Abstract
Cytomegalovirus is the most common congenital viral infection. Infection can cause developmental delay, sensorineural deafness and fetal death. Fetal damage is more severe when infection occurs in the first trimester of pregnancy. Prenatal ultrasound findings may be cerebral, such as ventriculomegaly, microcephaly and periventricular leukomalacia, as well as non-cerebral, such as echogenic bowel, ascites and pericardial effusion. We present a case of congenital cytomegalovirus infection in which the only ultrasound sign noted at routine second-trimester scan was low-grade echogenic bowel, a soft marker, which progressed to severe disease in the third trimester, when further investigation was prompted, leading to the diagnosis. Patients need to be counselled regarding the possible perinatal prognosis. Ultrasound markers can often but not always predict severity and, hence, counselling can be a challenge.
Conclusion: A meticulous anatomy survey in mid-trimester remains the norm and ultrasound soft markers should prompt comprehensive testing for viral infections in pregnancy.
Keywords: Congenital cytomegalovirus infection, IgG avidity, Fetal hydrops, Fetal pericardial effusion
Highlights
-
•
Fetal damage caused by infection with CMV is more severe when the infection occurs in the first trimester of pregnancy.
-
•
CMV is the most common congenital viral infection causingdevelopmental delay, sensorineural deafness and fetal death.
-
•
Ultrasound markers can often but not always predict the severity of congenital infection.
-
•
A meticulous anatomy survey in mid-trimester remains the norm.
-
•
Ultrasound soft markers should prompt comprehensive testing for viral infections in pregnancy.
1. Introduction
Cytomegalovirus (CMV) is a ubiquitous DNA herpesvirus [1]. Similar to other herpesviruses, it becomes latent after a primary infection but can reactivate with renewed viral shedding. Shedding can occur from multiple sites and for prolonged periods of time [2]. CMV is the most common congenital viral infection, with a birth prevalence of 0.48 to 1.3% in recent decades [3]. Congenital CMV is associated with developmental delay, sensorineural hearing loss (SNHL) and fetal death [4]. It is the leading infectious cause of hearing impairment in children, with 40–50% of infants born with symptomatic CMV infection and 7–15% of asymptomatic CMV-infected newborns developing SNHL in later years [5].
In the fetus, particularly, the virus replicates in the oropharynx and is then carried through the fetal circulation. The major target organs of CMV in the fetus are the brain, bone marrows, lungs, pancreas, kidneys and the liver [1].
Although the fetus can be affected by CMV throughout the whole pregnancy, the damage is more severe if infections occur during the first half of pregnancy [6,7]. The exact risk of symptomatic congenital CMV infection after maternal primary or secondary infection is unknown but it has been suggested to be as high as 15% after primary and 2% after secondary infection. [7]. Diagnosis can be difficult, as ultrasound features may not be evident until many weeks after fetal infection.
To this end, we present a case of congenital cytomegalovirus infection in pregnancy which was a diagnostic challenge in view of inconspicuous findings of a sole soft marker at the anatomy survey but in the third trimester, presenting with significant ultrasound features revealing a poor prognosis.
2. Case Report
A 26-year-old healthy primigravida woman was booked into hospital from early in the second trimester, having had early antenatal care with her local practitioner. Her booking BMI was 21.16 kg/m2. She was a non-smoker and denied alcohol and other substance abuse.
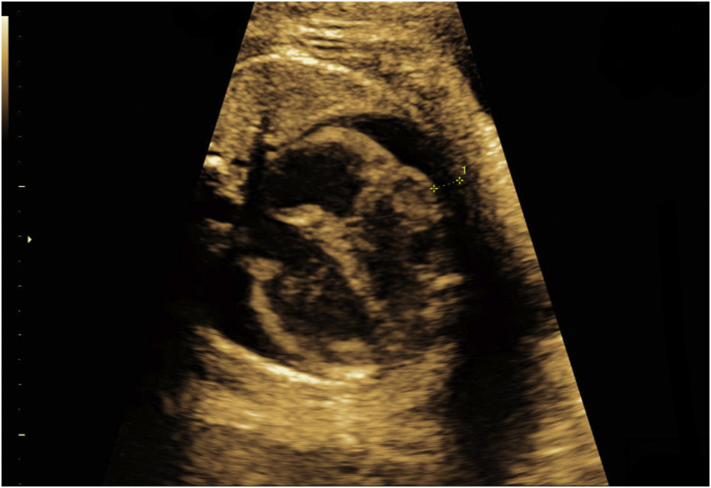
At her first trimester antenatal screen, she was noted to be Rhesus positive, negative for HIV, hepatitis B, hepatitis C and syphilis. She was also found to be Rubella immune. Her combined first-trimester screen was reported as low risk for trisomy 21, 18 and 13. She underwent a routine morphology scan at 19 weeks 5 days. The biparietal diameter (BPD) and head circumference (HC) were noted to be on the 5th centile, the abdominal circumference (AC) on the 57th centile with an estimated fetal weight (EFW) of 304 g, on the 40th centile. The nuchal fold thickness was 4.1 mm. A three-vessel cord was noted with normal amount of amniotic fluid for this gestation. The fetal anatomy appeared normal with the exception of low-grade echogenic bowel (Fig. 1). Fetal movements were visible. The placenta was posterior, not low lying and normal in appearance. At this visit, she was advised to have interval growth scans at 28 and 34 weeks.
Fig. 1.
Low-grade echogenic bowel noted at morphology scan (19w5d).
At 23 weeks 6 days on antenatal clinical exam, the uterine size appeared adequate for dates and the fetal heart with a normal rate was heard. A cystic fibrosis (CF) screen was negative. She underwent routine mid-trimester screening and was found to be anaemic, with a haemoglobin level of 95 g/dL, for which she was advised an iron infusion.
The planned third-trimester growth scan was performed at 30 weeks 6 days, and showed the BPD was on the 35th centile, HC on the 44th centile, AC > 95th centile, femur length(FL) on the 10th centile and the EFW was estimated at >95th centile(~2218 g). Several abnormalities were noted. The fetus had developed gross hydrops; gross ascites(Fig. 2), pericardial effusions of 6 mm on both sides of the fetal cardia (Fig. 3), ventriculomegaly of 12 mm was noted(Fig. 4) and the middle cerebral artery Doppler scans revealed a peak systolic velocity which was raised at >1.5 MoM (Fig. 5).
Fig. 2.
Fetal hydrops noted at growth scan (30wk6d). Gross fetal ascites in transverse view of abdomen.
Fig. 3.
Fetal hydrops noted at growth scan (30wk6d). Bilateral pericardial effusion in transverse view of the fetal chest.
Fig. 4.
Fetal hydrops noted at growth scan (30wk6d). Lateral ventricles of fetal brain measuring 12 mm showing ventriculomegaly.
Fig. 5.
Fetal hydrops noted at growth scan (30wk6d). Middle cerebral artery (MCA) Doppler showing high peak systolic velocity (PSV) measuring >1.5 MoM: 94.46 cm/s; Right MCA Systolic/diastolic (S/D): 3.99; Right MCA Pulsatility Index (PI): 1.38; Right MCA Resistance Index (RI): 0.75; Right MCA heart rate: 121 beats/min.
At 31 weeks of gestation, various possibilities were discussed with the patient, including aneuploidy, structural abnormalities, viral infection or a genetic syndrome. Urgent serum viral serology, and amniocentesis for fetal microarray was organized. While awaiting results, due to the possibility of severe fetal anaemia, cordocentesis and intrauterine fetal transfusion was performed at 31 weeks 1 day. Pre-transfusion full blood count revealed a haemoglobin of 79 g/dl, a haematocrit of 0.25% and a platelet count of 29.9 × 109/L. After 80 mL of intrauterine transfusion, the haemoglobin increased to 100 g/dl, the haematocrit to 0.30 and the platelet count was 30 × 109/L.
At 31 weeks 3 days, viral serology results revealed positive CMV IgG and IgM with high IgG avidity. Amniocentesis results showed normal fetal microarray, but with high CMV PCR. Based on this, the patient was explained that primary CMV infection is the most likely diagnosis. Given the presence of severe hydrops, ascites, pericardial effusion, pancytopenia with likely bone marrow involvement, as well as ventriculomegaly, poor perinatal prognosis was explained, including high chance of early neonatal death or significant long term neurological impact. The option of late termination was discussed; however, the patient wished for all reasonable measures for perinatal resuscitation to be taken. Use of CMV specific immunogloubulin and antivirals were also considered, but at 32 weeks of gestation the patient spontaneously ruptured membranes and went into spontaneous labour. In view of the fetus in breech presentation, the baby was delivered via lower segment caesarean section.
The neonatal intensive care unit (NICU) team performed active neonatal resuscitation and the neonate was admitted into the NICU for ongoing supportive care and management. On day 4 of life, a cranial ultrasound scan revealed severe periventricular leukomalacia, ventriculitis and diffuse calcifications. The poor long-term outlook was explained to the parents and the decision was taken to withdraw care.
3. Discussion
Our case highlights the challenges associated with diagnosing congenital CMV infection in pregnancy as well as managing a late diagnosis with poor prognosis. Prenatal ultrasound findings in CMV infected fetuses include cerebral abnormalities such as cerebral ventriculomegaly, occipital horn calcifications, periventricular leukomalacia and microcephaly, and non-cerebral multi-organ abnormalities such as echogenic bowel, ascitis, hepatomegaly and cardiomegaly caused by pericardial effusion such as seen in our case. [8,9] However, some features of congenital CMV disease such as neurodevelopmental defects, chorioretinitis and petechiae are not detectable by prenatal imaging; therefore, the absence of fetal abnormalities does not exclude fetal damage, and fetal death may also occur in those cases with almost no ultrasound features [10,11].
In our case, the only feature noted by ultrasound at the morphology scan was isolated low-grade echogenic bowel. Fetal echogenic bowel is an ultrasound soft marker and soft markers are considered a variant of the normal, distinct from fetal anatomical malformations. [12] It is a non-specific finding in routine second trimester ultrasound scans with an incidence of 0.2–1.8% [13]. An isolated finding before 20 weeks is usually transient in the vast majority but persistence into the third trimester would indicate serious underlying pathology [14]. The pathological causes for fetal echogenic bowel are fetal aneuploidy (being the most common cause ~4–25%) especially trisomy 21, duodenal/bowel atresia, oligohydramnios, Hirschsprung's disease, intrauterine growth restriction, intra-amniotc haemorrhage, cystic fibrosis and least commonly, congenital infections such as CMV, toxoplasmosis and parvovirus with an approximate incidence of 1–4% (CMV being the most common) [12]. In a study by Simon-Bouy et al., 682 cases of fetal echogenic bowel were examined, of which 65.5%(447/682) had no abnormality found and 2.8% were due to congenital infection [15].
Therefore, what practical steps are necessary in the second trimester for a fetus with hyperechogenic bowel, to avoid a late diagnosis of possible pathology? A detailed parental history is clearly essential because of the links with karyotype anomalies, intrauterine infection and CF. The sonographic fetal survey must be detailed and complete to exclude associated structural anomalies and features such as intestinal dilatation and fetal ascites [12]. Odibo et al. suggested that more invasive investigations such as fetal karyotyping are probably justified as upto 25% of fetuses can have an abnormal karyotype.[16]However, in the more than likely scenario, when parents may opt to wait and watch, maternal serum serology screen for congenital infection(Toxoplasmosis, CMV and parvovirus IgG and IgM) should certainly be offered and a plan for serial ultrasound assessments every fortnight may detect resolution of the hyperechogenicity or persistence, hence warranting further investigation [16]. O'Sullivan describes a case in which the only finding at the routine 20 weeks anomaly scan was isolated echogenic bowel. At this time, testing for cytomegalovirus, parvovirus, fetal aneuploidy and cystic fibrosis screen was offered. CMV IgG avidity was noted to be low and the possibility of congenital CMV infection was suspected. At 22 weeks, a repeat ultrasound still revealed only isolated echogenic bowel but only 5 days later, extensive cerebral abnormalities were noted revealing a poor prognosis [17].
The gold standard to identify primary infection in a pregnant woman currently is Anti CMV IgG avidity [18]. Studies conducted over the last 20 years convincingly demonstrate that measurement of CMV IgG avidity is both a sensitive and a specific method for identifying pregnant women with recent primary CMV infection and thus, at increased risk for vertical CMV transmission [19]. IgG avidity is defined as the strength with which IgG binds to antigenic epitopes expressed by a given protein; it matures gradually during the 6 months following primary infection. Low CMV IgG avidity is an accurate indicator of primary infection within the preceding 3 to 4 months, whereas high avidity excludes primary infection within the preceding 3 months [20]. In this regard, it is likely that in our case, with the finding of high IgG avidity at 30 weeks of gestation, primary infection occurred in the first trimester.
Predicting fetal prognosis and symptomatic neonatal infection is one of the most important factors in counselling parents. Clinical presentation of neonatal CMV infection varies widely between asymptomatic, intermediate forms and severely symptomatic newborns [21].
Pass et al. concluded that the severity of fetal/neonatal disease is greater when infection occurs in the first trimester as well as that such fetuses show a greater predisposition towards adverse neurological ultrasound findings [22], despite the fact that vertical transmission is higher when maternal infection occurs in the second and third trimesters [23]. Liesnard et al. reached similar conclusions by studying 55 cases of congenital CMV infection (testing Amniotic fluid) from 237 pregnancies undergoing prenatal evaluation and found that 10/38 (26%) cases infected before 20 weeks of gestation had severe disease compared with only 1/16 (6.2%) infected after 20 weeks [24].
Unfortunately, investigating further when maternal CMV infection is diagnosed can always pose to be a dilemma, as several weeks can elapse before symptomatic fetal infection occurs and abnormal ultrasound features are revealed, especially neurological abnormalities on imaging, sometimes as late as the third trimester [22]. The timing of amniocentesis is crucial and is recommended between 21 and 22 weeks of gestation. CMV replicates slowly and can take 6–9 weeks before it is excreted in the fetal urine in large enough amounts to be detected in the amniotic fluid [18,25]. If an invasive procedure is conducted too early, a false negative result could occur [25].
In addition, La torre et al. noted that the placentas of fetuses with CMV disease were significantly thicker than those free from disease and hence, suggested that CMV infection causes extensive placental inflammation [26]. This may be another clue to the diagnosis but was absent in our case.
In a study by Leruez-Ville et al., the positive predictive value was 79% and negative predictive value was 100% for severe disease when the combination of ultrasound findings such as ventriculomegaly and fetal blood parameters, i.e. thrombocytopenia, as well as amniotic fluid viral load (>100,000 copies/ml) were considered [27]. Similar findings were noted in a study by Benoist et al. in which the finding of any ultrasound abnormality and fetal thrombocytopenia remained significant independent predictors of poor outcome. It is interesting to note that in this particular study, the most common ultrasound abnormality noted was hyperechogenic bowel (26%), although not in isolation [28].
In our case, the parents wished for all resuscitative measures to be taken, despite the dismal picture through ultrasound imaging. As the diagnosis was still unknown and with laboratory investigations pending, in view of suspicion for fetal anaemia (possibly parvovirus infection), a decision for fetal transfusion was made. It is essential, that when fetal transfusion for red cells is considered and being undertaken, as was carried out in our case, it may prove beneficial to have pooled platelets, as standby, in the scenario that concurrent fetal thrombocytopenia is noted (as noted in our case).
In view of the essentiality of cerebral ultrasound findings in predicting neonatal outcome, the development of fetal MRI has become an asset in the assessment of infected fetuses. In a study by Picone et al., 46% of cases with only extracerebral abnormalities on ultrasound were found to have cerebral abnormalities on MRI. In cases with cerebral features seen on ultrasound, MRI maybe used to confirm these and to detect further abnormalities [29]. In our case described, in view of the severity of the obvious cerebral features on ultrasound, the multi-organ involvement as well as fetal thrombocytopenia, the prognosis was determined to be poor and the need for fetal MRI was deferred.
To this end, the thought arises as to how an earlier diagnosis of maternal infection in second trimester may have changed the clinical course and management. As stated above, the time lapse between maternal infection and fetal infection, if it at all occurs, may be several weeks and the correlation between severity of fetal infection and ultrasound findings is not well established. It can only be assumed that in the presence of only a soft marker at the anatomy survey, most patients may decline invasive testing, leaving the management to serial ultrasound scans until further abnormal features become apparent. In our case, whether these features would have appeared a few days after the routine second trimester ultrasound or possibly only a few weeks before the third trimester ultrasound remains a retrospective speculation.
4. Conclusion
Our case highlights the importance of meticulous anatomy survey at mid-trimester. Clinicians should be aware of potential, even subtle, ultrasound signs of CMV infection including small biometry, ventriculomegaly, echogenic bowel including low-grade findings and features of fetal hydrops. Presence of these findings should lead to prompt investigation which includes comprehensive testing for viral infections including CMV.
Contributors
Caroline Ruth Mathias conceptualized the case report, acquired data, contributed to the literature review, and drafted the case report.
Steven Jin Sung Joung conceptualized the case report, contributed to the literature review, and revised the draft manuscript.
Conflict of Interest
The authors declare that they have no conflict of interest regarding the publication of this case report.
Funding
No external or internal funding was sought or secured in relation to this case report.
Patient Consent
Informed consent was obtained from the subject in this case report.
Provenance and Peer Review
This case report was peer reviewed.
Contributor Information
Caroline Ruth Mathias, Email: caroline.r.mathias@gmail.com.
Steven Jin Sung Joung, Email: steven.joung@health.nsw.gov.au.
References
- 1.Gaytant M.A., Steegers E.A., Semmekrot B.A., Merkus H.M., Galama J.M. Congenital cytomegalovirus infection: review of the epidemiology and outcome. Obstet. Gynecol. Surv. 2002;57:245–256. doi: 10.1097/00006254-200204000-00024. (Level III) [DOI] [PubMed] [Google Scholar]
- 2.Alford C.A., Stagno S., Pass R.F., Britt W.J. Congenital and perinatal cytomegalovirus infections. Rev. Infect. Dis. 1990;12(Suppl. 7):S745–S753. doi: 10.1093/clinids/12.supplement_7.s745. [DOI] [PubMed] [Google Scholar]
- 3.Kenneson A., Cannon M.J. Review and meta-analysis of the epidemiology of congenital cytomegalovirus (CMV) infection. Rev. Med. Virol. 2007;17:253. doi: 10.1002/rmv.535. [DOI] [PubMed] [Google Scholar]
- 4.McMullan B., Palasanthiran P., Jones C. Congenital cytomegalovirus - time to diagnosis, management and clinical sequelae in Australia: opportunities for earlier identification. Med. J. Aust. 2011;194:625–629. doi: 10.5694/j.1326-5377.2011.tb03144.x. [DOI] [PubMed] [Google Scholar]
- 5.Dahle A., Fowler K., Wright J. Longitudinal investigation of hearing disorders in children with congenital cytomegalovirus. J. Am. Acad. Audiol. 2000;11:283–290. [PubMed] [Google Scholar]
- 6.Benshushan A., Brezinski A., Ben-David A., Nadjari M. Early recurrent CMV infection with severe outcome to the fetus. Acta Obstet. Gynecol. Scand. 1998;77:694–695. doi: 10.1034/j.1600-0412.1998.770620.x. [DOI] [PubMed] [Google Scholar]
- 7.Ornoy A. The effects of cytomegalovirus (CMV) infection during pregnancy on the developing human fetus. Harefuah. 2002;141:565–577. [PubMed] [Google Scholar]
- 8.Enders G., Bäder U., Lindemann L. Prenatal diagnosis of congenital cytomegalovirus infection in 189 pregnancies with known outcome. Prenat. Diagn. 2001;21:362–377. doi: 10.1002/pd.59. [DOI] [PubMed] [Google Scholar]
- 9.Twickler D., Perlman J., Maberry M. Congenital cytomegalovirus infection presenting as cerebral ventriculomegaly on antenatal sonography. Am. J. Perinatol. 1993;10:404–406. doi: 10.1055/s-2007-994773. [DOI] [PubMed] [Google Scholar]
- 10.Howard J., Hall B., Brennan L.E. Utility of newborn screening cards for detecting CMV infection in cases of stillbirth. J. Clin. Virol. 2009;44:215–218. doi: 10.1016/j.jcv.2008.12.013. [DOI] [PubMed] [Google Scholar]
- 11.Iwasenko J., Howard J., Arbuckle S. Human cytomegalovirus infection is detected frequently in stillbirths and is associated with fetal thrombotic vasculopathy. J. Infect. Dis. 2011;203:1526–1533. doi: 10.1093/infdis/jir121. [DOI] [PubMed] [Google Scholar]
- 12.De Oronzo Maria Antonietta. Hyperechogenic fetal bowel: an ultrasonography marker for adverse fetal and neonatal outcome? J. Prenat. Med. 2011 Jan-Mar;5(1):9–13. [PMC free article] [PubMed] [Google Scholar]
- 13.Bronshstein M., Balzer S. The gastrointestinal tract and abdominal wall. In: Callen P.W., editor. Ultrasonography in Obstetrics and Gynaecology. 5th Ed. Saunders Elsevier; Philadelphia, Pennsylvania: 2008. pp. 587–639. [Google Scholar]
- 14.Dicke J.M., Crane J.P. Sonographically detected hyperechoic fetal bowel: significance and implications for pregnancy management. Obstet. Gynecol. 1992;80:778–821. [PubMed] [Google Scholar]
- 15.Simon-Bouy B., Satre V. Hyperechogenic fetal bowel: a large French collaborative study of 682 cases. Am. J. Med. Genet. A. 2003 Sep 1;121A(3):209–213. doi: 10.1002/ajmg.a.20168. [DOI] [PubMed] [Google Scholar]
- 16.Odibo A.O., Katherine R. Goetzinger. Isolated echogenic bowel diagnosed on second-trimester ultrasound. Contemp. Ob/Gyn. 2011 Aug;56(8):24–28. [Google Scholar]
- 17.O'Sullivan Cynthia, Arulkumaran Shankari, Lakasing Lorin, Jauniaux Eric, Murphy Karl. Sequence and timing of intracranial changes in cytomegalovirus in pregnancy: a case report and literature review. Case Rep. Obstet. Gynaecol. 2017 doi: 10.1155/2017/5928398. (5 pages) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lazzarotto T., Guerra B., Lanari M., Gabrielli L., Landini M.P. New advances in the diagnosis of congenital cytomegalovirus infection. J. Clin. Virol. 2008;41(3):192–197. doi: 10.1016/j.jcv.2007.10.015. [DOI] [PubMed] [Google Scholar]
- 19.Manicklal S., Emery V.C., Lazzarotto T., Boppana S.B., Gupta R.K. The “silent” global burden of congenital cytomegalovirus. Clin. Microbiol. Rev. 2013;26:86–102. doi: 10.1128/CMR.00062-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Revello M.G., Gerna G. Diagnosis and management of human cytomegalovirus infection in the mother, fetus, and newborn infant. Clin. Microbiol. Rev. 2002;15:680–715. doi: 10.1128/CMR.15.4.680-715.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Benoist G., Leruez-Ville M., Magny J.F., Jacquemard F., Salomon L.J., Ville Y. Management of pregnancies with confirmed cytomegalovirus fetal infection. Fetal Diagn. Ther. 2013;33:203–214. doi: 10.1159/000342752. [DOI] [PubMed] [Google Scholar]
- 22.Pass R.F., Fowler K.B., Boppana S.B., Britt W.J., Stagno S. Congenital cytomegalovirus infection following first trimester maternal infection: symptoms at birth and outcome. J. Clin. Virol. 2006;35(2):216–220. doi: 10.1016/j.jcv.2005.09.015. [DOI] [PubMed] [Google Scholar]
- 23.Gindes L., Teperberg-Oikawa M., Sherman D., Pardo J., Rahav G. Congenital cytomegalovirus infection following primary maternal infection in the third trimester. BJOG. 2008;115:830–835. doi: 10.1111/j.1471-0528.2007.01651.x. [DOI] [PubMed] [Google Scholar]
- 24.Liesnard C., Donner C., Brancart F., Gosselin F., Delforge M.L., Rodesch F. Prenatal diagnosis of congenital cytomegalovirus infection: prospective study of 237 pregnancies at risk. Obstet. Gynecol. 2000;95:881–888. doi: 10.1016/s0029-7844(99)00657-2. [DOI] [PubMed] [Google Scholar]
- 25.Guerra B., Simonazzi G., Puccetti C. Ultrasound prediction of symptomatic congenital cytomegalovirus infection. Am. J. Obstet. Gynecol. 2008;198(4):380.e1–380.e7. doi: 10.1016/j.ajog.2007.09.052. [DOI] [PubMed] [Google Scholar]
- 26.La Torre R., Nigro G., Mazzocco M. Placental enlargement in women with primary maternal cytomegalovirus infection is associated with fetal and neonatal disease. Clin. Infect. Dis. 2006;43:994–1000. doi: 10.1086/507634. [DOI] [PubMed] [Google Scholar]
- 27.Leruez-Ville M., Stirnemann J., Sellier Y. Feasibility of predicting the outcome of fetal infection with cytomegalovirus at the time of prenatal diagnosis. Am. J. Obstet. Gynecol. 2016;215:342.e1. doi: 10.1016/j.ajog.2016.03.052. [DOI] [PubMed] [Google Scholar]
- 28.Benoist G., Salomon L.J., Jacquemard F., Daffos F., Ville Y. The prognostic value of ultrasound abnormalities and biological parameters in blood of fetuses infected with cytomegalovirus. BJOG. 2008;115:823–829. doi: 10.1111/j.1471-0528.2008.01714.x. [DOI] [PubMed] [Google Scholar]
- 29.Picone O., Simon I. Comparison between ultrasound and magnetic resonance imaging in assessment of fetal cytomegalovirus infection. Prenat. Diagn. 2008 Aug;28(8):753–758. doi: 10.1002/pd.2037. [DOI] [PubMed] [Google Scholar]