Abstract
Objective:
M235T polymorphism of the angiotensinogen (AGT) gene has been linked with cardiovascular disease (CVD). The aim of this meta-analysis was to investigate whether combined evidence supports this association.
Methods:
A systematic search was conducted for studies published up to October 2018 that evaluate the association between AGT M235T polymorphism and risk of CVD. Case–control studies were identified, and the association between AGT M235T polymorphism and CVD risk was assessed using genetic models.
Results:
Thirty-nine comparisons from 38 studies were collected, and a meta-analysis and subgroup analysis was performed based on ethnicity. In the overall population (9225 cases and 8406 controls), the occurrence of CVD was found to be associated with AGT M235T polymorphism in both allelic [T vs. M: odds ratio (OR)=1.16] and recessive (TT vs. MT+MM: OR=1.14) models. In subgroup analyses, a significant association was identified between AGT M235T polymorphism and CVD risk in East Asian subgroups in allelic (T vs. M: OR=1.46), homozygous (TT vs. MM: OR=1.78), dominant (MT+TT vs. MM: OR=1.47), and recessive (TT vs. MT+ MM: OR=1.68) models, but there was no significant association in Caucasian populations.
Conclusion:
Among East Asians, the AGT variant M235T is associated with CVD risk. However, current evidence suggests that there is no such association in the Caucasian population.
Keywords: angiotensinogen, genetic polymorphism, cardiovascular disease
Introduction
Cardiovascular disease (CVD) is the main cause of death and leads to over 30% of mortality annually worldwide (1). The general risk factors for CVD include smoking, high body mass index, hypertension, lipid metabolism disorders, and diabetes mellitus, among several other factors (2). Emerging evidence has demonstrated that genetic and environmental factors and polymorphisms also play a crucial role in the occurrence and development of CVD (3, 4). The advancement in single-nucleotide polymorphism (SNP) and genome-wide sequencing technologies has led to an increased number of in-depth studies on the genetics of CVD, and a number of candidate genes have been identified, such as those involved in the regulation of lipid metabolism (5), inflammatory cytokines (6), and the renin–angiotensin–aldosterone system (RAAS) (7).
The RAAS plays a critical role in the pathogenesis of coronary heart disease, and previous studies have determined that it is involved in the progression of hypertension and vascular and left ventricular remodeling (8). Much accumulated evidence has indicated that the RAAS is significantly associated with the initiation and progression of coronary atherosclerosis and thrombogenesis (9). In addition, studies involving angiotensin-converting enzyme (ACE) inhibition and angiotensin II receptor blockade have highlighted the vital role of the RAAS, and gene polymorphism of the RAAS may also affect the efficacy of drug (10). Recently, several genetic variants in the RAAS have been found to be significantly associated with CVD risk, such as an insertion/deletion polymorphism in the ACE gene, and T175M and M235T polymorphisms in the angiotensinogen (AGT) gene (11-13). AGT is a crucial determinant of angiotensin II levels, which is an important component of the RAAS. Furthermore, polymorphism in the AGT gene may contribute to atherogenesis in the coronary artery and may be related to the development of CVD (14, 15). The M235T polymorphism has been most widely studied; however, several inconsistent results regarding this polymorphism and CVD risk have been reported. Raygan et al. (16), Bonfim-Silva et al. (17), and Isordia-Salas et al. (18) detected positive correlations, whereas Renner et al. (19), Ranjith et al. (20), and Erbas et al. (21) determined that the AGT M235T polymorphism has no significant effect on the development of CVD. Meta-analyses have been performed to resolve these discrepancies; however, these analyses have been compromised by deficiencies in the sample size, and the results have been either inconclusive or only weakly significant (16). Some of the studies have been limited to Asian populations (22, 23), and several of the most recent studies have not been considered. The aim of the present study was to compile case–control research and updated meta-analyses to explore the association between AGT M235T polymorphism and susceptibility for CVD in a range of populations for more accurate assessment.
Methods
Search strategy
A systematic search of MEDLINE, Embase, China National Knowledge Infrastructure, OVID, ScienceDirect, and WanFang databases was performed to identify epidemiological studies on M235T polymorphisms of the AGT gene and CVD that were published up to October 2018. In the literature searches, various combinations of the keywords “angiotensinogen gene,” “AGT,” “M235T gene,” “genetic polymorphism,” “variants,” or “variations,” “coronary heart disease,” “coronary artery disease,” “cardiovascular disease,” “myocardial infarction,” “ischemic heart disease,” and “coronary stenosis” were used. Only studies published in English or Chinese were included in the study. The references of all full text papers were examined to identify additional relevant studies. Secondary searches of gray literature were not performed. All retrieved articles were organized using reference manager software (Endnote 6).
Inclusion and exclusion criteria
Inclusion criteria were the following: (1) the study evaluated AGT M235T and CVD risk, (2) original research (case–control studies) or AGT M235T genotype frequencies were provided by case–control status, (3) the study had sufficient data to allow the association between AGT M235T and CVD risk, (4) the study included original data, independent of other studies, and (5) the language of the report was in English or Chinese. Exclusion criteria were the following: (1) overlapping data, (2) missing information (particularly genotype distributions and studies without controls), after having not received the requested information from the corresponding author, and (3) genome scans investigating linkages without detailed genotype frequencies between cases and controls. Two reviewers independently screened the titles and abstracts for eligibility criteria. Thereafter, the reviewers read the full text of the studies that potentially met the inclusion criteria, and the literature was reviewed to determine the final inclusion of data. For each study, the following information was recorded: first author, year of publication, geographical area, ethnicity, number of cases and controls, genotypes for cases and controls, and evidence of Hardy–Weinberg equilibrium in the controls. If the two reviewers disagreed regarding the inclusions of a study, a consensus was reached through additional review and discussion.
Data extraction
The two reviewers extracted data from each study independently, and any discrepancies were resolved. The information extracted from each article in Tables 1 and 2, including first author, year of publication, country of origin, ethnicity of patients, numbers of cases and controls, AGT genotypes, allele distribution of cases and controls, and outcome, was summarized.
Table 1.
Characteristics and genotype frequencies for AGT M235T polymorphism in the included studies
| Study | Year | ID | Country | Enthic | Sample size Case/Control |
Genotypes and allele distribution | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cases | Controls | |||||||||||||||
| MM | MT | TT | M | T | MM | MT | TT | M | T | |||||||
| Tiret et al. | 1995 | 1 | France | Caucasian | 630 | 741 | 229 | 301 | 100 | 759 | 501 | 258 | 372 | 111 | 888 | 594 |
| Kamitani et al. | 1995 | 2 | Japan | East Asian | 103 | 103 | 6 | 31 | 66 | 43 | 163 | 10 | 41 | 52 | 61 | 145 |
| Ko et al. | 1997 | 3 | China | East Asian | 150 | 338 | 4 | 22 | 124 | 30 | 270 | 4 | 54 | 279 | 62 | 612 |
| Chen et al. | 1998 | 4 | China | East Asian | 57 | 76 | 4 | 13 | 40 | 21 | 93 | 13 | 31 | 32 | 57 | 95 |
| Sheu et al. | 1998 | 5 | China | East Asian | 102 | 145 | 1 | 26 | 75 | 28 | 176 | 1 | 37 | 107 | 39 | 251 |
| Pastinen et al. | 1998 | 6 | Finland | Caucasian | 122 | 122 | 48 | 66 | 37 | 162 | 140 | 53 | 64 | 34 | 170 | 132 |
| Frossard et al. | 1998 | 7 | UAE | Caucasian | 40 | 61 | 14 | 18 | 8 | 46 | 34 | 16 | 26 | 19 | 58 | 64 |
| Gardemann et al. | 1999 | 8 | Germany | Caucasian | 1058 | 511 | 319 | 582 | 157 | 1220 | 896 | 385 | 585 | 222 | 1355 | 1029 |
| Winkelmann et al. | 1999 | 9 | Germany | Caucasian | 122 | 92 | 38 | 54 | 30 | 130 | 114 | 28 | 53 | 11 | 109 | 75 |
| Batalla et al. | 2000 | 10 | Spain | Caucasian | 220 | 200 | 69 | 99 | 52 | 237 | 203 | 64 | 96 | 40 | 224 | 176 |
| Fomicheva et al. | 2000 | 11 | Russia | Caucasian | 198 | 152 | 63 | 85 | 50 | 211 | 185 | 43 | 75 | 34 | 161 | 143 |
| Olivieri et al. | 2001 | 12 | Italy | Caucasian | 247 | 245 | 63 | 124 | 60 | 250 | 244 | 54 | 76 | 27 | 184 | 130 |
| Xie et al. | 2001 | 13 | China | East Asian | 106 | 86 | 8 | 29 | 69 | 45 | 167 | 11 | 30 | 45 | 52 | 120 |
| Fernández-Arcás et al. | 2001 | 14 | Spain | Caucasian | 212 | 180 | 59 | 121 | 32 | 239 | 185 | 34 | 97 | 49 | 165 | 195 |
| Ermis et al. | 2002 | 15 | Turkey | Miscellaneous | 102 | 114 | 32 | 48 | 22 | 112 | 92 | 39 | 59 | 16 | 137 | 91 |
| Hooper et al. | 2002 | 16 | USA | Miscellaneous | 110 | 185 | 4 | 29 | 67 | 37 | 163 | 2 | 31 | 67 | 35 | 165 |
| Zhu et al. | 2002 | 17 | China | East Asian | 41 | 116 | 2 | 7 | 32 | 11 | 71 | 18 | 47 | 51 | 83 | 149 |
| Zhu et al. | 2002 | 18 | China | East Asian | 118 | 106 | 14 | 48 | 56 | 76 | 160 | 10 | 42 | 54 | 62 | 150 |
| Bis et al. | 2003 | 19 | USA | Caucasian | 208 | 717 | 71 | 98 | 39 | 240 | 176 | 215 | 349 | 153 | 779 | 655 |
| Gu et al. | 2003 | 20 | China | East Asian | 129 | 90 | 12 | 31 | 86 | 55 | 203 | 7 | 30 | 53 | 44 | 136 |
| Ranjith et al. | 2004 | 21 | India | Miscellaneous | 195 | 300 | 24 | 80 | 91 | 128 | 262 | 29 | 127 | 144 | 185 | 415 |
| Zhu et al. | 2004 | 22 | China | East Asian | 192 | 98 | 12 | 75 | 105 | 99 | 285 | 8 | 36 | 54 | 52 | 144 |
| Li et al. | 2004 | 23 | China | East Asian | 120 | 80 | 11 | 60 | 49 | 82 | 158 | 14 | 41 | 25 | 69 | 91 |
| Tobin et al. | 2004 | 24 | England | Caucasian | 547 | 505 | 212 | 252 | 83 | 676 | 418 | 197 | 226 | 82 | 620 | 390 |
| Ren et al. | 2005 | 25 | China | East Asian | 100 | 70 | 2 | 10 | 35 | 14 | 80 | 13 | 26 | 31 | 52 | 88 |
| Araujo et al. | 2005 | 26 | Brazil | Caucasian | 110 | 104 | 46 | 52 | 12 | 144 | 76 | 43 | 51 | 10 | 137 | 71 |
| Renner et al. | 2005 | 27 | Austria | Caucasian | 1370 | 733 | NA | NA | NA | 1537 | 1203 | NA | NA | NA | 832 | 634 |
| Liang et al. | 2006 | 28 | China | East Asian | 133 | 154 | 2 | 30 | 101 | 34 | 232 | 10 | 60 | 84 | 80 | 228 |
| Tsai et al. | 2007 | 29 | China | East Asian | 735 | 519 | 15 | 195 | 525 | 225 | 1245 | 5 | 111 | 403 | 121 | 917 |
| Niu et al. | 2008 | 30 | China | East Asian | 105 | 110 | 8 | 32 | 65 | 48 | 162 | 9 | 47 | 54 | 65 | 155 |
| Zhu et al. | 2010 | 31 | China | East Asian | 151 | 127 | 9 | 27 | 115 | 45 | 257 | 20 | 51 | 56 | 91 | 163 |
| Peng et al. | 2011 | 32 | China | East Asian | 196 | 200 | 14 | 54 | 128 | 82 | 155 | 18 | 86 | 96 | 122 | 278 |
| Konopka et al. | 2011 | 33 | Poland | Caucasian | 100 | 95 | 30 | 46 | 24 | 106 | 94 | 22 | 44 | 29 | 88 | 102 |
| Mehri et al. | 2011 | 34 | Tunisia | Miscellaneous | 123 | 144 | 29 | 53 | 41 | 111 | 135 | 53 | 61 | 30 | 167 | 121 |
| Raygan et al. | 2016 | 35 | Iran | Miscellaneous | 155 | 185 | 42 | 79 | 34 | 163 | 147 | 71 | 85 | 29 | 227 | 143 |
| Bonfim-Silva et al. | 2016 | 36 | Brazil | Miscellaneous | 153 | 113 | 23 | 69 | 61 | 115 | 191 | 13 | 63 | 37 | 89 | 137 |
| Caucasian | 306 | 142 | 73 | 145 | 88 | 291 | 321 | 34 | 68 | 40 | 136 | 148 | ||||
| Erbas et al. | 2017 | 37 | Turkey | Miscellaneous | 117 | 106 | 11 | 104 | 2 | 126 | 108 | 16 | 85 | 5 | 117 | 95 |
| Isordia-Salas et al. | 2018 | 38 | Mexico | Miscellaneous | 242 | 242 | 138 | 98 | 6 | 374 | 110 | 170 | 62 | 10 | 402 | 82 |
NA - not available
Table 2.
Baseline characteristics of the included studies
| Study | Enthic | Outcome | Genotyping-methods | Age | Gender (M/F) | HWE | ||
|---|---|---|---|---|---|---|---|---|
| Case | Control | Case | Control | |||||
| Tiret et al. | C | MI | PCR | 53.0±0.3 | 54.0±0.3 | 630/0 | 741/0 | Y |
| Kamitani et al. | EA | MI | PCR | 52±1 | 54±1 | 103/0 | 103/0 | Y |
| Ko et al. | EA | MI | PCR | 61.5±0.6 | 56.0±0.6 | NR | 181/157 | Y |
| Chen et al. | EA | MI | PCR | 67.7±8.5 | 65.7±8.2 | 50/7 | 69/7 | Y |
| Sheu et al. | EA | CAD | PCR | NR | NR | 102/0 | 145/0 | Y |
| Pastinen et al. | C | MI | PCR | 57.7±4.9 | 57.7±4.9 | 122/0 | 122/0 | Y |
| Frossard et al. | C | MI | PCR | 55.0±11.3 | 53.7±14.0 | 25/15 | 31/30 | Y |
| Gardemann et al. | C | MI | PCR | 62.2±9.5 | 58.5±10.6 | 1058/0 | 511/0 | Y |
| Winkelmann et al. | C | MI | PCR | 55.7±9.6 | 55.7±9.6 | 122/0 | 92/0 | Y |
| Batalla et al. | C | MI | PCR | 43±5 | 42±6 | 220/0 | 200/0 | Y |
| Fomicheva et al. | C | MI | PCR | 67 (55-85) | 11 (6-17) | 198/0 | 152/0 | Y |
| Olivieri et al. | C | MI | PCR | 57.7±12.8 | 59.6±9.5 | 160/85 | 221/26 | Y |
| Xie et al. | EA | CAD | PCR | 61.4±9.5 | 52.8±8.7 | 82/24 | 54/32 | Y |
| Fernández-Arcás et al. | C | MI | PCR | 54±13 | 56±15 | 212/0 | 180/0 | Y |
| Ermis et al. | M | MI | PCR | 42.1±11.8 | 40.3±12.8 | NR | NR | Y |
| Hooper et al. | M | MI | PCR | NR | NR | NR | NR | Y |
| Zhu et al. | EA | MI | PCR | 59.6±10.4 | 56.6±10.4 | 27/14 | 67/49 | Y |
| Zhu et al. | EA | CAD | PCR | NR | NR | NR | NR | Y |
| Bis et al. | C | MI | PCR | 63.6 | 64.4 | 128/80 | 371/346 | Y |
| Gu et al. | EA | CAD | PCR | 65.8±9.2 | 65.3±9.8 | 81/48 | 54/36 | Y |
| Ranjith et al. | M | MI | PCR | 18-45 | 18-45 | NR | NR | Y |
| Zhu et al. | EA | CAD | PCR | NR | NR | NR | NR | Y |
| Li et al. | EA | CAD | PCR | 61.5±11.8 | 59.3±10.5 | 80/40 | 47/33 | Y |
| Tobin et al. | C | MI | PCR | 61.9±9.2 | 58.6±10.7 | 372/175 | 313/192 | Y |
| Ren et al. | EA | CAD | PCR | 60.0±9.8 | 57.9±11.6 | 71/29 | 38/32 | Y |
| Arauji et al. | C | MI | PCR | >18 | >18 | 73/37 | 44/60 | Y |
| Renner et al. | C | MI | PCR | 63.1±10.4 | 58.4±12.1 | 1081/289 | 378/355 | NR |
| Liang et al. | EA | CAD | PCR | 64±8 | 63±8 | 100/33 | 116/38 | Y |
| Tsai et al. | EA | CAD | PCR | 63.8±11.4 | 58.6±13.1 | 531/204 | 269/250 | Y |
| Niu et al. | EA | CAD | PCR | 59±7 | 57±9 | 69/36 | 71/39 | Y |
| Zhu et al. | EA | CAD | PCR | 59.7±11.3 | 58.1±10.8 | 96/55 | 71/56 | Y |
| Peng et al. | EA | CAD | PCR | 70.0±8.3 | 69.0±6.4 | 128/68 | 132/68 | Y |
| Konopka et al. | C | MI | PCR | 57±10 | 38±11 | 79/21 | 76/19 | Y |
| Mehri et al. | M | MI | PCR | 62.3±11.8 | 60.4±10.3 | 71/52 | 83/61 | Y |
| Raygan et al. | M | MI | PCR | 62.4±3.2 | 61.7±4.3 | 102/53 | 127/58 | Y |
| Bonfim-Silva et al. | M | CAD | PCR | 55.7±7.9 | 51.8±8.4 | 99/54 | 49/64 | Y |
| C | CAD | PCR | 55.7±6.7 | 53.0±7.7 | 204/102 | 65/77 | Y | |
| Erbas et al. | M | CAD | PCR | 50.2±12.3 | 41.4±11.3 | 55/62 | 14/92 | Y |
| Isordia-Salas et al. | M | MI | PCR | 41.0±5.3 | 39.7±5.0 | 191/51 | 192/50 | Y |
C - Caucasian; EA - East Asian; M - Miscellaneous; MI - myocardial infarction; CAD - coronary artery disease; PCR - polymerase chain reaction; NR - no reported; HWE - Hardy-Weinberg equilibrium; Y - yes
Statistical analysis
Data analysis was conducted using STATA 12.0 software (StataCorp, College Station, TX, USA). The association between AGT M235T polymorphism and CVD susceptibility was assessed in the following genetic models: T versus M (allelic), TT versus MM (co-dominant), MT versus MM (co-dominant), MT+TT versus MM (dominant), and MT+MM versus TT (recessive). Inter-study heterogeneity was tested using Q-statistics. The Mantel–Haenszel method for fixed effects and the DerSimonian and Laird method for random effects were used to estimate pooled effects (24). The fixed effects method was used if the result of the Q test was not significant. Otherwise, the pooled odds ratio (OR) and 95% confidence interval (CI), assuming a random effects model, were calculated. Fixed effects assume that genetic factors have similar effects on CVD susceptibility across all studies, and that the observed variations among studies are caused by chance alone (25). The random effects model assumes that different studies may have substantial diversity and assesses both intra- and inter-study variations (26). A recently developed measure, I2, was used to quantify the inconsistency among the studies’ results for values of ≥50%, with large heterogeneity among values of ≥75% (27). Data are expressed as OR with 95% CI and two-tailed p-values. A p-value <0.05 was considered statistically significant. Assessment of publication bias was conducted both visually by using a funnel plot and statistically via Begg’s funnel plots and Egger’s bias test, which measures the degree of funnel plot asymmetry (28, 29). The Begg’s adjusted rank correlation test was used to assess the correlation between test accuracy estimates and their variances. The deviation of Spearman’s rho values from zero provides an estimate of funnel plot asymmetry, where positive values indicate a trend toward higher levels of test accuracy in studies with smaller sample sizes. The Egger’s bias test detects funnel plot asymmetry by determining whether the intercept deviates significantly from zero in a regression of the standardized effect estimates against their precision. Meta-regression analysis was applied to evaluate the heterogeneity of the studies.
Results
Search results and characteristics of included studies
Initially, 427 potentially relevant articles were obtained; however, after screening the abstracts, most were determined to be irrelevant to our analysis. Of the remaining 51 articles, 13 articles were removed because of an insufficient number of cases or unusable data. Eventually, 23 studies in English (12-21, 30-42) and 15 in Chinese (43-57), including 39 comparisons of the AGT M235T polymorphism that all adopted the observational study design, satisfied the eligibility criteria (Fig. 1). A total of 39 comparisons from the 38 studies of the AGT M235T polymorphism were included in this updated meta-analysis.
Figure 1.
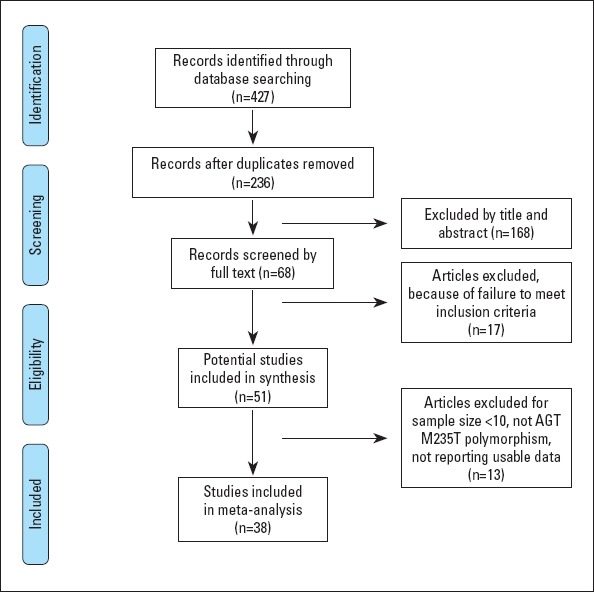
Flow of studies for meta-analysis
The relevant studies included 9225 cases and 8406 controls (Tables 1 and 2). Reference to the “overall population” indicates meta-analysis without ethnic subdivisions. Ethnicity-specific meta-analysis was categorized by Caucasian, East Asian, and other races (miscellaneous subgroup).
Association of the AGT M235T polymorphism with CVD risk in the overall population
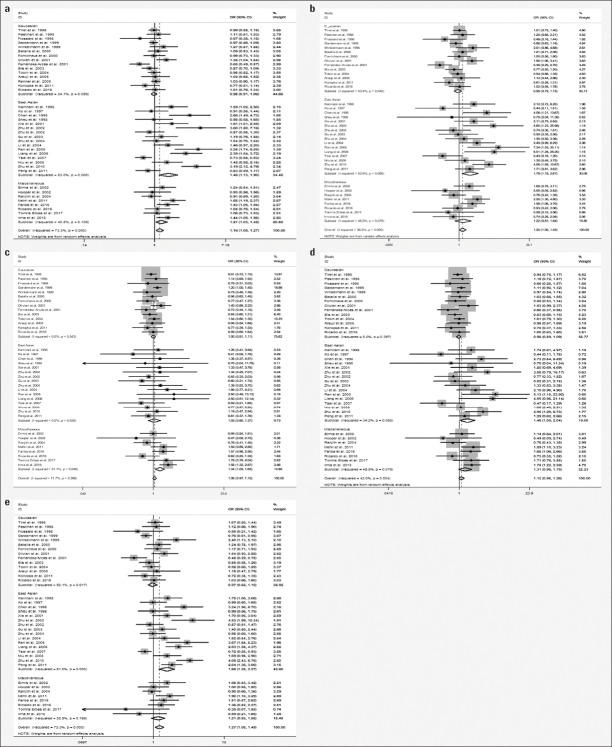
As shown in Figure 2, significant heterogeneity among studies was observed for the overall population (Ph<0.10 or I2≥50%). Using the random effect models, M235T was found to be associated with an increased risk of CVD in the allelic (T vs. M: OR=1.16, 95% CI=1.05–1.27, p<0.001) and recessive (TT vs. MT+MM: OR=1.14, 95% CI=1.06–1.23, p<0.001) models.
Figure 2.
Forest plots of the association between AGT M235T polymorphism and CVD risk. (a) Allelic model, (b) homozygote model, (c) heterozygote model, (d) dominant model, and (e) recessive model
Association of the M235T polymorphism of the AGT gene with CVD risk in subgroups analysis
When analyses were subdivided according to ethnicity, no associations were noted for Caucasians using any of the five genetic models. However, for the East Asian subgroup, M235T was significantly associated with CVD risk in allelic (T vs. M: OR=1.46, 95% CI=1.13–1.90, p<0.001), homozygous (TT vs. MM: OR=1.78, 95% CI=1.18–2.67, p=0.01), dominant (MT+TT vs. MM: OR=1.47, 95% CI=1.05–2.04, p=0.02), and recessive (TT vs. MT+MM: OR=1.68, 95% CI=1.25–2.27, p<0.001) models. In miscellaneous populations, a significant association between M235T and CVD risk was observed in the allelic model (T vs. M: OR=1.21, 95% CI=1.07–1.36, p<0.001), but no association was observed in the other four genetic models. In subgroup analysis, neither moderate nor large heterogeneity was observed among Caucasians, but true heterogeneity was noted among East Asians (T vs. M: Ph<0.10, I2=83% and TT vs. MT+MM: Ph<0.10, I2=81%) and miscellaneous populations (MT vs. MM: Ph=0.05, I2=51.1%) (Table 3).
Table 3.
Overall and subgroup meta-analysis of the association between AGT M235T polymorphism and risk of cardiovascular disease
| Categories | n | T vs. M | TT vs. MM | MT vs. MM | MT+TT vs. MM | TT vs. MM+MT | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| OR (95% CI) | P | I2 (%)/Ph | OR (95% CI) | P 0.00 | I2 (%)/Ph 0.00/0.00 | OR (95% CI) | P | I2 (%)/Ph | OR (95% CI) | P | I2 (%)/Ph | OR (95% CI) | P | I2 (%)/Ph | ||
| Overall | 22 | 1.16 (1.05-1.27) | 0.003 | 73.3/0.00 | 1.20 (1.00-1.45) | 0.05 | 56.5/0.00 | 1.06 (fixed) (0.97-1.15) | 0.20 | 11.7/0.27 | 1.12 (0.99-1.26) | 0.085 | 42.0/0.00 | 1.14 (1.06-1.23) | 0.003 | 73.3/0.00 |
| Subgroup (by population) | ||||||||||||||||
| Caucasian | 0.99 (fixed) (0.93-1.04) | 0.584 | 34.1/0.10 | 0.95 (0.79-1.16) | 0.63 | 43.4/0.04 | 1.00 (fixed) (0.91-1.11) | 0.94 | 00.0/0.54 | 0.99 (0.89-1.09) | 0.761 | 05.0/0.40 | 0.97 (0.82-1.16) | 0.743 | 50.1/0.02 | |
| East Asian | 1.46 (1.13-1.90) | 0.004 | 83.0/0.00 | 1.78 (1.18-2.67) | 0.01 | 53.8/0.01 | 1.05 (fixed) (0.80-1.38) | 0.73 | 00.0/0.84 | 1.47 (1.05-2.04) | 0.023 | 34.2/0.09 | 1.68 (1.25-2.27) | 0.001 | 81.0/0.00 | |
| Miscellaneous | 1.21 (fixed) (1.07-1.36) | 0.002 | 40.8/0.11 | 1.22 (0.81-1.84) | 0.33 | 45.0/0.08 | 1.23 (0.90-1.70) | 0.20 | 51.1/0.05 | 1.31 (0.98-1.75) | 0.065 | 45.5/0.08 | 1.20 (fixed) (0.98-1.46) | 0.082 | 32.8/0.17 | |
n- study numbers, OR- odds ratio, CI- confidence interval, bold values represent statistically significant findings, Ph - P heterogeneity (P<0.1 was considered as a significant difference), fixed - the fixed effects model
Publication bias and sensitivity analysis
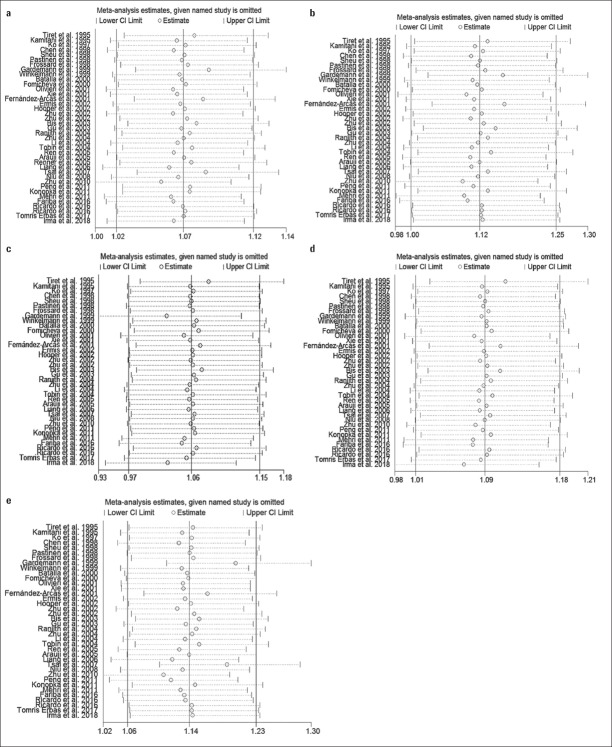
Publication bias was not detected in the analyses of the homozygote, heterozygote, or dominant models (p>0.05, for all). However, publication bias was noted in the analyses of the associations between M235T polymorphisms and CVD risk (allelic model: PEgger=0.01, PBegg=0.02 and recessive model: PEgger=0.01) (Table 4). Sensitivity analyses showed that the present meta-analysis was relatively stable and credible (Fig. 3).
Table 4.
Publication bias assessment of this meta-analysis
| Genetic model | Egger’s test t-value | P | Begg’s test t-value | P |
|---|---|---|---|---|
| Allelic model | 2.70 | 0.01 | 2.42 | 0.02 |
| Homozygote model | 1.84 | 0.07 | 1.28 | 0.20 |
| Heterozygote model | -0.2 | 0.85 | 0.10 | 0.92 |
| Dominant model | 1.18 | 0.24 | 0.85 | 0.39 |
| Recessive model | 2.83 | 0.01 | 1.91 | 0.06 |
P<0.05 was considered as a significant difference
Figure 3.
Sensitivity analyses of the association between AGT M235T polymorphism and CVD risk. (a) Allelic model, (b) homozygote model, (c) heterozygote model, (d) dominant model, and (e) recessive model
Meta-regression
A meta-regression analysis for several potential sources of heterogeneity, including published year, sample size, age, gender, outcome, and ethnic background, was performed. Single covariates were added in the allelic, homozygote, dominant, and recessive models. The results suggest that the East Asian population (allelic model: p=0.006, homozygote model: p=0.010, dominant model: p=0.022, and recessive model: p=0.005) and study size (homozygote model: p=0.042 and recessive model: p=0.010) contributed to the observed heterogeneity across all studies of the association between AGT M235T polymorphisms and CVD susceptibility.
Discussion
The AGT gene (on chromosome 1q42–43) comprises five exons and four introns spanning 12 kb, with the M235T polymorphism in exon 2. The M235T variant has been demonstrated to alter plasma AGT levels (58, 59), with elevated levels of serum AGT for patients carrying the T allele (60). Furthermore, a positive correlation exists between AGT M235T genotype and plasma AGT levels in survivors of myocardial infarction (MI) (14). Elevation circulating AGT levels are associated with an increase in the concentration of angiotensin II, which activates cardiomyocyte hypertrophy and fibroblast proliferation by stimulation the AT1 receptor (61, 62). In addition, angiotensin II stimulates vascular apoptosis and may promote the retention of low-density lipoprotein in the coronary arteries, oxidize, and be assimilated by phagocytes, ultimately contributing to the dysfunction of the vascular endothelium and myocardial ischemia and rupture of atherosclerotic plaque (63-65). These processes all play critical roles in promoting the pathological development of CVD.
In previous studies, the distribution of the AGT M235T variant has been shown to differ significantly among various populations. Katsuya et al. (66) demonstrated that homozygous AGT T235 is an independent risk factor, which carries a two-fold increased risk of CVD. In contrast, Tiret et al. (31) suggested that the AGT genetic polymorphism can lead to predisposition to hypertension, with no relationship to CVD. Renjith et al. (20) and Renner et al. (19) reported similar results showing that AGT genotypes are neither related to CVD nor high blood pressure. Batalla et al. (12) and Bonfim-Silva et al. (17) provided evidence of the synergistic effect between AGT polymorphism and CVD, suggesting that M235T polymorphism is significantly correlated with MI and hypertension. Recently, Raygan et al. (16) suggested that M235T polymorphism in Asians can be a useful biomarker for screening of individuals susceptible to MI. Given the disparity of the results, we sought to provide an updated meta-analysis to resolve the discrepancies among studies.
Our results clearly demonstrate a difference in the association of M235T polymorphism among Asians and Caucasians, suggesting that there is heterogeneity based on ethnicity. Our results suggest that the T allele is a genetic risk factor for CVD in Asians, as well as in the miscellaneous population. Previous meta-analyses based on Chinese populations have shown similar positive results (22, 23). Although two recent meta-analyses (16, 67) reported an association between the AGT M235T variant and MI, they did not include recently published studies, and the literature sample size was smaller than the current analysis. Thus, an update of previous meta-analyses that included diverse populations was warranted. Our results include the largest sample size to date and provide an ethnicity-based explanation for different results among studies. The association of AGT M235T with stroke was not addressed in the present study but would be an important topic for consideration in future studies to maintain a relatively narrow focus.
The following potential factors may account for differences observed among the various ethnic groups: (1) population diversity (68), (2) different habits among populations (69), and (3) environmental factors leading to differences in susceptibility to CVD (70). Furthermore, we speculate that the non-significant association among Caucasians may be due to the relatively low frequency of the TT genotype in this population. Moreover, owing to the limited number of these studies, the miscellaneous ethnic subgroups were not analyzed further. Therefore, more studies with a larger sample size may reveal factors that influence differences in the association of AGT M235T and CVD, especially among Caucasians and other ethnic populations. The association between AGT M235T and CVD among Asians is evident.
The heterogeneity of associations across all included studies should be noted as it may potentially affect the strength of the present study. Thus, the random effects model was used, and our analysis was based on different ethnic subgroups. Ethnic background and sample size were found to be factors of heterogeneity. However, heterogeneity was still high within the East Asian subgroup. The heterogeneity of results among those included in these studies may be explained by the quality of the included studies, classification of CVD, and sampling criteria. The heterogeneity of genetic effects among individual studies may also be caused by the existence of genetic and environmental or genetic interactions.
Study limitations
The primary limitations of our meta-analysis include: (1) significant publication bias in the allelic and recessive models, (2) insufficient genotyping data of AGT M235T in miscellaneous races, which limited the ability to draw conclusions regarding this population, and (3) potential heterogeneity of clinical variables, such as the general condition of subjects, their medical history, medication compliance, complications of CVD, and other factors.
Conclusion
The genetic polymorphism of AGT M235T is associated with a critical risk of CVD in East Asian populations, with no detectable association in Caucasian populations. However, further studies with multiple ethnicities and rigorous designs should be performed to confirm these conclusions.
Acknowledgment
The authors thank Professors Li and Liu for their great advices on studies of the unpublished literature.
Footnotes
Ethical approval: Ethical approval was not required for this study design.
Conflict of interest: None declared.
Peer-review: Externally peer-reviewed.
Authorship contributions: Concept – H.C.; Design – C.Z., H.C.; Supervision – C.Z., H.C.; Fundings – H.C; Materials – C.Z., H.Z., K.H.; Data collection &/or processing – C.Z., H.Z., Y.Z., Y.Y.Z.; Analysis &/or interpretation – C.Z., H.Z., K.H.; Literature search – K.H., Y.Z.; Writing – C.Z., Y.Y.Z.; Critical review – Y.Z., Y.Y.Z.
References
- 1.Roth GA, Huffman MD, Moran AE, Feigin V, Mensah GA, Naghavi M, et al. Global and regional patterns in cardiovascular mortality from 1990 to 2013. Circulation. 2015;132:1667–78. doi: 10.1161/CIRCULATIONAHA.114.008720. [DOI] [PubMed] [Google Scholar]
- 2.Ounpuu S, Negassa A, Yusuf S. INTER-HEART:A global study of risk factors for acute myocardial infarction. Am Heart J. 2001;141:711–21. doi: 10.1067/mhj.2001.114974. [DOI] [PubMed] [Google Scholar]
- 3.Roberts R, Stewart AF. Genes and coronary artery disease:where are we? J Am Coll Cardiol. 2012;60:1715–21. doi: 10.1016/j.jacc.2011.12.062. [DOI] [PubMed] [Google Scholar]
- 4.Meininger JC, Hayman LL, Coates PM, Gallagher PR. Genetic and environmental influences on cardiovascular disease risk factors in adolescents. Nurs Res. 1998;47:11–8. doi: 10.1097/00006199-199801000-00004. [DOI] [PubMed] [Google Scholar]
- 5.Xia J, Cai W, Peng C. Association of APOA5 T1131C polymorphism and risk of coronary artery disease. Int J Clin Exp Med. 2015;8:8986–94. [PMC free article] [PubMed] [Google Scholar]
- 6.Yuepeng J, Zhao X, Zhao Y, Li L. Gene polymorphism associated with TNF-α(G308A) IL-6 (C174G) and susceptibility to coronary atherosclerotic heart disease:A meta-analysis. Medicine (Baltimore) 2019;98:e13813. doi: 10.1097/MD.0000000000013813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ryu SK, Cho EY, Park HY, Im EK, Jang YS, Shin GJ, et al. Renin-angiotensin-aldosterone system (RAAS) gene polymorphism as a risk factor of coronary in-stent restenosis. Yonsei Med J. 2002;43:461–72. doi: 10.3349/ymj.2002.43.4.461. [DOI] [PubMed] [Google Scholar]
- 8.Wang JG, Staessen JA. Genetic polymorphisms in the renin-angiotensin system:relevance for susceptibility to cardiovascular disease. Eur J Pharmacol. 2000;410:289–302. doi: 10.1016/s0014-2999(00)00822-0. [DOI] [PubMed] [Google Scholar]
- 9.Koh KK, Han SH, Oh PC, Shin EK, Quon MJ. Combination therapy for treatment or prevention of atherosclerosis:focus on the lipid-RAAS interaction. Atherosclerosis. 2010;209:307–13. doi: 10.1016/j.atherosclerosis.2009.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Heidari F, Vasudevan R, Mohd ASZ, Ismail P, Etemad A, Pishva SR, et al. Association of insertion/deletion polymorphism of angiotensin-converting enzyme gene among Malay male hypertensive subjects in response to ACE inhibitors. J Renin Angiotensin Aldosterone Syst. 2015;16:872–9. doi: 10.1177/1470320314538878. [DOI] [PubMed] [Google Scholar]
- 11.Kondo H, Ninomiya T, Hata J, Hirakawa Y, Yonemoto K, Arima H, et al. Angiotensin I-converting enzyme gene polymorphism enhances the effect of hypercholesterolemia on the risk of coronary heart disease in a general Japanese population:the hisayama study. J Atheroscler Thromb. 2015;22:390–403. doi: 10.5551/jat.24166. [DOI] [PubMed] [Google Scholar]
- 12.Batalla A, Alvarez R, Reguero JR, Hevia S, Iglesias-Cubero G, Alvarez V, et al. Synergistic effect between apolipoprotein E and angiotensinogen gene polymorphisms in the risk for early myocardial infarction. Clin Chem. 2000;46:1910–5. [PubMed] [Google Scholar]
- 13.Gardemann A, Stricker J, Humme J, Nguyen QD, Katz N, Philipp M, et al. Angiotensinogen T174M and M235T gene polymorphisms are associated with the extent of coronary atherosclerosis. Atherosclerosis. 1999;145:309–14. doi: 10.1016/s0021-9150(99)00082-9. [DOI] [PubMed] [Google Scholar]
- 14.Winkelmann BR, Russ AP, Nauck M, Klein B, Böhm BO, Maier V, et al. Angiotensinogen M235T polymorphism is associated with plasma angiotensinogen and cardiovascular disease. Am Heart J. 1999;137:698–705. doi: 10.1016/s0002-8703(99)70226-7. [DOI] [PubMed] [Google Scholar]
- 15.Hooper WC, Dowling NF, Wenger NK, Dilley A, Ellingsen D, Evatt BL. Relationship of venous thromboembolism and myocardial infarction with the renin-angiotensin system in African-Americans. Am J Hematol. 2002;70:1–8. doi: 10.1002/ajh.10078. [DOI] [PubMed] [Google Scholar]
- 16.Raygan F, Karimian M, 1, Rezaeian A, Bahmani B, Behjati M. Angiotensinogen-M235T as a risk factor for myocardial infarction in Asian populations:a genetic association study and a bioinformatics approach. Croat Med J. 2016;57:351–62. doi: 10.3325/cmj.2016.57.351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bonfim-Silva R, Guimarães LO, Souza Santos J, Pereira JF, Leal Barbosa AA, Souza Rios DL. Case-control association study of polymorphisms in the angiotensinogen and angiotensin-converting enzyme genes and coronary artery disease and systemic artery hypertension in African-Brazilians and Caucasian-Brazilians. J Genet. 2016;95:63–9. doi: 10.1007/s12041-015-0599-5. [DOI] [PubMed] [Google Scholar]
- 18.Isordia-Salas I, Alvarado-Moreno JA, Jiménez-Alvarado RM, Hernández-Juárez J, Santiago-Germán D, Leaños-Miranda A, et al. Association of renin-angiotensin system genes polymorphisms and risk of premature ST elevation myocardial infarction in young Mexican population. Blood Coagul Fibrinolysis. 2018;29:267–74. doi: 10.1097/MBC.0000000000000714. [DOI] [PubMed] [Google Scholar]
- 19.Renner W, Nauck M, Winkelmann BR, Hoffmann MM, Scharnagl H, Mayer V, et al. Association of angiotensinogen haplotypes with angiotensinogen levels but not with blood pressure or coronary artery disease:the Ludwigshafen Risk and Cardiovascular Health Study. J Mol Med (Berl) 2005;83:235–9. doi: 10.1007/s00109-004-0618-0. [DOI] [PubMed] [Google Scholar]
- 20.Ranjith N, Pegoraro RJ, Rom L, Lanning PA, Naidoo DP. Renin-angiotensin system and associated gene polymorphisms in myocardial infarction in young South African Indians. Cardiovasc J S Afr. 2004;15:22–6. [PubMed] [Google Scholar]
- 21.Erbas T, Cinar N, Dagdelen S, Gedik A, Yorgun H, Canpolat U, et al. Association between ACE and AGT polymorphism and cardiovascular risk in acromegalic patients. Pituitary. 2017;20:569–77. doi: 10.1007/s11102-017-0819-5. [DOI] [PubMed] [Google Scholar]
- 22.Li X, Li Q, Wang Y, Li Y, Ye M, Ren J, et al. AGT gene polymorphisms (M235T, T174M) are associated with coronary heart disease in a Chinese population. J Renin Angiotensin Aldosterone Syst. 2013;14:354–9. doi: 10.1177/1470320312452029. [DOI] [PubMed] [Google Scholar]
- 23.Wang YJ, Pan Y. Angiotensinogen gene M235T polymorphism and risk of coronary artery disease:a meta-analysis. Mol Med Rep. 2012;6:884–8. doi: 10.3892/mmr.2012.1011. [DOI] [PubMed] [Google Scholar]
- 24.Robins J, Greenland S, Breslow NE. A general estimator for the variance of the Mantel-Haenszel odds ratio. Am J Epidemiol. 1986;124:719–23. doi: 10.1093/oxfordjournals.aje.a114447. [DOI] [PubMed] [Google Scholar]
- 25.Egger M, Smith GD, Phillips AN. Meta-analysis:principles and procedures. BMJ. 1997;315:1533–7. doi: 10.1136/bmj.315.7121.1533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7:177–88. doi: 10.1016/0197-2456(86)90046-2. [DOI] [PubMed] [Google Scholar]
- 27.Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002;21:1539–58. doi: 10.1002/sim.1186. [DOI] [PubMed] [Google Scholar]
- 28.Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315:629–34. doi: 10.1136/bmj.315.7109.629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994;50:1088–101. [PubMed] [Google Scholar]
- 30.Kamitani A, Rakugi H, Higaki J, Ohishi M, Shi SJ, Takami S, et al. Enhanced predictability of myocardial infarction in Japanese by combined genotype analysis. Hypertension. 1995;25:950–3. doi: 10.1161/01.hyp.25.5.950. [DOI] [PubMed] [Google Scholar]
- 31.Tiret L, Ricard S, Poirier O, Arveiler D, Cambou JP, Luc G, et al. Genetic variation at the angiotensinogen locus in relation to high blood pressure and myocardial infarction:the ECTIM Study. J Hypertens. 1995;13:311–7. [PubMed] [Google Scholar]
- 32.Pastinen T, Perola M, Niini P, Terwilliger J, Salomaa V, Vartiainen E, et al. Array-based multiplex analysis of candidate genes reveals two independent and additive genetic risk factors for myocardial infarction in the Finnish population. Hum Mol Genet. 1998;7:1453–62. doi: 10.1093/hmg/7.9.1453. [DOI] [PubMed] [Google Scholar]
- 33.Frossard PM, Hill SH, Elshahat YI, Obineche EN, Bokhari AM, Lestringant GG, et al. Associations of angiotensinogen gene mutations with hypertension and myocardial infarction in a gulf population. Clin Genet. 1998;54:285–93. doi: 10.1034/j.1399-0004.1998.5440405.x. [DOI] [PubMed] [Google Scholar]
- 34.Fomicheva EV, Gukova SP, Larionova-Vasina VI, Kovalev YR, Schwartz EI. Gene-gene interaction in the RAS system in the predisposition to myocardial infarction in elder population of St. Petersburg (Russia) Mol Genet Metab. 2000;69:76–80. doi: 10.1006/mgme.1999.2924. [DOI] [PubMed] [Google Scholar]
- 35.Fernández-Arcás N, Dieguez-Lucena JL, Muñoz-Morán E, Ruiz-Galdón M, Espinosa-Caliani S, Aranda-Lara P, et al. Both alleles of the M235T polymorphism of the angiotensinogen gene can be a risk factor for myocardial infarction. Clin Genet. 2001;60:52–7. doi: 10.1034/j.1399-0004.2001.600108.x. [DOI] [PubMed] [Google Scholar]
- 36.Olivieri O, Stranieri C, Girelli D, Pizzolo F, Grazioli S, Russo C, et al. Homozygosity for angiotensinogen 235T variant increases the risk of myocardial infarction in patients with multi-vessel coronary artery disease. J Hypertens. 2001;19:879–84. doi: 10.1097/00004872-200105000-00007. [DOI] [PubMed] [Google Scholar]
- 37.Ermis C, Tsai MY, Hanson NQ, Akar N, Aras O. Angiotensin I converting enzyme, angiotensin II type 1 receptor and angiotensinogen polymorphisms and early myocardial infarction in Turkish population. Thromb Haemost. 2002;88:693–4. [PubMed] [Google Scholar]
- 38.Bis JC, Smith NL, Psaty BM, Heckbert SR, Edwards KL, Lemaitre RN, et al. Angiotensinogen Met235Thr polymorphism, angiotensin-converting enzyme inhibitor therapy, and the risk of nonfatal stroke or myocardial infarction in hypertensive patients. Am J Hypertens. 2003;16:1011–7. doi: 10.1016/j.amjhyper.2003.07.018. [DOI] [PubMed] [Google Scholar]
- 39.Tobin MD, Braund PS, Burton PR, Thompson JR, Steeds R, Channer K, et al. Genotypes and haplotypes predisposing to myocardial infarction:a multilocus case-control study. Eur Heart J. 2004;25:459–67. doi: 10.1016/j.ehj.2003.11.014. [DOI] [PubMed] [Google Scholar]
- 40.Araújo MA, Goulart LR, Cordeiro ER, Gatti RR, Menezes BS, Lourenço C, et al. Genotypic interactions of renin-angiotensin system genes in myocardial infarction. Int J Cardiol. 2005;103:27–32. doi: 10.1016/j.ijcard.2004.07.009. [DOI] [PubMed] [Google Scholar]
- 41.Mehri S, Mahjoub S, Farhati A, Bousaada R, Ben Arab S, Baudin B, et al. Angiotensinogen gene polymorphism in acute myocardial infarction patients. J Renin Angiotensin Aldosterone Syst. 2011;12:42–7. doi: 10.1177/1470320310376425. [DOI] [PubMed] [Google Scholar]
- 42.Konopka A, Szperl M, Piotrowski W, Roszczynko M, Stępińska J. Influence of renin-angiotensin system gene polymorphisms on the risk of ST-segment-elevation myocardial infarction and association with coronary artery disease risk factors. Mol Diagn Ther. 2011;15:167–76. doi: 10.1007/BF03256407. [DOI] [PubMed] [Google Scholar]
- 43.Ko YL, Ko YS, Wang SM, Chu PH, Teng MS, Cheng NJ, et al. Angiotensinogen and angiotensin-I converting enzyme gene polymorphisms and the risk of coronary artery disease in Chinese. Hum Genet. 1997;100:210–4. doi: 10.1007/s004390050492. [DOI] [PubMed] [Google Scholar]
- 44.Sheu WH, Lee WJ, Jeng CY, Young MS, Ding YA, Chen YT. Angiotensinogen gene polymorphism is associated with insulin resistance in nondiabetic men with or without coronary heart disease. Am Heart J. 1998;136:125–31. doi: 10.1016/s0002-8703(98)70192-9. [DOI] [PubMed] [Google Scholar]
- 45.Chen D, Zhang M, Fan W, Shi H, Li Y, Chen Q, et al. A molecular variant of angiotensinogen gene is associated with myocardial infarction in Chinese. Zhonghua Yi Xue Yi Chuan Xue Za Zhi. 1998;15:133–5. [PubMed] [Google Scholar]
- 46.Xie Y, Zhang Z, Ma W, Yang D, Geng Z, Jia Y, et al. Association between gene polymorphism of angiotensiongen (M235T) and coronary heart disease. Jiangsu Med J. 2001;27:254–6. [Google Scholar]
- 47.Zhu T, Xu Z, Yang Z, Xie Y, Zhang Z, Jia Y, et al. The association of polymorphism of three genes of the renninangiotensin system with coronary heart disease. Jiangsu Med J. 2002;28:745–6. [Google Scholar]
- 48.Zhu Y, Hs Long H. Relationship of angiotensionogen gene polymorphism and myocardial infarction in Chinese. Chin J Cardiol. 2002;12:67. [Google Scholar]
- 49.Gu W, Zhu J, Pan M, Yuan Z, Gong Y, Cui Z, et al. Effects of angiotensinogen and apolipoprotein E gene polymorphisms on coronary heart disease. J NanTong Med Univ. 2003;4:394–5. [Google Scholar]
- 50.Li L FXH, Zhang YW. Association between angiotensinogen gene polymorphism and severity of coronary artery stenosis. J Med Forum. 2004;25:12–4. [Google Scholar]
- 51.Zhu T, Pan J, Shen Z, Zhao Y, Shen Z. Gene polymorphisms of reninangiotensin system and coronary thrombosis disease. Journal of Experimental Hematology. 2004;12:674–9. [PubMed] [Google Scholar]
- 52.Ren J, Jia Y, Lv J, Guo X. Relationship between angiotensinogen gene M235T polymorphism and coronary heart disease. Journal of Shanxi Medical University. 2005;36:439–42. [Google Scholar]
- 53.Liang Q DYG, Yang XL. Research of ACE, angiotensinogen and endothelial nitric oxide synthase (eNOS) gene polymorphisms in predisposition to CHD with chip technology. Hebei Medicine. 2006;12:403–6. [Google Scholar]
- 54.Tsai CT, Hwang JJ, Ritchie MD, Moore JH, Chiang FT, Lai LP, et al. Renin-angiotensin system gene polymorphisms and coronary artery disease in a large angiographic cohort:detection of high order gene-gene interaction. Atherosclerosis. 2007;195:172–80. doi: 10.1016/j.atherosclerosis.2006.09.014. [DOI] [PubMed] [Google Scholar]
- 55.Niu Y, Luo L, Peng J, Mei X, Peng Z, Gong W. Relationship between Renin-Angiotensin System Gene Polymorphism and Ischemic Chronic Heart Failure. Academic Journal of Sun Yat-sen University of Medi. 2008;29:168–72. [Google Scholar]
- 56.Zhu YG, Wang FJ, Fu ZQ, Xiang YD. Association between the gene polymorphism of angiotensinogen-converting enzyme and coronary artery disease. Chin J Arterioscler. 2010;18:405–8. [Google Scholar]
- 57.Peng J, Luo LY, Li T, Mei X, Niu YQ, Gong WX. Relationship between renninangiotensin system gene polymorphism and ischemic chronic heart failure in aged coronary artery disease patient. Chinese Circulation Journal. 2011;26:93–6. [Google Scholar]
- 58.Niu T, Chen X, Xu X. Angiotensin converting enzyme gene insertion/deletion polymorphism and cardiovascular disease:therapeutic implications. Drugs. 2002;62:977–93. doi: 10.2165/00003495-200262070-00001. [DOI] [PubMed] [Google Scholar]
- 59.Lanz JR, Pereira AC, Lemos PA, Martinez E, Krieger JE. Angiotensinogen M235T polymorphism is associated with coronary artery disease severity. Clin Chim Acta. 2005;362:176–81. doi: 10.1016/j.cccn.2005.06.004. [DOI] [PubMed] [Google Scholar]
- 60.Jeunemaitre X, Soubrier F, Kotelevtsev YV, Lifton RP, Williams CS, Charru A, et al. Molecular basis of human hypertension:role of angiotensinogen. Cell. 1992;71:169–80. doi: 10.1016/0092-8674(92)90275-h. [DOI] [PubMed] [Google Scholar]
- 61.Horiuchi M, Akishita M, Dzau VJ. Recent progress in angiotensin II type 2 receptor research in the cardiovascular system. Hypertension. 1999;33:613–21. doi: 10.1161/01.hyp.33.2.613. [DOI] [PubMed] [Google Scholar]
- 62.Henrion D, Kubis N, Lévy BI. Physiological and pathophysiological functions of the AT(2) subtype receptor of angiotensin II:from large arteries to the microcirculation. Hypertension. 2001;38:1150–7. doi: 10.1161/hy1101.096109. [DOI] [PubMed] [Google Scholar]
- 63.Dietz R, von Harsdorf R, Gross M, Krämer J, Gulba D, Willenbrock R, et al. Angiotensin II and coronary artery disease, congestive heart failure, and sudden cardiac death. Basic Res Cardiol. 1998;93(Suppl2):101–8. doi: 10.1007/s003950050230. [DOI] [PubMed] [Google Scholar]
- 64.Keidar S, Attias J, Heinrich R, Coleman R, Aviram M. Angiotensin II atherogenicity in apolipoprotein E deficient mice is associated with increased cellular cholesterol biosynthesis. Atherosclerosis. 1999;146:249–57. doi: 10.1016/s0021-9150(99)00145-8. [DOI] [PubMed] [Google Scholar]
- 65.Li W, Li J, Hao P, Chen W, Meng X, Li H, et al. Imbalance between angiotensin II and angiotensin-(1-7) in human coronary atherosclerosis. J Renin Angiotensin Aldosterone Syst. 2016;17 doi: 10.1177/1470320316659618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Katsuya T, Koike G, Yee TW, Sharpe N, Jackson R, Norton R, et al. Association of angiotensinogen gene T235 variant with increased risk of coronary heart disease. Lancet. 1995;345:1600–3. doi: 10.1016/s0140-6736(95)90115-9. [DOI] [PubMed] [Google Scholar]
- 67.Liang X, Qiu J, Liu X, Li X, Zhao S, Wang J, et al. Polymorphism of angiotensinogen gene M235T in myocardial infarction and brain infarction:a meta-analysis. Gene. 2013;529:73–9. doi: 10.1016/j.gene.2013.07.095. [DOI] [PubMed] [Google Scholar]
- 68.Walker AR, Walker BF, Segal I. Some puzzling situations in the onset, occurrence and future of coronary heart disease in developed and developing populations, particularly such in sub-Saharan Africa. J R Soc Promot Health. 2004;124:40–6. doi: 10.1177/146642400312400112. [DOI] [PubMed] [Google Scholar]
- 69.Micha R, Peñalvo JL, Cudhea F, Imamura F, Rehm CD, Mozaffarian D. Association Between Dietary Factors and Mortality From Heart Disease, Stroke, and Type 2 Diabetes in the United States. JAMA. 2017;317:912–24. doi: 10.1001/jama.2017.0947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Poelman M, Strak M, Schmitz O, Hoek G, Karssenberg D, Helbich M, et al. Relations between the residential fast-food environment and the individual risk of cardiovascular diseases in The Netherlands:A nationwide follow-up study. Eur J Prev Cardiol. 2018;25:1397–405. doi: 10.1177/2047487318769458. [DOI] [PMC free article] [PubMed] [Google Scholar]