An insidious belief pervades the modern ICU—that in cases of diagnostic uncertainty, broad-spectrum antibiotic therapy is “safest for the patient.” This idea is likely a vestige of that bygone era before multidrug-resistant bacteria, when faith in the everlasting efficacy of antimicrobials was strong, and responsible doctors protected their patients from infection without regard for pretest probability.
Fortunately, this belief has been vigorously challenged. Antibiotic overuse is now known to be a clear contributor to the spread of drug resistance, which the World Health Organization has declared to be one of the greatest current threats to human health. It has further predicted that in the absence of improved stewardship, we will enter a postantibiotic era by 2050, with an associated 10 million deaths due to multidrug-resistant bacteria per year (1).
If this apocalyptic prediction were not deterrence enough, it is also clear that inappropriate antimicrobial therapy poses an immediate threat to the patient (2). Risks include life-threatening drug reactions such as Stevens–Johnson syndrome and anaphylaxis, as well as more common complications such as drug–drug interactions and nephrotoxicity. A more recently appreciated hazard is the profound disruption of gut microbiota produced by antibiotics, a condition termed dysbiosis. Although the full consequences of dysbiosis have yet to be elucidated, it is clearly linked to Clostridioides difficile colitis, a highly prevalent and often deadly infection (3). Altogether, the adverse effects associated with unnecessary antibiotics have been shown to worsen mortality in a number of studies by independent groups (4, 5).
It is therefore imperative for both the community as a whole and the individual patient that we develop highly sensitive diagnostic tools to rule out bacterial infection and enable safe and prompt cessation of antibiotics. Such diagnostics are particularly needed for pneumonia, which is responsible for a substantial proportion of antibiotic misuse and is a well-established driver of resistance (6–8).
In this issue of the Journal, Walter and colleagues (pp. 1225–1237) take an important step toward solving this problem in the ICU (9). To do so, they make use of the defining host immune response in bacterial pneumonia, namely, neutrophilic alveolitis. Indeed, most of the clinical manifestations of pneumonia stem from this process, including 1) respiratory symptoms such as cough and purulent sputum; 2) systemic signs such as fever, which results from inflammatory signals derived in part from polymorphonuclear cells; and 3) radiographic infiltrates, which in pneumonia represent pus in the lung. Although relatively nonspecific, the BAL neutrophilia test is believed to be highly sensitive for pneumonia, as previously shown in critically ill (10) and nonneutropenic immunocompromised patients (11).
In keeping with this canonical pathophysiologic principle, Walter and colleagues demonstrate here that nonbronchoscopic BAL (NBBAL) neutrophil counts of <50% provided a negative predictive value of 91.5% in patients with suspected ventilator-associated pneumonia, and coupled with a negative gram stain, the negative predictive value approached 100%. Diagnosis of pneumonia was confirmed by quantitative culture, a method that is highly stringent but not widely available.
This is a remarkable finding in terms of not only test performance but also practicality. NBBAL does not require pulmonologists; it may be performed by trained respiratory therapists, which reduces the cost and potential delay involved with formal bronchoscopy, with no decrease in diagnostic yield (12). Furthermore, the laboratory analytics required—cell counts and Gram stain—are universally available, inexpensive, and rapid. For these reasons, the current findings have the potential to make a broad and immediate impact on clinical practice in the ICU.
The diagnostic insufficiency of BAL neutrophilia as a standalone test is worth discussing. One explanation may relate to a phenomenon that is observed in advanced sepsis and is known as neutrophil paralysis, wherein polymorphonuclear cell extravasation and chemotaxis are impaired (13). Thus, the 8.5% of patients in the study who had bacterial infection without neutrophilia may have had insufficient recruitment of neutrophils into the alveolar space due to sepsis-induced immunosuppression. Fortunately, these few cases could be identified via Gram stain.
Walter and colleagues also present a provocative series of genomic studies in which they assessed the ability of alveolar macrophage transcriptomes to predict the diagnosis of ventilator-associated pneumonia. Because alveolar macrophages serve as the central innate immune sensor of alveolar bacteria and principal coordinator of neutrophil recruitment, one might predict their transcriptional status to be a highly accurate reporter of lung infection. Surprisingly, however, the operating characteristics of an RNA sequencing–based test did not significantly outperform the NBBAL neutrophilia test alone. A number of reasons may explain this finding.
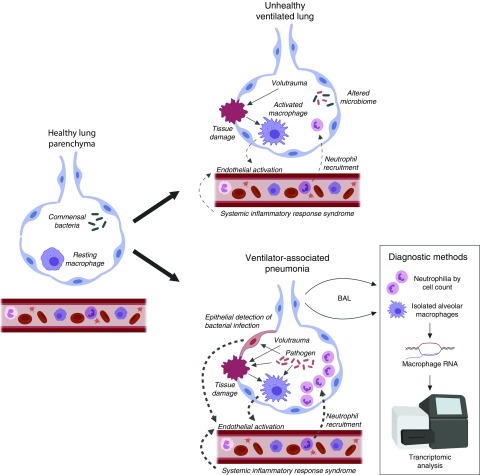
First, alveolar macrophages do not operate in isolation in the generation of pneumonic immune responses (Figure 1). Numerous other components of the innate immune system contribute to pathogen detection in the lung, including epithelial cells, innate lymphocytes, platelets, and plasma constituents (e.g., complement) (14). Indeed, immune signaling in epithelial cells alone was shown to be sufficient to recruit neutrophils and control infection in a murine model of Pseudomonas pneumonia (15). Therefore, alveolar macrophage–independent neutrophilic responses may help to explain the findings reported here.
Figure 1.
Pneumonic immune responses. Under healthy conditions (left panel), low levels of commensal bacteria and resting, noninflammatory macrophages predominate. The mechanically ventilated lung (top right panel) has numerous immune stimuli, for example, relating to volutrauma-induced tissue damage and systemic inflammation. These factors lead to endothelial activation and immune infiltrate, but not overt pneumonia. During ventilator-associated pneumonia (depicted in the bottom right panel), pathogenic bacteria further activate macrophages and induce both damage and stimulation of epithelial cells. Together, these drive a robust pyogenic inflammatory response, which may be detected by either one of the diagnostic methods described here.
Second, alveolar macrophages process immune signals from not only microbes (Figure 1) but also systemic inflammatory sources and damaged tissue (induced by pathogens, ventilator-induced volutrauma, etc.). It may be that the integration of these various stimuli produces genomic responses that are simply more similar than they are different. Studies in pneumonia and sepsis support this hypothesis and further indicate that the ability to distinguish features such as the site of infection, microbial etiology, and even the presence of infection appears to diminish as the illness progresses (16).
Thus, in patients with advanced critical illness, such as those studied here, host transcriptomics may be more challenging to interpret. Perhaps sampling patients earlier, combining transcriptomics with other biomarkers (including the alveolar neutrophilia described here), or synthesizing host genomics with microbiomic sequencing would lead to further diagnostic refinement. Further omics-based investigations of such questions are certainly warranted.
Although it is surprising that rapid, inexpensive, and widely available tests such as BAL cell count and Gram stain perform as well as cutting-edge personalized techniques like RNA sequencing, the finding is all the more valuable given the practical advantages of the former. Prospective trials are needed to demonstrate that the algorithm presented here can indeed reduce antibiotic use without compromising patient safety, but these results should certainly encourage more studies on the implementation of NBBAL sampling in the ICU. Finally, we submit that the present work, which describes a cost-effective rule-out test for infection inspired by sound pathophysiology, represents an important model for future studies aimed at reducing the unnecessary use of antibiotics—an often underappreciated but nonetheless rampant problem with potentially dire consequences for both society and the patient.
Footnotes
Originally Published in Press as DOI: 10.1164/rccm.201811-2163ED on December 13, 2018
Author disclosures are available with the text of this article at www.atsjournals.org.
References
- 1.O’Neill J. Tackling drug-resistant infections globally: final report and recommendations. The review on antimicrobial resistance; 2016 [accessed 2018 Apr 16]. Available from: https://amr-review.org/sites/default/files/160518_Final%20paper_with%20cover.pdf.
- 2.Gautam S, Sharma L, Dela Cruz CS. Personalizing the management of pneumonia. Clin Chest Med. 2018;39:871–900. doi: 10.1016/j.ccm.2018.08.008. [DOI] [PubMed] [Google Scholar]
- 3.Lessa FC, Mu Y, Bamberg WM, Beldavs ZG, Dumyati GK, Dunn JR, et al. Burden of Clostridium difficile infection in the United States. N Engl J Med. 2015;372:825–834. doi: 10.1056/NEJMoa1408913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Garnacho-Montero J, Gutiérrez-Pizarraya A, Escoresca-Ortega A, Corcia-Palomo Y, Fernández-Delgado E, Herrera-Melero I, et al. De-escalation of empirical therapy is associated with lower mortality in patients with severe sepsis and septic shock. Intensive Care Med. 2014;40:32–40. doi: 10.1007/s00134-013-3077-7. [DOI] [PubMed] [Google Scholar]
- 5.Schuetz P, Wirz Y, Sager R, Christ-Crain M, Stolz D, Tamm M, et al. Effect of procalcitonin-guided antibiotic treatment on mortality in acute respiratory infections: a patient level meta-analysis. Lancet Infect Dis. 2018;18:95–107. doi: 10.1016/S1473-3099(17)30592-3. [DOI] [PubMed] [Google Scholar]
- 6.Centers for Disease Control and Prevention. Antibiotic resistance threats in the United States; 2013 [accessed 2018 Apr 15]. Available from: http://www.cdc.gov/drugresistance/pdf/ar-threats-2013-508.pdf.
- 7.Cookson WOCM, Cox MJ, Moffatt MF. New opportunities for managing acute and chronic lung infections. Nat Rev Microbiol. 2018;16:111–120. doi: 10.1038/nrmicro.2017.122. [DOI] [PubMed] [Google Scholar]
- 8.Guest JF, Morris A. Community-acquired pneumonia: the annual cost to the National Health Service in the UK. Eur Respir J. 1997;10:1530–1534. doi: 10.1183/09031936.97.10071530. [DOI] [PubMed] [Google Scholar]
- 9.Walter JM, Ren Z, Yacoub T, Reyfman PA, Shah RD, Abdala-Valencia H, et al. Multidimensional assessment of the host response in mechanically ventilated patients with suspected pneumonia. Am J Respir Crit Care Med. 2019;199:1225–1237. doi: 10.1164/rccm.201804-0650OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Choi SH, Hong SB, Hong HL, Kim SH, Huh JW, Sung H, et al. Usefulness of cellular analysis of bronchoalveolar lavage fluid for predicting the etiology of pneumonia in critically ill patients. PLoS One. 2014;9:e97346. doi: 10.1371/journal.pone.0097346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Stolz D, Stulz A, Müller B, Gratwohl A, Tamm M. BAL neutrophils, serum procalcitonin, and C-reactive protein to predict bacterial infection in the immunocompromised host. Chest. 2007;132:504–514. doi: 10.1378/chest.07-0175. [DOI] [PubMed] [Google Scholar]
- 12.Lavigne MC. Nonbronchoscopic methods [nonbronchoscopic bronchoalveolar lavage (BAL), mini-BAL, blinded bronchial sampling, blinded protected specimen brush] to investigate for pulmonary infections, inflammation, and cellular and molecular markers: a narrative review. Clin Pulm Med. 2017;24:13–25. [Google Scholar]
- 13.Sônego F, Castanheira FV, Ferreira RG, Kanashiro A, Leite CA, Nascimento DC, et al. Paradoxical roles of the neutrophil in sepsis: protective and deleterious. Front Immunol. 2016;7:155. doi: 10.3389/fimmu.2016.00155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Quinton LJ, Walkey AJ, Mizgerd JP. Integrative physiology of pneumonia. Physiol Rev. 2018;98:1417–1464. doi: 10.1152/physrev.00032.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mijares LA, Wangdi T, Sokol C, Homer R, Medzhitov R, Kazmierczak BI. Airway epithelial MyD88 restores control of Pseudomonas aeruginosa murine infection via an IL-1-dependent pathway. J Immunol. 2011;186:7080–7088. doi: 10.4049/jimmunol.1003687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Burnham KL, Davenport EE, Radhakrishnan J, Humburg P, Gordon AC, Hutton P, et al. Shared and distinct aspects of the sepsis transcriptomic response to fecal peritonitis and pneumonia. Am J Respir Crit Care Med. 2017;196:328–339. doi: 10.1164/rccm.201608-1685OC. [DOI] [PMC free article] [PubMed] [Google Scholar]