Abstract
Brain aging induces neuropsychological changes, such as decreased memory capacity, language ability, and attention; and is also associated with neurodegenerative diseases. However, most of the studies on brain aging are focused on neurons, while senescence in astrocytes has received less attention. Astrocytes constitute the majority of cell types in the brain and perform various functions in the brain such as supporting brain structures, regulating blood-brain barrier permeability, transmitter uptake and regulation, and immunity modulation. Recent studies have shown that SIRT1 and SIRT2 play certain roles in cellular senescence in peripheral systems. Both SIRT1 and SIRT2 inhibitors delay tumor growth in vivo without significant general toxicity. In this study, we investigated the role of tenovin-1, an inhibitor of SIRT1 and SIRT2, on rat primary astrocytes where we observed senescence and other functional changes. Cellular senescence usually is characterized by irreversible cell cycle arrest and induces senescence-associated β-galactosidase (SA-β-gal) activity. Tenovin-1-treated astrocytes showed increased SA-β-gal-positive cell number, senescence-associated secretory phenotypes, including IL-6 and IL-1β, and cell cycle-related proteins like phospho-histone H3 and CDK2. Along with the molecular changes, tenovin-1 impaired the wound-healing activity of cultured primary astrocytes. These data suggest that tenovin-1 can induce cellular senescence in astrocytes possibly by inhibiting SIRT1 and SIRT2, which may play particular roles in brain aging and neurodegenerative conditions.
Keywords: Astrocyte, Tenovin-1, Cellular senescence, Wound healing, Senescence-associated secretory phenotype
INTRODUCTION
Brain aging can be caused by various reasons, such as DNA damage, oxidative stress, mitochondrial dysfunction, calcium homeostasis imbalance, chromatin breakdown, shortened or dysfunctional telomeres, cell cycle inhibitor expression, and oncogenic mutations (Munoz-Najar and Sedivy, 2011; Zhu et al., 2011). In this sense, the aged brain undergoes structural, chemical, neuropsychological and genetic changes. Significant structural alterations in the aged brain has been demonstrated, including decreased volume, diminished cortical thickness (Fjell et al., 2009a, 2009b) and hippocampal size (Sowell et al., 2003), increased ventricular size, and dys-regulated neural circuits and synaptic plasticity (Hof and Morrison, 2004). Chemical alterations of the aged brain include decreased dopamine synthesis (Ota et al., 2006), serotonin receptor expression (Wong et al., 1984), and glutamate concentration (Kaiser et al., 2005; Sailasuta et al., 2008). As a result, neuropsychological changes such as decreased memory capacity (Hof and Morrison, 2004), language ability (Burke and Mackay, 1997), and attention can be observed. Brain aging is associated with degenerative brain diseases such as Alzheimer’s disease, Parkinson’s disease, mild cognitive impairment, and cerebrovascular disease. With the rapid increase in the aging population and the deep linkage between aging and various neurodegenerative diseases, research focused on brain aging is gaining more attention.
Studies concerning cellular senescence have been performed for decades. Hayflick and Moorhead (1961) first introduced the term “senescence” in 1961. Cellular senescence induced various aberrations, such as shortened telomeres, DNA damage, and oncogene expression. Moreover, senescent cells are usually characterized by irreversible cell cycle arrest, enlarged size, flattened shape, induced senescence-associated β-galactosidase (SA-β-gal) activity, senescence-associated secretory phenotype (SASP), and the formation of senescence-associated heterochromatin foci (Salama et al., 2014). These senescent cell changes are related to tumor suppression, development, and aging (Baker et al., 2016). Thus, these cellular senescence features should also be evident in aged brain cells. For example, in normal brain aging and chronic age-related neurodegenerative diseases, microglia express increased pro-inflammatory cytokines (Bachstetter et al., 2011). Moreover, p16-positive neural stem cells are detected in the aging brain (Zou et al., 2012).
Astrocytes play various important roles in the brain, such as supporting brain structures during basal and traumatic conditions, blood-brain barrier regulation, transmitter uptake and regulation, and immune modulation. In this regard, it is important to note that the aged brain dysplays functional alterations that are normally regulated by astrocytes. Increased glial fibrillary acidic protein (GFAP) in astrocytes and disruption of the blood-brain barrier (BBB) are some of the commonly observed changes in the aged brain (Nichols et al., 1993; Cotrina and Nedergaard, 2002). Although the mechanism of BBB disruption in the aged brain is not clear, some studies have shown that astrocytes secrete inflammatory components that may increase BBB permeability (Abbott et al., 2006). With aging, SASPs including several interleukins (ILs), monocyte chemotactic proteins (aka CCLs), and growth-related oncogenes (aka CXCLs) accumulate in various human and mouse tissues (Chinta et al., 2015). In the aged brain, the expression of IL-6 in glial cells is higher than that in the young rat cortex, hippocampus, and striatum (Xie et al., 2003). SASPs may have potent effects on neighboring cells and systemic tissue milieus. These results suggest that brain aging is related with astrocyte senescence, yet the underlying mechanisms and relevance are still incompletely understood.
Sirtuins are a class of proteins that exhibit mono-ADP-ribosyltransferase or deacetylase activity. Sirtuin 1 (SIRT1) and sirtuin 2 (SIRT2) are NAD+-dependent histone deacetylases (HDACs) class III and are included in sirtuin class I. SIRT1 also regulates metabolism and inflammation, and physiologically deactivate the p53 protein in mammals (Saunders and Verdin, 2007). SIRT1 induces autophagy through the deacetylation of proteins as demonstrated in cultured cells and tissues. Some SIRT1 activators (SRT2104 and SRT1720) are reported to extend the lifespan and improve bone and muscle mass of mice (Mercken et al., 2014; Mitchell et al., 2014). Brain-specific SIRT1-overexpressing mice show delayed aging and lifespan-extension phenotypes that may be in association with SIRT1 activities in the central nervous system neurons (Satoh et al., 2013). EX-527, a SIRT1 inhibitor with low or no inhibitory effects on other SIRTs, increases levels of acetylated p53 in cells. However, it inhibits the deacetylation of mutant huntingtin proteins and thus, promotes the degradation of acetylated mutants by autophagy. Recently, EX-527 completed the phase 2 safety and tolerability study as a targeted drug therapy for Huntington’s disease (Kim et al., 2016). SIRT2 is involved in cell cycle and tumorigenesis, and assists in the repair of DNA. SIRT2 is transiently transported to the nucleus during the G2/M transition of the cell cycle, thereby regulating chromosomal condensation during mitosis (Vaquero et al., 2006). Finally, SIRT2-overexpressing cells exhibit marked prolongation of the cell cycle (Dryden et al., 2003). Despite the essential role of SIRT1 and SIRT2 on cellular senescence, the specific mechanisms on their role in astrocyte senescence are still unclear.
Tenovin-1 has been known for its role in inhibiting SIRT1 and SIRT2, inducing apoptosis in vitro, activating and elevating acetylated p53 level, and activating p21 and p53. Tenovin-6 is an analog of tenovin-1, which is more water-soluble than tenovin-1 and is also a small-molecule inhibitor of SIRT1 and SIRT2 in various colon cancer cell lines. Both SIRT1 and SIRT2 inhibitors delay tumor growth in vivo without significant general toxicity (Lain et al., 2008). HDAC inhibitors are known to trigger senescence through genome-wide chromatin decompression (van Deursen, 2014).
Although brain aging is related to various diseases including neurodegenerative diseases, cellular senescence studies are focused mainly on anti-cancer therapy in cancer cell lines. Moreover, studies on the aging of brain cells are mostly focused on neurons, while senescence in astrocytes has received less attention. In this study, we investigated the relationship between SIRT and astrocyte senescence using specific SIRT inibitor tenovin-1.
MATERIALS AND METHODS
Materials
The following materials were used in this study: Dulbecco’s modified Eagle medium (DMEM)/F12, Penicillin-Streptomycin (P/S), 0.25% trypsin-EDTA from Gibco BRL (Grand Island, NY, USA); 10% Fetal Bovine Serum (FBS) from Gibco BRL, and dimethyl sulfoxide from Invitrogen (Carlsbad, CA, USA); Tween® 20 and ECLTM Western blotting detection reagent from Amersham Life Science (Arlington Heights, IL, USA); tenovin-1 from Tocris bioscience (Minneapolis, MN, USA); anti-β-actin from Sigma (St. Louis, MO, USA); anti-iNOS from Abcam (Cambridgeshire, UK); anti-COX2 from Santa Cruz Biotechnology (CA, USA); anti-phospho-H3 and anti-histone H3 from Cell Signaling Technology (MA, USA); anti-CDK2 and anti-CDK4 from Santa Cruz Biotechnology; anti-GFAP from EMD Millipore (MA, USA); senescence detection kit from Abcam.
Astrocytes culture
Animal experimental procedures were carried out following protocols approved by the Institutional Animal Care and Use Committee (IACUC) of Konkuk University. Sprague-Dawley (SD) rats were purchased from Samtako Inc (Gyeonggi, Korea). Astrocytes were harvested from the brain cortex of post-natal day 2 (P2) SD-rat pup and were cultured as described previously (Cho et al., 2013; Kim et al., 2013). Concisely, brain cortices were divided and suspended into single cells by mechanical trituration. Cultured cells were seeded on poly-d-lysine (20 μg/ml) coated plate and maintained in DMEM/F12 with 100 U/ml of penicillin, 100 mg/ml of streptomycin, and 10% heat-inactivated FBS in a 95% CO2 incubator at 37°C. Cultured astrocyte medium was changed every 4 days. After 2 weeks, confluent cells were washed twice with serum-free medium and detached by 0.25% trypsin-EDTA. Isolated single cells were subcultured by replating in 6, 12, or 96-well plates. Subcultured astrocytes were positive for GFAP.
Drug treatment
Cultured astrocytes were rinsed twice with media and tenovin-1 was treated with 1, 2, and 4 μM concentrations for 24 h in serum condition. Vehicle group was treated with DMSO. In SA-β-gal staining, tenovin-1 was administered at its highest concentration (4 μM) for 24 h. In wound scratch assay, 4 μM of tenovin-1 was also administered and examined for 72 h. In these experiments, no cellular toxicity was observed.
SA-β-gal staining
SA-β-gal staining was performed using Senescence Detection Kit for the detection of senescent cells. The kit was designed for detecting SA-β-gal activity in cultured cells. Procedures were performed according to the manufacturer’s instructions (Dimri et al., 1995). Astrocytes were seeded in 12-well plates at 2.5×105 cells/ml. After 4 days, confluent cells were washed in phosphate-buffered saline (PBS) and then fixed for 15 min with 0.5 ml fixative solution at room temperature. Fixed cells were washed twice with 1 ml PBS and were incubated overnight in 0.5 ml staining solutions mix (staining solution, staining supplement, and 20 mg/ml X-gal in DMSO) at 37°C without providing CO2. The SA-β-gal-positive cells were observed with bright-field microscopy at 100× magnification.
Reverse transcription-polymerase chain reaction (RT-PCR)
IL-1β, IL-6, TNFα, iNOS, COX2, and Gapdh mRNA expression was determined in cultured astrocytes by semi-quantitative RT-PCR. RNA was extracted with TRIzol reagent (Invitrogen) and the concentration was measured using a spectrophotometer (Nanodrop Technologies, Wilmington, DE, USA). cDNA synthesis was performed using RNA and RT reaction mixture including RevertAid Reverse transcriptase, reaction buffer (Thermo Fisher Scientific, MA, USA) and dNTP (Promega, WI, USA). A total of 0.5 μl of cDNA sample was utilized for PCR amplification under the following cycle parameters: [94°C, 30 s; 60°C, 1 min; 72°C, 30 s]×30 cycles, then 72°C, 10 min for IL-1β, IL-6, TNFα, iNOS and COX2; [94°C, 30 s; 60°C, 1 min; 72°C, 30 s]×23 cycles, then 72°C, 10 min for Gapdh. Primers were designed for IL-1β (sense: 5′-AAA ATG CCT CGT GCT GTC TG-3′/ antisense: 5′-CTA TGT CCC GAC CAT TGC TG-3′), IL-6 (sense: 5′-TTG TGC AAT GGC AAT TCT GA-3′/ antisense: 5′-TGG AAG TTG GGG TAG GAA GG-3′), TNFα (sense: 5′-TAG CCC ACG TCG TAG CAA AC -3′/ antisense: 5′- GGA GGC TGA CTT TCT CCT GG-3′), iNOS (sense: 5′- CTG GCT GCC TTG TTC AGC TA-3′/ antisense: 5′-AGT GTA GCG TTT CGG GAT CT-3′), COX2 (sense: 5′-TGC ATG TGG CTG TGG ATG TCA TCA A-3′/ antisense: 5′-CAC TAA GAC AGA CCC GTC ATC TCC A-3′), and Gapdh (sense: 5′-GTG AAG GTC GGT GTG AAC GGA TTT-3′/ antisense: 5′-CAC AGT CTT CTG AGT GGC AGT GAT-3′). PCR products were then electrophoresed with 1.2% agarose gel and visualized with ethidium bromide (Sigma). PCR products were detected and measured using Image J (NIH) software program. Band intensity was normalized using Gapdh mRNA band intensity.
Western blot analysis
Cells were harvested with radioimmunoprecipitation assay buffer consisting of 2 mM EDTA, 0.1% (w/v) SDS, 50 mM Tris-HCl, 150 mM sodium chloride, 1% Triton X-100, and 1% (w/v) sodium deoxycholate. Extracted proteins from total cells were quantified using a BCA assay kit (Thermo Fisher Scientific) and boiled for 5 min at 105°C. Protein samples were separated by SDS-PAGE for 120 min at 100 V. Electrophoretically separated proteins were transferred to nitrocellulose membranes for 90 min. Blots were blocked with 1 μg/ml polyvinyl alcohol for 5 min at room temperature and were then washed with Tris-buffered saline and 0.1% Tween 20 (TBS-T). Blots were incubated overnight at 4°C with each primary antibodies (β-actin, 1:40,000; iNOS, 1:2,000; COX2, 1:2,000; p-H3, 1:2,000; histone H3, 1:2,000; CDK2, 1:2,000; CDK4, 1:2,000; GFAP, 1:2,000) diluted in TBS-T. On the next day, blots were washed 3 times and incubated with horseradish peroxidase-conjugated secondary antibody (Life Technologies, Carlsbad, CA, USA) at room temperature for 60 min. Blots were analyzed using a chemiluminescence detection system (Amersham, Buckinghamshire, UK). β-actin was used as the loading control.
Wound closure assay
For live cell analysis of wound closure assay, astrocytes at a density of 2.5×105 cells/ml were seeded on poly-D-lysine-coated 96-well plates and incubated until they reached confluence. Upon reaching confluence, we made a scratch that was 700–800 nm wide in each well by using a certified Essen Biosciences automated 96-wound-makerTM (Essen Biosciences, Hertfordshire, UK) on 96-well plates. The medium was replaced with serum. The closure of wound scratch width and relative wound density were measured using the IncuCyte ZOOM system (Essen Bioscience, MI, USA) by imaging each well every 3 h for 72 h. Wound width and relative wound density data were analyzed using the IncuCyte ZOOM microscope software 2015A (Essen Bioscience, MI, USA). Relative wound density was defined as cell density in the wound area expressed relative to the cell density outside of the wound area over time. This metric was normalized for changes in cell density by proliferation and/or pharmacological effects.
Statistical analysis
Experimental results were expressed as the mean ± SEM. Statistical comparisons were performed by t-test using GraphPad Prism 5 software (GraphPad Software Inc., CA, USA), and a value of p<0.05 was considered significant.
RESULTS
Determination of SA-β-gal-positive astrocytes in vitro
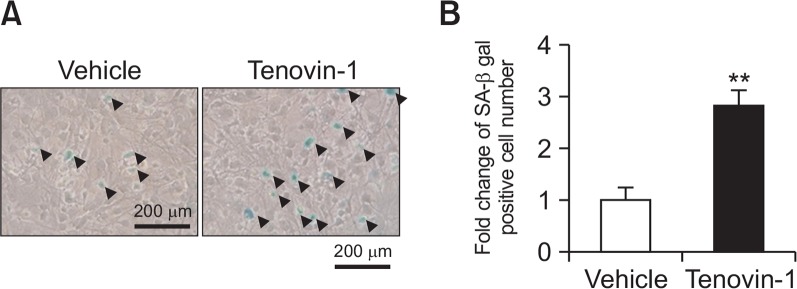
In the SA-β-gal staining experiments in cultured astrocytes, SA-β-gal-positive cells showed blue granular staining in the cytoplasm. The black triangles indicate SA-β-gal-positive cells (Fig. 1A). The astrocyte culture treated with tenovin-1 for 24 h showed about 3 times more SA-β-gal-positive cells than the vehicle-treated astrocyte culture (Fig. 1B).
Fig. 1.
Senescence-associated beta-galactosidase (SA-β-gal) staining of astrocytes in vitro. Rat primary astrocytes were treated with 4 μM tenovin-1 for 24 h. (A) Astrocytes were stained for SA-β-gal as indicated in Materials and Methods. Pictures were taken at 100× magnification. Black triangles indicate SA-β-gal-positive cells. (B) The graph represents fold-change in SA-β-gal-positive cell number in 4 μM tenovin-1-treated astrocytes. Values are expressed as the mean ± SEM. ** indicates p<0.01 vs. the vehicle-treated astrocytes.
Effects of tenovin-1 treatment on the mRNA expression of senescence-related genes in astrocytes
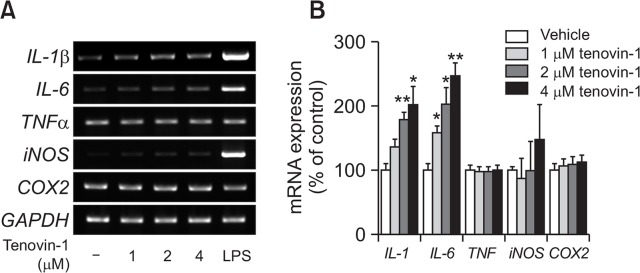
We examined the effects of tenovin-1 treatment on the mRNA expression of cell cycle-regulatory genes in astrocytes using RT-PCR analysis. Cultured rat primary astrocytes were treated with tenovin-1 for 24 h at 1, 2, and 4 μM concentrations. Astrocytes treated with 10 ng/ml of LPS were used as positive control. IL-1β mRNA expression was dose-dependently increased in tenovin-1-treated groups; the increase was significant in tenovin-1-treated astrocytes at 2 and 4 μM concentrations. It was 2-fold higher in astrocytes treated with 4 μM tenovin-1 than in the vehicle group. IL-6 mRNA expression was also dose-dependently and significantly upregulated in all tenovin-1-treated astrocytes. iNOS mRNA expression level showed an increasing tendency, albeit without a significant change. TNFα and COX2 mRNA expression level did not change, also in LPS treated astrocytes (Fig. 2A, 2B).
Fig. 2.
Senescence-related gene expression in senescent astrocytes. Rat primary astrocytes were treated with 0, 1, 2, and 4 μM tenovin-1 for 24 h. (A) Agarose gel images of mRNA expression level by RT-PCR. (B) The graph represents mRNA expression levels of markers in astrocytes. Values were normalized to GAPDH levels and are expressed as the mean ± SEM. * indicates p<0.05, ** indicates p<0.01 compared to the vehicle-treated astrocytes.
Effects of tenovin-1 treatment on levels of cell cycle marker proteins in astrocytes
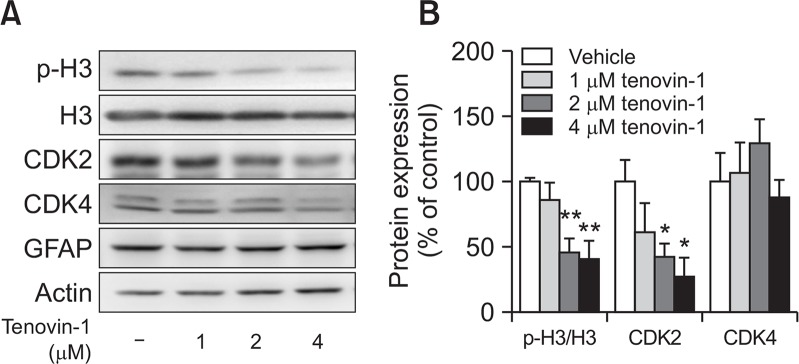
To confirm whether tenovin-1 affects the expression of cell cycle-related proteins, we evaluated the expression of phospho-histone H3, histone H3, CDK2, and CDK4 by western blot analysis. Phospho-histone H3/histone H3 and CDK2, but not CDK4, protein expression significantly decreased in 2 and 4 μM tenovin-1-treated astrocytes (Fig. 3A, 3B). Overall, the results suggest that tenovin-1 could downregulate phospho-histone H3/histone H3 and CDK2 protein expression, presumably due to cell cycle-regulation defects in astrocytes.
Fig. 3.
Senescence-related protein expression in senescent astrocytes. Rat primary astrocytes were treated with 0, 1, 2, and 4 μM tenovin-1 for 24 h. (A) Densitometry analysis of the protein expression levels of biomarkers. (B) The graph represents protein expression levels in astrocytes. Values were normalized to actin levels and are expressed as the mean ± SEM. * indicates p<0.05, ** indicates p<0.01 compared to the vehicle-treated astrocytes.
Effect of tenovin-1 treatment on wound-healing activity of astrocytes
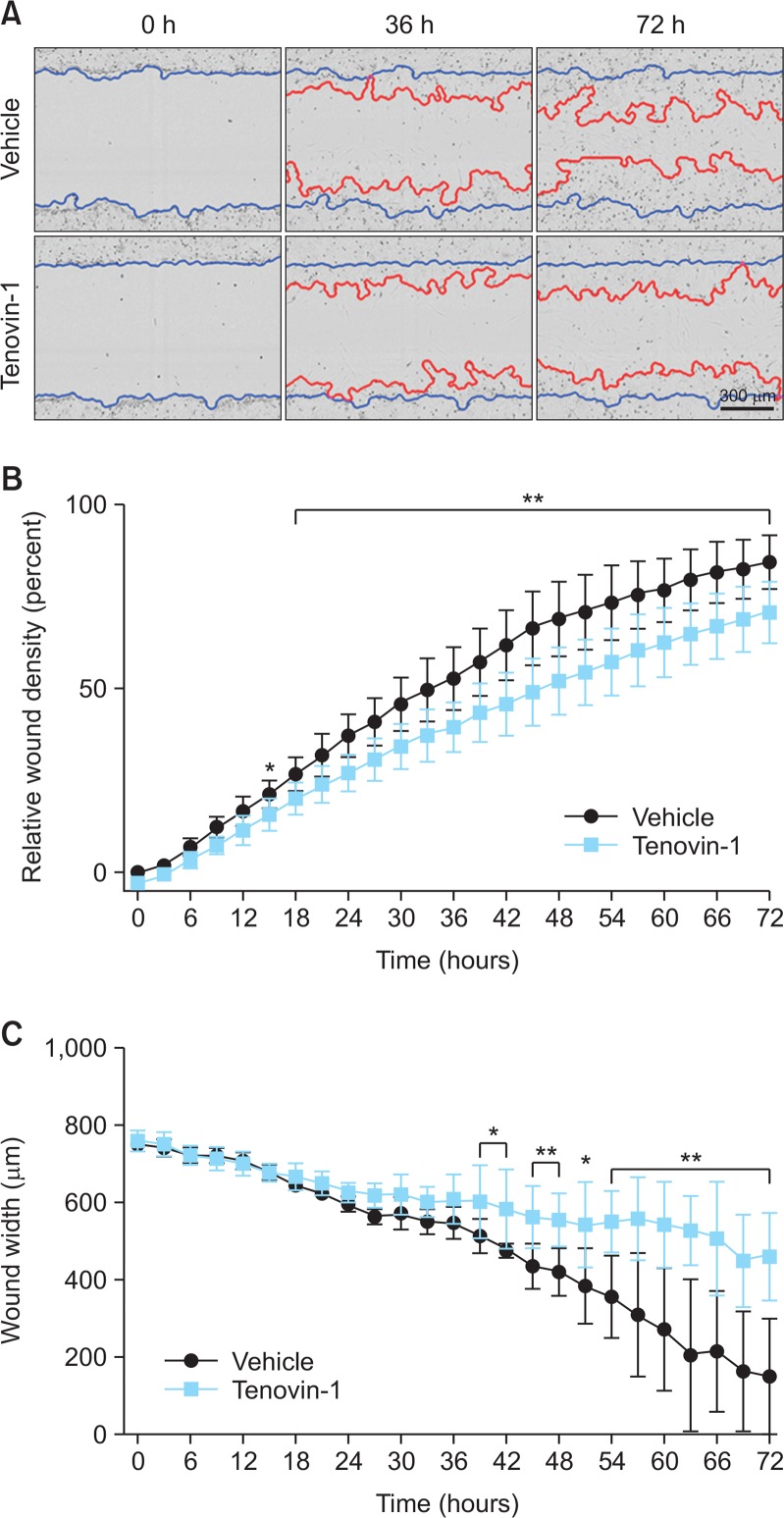
We checked the effect of tenovin-1 on wound healing in astrocytes through scratch-wound assay (Fig. 4A). Tenovin-1-treated astrocytes had lower relative wound density, which is the ratio of cell density compared to unwounded regions, than vehicle-treated astrocytes. Tenovin-1- and vehicle-treated astrocytes showed significant differences in relative wound density 15 h post-scratch and thereafter (Fig. 4B). Tenovin-1-treated astrocytes displayed slow closure of wound scratch than vehicle-treated astrocytes, and the change in wound width between the two groups was significant from 39 h post-scratch (Fig. 4C). These changes increased time-dependently.
Fig. 4.
Effects of tenovin-1 on astrocyte migration into the scratch wound. The monolayer of rat primary astrocytes was scratched and the cells were treated with 0 and 4 μM tenovin-1. (A) Representative images of cells in the wound-healing assay. Blue lines show initial scratch-wound area. Red lines indicates wound area at each timepoint, calculated by IncuCyte software. Images and data obtained using IncuCyte under the 10× objective. (B) The graph represents the percentage of relative wound density in the scratch-wound area every 3 h. (C) Representative graph of cell migration into the wound area shows wound width (μm) in the modified scratch-wound assay analysis at each 3-h timepoint. Values are expressed as the mean ± SEM. * indicates p<0.05, ** indicates p<0.01 compared to the vehicle-treated astrocytes.
DISCUSSION
Astrocytes perform various important functions in the brain and occupy a larger portion than neurons. Astrocytes play an active role in the neuron–neuroglia crosstalk, maintaining metabolic and ion homeostasis in neurons (Verkhratsky, 2010). In addition, astrocytes modulate the growth, signal regulation, and synaptic plasticity of neurons. In the brain, astrocytes interact with the capillary endothelial cells and regulate BBB permeability (Abbott et al., 2006). BBB permeability was shown to increase with aging (Popescu et al., 2009), and reduction of BBB function by aging leads to cognitive decline and dementia (Zeevi et al., 2010). Nevertheless, astrocytes have received less attention than neurons in aging-related studies.
In this study, we observed a senescent astrocyte phenotype in vitro using tenovin-1 treatment. Well-known features of senescent cells include increased SA-β-gal activity and SASP and decreased expression of cell cycle-related factors. To identify the individual senescent cells in vitro and in vivo, we employed SA-β gal as a biomarker (Geng et al., 2010; Pospelova et al., 2013). SA-β-gal-positive cells can be easily determined by counting the number of blue-stained cells in the cytosol (Debacq-Chainiaux et al., 2009). Although its expression is not definitive for senescence (Lee et al., 2006), it remains the most widely used biomarker for determining senescent and aging cells because it is easy to detect in both in vivo and in vitro conditions. Moreover, an increase in SA-β-gal activity in senescent cells increases the abundance of lysosomal enzymes (Kurz et al., 2000).
Senescent cells release cytokines, chemokines, and growth factors, collectively called SASP, which includes IL-6, IL-8, MMP3, IGFBP3, IGFBP5, IGFBP7, PAI-1, and CXCL (Kuilman and Peeper, 2009; Elkhattouti et al., 2015; Palmer et al., 2015). Senescence also increases IL-6 in human astrocytes (Bhat et al., 2012; Correale and Farez, 2015; Hou et al., 2017). A number of studies reported increased IL-6 secretion in mouse and human keratinocytes, melanocytes, monocytes, fibroblasts, and epithelial cells by DNA damage and oncogene-induced senescence (Coppe et al., 2008; Kuilman et al., 2008). Increased expression and secretion of IL-1α and IL-1β was observed in senescent endothelial cells (Maier et al., 1990), fibroblasts (Palmieri et al., 1999), and chemotherapy-induced senescent epithelial cells (Chang et al., 2002). Senescent endothelial, epithelial, and fibroblast cells also showed increased expression of IGFBP-2, -3, -4, -5 and -6 (Wang et al., 1996; Grillari et al., 2000; Coppe et al., 2008). In the brain, most of the basal expression of cytokines was localized in astrocytes, but not in the microglia and neurons (Campuzano et al., 2009). These data indicate that astrocytes have an important role in inflammation responses during the course of brain aging. In this study, the secretion of IL-6 and IL-1β was induced in senescent astrocytes using tenovin-1. However, levels of other immune-related proteins or the gene expression of TNFα, iNOS, and COX2 were not changed. Whether these results imply a different kinetic profile of the expression of inflammatory marker proteins by tenovin-1 treatment or the dominant role of IL-1 and IL-1β in the senescent inflammatory response of astrocytes remains to be determined.
Cellular senescence is defined as a condition in which the arrest of cell proliferation is irreversible (Sasaki et al., 2006; Lee et al., 2017). We observed decreased levels of cell cycle-related proteins such as phospho-histone H3 and CDK2, but not CDK4. Cell cycle arrest by cellular senescence is a result of replicative exhaustion in vitro (Hayflick and Moorhead, 1961). Upregulation of cell cycle suppressors, such as p16, p21, and p53, was also evident in senescent cells in vitro (Ressler et al., 2006), and whether tenovin-1 may downregulate these markers need to be determined in the future.
In our study, we have not only confirmed molecular changes, but also the functional alterations in senescence-induced astrocytes by tenovin-1. Several experiments have focused on delayed wound-healing effects on aged mice compared with young mice (Ashcroft et al., 1997; Keylock et al., 2008; Guo and Dipietro, 2010). We found significantly reduced wound-healing function in tenovin-1-treated astrocytes than in vehicle-treated astrocytes. Senescent 92.1 and Mel 270 cell lines, induced by tenovin-6, exhibited decreased wound-healing ability owing to the downregulation of mmp9 and mmp2 (Dai et al., 2016). Although the molecular mechanism of decreased would-healing activity in tenovin-1-treated astrocytes is unclear, the results suggest that tenovin-1 possibly inhibits SIRT1 and SIRT2 activity and reduces the adaptive response of astrocytes against cellular and physical brain injury.
Overall, we successfully demonstrated the induction of senescence in astrocytes using tenovin-1. Although further studies are needed to unequivocally demonstrate the role of SIRT1 and SIRT2 on astrocyte aging, this model should be used to further investigate the effects of aging astrocytes on neuronal and brain function both in vitro and in vivo, which may help investigate the effects of aging astrocytes on the manifestation of neurodegenerative diseases. For example, investigating the effects of SIRT activators including resveratrol on astrocyte senescence and its overall role in brain aging may provide interesting clues on the modulation of pathological brain aging processes.
Acknowledgments
This work was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT & Future Planning (NRF-2016R1A2B4014707) and the NRF grant funded by the Korea government (NRF-2016R1A5A2012284).
REFERENCES
- Abbott NJ, Ronnback L, Hansson E. Astrocyte-endothelial interactions at the blood-brain barrier. Nat. Rev. Neurosci. 2006;7:41–53. doi: 10.1038/nrn1824. [DOI] [PubMed] [Google Scholar]
- Ashcroft GS, Horan MA, Ferguson MW. Aging is associated with reduced deposition of specific extracellular matrix components, an upregulation of angiogenesis, and an altered inflammatory response in a murine incisional wound healing model. J. Invest. Dermatol. 1997;108:430–437. doi: 10.1111/1523-1747.ep12289705. [DOI] [PubMed] [Google Scholar]
- Bachstetter AD, Xing B, de Almeida L, Dimayuga ER, Watterson DM, Van Eldik LJ. Microglial p38α MAPK is a key regulator of proinflammatory cytokine up-regulation induced by toll-like receptor (TLR) ligands or beta-amyloid (Aβ). J. Neuroinflammation. 2011;8:79. doi: 10.1186/1742-2094-8-79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baker DJ, Childs BG, Durik M, Wijers ME, Sieben CJ, Zhong J, Saltness RA, Jeganathan KB, Verzosa GC, Pezeshki A, Khazaie K, Miller JD, van Deursen JM. Naturally occurring p16(Ink4a)-positive cells shorten healthy lifespan. Nature. 2016;530:184–189. doi: 10.1038/nature16932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhat R, Crowe EP, Bitto A, Moh M, Katsetos CD, Garcia FU, Johnson FB, Trojanowski JQ, Sell C, Torres C. Astrocyte senescence as a component of Alzheimer’s disease. PLoS ONE. 2012;7:e45069. doi: 10.1371/journal.pone.0045069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burke DM, Mackay DG. Memory, language, and ageing. Philos. Trans. R. Soc. Lond., B, Biol. Sci. 1997;352:1845–1856. doi: 10.1098/rstb.1997.0170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campuzano O, Castillo-Ruiz MM, Acarin L, Castellano B, Gonzalez B. Increased levels of proinflammatory cytokines in the aged rat brain attenuate injury-induced cytokine response after excitotoxic damage. J. Neurosci. Res. 2009;87:2484–2497. doi: 10.1002/jnr.22074. [DOI] [PubMed] [Google Scholar]
- Chang BD, Swift ME, Shen M, Fang J, Broude EV, Roninson IB. Molecular determinants of terminal growth arrest induced in tumor cells by a chemotherapeutic agent. Proc Natl Acad Sci USA. 2002;99:389–394. doi: 10.1073/pnas.012602599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chinta SJ, Woods G, Rane A, Demaria M, Campisi J, Andersen JK. Cellular senescence and the aging brain. Exp. Gerontol. 2015;68:3–7. doi: 10.1016/j.exger.2014.09.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cho KS, Kwon KJ, Jeon SJ, Joo SH, Kim KC, Cheong JH, Bahn GH, Kim HY, Han SH, Shin CY, Yang SI. Transcriptional upregulation of plasminogen activator inhibitor-1 in rat primary astrocytes by a proteasomal inhibitor MG132. Biomol. Ther. (Seoul) 2013;21:107–113. doi: 10.4062/biomolther.2012.102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coppe JP, Patil CK, Rodier F, Sun Y, Munoz DP, Goldstein J, Nelson PS, Desprez PY, Campisi J. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol. 2008;6:2853–2868. doi: 10.1371/journal.pbio.0060301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Correale J, Farez MF. The role of astrocytes in multiple sclerosis progression. Front. Neurol. 2015;6:180. doi: 10.3389/fneur.2015.00180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cotrina ML, Nedergaard M. Astrocytes in the aging brain. J. Neurosci. Res. 2002;67:1–10. doi: 10.1002/jnr.10121. [DOI] [PubMed] [Google Scholar]
- Dai W, Zhou J, Jin B, Pan J. Class III-specific HDAC inhibitor Tenovin-6 induces apoptosis, suppresses migration and eliminates cancer stem cells in uveal melanoma. Sci. Rep. 2016;6:22622. doi: 10.1038/srep22622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Debacq-Chainiaux F, Erusalimsky JD, Campisi J, Toussaint O. Protocols to detect senescence-associated beta-galactosidase (SA-betagal) activity, a biomarker of senescent cells in culture and in vivo. Nat. Protoc. 2009;4:1798–1806. doi: 10.1038/nprot.2009.191. [DOI] [PubMed] [Google Scholar]
- Dimri GP, Lee X, Basile G, Acosta M, Scott G, Roskelley C, Medrano EE, Linskens M, Rubelj I, Pereira-Smith O, Peacocke M, Campisi J. A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proc. Natl. Acad. Sci. U.S.A. 1995;92:9363–9367. doi: 10.1073/pnas.92.20.9363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dryden SC, Nahhas FA, Nowak JE, Goustin AS, Tainsky MA. Role for human SIRT2 NAD-dependent deacetylase activity in control of mitotic exit in the cell cycle. Mol. Cell. Biol. 2003;23:3173–3185. doi: 10.1128/MCB.23.9.3173-3185.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elkhattouti A, Hassan M, Gomez CR. Stromal fibroblast in age-related cancer: role in tumorigenesis and potential as novel therapeutic target. Front. Oncol. 2015;5:158. doi: 10.3389/fonc.2015.00158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fjell AM, Walhovd KB, Fennema-Notestine C, McEvoy LK, Hagler DJ, Holland D, Brewer JB, Dale AM. One-year brain atrophy evident in healthy aging. J. Neurosci. 2009a;29:15223–15231. doi: 10.1523/JNEUROSCI.3252-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fjell AM, Westlye LT, Amlien I, Espeseth T, Reinvang I, Raz N, Agartz I, Salat DH, Greve DN, Fischl B, Dale AM, Walhovd KB. High consistency of regional cortical thinning in aging across multiple samples. Cereb. Cortex. 2009b;19:2001–2012. doi: 10.1093/cercor/bhn232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geng YQ, Guan JT, Xu XH, Fu YC. Senescence-associated beta-galactosidase activity expression in aging hippocampal neurons. Biochem. Biophys. Res. Commun. 2010;396:866–869. doi: 10.1016/j.bbrc.2010.05.011. [DOI] [PubMed] [Google Scholar]
- Grillari J, Hohenwarter O, Grabherr RM, Katinger H. Subtractive hybridization of mRNA from early passage and senescent endothelial cells. Exp. Gerontol. 2000;35:187–197. doi: 10.1016/S0531-5565(00)00080-2. [DOI] [PubMed] [Google Scholar]
- Guo S, Dipietro LA. Factors affecting wound healing. J. Dent. Res. 2010;89:219–229. doi: 10.1177/0022034509359125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayflick L, Moorhead PS. The serial cultivation of human diploid cell strains. Exp. Cell Res. 1961;25:585–621. doi: 10.1016/0014-4827(61)90192-6. [DOI] [PubMed] [Google Scholar]
- Hof PR, Morrison JH. The aging brain: morphomolecular senescence of cortical circuits. Trends Neurosci. 2004;27:607–613. doi: 10.1016/j.tins.2004.07.013. [DOI] [PubMed] [Google Scholar]
- Hou J, Kim S, Sung C, Choi C. Ginsenoside Rg3 prevents oxidative stress-induced astrocytic senescence and ameliorates senescence paracrine effects on glioblastoma. Molecules. 2017;22:E1516. doi: 10.3390/molecules22091516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaiser LG, Schuff N, Cashdollar N, Weiner MW. Age-related glutamate and glutamine concentration changes in normal human brain: 1H MR spectroscopy study at 4 T. Neurobiol. Aging. 2005;26:665–672. doi: 10.1016/j.neurobiolaging.2004.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keylock KT, Vieira VJ, Wallig MA, DiPietro LA, Schrementi M, Woods JA. Exercise accelerates cutaneous wound healing and decreases wound inflammation in aged mice. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2008;294:R179–R184. doi: 10.1152/ajpregu.00177.2007. [DOI] [PubMed] [Google Scholar]
- Kim BS, Lee CH, Chang GE, Cheong E, Shin I. A potent and selective small molecule inhibitor of sirtuin 1 promotes differentiation of pluripotent P19 cells into functional neurons. Sci. Rep. 2016;6:34324. doi: 10.1038/srep34324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim JN, Kim MK, Cho KS, Choi CS, Park SH, Yang SI, Joo SH, Park JH, Bahn G, Shin CY, Lee HJ, Han SH, Kwon KJ. Valproic acid regulates alpha-synuclein expression through JNK pathway in rat primary astrocytes. Biomol. Ther. (Seoul) 2013;21:222–228. doi: 10.4062/biomolther.2013.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuilman T, Michaloglou C, Vredeveld LC, Douma S, van Doorn R, Desmet CJ, Aarden LA, Mooi WJ, Peeper DS. Oncogene-induced senescence relayed by an interleukin-dependent inflammatory network. Cell. 2008;133:1019–1031. doi: 10.1016/j.cell.2008.03.039. [DOI] [PubMed] [Google Scholar]
- Kuilman T, Peeper DS. Senescence-messaging secretome: SMS-ing cellular stress. Nat. Rev. Cancer. 2009;9:81–94. doi: 10.1038/nrc2560. [DOI] [PubMed] [Google Scholar]
- Kurz DJ, Decary S, Hong Y, Erusalimsky JD. Senescence-associated (beta)-galactosidase reflects an increase in lysosomal mass during replicative ageing of human endothelial cells. J. Cell Sci. 2000;113:3613–3622. doi: 10.1242/jcs.113.20.3613. [DOI] [PubMed] [Google Scholar]
- Lain S, Hollick JJ, Campbell J, Staples OD, Higgins M, Aoubala M, McCarthy A, Appleyard V, Murray KE, Baker L, Thompson A, Mathers J, Holland SJ, Stark MJ, Pass G, Woods J, Lane DP, Westwood NJ. Discovery, in vivo activity, and mechanism of action of a small-molecule p53 activator. Cancer Cell. 2008;13:454–463. doi: 10.1016/j.ccr.2008.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee BY, Han JA, Im JS, Morrone A, Johung K, Goodwin EC, Kleijer WJ, DiMaio D, Hwang ES. Senescence-associated beta-galactosidase is lysosomal beta-galactosidase. Aging Cell. 2006;5:187–195. doi: 10.1111/j.1474-9726.2006.00199.x. [DOI] [PubMed] [Google Scholar]
- Lee CS, Baek J, Han SY. The Role of kinase modulators in cellular senescence for use in cancer treatment. Molecules. 2017;22:E1411. doi: 10.3390/molecules22091411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maier JA, Voulalas P, Roeder D, Maciag T. Extension of the life-span of human endothelial cells by an interleukin-1 alpha antisense oligomer. Science. 1990;249:1570–1574. doi: 10.1126/science.2218499. [DOI] [PubMed] [Google Scholar]
- Mercken EM, Mitchell SJ, Martin-Montalvo A, Minor RK, Almeida M, Gomes AP, Scheibye-Knudsen M, Palacios HH, Licata JJ, Zhang Y, Becker KG, Khraiwesh H, Gonzalez-Reyes JA, Villalba JM, Baur JA, Elliott P, Westphal C, Vlasuk GP, Ellis JL, Sinclair DA, Bernier M, de Cabo R. SRT2104 extends survival of male mice on a standard diet and preserves bone and muscle mass. Aging Cell. 2014;13:787–796. doi: 10.1111/acel.12220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitchell SJ, Martin-Montalvo A, Mercken EM, Palacios HH, Ward TM, Abulwerdi G, Minor RK, Vlasuk GP, Ellis JL, Sinclair DA, Dawson J, Allison DB, Zhang Y, Becker KG, Bernier M, de Cabo R. The SIRT1 activator SRT1720 extends lifespan and improves health of mice fed a standard diet. Cell Rep. 2014;6:836–843. doi: 10.1016/j.celrep.2014.01.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Munoz-Najar U, Sedivy JM. Epigenetic control of aging. Antioxid. Redox Signal. 2011;14:241–259. doi: 10.1089/ars.2010.3250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nichols NR, Day JR, Laping NJ, Johnson SA, Finch CE. GFAP mRNA increases with age in rat and human brain. Neurobiol. Aging. 1993;14:421–429. doi: 10.1016/0197-4580(93)90100-P. [DOI] [PubMed] [Google Scholar]
- Ota M, Yasuno F, Ito H, Seki C, Nozaki S, Asada T, Suhara T. Age-related decline of dopamine synthesis in the living human brain measured by positron emission tomography with L-[beta-11C]DOPA. Life Sci. 2006;79:730–736. doi: 10.1016/j.lfs.2006.02.017. [DOI] [PubMed] [Google Scholar]
- Palmer AK, Tchkonia T, LeBrasseur NK, Chini EN, Xu M, Kirkland JL. Cellular senescence in type 2 diabetes: a therapeutic opportunity. Diabetes. 2015;64:2289–2298. doi: 10.2337/db14-1820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palmieri D, Watson JM, Rinehart CA. Age-related expression of PEDF/EPC-1 in human endometrial stromal fibroblasts: implications for interactive senescence. Exp. Cell Res. 1999;247:142–147. doi: 10.1006/excr.1998.4341. [DOI] [PubMed] [Google Scholar]
- Popescu BO, Toescu EC, Popescu LM, Bajenaru O, Muresanu DF, Schultzberg M, Bogdanovic N. Blood-brain barrier alterations in ageing and dementia. J. Neurol. Sci. 2009;283:99–106. doi: 10.1016/j.jns.2009.02.321. [DOI] [PubMed] [Google Scholar]
- Pospelova TV, Chitikova ZV, Pospelov VA. An integrated approach for monitoring cell senescence. Methods Mol. Biol. 2013;965:383–408. doi: 10.1007/978-1-62703-239-1_26. [DOI] [PubMed] [Google Scholar]
- Ressler S, Bartkova J, Niederegger H, Bartek J, Scharffetter-Kochanek K, Jansen-Durr P, Wlaschek M. p16INK4A is a robust in vivo biomarker of cellular aging in human skin. Aging Cell. 2006;5:379–389. doi: 10.1111/j.1474-9726.2006.00231.x. [DOI] [PubMed] [Google Scholar]
- Sailasuta N, Ernst T, Chang L. Regional variations and the effects of age and gender on glutamate concentrations in the human brain. Magn. Reson. Imaging. 2008;26:667–675. doi: 10.1016/j.mri.2007.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salama R, Sadaie M, Hoare M, Narita M. Cellular senescence and its effector programs. Genes Dev. 2014;28:99–114. doi: 10.1101/gad.235184.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sasaki M, Ikeda H, Sato Y, Nakanuma Y. Decreased expression of Bmi1 is closely associated with cellular senescence in small bile ducts in primary biliary cirrhosis. Am. J. Pathol. 2006;169:831–845. doi: 10.2353/ajpath.2006.051237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Satoh A, Brace CS, Rensing N, Cliften P, Wozniak DF, Herzog ED, Yamada KA, Imai S. Sirt1 extends life span and delays aging in mice through the regulation of Nk2 homeobox 1 in the DMH and LH. Cell Metab. 2013;18:416–430. doi: 10.1016/j.cmet.2013.07.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saunders LR, Verdin E. Sirtuins: critical regulators at the crossroads between cancer and aging. Oncogene. 2007;26:5489–5504. doi: 10.1038/sj.onc.1210616. [DOI] [PubMed] [Google Scholar]
- Sowell ER, Peterson BS, Thompson PM, Welcome SE, Henkenius AL, Toga AW. Mapping cortical change across the human life span. Nat. Neurosci. 2003;6:309–315. doi: 10.1038/nn1008. [DOI] [PubMed] [Google Scholar]
- van Deursen JM. The role of senescent cells in ageing. Nature. 2014;509:439–446. doi: 10.1038/nature13193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaquero A, Scher MB, Lee DH, Sutton A, Cheng HL, Alt FW, Serrano L, Sternglanz R, Reinberg D. SirT2 is a histone deacetylase with preference for histone H4 Lys 16 during mitosis. Genes Dev. 2006;20:1256–1261. doi: 10.1101/gad.1412706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verkhratsky A. Physiology of neuronal-glial networking. Neurochem. Int. 2010;57:332–343. doi: 10.1016/j.neuint.2010.02.002. [DOI] [PubMed] [Google Scholar]
- Wang S, Moerman EJ, Jones RA, Thweatt R, Goldstein S. Characterization of IGFBP-3, PAI-1 and SPARC mRNA expression in senescent fibroblasts. Mech. Ageing Dev. 1996;92:121–132. doi: 10.1016/S0047-6374(96)01814-3. [DOI] [PubMed] [Google Scholar]
- Wong DF, Wagner HN, Jr, Dannals RF, Links JM, Frost JJ, Ravert HT, Wilson AA, Rosenbaum AE, Gjedde A, Douglass KH, et al. Effects of age on dopamine and serotonin receptors measured by positron tomography in the living human brain. Science. 1984;226:1393–1396. doi: 10.1126/science.6334363. [DOI] [PubMed] [Google Scholar]
- Xie Z, Morgan TE, Rozovsky I, Finch CE. Aging and glial responses to lipopolysaccharide in vitro: greater induction of IL-1 and IL-6, but smaller induction of neurotoxicity. Exp. Neurol. 2003;182:135–141. doi: 10.1016/S0014-4886(03)00057-8. [DOI] [PubMed] [Google Scholar]
- Zeevi N, Pachter J, McCullough LD, Wolfson L, Kuchel GA. The blood-brain barrier: geriatric relevance of a critical brain-body interface. J. Am. Geriatr. Soc. 2010;58:1749–1757. doi: 10.1111/j.1532-5415.2010.03011.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu L, Yu J, Shi Q, Lu W, Liu B, Xu S, Wang L, Han J, Wang X. Strain- and age-related alteration of proteins in the brain of SAMP8 and SAMR1 mice. J. Alzheimers Dis. 2011;23:641–654. doi: 10.3233/JAD-2010-101389. [DOI] [PubMed] [Google Scholar]
- Zou Y, Zhang N, Ellerby LM, Davalos AR, Zeng X, Campisi J, Desprez PY. Responses of human embryonic stem cells and their differentiated progeny to ionizing radiation. Biochem. Biophys. Res. Commun. 2012;426:100–105. doi: 10.1016/j.bbrc.2012.08.043. [DOI] [PMC free article] [PubMed] [Google Scholar]