Significance
Zika virus (ZIKV) is a neurotropic and neurovirulent arbovirus that has severe detrimental impact on the developing human fetal brain. We used a genome-wide CRISPR-Cas9 knockout screen to identify ZIKV host genes in human neural progenitors. The screen identified host factors involved in heparan sulfation, endocytosis, endoplasmic reticulum processing, Golgi function, and interferon activity. Our findings provide insights into host-dependent mechanisms for ZIKV infection in the highly vulnerable human neural progenitor cells and identify molecular targets for potential therapeutic intervention.
Keywords: Zika virus, neural progenitors, CRISPR screen, fetal CNS infection, human pluripotent stem cells
Abstract
Zika virus (ZIKV) is a neurotropic and neurovirulent arbovirus that has severe detrimental impact on the developing human fetal brain. To date, little is known about the factors required for ZIKV infection of human neural cells. We identified ZIKV host genes in human pluripotent stem cell (hPSC)-derived neural progenitors (NPs) using a genome-wide CRISPR-Cas9 knockout screen. Mutations of host factors involved in heparan sulfation, endocytosis, endoplasmic reticulum processing, Golgi function, and interferon activity conferred resistance to infection with the Uganda strain of ZIKV and a more recent North American isolate. Host genes essential for ZIKV replication identified in human NPs also provided a low level of protection against ZIKV in isogenic human astrocytes. Our findings provide insights into host-dependent mechanisms for ZIKV infection in the highly vulnerable human NP cells and identify molecular targets for potential therapeutic intervention.
The recent outbreaks of Zika virus (ZIKV) in French Polynesia, the Caribbean, and Central and South America have been associated with a significant incidence of central and peripheral neuropathy (1, 2). Of particular concern are the severe fetal brain abnormalities such as microcephaly, lissencephaly, and cortical calcification after maternal infection during early pregnancy (3, 4). Accumulating evidence (in vitro and in vivo) demonstrates that human neural progenitors (NPs) are particularly sensitive to ZIKV infection (5, 6). Exposure to even a low level of ZIKV can result in robust infection of NPs, intracellular buildup of viral proteins, and extracellular release of functional virions, and ultimately lead to reduced differentiation and cell death (6, 7). This finding is in contrast to other central nervous system (CNS) cell types, including neurons, astrocytes, and microglia, which are all infectible by ZIKV but do not display a level of vulnerability comparable to NPs (5, 8). We and others have recently reported that intrinsic differences between NPs and other neural cell types in interferon (IFN) activity (8), RNA binding protein level (9), and signaling pathway dependence (10) may contribute to their enhanced vulnerability.
Unbiased forward genetic screens have proved powerful in identifying the cellular factors present in the host cells involved in viral propagation (11, 12). These host factors are often beneficial for viral entry, replication, packaging, and exit, or are involved in silencing host antiviral defense mechanisms. Genetic screens have been used to identify putative host factors for flaviviruses including dengue virus (13, 14), ZIKV (13), and West Nile virus (15). These studies used cancer cells such as kidney-derived HEK293, cervical HeLa cells, hepatic Huh7, and myeloid Hap1 and showed that some common pathways mediate the infection by different subtypes of flaviviruses. However, different flaviviruses vary significantly in their cellular tropism and manifest vastly different clinical pathology in patients. To date, ZIKV is the only example of a flavivirus that causes loss of proliferation and cell death in human NPs during early cortical development, which in turn results in fetal brain abnormality. Therefore, a better understanding of the host factors required for ZIKV infection in human NPs is needed to elucidate the mechanism of neural vulnerability and to develop future treatment options.
To date, the only genome-wide screen against ZIKV used culture-adapted cancer cell lines (13). Although that study identified genes important for ZIKV replication, it is possible that key genes involved in virus replication in the relevant cell types of the CNS were not expressed in the cancer cells and that viral host genes identified in cancer cells were not active in the CNS target cell types. In the present study, we performed a genome-wide CRISPR knockout screen in human NPs to identify host factors that, when mutated, confer resistance to ZIKV-induced cell death. Genes encoding proteins involved in heparan sulfation, endocytosis, endoplasmic reticulum (ER) protein processing, Golgi function, and IFN regulation were identified as putative host factors for ZIKV strains MR766 and PRVABC59. Pharmacological manipulation further validated the involvement of these pathways in mediating ZIKV infection in human NPs. Host factors were identified for ZIKV infection in NPs that were not previously reported in cancer cells. Furthermore, we show that the host factors identified in NPs partially mediate ZIKV infection in isogenic human astrocytes. These results emphasize the need to study viral mechanisms in biologically relevant cells. Our findings define host-dependent mechanisms that are essential for ZIKV propagation in the highly vulnerable human neural cells and identify molecular targets for potential therapeutic intervention.
Results
CRISPR-Cas9 Screens for ZIKV Host Factors in Human NPs.
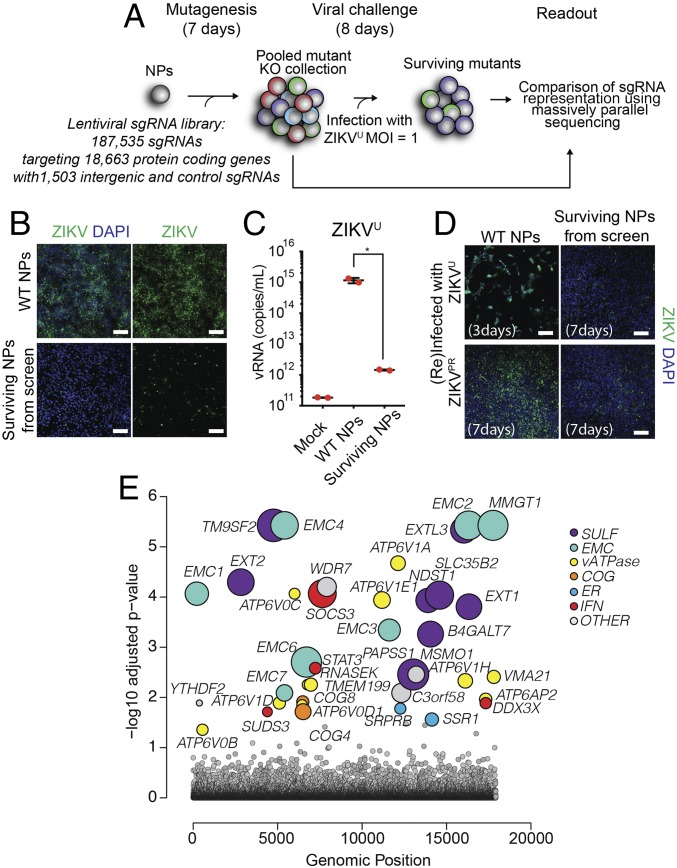
Human induced pluripotent stem cells (iPSCs)-derived NPs are highly vulnerable to ZIKV infection (6). We and others have shown that the prototypic ZIKV Uganda strain MR766 (ZIKVU) efficiently infects human NPs and leads to rapid cell death in 2D monocultures and 3D brain organoids (6, 8, 16). To identify host genes required for ZIKVU-mediated toxicity, we conducted a genome-wide CRISPR knockout screen. We used a pooled lentiviral library encoding Cas9 and 187,535 single guide RNAs (sgRNAs) targeting 18,663 protein-coding human genes that included 1,503 intergenic-targeted and nontargeting control sgRNAs (Fig. 1A). NPs derived from a wild-type human iPSC line (iPS-wt5) were transduced with the library and selected using puromycin (SI Appendix, Fig. S1A). These cells were then inoculated with ZIKVU at a multiplicity of infection of 1. While ZIKVU infection led to drastic death of the lentiviral library-carrying NPs starting from 48 h postinoculation, a small population was protected and survived. We confirmed that surviving NPs had substantially reduced ZIKV protein (Fig. 1B and SI Appendix, Fig. S1B) and viral RNA release (Fig. 1C) compared with ZIKV-infected wild-type NPs. Surviving NPs further showed reduced reinfection by both the initially screened MR766 strain (ZIKVU) as well as PRVABC59, an epidemic North American strain recently isolated in Puerto Rico (ZIKVPR) (17), as assessed by ZIKV protein staining 7 d postreinfection (Fig. 1D). High-throughput sequencing libraries were prepared from genomic DNA collected from surviving cells at day 8 and from an initial reference sample. The change in representation of each sgRNA in the population after ZIKVU exposure was calculated, and sgRNAs enriched in the surviving cells were identified. We found that knockouts of genes encoding proteins involved in heparan sulfation, endosome–lysosome acidification, ER protein complexes regulating translocation and proteostasis, Golgi complexes regulating glycosylation, and negative regulators of IFN response were protective (Fig. 1E and SI Appendix, Table S1). TP53, a gene that when knocked out confers growth advantage (18, 19), was also identified.
Fig. 1.
Genome-wide screen for ZIKVU resistance in human iPSC-derived NPs. (A) Schematic diagram of CRISPR screen with a genome-wide library in iPS-wt5 NPs. (B and C) Surviving NPs displayed minimal level of ZIKV protein (B) and greatly diminished viral RNA (vRNA) release into the medium (C). Control NPs were infected with multiplicity of infection (MOI) 1 ZIKVU and collected at 48 h (B) or 72 h (C) postinfection. (Scale bars in B: 100 μm.) (D) Surviving NPs rechallenged with MOI 1 ZIKVU and ZIKVPR displayed low ZIKV infectivity at 7 d postinfection. Control NPs infected with ZIKVU and ZIKVPR were collected at 3 d and 7 d postinfection, respectively. (Scale bars in D: 100 μm.) (E) Six main pathways were identified as protective against ZIKVU infection, upon gene knockouts. The size of the circles represents the magnitude of the increase in sgRNA representation after ZIKVU exposure. *P < 0.05.
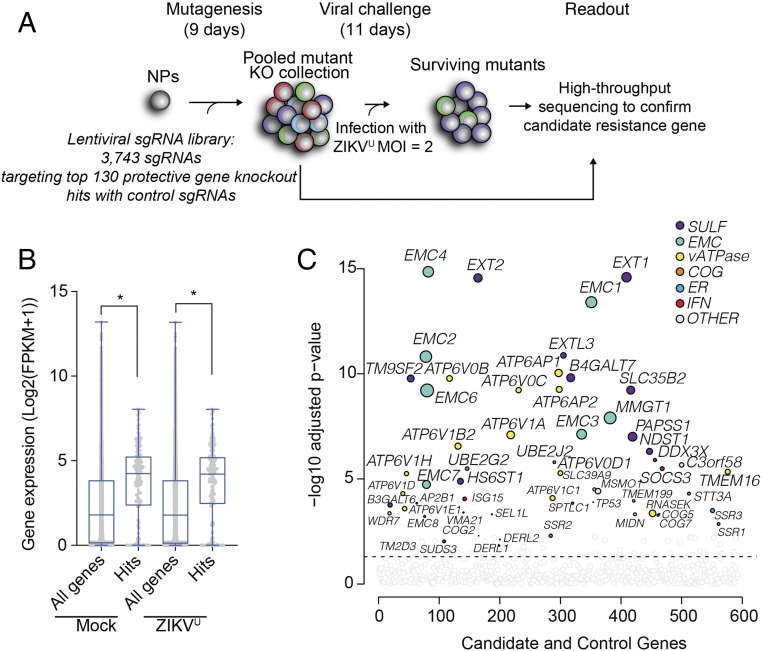
To further validate the candidates from the genome-wide screen, we next performed a focused screen using a custom library containing sgRNAs against candidate gene knockout hits (Fig. 2A). We started by confirming that candidate genes were expressed in mock and ZIKVU-infected NPs using RNA sequencing (Fig. 2B and SI Appendix, Table S2). We next constructed a secondary lentivirus CRISPR library encoding Cas9 and 3,743 sgRNAs targeting the top ∼130 protective gene knockout hits, plus 499 to 5,191 control sgRNAs targeting intergenic regions or protein-coding genes whose loss should not be protective to ZIKVU exposure (Fig. 2A and SI Appendix, Table S3). We transduced human NPs from two different genetic backgrounds, iPS-wt5 and WIBR3 hESCs, with the lentivirus library. As expected, a substantial fraction of lentivirus-transduced NPs survived ZIKVU infection (SI Appendix, Fig. S2A), and DNA was collected on day 8 or 10 after infection and sequenced. These secondary screens confirmed that top hits from the genome-wide screen were modifiers of ZIKVU toxicity in human NPs (Fig. 2C and SI Appendix, Fig. S2B).
Fig. 2.
Secondary CRISPR knockout screen in human NPs against ZIKVU. (A) Schematic diagram of CRISPR screen with a focused library in iPS-wt5 NPs. (B) Focused library contained gene hits with confirmed expression in human NPs on the baseline and 24 h after multiplicity of infection (MOI) 1 ZIKVU infection. (C) Focused screen in iPS-wt5 NPs identified most of the hits previously identified in the genome-wide screen. The size of the colored circles represents the increase in sgRNA representation after ZIKVU exposure. FPKM, fragments per kilobase of exon model per million reads mapped. *P < 0.05.
Multiple Pathways Mediate ZIKV Infectivity in Human NPs.
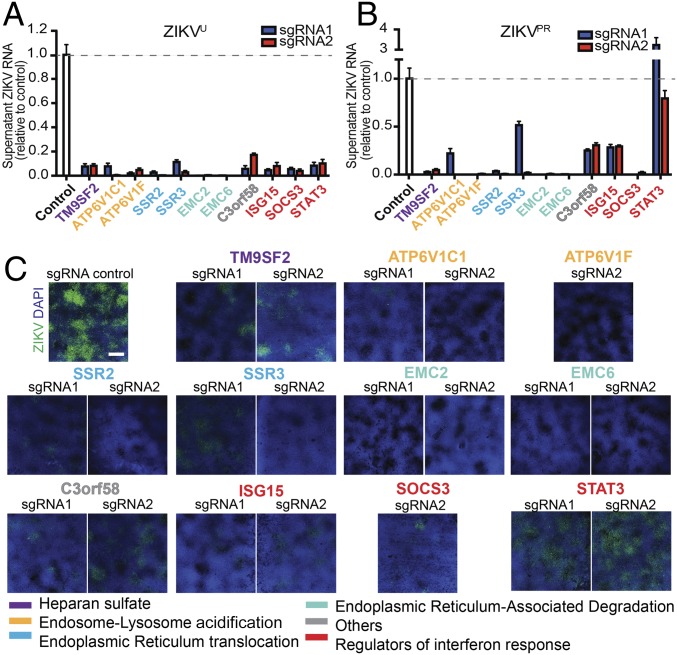
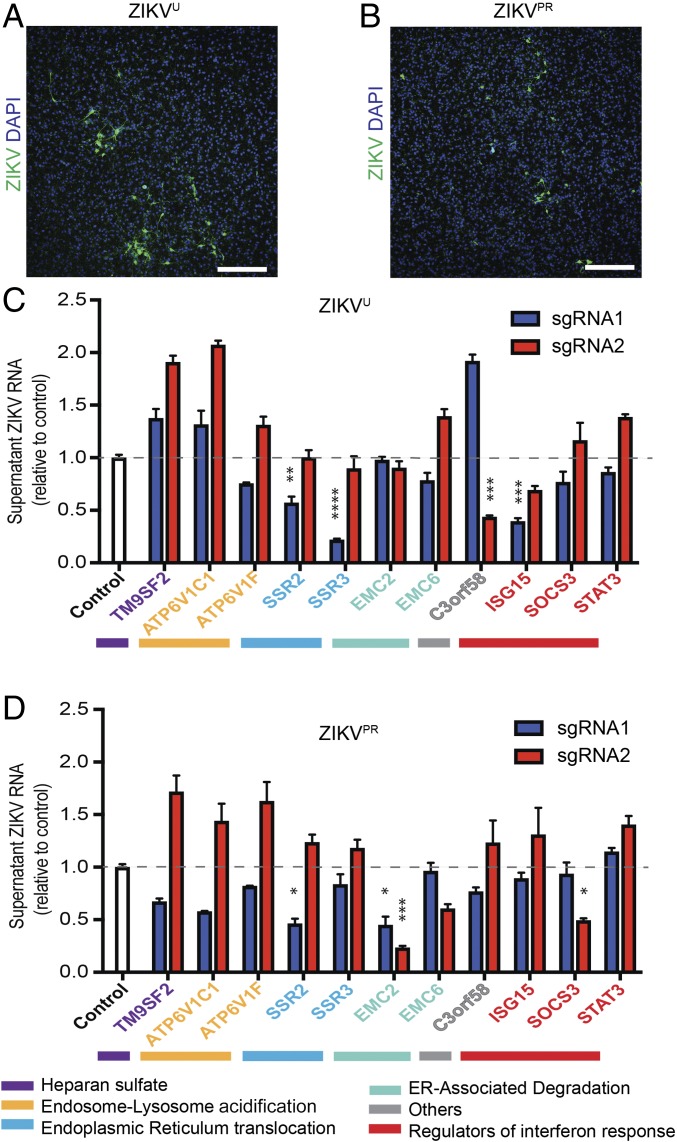
We targeted 11 host genes in iPS-wt5 NPs, using two independent sgRNAs targeting different regions of each gene. These genes were selected to represent the main functional categories identified through the pooled CRISPR screens. We confirmed that targeting of all 11 genes significantly reduced ZIKVU infection as measured by viral RNA released into the medium (Fig. 3A) and viral protein levels in the NPs (SI Appendix, Fig. S3). We further tested the effects of targeting these host genes on infection by ZIKVPR. Human NPs targeted for 10 of the 11 genes showed significantly less viral RNA release (Fig. 3B). Immunostaining for ZIKV envelope protein further confirmed reduced ZIKVPR infection in NPs (Fig. 3C). NPs targeted for STAT3, a regulator of IFN response, showed reduced infection by ZIKVU, but not by ZIKVPR (Fig. 3 and SI Appendix, Fig. S3). These findings demonstrate that ZIKV propagation depends on multiple pathways in human NPs.
Fig. 3.
Validation of ZIKV host factors with individual sgRNAs in human NPs. (A and B) Individually targeted iPS-wt5 NPs infected with multiplicity of infection (MOI) 0.5 ZIKVU (A) and ZIKVPR (B) showed reduced viral RNA released into the supernatant at 72 h postinfection, compared with ZIKV-infected control NPs. All experimental sgRNA results were significant (P < 0.001) compared with control sgRNA, except sgRNA-2 against STAT3 infected with ZIKVPR. (C) Immunostaining for ZIKV envelope (green) and DAPI (blue) showed reduced infection by MOI 0.5 ZIKVPR in targeted human NPs, 96 h postinfection. (Scale bar: 1,000 μm.) Three technical replicates. Results are mean ± SEM.
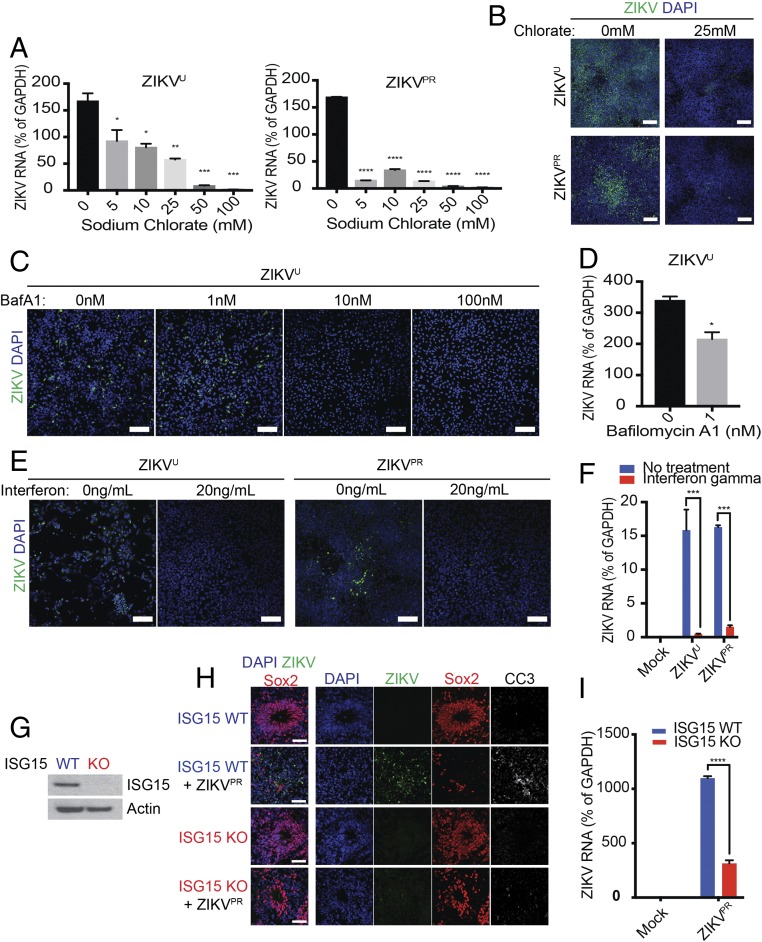
We investigated whether pharmacological modulation of these ZIKV host-factor pathways could reduce viral infection in NPs. We first examined the heparan sulfation pathway. Treatment of NPs with sodium chlorate, an inhibitor of sulfation, strongly inhibited infection by ZIKVU and ZIKVPR, as measured by viral RNA load and envelope protein staining (Fig. 4 A and B).
Fig. 4.
Validation of host pathways using pharmacological compounds and in gene-edited brain organoids. (A and B) Depletion of cellular heparan sulfate with sodium chlorate reduced viral RNA load (A) and envelope protein staining (B) upon ZIKVU and ZIKVPR exposure. iPS-wt5 NPs were treated with multiplicity of infection (MOI) 0.1 ZIKVU or MOI 1 ZIKVPR and collected at 48 h or 72 h postinfection, respectively. (Scale bars in B: 100 μm.) (C and D) Blockade of acidification with bafilomycin A1 (BafA1) decreased ZIKVU viral envelope protein staining. Low concentration (1 nM) bafilomycin A1 was sufficient to reduce ZIKVU infection as quantified by viral RNA load (D). NPs were infected with MOI 0.1 ZIKVU and collected at 24 h (C) or 48 h (D) postinfection. (Scale bars in C: 100 μm.) (E and F) NPs Pretreated with IFN-γ showed reduced infection by ZIKVU and ZIKVPR, as observed by viral RNA load (E) and ZIKV envelope protein staining (F). NPs were infected with MOI 1 ZIKVU and ZIKVPR and collected at 48 h (E) or 72 h (F) postinfection. (Scale bars in E: 100 μm.) (G–I) ISG15 mutant WIBR3-derived brain organoids lacked ISG15 protein (G) and showed reduced ZIKVPR infection, as shown by ZIKV protein (H) and viral RNA load (I). (Scale bars in H: 100 μm.) Two technical replicates. Results are mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001.
We noted that many genes involved in vacuolar acidification were enriched in the modifier genes, including RNASEK, VMA21, and 12 genes encoding subunits of vacuolar ATPase (V-ATPase). While some of these genes were previously associated with flavivirus infection (13), most were not identified in genome-wide screens performed on immortalized cell lines (14, 15). Indeed, many of these genes belong to the group of core fitness genes for human cancer cells (20, 21). To investigate the role of the endosome–lysosome pathway in mediating ZIKV infection of human NPs, we treated infected cells with bafilomycin A1, a specific inhibitor for vacuolar-type proton pumps. Treatment at 10 and 100 nM strongly protected NPs against ZIKVU infection (Fig. 4C). A significant protective effect was observed, even with a low concentration (1 nM) of bafilomycin A1 (Fig. 4D).
We have recently reported that low baseline IFN activity in NPs contributes to their high susceptibility to ZIKV infection (8). In the current genetic screen, we identified negative regulators of the IFN pathway such as ISG15 and SOCS3. Fig. 4 E and F shows that a 2-h treatment with IFN-γ before viral exposure was effective in reducing infection by ZIKVU and ZIKVPR. Mutations in one of these host genes, ISG15, has been linked to autoinflammatory disease and susceptibility to mycobacterial exposure (22, 23), consistent with these genes playing general role in controlling infection. Using CRISPR-Cas9–mediated gene editing, we generated human PSCs that lack ISG15 (Fig. 4G and SI Appendix, Fig. S4), which were then used to generate cerebral organoids. Organoids were exposed to ZIKVPR on day 30 of formation and examined 12 d after infection. We found that ZIKVPR efficiently infected control cerebral organoids, led to elevated apoptosis in Sox2+ NPs, and resulted in disruption of ventricle-like structures (Fig. 4 H and I). In contrast, ISG15 mutant organoids showed reduced viral infection and preserved Sox2+ NPs and maintained their ventricle-like structures (Fig. 4 H and I). Our results are consistent with the IFN pathway being critical for regulating ZIKV infection in human NPs.
Host Genes Identified in NPs Play Some Role During ZIKV Infection of Astrocytes.
In addition to NPs, other CNS cell types are also susceptible to ZIKV infection, including astrocytes, microglia, and neurons (8). Because astrocytes and NPs are both proliferative CNS cell types and closely related in lineage, we investigated whether mutation of host genes that reduced ZIKV infection in NPs could also protect astrocytes. Isogenic human astrocytes were differentiated from iPS-wt5 NPs and shown to express canonical markers of the astrocytic lineage and be infectible by ZIKVU and ZIKVPR (Fig. 5 A and B and SI Appendix, Fig. S5 A and B). We targeted the same 11 modifier genes and measured the susceptibility of the targeted astrocytes to ZIKV infection. We found that targeting of some of the modifier genes (such as EMC2, SSR2, SSR3, ISG15, and SOCS3) in astrocytes showed reduced infection by ZIKVU or ZIKVPR (Fig. 5 C and D and SI Appendix, Fig. S5 C and D). This suggests that both common and divergent signaling pathways may mediate ZIKV infection in these related neural cell types.
Fig. 5.
Investigation of the role of ZIKV host factors in human astrocytes. (A and B) Human astrocytes derived from iPS-wt5 were susceptible to ZIKVU (A) and ZIKVPR (B) infection. Astrocytes were infected with multiplicity of infection (MOI) 0.1 ZIKVU or MOI 0.6 ZIKVPR, and collected at 48 h. (Scale bars: 1,000 μm.) (C and D) Astrocytes targeted with individual sgRNAs show varying degrees of infection by ZIKVU (C) and ZIKVPR (D), as measured by viral RNA release into the medium. Astrocytes were infected with MOI 1 ZIKVU or ZIKVPR and collected at 72 h. Experimental sgRNA results that showed significant reduction compared with control sgRNA were noted. Two technical replicates. Results are mean ± SEM. *P < 0.05, ***P < 0.001, **P < 0.01, ****P < 0.0001.
Discussion
Selective vulnerability is a phenomenon often observed in neurodegenerative disorders. Subpopulations of progenitors, neurons, and glial cells can display increased susceptibility to certain genetic perturbations, environmental insults, and traumatic injuries. This leads to impairment of the development and function of the nervous system and can have dire consequences. Bacterial and viral infections of the CNS are often limited by the blood–brain barrier (BBB), but those that breach the BBB can lead to severe neuropathy, especially when infection occurs during early fetal development. Devastating consequences are often observed after fetal infection by a group of infectious agents (toxoplasma, other agents, rubella, cytomegalovirus, and herpes simplex) collectively known as TORCH. Historically, infection by flaviviruses during pregnancy has not been linked with fetal abnormalities (1). However, the emergence of ZIKV-related fetal brain malformations highlights the need to better understand the mechanism of selective neural vulnerability. Using an isogenic human PSC-derived in vitro system, we have recently reported that ZIKV and dengue virus are capable of infecting multiple cell types of the CNS and produce vastly different responses and cytopathic outcomes (8). While human NPs, neurons, astrocytes, microglia, and endothelial cells are all infectible by ZIKV, NPs display significantly higher levels of cytopathology and cell death. Compared with astrocytes and microglia, NPs have low baseline IFN activity and display muted IFN response upon ZIKV infection, likely contributing to their vulnerability (8). It has also been reported that upon infection, RNA and protein components of ZIKV sequester key proteins that maintain neural stem and progenitor identity (9) and interfere with the strength of the growth factor signaling pathway important for proliferation (10). One of these pathogen–host interactions was recently identified to be between ZIKV NS4A and ANKLE2 (24), which has previously been linked to primary microcephaly (25). These findings suggest that vulnerability of human NPs to ZIKV is multifaceted.
In the present study, we performed a systematic evaluation of ZIKV host factors in human NPs. We found that ZIKV utilizes multiple host cellular machineries to potentially facilitate entry (heparan sulfation, endocytosis); translation and replication of the viral genome (ER); assembly of virion; and egress from host (Golgi apparatus). Our results show that ZIKV relies on negative regulators of the IFN pathway to evade host antiviral defense. While some of these mechanisms have previously been identified in genomic screens on cancer cells against other flaviviruses, the active suppression of IFN activity was not seen in these systems. Interestingly, one of the suppressors of IFN activity is ISG15, a gene linked to an inherited form of autoinflammation reminiscent of Aicardi–Goutières syndrome (22, 23). Patients with ISG15 loss-of-function mutations have mycobacterial disease but no increased susceptibility to viral infection (23). Our findings suggest that lack of ISG15 in human NPs reduces ZIKV infection and has an acute protective effect. This is consistent with the protective effect of IFN treatment in NPs (8) and the finding that ISG15 mutant patient fibroblasts had enhanced antiviral protection (26). It has been recently reported that type I IFNs instigate ZIKV-mediated fetal demise in mice, while type III IFNs curb placental to fetal transmission (27, 28). Future investigation into the roles of different IFN pathways in the developing CNS may provide new insights into the mechanism of selective neural vulnerability.
Another category of ZIKV host factors differentially identified in our screen using human neural cells—compared with previous work done in cancer cell lines—is composed of components of the endosome–lysosome pathway. These putative ZIKV host factors include 12 genes encoding subunits of V-ATPase, as well as VMA21 and RNASEK, which facilitate V-ATPase assembly and function (29, 30). We validated that knockout of individual components of the V-ATPase (ATP6V1F, ATP6V1C1) reduced ZIKV infection in human NPs. This effect was recapitulated using bafilomycin A1, indicating that the canonical function of V-ATPase in vacuolar acidification is required for ZIKV infection. Our finding that the endosome–lysosome pathway is utilized by ZIKV is consistent with recent reports that the blockade of endocytosis using chloroquine reduced ZIKV infection (10, 31–33). Other recently identified protective small-molecule compounds against ZIKV infection have also been shown to interfere with this pathway (34, 35). We reason that many of these genes were not identified in screens using cancer cell lines because they are considered essential for cancer cells and their ablations disrupt cellular fitness (20, 21). This finding further highlights the value of investigating viral mechanism in biologically relevant cell types.
By comparing hits from our screens in NPs to previous studies in cell lines, we also highlight mechanisms that may be protective in other cell types but fail to be so in human NPs. For example, the receptor tyrosine kinase AXL was previously identified as an essential entry factor for ZIKV in human fibroblasts (36) and has been suggested to play a similar role in human neural stem cells based on expression pattern (37). However, genetic ablation of AXL was recently found to have no effect on ZIKV infection in mice (38) or in human NPs (39). AXL and other members of the receptor tyrosine kinase subfamily (TYRO3, MERTK) were not identified as protective against ZIKV in the current screens. This result is consistent with the notion that divergent entry mechanisms may be utilized by ZIKV on different cell types. The identification of genes in the heparan sulfation and endosome–lysosome pathways in the current screen supports their potential roles as attachment and entry mechanisms for ZIKV. Because different cell types may rely on different fitness genes, the current screens would be unable to identify host factors that are important for other cell types, if they are essential for the fitness of NPs. This may explain the identification of some (STT3A), but not other, members of the oligosaccharyltransferase complex as host factors in NPs. We also note that NPs used in the current study most closely resemble in vivo stem and progenitor cells from the developing dorsal forebrain. Because fetal exposure to ZIKV has been linked to structural abnormality in many brain regions, including forebrain, midbrain, and hindbrain (3, 4), future studies using NPs bearing midbrain and hindbrain regional identity could provide further insights into cell type-specific viral–host interactions. Lastly, the current study was conducted in two hPSC lines. In future studies, this effort could be expanded to more systematically analyze the genetic and environmental variabilities that impose protection or vulnerability against ZIKV infection, shedding new light on the mechanism of viral–host interaction and potential therapeutic strategies.
While ZIKV infection during early pregnancy is correlated with overt brain malformation such as microcephaly and lissencephaly, later infection is also linked to neural structural and functional abnormalities (1, 2). The diverse tropism of ZIKV toward CNS cell types highlights its potent ability to cause harm. During human fetal brain development, as neurons and glia emerge from NPs, or as microglia migrate into the brain, these cell types are susceptible to ZIKV infection. Evidence suggests that neuronal function can be impaired by ZIKV (40), and glial cells that are less acutely impacted could serve as viral reservoirs (8, 41). In the current study, we investigated whether ZIKV host factors in NPs also mediate infection of astrocytes. We used CRISPR-Cas9 to target the putative host factors in astrocytes. Compared with NPs, astrocytes divide less frequently and may therefore display a slower rate of protein depletion. Despite this possible limitation of incomplete protein depletion, we found that a subset of the factors that protected NPs against ZIKV infection also protected astrocytes. Future therapeutic strategies exploring these shared host factors may confer broad neural protection.
Materials and Methods
For a complete description of the materials and methods used in this study, please refer to SI Appendix, Materials and Methods. The human iPSC line iPS-wt5 and the hESC line WIBR3 were previously described (42, 43). ZIKV strains MR766 (ZIKVU) and PRVABC59 (ZIKVPR) were obtained from American Type Culture Collection and were expanded in Vero cells or in C6/36 mosquito cells.
Supplementary Material
Acknowledgments
We thank Tim Wang for substantial input on the design, execution, and interpretation of primary and secondary CRISPR screens and RNA sequencing experiments; Dongdong Fu and Raaji Alagappan for technical support; all members of the R.J., L.G., and D.M.S. laboratories for helpful discussions; and Alexsia Richards for critical reading of this manuscript. Confocal microscopy was performed at the Whitehead Institute W. M. Keck Biological Imaging Facility. RNA sequencing and CRISPR screen sequencing was performed by the Whitehead Institute Genome Technology Core. Y.L. received funding from a Simons Postdoctoral Fellowship, an International Rett Syndrome Foundation Postdoctoral Fellowship, and a National Alliance for Research on Schizophrenia and Depression (NARSAD) Young Investigator Grant. This research was part of the University of Toronto’s Medicine by Design initiative, which received funding from the Canada First Research Excellence Fund. J.M. received funding from the European Leukodystrophies Association, a NARSAD Young Investigator Grant, and the Canada Research Chairs program. A.O.J. was supported by a Jerome and Florence Brill Graduate Student Fellowship and Boehringer Ingelheim Fonds. Work for this project was supported by NIH Grants R01 MH104610 and R01 NS088538 (to R.J.), U19 AI131135 (to R.J., L.G., and D.M.S.), and R33 AI100190 (to L.G.); the Simons Foundation (SFARI 204106); the Emerald Foundation; and Biogen. D.M.S. is an investigator of the Howard Hughes Medical Institute and an American Cancer Society Research Professor.
Footnotes
Conflict of interest statement: R.J. is a cofounder of Fate Therapeutics, Fulcrum Therapeutics and Omega Therapeutics. I.B. and L.G. are cofounders of E25Bio, Inc.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1900867116/-/DCSupplemental.
References
- 1.Li H, Saucedo-Cuevas L, Shresta S, Gleeson JG. The neurobiology of Zika virus. Neuron. 2016;92:949–958. doi: 10.1016/j.neuron.2016.11.031. [DOI] [PubMed] [Google Scholar]
- 2.Cao-Lormeau VM, et al. Guillain-Barré syndrome outbreak associated with Zika virus infection in French Polynesia: A case-control study. Lancet. 2016;387:1531–1539. doi: 10.1016/S0140-6736(16)00562-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Martines RB, et al. Pathology of congenital Zika syndrome in Brazil: A case series. Lancet. 2016;388:898–904. doi: 10.1016/S0140-6736(16)30883-2. [DOI] [PubMed] [Google Scholar]
- 4.Mlakar J, et al. Zika virus associated with microcephaly. N Engl J Med. 2016;374:951–958. doi: 10.1056/NEJMoa1600651. [DOI] [PubMed] [Google Scholar]
- 5.Retallack H, et al. Zika virus cell tropism in the developing human brain and inhibition by azithromycin. Proc Natl Acad Sci USA. 2016;113:14408–14413. doi: 10.1073/pnas.1618029113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tang H, et al. Zika virus infects human cortical neural progenitors and attenuates their growth. Cell Stem Cell. 2016;18:587–590. doi: 10.1016/j.stem.2016.02.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Garcez PP, et al. Zika virus impairs growth in human neurospheres and brain organoids. Science. 2016;352:816–818. doi: 10.1126/science.aaf6116. [DOI] [PubMed] [Google Scholar]
- 8.Muffat J, et al. Human induced pluripotent stem cell-derived glial cells and neural progenitors display divergent responses to Zika and dengue infections. Proc Natl Acad Sci USA. 2018;115:7117–7122. doi: 10.1073/pnas.1719266115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chavali PL, et al. Neurodevelopmental protein Musashi-1 interacts with the Zika genome and promotes viral replication. Science. 2017;357:83–88. doi: 10.1126/science.aam9243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Liang Q, et al. Zika virus NS4A and NS4B proteins deregulate akt-mTOR signaling in human fetal neural stem cells to inhibit neurogenesis and induce autophagy. Cell Stem Cell. 2016;19:663–671. doi: 10.1016/j.stem.2016.07.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ramage H, Cherry S. Virus-host interactions: From unbiased genetic screens to function. Annu Rev Virol. 2015;2:497–524. doi: 10.1146/annurev-virology-100114-055238. [DOI] [PubMed] [Google Scholar]
- 12.Puschnik AS, Majzoub K, Ooi YS, Carette JE. A CRISPR toolbox to study virus-host interactions. Nat Rev Microbiol. 2017;15:351–364. doi: 10.1038/nrmicro.2017.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Savidis G, et al. Identification of Zika virus and dengue virus dependency factors using functional genomics. Cell Rep. 2016;16:232–246. doi: 10.1016/j.celrep.2016.06.028. [DOI] [PubMed] [Google Scholar]
- 14.Marceau CD, et al. Genetic dissection of Flaviviridae host factors through genome-scale CRISPR screens. Nature. 2016;535:159–163. doi: 10.1038/nature18631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zhang R, et al. A CRISPR screen defines a signal peptide processing pathway required by flaviviruses. Nature. 2016;535:164–168. doi: 10.1038/nature18625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Li Y, et al. Induction of expansion and folding in human cerebral organoids. Cell Stem Cell. 2017;20:385–396.e3. doi: 10.1016/j.stem.2016.11.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lanciotti RS, Lambert AJ, Holodniy M, Saavedra S, Signor LdelC. Phylogeny of Zika virus in Western Hemisphere, 2015. Emerg Infect Dis. 2016;22:933–935. doi: 10.3201/eid2205.160065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Merkle FT, et al. Human pluripotent stem cells recurrently acquire and expand dominant negative P53 mutations. Nature. 2017;545:229–233. doi: 10.1038/nature22312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Haapaniemi E, Botla S, Persson J, Schmierer B, Taipale J. CRISPR-Cas9 genome editing induces a p53-mediated DNA damage response. Nat Med. 2018;24:927–930. doi: 10.1038/s41591-018-0049-z. [DOI] [PubMed] [Google Scholar]
- 20.Hart T, et al. High-resolution CRISPR screens reveal fitness genes and genotype-specific cancer liabilities. Cell. 2015;163:1515–1526. doi: 10.1016/j.cell.2015.11.015. [DOI] [PubMed] [Google Scholar]
- 21.Wang T, et al. Identification and characterization of essential genes in the human genome. Science. 2015;350:1096–1101. doi: 10.1126/science.aac7041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhang X, et al. Human intracellular ISG15 prevents interferon-α/β over-amplification and auto-inflammation. Nature. 2015;517:89–93. doi: 10.1038/nature13801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bogunovic D, et al. Mycobacterial disease and impaired IFN-γ immunity in humans with inherited ISG15 deficiency. Science. 2012;337:1684–1688. doi: 10.1126/science.1224026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Shah PS, et al. Comparative flavivirus-host protein interaction mapping reveals mechanisms of dengue and Zika virus pathogenesis. Cell. 2018;175:1931–1945.e18. doi: 10.1016/j.cell.2018.11.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Shaheen R, et al. Genomic and phenotypic delineation of congenital microcephaly. Genet Med. 2018;21:545–552. doi: 10.1038/s41436-018-0140-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Speer SD, et al. ISG15 deficiency and increased viral resistance in humans but not mice. Nat Commun. 2016;7:11496. doi: 10.1038/ncomms11496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Yockey LJ, et al. Type I interferons instigate fetal demise after Zika virus infection. Sci Immunol. 2018;3:eaao1680. doi: 10.1126/sciimmunol.aao1680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Jagger BW, et al. Gestational stage and IFN-lambda signaling regulate ZIKV infection in utero. Cell Host Microbe. 2017;22:366–376.e3. doi: 10.1016/j.chom.2017.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ramachandran N, et al. VMA21 deficiency prevents vacuolar ATPase assembly and causes autophagic vacuolar myopathy. Acta Neuropathol. 2013;125:439–457. doi: 10.1007/s00401-012-1073-6. [DOI] [PubMed] [Google Scholar]
- 30.Perreira JM, et al. RNASEK is a V-ATPase-associated factor required for endocytosis and the replication of rhinovirus, influenza A virus, and dengue virus. Cell Rep. 2015;12:850–863. doi: 10.1016/j.celrep.2015.06.076. [DOI] [PubMed] [Google Scholar]
- 31.Delvecchio R, et al. Chloroquine, an endocytosis blocking agent, inhibits Zika virus infection in different cell models. Viruses. 2016;8:E322. doi: 10.3390/v8120322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Li C, et al. Chloroquine, a FDA-approved drug, prevents Zika virus infection and its associated congenital microcephaly in mice. EBioMedicine. 2017;24:189–194. doi: 10.1016/j.ebiom.2017.09.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Shiryaev SA, et al. Repurposing of the anti-malaria drug chloroquine for Zika virus treatment and prophylaxis. Sci Rep. 2017;7:15771. doi: 10.1038/s41598-017-15467-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Xu M, et al. Identification of small-molecule inhibitors of Zika virus infection and induced neural cell death via a drug repurposing screen. Nat Med. 2016;22:1101–1107. doi: 10.1038/nm.4184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Saiz JC, Oya NJ, Blázquez AB, Escribano-Romero E, Martín-Acebes MA. Host-directed antivirals: A realistic alternative to fight Zika virus. Viruses. 2018;10:E453. doi: 10.3390/v10090453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hamel R, et al. Biology of Zika virus infection in human skin cells. J Virol. 2015;89:8880–8896. doi: 10.1128/JVI.00354-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Nowakowski TJ, et al. Expression analysis highlights AXL as a candidate Zika virus entry receptor in neural stem cells. Cell Stem Cell. 2016;18:591–596. doi: 10.1016/j.stem.2016.03.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hastings AK, et al. TAM receptors are not required for Zika virus infection in mice. Cell Rep. 2017;19:558–568. doi: 10.1016/j.celrep.2017.03.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Wells MF, et al. Genetic ablation of AXL does not protect human neural progenitor cells and cerebral organoids from Zika virus infection. Cell Stem Cell. 2016;19:703–708. doi: 10.1016/j.stem.2016.11.011. [DOI] [PubMed] [Google Scholar]
- 40.Gaburro J, et al. Zika virus-induced hyper excitation precedes death of mouse primary neuron. Virol J. 2018;15:79. doi: 10.1186/s12985-018-0989-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Mesci P, et al. Modeling neuro-immune interactions during Zika virus infection. Hum Mol Genet. 2018;27:41–52. doi: 10.1093/hmg/ddx382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Muffat J, et al. Efficient derivation of microglia-like cells from human pluripotent stem cells. Nat Med. 2016;22:1358–1367. doi: 10.1038/nm.4189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Li Y, et al. Global transcriptional and translational repression in human-embryonic-stem-cell-derived Rett syndrome neurons. Cell Stem Cell. 2013;13:446–458. doi: 10.1016/j.stem.2013.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.