Abstract
Background
Differential diagnosis among psoriatic arthritis (PsA) and seronegative rheumatoid arthritis (Abneg RA) can be challenging particularly in the clinical setting of peripheral phenotype and autoantibodies seronegativity. The aim of the study was to identify synovial tissue (ST) biomarkers differentially expressed in PsA and Abneg RA and test their predictive value of therapeutic response.
Methods
Thirty-four PsA patients [12 DMARD naive and 22 non-responder to methotrexate (MTX-IR)] with peripheral joint involvement and 55 Abneg RA (27 DMARD naive and 28 MTX-IR) underwent US-guided ST biopsy and immunohistochemistry (IHC) for CD68+, CD3+, CD20+, CD21+, CD117+, and CD138+ cells. After study entry, each DMARD-naive patient started MTX therapy and was followed in an outpatient setting for at least 6 months to define the achievement of Minimal Disease Activity (PsA) and DAS remission (Abneg RA) status respectively. Each IR-MTX patient was treated according to EULAR recommendations.
Results
At study entry, IHC analysis revealed that PsA patients had comparable levels of lining and sublining CD68+ and sublining CD21+, CD20+, and CD3+ cells than Abneg RA, despite the therapeutic regimen. Moreover, regardless of the therapeutic scheme, PsA patients showed higher IHC score of CD117+ cells (p = 0.0004 and p = 0.0005 for naive and MTX-IR patients respectively) compared to Abneg RA patients. Conversely, Abneg RA patients showed higher IHC score of CD138+ cells, irrespective to the therapeutic scheme (p = 0.04 and p = 0.002 for naive and MTX-IR patients respectively). Analyzing the response rate to the therapeutic scheme, naive PsA patients reaching MDA status at 6 months follow-up, showed, at the study entry, lower IHC score of CD3+ cells compared to PsA patients not reaching this outcome (p = 0.02); conversely, naive Abneg RA patients reaching DAS remission status at 6 months follow-up, showed, at the study entry, lower IHC score of sublining CD68+ cells compared to Abneg RA patients not reaching this outcome (p < 0.001).
Conclusions
CD117+ and CD138+ cells are differentially distributed among PsA and Abneg RA. Histological analysis of ST may help to solve the clinical overlap between the two diseases and provides prognostic data about the therapy success.
Electronic supplementary material
The online version of this article (10.1186/s13075-019-1898-7) contains supplementary material, which is available to authorized users.
Keywords: Psoriatic arthritis, Rheumatoid arthritis, Synovial tissue, Autoantibodies, Response to therapy
Introduction
Psoriatic arthritis (PsA) is a chronic inflammatory disease characterized by remarkable heterogeneity of clinical presentation, including peripheral arthritis, axial involvement, enthesitis, dactylitis, nail dystrophy, uveitis, and osteitis, in addition to associated comorbidities such as cardiovascular disease, metabolic syndrome, and mood disorders [1, 2]. Most patients with PsA present with oligoarticular or polyarticular arthritis and can be differentiated from patients with rheumatoid arthritis (RA), the most common inflammatory joint disease, by specific non-articular clinical features being present, as well as the infrequent seropositivity for rheumatoid factor (RF) and anti-citrullinated peptide antibody (ACPA). These clinical features include an asymmetric distribution of the inflamed joints, the sacroiliitis or spinal involvement, the typical involvement of the distal interphalangeal joint (DIP), and the extra-articular manifestations [1]. However, in clinical practice, the differential diagnosis among PsA and RA can be challenging, particularly if the peripheral phenotype is present and RF and ACPA are negative. In recent years, there have been a number of advances made in synovial tissue biopsy techniques in patients with inflammatory joint diseases [3]. However, despite many research groups have focused on the analysis of possible differential biomarkers among PsA and RA, no studies have been performed considering a direct comparison between PsA with a RF/ACPA seronegative (Abneg) RA cohort [4, 5].
Based on that, the aims of the study were (i) to assess whether the histological characteristics of synovial tissue of PsA patients with peripheral arthritis phenotype compared to seronegative RA patients could differ in different disease phases and (ii) to evaluate possible predictive synovial biomarkers associated with treatment response in PsA and seronegative RA patients at disease onset and after c-DMARD failure.
Patients and methods
Patient enrollment
Eighty-nine patients were enrolled in the study [34 oligo-polyarticular PsA patients (12 naïve and 22 inadequately responder to methotrexate (MTX-IR) respectively) and 55 RA patients (27 naïve and 28 MTX-IR respectively)] fulfilling the classification criteria for PsA or RA [6, 7]. At baseline, demographical, clinical, and inflammatory parameters were collected for each patient. All RA patients were confirmed as being seronegative (Abneg) for IgA- and IgM-RF (Orgentec Diagnostika, Bouty, UK) and ACPA (Axis Shield Diagnostics, Bouty, UK) using commercial ELISA at study entry. Each naïve PsA and Abneg RA patient was then treated with methotrexate (up to 20 mg/week) and followed every 3 months for at least 6 months to assess the rate of achievement of Minimal Disease Activity (MDA) or DAS remission for PsA and Abneg RA patients respectively [8, 9]. MTX-IR PsA and Abneg RA patients were treated according to the current recommendations [10, 11]. The study protocol was approved by the local Ethical Committee, and all subjects provided signed informed consent.
Immunohistochemistry for CD68, CD21, CD3, CD20, CD138, CD117, and CD31 on synovial tissue
At study entry, each PsA and Abneg RA patient underwent ultrasound-guided synovial tissue biopsy of the knee. Joint inflammatory activity was tested for each patient using power Doppler as previously described [12]. Each tissue was tested through immunohistochemistry for the presence of CD68 (macrophages), CD21 (follicular dendritic cells), CD3 (T lymphocytes), CD20 (B lymphocytes), and CD31 (endothelial cells) staining following the already published protocol [12]. Other tissue sections were stained for plasma cell and mast cell detection, using CD138 mouse antihuman monoclonal antibody (clone MI15) or CD117 mouse antihuman monoclonal antibody (clone EP10) (all from Leica Biosystem, Newcastle, UK) by immunostainer BOND MAX III (Leica). Slides were examined by two independent evaluators using a light microscope (Leica DM 2000), and all tissues were evaluated using a numerical score based on the number of CD68+, CD21+, CD3+, CD20+, CD117+, and CD138+ cells (two different fields in each section), with a score of 0 indicating no positive cells, 1 indicating < 10% positive cells, 2 indicating 10–50% positive cells, and 3 indicating > 50% positive cells. CD31+ vessel count was done as mean of the values from three different fields in each section [12]. The inter-rater agreement coefficient was assessed for each single IHC marker (see Additional file 2: Table S1).
Statistical analysis
Statistical analysis was performed using SPSS V. 20.0 (SPSS. Chicago, IL, USA) and Prism software (GraphPad, San Diego, CA, USA). Categorical and quantitative variables were described as frequencies, percentage, and mean ± SD. Data on demographic and clinical features were compared between patients by the non-parametric Mann-Whitney U test or χ2 test, as appropriate. Spearman’s rank correlation test was used for correlation in all analyses. ROC analysis was performed to identify the best cut-off value for IHC scores associated with the highest rate of MDA or DAS remission achievement in PsA and Abneg RA patients respectively. A value of p ≤ 0.05 was considered statistically significant.
Results
Demographic and clinical characteristics of the enrolled study cohorts
Demographic and clinical characteristics of PsA and Abneg RA cohorts (naïve and MTX-IR respectively) enrolled in the study are summarized in Table 1. Comparing the different study cohorts, there were no significant differences according to age and gender (p > 0.05). PsA and Abneg RA patients naïve to treatment showed significant shorter disease duration (0.80 ± 0.24 years for naive PsA and 0.84 ± 0.95 Abneg RA patients respectively) compared to MTX-IR PsA and Abneg RA patients (5.83 ± 3.68 years for MTX-IR PsA, p < 0.001; 5.68 ± 5.28 for MTX-IR Abneg RA patients, p = 0.002 respectively). Considering the clinical parameters, there were no significant differences comparing PsA and Abneg RA patients based on swollen and tender joint counts or Disease Activity Scores in naïve and in MTX-IR subgroups (Table 1). However, considering inflammatory markers, ESR plasma levels were significantly higher in naïve Abneg RA patients (44.00 ± 25.40 mm/first hour) than MTX-IR Abneg RA patients (29.60 ± 36.94 mm/first hour; p = 0.03), whereas no differences were found for CRP plasma levels comparing naïve and MTX-IR PsA subgroups (Table 1).
Table 1.
Demographical, clinical, and immunological characteristics of the study cohorts
| PsA (n = 34) | p | Abneg RA (n = 55) | p 1 | p 2 | p 3 | |||
|---|---|---|---|---|---|---|---|---|
| Naive (n = 12) | MTX-IR (n = 22) | Naive (n = 27) | MTX-IR (n = 28) | |||||
| Age, years (mean ± SD) | 54.75 ± 17.18 | 60.18 ± 9.81 | 0.47 | 52.41 ± 17.70 | 58.36 ± 16.58 | 0.16 | 0.68 | 0.77 |
| Female, n (%) | 4 (33.3) | 14 (63.6) | 0.10 | 23 (85.2) | 23 (82.1) | 0.76 | 0.001 | 0.14 |
| Disease duration, years (mean ± SD) | 0.80 ± 0.24 | 5.83 ± 3.68 | < 0.001 | 0.84 ± 0.95 | 5.68 ± 5.28 | 0.002 | 0.44 | 0.49 |
| DAS44 (mean ± SD) | 3.00 ± 0.42 | 3.34 ± 0.91 | 0.34 | 3.37 ± 1.05 | 3.22 ± 0.79 | 0.71 | 0.52 | 0.81 |
| DAPSA (mean ± SD) | 22.55 ± 8.38 | 28.04 ± 10.14 | 0.10 | – | – | – | – | – |
| SJC (mean ± SD) | 4.00 ± 2.67 | 6.43 ± 5.69 | 0.35 | 8.22 ± 7.95 | 7.15 ± 5.53 | 0.93 | 0.18 | 0.54 |
| TJC (mean ± SD) | 5.00 ± 3.16 | 7.00 ± 6.13 | 0.66 | 8.22 ± 7.86 | 8.44 ± 5.46 | 0.33 | 0.51 | 0.24 |
| ESR, mm/1st hour (mean ± SD) | 29.60 ± 36.94 | 29.10 ± 19.59 | 0.44 | 44.00 ± 25.40 | 37.04 ± 27.25 | 0.19 | 0.03 | 0.44 |
| CRP, mg/l (mean ± SD) | 16.70 ± 29.16 | 18.11 ± 23.41 | 0.70 | 16.99 ± 19.43 | 16.77 ± 24.23 | 0.77 | 0.72 | 1.00 |
| Ab positivity, n (%) | 0 (0.0) | 0 (0.0) | 1.00 | 0 (0.0) | 0 (0.0) | 1.00 | 1.00 | 1.00 |
Data presented in italics have p < 0.05
PsA psoriatic arthritis, RA rheumatoid arthritis, Ab autoantibody, DAS Disease Activity Score, DAPSA Disease Activity in PSoriatic Arthritis, SJC swollen joint count, TJC tender joint count, ESR erythrocyte sedimentation rate, CRP C-reactive protein, SD standard deviation, MTX-IR methotrexate inadequately responder, p naive PsA vs MTX-IR PsA, p1 naive Abneg RA vs MTX-IR Abneg RA, p2 naive PsA vs naive Abneg RA, p3 MTX-IR PsA vs MTX-IR Abneg RA
PsA and Abneg RA show similar histological features in terms of synovial resident CD68+, CD21+, and CD3+ cells and microanatomical organization
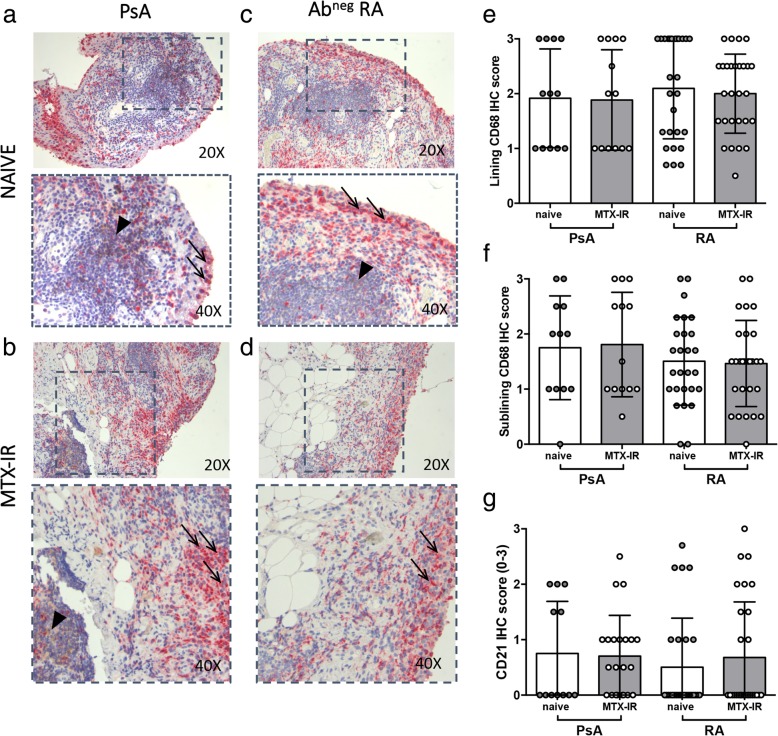
Each enrolled patient underwent US-guided ST biopsy, and IHC for CD68+, CD21+, CD20+, and CD3+ was performed. PsA patients showed similar IHC CD68+ cell score in the lining (1.95 ± 0.90) and sublining (1.81 ± 0.95) compared to Abneg RA patients (1.96 ± 0.75 for lining CD68+ cells, p = 0.94; 1.43 ± 0.76 for sublining CD68+ cells, p = 0.11 respectively) regardless of the treatment scheme (Fig. 1a–d). Furthermore, IHC analysis revealed that PsA and Abneg RA patients had comparable levels of CD21+ cells (0.72 ± 0.79 vs 0.60 ± 0.95; p = 0.18 respectively) regardless of the treatment scheme (Fig. 1a–d).
Fig. 1.
a–g IHC staining for CD68/CD21 on ST of patients with naïve or MTX-IR PsA and Abneg RA patients. Example photos of CD68 (RED)/CD21(DAB) staining of ST biopsies from patients with naïve (a) or MTX-IR (b) PsA patients and with naïve (c) or MTX-IR (d) Abneg RA (magnification × 20 and magnification × 40 in the corresponding inset). Black arrows indicate CD68+ cells (red), and black arrow head indicates CD21+ cells (brown) in the corresponding inset. e Lining IHC score for CD68+ cells in PsA and Abneg RA patients divided by treatment regimen. f Sublining IHC score for CD68+ cells in PsA and Abneg RA patients divided by treatment regimen. g IHC score for CD21+ cells in PsA and Abneg RA patients divided by treatment regimen. IHC immunohistochemistry, PsA psoriatic arthritis, RA rheumatoid arthritis, Ab autoantibody, MTX-IR methotrexate inadequately responder, CD cluster designation
Stratifying the study cohorts according to the treatment scheme, naïve PsA showed similar IHC scores for lining (2.00 ± 0.85) and sublining CD68+ cells (1.71 ± 0.96) compared to naïve Abneg RA patients (1.96 ± 0.79 for lining CD68+ cells, p = 1.00; 1.44 ± 0.80 for sublining CD68+ cells, p = 0.39 respectively). Moreover, naïve PsA showed similar IHC scores for CD21+ cells (0.75 ± 0.94) compared to naïve Abneg RA patients (0.52 ± 0.91; p = 0.43) (Fig. 1g), showing a direct correlation between ESR plasma levels and lining CD68+ cells IHC score (r = 0.66; p = 0.04). Similarly, MTX-IR PsA patients showed comparable IHC scores for lining (1.92 ± 0.95) and sublining CD68+ cells (1.86 ± 0.96) than MTX-IR Abneg RA patients (2.00 ± 0.72 for lining CD68+ cells, p = 0.79; 1.46 ± 0.78 for sublining CD68+ cells, p = 0.25) (Fig. 1e–h). Finally, MTX-IR PsA showed similar IHC scores for CD21+ cells (0.71 ± 0.73) compared to MTX-IR Abneg RA patients (0.68 ± 1.00; p = 0.91) (Fig. 1g).
Analyzing the microanatomical organization of the synovial tissue infiltrates, 15 (44.1%) PsA patients compared to 24 (43.6%, p = 0.51) Abneg RA patients showed follicular synovitis regardless of the treatment regimen. Moreover, there was no difference in the rate of follicular synovitis stratifying patients according to the therapeutic regimen (41.7% of naïve PsA patients with follicular synovitis compared to 45.5% of MTX-IR PsA patients with similar synovitis pattern, p = 0.79; 44.4% of naïve Abneg RA patients with follicular synovitis compared to 42.8% of MTX-IR Abneg RA patients with similar synovitis pattern, p = 0.51).
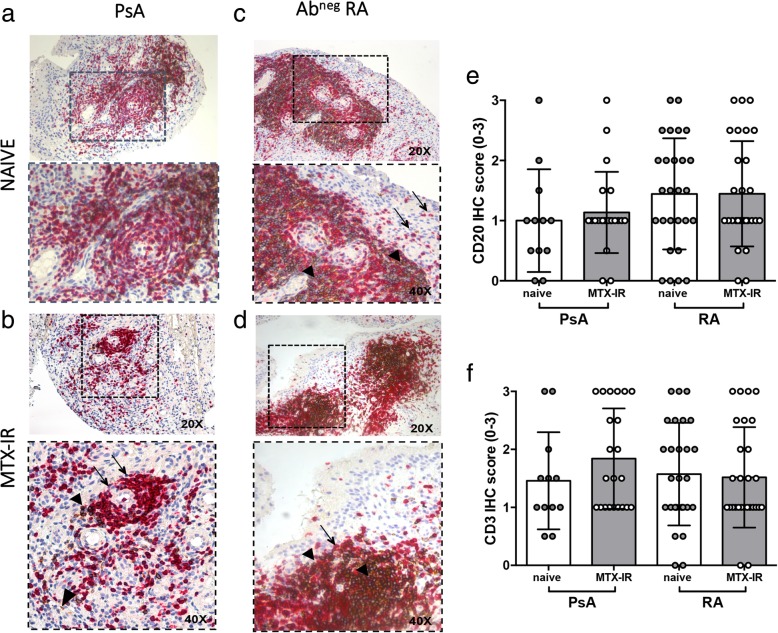
CD20 IHC revealed that PsA and Abneg RA patients had similar levels of CD20+ cells (1.08 ± 0.73 in PsA vs 1.44 ± 0.89 in Abneg RA, p = 0.07) (Fig. 2a–d) and after stratification based on the treatment regimen, PsA patients showed similar IHC CD20+ cell score than Abneg RA (1.00 ± 0.85 in naive PsA vs 1.44 ± 0.92 in naive Abneg RA, p = 0.16; 1.14 ± 0.67 in MTX-IR PsA vs 1.45 ± 0.87 MTX-IR Abneg RA, p = 0.18) (Fig. 2e).
Fig. 2.
a–f IHC staining for CD3/CD20 on ST of patients with naïve or MTX-IR PsA and Abneg RA patients. Example photos of CD3 (RED)/CD20 (DAB) staining of ST biopsies from patients with naïve (a) or MTX-IR (b) PsA patients and with naïve (c) or MTX-IR (d) Abneg RA patients (magnification × 20 and magnification × 40 in the corresponding inset). Thin black arrows indicate CD3+ cells (red), and black arrow heads indicate CD20+ cells (brown) in the corresponding inset. e IHC scores for CD20+ cells in PsA and Abneg RA patients divided by treatment regimen. f IHC scores for CD3+ cells in PsA and Abneg RA patients divided by treatment regimen. IHC immunohistochemistry, PsA psoriatic arthritis, RA rheumatoid arthritis, Ab autoantibody, MTX-IR methotrexate inadequately responder, CD cluster designation
Analyzing synovial CD3+ cell distribution, IHC showed that PsA and Abneg RA patients had similar levels of synovial CD3+ cells (1.71 ± 0.86 in PsA and 1.54 ± 0.86 in Abneg RA; p = 0.45 respectively) (Fig. 2a–d). Moreover, PsA and Abneg RA patients did not differ in terms of synovial CD3+ cells stratifying patients based on the treatment scheme (1.45 ± 0.83 in naive PsA vs 1.57 ± 0.88 in naïve Abneg RA, p = 0.70; 1.84 ± 0.86 in MTX-IR PsA vs 1.52 ± 0.87 MTX-IR Abneg RA, p = 0.19) (Fig. 2f).
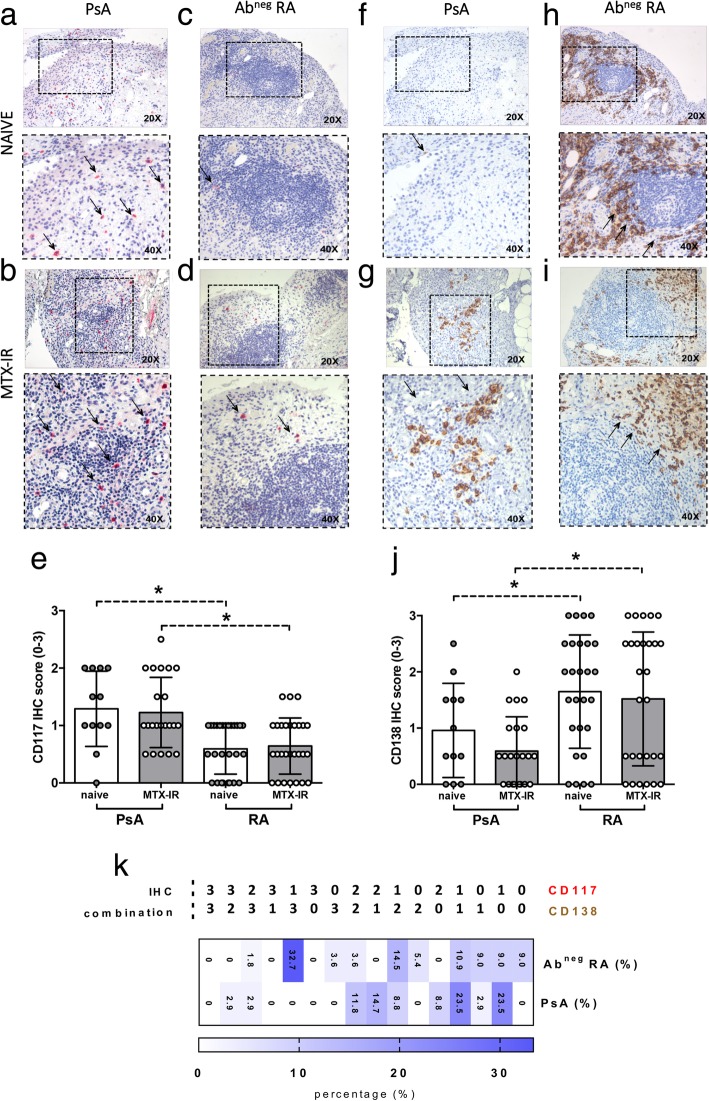
Synovial CD117+ and CD138+ cells are differentially distributed in PsA and Abneg RA patients
CD117 IHC showed that PsA patients are characterized by higher IHC scores for CD117+ cells (1.25 ± 0.61) compared to Abneg RA patients (0.62 ± 0.46, p < 0.001) regardless of the treatment scheme (Fig. 3a–d). Interestingly, stratifying the study cohorts according to the treatment scheme, naïve PsA patients showed higher CD117+ cell IHC scores (1.29 ± 0.65) compared to naïve Abneg RA patients (0.59 ± 0.44; p = 0.0004). Similarly, MTX-IR PsA patients showed higher CD117+ IHC score (1.23 ± 0.61) compared to MTX-IR Abneg RA patients (0.64 ± 0.48, p = 0.001) (Fig. 3e).
Fig. 3.
a–k IHC staining for CD117 and CD138 on ST of patients with naïve or MTX-IR PsA and Abneg RA patients. Example photos of CD117 (RED) staining of ST biopsies from patients with naïve (a) or MTX-IR (b) PsA and with naïve (c) or MTX-IR (d) Abneg RA (magnification × 20 and magnification × 40 in the corresponding inset). Thin black arrows indicate CD117+ cells (red) in the corresponding inset. e IHC scores for CD117+ cells in PsA and Abneg RA patients divided by treatment regimen; *p = 0.0004, naïve PsA vs naïve Abneg RA patients; *p = 0.0005, MTX-IR PsA vs MTX-IR Abneg RA patients. Example photos of CD138 (DAB) staining of ST biopsies from patients with naïve (f) or MTX-IR (g) PsA and with naïve (h) or MTX-IR (i) Abneg RA (magnification × 20 and magnification × 40 in the corresponding inset). Thin black arrows indicate CD138+ cells (brown) in the corresponding inset. j IHC scores for CD138+ cells in PsA and Abneg RA patients divided by treatment regimen; *p = 0.04, naive PsA vs naive AbnegRA patients; *p = 0.002, MTX-IR PsA vs MTX-IR Abneg RA patients. k Rate of distribution of CD117/CD138 IHC combination differentially distributed among PsA and Abneg RA patients. IHC immunohistochemistry, PsA psoriatic arthritis, RA rheumatoid arthritis, Ab autoantibody, MTX-IR methotrexate inadequately responder, CD cluster designation
Conversely, analyzing the distribution of CD138+ cells within the synovial tissue, Abneg RA patients showed higher IHC scores for CD138+ cells (1.58 ± 1.09) compared to PsA patients (0.72 ± 0.71; p < 0.001) independently of the treatment scheme (Fig. 3f–i). Considering the different study population subgroups, naive Abneg RA patients were characterized by higher CD138+ cell IHC score (1.65 ± 1.00) compared to naive PsA patients (0.96 ± 0.84; p = 0.04). Similarly, MTX-IR Abneg RA patients had higher CD138+ cell IHC score (1.51 ± 1.19) compared to MTX-IR PsA patients (0.59 ± 0.61, p = 0.002) (Fig. 3j). As shown in Fig. 3k, the combination of low IHC score for CD117+ (IHC score = 1) and high IHC score for CD138+ cells (IHC score = 3) significantly differentiates PsA than Abneg RA synovitis [OR (95% CI), 34.04 (1.535–2.398); p = 0.0002].
Synovial CD31+ vessel count is associated with the disease phase in PsA and Abneg RA patients
To assess the microvasculature at the synovial tissue level, each tissue was tested for the presence of CD31+ blood vessels. As shown in Fig. 4a–e, naïve PsA patients showed higher number of synovial CD31+ vessels (36.63 ± 11.02) compared to MTX-IR PsA patients (23.65 ± 11.67; p = 0.01). Similarly, naïve Abneg RA patients showed a higher number of synovial CD31+ vessels (35.43 ± 7.23) than MTX-IR Abneg RA patients (22.14 ± 5.32; p < 0.001). No significant differences in terms of CD31+ vessel count were found comparing PsA and Abneg RA patients stratified based on the same treatment category (Fig. 4e).
Fig. 4.
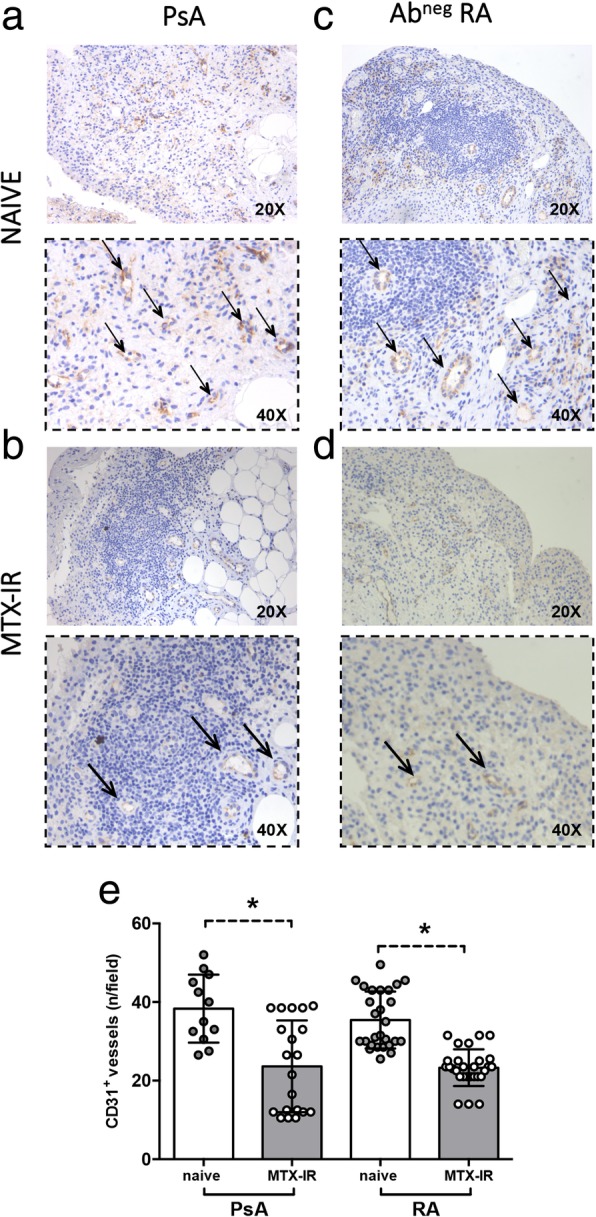
a–e IHC staining for CD31 on ST of patients with naïve or MTX-IR PsA and Abneg RA patients. Example photos of CD31 (DAB) staining of ST biopsies from patients with naïve (a) or MTX-IR (b) PsA and with naïve (c) or MTX-IR (d) Abneg RA (magnification × 20 and magnification × 40 in the corresponding inset). Thin black arrows indicate CD31+ vessels (brown) in the corresponding inset. e Synovial CD31+ vessel count in PsA and Abneg RA patients divided by treatment regimen; *p = 0.01, naïve vs MTX-IR PsA patients; *p < 0.001, naïve vs MTX-IR Abneg RA patients. IHC immunohistochemistry, PsA psoriatic arthritis, RA rheumatoid arthritis, Ab autoantibody, MTX-IR methotrexate inadequately responder, CD cluster designation
Baseline synovial CD3+ and CD68+ cell IHC scores are associated with Minimal Disease Activity and remission achievement in PsA and Abneg RA patients naïve to treatment respectively
After study enrollment, each naïve patient started methotrexate treatment and was followed for at least 6 months on outpatient setting and treatment response rate was recorded at each clinical assessment (MDA for PsA and DAS remission for Abneg RA patients respectively). After 6 months follow-up from methotrexate beginning, 6 (50.0%) PsA patients achieved MDA, while 11 (40.7%) Abneg RA patients achieved DAS remission. As shown in Table 2, naïve PsA patients who reached the MDA status at 6 months follow-up had lower CD3+ cell IHC score (0.91 ± 0.33) before methotrexate beginning, compared to naïve PsA patients who did not achieve the clinical endpoint at 6 months (1.99 ± 0.84; p = 0.02). Conversely, naïve Abneg RA patients who reached the DAS remission status at 6 months follow-up had lower sublining CD68+ cell IHC score (0.92 ± 0.58) before methotrexate beginning, compared to naïve Abneg RA patients who did not achieved the clinical endpoint at 6 months (2.13 ± 0.55; p < 0.001) (Table 2). To define the best cut-off value for CD3+ cell scores and CD68+ cell sublining scores in naive PsA and Abneg RA respectively, ROC analysis was performed for each parameter (Additional file 1: Figure S1a-b). In particular, naive PsA patients who reached a MDA status after 6 months follow-up had more likely baseline CD3+ cell scores< 1.25 (83.3%) than naive PsA patients who did not reach MDA status (16.7%, p = 0.02). Moreover, naive Abneg RA patients who reached DAS remission after 6 months follow-up had more likely baseline CD68+ cell sublining scores < 2.25 (65.0%) than naive Abneg RA patients who did not reach DAS remission (0.0%, p = 0.03). In relation to MTX-IR patients, there was no significant difference in MDA and DAS remission status achievement in PsA and Abneg RA patients respectively, even stratifying patients based on the therapeutic strategy after MTX failure (c-DMARD combination or addition of b-DMARDs) (40.0% MTX-IR PsA patients achieved MDA after c-DMARD combination vs 64.7% MTX-IR PsA patients achieved MDA after b-DMARD addition, p = 0.32; 44.4% MTX-IR Abneg RA patients achieved MDA after c-DMARD combination vs 50.0% MTX-IR Abneg RA patients achieved MDA after b-DMARD addition, p = 0.78).
Table 2.
IHC features of PsA and Abneg RA cohorts based on the achievement of MDA and DAS remission after 6 months follow-up
| PsA cohort | ||||||
| Naive (N = 12) | p | MTX-IR (N = 22) | p 1 | |||
| MDA (N = 6) | No MDA (N = 6) | MDA (N = 9) | No MDA (N = 13) | |||
| Follicular synovitis, n (%) | 3 (50.0) | 3 (50.0) | 1.00 | 3 (33.3) | 7 (53.8) | 0.34 |
| CD68 (L) (mean ± SD) | 1.58 ± 0.80 | 2.42 ± 0.73 | 0.09 | 1.86 ± 0.90 | 2.0 ± 1.07 | 0.77 |
| CD68 (SL) (mean ± SD) | 1.25 ± 1.08 | 2.16 ± 0.61 | 0.13 | 1.77 ± 0.93 | 2.0 ± 1.07 | 0.54 |
| CD21 (mean ± SD) | 0.58 ± 0.92 | 0.92 ± 1.02 | 0.59 | 0.96 ± 0.80 | 0.33 ± 0.43 | 0.06 |
| CD20 (mean ± SD) | 0.83 ± 0.82 | 1.17 ± 0.93 | 0.56 | 1.19 ± 0.83 | 1.06 ± 0.40 | 0.64 |
| CD3 (mean ± SD) | 0.91 ± 0.33 | 1.99 ± 0.84 | 0.02 | 1.65 ± 0.77 | 2.11 ± 0.96 | 0.29 |
| CD117 (mean ± SD) | 1.16 ± 0.82 | 1.42 ± 0.49 | 0.62 | 1.12 ± 0.62 | 1.39 ± 0.61 | 0.32 |
| CD138 (mean ± SD) | 0.67 ± 0.75 | 1.25 ± 0.88 | 0.25 | 0.58 ± 0.64 | 0.61 ± 0.60 | 0.84 |
| CD31+ vessels (mean ± SD) | 32.50 ± 13.41 | 40.75 ± 6.84 | 0.39 | 22.81 ± 11.31 | 25.21 ± 13.06 | 0.58 |
| Abneg RA cohort | ||||||
| Naive (N = 27) | p 2 | MTX-IR (N = 28) | p 3 | |||
| REM (N = 11) | No REM (N = 16) | REM (N = 11) | No REM (N = 17) | |||
| Follicular synovitis, n (%) | 6 (46.2) | 6 (42.6) | 0.86 | 3 (27.3) | 7 (41.2) | 0.45 |
| CD68 (L) (mean ± SD) | 1.86 ± 0.82 | 2.23 ± 0.68 | 0.27 | 2.10 ± 0.51 | 1.97 ± 0.83 | 0.82 |
| CD68 (SL) (mean ± SD) | 0.92 ± 0.58 | 2.13 ± 0.55 | < 0.001 | 1.35 ± 0.78 | 1.46 ± 0.76 | 0.66 |
| CD21 (mean ± SD) | 0.39 ± 0.74 | 0.65 ± 1.08 | 0.79 | 0.54 ± 0.79 | 0.77 ± 1.13 | 0.78 |
| CD20 (mean ± SD) | 1.64 ± 1.02 | 1.23 ± 0.78 | 0.26 | 1.46 ± 0.99 | 1.44 ± 0.83 | 0.85 |
| CD3 (mean ± SD) | 1.57 ± 0.95 | 1.57 ± 0.83 | 0.98 | 1.59 ± 0.89 | 1.47 ± 0.87 | 0.49 |
| CD117 (mean ± SD) | 0.57 ± 0.43 | 0.62 ± 0.46 | 0.79 | 0.77 ± 0.56 | 0.56 ± 0.43 | 0.28 |
| CD138 (mean ± SD) | 1.54 ± 0.99 | 1.77 ± 1.05 | 0.55 | 1.36 ± 1.07 | 1.62 ± 1.28 | 0.47 |
| CD31+ vessels (mean ± SD) | 35.64 ± 7.69 | 34.64 ± 6.89 | 0.54 | 22.35 ± 5.28 | 21.88 ± 5.78 | 0.52 |
Data presented in italics have p < 0.05
PsA psoriatic arthritis, Ab autoantibody, RA rheumatoid arthritis, MDA Minimal Disease Activity, REM remission, DAS Disease Activity Score, p naive PsA reaching MDA vs naive PsA not achieving MDA, p1 MTX-IR PsA reaching MDA vs MTX-IR PsA not achieving MDA, p2 naive ABneg RA reaching DAS remission vs naive ABneg RA not achieving DAS remission, p3 MTX-IR ABneg RA reaching DAS remission vs MTX-IR ABneg RA not achieving DAS remission, MTX-IR methotrexate inadequately responder
Discussion
As in RA, synovial membrane inflammation plays a key pathogenetic role in PsA and many studies have focused on this topic in the last decades, especially looking for differential synovial tissue biomarkers between different types of chronic inflammatory joint diseases. More recently, the new advances in collecting synovial tissue through minimally invasive techniques have provided insight into the pathogenetic mechanisms of such joint diseases and facilitate differential diagnosis, stratification prognosis, and identification of treatment effects and new therapeutic targets [13].
In our study, we investigated, for the first time, the synovial histological features of a selected cohort of PsA patients, with peripheral joint involvement, compared to ACPA/RF seronegative RA cohort stratified based on the disease phase (disease onset and after DMARD insufficient response respectively) finding differential histological features of synovial tissue inflammation composition and biomarkers of therapeutic response.
Both RA and PsA are systemic autoimmune diseases characterized by chronic inflammation of the joint which leads to the destruction of the cartilage and bone [14, 15]. In particular, the clinical presentation of PsA is heterogeneous, variably involving the synovium of peripheral joints, spine, and/or entheses [6]. Moreover, PsA patients may develop articular structural damage both in terms of erosions and new bone formation and may also develop systemic complications including the development of metabolic syndrome and increased cardiovascular risk with concomitant reduced life expectancy [16, 17]. The synovium in PsA represents a primary target of disease pathogenesis, together with the skin and entheses, with a distinct gene signature compared to healthy and other joint diseases [18]. Thus, it is an intriguing and plausible site in exploring important mechanisms of the disease.
Multiple studies have characterized the histological features of synovitis in PsA compared with RA, proving that there are some substantial histological differences; notably, the PsA synovitis was shown to be characterized by less pronounced lining layer hyperplasia and fewer monocytes/macrophages than are seen in RA [19–21]. Given that it has already been clearly shown that oligoarticular and polyarticular PsA presents comparable histopathological characteristics [4], in PsA, there is a higher grade of synovial vascularization, with a different vascular pattern, characterized by immature, tortuous, and branched vessels, compared with the straight blood vessels more likely observed in RA [22].
Despite these advances, currently, there are no differential synovial tissue biomarkers between PsA and RA especially if the latter is diagnosed in a clinical setting of negativity for ACPA and RF antibodies. This particular setting may create difficulties in terms of differential diagnosis and prognosis in the earliest undifferentiated phase of the disease. To address this issue, we have previously demonstrated that synovial tissue analysis in terms of histological, ultrasound, and epigenetic signature may support the clinician in the identification of patients with ACPA/RF seronegative undifferentiated arthritis with high likelihood chance of clinical differentiation towards definite arthritis (PsA or RA) [23]. In particular, synovial tissue enriched with CD68+ and CD3+ cells and high CD31+ vessels characterized ACPA/RF seronegative undifferentiated arthritis patients evolving into Abneg RA or PsA [23].
Comparing PsA and Abneg RA, histological analysis of synovial tissue composition revealed similarities in lining and sublining CD68+ and sublining CD21+, CD20+, and CD3+ cell distribution. Previously, controversial data were reported about CD3+ cell distribution among PsA and RA. However, no study has been conducted selecting Abneg RA patients only as the comparison group and multiple methods of synovial tissue collection were used (arthroscopic or needle biopsies vs tissue obtained during joint replacement surgery) [24–26]. Other inflammatory cells such as mast cells and plasma cells take part in the tissue inflammatory infiltrate in PsA and RA [27, 28]. In particular, CD117+ cells have been previously shown more likely in synovial tissue of SpA, including PsA, expressing significantly more interleukin-17 than in RA synovitis regardless of TNF inhibition [27]. In our cohort, synovial tissue of PsA patients was found to be enriched in CD117+ cells in the sublining area compared to Abneg RA irrespective of the disease phase. Interestingly, CD117+ distribution was found more likely in the context of tissue lymphoid aggregates. Conversely, synovial tissue of Abneg RA patients was found to be enriched in CD138+ cells compared to PsA synovial tissue, underlining the crucial role of B lymphocytes in RA pathogenesis and suggesting the need to investigate additional autoantibody specificities despite ACPA/RF negativity in such patient category. Moreover, the detection of lympho-neogenesis is a frequent feature of PsA synovitis with the expression of peripheral lymph node addressin-positive high endothelial venules and CXCL13/CCL21 expression demonstrating that the microanatomical bases for germinal center formation are present in PsA synovial tissue [29]. In this context, the concept of the autoimmune nature of PsA disease is strengthened by the recent detection of autoantibodies against modified antigens in the peripheral blood and synovial tissue of early naive to treatment PsA patients [30].
Increased vascularity has been reported in both psoriatic skin lesions and synovial tissue in PsA. In the dermis of the psoriatic skin, an abundance of dilated and tortuous blood vessels is present [31]. Multiple authors have reported that PsA synovium is characterized by an increase in macroscopically tortuous blood vessels, and this is more pronounced in, but not exclusive to, PsA than it is in RA synovium [20, 32]. In our study cohort, CD31 IHC revealed that the mean number of synovial CD31+ vessels does not differ among PsA and Abneg RA, suggesting that the microscopical level does not mirror the macroscopical view of synovial vasculature in PsA compared to RA once disease phase stratification is done.
The development of novel biomarkers of therapeutic response prediction is urgently needed for PsA and RA management. Previous studies have investigated the effect of histological markers of synovitis in PsA and RA mirroring the therapeutic response to conventional and biological DMARDs respectively [33–35]. In particular, Pontifex et al. proposed IHC score for sublining CD3+ cells as a useful biomarker of treatment response in PsA, enrolling a PsA cohort treated with TNF inhibitor [36] without providing clear cut-off value to be used at treatment initiation in naive PsA patients. In our study cohorts, we found that naive to treatment PsA with high likelihood of MDA achievement after DMARD treatment was more likely characterized, at baseline, by IHC score for CD3+ cells < 1.25 compared to PsA patients not achieving this clinical outcome. Conversely, validation studies in RA patients, regardless of the autoimmune profile, found that variation in IHC score of sublining CD68+ cells is a valuable synovial biomarker mirroring the treatment response to conventional and biological DMARDs (rituximab and TNF inhibitor) [37–39]. Interestingly, in our study, we confirmed these findings in a well-selected naive to treatment Abneg RA patient cohort in which Abneg RA patients with high likelihood chance of DAS remission achievement after DMARD treatment were more likely characterized, at baseline, by IHC score for sublining CD68+ cells < 2.5 compared to Abneg RA patients not achieving DAS remission after DMARD treatment.
Conclusions
In conclusion, our comparative study assessing the histological features of synovial tissues obtained from naive to treatment and MTX-IR PsA and Abneg RA patients revealed that PsA synovitis is characterized by being mast cell (CD117+) rich but plasma cell (CD138+) poor whereas Abneg RA synovitis by the reverse findings, being plasma cell (CD38+) rich but mast cell (CD117+) poor. These different histopathologic biomarkers may help to solve the diagnostic overlapping issue in the setting of ACPA/RF negativity at the disease onset. Moreover, baseline IHC scores of CD3+ and sublining CD68+ arose as useful biomarkers of treatment response to first-line DMARDs in PsA and Abneg RA respectively, suggesting the need to include both parameters in more extensive future synovial tissue biopsy-driven clinical trials for these inflammatory joint conditions.
Additional files
Figure S1. (a-b) ROC curve analysis for cut-off values for CD3+ cells and SL CD68+ cells IHC in naive PsA and Abneg RA patients. SL sublining, PsA psoriatic arthritis, Ab autoantibody, RA rheumatoid arthritis, IHC immunohistochemistry. (TIF 12416 kb)
Table S1. Inter-rater agreement coefficients for CD68, CD21, CD20, CD3, CD117, CD138, and CD31 IHC scores. (DOCX 12 kb)
Acknowledgements
Not applicable.
Funding
The authors received no specific funding for this work.
Availability of data and materials
The datasets generated and/or analyzed for the present study are available from the corresponding author on reasonable request.
Abbreviations
- Abneg
Autoantibody negative
- ACPA
Anti-citrullinated peptide antibody
- CD
Cluster designation
- CRP
C-reactive protein
- DAPSA
Disease Activity in PSoriatic Arthritis
- DAS
Disease Activity Score
- ESR
Erythrocyte sedimentation rate
- IHC
Immunohistochemistry
- MDA
Minimal Disease Activity
- MTX-IR
Methotrexate inadequately responder
- PsA
Psoriatic arthritis
- RA
Rheumatoid arthritis
- REM
Remission
- RF
Rheumatoid factor
- SJC
Swollen joint count
- TJC
Tender joint count
Authors’ contributions
SA, GF, and EG gave substantial contributions to the study conception and design. SA, DB, BT, LB, LP, MRG, DoB, ALF, GP, and FF gave substantial contributions to the acquisition of data. SA, DB, BT, GF, and EG gave substantial contributions to the analysis and interpretation of data. SA, DB, BT, LB, LP, MRG, DoB, ALF, GP, FF, GF, and EG drafted the article and revised it critically for important intellectual content; SA, DB, BT, LB, LP, MRG, DoB, ALF, GP, FF, GF, and EG gave final approval of the version of the article to be published.
Ethics approval and consent to participate
This study was conducted in accordance with the Declaration of Helsinki and was approved by the Ethics Committee of the Università Cattolica del Sacro Cuore (Protocol number: 6334/15). Signed informed consent was obtained from each patient included before any activity of the study was started.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Stefano Alivernini, Email: stefano.alivernini@unicatt.it.
Dario Bruno, Email: dbdariobruno@gmail.com.
Barbara Tolusso, Email: barbara.tolusso@policlinicogemelli.it.
Laura Bui, Email: laura.bui@policlinicogemelli.it.
Luca Petricca, Email: luca.petricca@policlinicogemelli.it.
Maria Rita Gigante, Email: mariaritagigante@gmail.com.
Domenico Birra, Email: dombirra@gmail.com.
Anna Laura Fedele, Email: annalaura.fedele@policlinicogemelli.it.
Giusy Peluso, Email: giusy.peluso@policlinicogemelli.it.
Francesco Federico, Email: francesco.federico@unicatt.it.
Gianfranco Ferraccioli, Email: gianfranco.ferraccioli@unicatt.it.
Elisa Gremese, Email: elisa.gremese@unicatt.it.
References
- 1.Veale DJ, Fearon U. The pathogenesis of psoriatic arthritis. Lancet. 2018;391:2273–2284. doi: 10.1016/S0140-6736(18)30830-4. [DOI] [PubMed] [Google Scholar]
- 2.Shah K, Paris M, Mellars L, Changolkar A, Mease PJ. Real-world burden of comorbidities in US patients with psoriatic arthritis. RMD Open. 2017;3:e000588. doi: 10.1136/rmdopen-2017-000588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Najm A, Le Goff B, Orr C, Thurlings R, Cañete JD, Humby F, et al. Standardisation of synovial biopsy analyses in rheumatic diseases: a consensus of the EULAR Synovitis and OMERACT Synovial Tissue Biopsy Groups. Arthritis Res Ther. 2018;20:265. doi: 10.1186/s13075-018-1762-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kruithof E, Baeten D, De Rycke L, Vandooren B, Foell D, Roth J, et al. Synovial histopathology of psoriatic arthritis, both oligo- and polyarticular, resembles spondyloarthropathy more than it does rheumatoid arthritis. Arthritis Res Ther. 2005;7:R569–R568. doi: 10.1186/ar1698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Veale DJ, Fearon U. What makes psoriatic and rheumatoid arthritis so different? RMD Open. 2015;1:e000025. doi: 10.1136/rmdopen-2014-000025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Taylor W, Gladman D, Helliwell P, Marchesoni A, Mease P, Mielants H, et al. Classification criteria for psoriatic arthritis. Development of new criteria from a large international study. Arthritis Rheumatol. 2006;54:2665–2673. doi: 10.1002/art.21972. [DOI] [PubMed] [Google Scholar]
- 7.Aletaha D, Neogi T, Silman AJ, Funovits J, Felson DT, Bingham CO, III, et al. Rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Ann Rheum Dis. 2010;69:1580–1588. doi: 10.1136/ard.2010.138461. [DOI] [PubMed] [Google Scholar]
- 8.Coates LC, Fransen J, Helliwell PS. Defining minimal disease activity in psoriatic arthritis: a proposed objective target for treatment. Ann Rheum Dis. 2010;69:48–53. doi: 10.1136/ard.2008.102053. [DOI] [PubMed] [Google Scholar]
- 9.Felson DT, Smolen JS, Wells G, Zhang B, van Tuyl LH, Funovits J, et al. American College of Rheumatology/European League against Rheumatism provisional definition of remission in rheumatoid arthritis for clinical trials. Ann Rheum Dis. 2011;70:404–413. doi: 10.1136/ard.2011.149765. [DOI] [PubMed] [Google Scholar]
- 10.Gossec L, Smolen JS, Ramiro S, de Wit M, Cutolo M, Dougados M, et al. European League Against Rheumatism (EULAR) recommendations for the management of psoriatic arthritis with pharmacological therapies: 2015 update. Ann Rheum Dis. 2016;75:499–510. doi: 10.1136/annrheumdis-2015-208337. [DOI] [PubMed] [Google Scholar]
- 11.Smolen JS, Landewé R, Bijlsma J, Burmester G, Chatzidionysiou K, Dougados M, et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2016 update. Ann Rheum Dis. 2017;76:960–977. doi: 10.1136/annrheumdis-2016-210715. [DOI] [PubMed] [Google Scholar]
- 12.Alivernini S, Tolusso B, Petricca L, Bui L, Di Sante G, Peluso G, et al. Synovial features of rheumatoid arthritis and psoriatic arthritis patients in clinical and ultrasound remission differ under anti-TNF therapy. A clue to interpret different chances of relapse after clinical remission? Ann Rheum Dis. 2017;76:1228–1236. doi: 10.1136/annrheumdis-2016-210424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Orr C, Sousa E, Boyle DL, Buch MH, Buckley CD, Cañete JD, et al. Synovial tissue research: a state-of-the-art review. Nat Rev Rheumatol. 2017;13:463–475. doi: 10.1038/nrrheum.2017.115. [DOI] [PubMed] [Google Scholar]
- 14.Smolen JS, Aletaha D, McInnes IB. Rheumatoid arthritis. Lancet. 2016;388:2023–38. [DOI] [PubMed]
- 15.Fitzgerald O, Winchester R. Psoriatic arthritis: from pathogenesis to therapy. Arthritis Res Ther. 2009;11:214. [DOI] [PMC free article] [PubMed]
- 16.Wong K, Gladman DD, Husted J, Long JA, Farewell VT. Mortality studies in psoriatic arthritis: results from a single outpatient clinic. I. Causes and risk of death. Arthritis Rheumatol. 1997;40:1868–1872. doi: 10.1002/art.1780401021. [DOI] [PubMed] [Google Scholar]
- 17.Gladman DD. Mortality in psoriatic arthritis. Clin Exp Rheumatol. 2008;26:S62–S65. [PubMed] [Google Scholar]
- 18.Belasco J, Louie JS, Gulati N, Wei N, Nograles K, Fuentes-Duculan J, et al. Comparative genomic profiling of synovium versus skin lesions in psoriatic arthritis. Arthritis Rheumatol. 2015;67:934–944. doi: 10.1002/art.38995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Veale D, Yanni G, Rogers S, Barnes L, Bresnihan B, Fitzgerald O. Reduced synovial membrane macrophage numbers, ELAM-1 expression, and lining layer hyperplasia in psoriatic arthritis as compared with rheumatoid arthritis. Arthritis Rheumatol. 1993;36:893–900. doi: 10.1002/art.1780360705. [DOI] [PubMed] [Google Scholar]
- 20.Reece RJ, Canete JD, Parsons WJ, Emery P, Veale DJ. Distinct vascular patterns of early synovitis in psoriatic, reactive, and rheumatoid arthritis. Arthritis Rheumatol. 1999;42:1481–1484. doi: 10.1002/1529-0131(199907)42:7<1481::AID-ANR23>3.0.CO;2-E. [DOI] [PubMed] [Google Scholar]
- 21.Baeten D, Demetter P, Cuvelier C, Van Den BF, Kruithof E, Van DN, et al. Comparative study of the synovial histology in rheumatoid arthritis, spondyloarthropathy, and osteoarthritis: influence of disease duration and activity. Ann Rheum Dis. 2000;59:945–953. doi: 10.1136/ard.59.12.945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Moll C, Bogas M, Gómez-Puerta JA, Celis R, Vázquez I, Rodríguez F, et al. Macroscopic features of knee synovitis in early untreated Behçet disease and psoriatic arthritis. Clin Rheumatol. 2009;28:1053–1057. doi: 10.1007/s10067-009-1205-0. [DOI] [PubMed] [Google Scholar]
- 23.Alivernini S, Tolusso B, Petricca L, Bui L, Di Mario C, Gigante MR, et al. Synovial predictors of differentiation to definite arthritis in patients with seronegative undifferentiated peripheral inflammatory arthritis: microRNA signature, histological, and ultrasound features. Front Medicine. 2018;5:186. doi: 10.3389/fmed.2018.00186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.van Kuijk AW, Reinders-Blankert P, Smeets TJ, Dijkmans BA, Tak PP. Detailed analysis of the cell infiltrate and the expression of mediators of synovial inflammation and joint destruction in the synovium of patients with psoriatic arthritis: implications for treatment. Ann Rheum Dis. 2006;65:1551–1557. doi: 10.1136/ard.2005.050963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Danning CL, Illei GG, Hitchon C, Greer MR, Boumpas DT, McInnes IB. Macrophage-derived cytokine and nuclear factor kappaB p65 expression in synovial membrane and skin of patients with psoriatic arthritis. Arthritis Rheumatol. 2000;43:1244–1256. doi: 10.1002/1529-0131(200006)43:6<1244::AID-ANR7>3.0.CO;2-2. [DOI] [PubMed] [Google Scholar]
- 26.Salvador G, Sanmarti R, Garcia-Peiro A, Rodriguez-Cros JR, Munoz-Gomez J, Canete JD. p53 expression in rheumatoid and psoriatic arthritis synovial tissue and association with joint damage. Ann Rheum Dis. 2005;64:183–187. doi: 10.1136/ard.2004.024430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Noordenbos T, Yeremenko N, Gofita I, van de Sande M, Tak PP, Caňete JD, et al. Interleukin-17-positive mast cells contribute to synovial inflammation in spondylarthritis. Arthritis Rheumatol. 2012;64:99–109. doi: 10.1002/art.33396. [DOI] [PubMed] [Google Scholar]
- 28.Rivellese F, Mauro D, Nerviani A, Pagani S, Fossati-Jimack L, Messemaker T, et al. Mast cells in early rheumatoid arthritis associate with disease severity and support B cell autoantibody production. Ann Rheum Dis. 2018;77:1773–1781. doi: 10.1136/annrheumdis-2018-213418. [DOI] [PubMed] [Google Scholar]
- 29.Cañete JD, Santiago B, Cantaert T, Sanmartí R, Palacin A, Celis R, et al. Ectopic lymphoid neogenesis in psoriatic arthritis. Ann Rheum Dis. 2007;66:720–726. doi: 10.1136/ard.2006.062042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Frasca L, Palazzo R, Chimenti MS, Alivernini S, Tolusso B, Bui L, et al. Anti-LL37 antibodies are present in psoriatic arthritis (PsA) patients: new biomarkers in PsA. Front Immunol. 2018;12(9):1936. doi: 10.3389/fimmu.2018.01936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Braverman IM, Yen A. Ultrastructure of the capillary loops in the dermal papillae of psoriasis. J Invest Dermatol. 1977;68:53–60. doi: 10.1111/1523-1747.ep12485169. [DOI] [PubMed] [Google Scholar]
- 32.Canete JD, Rodriguez JR, Salvador G, Gomez-Centeno A, Munoz-Gomez J, Sanmarti R. Diagnostic usefulness of synovial vascular morphology in chronic arthritis. A systematic survey of 100 cases. Semin Arthritis Rheum. 2003;32:378–387. doi: 10.1053/sarh.2002.50004. [DOI] [PubMed] [Google Scholar]
- 33.Kane D, Gogarty M, O’Leary J, Silva I, Bermingham N, Bresnihan B, et al. Reduction of synovial sublining layer inflammation and proinflammatory cytokine expression in psoriatic arthritis treated with methotrexate. Arthritis Rheumatol. 2004;50:3286–3295. doi: 10.1002/art.20518. [DOI] [PubMed] [Google Scholar]
- 34.Goedkoop AY, Kraan MC, Teunissen MB, Picavet DI, de Rie MA, Bos JD, et al. Early effects of tumour necrosis factor alpha blockade on skin and synovial tissue in patients with active psoriasis and psoriatic arthritis. Ann Rheum Dis. 2004;63:769–773. doi: 10.1136/ard.2003.018085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Canete JD, Pablos JL, Sanmarti R, Mallofre C, Marsal S, Maymo J, et al. Antiangiogenic effects of anti-tumor necrosis factor alpha therapy with infliximab in psoriatic arthritis. Arthritis Rheum. 2004;50:1636–1641. doi: 10.1002/art.20181. [DOI] [PubMed] [Google Scholar]
- 36.Pontifex EK, Gerlag DM, Gogarty M, Vinkenoog M, Gibbs A, Burgman I, et al. Change in CD3 positive T-cell expression in psoriatic arthritis synovium correlates with change in DAS28 and magnetic resonance imaging synovitis scores following initiation of biologic therapy--a single centre, open-label study. Arthritis Res Ther. 2011;13:R7. doi: 10.1186/ar3228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Humby F, Kelly S, Hands R, Rocher V, DiCicco M, Ng N, et al. Use of ultrasound-guided small joint biopsy to evaluate the histopathologic response to rheumatoid arthritis therapy: recommendations for application to clinical trials. Arthritis Rheumatol. 2015;67:2601–2610. doi: 10.1002/art.39235. [DOI] [PubMed] [Google Scholar]
- 38.Bresnihan B, Pontifex E, Thurlings RM, Vinkenoog M, El-Gabalawy H, Fearon U, et al. Synovial tissue sublining CD68 expression is a biomarker of therapeutic response in rheumatoid arthritis clinical trials: consistency across centers. J Rheumatol. 2009;36:1800–1802. doi: 10.3899/jrheum.090348. [DOI] [PubMed] [Google Scholar]
- 39.Haringman JJ, Gerlag DM, Zwinderman AH, Smeets TJ, Kraan MC, Baeten D, et al. Synovial tissue macrophages: a sensitive biomarker for response to treatment in patients with rheumatoid arthritis. Ann Rheum Dis. 2005;64:834–838. doi: 10.1136/ard.2004.029751. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figure S1. (a-b) ROC curve analysis for cut-off values for CD3+ cells and SL CD68+ cells IHC in naive PsA and Abneg RA patients. SL sublining, PsA psoriatic arthritis, Ab autoantibody, RA rheumatoid arthritis, IHC immunohistochemistry. (TIF 12416 kb)
Table S1. Inter-rater agreement coefficients for CD68, CD21, CD20, CD3, CD117, CD138, and CD31 IHC scores. (DOCX 12 kb)
Data Availability Statement
The datasets generated and/or analyzed for the present study are available from the corresponding author on reasonable request.