Abstract
Osteoarthritis (OA) is a common inflammatory joint disease. MicroRNAs (miRNAs/miRs) have been reported to be involved in the pathogenesis of OA; however, the role of miRNAs in OA remains largely unexplained. The purpose of the present study was to investigate the expression and role of miR-195-5p in OA, and to further explore the mechanism. The expression level of miR-195-5p was measured using reverse transcription-quantitative polymerase chain reaction (RT-qPCR). TargetScan and a luciferase reporter assay were used to reveal the associations between miR-195-5p and REGγ (also known as PSME3). To investigate the role of miR-195-5p in OA, a cell model of OA was established by treating ATDC5 cells with lipopolysaccharide (LPS). Then an MTT assay was conducted to detect cell proliferation ability, and an Annexin V-fluorescein isothiocyanate/propidium iodide apoptosis detection kit was used to measure cell apoptosis. In addition, the levels of interleukin (IL)-1β, IL-6 and tumor necrosis factor (TNF)-α were determined using ELISA. Furthermore, gene and protein expression was measured via RT-qPCR and western blot assay, respectively. The results revealed that miR-195-5p was significantly upregulated in the articular cartilage tissues of patients with OA and in LPS stimulated ATDC5 cells. REGγ was a direct target of miR-195-5p. The repressed cell proliferation ability and enhanced cell apoptosis of ATDC5 cells induced by LPS were reversed by miR-195-5p downregulation. Furthermore, LPS stimulation significantly upregulated the levels of IL-1β, IL-6 and TNF-α, while miR-195-5p downregulation markedly reduced the expression of inflammatory factors induced by LPS. The results also revealed that a miR-195-5p inhibitor inhibited the LPS induced repression of the Wnt/β-catenin signaling pathway and activation of nuclear factor (NF)-κB signaling pathway in ATDC5 cells. Notably, the results of the present study also indicated that all of the effects of the miR-195-5p inhibitor on ATDC5 cells were reversed by REGγ silencing. In conclusion, the results indicated that the miR-195-5p inhibitor served a protective role in OA by inhibiting chondrocyte apoptosis and inflammatory responses by regulating the Wnt/β-catenin and NF-κB signaling pathways.
Keywords: osteoarthritis, microRNA-195-5p, REGγ, apoptosis, Wnt/β-catenin pathway, nuclear factor-κB pathway
Introduction
Osteoarthritis (OA) is a type of degenerative joint disease that seriously affects the health of the elderly, and is characterized by articular cartilage degradation and joint inflammation (1–3). The main clinical manifestations of OA include recurrent joint pain and gradually increased joint movement disorder. OA seriously endangers the physical health of middle-aged and elderly people, and greatly affects patient quality of life, causing a heavy burden for individuals, families and even society (4). Articular cartilage chondrocytes and extracellular matrix (ECM) are the two components of articular cartilage. As the only cell component in articular cartilage, the main function of chondrocytes is to maintain the metabolic homeostasis of the ECM (5,6). Apoptosis of chondrocytes is one of the main pathological features of OA (7,8). Clinically, the primary treatment methods of OA are to relieve joint pain, maintain or improve the physiological function of the joint, and protect the tissue structure of the joint; however, the current effects are unsatisfactory. Therefore, identifying a safe, reliable and effective OA treatment is an urgently required, long-term task. Currently, the pathogenesis of OA remains largely unclear; the study of cartilage damage is important to elucidate the pathogenesis of OA, providing a theoretical basis for OA prevention and intervention.
MicroRNAs (miRNAs/miRs), a family of small non-coding RNAs, 19–25 nucleotides in length, regulate gene expression by binding to the 3′ untranslated region (3′UTR) of the target genes (9,10). miRNA is involved in the regulation of many survival-associated activities such as organ development, cell proliferation, cell differentiation and apoptosis (9,11,12). Recently, an increasing body of research has shown that miRNAs serve an important role in cartilage formation and OA development (13,14). miR-195-5p, a member of the miR-15a/b/16/195/497 family, is located on chromosome 17p13.1 (15). In recent years, the role of miR-195-5p in tumors has been extensively studied (16–19). However, the function of miR-195-5p in OA is largely unknown.
The purpose of the present study was to investigate the differential expression level of miR-195-5p in normal and OA cartilage tissues, and to study the biological effects and mechanisms of miR-195-5p on LPS induced chondrocyte injury. We hope to explore novel therapeutic targets and provide new theoretical basis for the treatment of OA.
Materials and methods
Clinical samples
A total of 30 paired articular cartilage tissue samples (n=60 samples in total) from 30 OA patients (male, 17; female, 13; age 59.4±4.2 years) and 30 non-OA patients (no history of OA or rheumatoid arthritis; male, 16; female, 14; age 59.1±6.2 years) were collected at the Sixth Hospital of Wuhan (Hubei, China) from March 2016 to March 2017. The present study was approved by the Ethics Committee of the Sixth Hospital of Wuhan, and all patients provided written informed consent.
Cell culture and OA cell model establishment
The murine chondrogenic cell line ATDC5 was obtained from American Type Culture Collection (Manassas, VA, USA). ATDC5 cells were grown in Dulbecco's modified Eagle's medium/Ham's Nutrient Mixture F-12 (Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA) containing 10% fetal bovine serum (Gibco; Thermo Fisher Scientific, Inc.) and 2 mM Glutamine (Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) at 37°C with 5% CO2.
To establish the cell model of OA, ATDC5 cells were treated with 5 µg/ml lipopolysaccharide (LPS; Sigma-Aldrich; Merck KGaA) at 37°C for 5 h. Cells without any treatments were used as the control.
Cell transfection
Inhibitor control, miR-195-5p inhibitor, control-small interfering (si)-RNA and the REGγ-siRNA were purchased from Guangzhou RiboBio Co., Ltd. (Guangzhou, China). ATDC5 cells were seeded into a 6-well plate (1×106 cells/well) and cultured at 37°C for 24 h. Then, ATDC5 cells were transfected with 100 nM miR-195-5p inhibitor (5′-GCCAAUAUUUCUGUGCUGCUA-3′), 100 nM inhibitor control (5′-CAGUACUUUUGUGUAGUACAA-3′), 2 µl control-siRNA [5′-UUCUCCGAACGUGUCACGUTT-3′ (sense) and 5′-ACGUGACACGUUCGGAGAATT-3′ (antisense)], 2 µl REGγ-siRNA [5′-CAGAAGACUUGGUGGCAAATT-3′ (sense) and 5′-UUUGCCACCAAGUCUUCUGTT-3′ (antisense)] or 100 nM miR-195-5p inhibitor+2 µl REGγ-siRNA using Lipofectamine 3000® regent (Thermo Fisher Scientific, Inc.) according to the manufacturer's protocol. At 24 h post-cell transfection, transfection efficiency was detected via reverse transcription-quantitative polymerase chain reaction (RT-qPCR).
MTT assay
ATDC5 cells (5×103 cells/well) were cultured in 96-well plates. At 24 h post-transfection with either miR-195-5p inhibitor, inhibitor control or miR-195-5p inhibitor+REGγ-siRNA at 37°C, the cells were treated with LPS for 5 h at 37°C. Cells without LPS treatment were used as the control group. Following stimulation, MTT solution (20 µl/well) was added into the culture medium. Then the plates were incubated for 4 h at 37°C in humidified 95% air and 5% CO2. Then dimethyl sulfoxide (150 µl) was used to dissolve the purple formazan. Cell proliferation ability was assessed by measuring the absorbance at 490 nm using a Microplate Reader (Bio-Rad Laboratories, Inc., Hercules, CA, USA).
Apoptosis assay
To analyze cell apoptosis, flow cytometry was performed with the Annexin V-fluorescein isothiocyanate (FITC)/propidium iodide (PI) apoptosis Detection kit (cat. no. 70-AP101-100; Hangzhou MultiSciences Biotech, Co., Ltd., Hangzhou, China). Briefly, following the specific treatments, ATDC5 cells (1×105 cells/well) were collected and washed with cold PBS. Then the cells were stained with FITC-conjugated Annexin V and propidium iodide without light according to the manufacturer's instructions. Finally, the number of apoptotic cells was determined using a flow cytometer (BD Biosciences; Becton-Dickinson and Company, Franklin Lakes, NJ, USA), and FlowJo software version 7.6.1 (FlowJo LLC, Ashland, OR, USA) was used for data analysis.
Enzyme-linked immunosorbent assay (ELISA)
To detect the levels of IL-1β, IL-6 and TNF-α in cell supernatants, ELISA was performed. Briefly, the cell culture supernatant was collected by centrifugation (1,000 × g at 4°C for 10 min), then the concentrations of the inflammatory cytokines (IL-1β, cat. no. ab100704; IL-6, cat. no. ab100712 and TNF-α, cat. no. ab208348) were determined by ELISA (Abcam, Cambridge, UK) according to the manufacturer's instructions of each kit.
RT-qPCR
The total RNA was extracted from tissues and cells using TRIzol reagent (Invitrogen; Thermo Fisher Scientific, Inc.) as per the manufacturer's instructions. Then the TaqMan MicroRNA Reverse Transcription kit (Applied Biosystems; Thermo Fisher Scientific, Inc.) was used to perform the RT experiment. Amplification conditions for RT experiment were 50°C for 15 min and 85°C for 2 min. Finally, the synthesized cDNAs were analyzed using TaqMan Universal Master Mix II (Applied Biosystems; Thermo Fisher Scientific, Inc.) following the manufacturer's protocols. Amplification conditions were: 10 min at 95°C, followed by 35 cycles of 15 sec at 95°C and 40 sec at 55°C. GAPDH and U6 were used in the present study for normalizing mRNA and miRNA levels. Primer sequences were as following: GAPDH forward, 5′-CTTTGGTATCGTGGAAGGACTC-3′ and reverse, 5′-GTAGAGGCAGGGATGATGTTCT-3′; U6 forward, 5′-GCTTCGGCAGCACATATACTAAAAT-3′ and reverse, 5′-CGCTTCACGAATTTGCGTGTCAT-3′; TNF-α forward, 5′-GAACTGGCAGAAGAGGCACT-3′ and reverse, 5′-GGTCTGGGCCATAGAACTGA-3′; IL-1β forward, 5′-TGTGAAATGCCACCTTTTGA-3′ and reverse, 5′-TGAGTGATACTGCCTGCCTG-3′; IL-6 forward, 5′-CCGGAGAGGAGACTTCACAG-3′ and reverse, 5′-CAGAATTGCCATTGCACA-3′; miR-195-5p forward, 5′-GGGGTAGCAGCACAGAAAT-3′ and reverse, 5′-TCCAGTGCGTGTCGTGGA-3′; REGγ forward, 5′-AAGGTTGATTCTTTCAGGGAGC-3′ and reverse, 5′-AGTGGATCTGAGTTAGGTCATGG-3′. Relative gene expressions were calculated by the 2−ΔΔCq method (20).
Dual luciferase activity assay
The targets of miR-195-5p were predicted using TargetScan bioinformatics software 7.1 (www.targetscan.org/vert_71), and REGγ was revealed to be a potential target of miR-195-5p. To verify this prediction, the wild-type (REGγ-WT) and mutant (REGγ-MUT) 3′UTR of REGγ were cloned into a pmiR-RB-Report™ dual luciferase reporter gene plasmid vector (Guangzhou RiboBio Co., Ltd.). ATDC5 cells (5×104 cells/well) were co-transfected with 100 ng REGγ-WT or 100 ng REGγ-MUT and 50 nM miR-195-5p mimic (5′-UAGCAGCACAGAAAUAUUGGC-3′) or 50 nM mimic control (5′-UUCUCCGAACGUGUCACGUTT-3′) using Lipofectamine 3000® regent (Thermo Fisher Scientific, Inc.) according to the manufacturer's protocols. At 48 h post-transfection, the dual-luciferase assay system (Promega Corporation, Madison, WI, USA) was used to determine luciferase activity, which was normalized to Renilla luciferase activity.
Western blot assay
Whole cell proteins were extracted from cells (5×105 cells/well) using Radioimmunoprecipitation Assay Lysis buffer (Beyotime Institute of Biotechnology, Shanghai, China) according to the manufacturer's protocols. The protein samples were quantified using the BCA™ Protein Assay kit (Pierce; Thermo Fisher Scientific, Inc.). Western blotting was performed using a Bio-Rad Bis-Tris Gel system (Bio-Rad Laboratories, Inc.) following the manufacturer's instructions. Briefly, protein samples (30 µg/lane) were separated by 12% SDS-PAGE and then transferred onto polyvinylidene difluoride membranes. Then the membranes were blocked with 5% non-fat milk at room temperature for 1.5 h, incubated with primary antibodies at 4°C overnight, and subsequently incubated with a secondary antibody at room temperature for 4 h. The primary antibodies used in this study were REGγ (cat. no. ab157157; 1:1,000; Abcam), Bcl-2 (cat. no. 4223; 1:1,000), Bax (cat. no. 14796; 1:1,000), β-catenin (cat. no. 25362; 1:1,000), c-Myc (cat. no. 5605; 1:1,000), Cyclin D1 (cat. no. 2978; 1:1,000), p-p65 (cat. no. 3033; 1:1,000) and β-actin (cat. no. 4970; 1:1,000) (all from Cell Signaling Technology, Inc., Danvers, MA, USA). The horseradish peroxidase-conjugated secondary antibody was obtained from Cell Signaling Technology, Inc. (cat. no. 7074; 1:2,000). At the end of this experiment, protein bands were visualized using the enhanced chemiluminescence detection system (Super Signal West Dura Extended Duration Substrate; Pierce; Thermo Fisher Scientific, Inc.) according to the manufacturer's instructions. Gel-Pro Analyzer densitometry software (version 6.3; Media Cybernetics, Inc., Rockville, MD, USA) was used for band density quantification.
Statistical analysis
All experiments were repeated three times. The data were presented as the mean ± standard deviation. Statistical analyses were carried out using SPSS 17.0 statistical software (SPSS Inc., Chicago, IL, USA). Comparisons between two groups were evaluated by Student's t-test, and comparisons between multiple groups were analyzed using one-way analysis of variance followed by Tukey's post hoc test. P<0.05 was considered statistically significant.
Results
miR-195-5p is upregulated in OA
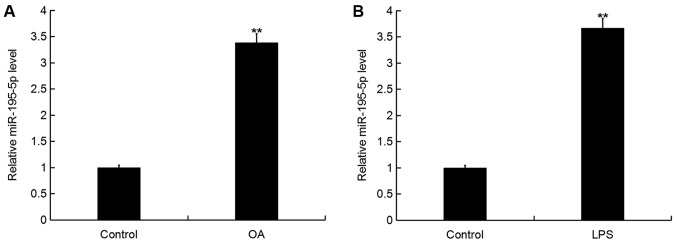
To determine the miR-195-5p expression level in OA, RT-qPCR was performed. As shown in Fig. 1A, when compared with the control group, the level of miR-195-5p was significantly upregulated in the articular cartilage tissues of patients with OA. In addition, the level of miR-195-5p in the murine chondrogenic cell line ATDC5 was significantly increased following LPS treatment for 5 h in comparison with the control group (Fig. 1B).
Figure 1.
miR-195-5p expression in OA. (A) The level of miR-195-5p in the articular cartilage tissues of patients with or without OA. (B) The level of miR-195-5p in the murine chondrogenic cell line ATDC5 with or without LPS treatment. Data were displayed as the mean ± standard deviation. **P<0.01 vs. Control group. miR, microRNA; OA, osteoarthritis; LPS, lipopolysaccharide.
REGγ is a target of miR-195-5p
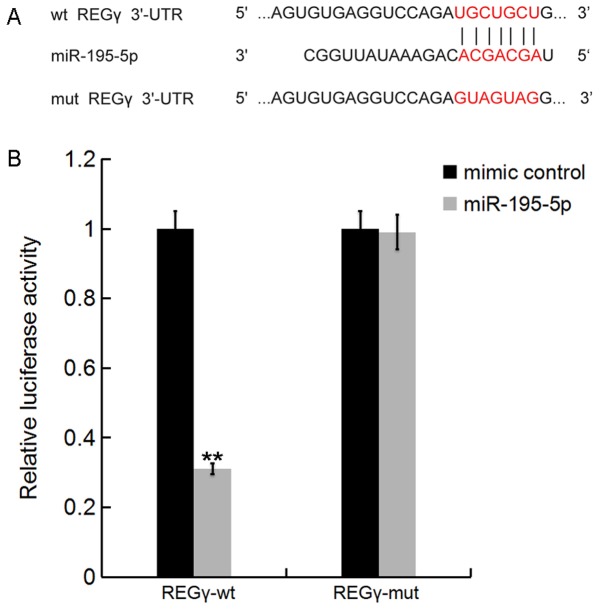
Bioinformatics tools on TargetScan (www.targetscan.org) were used to predict the potential targets of miR-195-5p. The results revealed that REGγ was a potential target for miR-195-5p (Fig. 2A). To determine whether miR-195-5p directly modulated REGγ expression via interactions with potential binding sites, a luciferase reporter assay was performed. As shown in Fig. 2B, compared with co-transfection with REGγ-MUT and miR-195-5p mimic, luciferase activity was markedly decreased by co-transfection with REGγ-WT and miR-195-5p mimic. The results indicated that miR-195-5p directly targeted REGγ.
Figure 2.
REGγ is a target of miR-195-5p. (A) Interactions between miR-195-5p and the 3′UTR of REGγ were predicted using microRNA target site prediction software. (B) Luciferase activity of a reporter containing a WT REGγ 3′UTR or a MUT REGγ 3′UTR are presented. ‘REGγ-MUT’ indicates the REGγ 3′UTR with a mutation in the miR-195-5p binding site. All data are presented as the mean ± standard deviation of three independent experiments. **P<0.01 vs. mimic control group. WT, wild-type; MUT, mutant; UTR, untranslated region; miR, microRNA.
miR-195-5p inhibitor enhances the ATDC5 cell proliferation ability inhibited by LPS
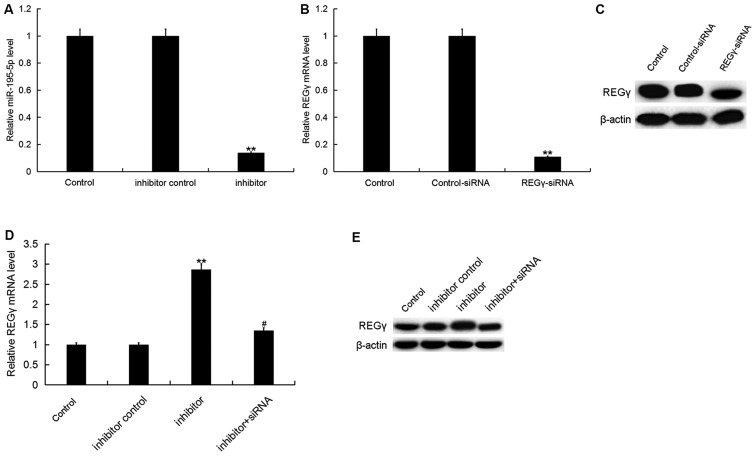
To investigate the effect of miR-195-5p on OA, the present study firstly transfected ATDC5 cells with miR-195-5p inhibitor, inhibitor control, control-siRNA, REGγ-siRNA or miR-195-5p inhibitor+REGγ-siRNA respectively, and the transfection efficiency was detected 24 h following cell transfection. As shown in Fig. 3A, compared with the control group, miR-195-5p inhibitor significantly downregulated the miR-195-5p level in ATDC5 cells. REGγ-siRNA significantly reduced the protein and mRNA levels of REGγ in ATDC5 cells (Fig. 3B and C). In addition, the miR-195-5p inhibitor significantly upregulated the protein and mRNA levels of REGγ, and this upregulation was attenuated by REGγ-siRNA (Fig. 3D and E).
Figure 3.
miR-195-5p inhibitor enhances REGγ expression in ATDC5 cells. (A) Relative miR-195-5p expression in ATDC5 cells in the different groups. (B and D) Relative REGγ mRNA expression in ATDC5 cells in the different groups. (C and E) Protein levels of REGγ in ATDC5 cells in the different groups. Data were displayed as the mean ± standard deviation. **P<0.01 vs. control group; #P<0.05 vs. inhibitor group. miR, microRNA; siRNA, small interfering RNA.
Then, an MTT assay was conducted to determine the effect of miR-195-5p on ATDC5 cell proliferation ability. The data showed that the miR-195-5p inhibitor significantly enhanced the proliferation ability of ATDC5 cells, which was inhibited by LPS treatment, and this enhancement was markedly reversed by REGγ-siRNA (Fig. 4).
Figure 4.
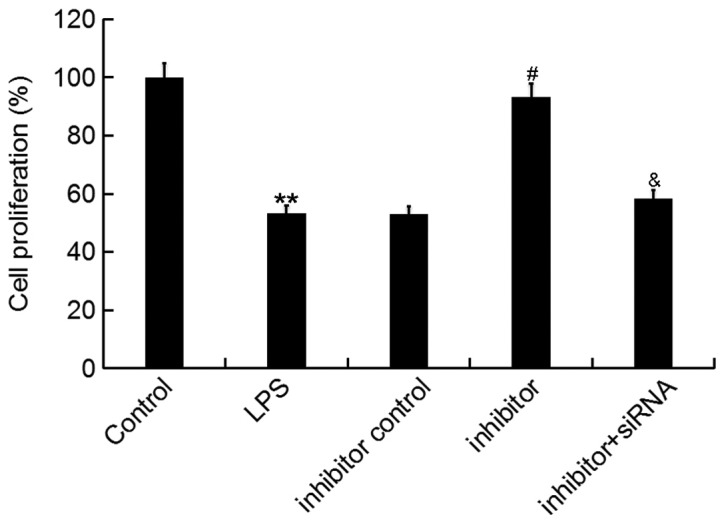
Effect of the miR-195-5p inhibitor on ATDC5 cell proliferation ability. At 24 h post-transfection with either the miR-195-5p inhibitor, inhibitor control, or miR-195-5p inhibitor+REGγ-siRNA at 37°C, the cells were treated with LPS for 5 h. Cells without any treatment were used as the control group. Then, ATDC5 cell proliferation ability was assessed by an MTT assay. Data were presented as the mean ± standard deviation. **P<0.01 vs. control group; #P<0.05 vs. LPS group; &P<0.05 vs. inhibitor group. miR, microRNA; siRNA, small interfering RNA; LPS, lipopolysaccharide.
miR-195-5p inhibitor reduces the ATDC5 cell apoptosis induced by LPS
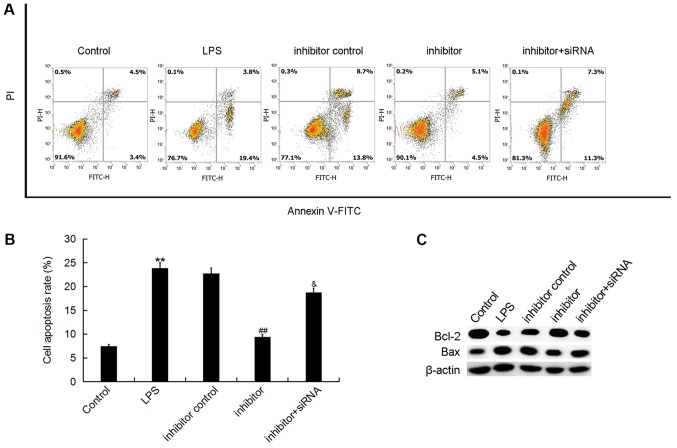
The present study further investigated the effect of miR-195-5p on ATDC5 cell apoptosis via flow cytometry. The results indicated that LPS treatment significantly induced ATDC5 cell apoptosis, but this increase in cell apoptosis was inhibited by the miR-195-5p inhibitor. Notably, the results also indicated that REGγ-siRNA significantly eliminated the effect of the miR-195-5p inhibitor on ATDC5 cell apoptosis (Fig. 5A and B). In addition, we the levels of increased Bax and decreased Bcl-2 levels induced by LPS treatment were reversed by the miR-195-5p inhibitor. Furthermore, REGγ-siRNA significantly eliminated the effect of the miR-195-5p inhibitor on the expression of Bcl-2 and Bax in LPS treated ATDC5 cells (Fig. 5C).
Figure 5.
Effect of miR-195-5p inhibitor on ATDC5 cell apoptosis. At 24 h post-transfection with either the miR-195-5p inhibitor, inhibitor control, or miR-195-5p inhibitor+REGγ-siRNA at 37°C, the cells were treated with LPS for 5 h. Cells without any treatments were used as the control group. Then, (A) ATDC5 cell apoptosis was assessed by using flow cytometry and (B) the cell apoptosis rate was calculated. (C) The protein levels of Bax and Bcl-2 were determined by western blotting. Data were displayed as the mean ± standard deviation. **P<0.01 vs. control group; ##P<0.01 vs. LPS group; &P<0.05 vs. inhibitor group. miR, microRNA; siRNA, small interfering RNA; LPS, lipopolysaccharide; Bcl-2, B-cell lymphoma-2; Bax, Bcl-2-associated X; FITC, fluorescein isocyanate; PI, propidium iodide.
miR-195-5p inhibitor prevents the inflammatory response in ATDC5 cells induced by LPS
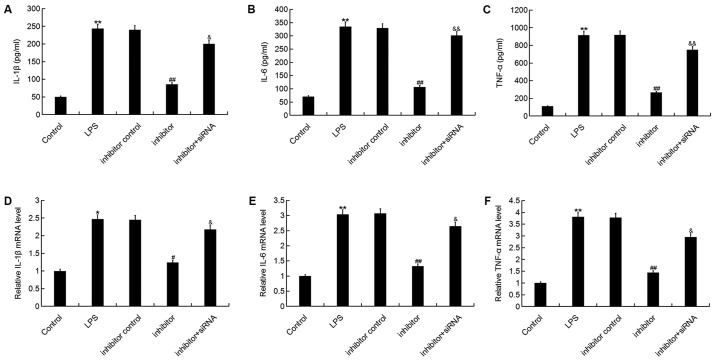
The present study then investigated whether miR-195-5p has an effect on the inflammatory response during OA development. The results demonstrated that the mRNA and protein levels of IL-1β, IL-6 and TNF-α were significantly enhanced by LPS stimulation. The miR-195-5p inhibitor significantly reduced the expression levels of IL-1β, IL-6 and TNF-α, and these reductions were reversed by REGγ downregulation (Fig. 6). These results indicated that the miR-195-5p inhibitor prevented the inflammatory response in ATDC5 cells induced by LPS.
Figure 6.
Effect of the miR-195-5p inhibitor on inflammatory factor expression in ATDC5 cells. At 24 h post-transfection with either the miR-195-5p inhibitor, inhibitor control, or miR-195-5p inhibitor+REGγ-siRNA at 37°C, the cells were treated with LPS for 5 h. Cells without any treatment were used as the control group. Then, the (A-C) protein and (D-F) mRNA levels of (A and D) IL-1β, (B and E) IL-6 and (C and F) TNF-α in ATDC5 cells were detected by reverse transcription-quantitative polymerase chain reaction and ELISA assay. Data were presented as the mean ± standard deviation. *P<0.05 and **P<0.01 vs. control group; #P<0.05 and ##P<0.01 vs. LPS group; &P<0.05 and &&P<0.01 vs. inhibitor group. miR, microRNA; siRNA, small interfering RNA; LPS, lipopolysaccharide; IL, interleukin; TNF, tumor necrosis factor.
miR-195-5p inhibitor represses LPS-induced Wnt/β-catenin and nuclear factor (NF)-κB signaling pathway activation in ATDC5 cells
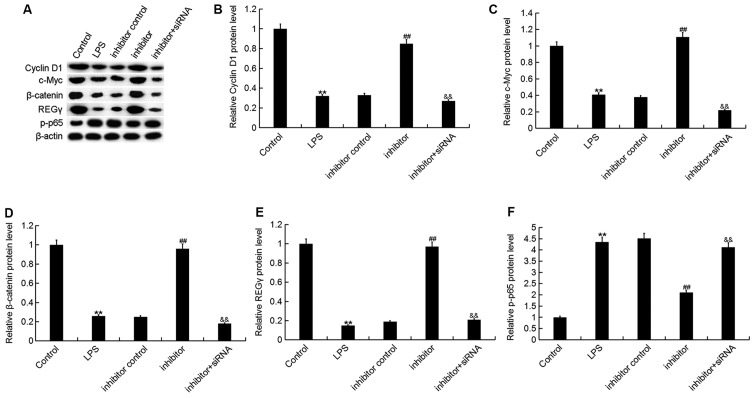
Finally, to explore the molecular mechanism underlying the effects of the miR-195-5p inhibitor on ATDC5 cells, the Wnt/β-catenin and NF-κB signaling pathways were analyzed. As shown in Fig. 7, LPS stimulation significantly decreased the protein expression of REGγ, β-catenin, c-Myc and Cyclin D1, and enhanced the phosphorylation of NF-κB [phosphorylated (p)-p65] in ATDC5 cells, while the miR-195-5p inhibitor markedly increased REGγ, β-catenin, c-Myc and Cyclin D1 protein expression and reduced p-NF-κB (p-p65) protein expression. As expected, REGγ-siRNA significantly attenuated the effect of the miR-195-5p inhibitor on REGγ, β-catenin, c-Myc, Cyclin D1 and p-p65 expression in ATDC5 cells.
Figure 7.
Effect of the miR-195-5p inhibitor on Wnt/β-catenin and NF-κB signaling pathway activation in ATDC5 cells. At 24 h post-transfection with either the miR-195-5p inhibitor, inhibitor control, or miR-195-5p inhibitor+REGγ-siRNA at 37°C, the cells were treated with LPS for 5 h. Cells without any treatment were used as the control group. (A) Then, the protein levels of (B) Cyclin D1, (C) c-Myc, (D) β-catenin, (E) REGγ and (F) p-p65 were detected using western blot assay and Gel-Pro Analyzer densitometry software was used for band density quantification. Data were displayed as the mean ± standard deviation. **P<0.01 vs. control group; ##P<0.01 vs. LPS group; &&P<0.01 vs. inhibitor group. miR, microRNA; siRNA, small interfering RNA; LPS, lipopolysaccharide; p-, phosphorylated.
Discussion
In the present study, miR-195-5p expression was significantly increased in patients with OA and the OA cell model. miR-195-5p downregulation repressed the effects of LPS on reduced cell proliferation ability, enhanced cell apoptosis and increased inflammation-associated factor expression by targeting REGγ. In addition, the results suggested that miR-195-5p downregulation inhibited LPS-induced Wnt/β-catenin signaling pathway repression and activation of the NF-κB signaling pathway in ATDC5 cells. The results of the present study indicated that the miR-195-5p inhibitor protected chondrocytes from LPS-induced injury, therefore it may be a promising therapeutic target for OA treatment.
OA is an orthopedic multiple degenerative inflammatory disease. The main pathological manifestation of OA is the destruction of the structural and functional integrity of articular cartilage and the inflammatory response (21). The onset of OA is a complex process involving multiple factors. Currently, the etiology and pathogenesis of OA are not fully understood (22). Chondrocytes serve a very important role in the pathogenesis of OA (23). Therefore, studies on chondrocyte injury have potential significance for identifying effective methods for treating OA.
Recently, an increasing body of evidence has revealed that miRNAs serve an important role in cartilage formation and OA development (13,14). miR-195-5p, a member of the miR-15a/b/16/195/497 family, is currently extensively studied in the development of tumors (16–19). However, the role of miR-195-5p in OA is unknown. Therefore, we conducted the present study.
The present study firstly detected the expression level of miR-195-5p in OA, and the results revealed that miR-195-5p was significantly upregulated in the articular cartilage tissues of patients with OA and in the LPS-induced OA cell model, indicating the potential role of miR-195-5p in OA. Then, it was identified that REGγ, a proteasome activator, was a direct target of miR-195-5p.
During the progression of OA, areas with severe matrix degradation and destruction are often associated with the excessive apoptosis of chondrocytes (24). The excessive apoptosis of chondrocytes is considered to be one of the key factors in the pathogenesis of OA (25). The present study demonstrated that the inhibited cell proliferation ability, and increased cell apoptosis of chondrocytes induced by LPS treatment were inhibited by miR-195-5p downregulation. Another important discovery was that REGγ knockdown significantly eliminated the effect of miR-195-5p on chondrocyte apoptosis.
Chondrocytes can promote cartilage degradation through the secretion of inflammatory factors (26). A large number of cytokines, especially inflammatory factors, are involved in the regulation and maintain the dynamic balance of articular cartilage. Inflammatory factors can stimulate cascades in many ways, continuously stimulating chondrocytes to secrete cytokines, which in turn triggers OA (27–29). In the present study, LPS significantly enhanced the levels of IL-1β, IL-6 and TNF-α, while miR-195-5p inhibitor reversed these enhancements. It is worth mentioning that the effect of the miR-195-5p inhibitor on the expression of inflammatory factors in chondrocytes was eliminated by REGγ silencing.
Finally, to explore the underlying mechanism of the effects of the miR-195-5p inhibitor on LPS-induced chondrocyte injury, the Wnt/β-catenin and NF-κB signaling pathways were analyzed. The results revealed that the miR-195-5p inhibitor inhibited the LPS-induced Wnt/β-catenin signaling pathway repression and NF-κB signaling pathway activation in chondrocytes, and this inhibition was reversed by REGγ knockdown.
In conclusion, to the best of our knowledge the present study has reported for the first time that miR-195-5p was abnormally high in OA, and its inhibition could inhibit chondrocyte apoptosis and the inflammatory response in an OA cell model by targeting REGγ. miR-195-5p may be a novel and promising therapeutic target for the treatment of OA. However, this is a preliminary study of the role of miR-195-5p in OA, and in order to make the role of miR-195-5p in OA more convincing, a lot of further research is still required. For example, the effect of miR-195-5p overexpression on the OA cell model still requires investigation. In addition, the expression of REGγ in the articular cartilage tissues of patients with OA and the LPS-induced cell model should be revealed. Furthermore, the in vitro study of OA is significantly different from the in vivo OA in humans, and some in vivo and clinical studies of miR-195-5p in OA are required to confirm the role of miR-195-5p in OA. In the future, we will perform in-depth research on these issues.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
YS contributed to study design and data collection; JL contributed to statistical analysis; WG contributed to data interpretation; WY contributed to manuscript preparation and statistical analysis.
Ethics approval and consent to participate
The present study was approved by the Ethics Committee of the Sixth Hospital of Wuhan, and all patients provided written informed consent.
Patient consent for publication
Informed patient consent was obtained to publish.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Johnson VL, Hunter DJ. The epidemiology of osteoarthritis. Best Pract Res Clin Rheumatol. 2014;28:5–15. doi: 10.1016/j.berh.2014.01.004. [DOI] [PubMed] [Google Scholar]
- 2.Loeser RF, Goldring SR, Scanzello CR, Goldring MB. Osteoarthritis: A disease of the joint as an organ. Arthritis Rheum. 2012;64:1697–1707. doi: 10.1002/art.34453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Burr DB, Gallant MA. Bone remodelling in osteoarthritis. Nat Rev Rheumatol. 2012;8:665–673. doi: 10.1038/nrrheum.2012.130. [DOI] [PubMed] [Google Scholar]
- 4.Salmon JH, Rat AC, Sellam J, Michel M, Eschard JP, Guillemin F, Jolly D, Fautrel B. Economic impact of lower-limb osteoarthritis worldwide: A systematic review of costof-illness studies. Osteoarthritis Cartilage. 2016;24:1500–1508. doi: 10.1016/j.joca.2016.03.012. [DOI] [PubMed] [Google Scholar]
- 5.Goldring MB, Goldring SR. Osteoarthritis. J Cell Physiol. 2007;213:626–634. doi: 10.1002/jcp.21258. [DOI] [PubMed] [Google Scholar]
- 6.Qin J, Shang L, Ping AS, Li J, Li XJ, Yu H, Magdalou J, Chen LB, Wang H. Response to ‘TNF/TNFR signal transduction pathway-mediated anti-apoptosis and anti-inflammatory effects of sodium ferulate on IL-1β-induced rat osteoarthritis chondrocytes in vitro’-authors' reply. Arthritis Res Ther. 2013;15:409. doi: 10.1186/ar4227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kim HA, Lee YJ, Seong SC, Choe KW, Song YW. Apoptotic chondrocyte death in human osteoarthritis. J Rheumatol. 2000;27:455–462. [PubMed] [Google Scholar]
- 8.Yan S, Wang M, Zhao J, Zhang H, Zhou C, Jin L, Zhang Y, Qiu X, Ma B, Fan Q. MicroRNA-34a affects chondrocyte apoptosis and proliferation by targeting the SIRT1/p53 signaling pathway during the pathogenesis of osteoarthritis. Int J Mol Med. 2016;38:201–209. doi: 10.3892/ijmm.2016.2618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bartel DP. MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell. 2004;116:281–297. doi: 10.1016/S0092-8674(04)00045-5. [DOI] [PubMed] [Google Scholar]
- 10.Kim J, Yao F, Xiao Z, Sun Y, Ma L. MicroRNAs and metastasis: Small RNAs play big roles. Cancer Metastasis Rev. 2018;37:5–15. doi: 10.1007/s10555-017-9712-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chen CZ, Li L, Lodish HF, Bartel DP. MicroRNAs modulate hematopoietic lineage differentiation. Science. 2004;303:83–86. doi: 10.1126/science.1091903. [DOI] [PubMed] [Google Scholar]
- 12.Fujii T, Shimada K, Nakai T, Ohbayashi C. MicroRNAs in smoking-related carcinogenesis: Biomarkers, functions, and therapy. J Clin Med. 2018;7:E98. doi: 10.3390/jcm7050098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wu C, Tian B, Qu X, Liu F, Tang T, Qin A, Zhu Z, Dai K. MicroRNAs play a role in chondrogenesis and osteoarthritis (Review) Int J Mol Med. 2014;34:13–23. doi: 10.3892/ijmm.2014.1743. [DOI] [PubMed] [Google Scholar]
- 14.Trachana V, Ntoumou E, Anastasopoulou L, Tsezou A. Studying microRNAs in osteoarthritis: Critical overview of different analytical approaches. Mech Ageing Dev. 2018;171:15–23. doi: 10.1016/j.mad.2018.02.006. [DOI] [PubMed] [Google Scholar]
- 15.He JF, Luo YM, Wan XH, Jiang D. Biogenesis of miRNA-195 and its role in biogenesis, the cell cycle, and apoptosis. J Biochem Mol Toxicol. 2011;25:404–408. doi: 10.1002/jbt.20396. [DOI] [PubMed] [Google Scholar]
- 16.Wang Y, Zhang X, Zou C, Kung HF, Lin MC, Dress A, Wardle F, Jiang BH, Lai L. miR-195 inhibits tumor growth and angiogenesis through modulating IRS1 in breast cancer. Biomed Pharmacother. 2016;80:95–101. doi: 10.1016/j.biopha.2016.03.007. [DOI] [PubMed] [Google Scholar]
- 17.Guo J, Wang M, Liu X. MicroRNA-195 suppresses tumor cell proliferation and metastasis by directly targeting BCOX1 in prostate carcinoma. J Exp Clin Cancer Res. 2015;34:91. doi: 10.1186/s13046-015-0209-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wang M, Zhang J, Tong L, Ma X, Qiu X. miR-195 is a key negative regulator of hepatocellular carcinoma metastasis by targeting FGF2 and VEGFA. Int J Clin Exp Pathol. 2015;8:14110–14120. [PMC free article] [PubMed] [Google Scholar]
- 19.Chen S, Wang L, Yao X, Chen H, Xu C, Tong L, Shah A, Huang T, Chen G, Chen J, et al. miR-195-5p is critical in REGγ-mediated regulation of wnt/β-catenin pathway in renal cell carcinoma. Oncotarget. 2017;8:63986–64000. doi: 10.18632/oncotarget.19256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 21.Taruc-Uy RL, Lynch SA. Diagnosis and treatment of osteoarthritis. Prim Care. 2013;40(vii):821–836. doi: 10.1016/j.pop.2013.08.003. [DOI] [PubMed] [Google Scholar]
- 22.Creamer P, Hochberg MC. Osteoarthritis. Lancet. 1997;350:503–508. doi: 10.1016/S0140-6736(05)62494-4. [DOI] [PubMed] [Google Scholar]
- 23.Sanded LJ, Aigner T. Articular cartilage and changes in arthritis. An introduction: Cell biology of osteoarthritis. Arthritis Res. 2001;3:107–113. doi: 10.1186/ar148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Héraud F, Héraud A, Harmand M. Apoptosis in normal and human articular cartilage. Ann Rheum Dis. 2000;59:959–965. doi: 10.1136/ard.59.12.959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kawaguchi H. Endochondral ossification signals in cartilage degradation osteoarthritis progression in experimental mouse models. Mol cells. 2008;25:1–6. [PubMed] [Google Scholar]
- 26.Henrotin YE, De Groote DD, Labasse AH, Gaspar SE, Zheng SX, Geenen VG, Reginster JY. Effects of exogenous IL-1 beta, TNF alpha, IL-6, IL-8 and LIF on cytokine production by human articular chondrocytes. Osteoarthritis Cartilage. 1996;4:163–173. doi: 10.1016/S1063-4584(96)80012-4. [DOI] [PubMed] [Google Scholar]
- 27.Marks PH, Donaldson ML. Inflammatory cytokine profiles associated with damage in the anterior cruciate ligament-deficient knee. Arthroscopy. 2005;21:1342–1347. doi: 10.1016/j.arthro.2005.08.034. [DOI] [PubMed] [Google Scholar]
- 28.Stannus OP, Jones G, Blizzard L, Cicuttini FM, Ding C. Associations between serum levels of inflammatory markers and change in knee pain over 5 years in older adults: A prospective cohort study. Ann Rheum Dis. 2013;72:535–540. doi: 10.1136/annrheumdis-2011-201047. [DOI] [PubMed] [Google Scholar]
- 29.Hu G, Zhao X, Wang C, Geng Y, Zhao J, Xu J, Zuo B, Zhao C, Wang C, Zhang X. MicroRNA-145 attenuates TNF-α-driven cartilage matrix degradation in osteoarthritis via direct suppression of MKK4. Cell Death Dis. 2017;8:e3140. doi: 10.1038/cddis.2017.522. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.