Abstract
Enzyme replacement therapy (ERT) with rhGAA has improved clinical outcomes in infantile Pompe disease (IPD). A subset of CRIM-positive IPD patients develop high and sustained antibody titers (HSAT; ≥51,200) and/or sustained intermediate titer (SIT; ≥12,800 and <51,200), similar to CRIM-negative patients. To date there has been no systematic study to analyze the extent of IgG antibody response in CRIM-positive IPD. Such data would be critical and could serve as a comparator group for potential immune modulation approaches. A retrospective analysis of the dataset from the original rhGAA clinical trials final reports was conducted. CRIM-positive patients who received ERT monotherapy and had >6 months of antibody titer data available, were included in the study. Patients were classified based on their longitudinal antibody titers into HSAT, SIT, and low titer (LT; <12,800) groups. Of the 37 patients that met inclusion criteria, five (13%), seven (19%), and 25 (68%) developed HSAT, SIT, and LT, respectively. Median peak titers were 204,800 (51,200–409,600), 25,600 (12,800–51,200), and 800 (200–12,800) for HSAT, SIT, and LT groups, respectively. Median last titers were 102,400 (51,200–409,600), 1600 (200–25,600), and 400 (0–12,800) at median time since ERT initiation of 94 weeks (64–155 weeks), 104 weeks (86–144 weeks), and 130 weeks (38–182 weeks) for HSAT, SIT, and LT groups, respectively. 32% (12/37) of CRIM-positive IPD patients developed HSAT/SIT which may lead to limited ERT response and clinical decline. Further Studies are needed to identify CRIM-positive IPD patients at risk of developing HSAT/SIT, especially with the addition of Pompe disease to the newborn screening.
Keywords: Pompe disease, Glycogen storage disease type II, Neuromuscular disease, Enzyme replacement therapy, Anti-rhGAA Ig antibodies, Antidrug antibodies
Abbreviations: IPD, Infantile Pompe disease; ERT, Enzyme replacement therapy; GAA, Acid α-glucosidase; GAA, Gene encoding acid α-glucosidase; rhGAA, Recombinant human acid α-glucosidase; CRIM, Cross-reactive immunological material; HSAT, High and sustained antibody titers; SIT, Sustained intermediate titers; LT, Low titers; LVMI, Left ventricular mass index; AIMS, Alberta infant motor scale; Glc4, Glucose tetrasaccharide; EOW, Every other week; IgG, Immunoglobulin G; CI-MPR, Cation-independent mannose 6-phosphate receptor; RUSP, Recommended universal screening panel; HLA, Human leukocyte antigen; MHC, Major histocompatibility complex; iTEM, Individualized T-cell epitope measure
1. Introduction
Pompe disease (glycogen storage disease type II, OMIM # 232300) is an autosomal recessive glycogen storage disorder caused by deficiency of lysosomal hydrolyzing enzyme acid α-glucosidase (GAA) [7]. Deficiency of GAA leads to progressive accumulation of lysosomal glycogen in multiple tissues, particularly skeletal, cardiac, and smooth muscles [13]. Infantile Pompe disease (IPD) is a spectrum ranging from “classic” to “non-classic”. All patients with IPD present with cardiomyopathy in the first year of life. Classic IPD, the most severe end of the disease spectrum, presents in first few days to week of life with severe cardiomyopathy and without treatment, patient rarely survive beyond two year of age. Whereas, patients with “non-classic” IPD present in the first year of life with less severe cardiomyopathy, no left ventricular outflow tract obstruction and can survive beyond two years of age without treatment [7,13,22,25].
In 2006, the FDA approved enzyme replacement therapy (ERT) with recombinant human acid α-glucosidase (rhGAA, alglucosidase alfa). Advent of ERT with alglucosidase alfa has improved clinical outcomes and prolonged overall and ventilator-free survival in patients with IPD [9,10,16]. Despite improved cohort outcomes, individual response to ERT is heterogeneous and influenced by many factors such as age at treatment initiation, cross-reactive immunological material (CRIM) status, high and sustained antibody titers (HSAT), sustained intermediate titers (SIT), extent of preexisting pathology, muscle fiber type involvement, ACE genotype, and ACTN genotype [3,5,17,21].
Previous studies have demonstrated that CRIM status is an important predictor of clinical response to ERT [3,12]. CRIM-negative IPD patients with deleterious null GAA variants have poor clinical outcomes due to development of HSAT [12]. The majority of CRIM-negative patients on ERT develop anti-rhGAA IgG antibodies and are ventilator-dependent or deceased by age 27.1 months [3,4,12]. CRIM-positive patients with some endogenous GAA typically maintain none to low anti-rhGAA IgG antibody titers and, as a cohort, have better response to ERT. Kishnani et al. reported that by week 52 on ERT, 4.8% of CRIM-positive IPD patients were deceased or invasively ventilated compared to 54.5% of CRIM-negative IPD patients [12]. However, it is recognized that a subset of CRIM-positive IPD patients develop anti-rhGAA IgG antibodies similar to CRIM-negative patients with equally poor clinical outcomes. Banugaria et al. showed that a subset (~40%) of CRIM-positive IPD patients developed an immune response similar to CRIM-negative patients and had poor clinical outcomes; measured as overall survival, ventilator-free survival, left ventricular mass index (LVMI), Alberta Infant Motor Scale (AIMS), and urinary glucose tetrasaccharide (Glc4) [3]. In this study, we aimed to systematically assess and characterize anti-rhGAA IgG antibody response in all CRIM-positive IPD patients from all previous rhGAA clinical trials, who had received ERT without immune modulation.
2. Methods
A retrospective analysis of all CRIM-positive IPD patients from alglucosidase alfa clinical trials (GAA-CL-001, GAA-CL-002, AGLU-008-01, AGLU-001-00, AGLU-009-02, AGLU01702, AGLU02203, AGLU01602/AGLU02403) was performed [1,9,10,14,16]. The datasets included in the final reports at the end of clinical trials were reviewed for data extraction and data analysis. Patients were selected based on the following criteria; 1) a confirmed diagnosis of Pompe disease as described previously [13,25], 2) CRIM-positive status determined as described previously [2], 3) received ERT with alglucosidase alfa at a cumulative dose of 20 mg/kg or 40 mg/kg every other week (EOW) without immune modulation, and 4) availability of at least 6 months of anti-rhGAA IgG antibody titers since initiation of ERT.
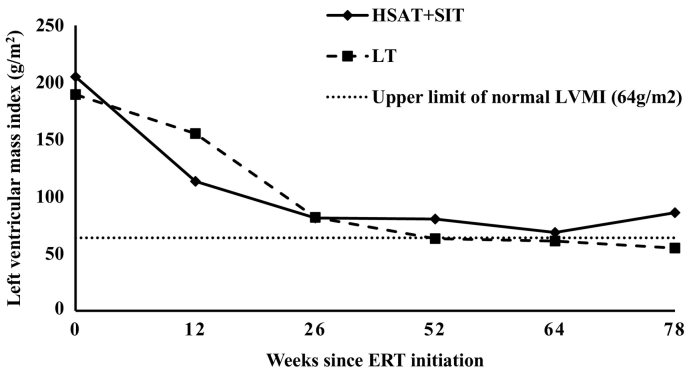
Data analyzed included age at ERT initiation, pathogenic GAA variant data, ERT dose and frequency, left ventricular mass index (LVMI), and longitudinal anti-rhGAA IgG antibody titers of all qualifying CRIM-positive IPD patients. Patients were classified into three groups based on anti-rhGAA IgG antibody titers; 1) high and sustained antibody titer (HSAT), defined as titers of ≥51,200 on two or more occasions at or beyond 6 months on ERT [3], 2) sustained intermediate titer (SIT), defined as titers of ≥12,800 and <51,200 within 12 months on ERT (Lumizyme Package Insert) [15], and 3) low titer (LT), defined as titers of <12,800 within 12 months on ERT. Age at ERT initiation, pathogenic GAA variants, time to seroconversion, peak anti-rhGAA IgG titers, and last available anti-rhGAA IgG titers were compared among HSAT, SIT, and LT groups. The anti-rhGAA IgG antibody titer data were analyzed to find the first time point when a patient developed titer of ≥12,800. Left ventricular mass index (LVMI) was analyzed to investigate effects of antibody titers of ≥12,800 on efficacy of ERT. LVMI of patients in HSAT and SIT groups were combined for comparison with patients in LT group. Median LVMI at week 0, 26, 52, and 78 were compared between HSAT+SIT group versus LT group (Fig. 2).
Fig. 2.
Comparison of left ventricular mass index in HSAT + SIT group versus LT group. ERT, enzyme replacement therapy; HSAT, high and sustained titer; SIT, sustained intermediate titer; LT, low titer; LVMI, left ventricular mass index.
CRIM-status was determined by western blot analysis and correlated with GAA genotypes for patients in this cohort. Anti-rhGAA IgG antibody testing was performed by Sanofi Genzyme (Framingham, MA) using enzyme-linked immunosorbent assays and confirmed using radio immunoprecipitation at baseline, week 4, week 8, week 12, week 16, week 20, week 24, week 38, week 52, and week 64 since ERT initiation.
Age at ERT initiation and LVMI were compared using Wilcoxon/Kruskal-Wallis rank-sum test. Frequency of GAA variants were compared using Fisher's exact test. Analyses were performed with STATA version (StataCorp. LP). Descriptive data are presented as medians.
3. Results
3.1. Patient demographics
Within the entire IPD cohort (n = 83) from the various alglucosidase alfa clinical trials, 48 were identified as CRIM-positive, 14 were CRIM-negative, and for 21 patients CRIM status unknown. Of the 48 CRIM-positive IPD patients, 37 patients met inclusion criteria. Patient demographics and pathogenic GAA variant data are presented in Table 1. Five (13%), seven (19%), and 25 (68%) developed HSAT, SIT, and LT, respectively. Median age at ERT initiation was 7.0 months (range, 5.0–8.4 months), 4.6 months (range, 1.9–9.3 months), and 6.9 months (range, 0.5–43.1 months) in HSAT, SIT and LT groups, respectively. There was no significant difference (p = 2700) in age at ERT initiation between the three groups. Of these 37 CRIM-positive IPD patients, 84% (31/37) and 16% (6/37) patients receive ERT at cumulative dose of 20 mg/kg every other week (EOW) and 40 mg/kg EOW, respectively. In 20 mg/kg EOW group (n = 31), 71% (22/31) patients maintained LT and 29% (9/31) developed HSAT/SIT. In 40 mg/kg EOW group (n = 6), 50% (3/6) patients maintained LT and 50% (3/6) developed HSAT/SIT. We did not observe any correlation between ERT dosing and development of anti-rhGAA IgG antibodies.
Table 1.
Demographics and GAA variant data.
| ID/Gender | Age at ERT initiation (months) | Complementary DNA change |
GAA Variant type |
GAA Variant effecta |
ERT dose (mg/kg) (every other week) | |||
|---|---|---|---|---|---|---|---|---|
| Amino acid change | ||||||||
| GAA Variant 1 | GAA Variant 2 | GAA Variant 1 | GAA Variant 2 | GAA Variant 1 | GAA Variant 2 | |||
| HSAT1/M | 8.0 | c.2560C>T | c.1129G>C | Nonsense | Missense | Very severe | Potentially less severe | 20 |
| p.Arg854X | p.Gly377Arg | |||||||
| HSAT2/F | 8.4 | c .2189+459_3405del | c.2012T>G | Large deletion | Missense | Potentially severe | Potentially less severe | 20 |
| p.Glu730_Cys952del | p.Met671Arg | |||||||
| HSAT3/M | 5.0 | c.437delT | c.2481+102_2646+31del | Frameshift deletion | In-frame deletion | Very severe | Very severe | 40 |
| p.Met146ArgfsX7 | p.Gly828_Asn882del | |||||||
| HSAT4/M | 5.4 | c.2481+102_2646+31del | c.2481+102_2646+31del | In-frame deletion | In-frame deletion | Very severe | Very severe | 40 |
| p.Gly828_Asn882del | p.Gly828_Asn882del | |||||||
| HSAT5/F | 7.0 | c.2804T>C | c.2804T>C | Missense | Missense | Potentially less severe | Potentially less severe | 20 |
| p.Leu935Pro | p.Leu935Pro | |||||||
| SIT1/M | 4.6 | c.2238G>A | c.1843G>A | Nonsense | Missense | Very severe | Potentially less severe | 20 |
| p.Trp746X | p.Gly615Arg | |||||||
| SIT2/M | 3.1 | c.1933G>A | c.1933G>A | Missense | Missense | Potentially less severe | Potentially less severe | 20 |
| p.Asp645Asn | p.Asp645Asn | |||||||
| SIT3/F | 2.9 | Not available | Not available | Not available | Not available | Not available | Not available | 20 |
| SIT4/M | 7.0 | c.1064T>C | c.1064T>C | Missense | Missense | Potentially less severe | Potentially less severe | 20 |
| p.Leu355Pro | p.Leu355Pro | |||||||
| SIT5/M | 7.3 | c.2297A>C | c.2297A>C | Missense | Missense | Potentially less severe | Potentially less severe | 40 |
| p.Tyr766Ser | p.Tyr766Ser | |||||||
| SIT6/M | 1.9 | c.2741_2742delAG; c.2743_2746insCAGG | c.2741_2742delAG; c.2743_2746insCAGG | Frameshift deletion | Frameshift deletion | Very severe | Very severe | 20 |
| p.Gln914ProfsX30 | p.Gln914ProfsX30 | |||||||
| SIT7/M | 9.3 | c.1655T>C | c.2237G>A | Missense | Nonsense | Potentially less severe | Very severe | 20 |
| p.Leu552Pro | p.Trp746X | |||||||
| LT1/M | 2.4 | c.1438-1G>T | c. 1655T>C | Splicing mutation | Missense | Potentially less severe | Potentially less severe | 20 |
| p.Lue552Pro | ||||||||
| LT2/F | 5.7 | Not available | Not available | Not available | Not available | Not available | Not available | 20 |
| LT3/M | 5.9 | c.872T>C | c.872T>C | Missense | Missense | Potentially less severe | Potentially less severe | 20 |
| p.Leu291Pro | p.Leu291Pro | |||||||
| LT4/M | 6.9 | c.1710C>G | c.2560C>T | Missense | Nonsense | Potentially less severe | Very severe | 40 |
| p.Asn570Lys | p.Arg854X | |||||||
| LT5/M | 4.3 | c.1465G>A | c.40_47delGCCGTCTG | Missense | Frameshift deletion | Potentially less severe | Very severe | 40 |
| p.Asp489Asn | p.Ala14ArgfsX18 | |||||||
| LT6/F | 5.3 | c.1802C>T | c.1099T>C | Missense | Missense | Potentially less severe | Potentially less severe | 20 |
| p.Ser601Leu | p.Trp367Arg | |||||||
| LT7/M | 6.4 | c.1935C>A | c.1194+2T>C | Missense | Splice site | Potentially less severe | Very severe | 20 |
| p.Asp645Glu | ||||||||
| LT8/F | 6.9 | c.2560C>T | c.1979G>A | Nonsense | Missense | Very severe | Potentially less severe | 20 |
| p.Arg854X | p.Arg660His | |||||||
| LT9/F | 17.0 | c.670C>T | c.925G>A | Missense | Missense | Less severe | Potentially less severe | 20 |
| p.Arg224Trp | p.Gly309Arg | |||||||
| LT10/M | 37.3 | c.1556T>C | c.1441T>C | Missense | Missense | Potentially less severe | Potentially less severe | 20 |
| p.Met519Thr | p.Trp481Arg | |||||||
| LT11/M | 8.2 | c.1735G>A | c.655G>A | Missense | Missense | Potentially less severe | Potentially less severe | 20 |
| p.Glu579Lys | p.Gly219Arg | |||||||
| LT12/M | 16.2 | c.525delT | c.1448G>T | Frameshift deletion | Missense | Very severe | Potentially less severe | 20 |
| p.Glu176ArgfsX45 | p.Gly483Val | |||||||
| LT13/F | 43.1 | c.3G>A | c.923A>C | Initiator codon | Missense | Very severe | Potentially less severe | 20 |
| p.Met1? | p.His308Pro | |||||||
| LT14/M | 36.6 | c.1064T>C | c.1210G>A | Missense | Missense | Potentially less severe | Potentially less severe | 20 |
| p.Leu355Pro | p.Asp404Asn | |||||||
| LT15/M | 9.8 | c.2560C>T | c.1933G>A | Nonsense | Missense | Very severe | Potentially less severe | 20 |
| p.Arg854X | p.Asp645Asn | |||||||
| LT16/F | 24.1 | c.796C>T | c.1316T>A | Missense | Missense | Potentially less severe | Potentially less severe | 20 |
| p.Pro266Ser | p.Met439Lys | |||||||
| LT17/M | 15.0 | c.1655T>C | c.2560C>T | Missense | Nonsense | Potentially less severe | Very severe | 20 |
| p.Leu552Pro | p.Arg854X | |||||||
| LT18/M | 18.1 | c.1978C>T | c.784G>A | Missense | Missense | Potentially less severe | Potentially less severe | 20 |
| p.Arg660Cys | p.Glu262Lys | |||||||
| LT19/F | 14.3 | c.1040C>G | c.1003G>A | Missense | Missense | Potentially less severe | Potentially less severe | 20 |
| p.Pro347Arg | p.Gly335Arg | |||||||
| LT20/F | 17.8 | c.670C>T | c.670C>T | Missense | Missense | Less severe | Less severe | 20 |
| p.Arg224Trp | p.Arg224Trp | |||||||
| LT21/M | 0.5 | c.525delT | c.1642G>T, c.1880C>T | Frameshift deletion | Missense | Very severe | Potentially less severe | 20 |
| p.Glu176ArgfsX45 | p.Val548Phe; p.Ser627Phe | |||||||
| LT22/M | 2.9 | c.1933G>A | c.1933G>A | Missense | Missense | Potentially less severe | Potentially less severe | 20 |
| p.Asp645Asn | p.Asp645Asn | |||||||
| LT23/F | 6.0 | c.1210G>A | c.2481+102_2646+31del | Missense | In-frame deletion | Potentially less severe | Very severe | 20 |
| p.Asp404Asn | p.Gly828_Asn882del | |||||||
| LT24/F | 5.1 | Not available | Not available | Not available | Not available | Not available | Not available | 20 |
| LT25/M | 1.2 | c.1210G>A | c.1064T>C | Missense | Missense | Potentially less severe | Potentially less severe | 40 |
| p.Asp404Asn | p.Leu355Pro | |||||||
ERT, enzyme replacement therapy; M, male; F, female; HSAT, high and sustained antibody titer; SIT, sustained intermediate titer; LT, low titer; GAA, gene encoding acid α-glucosidase.
Data from Erasmus MC Rotterdam; http://www.pompecenter.nl.; published literature.
3.2. GAA genotype in HSAT, SIT and LT groups
GAA genotype data was available on 34 patients. A total of sixty-eight pathogenic variants were identified with 44 unique GAA variants (Table 1). The frequency of in-frame deletion variants was significantly higher in HSAT group (30%) as compared to LT group (2%) (p = 0012) (Fig. 1). A large deletion (patient HSAT2), frameshift variants (patient SIT6) and in-frame deletion (patient HSAT4) variants in homozygosity, and/or in combination with another variant (patient HSAT3) as compound heterozygotes were exclusively seen in HSAT and SIT groups (Table 1, Fig. 1). Missense variants in homozygosity were noted in 12 LT patients, one HSAT and three SIT patients, with no statistically significant difference (p = 3991) (Table 1, Fig. 1).
Fig. 1.
Frequency of GAA variants in HSAT, SIT, and LT groups. HSAT, high and sustained antibody titer; SIT, sustained intermediate titer; LT, low titer.
3.3. Left ventricular mass index in HSAT+SIT versus LT groups
At baseline, median LVMI for HSAT + SIT and LT group was 205.0 g/m2 (range, 99.5–352.0 g/m2) and 189.4 g/m2 (range, 54.9–417.6 g/m2), respectively with no statistically significant difference (p = 4218). Continued decline in LVMI was observed in HSAT + SIT (median LVMI = 81.3 g/m2; range, 61.1–149.7 g/m2) and LT (median LVMI = 81.8 g/m2; range, 33.0–238.2 g/m2) up to week 26 post ERT initiation. From week 26 to week 52, median LVMI in LT group continued to decrease (median LVMI = 63.2 g/m2; range, 41.0–211.6 g/m2) where as in HSAT + SIT group LVMI plateaued (median LVMI = 80.4 g/m2; range, 45.0–113.4 g/m2). From week 52 to 78, LVMI in LT group improved further (median LVMI = 54.95 g/m2; range, 40.8–140.6 g/m2) whereas LVMI worsened in HSAT + SIT group (median LVMI = 85.99 g/m2; range, 50.2–159.5 g/m2). There was no statistically significant difference in median LVMI between these two groups at baseline (p = 4218), week 26 (p = 7831), and week 52 (p = 3960) but there was a statistically significant difference at week 78 (p = 0041), with LVMI significantly greater in the HSAT + SIT group compared to the LT group (Fig. 2).
3.4. Immune response in HSAT, SIT, and LT groups
Individual peak anti-rhGAA IgG antibody titer, last available anti-rhGAA IgG antibody titer, and time to seroconversion are presented in Table 2. Antibody data points varied from patient to patient with median number of data points available per patient being 15 (range, 6–44). Two patients (patients LT24 and LT25) in the LT group did not seroconvert. All patients (n = 5) in HSAT group seroconverted by 4 weeks after initiation of ERT. Median time to seroconversion was 4 weeks (range, 4–8 weeks; n = 7) and 8 weeks (range, 4–64 weeks; n = 23) since initiation of ERT for SIT and LT groups, respectively. In HSAT group (n = 5), median peak titer was 204,800 (range, 51,200–409,600) at median time since ERT initiation of 82 weeks (range, 24–130 weeks) and median last available titer was 102,400 (range, 51,200–409,600) at median time since ERT initiation of 94 weeks (range, 64–155 weeks). In SIT group (n = 7), median peak titer was 25,600 (range, 12,800–51,200) at median time since ERT initiation of 12 weeks (range, 8–24 weeks) and median last available titer was 1600 (range, 200–25,600) at median time since ERT initiation of 104 weeks (range, 86–144 weeks). In LT group (n = 23), median peak titer was 800 (range, 200–12,800) at median time since ERT initiation of 38 weeks (range, 8–172 weeks) and median last available titer was 400 (range, 0–12,800) at median time since ERT initiation of 130 weeks (range, 38–182 weeks).
Table 2.
Anti-rhGAA IgG antibody response in HSAT, SIT, and LT groups.
| ID | Time to Seroconversion (weeks) | Peak antibody titer | Time since ERT at peak titer (weeks) | Last available antibody titer | Time since ERT at peak titer (weeks) | Time on ERT (weeks) at antibody titer of >12,800 |
|---|---|---|---|---|---|---|
| HSAT1 | 4 | 102,400 | 24 | 102,400 | 93 | 4 |
| HSAT2 | 4 | 204,800 | 24 | 102,400 | 64 | 8 |
| HSAT3 | 4 | 204,800 | 119 | 102,400 | 146 | 12 |
| HSAT4 | 4 | 409,600 | 82 | 409,600 | 94 | 8 |
| HSAT5 | 4 | 51,200 | 130 | 51,200 | 155 | 8 |
| SIT1 | 4 | 25,600 | 12 | 800 | 101 | 8 |
| SIT2 | 8 | 51,200 | 24 | 1600 | 91 | 12 |
| SIT3 | 4 | 25,600 | 8 | 25,600 | 86 | 4 |
| SIT4 | 4 | 12,800 | 12 | 200 | 144 | 12 |
| SIT5 | 4 | 25,600 | 24 | 1600 | 119 | 24 |
| SIT6 | 8 | 12,800 | 16 | 400 | 122 | 16 |
| SIT7 | 4 | 12,800 | 12 | 3200 | 104 | 12 |
| LT1 | 4 | 12,800 | 172 | 12,800 | 182 | N/A |
| LT2 | 4 | 6400 | 20 | 200 | 52 | N/A |
| LT3 | 64 | 1600 | 130 | 1600 | 130 | N/A |
| LT4 | 8 | 3200 | 8 | 0 | 130 | N/A |
| LT5 | 12 | 400 | 20 | 200 | 122 | N/A |
| LT6 | 8 | 800 | 82 | 200 | 122 | N/A |
| LT7 | 8 | 800 | 118 | 100 | 129 | N/A |
| LT8 | 8 | 400 | 38 | 200 | 105 | N/A |
| LT9 | 12 | 800 | 38 | 400 | 168 | N/A |
| LT10 | 12 | 6400 | 38 | 1600 | 168 | N/A |
| LT11 | 4 | 3200 | 52 | 1600 | 155 | N/A |
| LT12 | 4 | 3200 | 8 | 400 | 156 | N/A |
| LT13 | 4 | 6400 | 8 | 1600 | 156 | N/A |
| LT14 | 8 | 12,800 | 78 | 6400 | 156 | N/A |
| LT15 | 8 | 400 | 8 | 0 | 156 | N/A |
| LT16 | 38 | 400 | 52 | 400 | 52 | N/A |
| LT17 | 8 | 400 | 134 | 200 | 143 | N/A |
| LT18 | 8 | 3200 | 52 | 800 | 132 | N/A |
| LT19 | 4 | 800 | 12 | 400 | 120 | N/A |
| LT20 | 8 | 400 | 12 | 0 | 104 | N/A |
| LT21 | 12 | 200 | 16 | 200 | 52 | N/A |
| LT22 | 64 | 400 | 64 | 400 | 78 | N/A |
| LT23 | 8 | 400 | 20 | 400 | 38 | N/A |
| LT24 | N/A | 0 | 106 | 0 | 106 | N/A |
| LT25 | N/A | 0 | 104 | 0 | 104 | N/A |
ERT, enzyme replacement therapy; N/A, not applicable; HSAT, high and sustained titer; SIT, sustained intermediate titer; LT, low titer.
3.5. Time since ERT initiation for development of deleterious immune response
Out of 37 patients, 12 patients developed titers >12,800 so time on ERT at antibody is provided for these 12 patients (Table 2); whereas, 25 patients did not develop titers >12,800 so time on ERT for antibody titer >12,800 was not applicable for these 25 patients. Twelve patients (32%) in HSAT and SIT groups developed titers of ≥12,800 at median time since ERT initiation of 10 weeks (range, 4–24 weeks) as shown in Table 2 below. In HSAT group, median time since ERT for patients to develop titer of ≥12,800 for the first time was 8 weeks (range, 4–12 weeks) compared to 12 weeks (range, 4–24 weeks) in SIT group. Patients who developed HSAT or SIT showed upward trend in anti-rhGAA antibody titers within 24 weeks on ERT.
4. Discussion
Enzyme replacement therapy (ERT) with alglucosidase alfa has led to prolonged survival and improved clinical outcomes in patients with IPD. Subsequent studies in patients with IPD have identified various factors that negatively impact clinical response to ERT, including CRIM-negative status and the development of HSAT [3,12]. Despite overall better prognosis of CRIM-positive patients as compared to CRIM-negative status patients, a subset of CRIM-positive IPD patients develop HSAT resulting in a poor clinical response to treatment [3,17]. As stated in the Lumizyme® package insert, development of anti-rhGAA IgG antibody titers of ≥12,800 can lead to a 50% increase in clearance of ERT from week 1 to week 12 (Lumizyme Package Insert). This systematic evaluation of a large cohort of CRIM-positive IPD patients is the first ever study, to our knowledge, to assess the extent of immunogenicity in patients with CRIM-positive IPD. In our cohort of 37 CRIM-positive IPD patients, 32% developed HSAT or SIT, which was significantly associated with poor clinical outcome as measured by LVMI.
Previous studies in Pompe mice demonstrated that a large fraction of infused rhGAA is diverted to liver instead of target tissues- cardiac and skeletal muscles [19,20]. In addition, uptake of alglucosidase alfa is substantially higher in cardiac muscle compared to skeletal muscle and uptake of alglucosidase alfa in skeletal muscle also varies within different muscle types [19,20]. Pompe mice model also showed that limited uptake of ERT was sufficient to clear the glycogen in the heart and diaphragm, however, same level of enzyme activity was ineffective in clearing glycogen from skeletal muscle completely. This very low uptake of alglucosidase alfa can be attributed to low levels of cation-independent mannose 6-phosphate receptor (CI-MPR) in skeletal muscles as compared to cardiac and other fast twitch muscles. Studies in Pompe mice showed that only a small fraction of infused ERT reaches the skeletal muscle and any further reduction will likely impact the clinical outcome. As previously reported development of antibodies can affect the clearance of ERT and further reduce the fraction of alglucosidase alfa that reaches the target tissue. Patients with IPD who develop HSAT/SIT have a poorer clinical response to ERT as antibody titers increase. Minimizing immunogenicity to ERT is important to avoid any further reduction of already low uptake of alglucosidase alfa in skeletal muscle.
We reviewed the GAA variants and variant combinations reported in patients who developed HSAT, SIT and LT and found that development of anti-rhGAA IgG antibodies cannot be predicted based on GAA variants alone. However, statistically significant trends in frequency of specific GAA variants and GAA variant combinations were identified. Although in-frame deletions and frameshift deletion variants were present in all three groups, the combination of variants differed between the three groups. Homozygous or compound heterozygous frameshift deletion variants, in-frame deletions and large deletion were exclusively seen in HSAT and SIT groups. In contrast, in the LT group, frameshift deletion and in-frame deletions were only seen in heterozygosity in combination with missense variants. Frameshift deletions, in-frame deletions, and large deletions significantly change the amino acid sequence of endogenous GAA which may lead to variations and changes in the immune response to ERT in these patients, that is, immune system failing to recognize alglucosidase alfa as a self-protein. This may be the driving force behind the development of high antibody titers observed in these patients. Although many missense mutations were common to the three groups (HSAT, SIT, LT) but location of the variant within the protein may play a crucial role in eliciting an immune response in CRIM-positive IPD patients, as has been reported before [23,24].
With inclusion of Pompe disease in recommended universal screening panel (RUSP) for newborn screening and the knowledge that early ERT (within first month of life) does not prevent development of HSAT [11,18], it is of paramount importance to identify patients who are at risk for developing significantly high antibody titers prior to the onset of treatment, as prophylactic immune modulation strategies are already in place. This further emphasizes the need of a method to predict which CRIM-positive IPD patients will mount a significant immune response to ERT. The use of tools based on omics, human leukocyte antigen (HLA) type, and T-cell epitopes may help identify CRIM-positive patients at higher risk of developing anti-rhGAA IgG antibodies against ERT [6]. Major histocompatibility complex (MHC) class II molecules, known as HLA in humans, are found on antigen-presenting cells. HLA plays an important role in cellular immune response by presenting specific foreign peptide to helper T-cells. Epitope-specific T-cells, in turn, help to regulate antibody class switching and affinity maturation in B cells. One novel method, individualized T-cell epitope measure (iTEM), utilizes GAA genotype and HLA haplotype to identify the patients with Pompe disease at risk of developing significant antibody titers to alglucosidase alfa [6]. This algorithm identifies T-cell epitopes within alglucosidase alfa sequence that are absent in patient's endogenous GAA to calculate a score to identify high-risk patients. This unique approach can enable clinician in identification of patients with IPD who are at high-risk of developing anti-rhGAA IgG antibodies at the outset and implementation of immunomodulation at ERT initiation.
Immune tolerance induction (ITI) protocols have shown to be effective in the ERT-naïve setting and also in patients who develop HSAT; however, the latter has been challenging, requiring a more extensive protocol administered over a prolonged duration. It has become increasingly clear that CRIM-negative patients, while an important subgroup, represent only a fraction of Pompe patients at risk for an immune response to ERT. This study adds to the knowledge of the extent of immunogenicity in CRIM-positive patients with IPD. To our knowledge this is the largest cohort of CRIM-positive IPD patients systematically studied to characterize the immune response. Further studies on a larger cohort of CRIM-positive IPD patients are needed to better our understanding of immune responses and association with GAA variants, if any. With immune modulation strategies tailored for CRIM-positive patients [8] and prediction tools to identify the high-risk patients on the horizon [6], this cohort of CRIM-positive IPD patients on ERT monotherapy can serve as a nice comparative group to measure the efficacy of ITI approaches.
Authors' contributions
Conception and study design were performed by AKD, ZBK, and PSK. All authors participated in analysis and interpretation of the data. All authors provided substantive revisions to the manuscript, approved the final version for submission, contributed to journal selection, and accept responsibility for the integrity of the published work.
Ethics approval and consent to participate
The retrospective analysis of deidentified data from alglucosidase alfa clinical trials was conducted as part of Duke University Institutional Review Board approved protocol (Pro00010830; Pompe long-term follow-up study).
Conflict of interest disclosures
AKD has received grant support from Sanofi Genzyme. ZBK has received grant support from Sanofi Genzyme. DSB has received research grant support and travel funds from Genzyme Sanofi, Baebies Inc., Biomarin, and Alexion Inc. PSK has received grant support from Sanofi Genzyme, Valerion Therapeutics, Shire Pharmaceuticals, and Amicus Therapeutics. PSK has received consulting fees and honoraria from Sanofi Genzyme, Shire Pharmaceuticals, Amicus Therapeutics, Vertex Pharmaceuticals, and Asklepios BioPharmaceutical, Inc. (AskBio). PSK is a member of the Pompe and Gaucher Disease Registry Advisory Board for Sanofi Genzyme. PSK has equity in Actus Therapeutics, which is developing gene therapy for Pompe disease.
Funding
No external funding source.
Acknowledgement
The authors acknowledge editorial assistance of Heidi Cope, MS, CGC of Duke University Medical Center.
References
- 1.Amalfitano A., Bengur A.R., Morse R.P., Majure J.M., Case L.E., Veerling D.L., Mackey J., Kishnani P., Smith W., McVie-Wylie A., Sullivan J.A., Hoganson G.E., Phillips J.A., III, Schaefer G.B., Charrow J., Ware R.E., Bossen E.H., Chen Y.T. Recombinant human acid alpha-glucosidase enzyme therapy for infantile glycogen storage disease type II: results of a phase I/II clinical trial. Genet Med. 2001;3(2):132–138. [PubMed] [Google Scholar]
- 2.Bali D.S., Goldstein J.L., Banugaria S., Dai J., Mackey J., Rehder C., Kishnani P.S. Predicting cross-reactive immunological material (CRIM) status in Pompe disease using GAA mutations: lessons learned from 10 years of clinical laboratory testing experience. Am. J. Med. Genet. C: Semin. Med. Genet. 2012;160C(1):40–49. doi: 10.1002/ajmg.c.31319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Banugaria S.G., Prater S.N., Ng Y.K., Kobori J.A., Finkel R.S., Ladda R.L., Chen Y.T., Rosenberg A.S., Kishnani P.S. The impact of antibodies on clinical outcomes in diseases treated with therapeutic protein: lessons learned from infantile Pompe disease. Genet Med. 2011;13(8):729–736. doi: 10.1097/GIM.0b013e3182174703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Berrier K.L., Kazi Z.B., Prater S.N., Bali D.S., Goldstein J., Stefanescu M.C., Rehder C.W., Botha E.G., Ellaway C., Bhattacharya K., Tylki-Szymanska A., Karabul N., Rosenberg A.S., Kishnani P.S. CRIM-negative infantile Pompe disease: characterization of immune responses in patients treated with ERT monotherapy. Genet Med. 2015;17(11):912–918. doi: 10.1038/gim.2015.6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.De Filippi P., Saeidi K., Ravaglia S., Dardis A., Angelini C., Mongini T., Morandi L., Moggio M., Di Muzio A., Filosto M., Bembi B., Giannini F., Marrosu G., Rigoldi M., Tonin P., Servidei S., Siciliano G., Carlucci A., Scotti C., Comelli M., Toscano A., Danesino C. Genotype-phenotype correlation in Pompe disease, a step forward. Orphanet J Rare Dis. 2014;9:102. doi: 10.1186/s13023-014-0102-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.De Groot A.S., Kazi Z.B., Martin R.F., Terry F.E., Desai A.K., Martin W.D., Kishnani P.S. HLA- and genotype-based risk assessment model to identify infantile onset pompe disease patients at high-risk of developing significant anti-drug antibodies (ADA) Clin. Immunol. 2019;200:66–70. doi: 10.1016/j.clim.2019.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hirschhorn R., Reuser A.J.J. Glycogen storage disease type II: acid a-glucosidase (acid maltase) deficiency. In: Valle D., Scriver C.R., editors. Scriver's OMMBID the Online Metabolic & Molecular Bases of Inherited Disease. McGraw-Hill; New York: 2009. [Google Scholar]
- 8.Kazi Z.B., Desai A.K., Troxler R.B., Kronn D., Packman S., Sabbadini M., Rizzo W.B., Scherer K., Abdul-Rahman O., Tanpaiboon P., Nampoothiri S., Gupta N., Feigenbaum A., Niyazov D.M., Sherry L., Segel R., McVie-Wylie A., Sung C., Joseph A.M., Richards S., Kishnani P.S. An immune tolerance approach using transient low-dose methotrexate in the ERT-naive setting of patients treated with a therapeutic protein: experience in infantile-onset Pompe disease. Genet Med. 2018;21:887–895. doi: 10.1038/s41436-018-0270-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kishnani P.S., Corzo D., Leslie N.D., Gruskin D., Van der Ploeg A., Clancy J.P., Parini R., Morin G., Beck M., Bauer M.S., Jokic M., Tsai C.E., Tsai B.W., Morgan C., O'Meara T., Richards S., Tsao E.C., Mandel H. Early treatment with alglucosidase alpha prolongs long-term survival of infants with Pompe disease. Pediatr. Res. 2009;66(3):329–335. doi: 10.1203/PDR.0b013e3181b24e94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kishnani P.S., Corzo D., Nicolino M., Byrne B., Mandel H., Hwu W.L., Leslie N., Levine J., Spencer C., McDonald M., Li J., Dumontier J., Halberthal M., Chien Y.H., Hopkin R., Vijayaraghavan S., Gruskin D., Bartholomew D., van der Ploeg A., Clancy J.P., Parini R., Morin G., Beck M., De la Gastine G.S., Jokic M., Thurberg B., Richards S., Bali D., Davison M., Worden M.A., Chen Y.T., Wraith J.E. Recombinant human acid [alpha]-glucosidase: major clinical benefits in infantile-onset Pompe disease. Neurology. 2007;68(2):99–109. doi: 10.1212/01.wnl.0000251268.41188.04. [DOI] [PubMed] [Google Scholar]
- 11.Kishnani P.S., Dickson P.I., Muldowney L., Lee J.J., Rosenberg A., Abichandani R., Bluestone J.A., Burton B.K., Dewey M., Freitas A., Gavin D., Griebel D., Hogan M., Holland S., Tanpaiboon P., Turka L.A., Utz J.J., Wang Y.M., Whitley C.B., Kazi Z.B., Pariser A.R. Immune response to enzyme replacement therapies in lysosomal storage diseases and the role of immune tolerance induction. Mol. Genet. Metab. 2016;117(2):66–83. doi: 10.1016/j.ymgme.2015.11.001. [DOI] [PubMed] [Google Scholar]
- 12.Kishnani P.S., Goldenberg P.C., DeArmey S.L., Heller J., Benjamin D., Young S., Bali D., Smith S.A., Li J.S., Mandel H., Koeberl D., Rosenberg A., Chen Y.T. Cross-reactive immunologic material status affects treatment outcomes in Pompe disease infants. Mol. Genet. Metab. 2010;99(1):26–33. doi: 10.1016/j.ymgme.2009.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kishnani P.S., Hwu W.L., Mandel H., Nicolino M., Yong F., Corzo D., G. Infantile-Onset Pompe Disease Natural History Study A retrospective, multinational, multicenter study on the natural history of infantile-onset Pompe disease. J. Pediatr. 2006;148(5):671–676. doi: 10.1016/j.jpeds.2005.11.033. [DOI] [PubMed] [Google Scholar]
- 14.Kishnani P.S., Nicolino M., Voit T., Rogers R.C., Tsai A.C., Waterson J., Herman G.E., Amalfitano A., Thurberg B.L., Richards S., Davison M., Corzo D., Chen Y.T. Chinese hamster ovary cell-derived recombinant human acid alpha-glucosidase in infantile-onset Pompe disease. J. Pediatr. 2006;149(1):89–97. doi: 10.1016/j.jpeds.2006.02.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lumizyme R [Package Insert] Genzyme Corporation; Cambridge, MA: October 2016. Lumizyme Package Insert. [Google Scholar]
- 16.Nicolino M., Byrne B., Wraith J.E., Leslie N., Mandel H., Freyer D.R., Arnold G.L., Pivnick E.K., Ottinger C.J., Robinson P.H., Loo J.C., Smitka M., Jardine P., Tato L., Chabrol B., McCandless S., Kimura S., Mehta L., Bali D., Skrinar A., Morgan C., Rangachari L., Corzo D., Kishnani P.S. Clinical outcomes after long-term treatment with alglucosidase alfa in infants and children with advanced Pompe disease. Genet Med. 2009;11(3):210–219. doi: 10.1097/GIM.0b013e31819d0996. [DOI] [PubMed] [Google Scholar]
- 17.Prater S.N., Banugaria S.G., DeArmey S.M., Botha E.G., Stege E.M., Case L.E., Jones H.N., Phornphutkul C., Wang R.Y., Young S.P., Kishnani P.S. The emerging phenotype of long-term survivors with infantile Pompe disease. Genet Med. 2012;14(9):800–810. doi: 10.1038/gim.2012.44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Prater S.N., Banugaria S.G., Morgan C., Sung C.C., Rosenberg A.S., Kishnani P.S. Letter to the editors: concerning “CRIM-negative Pompe disease patients with satisfactory clinical outcomes on enzyme replacement therapy”. J. Inherit. Metab. Dis. 2014;37(1):141–143. doi: 10.1007/s10545-013-9637-8. (by Al Khallaf et al.) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Raben N., Danon M., Gilbert A.L., Dwivedi S., Collins B., Thurberg B.L., Mattaliano R.J., Nagaraju K., Plotz P.H. Enzyme replacement therapy in the mouse model of Pompe disease. Mol. Genet. Metab. 2003;80(1–2):159–169. doi: 10.1016/j.ymgme.2003.08.022. [DOI] [PubMed] [Google Scholar]
- 20.Raben N., Fukuda T., Gilbert A.L., de Jong D., Thurberg B.L., Mattaliano R.J., Meikle P., Hopwood J.J., Nagashima K., Nagaraju K., Plotz P.H. Replacing acid alpha-glucosidase in Pompe disease: recombinant and transgenic enzymes are equipotent, but neither completely clears glycogen from type II muscle fibers. Mol. Ther. 2005;11(1):48–56. doi: 10.1016/j.ymthe.2004.09.017. [DOI] [PubMed] [Google Scholar]
- 21.Raben N., Takikita S., Pittis M.G., Bembi B., Marie S.K., Roberts A., Page L., Kishnani P.S., Schoser B.G., Chien Y.H., Ralston E., Nagaraju K., Plotz P.H. Deconstructing Pompe disease by analyzing single muscle fibers: to see a world in a grain of sand. Autophagy. 2007;3(6):546–552. doi: 10.4161/auto.4591. [DOI] [PubMed] [Google Scholar]
- 22.Slonim A.E., Bulone L., Ritz S., Goldberg T., Chen A., Martiniuk F. Identification of two subtypes of infantile acid maltase deficiency. J. Pediatr. 2000;137(2):283–285. doi: 10.1067/mpd.2000.107112. [DOI] [PubMed] [Google Scholar]
- 23.Sugawara K., Saito S., Sekijima M., Ohno K., Tajima Y., Kroos M.A., Reuser A.J., Sakuraba H. Structural modeling of mutant alpha-glucosidases resulting in a processing/transport defect in Pompe disease. J. Hum. Genet. 2009;54(6):324–330. doi: 10.1038/jhg.2009.32. [DOI] [PubMed] [Google Scholar]
- 24.Tajima Y., Matsuzawa F., Aikawa S., Okumiya T., Yoshimizu M., Tsukimura T., Ikekita M., Tsujino S., Tsuji A., Edmunds T., Sakuraba H. Structural and biochemical studies on Pompe disease and a “pseudodeficiency of acid alpha-glucosidase”. J. Hum. Genet. 2007;52(11):898–906. doi: 10.1007/s10038-007-0191-9. [DOI] [PubMed] [Google Scholar]
- 25.van den Hout H.M., Hop W., van Diggelen O.P., Smeitink J.A., Smit G.P., Poll-The B.T., Bakker H.D., Loonen M.C., de Klerk J.B., Reuser A.J., van der Ploeg A.T. The natural course of infantile Pompe's disease: 20 original cases compared with 133 cases from the literature. Pediatrics. 2003;112(2):332–340. doi: 10.1542/peds.112.2.332. [DOI] [PubMed] [Google Scholar]