Abstract
Background/Aims
Endoscopic resection is a standard treatment for stage T1a esophageal cancer, with esophagectomy or radical radiation therapy (RT) performed for stage T1b lesions. This study aimed to compare treatment outcomes of each modality for clinical stage T1 esophageal cancer.
Methods
In total, 179 patients with clinical T1N0M0-stage esophageal cancer treated from 2006 to 2016 were retrospectively evaluated. Sixty-two patients with clinical T1a-stage cancer underwent endoscopic resection. Among 117 patients with clinical T1b-stage cancer, 82 underwent esophagectomy, and 35 received chemoradiotherapy or RT. We compared overall survival (OS) and recurrence-free survival (RFS) rates for each treatment modality.
Results
The median follow-up time was 32 months (range, 1 to 120 months). The 5-year OS and RFS rates for patients with stage T1a cancer receiving endoscopic resection were 100% and 85%, respectively. For patients with stage T1b, the 5-year OS and RFS rates were 78% and 77%, respectively, for the esophagectomy group; 80% and 44%, respectively, for the RT alone group; and 96% and 80%, respectively, for the chemoradiation group. The esophagectomy group showed significantly higher RFS than the RT alone group (p=0.04). There was no significant difference in RFS between the esophagectomy and chemoradiation groups (p=0.922). Grade 4 or higher treatment-related complications occurred in four patients who underwent esophagectomy.
Conclusions
Endoscopic resection appeared to be an adequate treatment for patients with T1a-stage esophageal cancer. The multidisciplinary approach involving chemoradiation was comparable to esophagectomy in terms of survival outcome without serious complications for T1b-stage esophageal cancer.
Keywords: Esophageal neoplasm, Endoscopic mucosal resection, Esophagectomy, Radiotherapy, Chemoradiotherapy
INTRODUCTION
Esophageal cancer is one of the most common and aggressive cancers worldwide. It is a frequently diagnosed cancer, with approximately 460,000 new cases and 400,000 deaths worldwide in 2012.1 The incidence of this disease has increased dramatically during the past 3 decades.2 With this increasing incidence, the overall prognosis of patients with esophageal cancer remains poor, with a 5-year survival rate of 14%.3 However, with the increasing use of endoscopic surveillance and early detection, early-stage cancers are being diagnosed more frequently. In early-stage (T1N0M0) esophageal cancer, lesions are limited to the mucosa (T1a) or submucosa (T1b), and this diagnosis accounts for approximately 20% of all initial diagnoses.4 These early-stage cancers present the best opportunity for a cure at diagnosis, which can affect the overall prognosis of all esophageal cancers. The 5-year survival rate can be as high as 85%; a stark contrast to 10% when diagnosed at an advanced stage.5
Esophagectomy has been the standard treatment for T1b esophageal cancer, although with some debate. Recently, there has been increasing interest in endoscopic therapies for lesions of this stage. These include ablative techniques, such as photodynamic therapy and endoscopic mucosal resection (EMR).6,7 Radiation therapy (RT) can also be used for superficial esophageal cancer, and favorable outcomes have also been reported for this noninvasive treatment.8 Moreover, RT has undergone notable advances during the past decade. Three-dimensional (3D) conformal RT, intensity-modulated radiation therapy (IMRT) based on computed tomography (CT) images and multileaf collimators have contributed to the accurate delivery of high doses to the target, sparing normal tissues. However, the role of definitive RT in treating superficial esophageal cancer compared with other treatment modalities, such as esophagectomy and endoscopic therapies, has not yet been elucidated.
In Yonsei Cancer Center, we perform esophagectomy, endoscopic therapies, or RT for T1 esophageal cancers according to the physicians’ decision after a multidisciplinary discussion. This study aimed to compare the outcomes of each treatment modality while examining the experience of a single institution with the treatment of early-stage esophageal cancers.
MATERIALS AND METHODS
1. Patients
We retrospectively identified 208 patients who received treatments for clinical T1N0M0 esophageal cancer between February 2006 and December 2016 at Severance Hospital, Seoul, Korea. Among 208 patients, 27 with other malignancies and two with involvement of the esophagogastric junction were excluded. A total of 179 patients were included in the final analysis. Data were collected from patients’ medical and surgical records and a departmental database (IRB exemption number: 2018-3377-001).
2. Treatment
Before the treatment decision, endoscopic ultrasonography (EUS) was performed for all patients to evaluate the depth of cancer invasion. The depth of tumor infiltration for clinical T categorization was determined by EUS based on the deepest invasion depth. According to the American Joint Committee on Cancer guidelines 7th edition, the depth of invasion was categorized as T1a if the tumor invaded the lamina propria or muscularis mucosae and as T1b if there was submucosal invasion. Careful examination of the ultrasound image revealed the presence of abnormal or enlarged lymph nodes that were likely to harbor cancer and occasionally signs of distant spread, such as a lesion in the surrounding organs. In addition, the nodal status and occurrence of distant metastasis were evaluated by CT and positron emission tomography-CT.
The treatment decision was made fundamentally according to the National Comprehensive Cancer Network guidelines for these stages after a multidisciplinary discussion. Physicians preferentially considered endoscopic therapy for T1a tumors and esophagectomy for T1b tumors. RT was performed in cases of surgical difficulties because of the tumor location, advanced patient age, or comorbidities and for patients who refused surgery. Patients who were initially diagnosed as having clinical T1a esophageal cancer but diagnosed with pathological T1b disease after endoscopic therapy and received complete surgery or RT were included in this study. These patients were examined separately from those who were finally diagnosed with pT1a disease.
1) Endoscopic therapy
Endoscopic resection was conducted by EMR consisting of EMR using a transparent cap procedure, EMR with precutting, and endoscopic submucosal dissection (ESD) with various knives, such as a hook knife, IT knife-2, or dual knife. The detailed methods used for endoscopic resection were described in our institution’s previous paper.9
2) Radiotherapy
The total dose was determined by a radiation oncologist considering the patient’s condition (e.g., age, performance status, and coexisting disease) and tolerance dose of adjacent normal tissues (e.g., spinal cord, lungs, and heart). RT was delivered 5 days a week at 1.8 to 2.1 Gy (median, 1.8 Gy) per fraction. The total dose ranged from 50.4 to 64.0 Gy (median, 63.0 Gy) in 28 to 35 fractions. The gross tumor volume (GTV) was defined for primary and nodal disease and was drawn on axial planning CT images using the EUS-derived ab oral distances and EUS/CT findings as references. The clinical target volume (CTV) was defined as the GTV plus a 3 to 5 cm margin in the longitudinal direction and a 0.5 to 1 cm margin in the lateral and anteroposterior directions. The planning target volume was defined as the CTV plus an additional 0.3 to 0.5 cm margin to account for setup error and internal organ motion. The RT modality (3D conformal RT or IMRT) was selected considering the tumor location and temporal changes. The proportion of patients who underwent 3D conformal RT was 82.9%, and that of patients who underwent IMRT was 17.1%.
Rather than RT alone, concurrent chemoradiotherapy (CCRT) was performed for patients with a good general condition. For CCRT, two courses of chemotherapy were administered during RT on days 1 to 5 at 3-week intervals. Chemotherapy consisted of 5-fluorouracil (700 mg/m2 of body surface area per day by 120-hour infusion) and cisplatin (15 mg/m2 of body surface area per day intravenously).
3) Esophagectomy
For patients with middle and lower esophageal cancers, a two-field lymph node dissection and intrathoracic esophagogastrostomy were performed using the whole stomach as a conduit, and anastomosis was performed with a 28-mm end-to-end anastomosis stapler. For patients with upper esophageal cancer, three-field lymph node dissection was routinely performed. If intrathoracic esophagogastrostomy was possible, intrathoracic anastomosis was performed using the entire stomach. For all others, cervical esophagogastrostomy with a gastric tube was performed. The stomach was positioned in the posterior mediastinum. The detailed methods are available in another previous report.10
4) Salvage treatment
At a multidisciplinary clinic, the optimal treatment was discussed. Reirradiation was also considered when the interval from previous RT was long and the radiation doses were acceptable for the adjacent normal organs. In contrast, surgical resection was considered when reirradiation was thought to be difficult or pathological confirmation was required. Only local or regional recurrence was considered for salvage treatment. However, systemic chemotherapy was performed after local treatment if distant metastasis occurred.
3. Evaluation of response
Patients who underwent endoscopic therapy or RT were followed using endoscopy with biopsies, CT, and EUS every 6 months after treatment for 3 years and every year thereafter. The opinion of the experienced physicians who performed the endoscopy was also important for discriminating between true recurrence and treatment-related changes. A complete response (CR) was defined when no residual tumor findings were seen in any examination. A partial response (PR) was defined when residual tumor findings were identified by all examinations.
Patients who underwent esophagectomy were followed using CT every 6 months after treatment for 3 years and every year thereafter. An endoscopic evaluation was performed every year and when abnormalities were seen on follow-up CT.
Patterns of failure were classified according to the recurrence sites, as follows. Local recurrence was defined as recurrence in the esophagus, and regional recurrence was defined as recurrence in regional nodes. Distant recurrence was defined as recurrence at any other site.
4. Statistical analysis
Overall survival (OS) was defined as the time from initial diagnosis to death from any cause or the last follow-up. Recurrence- free survival (RFS) was measured from the initial diagnosis to any recurrence or the last follow-up. Both the OS and RFS rates were calculated using the Kaplan-Meier method, and the significance of differences in survival was assessed using the log-rank test. The Cox regression was used for the identification of independent prognostic factors by multivariate analysis. Furthermore, the chi-square test was used to compare categorical variables, and the t-test was used to compare continuous variables. A p-value <0.05 was considered statistically significant. SPSS version 23.0 (IBM Corp., Armonk, NY, USA) was used for all statistical analyses.
RESULTS
1. Patients
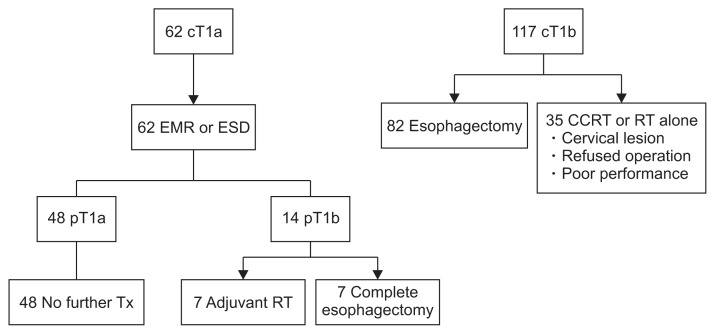
Among the 179 patients who met the inclusion criteria in this study, 62 were initially diagnosed with cT1a esophageal cancer, and the rest were diagnosed with cT1b esophageal cancer. As shown in Fig. 1, 62 patients first underwent endoscopic resection (28 by EMR, 34 by ESD). Further treatments were performed according to the pathological results of subsequent endoscopic tests. A total of 14 patients were finally diagnosed with pT1b esophageal cancer; half of them received adjuvant RT, and the rest underwent complete esophagectomy. Among 117 patients with cT1b esophageal cancer, 82 underwent esophagectomy and 35 received CCRT (n=25) or RT alone (n=10).
Fig. 1.
Treatment flow of all patients (n=179).
cT, clinical T stage; EMR, endoscopic mucosal resection; ESD, endoscopic submucosal resection; CCRT, concurrent chemoradiotherapy; RT, radiation therapy; pT, pathologic T stage; Tx, therapy.
The baseline characteristics and cancer-related features of the four groups (EMR/ESD, esophagectomy, CCRT, and RT alone) are summarized in Table 1. There was a significant difference in age and tumor location according to the treatment method (p=0.003 and p<0.001, respectively). The median patient age (years) was 68 in the endoscopy group, 64 in the esophagectomy group, 66 in the CCRT group, and 75 in the RT alone group. The median patient age was significantly higher in the RT alone group than in the other groups. There were significantly more tumors in the cervical or upper thoracic area in the CCRT group than in the other groups. Because performing laryngopharyn-goesophagectomy for cervically located tumors might subsequently reduce the quality of life, we selected CCRT or RT alone for patients with these lesions. Otherwise, no other significant differences were found in the patient characteristics.
Table 1.
Patient Characteristics
| Variable | EMR/ESD | Esophagectomy | CCRT | RT alone | p-value |
|---|---|---|---|---|---|
| Sex | 0.607 | ||||
| Male | 57 (92) | 75 (92) | 25 (100) | 10 (100) | |
| Female | 5 (8) | 7 (8) | 0 | 0 | |
| Age, yr | 68 (44–82) | 64 (50–86) | 66 (55–85) | 75 (65–82) | 0.003 |
| Histology | 0.834 | ||||
| SCCa | 61 (98) | 81 (99) | 25 (100) | 10 (100) | |
| Adenosquamous | 1 (2) | 1 (1) | 0 | 0 | |
| cT stage | <0.001 | ||||
| cT1a | 62 (100) | 0 | 0 | 0 | |
| cT1b | 0 | 82 (100) | 25 (100) | 10 (100) | |
| Tumor location | <0.001 | ||||
| Cervical | 0 | 0 | 5 (20) | 1 (10) | |
| Upper thoracic | 6 (10) | 10 (12) | 8 (32) | 0 | |
| Middle thoracic | 17 (27) | 34 (42) | 4 (16) | 3 (30) | |
| Lower thoracic | 39 (63) | 38 (46) | 8 (32) | 6 (60) |
Data are presented as number (%) or median (range).
EMR, endoscopic mucosal resection; ESD, endoscopic submucosal dissection; CCRT, concurrent chemoradiotherapy; RT, radiotherapy; SCCa, squamous cell carcinoma; cT stage, clinical T stage.
2. Treatment outcomes
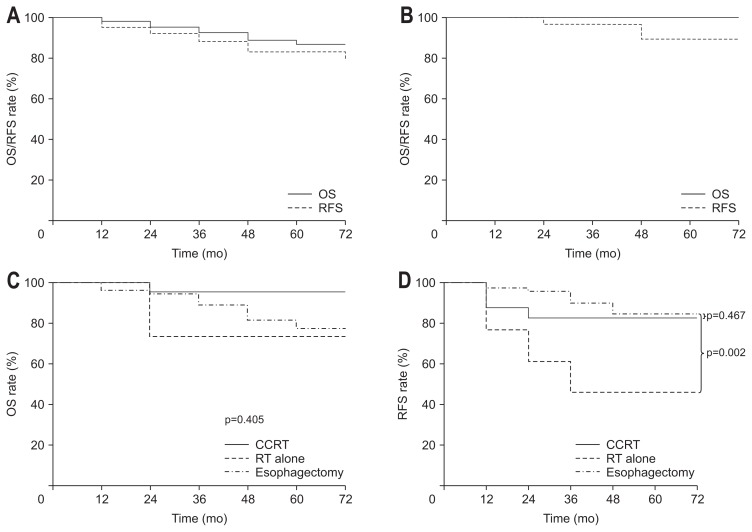
The median follow-up period among all patients was 32 months (range, 1 to 120 months). The 5-year OS and RFS rates of all patients were 86% and 77%, respectively (Fig. 2A). All patients with T1a lesions underwent endoscopic therapy (EMR/ESD). Except for the 14 patients who were diagnosed with pathological T1b disease through endoscopic therapy, the 5-year OS and RFS rates in the EMR/ESD group were 100% and 85%, respectively (Fig. 2B). Patients diagnosed with T1b disease were excluded from the outcome analysis of the EMR/ESD group because they received salvage treatment. Patients with T1b lesions underwent esophagectomy, CCRT, or RT. The 5-year OS and RFS rates were 78% and 77%, respectively, in the esophagectomy group, 96% and 80%, respectively, in the CCRT group, and 80% and 44%, respectively, in the RT alone group (Fig. 2C and D). There was no significant difference in OS according to the treatment modality in patients with T1b disease (p=0.405). The RFS rate was significantly higher in the esophagectomy group than in the RT alone group (p=0.04). There was no significant difference in RFS between the esophagectomy and CCRT groups (p=0.922). There were 35 patients with pathologically diagnosed T1a disease after surgery. Excluding these patients, the esophagectomy group had a 5-year OS rate of 63% and a 5-year RFS rate of 66%. At this time, no significant differences were found in OS or RFS among the esophagectomy, CCRT, and RT alone groups. Regarding OS, age, T stage, and treatment modality were significant factors in both the univariate and multivariate analyses (p=0.007, p=0.056, and p=0.026, respectively). Regarding RFS, there were no significant factors in the univariate or multivariate analysis (Supplementary Table 1).
Fig. 2.
Comparison of Kaplan-Meier overall survival (OS) curves; (A) OS and recurrence-free survival (RFS) of all patients, (B) OS and RFS of patients with cT1a lesions after endoscopic resection, (C) OS and (D) RFS of patients with cT1b lesions according to the treatment group.
CCRT, concurrent chemoradiotherapy; RT, radiotherapy.
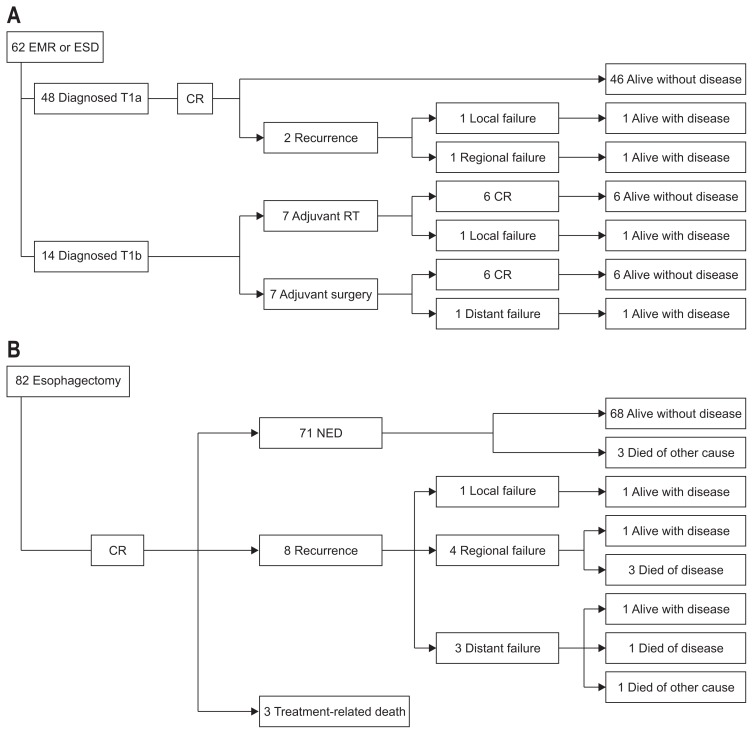
In the EMR/ESD group (n=62), 48 patients were diagnosed with pT1a disease and 14 patients were diagnosed with pT1b disease. Of the 48 patients diagnosed with pT1a disease, 46 are alive without disease after no adjuvant treatment, and two experienced local or regional recurrence. Seven of 14 patients with pT1b disease underwent adjuvant RT, and seven underwent adjuvant surgery sequentially. Most patients, except the two with recurrence, have maintained CR and are alive without recurrence (Fig. 3A).
Fig. 3.
Flow of treatment outcomes for each treatment group. Outcome for endoscopic therapy group (A), esophagectomy group (B), concurrent chemoradiotherapy (CCRT) group (C), radiotherapy (RT) group (D).
EMR, endoscopic mucosal resection; ESD, endoscopic submucosal resection; CR, complete response; NED, no evidence of disease; PR, partial response.
In the esophagectomy group (n=82), all esophageal cancers were completely removed through surgery. However, three patients died from treatment-related causes (sepsis after pneumonia or esophageal perforation). Otherwise, 71 patients are alive without any recurrence. Eight patients experienced recurrence (local=1, regional=4, and distant=3), and half of them died after disease progression (Fig. 3B).
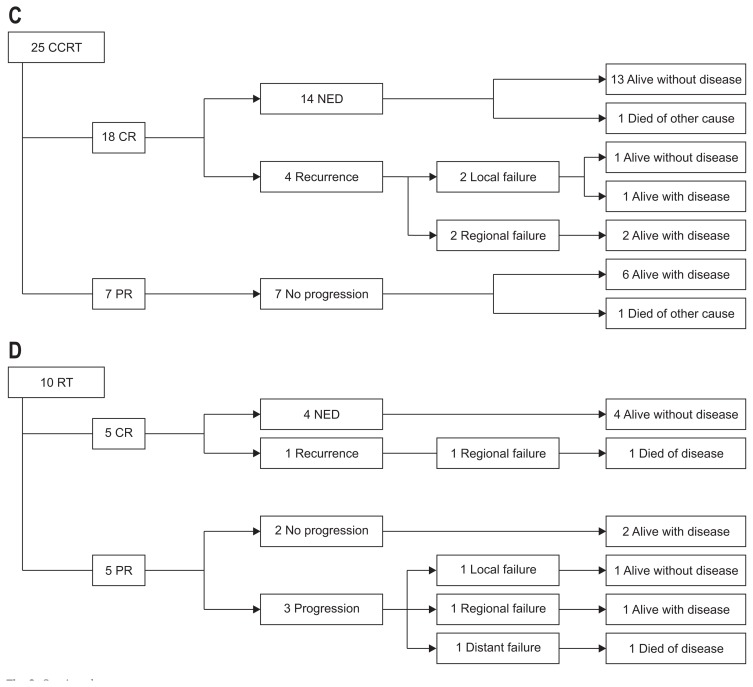
In the CCRT group (n=25), 18 patients had CR, and seven had PR. Of the patients with CR, all are alive without disease, except one who died of another medical cause. Of the patients with PR, none showed disease progression, and all are alive even without any additional treatment, except one who died of another medical cause (Fig. 3C).
In the RT alone group (n=10), half of the patients had CR, and the other half had PR. Of the patients with CR, only one developed regional recurrence, and the rest are alive with or without the disease. Of the patients with PR, three patients experienced recurrence (local=1, regional=1, and distant=1), and the patient with distant recurrence died of the disease (Fig. 3D). An overview of the outcomes in each treatment subgroup is shown in Fig. 3.
3. Patterns of failure
Table 2 lists the site of the first recurrence for patients in the four subgroups. Twenty of the 179 patients experienced recurrence. The group with the highest rate of recurrence was the RT alone group (7% in the EMR/ESD group, 9% in the esophagectomy group, 16% in the CCRT group, and 40% in the RT alone group). In the EMR/ESD group, although the overall rate of recurrence was low (n=4), local recurrence occurred in three patients (75%), and regional recurrence occurred in one patient (25%). In the esophagectomy group, regional recurrence was the most frequent recurrence pattern (50%), followed by distant (38%) and local recurrence (12%). In the CCRT group, half of the overall instances of recurrence were local, and the rest were regional. There were no instances of distant recurrence after CCRT. In the RT alone group, the regional recurrence rate was the most frequent, at 50%, followed by local (25%) and distant recurrence (25%).
Table 2.
Patterns of Failure According to the Treatment Group
| Type | EMR/ESD | Esophagectomy | CCRT | RT alone |
|---|---|---|---|---|
| Local recurrence | 3 (5) | 1 (1) | 2 (8) | 1 (10) |
| Regional recurrence | 1 (2) | 4 (5) | 2 (8) | 2 (20) |
| Distant metastasis | 0 | 3 (4) | 0 | 1 (10) |
| Total | 4 (7) | 8 (9) | 4 (16) | 4 (40) |
Data are presented as number (%).
EMR, endoscopic mucosal resection; ESD, endoscopic submucosal resection; CCRT, concurrent chemoradiotherapy; RT, radiation therapy.
4. Complications
Table 3 lists the complications of patients in the three subgroups, which included only T1b lesions. The overall rates of acute complications were 34%, 52%, and 50% in the esophagectomy, CCRT, and RT alone groups, respectively. The most common acute complications were esophageal stricture (17%) in the esophagectomy group, esophagitis (52%) in the CCRT group, and esophagitis (40%) in the RT alone group. Grade 4 or higher complications only developed in four patients (3%) in the esophagectomy group. Three patients had pneumonia at a median of 1 month after surgery, and one patient experienced perforation 1 week after surgery. Three patients died after these complications. Otherwise, all complications were grade 2, except two complications in the CCRT group and four complications in the RT alone group (all manageable esophagitis during RT). Late complications were only found in the esophagectomy group (24%), most of which were esophageal stricture, dysphagia, or regurgitation.
Table 3.
Acute and Late Complication Rates According to Treatment Group in Patients with T1b Lesions
| Complication | Esophagectomy | CCRT | RT alone |
|---|---|---|---|
| Acute | |||
| Esophagitis | |||
| Grade 2 | 0 | 11 (44) | 0 |
| Grade 3 | 0 | 2 (8) | 4 (40) |
| Grade 4 or 5 | 0 | 0 | 0 |
| Esophageal stricture | |||
| Grade 2 | 1 (1) | 0 | 0 |
| Grade 3 | 13 (16) | 0 | 0 |
| Grade 4 or 5 | 0 | 0 | 0 |
| Pneumonia | |||
| Grade 2 | 0 | 0 | 0 |
| Grade 3 | 2 (2) | 0 | 0 |
| Grade 4 or 5 | 3 (4) | 0 | 0 |
| Other complication | |||
| Grade 2 | 2 (2) | 0 | 1 (10) |
| Grade 3 | 6 (7) | 0 | 0 |
| Grade 4 or 5 | 1 (1) | 0 | 0 |
| Late | |||
| Esophagitis | |||
| Grade 2 | 0 | 0 | 0 |
| Grade 3 | 0 | 0 | 0 |
| Grade 4 or 5 | 0 | 0 | 0 |
| Esophageal stricture | |||
| Grade 2 | 5 (6) | 0 | 0 |
| Grade 3 | 10 (12) | 0 | 0 |
| Grade 4 or 5 | 0 | 0 | 0 |
| Pneumonia | |||
| Grade 2 | 0 | 0 | 0 |
| Grade 3 | 0 | 0 | 0 |
| Grade 4 or 5 | 0 | 0 | 0 |
| Other complication | |||
| Grade 2 | 4 (5) | 0 | 0 |
| Grade 3 | 1 (1) | 0 | 0 |
| Grade 4 or 5 | 0 | 0 | 0 |
Data are presented as number (%).
CCRT, concurrent chemoradiotherapy; RT, radiation therapy.
DISCUSSION
In this study, we compared the outcomes of endoscopic therapy, esophagectomy, CCRT, and RT in patients with early-stage esophageal cancer. First, patients with T1a esophageal cancer had a good prognosis with endoscopic eradication therapies. Second, esophagectomy did not improve OS compared with other noninvasive therapies in patients with T1b esophageal cancer. Esophagectomy prolonged RFS significantly compared with RT alone; however, there was no notable difference compared with CCRT. The outcomes of CCRT and RT should be assessed further, given that more patients of an advanced age or with a poor performance status are treated with either CCRT or RT rather than surgery.
Traditionally, the gold standard approach to the management of early-stage esophageal cancer has been esophagectomy, either alone or in combination with chemotherapy and/or RT. Although shown to improve survival, this approach has been associated with significant risks, such as surgical complications (e.g., anastomotic leak, infection, pneumonia, or stenosis) and even mortality. Recent efforts to improve the outcomes of early-stage esophageal cancer treatment, the primary goal of which is cure, have focused on the development of less invasive endoscopic treatments designed to destroy or remove cancerous lesions in the esophageal wall while sparing healthy tissue. These treatments have been used either alone or in combination with other techniques without compromising oncologic outcomes. As in our results, grade 4 or higher complications, including pneumonia and esophageal perforation, have only been observed in the esophagectomy group, and no severe late complications have appeared in patients undergoing CCRT or RT.
Currently, the National Cancer Institute recommends surgery as the standard treatment for T1b esophageal cancer, and this recommendation is based on the observation that T1b esophageal squamous cell carcinoma has a high incidence of lymph node metastasis.11 In our study, the patterns of failure data showed that the overall recurrence rate in patients with T1a disease was as low as 7%. In the esophagectomy group, the local recurrence rate was 1%, the regional recurrence rate was 5%, and the distant metastasis rate was 4%, which means that surgical resection is good for local control. In the CCRT group, the local and regional recurrence rates were both 8%, with no distant metastasis. In the RT alone group, the regional recurrence rate was as high as 20%, and the local and distant metastasis rates were both 10%. Comparing the CCRT group with the RT alone group, the absence of distant metastasis in the CCRT group indicates that the combination of chemotherapy with RT might be effective enough for systemic control. Considering that patients with T1b lesions had a regional recurrence rate of 7.6%, we recommend including the regional lymph nodes in the RT field, rather than simply irradiating the primary mass. Chemotherapy for systemic control should also be considered.
In patients with T1a disease, the 5-year OS and RFS rates were 100% and 85%, respectively. Among patients with T1b disease, there was no significant difference in outcome between patients who underwent esophagectomy and those who underwent CCRT. This suggests that CCRT is sufficiently comparable to esophagectomy even after considering possible selection bias, which may have occurred because more patients with a poor condition were included in the CCRT or RT alone group.
One previous study demonstrated the superior outcome of esophagectomy in patients with cT1 esophageal cancer.11 Matsumoto et al.11 compared the outcomes of 29 patients with radical esophagectomy and 38 patients with definitive RT. Although there was no difference in the OS rate, the RFS rate was significantly higher in the surgery group than in the radiation group (5-year RFS 62% vs 39%, p=0.005). In particular, a significantly higher RFS rate was achieved in patients with tumors that invaded the submucosa. However, before concluding that surgery is the most appropriate treatment for cT1 esophageal cancer, it should be noted that the authors did not distinguish patients who underwent RT only from patients who underwent CCRT. Considering that the RT alone group showed an inferior result in our paper, the results of Matsumoto’s study are considered to be similar to the results of our study. The outcome of CCRT was superior to that of RT alone in esophageal cancer according to a previous prospective randomized trial.12 Therefore, we propose that CCRT should be in an equal position rather than considered a simple alternative treatment, based on the results of those studies and other favorable data supporting RT.13,14
After esophagectomy, 35 patients were diagnosed with pathological T1a disease, but not T1b. When the outcome of the esophagectomy group was excluded except for these patients, it showed a poorer outcome than before, which is probably to be expected. This outcome was not statistically significant when compared with those of the other groups. In other words, the significant differences observed in RFS for the RT alone group disappeared and showed an inferior tendency, although not significantly so, compared with the CCRT group. However, in the CCRT and RT alone groups, T1b disease was diagnosed using EUS, but there were actually patients with T1a disease in the group. Therefore, it is difficult to conclude that esophagectomy is inferior to CCRT.
When we performed multivariate analysis in this study, the factors significantly associated with OS were age, T stage, and treatment modality. T stage has been reported as a significant factor in several studies;1,15 treatment modality could also be a significant factor because there was a difference in the baseline characteristics according to treatment modality, which was related to T stage. Conversely, there might be no significant difference in OS according to treatment modality when patients with the same stage disease are analyzed. Other studies have shown that T stage, age, facility, and follow-up frequency in early-stage esophageal cancer are significantly associated with outcome.15,16 Most of these studies were similar to ours; this means that our cohort can be a representative cohort of early-stage esophageal cancer patients. Moreover, our study was a single-institution study with no differences in follow-up schedules or facilities among the treatment groups.
There are several limitations to this study. First, there could be a selection bias because of the retrospective nature of this study. Although the main purpose of this study was to compare each treatment modality in early-stage esophageal cancer, there was a trend to treat patients with T1b disease with esophagectomy unless the patient refused surgery. Definitive CCRT was performed because of the difficulty of surgery when the lesion was located at a cervical level. Furthermore, if the patient had any medical comorbidities or a poor performance status, the physicians decided to perform CCRT or RT. Physicians’ preferences or differences in patient characteristics could affect the treatment outcomes unexpectedly. Second, this study could not analyze the effect of RT technique development on the treatment outcome. Recently, our institution has been treating many patients with esophageal cancer using IMRT. This study included patients from the past, most of whom were treated with 3-dimensional (3D)-conformal radiation therapy (3D-conformal radiation therapy 82.8% vs IMRT 17.2%). We could not find any difference according to RT modality in this small group of patients. Lastly, we performed response evaluation using biopsy for patients who underwent definitive CCRT or RT. However, because endoscopic biopsy after CCRT/RT sometimes shows low sensitivity/specificity in the detection of recurring carcinoma, this may have caused some bias in the outcomes.
Nevertheless, this study has strength in that it analyzed the outcome of patients with early esophageal cancer who received treatment with various modalities based on the medical records of a single institution. Patients were included in this study according to stricter eligibility criteria than those of previous studies, which in turn enhanced the homogeneity of this study. In our institution, many patients with esophageal cancer are referred to a multidisciplinary clinic, where the optimal treatment based on institutional policy is selected. In addition, unlike previous studies, a more precise comparative study could be performed by comparing the patient subgroups. To overcome the limitations caused by the small number of patients receiving each treatment and perform a more structured comparative study, the Korean Radiation Oncology Group plans to perform a multi-institutional retrospective study on the optimal treatment policy for early-stage esophageal cancer.
In conclusion, we assessed the clinical outcomes of clinical T1N0M0 esophageal cancer patients according to each different treatment modality. In patients with T1a disease, endoscopic therapy was found to be a satisfactory treatment. In patients with T1b disease, CCRT and esophagectomy showed a comparably good outcome, although RT alone showed an inferior outcome compared with the modalities. However, the esophagectomy group included some patients with ≥ grade 4 complications and included patients with more favorable conditions than those in the other groups, which might have caused selection bias. Considering those results, CCRT should be considered an alternative to esophagectomy for the treatment of patients with T1b disease. To clarify our findings, further studies with a randomized controlled design or larger cohort are warranted.
Supplementary Material
Footnotes
CONFLICTS OF INTEREST
No potential conflict of interest relevant to this article was reported.
REFERENCES
- 1.Zeng Y, Liang W, Liu J, He J. Endoscopic treatment versus esophagectomy for early-stage esophageal cancer: a population-based study using propensity score matching. J Gastrointest Surg. 2017;21:1977–1983. doi: 10.1007/s11605-017-3563-2. [DOI] [PubMed] [Google Scholar]
- 2.Pennathur A, Farkas A, Krasinskas AM, et al. Esophagectomy for T1 esophageal cancer: outcomes in 100 patients and implications for endoscopic therapy. Ann Thorac Surg. 2009;87:1048–1054. doi: 10.1016/j.athoracsur.2008.12.060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Enzinger PC, Mayer RJ. Esophageal cancer. N Engl J Med. 2003;349:2241–2252. doi: 10.1056/NEJMra035010. [DOI] [PubMed] [Google Scholar]
- 4.Rice TW, Rusch VW, Ishwaran H, Blackstone EH Worldwide Esophageal Cancer Collaboration. Cancer of the esophagus and esophagogastric junction: data-driven staging for the seventh edition of the American Joint Committee on Cancer/International Union Against Cancer Cancer Staging Manuals. Cancer. 2010;116:3763–3773. doi: 10.1002/cncr.25146. [DOI] [PubMed] [Google Scholar]
- 5.Wang S, Huang Y, Xie J, et al. Does delayed esophagectomy after endoscopic resection affect outcomes in patients with stage T1 esophageal cancer? A propensity score-based analysis. Surg Endosc. 2018;32:1441–1448. doi: 10.1007/s00464-017-5830-4. [DOI] [PubMed] [Google Scholar]
- 6.Rice TW, Blackstone EH, Goldblum JR, et al. Superficial adenocarcinoma of the esophagus. J Thorac Cardiovasc Surg. 2001;122:1077–1090. doi: 10.1067/mtc.2001.113749. [DOI] [PubMed] [Google Scholar]
- 7.Pennathur A, Landreneau RJ, Luketich JD. Surgical aspects of the patient with high-grade dysplasia. Semin Thorac Cardiovasc Surg. 2005;17:326–332. doi: 10.1053/j.semtcvs.2005.10.003. [DOI] [PubMed] [Google Scholar]
- 8.Cao C, Luo J, Gao L, et al. Definitive radiotherapy for cervical esophageal cancer. Head Neck. 2015;37:151–155. doi: 10.1002/hed.23572. [DOI] [PubMed] [Google Scholar]
- 9.Lee HJ, Lee H, Park JC, Shin SK, Lee SK, Lee YC. Treatment strategy after endoscopic resection of superficial esophageal squamous cell carcinoma: a single institution experience. Gut Liver. 2015;9:714–719. doi: 10.5009/gnl14142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kim DJ, Park SY, Lee S, Kim HI, Hyung WJ. Feasibility of a robot-assisted thoracoscopic lymphadenectomy along the recurrent laryngeal nerves in radical esophagectomy for esophageal squamous carcinoma. Surg Endosc. 2014;28:1866–1873. doi: 10.1007/s00464-013-3406-5. [DOI] [PubMed] [Google Scholar]
- 11.Matsumoto S, Takayama T, Tamamoto T, et al. A comparison of surgery and radiation therapy for cT1 esophageal squamous cell carcinoma. Dis Esophagus. 2011;24:411–417. doi: 10.1111/j.1442-2050.2010.01163.x. [DOI] [PubMed] [Google Scholar]
- 12.Herskovic A, Martz K, al-Sarraf M, et al. Combined chemotherapy and radiotherapy compared with radiotherapy alone in patients with cancer of the esophagus. N Engl J Med. 1992;326:1593–1598. doi: 10.1056/NEJM199206113262403. [DOI] [PubMed] [Google Scholar]
- 13.Nemoto K, Yamada S, Hareyama M, Nagakura H, Hirokawa Y. Radiation therapy for superficial esophageal cancer: a comparison of radiotherapy methods. Int J Radiat Oncol Biol Phys. 2001;50:639–644. doi: 10.1016/S0360-3016(01)01481-X. [DOI] [PubMed] [Google Scholar]
- 14.Sasaki T, Nakamura K, Shioyama Y, et al. Treatment outcomes of radiotherapy for patients with stage I esophageal cancer: a single institute experience. Am J Clin Oncol. 2007;30:514–519. doi: 10.1097/COC.0b013e31805c1410. [DOI] [PubMed] [Google Scholar]
- 15.Wani S, Drahos J, Cook MB, et al. Comparison of endoscopic therapies and surgical resection in patients with early esophageal cancer: a population-based study. Gastrointest Endosc. 2014;79:224–232.e1. doi: 10.1016/j.gie.2013.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Moreno AC, Verma V, Hofstetter WL, Lin SH. Patterns of care and treatment outcomes of elderly patients with stage I esophageal cancer: analysis of the national cancer data base. J Thorac Oncol. 2017;12:1152–1160. doi: 10.1016/j.jtho.2017.04.004. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.