Abstract
The amplification of estrogen receptor alpha (ERα) encoded by the ESR1 gene has been described as having a prognostic role in breast cancer patients. However, increased dosage of the ESR1 gene (tested by real-time PCR) is also observed in ER-negative breast cancers, which might suggest the expression of alternative isoforms of ERα (other than classical ERα of 66 kDa). In the current work, we have investigated the ESR1 gene dosage in 402 primary breast cancer patients as well as the expression of ERα isoforms—ERα66 and ERα36—on mRNA and protein levels. The obtained results were correlated with clinicopathological data of the patients. Results showed that increased ESR1 gene dosage is not related to ESR1 gene amplification measured by fluorescent in situ hybridization (FISH), but it correlates with the decreased expression of ERα66 isoform (p = 0.01). Interestingly, the short ER isoform ERα36 was expressed in samples with increased ESR1 gene dosage, suggesting that genomic aberration might influence the expression of that particular isoform. Similarly to ESR1 increased gene dosage, high ERα36 expression was linked with the decreased disease-free survival of the patients (p = 0.05), which was independent of the status of the classical ERα66 level in breast tumors.
Keywords: breast cancer; estrogen receptor; ERα36, ERα66, gene amplification; prognostic factor
1. Introduction
Estrogen receptor alpha (ERα) is an important prognostic and predictive factor in breast cancer. It is a ligand-activated transcription factor and its signaling governs the growth, proliferation, and survival of cancer cells. This makes it a crucial target for endocrine therapies [1]. In breast cancer, nuclear ERα levels are routinely assessed by immunohistochemical methods particularly to determine the dependency of a tumour on estrogen-driven growth [2]; together with human epidermal growth factor receptor 2 (HER2), these are the basis of the molecular subtyping of breast cancers [3].
ERα is encoded by the ESR1 gene located on chromosome 6. Due to its prognostic and predictive potential, ESR1 gene alterations have been studied intensively. Gene amplification, as a mechanism of gene overexpression, may influence levels of the ERα protein, resulting in altered growth-stimulating signaling. Thus, changes in ESR1 copy number are a subject of ever-present debate. Reported ESR1 amplification rates in breast cancer range from 0% to 75% of patients [4,5,6,7,8,9]. Observed discrepancies have been related to different analytical techniques used for ESR1 copy number analysis. We have previously developed a qPCR-based protocol measuring gene copy number alterations in topoisomerase IIα (TOP2A), which we showed was able to detect smaller structural changes in the TOP2A gene than large fluorescent in situ hybridization (FISH) probes [10]. We have applied a similar technique for the analysis of ESR1 genomic sequence alteration [6]. With this method, we have shown that the ESR1 copy number changes (gene dosage changes) occur also in ER-negative patients and have prognostic significance. In the current study, we aimed at exploring this seemingly paradoxical observation of absent (or low) ER protein level in the presence of increased ESR1 gene dosage. Since the routine evaluation of ER in breast cancer focuses on the classical ER isoform of the molecular mass of 66 kDa (hence its name ERα66), aberrations in the ESR1 gene were thus far correlated with levels of classical ER isoforms but omitted other ER isoforms. In 2005, Wang et al. described a 36-kDa splicing variant of the ESR1 gene, which was called ERα36 [11,12]. It differs from the ERα66 isoform by lacking both transcriptional activation domains, but retains the DNA-binding domains [13,14], although new results indicate that it might act as a transcription factor [15]. Dissimilar to ERα66, which is usually detected in the cell nucleus, ERα36 localizes mainly to the cytosol and cell membrane [11,16], although nuclear localization is also observed [15,17,18]. ERα36 expression was described in ER-positive as well as in ER-negative breast cancer cell lines [17] and breast cancers [15,19,20]. The study performed by Lee et al. on 31 breast cancer patients describes that ERα36 assessed by immunohistochemical staining (IHC) is commonly expressed in breast cancers with different ER status [21]. A retrospective study of 896 breast cancer patients reveals that ERα66-positive patients with a high expression of ERα36 are less likely to benefit from hormonal therapy [19]. In vitro studies performed using various breast cancer cells indicate that ERα36 rapidly activates the MAPK signalling pathway, leading to uncontrolled proliferation and anti-apoptotic events [22].
Thus, we decided to explore whether the ESR1 gene dosage measured by qPCR is related to the amplification of the whole ESR1 gene and if it correlates with the expression of classical ERα66 as well as the short ER isoform ERα36. Additionally, the clinical significance of the measured parameters was assessed in the group of operable breast cancer patients.
2. Results
2.1. ESR1 Gene Dosage and Copy Number in Breast Cancers
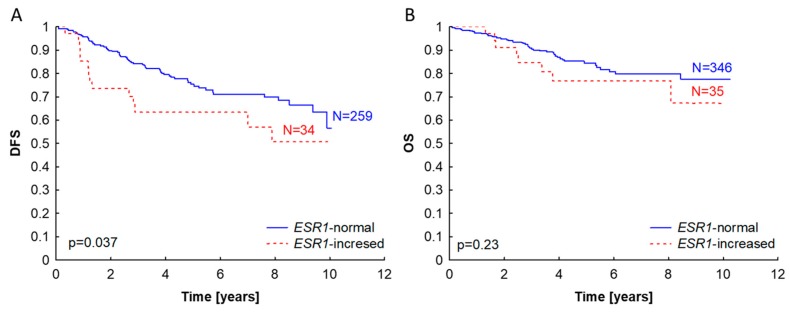
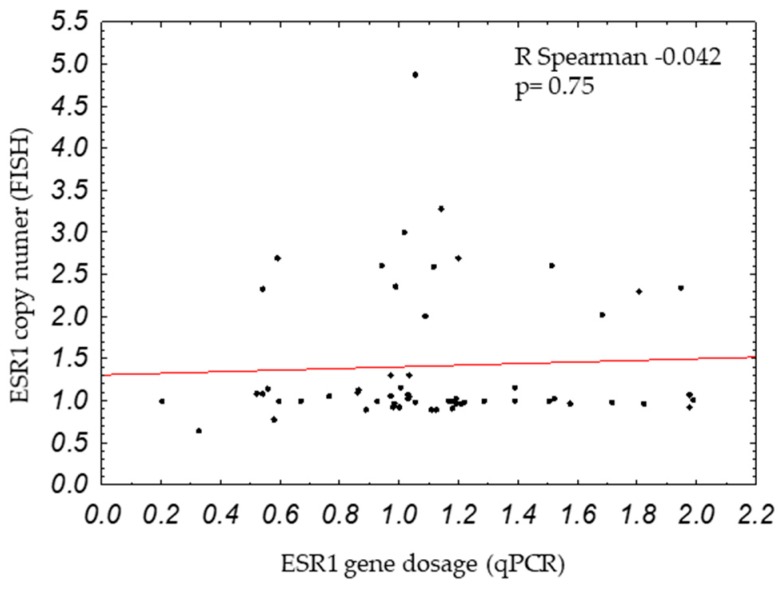
ESR1 gene dosage was examined by qPCR in 402 samples of primary breast cancers. Increased ESR1 gene dosage (ESR1/APP ratio ≥2) was observed in 9.2% (37/402) of the patients. As previously reported on a smaller group of patients [4], increased ESR1 gene dosage was linked with the poor survival of the patients (Figure 1). As the literature describes the opposite findings, when ESR1 gene status was tested by FISH, it made us wonder if increased ESR1 gene dosage measured by qPCR would also correspond to ESR1 amplification analyzed by the golden standard method for gene amplification testing: FISH. For this analysis, we selected 80 breast cancer samples that were additionally tested by FISH (successful results with both methods, qPCR and FISH, were available for 58 samples). In one case, polyploidy occurred with the average number of 3.5 CEN6 and 3.7 ESR1 copies per cells; the sample was removed from the analysis. ESR1 gene status measured by qPCR and FISH did not correlate (r = −0.042, p = 0.75, Figure 2), which suggests that both methods detect different types of alterations. FISH and qPCR assays detect different fragments in the ESR1 gene: the FISH probe binds a region of 395 kbp, whereas with our qPCR assay, we detected an amplicon of 60 bp. Thus, FISH might detect larger, whole gene amplification, whereas qPCR might detect small changes in the ESR1 gene that go undetected by the long FISH probe. Therefore, if ESR1 gene dosage is not related to the amplification of the whole ESR1 gene, we asked if ESR1 gene dosage might be related to the altered expression of ER, including other than the classical ERα66 isoform, which could be the underlying factor conferring poor prognosis to breast cancer patients.
Figure 1.
Kaplan–Meier survival curves according to ESR1 gene dosage status (measured by qPCR) in primary breast tumors. ESR1-normal status was described as ESR1/APP ratio <2, ESR1-increased as ESR1/APP ratio ≥2. The probability of disease-free survival (A) and overall survival (B) are shown.
Figure 2.
Correlation between ESR1 copy number (measured by fluorescent in situ hybridization, or FISH) and ESR1 gene dosage (measured by qPCR).
2.2. Genomic ESR1 Level versus mRNA and Protein Isoforms Expression
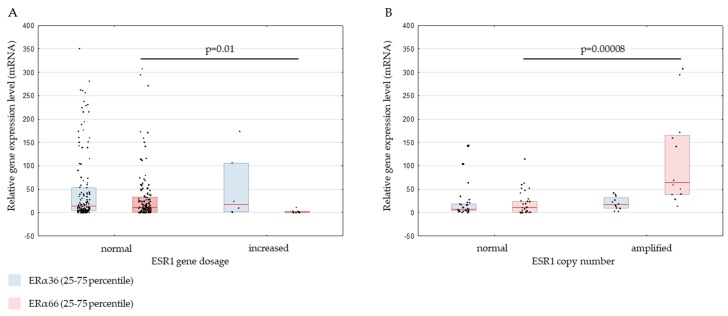
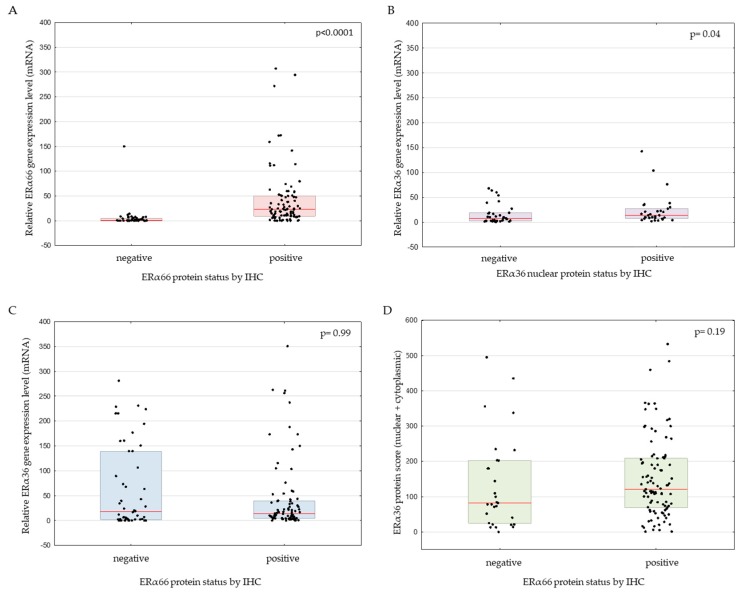
We have analyzed the expression of ERα36 and ERα66 on mRNA level by qPCR in frozen breast cancer samples and on protein level by IHC in formalin-fixed, paraffin-embedded (FFPE) samples. The results were correlated with ESR1 gene tested by qPCR and FISH. On the mRNA level, reduced expression of the full-length ERα66 isoform was observed in samples with increased ESR1 gene dosage (median relative expression 10.63 in ESR1-normal gene dosage and 0 in ESR1-increased gene dosage, p = 0.01; Figure 3A). On the contrary, increased ERα66 expression was found in samples with amplified ESR1 measured by FISH (median gene expression level in ESR1-amplified samples [ESR1/CEN-6 ratio <2] −64.14 and in ESR1-normal samples [ESR1/CEN-6 ratio ≥2] −10.28; p = 0.00008, Figure 3B). However, there was no correlation between ERα36 expression and ESR1 gene dosage (measured by qPCR; Figure 3A) or ESR1 copy number (measured by FISH; Figure 3B). The expression of ERα36 was observed in samples with normal and increased ESR1 gene dosage (Figure 3A), which also included ERα66-negative samples. However, depending on the ESR1 gene dosage, the ratio of median ERα36 to ERα66 mRNA expression level (ERα36/ERα66) was markedly different: in ESR1-increased gene dosage samples, the ERα36/ERα66 ratio was 86, in comparison to ESR1-normal gene dosage samples, which had the ratio of 1.4. For ESR1-amplified and ESR1–normal samples by FISH, the ERα36/ERα66 ratio was almost unaffected (0.3 versus 0.7, respectively).
Figure 3.
ERα36 and ERα66 relative gene expression levels in primary breast cancers classified according to ESR1 gene dosage status measured by qPCR (A) or ESR1 gene copy number measured by FISH (B). The vertical red line represents the median gene expression level. * p < 0.05, ** p < 0.0001. ERα: estrogen receptor alpha.
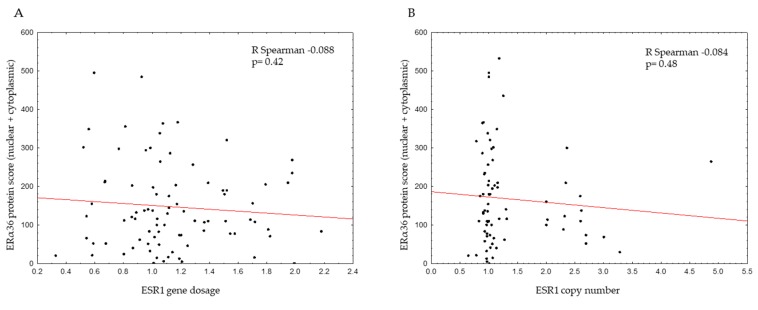
The ESR1 gene dosage was also inversely correlated with ERα66 protein level (Table 1, p = 0.001) and mRNA expression (p = 0.0086; Table 1, Figure S3A). In samples with ESR1-increased gene dosage, 65% of samples were negative for ERα66 protein (according to the Allred score), whereas in samples with normal ESR1 gene dosage, only 37% of the samples were ERα66-negative. Therefore, in ERα66-negative tumors, an ESR1-increased gene dosage was found in 15% (24/158) of the samples, in comparison to 5% (13/240) of the ER-positive samples. For ESR1 gene status tested by FISH, there was a good positive correlation: all the ESR1-amplified tumors were ERα66-positive, and none of the ERα66-negative tumors carried ESR1 amplification (p = 0.0038). ESR1 gene copy number was also positively correlated with ERα66 gene expression (p = 0.00002; Table 1, Figure S3B). For ERα36 protein level (presented as the total IHC score: the sum of cytoplasmic and nuclear staining scores), no correlation was found between ESR1 gene dosage or copy number (Figure 4A,B); similarly, there was no correlation between ERα36 mRNA expression and the ESR1 gene by FISH and qPCR (Figure S3C,D).
Table 1.
Correlation between ERα66 protein status, ERα66 gene expression, and ESR1 gene status measured by qPCR or FISH.
| Header |
ESR1
Gene Dosage Status (qPCR)
[Number of Cases (%)] |
Total | p |
ESR1
Gene Copy Number (FISH)
[Number of Cases (%)] |
Total | p | ||
| Normal | Increased | Normal | Amplified | |||||
| ERα66 Protein Status | ||||||||
| negative | 134 (37%) | 24 (65%) | 158 | 0.001 | 21 (36%) | 0 | 21 | 0.0038 |
| positive | 227 (63%) | 13 (25%) | 240 | 37 (64%) | 15 (100%) | 52 | ||
| Total | 361 (100%) | 37 (100%) | 58 (100%) | 15 (100%) | 73 | |||
| ESR1 Gene Dosage Status (qPCR) | Total | p | ESR1 Gene Copy Number (FISH) | Total | p | |||
| Normal | Increased | Normal | Amplified | |||||
| ERa66 mRNA Expression, Median (25–75 Percentile) | 10.63 (0.84–33.1) |
0.21 (0–3.22) |
145 | 0.0086 | 10.28 (0.2–24.5) |
64.14 (39–165.3) |
45 | 0.00002 |
Figure 4.
Correlation between ERα36 protein score (total nuclear and cytoplasmic score and ESR1 gene dosage (measured by qPCR) (A) or ESR1 copy number (measured by FISH) (B).
2.3. Correlation between ERα66 and ERα36 Isoforms on mRNA and Protein Level
As expected, the level of ERα66 mRNA was increased in samples positive for ERα66 protein (p < 0.0001, Figure 5A); in the case of ERα36, correlation between mRNA and protein was observed, but only for the nuclear localization (p = 0.04, Figure 5B). Interestingly though, ERα36 mRNA was expressed on a similar level in IHC ERα66-negative and ERα66-positive samples (median relative gene expression levels of 17.8 and 13.7, respectively; p = 0.99, Figure 5C). Also on the protein level, ERα36 was not statistically different between ERα66-negative (median IHC score 82) and ERα66-positive samples (median IHC score 120; p = 0.19, Mann–Whitney test, n = 126, Figure 5D).
Figure 5.
ERα66 relative expression according to ERα66 immunohistochemical (IHC) protein status (A), ERα36 relative expression according to ERα36 nuclear protein (B), ERα36 expression according to ERα66 protein status (C), and ERα36 protein immunohistochemical score according to ERα66 protein status (D).
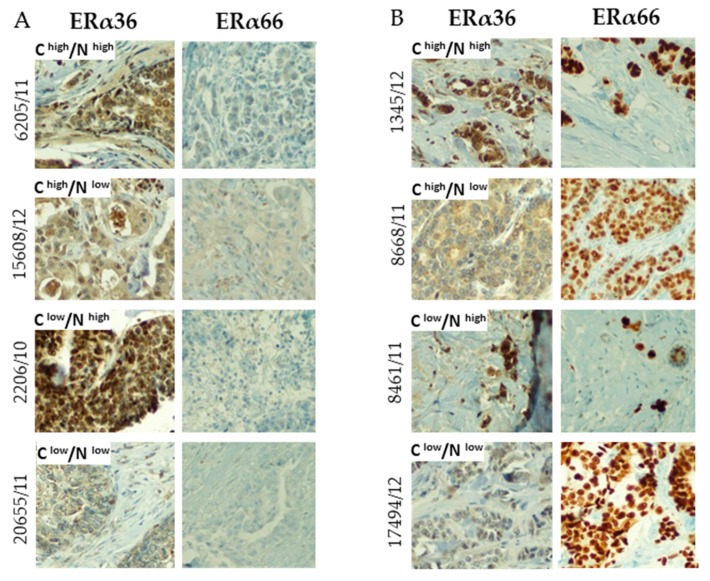
The ERα36 protein showed a different staining pattern than ERα66: whereas ERα66 was mostly located in the nucleus, ERα36 was observed both in the nucleus and cytoplasm, and also with different immunohistochemical scores in nuclear and cytoplasmic localization (Figure 6). The presence of ERα66 was not associated with the localization of the ERα36 isoform; both nuclear and/or cytoplasmic ERα36 was observed in ERα66-negative (Figure 6A) and positive samples (Figure 6B).
Figure 6.
Exemplary photos of immunohistochemical staining of breast cancer samples with different ERα66 status (panel A – negative, panel B – positive according to Allred score) and ERα36 status. The ERa36 staining pattern has additionally been divided into positive and negative in cytoplasmic (C) and nuclear (N) localization based on the immunohistochemical score of the samples. All photos were taken under 20× magnification.
2.4. Clinical Significance of ERα Isoforms Expression
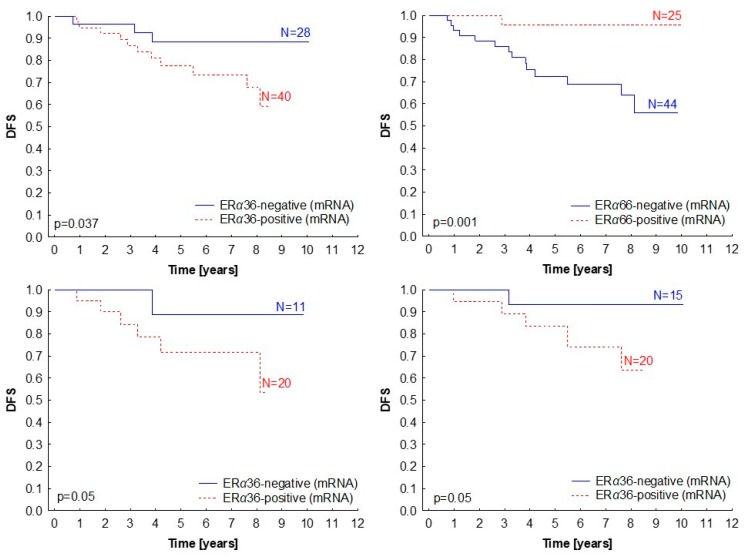
Since we have observed ERα36 isoform expression in patients with ESR1 increased gene dosage and in ERα66-negative patients, we were interested to see how it influences the clinicopathological characteristics of the patients (Table 2). The ERα36 expression did not correlate with stage, grade, lymph node status, breast cancer molecular subtype, or histological tumor subtype, but its elevated expression (above median) was linked with shorter disease-free survival (p = 0.037; Figure 7A). Interestingly, ERα66 showed the exact opposite effect (p = 0.001; Figure 7B). Survival analysis in subgroups showed that the prognostic significance of the ERα36 isoform was sustained in both the ER-negative and ER-positive groups (Figure 7C,D), indicating that the ERα36 effect is independent of the presence of the full-length ERα66 isoform.
Table 2.
Correlation between ERα36 relative expression level (measured by qPCR) and the clinicopathological characteristics of the patients. HER2: human epidermal growth factor receptor 2.
| Variable | N | Median ERα36 Expression (25–75th Percentile) |
p |
|---|---|---|---|
| T stage | p = 0.41 | ||
| T1–2 | 127 | 13.17 (3.75–42.90) | |
| T3–4 | 10 | 58.02 (7.37–160.88) | |
| N stage | p = 0.69 | ||
| N0 | 78 | 17.20 (3.52–63.44) | |
| N1 | 58 | 9.95 (4.23–40.05) | |
| Grading | p = 0.92 | ||
| 1 | 13 | 18.84 (5.34–27.19) | |
| 2 | 63 | 9.89 (3.75–37.41) | |
| 3 | 42 | 9.97 (2.89–39.08) | |
| Histological subtype | p = 0.21 | ||
| Ductal | 54 | 31.28 (4.1–144.7) | |
| Lobular | 7 | 74.39 (1.1–180.7) | |
| Other | 4 | 162.13 (78.1–205.5) | |
| Molecular type | p = 0.84 | ||
| Luminal A | 17 | 16.70 (4.68–150.35) | |
| Luminal B HER2– | 13 | 11.18 (3.98–24.24) | |
| Luminal B HER2+ | 2 | 97.21 (0–194.42) | |
| Non luminal HER2+ | 6 | 83.57 (0–160.88) | |
| Triple negative | 13 | 39.56 (2.88–160.1) | |
| ER status | p = 0.99 | ||
| 0 | 50 | 17.84 (1.91–139.11) | |
| 1 | 87 | 13.71 (4.41–39.08) | |
| PR status | p = 0.08 | ||
| 0 | 52 | 10.46 (1.52–53.17) | |
| 1 | 85 | 15.77 (5.34–54.09) | |
| HER2 status | p = 0.35 | ||
| 0 | 104 | 13.82 (3.94–59.45) | |
| 1 | 24 | 10.41 (2.56–31.21) |
Figure 7.
Kaplan–Meier survival curves presenting disease-free survival (DFS) according to ERα36 (A) and ERα66 (B) mRNA levels (measured by qPCR) in primary breast tumors. (C,D) Subgroup analysis for ERα36 expression in ERα66 protein-negative (p = 0.05) (C) and positive (D) patients (p = 0.05). Median relative gene expression was a cut-off value for the classification of samples into negative and positive.
On the protein level, nuclear ERα36 was connected with decreased overall survival (p = 0.04), and cytoplasmic ERα36 showed a similar trend, but the result was not statistically significant (p = 0.12; Supplementary Figure S2). Data on disease-free survival (DFS) was not available for this set of patients.
2.5. The Role of ERα36 Isoform in Breast Cancer Cell Lines
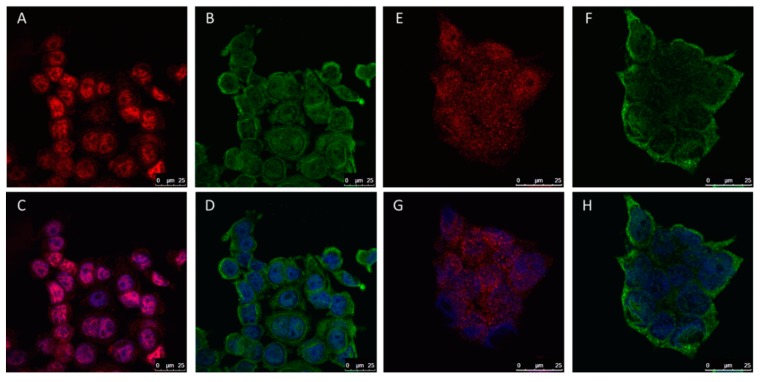
Similarly to clinical samples, we observed that in ERα66-positive cell lines, the MCF7 and BT474, ERα36 isoform localized independently from ERα66. ERα36 was found in the cytosol and nucleus, whereas ERα66 was found predominantly in cell nucleus (Figure 8).
Figure 8.
Immunofluorescent staining of two breast cancer cell lines: MCF7 (A–D) and BT474 (E–H). Staining for: ERα66 (red), ERα36 (green), and nucleus (blue).
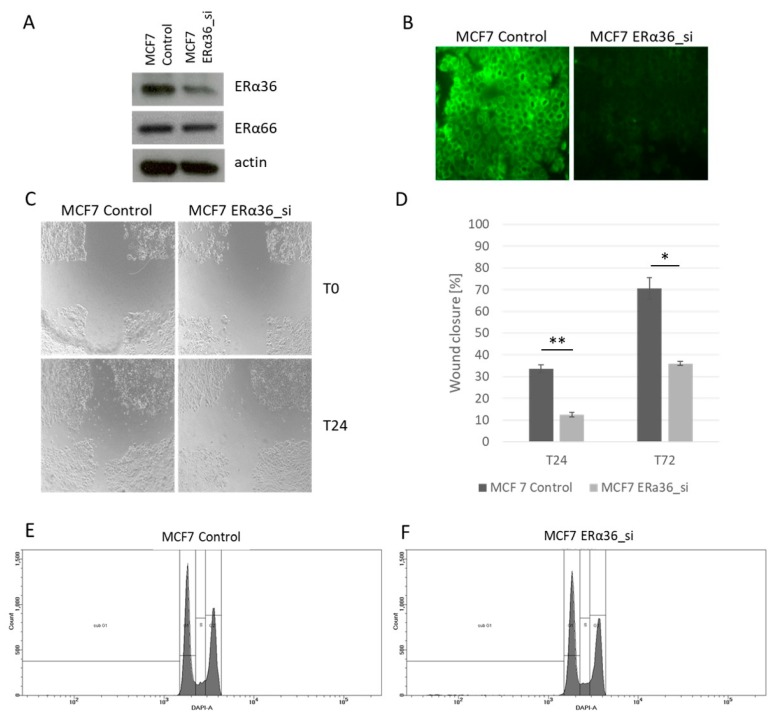
Basing on our observations from the clinical samples, where the prognostic significance of ERα36 was observed, we asked if silencing ERα36 will translate to decreased migration and changes in the cell cycle of breast cancer cell lines. Three siRNAs against ERα36 were tested, and siRNA#3 was chosen as it resulted in the strongest decrease in ERα36 protein level measured by Wester blot (Figure 9A,B). As a negative control, non-targeting siRNA was used in all the experiments. MCF7 cells with silenced ERα36 showed a significantly lower ability to migrate in wound-healing assay (Figure 9C,D). Silencing had no effect on cell cycle analyzed by flow cytometry (Figure 9E,F).
Figure 9.
Silencing the ERα36 by siRNA confirmed by Western blotting (A) and immunofluorescence (B). Decrease in cell migration measured by wound healing assay in cells with silenced ERα36 (C,D, * p = 0.005, ** p = 0.002). Cell cycle analysis by flow cytometry in the MCF7 wild-type cell line (MCF7 Control) and with ERα36 silenced (MCF7 ERα36_si) showed no differences in the number of cells in a given cell cycle phase (E,F).
3. Discussion
As the estrogen receptor is one of the most important molecular targets in breast cancer, it is crucial to deeply understand every aspect of its functioning, as it might help to predict treatment response or disease course. The amplification of ESR1 was described in mastopathic breast tissue, which progressed to invasive cancer [23] or pre-malignant endometrial cancers [24], implying early ESR1 amplification and its role in carcinogenesis. Moreover, the amplification of ESR1 was shown to be linked with good prognosis in tamoxifen-treated breast cancer patients [4,25]. Nevertheless, large variations are reported in the frequency of ESR1 amplification in breast cancer (0–75%, reviewed in [9]), depending on the methodology and applied cut-off values. Regarding the correlation between ESR1 amplification and the expression of ER, studies results are divided. As a general rule, in morphology-guided methods of ESR1 amplification analysis, such as FISH, correlations between gene and protein levels were more frequently observed [4,5,23,26,27]. It is still being debated if the analysis of ESR1 amplification assessed by FISH can be influenced by the FISH probe binding to the ESR1 mRNA, generating false-positive signals [5,7,28]. In our analysis, we have used validated FISH probes, which give similar results (ESR1 copy number) with and without RNase treatment [5], making RNase treatment a dispensable step.
We have previously reported that increased ESR1 gene dosage measured by qPCR has prognostic significance in breast cancer patients [6]. Now, we have performed additional ESR1 gene dosage analysis using the previously developed qPCR method. We observed increased ESR1 gene dosage (ESR1/APP ratio ≥2) in 9.4% of all patients. Interestingly, ESR1 gene dosage increase was observed more frequently in ER-negative compared to ER-positive patients (15% versus 5% of the patients). Since gene amplification is a known mechanism for protein overexpression, one would expect the biological effect to be observed only in patients with ER-positive tumors. Therefore, we have selected a subset of sample for which ESR1 gene status was measured both with qPCR and a golden standard method: FISH. We have shown that there is no correlation between ESR1 gene dosage (measured by qPCR) and ESR1 copy number (measured by FISH), which underlines that qPCR detects different changes than FISH. We had a similar observation when analyzing changes in the topoisomerase II α gene (encoded by TOP2A) in breast cancers [29]. Possibly, much smaller changes are detected by qPCR than by large FISH probes spanning the whole gene sequence [10]. This would make the aberrations measured by qPCR go undetected by large FISH probes.
To our knowledge, this is the first study describing aberrations in the ESR1 gene in the context of the expression of ER isoforms. In the current work, we have tested the expression of two ER isoforms on mRNA and protein levels: classical ERα66 and a short variant, ERα36, which was recently described as playing a role in breast cancer aggressiveness [11]. Regarding the expression levels of isoforms, ESR1-increased gene dosage measured by qPCR was linked with decreased ERα66 expression (on the mRNA and protein level), as opposed to ESR1 amplification measured by FISH, which correlated with increased ERα66 protein and mRNA. In the case of the ERα36 isoform, no correlation was observed between ESR1 measured by qPCR or FISH. The qPCR amplicon, which is designed to detect changes in ESR1 gene dosage, is located in exon 1 of the ESR1 gene. Since small aberrations in regulatory regions might affect the expression level of a gene or its splice variants [30], it is possible that the ESR1 gene dosage alterations that we have measured by qPCR decrease the ERα66 level, but do not change ERα36 expression. Indeed, in samples with ESR1-increased gene dosage, the ERα36/ERα66 ratio was over 60 times higher than that in the ESR1-normal samples. Therefore, we have investigated what is the prognostic role of ER isoforms, being especially interested in the novel ERα36, for which data is very limited. We observed that the two investigated isoforms have a dramatically different impact on survival. Whereas classical ERα66 is a marker of good prognosis, ERα36 was related to decreased disease-free survival, which is similar to the increased ESR1 gene dosage measured by qPCR. The prognostic effect of ERα36 was visible in both ERα66-negative and ERα66-positive patients. This points to ERα66-independent mechanism of ERα36 action; even in samples with high ERα66 expression, ERα36 still conferred poor prognosis. This confirms the results by Wang et al., who also reported the negative influence of ERα36 expression on the survival of both ERα66-negative and ERα66-positive breast cancer patients, especially those treated with tamoxifen [15]. The study of Shi et al. also showed the prognostic role of ERα36, but only in ERα66-positive patients treated with tamoxifen (and not in ERα66-negative patients) [19]. Further studies are required to uncover the dependence between ERα isoforms and their impact on the survival of breast cancer patients during treatment.
Other groups have reported on the expression of the ERα36 isoform in breast cancer cell lines [11,17,20,31,32,33,34] and breast cancer patients [15,19,20,34,35,36]. Deng et al. [31,32] as well as Yin et al. [33] showed that increased ERα36 expression is important for the maintenance of the stem cell population in breast cancers, and Zhao indicated its role in tamoxifen resistance, which is possibly related to non-genomic signaling via the MAPK and Akt pathways [34]. On the other hand, Wang et al. proposed a mechanism in which after binding to estrogen or Tamoxifen, ERα36 translocates to the nucleus and acts as a transcription factor for stem cell marker ALDH1A1 [15]. The tamoxifen resistance of the MCF7 cell line was also attributed to the role of ERα36 in the downregulation of ERα66 [37]. Also, Zhang et al. described an inversed association between the expression of ERα36 and ERα66 isoforms [17]. We have not observed any correlation between the expression of the two ER isoforms, but this effect might be specific to the tumor cell type and state (such as the condition of acquired tamoxifen resistance). In the in vitro functional tests, we showed that the silencing of ERα36 decreased the migration of the MCF7 breast cancer cell line, which was also described by Li et al. [37]. Thus, ERα36 might be important for the invasive potential of cancer cells, which translates to the poor outcome of the patients.
4. Materials and Methods
Four hundred and eighteen primary tumours from breast cancer patients (stages I–IV) treated in the Medical University of Gdansk were investigated. Their detailed clinical characteristics are listed in Table 3. The study was granted permission from the Bioethical Committee of the Medical University of Gdansk. ESR1 gene dosage was tested using qPCR on frozen primary tumours, the data was successfully obtained for 402 samples. ESR1 gene amplification was measured using fluorescent in situ hybridization (FISH) on tissue microarrays containing 80 formalin-fixed, paraffin-embedded (FFPE) tissues samples. For ERα36 and ERα66 gene expression analysis, 149 frozen primary tumor samples were used. The median age of the patients was 58 years (27–61 years). Informed consent was collected from all the participants who were included in the study.
Table 3.
Patient characteristics.
| Variable | Number of Cases (%) | |
|---|---|---|
| Age | ||
| <50 | 120 | (29) |
| >50 | 298 | (71) |
| T Stage | ||
| 1 | 140 | (33) |
| 2 | 194 | (46) |
| 3 | 40 | (10) |
| 4 | 39 | (9) |
| Missing data | 5 | (2) |
| N Stage | ||
| negative | 207 | (49) |
| positive | 206 | (49) |
| Missing data | 5 | (2) |
| Grade | ||
| 1 | 30 | (7) |
| 2 | 171 | (41) |
| 3 | 135 | (32) |
| Missing data | 82 | (20) |
| Histologic Type | ||
| Ductal | 218 | (52) |
| Lobular | 54 | (13) |
| Other | 26 | (6) |
| Missing data | 120 | (29) |
| ER Status | ||
| negative | 164 | (39) |
| positive | 250 | (59) |
| Missing data | 4 | (2) |
| PR Status | ||
| negative | 175 | (42) |
| positive | 239 | (57) |
| Missing data | 4 | (2) |
| HER2 Status | ||
| negative | 274 | (66) |
| positive | 59 | (14) |
| Missing data | 85 | (20) |
4.1. RNA and DNA Isolation, Reverse Transcription, and Gene Expression Analysis
Primary tumor samples were collected during surgery, snap frozen, and stored at –80 °C. DNA and RNA isolation was performed usingan AllPrep DNA/RNA Kit (Qiagen, Hilden, Germany) according to the manufacturer protocol. Briefly, 30 mg of frozen tissue was lysed in lysis buffer and ceramic beads using a MagNA Lyser (Roche, Basile, Switzerland), then added to the DNA-binding column and processed further for DNA isolation. Flow-through was used to isolate RNA with the DNase digestion step. Purified RNA (1000 ng) was reverse-transcribed with random hexamers to cDNA using a Transcriptor First Strand cDNA Synthesis Kit (Roche, Basil, Switzerland) and Mastercycler Gradient Thermal Cycler (Eppendorf, Hamburg, Germany) according to the manufacturer protocol. The expression of ERα36 and ERα66 was measured using real-time PCR with Universal PCR mastermix (Applied Biosystems, Foster City, CA, USA), giving 10 ng of cDNA per reaction. Two reference genes GAPDH and YWHAZ were chosen based on their expression stability in the tested samples. Sequences of the primers were as follows. For ERa36: forward, 5′-CCAAGAATGTTCAACCACAACCT-3′; reverse, 5′-GCACGGTTCATTAACATCTTTCTG-3′. For ERα66: forward, 5′-AAGAAAGAACAACATCAGCAGTAAAGTC-3′; reverse, 5′-GGGCTATGGCTTGGTTAAACAT-3′. For GAPDH: forward, 5′-ACAACTTTGGTATCGTGGAAGG-3′; reverse, 5′-GCCATCACGCCACAGTTTC-3′. For YWHAZ, forward, 5′-TGTAGGAGCCCGTAGGTCATC-3′; reverse, 5′-GTGAAGCATTGGGGATCAAGA-3′. The PCR programme used was: 95 °C for 10 min; then, 40 cycles of 95 °C for 15 s, 60 °C for 1 min, and 95 °C for 10 s. The melting curve was performed by monitoring fluorescence in the samples, which were heated up from 65 °C to 95 °C in 0.5 °C increments.
4.2. ESR1 Gene Dosage Analysis with qPCR
ESR1 gene dosage in frozen primary tumors was tested according to our method described before [6]. Briefly, ESR1 gene dosage was measured in a relative manner (to APP reference gene and full blood as a calibrator) in real-time PCR (CFX96 cycler, Bio-Rad, Hercules, CA, USA) with Locked Nucleic Acid hydrolysis probes. Gene dosage equal to or higher than two was defined as ESR1 increased gene dosage.
4.3. ESR1 Copy Number Analysis with Fluorescent in situ Hybridization
FISH analysis was performed on tissue microarray (TMA) sections of FFPE samples from 80 breast cancer patients using a ZytoLight SPEC ESR1/CEN 6 Dual Color Probe (ZytoVision, Bremerhaven, Germany) and ZytoLight FISH-Tissue Implementation Kit (ZytoVision, Bremerhaven, Germany) according to the guidelines of the manufacturer. The evaluation of fluorescent signals was performed by the analysis of images under the fluorescent microscope. Twenty to thirty nuclei cells were evaluated per sample; the number of fluorescent signals from the ESR1 probe and centromere 6 (CEN6) probe was counted, averaged, and converted into a ratio of ESR1/CEN6 signal per cell. The ESR1/CEN6 ratio of ≥2 was classified as ESR1 amplification, while the ESR1/CEN6 ratio between 1.3–2 was classified as gain (according to [5]). Exemplary photos of breast cancer samples analyzed for ESR1 gene amplification with FISH are shown in Figure S1.
4.4. ERα36 Protein Level Analysis with Immunohistochemistry
Tissue microarrays (TMA) were prepared by sampling up to five non-adjacent tissue cores of 1-mm diameter from each FFPE primary tumor. Serial sections were analyzed by manual immunohistochemical staining with commercially available rabbit antibodies against ERα36 specific to unique C-terminal sequence (Cell Applications Inc. San Diego, CA, USA, Cat# CY-1109; dilution 1:800, incubation time 1 h). Secondary anti-rabbit antibodies conjugated with horseradish peroxidase (HRP) were used together with the Novolink Max-Polymer Detection System (Leica Novocastra, Wetzlar, Germany) for the detection of the ERα36 protein. Intensity (scale 0–3) and the percentage of positively stained cells were evaluated in the nucleus and cytoplasm/membrane, giving an immunohistochemical score between 0–300. The results of the immunochistochemical staining of FFPE breast cancer samples with low and high immunohistochemical scores and the Western blotting analysis of the corresponding frozen breast cancer samples are presented in the Supplementary Figure S5.
4.5. ERα66, PR, and HER2 Status Analysis in Breast Cancer Samples
Hormone receptor status in breast cancer samples was assessed by IHC with mouse monoclonal antibody against ERα66 (clone 1D5, Dako Agilent, Santa Clara, CA, USA) and PR (clone 636, Dako Agilent, Santa Clara, CA, USA) according to the manufacturer’s instructions. Antibodies were used at a dilution of 1:50; antigen retrieval was performed at neutral pH by water-bath heating at 90 °C for 30 min. Visualization was performed with the Envision Dako (Dako Agilent, Santa Clara, CA, USA) system. For ER and PR, evaluation of the immunohistochemical nuclear staining was performed based on an Allred score or the immunoreactivity of any intensity in at least 10% of the tumor cells (for older tumor samples). HER2 receptor status was analyzed by IHC: a 3+ score was considered positive, and 2+ cases were equivocal and were tested for HER2 gene amplification with FISH with the use of PathVision HER2 DNA Probe Kit (Abbott Molecular, Abbott Park, IL, USA), according to the manufacturer’s instructions. The mean numbers of HER2 and centromer 17 signals were estimated for each tumor sample. A ratio of HER2/CEP-17 ≥2 was considered as HER2 amplification and an HER2-positive result.
4.6. Cell Culture
MCF7 and BT474 cells were purchased from the American Tissue Culture Collection (ATCC, Manassas, VA, USA). Cells were passaged for a maximum of 3 to 4 months post resuscitation and routinely tested for mycoplasma contamination. MCF7 cells were cultured in DMEM supplemented with 10% fetal bovine serum (FBS). BT474 cells were maintained in RPMI-1640 with 10% FBS and 5 μg/mL insulin. All the media and their supplements were from Sigma-Aldrich (Saint Louis, MO, USA) or HyClone (GE Healthcare, Chicago, IL, USA).
4.7. Gene Knock-Down with siRNA and Western Blotting
Cells were seeded and transfected after 24 h using Lipofectamine 2000 according to the producers’ protocol. The siRNA used in this experiment was designed to interfere with ERα36 mRNA. The following sequences were chosen: sense 5′-AUGCCAAUAGGUACUGAA-3′ and antisense 5′-UUCAGTACCUAUUGGCAU-3′. We confirmed gene silencing in the MCF7 cell line by Western blotting. Cell lysate was prepared using RIPA buffer (Sigma Aldrich, Saint Louis, MO, USA), while protein concentration was measured by a BCA assay kit (Thermo Fisher Scientific, Waltham, MA, USA). Proteins were separated using 12% polyacrylamide TGX gels (Bio-Rad, Hercules, CA, USA) and transferred onto the PVDF membrane by semi-dry transfer (Bio-Rad, Hercules, CA, USA). For detection, we used primary rabbit anti-ERα36 antibody (Cell Applications Inc. San Diego, CA, USA; dilution 1:500), mouse anti-ERα66 antibody (SantaCruz, Dallas, TA, USA; dilution 1:1000), and anti-β-actin antibody (Sigma Aldrich, Saint Louis, MO, USA; dilution 1:10,000). Appropriate, secondary anti-rabbit and anti-mouse HRP-conjugated antibodies were used (Sigma Aldrich, Saint Louis, MO, USA; dilution 1:100,000).
Analysis of ERα36 levels in frozen breast cancer samples with Western blotting was performed according to the protocol described above, apart from the homogenization step, where frozen tumor samples were cut and suspended in RIPA buffer (Sigma Aldrich, Saint Louis, MO, USA); then, tissue was minced using a sterile scalpel and centrifuged at 10,000× g for 10 min in 4 °C. Homogenization was followed by the measurement of protein concentration by a BCA assay kit (Thermo Fisher Scientific, Waltham, MA, USA).
4.8. Immunofluorescent Staining
Cells were seeded on the sterilized cover glass and after 24 h were fixed and permeabilized using a methanol/acetone mix for 15 min. For blocking, 5% BSA in PBS was used. Primary antibodies were diluted in Antibody Diluent (Dako Agilent, Santa Clara, CA, USA) and incubated with cells for 30 min. The following antibodies were used: rabbit anti-ERα36 (Cell Applications Inc. San Diego, CA, US, dilution 1:1000) and mouse anti-ERα clone 1D5 (Dako Agilent, Santa Clara, CA, USA; dilution 1:400). As secondary antibodies, appropriately anti-rabbit IgG DyLight 488 and anti-mouse IgG DyLight 594 were used (Thermo Fisher Scientific, Waltham, MA, USA; dilution 1:2000).
4.9. Wound-Healing Assay
Cells were seeded into non-coated 24-well plates in full medium (DMEM with 10% serum). Wounds were made with a sterile pipette tip after cells reached confluence, and cell debris was washed; then, incubation was performed in DMEM medium with 2% serum in order to inhibit cell proliferation. Wound closure was measured after 24 h using a phase-contrast microscope. Data was analyzed using the “MRI Wound Healing” ImageJ plugin, and statistical analyses (t-test) were performed using MS Office Excel software.
4.10. Cell Cycle Analysis
Cells were seeded on the six-well plates, and after 24 h, siRNA was added to appropriate samples. After 48 h, cells were harvested washed with PBS and fixed with 70% ice-cold ethanol for 30 min. After the depletion of ethanol, the mixture of 10 µg/mL DAPI and 2 mg/mL RNase free from DNases in PBS was added for 15 min. Then, cells were analyzed using a LSR II flow cytometer (BD Biosciences, Franklin Lakes, NJ, USA).
4.11. Statistical Analysis
All the analyses were performed using Statistica version 12 (StatSoft Dell, Round Rock, TA, USA) software. Categorical variables were compared by χ2 test. Continuous variables were compared by Spearman’s rank order test. The Mann–Whitney test was used to examine the differences between continuous values in two groups. Kaplan–Meier curves for disease-free survival were compared using an F-Cox test.
5. Conclusions
We have shown that increased ESR1 gene dosage is found more frequently in ERα66-negative patients. The lack of a full-length ER isoform, ERα66, did not preclude the expression of a short ERα36 variant. The expression of both isoforms was related to patients’ survival, but their effect was opposite—high ERα66 levels conferred good prognosis, whereas high ERα36 conferred poor prognosis. The obtained results underline the complexity of molecular networks in which estrogen receptor is involved. Together with other findings linking the expression of ERα36 to resistance to endocrine therapies, the role of alternative ER isoforms should be further investigated in order to uncover their biological function and clinical utility.
Abbreviations
| ESR1 | estrogen receptor gene |
| ER | estrogen receptor alpha |
| ERα36 | estrogen receptor alpha, 36kDa isoform |
| ERα66 | estrogen receptor alpha, 66kDa isoform |
| FISH | fluorescent in situ hybridization |
| HER2 | human epidermal growth factor receptor |
| IHC | immunohistochemistry |
| PR | progesterone receptor alpha |
| OS | overall survial |
| DFS | disease free survival |
Supplementary Materials
The following are available online at https://www.mdpi.com/1422-0067/20/8/1881/s1. Figure S1: ESR1 gene copy number analysis with by fluorescent in situ hybridization (FISH). Figure S2. Prognostic significance of ERa36 on protein level. Kaplan–Meier survival curves presenting overall survival (OS) according to ERα36 protein level in the nucleus (A) and cytoplasm (B). Division into positive/negative samples was made based on median immunohistochemical score (percentage of positively stained cells x intensity of the staining). Figure S3. Correlation between ERα66 (A,B) and ERα36 (C,D) mRNA expression and ESR1 gene status. ESR1 gene dosage assessed by qPCR (A, C) and ESR1 copy number assessed by FISH (B, D). Figure S4: Correlation between nuclear (A, B) as well as cytoplasmic (C, D) ERα36 levels and ERα66 assessed by immunohistochemistry. ERα36 levels are presented as either a percentage of positive cells (A, C) or immunohistochemical score (percentage of positive cells (0–100%) x staining intensity (0–3); B, D). Figure S5: ERα36 protein levels assessed by Western blot in frozen breast cancer samples and matched FFPE samples using immunohistochemistry. ERα36 low – samples with low total ERα36 immunohistochemical score; ERα36 high – samples with high total ERα36 immunohistochemical score.
Author Contributions
Conceptualization A.N., A.M. and A.Z.; methodology A.N., J.S. (Jolanta Szade), R.S., A.Z., A.M.; formal analysis A.N., A.M.; investigation A.N., J.S. (Jolanta Szade), J.E., M.I., G.S., A.M.; resources M.W.J., J.S. (Jaroslaw Skokowski), J.B, A.Z., A.M.; writing—original draft preparation A.N., A.M.; writing—review and editing A.N., R.S., A.Z., A.M.; visualization A.N., J.S. (Jolanta Szade), M.I., G.S., A.M.; supervision A.M.; project administration A.M.; funding acquisition A.M.”
Funding
This research was funded by the Ministry of Science and Higher Education program “Iuventus Plus” No. IP 2014 028473 between 2015 and 2018.
Conflicts of Interest
The authors declare no conflict of interest.
References
- 1.Orlando L., Schiavone P., Fedele P., Calvani N., Nacci A., Rizzo P., Marino A., D’Amico M., Sponziello F., Mazzoni E., et al. Molecularly targeted endocrine therapies for breast cancer. Cancer Treat Rev. 2010;36:67–71. doi: 10.1016/S0305-7372(10)70023-2. [DOI] [PubMed] [Google Scholar]
- 2.Hammond M.E., Hayes D.F., Dowsett M., Allred D.C., Hagerty K.L., Badve S., Fitzgibbons P.L., Francis G., Goldstein N.S., Hayes M., et al. American Society of Clinical Oncology/College of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. Arch. Pathol. Lab. Med. 2010;134:907–922. doi: 10.1043/1543-2165-134.6.907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Goldhirsch A., Winer E.P., Coates A.S., Gelber R.D., Piccart-Gebhart M., Thürlimann B., Senn H.J., Panel members Personalizing the treatment of women with early breast cancer: Highlights of the St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2013. Ann. Oncol. 2013;24:2206–2223. doi: 10.1093/annonc/mdt303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Holst F., Stahl P.R., Ruiz C., Hellwinkel O., Jehan Z., Wendland M., Lebeau A., Terracciano L., Al-Kuraya K., Jänicke F., et al. Estrogen receptor alpha (ESR1) gene amplification is frequent in breast cancer. Nat. Genet. 2007;39:655–660. doi: 10.1038/ng2006. [DOI] [PubMed] [Google Scholar]
- 5.Moelans C.B., Holst F., Hellwinkel O., Simon R., van Diest P.J. ESR1 amplification in breast cancer by optimized RNase FISH: Frequent but low-level and heterogeneous. PLoS ONE. 2013;8:e84189. doi: 10.1371/journal.pone.0084189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Markiewicz A., Welnicka-Jaskiewicz M., Skokowski J., Jaskiewicz J., Szade J., Jassem J., Zaczek A.J. Prognostic significance of ESR1 amplification and ESR1 PvuII, CYP2C19*2, UGT2B15*2 polymorphisms in breast cancer patients. PLoS ONE. 2013;8:e72219. doi: 10.1371/journal.pone.0072219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ooi A., Inokuchi M., Harada S., Inazawa J., Tajiri R., Kitamura S.S., Ikeda H., Kawashima H., Dobashi Y. Gene amplification of ESR1 in breast cancers-fact or fiction? A fluorescence in situ hybridization and multiplex ligation-dependent probe amplification study. J. Pathol. 2012;227:8–16. doi: 10.1002/path.3974. [DOI] [PubMed] [Google Scholar]
- 8.Moelans C.B., Monsuur H.N., de Pinth J.H., Radersma R.D., de Weger R.A., van Diest P.J. ESR1 amplification is rare in breast cancer and is associated with high grade and high proliferation: A multiplex ligation-dependent probe amplification study. Cell Oncol. (Dordr) 2011;34:489–494. doi: 10.1007/s13402-011-0045-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Holst F. Estrogen receptor alpha gene amplification in breast cancer: 25 years of debate. World J. Clin. Oncol. 2016;7:160–173. doi: 10.5306/wjco.v7.i2.160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zaczek A.J., Markiewicz A., Seroczynska B., Skokowski J., Jaskiewicz J., Pienkowski T., Olszewski W.P., Szade J., Rhone P., Welnicka-Jaskiewicz M., et al. Prognostic Significance of TOP2A Gene Dosage in HER-2-Negative Breast Cancer. Oncologist. 2012;17:1246–1255. doi: 10.1634/theoncologist.2012-0023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wang Z., Zhang X., Shen P., Loggie B.W., Chang Y., Deuel T.F. A variant of estrogen receptor-{alpha}, hER-{alpha}36: Transduction of estrogen- and antiestrogen-dependent membrane-initiated mitogenic signaling. Proc. Natl. Acad. Sci. USA. 2006;103:9063–9068. doi: 10.1073/pnas.0603339103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wang Z., Zhang X., Shen P., Loggie B.W., Chang Y., Deuel T.F. Identification, cloning, and expression of human estrogen receptor-alpha36, a novel variant of human estrogen receptor-alpha66. Biochem Biophys Res. Commun. 2005;336:1023–1027. doi: 10.1016/j.bbrc.2005.08.226. [DOI] [PubMed] [Google Scholar]
- 13.Sołtysik K., Czekaj P. ERα36--Another piece of the estrogen puzzle. Eur. J. Cell. Biol. 2015;94:611–625. doi: 10.1016/j.ejcb.2015.10.001. [DOI] [PubMed] [Google Scholar]
- 14.Chaudhri R.A., Schwartz N., Elbaradie K., Schwartz Z., Boyan B.D. Role of ERα36 in membrane-associated signaling by estrogen. Steroids. 2014;81:74–80. doi: 10.1016/j.steroids.2013.10.020. [DOI] [PubMed] [Google Scholar]
- 15.Wang Q., Jiang J., Ying G., Xie X.Q., Zhang X., Xu W., Song E., Bu H., Ping Y.F., Yao X.H., et al. Tamoxifen enhances stemness and promotes metastasis of ERα36 + breast cancer by upregulating ALDH1A1 in cancer cells. Cell. Res. 2018;28:336–358. doi: 10.1038/cr.2018.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chaudhri R.A., Olivares-Navarrete R., Cuenca N., Hadadi A., Boyan B.D., Schwartz Z. Membrane estrogen signaling enhances tumorigenesis and metastatic potential of breast cancer cells via estrogen receptor-α36 (ERα36) J. Biol. Chem. 2012;287:7169–7181. doi: 10.1074/jbc.M111.292946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhang X.T., Kang L.G., Ding L., Vranic S., Gatalica Z., Wang Z.Y. A positive feedback loop of ER-α36/EGFR promotes malignant growth of ER-negative breast cancer cells. Oncogene. 2011;30:770–780. doi: 10.1038/onc.2010.458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wallacides A., Chesnel A., Ajj H., Chillet M., Flament S., Dumond H. Estrogens promote proliferation of the seminoma-like TCam-2 cell line through a GPER-dependent ERα36 induction. Mol. Cell. Endocrinol. 2012;350:61–71. doi: 10.1016/j.mce.2011.11.021. [DOI] [PubMed] [Google Scholar]
- 19.Shi L., Dong B., Li Z., Lu Y., Ouyang T., Li J., Wang T., Fan Z., Fan T., Lin B., et al. Expression of ER-{alpha}36, a novel variant of estrogen receptor {alpha}, and resistance to tamoxifen treatment in breast cancer. J. Clin. Oncol. 2009;27:3423–3429. doi: 10.1200/JCO.2008.17.2254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhang J., Li G., Li Z., Yu X., Zheng Y., Jin K., Wang H., Gong Y., Sun X., Teng X., et al. Estrogen-independent effects of ER-α36 in ER-negative breast cancer. Steroids. 2012;77:666–673. doi: 10.1016/j.steroids.2012.02.013. [DOI] [PubMed] [Google Scholar]
- 21.Lee L.M., Cao J., Deng H., Chen P., Gatalica Z., Wang Z.Y. ER-alpha36, a novel variant of ER-alpha, is expressed in ER-positive and -negative human breast carcinomas. Anticancer Res. 2008;28:479–483. [PMC free article] [PubMed] [Google Scholar]
- 22.Schwartz N., Verma A., Bivens C.B., Schwartz Z., Boyan B.D. Rapid steroid hormone actions via membrane receptors. Biochim. Biophys Acta. 2016;1863:2289–2298. doi: 10.1016/j.bbamcr.2016.06.004. [DOI] [PubMed] [Google Scholar]
- 23.Soysal S.D., Kilic I.B., Regenbrecht C.R., Schneider S., Muenst S., Kilic N., Güth U., Dietel M., Terracciano L.M., Kilic E. Status of estrogen receptor 1 (ESR1) gene in mastopathy predicts subsequent development of breast cancer. Breast Cancer Res. Treat. 2015;151:709–715. doi: 10.1007/s10549-015-3427-y. [DOI] [PubMed] [Google Scholar]
- 24.Lebeau A., Grob T., Holst F., Seyedi-Fazlollahi N., Moch H., Terracciano L., Turzynski A., Choschzick M., Sauter G., Simon R. Oestrogen receptor gene (ESR1) amplification is frequent in endometrial carcinoma and its precursor lesions. J. Pathol. 2008;216:151–157. doi: 10.1002/path.2405. [DOI] [PubMed] [Google Scholar]
- 25.Tomita S., Zhang Z., Nakano M., Ibusuki M., Kawazoe T., Yamamoto Y., Iwase H. Estrogen receptor alpha gene ESR1 amplification may predict endocrine therapy responsiveness in breast cancer patients. Cancer Sci. 2009;100:1012–1017. doi: 10.1111/j.1349-7006.2009.01145.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Laenkholm A.V., Knoop A., Ejlertsen B., Rudbeck T., Jensen M.B., Muller S., Lykkesfeldt A.E., Rasmussen B.B., Nielsen K.V. ESR1 gene status correlates with estrogen receptor protein levels measured by ligand binding assay and immunohistochemistry. Mol. Oncol. 2012;6:428–436. doi: 10.1016/j.molonc.2012.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lin C.H., Liu J.M., Lu Y.S., Lan C., Lee W.C., Kuo K.T., Wang C.C., Chang D.Y., Huang C.S., Cheng A.L. Clinical significance of ESR1 gene copy number changes in breast cancer as measured by fluorescence in situ hybridisation. J. Clin. Pathol. 2013;66:140–145. doi: 10.1136/jclinpath-2012-200929. [DOI] [PubMed] [Google Scholar]
- 28.Tomita S., Abdalla M.O., Fujiwara S., Matsumori H., Maehara K., Ohkawa Y., Iwase H., Saitoh N., Nakao M. A cluster of noncoding RNAs activates the ESR1 locus during breast cancer adaptation. Nat. Commun. 2015;6:6966. doi: 10.1038/ncomms7966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zaczek A., Markiewicz A., Jaskiewicz J., Pienkowski T., Rhone P., Jassem J., Welnicka-Jaskiewicz M. Clinical evaluation of developed PCR-based method with hydrolysis probes for TOP2A copy number evaluation in breast cancer samples. Clinical. Biochemistry. 2010;43:891–898. doi: 10.1016/j.clinbiochem.2010.04.060. [DOI] [PubMed] [Google Scholar]
- 30.Pagani F., Baralle F.E. Genomic variants in exons and introns: Identifying the splicing spoilers. Nat. Rev. Genet. 2004;5:389–396. doi: 10.1038/nrg1327. [DOI] [PubMed] [Google Scholar]
- 31.Deng H., Yin L., Zhang X.T., Liu L.J., Wang M.L., Wang Z.Y. ER-α variant ER-α36 mediates antiestrogen resistance in ER-positive breast cancer stem/progenitor cells. Pt BJ. Steroid Biochem. Mol. Biol. 2014;144:417–426. doi: 10.1016/j.jsbmb.2014.08.017. [DOI] [PubMed] [Google Scholar]
- 32.Deng H., Zhang X.T., Wang M.L., Zheng H.Y., Liu L.J., Wang Z.Y. ER-α36-mediated rapid estrogen signaling positively regulates ER-positive breast cancer stem/progenitor cells. PLoS ONE. 2014;9:e88034. doi: 10.1371/journal.pone.0088034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Yin L., Pan X., Zhang X.T., Guo Y.M., Wang Z.Y., Gong Y., Wang M. Downregulation of ER-α36 expression sensitizes HER2 overexpressing breast cancer cells to tamoxifen. Am. J. Cancer Res. 2015;5:530–544. [PMC free article] [PubMed] [Google Scholar]
- 34.Zhao Y., Deng C., Lu W., Xiao J., Ma D., Guo M., Recker R.R., Gatalica Z., Wang Z., Xiao G.G. let-7 microRNAs induce tamoxifen sensitivity by downregulation of estrogen receptor α signaling in breast cancer. Mol. Med. 2011;17:1233–1241. doi: 10.2119/molmed.2010.00225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Li L., Wang Q., Lv X., Sha L., Qin H., Wang L. Expression and localization of estrogen receptor in human breast cancer and its clinical significance. Cell Biochem. Biophys. 2015;71:63–68. doi: 10.1007/s12013-014-0163-6. [DOI] [PubMed] [Google Scholar]
- 36.Chamard-Jovenin C., Jung A.C., Chesnel A., Abecassis J., Flament S., Ledrappier S., Macabre C., Boukhobza T., Dumond H. From ERα66 to ERα36: A generic method for validating a prognosis marker of breast tumor progression. BMC Syst. Biol. 2015;9:28. doi: 10.1186/s12918-015-0178-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Li G., Zhang J., Jin K., He K., Zheng Y., Xu X., Wang H., Li Z., Yu X., Teng X., et al. Estrogen receptor-α36 is involved in development of acquired tamoxifen resistance via regulating the growth status switch in breast cancer cells. Mol. Oncol. 2013;7:611–624. doi: 10.1016/j.molonc.2013.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.