Abstract
Background:
Individuals with Down syndrome (DS) experience autonomic dysfunction, with reduced sympathetic and parasympathetic control. This results in alterations in resting heart rate and blood pressure and attenuated responses to sympathoexcitatory stimuli. It is unknown to what extent this impacts the regulation of peripheral blood flow in response to sympathetic stimuli, which is an important prerequisite to exercise and perform work. Therefore, we aimed to investigate differences in peripheral blood flow regulation in response to lower body negative pressure (LBNP) between individuals with and without DS.
Methods:
Participants (n=10 males with DS and n=11 male controls, mean age 23.7 years ± 3.2) underwent 5 min of LBNP stimulations (−20 mmHg), after resting supine for 10 min. One minute steady state blood pressure and blood flow at baseline and during LBNP were obtained for analysis. Mean flow velocity and arterial diameters were recorded with ultrasonography; foreram blood flow (FBF), shear rate and forearm vascular conductance (FVC) were calculated using brachial blood pressure measured right before ultrasound recordings.
Results:
Participants with DS responded differently (consistent with reduced vasoconstrictive control) to the LBNP stimulus (significant ConditionxGroup interaction effect) for mean velocity (p=0.02), FBF (p=0.04), shear rate (p=0.02) and FVC (p=0.03), compared to participants without DS.
Conclusion:
Young males with DS exhibit reduced peripheral regulation of blood flow in response to LBNP compared to controls, indicating a blunted sympathetic control of blood flow. Further research is necessary to explore the impact of these findings on exercise and work capacity.
Keywords: Down syndrome, blood flow, sympathetic stimulus, autonomic nervous system
Introduction
Down syndrome (DS), a genetic condition also referred to as trisomy 21, impacts about 1 in every 700 newborns1. While life expectancy is improving in this population2, 3, individuals with DS are still at risk for numerous conditions impairing quality of life and health, such as diabetes mellitus, dyslipidemia, congenital heart defects, leukemia, thyroid disease, cognitive decline and Alzheimer’s disease4, 5. In addition, individuals with DS present with a higher prevalence of cardiovascular disease (CVD) risk factors, such as obesity6, very low physical activity7, low fitness8, and an unfavorable metabolic risk profile9, 10. Despite this CVD risk profile, individuals with DS exhibit lower rates of atherosclerotic disease, and DS has been referred to as an atheroma-free model9, 11, 12. Consequently, a better understanding of the physiology of people with DS may yield important insight regarding the pathology and/or prevention of not only atherosclerotic disease but also several other chronic conditions.
In individuals with DS, these CVD risk factors may not be only attributable to lifestyle or to DS-specific physical characteristics like low muscle tone and mass13. Lower fitness in individuals with DS has been attributed to autonomic dysfunction14. In turn, autonomic dysfunction has been implicated in increased metabolic risk15, 16 and could potentially alter disease risk in this population.
To further understand the role of autonomic regulation in DS and its potential effects on work capacity, our group has conducted a number of in-depth physiological studies (as reviewed in14). In brief, we have shown individuals with DS exhibit 1) chronotropic incompetence, which is the inability to increase heart rate proportionate to increased activity17, 18; 2) blunted heart rate and blood pressure responses to sympathoexcitatory tasks (orthostasis, cold pressor test, handgrip exercise)19–22; and 3) suppressed catecholamine release in response to maximal exercise23. Cumulatively, this data suggests reduced vagal withdrawal and sympathetic activation and decreased baroreceptor sensitivity in individuals with DS14. Importantly, this autonomic dysfunction may also impair the ability to regulate blood flow to working muscles during exercise, and further compromise work capacity, which provides the basis of the current study.
Our current work is the first step to test the hypothesis that impaired peripheral blood flow control could potentially explain fatigue and lower work capacity in individuals with DS. Reduced sympathetic activation during exercise may impair the ability to adequately shunt blood flow to the working muscle. One means of examining this hypothesis would be to evaluate the vascular responses to a blood volume distribution challenge, such as lower body negative pressure (LBNP). LBNP provides a sympathetic stimulus and redistribution of blood flow that may reveal differential blood flow regulation in individuals with DS. As such, the purpose of the present study was to investigate the effects of a mild sympathoexcitatory stimulus (−20 lower LBNP) on brachial blood flow in individuals with and without DS. We hypothesized that individuals with DS would demonstrate less vasoconstriction and smaller reductions in brachial blood flow than the control group.
Methods
Participants
Participants were recruited from the UIC campus and the Chicago community via support groups and organizations for individuals with DS, word of mouth, as well as online postings. Potential participants were invited for an on-site screening visit in order to determine eligibility for the study.
Individuals were included if between 18–40 years of age, non-athletic, in general good health, and diagnosed with DS for the DS group. Exclusion criteria were congenital heart disease, cardiovascular disease, a BMI of over 40 kg/m2, any conditions listed as absolute or relative contraindications to exercise according to The American College of Sports Medicine, blood pressure over 140/90 mmHg and impaired fasting glucose or diabetes (fasting glucose > 100 mg/dl). For the preliminary analyses in this paper only males were included to avoid the influence of varying reproductive hormones in women. This study was approved by the University of Illinois at Chicago Institutional Review Board and all participants and their parent or caregiver provided written informed consent.
Experimental protocol
For participants with DS, a familiarization visit was completed prior to the experimental visit to allow the participant to become comfortable with research personnel, the laboratory environment and the research protocol. Participants were instructed to abstain from caffeine, alcohol, multivitamins and exercise for at least 12 hours and a minimum 4 hour fast (only water allowed) prior to the experimental study visit. Upon arrival, height, weight, and waist circumference were measured and a health history and physical activity questionnaire were completed. BMI was determined using the standard calculation (kg/m2). Following anthropometric measurements, participants were instrumented for 3lead electrocardiogram (ECG) and respiratory monitoring. Participants were then guided into a supine position in the lower body negative pressure (LBNP) chamber sealed at the waist. Blood pressure was continuously monitored non-invasively using finger plethysmography (Finometer Pro, Finapres Medical System, Netherlands). Analog signals from ECG, Finometer, respiration and the LBNP chamber were continuously recorded during the protocol using Acknowledge software (BIOPAC System, Inc., CA, USA) for storage and offline analysis.
In the last minute of 10 minutes of quiet rest, measurements of brachial artery diameter and blood velocity were obtained. LBNP was then applied at −20 mmHg to unload the cardiopulmonary baroreceptors and trigger a reflex sympathetic response, which has shown to cause little to no change in blood pressure24, 25. Brachial artery diameter and blood velocity were assessed again after 5 min of adjustment to −20 mmHg LBNP. An individualised treadmill test was then performed to measure work capacity, by measuring peak oxygen uptake (VO2peak) (ParvoMedics True One, Sandy, Utah). As previously described26, treadmill speed was held constant at a fast walk while grade increased 2.5% every 2 minutes up to 12.5% grade. From this point, grade remained constant and speed was increased 1.6 km/h every minute until volitional fatigue. This protocol has been shown to be both valid and reliable for testing individuals with and without Down syndrome27, 28. The highest VO2 and heart rate were recorded as VO2peak and HRpeak, if the respiratory exchange ratio was over 1.0.
Measurements
Brachial diameter and mean blood flow velocity were measured on the upper arm using high fidelity ultrasound (Hitachi Aloka Alpha 7, Japan), with the probe secured over the brachial artery with a ≤60° probe insonation angle29. Measurements were obtained during the last minute of the 10 min baseline and the minute right after 5 min LBNP to calculate forearm blood flow (FBF, FBF=velocity*πr2*60) and shear rate (shear rate=4*velocity*diameter). Forearm vascular conductance (FVC, FVC=FBF/MAP*100) was calculated using brachial mean arterial pressure (MAP) measured in conjunction with ultrasound recordings.
Statistical analysis
Data were checked for normality and outliers. Baseline differences between groups were tested with independent t-tests. Mixed ANOVA was used to test the effects of Group, Condition and the Group x Condition interaction. Post-hoc pairwise comparisons were performed in case of a significant interaction effect, and the Bonferroni correction was used to correct for multiple comparisons. Statistical analyses were performed with SPSS 23.0, and all p-values are 2-sided, with an a priori αlevel of 0.05 determined to be significant.
Results
Twenty-one healthy, young male adults (DS n=10) participated in the study. Individuals with DS had a higher BMI (p=0.04), lower VO2peak (p<0.01) and lower HRpeak (p<0.01) than the control group (Table 1).
Table 1.
Descriptive characteristics
| DS (n=10) | Control (n=11) | |
|---|---|---|
| Age (years) | 24 ± 3 | 24 ± 3 |
| BMI (kg/m2) | 29.5 ± 4.0 | 25.1 ± 5.0 * |
| VO2peak (ml/kg/min) | 28.2 ± 4.5 | 42.6 ± 6.0 ** |
| HRpeak (bpm) | 170 ± 13 | 195 ± 10 ** |
Data mean ± SD. BMI, body mass index; HR, heart rate
= significant difference with DS p< 0.05
= significant difference with DS p<0.01
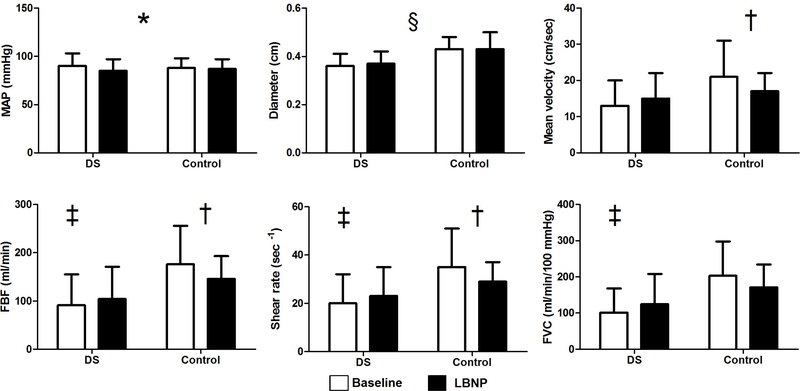
MAP decreased in both groups from baseline to LBNP (F=5.320, p=0.033). Brachial artery diameter was smaller in participants with DS compared to the control group (F=5.871, p=0.026). Significant interaction effects (p<0.05) were observed for mean velocity (F=6.300, p=0.021), FBF (F=5.039, p=0.037), shear rate (F=5.970, p=0.024) and FVC (F=5.195, p=0.034) (Figure 1). Post-hoc pairwise comparisons demonstrated a difference in FBF, shear rate and FVC between participants with DS and the control group at baseline, not during LBNP (p<0.05). From baseline to LBNP, the control group reduced mean velocity, FBF and shear rate (p<0.05). No changes were seen for participants with DS (p>0.05).
Figure 1.
Outcome measures in DS and control group at baseline and during LBNP MAP, mean arterial pressure; FBF, forearm blood flow; FVC, forearm vascular conductance
*= overall Condition effect
§ = overall Group effect
† = difference between baseline and LBNP
‡ = difference between DS and control group at baseline
Significance level is set at p<0.05
Discussion
FBF, shear rate, and FVC decreased in the control group during LBNP, indicating peripheral vasoconstriction as a result of sympathoexcitation. However, individuals with DS did not show any evidence of vasoconstriction, indicating a lack of forearm blood flow control in response to sympathotexcitation. Furthermore, both groups showed a slight decrease in MAP from baseline to LBNP, and individuals with DS had smaller brachial diameters than the control group.
During LBNP, the control group reduced mean brachial blood flow velocity, resulting in decreased FBF and shear rate. Brachial artery diameter did not change from baseline during the LBNP, suggesting that the vasoconstriction likely occurred downstream in the resistance arteries, resulting in decreased FBF. This is in line with previous research demonstrating a decrease in brachial blood flow24 and FVC30 during LBNP in healthy subjects. In individuals with DS however, none of the vascular outcomes were altered during LBNP. These differential responses between persons with DS and the control group were not driven by blood pressure as both groups exhibited similar changes in MAP during the LBNP. These findings suggest reduced peripheral regulation of blood flow due to blunted sympathetic control in individuals with DS.
Interestingly, brachial artery diameter was smaller in individuals with DS, and the mean velocity at baseline was also lower compared to the control group. A possible explanation could be a lower metabolic demand for oxygen from the periphery in individuals with DS, likely driven by features that are specific to DS, such as lower muscle mass13 and higher oxidative stress31, 32. Another possible explanation could be that the blunted vagal withdrawal and sympathetic activation in individuals with DS14, resulting in a lower heart rate and lower systemic blood pressure, may also result in lower mean velocities throughout the systemic circulation. Combined with blunted control of peripheral blood flow, this is likely to impact exercise capacity. To our knowledge, only one other research group has assessed brachial blood flow in individuals with DS33. Contrary to our results, there was no difference between individuals with DS and healthy control participants in resting brachial blood flow. As only the second group to report brachial blood flow in DS with both studies having small sample sizes, future investigations with larger sample sizes are necessary to determine whether differences in resting values exist.
This study demonstrates for the first time that autonomic dysfunction in individuals with DS is not only impacting systemic control of heart rate and blood pressure, but also peripheral blood flow. As described earlier, autonomic dysfunction negatively impacts work capacity, as the ability to vasoconstrict in the non-active organs and muscles is critical to maintain appropriate blood pressure, heart rate and thus cardiac output during exercise. An appropriate increase in cardiac output during exercise is necessary to deliver blood to the working muscles. Furthermore, vasodilation in the working muscles, coupled with vasoconstriction in non-working muscle and other organs is required to ensure oxygen delivery to working muscle fibers34. Vasodilation is largely controlled by local mechanisms, in which the endothelium plays a major role. Individuals with DS have increased levels of oxidative stress, caused by a DS-specific mitochondrial dysfunction, which leads to endothelial dysfunction32. Only a few studies have investigated vasodilatory capacity and vascular function in individuals with DS. They found individuals with DS have either a smaller9 or equal35 intima media thickness, and a higher35 or lower arterial stiffness36, 37, and reduced ability to vasodilate in response to increased shear stress33. While no previous studies have evaluated vascular function during exercise in this population, we speculate that individuals with DS would exhibit a blunted vasodilatory response to exercise in working muscles, coupled with a lack of vasoconstriction in non-working tissue. This impairment in blood flow regulation could further limit work capacity in individuals with DS, however, future studies are required to evaluate this hypothesis.
Strengths of this study were the familiarization of the individuals with DS with the protocol, and the application of high-quality, established methods to a population usually excluded from this type of experimental research. Potential limitations are the small sample size and the baseline differences in BMI and VO2peak, which may have confounded the differences in the outcome variables as obesity and fitness are known determinants of autonomic function. Due to the unique phenotype of DS, it is difficult to match body size and work capacity between groups. However, our previous research demonstrated that BMI had only a minimal impact on maximal heart rate and VO2peak in individuals with DS38.
Future research needs to focus on understanding peripheral blood flow regulation in individuals with DS. This is a necessary step to further elucidate the physiology behind low physical activity and reduced work capacity in this population and the comorbid conditions that accompany it. Dynamic handgrip exercise is often used to elucidate characteristics of peripheral blood flow regulation39 and would be the next step to further understand blood flow regulation in individuals with DS. Handgrip exercise should elicit vasodilation in the smaller arteries and microvasculature of the working muscle to increase brachial blood flow34, 40, even when challenged with a sympathetic stimulus30. We are currently conducting a study investigating this interplay between vasoconstriction and vasodilation in individuals with DS during handgrip exercise and a sympathetic stimulus.
Conclusion
Young males with DS exhibit reduced peripheral regulation of blood flow in response to LBNP compared to the control group, indicating a blunted sympathetic control of blood flow. Further research is necessary to investigate the peripheral regulation of blood flow in the resistance arteries and microvasculature in individuals with DS, and to explore the impact of these findings on exercise, work capacity and health in this population.
Highlights.
Males with DS show reduced peripheral regulation of blood flow during LBNP
Autonomic dysfunction in DS impacts systemic and peripheral blood flow regulation
Work capacity in DS is likely reduced due to insufficient control of blood flow
Further research should focus on the impact of the findings on exercise and health
Acknowledgements
We would like to thank all the participants, their parents and caregivers for supporting this research by investing their time and energy in this research study.
Funding sources
This work has been supported by the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NIH K99/R00 1 K99 HD092606–01). This funding body had no role in study design; in the collection, analysis and interpretation of data; in the writing of the report; and in the decision to submit the article for publication.
Footnotes
Declarations of interest
None.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- [1].Parker SE, Mai CT, Canfield MA, Rickard R, Wang Y, Meyer RE, et al. Updated National Birth Prevalence estimates for selected birth defects in the United States, 2004–2006. Birth Defects Res A Clin Mol Teratol 2010;88:1008–16. [DOI] [PubMed] [Google Scholar]
- [2].Janicki MP, Dalton AJ, Henderson CM, Davidson PW. Mortality and morbidity among older adults with intellectual disability: health services considerations. Disability and Rehabilitation 1999;21:284–94. [DOI] [PubMed] [Google Scholar]
- [3].Perkins EA, Moran JA. Aging adults with intellectual disabilities. Journal of the American Medical Association 2010;304:91–2. [DOI] [PubMed] [Google Scholar]
- [4].Colvin KL, Yeager ME. What people with Down Syndrome can teach us about cardiopulmonary disease. Eur Respir Rev 2017;26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Capone GT, Chicoine B, Bulova P, Stephens M, Hart S, Crissman B, et al. Co-occurring medical conditions in adults with Down syndrome: A systematic review toward the development of health care guidelines. Am J Med Genet A 2018;176:116–33. [DOI] [PubMed] [Google Scholar]
- [6].de Winter CF, Bastiaanse LP, Hilgenkamp TI, Evenhuis HM, Echteld MA. Overweight and obesity in older people with intellectual disability. Research in Developmental Disabilities 2012;33:398–405. [DOI] [PubMed] [Google Scholar]
- [7].Temple VA, Stanish HI. Pedometer-measured physical activity of adults with intellectual disability: predicting weekly step counts. American Journal on Intellectual and Developmental Disabilities 2009;114:15–22. [DOI] [PubMed] [Google Scholar]
- [8].Hilgenkamp TI, van Wijck R, Evenhuis HM. Subgroups associated with lower physical fitness in older adults with ID: results of the HA-ID study. Research in Developmental Disabilities 2014;35:439–47. [DOI] [PubMed] [Google Scholar]
- [9].Draheim CC, Geijer JR, Dengel DR. Comparison of intima-media thickness of the carotid artery and cardiovascular disease risk factors in adults with versus without the Down syndrome. American Journal of Cardiology 2010;106:1512–6. [DOI] [PubMed] [Google Scholar]
- [10].Draheim CC, McCubbin JA, Williams DP. Differences in cardiovascular disease risk between nondiabetic adults with mental retardation with and without Down syndrome. American Journal of Mental Retardation 2002;107:201–11. [DOI] [PubMed] [Google Scholar]
- [11].Murdoch JC, Rodger JC, Rao SS, Fletcher CD, Dunnigan MG. Down’s syndrome: an atheroma-free model? Br Med J 1977;2:226–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Yla-Herttuala S, Luoma J, Nikkari T, Kivimaki T. Down’s syndrome and atherosclerosis. Atherosclerosis 1989;76:269–72. [DOI] [PubMed] [Google Scholar]
- [13].Rigoldi C, Galli M, Albertini G. Gait development during lifespan in subjects with Down syndrome. Research in Developmental Disabilities 2011;32:158–63. [DOI] [PubMed] [Google Scholar]
- [14].Fernhall B, Mendonca GV, Baynard T. Reduced work capacity in individuals with down syndrome: a consequence of autonomic dysfunction? Exercise and sport sciences reviews 2013;41:138–47. [DOI] [PubMed] [Google Scholar]
- [15].Thayer JF, Yamamoto SS, Brosschot JF. The relationship of autonomic imbalance, heart rate variability and cardiovascular disease risk factors. International Journal of Cardiology 2010;141:122–31. [DOI] [PubMed] [Google Scholar]
- [16].Chang YW, Lin JD, Chen WL, Yen CF, Loh CH, Fang WH, et al. Metabolic syndrome and short-term heart rate variability in adults with intellectual disabilities. Research in Developmental Disabilities 2012;33:1701–7. [DOI] [PubMed] [Google Scholar]
- [17].Fernhall B, McCubbin JA, Pitetti KH, Rintala P, Rimmer JH, Millar AL, et al. Prediction of maximal heart rate in individuals with mental retardation. Medicine and Science in Sports and Exercise 2001;33:1655–60. [DOI] [PubMed] [Google Scholar]
- [18].Guerra M, Llorens N, Fernhall B. Chronotropic incompetence in persons with down syndrome. Archives of Physical Medicine and Rehabilitation 2003;84:1604–8. [DOI] [PubMed] [Google Scholar]
- [19].Fernhall B, Otterstetter M. Attenuated responses to sympathoexcitation in individuals with Down syndrome. Journal of Applied Physiology 2003;94:2158–65. [DOI] [PubMed] [Google Scholar]
- [20].Figueroa A, Collier SR, Baynard T, Giannopoulou I, Goulopoulou S, Fernhall B. Impaired vagal modulation of heart rate in individuals with Down syndrome. Clinical autonomic research 2005;15:45–50. [DOI] [PubMed] [Google Scholar]
- [21].Iellamo F, Galante A, Legramante JM, Lippi ME, Condoluci C, Albertini G, et al. Altered autonomic cardiac regulation in individuals with Down syndrome. Am J Physiol Heart Circ Physiol 2005;289:H2387–91. [DOI] [PubMed] [Google Scholar]
- [22].Agiovlasitis S, Collier SR, Baynard T, Echols GH, Goulopoulou S, Figueroa A, et al. Autonomic response to upright tilt in people with and without Down syndrome. Research in Developmental Disabilities 2010;31:857–63. [DOI] [PubMed] [Google Scholar]
- [23].Fernhall B, Baynard T, Collier SR, Figueroa A, Goulopoulou S, Kamimori GH, et al. Catecholamine response to maximal exercise in persons with Down syndrome. American Journal of Cardiology 2009;103:724–6. [DOI] [PubMed] [Google Scholar]
- [24].Kitano A, Shoemaker JK, Ichinose M, Wada H, Nishiyasu T. Comparison of cardiovascular responses between lower body negative pressure and head-up tilt. J Appl Physiol (1985) 2005;98:2081–6. [DOI] [PubMed] [Google Scholar]
- [25].Halliwill JR, Lawler LA, Eickhoff TJ, Joyner MJ, Mulvagh SL. Reflex responses to regional venous pooling during lower body negative pressure in humans. J Appl Physiol (1985) 1998;84:454–8. [DOI] [PubMed] [Google Scholar]
- [26].Baynard T, Pitetti KH, Guerra M, Unnithan VB, Fernhall B. Age-related changes in aerobic capacity in individuals with mental retardation: a 20-yr review. Medicine and Science in Sports and Exercise 2008;40:1984–9. [DOI] [PubMed] [Google Scholar]
- [27].Fernhall B, Tymeson G. Graded exercise testing of mentally retarded adults: a study of feasibility. Archives of Physical Medicine and Rehabilitation 1987;68:363–5. [PubMed] [Google Scholar]
- [28].Fernhall B, Pitetti KH, Rimmer JH, McCubbin JA, Rintala P, Millar AL, et al. Cardiorespiratory capacity of individuals with mental retardation including Down syndrome. Medicine and Science in Sports and Exercise 1996;28:366–71. [DOI] [PubMed] [Google Scholar]
- [29].Thijssen DH, Black MA, Pyke KE, Padilla J, Atkinson G, Harris RA, et al. Assessment of flow-mediated dilation in humans: a methodological and physiological guideline. Am J Physiol Heart Circ Physiol 2011;300:H2–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [30].Tschakovsky ME, Hughson RL. Rapid blunting of sympathetic vasoconstriction in the human forearm at the onset of exercise. J Appl Physiol (1985) 2003;94:1785–92. [DOI] [PubMed] [Google Scholar]
- [31].Pagano G, Castello G. Oxidative stress and mitochondrial dysfunction in Down syndrome. Adv Exp Med Biol 2012;724:291–9. [DOI] [PubMed] [Google Scholar]
- [32].Valenti D, Braidy N, De Rasmo D, Signorile A, Rossi L, Atanasov AG, et al. Mitochondria as pharmacological targets in Down syndrome. Free radical biology & medicine 2018;114:69–83. [DOI] [PubMed] [Google Scholar]
- [33].Cappelli-Bigazzi M, Santoro G, Battaglia C, Palladino MT, Carrozza M, Russo MG, et al. Endothelial cell function in patients with Down’s syndrome. American Journal of Cardiology 2004;94:392–5. [DOI] [PubMed] [Google Scholar]
- [34].Saltin B, Radegran G, Koskolou MD, Roach RC. Skeletal muscle blood flow in humans and its regulation during exercise. Acta Physiol Scand 1998;162:421–36. [DOI] [PubMed] [Google Scholar]
- [35].Hu M, Yan H, Ranadive SM, Agiovlasitis S, Fahs CA, Atiq M, et al. Arterial stiffness response to exercise in persons with and without Down syndrome. Research in Developmental Disabilities 2013;34:3139–47. [DOI] [PubMed] [Google Scholar]
- [36].Parra P, Costa R, de Asua DR, Moldenhauer F, Suarez C. Atherosclerotic Surrogate Markers in Adults With Down Syndrome: A Case-Control Study. J Clin Hypertens (Greenwich) 2017;19:205–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [37].Rodrigues AN, Coelho LC, Goncalves WL, Gouvea SA, Vasconcellos MJ, Cunha RS, et al. Stiffness of the large arteries in individuals with and without Down syndrome. Vasc Health Risk Manag 2011;7:375–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38].Wee SO, Pitetti KH, Goulopoulou S, Collier SR, Guerra M, Baynard T. Impact of obesity and Down syndrome on peak heart rate and aerobic capacity in youth and adults. Research in Developmental Disabilities 2014;36C:198–206. [DOI] [PubMed] [Google Scholar]
- [39].Richards JC, Crecelius AR, Larson DG, Dinenno FA. Acute ascorbic acid ingestion increases skeletal muscle blood flow and oxygen consumption via local vasodilation during graded handgrip exercise in older adults. Am J Physiol Heart Circ Physiol 2015;309:H360–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [40].Clifford PS, Hellsten Y. Vasodilatory mechanisms in contracting skeletal muscle. J Appl Physiol (1985) 2004;97:393–403. [DOI] [PubMed] [Google Scholar]