Abstract
Objective
Patients with rheumatoid arthritis (RA) suffer from more cardiovascular disease (CVD), and develop cardiovascular risk factors at an earlier age than the general population. Cardiorespiratory fitness (CRF) is an important predictor of cardiovascular health. There are few data regarding CRF of RA patients, measured as peak oxygen uptake (VO2peak) by the gold standard method; cardiopulmonary exercise testing. We compared CRF in RA patients to those from a healthy population, and investigated if risk factors for CVD and RA-specific variables including subjective and objective disease activity measures were associated with CRF in RA patients.
Methods
VO2peak tests of RA patients (n=93) were compared to those of an age-matched and gender-matched healthy population (n=4631) from the Nord-Trøndelag Health Study. Predictors of VO2peak were found using Lasso (least absolute shrinkage and selection operator) regression, followed by standardised multiple linear regression.
Results
Women with RA ≥40 years and men with RA aged 40–49 years or 60–69 years had up to 20% lower CRF than the healthy population in the same age groups. By relative importance, body mass index (standardised coefficient=−0.25, p<0.001), physical activity level (coefficient=0.21, p<0.001), patient global assessment (PGA; coefficient=−0.14, p=0.006), systolic blood pressure (coefficient=−0.12, p=0.016), resting heart rate (coefficient=−0.11, p=0.032) and smoking (coefficient=−0.10, p=0.046) were significant predictors of CRF (R2=0.82, gender-adjusted and age-adjusted).
Conclusion
CRF in RA patients was lower than in a healthy population. CRF was associated with common risk factors for CVD and the PGA score. Focusing on fitness in RA patients may improve cardiovascular health.
Keywords: rheumatoid arthritis, cardiovascular disease, patient perspective, epidemiology
Key messages.
What is already known about this subject?
Cardiovascular disease (CVD) is inversely associated with cardiorespiratory fitness (CRF), and patients with rheumatoid arthritis (RA) suffer from more CVD and develop cardiovascular risk factors at an earlier age than the general population.
What does this study add?
The variables most strongly associated with the CRF level in RA patients were body mass index (BMI), physical activity level and the patient global assessment (PGA).
Contradictory to earlier suggestions that objective measures of RA disease activity are related to CRF, the subjective patient assessment captured as the PGA was the only RA-related variable associated with CRF in this study.
How does this impact on clinical practice?
Physical activity should be assessed and acted on in RA patients because it may positively change both their BMI and CRF level.
Assessing the PGA may be a simple method to capture the patient’s subjective factors influencing the physical activity level.
Introduction
Cardiorespiratory fitness (CRF) is inversely associated with the risk for cardiovascular disease (CVD) in the general population.1–6 The CRF level influences prognosis after myocardial infarction and coronary artery bypass surgery,7 8 and the American Heart Association now regards CRF as a clinical vital sign which associates inversely with prognosis after several diseases and conditions.9 Patients with rheumatoid arthritis (RA) also suffer from more CVD, develop cardiovascular risk factors at an earlier age,10 11 and have an increased death rate due to CVD compared with age-matched controls.11–15 Both RA-specific and general risk factors have been implicated, including the RA-associated systemic inflammation process that affects the vasculature of internal organs.
Studies suggest that RA patients might be less physically active due to fatigue, incomplete RA disease control, pain and/or structural changes of the joints.16 17 In practice, changing frequency, duration and/or intensity of physical activity is the only way to improve CRF. Physical inactivity might therefore contribute to worsened CRF and greater prevalence of CVD.18 19 Thus, low CRF may contribute to the risk of CVD in RA patients in addition to an increased burden of known cardiovascular risk factors, such as smoking, lipid levels and hypertension.11–15
Cardiopulmonary exercise testing with direct measurement of peak oxygen uptake (VO2peak) is the gold standard method for CRF assessment. However, VO2peak in RA patients has rarely been compared with VO2peak in healthy age-matched and gender-matched controls from the same population.18 20 21 Moreover, many studies were performed before biological disease-modifying anti-rheumatic drugs (DMARDs) became part of standard treatment regimens.
Given the excess of CVD and indications of limited physical activity in RA patients, we hypothesised that CRF in RA patients is lower than in a healthy age-matched and gender-matched population. This study therefore had three aims: (1) to measure VO2peak in RA patients and compare the results with VO2peak measurements from a healthy age-matched and gender-matched population; (2) to investigate variables that potentially were associated with VO2peak in RA patients, including both RA-specific variables and general risk factors for CVD, and evaluate their relative importance in our population.
Patients and methods
RA patients fulfilling the 1987 American College of Rheumatology (ACR)22 and/or the 2010 ACR/European League Against Rheumatism (EULAR) classification criteria for RA23 24 were recruited from St. Olavs Hospital’s Rheumatology outpatient clinic from 17 February 2017 to 04 January 2018. Since 01 May 2013, RA patients with stable disease activity have been transferred to a patient-centred follow-up programme where the patient’s general practitioner (GP) performs check-ups. If necessary, either the GP or the patient may contact the rheumatology clinic for an appointment. The remaining RA patients attend regular visits at the outpatient clinic. To include participants representing different disease activities, both types of patients were recruited to the present study. Exclusion criteria were unstable heart conditions, chronic obstructive/restrictive pulmonary disease (COPD/CRPD) necessitating use of oxygen therapy, or physical disability making a treadmill test impossible.
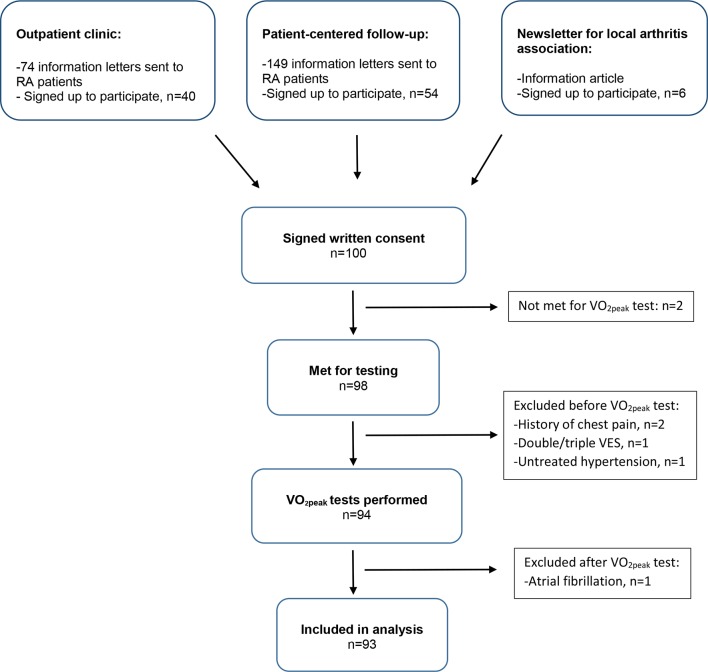
Three different recruitment schemes were used (figure 1): (1) RA patients attending regular visits were recruited during an appointment at the clinic; (2) a random selection of RA patients from the patient-centred follow-up list were contacted by mail; (3) a few RA patients signed up after reading information from the local arthritis association.
Figure 1.
Recruitment to the study. RA, rheumatoid arthritis; VES, ventricular extrasystoles.
Power calculations were based on the following assumptions: from the literature, we assumed that the most relevant variables associated with fitness in the general population (ie, age, gender, waist circumference, resting heart rate [RHR] and an index of physical activity)25 would explain 60% of the variance in measured VO2peak (ie, R2=0.60). Given α=0.05 and a planned inclusion of 100 RA patients, we would be able to identify one or more RA-related variables that would increase R2 to 0.65 with a power of 0.96, which was considered very satisfactory. In reality, useful data were available from 93 patients, which resulted in a power of 0.95 to detect this increase in R2.
Cardiopulmonary exercise testing to measure VO2peak was performed at the NeXt Move core facility at NTNU—Norwegian University of Science and Technology. Because RA patients sometimes exhibit physical limitations, experienced personnel determined the best individual cardiopumonary exercise testing (CPET) regimen during a 6 min warm-up on a treadmill (Woodway PPS 55, Waukesha, Wisconsin, USA), by detecting functional walking or running speed and inclination, as well as subjective moderate aerobic intensity based on rated perceived exertion (RPE Borg scale 6–20).26 Participants were then fitted with a heart rate monitor (H7, Polar Electro, Kempele, Finland) and facemask (7450 Series V2 CPET mask, Hans Rudolph, Shawnee, Kansas, USA). During an initial period of 4 min at fixed submaximal workload serving as an extended warm-up, work economy measurements were made.
An individualised ramp protocol was used, until either exhaustion or fulfilment of the criteria for VO2max or VO2peak. Workload was gradually increased, and gas measurements were recorded every 10th second using a mixing chamber ergospirometry system (Metalyzer II, Cortex Biophysik Gmbh, Leipzig, Germany). Maximal oxygen uptake (VO2max) was defined using the following criteria: (1) VO2 levelling off (<2 mL/(kg x min)) despite increase in workload and (2) respiratory exchange ratio ≥1.05. If these criteria were not met, the term VO2peak was used. A participant’s VO2peak was defined as the mean of the three successive highest VO2 registrations achieved during the CPET. Of the 93 patients tested, 17.2% qualified for VO2peak. For simplicity, the term VO2peak is used for all patients.
Participants rated their RPE on the Borg scale before, during and at the peak of the test, using a 6–20 scale.26 At the same time points, they also graded their lower extremities joint pain due to RA. Grading was similar to the RPE Borg scale, but instead focusing on pain: 6=‘Very, very light pain,’ 9=‘Very light pain,’ 11=‘Fairly light pain,’ 13=‘Somewhat painful,’ 15=‘Strong pain,’ 17=‘Very strong pain’ and 20=‘Very, very strong pain.’ Ratings below 6 were equivalent to ‘no pain.’ Age-predicted maximal heart rate was not used because it is does not account for the large normal variation. Lactate measurements were considered unnecessary because we measured the increase in respiratory exchange ratio, which is caused by and strongly correlated to lactate. The protocol for VO2peak testing did not include a verification phase because there is no general agreement on how it should be performed.
Before the physical test, a blood sample for measurement of high sensitivity C reactive protein (hsCRP) was drawn. A rheumatologist recorded the number of tender and swollen joints, height, weight, physician global assessment,27 rheumatoid factor (RF), anti-citrullinated protein antibody, present/previous RA medication, disease duration and information on comorbidity (cancer, CVD, diabetes, COPD/CRPD) from an interview and medical records. Blood pressure (BP) and RHR were measured after 10 min of rest. RHR was electronically measured using a pulse watch, and compared with manually counted RHR in 15 randomly selected participants to ensure that readings were correct. Participants filled in the modified Health Assessment Questionnaire (mHAQ)28 and the patient global assessment (PGA),27 as well as self-reported smoking (never vs ever). A questionnaire from the Nord-Trøndelag Health Study29 on physical activity habits, grading frequency, duration and intensity of physical activity was completed, and a physical activity summary index (PA index) was calculated from a previously published formula.25 In the statistical analysis, the PA index was used as a continuous variable. For descriptive purposes, patients were categorised into two physical activity categories, depending on whether they fulfilled the American College of Sports Medicine and American Heart Association’s recommendations for physical activity or not (table 1).30 The Disease Activity Score 28 (DAS28),31 Simplified Disease Activity Index (SDAI) and Clinical Disease Activity Index (CDAI)32 were also calculated.
Table 1.
Patient characteristics
| Patient characteristics | Total n=93 | Women n=68 | Men n=25 |
| Age median, (IQR) | 60 (52–66) | 60 (51–67) | 60 (52–66) |
| Height (m), mean (SD) | 1.69 (0.90) | 1.66 (0.63) | 1.80 (0.71) |
| Weight (kg), mean (SD) | 76.4 (12.3) | 72.7 (10.9) | 86.8 (9.7) |
| Body mass index (kg/m2), mean (SD) | 26.7 (3.9) | 26.6 (4.1) | 26.9 (3.4) |
| Comorbidity, n (%) | 38 (41) | 30 (44) | 8 (32) |
| Cardiovascular (HT, angina, MI) | 21 (23) | 17 (25) | 4 (16) |
| Respiratory (COPD and/or CRPD) | 18 (19) | 15 (22) | 3 (12) |
| Diabetes | 4 (4) | 3 (4) | 1 (4) |
| Cancer (previous or present) | 5 (5) | 3 (4) | 2 (8) |
| Smoking, n (%)* | |||
| Never smoked | 35 (38) | 27 (40) | 8 (32) |
| Previous smoker | 51 (55) | 37 (54) | 14 (56) |
| Present smoker | 7 (8) | 4 (6) | 3 (12) |
| Resting heart rate (beats per min), mean (SD) | 66 (10) | 67 (9) | 65 (11) |
| Physical activity categories, n (%) | |||
| Does not fulfil ACSM/AHA recommendations | 64 (69) | 44 (66) | 19 (76) |
| Fulfils ACSM/AHA recommendations | 29 (31) | 23 (34) | 6 (24) |
| Seropositivity (ACPA and/or RF), n (%) | 75 (81) | 54 (79) | 21 (84) |
| Disease duration (years), median (IQR) | 10 (5–19) | 10 (5–20) | 11 (6–16) |
| Patient global assessment (0–100 mm), median (IQR) | 24 (10–36) | 27 (16–42) | 12 (5–24) |
| Physician global assessment (0–100 mm), median (IQR) | 10 (0–12) | 8 (0–18) | 5 (0–10) |
| mHAQ, mean (SD) | 0.26 (0.31) | 0.29 (0.33) | 0.17 (0.23) |
| hsCRP, median (IQR) | 1.75 (0.75–3.13) | 1.64 (0.71–3.13) | 2.39 (0.98–3.20) |
| SDAI, n (%) | |||
| Remission | 22 (24) | 12 (18) | 10 (40) |
| Low disease activity | 41 (44) | 32 (47) | 9 (36) |
| Moderate disease activity | 24 (25) | 21 (31) | 3 (12) |
| High disease activity | 6 (7) | 3 (4) | 3 (12) |
| Mean (SD) | 10.2 (8.7) | 10.6 (8.0) | 9.3 (10.3) |
| DAS28 (hsCRP), n (%) | |||
| Remission | 39 (42) | 25 (37) | 14 (56) |
| Low disease activity | 23 (25) | 18 (27) | 5 (20) |
| Moderate disease activity | 28 (30) | 23 (34) | 5 (20) |
| High disease activity | 3 (3) | 2 (3) | 1 (4) |
| Mean (SD) | 2.56 (1.04) | 2.67 (1.01) | 2.27 (1.07) |
| ACR/EULAR remission, n (%) | 25 (27) | 13 (19) | 12 (48) |
| Medication, n (%) | |||
| bDMARDs (present) | 54 (58) | 41 (60) | 13 (52) |
| cDMARDs (present) | 74 (80) | 54 (79) | 20 (80) |
| Corticosteroids (any form during last year) | 39 (42) | 29 (43) | 10 (40) |
*Total sum is 101% due to rounding.
ACPA, anti-citrullinated protein antibody;ACR, American College of Rheumatology;ACSM, American College of Sports Medicine;AHA, American Heart Association;bDMARDs, biological disease-modifying anti-rheumatic drugs;cDMARDs, conventional disease-modifying anti-rheumatic drugs;COPD, chronic obstructive pulmonary disease;CRPD, chronic restrictive pulmonary disease;DAS28, disease activity score index;EULAR, European League Against Rheumatism;hsCRP, high-sensitivity C reactive protein;HT, hypertension;mHAQ, modified Health Assessment Questionnaire;MI, myocardial infarction;PA index, physical activity summary index;RF, rheumatoid factor;SDAI, Simple Disease Activity Index.
We compared our results with VO2peak measurements from the general population using published data from the HUNT3 Fitness study, which was part of the third survey of the Nord-Trøndelag Health Study (HUNT3, ntnu.edu/hunt).29 In HUNT3 (2006–2008), the entire population >20 years old in the northern region of Trøndelag (previously, Nord-Trøndelag county), Norway, were invited, with a participation rate of 54%. In the HUNT3 Fitness sub-study 4631 participants of both genders free from CVD, respiratory symptoms, cancer and the use of anti-hypertensives also completed VO2peak tests.1 33
Statistical analysis
Data are given as counts and percentages, mean with SD or median with IQR in parenthesis.
Mean VO2peak in 10 years age and gender categories in our study were compared with the corresponding mean and 95% CI in the HUNT3 Fitness population. Due to small numbers in our study, age groups 20–29 years and 30–39 years were combined, and compared with the HUNT3 Fitness age group 30–39 years. The results were similar if the RA patients from 20 to 29 years were omitted from the comparison.
To evaluate variables associated with VO2peak in RA patients, multivariable linear regression analyses with VO2peak (mL/(kg x min)) as the dependent variable were performed. The following explanatory variables were included based on previous literature: age, gender and the age×gender interaction, body mass index (BMI, calculated as weight in kg/height in m2), smoking (present or previous vs never smoker), RHR and the PA index. Other potential explanatory variables included the systolic blood pressure (SBP), comorbidity (cancer, CVD [hypertension/angina/myocardial infarction], diabetes, COPD/CRPD) coded as a single yes/no variable. The following RA-specific variables were then considered: PGA and the physician global assessment, mHAQ, DAS28, SDAI, CDAI, remission criteria (DAS28 or ACR/EULAR),34–36 time since diagnosis, seropositivity and medication (present use of biological DMARDs and conventional DMARDs, and corticosteroids used during the last year). In order not to overfit the linear regression model, variable selection for these variables was first performed using Lasso (least absolute shrinkage and selection operator) regression using n=1000 repetitions. Lasso regression identifies the smallest useful set of variables among variables that may be highly correlated, setting the coefficients of irrelevant variables to 0. Only variables with a coefficient different from 0 in the Lasso regression were included in the multivariable linear regression model. The full model was then reduced to the final model by removal of non-significant variables. Finally, the reduced model was standardised in order to permit direct comparison of the importance of the included variables by the size of their coefficients, which are all measured on the same scale (ie, SD). P values <0.05 were considered significant and assumptions were evaluated using residual plots.
Results
A 100 RA patients signed up for the project, and 93 patients completed a valid VO2peak test (figure 1). From those receiving an invitation letter by mail, a higher percentage of men (40%) compared with women (34%) replied when recruited from the patient-centred follow-up lists, whereas a higher percentage of women (62%) compared with men (33%) signed up from patients attending regular visits. More women (n=68) than men (n=25) entered.
Patient characteristics are given in table 1.
Approximately 2/3 of the women and 3/4 of the men did not fulfil the current recommendations for physical activity. Higher proportions of men were in DAS28 and ACR/EULAR remission, and men had lower disease activity compared with women. At the test peak, only 10% of participants reported complaints above 13=‘Somewhat painful’ due to RA in the lower extremities, whereas 75% of participants reported the test being 17=‘Very Hard’ or above (RPE Borg scale).26
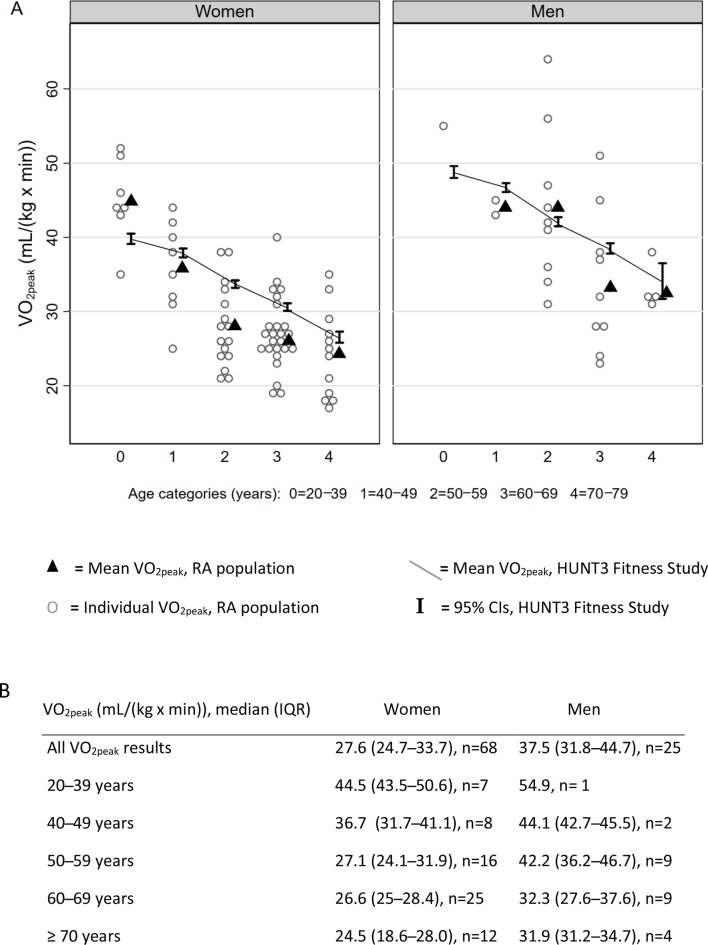
VO2peak was lower in older age groups, and women had lower VO2peak than men (figures 2 and 3A). Compared with the general population, women with RA aged ≥40 years had significantly lower VO2peak as indicated by group means below the 95% CI for the HUNT3 Fitness population, and men with RA in the age groups 40–49 and 60–69 years had a significantly lower VO2peak (figure 2). In most age groups, patients recruited from the patient-centred follow-up list had higher VO2peak compared with the other patients (age group <70 years: p=0.02 for patient-centred follow-up vs other patients in linear regression adjusted for gender).
Figure 2.
VO2peak in RA patients compared with healthy controls. (A) Mean VO2peak in RA population compared with means and 95% CIs of HUNT3 Fitness study. (B) VO2peak results for RA population, median (IQR). RA, rheumatoid arthritis.
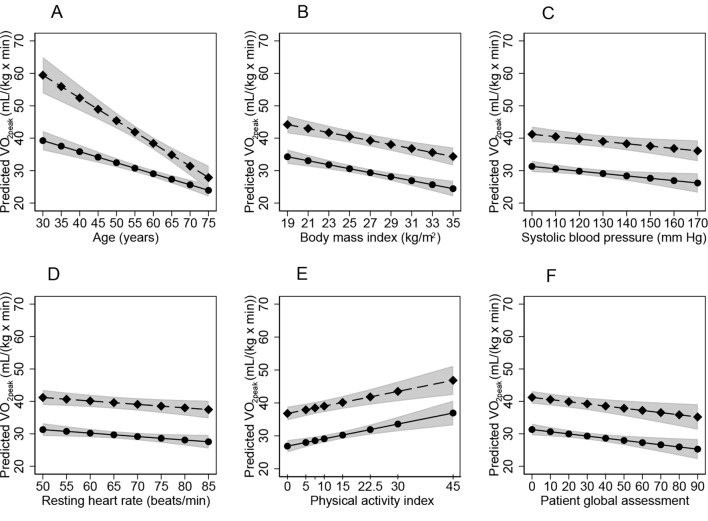
Figure 3.
Associations of VO2peak to significant predictors. Influence of age (A), body mass index (BMI) (B), systolic blood pressure (SBP) (C), resting heart rate (RHR) (D), physical activity index (E) and patient global assessment (PGA) (F) on VO2peak, based on the multivariable model including age, gender, BMI, RHR, smoking, SBP, physical activity index and PGA. Circles: women; diamonds: men; grey areas: 95% CIs.
In the Lasso regression with VO2peak as the dependent variable, five explanatory variables were significant: comorbidity (yes/no), disease duration, SBP, ACR/EULAR remission (yes/no) and the PGA. These variables were included in the full regression model in addition to explanatory variables included based on previous literature; that is, age, gender, the age×gender interaction, BMI, smoking, RHR and the PA index. After stepwise removal of non-significant variables (disease duration [p=0.90], comorbidity [p=0.33] and ACR/EULAR remission [p=0.25]), and after adjusting for age (p<0.001), gender (p<0.001) and the age×gender interaction term (p=0.001), the final reduced model explained 82% of the variation in VO2peak. The final predictors in the multiple linear regression model and their standardised coefficients are shown in table 2. The influence of each variable on VO2peak is shown in figure 3.
Table 2.
Variables associated with VO2peak in the standardised regression model*
| Variable | P value | Standardised coefficient |
| Body mass index | <0.001 | −0.25 |
| Physical activity summary index | <0.001 | 0.21 |
| Patient global assessment | 0.006 | −0.14 |
| Systolic blood pressure | 0.016 | −0.12 |
| Resting heart rate | 0.032 | −0.11 |
| Smoking | 0.046 | −0.10 |
*The model was adjusted for age and gender. The standardised coefficient gives the change of VO2peak (mL/(kg x min)) for one SD increase in each variable.
Discussion
The main finding in this study was that in most age groups, patients with RA had significantly lower CRF measured as VO2peak compared with healthy controls of similar age and gender. The difference in CRF level between RA patients and the general population was more pronounced in women. CRF was associated with common risk factors for CVD, with BMI having the strongest association, followed by physical activity measured by the PA index, and the patients’ own impression of RA disease activity measured as the PGA. The only RA-specific variable associated with CRF was the PGA.
Predictors of VO2peak
The associations between CRF and common risk factors for cardiovascular health in RA patients (high BMI, high RHR, high SBP and previous/present smoking) are supported by other studies.18 21 CRF is a predictor of cardiovascular health, and improvement of CRF strengthens cardiovascular health in the general population6 and improves risk factors for CVD in RA patients.21 Thus, improvement of CRF could probably contribute to better cardiovascular health in RA patients.1–8 18
The degree of physical activity, measured as the PA index, contributed strongly to the prediction of CRF. Increased physical activity might also reduce BMI. These two factors showed the highest relative importance for the CRF, supporting the view that physical activity should be assessed and acted on in RA patients. Information on the degree of physical activity might reveal possibilities and barriers for improving CRF, which is associated with cardiovascular health.
Contradictory to our hypothesis, CRF was not associated with objective RA-specific variables. The PGA score is strongly subjective, and completely determined by the patient’s own impression of disease impact. In addition to disease activity, pain and functional incapacity, the PGA is driven by factors like fatigue, psychological distress and the coexistence of fibromyalgia.37 Such factors may represent aspects of RA that are difficult for physicians to capture, and might partly explain the discrepancy between the patient and physician global assessment (table 1). Physicians tend to examine joints and evaluate markers of inflammation before deciding on the global assessment, and joint counts and CPR concentrations are not strong drivers of the PGA.37 Perhaps disease activity is not what stops RA patients from being physically active, but rather symptoms like fatigue, psychological distress and coexistence of fibromyalgia that influence motivation for physical activity. The association between the PGA with CRF merits further investigation.
In the HUNT Fitness Study, patients were excluded if they had comorbidities and the participants therefore represent a selected, healthy population. In the present study, no such exclusions were made. The results indicate that the reason for lower fitness in the RA patients was not the coexistence of CVD, diabetes, cancer or pulmonary disease, but was rather related to other differences from the healthy population. However, the study was not designed to investigate the mechanisms behind the observed fitness level. Further, a larger study would enable investigating the influence of separate comorbidities, avoiding possible biases from using a combined and dichotomised comorbidity variable.
Patient representability
Disease activity in the present RA population was comparable with disease activity in other Norwegian RA patients,38 but the mHAQ was slightly lower. The discrepancy in mHAQ may be due to biassed selection of participants with fewer physical restraints to the VO2peak test. Due to few included women <40 years and men in all age groups, the results for these groups should be evaluated cautiously. A higher proportion of men from the patient-centred follow-up programme agreed to participate, to a large extent representing patients with more stable disease than those with regular clinical appointments. This selection bias may explain why the findings of lower fitness were clearer for women than men when comparing to the healthy population and limits the generalisability of the results for men. We cannot exclude that a bigger study with more patients with moderate or high disease activity would have resulted in different findings regarding the importance of disease activity. However, the study was well powered to identify the most important variables among a large selection of potentially relevant disease-related variables. Furthermore, reaching an R2 of ~0.80, only a small part of the variance in the data remained unexplained.
It is difficult to evaluate whether the participants were representative of Norwegian RA patients with respect to physical activity, because the PA index of those who declined to attend is unknown. Physical activity was based on self-report and could be both underestimated and overestimated by the participants. The PA index used in our study has not been validated in RA patients. Objective measures like accelerometry would have been useful, but were not available in our setting. We may speculate that RA patients who are familiar with working out might be more likely to sign up for a study with CRF testing. However, most participants did not fulfil the current recommendations for physical activity.30
Validity of VO2peak test results
In comparison to some studies measuring VO2peak in RA patients, our results are quite high, but many studies are old, and have different inclusion criteria. For instance, some interventional studies excluded participants not leading a sedentary life21 39 or those undertaking more than 30 min aerobic exercise three times a week.40 The baseline of VO2peak results of such studies are therefore not comparable to those of our study where no such exclusions were made. In addition, over the years, there has been a tremendous change in treatment strategies for RA,41 42 and treatment strategies and traditions of physical activity might differ between countries. This further complicates comparisons with VO2peak results of other studies. In one Swedish study from 2014, RA patient’s mean (SD) VO2peak was 31.2 (7.0) in women and 40.0 (8.2) in men,43 which closely resembles the results of our study. The RA population of Sweden is expected to be comparable to that of Norway.
Previous studies have shown benefits of exercise and few safety issues in RA patients.20 21 39 44 45 The present study demonstrated that RA patients are able to complete treadmill tests without premature termination due to disease complaints. At peak, only a small proportion of participants reported RA-associated joint pain in the lower extremities, supporting that RA patients probably terminated the VO2peak test because of cardiorespiratory limitations, rather than RA complaints. Adequate VO2peak test using a cycle ergometer requires cycling experience and may lead to lower measured VO2peak due to local fatigue.46 47 On the other hand, everyone is familiar with walking. The NeXt Move core facility has strict routines for calibration and maintenance of the testing equipment. We therefore consider the VO2peak test results to be reliable.
In summary, RA patients had decreased CRF compared with a healthy population of similar age and gender. Their CRF was associated with common risk factors for CVD, implying that life-style changes may improve CRF, which is associated with improved cardiovascular health. Reduction of BMI and increased physical activity would most strongly improve the CRF. The CRF level adds important information to the evaluation of RA patients, but the VO2peak test is a resource-intensive method. Models for estimation of CRF without a physical test have been developed for healthy people. Until similar tools exist for RA patients, increasing and monitoring physical activity, advice on smoking cessation, measurement of BP, RHR and BMI are practical interventions for CVD prevention. RA-specific variables were not of importance for CRF, except for PGA, indicating that subjective factors have a stronger bearing on CRF than objective measures of disease activity in RA patients. Future studies are warranted that test the use of CRF to identify RA patients at increased risk of CVD, and whether effective prevention strategies including reduction of sedentary behaviour and improvement of fitness may be developed and implemented in this population.
Acknowledgments
We are grateful to the RA patients for their participation. Cardiopulmonary exercise testing was provided by NeXt Move, NTNU—Norwegian University of Science and Technology. Some results from the study were presented at the Annual European Congress of Rheumatology—EULAR (12–15 June 2018, Amsterdam).
Footnotes
Contributors: Study conception and design: MHL, MH, UW, RT, VV. Acquisition and analysis of data: MHL, TF, VV. Interpretation of data: MH, UW, RT, VV. Drafting the manuscript: MHL, TF, VV. Revising the manuscript critically for important intellectual content: MH, TF, UW, RT, VV. All authors approved the final version of the manuscript.
Funding: This project is funded by a grant to Marthe Halsan Liff from The Central Norway Regional Health Authority, allocated via The Liaison Committee for Education, Research and Innovation in Central Norway (2016/29014). NeXt Move is funded by the Faculty of Medicine at NTNU and Central Norway Regional Health Authority.
Competing interests: None declared.
Patient consent for publication: All participants provided written informed consent.
Ethics approval: The Regional Committee for Medical and Health Research Ethics approved the study (2016/275), which was performed in compliance with the Declaration of Helsinki.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data availability statement: No additional data are available.
References
- 1.Aspenes ST, Nilsen TIL, Skaug E-A, et al. Peak oxygen uptake and cardiovascular risk factors in 4631 healthy women and men. Med Sci Sports Exerc 2011;43:1465–73. 10.1249/MSS.0b013e31820ca81c [DOI] [PubMed] [Google Scholar]
- 2.DeFina LF, Haskell WL, Willis BL, et al. Physical activity versus cardiorespiratory fitness: two (partly) distinct components of cardiovascular health? Prog Cardiovasc Dis 2015;57:324–9. 10.1016/j.pcad.2014.09.008 [DOI] [PubMed] [Google Scholar]
- 3.Harber MP, Kaminsky LA, Arena R, et al. Impact of cardiorespiratory fitness on all-cause and disease-specific mortality: advances since 2009. Prog Cardiovasc Dis 2017;60:11–20. 10.1016/j.pcad.2017.03.001 [DOI] [PubMed] [Google Scholar]
- 4.Kodama S, Saito K, Tanaka S, et al. Cardiorespiratory fitness as a quantitative predictor of all-cause mortality and cardiovascular events in healthy men and women: a meta-analysis. JAMA 2009;301:2024–35. 10.1001/jama.2009.681 [DOI] [PubMed] [Google Scholar]
- 5.Myers J, McAuley P, Lavie CJ, et al. Physical activity and cardiorespiratory fitness as major markers of cardiovascular risk: their independent and interwoven importance to health status. Prog Cardiovasc Dis 2015;57:306–14. 10.1016/j.pcad.2014.09.011 [DOI] [PubMed] [Google Scholar]
- 6.Sui X, Sarzynski MA, Lee D-C, et al. Impact of changes in cardiorespiratory fitness on hypertension, dyslipidemia and survival: an overview of the epidemiological evidence. Prog Cardiovasc Dis 2017;60:56–66. 10.1016/j.pcad.2017.02.006 [DOI] [PubMed] [Google Scholar]
- 7.Vanhees L, Fagard R, Thijs L, et al. Prognostic significance of peak exercise capacity in patients with coronary artery disease. J Am Coll Cardiol 1994;23:358–63. 10.1016/0735-1097(94)90420-0 [DOI] [PubMed] [Google Scholar]
- 8.Kavanagh T, Mertens DJ, Hamm LF, et al. Prediction of long-term prognosis in 12 169 men referred for cardiac rehabilitation. Circulation 2002;106:666–71. 10.1161/01.CIR.0000024413.15949.ED [DOI] [PubMed] [Google Scholar]
- 9.Ross R, Blair SN, Arena R, et al. Importance of assessing cardiorespiratory fitness in clinical practice: a case for fitness as a clinical vital sign: a scientific statement from the American Heart Association. Circulation 2016;134:e653–99. 10.1161/CIR.0000000000000461 [DOI] [PubMed] [Google Scholar]
- 10.Dhawan SS, Quyyumi AA. Rheumatoid arthritis and cardiovascular disease. Curr Atheroscler Rep 2008;10:128–33. 10.1007/s11883-008-0019-x [DOI] [PubMed] [Google Scholar]
- 11.Widdifield J, Paterson JM, Huang A, et al. Causes of death in rheumatoid arthritis: how do they compare to the general population? Arthritis Care Res 2018;70:1748–55. 10.1002/acr.23548 [DOI] [PubMed] [Google Scholar]
- 12.Avina-Zubieta JA, Thomas J, Sadatsafavi M, et al. Risk of incident cardiovascular events in patients with rheumatoid arthritis: a meta-analysis of observational studies. Ann Rheum Dis 2012;71:1524–9. 10.1136/annrheumdis-2011-200726 [DOI] [PubMed] [Google Scholar]
- 13.Humphreys JH, Warner A, Chipping J, et al. Mortality trends in patients with early rheumatoid arthritis over 20 years: results from the Norfolk Arthritis Register. Arthritis Care Res 2014;66:1296–301. 10.1002/acr.22296 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sokka T, Abelson B, Pincus T. Mortality in rheumatoid arthritis: 2008 update. Clin Exp Rheumatol 2008;26(5 Suppl 51):S35–61. [PubMed] [Google Scholar]
- 15.van den Hoek J, Boshuizen HC, Roorda LD, et al. Mortality in patients with rheumatoid arthritis: a 15-year prospective cohort study. Rheumatol Int 2017;37:487–93. 10.1007/s00296-016-3638-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Law R-J, Markland DA, Jones JG, et al. Perceptions of issues relating to exercise and joint health in rheumatoid arthritis: a UK-based questionnaire study. Musculoskeletal Care 2013;11:147–58. 10.1002/msc.1037 [DOI] [PubMed] [Google Scholar]
- 17.Sokka T, Häkkinen A, Kautiainen H, et al. Physical inactivity in patients with rheumatoid arthritis: data from twenty-one countries in a cross-sectional, International study. Arthritis Rheum 2008;59:42–50. 10.1002/art.23255 [DOI] [PubMed] [Google Scholar]
- 18.Metsios GS, Koutedakis Y, Veldhuijzen van Zanten JJCS, et al. Cardiorespiratory fitness levels and their association with cardiovascular profile in patients with rheumatoid arthritis: a cross-sectional study. Rheumatology 2015;54:kev035–20. 10.1093/rheumatology/kev035 [DOI] [PubMed] [Google Scholar]
- 19.Metsios GS, Stavropoulos-Kalinoglou A, Veldhuijzen van Zanten JJCS, et al. Rheumatoid arthritis, cardiovascular disease and physical exercise: a systematic review. Rheumatology 2008;47:239–48. 10.1093/rheumatology/kem260 [DOI] [PubMed] [Google Scholar]
- 20.Sandstad J, Stensvold D, Hoff M, et al. The effects of high intensity interval training in women with rheumatic disease: a pilot study. Eur J Appl Physiol 2015;115:2081–9. 10.1007/s00421-015-3186-9 [DOI] [PubMed] [Google Scholar]
- 21.Stavropoulos-Kalinoglou A, Metsios GS, Veldhuijzen van Zanten JJCS, et al. Individualised aerobic and resistance exercise training improves cardiorespiratory fitness and reduces cardiovascular risk in patients with rheumatoid arthritis. Ann Rheum Dis 2013;72:1819–25. 10.1136/annrheumdis-2012-202075 [DOI] [PubMed] [Google Scholar]
- 22.Arnett FC, Edworthy SM, Bloch DA, et al. The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum 1988;31:315–24. 10.1002/art.1780310302 [DOI] [PubMed] [Google Scholar]
- 23.Aletaha D, Neogi T, Silman AJ, et al. 2010 rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Ann Rheum Dis 2010;69:1580–8. 10.1136/ard.2010.138461 [DOI] [PubMed] [Google Scholar]
- 24.Kourilovitch M, Galarza-Maldonado C, Ortiz-Prado E. Diagnosis and classification of rheumatoid arthritis. J Autoimmun 2014;48-49:26–30. 10.1016/j.jaut.2014.01.027 [DOI] [PubMed] [Google Scholar]
- 25.Nes BM, Janszky I, Vatten LJ, et al. Estimating V·O 2peak from a nonexercise prediction model: the HUNT study, Norway. Med Sci Sports Exerc 2011;43:2024–30. 10.1249/MSS.0b013e31821d3f6f [DOI] [PubMed] [Google Scholar]
- 26.Borg GAV, Noble BJ, exertion P. Perceived exertion. Exerc Sport Sci Rev 1974;2:131–53. 10.1249/00003677-197400020-00006 [DOI] [PubMed] [Google Scholar]
- 27.Felson DT, Anderson JJ, Boers M, et al. The American College of Rheumatology preliminary core set of disease activity measures for rheumatoid arthritis clinical trials. The Committee on Outcome Measures in Rheumatoid Arthritis Clinical Trials. Arthritis Rheum 1993;36:729–40. [DOI] [PubMed] [Google Scholar]
- 28.Pincus T, Summey JA, Soraci SA, et al. Assessment of patient satisfaction in activities of daily living using a modified Stanford Health Assessment Questionnaire. Arthritis Rheum 1983;26:1346–53. 10.1002/art.1780261107 [DOI] [PubMed] [Google Scholar]
- 29.Krokstad S, Langhammer A, Hveem K, et al. Cohort profile: the HUNT study, Norway. Int J Epidemiol 2013;42:968–77. 10.1093/ije/dys095 [DOI] [PubMed] [Google Scholar]
- 30.Haskell WL, Lee I-M, Pate RR, et al. Physical activity and public health: updated recommendation for adults from the American College of Sports Medicine and the American Heart Association. Circulation 2007;116:1081–93. 10.1161/CIRCULATIONAHA.107.185649 [DOI] [PubMed] [Google Scholar]
- 31.Prevoo ML, van 't Hof MA, Kuper HH, et al. Modified disease activity scores that include twenty-eight-joint counts. Development and validation in a prospective longitudinal study of patients with rheumatoid arthritis. Arthritis Rheum 1995;38:44–8. 10.1002/art.1780380107 [DOI] [PubMed] [Google Scholar]
- 32.Aletaha D, Smolen JS. The simplified disease activity index (SDAI) and clinical disease activity index (CDAI) to monitor patients in standard clinical care. Best Pract Res Clin Rheumatol 2007;21:663–75. 10.1016/j.berh.2007.02.004 [DOI] [PubMed] [Google Scholar]
- 33.Loe H, Steinshamn S, Wisløff U. Cardio-respiratory reference data in 4631 healthy men and women 20-90 years: the HUNT 3 fitness study. PLoS One 2014;9:e113884 10.1371/journal.pone.0113884 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bykerk VP, Massarotti EM. The new ACR/EULAR remission criteria: rationale for developing new criteria for remission. Rheumatology 2012;51 Suppl 6(suppl 6):vi16–20. 10.1093/rheumatology/kes281 [DOI] [PubMed] [Google Scholar]
- 35.Felson DT, Smolen JS, Wells G, et al. American College of Rheumatology/European League Against Rheumatism provisional definition of remission in rheumatoid arthritis for clinical trials. Ann Rheum Dis 2011;70:404–13. 10.1136/ard.2011.149765 [DOI] [PubMed] [Google Scholar]
- 36.Fleischmann RM, van der Heijde D, Gardiner PV, et al. DAS28-CRP and DAS28-ESR cut-offs for high disease activity in rheumatoid arthritis are not interchangeable. RMD Open 2017;3:e000382 10.1136/rmdopen-2016-000382 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Nikiphorou E, Radner H, Chatzidionysiou K, et al. Patient global assessment in measuring disease activity in rheumatoid arthritis: a review of the literature. Arthritis Res Ther 2016;18:151 10.1186/s13075-016-1151-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Haugeberg G, Hansen IJW, Soldal DM, et al. Ten years of change in clinical disease status and treatment in rheumatoid arthritis: results based on standardized monitoring of patients in an ordinary outpatient clinic in southern Norway. Arthritis Res Ther 2015;17:219 10.1186/s13075-015-0716-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Bartlett DB, Willis LH, Slentz CA, et al. Ten weeks of high-intensity interval walk training is associated with reduced disease activity and improved innate immune function in older adults with rheumatoid arthritis: a pilot study. Arthritis Res Ther 2018;20:127 10.1186/s13075-018-1624-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Neuberger GB, Aaronson LS, Gajewski B, et al. Predictors of exercise and effects of exercise on symptoms, function, aerobic fitness, and disease outcomes of rheumatoid arthritis. Arthritis Rheum 2007;57:943–52. 10.1002/art.22903 [DOI] [PubMed] [Google Scholar]
- 41.Combe B, Landewe R, Daien CI, et al. 2016 update of the EULAR recommendations for the management of early arthritis. Ann Rheum Dis 2017;76:948–59. 10.1136/annrheumdis-2016-210602 [DOI] [PubMed] [Google Scholar]
- 42.Smolen JS, Landewé R, Bijlsma J, et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2016 update. Ann Rheum Dis 2017;76:960–77. 10.1136/annrheumdis-2016-210715 [DOI] [PubMed] [Google Scholar]
- 43.Nordgren B, Fridén C, Jansson E, et al. Criterion validation of two submaximal aerobic fitness tests, the self-monitoring Fox-walk test and the Åstrand cycle test in people with rheumatoid arthritis. BMC Musculoskelet Disord 2014;15:305 10.1186/1471-2474-15-305 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.de Jong Z, Munneke M, Zwinderman AH, et al. Long term high intensity exercise and damage of small joints in rheumatoid arthritis. Ann Rheum Dis 2004;63:1399–405. 10.1136/ard.2003.015826 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Rausch Osthoff A-K, Niedermann K, Braun J, et al. 2018 EULAR recommendations for physical activity in people with inflammatory arthritis and osteoarthritis. Ann Rheum Dis 2018;77:1251–60. 10.1136/annrheumdis-2018-213585 [DOI] [PubMed] [Google Scholar]
- 46.Carter H, Jones AM, Barstow TJ, et al. Oxygen uptake kinetics in treadmill running and cycle ergometry: a comparison. J Appl Physiol 2000;89:899–907. 10.1152/jappl.2000.89.3.899 [DOI] [PubMed] [Google Scholar]
- 47.McKay GA, Banister EW. A comparison of maximum oxygen uptake determination by bicycle ergometry at various pedaling frequencies and by treadmill running at various speeds. Eur J Appl Physiol Occup Physiol 1976;35:191–200. 10.1007/BF02336193 [DOI] [PubMed] [Google Scholar]