J, Lokki ML, Perola M, Havulinna A, de Faire U, Gigante B, Ingelsson E, Zeller T, Wild P, de Bakker PIW, Klungel OH, Maitland-van der Zee AH, Peters BJM, de Boer A, Grobbee DE, Kamphuisen PW, Deneer VHM, Elbers CC, Onland-Moret NC, Hofker MH, Wijmenga C, Verschuren WMM, Boer JMA, van der Schouw YT, Rasheed A, Frossard P, Demissie S, Willer C, Do R, Ordovas JM, Abecasis GR, Boehnke M, Mohlke KL, Daly MJ, Guiducci C, Burtt NP, Surti A, Gonzalez E, Purcell S, Gabriel S, Marrugat J, Peden J, Erdmann J, Diemert P, Willenborg C, König IR, Fischer M, Hengstenberg C, Ziegler A, Buysschaert I, Lambrechts D, van de Werf F, Fox KA, el Mokhtari NE, Rubin D, Schrezenmeir J, Schreiber S, Schäfer A, Danesh J, Blankenberg S, Roberts R, McPherson R, Watkins H, Hall AS, Overvad K, Rimm E, Boerwinkle E, Tybjaerg-Hansen A, Cupples LA, Reilly MP, Melander O, Mannucci PM, Ardissino D, Siscovick D, Elosua R, Stefansson K, O'Donnell CJ, Salomaa V, Rader DJ, Peltonen L, Schwartz SM, Altshuler D, Kathiresan S. Plasma HDL cholesterol and risk of myocardial infarction: a mendelian randomisation study. Lancet. 2012;380:572–580. doi: 10.1016/S0140-6736(12)60312-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Barter PJ, Caulfield M, Eriksson M, Grundy SM, Kastelein JJP, Komajda M, Lopez-Sendon J, Mosca L, Tardif JC, Waters DD, Shear CL, Revkin JH, Buhr KA, Fisher MR, Tall AR, Brewer B. Effects of torcetrapib in patients at high risk for coronary events. N Engl J Med. 2007;357:2109–2122. doi: 10.1056/NEJMoa0706628. [DOI] [PubMed] [Google Scholar]
- 4.Schwartz GG, Olsson AG, Abt M, Ballantyne CM, Barter PJ, Brumm J, Chaitman BR, Holme IM, Kallend D, Leiter LA, Leitersdorf E, McMurray JJV, Mundl H, Nicholls SJ, Shah PK, Tardif JC, Wright RS. Effects of dalcetrapib in patients with a recent acute coronary syndrome. N Engl J Med. 2012;367:2089–2099. doi: 10.1056/NEJMoa1206797. [DOI] [PubMed] [Google Scholar]
- 5.Lincoff AM, Nicholls SJ, Riesmeyer JS, Barter PJ, Brewer HB, Fox KAA, Gibson CM, Granger C, Menon V, Montalescot G, Rader D, Tall AR, McErlean E, Wolski K, Ruotolo G, Vangerow B, Weerakkody G, Goodman SG, Conde D, McGuire DK, Nicolau JC, Leiva-Pons JL, Pesant Y, Li W, Kandath D, Kouz S, Tahirkheli N, Mason D, Nissen SE. Evacetrapib and cardiovascular outcomes in high-risk vascular disease. N Engl J Med. 2017;376:1933–1942. doi: 10.1056/NEJMoa1609581. [DOI] [PubMed] [Google Scholar]
- 6.The AIM-HIGH Investigators. Niacin in patients with low hdl cholesterol levels receiving intensive statin therapy. N Engl J Med. 2011;365:2255–2267. doi: 10.1056/NEJMoa1107579. [DOI] [PubMed] [Google Scholar]
- 7.The HPS2-THRIVE Collaborative Group Effects of extended-release niacin with laropiprant in high-risk patients. N Engl J Med. 2014;371:203–212. doi: 10.1056/NEJMoa1300955. [DOI] [PubMed] [Google Scholar]
- 8.Gordon T, Castelli WP, Hjortland MC, Kannel WB, Dawber TR. High density lipoprotein as a protective factor against coronary heart disease. Am J Med. 1977;62:707–714. doi: 10.1016/0002-9343(77)90874-9. [DOI] [PubMed] [Google Scholar]
- 9.Castelli WP, Garrison RJ, Wilson PW, Abbott RD, Kalousdian S, Kannel WB. Incidence of coronary heart disease and lipoprotein cholesterol levels. Framingham Study JAMA. 1986;256:2835–2838. [PubMed] [Google Scholar]
- 10.Di Angelantonio E, Sarwar N, Perry P, Kaptoge S, Ray KK, Thompson A, et al. Major lipids, apolipoproteins, and risk of vascular disease. JAMA. 2009;302:1993. doi: 10.1001/jama.2009.1619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sharrett AR, Ballantyne CM, Coady SA, Heiss G, Sorlie PD, Catellier D, Patsch W. Coronary heart disease prediction from lipoprotein cholesterol levels, triglycerides, lipoprotein(a), apolipoproteins A-I and B, and HDL density subfractions: the atherosclerosis risk in communities (ARIC) study. Circulation. 2001;104:1108–1113. doi: 10.1161/hc3501.095214. [DOI] [PubMed] [Google Scholar]
- 12.Hegele RA. CETP inhibitors—a new inning? N Engl J Med. 2017;377:1284–1285. doi: 10.1056/NEJMe1711407. [DOI] [PubMed] [Google Scholar]
- 13.Chapman MJ, Le Goff W, Guerin M, Kontush A. Cholesteryl ester transfer protein: at the heart of the action of lipid-modulating therapy with statins, fibrates, niacin, and cholesteryl ester transfer protein inhibitors. Eur Heart J. 2010;31:149–164. doi: 10.1093/eurheartj/ehp399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Besler C, Lüscher TF, Landmesser U. Molecular mechanisms of vascular effects of high-density lipoprotein: alterations in cardiovascular disease. EMBO Mol Med. 2012;4:251–268. doi: 10.1002/emmm.201200224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fogelman AM. Further evidence that high-density lipoprotein is a chameleon-like lipoprotein. Eur Heart J. 2015;ehv465. [DOI] [PMC free article] [PubMed]
- 16.Kontush A, Lhomme M, Chapman MJ. Unraveling the complexities of the HDL lipidome. J Lipid Res. 2013;54:2950–2963. doi: 10.1194/jlr.R036095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Shah AS, Tan L, Long JL, Davidson WS. Proteomic diversity of high density lipoproteins: our emerging understanding of its importance in lipid transport and beyond. J Lipid Res. 2013;54:2575–2585. doi: 10.1194/jlr.R035725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Miller NE. HDL metabolism and its role in lipid transport. Eur Heart J. 1990;11:1–3. doi: 10.1093/eurheartj/11.suppl_h.1. [DOI] [PubMed] [Google Scholar]
- 19.Glomset JA. The plasma lecithins:cholesterol acyltransferase reaction. J Lipid Res. 1968;9:155–167. [PubMed] [Google Scholar]
- 20.Adorni MP, Zimetti F, Billheimer JT, Wang N, Rader DJ, Phillips MC, Rothblat GH. The roles of different pathways in the release of cholesterol from macrophages. J Lipid Res. 2007;48:2453–2462. doi: 10.1194/jlr.M700274-JLR200. [DOI] [PubMed] [Google Scholar]
- 21.Thuahnai ST, Lund-Katz S, Dhanasekaran P, de la Llera-Moya M, Connelly MA, Williams DL, Rothblat GH, Phillips MC. Scavenger receptor class B type I-mediated cholesteryl ester-selective uptake and efflux of unesterified cholesterol. J Biol Chem. 2004;279:12448–12455. doi: 10.1074/jbc.M311718200. [DOI] [PubMed] [Google Scholar]
- 22.Francis GA, Knopp RH, Oram JF. Defective removal of cellular cholesterol and phospholipids by apolipoprotein A-I in Tangier disease. J Clin Invest. 1995;96:78–87. doi: 10.1172/JCI118082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Li X-M, Tang WHW, Mosior MK, Huang Y, Wu Y, Matter W, Gao V, Schmitt D, DiDonato JA, Fisher EA, Smith JD, Hazen SL. Paradoxical association of enhanced cholesterol efflux with increased incident cardiovascular risks. Arterioscler Thromb Vasc Biol. 2013;33:1696–1705. doi: 10.1161/ATVBAHA.113.301373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yancey PG, Bortnick AE, Kellner-Weibel G, de la Llera-Moya M, Phillips MC, Rothblat GH. Importance of different pathways of cellular cholesterol efflux. Arterioscler Thromb Vasc Biol. 2003;23:712–719. doi: 10.1161/01.ATV.0000057572.97137.DD. [DOI] [PubMed] [Google Scholar]
- 25.Phillips MC. Molecular mechanisms of cellular cholesterol efflux. J Biol Chem. 2014;289:24020–24029. doi: 10.1074/jbc.R114.583658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Naseem K. The role of nitric oxide in cardiovascular diseases. Mol Asp Med. 2005;26:33–65. doi: 10.1016/j.mam.2004.09.003. [DOI] [PubMed] [Google Scholar]
- 27.Yuhanna IS, Zhu Y, Cox BE, Hahner LD, Osborne-Lawrence S, Lu P, Marcel YL, Anderson RGW, Mendelsohn ME, Hobbs HH, Shaul PW. High-density lipoprotein binding to scavenger receptor-BI activates endothelial nitric oxide synthase. Nat Med. 2001;7:853–857. doi: 10.1038/89986. [DOI] [PubMed] [Google Scholar]
- 28.Nofer J-R, van der Giet M, Tölle M, Wolinska I, von Wnuck LK, Baba HA, et al. HDL induces NO-dependent vasorelaxation via the lysophospholipid receptor S1P3. J Clin Invest. 2004;113:569–581. doi: 10.1172/JCI18004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Mineo C, Yuhanna IS, Quon MJ, Shaul PW. High density lipoprotein-induced endothelial nitric-oxide synthase activation is mediated by Akt and MAP kinases. J Biol Chem. 2003;278:9142–9149. doi: 10.1074/jbc.M211394200. [DOI] [PubMed] [Google Scholar]
- 30.Terasaka N, Yu S, Yvan-Charvet L, Wang N, Mzhavia N, Langlois R, Pagler T, Li R, Welch CL, Goldberg IJ, Tall AR. ABCG1 and HDL protect against endothelial dysfunction in mice fed a high-cholesterol diet. J Clin Invest. 2008;118:3701–3713. doi: 10.1172/JCI35470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Terasaka N, Westerterp M, Koetsveld J, Fernández-Hernando C, Yvan-Charvet L, Wang N, et al. ATP-binding cassette transporter G1 and high-density lipoprotein promote endothelial NO synthesis through a decrease in the interaction of caveolin-1 and endothelial NO synthase. Arterioscler Thromb Vasc Biol. 2010;30:2219–2225. doi: 10.1161/ATVBAHA.110.213215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hessler JR, Robertson AL, Chisolm GM. LDL-induced cytotoxicity and its inhibition by HDL in human vascular smooth muscle and endothelial cells in culture. Atherosclerosis. 1979;32:213–229. doi: 10.1016/0021-9150(79)90166-7. [DOI] [PubMed] [Google Scholar]
- 33.Parthasarathy S, Barnett J, Fong LG. High-density lipoprotein inhibits the oxidative modification of low-density lipoprotein. Biochim Biophys Acta. 1990;1044:275–283. doi: 10.1016/0005-2760(90)90314-n. [DOI] [PubMed] [Google Scholar]
- 34.van Hinsbergh VW, Scheffer M, Havekes L, Kempen HJ. Role of endothelial cells and their products in the modification of low-density lipoproteins. Biochim Biophys Acta. 1986;878:49–64. doi: 10.1016/0005-2760(86)90343-7. [DOI] [PubMed] [Google Scholar]
- 35.Silva IT, Mello APQ, Damasceno NRT. Antioxidant and inflammatory aspects of lipoprotein-associated phospholipase A2 (Lp-PLA2): a review. Lipids Health Dis. 2011;10:170. doi: 10.1186/1476-511X-10-170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Vohl M-C, Neville TA-M, Kumarathasan R, Braschi S, Sparks DL. A novel lecithin-cholesterol acyltransferase antioxidant activity prevents the formation of oxidized lipids during lipoprotein oxidation. Biochemistry. 1999;38:5976–5981. doi: 10.1021/bi982258w. [DOI] [PubMed] [Google Scholar]
- 37.Mackness MI, Arrol S, Durrington PN. Paraoxonase prevents accumulation of lipoperoxides in low-density lipoprotein. FEBS Lett. 1991;286:152–154. doi: 10.1016/0014-5793(91)80962-3. [DOI] [PubMed] [Google Scholar]
- 38.Shih DM, Gu L, Xia Y-R, Navab M, Li W-F, Hama S, Castellani LW, Furlong CE, Costa LG, Fogelman AM, Lusis AJ. Mice lacking serum paraoxonase are susceptible to organophosphate toxicity and atherosclerosis. Nature. 1998;394:284–287. doi: 10.1038/28406. [DOI] [PubMed] [Google Scholar]
- 39.Gaidukov L, Tawfik DS. High affinity, stability, and lactonase activity of serum paraoxonase PON1 anchored on HDL with ApoA-I. Biochemistry. 2005;44:11843–11854. doi: 10.1021/bi050862i. [DOI] [PubMed] [Google Scholar]
- 40.Navab M, Hama SY, Anantharamaiah GM, Hassan K, Hough GP, Watson AD, et al. Normal high density lipoprotein inhibits three steps in the formation of mildly oxidized low density lipoprotein: steps 2 and 3. J Lipid Res. 2000;41:1495–1508. [PubMed] [Google Scholar]
- 41.Navab M, Hama SY, Cooke CJ, Anantharamaiah GM, Chaddha M, Jin L, et al. Normal high density lipoprotein inhibits three steps in the formation of mildly oxidized low density lipoprotein: step 1. J Lipid Res. 2000;41:1481–1494. [PubMed] [Google Scholar]
- 42.O’Neill F, Riwanto M, Charakida M, Colin S, Manz J, McLoughlin E, et al. Structural and functional changes in HDL with low grade and chronic inflammation. Int J Cardiol. 2015;188:111–116. doi: 10.1016/j.ijcard.2015.03.058. [DOI] [PubMed] [Google Scholar]
- 43.Shroff R, Speer T, Colin S, Charakida M, Zewinger S, Staels B, Chinetti-Gbaguidi G, Hettrich I, Rohrer L, O'Neill F, McLoughlin E, Long D, Shanahan CM, Landmesser U, Fliser D, Deanfield JE. HDL in children with CKD promotes endothelial dysfunction and an abnormal vascular phenotype. J Am Soc Nephrol. 2014;25:2658–2668. doi: 10.1681/ASN.2013111212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Sorrentino SA, Besler C, Rohrer L, Meyer M, Heinrich K, Bahlmann FH, Mueller M, Horváth T, Doerries C, Heinemann M, Flemmer S, Markowski A, Manes C, Bahr MJ, Haller H, von Eckardstein A, Drexler H, Landmesser U. Endothelial-vasoprotective effects of high-density lipoprotein are impaired in patients with type 2 diabetes mellitus but are improved after extended-release niacin therapy. Circulation. 2010;121:110–122. doi: 10.1161/CIRCULATIONAHA.108.836346. [DOI] [PubMed] [Google Scholar]
- 45.Tölle M, Pawlak A, Schuchardt M, Kawamura A, Tietge UJ, Lorkowski S, et al. HDL-associated lysosphingolipids inhibit NAD(P)H oxidase-dependent monocyte chemoattractant protein-1 production. Arterioscler Thromb Vasc Biol. 2008;28:1542–1548. doi: 10.1161/ATVBAHA.107.161042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Cockerill GW, Rye KA, Gamble JR, Vadas MA, Barter PJ. High-density lipoproteins inhibit cytokine-induced expression of endothelial cell adhesion molecules. Arterioscler Thromb Vasc Biol. 1995;15:1987–1994. doi: 10.1161/01.atv.15.11.1987. [DOI] [PubMed] [Google Scholar]
- 47.Van Lenten BJ, Hama SY, de Beer FC, Stafforini DM, McIntyre TM, Prescott SM, et al. Anti-inflammatory HDL becomes pro-inflammatory during the acute phase response. Loss of protective effect of HDL against LDL oxidation in aortic wall cell cocultures. J Clin Invest. 1995;96:2758–2767. doi: 10.1172/JCI118345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Tang C, Liu Y, Kessler PS, Vaughan AM, Oram JF. The macrophage cholesterol exporter ABCA1 functions as an anti-inflammatory receptor. J Biol Chem. 2009;284:32336–32343. doi: 10.1074/jbc.M109.047472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Annema W, von Eckardstein A. High-density lipoproteins. Multifunctional but vulnerable protections from atherosclerosis. Circ J. 2013;77:2432–2448. doi: 10.1253/circj.cj-13-1025. [DOI] [PubMed] [Google Scholar]
- 50.Artl A, Marsche G, Lestavel S, Sattler W, Malle E. Role of serum amyloid A during metabolism of acute-phase HDL by macrophages. Arterioscler Thromb Vasc Biol. 2000;20:763–772. doi: 10.1161/01.atv.20.3.763. [DOI] [PubMed] [Google Scholar]
- 51.Vaisar T, Tang C, Babenko I, Hutchins P, Wimberger J, Suffredini AF, Heinecke JW. Inflammatory remodeling of the HDL proteome impairs cholesterol efflux capacity. J Lipid Res. 2015;56:1519–1530. doi: 10.1194/jlr.M059089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Annema W, Nijstad N, Tölle M, de Boer JF, Buijs RVC, Heeringa P, van der Giet M, Tietge UJF. Myeloperoxidase and serum amyloid A contribute to impaired in vivo reverse cholesterol transport during the acute phase response but not group IIA secretory phospholipase A(2) J Lipid Res. 2010;51:743–754. doi: 10.1194/jlr.M000323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Cai L, de Beer MC, de Beer FC, van der Westhuyzen DR. Serum amyloid A is a ligand for scavenger receptor class B type I and inhibits high density lipoprotein binding and selective lipid uptake. J Biol Chem. 2005;280:2954–2961. doi: 10.1074/jbc.M411555200. [DOI] [PubMed] [Google Scholar]
- 54.van der Westhuyzen DR, Cai L, de Beer MC, de Beer FC. Serum amyloid A promotes cholesterol efflux mediated by scavenger receptor B-I. J Biol Chem. 2005;280:35890–35895. doi: 10.1074/jbc.M505685200. [DOI] [PubMed] [Google Scholar]
- 55.Chiba T, Chang MY, Wang S, Wight TN, McMillen TS, Oram JF, et al. Serum amyloid A facilitates the binding of high-density lipoprotein from mice injected with lipopolysaccharide to vascular proteoglycans. Arterioscler Thromb Vasc Biol. 2011;31:1326–1332. doi: 10.1161/ATVBAHA.111.226159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.DiDonato JA, Huang Y, Aulak KS, Even-Or O, Gerstenecker G, Gogonea V, et al. Function and distribution of apolipoprotein A1 in the artery wall are markedly distinct from those in plasma. Circulation. 2013;128:1644–1655. doi: 10.1161/CIRCULATIONAHA.113.002624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Pennathur S, Bergt C, Shao B, Byun J, Kassim SY, Singh P, Green PS, McDonald TO, Brunzell J, Chait A, Oram JF, O'Brien K, Geary RL, Heinecke JW. Human atherosclerotic intima and blood of patients with established coronary artery disease contain high density lipoprotein damaged by reactive nitrogen species. J Biol Chem. 2004;279:42977–42983. doi: 10.1074/jbc.M406762200. [DOI] [PubMed] [Google Scholar]
- 58.Shao B, Pennathur S, Heinecke JW. Myeloperoxidase targets apolipoprotein A-I, the major high density lipoprotein protein, for site-specific oxidation in human atherosclerotic lesions. J Biol Chem. 2012;287:6375–6386. doi: 10.1074/jbc.M111.337345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Huang Y, DiDonato JA, Levison BS, Schmitt D, Li L, Wu Y, et al. An abundant dysfunctional apolipoprotein A1 in human atheroma. Nat Med. 2014;20:193–203. doi: 10.1038/nm.3459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Zheng L, Nukuna B, Brennan M-L, Sun M, Goormastic M, Settle M, Schmitt D, Fu X, Thomson L, Fox PL, Ischiropoulos H, Smith JD, Kinter M, Hazen SL. Apolipoprotein A-I is a selective target for myeloperoxidase-catalyzed oxidation and functional impairment in subjects with cardiovascular disease. J Clin Invest. 2004;114:529–541. doi: 10.1172/JCI21109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Zheng L, Settle M, Brubaker G, Schmitt D, Hazen SL, Smith JD, Kinter M. Localization of nitration and chlorination sites on apolipoprotein A-I catalyzed by myeloperoxidase in human atheroma and associated oxidative impairment in ABCA1-dependent cholesterol efflux from macrophages. J Biol Chem. 2005;280:38–47. doi: 10.1074/jbc.M407019200. [DOI] [PubMed] [Google Scholar]
- 62.Shao B, Cavigiolio G, Brot N, Oda MN, Heinecke JW. Methionine oxidation impairs reverse cholesterol transport by apolipoprotein A-I. Proc Natl Acad Sci. 2008;105:12224–12229. doi: 10.1073/pnas.0802025105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Shao B, Pennathur S, Pagani I, Oda MN, Witztum JL, Oram JF, Heinecke JW. Modifying apolipoprotein A-I by malondialdehyde, but not by an array of other reactive carbonyls, blocks cholesterol efflux by the ABCA1 pathway. J Biol Chem. 2010;285:18473–18484. doi: 10.1074/jbc.M110.118182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Skeggs JW, Morton RE. LDL and HDL enriched in triglyceride promote abnormal cholesterol transport. J Lipid Res. 2002;43:1264–1274. [PubMed] [Google Scholar]
- 65.Fournier N, Francone O, Rothblat G, Goudouneche D, Cambillau M, Kellner-Weibel G, Robinet P, Royer L, Moatti N, Simon A, Paul JL. Enhanced efflux of cholesterol from ABCA1-expressing macrophages to serum from type IV hypertriglyceridemic subjects. Atherosclerosis. 2003;171:287–293. doi: 10.1016/j.atherosclerosis.2003.08.011. [DOI] [PubMed] [Google Scholar]
- 66.Yassine HN, Belopolskaya A, Schall C, Stump CS, Lau SS, Reaven PD. Enhanced cholesterol efflux to HDL through the ABCA1 transporter in hypertriglyceridemia of type 2 diabetes. Metabolism. 2014;63:727–734. doi: 10.1016/j.metabol.2014.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Greene DJ, Skeggs JW, Morton RE. Elevated triglyceride content diminishes the capacity of high density lipoprotein to deliver cholesteryl esters via the scavenger receptor class B type I (SR-BI) J Biol Chem. 2001;276:4804–4811. doi: 10.1074/jbc.M008725200. [DOI] [PubMed] [Google Scholar]
- 68.Moerland M, Samyn H, van Gent T, Jauhiainen M, Metso J, van Haperen R, Grosveld F, van Tol A, de Crom R. Atherogenic, enlarged, and dysfunctional HDL in human PLTP/apoA-I double transgenic mice. J Lipid Res. 2007;48:2622–2631. doi: 10.1194/jlr.M700020-JLR200. [DOI] [PubMed] [Google Scholar]
- 69.Charakida M, Besler C, Batuca JR, Wang G. Vascular abnormalities, paraoxonase in primary antiphospholipid syndrome. JAMA. 2009;302:1210–1217. doi: 10.1001/jama.2009.1346. [DOI] [PubMed] [Google Scholar]
- 70.Speer T, Rohrer L, Blyszczuk P, Shroff R, Kuschnerus K, Kränkel N, Kania G, Zewinger S, Akhmedov A, Shi Y, Martin T, Perisa D, Winnik S, Müller MF, Sester U, Wernicke G, Jung A, Gutteck U, Eriksson U, Geisel J, Deanfield J, von Eckardstein A, Lüscher TF, Fliser D, Bahlmann FH, Landmesser U. Abnormal high-density lipoprotein induces endothelial dysfunction via activation of Toll-like receptor-2. Immunity. 2013;38:754–768. doi: 10.1016/j.immuni.2013.02.009. [DOI] [PubMed] [Google Scholar]
- 71.Besler C, Heinrich K, Rohrer L, Doerries C, Riwanto M, Shih DM, Chroni A, Yonekawa K, Stein S, Schaefer N, Mueller M, Akhmedov A, Daniil G, Manes C, Templin C, Wyss C, Maier W, Tanner FC, Matter CM, Corti R, Furlong C, Lusis AJ, von Eckardstein A, Fogelman AM, Lüscher TF, Landmesser U. Mechanisms underlying adverse effects of HDL on eNOS-activating pathways in patients with coronary artery disease. J Clin Invest. 2011;121:2693–2708. doi: 10.1172/JCI42946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Huang Y, Wu Z, Riwanto M, Gao S, Levison BS, Gu X, Fu X, Wagner MA, Besler C, Gerstenecker G, Zhang R, Li XM, DiDonato AJ, Gogonea V, Tang WHW, Smith JD, Plow EF, Fox PL, Shih DM, Lusis AJ, Fisher EA, DiDonato JA, Landmesser U, Hazen SL. Myeloperoxidase, paraoxonase-1, and HDL form a functional ternary complex. J Clin Invest. 2013;123:3815–3828. doi: 10.1172/JCI67478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Chang F-J, Yuan H-Y, Hu X-X, Ou Z-J, Fu L, Lin Z-B, Wang ZP, Wang SM, Zhou L, Xu YQ, Wang CP, Xu Z, Zhang X, Zhang CX, Ou JS. High density lipoprotein from patients with valvular heart disease uncouples endothelial nitric oxide synthase. J Mol Cell Cardiol. 2014;74:209–219. doi: 10.1016/j.yjmcc.2014.05.015. [DOI] [PubMed] [Google Scholar]
- 74.Baldus S, Eiserich JP, Mani A, Castro L, Figueroa M, Chumley P, Ma W, Tousson A, White CR, Bullard DC, Brennan ML, Lusis AJ, Moore KP, Freeman BA. Endothelial transcytosis of myeloperoxidase confers specificity to vascular ECM proteins as targets of tyrosine nitration. J Clin Invest. 2001;108:1759–1770. doi: 10.1172/JCI12617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Navab M, Hama SY, Hough GP, Subbanagounder G, Reddy ST, Fogelman AM. A cell-free assay for detecting HDL that is dysfunctional in preventing the formation of or inactivating oxidized phospholipids. J Lipid Res. 2001:1308–17. [PubMed]
- 76.Zewinger S, Drechsler C, Kleber ME, Dressel A, Riffel J, Triem S, et al. Serum amyloid A: high-density lipoproteins interaction and cardiovascular risk. Eur Heart J. 2015;36:3007–3016. doi: 10.1093/eurheartj/ehv352. [DOI] [PubMed] [Google Scholar]
- 77.Zewinger S, Kleber ME, Rohrer L, Lehmann M, Triem S, Jennings RT, Petrakis I, Dressel A, Lepper PM, Scharnagl H, Ritsch A, Thorand B, Heier M, Meisinger C, de las Heras Gala T, Koenig W, Wagenpfeil S, Schwedhelm E, Böger RH, Laufs U, von Eckardstein A, Landmesser U, Lüscher TF, Fliser D, März W, Meinitzer A, Speer T. Symmetric dimethylarginine, high-density lipoproteins and cardiovascular disease. Eur Heart J. 2017;38:1597–1607. doi: 10.1093/eurheartj/ehx118. [DOI] [PubMed] [Google Scholar]
- 78.Corsetti JP, Gansevoort RT, Sparks CE, Dullaart RPF. Inflammation reduces HDL protection against primary cardiac risk. Eur J Clin Investig. 2010;40:483–489. doi: 10.1111/j.1365-2362.2010.02287.x. [DOI] [PubMed] [Google Scholar]
- 79.Jensen MK, Rimm EB, Furtado JD, Sacks FM. Apolipoprotein C-III as a potential modulator of the association between HDL-cholesterol and incident coronary heart disease. J Am Heart Assoc. 2012;1. [DOI] [PMC free article] [PubMed]
- 80.Zhang J, Xu J, Wang J, Wu C, Xu Y, Wang Y, Deng F, Wang Z, Chen X, Wu M, Chen Y. Prognostic usefulness of serum cholesterol efflux capacity in patients with coronary artery disease. Am J Cardiol. 2016;117:508–514. doi: 10.1016/j.amjcard.2015.11.033. [DOI] [PubMed] [Google Scholar]
- 81.Griffiths K, Pazderska A, Ahmed M, McGowan A, Maxwell AP, McEneny J, Gibney J, McKay GJ. Type 2 diabetes in young females results in increased serum amyloid A and changes to features of high density lipoproteins in both HDL2 and HDL3. J Diabetes Res. 2017;2017:1–9. doi: 10.1155/2017/1314864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Vergès B. Lipid modification in type 2 diabetes: the role of LDL and HDL. Fundam Clin Pharmacol. 2009;23:681–685. doi: 10.1111/j.1472-8206.2009.00739.x. [DOI] [PubMed] [Google Scholar]
- 83.Vaisar T, Couzens E, Hwang A, Russell M, Barlow CE, DeFina LF, et al. Type 2 diabetes is associated with loss of HDL endothelium protective functions. PLoS One. 2018;13:e0192616. doi: 10.1371/journal.pone.0192616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Farbstein D, Levy AP. HDL dysfunction in diabetes: causes and possible treatments. Expert Rev Cardiovasc Ther. 2012;10:353–361. doi: 10.1586/erc.11.182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Apro J, Tietge UJF, Dikkers A, Parini P, Angelin B, Rudling M. Impaired cholesterol efflux capacity of high-density lipoprotein isolated from interstitial fluid in type 2 diabetes mellitus—brief report. Arterioscler Thromb Vasc Biol. 2016;36:787–791. doi: 10.1161/ATVBAHA.116.307385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Nakhjavani M, Esteghamati A, Esfahanian F, Ghanei A, Rashidi A, Hashemi S. HbA1c negatively correlates with LCAT activity in type 2 diabetes. Diabetes Res Clin Pract. 2008;81:38–41. doi: 10.1016/j.diabres.2008.01.018. [DOI] [PubMed] [Google Scholar]
- 87.Holzer M, Birner-Gruenberger R, Stojakovic T, El-Gamal D, Binder V, Wadsack C, et al. Uremia alters HDL composition and function. J Am Soc Nephrol. 2011;22:1631–1641. doi: 10.1681/ASN.2010111144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Marsche G, Frank S, Hrzenjak A, Holzer M, Dirnberger S, Wadsack C, Scharnagl H, Stojakovic T, Heinemann A, Oettl K. Plasma-advanced oxidation protein products are potent high-density lipoprotein receptor antagonists in vivo. Circ Res. 2009;104:750–757. doi: 10.1161/CIRCRESAHA.108.193169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Ganjali S, Dallinga-Thie GM, Simental-Mendía LE, Banach M, Pirro M, Sahebkar A. HDL functionality in type 1 diabetes. Atherosclerosis. 2017;267:99–109. doi: 10.1016/j.atherosclerosis.2017.10.018. [DOI] [PubMed] [Google Scholar]
- 90.Gómez Rosso L, Lhomme M, Meroño T, Sorroche P, Catoggio L, Soriano E, Saucedo C, Malah V, Dauteuille C, Boero L, Lesnik P, Robillard P, John Chapman M, Brites F, Kontush A. Altered lipidome and antioxidative activity of small, dense HDL in normolipidemic rheumatoid arthritis: relevance of inflammation. Atherosclerosis. 2014;237:652–660. doi: 10.1016/j.atherosclerosis.2014.09.034. [DOI] [PubMed] [Google Scholar]
- 91.Charles-Schoeman C, Watanabe J, Lee YY, Furst DE, Amjadi S, Elashoff D, Park G, McMahon M, Paulus HE, Fogelman AM, Reddy ST. Abnormal function of high-density lipoprotein is associated with poor disease control and an altered protein cargo in rheumatoid arthritis. Arthritis Rheum. 2009;60:2870–2879. doi: 10.1002/art.24802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.McMahon M, Grossman J, Skaggs B, Fitzgerald J, Sahakian L, Ragavendra N, et al. Dysfunctional proinflammatory high-density lipoproteins confer increased risk of atherosclerosis in women with systemic lupus erythematosus. Arthritis Rheum. 2009;60:2428–2437. doi: 10.1002/art.24677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Manjunatha S, Distelmaier K, Dasari S, Carter RE, Kudva YC, Nair KS. Functional and proteomic alterations of plasma high density lipoproteins in type 1 diabetes mellitus. Metabolism. 2016;65:1421–1431. doi: 10.1016/j.metabol.2016.06.008. [DOI] [PubMed] [Google Scholar]
- 94.Machado-Lima A, Iborra RT, Pinto RS, Sartori CH, Oliveira ER, Nakandakare ER, Stefano JT, Giannella-Neto D, Corrêa-Giannella MLC, Passarelli M. Advanced glycated albumin isolated from poorly controlled type 1 diabetes mellitus patients alters macrophage gene expression impairing ABCA-1-mediated reverse cholesterol transport. Diabetes Metab Res Rev. 2013;29:66–76. doi: 10.1002/dmrr.2362. [DOI] [PubMed] [Google Scholar]
- 95.Mackness MI, Harty D, Bhatnagar D, Winocour PH, Arrol S, Ishola M, Durrington PN. Serum paraoxonase activity in familial hypercholesterolaemia and insulin-dependent diabetes mellitus. Atherosclerosis. 1991;86:193–199. doi: 10.1016/0021-9150(91)90215-o. [DOI] [PubMed] [Google Scholar]
- 96.Savu O, Serafinceanu C, Grajdeanu IV, Iosif L, Gaman L, Stoian I. Paraoxonase lactonase activity, inflammation and antioxidant status in plasma of patients with type 1 diabetes mellitus. J Int Med Res. 2014;42:523–529. doi: 10.1177/0300060513516287. [DOI] [PubMed] [Google Scholar]
- 97.Denimal D, Pais de Barros J-P, Petit J-M, Bouillet B, Vergès B, Duvillard L. Significant abnormalities of the HDL phosphosphingolipidome in type 1 diabetes despite normal HDL cholesterol concentration. Atherosclerosis. 2015;241:752–760. doi: 10.1016/j.atherosclerosis.2015.06.040. [DOI] [PubMed] [Google Scholar]
- 98.Costacou T, Evans RW, Orchard TJ. High-density lipoprotein cholesterol in diabetes: is higher always better? J Clin Lipidol. 2011;5:387–394. doi: 10.1016/j.jacl.2011.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.O’Neill F, Charakida M, Topham E, McLoughlin E, Patel N, Sutill E, et al. Anti-inflammatory treatment improves high-density lipoprotein function in rheumatoid arthritis. Heart. 2017;103:766–773. doi: 10.1136/heartjnl-2015-308953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Charles-Schoeman C, Gugiu GB, Ge H, Shahbazian A, Lee YY, Wang X, Furst DE, Ranganath VK, Maldonado M, Lee T, Reddy ST. Remodeling of the HDL proteome with treatment response to abatacept or adalimumab in the AMPLE trial of patients with rheumatoid arthritis. Atherosclerosis. 2018;275:107–114. doi: 10.1016/j.atherosclerosis.2018.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Nazir MA. Prevalence of periodontal disease, its association with systemic diseases and prevention. Int J Health Sci (Qassim) 2017;11:72–80. [PMC free article] [PubMed] [Google Scholar]
- 102.Rohatgi A, Khera A, Berry JD, Givens EG, Ayers CR, Wedin KE, et al. HDL cholesterol efflux capacity and incident cardiovascular events. N Engl J Med. 2014;317:2383–2393. doi: 10.1056/NEJMoa1409065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Saleheen D, Scott R, Javad S, Zhao W, Rodrigues A, Picataggi A, Lukmanova D, Mucksavage ML, Luben R, Billheimer J, Kastelein JJP, Boekholdt SM, Khaw KT, Wareham N, Rader DJ. Association of HDL cholesterol efflux capacity with incident coronary heart disease events: a prospective case-control study. Lancet Diabetes Endocrinol. 2015;3:507–513. doi: 10.1016/S2213-8587(15)00126-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Ritsch A, Scharnagl HMW. HDL cholesterol efflux capacity and cardiovascular events. N Engl J Med. 2015;372:1869–1872. doi: 10.1056/NEJMc1503139. [DOI] [PubMed] [Google Scholar]
- 105.Khera AV, Cuchel M, de la Llera-Moya M, Rodrigues A, Burke MF, Jafri K, French BC, Phillips JA, Mucksavage ML, Wilensky RL, Mohler ER, Rothblat GH, Rader DJ. Cholesterol efflux capacity, high-density lipoprotein function, and atherosclerosis. N Engl J Med. 2011;364:127–135. doi: 10.1056/NEJMoa1001689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Shah PK, Yano J, Reyes O, Chyu KY, Kaul S, Bisgaier CL, Drake S, Cercek B. High-dose recombinant apolipoprotein A-I(milano) mobilizes tissue cholesterol and rapidly reduces plaque lipid and macrophage content in apolipoprotein e-deficient mice. Potential implications for acute plaque stabilization. Circulation. 2001;103:3047–3050. doi: 10.1161/hc2501.092494. [DOI] [PubMed] [Google Scholar]
- 107.Nissen SE, Tsunoda T, Tuzcu EM, Schoenhagen P, Cooper CJ, Yasin M, Eaton GM, Lauer MA, Sheldon WS, Grines CL, Halpern S, Crowe T, Blankenship JC, Kerensky R. Effect of recombinant ApoA-I Milano on coronary atherosclerosis in patients with acute coronary syndromes. JAMA. 2003;290:2292. doi: 10.1001/jama.290.17.2292. [DOI] [PubMed] [Google Scholar]
- 108.Nicholls SJ, Andrews J, Kastelein JJP, Merkely B, Nissen SE, Ray KK, Schwartz GG, Worthley SG, Keyserling C, Dasseux JL, Griffith L, Kim SW, Janssan A, di Giovanni G, Pisaniello AD, Scherer DJ, Psaltis PJ, Butters J. Effect of serial infusions of CER-001, a pre-β high-density lipoprotein mimetic, on coronary atherosclerosis in patients following acute coronary syndromes in the CER-001 atherosclerosis regression acute coronary syndrome trial. JAMA Cardiol. 2018;3:815. doi: 10.1001/jamacardio.2018.2121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Nicholls SJ, Puri R, Ballantyne CM, Jukema JW, Kastelein JJP, Koenig W, Wright RS, Kallend D, Wijngaard P, Borgman M, Wolski K, Nissen SE. Effect of infusion of high-density lipoprotein mimetic containing recombinant apolipoprotein A-I Milano on coronary disease in patients with an acute coronary syndrome in the MILANO-PILOT trial. JAMA Cardiol. 2018;3:806. doi: 10.1001/jamacardio.2018.2112. [DOI] [PMC free article] [PubMed] [Google Scholar]