Abstract
Background
This post-marketing surveillance (PMS) study evaluated the safety and effectiveness of long-term darbepoetin alfa (darbepoetin) for the treatment of renal anemia in Japanese non-dialysis chronic kidney disease patients.
Methods
Patients were treated with darbepoetin and followed up for 3 years. Adverse events (AEs), adverse drug reactions (ADRs), hemoglobin (Hb) levels, and renal function were assessed. Patients were stratified by Hb level at the time of occurrence of cardiovascular-related AEs. Statistical analyses were performed to explore factors affecting the occurrence of AEs, cardiovascular-related AEs, and composite renal endpoints.
Results
In the safety analysis set (5547 patients), AEs and ADRs occurred in 44.4 and 7.1% of patients, respectively. Cardiovascular-related AEs were observed in 12.6% of the overall population. The proportion of patients who presented cardiovascular-related AEs was lower among those with a higher Hb level at the time of occurrence. In the effectiveness analysis set (5024 patients), mean Hb levels remained between 10.0 and 10.6 g/dL (Weeks 4–156). Three months after darbepoetin administration, patients with Hb ≥ 11 g/dL presented fewer composite renal endpoints than those with Hb < 11 g/dL (p = 0.0013), and the cumulative proportion of renal survival was higher in those with Hb ≥ 11 g/dL vs. Hb < 11 g/dL (p < 0.0001).
Conclusions
This PMS study showed the safety and effectiveness of long-term use of darbepoetin in a large number of patients. Patients with Hb ≥ 11 g/dL presented fewer composite renal endpoints than those with Hb < 11 g/dL, without an increase in the incidence of cardiovascular-related AEs.
Electronic supplementary material
The online version of this article (10.1007/s10157-018-1632-9) contains supplementary material, which is available to authorized users.
Keywords: Non-dialysis chronic kidney disease, Darbepoetin alfa, Post-marketing surveillance, Cardiovascular-related adverse events, Composite renal endpoints
Introduction
Renal anemia is a common complication of chronic kidney disease (CKD). The majority of patients with CKD stage 5 suffer from anemia [1]. Anemia leads to a decrease in oxygen delivery to vital organs, which is initially compensated by increased cardiac output, but may eventually result in maladaptive left ventricular hypertrophy, a well-recognized risk factor for cardiovascular (CV) disease and all-cause mortality [2, 3]. CKD patients with anemia were reported to have about twofold higher risk of CV disease than those without anemia [2, 3]. It is also suggested that progression of renal anemia not only increases the risk of CV disease, but also is an independent risk factor for the deterioration of renal function, causing the vicious cycle known as cardio-renal anemia syndrome [4].
The primary cause of renal anemia is the deficiency of endogenous erythropoietin, which is mainly produced by the kidneys [1]. Clinical studies have shown that the correction of renal anemia with erythropoiesis-stimulating agents (ESAs) might retard the progression of CKD [5, 6] and decrease left ventricular hypertrophy [7], but this is not a consistent finding as summarized in latter meta-analyses [8–12]. Moreover, lines of evidence from large-scale clinical studies in Europe and the US, which include CREATE [13], CHOIR [14] and TREAT [15], raised questions about correcting almost normal hemoglobin (Hb) levels with ESAs. Meta-analyses including these clinical studies claimed that targeting higher Hb levels in CKD increases CV risk and probably increases the risk of end-stage renal disease and death [16, 17]. However, the majority of patients recruited in these studies had diabetes or a high risk of CV disease; thus, the ideal target Hb level in the more general population of non-dialysis CKD patients, especially in Japanese patients, remains to be determined [18].
In Japan, darbepoetin alfa (darbepoetin), a long-acting ESA, became available for the treatment of renal anemia in non-dialysis CKD patients in 2010. Clinical trials of darbepoetin showed greater benefits in terms of quality of life and preserving cardiac and renal functions in Japanese non-dialysis CKD patients [19–21]. Later, a post-marketing surveillance study (Darbepoetin Alfa for Renal Anemia Management in Japan: DREAM-J, UMIN identifier: UMIN000017252) was conducted to evaluate the safety and effectiveness of long-term use of darbepoetin for the treatment of renal anemia in Japanese non-dialysis CKD patients. In addition, based on the results of clinical trials, this post-marketing surveillance study prospectively aimed at exploring the factors associated with renal and CV outcomes.
Here, we report on the adverse events (AEs) and adverse drug reactions (ADRs) of patients treated with darbepoetin in the DREAM-J study, which is thought to reflect the Japanese real-world clinical setting. Finally, we describe the relationship between treatment with darbepoetin and renal or CV outcomes in Japanese non-dialysis CKD patients.
Materials and methods
This was a multicenter, prospective, observational post-marketing surveillance study. The protocol of the current study was approved by the Pharmaceuticals and Medical Devices Agency (PMDA). This post-marketing surveillance was part of the mandatory actions determined and reviewed by the PMDA. The study was conducted in accordance with the Japanese regulation (Ministry of Health, Labour and Welfare Ministerial Ordinance No 171) for Good Post-Marketing Study Practice (GPSP).
Patients
Participants in the study were non-dialysis CKD patients with renal anemia who were treatment naïve for darbepoetin. Patients with planned renal replacement therapy (for dialysis or kidney transplantation) within 6 months were excluded. The target sample size was set at 3000 patients to detect at least one patient with unknown ADR occurring at a frequency of 0.1%, with a probability of ≥ 95%.
Method
Using a central registration system, patients were registered between August 2010 and December 2011, and the survey period was from August 2010 to June 2015. The observation period continued for 3 years post-initial darbepoetin administration, and survey sheets were collected four times, at 6 months and 1, 2, and 3 years after initiation of treatment.
Discontinuation/dropout criteria included the following: discontinuation of drug administration at the physician’s discretion (excluding temporary cessation of drug administration), change to renal replacement therapy, death, and loss to follow-up due to transfer to another hospital.
Administration of darbepoetin was made according to the approved dosage and at the discretion of the treating physician at medical institutions participating in this study. Survey items were as follows: AEs including abnormal laboratory values, ADRs (i.e., AEs for which a causal relationship with darbepoetin could not be ruled out), status of darbepoetin administration (date and dosage), Hb levels, blood pressure, serum creatinine levels, urine protein (spot urine), urine creatinine, and need of transfusion. AEs and ADRs were recorded according to the International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use Medical Dictionary for Regulatory Activities/Japanese version (MedDRA/J version 18.1). A CV-related AE was defined to include myocardial infarction, congestive heart failure, cerebrovascular disorders, aortic dissection, angina pectoris, chronic arteriosclerosis obliterans, and arrhythmia.
Safety evaluation involved the analysis of the occurrence of AEs and ADRs by patient characteristics. Hb levels at the time of occurrence of CV-related AEs and factors that affect the occurrence of CV-related AEs were explored.
Effectiveness evaluation included the investigation of changes in Hb levels and estimated glomerular filtration rates (eGFR) after darbepoetin administration, changes in dosage and frequency of darbepoetin administration at each point of evaluation, and factors that affect composite renal endpoints (50% reduction in eGFR, initiation of dialysis, or kidney transplantation). Patients without composite renal endpoints were defined as renal survivors.
Statistical analysis
Proportions of patients in the safety analysis set with AEs and ADRs were analyzed by patient characteristics using Fisher’s exact test or chi-square tests. Tests were performed at a two-sided significance level of 0.05. Independent associations of potential factors with the occurrence of CV-related AEs and composite renal endpoints were assessed by Cox proportional hazards regression analysis.
The cumulative proportions of renal survival, classified by achievement or non-achievement of target Hb levels, were presented as Kaplan–Meier survival curves. For inter-group comparison, proportional hazard assumption was confirmed in each group. The log-rank test was used when the proportional hazard assumption was met (p < 0.05), and the generalized Wilcoxon test was used when the assumption was not met.
For multivariate Cox regression analysis using CV-related AEs as objective variables, the following variables were adjusted for potential confounders: sex, age, body mass index (BMI), history of CV disease, hypertension, hyperlipidemia, diabetes mellitus (comorbidity), previous treatment with recombinant human erythropoietin (rHuEPO), transfusion, baseline systolic blood pressure, baseline diastolic blood pressure, baseline eGFR, increased Hb level during 4 weeks of darbepoetin administration, continuous quantity of baseline urine protein, dose of darbepoetin administered per week during the study, and use of antihypertensive agents, antithrombotic agents, and lipid-lowering agents at baseline. The Cox regression analysis for CV-related AEs was performed using the safety analysis set, regardless of the darbepoetin administration period.
For multivariate Cox regression analysis using composite renal endpoints as objective variables, the following variables were selected as explanatory variables: Hb level 3 months after the start of darbepoetin administration, sex, age, BMI, previous medical history, diabetes (comorbidity), previous treatment with rHuEPO, transfusion, baseline systolic blood pressure, baseline eGFR, increased Hb level during 4 weeks of darbepoetin administration, continuous quantity of baseline urine protein, dose of darbepoetin administered per week during this study, and use of antihypertensive agents, antithrombotic agents, and lipid-lowering agents at baseline. Cox regression analysis of composite renal endpoints, in relation to Hb levels and eGFR, was performed in patients in the effectiveness analysis set who received darbepoetin for > 3 months and who did not develop any renal endpoints within 3 months.
All statistical analyses were conducted by the contract research organization Asklep Inc. (Tokyo, Japan) using SAS software, version 9.3 (SAS Institute, Inc., Cary, NC, USA).
Results
Patient characteristics
Between August 2010 and December 2011, 5772 patients from 966 sites were initially registered, of whom 5607 were eligible for final registration. Survey sheets were collected from 5594 patients 6 months after the start of darbepoetin administration, from 4258 patients at 1 year, 3109 patients at 2 years, and 1877 patients at 3 years. The mean observation period was 1.8 years.
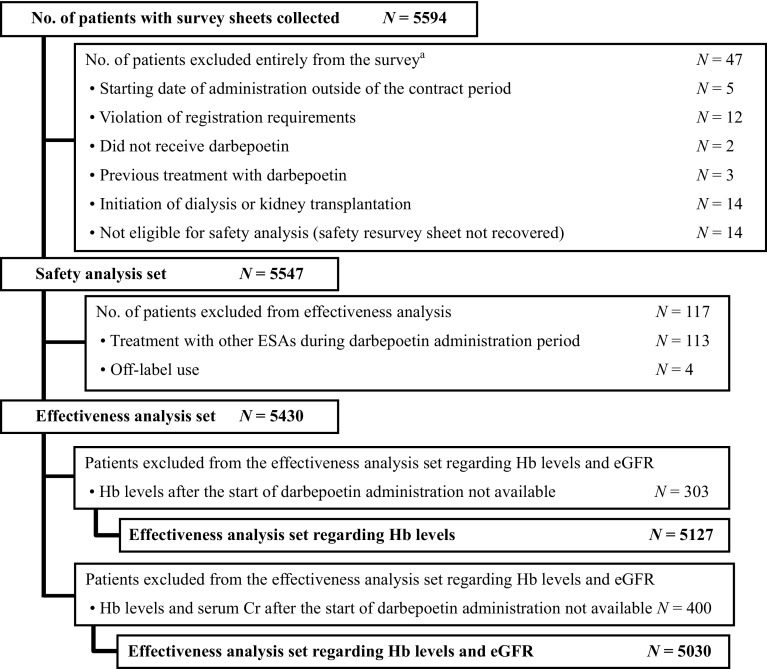
Figure 1 shows the patient disposition. A total of 47 patients were excluded from the survey, including those who initiated dialysis or underwent kidney transplantation before the start of darbepoetin administration. The remaining 5547 patients were included in the safety analysis set. Of these, the 3-year observation period was discontinued for 4309 patients; principal reasons included the initiation of dialysis (N = 1749) and transfer to another hospital (N = 1032). Of the 5547 patients in the safety analysis set, 117 patients were excluded, and thus 5430 patients were included in the main effectiveness analysis set. Patients in the effectiveness analysis with available Hb values after darbepoetin administration were further defined as the effectiveness analysis set regarding Hb levels (N = 5127). Those whose Hb values and serum creatinine values were available after darbepoetin administration were defined as the effectiveness analysis set regarding Hb levels and eGFR (N = 5030). Of the 5030 patients in the effectiveness analysis set regarding Hb levels and eGFR, these parameters were available in 5024 patients at the same evaluation point.
Fig. 1.
Patient disposition. aIf patients had more than one exclusion criterion, the patients were counted for each respective reason. Cr creatinine, eGFR estimated glomerular filtration rate, ESAs erythropoietin-stimulating agents, Hb hemoglobin
Patient characteristics of the safety analysis set (N = 5547) are presented in Table 1. Of these patients, 77.7% were elderly, 41.6% had CV disease, 5.9% had malignant tumors, and 40.5% were switched from rHuEPO. The baseline Hb level (mean ± standard deviation [SD]) was 9.5 ± 1.1 g/dL, and the baseline eGFR (mean ± SD) was 18.7 ± 10.3 mL/min/1.73 m2.
Table 1.
Patient characteristics
| Characteristics | Safety analysis set N = 5547 |
|---|---|
| Sex (male) | 3053 (55.0) |
| Age | 73 ± 12 |
| ≥ 65 years | 4308 (77.7) |
| BMI | 22.7 ± 3.8 |
| ≥ 25 kg/m2 | 1014 (18.3) |
| Underlying disease | |
| Diabetic nephropathy | 1818 (32.8) |
| Chronic glomerulonephritis | 1182 (21.3) |
| Nephrosclerosis | 1623 (29.3) |
| Other | 924 (16.7) |
| Medical history | |
| CV disease | 735 (13.3) |
| Malignant tumors | 412 (7.4) |
| Comorbidity | |
| CV disease | 2306 (41.6) |
| Malignant tumors | 330 (5.9) |
| Previous treatment with rHuEPO | 2244 (40.5) |
| Systolic blood pressure (mmHg) | 134 ± 20 |
| Hb level (g/dL) | 9.5 ± 1.1 |
| eGFR (mL/min/1.73 m2) | 18.7 ± 10.3 |
| Serum Cr (mg/dL) | 3.1 ± 1.6 |
Data are shown as n (%) or mean ± SD
BMI body mass index, Cr creatinine, CV cardiovascular, eGFR estimated glomerular filtration rate, Hb hemoglobin, rHuEPO recombinant human erythropoietin, SD standard deviation
Safety
In the safety analysis set, a total of 5735 AEs occurred in 2462 patients (44.4%), and 605 ADRs occurred in 394 patients (7.1%, Table 2). Major ADRs were hypertension (0.7%) and blood pressure increased (0.6%). Major ADRs that could not be specified in the “Precautions for Use” section of the product package (4th edition revised on December 2014) were heart failure, death, sudden death, congestive heart failure, pneumonia, and gastric cancer.
Table 2.
Occurrence of adverse events and adverse drug reactions
| DREAM-J N (%) | Phase 1–3 studies on non-dialysis CKD patients in JapanaN (%) | ||||
|---|---|---|---|---|---|
| Safety analysis set | 5547 | 439 | |||
| No. of AEs | 5735 | ||||
| No. of patients with AEs | 2462 (44.4) | ||||
| No. of ADRs | 605 | 205 | |||
| No. of patients with ADRs | 394 (7.1) | 135 (30.75) | |||
| Major ADRs | |||||
| Cardiac disorders | 66 (1.2) | 6 (1.36) | |||
| Cardiac failure | 22 (0.4) | – | |||
| Congestive heart failure | 10 (0.2) | – | |||
| Angina pectoris | 8 (0.1) | 1 (0.22) | |||
| Nervous system disorders | 49 (0.9) | 16 (3.64) | |||
| Cerebral infarction | 20 (0.4) | 2 (0.45) | |||
| Vascular disorders | 49 (0.9) | 29 (6.60) | |||
| Hypertension | 40 (0.7) | 27 (6.15) | |||
| Benign and malignant neoplasms | 37 (0.7) | – | |||
| Gastric cancer | 7 (0.1) | – | |||
| Colon cancer | 6 (0.1) | – | |||
| Lung neoplasm malignant | 6 (0.1) | – | |||
| Blood and lymphatic system disorders | 30 (0.5) | 3 (0.68) | |||
| Iron deficiency anemia | 23 (0.4) | – | |||
| Renal and urinary disorders | 25 (0.5) | 8 (1.82) | |||
| General disorders and others | 44 (0.8) | 7 (1.59) | |||
| Death | 12 (0.2) | – | |||
| Sudden death | 10 (0.2) | – | |||
| Skin and subcutaneous tissue disorders | 31 (0.6) | 8 (1.82) | |||
| Investigationsb | 68 (1.2) | 76 (17.31) | |||
| Blood pressure increased | 36 (0.6) | 51 (11.61) | |||
ADRs are classified according to MedDRA 18.1
ADR adverse drug reaction, AE adverse event, CKD chronic kidney disease
aQuoted from an interview form in Japan [http://www.info.pmda.go.jp/go/interview/1/230124_3999425S5028_1_006_1F]
bInvestigations include laboratory abnormalities such as blood pressure increased
Table 3 shows the occurrence of AEs and ADRs stratified by patient characteristics. CV-related AEs and malignant tumor-related AEs occurred in 12.6 and 4.1% of patients, respectively. CV-related ADRs and malignant tumor-related ADRs occurred in 2.0 and 0.6%, respectively.
Table 3.
Adverse events and adverse drug reactions stratified by patient characteristics
| Factors | No. of patients | All events (%) | CV-related events (%) | Malignant tumors (%) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AEs | ADRs | AEs | ADRs | AEs | ADRs | |||||||||
| Total | 5547 | 2462 (44.4) | 394 (7.1) | 697 (12.6) | 109 (2.0) | 228 (4.1) | 35 (0.6) | |||||||
| Sex | Male | 3053 | 1334 (43.7) | 228 (7.5) | 394 (12.9) | 61 (2.0) | 149 (4.9) | 25 (0.8) | ||||||
| Female | 2494 | 1128 (45.2) | 166 (6.7) | 303 (12.1) | 48 (1.9) | 79 (3.2) | 10 (0.4) | |||||||
| n.s. | n.s. | n.s. | n.s. | p = 0.0014 | n.s. | |||||||||
| Age | < 65 years | 1239 | 439 (35.4) | 53 (4.3) | 87 (7.0) | 12 (1.0) | 33(2.7) | 1 (0.1) | ||||||
| ≥ 65 years | 4308 | 2023 (47.0) | 341 (7.9) | 610 (14.2) | 97 (2.3) | 195 (4.5) | 34 (0.8) | |||||||
| p < 0.0001 | p < 0.0001 | p < 0.0001 | p = 0.0034 | p = 0.0033 | p = 0.0033 | |||||||||
| Underlying disease | Diabetic nephropathy | 1818 | 749 (41.2) | 100 (5.5) | 257 (14.1) | 34 (1.9) | 63 (3.5) | 9 (0.5) | ||||||
| Chronic glomerulonephritis | 1182 | 494 (41.8) | 87 (7.4) | 103 (8.7) | 17 (1.4) | 53 (4.5) | 8 (0.7) | |||||||
| Nephrosclerosis | 1623 | 760 (46.8) | 131(8.1) | 239 (14.7) | 37 (2.3) | 61 (3.8) | 12 (0.7) | |||||||
| Others | 924 | 459 (49.7) | 76 (8.2) | 98 (10.6) | 21 (2.3) | 51 (5.5) | 6 (0.6) | |||||||
| p < 0.0001 | p = 0.0104 | p < 0.0001 | n.s. | n.s. | n.s. | |||||||||
| History of CV disease | No | 4812 | 2072 (43.1) | 317 (6.6) | 534 (11.1) | 86 (1.8) | 193 (4.0) | 29 (0.6) | ||||||
| Yes | 735 | 390 (53.1) | 77 (10.5) | 163 (22.2) | 23 (3.1) | 35 (4.8) | 6 (0.8) | |||||||
| p < 0.0001 | p = 0.0003 | p < 0.0001 | p = 0.0214 | n.s. | n.s. | |||||||||
| History of malignant tumors | No | 5135 | 2227 (43.4) | 349 (6.8) | 624 (12.2) | 99 (1.9) | 193 (3.8) | 26 (0.5) | ||||||
| Yes | 412 | 235 (57.0) | 45 (10.9) | 73 (17.7) | 10 (2.4) | 35 (8.5) | 9 (2.2) | |||||||
| p < 0.0001 | p = 0.0036 | p = 0.0019 | n.s. | p < 0.0001 | p = 0.0008 | |||||||||
| Comorbidity of CV disease | No | 3241 | 1269 (39.2) | 206 (6.4) | 243 (7.5) | 43 (1.3) | 129 (4.0) | 16 (0.5) | ||||||
| Yes | 2306 | 1193 (51.7) | 188 (8.2) | 454 (19.7) | 66 (2.9) | 99 (4.3) | 19 (0.8) | |||||||
| p < 0.0001 | p = 0.0109 | p < 0.0001 | p < 0.0001 | n.s | n.s. | |||||||||
| Comorbidity of malignant tumors | No | 5217 | 2258 (43.3) | 365 (7.0) | 652 (12.5) | 100 (1.9) | 150 (2.9) | 26 (0.5) | ||||||
| Yes | 330 | 204 (61.8) | 29 (8.8) | 45 (13.6) | 9 (2.7) | 78 (23.6) | 9 (2.7) | |||||||
| p < 0.0001 | n.s. | n.s. | n.s. | p < 0.0001 | p = 0.0001 | |||||||||
| rHuEPO administration in past 3 months | No | 3202 | 1373 (42.9) | 217 (6.8) | 384 (12.0) | 55 (1.7) | 119 (3.7) | 16 (0.5) | ||||||
| Yes | 2244 | 1048 (46.7) | 170 (7.6) | 303 (13.5) | 52 (2.3) | 104 (4.6) | 17 (0.8) | |||||||
| Unknown, not recorded | 101 | 41 (40.6) | 7 (6.9) | 10 (9.9) | 2 (2.0) | 5 (5.0) | 2 (2.0) | |||||||
| p = 0.0056 | n.s. | n.s. | n.s. | n.s. | n.s. | |||||||||
Differences between factors were analyzed by Fisher’s exact test or the chi-square test
ADR adverse drug reaction, AE adverse event, CV cardiovascular, rHuEPO recombinant human erythropoietin, n.s. not significant
Table 4 shows all AEs and CV-related AEs stratified by Hb level at the time of the AE occurrence for the 5517 patients in the safety analysis set with available Hb values. The proportion of patients with CV-related AEs was lower in those with high Hb levels than in those with low Hb levels. The same tendency was observed for all AEs, AEs by MedDRA system organ class (SOC), and major CV-related AEs.
Table 4.
All adverse events and cardiovascular-related adverse events stratified by hemoglobin level at the time of the event occurrence
| Hb level at the time of all AEs and CV-related AE occurrences | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| < 10 g/dL | ≥ 10 g/dL < 11 g/dL |
≥ 11 g/dL < 12 g/dL |
≥ 12 g/dL < 13 g/dL |
≥ 13 g/dL | ||||||
| No. of all AEs | 2763 | 1598 | 983 | 390 | 126 | |||||
| No. of patients with all AEs (%) | 1456 (26.4) | 957 (17.3) | 622 (11.3) | 268 (4.9) | 88 (1.6) | |||||
| No. of CV-related AEs | 440 | 211 | 122 | 60 | 11 | |||||
| No. of patients with CV-related AEs (%) | 394 (7.1) | 188 (3.4) | 113 (2.0) | 57 (1.0) | 11 (0.2) | |||||
| No. of patients with CV-related AEs by type (MedDRA SOC) | ||||||||||
| Nervous system disordersa | 53 (1.0) | 38 (0.7) | 23 (0.4) | 18 (0.3) | 3 (0.1) | |||||
| Cardiac disordersb | 318 (5.8) | 138 (2.5) | 84 (1.5) | 34 (0.6) | 7 (0.1) | |||||
| Vascular disordersc | 24 (0.4) | 12 (0.2) | 5 (0.1) | 7 (0.1) | – | |||||
| General disorders and administration site conditionsd | 12 (0.2) | 6 (0.1) | 5 (0.1) | 1 (0.0) | 1 (0.0) | |||||
| Major CV-related AEs | ||||||||||
| Cardiac failure | 151 (2.7) | 59 (1.1) | 27 (0.5) | 11 (0.2) | 2 (0.0) | |||||
| Congestive heart failure | 65 (1.2) | 33 (0.6) | 21 (0.4) | 8 (0.1) | 1 (0.0) | |||||
| Cerebral infarction | 20 (0.4) | 27 (0.5) | 5 (0.1) | 11 (0.2) | 1 (0.0) | |||||
Patients in the safety analysis set with available Hb values (n = 5517) were subject to analysis
AEs adverse events, CV cardiovascular, Hb hemoglobin, MedDRA Medical Dictionary for Regulatory Activities, SOC system organ class
aCerebral infarction, cerebral hemorrhage, transient ischemic attack
bCardiac failure, angina pectoris, myocardial infarction
cPeripheral arterial occlusive disease, aortic dissection, aortic aneurysm
dSudden death, cardiac death
Among the patients in the safety analysis set, 39 patients had a history of cerebrovascular disease that was found to be correlated with a history of CV disease. After excluding these patients, factors affecting the occurrence of CV-related AEs were assessed in the remaining 5508 patients by Cox regression analysis. The following factors were found to be significantly associated with the risk of CV-related AEs: advanced age (≥ 65 years), history of CV disease, presence of diabetes, transfusion, baseline eGFR < 30 mL/min/1.73 m2, high baseline urine protein, median darbepoetin dose administered per week of ≥ 17.1 µg, and concomitant use of antithrombotic agents (Table 5).
Table 5.
Multivariate Cox regression analysis of cardiovascular-related adverse events
| Factors | No. of patients | No. of patients with CV-related AEs (%) | HR (95% CI) | p value | |
|---|---|---|---|---|---|
| Age (years) | < 65 | 1232 | 79 (6.4) | Ref | |
| ≥ 65 to < 75 | 1459 | 171 (11.7) | 1.931 (1.258–2.966) | 0.0026 | |
| ≥ 75 | 2817 | 404 (14.3) | 2.457 (1.632–3.699) | < 0.0001 | |
| History of CV disease | No | 4782 | 502 (10.5) | Ref | |
| Yes | 726 | 152 (20.9) | 2.216 (1.656–2.966) | < 0.0001 | |
| Concurrent diabetes | No | 3162 | 343 (10.8) | Ref | |
| Yes | 2346 | 311 (13.3) | 1.561 (1.199–2.032) | 0.0009 | |
| Transfusion | No | 4995 | 547 (11.0) | Ref | |
| Yes | 475 | 105 (22.1) | 1.913 (1.367–2.676) | 0.0002 | |
| Baseline eGFR (mL/min/1.73 m2) | < 15 | 2227 | 255 (11.5) | 1.849 (1.122–3.046) | 0.0159 |
| ≥ 15 to < 30 | 2074 | 279 (13.5) | 1.857 (1.142–3.019) | 0.0125 | |
| ≥ 30 | 655 | 61 (9.3) | Ref | ||
| Baseline urine protein (mg/dL) | Continuous quantity | 2451 | 260 (10.6) | 1.001 (1.000–1.001) | 0.0026 |
| Dose of darbepoetin per week | < Median | 2754 | 309 (11.2) | Ref | |
| ≥ Median | 2754 | 345 (12.5) | 1.361 (1.049–1.766) | 0.0201 | |
| Use of antithrombotic agent | No | 5018 | 511 (10.2) | Ref | |
| Yes | 490 | 143 (29.2) | 2.377 (1.701–3.322) | < 0.0001 | |
Patients from the safety analysis set (n = 5547) without a history of cerebrovascular disease (n = 39) were included in this multivariate analysis (n = 5508)
The following variables were selected as explanatory variables: sex, age, body mass index, history of CV disease, hypertension, hyperlipidemia, diabetes, previous treatment with recombinant human erythropoietin, transfusion, baseline systolic blood pressure, baseline diastolic blood pressure, baseline eGFR, increase rate of hemoglobin level during darbepoetin administration for 4 weeks, baseline urine protein (spot urine), dose of darbepoetin administered per week, antihypertensive agent, antithrombotic agent, and lipid-lowering agent. As history of CV disease and cerebrovascular disease were strongly correlated (Spearman’s rank correlation coefficient < 0.7), only history of CV disease was selected for the model
AE adverse event, CI confidence interval, CV cardiovascular, eGFR estimated glomerular filtration rate, HR hazard ratio
P values for trend (Wald chi-square test [type 3]) were p < 0.0001 for age and p = 0.0375 for baseline eGFR
The median (25, 75 percentiles) of darbepoetin dose administered per week was 17.1 (11.4, 26.5) µg
Effectiveness
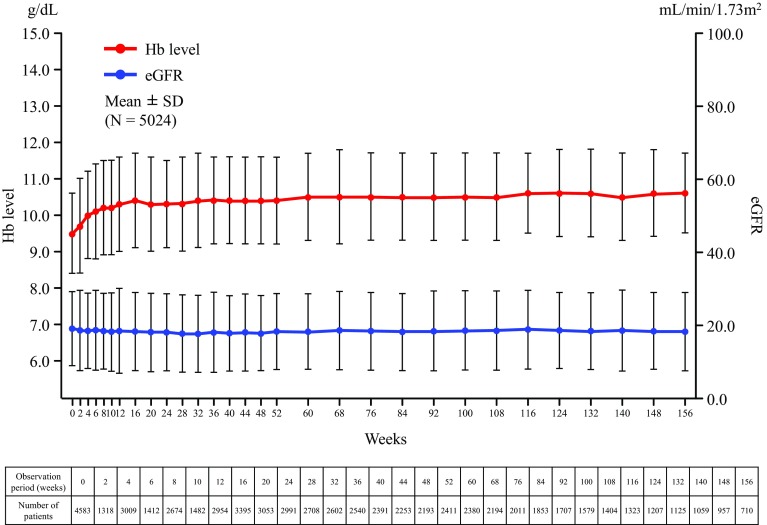
Of the 5430 patients in the effectiveness analysis set, 5024 patients with available Hb levels and eGFR at the same evaluation point were included in the analysis. The mean Hb level at baseline of 9.5 g/dL started to increase from Week 2 of darbepoetin administration and remained between 10.0 and 10.6 g/dL from Week 4 to Week 156 (Fig. 2). In the same patients, the eGFR remained almost constant throughout the observation period (18.7 ± 10.3 mL/min/1.73 m2 at baseline and 17.9 ± 10.8 mL/min/1.73 m2 at Week 156 of darbepoetin treatment; Fig. 2). The administered dose of darbepoetin (mean ± SD, µg/week) in the effectiveness analysis set regarding Hb and serum creatinine levels (N = 5030) was 49.9 ± 27.7 at baseline and increased gradually, reaching 76.4 ± 45.9 at Week 156. When stratified by dosing intervals, Hb levels were maintained at approximately 10.0 g/dL with each dosing interval from 3 months to 3 years of treatment (Online Resource 1).
Fig. 2.
Changes in Hb levels and eGFR. eGFR estimated glomerular filtration rate, Hb hemoglobin, SD standard deviation
In the effectiveness analysis set regarding Hb levels and eGFR, 4444 patients who received darbepoetin for more than 3 months and did not develop any of the renal endpoints within 3 months were followed up for 3 years for further assessment of renal endpoints (50% reduction in eGFR, initiation of dialysis, or kidney transplantation). Among these patients, 1813 patients (40.8%) developed composite renal endpoints.
The results of multivariate Cox regression analysis of factors affecting the occurrence of composite renal endpoints are shown in Table 6. Composite renal endpoints occurred in 39.5% of patients with Hb ≥ 11 g/dL at 3 months after initial darbepoetin administration compared with 44.8% of patients with Hb < 11 g/dL, demonstrating that patients with Hb ≥ 11 g/dL at 3 months after the start of darbepoetin administration had a lower risk of composite renal endpoints (hazard ratio, 0.731; 95% confidence interval, 0.604–0.884; p = 0.0013).
Table 6.
Multivariate Cox regression analysis of composite renal endpoints
| Factors | No. of patients | No. of patients with renal endpoints (%) | HR (95% CI) | p value | |
|---|---|---|---|---|---|
| Hb level at 3 months after the start of darbepoetin administration | < 11 g/dL | 2380 | 1066 (44.8) | Ref | |
| ≥ 11 g/dL | 1093 | 432 (39.5) | 0.731 (0.604–0.884) | 0.0013 | |
| Sex | Male | 2412 | 1071 (44.4) | Ref | |
| Female | 2032 | 742 (36.5) | 0.634 (0.536–0.749) | < 0.0001 | |
| Age (years) | < 65 | 999 | 580 (58.1) | Ref | |
| ≥ 65 to < 75 | 1195 | 558 (46.7) | 0.794 (0.652–0.967) | 0.0216 | |
| ≥ 75 | 2250 | 675 (30.0) | 0.669 (0.547–0.819) | < 0.0001 | |
| Diabetes | No | 2567 | 942 (36.7) | Ref | |
| Yes | 1877 | 871 (46.4) | 1.186 (1.004–1.401) | 0.0444 | |
| Transfusion | No | 4047 | 1631 (40.3) | Ref | |
| Yes | 364 | 176 (48.4) | 1.424 (1.062–1.910) | 0.0183 | |
| Baseline systolic blood pressure (mmHg) | < 130 | 1519 | 512 (33.7) | Ref | |
| ≥ 130 to < 140 | 851 | 366 (43.0) | 1.468 (1.181–1.824) | 0.0005 | |
| ≥ 140 | 1480 | 751 (50.7) | 1.434 (1.186–1.734) | 0.0002 | |
| Baseline eGFR (mL/min/1.73 m2) | < 15 | 1791 | 1082 (60.4) | 3.227 (2.245–4.639) | < 0.0001 |
| 15 to < 30 | 1729 | 544 (31.5) | 1.344 (0.926–1.951) | 0.1204 | |
| ≥ 30 | 538 | 95 (17.7) | Ref | ||
| Baseline urine protein (mg/dL) | Continuous quantity | 2033 | 882 (43.4) | 1.001 (1.001–1.001) | < 0.0001 |
| Dose of darbepoetin administered per week | < Median | 2220 | 741 (33.4) | Ref | |
| ≥ Median | 2224 | 1072 (48.2) | 1.514 (1.282–1.790) | < 0.0001 | |
| Concurrent use of antihypertensive agent | No | 650 | 175 (26.9) | Ref | |
| Yes | 3794 | 1638 (43.2) | 1.654 (1.158–2.360) | 0.0056 | |
Patients in the effectiveness analysis set regarding Hb levels and eGFR who received darbepoetin for more than 3 months and did not develop any of the endpoints (50% reduction in eGFR, initiation of dialysis, and kidney transplantation) evaluated within 3 months of darbepoetin administration were included in this analysis (n = 4444)
The following variables were selected as explanatory variables: Hb level at 3 months after the start of darbepoetin administration, sex, age, body mass index, history, diabetes, previous treatment with recombinant human erythropoietin, transfusion, baseline systolic blood pressure, baseline eGFR, increase rate of Hb level during darbepoetin administration for 4 weeks, baseline urine protein (spot urine), dose of darbepoetin administered per week, antihypertensive agent, antithrombotic agent, and lipid-lowering agent
Time (days) to composite renal endpoints (either 50% reduction in eGFR, initiation of dialysis, or kidney transplantation)
CI confidence interval, eGFR estimated glomerular filtration rate, Hb hemoglobin, HR hazard ratio
P values for trend (Wald chi-square test [type 3]) were p = 0.0004 for age, p = 0.0002 for baseline systolic blood pressure, and p < 0.0001 for baseline eGFR
The median (25, 75 percentiles) of darbepoetin dose administered per week was 15.4 (10.3, 23.2) µg
Female patients or those with advanced age (≥ 65 years) had a lower risk of composite renal endpoints than did male or younger patients, while a high risk of composite renal endpoints was found in patients with diabetes, transfusion, baseline systolic blood pressure ≥ 30 mmHg, baseline eGFR < 30 mL/min/1.73 m2, higher baseline urine protein, median dose of darbepoetin administered per week ≥ 15.4 µg, and concomitant use of antihypertensive agents.
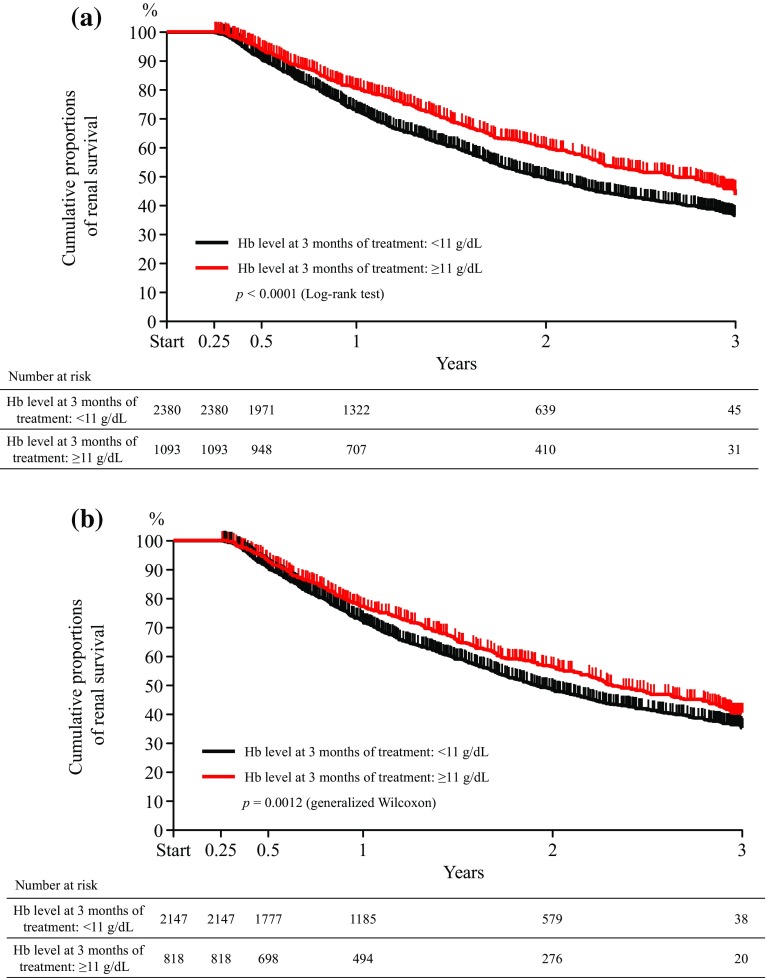
Of the patients in the effectiveness analysis set regarding Hb levels and eGFR, 3473 patients with available Hb levels at 3 months after the start of darbepoetin administration were divided into two groups: those with Hb < 11 g/dL (N = 2380) and those with Hb ≥ 11 g/dL at 3 months after the start of darbepoetin administration (N = 1093). Time to occurrence of composite renal endpoints was assessed using Kaplan–Meier survival analysis (Fig. 3a). The cumulative proportion of renal survival during 3 years of darbepoetin administration was significantly higher in those with Hb ≥ 11 g/dL than those with Hb < 11 g/dL at 3 months after the start of darbepoetin administration (p < 0.0001, log-rank). A sensitivity analysis, which was performed for patients with Hb < 11 g/dL at baseline, showed a similar result that the cumulative proportion of renal survival was higher in those with Hb ≥ 11 g/dL than those with Hb < 11 g/dL at 3 months after the start of darbepoetin administration (p = 0.0012, generalized Wilcoxon test, Fig. 3b).
Fig. 3.
Analysis of time to first outcome on composite renal endpoints in the effectiveness analysis set for whom available Hb levels at 3 months after the start of darbepoetin administration were stratified by < 11 g/dL or ≥ 11 g/dL. a All patients. b Patients whose baseline Hb levels were < 11 g/dL. Patients who died during the course of follow-ups were excluded from the analysis. Composite renal endpoints: 50% reduction in eGFR, initiation of dialysis, or kidney transplantation. eGFR estimated glomerular filtration rate, Hb hemoglobin
Multivariate Cox regression analysis and Kaplan–Meier survival analysis of time to event provided similar results even after death was included in composite renal endpoints (Online Resources 2 and 3a, b).
Discussion
This paper reports on the results of the DREAM-J surveillance study conducted to explore the factors that affect the occurrence of ADRs, safety, and effectiveness of long-term use of darbepoetin in non-dialysis CKD patients with renal anemia. In this study, ADRs occurred in 7.1% of patients. The ADR rate in this study was not higher than that in phase 1–3 studies on non-dialysis CKD patients in Japan [19–21]. Furthermore, major ADRs that could not be specified in the “Precautions for Use” section of the product package (4th edition revised on December 2014) were heart failure, death, sudden death, congestive heart failure, pneumonia, and gastric cancer. These ADRs had been demonstrated in clinical studies in other countries [15, 22]. According to these safety results, no new safety concerns were identified in the present study.
The incidence rate of ADRs in the present study (7.1%) was lower than that reported in pre-approval Japanese clinical trials (30.75%) [19–21]. Regarding ADRs classified by SOC, cardiac disorders occurred in 1.2% of patients, which was similar to the occurrence of cardiac disorders reported in the pre-approval Japanese clinical studies (1.36%) [19–21]. Although no malignant tumors occurred in the pre-approval Japanese clinical studies, they occurred in 0.6% of the patients in this study. Data from the Japan Cancer Surveillance Research Group (Cancer Information Service, National Cancer Center, Japan) have shown that crude incidence rates of cancer in Japan were 0.76% for men and 0.54% for women in 2009 [23], suggesting that the incidence rate of 0.6% in this study (average, 1.8 years; maximum, 3 years) is roughly comparable to that of the general population in Japan. Therefore, the discrepancy between this study and the clinical studies may be attributable to the fact that patients with a history of malignant tumor were not excluded from this study and that the observation period was rather long. Finally, hypertension, increase in blood pressure, and other ADRs that occur relatively frequently developed at similar frequencies as reported in the pre-approval Japanese clinical studies and overseas large-scale clinical studies [13–15].
In the DREAM-J surveillance study, the incidence of CV-related AEs did not tend to increase in patients with higher Hb levels. In large-scale clinical studies outside Japan, concern was raised that treatment of non-dialysis CKD patients with renal anemia using ESA with the aim of normalizing Hb levels may be associated with increased risk of CV disease. In the CHOIR study (target Hb, 13.5 g/dL) [14], there were increased incidences of myocardial infarction, hospitalization due to heart failure, and stroke in patients with higher Hb levels; whereas, in the TREAT study (target Hb, ≥ 13.0 g/dL) [15], an increased incidence of cerebral infarction in patients with higher Hb levels was noted. However, in randomized controlled studies of non-dialysis CKD patients with renal anemia conducted in Japan, no difference was found between the darbepoetin group (target Hb, 11.0–13.0 g/dL) and the rHuEPO group (target Hb, 9.0–11.0 g/dL) in terms of safety, including risk for CV events. However, there was a significant decrease in the left ventricular mass index in the darbepoetin group [19, 20]. The present study revealed no association between Hb levels and the risk of CV-related AEs, including cerebral infarction, cardiac failure, and congestive heart failure, thus supporting the results obtained in Japanese randomized controlled trials [19–21]. The trends for all AEs and CV-related AEs by Hb values were similar. Taken together, these results suggest that there is no apparent risk for CV events under clinical conditions where darbepoetin treatment is prescribed in accordance with Japanese guidelines for the management of anemia (2008) [24].
This study demonstrated that a decrease in composite renal endpoints is associated with the achievement of target Hb levels being maintained by darbepoetin. Kaplan–Meier survival analysis of time to occurrence of composite renal endpoints revealed a significantly lower incidence of events in patients who achieved an Hb ≥ 11 g/dL at 3 months after the start of darbepoetin administration. Moreover, multivariate Cox regression analysis identified the maintenance of Hb ≥ 11 g/dL 3 months after the start of darbepoetin administration as a significant factor for decreased risk of composite renal endpoints. These results are consistent with those obtained with rHuEPO in previous studies [5, 6, 25]. These results also support the findings of a study in which darbepoetin treatment was given for 3 years while Hb levels were maintained at a target of 11.0–13.0 g/dL, and a 29% risk reduction for composite renal endpoints was found [21].
However, reports of studies outside Japan suggest that there is no relationship between ESA treatment and preservation of renal function. In the CREATE study [13], dialysis was required in a significantly higher proportion of patients with high Hb levels, while no difference was seen between high and low Hb groups in terms of reduction in eGFR. In the CREATE study, the target Hb value in the high Hb group was as high as 13.0–15.0 g/dL, and hypertension (defined as a systolic blood pressure of ≥ 160 mmHg) was reported as an AE in more patients in the high Hb group (20% of patients in the low Hb group vs. 30% in the high Hb group). ESA treatment has been reported to exert kidney protection independent from its correction of anemia and result in the improvement of renal hemodynamics, as shown by the induction of an anti-oxidative enzyme involved in endothelial function [26], and an anti-apoptotic effect in glomerular epithelial cells [27]. In this study, increased systolic blood pressure was shown to be a significant risk factor for composite renal endpoints. Given that ESAs often cause increased blood pressure, appropriate management of this is likely to contribute to preventing composite renal endpoints.
In the multivariate Cox models, patients who received higher doses of darbepoetin had increased risks of composite renal endpoints and CV-related AEs. It is possible that this is related to ESA hyporesponsiveness. ESA hyporesponsiveness caused various clinical conditions including bleeding/blood loss, hematopoietic disorder (infection, inflammation, and malignant tumor), deficiency of elements required for erythropoiesis (iron, folic acid, and vitamin B12 deficiency), and anti-erythropoietin antibody [28]. In the secondary analysis of the TREAT study, patients with ESA hyporesponsiveness had significantly higher rates of death and cardiovascular events [29]. In that study, there were no apparent increases in CV-related AEs at higher Hb levels. Therefore, ESA hyporesponsiveness may increase renal and CV risk in patients who receive higher doses of darbepoetin. In the present study, a lower incidence of CV-related AEs was observed in patients with Hb ≥ 11 g/dL than in patients with Hb < 11 g/dL. The latest guidelines at the start of the current study (2008) [24] recommended an Hb level of 11.0–13.0 g/dL. In the most recent guideline (2015), the same target range has been adopted [28]. The results of the current study validate the lower range of the target Hb level (11.0 g/dL) recommended by the Japanese guidelines [24, 28]. The examination of CV-related AEs by patient characteristics revealed that an increase in CV-related AEs was found in patients with a history of CV disease and of CV-related comorbidity. Multivariate Cox regression analysis identified advanced age, diabetes, transfusion, low eGFR values, high urine protein, and use of antithrombotic agents as independent risk factors for increased CV-related AEs. Careful consideration should, therefore, be given to CV-related AEs when darbepoetin is administered to such patients.
Limitations
This post-marketing surveillance is an observational study conducted under the rules of GPSP; as such, it is expected to show reporting bias concerning AEs compared with clinical trials (intervention studies) conducted under GCP guidelines. There was no control group to allow a comparison of safety and effectiveness and no comparison of AEs and prognosis. Additionally, data from patients who withdrew from the study or switched to renal replacement therapies were not provided. As part of the design of this study, AEs were collected according to spontaneous reports, and it appears that the incidence of AEs was lower than in clinical studies [19–21]. Although caution should be applied when comparing the incidence in this study and clinical studies, it is thought that this result may reflect the underlying medical condition as this drug was used for a wide range of patients. In this study, we could not evaluate multivariate Cox models adjusted for iron status, because we did not obtain laboratory results on iron status, or information regarding iron intake. Furthermore, in this study, the prescription of darbepoetin is based solely on the clinical judgment of the nephrologists in charge of the patients. Therefore, the influence of facility or physician bias cannot be excluded. Despite these limitations, this is a large-scale, prospective study conducted under clinical conditions in Japan and reflects the status quo of medical practice in Japan. Further large-scale, prospective studies are required to verify the effects of the correction of Hb levels using darbepoetin on CV-related events and composite renal endpoints.
Conclusions
The DREAM-J surveillance study demonstrated the safety profile of darbepoetin under clinical conditions in Japan. The study results did not suggest an increase in the risk of CV-related AEs under high Hb levels. Additionally, our findings illustrate that darbepoetin treatment with a target Hb level of ≥ 11 g/dL, as recommended by the guidelines, is associated with a lower occurrence of composite renal endpoints at an early stage of CKD. Results from this surveillance study provide further insights into CKD in Japan.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
The authors wish to thank Dr. Allison Kirsop and Dr. Sarah Williams of Edanz Medical Writing for providing medical writing support during the development of this manuscript, which was funded by Kyowa Hakko Kirin Co., Ltd.
Funding
This study was funded by Kyowa Hakko Kirin Co., Ltd.
Conflict of interest
Kyowa Hakko Kirin Co., Ltd. provided funding for the DREAM-J surveillance study. Masaomi Nangaku, Enyu Imai, Yoshiharu Tsubakihara, and Tadao Akizawa are members of the scientific advisory board of the DREAM-J surveillance study. Tetsuhiro Tanaka has received lecture fees from Kyowa Hakko Kirin Co., Ltd. Masaomi Nangaku has received lecture fees from Kyowa Hakko Kirin Co., Ltd., Daiichi Sankyo Company, Limited, MSD K.K., Astellas Pharma Inc., AstraZeneca, Alexion Pharmaceuticals, Inc., Mitsubishi Tanabe Pharma Corporation, Japan Tobacco Inc., GlaxoSmithKline K.K., and Otsuka Pharmaceutical Co., Ltd.; manuscript fees from Kyowa Hakko Kirin Co., Ltd.; and scholarship/grant/research support from Kyowa Hakko Kirin Co., Ltd., Astellas Pharma Inc., Daiichi Sankyo Company, Limited, Chugai Pharmaceutical Co., Ltd., Mitsubishi Tanabe Pharma Corporation, and Torii Pharmaceutical Co., Ltd. Enyu Imai has received lecture fees from Kyowa Hakko Kirin Co., Ltd., Sumitomo Dainippon Pharma Co., Ltd., Takeda Pharmaceutical Company Limited, Daiichi Sankyo Company, Limited, Nippon Boehringer Ingelheim Co., Ltd., and Otsuka Pharmaceutical Co., Ltd., and manuscript fees from Kyowa Hakko Kirin Co., Ltd. Yoshiharu Tsubakihara has received lecture fees from Chugai Pharmaceutical Co., Ltd., Kyowa Hakko Kirin Co., Ltd., Mitsubishi Tanabe Pharma Corporation, Torii Pharmaceutical Co., Ltd., and Otsuka Pharmaceutical Co., Ltd.; scholarship/grant/research support from Bayer Yakuhin, Ltd.; and sponsorship for courses from Chugai Pharmaceutical Co., Ltd., and Baxter Limited. Tadao Akizawa is a board member/advisor/consultant for Kyowa Hakko Kirin Co., Ltd., Astellas Pharma Inc., Bayer Yakuhin, Ltd., Fuso Pharmaceutical Industries, Ltd., Japan Tobacco Inc., Ono Pharmaceutical Co., Ltd., and NIPRO; has received lecture fees from Kyowa Hakko Kirin Co., Ltd., Chugai Pharmaceutical Co., Ltd., Bayer Yakuhin, Ltd., Kissei Pharmaceutical Co., Ltd., and Torii Pharmaceutical Co., Ltd.; and has received manuscript fees from Astellas Pharma Inc. Masatoshi Kamai, Michihito Wada, and Shinji Asada are employees of Kyowa Hakko Kirin Co., Ltd.
Ethical approval and informed consent
Informed consent and institutional review board approval were not required in the present surveillance according to GPSP. This article does not contain any studies with animals performed by any of the authors.
References
- 1.Babitt JL, Lin HY. Mechanisms of anemia in CKD. J Am Soc Nephrol. 2012;23:1631–1634. doi: 10.1681/ASN.2011111078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Vlagopoulos PT, Tighiouart H, Weiner DE, Griffith J, Pettitt D, Salem DN, et al. Anemia as a risk factor for cardiovascular disease and all-cause mortality in diabetes: the impact of chronic kidney disease. J Am Soc Nephrol. 2005;16:3403–3410. doi: 10.1681/ASN.2005030226. [DOI] [PubMed] [Google Scholar]
- 3.Muntner P, He J, Astor BC, Folsom AR, Coresh J. Traditional and nontraditional risk factors predict coronary heart disease in chronic kidney disease: results from the Atherosclerosis Risk in Communities study. J Am Soc Nephrol. 2005;16:529–538. doi: 10.1681/ASN.2004080656. [DOI] [PubMed] [Google Scholar]
- 4.Silverberg D. Outcomes of anaemia management in renal insufficiency and cardiac disease. Nephrol Dial Transplant. 2003;18(Suppl 2):ii7–ii12. [PubMed] [Google Scholar]
- 5.Kuriyama S, Tomonari H, Yoshida H, Hashimoto T, Kawaguchi Y, Sakai O. Reversal of anemia by erythropoietin therapy retards the progression of chronic renal failure, especially in nondiabetic patients. Nephron. 1997;77:176–185. doi: 10.1159/000190270. [DOI] [PubMed] [Google Scholar]
- 6.Gouva C, Nikolopoulos P, Ioannidis JP, Siamopoulos KC. Treating anemia early in renal failure patients slows the decline of renal function: a randomized controlled trial. Kidney Int. 2004;66:753–760. doi: 10.1111/j.1523-1755.2004.00797.x. [DOI] [PubMed] [Google Scholar]
- 7.Ayus JC, Go AS, Valderrabano F, Verde E, de Vinuesa SG, Achinger SG, et al. Effects of erythropoietin on left ventricular hypertrophy in adults with severe chronic renal failure and hemoglobin < 10 g/dL. Kidney Int. 2005;68:788–795. doi: 10.1111/j.1523-1755.2005.00458.x. [DOI] [PubMed] [Google Scholar]
- 8.Parfrey PS, Lauve M, Latremouille-Viau D, Lefebvre P. Erythropoietin therapy and left ventricular mass index in CKD and ESRD patients: a meta-analysis. Clin J Am Soc Nephrol. 2009;4:755–762. doi: 10.2215/CJN.02730608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Palmer SC, Saglimbene V, Craig JC, Navaneethan SD, Strippoli GF. Darbepoetin for the anaemia of chronic kidney disease. Cochrane Database Syst Rev. 2014;3:CD009297. doi: 10.1002/14651858.CD009297.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Palmer SC, Saglimbene V, Mavridis D, Salanti G, Craig JC, Tonelli M, et al. Erythropoiesis-stimulating agents for anaemia in adults with chronic kidney disease: a network meta-analysis. Cochrane Database Syst Rev. 2014;12:CD010590. doi: 10.1002/14651858.CD010590.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Covic A, Nistor I, Donciu MD, Dumea R, Bolignano D, Goldsmith D. Erythropoiesis-stimulating agents (ESA) for preventing the progression of chronic kidney disease: a meta-analysis of 19 studies. Am J Nephrol. 2014;40:263–279. doi: 10.1159/000366025. [DOI] [PubMed] [Google Scholar]
- 12.Cody JD, Hodson EM. Recombinant human erythropoietin versus placebo or no treatment for the anaemia of chronic kidney disease in people not requiring dialysis. Cochrane Database Syst Rev. 2016;1:CD003266. doi: 10.1002/14651858.CD003266.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Drüeke TB, Locatelli F, Clyne N, Eckardt KU, Macdougall IC, Tsakiris D, et al. CREATE investigators. Normalization of hemoglobin level in patients with chronic kidney disease and anemia. N Engl J Med. 2006;355:2071–2084. doi: 10.1056/NEJMoa062276. [DOI] [PubMed] [Google Scholar]
- 14.Singh AK, Szczech L, Tang KL, Barnhart H, Sapp S, Wolfson M, et al. CHOIR investigators. Correction of anemia with epoetin alfa in chronic kidney disease. N Engl J Med. 2006;355:2085–2098. doi: 10.1056/NEJMoa065485. [DOI] [PubMed] [Google Scholar]
- 15.Pfeffer MA, Burdmann EA, Chen CY, Cooper ME, de Zeeuw D, Eckardt KU, et al. TREAT investigators. A trial of darbepoetin alfa in type 2 diabetes and chronic kidney disease. N Engl J Med. 2009;361:2019–2032. doi: 10.1056/NEJMoa0907845. [DOI] [PubMed] [Google Scholar]
- 16.Phrommintikul A, Haas SJ, Elsik M, Krum H. Mortality and target haemoglobin concentrations in anaemic patients with chronic kidney disease treated with erythropoietin: a meta-analysis. Lancet. 2007;369:381–388. doi: 10.1016/S0140-6736(07)60194-9. [DOI] [PubMed] [Google Scholar]
- 17.Palmer SC, Navaneethan SD, Craig JC, Johnson DW, Tonelli M, Garg AX, et al. Meta-analysis: erythropoiesis-stimulating agents in patients with chronic kidney disease. Ann Intern Med. 2010;153:23–33. doi: 10.7326/0003-4819-153-1-201007060-00252. [DOI] [PubMed] [Google Scholar]
- 18.Mimura I, Tanaka T, Nangaku M. How the target hemoglobin of renal anemia should be. Nephron. 2015;131:202–209. doi: 10.1159/000440849. [DOI] [PubMed] [Google Scholar]
- 19.Akizawa T, Gejyo F, Nishi S, Iino Y, Watanabe Y, Suzuki M, et al. Positive outcomes of high hemoglobin target in patients with chronic kidney disease not on dialysis: a randomized controlled study. Ther Apher Dial. 2011;15:431–440. doi: 10.1111/j.1744-9987.2011.00931.x. [DOI] [PubMed] [Google Scholar]
- 20.Akaishi M, Hiroe M, Hada Y, Suzuki M, Tsubakihara Y, Akizawa T, et al. Effect of anemia correction on left ventricular hypertrophy in patients with modestly high hemoglobin level and chronic kidney disease. J Cardiol. 2013;62:249–256. doi: 10.1016/j.jjcc.2013.04.008. [DOI] [PubMed] [Google Scholar]
- 21.Tsubakihara Y, Gejyo F, Nishi S, Iino Y, Watanabe Y, Suzuki M, et al. High target hemoglobin with erythropoiesis-stimulating agents has advantages in the renal function of non-dialysis chronic kidney disease patients. Ther Apher Dial. 2012;16:529–540. doi: 10.1111/j.1744-9987.2012.01082.x. [DOI] [PubMed] [Google Scholar]
- 22.Swedberg K, Young JB, Anand IS, Cheng S, Desai AS, Diaz R, et al. Treatment of anemia with darbepoetin alfa in systolic heart failure. N Engl J Med. 2013;368:1210–1219. doi: 10.1056/NEJMoa1214865. [DOI] [PubMed] [Google Scholar]
- 23.Hori M, Matsuda T, Shibata A, Katanoda K, Sobue T, Nishimoto H, et al. Cancer incidence and incidence rates in Japan in 2009: a study of 32 population-based cancer registries for the Monitoring of Cancer Incidence in Japan (MCIJ) project. Jpn J Clin Oncol. 2015;45:884–891. doi: 10.1093/jjco/hyv088. [DOI] [PubMed] [Google Scholar]
- 24.Tsubakihara Y, Nishi S, Akiba T, Hirakata H, Iseki K, Kubota M, et al. 2008 Japanese Society for Dialysis Therapy: guidelines for renal anemia in chronic kidney disease. Ther Apher Dial. 2010;14:240–275. doi: 10.1111/j.1744-9987.2010.00836.x. [DOI] [PubMed] [Google Scholar]
- 25.Akizawa T, Saito A, Gejyo F, Suzuki M, Nishizawa Y, Tomino Y, et al. Co-JET Study Group. Impacts of recombinant human erythropoietin treatment during predialysis periods on the progression of chronic kidney disease in a large-scale cohort study (Co-JET study) Ther Apher Dial. 2014;18:140–148. doi: 10.1111/1744-9987.12066. [DOI] [PubMed] [Google Scholar]
- 26.Katavetin P, Inagi R, Miyata T, Shao J, Sassa R, Adler S, et al. Erythropoietin induces hemeoxygenase-1 expression and attenuates oxidative stress. Biochem Biophys Res Commun. 2007;359:928–934. doi: 10.1016/j.bbrc.2007.05.207. [DOI] [PubMed] [Google Scholar]
- 27.Eto N, Wada T, Inagi R, Takano H, Shimizu A, Kato H, et al. Podocyte protection by darbepoetin: preservation of the cytoskeleton and nephrin expression. Kidney Int. 2007;72:455–463. doi: 10.1038/sj.ki.5002311. [DOI] [PubMed] [Google Scholar]
- 28.Yamamoto H, Nishi S, Tomo T, Masakane I, Saito K, Nangaku M, et al. 2015 Japanese Society for Dialysis Therapy: guidelines for renal anemia in chronic kidney disease. Ren Replace Ther. 2017;3:36. doi: 10.1186/s41100-017-0114-y. [DOI] [Google Scholar]
- 29.Solomon SD, Uno H, Lewis EF, Eckardt KU, Lin J, Burdmann EA, et al. Erythropoietic response and outcomes in kidney disease and type 2 diabetes. N Engl J Med. 2010;363:1146–1155. doi: 10.1056/NEJMoa1005109. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.