Abstract
Background
As reports on the influence of cigarette smoking, an important cardiovascular risk factor, on platelet ADP–P2Y12 receptor inhibitors lack consistency, we aimed to assess the effectiveness and safety of platelet ADP–P2Y12 receptor inhibitors influenced by smoking status.
Methods and Results
PubMed, Web of Science, EMBASE, Clinical Trials, and the Cochrane Library were searched from inception until June 2018. Among the 5076 citations retrieved, 22 studies, including 163 011 patients with or without percutaneous coronary intervention, were included for meta‐analysis. Compared with nonsmokers within the first year of follow‐up, the reductions of stroke and major adverse cardiovascular event rate were 18% (P=0.008) and 26% (P=0.02), respectively. A 20% reduction in stroke (P=0.02) and a 34% reduction in major adverse cardiovascular event (P=0.0001) rates were observed in smoking patients without percutaneous coronary intervention. No significant difference was observed in clinical outcome rates among prasugrel, ticagrelor, and clopidogrel in different smoking status. No significant difference was found in myocardial infarction and bleeding event incidence between current smokers and nonsmokers.
Conclusions
We concluded that current smokers had a lower incidence of major adverse cardiovascular events and stroke events than did nonsmokers, particularly in the early period (1 year) and among patients without percutaneous coronary intervention. However, because of the lack of original adjusted data, smoker's paradox still needs to consider the impact of age and other covariates. Thus, a differential risk‐benefit evaluation should be considered, according to different smoking status, patient conditions, and therapy time points.
Keywords: effectiveness, meta‐analysis, platelet aggregation inhibitors, safety, smoking
Subject Categories: Meta Analysis, Mortality/Survival, Thrombosis, Lifestyle, Treatment
Clinical Perspective
What Is New?
A systematic review and meta‐analysis was conducted to evaluate the effectiveness and safety of platelet ADP–P2Y12 receptor inhibitors influenced by smoking status.
This meta‐analysis concluded that current smokers had a lower incidence of major adverse cardiovascular events and stroke events than did nonsmokers, particularly in the early period (1 year) and among patients without percutaneous coronary intervention.
What Are the Clinical Implications?
A differential risk‐benefit evaluation should be considered, according to different smoking status, patient conditions, and therapy time points.
In addition, future studies can clearly define smoking status and record the specific intake of tobacco to better explore the effects of smoking on clinical outcomes.
Introduction
Atherothrombosis is the most common cause of the development of acute coronary syndrome and plays a critical role in complications occurring around the coronary stent implantation.1 Myocardial infarction (MI) and ischemic stroke were estimated to cause >14 million deaths/year.2
P2Y12 receptor inhibitors reduce the risk for repeated MI, stroke, cardiovascular death, and stent thrombosis in patients with acute coronary syndrome or undergoing coronary stent implantation,3, 4, 5 which has been the most attractive target of antiplatelet drug development because of its central role in platelet activation and aggregation.6 Three available oral P2Y12 antagonists were used in the contemporary clinical practice (namely, clopidogrel, ticagrelor, and prasugrel). Recently, new oral P2Y12 antagonists, ticagrelor and prasugrel, provided more consistent and powerful platelet inhibition than clopidogrel,7, 8 and their safety and effectiveness in Asian patients have been demonstrated in recent clinical trials.9, 10, 11
Multiple reasons can influence an individual's response to P2Y12 antagonists that can be consequent to genetic, clinical, or cellular factors.1 Cigarette smoking was reported to influence the inception and progression of atherosclerosis12 and poses hazards to cardiovascular and cerebrovascular systems, which can trigger thrombotic complications, such as MI, ischemic stroke, and cardiovascular death.13 However, the interaction between cigarette smoking and cardiovascular outcomes of oral P2Y12 inhibitors is complex.14 Several studies have reported reduced recurrence of cardiovascular events and improved survival in smokers with antithrombotic therapy after an index cardiovascular event, a phenomenon called “smoker's paradox.”15, 16 One potential mechanism to explain the smoker's paradox is the induction of cytochrome 1A2 and 2B6 activity by cigarette smoking,17, 18 resulting in greater clopidogrel active metabolite generation, an enhanced pharmacodynamic effect, and less ischemic event occurrence in current smokers than nonsmokers. Polycyclic aromatic hydrocarbons, products of incomplete combustion present in tobacco smoke, significantly increase cytochrome 1A2 activity. Cytochrome 1A2 is one of the major hepatic cytochromes and plays an important role in the biotransformation of clopidogrel.19 Cigarette smoking significantly impacts the pharmacokinetics of the cytochrome 1A2 drugs,20 and it was reported that cytochrome 1A2 inducers were associated with a reduced rate of clopidogrel nonresponsiveness in patients with ischemic stroke.21 Cytochrome 2B6 expression is upregulated in smokers, and cigarette smoke extract induced cytochrome 2B6 mRNA expression in hepatocytes.17 Because cytochrome 2B6 is involved in both steps of clopidogrel oxidation and in the second step in prasugrel metabolism, the possible contribution of smoking‐induced cytochrome 2B6 activity to thienopyridine metabolism should be further explored. Among the patients at high vascular risk in the CHARISMA (Clopidogrel for High Atherothrombotic Risk and Ischemic Stabilization, Management, and Avoidance) trial, a reduction in mortality associated with clopidogrel use was only observed among smokers in a post hoc analysis.15 Similarly, in the CLARITY–TIMI (Clopidogrel as Adjunctive Reperfusion Therapy–Thrombolysis in Myocardial Infarction) 28 trial of patients presenting with ST‐segment–elevation MI, smokers had better clinical outcomes.16 In contrast to the above reports, Zhang et al22 suggested that no significant difference was observed between smoking and bleeding events in patients who used antiplatelet agents. In the SYNTAX (Synergy Between Percutaneous Coronary Intervention With Taxus and Cardiac Surgery) trial of a 5‐year follow‐up study,23 researchers found that among patients with complex cardiovascular disease undergoing revascularization with either percutaneous coronary intervention (PCI) or coronary artery bypass grafting, those who are smokers have a higher incidence of experiencing adverse outcomes, such as MI, than those who never smoked or stopped smoking.
PCI for unblocking a narrowed coronary artery is a widely used technique in treating patients with angina or an acute coronary event, the most common form of myocardial revascularization.24, 25 The average annual growth of PCI surgery was ≈30% to 40%, and the total number of PCI surgeries was 666 495 in 2016 in the mainland.26 In addition, most studies23, 24, 25, 26, 27, 28 were performed in the era of PCI without stents or with bare‐metal stents, providing little insight into the impact of smoking in patients undergoing revascularization. Thus, the effect of smoking on clinical events in patients undergoing PCI remains undefined.
On the basis of the previously mentioned unsolved problems, we conducted a meta‐analysis to examine the association between smoking status and the effectiveness and safety of P2Y12 receptor inhibitors in patients with or without PCI. We hypothesize that smoking is a protective factor in patients using P2Y12 inhibitors. Thus, this study was conducted to explore the following issues: (1) to comprehensively evaluate the effectiveness and safety of platelet ADP–P2Y12 receptor inhibitors influenced by smoking status during different follow‐up periods; (2) to compare the clinical outcomes in patients with and without PCI; (3) and to identify studies on new oral P2Y12 receptor inhibitors to evaluate the association comprehensively.
Methods
The data, analytic methods, and study materials will be made available to other researchers for purposes of reproducing the results or replicating the procedure. Researchers can contact us by e‐mail.
Search Strategy and Study Selection
A search of the medical literature was conducted using PubMed, Web of Science, EMBASE, Clinical Trials, and the Cochrane Library from inception to June 2018. The search terms used in these databases are as follows: “clopidogrel,” “prasugrel,” “ticagrelor,” “P2Y12,” “smoking,” “smoker,” and “tobacco” (Data S1 Search strategies in PubMed).
Citations recalled were initially screened with title and abstract, and then 2 investigators (Z.L. and Q. Xiang) retrieved and assessed the full texts of potentially relevant studies for their eligibility. Any disagreement between investigators was resolved by consensus. No language restrictions were applied, and both full‐text articles and abstracts were considered.
A study was included for analysis if it met the following prespecified criteria: (1) enrollment of patients using ADP–P2Y12 receptor inhibitors, including clopidogrel, prasugrel, or ticagrelor treatment; (2) patients were recorded with different smoking status; and (3) clinical outcomes, such as MI, stroke, death, bleeding, or major adverse cardiovascular events (MACEs), were reported and compared with different smoking status. The exclusion criteria were as follows: (1) patients not treated with ADP–P2Y12 receptor inhibitors; (2) studies reported without clinical outcomes, or clinical outcomes were not compared with different smoking status; and (3) editorials, commentary letters, reviews, case reports/series, economic or modeling studies, extensions of previously completed studies, and studies that did not report the results needed for these analyses.
Outcome Assessment
The primary efficacy/effectiveness outcomes were stroke and/or MI. The primary safety outcomes were death, bleeding, or MACE. MI was defined according to the recommendations of the European Society of Cardiology/American College of Cardiology Foundation/American Heart Association/World Heart Federation task force.27 Bleeding was defined on the basis of the International Society on Thrombosis and Haemostasis definition (ie, fatal bleeding, symptomatic bleeding in a critical area or organ, and/or bleeding causing a decrease in hemoglobin level of ≥20 g/L or leading to transfusion of ≥2 units of whole blood or red blood cells)28 and any bleeding that required emergency department visits and/or hospitalizations commonly used in observational studies. Moreover, all‐cause and cardiovascular deaths were all considered as death in the study. MACEs were defined as a composite of death from cardiovascular causes, spontaneous MI, unstable angina, stent thrombosis, target vessel revascularization, non–target vessel revascularization, and ischemic stroke.
Smoking status was classified into current smokers, former smokers, and never smokers. Nonsmokers were defined as never smokers plus former smokers in this study. For studies that presented current smoker data stratified by smoking intensity, these subgroup data were combined using a formula described elsewhere.29
Data Extraction and Quality Assessment
Details were extracted with a prespecified table on the study and patient characteristics, smoking status and definition, drug regimens, clinical outcomes or parameters assessed, and follow‐up time, among others. The Newcastle‐Ottawa Scale was used to assess the methodological quality of included studies.30, 31 In brief, eligible studies were assessed on the basis of 3 perspectives: the selection of study groups, the comparability of the groups, and the ascertainment of outcomes or test results of interest in studies. Each study with Newcastle‐Ottawa Scale scores ≥7 was considered as a high‐quality study, whereas studies with Newcastle‐Ottawa Scale scores <7 were considered as low‐quality studies. Assessment was performed independently by 2 investigators, with disagreements resolved by discussion.
Data Synthesis and Statistical Analyses
Data were pooled using a random‐ or fixed‐effects model to obtain a more conservative estimate of smoking effects on clinical outcomes of platelet ADP–P2Y12 receptor inhibitors.
Measures of association in the form of odds ratios (ORs) were pooled, and the 95% CI was selected as the summary statistic. The I2 statistic and the P value for Q statistics were used to estimate the percentage of variability across studies that was attributable to heterogeneity. The P value for statistical significance was 0.05 in all cases, except the test for heterogeneity, in which the level was set at 0.10.
Review Manager Version 5.3 (RevMan for Windows; Nordic Cochrane Centre, Copenhagen, Denmark) was used to generate forest plots of pooled ORs for primary outcomes with 95% CIs. Sensitivity analyses were performed by excluding one study at a time and thereafter computing the OR of the remaining studies. We also explored the subgroup analyses to better assess between‐study variability. These variables included different follow‐up times, patients with or without PCI, age, race, and different P2Y12 receptor inhibitor therapies. Because age is not a normal distribution between each study, median age (63 years old) was selected for subgroup analysis. Population race can be divided into Asian, white, and the mixed races.
Sensitivity analysis, publication bias assessment, and meta‐regression were performed using STATA software version 15.1 (Stata Corporation, College Station, TX).
This review and meta‐analysis was conducted and reported according to the Preferred Reporting Items for Systematic Reviews and Meta‐Analysis and Meta‐Analysis of Observational Studies in Epidemiology statements32, 33 and was registered with PROSPERO (International Prospective Register of Systematic Reviews) (CRD42018100183).
Results
The search strategy identified 5076 citations. The titles and abstracts were reviewed, and 117 articles that were thought to be potentially eligible for inclusion were retrieved and evaluated. After screening the 117 studies for eligibility, 2215, 16, 22, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52 reporting the effectiveness and safety outcomes were included in the meta‐analysis. A flow diagram of study identification and selection is shown in Figure 1.
Figure 1.
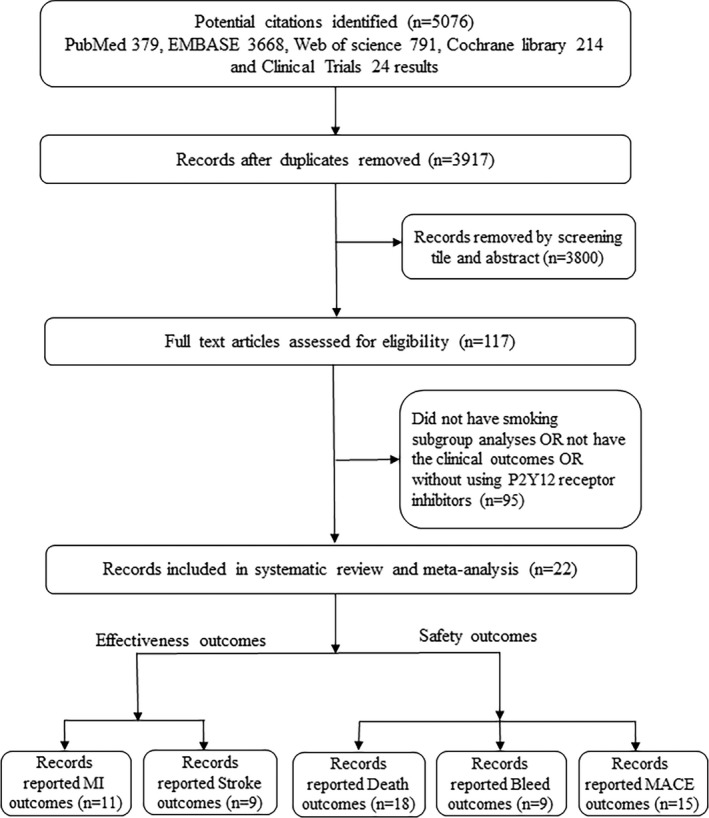
Flow diagram of study identification and selection. MACE indicates major adverse cardiovascular event; MI, myocardial infarction.
For the effectiveness outcomes, 11 studies1 with a total of 83 677 patient‐reported MI events associated with different smoking status and 9 studies22, 37, 38, 41, 42, 43, 47, 49, 53 with 59 892 patient‐reported stroke outcomes were included. For the safety outcomes, 18 studies2 reported death events (115 156 patients), 9 studies3 reported bleeding events (93 744 patients), and 15 studies4 reported MACE outcomes (107 188 patients); all of them were categorized by smoking status. In addition, different platelet ADP–P2Y12 receptor inhibitor therapies were compared in 3 studies45, 49, 50 based on smoking status.
Among the patient population involved, the prevalence of current smoking ranged from 19% to 62%. Compared with nonsmokers, current smokers were approximately diagnosed with cardiovascular diseases 10 years younger (mean age, 65 versus 55 years) and more men were found (men account for ≈70%‐96%). Patients with cardiovascular disease with or without stents were involved in the study, and all were treated with ADP–P2Y12 receptor inhibitors, including clopidogrel at a dosage of 75 mg/d, prasugrel, 5 to 10 mg/d, or ticagrelor, 90 mg twice daily, for at least 1 month. Clinical outcome follow‐up times ranged from in hospital to 5 years; therefore, we conducted subgroup analyses to comprehensively explore the influence of smoking status on clinical outcomes after taking antiplatelet drugs.
Details of the included studies are summarized in Tables 1 and 2, which list the demographic characteristics of the participants, smoking status, medication therapy, clinical outcomes, and follow‐up time, among others. On quality assessment, 4 studies had a score of 8, 9 studies had a score of 7, 8 studies had a score of 6, and the remaining 1 study had a score of 5 (Table S1 Quality assessment of the included studies by Newcastle‐Ottawa Scale).
Table 1.
Characteristics of Studies Included in the Systematic Review and Meta‐Analysis
| Study | No. Analyzed | Population | Country | Study Design | Age, y | Male sex, % | Drug Regimen | Clinical Outcomes | Follow‐Up Time | ||
|---|---|---|---|---|---|---|---|---|---|---|---|
| CS | NS | CS | NS | ||||||||
| Sibbald et al, 201034 | 24 456 | Patients with ACS | Multicenter | Prospective cohort study | 56±11.1 | 69±13.3 | 78.6 | 66.6 | Clopidogrel, 75 mg/d | Death, MI, bleeding | 6 mo |
| Kang et al, 201335 | 24 257 | Patients with AMI | Korea | Prospective cohort study | 54.5±12.1 | 63.8±11.4 | 92.1 | 54.4 | DAT | Death, MI, MACE | 1 y |
| Zhang et al, 201636 | 600 | Patients with AMI | China | Prospective cohort study | 62.1±6.7 | 72 | DAT | Death | 5 y | ||
| Kim et al, 201437 | 457 | Patients with PCI | Korea | Prospective study | 59.1±10.0 | 64.0±8.6 | 95.8 | 61.1 | DAT | Death, MI, stroke, MACE | 2 y |
| Zhang et al, 201722 | 623 | Patients with CVD | China | Retrospective study | 58.3±11.7 | 64.9±12.7 | 95.8 | 40.8 | Clopidogrel, 75 mg/d | Stroke, MI, death, bleeding, MACE | 1 y |
| Ashby et al, 200238 | 5592 | Patients with PCI | United States | Retrospective cohort study | 57.3±11.2 | 65.5±11.0 | 71.5 | 67.8 | Clopidogrel, 75 mg/d | Death, MI, stroke | In hospital |
| Tan et al, 201439 | 1051 | Patients with STEMI | Canada | Retrospective cohort study | 54±8.1 | 60±14.1 | 82.4 | 77.4 | Clopidogrel, 70 g/d | Death, MACE | 1 mo |
| Adamo et al, 201540 | 1962 | Patients with PCI | Italy | Retrospective cohort study | 60.3±11.6 | 71.4±10.2 | 82.1 | 75.2 | DAT | MACE | 2 y |
| Bossard et al, 201741 | 17 263 | Patients with ACS and PCI | Multicenter | Prospective cohort study | 55.3±9.9 | 64.6±11.2 | 84 | 70.5 | Clopidogrel, LD+75 mg/d | Death, MI, bleeding, stroke, MACE | 1 mo |
| Ovbiagele et al, 201742 | 5170 | Patients with CVD | China | Retrospective cohort study | NA | DAT | Stroke | 3 mo | |||
| Kodaira et al, 201643 | 6195 | Patients with ACS and PCI | Japan | Retrospective cohort study | 62.0±11.7 | 70.6±11.0 | 91 | 68.7 | Clopidogrel, 75 mg/d | Death, MI, bleeding, stroke, MACE | In hospital |
| Siasos et al, 201644 | 229 | Patients with stable CAD | Greece | Prospective cohort study | 58±10 | 64±10 | 90 | 90 | DAT | Death | 3 mo |
| Chandrasekhar et al, 2015 45 | 19 906 | Patients with ACS and PCI | United States | Retrospective cohort study | NA | Clopidogrel/prasugrel | MACE | 1 y | |||
| Wakabayashi et al, 201146 | 1424 | Patients with AMI and PCI | United States | Prospective cohort study | 55.6±10.2 | 66.9±12.5 | 72.4 | 63.5 | Clopidogrel, LD+75 mg/d | Death, MI, MACE | 1 mo |
| Ferreiro et al, 201447 | 9534 | Patients with CVD | United States | Retrospective cohort study | 58.3±10.8 | 64.1±10.8 | 76.4 | 70.8 | Clopidogrel, 75 mg/d | Stroke, bleeding | 3 y |
| Lee et al, 201648 | 1527 | Patients with PCI | Keron, Nepal | Retrospective cohort study | 57.9±11.2 | 65.4±10.4 | 91 | 52.2 | DAT | Death, MI, MACE | 1 y |
| Cornel et al, 201449 | 7062 | Patients with UA/NSTEMI | Multicenter | Retrospective cohort study | 59±8.9 | 64±8.1 | 79.6 | 59.2 | Prasugrel, 10/5 mg/d; clopidogrel, 75 mg/d | MI, stroke, death, bleeding, MACE | 2.5 y |
| Desai et al, 200916 | 1726 | Patients with STEMI | Multicenter | Retrospective cohort study | 53.1±9.2 | 61.1±9.6 | 83.9 | 77 | Clopidogrel, LD+75 mg/d | MACE, death | 1 mo |
| Cornel et al, 201250 | 18 610 | Patients with STE/NSTE ACS | Multicenter | Retrospective cohort study | 56±9.6 | 66±11.1 | 81.7 | 65.9 | Ticagrelor, LD+90 mg twice daily; clopidogrel, LD+75 mg/d | Death, bleeding, MACE | 9.1 mo |
| Ciccarelli et al, 201751 | 713 | Patients with STEMI and PCI | Italy | Retrospective cohort study | NA | 77.9 | 72.9 | DAT | Stroke, death, MACE | 1.5 y | |
| Berger et al, 200915 | 6071 | Patients with CVD | Multicenter | Retrospective cohort study | 60.4±8.5 | 64.9±9.6 | 75.4 | 71.7 | Clopidogrel, 75 mg/d | Death, bleeding | 2 y |
| Weisz et al, 201352 | 8583 | Patients with PCI | United States | Retrospective cohort study | 59.6±9.6 | 65.6±10.4 | NA | DAT | Death, MI, bleeding, MACE | 1 y | |
ACS indicates acute coronary syndrome; AMI, acute MI; CAD, coronary artery disease; CS, current smoker; CVD, cardiovascular disease; DAT, dual antiplatelet therapy, clopidogrel (75 mg/d) and aspirin (100 mg/d), multicenter (at least 3 countries), including the United States, Canada, Poland, Scotland, India, France, and Australia; LD, loading dose; MACE, major adverse cardiovascular event; MI, myocardial infarction; NA, no valid data can be obtained; NS, never smoker; NSTE, non‐STE; NSTEMI, non‐STEMI; PCI, percutaneous coronary intervention; STE, ST‐segment elevation; STEMI, STE myocardial infarction.
Table 2.
Definition of Smoking Status and Quality Assessment Scales of the Included Studies
| Study | No. Analyzed | Smoker,% | Smoking Status, n | Definition of Smoking Status | NOS Scales | ||||
|---|---|---|---|---|---|---|---|---|---|
| CS | FS | NS | CS | FS | NS | ||||
| Sibbald et al, 201034 | 24 456 | 30.0 | 7326 | 17 130 | Cigarette smoking within 1 mo before admission | Quitting >1 mo before admission | Never smoking | 8 | |
| Kang et al, 201335 | 24 257 | 42.3 | 10 251 | 14 006 | Cigarette smoking within 1 mo before admission | ··· | Ex‐smokers and never smokers | 7 | |
| Zhang et al, 201636 | 600 | 55.8 | 335 | 106 | 159 | Smoking >10 y (>10 cigarettes/d); smoking cessation <4 wk | Smoking <10 y (>10 cigarettes/d); smoking cessation <1 y | Never smoking | 8 |
| Kim et al, 201437 | 457 | 26.3 | 120 | 337 | Smoked within 3 mo of index PCI | ··· | The other patients | 8 | |
| Zhang et al, 201722 | 623 | 61.5 | 383 | 240 | ≥1 Cigarette/d during the month before admission | ··· | Never smoking | 7 | |
| Ashby et al, 200238 | 5592 | 18.3 | 1025 | 2155 | 2412 | Smoking at the time of PCI or had smoked within 3 mo | Smoked more than half a packet of cigarettes/d for 1 y and quit in the 3 mo | Never smoking | 6 |
| Tan et al, 201439 | 1051 | 42.6 | 448 | 603 | NA | ··· | NA | 5 | |
| Adamo et al, 201540 | 1962 | 23.9 | 469 | 1493 | NA | ··· | NA | 6 | |
| Bossard et al, 201741 | 17 263 | 37.0 | 6394 | 10 862 | ≥10 Cigarettes/d | ··· | <10 Cigarettes/d | 6 | |
| Ovbiagele et al, 201742 | 5170 | 33.0 | 1705 | 2949 | >5 Cigarettes/d within 1 mo | >5 Cigarettes previously and had stopped for ˃1 mo | Never smoked or ≤5 cigarettes/d currently or previously | 6 | |
| Kodaira et al, 201643 | 6195 | 38.8 | 2403 | 3792 | Current smokers within 1 y | ··· | Quitting smoking >1 y; never smoking | 6 | |
| Siasos et al, 201644 | 229 | 23.1 | 53 | 176 | ≥1 Cigarette/d | ··· | <1 Cigarette/d | 8 | |
| Chandrasekhar et al, 201545 | 19 906 | 25.1 | 5006 | 14 900 | NA | ··· | NA | 6 | |
| Wakabayashi et al, 201146 | 1424 | 34.1 | 486 | 349 | 589 | Currently smoking or had stopped within 1 y | Smoked ≥1 y and those who quit at least 1 y | Never smoking | 7 |
| Ferreiro et al, 201447 | 9534 | 29.5 | 2808 | 6726 | NA | NA | NA | 6 | |
| Lee et al, 201648 | 1527 | 40.9 | 624 | 903 | Smoked within 1 mo | ··· | Never smoking | 7 | |
| Cornel et al, 201449 | 7062 | 22.2 | 1566 | 5494 | Smoking ≥1 cigarette/d or stopped <1 mo previously | Stopped >1 mo previously | Never smoking | 7 | |
| Desai et al, 200916 | 1726 | 50.1 | 864 | 862 | Smoked 1–9, 10–19, 20–29, and 30 cigarettes/d | ··· | Not smoking currently | 7 | |
| Cornel et al, 201250 | 18 610 | 35.9 | 6678 | 11 932 | Smoking ≥1 cigarette/d | Stopped for at least 1 mo; smoke <1 cigarette/d | Never smoking | 7 | |
| Ciccarelli et al, 201751 | 713 | 56.5 | 403 | 310 | NA | ··· | NA | 6 | |
| Berger et al, 200915 | 6071 | 19.8 | 1204 | 3111 | 1756 | Smoked ≥1 cigarette/d within 1 mo | Smoked ≥1 cigarette/d before the month before enrollment | Never smoking | 7 |
| Weisz et al, 201352 | 8583 | 22.6 | 1939 | 6644 | Smoking within 1 mo | ··· | No smoking within 1 mo | 7 | |
CS indicates current smoker; FS, former smoker; NA, no valid data can be obtained; NOS, Newcastle‐Ottawa Scale; NS, never smoker; PCI, percutaneous coronary intervention.
Effectiveness Outcomes Influenced by Smoking Status
MI and smoking status
A total of 83 677 patients in 11 studies5 were included in the analyses, and a random‐effects model was used because of interstudy heterogeneity. As a whole, no significant difference was found in MI event incidence between current smokers and nonsmokers (pooled OR, 0.93; 95% CI, 0.79–1.09; I2=59%; P=0.36; Figure 2). Compared with nonsmokers, current smokers had no difference found at various follow‐up times in the incidence of MI events (>1 year: P=0.87; <1 year: P=0.22).
Figure 2.
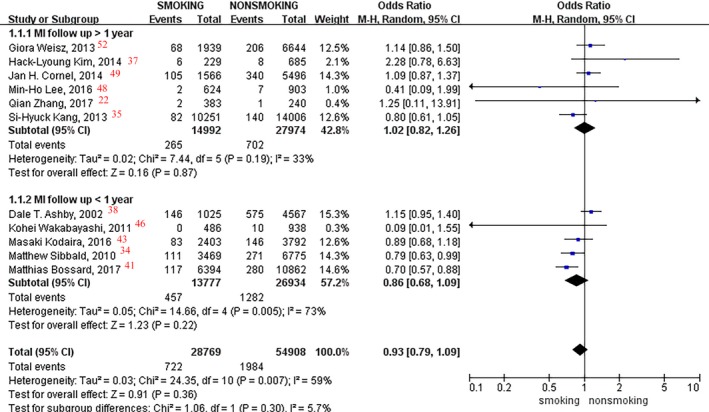
Forest plot of the effects of smoking status on myocardial infarction (MI) events.
We performed prespecified subgroup analyses in patients with or without PCI. However, only one study34 on patients without PCI was included, suggesting a 21% lower incidence of MI events in current smokers compared with nonsmokers (OR, 0.79; 95% CI, 0.63–0.99; P=0.04; Figure 3) in <1 year of follow‐up; no difference was found in patients with PCI (OR, 0.88; P=0.42). Moreover, no significant difference was observed between smoking status and MI event rate in patients with or without PCI after >1 year of follow‐up (P=0.87).
Figure 3.
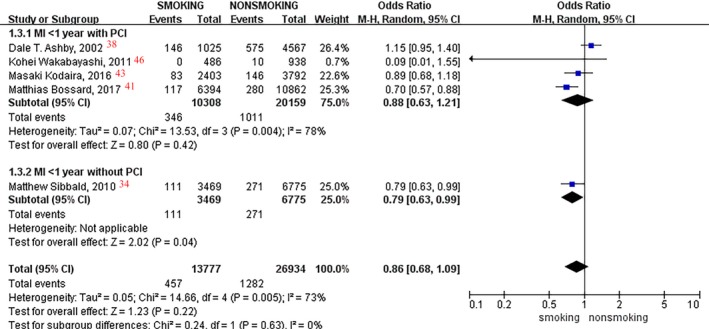
Forest plot of the effects of smoking status on myocardial infarction (MI) events in patients with or without percutaneous coronary intervention (PCI) in <1 year of follow‐up.
In the subgroup analysis of age, patients <63 years old had a 26% lower incidence of MI events in current smokers than in nonsmokers (OR, 0.74; 95% CI, 0.62–0.87; P=0.0004; Figure 4), and no difference was found in patients >65 years old (OR, 1.02; P=0.86). Moreover, no significant difference was observed between smoking status and MI event rate in race subgroup analysis (Asian, white, and mixed races: P=0.22, P=0.34, and P=0.20, respectively; Figure 5).
Figure 4.
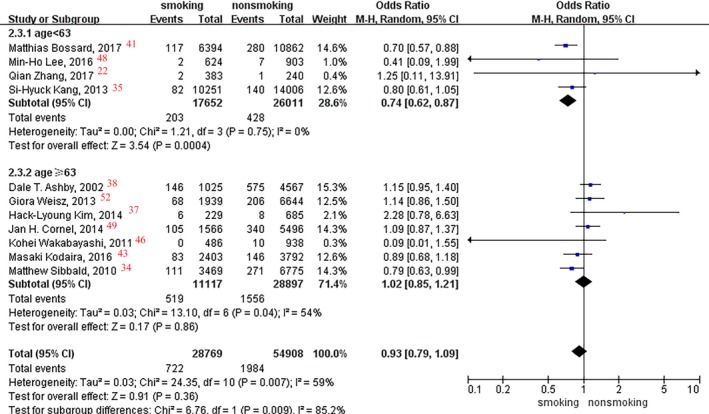
Forest plot of the effects of smoking status on myocardial infarction events in the age subgroup analysis.
Figure 5.
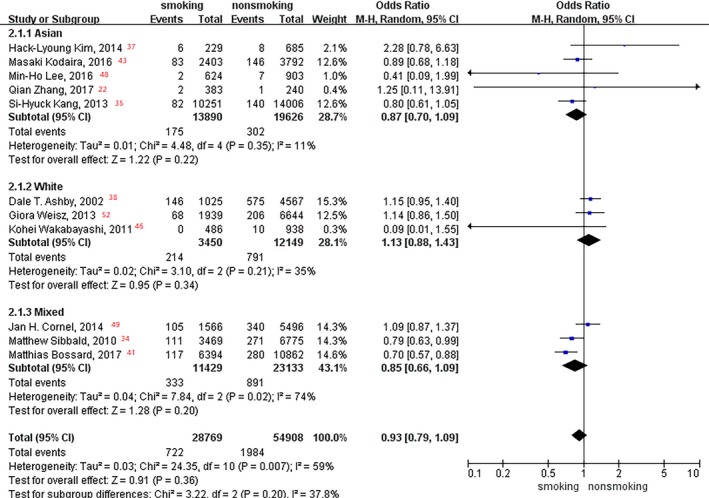
Forest plot of the effects of smoking status on myocardial infarction events in the race subgroup analysis.
Stroke and smoking status
A total of 59 892 patients in 9 studies22, 37, 38, 41, 42, 43, 47, 49, 51 that reported stroke events were analyzed. A fixed‐effects model was used in the analyses because of no significant interstudy heterogeneity. Pooled data for stroke events within 1 year showed an 18% lower rate in current smokers than that in nonsmokers (OR, 0.82; 95% CI, 0.70–0.95; I2=0%; P=0.008; Figure 6). However, no difference was found for those taking the medication for >1 year between the current smokers and nonsmokers (OR, 1.05; 95% CI, 0.88–1.25; I2=0%; P=0.62; Figure 6).
Figure 6.
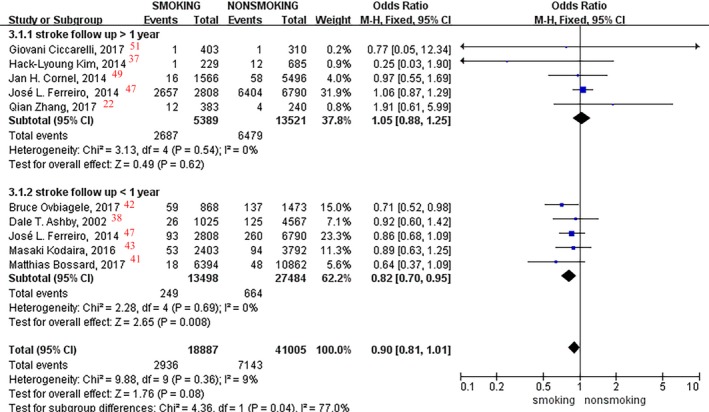
Forest plot of the effects of smoking status on stroke events.
Subgroup analyses in patients with or without PCI were conducted, with an 18% incidence reduction found in current smokers versus nonsmokers within 1 year of follow‐up (OR, 0.82; 95% CI, 0.70–0.95; I2=0%; P=0.008; Figure 7). Among the patients without PCI, a 20% lower stroke event incidence rate was found in current smokers versus nonsmokers within 1 year of follow‐up (OR, 0.80; 95% CI, 0.66‐0.97; I2=0%; P=0.02; Figure 7), whereby no difference was found in patients with PCI (OR, 0.84; I2=0%; P=0.15).
Figure 7.
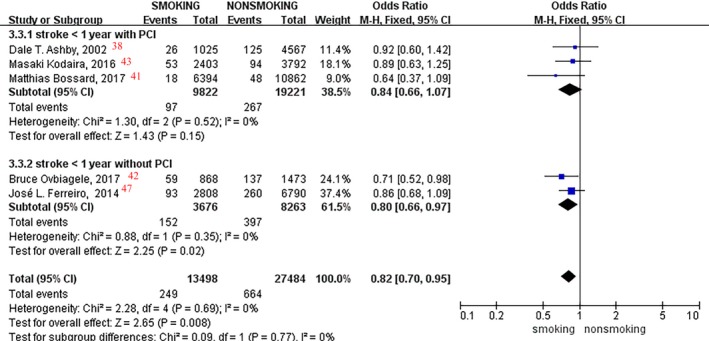
Forest plot of the effects of smoking status on stroke events in patients with or without percutaneous coronary intervention (PCI) in <1 year of follow‐up.
In the subgroup analyses of age and race, no significant difference was observed between smoking status and stroke event rate (aged <63 and ≥63 years: P=0.87 and P=0.32, respectively [Figure 8]; Asian, white, and mixed races: P=0.24, P=0.70, and P=0.24, respectively [Figure 9]).
Figure 8.
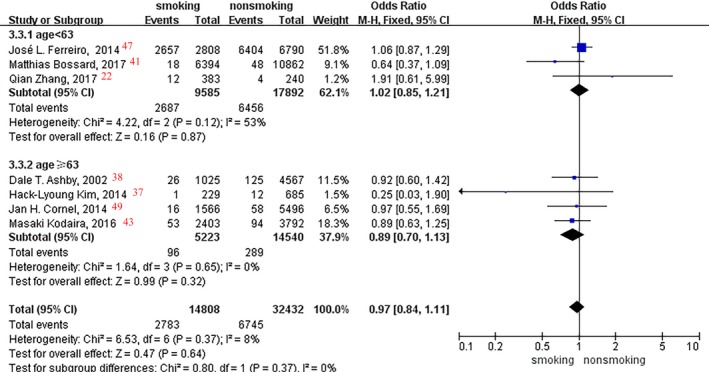
Forest plot of the effects of smoking status on stroke events in the age subgroup analysis.
Figure 9.
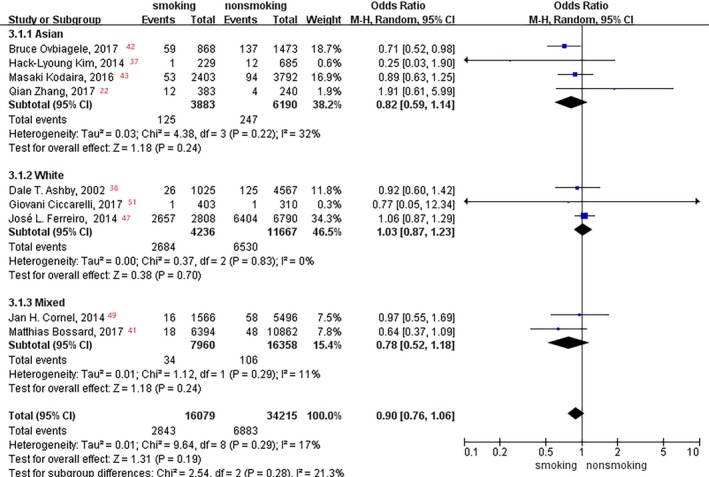
Forest plot of the effects of smoking status on stroke events in the race subgroup analysis.
Safety Outcomes Influenced by Smoking Status
Death and smoking status
A total of 18 studies,6 including 38 844 smokers and 76 312 nonsmokers, were analyzed, and a significant difference in death event rates was found between them (OR, 0.78; 95% CI, 0.63–0.98; I2=89%; P=0.03; Figure 10). Pooled data within 1 year showed a 28% lower death rate in current smokers versus nonsmokers (OR, 0.72; 95% CI, 0.53–0.99; I2=87%; P=0.04), whereas no difference was found in the follow‐up time of >1 year between current smokers and nonsmokers (OR, 0.85; 95% CI, 0.59–1.22; I2=90%; P=0.38; Figure 10). No significant difference was found in the subgroup analyses among patients with (P=0.06) or without (P=0.43) PCI within the 1‐year follow‐up.
Figure 10.
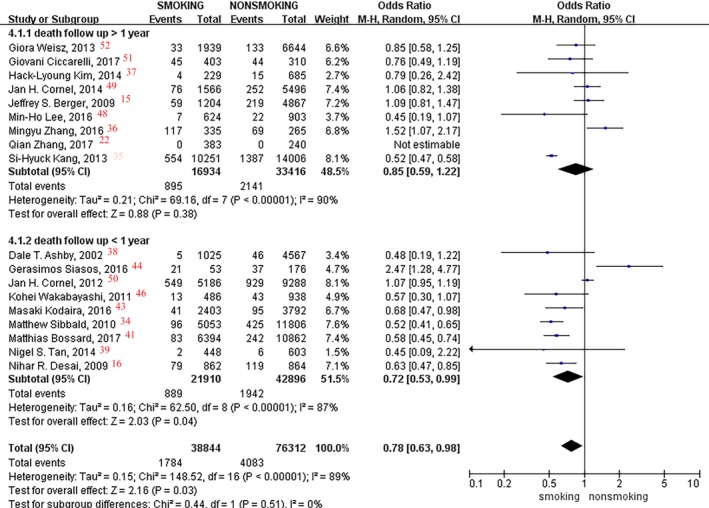
Forest plot of the effects of smoking status on death events.
In the subgroup analyses of age and race, no significant difference was observed between smoking status and death event rate (aged <63 and ≥63 years: P=0.09 and P=0.24, respectively [Figure 11]; Asian, white, and mixed races: P=0.23, P=0.41, and P=0.11, respectively [Figure 12).
Figure 11.
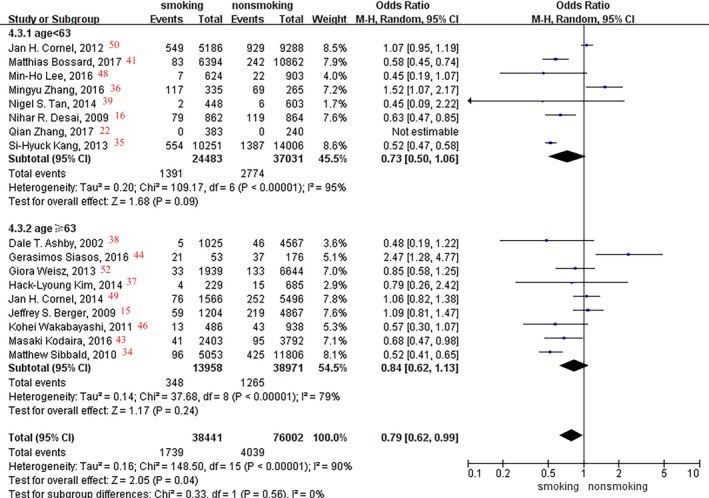
Forest plot of the effects of smoking status on death events in the age subgroup analysis.
Figure 12.
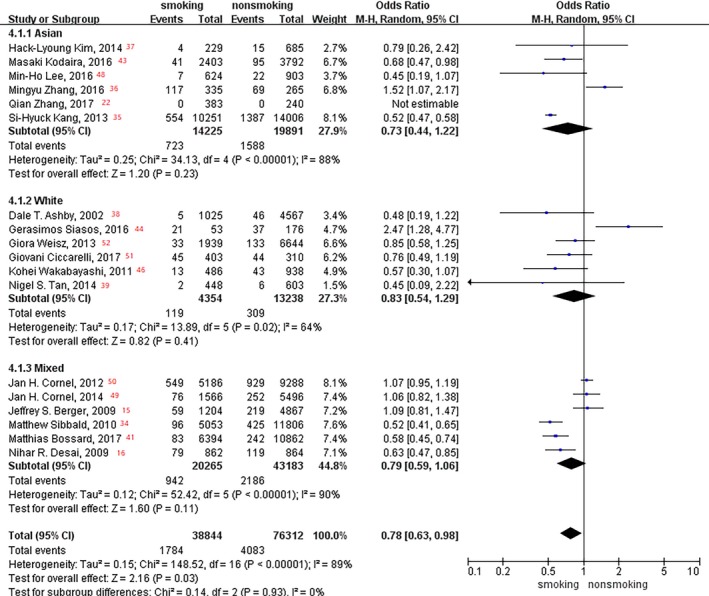
Forest plot of the effects of smoking status on death events in the race subgroup analysis.
Bleeding and smoking status
Nine studies,7 including 28 980 smokers and 64 764 nonsmokers, were analyzed, and no significant difference in the bleeding event rate was observed between them (OR, 0.86; 95% CI, 0.73–1.00; I2=75%; P=0.05; Figure 13). Subgroup analyses in patients with or without PCI were conducted; only one study reported bleeding outcomes in patients with PCI,52 showing a significant difference between smokers and nonsmokers after >1 year of follow‐up (OR, 0.73; P=0.006).
Figure 13.
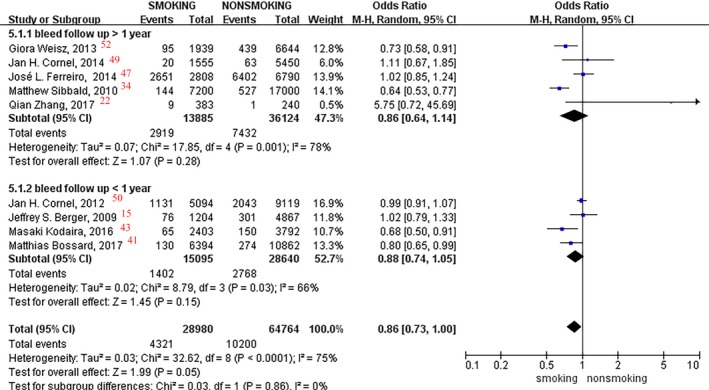
Forest plot of the effects of smoking status on bleeding events.
In the subgroup analysis of age, patients >63 years old had a 22% lower incidence of bleeding events in current smokers versus nonsmokers (OR, 0.78; 95% CI, 0.64–0.96; P=0.02; Figure 14), and no difference was found in patients <63 years old (OR, 0.95; P=0.54). Moreover, no significant difference was observed between smoking status and bleeding event rate in race subgroup analysis (Asian, white, and mixed races: P=0.68, P=0.41, and P=0.19, respectively; Figure 15).
Figure 14.
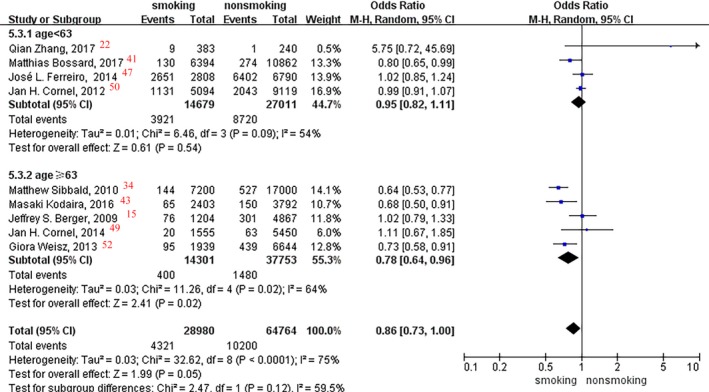
Forest plot of the effects of smoking status on bleeding events in the age subgroup analysis.
Figure 15.
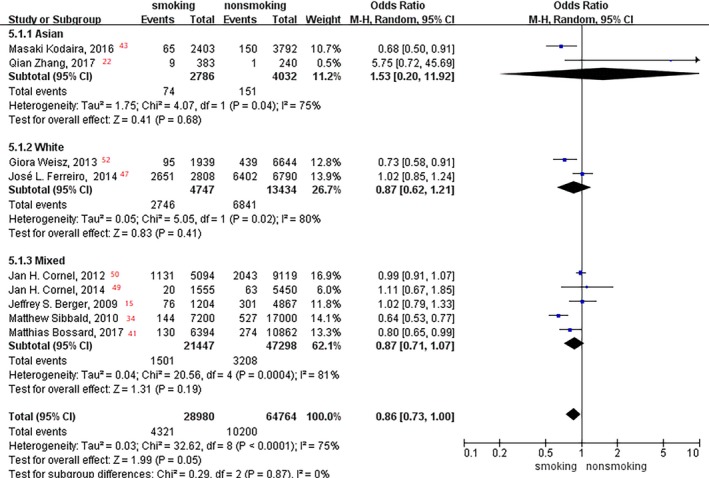
Forest plot of the effects of smoking status on bleeding events in the race subgroup analysis.
MACEs and smoking status
Fifteen studies8 with 107 188 patients were included in the analyses of MACEs. Compared with nonsmokers, smokers had a 16% lower MACE incidence rate (OR, 0.84; 95% CI, 0.72–0.98; I2=90%; P=0.03; Figure 16). Meanwhile, a 26% reduction rate was found in current smokers versus nonsmokers, especially at the follow‐up time in the within 1‐year group (OR, 0.74; 95% CI, 0.58–0.95; I2=88%; P=0.02; Figure 16).
Figure 16.
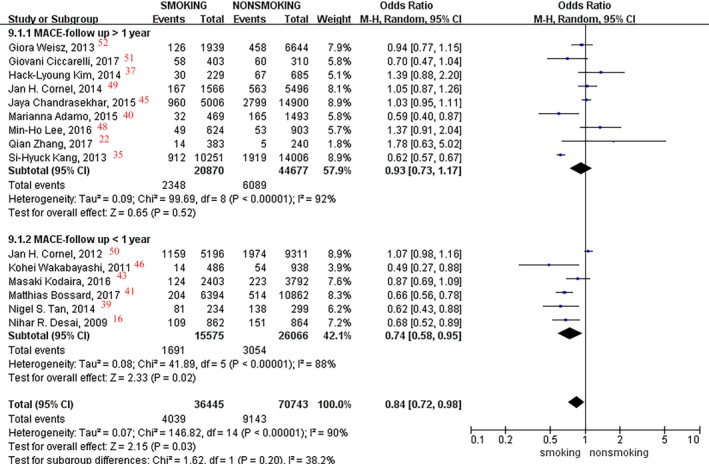
Forest plot of the effects of smoking status on major adverse cardiovascular events (MACEs).
In the subgroup analyses of patients with or without PCI, a 34% reduction of the MACE incidence rate was found among patients without PCI within 1 year of follow‐up (OR, 0.66; 95% CI, 0.53–0.81; I2=0%; P=0.0001; Figure 17), whereby no difference was found in patients with PCI (P=0.13).
Figure 17.
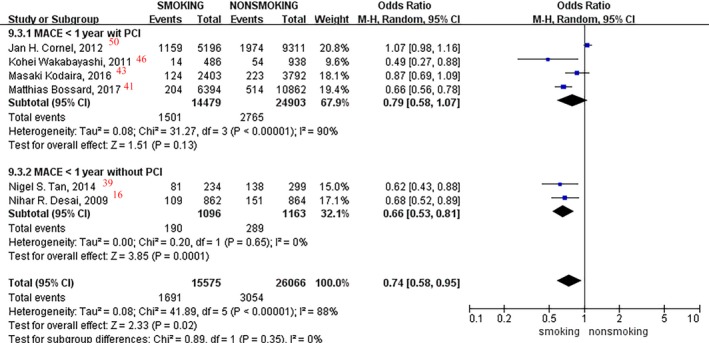
Forest plot of the effects of smoking status on major adverse cardiovascular events (MACEs) in patients with or without percutaneous coronary intervention (PCI) in <1 year of follow‐up.
In the subgroup analysis of age, no significant difference was observed between smoking status and MACE incidence rate (aged <63 and ≥63 years: P=0.15 and P=0.22, respectively; Figure 18). White patients had a 24% lower incidence of MACEs in current smokers versus nonsmokers (OR, 0.76; 95% CI, 0.60–0.95; P=0.02; Figure 19), and no difference was found in Asian and mixed race patients (Asians and mixed races: P=0.92 and P=0.24, respectively).
Figure 18.
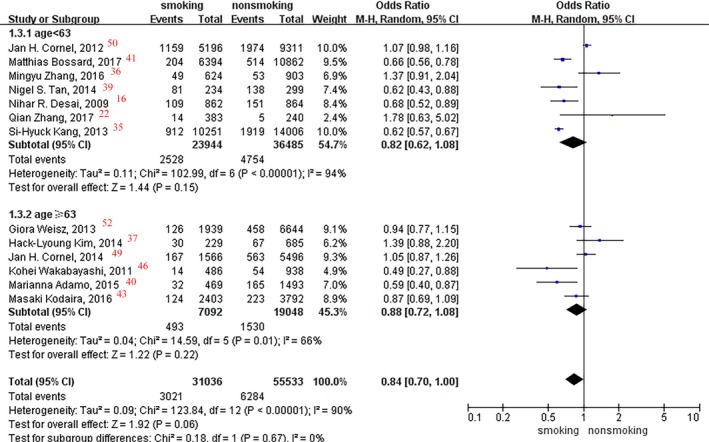
Forest plot of the effects of smoking status on major adverse cardiovascular events in the age subgroup analysis.
Figure 19.
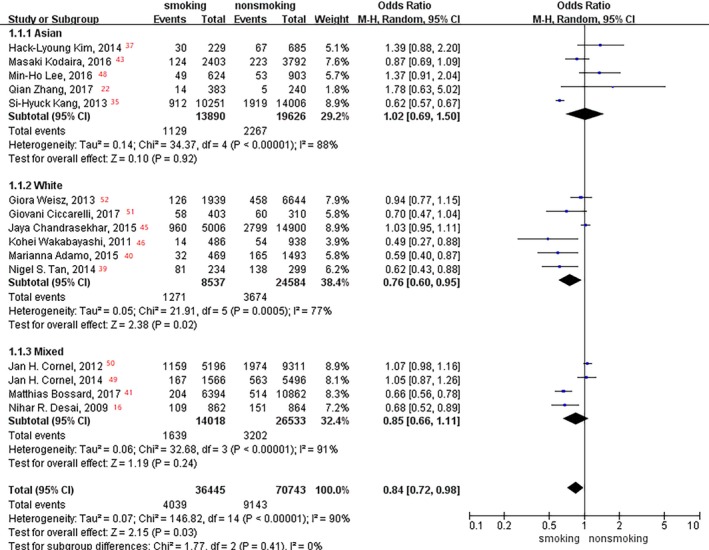
Forest plot of the effects of smoking status on major adverse cardiovascular events in the race subgroup analysis.
Comparison Between Different ADP–P2Y12 Receptor Inhibitors
We included studies that used different ADP–P2Y12 receptor inhibitors; therefore, different drugs were compared to assess the influence of smoking on their clinical outcomes. Three studies45, 49, 50 using different therapeutic drugs that reported MACE outcomes with different smoking status were analyzed. However, no significant difference was found in MACE rates between prasugrel, ticagrelor, and clopidogrel in different smoking status (OR, 1.08; 95% CI, 0.95–1.23; I2=77%; P = 0.24; Figure 20).
Figure 20.
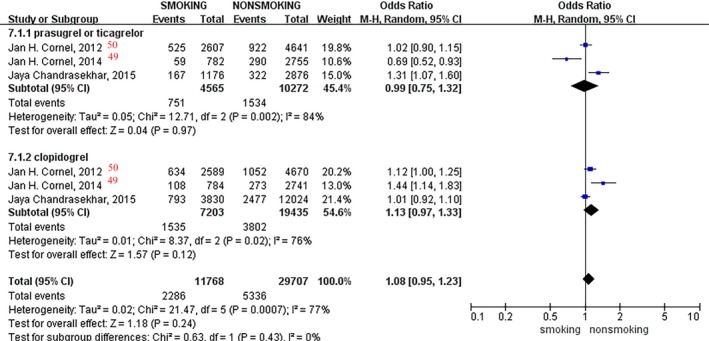
Forest plot of the effects of smoking status on major adverse cardiovascular events in patients taking different drugs.
Meta‐Regression
We also used meta‐regression to determine whether age and race affected the association between clinical outcomes and different smoking status, but we found no significant association between MI, MACE, death events, age, and race (P>0.05; Table 3). The proportions of between‐study variance explained by age and race in MACE incidence rate were −16.43% and 5.48%, respectively, but there were no significant statistical differences (P=0.35 and P=0.38, respectively). The meta‐regression results of race and age are shown in Table 3.
Table 3.
Results From Meta‐Regression
| Outcomes | Factors | No. of Studies | τ2 a | I2 b | Adjusted R 2, %c | P Value | t Value | Regression Coefficient | SEM | 95% CI |
|---|---|---|---|---|---|---|---|---|---|---|
| MI | Age | 11 | 0 | 0 | 0 | 0.73 | 0.35 | 0.01 | 0.02 | −0.03 to 0.05 |
| Race | 11 | 0 | 0 | 0 | 0.98 | 0.02 | 0.001 | 0.05 | −0.11 to 0.11 | |
| MACE | Age | 13 | 0.01 | 56.35 | −16.43 | 0.35 | −0.97 | −0.01 | 0.01 | −0.04 to 0.02 |
| Race | 15 | 0.01 | 39.48 | 5.48 | 0.38 | 0.92 | 0.04 | 0.04 | −0.005 to 0.12 | |
| Death | Age | 17 | 0 | 0 | 0 | 0.89 | 0.14 | 0.002 | 0.01 | −0.03 to 0.03 |
| Race | 18 | 0.002 | 0 | 0 | 0.62 | −0.05 | −0.02 | 0.03 | −0.09 to 0.06 |
Meta‐regression with Knapp‐Hartung modification. MACE indicates major adverse cardiovascular event; MI, myocardial infarction.
REML (restricted maximum likelihood) estimate of between‐study variance.
Percentage residual variation attributable to heterogeneity.
Proportion of between‐study variance explained.
As the age meta‐regression analysis reflected the between‐study variance, we were unable to identify age potential influence on personal clinical outcomes. Studies reported unadjusted and adjusted hazard ratio (HR) results, and the adjusted HR results were adjusted by multiple factors (including age, sex, and body mass index) in original research. Therefore, we conducted merged HR analysis in the study. In adjusted MACE‐HR subgroup analysis, smokers had a lower MACE incidence rate than nonsmokers (HR, 0.90; 95% CI, 0.82–0.98; P=0.01; Figure 21), but no difference was found in unadjusted subgroup analysis (P=0.73). In unadjusted death‐HR subgroup analysis, smokers had a lower death incidence rate than nonsmokers (HR, 0.52; 95% CI, 0.47–0.58; P<0.001; Figure 22), whereas no difference was found in adjusted subgroup analysis (P=0.28). No significant difference was observed between smoking status and MI and stroke incidence rate in unadjusted (MI: P=0.06) and adjusted (MI and stroke: P=0.83 and P=0.43, respectively; Figures 23 and 24) subgroup analysis.
Figure 21.

Forest plot of combined hazard ratio (HR) on major adverse cardiovascular events (MACEs) in unadjusted and adjusted subgroup analysis.
Figure 22.

Forest plot of combined hazard ratio (HR) on death events in unadjusted and adjusted subgroup analysis.
Figure 23.
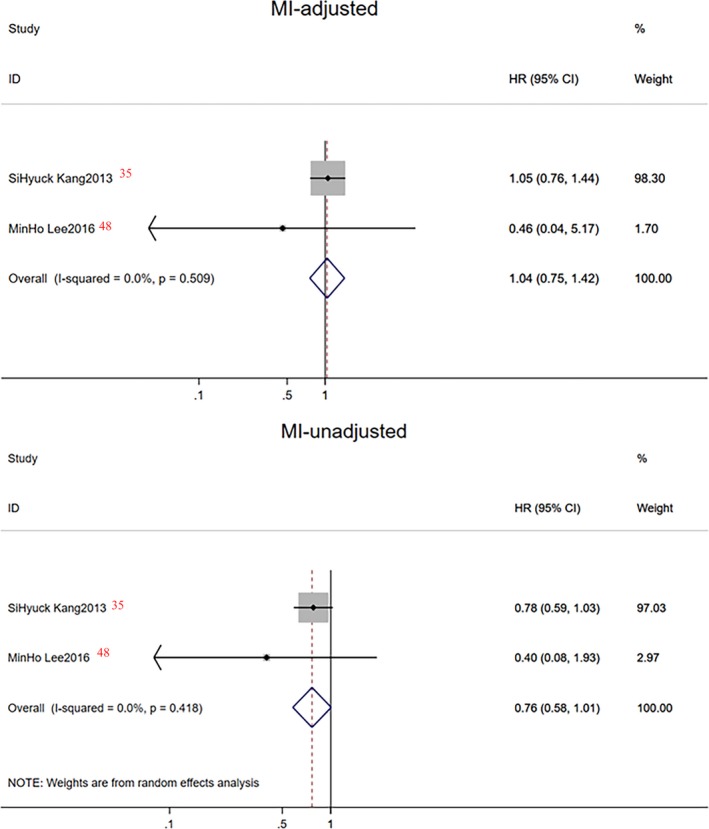
Forest plot of combined hazard ratio (HR) on myocardial infarction (MI) events in unadjusted and adjusted subgroup analysis.
Figure 24.
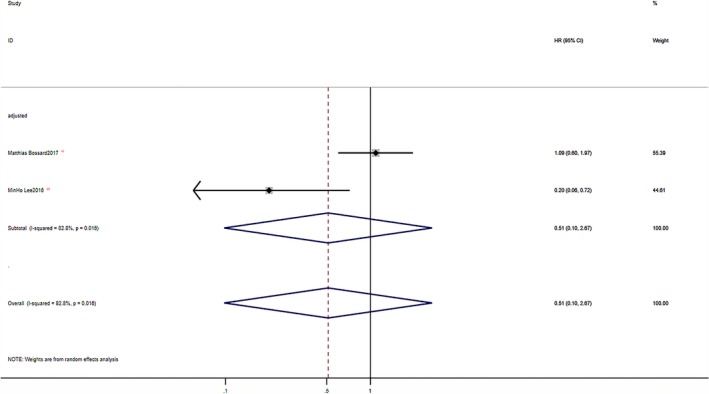
Forest plot of combined hazard ratio (HR) on stroke events in unadjusted and adjusted subgroup analysis.
Sensitivity Analyses and Publication Bias
The exclusion of an individual study did not significantly influence the pooled results and the conclusion in the study. For MI, stroke, and MACE studies, no major publication biases were found through the Egger's test, with P=0.16, P=0.10, and P=0.08, respectively. However, for death studies, publication bias was observed through the Egger's test, with P=0.01. The funnel plot (Figure 25) indicated that studies of Siasos et al44 and Zhang et al36 might influence the bias because both of them were small sample size studies (n=229 and n=600, respectively).
Figure 25.
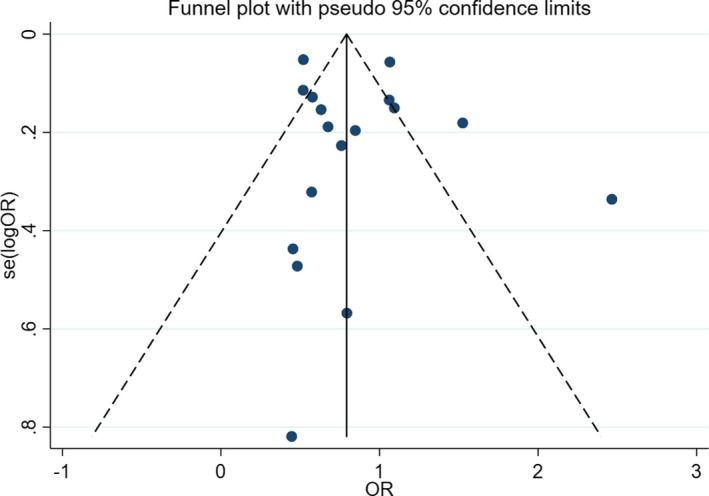
Funnel plot of the effects of smoking status on death events. OR indicates odds ratio.
Discussion
We noted that current smokers had a lower incidence of MACEs and stroke events during the first year of follow‐up than did nonsmokers, especially with respect to patients without PCI. Compared with nonsmokers, a16% reduction in the MACE rate (OR, 0.84; 95% CI, 0.72–0.98; P=0.03) was found among current smokers. Compared with nonsmokers within 1 year of follow‐up, stroke and MACE rates were reduced by 18% (P=0.008) and 26% (P=0.02) in smokers, respectively. For smoking patients without PCI, the stroke and MACE rates were reduced by 20% (OR, 0.80; 95% CI, 0.66–0.97; P=0.02) and 34% (OR, 0.66; 95% CI, 0.53–0.81; P=0.0001), respectively. No significant difference in clinical outcomes (MACEs) was observed between prasugrel, ticagrelor, and clopidogrel in different smoking status (OR, 1.08; 95% CI, 0.95–1.23; P=0.24). No significant difference was found in MI and bleeding event incidence between current smokers and nonsmokers.
The study mainly included new points, as follows:
-
More patients were involved.
Compared with the previous meta‐analyses (by Zhao et al,53 including 7 studies with 66 706 patients; and Gagne et al,55 including 9 studies with 74 489 patients), this study involved additional 14 new studies from 2014 to 2018 and a total of 163 011 patients were analyzed.
-
Different outcome indicators were compared.
The meta‐analyses conducted by Zhao et al53 and Gagne et al55 only compared outcomes of MACE incidence. Zhao et al53 showed that clopidogrel was associated with a 10% reduction in MACE among noncurrent smokers, whereas this clinical benefit was boosted by 2.9‐fold in current smokers. Gagne et al55 showed that clopidogrel was associated with a 25% reduction in MACEs in smokers; however, only an 8% reduction was found among nonsmokers. Ferreiro et al56 found that the reduction of ischemic events and death was higher among patients treated with clopidogrel in current smokers compared with those who never smoked/ex‐smokers, whereas no interaction between smoking status and study treatment was observed for bleeding events. Thus, to comprehensively estimate the influence of cigarette smoking, we compared MI, stroke, death, bleeding, and MACEs according to different smoking status in the ADP–P2Y12 receptor inhibitor treatment groups. Compared with nonsmokers within 1 year of follow‐up, the reductions of stroke and MACE rates were 18% and 26%, respectively, in the study. No significant difference was found in MI and bleeding event incidence between current smokers and nonsmokers. The result indicated that the influence of smoking on stroke incidence was more significant than that of MI and bleeding events. A 22% reduction in death event rate was found among current smokers; however, as for the unstable results and publication bias, more studies are needed to verify our conclusion.
-
Different follow‐up time was compared by subgroup analysis.
Compared with never smokers, current smokers with a recent minor stroke or transient ischemic attack had a greater benefit in stroke prevention at 90 days since clopidogrel intake.42 The meta‐analysis conducted by Zhao et al53 compared MACEs based on a mean follow‐up time of 11.2 months; and in the meta‐analysis conducted by Gagne et al,55 the follow‐up time ranged from 30 days to 3 years. Thus, we aimed to explore the influence of different follow‐up times on the association between smoking and clinical outcomes. The follow‐up time was divided into within 1 year and >1 year after taking P2Y12 receptor inhibitors into the study. Within the first year of follow‐up, we found a significant reduction in stroke and MACEs among current smokers versus nonsmokers, whereas no significant difference in the effectiveness and safety outcomes was found between smokers and nonsmokers at >1‐year follow‐up time. Thus, we concluded that the difference in clinical outcomes between current smokers and nonsmokers was predominantly found at an early time point.
-
Influence of patients with or without PCI on the association between the clinical outcomes and smoking.
A previous meta‐analysis did not compare patients with or without PCI; thus, we conducted a subgroup analysis to determine the PCI influence on the association between smoking and clinical outcomes. For smokers without PCI, the stroke and MACE rates were reduced by 20% and 34%, respectively. Meanwhile, no significant difference was found in patients with PCI on the association between stroke, MACEs, and smoking status. In line with the study, other studies found that smokers have more P2Y12 receptor binding than nonsmokers among patients with stable coronary artery disease57 and smokers do not modify the bleeding outcomes in patients undergoing coronary stenting.40 The results indicated that the difference in clinical outcomes between different smokers after taking antiplatelet drugs was more pronounced in patients without PCI.
-
Different P2Y12 receptor inhibitors influence the association between clinical outcomes and smoking status.
When comparing clopidogrel use, prasugrel was more associated with an accentuated benefit in nonsmokers than smokers,45 whereas Hochholzer et al58 found the clinical treatment effects of clopidogrel versus prasugrel were not affected by the smoking status at presentation. Thus, with more studies involved, we compared the influence of different P2Y12 receptor inhibitors on the association between clinical outcomes and smoking. Three studies45, 49, 50 using different P2Y12 receptor inhibitors that reported MACE outcomes with different smoking status were analyzed. However, no significant difference on MACE rate was found between prasugrel, ticagrelor, and clopidogrel in patients with different smoking status in this study. Because of the limited research data, the relationship between new P2Y12 receptor inhibitors and clinical outcomes of different smoking status cannot be concluded yet.
-
Taking confounding covariates into account.
In the study, we found that current smokers were, on average, 10 years younger than nonsmokers. Ashby et al38 also found that the smoker's paradox, in terms of total mortality, disappeared in the current smokers after they adjusted the results for differences (age, diabetes mellitus, and systemic hypertension) in the baseline variables. Thus, they speculated that the reason for this paradox might be the fact that smokers are significantly younger as a group and have fewer additional cardiac risk factors, such as diabetes mellitus and systemic hypertension. However, Kodaira et al43 found that unadjusted in‐hospital mortality rate, general complication rate, and bleeding complication rate were lower in smokers than in nonsmokers. After adjustment, the trend persisted and smoking was not associated with overall mortality and was associated with lower overall and bleeding complication events. Consequently, subgroup analyses and meta‐regression were conducted to determine the confounding effect of age. In the subgroup analysis of age, patients >63 years old had a significantly lower incidence of bleeding events in current smokers versus nonsmokers, whereas a lower incidence of MI events was found in smokers <63 years old. In the meta‐regression, age did not significantly influence the association between clinical outcomes and different smoking status (P>0.05). However, the age meta‐regression analysis reflected the between‐study variance; we were unable to identify the potential influence of age on personal clinical outcomes. As the mean age of the studies was similar, no correlation could be found in the meta‐regression, even that there might be a strong correlation between age and clinical outcomes in each study. This is one of the problems of integrating individual outcomes and is called aggregation bias, ecological bias, or ecological fallacy.54, 59, 60 Thus, we conducted unadjusted/adjusted HR subgroup analyses in the original research. In adjusted HR subgroup analysis, smokers might have had a lower MACE incidence rate than nonsmokers (P=0.01), but no difference was found in unadjusted HR subgroup analysis (P=0.73).
On the basis of the above subgroup analyses, meta‐regression, and adjusted data analyses, we found that the MACE rate of smokers was lower than that of nonsmokers. However, because of the lack of original adjusted data, smoker's paradox still needs to consider the impact of age and other covariates. We also hope that more studies can do data comparison analyses after adjusting the results for differences (eg, age) in the baseline variables to obtain more reliable conclusions.
Conclusion
This systematic review and meta‐analysis suggested that the smoker's paradox does exist, with better clinical efficacy and safety of P2Y12 receptor inhibitors in current smokers, especially for stroke and MACEs. However, because of the lack of original adjusted data, smoker's paradox still needs to consider the impact of age and other covariates. No significant difference was found in MI, bleeding event incidence, and different P2Y12 receptor inhibitors between current smokers and nonsmokers in the study. As for the unstable results and publication bias in death event analysis, more studies are needed to verify our conclusion. Moreover, on the basis of the subanalysis, we noted that the difference in efficacy and safety outcomes between current smokers and nonsmokers was predominantly found at an early time point and was more pronounced in patients without PCI. Thus, a differential risk‐benefit evaluation should be considered, according to different smoking status, patient conditions, and therapy time points.
Limitations
The definition of smoking was not consistent in some studies; thus, the heterogeneity might be partially attributed to the confounding effects of former smokers when comparing smokers with nonsmokers. We hope that future studies can clearly define smoking status and record the specific intake of tobacco to better explore the effects of smoking on clinical outcomes.
Because of the lack of original adjusted data, smoker's paradox still needs to consider the impact of age and other covariates. We also hope that more studies can do data comparison analyses after adjusting the results for differences (eg, age) in the baseline variables to obtain more reliable conclusions.
Author Contributions
Yimin Cui and Qian Xiang conceived and designed the study. Zhiyan Liu and Guangyan Mu searched the databases and checked them according to the eligible criteria and exclusion criteria. Yanjun Gong and Jie Jiang helped develop search strategies. Zining Wang and Shuang Zhou extracted the quantitative data. Zhiyan Liu, Shuqing Chen, Qiufen Xie, and Kun Hu analyzed the data. Zhiyan Liu wrote the draft of the article. All authors contributed in writing, reviewing, or revising the article.
Sources of Funding
This study was supported by grants from the National Natural Science Foundation of China (Nos. 81673509 and 81573504), the Beijing Municipal Natural Science Foundation (No. 7171012), the National Key Research and Development Program of China (No. 2016YFC0904900), and the National Science and Technology Major Projects for “Major New Drugs Innovation and Development” of China (Nos. 2017ZX09304028 and 2017ZX09101001).
Disclosures
None.
Supporting information
Data S1. Search strategies in PubMed.
Table S1. Quality Assessment of the Included Studies Using the Newcastle–Ottawa Scale
Acknowledgments
The authors thank the participants of this study for their important contributions.
(J Am Heart Assoc. 2019;8:e010889 DOI: 10.1161/JAHA.118.010889.)
Notes
References
- 1. Ferri N, Corsini A, Bellosta S. Pharmacology of the new P2Y12 receptor inhibitors: insights on pharmacokinetic and pharmacodynamic properties. Drugs. 2013;73:1681–1709. [DOI] [PubMed] [Google Scholar]
- 2. GBD 2013 Mortality and Causes of Death Collaborators . Global, regional, and national age‐sex specific all‐cause and cause‐specific mortality for 240 causes of death, 1990–2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet. 2015;385:117–171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Mehta SR, Bassand JP, Chrolavicius S, Diaz R, Fox KA, Granger CB, Jolly S, Rupprecht HJ, Widimsky P, Yusuf S; CURRENT‐OASIS 7 Steering Committee . Design and rationale of CURRENT‐OASIS 7: a randomized, 2 x 2 factorial trial evaluating optimal dosing strategies for clopidogrel and aspirin in patients with ST and non‐ST elevation acute coronary syndromes managed with an early invasive strategy. Am Heart J. 2008;156:1080–1088. [DOI] [PubMed] [Google Scholar]
- 4. CURRENT‐OASIS 7 Investigators , Mehta SR, Bassand JP, Chrolavicius S, Diaz R, Eikelboom JW, Fox KA, Granger CB, Jolly S, Joyner CD, Rupprecht HJ, Widimsky P, Afzal R, Pogue J, Yusuf S. Dose comparisons of clopidogrel and aspirin in acute coronary syndromes. N Engl J Med. 2010;363:930–942. [DOI] [PubMed] [Google Scholar]
- 5. Mehta SR, Tanguay JF, Eikelboom JW, Jolly SS, Joyner CD, Granger CB, Faxon DP, Rupprecht HJ, Budaj A, Avezum A, Widimsky P, Steg PG, Bassand JP, Montalescot G, Macaya C, Di Pasquale G, Niemela K, Ajani AE, White HD, Chrolavicius S, Gao P, Fox KA, Yusuf S; CURRENT‐OASIS 7 trial investigators . Double‐dose versus standard‐dose clopidogrel and high‐dose versus low‐dose aspirin in individuals undergoing percutaneous coronary intervention for acute coronary syndromes (CURRENT‐OASIS 7): a randomized factorial trial. Lancet. 2010;376:1233–1243. [DOI] [PubMed] [Google Scholar]
- 6. Siller‐Matula JM, Trenk D, Krähenbühl S, Michelson AD, Delle‐Karth G. Clinical implications of drug‐drug interactions with P2Y12 receptor inhibitors. J Thromb Haemost. 2014;12:2–14. [DOI] [PubMed] [Google Scholar]
- 7. Amsterdam EA, Wenger NK, Brindis RG, Casey DE Jr, Ganiats TG, Holmes DR Jr, Jaffe AS, Jneid H, Kelly RF, Kontos MC, Levine GN, Liebson PR, Mukherjee D, Peterson ED, Sabatine MS, Smalling RW, Zieman SJ; ACC/AHA Task Force Members . 2014 AHA/ACC guideline for the management of patients with non‐ST‐elevation acute coronary syndromes: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014;130:344–426. [Google Scholar]
- 8. Windecker S, Kolh P, Alfonso F, Collet JP, Cremer J, Falk V, Filippatos G, Hamm C, Head SJ, Jüni P, Kappetein AP, Kastrati A, Knuuti J, Landmesser U, Laufer G, Neumann FJ, Richter DJ, Schauerte P, Uva MS, Stefanini GG, Taggart DP, Torracca L, Valgimigli M, Wijns W, Witkowski A; Grupa Robocza Europejskiego Towarzystwa Kardiologicznego (ESC); Europejskie Stowarzyszenie Chirurgii Serca i Klatki Piersiowej (EACTS) do spraw rewaskularyzacji mięśnia sercowego; European Association for Percutaneous Cardiovascular Interventions (EAPCI) . 2014 ESC/EACTS guidelines on myocardial revascularization [in Polish]. Kardiol Pol. 2014;72:1253–1379. [DOI] [PubMed] [Google Scholar]
- 9. Saito S, Isshiki T, Kimura T, Ogawa H, Yokoi H, Nanto S, Takayama M, Kitagawa K, Nishikawa M, Miyazaki S, Nakamura M. Efficacy and safety of adjusted‐dose prasugrel compared with clopidogrel in Japanese patients with acute coronary syndrome: the PRASFIT‐ACS study. Circ J. 2014;78:1684–1692. [DOI] [PubMed] [Google Scholar]
- 10. Nishi T, Ariyoshi N, Nakayama T, Fujimoto Y, Sugimoto K, Takahara M, Wakabayashi S, Koshizaka M, Hanaoka H, Kobayashi Y. Increased platelet inhibition after switching from maintenance clopidogrel to prasugrel in Japanese patients with stable coronary artery disease. Circ J. 2015;79:2439–2444. [DOI] [PubMed] [Google Scholar]
- 11. Goto S, Huang CH, Park SJ, Emanuelsson H, Kimura T. Ticagrelor vs. clopidogrel in Japanese, Korean and Taiwanese patients with acute coronary syndrome: randomized, double‐blind, phase III PHILO study. Circ J. 2015;79:2452–2460. [DOI] [PubMed] [Google Scholar]
- 12. Rollini F, Franchi F, Cho JR, Degroat C, Bhatti M, Ferrante E, Patel R, Darlington A, Tello‐Montoliu A, Desai B, Ferreiro J, Muniz‐Lozano A, Zenni MM, Guzman LA, Bass TA, Angiolillo DJ. Cigarette smoking and antiplatelet effects of aspirin monotherapy versus clopidogrel monotherapy in patients with atherosclerotic disease: results of a prospective pharmacodynamic study. J Cardiovasc Transl Res. 2014;7:53–63. [DOI] [PubMed] [Google Scholar]
- 13. U.S. Department of Health and Human Services . How Tobacco Smoke Causes Disease: The Biology and Behavioral Basis for Smoking‐Attributable Disease: A Report of the Surgeon General. Atlanta, GA: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health ; 2010. [Google Scholar]
- 14. Gurbel PA, Baker BA, Bailey WL, Bliden KP, Tantry US. Unravelling the smokers’ paradox: cigarette smoking, high‐risk coronary artery disease and enhanced clinical efficacy of oral P2Y12 inhibitors. Thromb Haemost. 2014;111:1187–1190. [DOI] [PubMed] [Google Scholar]
- 15. Berger JS, Bhatt DL, Steinhubl SR, Shao M, Steg PG, Montalescot G, Hacke W, Fox KA, Lincoff AM, Topol EJ, Berger PB; CHARISMA Investigators . Smoking, clopidogrel, and mortality in patients with established cardiovascular disease. Circulation. 2009;120:2337–2344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Desai NR, Mega JL, Jiang S, Cannon CP, Sabatine MS. Interaction between cigarette smoking and clinical benefit of clopidogrel. J Am Coll Cardiol. 2009;53:1273–1278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Washio I, Maeda M, Sugiura C, Shiga R, Yoshida M, Nonen S, Fujio Y, Azuma J. Cigarette smoke extract induces CYP2B6 through constitutive androstane receptor in hepatocytes. Drug Metab Dispos. 2011;39:1–3. [DOI] [PubMed] [Google Scholar]
- 18. Bliden KP, Baker BA, Nolin TD, Jeong YH, Bailey WL, Tantry US, Gurbel PA. Thienopyridine efficacy and cigarette smoking status. Am Heart J. 2013;165:693–703. [DOI] [PubMed] [Google Scholar]
- 19. Farid NA, Kurihara A, Wrighton SA. Metabolism and disposition of the thienopyridine antiplatelet drugs ticlopidine, clopidogrel, and prasugrel in humans. J Clin Pharmacol. 2010;50:126–142. [DOI] [PubMed] [Google Scholar]
- 20. Zhou SF, Wang B, Yang LP, Liu JP. Structure, function, regulation and polymorphism and the clinical significance of human cytochrome P450 1A2. Drug Metab Rev. 2010;42:268–354. [DOI] [PubMed] [Google Scholar]
- 21. Gengo F, Robson M, Rainka M. CYP1A2‐inducing medications improve platelet responsiveness to clopidogrel. J Clin Pharmacol. 2010;50:1069. [Google Scholar]
- 22. Zhang Q, Wang Y, Song H, Hou C, Cao Q, Dong K, Huang X, Feng W, Ovbiagele B, Wang M, Ji X. Clopidogrel and ischemic stroke outcomes by smoking status: smoker's paradox? J Neurol Sci. 2017;373:41–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Zhang YJ, Iqbal J, van Klaveren D, Campos CM, Holmes DR, Kappetein AP, Morice MC, Banning AP, Grech ED, Bourantas CV, Onuma Y, Garcia‐Garcia HM, Mack MJ, Colombo A, Mohr FW, Steyerberg EW, Serruys PW. Smoking is associated with adverse clinical outcomes in patients undergoing revascularization with PCI or CABG: the SYNTAX trial at 5‐year follow‐up. J Am Coll Cardiol. 2015;65:1107–1115. [DOI] [PubMed] [Google Scholar]
- 24. Weintraub WS, Grau‐Sepulveda MV, Weiss JM, Delong ER, Peterson ED, O'Brien SM, Kolm P, Klein LW, Shaw RE, McKay C, Ritzenthaler LL, Popma JJ, Messenger JC, Shahian DM, Grover FL, Mayer JE, Garratt KN, Moussa ID, Edwards FH, Dangas GD. Prediction of long‐term mortality after percutaneous coronary intervention in older adults: results from the National Cardiovascular Data Registry. Circulation. 2012;125:1501–1510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Task Force on Myocardial Revascularization of the European Society of Cardiology (ESC) and the European Association for Cardio‐Thoracic Surgery (EACTS); European Association for Percutaneous Cardiovascular Interventions (EAPCI) , Wijns W, Kolh P, Danchin N, Di Mario C, Falk V, Folliguet T, Garg S, Huber K, James S, Knuuti J, Lopez‐Sendon J, Marco J, Menicanti L, Ostojic M, Piepoli MF, Pirlet C, Pomar JL, Reifart N, Ribichini FL, Schalij MJ, Sergeant P, Serruys PW, Silber S, Uva MS, Taggart D. Guidelines on myocardial revascularization. Eur Heart J. 2010;31:2501–2555. [DOI] [PubMed] [Google Scholar]
- 26. Chen W, Gao RL, Liu L, Zhu ML, Wang W, Wang YJ, Wu ZS, Li HJ, Gu DF, Yang Y, Zheng Z, Jiang LX, Hu ST. 2017‐Cardiovascular disease report in China outline. Chinese Circ J. 2018;33:1–8. [Google Scholar]
- 27. Thygesen K, Alpert JS, Jaffe AS, Simoons ML, Chaitman BR, White HD; Joint ESC/ACCF/AHA/WHF Task Force for Universal Definition of Myocardial Infarction; Authors/Task Force Members Chairpersons , Thygesen K, Alpert JS, White HD; Biomarker Subcommittee , Jaffe AS, Katus HA, Apple FS, Lindahl B, Morrow DA; ECG Subcommittee , Chaitman BR, Clemmensen PM, Johanson P, Hod H; Imaging Subcommittee , Underwood R, Bax JJ, Bonow JJ, Pinto F, Gibbons RJ; Classification Subcommittee , Fox KA, Atar D, Newby LK, Galvani M, Hamm CW; Intervention Subcommittee , Uretsky BF, Steg PG, Wijns W, Bassand JP, Menasche P, Ravkilde J; Trials & Registries Subcommittee , Ohman EM, Antman EM, Wallentin LC, Armstrong PW, Simoons ML; Trials & Registries Subcommittee , Januzzi JL, Nieminen MS, Gheorghiade M, Filippatos G; Trials & Registries Subcommittee , Luepker RV, Fortmann SP, Rosamond WD, Levy D, Wood D; Trials & Registries Subcommittee , Smith SC, Hu D, Lopez‐Sendon JL, Robertson RM, Weaver D, Tendera M, Bove AA, Parkhomenko AN, Vasilieva EJ, Mendis S; ESC Committee for Practice Guidelines (CPG) , Bax JJ, Baumgartner H, Ceconi C, Dean V, Deaton C, Fagard R, Funck‐Brentano C, Hasdai D, Hoes A, Kirchhof P, Knuuti J, Kolh P, McDonagh T, Moulin C, Popescu BA, Reiner Z, Sechtem U, Sirnes PA, Tendera M, Torbicki A, Vahanian A, Windecker S; Document Reviewers , Morais J, Aguiar C, Almahmeed W, Arnar DO, Barili F, Bloch KD, Bolger AF, Botker HE, Bozkurt B, Bugiardini R, Cannon C, de Lemos J, Eberli FR, Escobar E, Hlatky M, James S, Kern KB, Moliterno DJ, Mueller C, Neskovic AN, Pieske BM, Schulman SP, Storey RF, Taubert KA, Vranckx P, Wagner DR. Third universal definition of myocardial infarction. J Am Coll Cardiol. 2012;60:1581–1598.22958960 [Google Scholar]
- 28. Kikkert WJ, van Geloven N, van der Laan MH, Vis MM, Baan J Jr, Koch KT, Peters RJ, de Winter RJ, Piek JJ, Tijssen JG, Henriques JP. The prognostic value of bleeding academic research consortium (BARC)‐defined bleeding complications in ST‐segment elevation myocardial infarction: a comparison with the TIMI (Thrombolysis In Myocardial Infarction), GUSTO (Global Utilization of Streptokinase and Tissue Plasminogen Activator for Occluded Coronary Arteries), and ISTH (International Society on Thrombosis and Haemostasis) bleeding classifications. J Am Coll Cardiol. 2014;63:1866–1875. [DOI] [PubMed] [Google Scholar]
- 29. Higgins JPT, Deeks JJ. The Cochrane Collaboration. Chapter 7: selecting studies and collecting data. Cochrane handbook for systematic reviews of interventions version 5.1. 2011. http://www.cochrane-handbook.org. Accessed March 1, 2011.
- 30. Oremus M, Oremus C, Hall GB, McKinnon MC; ECT & Cognition Systematic Review Team . Inter‐rater and test‐retest reliability of quality assessments by novice student raters using the Jadad and Newcastle‐Ottawa Scales. BMJ Open. 2012;2:e001368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Sanderson S, Tatt ID, Higgins JP. Tools for assessing quality and susceptibility to bias in observational studies in epidemiology: a systematic review and annotated bibliography. Int J Epidemiol. 2007;36:666–676. [DOI] [PubMed] [Google Scholar]
- 32. Knobloch K, Yoon U, Vogt PM. Preferred reporting items for systematic reviews and meta‐analysis (PRISMA) statement and publication bias. J Craniomaxillofac Surg. 2011;39:91–92. [DOI] [PubMed] [Google Scholar]
- 33. Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, Rennie D, Moher D, Becker BJ, Sipe TA, Thacker SB; Meta‐analysis of Observational Studies in Epidemiology (MOOSE) group. Meta‐analysis of observational studies in epidemiology: a proposal for reporting. JAMA. 2000;283:2008–2012. [DOI] [PubMed] [Google Scholar]
- 34. Sibbald M, Yan AT, Huang W, Fox KA, Gore JM, Steg PG, Eagle KA, Brieger D, Montalescot G, Goodman SG. Association between smoking, outcomes, and early clopidogrel use in patients with acute coronary syndrome: insights from the Global Registry of Acute Coronary Events. Am Heart J. 2010;160:855–861. [DOI] [PubMed] [Google Scholar]
- 35. Kang SH, Suh JW, Choi DJ, Chae IH, Cho GY, Youn TJ, Cho YS, Yoon CH, Oh IY, Cho MC, Kim YJ, Chae SC, Kim JH, Ahn YK, Jeong MH; KAMIR/KorMI Registry . Cigarette smoking is paradoxically associated with low mortality risk after acute myocardial infarction. Nicotine Tob Res. 2013;15:1230–1238. [DOI] [PubMed] [Google Scholar]
- 36. Zhang M, Liu X, Wang L, Wang Y, Ju L, Li J. Cigarette smoking might weaken the prognostic significance of cytochrome P4502C19*2 polymorphism in acute myocardial infarction patients. J Cell Mol Med. 2016;20:1247–1254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Kim HL, Suh JW, Lee SP, Kang HJ, Koo BK, Cho YS, Youn TJ, Chae IH, Choi DJ, Rha SW, Bae JH, Kwon TG, Bae JW, Cho MC, Kim HS. Cilostazol eliminates adverse smoking outcome in patients with drug‐eluting stent implantation. Circ J. 2014;78:1420–1427. [DOI] [PubMed] [Google Scholar]
- 38. Ashby DT, Dangas G, Mehran R, Lansky AJ, Fahy MP, Iakovou I, Satler LF, Pichard AD, Kent KM, Stone GW, Leon MB. Comparison of one‐year outcomes after percutaneous coronary intervention among current smokers, ex‐smokers, and nonsmokers. Am J Cardiol. 2002;89:221–224. [DOI] [PubMed] [Google Scholar]
- 39. Tan NS, Goodman SG, Cantor WJ, Tan MK, Yan RT, Bagnall AJ, Mehta SR, Fitchett D, Strauss BH, Yan AT; TRANSFER‐AMI Investigators . Comparison of the efficacy of pharmacoinvasive management for ST‐segment elevation myocardial infarction in smokers versus non‐smokers (from the Trial of Routine Angioplasty and Stenting After Fibrinolysis to Enhance Reperfusion in Acute Myocardial Infarction). Am J Cardiol. 2014;114:955–961. [DOI] [PubMed] [Google Scholar]
- 40. Adamo M, Costa F, Vranckx P, Leonardi S, Navarese EP, Garcia‐Garcia HM, Valgimigli M. Does smoking habit affect the randomized comparison of 6 versus 24‐month dual antiplatelet therapy duration? insights from the PRODIGY trial. Int J Cardiol. 2015;190:242–245. [DOI] [PubMed] [Google Scholar]
- 41. Bossard M, Granger CB, Tanguay JF, Montalescot G, Faxon DP, Jolly SS, Widimsky P, Niemela K, Steg PG, Natarajan MK, Gao P, Fox KAA, Yusuf S, Mehta SR. Double‐dose versus standard‐dose clopidogrel according to smoking status among patients with acute coronary syndromes undergoing percutaneous coronary intervention. J Am Heart Assoc. 2017;6:e006577 DOI: 10.1161/JAHA.117.006577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Ovbiagele B, Wang J, Johnston SC, Wang A, Wang D, Wang Y, Zhao X, Wang Y. Effect of clopidogrel by smoking status on secondary stroke prevention. Circulation. 2017;135:315–316. [DOI] [PubMed] [Google Scholar]
- 43. Kodaira M, Miyata H, Numasawa Y, Ueda I, Maekawa Y, Sueyoshi K, Ishikawa S, Ohki T, Negishi K, Fukuda K, Kohsaka S. Effect of smoking status on clinical outcome and efficacy of clopidogrel in acute coronary syndrome. Circ J. 2016;80:1590–1599. [DOI] [PubMed] [Google Scholar]
- 44. Siasos G, Kioufis S, Oikonomou E, Zaromitidou M, Maniatis K, Vavuranakis M, Kokkou E, Tousoulis D. Author information impact of C34T P2Y12 ADP receptor polymorphism and smoking status on cardiovascular outcome in coronary artery disease patients receiving clopidogrel. Int J Cardiol. 2016;210:161–163. [DOI] [PubMed] [Google Scholar]
- 45. Chandrasekhar J, Baber U, Sartori S, Aquino M, DeFranco A, Muhlestein JB, Weiss S, Henry TD, Effron MB, Strauss CE, Keller SY, Baker BA, Weintraub W, Chao J, Poddar KL, Rao S, Kapadia S, Kini A, Mehran R. Impact of prasugrel versus clopidogrel in smokers and non‐smokers undergoing PCI for ACS: results from the PROMETHEUS Study. 27th Annual Symposium on Transcatheter Cardiovascular Therapeutics (TCT). 2015;66:85–86. San Francisco. CA, United States. Accessed October 11, 2015.
- 46. Wakabayashi K, Romaguera R, Laynez‐Carnicero A, Maluenda G, Ben‐Dor I, Sardi G, Gaglia MA Jr, Mahmoudi M, Gonzalez MA, Delhaye C, Torguson R, Xue Z, Suddath WO, Satler LF, Kent KM, Pichard AD, Lindsay J, Waksman R. Impact of smoking on acute phase outcomes of myocardial infarction. Coron Artery Dis. 2011;22:217–222. [DOI] [PubMed] [Google Scholar]
- 47. Ferreiro JL, Bhatt DL, Ueno M, Bauer D, Angiolillo DJ. Impact of smoking on long‐term outcomes in patients with atherosclerotic vascular disease treated with aspirin or clopidogrel: insights from the CAPRIE trial (Clopidogrel Versus Aspirin in Patients at Risk of Ischemic Events). J Am Coll Cardiol. 2014;63:769–777. [DOI] [PubMed] [Google Scholar]
- 48. Lee MH, Park JJ, Yoon CH, Cha MJ, Park SD, Oh IY, Suh JW, Cho YS, Youn TJ, Rha SW, Yu CW, Gwon HC, Jang Y, Kim HS, Chae IH, Choi DJ. Impact of smoking status on clinical outcomes after successful chronic total occlusion intervention: Korean national registry of CTO intervention. Catheter Cardiovasc Interv. 2016;87:1050–1062. [DOI] [PubMed] [Google Scholar]
- 49. Cornel JH, Ohman EM, Neely B, Clemmensen P, Sritara P, Zamoryakhin D, Armstrong PW, Prabhakaran D, White HD, Fox KA, Gurbel PA, Roe MT; TRILOGY ACS Investigators . Impact of smoking status on platelet function and clinical outcomes with prasugrel vs. clopidogrel in patients with acute coronary syndromes managed without revascularization: insights from the TRILOGY ACS trial. Am Heart J. 2014;168:76–87. [DOI] [PubMed] [Google Scholar]
- 50. Cornel JH, Becker RC, Goodman SG, Husted S, Katus H, Santoso A, Steg G, Storey RF, Vintila M, Sun JL, Horrow J, Wallentin L, Harrington R, James S. Prior smoking status, clinical outcomes, and the comparison of ticagrelor with clopidogrel in acute coronary syndromes: insights from the PLATelet inhibition and patient Outcomes (PLATO) trial. Am Heart J. 2012;164:334–342. [DOI] [PubMed] [Google Scholar]
- 51. Ciccarelli G, Barbato E, Golino M, Cimmino G, Bartunek J, Serafino L, Di Girolamo D, De Bruyne B, Wijns W, Golino P. Prognostic factors in patients with STEMI undergoing primary PCI in the clopidogrel era: role of dual antiplatelet therapy at admission and the smoking paradox on long‐term outcome. J Interv Cardiol. 2017;30:5–15. [DOI] [PubMed] [Google Scholar]
- 52. Weisz G, Kirtane AJ, Xu K, Stuckey T, Witzenbichler B, Rinaldi M, Neumann FJ, Metzger DC, Henry TD, Cox D, Duffy PL, Brodie BR, Mazzaferri EL, Parise H, Mehran R, Stone G. The effect of smoking status on platelet reactivity and long‐term events after PCI: does a clopidogrel‐related “smoker's paradox” exist? 25th Annual Symposium on Transcatheter Cardiovascular Therapeutics (TCT). 2013;62:50. San Francisco, CA, United States. Accessed October 27, 2013.
- 53. Zhao ZG, Chen M, Peng Y, Chai H, Liu W, Li Q, Ren X, Wang XQ, Luo XL, Zhang C, Huang DJ. The impact of smoking on clinical efficacy and pharmacodynamic effects of clopidogrel: a systematic review and meta‐analysis. Heart. 2014;100:192–199. [DOI] [PubMed] [Google Scholar]
- 54. Greenland S. Quantitative methods in the review of epidemiologic literature. Epidemiol Rev. 1987;9:1–30. [DOI] [PubMed] [Google Scholar]
- 55. Gagne JJ, Bykov K, Choudhry NK, Toomey TJ, Connolly JG, Avorn J. Effect of smoking on comparative efficacy of antiplatelet agents: systematic review, meta‐analysis, and indirect comparison. BMJ. 2013;347:5307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Ferreiro JL, Bhatt D, Ueno M, Bauer D, Angiolillo D. Differential effects of smoking on long‐term outcomes in patients with atherosclerotic vascular disease treated with aspirin or clopidogrel: insight from the clopidogrel versus aspirin in patients at risk of ischemic events trial. 61st Annual Scientific Session and Expo of the American‐College‐of‐Cardiology (ACC). 2012;13:257. Chicago, IL, United States. Accessed March 24, 2012.
- 57. Cho JR, Desai B, Haas MJ, Rollini F, Franchi F, Muñiz‐Lozano A, Tello‐Montoliu A, Ferrante E, Guzman LA, Bass TA, Angiolillo DJ. Impact of cigarette smoking on P2Y12 receptor binding activity before and after clopidogrel therapy in patients with coronary artery disease. J Cardiovasc Transl Res. 2014;7:47–52. [DOI] [PubMed] [Google Scholar]
- 58. Hochholzer W, Trenk D, Mega JL, Morath T, Stratz C, Valina CM, O'Donoghue ML, Bernlochner I, Contant CF, Guo J, Sabatine MS, Schömig A, Neumann FJ, Kastrati A, Wiviott SD, Sibbing D. Impact of smoking on antiplatelet effect of clopidogrel and prasugrel after loading dose and on maintenance therapy. Am Heart J. 2011;162:518–526. [DOI] [PubMed] [Google Scholar]
- 59. Morgenstern H. Uses of ecologic analysis in epidemiologic research. Am J Public Health. 1982;72:1336–1344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Berlin JA, Santanna J, Schmid CH, Szczech LA, Feldman KA; Anti‐Lymphocyte Antibody Induction Therapy Study Group . Individual patient‐ versus group‐level data meta‐regressions for the investigation of treatment effect modifiers: ecological bias rears its ugly head. Stat Med. 2002;21:371–387. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data S1. Search strategies in PubMed.
Table S1. Quality Assessment of the Included Studies Using the Newcastle–Ottawa Scale


