Abstract
Cardio-oncology has organically developed as a new discipline within cardiovascular medicine as a result of the cardiac and vascular adverse sequelae of the major advances in cancer treatment. Patients with cancer and cancer survivors are at increased risk of vascular disease for a number of reasons. First, many new cancer therapies, including several targeted therapies, are associated with vascular and metabolic complications. Second, cancer itself serves as a risk factor for vascular disease, especially by increasing the risk for thromboembolic events. Finally, recent data suggest that common modifiable and genetic risk factors predispose to both malignancies and cardiovascular disease. Vascular complications in patients with cancer represent a new challenge for the clinician and a new frontier for research and investigation. Indeed, vascular sequelae of novel targeted therapies may provide insights into vascular signaling in humans. Clinically, emerging challenges are best addressed by a multidisciplinary approach in which cardiovascular medicine specialists and vascular biologists work closely with oncologists in the care of patients with cancer and cancer survivors. This novel approach realizes the goal of providing superior care through the creation of cardio-oncology consultative services and the training of a new generation of cardiovascular specialists with a broad understanding of cancer treatments.
Keywords: AHA Scientific Statements, cardiovascular diseases, medical oncology, therapeutics
The vascular and metabolic adverse effects of cancer and cancer therapies have spurred the growth of cardio-oncology as a field. Unlike the left ventricular (LV) dysfunction associated with some of the early chemotherapies in oncology,1 vascular effects are diverse and less well characterized. In this document, we focus on vascular cardio-oncology as a new and significant research and clinical dimension in cardio-oncology. Whereas traditional cancer treatments have been associated with vascular complications in an often-unpredictable fashion, new cancer therapies frequently target the interaction between cancer and the endothelium and may result in predictable vascular and metabolic sequelae. Because of the more clearly defined mechanisms of action, these novel targeted oncology therapies can introduce new paradigms in vascular biology.2 At the same time, the intersection of cancer and cardiovascular disease (CVD) extends beyond pharmacology. New data suggest that common risk factors, including genetic factors, can underlie the pathogenesis of cancer and CVD, a paradigm that can have significant public health implications, especially for the >16 million Americans who are cancer survivors.3,4 In this document, we highlight these new paradigms in the field of cardio-oncology and bring to the forefront the many unanswered questions and future directions.
VASCULAR COMPLICATIONS OF TRADITIONAL THERAPIES
Despite the advent of targeted cancer agents and immunotherapies, traditional cytotoxic chemotherapies and radiation therapy (RT) remain the cornerstone of many treatment protocols. Vascular complications of these traditional cancer therapies are explored in this section (Table 1).
Table 1.
Summary of Main Vascular Toxicities Associated With Traditional Cancer Therapies and Proposed Mechanisms for Toxicities
| Cancer Therapy | Proposed Mechanisms of Vascular Toxicity | Vascular Toxicities* |
|---|---|---|
| Antimetabolites: fluoropyrimidines | Endothelial
injury Vasospasm Increased endothelin-1 bioactivity |
Coronary vasospasm Raynaud phenomenon |
| Antimicrotubule agents: taxanes | Interference with basic endothelial cell functions by affecting the cytoskeleton | Capillary leak Peripheral neuropathy |
| Antimicrotubule agents: vinca alkaloids (vincristine, vinblastine) | Caspase-mediated apoptosis Inhibition of endothelial cell proliferation |
Chest pain
presentations Hypertension Myocardial ischemia Raynaud phenomenon Thromboembolism |
| Alkyl-like agents: platinum compounds | Injury to endothelial cells Increased platelet aggregation Reduced NO availability |
Cerebrovascular
events Hypertension Myocardial ischemia/MI Raynaud phenomenon Venous thromboembolic disease |
| Alkylating agents: cyclophosphamide | Injury to endothelial cells Increased platelet aggregation Decreased angiotensin-converting enzyme activity |
Cerebrovascular events Hepatic veno-occlusive disease Hypertension Myocardial ischemia/MI Pulmonary hypertension Raynaud phenomenon |
| Antitumor antibiotics: anthracycline | Production of reactive oxygen
species DNA double-stranded breaks Mitochondrial dysfunction Injury to endothelial cells |
Endothelial dysfunction |
| Antitumor antibiotics: bleomycin | Inhibition of endothelial cell
proliferation/migration Endothelial cell apoptosis |
Myocardial ischemia/MI Pulmonary hypertension Raynaud phenomenon |
| Other older therapies: IL-2 | Cytotoxic effects by lymphokine-activated
killer cells Direct effects of IL-2 on endothelial cells Induction of inflammatory cytokines |
Vascular leak syndrome |
IL indicates interleukin; MI, myocardial infarction; and NO, nitric oxide.
Vascular toxicities are presented in alphabetical order. Order does not reflect prevalence of respective toxicities.
Antimetabolites
The fluoropyrimidines are important antimetabolites and include 5-fluorouracil (5-FU) and its oral prodrug, capecitabine. These agents are used in the treatment of gastrointestinal, breast, and head and neck tumors. Fluoropyrimidines may cause myocardial ischemia by inducing coronary artery spasm, which may occur in the absence of angiographic coronary artery disease (CAD).5 Multiple mechanisms have been reported to underlie vasospasm, including endothelial cell damage with cytolysis and denudation, as well as increased endothelin-1 bioactivity, leading to enhanced contractility of vascular smooth muscle cells and vasoconstriction.6–8 The incidence of coronary vasospasm varies by agent and schedule of administration. When high-dose 5-FU–based chemotherapy was given as a continuous intravenous infusion, events consistent with coronary vasospasm (angina, arrhythmia, or sudden death) were reported in up to 5.4% of patients.9 In a prospective cohort, short-term 5-FU and leucovorin administration was associated with the occurrence of cardiac-related events in 2.4% of patients.10 Vascular toxicity of 5-FU is observed predominantly within 72 hours of the first cycle.11 In a retrospective analysis from the Dutch Colorectal Cancer Group, ischemia/infarction was observed in 2.9% of the patients treated with capecitabine, and the highest incidence of cardiac events was observed in patients treated with capecitabine combined with oxaliplatin and bevacizumab.12 Symptoms are generally reversible after cessation of the fluoropyrimidine and with the administration of vasodilators. However, despite the reversibility of vasospasm, death can occur.11 There is a high risk of relapse with fluoropyrimidine rechallenge,13 and relapse is associated with higher mortality.11 Therefore, rechallenge should be reserved for those without reasonable alternative cancer therapies and should occur only in the context of informed consent, aspirin therapy, vasodilator therapy with L-type calcium channel blockers or nitrates or both, continuous electrocardiographic monitoring (ideally in a coronary care unit), and bolus, rather than continuous, 5-FU infusion.14,15 Of note, Clasen and colleagues16 recently reported that cardioprotective pretreatment with 2 calcium channel blockers (nifedipine and diltiazem) and long-acting isosorbide mononitrate allowed successful rechallenge with bolus intravenous 5-FU or oral capecitabine in 11 patients.
Antimicrotubule Agents (Taxanes and Vinca Alkaloids)
Taxanes cause mitotic arrest and activate caspase-dependent apoptosis through microtubule destabilization.17 Direct effects on endothelial and smooth muscle cells, which may occur at concentrations below those inducing cytotoxicity, lead to antiangiogenic effects or vascular disruption.18–20 Clinical manifestations of vascular toxicity induced by taxanes include peripheral neuropathy mediated by damage to the vasa nervorum,21 capillary hyperpermeability with fluid retention,22 and myocardial ischemia.23 The incidence and severity of these vascular toxicities exhibit a dose-response relationship.24
Vinca Alkaloids
Reported vascular toxicities after exposure to vincristine alone or in combination with other drugs include chest pain,25 myocardial infarction (MI),26,27 hypertension,28 Raynaud phenomenon,25,29 and thromboembolism.30 Caspase-mediated apoptosis and inhibition of endothelial cell proliferation are implicated in the pathogenesis of these toxicities.24
Alkyl-Like and Alkylating Agents
The platinum compounds such as cisplatin are alkyl-like agents that are associated with Raynaud phenomenon, hypertension, MI, stroke, arterial thrombosis, acute limb ischemia, deep vein thrombosis (DVT), and pulmonary embolism.31–34 Furthermore, chest pain has been reported in as many as 38% of patients with testicular cancer treated with cisplatin in combination with vinca alkaloids and bleomycin.25 These adverse events are likely the result of direct toxic effects on endothelial cells, platelet activation, and decreased nitric oxide availability.35,36 The alkylating agent cyclophosphamide is associated with a comparable range of vascular adverse effects mediated by similar mechanisms, particularly when used at high doses before bone marrow or stem cell transplantation.37–39
Antitumor Antibiotics
Anthracyclines are cytotoxic antibiotics used for a variety of hematologic and solid malignancies. The risk of anthracycline-induced cardiomyopathy is well recognized and follows a dose-response relationship.40–42 In addition to direct cardiomyocyte toxicity through DNA double-stranded breaks (via topoisomerase II), production of reactive oxygen species, and mitochondrial dysfunction,43 anthracyclines may injure the vascular endothelium.44,45 Such endothelial toxicity can occur immediately,46 and endothelial dysfunction can persist for months to years after exposure.45,47 The implications of endothelial dysfunction for the vascular health of anthracycline-treated patients require more investigation, and current clinical practice focuses largely on surveillance for myocardial dysfunction rather than vascular toxicities in this cohort.
Bleomycin, a DNA-damaging chemotherapy drug used in the treatment of lymphomas and head, neck, and testicular cancers, exerts antiangiogenic effects by inhibiting endothelial cell proliferation and migration and by inducing endothelial cell apoptosis.48 Bleomycin, alone or in combination with vinca alkaloids, cisplatin, or etoposide, is associated with Raynaud phenomenon,49 MI,50 and pulmonary hypertension.51 In particular, bleomycin is associated with a 3-fold increased risk of Raynaud phenomenon among testicular cancer survivors, and this risk is dose related.52
Other Agents
Trastuzumab is a humanized monoclonal antibody that targets the extracellular domain of the HER2 (human epidermal growth factor receptor-2) receptor, and its use has improved cancer outcomes in HER2-positive breast cancer.53 The potential for symptomatic or asymptomatic reductions in LV systolic function is well recognized, and serial assessment of LV function throughout treatment is considered standard of care.54,55 In initial trials with trastuzumab, patients were concomitantly treated with anthracyclines, which resulted in considerable risk for cardiomyopathy (up to 27% in an initial trial).53 This risk was attenuated in subsequent trials in which anthracyclines were given before trastuzumab or trastuzumab was used alone without anthracyclines.55,56 Less clear with respect to cardiomyopathy risk is the case of dual HER2 blockade (in which newer compounds such as pertuzumab are combined with trastuzumab), although early data suggest cardiomyopathy signals similar to those with trastuzumab monotherapy.57,58 The HER2 receptor is also expressed in vascular endothelial cells,59 and disruption of the HER2 signaling may contribute to the pathophysiology of myocardial injury.60,61
IL (interleukin)-2 is an immunotherapeutic agent used to treat metastatic melanoma and renal cell carcinoma. Administration of high-dose IL-2 is associated with vascular leak syndrome, a potentially fatal condition of increased vascular permeability in multiple organs leading to pulmonary edema, hypotension, and acute renal and cardiovascular failure.62 The pathogenesis of vascular damage in vascular leak syndrome includes cytotoxic effects by lymphokine-activated killer cells,63,64 direct effects of IL-2 on endothelial cells,65 and induction of inflammatory cytokines such as tumor necrosis factor-α and IL-1.65
Radiation Therapy
More than 50% of patients with cancer receive RT during treatment.66 Vascular structures within the radiation field are vulnerable to injury.67,68 Acute vascular effects of RT include endothelial dysfunction69 and infiltration of inflammatory cells,70 which can lead to persistent inflammation and progressive damage to the microvasculature71 and to conduit arteries.72 In addition, radiation injury to the vasa vasorum can precipitate ischemia of the vessel wall.73,74 Thoracic RT is associated with an increased risk of premature CAD; it has been observed in Hodgkin lymphoma75–77 and breast cancer78,79 survivors. RT to the head and neck confers a significantly increased risk of carotid disease, transient ischemic attack, and ischemic stroke.80,81 RT can also cause large- and medium-vessel vasculopathy, as exemplified by axillary artery stenosis after RT to the axilla in patients with breast cancer and porcelain aorta after mediastinal RT.82 The nonselective damage to any vascular structures included in the irradiated field is highlighted by reports of radiation-induced DVT,83 venous stenosis,84 and renal artery stenosis.85 The total cumulative dose is an important risk factor for RT-mediated vascular injury; a linear radiation dose-response relationship with subsequent risk of CAD has been described.78,86 Risk from RT also increases over time.79,87 Other risk factors for radiation-induced vasculopathy include a higher dose of radiation fractions, young age at time of treatment, concomitant cancer therapies, and superficial location of vessels.76,88 Radiation-induced vasculopathy may occur in the absence of traditional risk factors. Follow-up of patients at risk of radiation-induced vasculopathy should include careful longitudinal assessment for signs and symptoms of vascular disease and optimization of modifiable cardiovascular risk factors.
In addition to vascular damage, cranial, neck, and mediastinal radiation can cause impaired autonomic regulation of the cardiovascular system and can result in labile blood pressure, labile heart rate, and orthostatic intolerance.89–92 In a study that compared 263 Hodgkin lymphoma survivors clinically referred for exercise treadmill testing after a median interval of 19 years (interquartile range, 12–26 years) after mediastinal radiation with 526 matched controls, survivors of mantle radiation had an almost 4 times higher likelihood of elevated resting heart rate and a >5 times higher like-lihood of abnormal heart rate recovery after cessation of exercise in adjusted analyses.91 These autonomic abnormalities increased in prevalence with time from RT and were associated with significant reductions in exercise performance. Furthermore, abnormal heart rate recovery was associated with a 4-fold increased risk of all-cause mortality during follow-up of survivors of mediastinal radiation.
For patients receiving mediastinal RT, some expert groups have proposed that periodic surveillance with functional noninvasive stress testing for CAD detection should be performed starting 5 to 10 years after treatment and continued every 5 years thereafter.88,93 Similarly, ultrasound scanning of the carotid arteries for patients treated with prior neck RT has also been suggested.88,93,94 Management of coronary, carotid, or other vascular disease in the aftermath of RT is based largely on data extrapolated from nonradiation cohorts. Outcomes after percutaneous coronary intervention95 and cardiac surgery96,97 among patients receiving RT are worse compared with those in nonradiation cohorts, and carotid interventions are associated with higher rates of in-stent restenosis.98 However, the reported data are from retrospective, observational, or nonrandomized studies, and randomized controlled trials are needed to investigate outcomes in these patients. Refinements in contemporary radiation protocols that include lower cumulative radiation doses, cardiac shielding, tangential fields, 3-D image–guided treatment planning, and respiratory gating have successfully reduced incidental radiation exposure to cardiovascular structures.99–102 It is hoped that such refinements will reduce subsequent risk of CVD.
VASCULAR COMPLICATIONS WITH TARGETED THERAPIES
The introduction of targeted cancer therapies has significantly augmented the arsenal of treatment options for patients with cancer. By targeting specific signaling pathways that are hijacked by the cancer cell, these therapies have resulted in the introduction of precision medicine in the clinic and in the improvement of patient outcomes. For example, the realization that, in many cancers, kinases become inappropriately active has fostered the development of kinase inhibitors as a therapeutic strategy.103 In particular, small-molecule kinase inhibitors, which may be administered orally, have shown efficacy for multiple cancer types and have dramatically changed the natural history of several malignancies. In chronic myelogenous leukemia (CML), for example, recognition of activation of ABL1 (Abelson murine leukemia viral oncogene homolog 1) kinase driven by a specific chromosomal translocation (the so-called Philadelphia chromosome) allowed specific therapeutic targeting. The introduction of imatinib, the first of several such small-molecule inhibitors, significantly improved outcomes in patients with CML, effectively transforming it into a chronic disease.104,105
In many cases, kinases and their downstream path-ways that are usurped by the cancer cell also play critical roles for vascular and metabolic homeostasis in normal cells.2 Inhibitors of these kinases may cause cardiovascular sequelae, depending on the individual compound and the specific kinase target (Table 2). For example, inhibition of the vascular endothelial growth factor (VEGF) signaling pathway results in hypertension, proteinuria, cardiomyopathy, and vascular disease in a subset of patients.106 Dasatinib, nilotinib, and ponatinib, new-generation ABL1 kinase inhibitors used for the treatment of CML, are associated with pulmonary hypertension (dasatinib), hyperglycemia and atherosclerosis (nilotinib), and hypertension and vascular disease (ponatinib).107 The most concerning vascular toxicities that may occur with the new agents include arterial is-chemic events such as MI, stroke, and limb ischemia, as well as venous thromboembolic (VTE) events.14
Table 2.
Summary of Main Vascular Toxicities Associated With Targeted Therapies and Proposed Mechanisms for Toxicities
| Cancer Therapy | Proposed Mechanisms of Vascular Toxicity | Vascular Toxicities* |
|---|---|---|
| Antibody-related targeted therapies: VEGF-A monoclonal antibody (bevacizumab), VEGF-R2 monoclonal antibody (ramucirumab), VEGF-R1/R2 fused to Fc portion of IgG1 (aflibercept) | Reduction of PI3K-Akt, PLCγ-PKC/IP3,
and Erk-MAPK signaling pathway activity in endothelial cells with
reduction in eNOS activity, NO production, endothelial function, and
cell survival and proliferation (capillary rarefaction) Increase in mitochondrial oxidative stress and eNOS uncoupling, reducing NO bioavailability |
Cerebrovascular events Myocardial ischemia/MI Proteinuria Renal thrombotic microangiopathy Reversible posterior leukoencephalopathy syndrome Systemic hypertension Venous thromboembolic disease |
| Tyrosine kinase–related targeted
therapies: primarily VEGF-R
directed Sorafenib Sunitinib Pazopanib Axitinib Regorafenib Cabozantinib Vandetanib Lenvatinib |
Reduction of PI3K-Akt, PLCγ-PKC/IP3,
and Erk-MAPK signaling pathway activity in endothelial cells with
reduction in eNOS activity, NO production, endothelial function, and
cell survival and proliferation (capillary rarefication) Increase in mitochondrial oxidative stress and eNOS uncoupling, reducing NO bioavailability Increase in endothelin-1 production Rightward shift of the renal pressure–natriuresis curve, impaired sodium excretion, fluid retention, and salt-dependent hypertension Inhibition of PDGFβ-R signaling and pericyte function and survival with reduced VEGF and Ang-1 production and reduced VEGF-R and Tie-2 signaling activity in endothelial cells |
Cerebrovascular events Myocardial ischemia/MI Proteinuria Renal thrombotic microangiopathy Systemic hypertension Venous thromboembolic disease Reversible posterior leukoencephalopathy syndrome |
| Tyrosine kinase-related targeted therapies:
primarily ABL
directed Nilotinib Ponatinib Dasatinib |
Reduction in endothelial cell c-Abl signaling
and cell survival Reduction in VEGF-R2 signaling with reduction in endothelial function, survival, and proliferation |
Cerebrovascular events Myocardial ischemia/MI Pulmonary hypertension (especially dasatinib) Systemic hypertension (especially ponatinib) Venous thromboembolic disease |
| Proteasome inhibitors (bortezomib, carfilzomib) | Induction of vascular oxidative
stress Endothelial dysfunction and injury Inhibition of endothelial cell proliferation |
Cerebrovascular events Myocardial ischemia/MI Systemic and pulmonary hypertension Venous thromboembolic disease |
| Immunomodulatory agents (thalidomide, lenalidomide) | Inhibition of endothelial cell
migration Induction of homeostatic imbalance |
Cerebrovascular events Myocardial ischemia/MI Systemic hypertension Venous thromboembolic disease |
| Immune checkpoint inhibitors: ipilimumab (CTLA-4), nivolumab (PD-1), permbrolizumab (PD-1), atezolizumab (PD-L1), avelumabn (PD-L1), durvalumab (PD-L1) | Activation of immune cells (T cells) | Myocarditis (vasculo-mediated) Vasculitis |
ABL indicates Abelson murine leukemia viral oncogene homolog; Ang-1, angiopoietin 1; CTLA-4, cytotoxic T lymphocyte–associated protein 4; eNOS, endothelial nitric oxide synthase; Erk, extracellular signal–regulated kinase; Ig, immunoglobulin; IP3, inositol trisphosphate; MAPK, mitogen-activated protein kinase; MI, myocardial infarction; NO, nitric oxide; PDGFβ-R, platelet-derived growth factor-β receptor; PD-1, programmed cell death protein 1; PD-L1, programmed death ligand 1; PI3K, phosphatidylinositol-4,5-bisphosphate 3-kinase; PKC, protein kinase C; PLCγ, phosphoinositide phospholipase Cγ; Tie-2, tyrosine kinase with Ig and endothelial growth factor homology domains type 2; VEGF, vascular endothelial growth factor; and VEGF-R, vascular endothelial growth factor receptor.
Vascular toxicities are presented in alphabetical order. Order does not reflect prevalence of respective toxicities.
VEGF inhibitors, which include both biologics (eg, bevacizumab, a monoclonal antibody targeting circulating VEGF) and small-molecule inhibitors targeting VEGF receptors, lead to increased blood pressure within days to a week of starting therapy, resulting in hypertension in at least a quarter of patients. The level of variation in the observed blood pressure response and in the criteria used to define systemic hypertension in clinical practice and clinical trials has bestowed a wide range of incidence estimates. Despite this uncertainty, it is well documented that newer VEGF inhibitors can result in hypertension in an even larger percentage of patients, in some cases >50%.108 For example, treatment-induced hypertension has recently been reported in 57% of antineoplastic-naïve patients with metastatic renal cell carcinoma newly started on pazopanib.109 VEGF is a critical growth and survival factor for endothelial cells and exerts important homeostatic functions. Inhibition of VEGF signaling may elevate blood pressure by reducing the bioavailability of nitric oxide, a pivotal vasodilator and antithrombotic and anti-inflammatory molecule, and by increasing the activity of the potent vasoconstrictor peptide endothelin-1. In addition, it can lead to capillary rarefaction, resulting in increased resistance in the microcirculation.110 Other proposed mechanisms of VEGF inhibitor–associated hypertension include a rightward shift of the renal pressure–natriuresis curve, impaired sodium excretion with consequent fluid retention, and salt-dependent hypertension.110,111 On the basis of this evidence, the association between VEGF inhibitors and hypertension is not surprising; however, the best antihypertensive approach remains undefined. In the absence of a directly tested protocol for the management of VEGF inhibitor– related hypertension, general practice includes the avoidance of the calcium channel blockers diltiazem and verapamil because of the risk of drug-drug interaction related to the induction of CYP3A4 and the preferred use of angiotensin-converting enzyme inhibitors and the dihydropyridine calcium channel blocker amlodipine.112 Of note, angiotensin-converting enzyme inhibitors have been associated with superior outcomes in patients with renal cell cancer in small studies.113,114
Recent reports indicate that VEGF inhibitor therapy might lead to adverse vascular events, including aortic dissection,115 stroke,116,117 and arterial and venous thrombosis. Bevacizumab is associated with the highest incidence of VTE among VEGF inhibitors, with VTEs occurring in nearly 12% of patients118 compared with 2% to 6% of patients treated with VEGF receptor tyrosine kinase inhibitors.108 Nearly half of these events are high-grade VTEs, defined as thrombotic episodes leading to clinical events, medical interventions, or death.
The vascular toxicity associated with small molecules inhibiting VEGF may also contribute to the cardiomyopathy observed with these agents. For example, the earliest drugs approved in this class, sunitinib, sorafenib, and pazopanib, which target other angiogenic kinase receptors such platelet-derived growth factor receptors, were associated with the occurrence of cardiomyopathy.119–122 Mouse models of VEGF inhibitor–associated cardiomyopathy suggest that these drugs may lead to capillary rarefaction in the myocardium, subsequent myocyte hypoxia, and induction of hypoxia-inducible factors, which is sufficient to cause a reversible cardiomyopathy.123–126 Consistent with these models, the cardiomyopathy seen with sunitinib and sorafenib is often reversible.119,127 Although additional research is needed, these data suggest a mechanistic linkage between vascular and myocardial pathologies and the general contribution of the former to myocardial disease both in cardio-oncology and in other forms of heart disease.2
There are intriguing similarities between vascular toxicities associated with VEGF inhibitors and pregnancy-associated CVD.2 As in preeclampsia, proteinuria often accompanies hypertension in patients treated with VEGF inhibitors.128 Patients may also have evidence of thrombotic angiopathy on renal biopsy, similar to patients with severe preeclampsia.129 Emerging evidence that pregnancy-related CVD (including peripartum cardiomyopathy) is at least partly caused by VEGF inhibition (via placental secretion of sFLT-1 [soluble fms-like tyrosine kinase 1], a soluble splice variant of the VEGF receptor that functions as a decoy receptor) provides biological plausibility of a common underlying mechanism.2,130,131 The above observations also suggest that cardiovascular sequelae that arise as a result of VEGF inhibitors are probably the result of “on-target” effects.
Whereas vascular toxicities seen with VEGF inhibitors may be expected (and even predicted) on the basis of the underlying biology, the spectrum of vascular sequelae of kinase inhibitors targeting ABL1 in patients with CML has been surprisingly broad. Imatinib, the first kinase inhibitor in this class, has a safe clinical profile, and some data suggest a vascular protective effect.107,132 In contrast, nilotinib is associated with peripheral and coronary artery events.14,107,108 A single-center observation of the occurrence of ankle-brachial index reductions in patients treated with nilotinib suggests atherosclerosis as the main pathophysiological mechanism.133 Ponatinib, a third-generation kinase inhibitor for CML and the only drug active against a number of resistant CML subtypes, is associated with significant peripheral and coronary artery ischemic events. The severity of these events led to transient withdrawal of drug approval by the US Food and Drug Administration (FDA).107 Dasatinib has been associated with pulmonary hypertension and a slightly higher incidence of MI and stroke compared with imatinib.134,135 The diverse vascular effects of kinase inhibitors in CML suggest that these toxicities are likely dissociable from the cancer target (in this case, the ABL1 kinase) and suggest “off-target” vascular effects.107
Other classes of novel oncology therapies work through different mechanisms but are associated with significant vascular disease. Immunomodulators such as thalidomide and lenalidomide and proteasome inhibitors such as carfilzomib target the cellular protein degradation machinery and have been highly effective for a number of B-cell malignancies, including multiple myeloma.136 Immunomodulators are associated with the occurrence of thromboembolic events, which occur predominantly in the venous circulation and in patients receiving concomitant multiagent chemotherapy and dexamethasone.136 The prothrombotic effect could be extrinsic, arising from stimulation of the coagulation cascade consequent to endothelial injury. Alternatively, in vitro data support the hypothesis that thalidomide and lenalidomide induce a hypercoagulable state through increased endothelial tissue factor expression; that is, stimulation of the intrinsic coagulation pathway.137 The mechanisms for the increased risk of thrombotic events with thalidomide and its analogs are ill-defined. For clinical practice, risk factors have been identified that enable a practical approach within the framework of 3 risk categories.138 Patients on single-agent thalidomide (or thalidomide analogs) are at low risk (<5%) and do not require prophylaxis. Patients with no or 1 risk factor who are not receiving multiagent chemotherapy or high-dose dexamethasone are at standard risk (up to 20%) and should receive prophylaxis with aspirin (81 mg/d may suffice). Patients with ≥2 risk factors and any patient receiving multiagent chemotherapy or high-dose dexamethasone are considered high risk and should receive prophylaxis with either warfarin or low-molecular-weight heparin (LMWH).138
In the past 2 decades, immune checkpoint inhibitors have emerged as one of the most revolutionary paradigms in cancer therapy. The best known of these are monoclonal antibodies that block CTLA-4 (cytotoxic T lymphocyte–associated protein 4) and PD-1 (programmed cell death protein 1) T-lymphocyte receptor pathways, thus activating the immune system.139 Drugs such as ipilimumab (targeting CTLA-4) or nivolumab and pembrolizumab (targeting PD-1) have produced durable regressions in patients with a widening spectrum of malignancies.140 Vascular events (specifically vasculitis) have been described in a number of recent case reports in patients treated with immune checkpoint inhibitors.141,142 Fulminant myocarditis has been described as a rare toxicity associated with these therapies.143,144 Given the explosion of these therapies for all cancer types, alone and in combination with other cancer agents with known vascular toxicity, it will be important to monitor patients for the occurrence of cardiovascular events and to better define the specific toxicities associated with these therapies.
The immune system may be harnessed in other ways to fight cancer. For example, there has been considerable interest in chimeric antigen receptor (CAR)–modified T cells. Broadly, this personalized therapeutic approach involves removal of the patient’s T cells, followed by in vitro activation, genetic modification, and infusion of the cells back into the patient. Recently, autologous anti-CD19 CAR T-cell therapy showed considerable efficacy in patients with refractory large B-cell lymphoma and acute B-cell lymphoblastic leukemia.145,146 The main adverse event after the infusion of CAR T cells is the onset of the cytokine release syndrome, characterized by immune activation with elevated inflammatory cytokines, including interferon-γ, granulocyte macrophage colony-stimulating factor, IL-10, and IL-6.147 It is too early to determine whether cardiovascular complications are a concern with CAR T-cell treatment, although vascular leak syndrome with hypotension, QT prolongation, tachycardia and other arrhythmias, troponin elevation, and LV systolic dysfunction have been reported in small subsets of patients.144
METABOLIC COMPLICATIONS OF CANCER THERAPIES
The link between metabolic dysregulation and CVD has been under investigation for decades. Abnormalities in glucose and lipid levels and increased blood pressure are key elements of the metabolic syndrome. Increased levels of each of these components are associated with increased rates of CVD.148 Thus, it should not be surprising that therapies that adversely affect metabolism may also be associated with cardiac and vascular sequelae. The use of androgen deprivation in prostate cancer serves as a good example. Androgen deprivation therapy (ADT) has been used to treat this hormone-sensitive malignancy for decades and is accepted as front-line therapy.149 In 2006, using a large population-based study of older men, Keating and colleagues150 demonstrated that ADT, in the form of gonadotropin-releasing hormone agonism, was associated with a significantly increased risk of incident diabetes mellitus, CAD, MI, and sudden cardiac death by 44%, 16%, 11%, and 16%, respectively. Further retrospective studies confirmed these results, again using real-world populations.151,152 On the other hand, data from randomized oncology clinical trials demonstrated that ADT increased mortality only in patients with underlying CAD or heart failure (HF).153–156 In exploring the mechanisms of these adverse cardiovascular events, multiple studies have shown that ADT increased insulin resistance and the rate of incident diabetes mellitus.150,151,157–161 ADT has also been shown to consistently increase total cholesterol and low-density lipoprotein levels and to have mixed effects on high-density lipoprotein levels.162–166 Despite the largely adverse changes in metabolism, endothelial function is preserved or enhanced with ADT.167,168 Moreover, the changes in metabolism and vascular function return to baseline on ADT cessation.169
Because cancer itself represents a dysregulation of metabolism to promote cell growth and survival, it should not be surprising that metabolism has become a therapeutic target. In many cases, these targets also play critical roles in normal metabolic homeostasis. For example, PI3Ks (phosphoinositide 3-kinases) are lipid kinases that mediate response to insulin. The PI3K pathway is also frequently altered in cancer; small-molecule inhibitors targeting PI3K or immediate downstream targets have been introduced at a rapid pace, and several have already been approved for specific cancer types. Not surprisingly, glucose can be affected by PI3K inhibition because upregulation of the glucose transporter, glut4, occurs in part through insulin-mediated PI3K activation.170 Copanlisib, which is indicated for the treatment of adults with relapsed follicular lymphoma who have received at least 2 prior systemic therapies, is commonly associated with hyperglycemia.171–174 Although glucose dysregulation as a result of PI3K inhibitors may be expected, metabolic abnormalities resulting from other novel therapies may suggest new targets for scientific study. Small-molecule inhibitors that target VEGF and platelet-derived growth factor signaling pathways (eg, sunitinib and sorafenib) seem to improve glycemia.175–179 In some instances, agents used to treat the same malignancy have opposite effects on glycemia. Such is the case in the treatment of CML. Imatinib improves glycemia, whereas nilotinib can worsen it.180–184 Taken together, these findings indicate more complex pharmacological effects or regulation of metabolism by these agents than is currently appreciated. In addition, they emphasize the need for increased clinical vigilance when novel therapeutic agents are applied.
THROMBOTIC COMPLICATIONS IN CANCER AND ITS TREATMENT
Venous thrombosis, including superfi thrombophlebitis, DVT, in-dwelling catheter–associated thrombosis, and pulmonary embolism, likely represents the most common cardiovascular complication of malignancy. In a Dutch series of 3220 consecutive patients with a first DVT or pulmonary embolism, the presence of malignancy increased the rate of VTE 7-fold compared with patients without cancer.185 In this series, hematologic malignancies increased the odds ratio of VTE 28-fold, whereas both lung and gastrointestinal tumors increased the odds >20-fold. In a Danish study of 57 951 patients with cancer and 287 476 individuals in a general population cohort, cancer increased the risk of VTE 8-fold.186 The risk of developing VTE was highest (15-fold) in the first year after diagnosis. As would be expected, the presence of metastases, particularly at distant sites, also increases the VTE risk (Figure 1).187 In a large Californian cancer registry, for various types of malignancy, the 2-year cumulative incidence of VTE increased with progression of the disease from localized to regional to remote.188 Patients with cancer represent ≈20% of the overall VTE burden, and the annual incidence in these patients is 0.5% compared with 0.1% in the general population.189 Despite a stable background population rate of VTE, the incidence of cancer-associated VTE is increasing over time.187
Figure 1. Two-year incidence of venous thromboembolism (VTE) by type and spread of cancer.
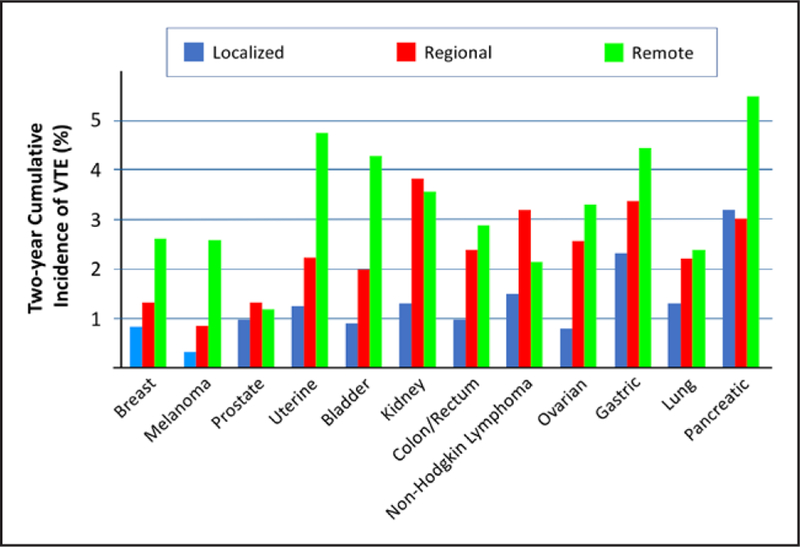
Modified from Timp et al187 with permission of the American Society of Hematology; permission conveyed through Copyright Clearance Center, Inc. Copyright © 2013, American Society of Hematology.
Thrombosis in patients with cancer is also characterized by a particularly high clot burden. Imberti and colleagues190 have demonstrated that the rates of bilateral lower extremity DVT, iliocaval thrombosis, and upper limb DVT were elevated in patients with cancer compared with patients without cancer. Furthermore, patients with cancer have significantly increased rates of VTE recurrence. In a study of 840 patients with DVT, individuals with cancer had an ≈21% recurrence rate at 1 year compared with ≈7% in the patients without malignancy.191 In the RIETE registry (Registro Informatizado de Enfermedad TromboEmbólica), which included nearly 19 000 subjects, the relative risk of recurrence was 2.4-fold for DVT and 2-fold for pulmonary embolism.192 As discussed in the next paragraph, the high rate of recurrence has led to recommendations for extended or indefinite anticoagulation. Louzada and colleagues193 developed a clinical prediction rule for recurrent VTE with cancer-associated thrombosis. The authors identified 4 predictors: sex, primary tumor site, stage, and prior VTE (Table 3). The score had a 100% sensitivity, a % negative predictive value, and a negative likelihood ratio of 0.16. Scores ranged from −3 to 3; a score of ≤0 indicated a risk of <4.5%, whereas a score of ≥1 was associated with a risk of recurrence of 19%. The score was validated in 2 randomized controlled trials of anticoagulation in cancer-associated VTE. The development of a VTE event is a poor prognostic sign in a patient with malignancy. Patients with cancer-associated VTE have an increased risk of bleeding compared with patients with cancer without VTE. Worse, cancer-associated VTE confers a significantly increased risk of death. In a Norwegian study of 740 patients and a first VTE, mortality was 5-fold higher in patients with cancer compared with those without cancer.194 In the RIETE registry, the risk was 6-fold.195 Even within a cancer-only population, VTE increases the rate of death from 1.6- to 4.2-fold.188 Causality is often not easy to establish, and there is no recommendation for routine VTE prophylaxis in cancer outpatient practice except for patients with multiple myeloma (as outlined in the Vascular Complications With Targeted Therapies section).
Table 3.
Ottawa Score for Recurrent Venous Thromboembolism in Cancer-Associated Thrombosis
| Variable | Points |
|---|---|
| Female | 1 |
| Lung cancer | 1 |
| Breast cancer | −1 |
| TNM stage 1 | −2 |
| Previous VTE | 1 |
| Clinical probability | |
| Low (≤0) | −3 to 0 |
| High (≥1) | 1 to 3 |
TNM indicates tumor, node, and metastases; and VTE, venous thromboembolism.
Modified from Louzada et al193 with permission. Copyright © 2012, American Heart Association, Inc.
The presence of cancer changes the duration of therapy of VTE and requires the use of specific drugs. Because of the increased risk of recurrence, the American College of Chest Physician guidelines recommend “extended anticoagulant therapy (no scheduled stop date)” even in the presence of a high bleeding risk.196 Three randomized clinical trials have compared LMWH therapy with vitamin K antagonism with an international normalized ratio target of 2.0 to 3.0. Both dalteparin and enoxaparin showed superiority in the prevention of recurrent VTE compared with vitamin K antagonism therapy.197,198 As a result of these 2 clinical trials, LMWH is the preferred therapy at this time; however, limiting factors include costs, the need for long-term injections, and the inability to easily reverse the effects. The reduction of recurrent VTE in a study comparing tinzaparin with vitamin K antagonism trended toward better outcomes with LMWH, but it did not reach statistical significance.199 Over the past decade, direct oral anticoagulants (DOACs) have become a standard therapy in the management of VTE. A meta-analysis of the large approval trials of DOACs showed preliminary evidence that these agents are as effective and safe as conventional treatment for the prevention of VTE in patients with cancer.200 More recently, the first 2 of several trials of DOACs in a cancer population have been published. In a study of 1050 patients with cancer and acute VTE, participants were randomized to dalteparin or edoxaban for 6 to 12 months.201 The primary outcome, recurrent VTE or major bleeding, was met by 12.8% of the subjects randomized to edoxaban and 13.5% of the subjects who received dalteparin (P=0.006 for noninferiority). In a pilot study, the risk of recurrent VTE in cancer patients was lower with rivaroxaban compared with dalteparin (HR, 0.43 [95% CI, 0.19–0.99]). However, the risk of clinically relevant non-major bleeding was higher with rivaroxaban than with dalteparin (HR, 3.76 [95% CI, 1.63–8.69]).201a A study comparing apixaban with dalteparin has been presented in abstract form. Treatment with apixaban was associated with a significantly lower rate of VTE recurrence compared with dalteparin (3.4% vs 14.1%, respectively; HR, 0.26 [95% CI, 0.09–0.80]; P=0.0182) with superior quality of life and very low bleeding rates.201b Finally, the Rivaroxaban in the Treatment of Venous Thromboembolism (VTE) in Cancer Patients clinical trial (NCT02583191; comparing rivaroxaban with standard LMWH therapy) is currently ongoing.
From the accumulating evidence, it is likely that in the near future oral DOAC therapy may become a standard therapy for cancer-associated VTE, but several caveats apply, including the impact of renal and liver dysfunction on dosing, drug-drug interactions, and the problem of reversing anticoagulation effects.
COMMON RISK FACTORS BETWEEN CANCER AND CVD
An area in need of further exploration in cardio-oncology has emerged from the growing evidence that common risk factors can predispose to both cancer and CVD.202 Notably, data from the ARIC study (Atherosclerosis Risk in Communities) indicate that adherence to the 7 American Heart Association 2020 Strategic Impact Goal cardiovascular health metrics is inversely associated not only with CVD but also with cancer, most strongly breast, colorectal, and lung cancer.203 Some risk factors (eg, tobacco) have a wellknown association with certain cancer types (eg, lung cancer) and CVD.204 More recently, other cardiovascular risk factors have also emerged as potentially important risk factors in cancer. For example, epidemiological data suggest that hyperlipidemia can serve as a risk factor for estrogen receptor–positive breast cancer.205 Data from the Canadian National Cancer Surveillance System study suggest that postmenopausal women within the top quartile of dietary cholesterol intake had a 48% increase in the risk of breast cancer.206 Mechanistically, 27-hydroxycholesterol, a cholesterol metabolite, may serve as the biochemical link between lipid metabolism and cancer. 27-Hydroxycholesterol can act as a direct estrogen receptor agonist in breast cancer cells, thus stimulating the growth and metastatic spread of tumors in several models of breast cancer.207 Alternatively, a growing body of literature suggests that inflammation is a risk for both cancer and CVD. For example, the recent results of CANTOS (Canakinumab Antiinflammatory Thrombosis Outcome Study) showed that pharmacological inhibition of IL-1β reduced both cardiac events and lung cancer incidence and mortality.208,209
Genetic risk factors have also emerged as important common risk factors for cancer and CVD. Clonal hematopoiesis of indeterminate potential, which is defined as the presence of an expanded somatic blood cell clone in individuals without other hematologic abnormalities, is common in older populations and is associated with an increased risk of hematologic cancer.210,211 Surprisingly, clonal hematopoiesis of indeterminate potential also serves as risk factor for MI, stroke, and all-cause mortality in this population.210 Clonal hematopoiesis of indeterminate potential results from an expansion of cells that harbor an initiating driver mutation, with frequent somatic mutations in 3 genes: DNMT3A, ASXL1, and TET2.210,211 Mutations in these 3 genes are each individually associated with coronary heart disease, and basic models suggest that these genes may participate in the pathogenesis of atherosclerosis.212,213
These common links between cancer and CVD have enormous public health implications.202 For example, the link between cholesterol and breast cancer provides the rationale for the clinical evaluation of pharmacologic approaches that interfere with cholesterol/27-hydroxycholesterol synthesis (ie, a statin) as a means to mitigate breast cancer pathogenesis. These observations may be particularly relevant to cancer survivors. The >16 million American cancer survivors are at risk of 2 major diseases that contribute to morbidity and early mortality: recurrence of their cancer and CVD. The ability to address these patients’ cardiovascular risks may have the added advantage of protecting them from cancer recurrence. In addition, identifying new risk factors such as genetic risks may inform new approaches to preventing and treating both conditions in this population. The relevance of the intersection between CVD and cancer is being increasingly recognized within the cardiovascular community, as highlighted in a recent scientific statement from the American Heart Association,214 which should serve to stimulate much-needed research in this area.
CARDIO-ONCOLOGY RESEARCH DIRECTIONS
The major change in cancer treatment has involved a shift from nonselective toxins to therapies aimed at specific pathways important for cancer growth and survival. These therapeutic options have expanded through investigation of the pathways involved in tumorigenesis. These pathways often play critical roles in cardiovascular homeostasis and may exert effects on the heart and vasculature. As the number of small-molecule kinase inhibitors, which can target multiple kinases, expands, it will be important to define the “off-target” kinase effects of these therapies to better predict possible toxicities when these agents are being tested in humans.215,216 Understanding the vascular sequelae of new targeted cancer therapies also offers an opportunity for basic and translational investigations in which pathways critical for vascular signaling may be uncovered.2 Consequently, robust programs in cardio-oncology engage in a multidisciplinary approach in which cardiovascular and oncology teams informed of the most recent research findings closely collaborate to scrutinize for new vascular sequelae and strive to tailor therapies accordingly. Recent primary prevention studies in patients with breast cancer receiving anthracyclines or HER2-targeted therapies point out the limitations of a “universal prevention” (or “wide-gun”) approach such as the use of either β-blockers or angiotensin-converting enzyme inhibitors in all patients undergoing breast cancer treatment.217–220 There is a critical unmet need to develop personalized, risk-based approaches based on a detailed knowledge of the underlying pathophysiology.
Further collaboration with basic and translational scientists will permit investigations to elucidate the mechanism of the toxicities and to define patients at risk, such that preventive and treatment efforts can be focused on these high-risk patients. As the fi of cardio-oncology matures, integration of basic and translational research teams will be needed to most efficiently define the cardiovascular implications of new therapies, to elucidate the mechanisms of toxicity, and to develop management strategies for patients.215 More robust basic and translational research models, however, will be needed to determine the mechanisms of toxicity. In cardiomyocyte biology, the introduction of induced pluripotent stem cells differentiated into cardiomyocytes offers a human-based platform on which cardiac toxicity can be modeled.221 Differentiation of induced pluripotent stem cells into endothelial cells and smooth muscle cells could likewise enable modeling of vascular toxicity. In addition, induced pluripotent stem cells can be derived from individual patients with or without clinical toxicity, which could further advance the personalized approach221 and help to define vascular and metabolic mechanisms of toxicity (Table 4).
Table 4.
Future Research Directions in Vascular Cardio-Oncology
| More rigorous identification of cardiovascular and cardiometabolic side effects during clinical trials and in the real-world population after drug approval |
| Cardiovascular adjudications by an independent committee during clinical trials |
| Multi-institutional registries for identifying vascular and metabolic toxicities once a drug is approved |
| Open-source data sharing among pharmaceutical companies with cardiovascular toxicities of cancer therapies |
| Comprehensive and systematic vascular phenotyping via biomarkers and imaging |
| Personalized/precision medicine in cardio-oncology |
| Better identification of patients at risk for cardiovascular toxicities during cancer treatment |
| Single integrated registry with researchers, patients, providers, and clinical diagnostic laboratories entering family history, clinical and research data, and accompanying biospecimens (including DNA) in a deidentified manner |
| Genetic inquiries for risk of toxicity |
| Development of better vascular imaging and use in cardio-oncology population |
| Integration of basic, translational, and clinical research programs in academic cardio-oncology |
| Cardiovascular clinical and translational models to help elucidate mechanisms of toxicity |
| Development of more robust model cell systems (eg, induced pluripotent stem cells) and animal models for preclinical testing of novel compounds |
| Research on mechanisms of common risk factors (including genetic risk factors) that are shared between cancer and cardiovascular disease |
| Education of clinicians and patients about cardiovascular toxicities of cancer therapies |
| Web-based platforms for access to known vascular toxic effects of novel anticancer drugs |
An example of rigorous clinical observations leading to basic and mechanistic insights is the early reports of cardiomyopathy after treatment with small-molecule kinase inhibitors targeting VEGF and platelet-derived growth factor receptors.120,121 From these clinical observations, several mouse models were created that demonstrated the contribution of impaired vascular function to the cardiomyopathy. One such model involved a mouse expressing a “tunable” transgene encoding a VEGF trap, recapitulating the effects of bevacizumab. In this mouse model, the induction of the VEGF trap leads to decreased myocardial capillary density (capillary rarefaction), induction of hypoxia and hypoxia-inducible genes in the myocardium, and cardiac dysfunction, which is reversible on removal of the transgene.123 Similarly, mice in which PDGF (platelet-derived growth factor) receptor β is genetically deleted in the heart exhibited decreased capillary density, increased myocardial hypoxia, and accentuated HF after transverse aortic constriction.124,222 Both of these models resulted in myocardial hypoxia leading to the stabilization and activation of the master transcriptional factor hypoxia-inducible factor and induction of hypoxia-inducible factor–regulated genes. Chronic stabilization of hypoxia-inducible factor proved sufficient to lead to cardiomyopathy in mice.125,126 Although it remains to be seen whether myocardial hypoxia resulting from VEGF inhibitor–mediated capillary rarefaction plays a causal role in cardiomyopathy in humans, preclinical models predict that the cardiomyopathy is reversible and more consistent with myocardial hibernation rather than necrosis, which is consistent with early clinical observations.119,127
HOW TO STRUCTURE A CARDIO-ONCOLOGY SERVICE
Although much has been written about the need for a cardio-oncology service to provide comprehensive cardiovascular care to patients with cancer and cancer survivors, the actual components and structure of such a service have not been established. In many instances, cardiology consultative services for preoperative cancer surgery assessment or management of symptomatic CVD such as ischemia, arrhythmia, and HF occurring during cancer treatment serve as a focal point for home-grown programs. With the growth of new cancer therapeutics and unprecedented improvement in life expectancy in many patients with cancer, there is an unmet need to develop and implement cardiovascular care across the cancer treatment continuum. This approach requires dynamic recognition and management of cardiovascular care needs before, during, and after cancer treatment and effective integration of cardiac and oncology health teams.
Components of Cardio-Oncology Service
A critical theme of the cardio-oncology service is active collaboration and partnership between cardio-vascular and oncology teams. Different models have been proposed, mostly reflecting differences in individual cancer programs, from comprehensive National Cancer Institute–designated sites and tertiary referral centers to community-based oncology offies.223,224 These programs share the common premise of multidisciplinary collaboration among medical and radiation oncologists, hematologists, surgeons, palliative care specialists, pharmacists, and cardiologists (including cardiovascular imaging, HF, interventional cardiology, electrophysiology, and more recently, vascular medicine subspecialties). The complexity and heterogeneity of the cardio-oncology team reflect not only the recognition of new cardiovascular effects of cancer therapy but also advances in cardiovascular treatment such as new anticoagulation approaches, interventional strategies for thrombotic complications of cancer and its treatments, cardioprotective and vasculoprotective strategies, transcatheter valve replacement, and arrhythmia management that may offer specific advantages to patients with cancer. Because a single cardio-oncology service model is unlikely to meet these diverse needs, we highlight the examples proposed by Snipelisky and colleagues223 and summarize the key elements to consider in program development.
Definition of Need, Content, and Scope of Cardio-Oncology Service
Cardiovascular manifestations in the oncology population span a spectrum of conditions (Figure 2), and the initial step in collaboration requires identification of the individual site priorities along the continuum of cancer treatment. Primary prevention in this field is often defined as cardiovascular care before cancer treatment and may include optimization of existing cardiovascular risk factors and CVD, as well as risk stratification based on the planned cancer management. The recent American Society of Clinical Oncology clinical practice guideline provides a useful tool to standardize identification of patients at risk for cardiac dysfunction226 (although the document pertains primarily to patients with cardiomyopathy from anthracyclines and HER2-targeted therapies). Timeliness of cardiovascular evaluation, particularly in high-risk patients, is of utmost importance in this patient group and in patients undergoing therapy. Development of new conditions or progression of previous cardiovascular conditions during cancer treatment requires urgent attention. Prompt access to cardiovascular imaging and intervention should be anticipated, and coordination of respective services should be established. Examples of common conditions include acute ischemic and thrombotic events, symptomatic HF, and arrhythmias, which may present in the setting of systemic illness, as adverse effects of treatment, or concurrently with cancer progression. Although the majority of cancer care occurs in the outpatient setting, this group of patients may be more likely to need hosptalization and require coordination of cardio-oncology with inpatient services.
Figure 2. Cardiovascular (CV) complications in the oncology population and comprehensive cardio-oncology services across the cancer treatment continuum.
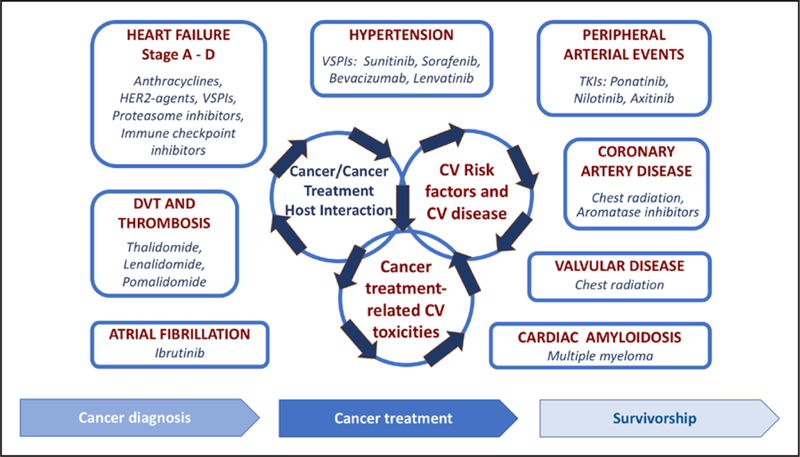
DVT indicates deep vein thrombosis; HER2, human epidermal growth factor receptor-2; TKI, tyrosine kinase inhibitor; and VSPI, vascular endothelial growth factor signaling pathway inhibitors. Modified from Barac et al225 with permission from the American College of Cardiology Foundation. Copyright © 2015, American College of Cardiology Foundation.
Another distinct area of need is long-term cardiac follow-up for patients undergoing cancer treatment for more indolent malignancies such as CML or met static solid tumors with long survivorship such as breast cancer. At the present time, there are limited recommendations for longitudinal cardiovascular surveillance after treatment with anthracyclines and HER2-targeted therapies (monitoring of LV systolic function) and VEGF inhibitors (monitoring of hypertension) has been completed,223 reflecting the gap in knowledge and the need for further research in this area. Cardio-oncology services have a unique opportunity to contribute to developing evidence for multidisciplinary approaches to diagnosing and treating complex phenotypes of cardiovascular toxicities.
Cancer survivorship is another growing area in which cardiovascular needs are being recognized, with the projected number of cancer survivors reaching 20 million by 2026 in the United States.227 In 2006, the Institute of Medicine issued a statement on the need to improve the quality of care of cancer survivors and the importance of individual survivorship care plans stating goals of care, surveillance recommendations, symptoms monitoring, and health maintenance goals.228 At the present time, recommendations for cardiovascular screening in asymptomatic individuals remain limited to survivors who received therapeutic radiation88 or received anthracycline-based regimen as part of childhood and adolescent cancer40 or adult cancer treatment.224
Structure and Institutional Support
The structure of the cardio-oncology service will be determined by the individual oncology programs and resulting patient needs, as well as the resources available. In addition to identification of the content and patient population of interest, considerations need to include staffing and location of cardio-oncology clinics, coordination of care, and institutional support. Different models have been proposed, including (1) a clinic staffed by a cardiologist with communication to oncology practices, (2) a clinic staffed by an oncologist with communication to cardiology, and (3) a truly multidisciplinary approach with the clinic staffed by oncology, cardiovascular, and often other specialists.223 The last model fosters interactions; however, it is resource intensive and may be limited to centers with large volumes. Models 1 and 2 reflect the fact that “ownership” of cardio-oncology services may reside in either specialty as long as expertise and built-in mechanisms for communication are available. The advantages of locating outpatient cardio-oncology clinics within a cancer center include easy patient access and facilitated interactions with oncology services; however, the availability of onsite cardiac imaging and subspecialized cardiovascular services will likely be limited. On the other hand, the alternative model of cardio-oncology clinics within cardiovascular centers may limit access to different oncology specialists and hamper direct communications that promote efficient decision making.40 As the field of cardio-oncology evolves, novel model systems of care will be necessary to address these challenging problems.
With recent emphasis on survivorship care, new models of comprehensive cancer survivorship services are emerging, centered mostly within oncology centers and led by nurse practitioners or physician assistants.229 These programs will offer an important opportunity for the identification of cardiovascular needs and development of pathways for further integration of cardiovascular services into survivorship care. Institutional support is an essential component for the development and sustainability of cardio-oncology services. As cardio-oncology expands as a field, the development of metrics of success will be important to ensure efficiency and quality of care.225
Continued Integration, Education, and Training
The creation of an integrated multidisciplinary approach and focus on patient care have been identified as essential components of a cardio-oncology service (Figure 3). In 2015, a cardio-oncology survey by the American College of Cardiology identified the lack of national guidelines and funding as the main barrier to growth and expansion of cardio-oncology programs, followed by limitations in infrastructure, interest, and educational opportunities.230 In <3 years, progress has been made in several key areas. Recent examples clude FDA Public Workshops dedicated to this topic231 and National Institutes of Health research funding announcements focused on improving outcomes in cancer treatment–related cardiotoxicity,232 which are critical developments that will foster the growth of cardio-oncology. Several multidisciplinary professional society guidelines and statements have been published in a span of <2 years addressing the intersection between CVD and cancer care.40,214,233 These comprehensive documents have increased the educational resource base for providers focused on the cardiovascular needs and care of oncology populations and have spurred mainstream educational sessions in cardio-oncology at major scientific meetings. The need for dedicated cardio-oncology training within both specialties has been recognized, and the opportunity to implement and disseminate training will further define centers of excellence in cardio-oncology.
Figure 3. Integrated multidisciplinary approach and focus on patient care, research (basic, translational, clinical, and population science), education, and clinical training.
CV indicates cardiovascular.
In summary, the operational considerations of a cardio-oncology service include full engagement by the healthcare team to provide patient-centered care approaches; proximity of necessary services and providers; timely scheduling of diagnostic studies with prompt care on the basis of results; timely communication of changes in cancer treatment plans and changes in cardiovascular status; patient education and engagement; and enhanced clinical flow with physician extenders for routine follow-up and preventive care. The need to aggressively address modifiable lifestyle factors cannot be overstated, with an emphasis on referral to cardiac rehabilitation, dieticians, exercise physiologists, and lifestyle education as required.
ROLE OF THE CARDIO-ONCOLOGY FELLOWSHIP
As the field of cardio-oncology matures, there will be a need for more formal training to better prepare the next generation of physicians for what has emerged as a new discipline in cardiology. Cardio-oncology initially emerged as an HF specialty given the toxicities of older agents such as anthracyclines and trastuzumab. However, in 2019, cardio-oncology is truly a general cardiology specialty, and a successful cardio-oncology training program will need to incorporate all elements of a cardiology division, including vascular medicine. Although several institutions have started cardio-oncology fellowship programs, there is a need to better define what makes up a comprehensive fellowship and thus compe intency in the field. In the coming years, a formal training paradigm in the model of the Core Cardiovascular Training Statement will need to be developed like the one created for vascular medicine training more than a decade ago.234
BENCHMARKS AND PUBLIC REPORTING OF NOVEL THERAPIES
Whereas a robust research program in cardio-oncology is critical for better mechanistic understanding of approved cancer therapies, oncology clinical trials offer the opportunity to detect vascular and metabolic toxicities. Oncology clinical trial end points serve different purposes. In conventional drug development, early-phase clinical trials evaluate safety, whereas later-phase studies primarily evaluate whether a drug provides a clinical benefi In general, the FDA recommends at least 2 adequate and well-controlled clinical trials. However, for drugs approved to treat patients with a malignancy, evidence from 1 trial may be sufficient Clinical benefi that supported drug approval have included important primary outcomes (eg, increased survival, symptomatic improvement) and effects on established surrogate end points. The accelerated-approval regulations permit the use of surrogate end points for the approval of drugs or biological products that are intended either to treat serious or life-threatening diseases or to demonstrate an improvement over available therapy, particularly when no proven therapy exists. In the case of accelerated approval, the manufacturer is expected to conduct clinical studies to verify and characterize the actual clinical benefit Although the FDA may grant accelerated approval based on the effects of a surrogate end point that is “reasonably likely” to predict clinical benefi it may lack sufficient information about the risk of the drug to appropriately articulate it in the labeling. In such a case, the FDA can mandate a postmarketing study or establishment of a registry to collect data as a postmarketing requirement. An observational pharmacoepidemiological study can also be designed with input from the FDA to identify serious risks associated with a drug exposure, to quantify the risks, or to evaluate factors that affect the risk of toxicity such as drug dose, timing of exposure, and patient characteristics. Data sources for observational studies can include administrative healthcare claims data, electronic medical records, prospectively collected observational data, and registries. Some registries are required either before or after drug approval by the FDA as part of Risk Evaluation and Mitigation Strategies. When part of a Risk Evaluation and Mitigation Strategy, these registries are considered essential for drug safety and are not designed as a study with completion dates. The FDA can require a Risk Evaluation and Mitigation Strategy if the agency determines that safety measures are needed beyond the professional labeling to ensure that the benefi of a drug outweigh its risks.
Because of these measures implemented by the FDA, vascular and metabolic complications may arise after the introduction of new therapies and lead to changes in their use. As an example, ponatinib is the only tyrosine kinase inhibitor effective against many resistant cases of CML. On the basis of encouraging early results of the phase 2 PACE trial (Ponatinib for Chronic Myeloid Leukemia [CML] Evaluation and Ph+ Acute Lymphoblastic Leukemia [ALL]), the FDA granted ponatinib an accelerated approval in 2012.235 Updated safety information from the PACE trial in late 2013 showed the occurrence of arterial and venous thrombosis and occlusions in at least 27% of patients treated with ponatinib.236 The sponsor temporarily withdrew ponatinib from the market, and the FDA allowed reintroduction of marketing with implementation of revised labeling, a Risk Evaluation and Mitigation Strategy program, and additional postmarketing safety requirements. Nevertheless, existing data from the ponatinib trial gave very little insight into the mechanisms of vascular toxicity.237 Whether these cases represent atherosclerotic events, thromboembolic events, or some other vascular process such as vasospasm is unclear. These distinctions are critical because cardiovascular specialists may approach each condition differently in terms of both prevention and treatment. Cases such as the one with ponatinib argue for possible cardiovascular adjudication in oncology trials by an independent committee. Furthermore, similar to the recent emphasis on CVD end points in the approval of medications for diabetes mellitus, stricter oversight for adverse cardiovascular events in the approval process may be warranted. A number of strategies and checklists have been proposed to detect cardiovascular safety signals with cancer drugs early and in a more standardized manner.238
CONCLUSIONS
The remarkable advances in the understanding of cancer biology have led to breakthrough treatments and an ever-growing number of cancer survivors. This progress has come with both new challenges and unexpected discoveries that have blurred the border between oncology and cardiovascular medicine. Traditional and new cancer treatments, including several targeted therapies, are associated with vascular injury and metabolic complications. These untoward effects increase the short-and long-term risk of cardiovascular events above and beyond the already elevated risk often present in patients with cancer and cancer survivors. An improved understanding of the mechanisms of toxicity of these therapies may lead to the identification of novel targets to reduce vascular complications while providing biological insights into cardiovascular pathophysiology and informing new platforms for drug discovery. Clinically, optimal management of patients with cancer and cancer survivors is best addressed by a multidisciplinary approach whereby cardiovascular medicine specialists work closely with oncologists to assess cardiovascular risk, to minimize vascular toxicity, and to manage long-term adverse effects. This multidisciplinary approach will require the creation of cardio-oncology services and the training of a new generation of cardiovascular specialists with a broad understanding of cancer treatments.
Disclosures
Writing Group Disclosures
| Writing Group Member | Employment | Research Grant | Other Research Support | Speakers’ Bureau/ Honoraria | Expert Witness | Ownership Interest | Consultant/ Advisory Board | Other |
|---|---|---|---|---|---|---|---|---|
| Umberto Campia | Brigham and Women’s Hospital–Harvard Medical School | None | None | None | None | None | None | None |
| Javid J. Moslehi | Vanderbilt University Medical Center | Bristol-Myers Squibb†; NIH (R56 HL141466)†; Pfizer† | None | None | None | None | Bristol-Myers Squibb*; Ipsen*; Myokardia*; Novartis*; Pfizer*; Regeneron*; Takeda* | None |
| Laleh Amiri-Kordestani | FDA | None | None | None | None | None | None | None |
| Ana Barac | Medstar Heart and Vascular Institute, Medstar Washington Hospital Center | Genentech (serves as a cardiology PI on an investigator-initiated study supported by Genentech)* | None | None | None | None | None | None |
| Joshua A. Beckman | Vanderbilt University | Bayer (DSMB)†; Novartis (DSMB)* | None | None | None | None | AstraZeneca*; Boehringer Ingelheim*; Bristol Myers Squibb*; Merck*; Sonofi* | None |
| David D. Chism | Vanderbilt University Medical Center | None | None | None | None | None | None | None |
| Paul Cohen | Rockefeller University Laboratory of Molecular Metabolism | None | None | None | None | None | None | None |
| John D. Groarke | Brigham and Women’s Hospital | Amgen† | None | None | None | None | None | None |
| Joerg Herrmann | Mayo Clinic Rochester | Amgen† | None | None | None | None | None | None |
| Carolyn M. Reilly | Emory University School of Nursing | None | None | None | None | None | None | None |
| Neil L. Weintraub | Medical College of Georgia at Augusta University | NIH (AR070029, HL134354, HL126949, HL142097)† | None | None | None | None | None | None |
This table represents the relationships of writing group members that may be perceived as actual or reasonably perceived conflicts of interest as reported on the Disclosure Questionnaire, which all members of the writing group are required to complete and submit. A relationship is considered to be “significant” if the person receives $10 000 or more during any 12-month period, or 5% or more of the person’s gross income; or (b) the person owns 5% or more of the voting stock or share of the entity or owns $10 000 or more of the fair market value of the entity. A relationship is considered to be “modest” if it is less than “significant” under the preceding definition.
Modest.
Significant.
Reviewer Disclosures
| Reviewer | Employment | Research Grant | Other Research Support | Speakers’ Bureau/ Honoraria | Expert Witness | Ownership Interest | Consultant/Advisory Board | Other |
|---|---|---|---|---|---|---|---|---|
| Geoffrey D. Barnes | University of Michigan | BMS/Pfizer† | None | None | None | None | BMS/Pfizer*; Jansen* | None |
| Michael Fradley | University of South Florida | None | None | None | None | None | Novartis* | None |
| Eric H. Yang | University of California at Los Angeles | None | None | None | None | None | Voluntis* | None |
This table represents the relationships of reviewers that may be perceived as actual or reasonably perceived conflicts of interest as reported on the Disclosure Questionnaire, which all reviewers are required to complete and submit. A relationship is considered to be “significant” if (a) the person receives $10000 or more during any 12-month period, or 5% or more of the person’s gross income; or (b) the person owns 5% or more of the voting stock or share of the entity, or owns $10 000 or more of the fair market value of the entity. A relationship is considered to be “modest” if it is less than “significant” under the preceding definition.
Modest.
Significant.
Footnotes
The American Heart Association makes every effort to avoid any actual or potential conflicts of interest that may arise as a result of an outside relationship or a personal, professional, or business interest of a member of the writing panel. Specifically, all members of the writing group are required to complete and submit a Disclosure Questionnaire showing all such relationships that might be perceived as real or potential conflicts of interest.
Publisher's Disclaimer: The expert peer review of AHA-commissioned documents (eg, scientific statements, clinical practice guidelines, systematic reviews) is conducted by the AHA Office of Science Operations. For more on AHA statements and guidelines development, visit http://professional.heart.org/statements. Select the “Guide-lines & Statements” drop-down menu, then click “Publication Development.”
REFERENCES
- 1.Ky B, Vejpongsa P, Yeh ET, Force T, Moslehi JJ. Emerging paradigms in cardiomyopathies associated with cancer therapies. Circ Res 2013;113:754–764. doi: 10.1161/CIRCRESAHA.113.300218 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bellinger AM, Arteaga CL, Force T, Humphreys BD, Demetri GD, Druker BJ, Moslehi JJ. Cardio-oncology: how new targeted cancer therapies and precision medicine can inform cardiovascular discovery. Circulation 2015;132:2248–2258. doi: 10.1161/CIRCULATIONAHA.115.010484 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bluethmann SM, Mariotto AB, Rowland JH. Anticipating the “silver tsunami”: prevalence trajectories and comorbidity burden among older cancer survivors in the United States. Cancer Epidemiol Biomarkers Prev 2016;25:1029–1036. doi: 10.1158/1055-9965.EPI-16-0133 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. CA Cancer J Clin 2018;68:7–30. doi: 10.3322/caac.21442 [DOI] [PubMed] [Google Scholar]
- 5.Luwaert RJ, Descamps O, Majois F, Chaudron JM, Beauduin M. Coronary artery spasm induced by 5-fluorouracil. Eur Heart J 1991;12:468–470. [DOI] [PubMed] [Google Scholar]
- 6.Südhoff T, Enderle MD, Pahlke M, Petz C, Teschendorf C, Graeven U, Schmiegel W. 5-Fluorouracil induces arterial vasocontractions. Ann Oncol 2004;15:661–664. [DOI] [PubMed] [Google Scholar]
- 7.Mosseri M, Fingert HJ, Varticovski L, Chokshi S, Isner JM. In vitro evidence that myocardial ischemia resulting from 5-fluorouracil chemotherapy is due to protein kinase C-mediated vasoconstriction of vascular smooth muscle. Cancer Res 1993;53:3028–3033. [PubMed] [Google Scholar]
- 8.Thyss A, Gaspard MH, Marsault R, Milano G, Frelin C, Schneider M. Very high endothelin plasma levels in patients with 5-FU cardiotoxicity. Ann Oncol 1992;3:88. [DOI] [PubMed] [Google Scholar]
- 9.de Forni M, Malet-Martino MC, Jaillais P, Shubinski RE, Bachaud JM, Lemaire L, Canal P, Chevreau C, Carrié D, Soulié P. Cardiotoxicity of high-dose continuous infusion fluorouracil: a prospective clinical study. J Clin Oncol 1992;10:1795–1801. doi: 10.1200/JCO.1992.10.11.1795 [DOI] [PubMed] [Google Scholar]
- 10.Kosmas C, Kallistratos MS, Kopterides P, Syrios J, Skopelitis H, Mylonakis N, Karabelis A, Tsavaris N. Cardiotoxicity of fluoropyrimidines in different schedules of administration: a prospective study. J Cancer Res Clin Oncol 2008;134:75–82. doi: 10.1007/s00432-007-0250-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Saif MW, Shah MM, Shah AR. Fluoropyrimidine-associated cardiotoxicity: revisited. Expert Opin Drug Saf 2009;8:191–202. doi: 10.1517/14740330902733961 [DOI] [PubMed] [Google Scholar]
- 12.Kwakman JJ, Simkens LH, Mol L, Kok WE, Koopman M, Punt CJ. Incidence of capecitabine-related cardiotoxicity in different treatment schedules of metastatic colorectal cancer: a retrospective analysis of the CAIRO studies of the Dutch Colorectal Cancer Group. Eur J Cancer 2017;76:93–99. doi: 10.1016/j.ejca.2017.02.009 [DOI] [PubMed] [Google Scholar]
- 13.Becker K, Erckenbrecht JF, Häussinger D, Frieling T. Cardiotoxicity of the antiproliferative compound fluorouracil. Drugs 1999;57:475–484. doi: 10.2165/00003495-199957040-00003 [DOI] [PubMed] [Google Scholar]
- 14.Herrmann J, Yang EH, Iliescu CA, Cilingiroglu M, Charitakis K, Hakeem A, Toutouzas K, Leesar MA, Grines CL, Marmagkiolis K. Vascular toxicities of cancer therapies: the old and the new–an evolving avenue. Circulation 2016;133:1272–1289. doi: 10.1161/CIRCULATIONAHA.115.018347 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dalzell JR, Abu-Arafeh A, Campbell RT. Cardiotoxicity with 5-fluorouracil based agents: rechallenge cannot currently be safely advised. Am J Cardiol 2013;111:454. doi: 10.1016/j.amjcard.2012.09.006 [DOI] [PubMed] [Google Scholar]
- 16.Clasen SC, Ky B, O’Quinn R, Giantonio B, Teitelbaum U, Carver JR. Fluoropyrimidine-induced cardiac toxicity: challenging the current paradigm. J Gastrointest Oncol 2017;8:970–979. doi: 10.21037/jgo.2017.09.07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Verweij J, Clavel M, Chevalier B. Paclitaxel (Taxol) and docetaxel (Taxotere): not simply two of a kind. Ann Oncol 1994;5:495–505. [DOI] [PubMed] [Google Scholar]
- 18.Schwartz EL. Antivascular actions of microtubule-binding drugs. Clin Cancer Res 2009;15:2594–2601. doi: 10.1158/1078-0432.CCR-08-2710 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Belotti D, Vergani V, Drudis T, Borsotti P, Pitelli MR, Viale G, Giavazzi R, Taraboletti G. The microtubule-affecting drug paclitaxel has antiangiogenic activity. Clin Cancer Res 1996;2:1843–1849. [PubMed] [Google Scholar]
- 20.Sweeney CJ, Miller KD, Sissons SE, Nozaki S, Heilman DK, Shen J, Sledge GW Jr. The antiangiogenic property of docetaxel is synergistic with a recombinant humanized monoclonal antibody against vascular endothelial growth factor or 2-methoxyestradiol but antagonized by endothelial growth factors. Cancer Res 2001;61:3369–3372. [PubMed] [Google Scholar]
- 21.Tofthagen C, McAllister RD, Visovsky C. Peripheral neuropathy caused by paclitaxel and docetaxel: an evaluation and comparison of symptoms. J Adv Pract Oncol 2013;4:204–215. [PMC free article] [PubMed] [Google Scholar]
- 22.Béhar A, Pujade-Lauraine E, Maurel A, Brun MD, Chauvin FF, Feuilhade de Chauvin F, Oulid-Aissa D, Hille D. The pathophysiological mechanism of fluid retention in advanced cancer patients treated with docetaxel, but not receiving corticosteroid comedication. Br J Clin Pharmacol 1997;43:653–658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Rowinsky EK, McGuire WP, Guarnieri T, Fisherman JS, Christian MC, Done-hower RC. Cardiac disturbances during the administration of Taxol. J Clin Oncol 1991;9:1704–1712. doi: 10.1200/JCO.1991.9.9.1704 [DOI] [PubMed] [Google Scholar]
- 24.Soultati A, Mountzios G, Avgerinou C, Papaxoinis G, Pectasides D, Dimopoulos MA, Papadimitriou C. Endothelial vascular toxicity from chemotherapeutic agents: preclinical evidence and clinical implications. Cancer Treat Rev 2012;38:473–483. doi: 10.1016/j.ctrv.2011.09.002 [DOI] [PubMed] [Google Scholar]
- 25.Stefenelli T, Kuzmits R, Ulrich W, Glogar D. Acute vascular toxicity after combination chemotherapy with cisplatin, vinblastine, and bleomycin for testicular cancer. Eur Heart J 1988;9:552–556. [DOI] [PubMed] [Google Scholar]
- 26.Lejonc JL, Vernant JP, Macquin J, Castaigne A. Myocardial infarction following vinblastine treatment. Lancet 1980;2:692. [DOI] [PubMed] [Google Scholar]
- 27.Subar M, Muggia FM. Apparent myocardial ischemia associated with vin-blastine administration. Cancer Treat Rep 1986;70:690–691. [PubMed] [Google Scholar]
- 28.Stoter G, Koopman A, Vendrik CP, Struyvenberg A, Sleyfer DT, Willemse PH, Schraffordt Koops H, van Oosterom AT, ten Bokkel Huinink WW, Pinedo HM. Ten-year survival and late sequelae in testicular cancer patients treated with cisplatin, vinblastine, and bleomycin. J Clin Oncol 1989;7:1099–1104. doi: 10.1200/JCO.1989.7.8.1099 [DOI] [PubMed] [Google Scholar]
- 29.Vogelzang NJ, Bosl GJ, Johnson K, Kennedy BJ. Raynaud’s phenomenon: a common toxicity after combination chemotherapy for testicular cancer. Ann Intern Med 1981;95:288–292. [DOI] [PubMed] [Google Scholar]
- 30.Clarke CS, Otridge BW, Carney DN. Thromboembolism: a complication of weekly chemotherapy in the treatment of non-Hodgkin’s lymphoma. Cancer 1990;66:2027–2030. [DOI] [PubMed] [Google Scholar]
- 31.Numico G, Garrone O, Dongiovanni V, Silvestris N, Colantonio I, Di Costanzo G, Granetto C, Occelli M, Fea E, Heouaine A, Gasco M, Merlano M. Prospective evaluation of major vascular events in patients with nonsmall cell lung carcinoma treated with cisplatin and gemcitabine. Cancer 2005;103:994–999. doi: 10.1002/cncr.20893 [DOI] [PubMed] [Google Scholar]
- 32.Doll DC, List AF, Greco FA, Hainsworth JD, Hande KR, Johnson DH. Acute vascular ischemic events after cisplatin-based combination chemotherapy for germ-cell tumors of the testis. Ann Intern Med 1986;105:48–51. [DOI] [PubMed] [Google Scholar]
- 33.Moore RA, Adel N, Riedel E, Bhutani M, Feldman DR, Tabbara NE, Soff G, Parameswaran R, Hassoun H. High incidence of thromboembolic events in patients treated with cisplatin-based chemotherapy: a large retrospective analysis. J Clin Oncol 2011;29:3466–3473. doi: 10.1200/JCO.2011.35.5669 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Abdel-Razeq H, Mansour A, Abdulelah H, Al-Shwayat A, Makoseh M, Ibrahim M, Abunasser M, Rimawi D, Al-Rabaiah A, Alfar R, Abufara A, Ibrahim A, Bawaliz A, Ismael Y. Thromboembolic events in cancer patients on active treatment with cisplatin-based chemotherapy: another look! Thromb J 2018;16:2. doi: 10.1186/s12959-018-0161-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Nuver J, De Haas EC, Van Zweeden M, Gietema JA, Meijer C. Vascular damage in testicular cancer patients: a study on endothelial activation by bleomycin and cisplatin in vitro. Oncol Rep 2010;23:247–253. [PubMed] [Google Scholar]
- 36.Daher IN, Yeh ET. Vascular complications of selected cancer therapies. Nat Clin Pract Cardiovasc Med 2008;5:797–805. doi: 10.1038/ncpcardio1375 [DOI] [PubMed] [Google Scholar]
- 37.Cazin B, Gorin NC, Laporte JP, Gallet B, Douay L, Lopez M, Najman A, Duhamel G. Cardiac complications after bone marrow transplantation: a report on a series of 63 consecutive transplantations. Cancer 1986;57:2061–2069. [DOI] [PubMed] [Google Scholar]
- 38.Braverman AC, Antin JH, Plappert MT, Cook EF, Lee RT. Cyclophospha mide cardiotoxicity in bone marrow transplantation: a prospective evaluation of new dosing regimens. J Clin Oncol 1991;9:1215–1223. doi: 10.1200/JCO.1991.9.7.1215 [DOI] [PubMed] [Google Scholar]
- 39.Goldberg MA, Antin JH, Guinan EC, Rappeport JM. Cyclophosphamide cardiotoxicity: an analysis of dosing as a risk factor. Blood 1986;68:1114–1118. [PubMed] [Google Scholar]
- 40.Armenian SH, Hudson MM, Mulder RL, Chen MH, Constine LS, Dwyer M, Nathan PC, Tissing WJ, Shankar S, Sieswerda E, Skinner R, Steinberger J, van Dalen EC, van der Pal H, Wallace WH, Levitt G, Kremer LC; International Late Effects of Childhood Cancer Guide-line Harmonization Group. Recommendations for cardiomyopathy surveillance for survivors of childhood cancer: a report from the International Late Effects of Childhood Cancer Guideline Harmonization Group. Lancet Oncol 2015;16:e123–e136. doi: 10.1016/S1470-2045(14)70409-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Lipshultz SE, Franco VI, Miller TL, Colan SD, Sallan SE. Cardiovascular disease in adult survivors of childhood cancer. Annu Rev Med 2015;66:161–176. doi: 10.1146/annurev-med-070213-054849 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lipshultz SE, Colan SD, Gelber RD, Perez-Atayde AR, Sallan SE, Sanders SP. Late cardiac effects of doxorubicin therapy for acute lymphoblastic leukemia in childhood. N Engl J Med 1991;324:808–815. doi: 10.1056/NEJM199103213241205 [DOI] [PubMed] [Google Scholar]
- 43.Vejpongsa P, Yeh ET. Prevention of anthracycline-induced cardiotoxicity: challenges and opportunities. J Am Coll Cardiol 2014;64:938–945. doi: 10.1016/j.jacc.2014.06.1167 [DOI] [PubMed] [Google Scholar]
- 44.Ulu N, Buikema H, van Gilst WH, Navis G. Vascular dysfunction in adriamycin nephrosis: different effects of adriamycin exposure and nephrosis. Nephrol Dial Transplant 2008;23:1854–1860. doi: 10.1093/ndt/gfm911 [DOI] [PubMed] [Google Scholar]
- 45.Chow AY, Chin C, Dahl G, Rosenthal DN. Anthracyclines cause endothelial injury in pediatric cancer patients: a pilot study. J Clin Oncol 2006;24:925–928. doi: 10.1200/JCO.2005.03.5956 [DOI] [PubMed] [Google Scholar]
- 46.Duquaine D, Hirsch GA, Chakrabarti A, Han Z, Kehrer C, Brook R, Joseph J, Schott A, Kalyanaraman B, Vasquez-Vivar J, Rajagopalan S. Rapid-onset endothelial dysfunction with adriamycin: evidence for a dysfunctional nitric oxide synthase. Vasc Med 2003;8:101–107. doi: [DOI] [PubMed] [Google Scholar]
- 47.Dengel DR, Ness KK, Glasser SP, Williamson EB, Baker KS, Gurney JG. Endothelial function in young adult survivors of childhood acute lymphoblastic leukemia. J Pediatr Hematol Oncol 2008;30:20–25. doi: 10.1097/MPH.0b013e318159a593 [DOI] [PubMed] [Google Scholar]
- 48.Mabeta P, Pepper MS. A comparative study on the anti-angiogenic effects of DNA-damaging and cytoskeletal-disrupting agents. Angiogenesis 2009;12:81–90. doi: 10.1007/s10456-009-9134-8 [DOI] [PubMed] [Google Scholar]
- 49.Singh S, De Trafford JC, Baskerville PA, Martin JF. Response of digital arteries to endothelium dependent and independent vasodilators in patients with Raynaud’s phenomenon. Eur J Clin Invest 1995;25:182–185. [DOI] [PubMed] [Google Scholar]
- 50.van den Belt-Dusebout AW, Nuver J, de Wit R, Gietema JA, ten Bokkel Huinink WW, Rodrigus PT, Schimmel EC, Aleman BM, van Leeuwen FE. Long-term risk of cardiovascular disease in 5-year survivors of testicular cancer. J Clin Oncol 2006;24:467–475. doi: 10.1200/JCO.2005.02.7193 [DOI] [PubMed] [Google Scholar]
- 51.Van Rheen Z, Fattman C, Domarski S, Majka S, Klemm D, Stenmark KR, Nozik-Grayck E. Lung extracellular superoxide dismutase overexpression lessens bleomycin-induced pulmonary hypertension and vascular remodeling. Am J Respir Cell Mol Biol 2011;44:500–508. doi: 10.1165/rcmb.2010-0065OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Glendenning JL, Barbachano Y, Norman AR, Dearnaley DP, Horwich A, Huddart RA. Long-term neurologic and peripheral vascular toxicity after chemotherapy treatment of testicular cancer. Cancer 2010;116:2322–2331. doi: 10.1002/cncr.24981 [DOI] [PubMed] [Google Scholar]
- 53.Slamon DJ, Leyland-Jones B, Shak S, Fuchs H, Paton V, Bajamonde A, Fleming T, Eiermann W, Wolter J, Pegram M, Baselga J, Norton L. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N Engl J Med 2001;344:783–792. doi: 10.1056/NEJM200103153441101 [DOI] [PubMed] [Google Scholar]
- 54.Romond EH, Jeong JH, Rastogi P, Swain SM, Geyer CE Jr, Ewer MS, Rathi V, Fehrenbacher L, Brufsky A, Azar CA, Flynn PJ, Zapas JL, Polikoff J, Gross HM, Biggs DD, Atkins JN, Tan-Chiu E, Zheng P, Yothers G, Mamounas EP, Wolmark N. Seven-year follow-up assessment of cardiac function in NSABP B-31, a randomized trial comparing doxorubicin and cyclophosphamide followed by paclitaxel (ACP) with ACP plus trastuzumab as adjuvant therapy for patients with node-positive, human epidermal growth factor receptor 2-positive breast cancer. J Clin Oncol 2012;30:3792–3799. doi: 10.1200/JCO.2011.40.0010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Slamon D, Eiermann W, Robert N, Pienkowski T, Martin M, Press M, Mackey J, Glaspy J, Chan A, Pawlicki M, Pinter T, Valero V, Liu MC, Sauter G, von Minckwitz G, Visco F, Bee V, Buyse M, Bendahmane B, Tabah-Fisch I, Lindsay MA, Riva A, Crown J; Breast Cancer International Research Group. Adjuvant trastuzumab in HER2-positive breast cancer. N Engl J Med 2011;365:1273–1283. doi: 10.1056/NEJMoa0910383 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Dang C, Guo H, Najita J, Yardley D, Marcom K, Albain K, Rugo H, Miller K, Ellis M, Shapira I, Wolff AC, Carey LA, Moy B, Groarke J, Moslehi J, Krop I, Burstein HJ, Hudis C, Winer EP, Tolaney SM. Cardiac outcomes of patients receiving adjuvant weekly paclitaxel and trastuzumab for nodenegative, ERBB2-positive breast cancer. JAMA Oncol 2016;2:29–36. doi: 10.1001/jamaoncol.2015.3709 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Gianni L, Pienkowski T, Im YH, Roman L, Tseng LM, Liu MC, Lluch A, Staroslawska E, de la Haba-Rodriguez J, Im SA, Pedrini JL, Poirier B, Morandi P, Semiglazov V, Srimuninnimit V, Bianchi G, Szado T, Ratnayake J, Ross G, Valagussa P. Efficacy and safety of neoadjuvant pertuzumab and trastuzumab in women with locally advanced, inflammatory, or early HER2-positive breast cancer (NeoSphere): a randomised multicentre, open-label, phase 2 trial. Lancet Oncol 2012;13:25–32. doi: 10.1016/S1470-2045(11)70336-9 [DOI] [PubMed] [Google Scholar]
- 58.Lenihan D, Suter T, Brammer M, Neate C, Ross G, Baselga J. Pooled analysis of cardiac safety in patients with cancer treated with pertuzumab. Ann Oncol 2012;23:791–800. doi: 10.1093/annonc/mdr294 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Odiete O, Hill MF, Sawyer DB. Neuregulin in cardiovascular development and disease. Circ Res 2012;111:1376–1385. doi: 10.1161/CIRCRESAHA.112.267286 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Hedhli N, Dobrucki LW, Kalinowski A, Zhuang ZW, Wu X, Russell RR 3rd, Sinusas AJ, Russell KS. Endothelial-derived neuregulin is an important mediator of ischaemia-induced angiogenesis and arteriogenesis. Cardiovasc Res 2012;93:516–524. doi: 10.1093/cvr/cvr352 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Hedhli N, Huang Q, Kalinowski A, Palmeri M, Hu X, Russell RR, Russell KS. Endothelium-derived neuregulin protects the heart against ischemic injury. Circulation 2011;123:2254–2262. doi: 10.1161/CIRCULATIONAHA.110.991125 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Baluna R, Vitetta ES. Vascular leak syndrome: a side effect of immunotherapy. Immunopharmacology 1997;37:117–132. [DOI] [PubMed] [Google Scholar]
- 63.Assier E, Jullien V, Lefort J, Moreau JL, Di Santo JP, Vargaftig BB, Lapa e Silva JR, Thèze J. NK cells and polymorphonuclear neutrophils are both critical for IL-2-induced pulmonary vascular leak syndrome. J Immunol 2004;172:7661–7668. [DOI] [PubMed] [Google Scholar]
- 64.Damle NK, Doyle LV. IL-2-activated human killer lymphocytes but not their secreted products mediate increase in albumin flux across cultured endothelial monolayers: implications for vascular leak syndrome. J Immunol 1989;142:2660–2669. [PubMed] [Google Scholar]
- 65.Baluna R, Rizo J, Gordon BE, Ghetie V, Vitetta ES. Evidence for a structural motif in toxins and interleukin-2 that may be responsible for binding to endothelial cells and initiating vascular leak syndrome. Proc Natl Acad Sci USA 1999;96:3957–3962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Cutter DJ, Darby SC, Yusuf SW. Risks of heart disease after radiotherapy. Tex Heart Inst J 2011;38:257–258. [PMC free article] [PubMed] [Google Scholar]
- 67.Piedbois P, Becquemin JP, Blanc I, Mazeron JJ, Lange F, Melliere D, Le Bourgeois JP. Arterial occlusive disease after radiotherapy: a report of fourteen cases. Radiother Oncol 1990;17:133–140. [DOI] [PubMed] [Google Scholar]
- 68.Basavaraju SR, Easterly CE. Pathophysiological effects of radiation on atherosclerosis development and progression, and the incidence of cardiovascular complications. Med Phys 2002;29:2391–2403. doi: 10.1118/1.1509442 [DOI] [PubMed] [Google Scholar]
- 69.Beckman JA, Thakore A, Kalinowski BH, Harris JR, Creager MA. Radiation therapy impairs endothelium-dependent vasodilation in humans. J Am Coll Cardiol 2001;37:761–765. [DOI] [PubMed] [Google Scholar]
- 70.Hendry JH, Akahoshi M, Wang LS, Lipshultz SE, Stewart FA, Trott KR. Radiation-induced cardiovascular injury. Radiat Environ Biophys 2008;47:189–193. doi: 10.1007/s00411-007-0155-7 [DOI] [PubMed] [Google Scholar]
- 71.Seemann I, Gabriels K, Visser NL, Hoving S, te Poele JA, Pol JF, Gijbels MJ, Janssen BJ, van Leeuwen FW, Daemen MJ, Heeneman S, Stewart FA. Irradiation induced modest changes in murine cardiac function despite progressive structural damage to the myocardium and microvasculature. Radiother Oncol 2012;103:143–150. doi: 10.1016/j.radonc.2011.10.011 [DOI] [PubMed] [Google Scholar]
- 72.Tapio S. Pathology and biology of radiation-induced cardiac disease. J Radiat Res 2016;57:439–448. doi: 10.1093/jrr/rrw064 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Zidar N, Ferluga D, Hvala A, Popović M, Soba E. Contribution to the pathogenesis of radiation-induced injury to large arteries. J Laryngol Otol 1997;111:988–990. [DOI] [PubMed] [Google Scholar]
- 74.Kalman PG, Lipton IH, Provan JL, Walker PM, Miles JT, Yeung HP. Radiation damage to large arteries. Can J Surg 1983;26:88–91. [PubMed] [Google Scholar]
- 75.Reinders JG, Heijmen BJ, Olofsen-van Acht MJ, van Putten WL, Levendag PC. Ischemic heart disease after mantlefield irradiation for Hodgkin’s disease in long-term follow-up. Radiother Oncol 1999;51:35–42. [DOI] [PubMed] [Google Scholar]
- 76.Hancock SL, Tucker MA, Hoppe RT. Factors affecting late mortality from heart disease after treatment of Hodgkin’s disease. JAMA 1993;270:1949–1955. [PubMed] [Google Scholar]
- 77.Heidenreich PA, Schnittger I, Strauss HW, Vagelos RH, Lee BK, Mariscal CS, Tate DJ, Horning SJ, Hoppe RT, Hancock SL. Screening for coronary artery disease after mediastinal irradiation for Hodgkin’s disease. J Clin Oncol 2007;25:43–49. doi: 10.1200/JCO.2006.07.0805 [DOI] [PubMed] [Google Scholar]
- 78.Darby SC, Ewertz M, McGale P, Bennet AM, Blom-Goldman U, Brønnum D, Correa C, Cutter D, Gagliardi G, Gigante B, Jensen MB, Nisbet A, Peto R, Rahimi K, Taylor C, Hall P. Risk of ischemic heart disease in women after radiotherapy for breast cancer. N Engl J Med 2013;368:987–998. doi: 10.1056/NEJMoa1209825 [DOI] [PubMed] [Google Scholar]
- 79.Henson KE, McGale P, Taylor C, Darby SC. Radiation-related mortality from heart disease and lung cancer more than 20 years after radiotherapy for breast cancer. Br J Cancer 2013;108:179–182. doi: 10.1038/bjc.2012.575 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Plummer C, Henderson RD, O’Sullivan JD, Read SJ. Ischemic stroke and transient ischemic attack after head and neck radiotherapy: a review. Stroke 2011;42:2410–2418. doi: 10.1161/STROKEAHA.111.615203 [DOI] [PubMed] [Google Scholar]
- 81.De Bruin ML, Dorresteijn LD, van’t Veer MB, Krol AD, van der Pal HJ, Kappelle AC, Boogerd W, Aleman BM, van Leeuwen FE. Increased risk of stroke and transient ischemic attack in 5-year survivors of Hodgkin lymphoma. J Natl Cancer Inst 2009;101:928–937. doi: 10.1093/jnci/djp147 [DOI] [PubMed] [Google Scholar]
- 82.Groarke JD, Nguyen PL, Nohria A, Ferrari R, Cheng S, Moslehi J. Cardio-vascular complications of radiation therapy for thoracic malignancies: the role for non-invasive imaging for detection of cardiovascular disease. Eur Heart J 2014;35:612–623. doi: 10.1093/eurheartj/eht114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Franklin WJ, Strickman NE, Hall RJ. Stent deployment for peripheral venous stenosis as a result of radiation therapy. Catheter Cardiovasc Interv 2003;59:60–62. doi: 10.1002/ccd.10488 [DOI] [PubMed] [Google Scholar]
- 84.Zhou W, Bush RL, Lin PH, Lumsden AB. Radiation-associated venous stenosis: endovascular treatment options. J Vasc Surg 2004;40:179–182. doi: 10.1016/j.jvs.2004.03.039 [DOI] [PubMed] [Google Scholar]
- 85.Fakhouri F, La Batide Alanore A, Rérolle JP, Guéry B, Raynaud A, Plouin PF. Presentation and revascularization outcomes in patients with radiation-induced renal artery stenosis. Am J Kidney Dis 2001;38:302–309. [DOI] [PubMed] [Google Scholar]
- 86.van Nimwegen FA, Schaapveld M, Cutter DJ, Janus CP, Krol AD, Hauptmann M, Kooijman K, Roesink J, van der Maazen R, Darby SC, Aleman BM, van Leeuwen FE. Radiation dose-response relationship for risk of coronary heart disease in survivors of Hodgkin lymphoma. J Clin Oncol 2016;34:235–243. doi: 10.1200/JCO.2015.63.4444 [DOI] [PubMed] [Google Scholar]
- 87.Weintraub NL, Jones WK, Manka D. Understanding radiation-induced vascular disease. J Am Coll Cardiol 2010;55:1237–1239. doi: 10.1016/j.jacc.2009.11.053 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Lancellotti P, Nkomo VT, Badano LP, Bergler-Klein J, Bergler J, Bogaert J, Davin L, Cosyns B, Coucke P, Dulgheru R, Edvardsen T, Gaemperli O, Galderisi M, Griffin B, Heidenreich PA, Nieman K, Plana JC, Port SC, Scherrer-Crosbie M, Schwartz RG, Sebag IA, Voigt JU, Wann S, Yang PC; European Society of Cardiology Working Groups on Nuclear Cardiology and Cardiac Computed Tomography and Cardiovascular Magnetic Resonance; American Society of Nuclear Cardiology, Society for Cardiovascular Magnetic Resonance, and Society of Cardiovascular Computed Tomography. Expert consensus for multi-modality imaging evaluation of cardiovascular complications of radiotherapy in adults: a report from the European Association of Cardiovascular Imaging and the American Society of Echocardiography. J Am Soc Echocardiogr 2013;26:1013–1032. doi: 10.1016/j.echo.2013.07.005 [DOI] [PubMed] [Google Scholar]
- 89.Kamath MV, Halton J, Harvey A, Turner-Gomes S, McArthur A, Barr RD. Cardiac autonomic dysfunction in survivors of acute lymphoblastic leukemia in childhood. Int J Oncol 1998;12:635–640. [DOI] [PubMed] [Google Scholar]
- 90.Sharabi Y, Dendi R, Holmes C, Goldstein DS. Baroreflex failure as a late sequela of neck irradiation. Hypertension 2003;42:110–116. doi: 10.1161/01.HYP.0000077441.45309.08 [DOI] [PubMed] [Google Scholar]
- 91.Groarke JD, Tanguturi VK, Hainer J, Klein J, Moslehi JJ, Ng A, Forman DE, Di Carli MF, Nohria A. Abnormal exercise response in long-term survivors of hodgkin lymphoma treated with thoracic irradiation: evidence of cardiac autonomic dysfunction and impact on outcomes. J Am Coll Cardiol 2015;65:573–583. doi: 10.1016/j.jacc.2014.11.035 [DOI] [PubMed] [Google Scholar]
- 92.Adams MJ, Lipsitz SR, Colan SD, Tarbell NJ, Treves ST, Diller L, Greenbaum N, Mauch P, Lipshultz SE. Cardiovascular status in long-term survivors of Hodgkin’s disease treated with chest radiotherapy. J Clin Oncol 2004;22:3139–3148. doi: 10.1200/JCO.2004.09.109 [DOI] [PubMed] [Google Scholar]
- 93.Zamorano JL, Lancellotti P, Rodriguez Muñoz D, Aboyans V, Asteggiano R, Galderisi M, Habib G, Lenihan DJ, Lip GYH, Lyon AR, Lopez Fernandez T, Mohty D, Piepoli MF, Tamargo J, Torbicki A, Suter TM; ESC Scientific Document Group. 2016 ESC position paper on cancer treatments and cardiovascular toxicity developed under the auspices of the ESC Committee for Practice Guidelines: the Task Force for Cancer Treatments and Cardiovascular Toxicity of the European Society of Cardiology (ESC). Eur Heart J 2016;37:2768–2801. doi: 10.1093/eurheartj/ehw211 [DOI] [PubMed] [Google Scholar]
- 94.Lipshultz SE, Adams MJ, Colan SD, Constine LS, Herman EH, Hsu DT, Hudson MM, Kremer LC, Landy DC, Miller TL, Oeffinger KC, Rosenthal DN, Sable CA, Sallan SE, Singh GK, Steinberger J, Cochran TR, Wilkinson JD; on behalf of the American Heart Association Congenital Heart Defects Committee of the Council on Cardiovascular Disease in the Young, Council on Basic Cardiovascular Sciences, Council on Cardiovascular and Stroke Nursing, Council on Cardiovascular Radiolo. Long-term cardiovascular toxicity in children, adolescents, and young adults who receive cancer therapy: pathophysiology, course, monitoring, management, prevention, and research directions: a scientific statement from the American Heart Association [published correction appears in Circulation. 2013;128:e394]. Circulation 2013;128:1927–1995. doi: 10.1161/CIR.0b013e3182a88099 [DOI] [PubMed] [Google Scholar]
- 95.Reed GW, Masri A, Griffin BP, Kapadia SR, Ellis SG, Desai MY. Long-term mortality in patients with radiation-associated coronary artery disease treated with percutaneous coronary intervention. Circ Cardiovasc Interv 2016;9:e003483. doi: 10.1161/CIRCINTERVENTIONS.115.003483 [DOI] [PubMed] [Google Scholar]
- 96.Wu W, Masri A, Popovic ZB, Smedira NG, Lytle BW, Marwick TH, Griffin BP, Desai MY. Long-term survival of patients with radiation heart disease undergoing cardiac surgery: a cohort study. Circulation 2013;127:1476–1485. doi: 10.1161/CIRCULATIONAHA.113.001435 [DOI] [PubMed] [Google Scholar]
- 97.Donnellan E, Masri A, Johnston DR, Pettersson GB, Rodriguez LL, Popovic ZB, Roselli EE, Smedira NG, Svensson LG, Griffin BP, Desai MY. Long-term outcomes of patients with mediastinal radiation-associated severe aortic stenosis and subsequent surgical aortic valve replacement: a matched cohort study. J Am Heart Assoc 2017;6:e005396. doi: 10.1161/JAHA.116.005396 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Yu SC, Zou WX, Soo YO, Wang L, Hui JW, Chan AY, Lee KT, Ip VH, Fan FS, Chan AL, Wong LK, Leung TW. Evaluation of carotid angioplasty and stenting for radiation-induced carotid stenosis. Stroke 2014;45:1402–2407. doi: 10.1161/STROKEAHA.113.003995 [DOI] [PubMed] [Google Scholar]
- 99.Bruzzaniti V, Abate A, Pinnaro P, D’Andrea M, Infusino E, Landoni V, Soriani A, Giordano C, Ferraro A, Strigari L. Dosimetric and clinical advantages of deep inspiration breath-hold (DIBH) during radiotherapy of breast cancer. J Exp Clin Cancer Res 2013;32:88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Pedersen AN, Korreman S, Nystrom H, Specht L. Breathing adapted radiotherapy of breast cancer: reduction of cardiac and pulmonary doses using voluntary inspiration breath-hold. Radiother Oncol 2004;72:53–60. [DOI] [PubMed] [Google Scholar]
- 101.Hodgson DC. Late effects in the era of modern therapy for Hodgkin lymphoma. Hematology Am Soc Hematol Educ Program 2011;2011:323–329. doi: 10.1182/asheducation-2011.1.323 [DOI] [PubMed] [Google Scholar]
- 102.Beck RE, Kim L, Yue NJ, Haffty BG, Khan AJ, Goyal S. Treatment techniques to reduce cardiac irradiation for breast cancer patients treated with breast-conserving surgery and radiation therapy: a review. Front Oncol 2014;4:327. doi: 10.3389/fonc.2014.00327 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Krause DS, Van Etten RA. Tyrosine kinases as targets for cancer therapy. N Engl J Med 2005;353:172–187. doi: 10.1056/NEJMra044389 [DOI] [PubMed] [Google Scholar]
- 104.Druker BJ, Talpaz M, Resta DJ, Peng B, Buchdunger E, Ford JM, Lydon NB, Kantarjian H, Capdeville R, Ohno-Jones S, Sawyers CL. Efficacy and safety of a specific inhibitor of the BCR-ABL tyrosine kinase in chronic myeloid leukemia. N Engl J Med 2001;344:1031–1037. doi: 10.1056/NEJM200104053441401 [DOI] [PubMed] [Google Scholar]
- 105.Hochhaus A, Larson RA, Guilhot F, Radich JP, Branford S, Hughes TP, Baccarani M, Deininger MW, Cervantes F, Fujihara S, Ortmann CE, Menssen HD, Kantarjian H, O’Brien SG, Druker BJ; IRIS Investigators. Long-term outcomes of imatinib treatment for chronic myeloid leukemia. N Engl J Med 2017;376:917–927. doi: 10.1056/NEJMoa1609324 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Bair SM, Choueiri TK, Moslehi J. Cardiovascular complications associated with novel angiogenesis inhibitors: emerging evidence and evolving perspectives. Trends Cardiovasc Med 2013;23:104–113. doi: 10.1016/j.tcm.2012.09.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Moslehi JJ, Deininger M. Tyrosine kinase inhibitor-associated cardiovascular toxicity in chronic myeloid leukemia. J Clin Oncol 2015;33:4210–4218. doi: 10.1200/JCO.2015.62.4718 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Li W, Croce K, Steensma DP, McDermott DF, Ben-Yehuda O, Moslehi J. Vascular and metabolic implications of novel targeted cancer therapies: focus on kinase inhibitors. J Am Coll Cardiol 2015;66:1160–1178. doi: 10.1016/j.jacc.2015.07.025 [DOI] [PubMed] [Google Scholar]
- 109.Pinkhas D, Ho T, Smith S. Assessment of pazopanib-related hypertension, cardiac dysfunction and identify of clinical risk factors for their development. Cardio-oncology 2017;3:5. doi: 10.1186/s40959-017-0024-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Pandey AK, Singhi EK, Arroyo JP, Ikizler TA, Gould ER, Brown J, Beckman JA, Harrison DG, Moslehi J. Mechanisms of VEGF (vascular endothelial growth factor) inhibitor-associated hypertension and vascular disease. Hypertension 2018;71:e1–e8. doi: 10.1161/HYPERTENSIONAHA.117.10271 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Touyz RM, Lang NN, Herrmann J, van den Meiracker AH, Danser AHJ. Recent advances in hypertension and cardiovascular toxicities with vascular endothelial growth factor inhibition. Hypertension 2017;70:220–226. doi: 10.1161/HYPERTENSIONAHA.117.08856 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Nazer B, Humphreys BD, Moslehi J. Effects of novel angiogenesis inhibitors for the treatment of cancer on the cardiovascular system: focus on hypertension. Circulation 2011;124:1687–1691. doi: 10.1161/CIRCULATIONAHA.110.992230 [DOI] [PubMed] [Google Scholar]
- 113.McKay RR, Rodriguez GE, Lin X, Kaymakcalan MD, Hamnvik OP, Sabbisetti VS, Bhatt RS, Simantov R, Choueiri TK. Angiotensin system inhibitors and survival outcomes in patients with metastatic renal cell carcinoma. Clin Cancer Res 2015;21:2471–2479. doi: 10.1158/1078-0432.CCR-14-2332 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Izzedine H, Derosa L, Le Teuff G, Albiges L, Escudier B. Hypertension and angiotensin system inhibitors: impact on outcome in sunitinib-treated patients for metastatic renal cell carcinoma. Ann Oncol 2015;26:1128–1133. doi: 10.1093/annonc/mdv147 [DOI] [PubMed] [Google Scholar]
- 115.Oshima Y, Tanimoto T, Yuji K, Tojo A. Association between aortic dissection and systemic exposure of vascular endothelial growth factor path-way inhibitors in the Japanese Adverse Drug Event Report Database. Circulation 2017;135:815–817. doi: 10.1161/CIRCULATIONAHA.116.025144 [DOI] [PubMed] [Google Scholar]
- 116.Ueta T, Mori H, Kunimatsu A, Yamaguchi T, Tamaki Y, Yanagi Y. Stroke and anti-VEGF therapy. Ophthalmology 2011;118:2093–2093.e2. doi: 10.1016/j.ophtha.2011.06.001 [DOI] [PubMed] [Google Scholar]
- 117.Jang S, Zheng C, Tsai HT, Fu AZ, Barac A, Atkins MB, Freedman AN, Minasian L, Potosky AL. Cardiovascular toxicity after antiangiogenic therapy in persons older than 65 years with advanced renal cell carcinoma. Cancer 2016;122:124–130. doi: 10.1002/cncr.29728 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Nalluri SR, Chu D, Keresztes R, Zhu X, Wu S. Risk of venous thromboembolism with the angiogenesis inhibitor bevacizumab in cancer patients: a meta-analysis. JAMA 2008;300:2277–2285. doi: 10.1001/jama.2008.656 [DOI] [PubMed] [Google Scholar]
- 119.Chu TF, Rupnick MA, Kerkela R, Dallabrida SM, Zurakowski D, Nguyen L, Woulfe K, Pravda E, Cassiola F, Desai J, George S, Morgan JA, Harris DM, Ismail NS, Chen JH, Schoen FJ, Van den Abbeele AD, Demetri GD, Force T, Chen MH. Cardiotoxicity associated with tyrosine kinase inhibitor sunitinib. Lancet 2007;370:2011–2019. doi: 10.1016/S0140-6736(07)61865-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Richards CJ, Je Y, Schutz FA, Heng DY, Dallabrida SM, Moslehi JJ, Choueiri TK. Incidence and risk of congestive heart failure in patients with renal and nonrenal cell carcinoma treated with sunitinib. J Clin Oncol 2011;29:3450–3456. doi: 10.1200/JCO.2010.34.4309 [DOI] [PubMed] [Google Scholar]
- 121.Hall PS, Harshman LC, Srinivas S, Witteles RM. The frequency and severity of cardiovascular toxicity from targeted therapy in advanced renal cell carcinoma patients. JACC Heart Fail 2013;1:72–78. doi: 10.1016/j.jchf.2012.09.001 [DOI] [PubMed] [Google Scholar]
- 122.Motzer RJ, Hutson TE, Cella D, Reeves J, Hawkins R, Guo J, Nathan P, Staehler M, de Souza P, Merchan JR, Boleti E, Fife K, Jin J, Jones R, Uemura H, De Giorgi U, Harmenberg U, Wang J, Sternberg CN, Deen K, McCann L, Hackshaw MD, Crescenzo R, Pandite LN, Choueiri TK. Pazopanib versus sunitinib in metastatic renal-cell carcinoma. N Engl J Med 2013;369:722–731. doi: 10.1056/NEJMoa1303989 [DOI] [PubMed] [Google Scholar]
- 123.May D, Gilon D, Djonov V, Itin A, Lazarus A, Gordon O, Rosenberger C, Keshet E. Transgenic system for conditional induction and rescue of chronic myocardial hibernation provides insights into genomic programs of hibernation. Proc Natl Acad Sci USA 2008;105:282–287. doi: 10.1073/pnas.0707778105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Chintalgattu V, Rees ML, Culver JC, Goel A, Jiffar T, Zhang J, Dunner K Jr, Pati S, Bankson JA, Pasqualini R, Arap W, Bryan NS, Taegtmeyer H, Langley RR, Yao H, Kupferman ME, Entman ML, Dickinson ME, Khakoo AY. Coronary microvascular pericytes are the cellular target of sunitinib malate-induced cardiotoxicity. Sci Transl Med 2013;5:187ra69. doi: 10.1126/scitranslmed.3005066 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Moslehi J, Minamishima YA, Shi J, Neuberg D, Charytan DM, Padera RF, Signoretti S, Liao R, Kaelin WG Jr. Loss of hypoxia-inducible factor prolyl hydroxylase activity in cardiomyocytes phenocopies ischemic cardiomyopathy. Circulation 2010;122:1004–1016. doi: 10.1161/CIRCULATIONAHA.109.922427 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Bekeredjian R, Walton CB, MacCannell KA, Ecker J, Kruse F, Outten JT, Sutcliffe D, Gerard RD, Bruick RK, Shohet RV. Conditional HIF-1alpha expression produces a reversible cardiomyopathy. PLoS One 2010;5:e11693. doi: 10.1371/journal.pone.0011693 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Uraizee I, Cheng S, Moslehi J. Reversible cardiomyopathy associated with sunitinib and sorafenib. N Engl J Med 2011;365:1649–1650. doi: 10.1056/NEJMc1108849 [DOI] [PubMed] [Google Scholar]
- 128.Patel TV, Morgan JA, Demetri GD, George S, Maki RG, Quigley M, Humphreys BD. A preeclampsia-like syndrome characterized by reversible hypertension and proteinuria induced by the multitargeted kinase inhibitors sunitinib and sorafenib. J Natl Cancer Inst 2008;100:282–284. doi: 10.1093/jnci/djm311 [DOI] [PubMed] [Google Scholar]
- 129.Vigneau C, Lorcy N, Dolley-Hitze T, Jouan F, Arlot-Bonnemains Y, Laguerre B, Verhoest G, Goujon JM, Belaud-Rotureau MA, Rioux-Leclercq N. All anti-vascular endothelial growth factor drugs can induce ‘pre-eclampsialike syndrome’: a RARe study. Nephrol Dial Transplant 2014;29:325–332. doi: 10.1093/ndt/gft465 [DOI] [PubMed] [Google Scholar]
- 130.Maynard SE, Min JY, Merchan J, Lim KH, Li J, Mondal S, Libermann TA, Morgan JP, Sellke FW, Stillman IE, Epstein FH, Sukhatme VP, Karumanchi SA. Excess placental soluble fms-like tyrosine kinase 1 (sFlt1) may contribute to endothelial dysfunction, hypertension, and proteinuria in preeclampsia. J Clin Invest 2003;111:649–658. doi: 10.1172/JCI17189 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Patten IS, Rana S, Shahul S, Rowe GC, Jang C, Liu L, Hacker MR, Rhee JS, Mitchell J, Mahmood F, Hess P, Farrell C, Koulisis N, Khankin EV, Burke SD, Tudorache I, Bauersachs J, del Monte F, Hilfiker-Kleiner D, Karumanchi SA, Arany Z. Cardiac angiogenic imbalance leads to peripartum cardiomyopathy. Nature 2012;485:333–338. doi: 10.1038/nature11040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Lassila M, Allen TJ, Cao Z, Thallas V, Jandeleit-Dahm KA, Candido R, Cooper ME. Imatinib attenuates diabetes-associated atherosclerosis. Arterioscler Thromb Vasc Biol 2004;24:935–942. doi: 10.1161/01.ATV.0000124105.39900.db [DOI] [PubMed] [Google Scholar]
- 133.Kim TD, Rea D, Schwarz M, Grille P, Nicolini FE, Rosti G, Levato L, Giles FJ, Dombret H, Mirault T, Labussière H, Lindhorst R, Haverkamp W, Buschmann I, Dörken B, le Coutre PD. Peripheral artery occlusive disease in chronic phase chronic myeloid leukemia patients treated with nilotinib or imatinib. Leukemia 2013;27:1316–1321. doi: 10.1038/leu.2013.70 [DOI] [PubMed] [Google Scholar]
- 134.Montani D, Bergot E, Günther S, Savale L, Bergeron A, Bourdin A, Bouvaist H, Canuet M, Pison C, Macro M, Poubeau P, Girerd B, Natali D, Guignabert C, Perros F, O’Callaghan DS, Jaïs X, Tubert-Bitter P, Zalcman G, Sitbon O, Simonneau G, Humbert M. Pulmonary arterial hypertension in patients treated by dasatinib. Circulation 2012;125:2128–2137. doi: 10.1161/CIRCULATIONAHA.111.079921 [DOI] [PubMed] [Google Scholar]
- 135.Dahlén T, Edgren G, Lambe M, Höglund M, Björkholm M, Sandin F, Själander A, Richter J, Olsson-Strömberg U, Ohm L, Bäck M, Stenke L; Swedish CML Group and the Swedish CML Register Group. Cardiovascular events associated with use of tyrosine kinase inhibitors in chronic myeloid leukemia: a population-based cohort study. Ann Intern Med 2016;165:161–166. doi: 10.7326/M15-2306 [DOI] [PubMed] [Google Scholar]
- 136.Li W, Garcia D, Cornell RF, Gailani D, Laubach J, Maglio ME, Richardson PG, Moslehi J. Cardiovascular and thrombotic complications of novel multiple myeloma therapies: a review. JAMA Oncol 2017;3:980–988. doi: 10.1001/jamaoncol.2016.3350 [DOI] [PubMed] [Google Scholar]
- 137.Valsami S, Ruf W, Leikauf MS, Madon J, Kaech A, Asmis LM. Immunomodulatory drugs increase endothelial tissue factor expression in vitro. Thromb Res 2011;127:264–271. doi: 10.1016/j.thromres.2010.11.018 [DOI] [PubMed] [Google Scholar]
- 138.Palumbo A, Rajkumar SV, Dimopoulos MA, Richardson PG, San Miguel J, Barlogie B, Harousseau J, Zonder JA, Cavo M, Zangari M, Attal M, Belch A, Knop S, Joshua D, Sezer O, Ludwig H, Vesole D, Bladé J, Kyle R, Westin J, Weber D, Bringhen S, Niesvizky R, Waage A, von Lilienfeld-Toal M, Lonial S, Morgan GJ, Orlowski RZ, Shimizu K, Anderson KC, Boccadoro M, Durie BG, Sonneveld P, Hussein MA; International Myeloma Working Group. Prevention of thalidomide- and lenalidomide- associated thrombosis in myeloma. Leukemia 2008;22:414–423. doi: 10.1038/sj.leu.2405062 [DOI] [PubMed] [Google Scholar]
- 139.Callahan MK, Postow MA, Wolchok JD. Targeting T cell co-receptors for cancer therapy. Immunity 2016;44:1069–1078. doi: 10.1016/j.immuni.2016.04.023 [DOI] [PubMed] [Google Scholar]
- 140.Hodi FS, O’Day SJ, McDermott DF, Weber RW, Sosman JA, Haanen JB, Gonzalez R, Robert C, Schadendorf D, Hassel JC, Akerley W, van den Eertwegh AJ, Lutzky J, Lorigan P, Vaubel JM, Linette GP, Hogg D, Ottensmeier CH, Lebbé C, Peschel C, Quirt I, Clark JI, Wolchok JD, Weber JS, Tian J, Yellin MJ, Nichol GM, Hoos A, Urba WJ. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med 2010;363:711–723. doi: 10.1056/NEJMoa1003466 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Läubli H, Hench J, Stanczak M, Heijnen I, Papachristofilou A, Frank S, Zippelius A, Stenner-Liewen F. Cerebral vasculitis mimicking intracranial metastatic progression of lung cancer during PD-1 blockade. J Immunother Cancer 2017;5:46. doi: 10.1186/s40425-017-0249-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Gambichler T, Strutzmann S, Tannapfel A, Susok L. Paraneoplastic acral vascular syndrome in a patient with metastatic melanoma under immune checkpoint blockade. BMC Cancer 2017;17:327. doi: 10.1186/s12885-017-3313-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Johnson DB, Balko JM, Compton ML, Chalkias S, Gorham J, Xu Y, Hicks M, Puzanov I, Alexander MR, Bloomer TL, Becker JR, Slosky DA, Phillips EJ, Pilkinton MA, Craig-Owens L, Kola N, Plautz G, Reshef DS, Deutsch JS, Deering RP, Olenchock BA, Lichtman AH, Roden DM, Seidman CE, Koralnik IJ, Seidman JG, Hoffman RD, Taube JM, Diaz LA Jr, Anders RA, Sosman JA, Moslehi JJ. Fulminant myocarditis with combination immune checkpoint blockade. N Engl J Med 2016;375:1749–1755. doi: 10.1056/NEJMoa1609214 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Wang DY, Okoye GD, Neilan TG, Johnson DB, Moslehi JJ. Cardiovascular toxicities associated with cancer immunotherapies. Curr Cardiol Rep 2017;19:21. doi: 10.1007/s11886-017-0835-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Neelapu SS, Locke FL, Bartlett NL, Lekakis LJ, Miklos DB, Jacobson CA, Braunschweig I, Oluwole OO, Siddiqi T, Lin Y, Timmerman JM, Stiff PJ, Friedberg JW, Flinn IW, Goy A, Hill BT, Smith MR, Deol A, Farooq U, McSweeney P, Munoz J, Avivi I, Castro JE, Westin JR, Chavez JC, Ghobadi A, Komanduri KV, Levy R, Jacobsen ED, Witzig TE, Reagan P, Bot A, Rossi J, Navale L, Jiang Y, Aycock J, Elias M, Chang D, Wiezorek J, Go WY. Axicabtagene ciloleucel CAR t-cell therapy in refractory large B-cell lymphoma. N Engl J Med 2017;377:2531–2544. doi: 10.1056/NEJMoa1707447 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Maude SL, Laetsch TW, Buechner J, Rives S, Boyer M, Bittencourt H, Bader P, Verneris MR, Stefanski HE, Myers GD, Qayed M, De Moerloose B, Hiramatsu H, Schlis K, Davis KL, Martin PL, Nemecek ER, Yanik GA, Peters C, Baruchel A, Boissel N, Mechinaud F, Balduzzi A, Krueger J, June CH, Levine BL, Wood P, Taran T, Leung M, Mueller KT, Zhang Y, Sen K, Lebwohl D, Pulsipher MA, Grupp SA. Tisagenlecleucel in children and young adults with B-cell lymphoblastic leukemia. N Engl J Med 2018;378:439–448. doi: 10.1056/NEJMoa1709866 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Bonifant CL, Jackson HJ, Brentjens RJ, Curran KJ. Toxicity and management in CAR T-cell therapy. Mol Ther Oncolytics 2016;3:16011. doi: 10.1038/mto.2016.11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Grundy SM, Cleeman JI, Daniels SR, Donato KA, Eckel RH, Franklin BA, Gordon DJ, Krauss RM, Savage PJ, Smith SC Jr, Spertus JA, Costa F. Diagnosis and management of the metabolic syndrome: an American Heart Association/National Heart, Lung, and Blood Institute scientific statement [published corrections appear in Circulation. 2005;112:e297 and Circulation. 2005;112:e298]. Circulation 2005;112:2735–2752. doi: 10.1161/CIRCULATIONAHA.105.169404 [DOI] [PubMed] [Google Scholar]
- 149.Huggins C Prostatic cancer treated by orchiectomy; the five year results. J Am Med Assoc 1946;131:576–581. [DOI] [PubMed] [Google Scholar]
- 150.Keating NL, O’Malley AJ, Smith MR. Diabetes and cardiovascular disease during androgen deprivation therapy for prostate cancer. J Clin Oncol 2006;24:4448–4456. doi: 10.1200/JCO.2006.06.2497 [DOI] [PubMed] [Google Scholar]
- 151.Keating NL, O’Malley AJ, Freedland SJ, Smith MR. Diabetes and cardiovascular disease during androgen deprivation therapy: observational study of veterans with prostate cancer [published correction appears in J Natl Cancer Inst. 2012;104:1518–1523]. J Natl Cancer Inst 2010;102:39–46. doi: 10.1093/jnci/djp404 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Bosco C, Bosnyak Z, Malmberg A, Adolfsson J, Keating NL, Van Hemelrijck M. Quantifying observational evidence for risk of fatal and nonfatal cardiovascular disease following androgen deprivation therapy for prostate cancer: a meta-analysis. Eur Urol 2015;68:386–396. doi: 10.1016/j.eururo.2014.11.039 [DOI] [PubMed] [Google Scholar]
- 153.Nguyen PL, Je Y, Schutz FA, Hoffman KE, Hu JC, Parekh A, Beckman JA, Choueiri TK. Association of androgen deprivation therapy with cardiovascular death in patients with prostate cancer: a meta-analysis of randomized trials. JAMA 2011;306:2359–2366. doi: 10.1001/jama.2011.1745 [DOI] [PubMed] [Google Scholar]
- 154.Nguyen PL, Chen MH, Beckman JA, Beard CJ, Martin NE, Choueiri TK, Hu JC, Hoffman KE, Dosoretz DE, Moran BJ, Salenius SA, Braccioforte MH, Kantoff PW, D’Amico AV, Ennis RD. Influence of androgen deprivation therapy on all-cause mortality in men with high-risk prostate cancer and a history of congestive heart failure or myocardial infarction. Int J Radiat Oncol Biol Phys 2012;82:1411–1416. doi: 10.1016/j.ijrobp.2011.04.067 [DOI] [PubMed] [Google Scholar]
- 155.Parekh A, Chen MH, D’Amico AV, Dosoretz DE, Ross R, Salenius S, Graham PL, Beckman JA, Beard CJ, Choueiri TK, Ennis RD, Hoffman KE, Hu JC, Ma J, Martin NE, Nguyen PL. Identification of comorbidities that place men at highest risk of death from androgen deprivation therapy before brachytherapy for prostate cancer. Brachytherapy 2013;12:415–421. doi: 10.1016/j.brachy.2013.02.005 [DOI] [PubMed] [Google Scholar]
- 156.Ziehr DR, Chen MH, Zhang D, Braccioforte MH, Moran BJ, Mahal BA, Hyatt AS, Basaria SS, Beard CJ, Beckman JA, Choueiri TK, D’Amico AV, Hoffman KE, Hu JC, Martin NE, Sweeney CJ, Trinh QD, Nguyen PL. Association of androgen-deprivation therapy with excess cardiac-specific mortality in men with prostate cancer. BJU Int 2015;116:358–365. doi: 10.1111/bju.12905 [DOI] [PubMed] [Google Scholar]
- 157.Haidar A, Yassin A, Saad F, Shabsigh R. Effects of androgen deprivation on glycaemic control and on cardiovascular biochemical risk factors in men with advanced prostate cancer with diabetes. Aging Male 2007;10:189–196. doi: 10.1080/13685530701653538 [DOI] [PubMed] [Google Scholar]
- 158.Smith MR, O’Malley AJ, Keating NL. Gonadotrophin-releasing hormone agonists, diabetes and cardiovascular disease in men with prostate cancer: which metabolic syndrome? BJU Int 2008;101:1335–1336. doi: 10.1111/j.1464-410X.2008.07707.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Saylor PJ, Keating NL, Freedland SJ, Smith MR. Gonadotropin-releasing hormone agonists and the risks of type 2 diabetes and cardiovascular disease in men with prostate cancer. Drugs 2011;71:255–261. doi: 10.2165/11588930-000000000-00000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Collier A, Ghosh S, McGlynn B, Hollins G. Prostate cancer, androgen deprivation therapy, obesity, the metabolic syndrome, type 2 diabetes, and cardiovascular disease: a review. Am J Clin Oncol 2012;35:504–509. doi: 10.1097/COC.0b013e318201a406 [DOI] [PubMed] [Google Scholar]
- 161.Keating NL, Liu PH, O’Malley AJ, Freedland SJ, Smith MR. Androgendeprivation therapy and diabetes control among diabetic men with prostate cancer. Eur Urol 2014;65:816–824. doi: 10.1016/j.eururo.2013.02.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Mohamedali HZ, Breunis H, Timilshina N, Alibhai SM. Changes in blood glucose and cholesterol levels due to androgen deprivation therapy in men with non-metastatic prostate cancer. Can Urol Assoc J 2011;5:28–32. doi: 10.5489/cuaj.09172 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Sağlam HS, Köse O, Kumsar S, Budak S, Adsan O. Fasting blood glucose and lipid profile alterations following twelve-month androgen deprivation therapy in men with prostate cancer. ScientificWorldJournal 2012;2012:696329. doi: 10.1100/2012/696329 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Salvador C, Planas J, Agreda F, Placer J, Trilla E, Lopez MA, Morote J. Analysis of the lipid profile and atherogenic risk during androgen deprivation therapy in prostate cancer patients. Urol Int 2013;90:41–44. doi: 10.1159/000342814 [DOI] [PubMed] [Google Scholar]
- 165.Oka R, Utsumi T, Endo T, Yano M, Kamijima S, Kamiya N, Shirai K, Suzuki H. Effect of androgen deprivation therapy on arterial stiffness and serum lipid profile changes in patients with prostate cancer: a prospective study of initial 6-month follow-up. Int J Clin Oncol 2016;21:389–396. doi: 10.1007/s10147-015-0891-7 [DOI] [PubMed] [Google Scholar]
- 166.Mitsuzuka K, Kyan A, Sato T, Orikasa K, Miyazato M, Aoki H, Kakoi N, Narita S, Koie T, Namima T, Toyoda S, Fukushi Y, Habuchi T, Ohyama C, Arai Y; Tohoku Evidence-Based Medicine Study Group; Michinoku Urological Cancer Study Group. Influence of 1 year of androgen deprivation therapy on lipid and glucose metabolism and fat accumulation in Japanese patients with prostate cancer. Prostate Cancer Prostatic Dis 2016;19:57–62. doi: 10.1038/pcan.2015.50 [DOI] [PubMed] [Google Scholar]
- 167.Gilbert SE, Tew GA, Bourke L, Winter EM, Rosario DJ. Assessment of endothelial dysfunction by flow-mediated dilatation in men on long-term androgen deprivation therapy for prostate cancer. Exp Physiol 2013;98:1401–1410. doi: 10.1113/expphysiol.2013.073353 [DOI] [PubMed] [Google Scholar]
- 168.Herman SM, Robinson JT, McCredie RJ, Adams MR, Boyer MJ, Celermajer DS. Androgen deprivation is associated with enhanced endothelium-dependent dilatation in adult men. Arterioscler Thromb Vasc Biol 1997;17:2004–2009. [DOI] [PubMed] [Google Scholar]
- 169.Nguyen PL, Jarolim P, Basaria S, Zuflacht JP, Milian J, Kadivar S, Graham PL, Hyatt A, Kantoff PW, Beckman JA. Androgen deprivation therapy reversibly increases endothelium-dependent vasodilation in men with prostate cancer. J Am Heart Assoc 2015;4:e001914. doi: 10.1161/JAHA.115.001914 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Patel TB. Single transmembrane spanning heterotrimeric g protein-coupled receptors and their signaling cascades. Pharmacol Rev 2004;56:371–385. doi: 10.1124/pr.56.3.4 [DOI] [PubMed] [Google Scholar]
- 171.Patnaik A, Appleman LJ, Tolcher AW, Papadopoulos KP, Beeram M, Rasco DW, Weiss GJ, Sachdev JC, Chadha M, Fulk M, Ejadi S, Mountz JM, Lotze MT, Toledo FG, Chu E, Jeffers M, Peña C, Xia C, Reif S, Genvresse I, Ramanathan RK. First-in-human phase I study of copanlisib (BAY 80– 6946), an intravenous pan-class I phosphatidylinositol 3-kinase inhibitor, in patients with advanced solid tumors and non-Hodgkin’s lymphomas. Ann Oncol 2016;27:1928–1940. doi: 10.1093/annonc/mdw282 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Doi T, Fuse N, Yoshino T, Kojima T, Bando H, Miyamoto H, Kaneko M, Osada M, Ohtsu A. A phase I study of intravenous PI3K inhibitor copanlisib in Japanese patients with advanced or refractory solid tumors. Cancer Chemother Pharmacol 2017;79:89–98. doi: 10.1007/s00280-016-3198-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Dreyling M, Morschhauser F, Bouabdallah K, Bron D, Cunningham D, Assouline SE, Verhoef G, Linton K, Thieblemont C, Vitolo U, Hiemeyer F, Giurescu M, Garcia-Vargas J, Gorbatchevsky I, Liu L, Koechert K, Peña C, Neves M, Childs BH, Zinzani PL. Phase II study of copanlisib, a PI3K inhibitor, in relapsed or refractory, indolent or aggressive lymphoma. Ann Oncol 2017;28:2169–2178. doi: 10.1093/annonc/mdx289 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Dreyling M, Santoro A, Mollica L, Leppä S, Follows GA, Lenz G, Kim WS, Nagler A, Panayiotidis P, Demeter J, Özcan M, Kosinova M, Bouabdallah K, Morschhauser F, Stevens DA, Trevarthen D, Giurescu M, Cupit L, Liu L, Köchert K, Seidel H, Peña C, Yin S, Hiemeyer F, Garcia-Vargas J, Childs BH, Zinzani PL. Phosphatidylinositol 3-kinase inhibition by copanlisib in relapsed or refractory indolent lymphoma. J Clin Oncol 2017;35:3898–3905. doi: 10.1200/JCO.2017.75.4648 [DOI] [PubMed] [Google Scholar]
- 175.Billemont B, Medioni J, Taillade L, Helley D, Meric JB, Rixe O, Oudard S. Blood glucose levels in patients with metastatic renal cell carcinoma treated with sunitinib. Br J Cancer 2008;99:1380–1382. doi: 10.1038/sj.bjc.6604709 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Agostino NM, Chinchilli VM, Lynch CJ, Koszyk-Szewczyk A, Gingrich R, Sivik J, Drabick JJ. Effect of the tyrosine kinase inhibitors (sunitinib, sorafenib, dasatinib, and imatinib) on blood glucose levels in diabetic and nondiabetic patients in general clinical practice. J Oncol Pharm Pract 2011;17:197–202. doi: 10.1177/1078155210378913 [DOI] [PubMed] [Google Scholar]
- 177.Oh JJ, Hong SK, Joo YM, Lee BK, Min SH, Lee S, Byun SS, Lee SE. Impact of sunitinib treatment on blood glucose levels in patients with metastatic renal cell carcinoma. Jpn J Clin Oncol 2012;42:314–317. doi: 10.1093/jjco/hys002 [DOI] [PubMed] [Google Scholar]
- 178.Chen J, Wang C, Han J, Luan Y, Cui Y, Shen R, Sha D, Cong L, Zhang Z, Wang W. Therapeutic effect of sunitinib malate and its influence on blood glucose concentrations in a patient with metastatic insulinoma. Expert Rev Anticancer Ther 2013;13:737–743. doi: 10.1586/era.13.45 [DOI] [PubMed] [Google Scholar]
- 179.Lutz SZ, Ullrich A, Häring HU, Ullrich S, Gerst F. Sunitinib specifically augments glucose-induced insulin secretion. Cell Signal 2017;36:91–97. doi: 10.1016/j.cellsig.2017.04.018 [DOI] [PubMed] [Google Scholar]
- 180.Ono K, Suzushima H, Watanabe Y, Kikukawa Y, Shimomura T, Furukawa N, Kawaguchi T, Araki E. Rapid amelioration of hyperglycemia facilitated by dasatinib in a chronic myeloid leukemia patient with type 2 diabetes mellitus. Intern Med 2012;51:2763–2766. [DOI] [PubMed] [Google Scholar]
- 181.Martinez Marignac VL, Smith S, Toban N, Bazile M, Aloyz R. Resistance to Dasatinib in primary chronic lymphocytic leukemia lymphocytes involves AMPK-mediated energetic re-programming. Oncotarget 2013;4:2550–2566. doi: 10.18632/oncotarget.1508 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Deremer DL, Ustun C, Natarajan K. Nilotinib: a second-generation tyrosine kinase inhibitor for the treatment of chronic myelogenous leukemia. Clin Ther 2008;30:1956–1975. doi: 10.1016/j.clinthera.2008.11.014 [DOI] [PubMed] [Google Scholar]
- 183.Racil Z, Razga F, Drapalova J, Buresova L, Zackova D, Palackova M, Semerad L, Malaskova L, Haluzik M, Mayer J. Mechanism of impaired glucose metabolism during nilotinib therapy in patients with chronic myelogenous leukemia. Haematologica 2013;98:e124–e126. doi: 10.3324/haematol.2013.086355 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Breccia M, Muscaritoli M, Gentilini F, Latagliata R, Carmosino I, Rossi Fanelli F, Alimena G. Impaired fasting glucose level as metabolic side effect of nilotinib in non-diabetic chronic myeloid leukemia patients resistant to imatinib. Leuk Res 2007;31:1770–1772. doi: 10.1016/j.leukres.2007.01.024 [DOI] [PubMed] [Google Scholar]
- 185.Blom JW, Vanderschoot JP, Oostindiër MJ, Osanto S, van der Meer FJ, Rosendaal FR. Incidence of venous thrombosis in a large cohort of 66,329 cancer patients: results of a record linkage study. J Thromb Haemost 2006;4:529–535. doi: 10.1111/j.1538-7836.2006.01804.x [DOI] [PubMed] [Google Scholar]
- 186.Cronin-Fenton DP, Søndergaard F, Pedersen LA, Fryzek JP, Cetin K, Acquavella J, Baron JA, Sørensen HT. Hospitalisation for venous thromboembolism in cancer patients and the general population: a population-based cohort study in Denmark, 1997–2006. Br J Cancer 2010;103:947–953. doi: 10.1038/sj.bjc.6605883 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Timp JF, Braekkan SK, Versteeg HH, Cannegieter SC. Epidemiology of cancer-associated venous thrombosis. Blood 2013;122:1712–1723. doi: 10.1182/blood-2013-04-460121 [DOI] [PubMed] [Google Scholar]
- 188.Chew HK, Wun T, Harvey D, Zhou H, White RH. Incidence of venous thromboembolism and its effect on survival among patients with common cancers. Arch Intern Med 2006;166:458–464. doi: 10.1001/archinte.166.4.458 [DOI] [PubMed] [Google Scholar]
- 189.Heit JA, Spencer FA, White RH. The epidemiology of venous thromboembolism. J Thromb Thrombolysis 2016;41:3–14. doi: 10.1007/s11239-015-1311-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Imberti D, Agnelli G, Ageno W, Moia M, Palareti G, Pistelli R, Rossi R, Verso M; MASTER Investigators. Clinical characteristics and management of cancer-associated acute venous thromboembolism: findings from the MASTER Registry. Haematologica 2008;93:273–278. doi: 10.3324/haematol.11458 [DOI] [PubMed] [Google Scholar]
- 191.Prandoni P, Lensing AW, Piccioli A, Bernardi E, Simioni P, Girolami B, Marchiori A, Sabbion P, Prins MH, Noventa F, Girolami A. Recurrent venous thromboembolism and bleeding complications during anticoagulant treatment in patients with cancer and venous thrombosis. Blood 2002;100:3484–3488. doi: 10.1182/blood-2002-01-0108 [DOI] [PubMed] [Google Scholar]
- 192.Trujillo-Santos J, Nieto JA, Tiberio G, Piccioli A, Di Micco P, Prandoni P, Monreal M; RIETE Registry. Predicting recurrences or major bleeding in cancer patients with venous thromboembolism: findings from the RIETE Registry. Thromb Haemost 2008;100:435–439. [PubMed] [Google Scholar]
- 193.Louzada ML, Carrier M, Lazo-Langner A, Dao V, Kovacs MJ, Ramsay TO, Rodger MA, Zhang J, Lee AY, Meyer G, Wells PS. Development of a clinical prediction rule for risk stratification of recurrent venous thromboembolism in patients with cancer-associated venous thromboembolism. Circulation 2012;126:448–454. doi: 10.1161/CIRCULATIONAHA.111.051920 [DOI] [PubMed] [Google Scholar]
- 194.Gussoni G, Frasson S, La Regina M, Di Micco P, Monreal M; RIETE Investigators. Three-month mortality rate and clinical predictors in patients with venous thromboembolism and cancer: findings from the RIETE registry. Thromb Res 2013;131:24–30. doi: 10.1016/j.thromres.2012.10.007 [DOI] [PubMed] [Google Scholar]
- 195.Naess IA, Christiansen SC, Romundstad P, Cannegieter SC, Rosendaal FR, Hammerstrøm J. Incidence and mortality of venous thrombosis: a population-based study. J Thromb Haemost 2007;5:692–699. doi: 10.1111/j.1538-7836.2007.02450.x [DOI] [PubMed] [Google Scholar]
- 196.Kearon C, Akl EA, Ornelas J, Blaivas A, Jimenez D, Bounameaux H, Huisman M, King CS, Morris TA, Sood N, Stevens SM, Vintch JRE, Wells P, Woller SC, Moores L. Antithrombotic therapy for VTE disease: CHEST Guideline and Expert Panel Report. Chest 2016;149:315–352. doi: 10.1016/j.chest.2015.11.026 [DOI] [PubMed] [Google Scholar]
- 197.Lee AY, Levine MN, Baker RI, Bowden C, Kakkar AK, Prins M, Rickles FR, Julian JA, Haley S, Kovacs MJ, Gent M; Randomized Comparison of Low-Molecular-Weight Heparin versus Oral Anticoagulant Therapy for the Prevention of Recurrent Venous Thromboembolism in Patients with Cancer (CLOT) Investigators. Low-molecular-weight heparin versus a coumarin for the prevention of recurrent venous thromboembolism in patients with cancer. N Engl J Med 2003;349:146–153. doi: 10.1056/NEJMoa025313 [DOI] [PubMed] [Google Scholar]
- 198.Meyer G, Marjanovic Z, Valcke J, Lorcerie B, Gruel Y, Solal-Celigny P, Le Maignan C, Extra JM, Cottu P, Farge D. Comparison of low-molecular-weight heparin and warfarin for the secondary prevention of venous thromboembolism in patients with cancer: a randomized controlled study. Arch Intern Med 2002;162:1729–1735. [DOI] [PubMed] [Google Scholar]
- 199.Lee AYY, Kamphuisen PW, Meyer G, Bauersachs R, Janas MS, Jarner MF, Khorana AA; CATCH Investigators. Tinzaparin vs warfarin for treatment of acute venous thromboembolism in patients with active cancer: a randomized clinical trial. JAMA 2015;314:677–686. doi: 10.1001/jama.2015.9243 [DOI] [PubMed] [Google Scholar]
- 200.Vedovati MC, Germini F, Agnelli G, Becattini C. Direct oral anticoagulants in patients with VTE and cancer: a systematic review and meta-analysis. Chest 2015;147:475–483. doi: 10.1378/chest.14-0402 [DOI] [PubMed] [Google Scholar]
- 201.Raskob GE, van Es N, Verhamme P, Carrier M, Di Nisio M, Garcia D, Grosso MA, Kakkar AK, Kovacs MJ, Mercuri MF, Meyer G, Segers A, Shi M, Wang TF, Yeo E, Zhang G, Zwicker JI, Weitz JI, Büller HR; Hokusai VTE Cancer Investigators. Edoxaban for the treatment of cancer-associated venous thromboembolism. N Engl J Med 2018;378:615–624. doi: 10.1056/NEJMoa1711948 [DOI] [PubMed] [Google Scholar]
- 201a.Young AM, Marshall A, Thirlwall J, Chapman O, Lokare A, Hill C, Hale D, Dunn JA, Lyman GH, Hutchinson C, MacCallum P, Kakkar A, Hobbs FDR, Petrou S, Dale J, Poole CJ, Maraveyas A, Levine M. Comparison of an oral factor Xa inhibitor with low molecular weight heparin in patients with cancer with venous thromboembolism: results of a randomized trial (SELECT-D). J Clin Oncol 2018;36:2017–2023. [DOI] [PubMed] [Google Scholar]
- 201b.McBane RD, Wysokinski WE, Le-Rademacher J, Ashrani AA, Tafur AJ, Gundabolu K, Perez-Botero J, Perepu U, Anderson DM, Kuzma C, Leon Ferre R, Henkin S, Lenz C, Loprinzi C. Apixaban, dalteparin, in active cancer associated venous thromboembolism, the ADAM VTE trial. Blood 2018;132 (suppl 1): Abstract 421. http://www.bloodjournal.org/content/132/Suppl_1/421. Accessed January 11, 2019. [Google Scholar]
- 202.Moslehi JJ. Cardiovascular toxic effects of targeted cancer therapies. N Engl J Med 2016;375:1457–1467. doi: 10.1056/NEJMra1100265 [DOI] [PubMed] [Google Scholar]
- 203.Rasmussen-Torvik LJ, Shay CM, Abramson JG, Friedrich CA, Nettleton JA, Prizment AE, Folsom AR. Ideal cardiovascular health is inversely associated with incident cancer: the Atherosclerosis Risk In Communities study. Circulation 2013;127:1270–1275. doi: 10.1161/CIRCULATIONAHA.112.001183 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Carter BD, Abnet CC, Feskanich D, Freedman ND, Hartge P, Lewis CE, Ockene JK, Prentice RL, Speizer FE, Thun MJ, Jacobs EJ. Smoking and mortality: beyond established causes. N Engl J Med 2015;372:631–640. doi: 10.1056/NEJMsa1407211 [DOI] [PubMed] [Google Scholar]
- 205.Danilo C, Frank PG. Cholesterol and breast cancer development. Curr Opin Pharmacol 2012;12:677–682. doi: 10.1016/j.coph.2012.07.009 [DOI] [PubMed] [Google Scholar]
- 206.Hu J, La Vecchia C, de Groh M, Negri E, Morrison H, Mery L; Canadian Cancer Registries Epidemiology Research Group. Dietary cholesterol intake and cancer. Ann Oncol 2012;23:491–500. doi: 10.1093/annonc/mdr155 [DOI] [PubMed] [Google Scholar]
- 207.Nelson ER, Wardell SE, Jasper JS, Park S, Suchindran S, Howe MK, Carver NJ, Pillai RV, Sullivan PM, Sondhi V, Umetani M, Geradts J, McDonnell DP. 27-Hydroxycholesterol links hypercholesterolemia and breast cancer pathophysiology. Science 2013;342:1094–1098. doi: 10.1126/science.1241908 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Ridker PM, Everett BM, Thuren T, MacFadyen JG, Chang WH, Ballantyne C, Fonseca F, Nicolau J, Koenig W, Anker SD, Kastelein JJP, Cornel JH, Pais P, Pella D, Genest J, Cifkova R, Lorenzatti A, Forster T, Kobalava Z, Vida-Simiti L, Flather M, Shimokawa H, Ogawa H, Dellborg M, Rossi PRF, Troquay RPT, Libby P, Glynn RJ; CANTOS Trial Group. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N Engl J Med 2017;377:1119–1131. doi: 10.1056/NEJMoa1707914 [DOI] [PubMed] [Google Scholar]
- 209.Ridker PM, MacFadyen JG, Thuren T, Everett BM, Libby P, Glynn RJ; CANTOS Trial Group. Effect of interleukin-1β inhibition with canakinumab on incident lung cancer in patients with atherosclerosis: exploratory results from a randomised, double-blind, placebo-controlled trial. Lancet 2017;390:1833–1842. doi: 10.1016/S0140-6736(17)32247-X [DOI] [PubMed] [Google Scholar]
- 210.Jaiswal S, Fontanillas P, Flannick J, Manning A, Grauman PV, Mar BG, Lindsley RC, Mermel CH, Burtt N, Chavez A, Higgins JM, Moltchanov V, Kuo FC, Kluk MJ, Henderson B, Kinnunen L, Koistinen HA, Ladenvall C, Getz G, Correa A, Banahan BF, Gabriel S, Kathiresan S, Stringham HM, McCarthy MI, Boehnke M, Tuomilehto J, Haiman C, Groop L, Atzmon G, Wilson JG, Neuberg D, Altshuler D, Ebert BL. Age-related clonal hematopoiesis associated with adverse outcomes. N Engl J Med 2014;371:2488–2498. doi: 10.1056/NEJMoa1408617 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Genovese G, Kähler AK, Handsaker RE, Lindberg J, Rose SA, Bakhoum SF, Chambert K, Mick E, Neale BM, Fromer M, Purcell SM, Svantesson O, Landén M, Höglund M, Lehmann S, Gabriel SB, Moran JL, Lander ES, Sullivan PF, Sklar P, Grönberg H, Hultman CM, McCarroll SA. Clonal hematopoiesis and blood-cancer risk inferred from blood DNA sequence. N Engl J Med 2014;371:2477–2487. doi: 10.1056/NEJMoa1409405 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Jaiswal S, Natarajan P, Silver AJ, Gibson CJ, Bick AG, Shvartz E, McConkey M, Gupta N, Gabriel S, Ardissino D, Baber U, Mehran R, Fuster V, Danesh J, Frossard P, Saleheen D, Melander O, Sukhova GK, Neuberg D, Libby P, Kathiresan S, Ebert BL. Clonal hematopoiesis and risk of atherosclerotic cardiovascular disease. N Engl J Med 2017;377:111–121. doi: 10.1056/NEJMoa1701719 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Fuster JJ, MacLauchlan S, Zuriaga MA, Polackal MN, Ostriker AC, Chakraborty R, Wu CL, Sano S, Muralidharan S, Rius C, Vuong J, Jacob S, Muralidhar V, Robertson AA, Cooper MA, Andrés V, Hirschi KK, Martin KA, Walsh K. Clonal hematopoiesis associated with TET2 deficiency accelerates atherosclerosis development in mice. Science 2017;355:842–847. doi: 10.1126/science.aag1381 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Mehta LS, Watson KE, Barac A, Beckie TM, Bittner V, Cruz-Flores S, Dent S, Kondapalli L, Ky B, Okwuosa T, Piña IL, Volgman AS; on behalf of the American Heart Association Cardiovascular Disease in Women and Special Populations Committee of the Council on Clinical Cardiology; Council on Cardiovascular and Stroke Nursing; and Council on Quality of Care and Outcomes Research. Cardiovascular disease and breast cancer: where these entities intersect: a scientific statement from the American Heart Association. Circulation 2018;137:e30–e66. doi: 10.1161/CIR.0000000000000556 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Sheng CC, Amiri-Kordestani L, Palmby T, Force T, Hong CC, Wu JC, Croce K, Kim G, Moslehi J. 21st Century cardio-oncology: identifying cardiac safety signals in the era of personalized medicine. JACC Basic Transl Sci 2016;1:386–398. doi: 10.1016/j.jacbts.2016.05.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Louvet C, Szot GL, Lang J, Lee MR, Martinier N, Bollag G, Zhu S, Weiss A, Bluestone JA. Tyrosine kinase inhibitors reverse type 1 diabetes in non-obese diabetic mice. Proc Natl Acad Sci USA 2008;105:18895–18900. doi: 10.1073/pnas.0810246105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Pituskin E, Mackey JR, Koshman S, Jassal D, Pitz M, Haykowsky MJ, Pagano JJ, Chow K, Thompson RB, Vos LJ, Ghosh S, Oudit GY, Ezekowitz JA, Paterson DI. Multidisciplinary Approach to Novel Therapies in Cardio-oncology Research (MANTICORE 101-Breast): a randomized trial for the prevention of trastuzumab-associated cardiotoxicity. J Clin Oncol 2017;35:870–877. doi: 10.1200/JCO.2016.68.7830 [DOI] [PubMed] [Google Scholar]
- 218.Gulati G, Heck SL, Ree AH, Hoffmann P, Schulz-Menger J, Fagerland MW, Gravdehaug B, von Knobelsdorff-Brenkenhoff F, Bratland Å, Storås TH, Hagve TA, Røsjø H, Steine K, Geisler J, Omland T. Prevention of Cardiac Dysfunction During Adjuvant Breast Cancer Therapy (PRADA): a 2 × 2 factorial, randomized, placebo-controlled, double-blind clinical trial of candesartan and metoprolol. Eur Heart J 2016;37:1671–1680. doi: 10.1093/eurheartj/ehw022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Barac A, Swain SM. Cardiac protection in HER2-targeted treatment: how should we measure new strategies? JAMA Oncol 2016;2:1037–1039. doi: 10.1001/jamaoncol.2016.0283 [DOI] [PubMed] [Google Scholar]
- 220.Avila MS, Ayub-Ferreira SM, de Barros Wanderley Junior MR, Cruz FDD, Goncalves Brandao SM, Carvalho Rigaud VO, Higushi-Dos-Santos M, Hajjar LA, Filho RK, Hoff PM, Sahade M, Ferrari MSM, de Paula Costa RL, Mano MS, Bittencourt Viana Cruz CB, Abduch MC, Lofrano Alves MS, Guimaraes GV, Issa VS, Bittencourt MS, Bocchi EA. Carvedilol for prevention of chemotherapy related cardiotoxicity: the CECCY trial. J Am Coll Cardiol 2018;71:2281–2290. doi: 10.1016/j.jacc.2018.02.049 [DOI] [PubMed] [Google Scholar]
- 221.Stack JP, Moslehi J, Sayed N, Wu JC. Cancer therapy-induced cardiomyopathy: can human induced pluripotent stem cell modelling help prevent it? [published online January 25, 2018]. Eur Heart J doi: 10.1093/eurheartj/ehx811. https://academic.oup.com/eurheartj/advance-article-abstract/doi/10.1093/eurheartj/ehx811/4825036?redirectedFrom=fulltext [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Chintalgattu V, Ai D, Langley RR, Zhang J, Bankson JA, Shih TL, Reddy AK, Coombes KR, Daher IN, Pati S, Patel SS, Pocius JS, Taffet GE, Buja LM, Entman ML, Khakoo AY. Cardiomyocyte PDGFR-beta signaling is an essential component of the mouse cardiac response to load-induced stress. J Clin Invest 2010;120:472–484. doi: 10.1172/JCI39434 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Snipelisky D, Park JY, Lerman A, Mulvagh S, Lin G, Pereira N, Rodriguez-Porcel M, Villarraga HR, Herrmann J. How to develop a cardio-oncology clinic. Heart Fail Clin 2017;13:347–359. doi: 10.1016/j.hfc.2016.12.011 [DOI] [PubMed] [Google Scholar]
- 224.Okwuosa TM, Akhter N, Williams KA, DeCara JM. Building a cardio-oncology program in a small- to medium-sized, nonprimary cancer center, academic hospital in the USA: challenges and pitfalls. Future Cardiol 2015;11:413–420. doi: 10.2217/FCA.15.43 [DOI] [PubMed] [Google Scholar]
- 225.Barac A, Murtagh G, Carver JR, Chen MH, Freeman AM, Herrmann J, Iliescu C, Ky B, Mayer EL, Okwuosa TM, Plana JC, Ryan TD, Rzeszut AK, Douglas PS. Cardiovascular health of patients with cancer and cancer survivors: a roadmap to the next level. J Am Coll Cardiol 2015;65:2739–2746. doi: 10.1016/j.jacc.2015.04.059 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Armenian SH, Lacchetti C, Barac A, Carver J, Constine LS, Denduluri N, Dent S, Douglas PS, Durand JB, Ewer M, Fabian C, Hudson M, Jessup M, Jones LW, Ky B, Mayer EL, Moslehi J, Oeffinger K, Ray K, Ruddy K, Lenihan D. Prevention and monitoring of cardiac dysfunction in survivors of adult cancers: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol 2017;35:893–911. doi: 10.1200/JCO.2016.70.5400 [DOI] [PubMed] [Google Scholar]
- 227.Miller KD, Siegel RL, Lin CC, Mariotto AB, Kramer JL, Rowland JH, Stein KD, Alteri R, Jemal A. Cancer treatment and survivorship statistics, 2016. CA Cancer J Clin 2016;66:271–289. doi: 10.3322/caac.21349 [DOI] [PubMed] [Google Scholar]
- 228.Institute of Medicine and National Research Council. From Cancer Patient to Cancer Survivor: Lost in Transition Washington, DC: The National Academies Press; 2006. 10.17226/11468. Accessed December 27, 2018. [DOI] [Google Scholar]
- 229.Mead H, Pratt-Chapman M, Gianattasio K, Cleary S, Gerstein M. Identifying models of cancer survivorship care. J Clin Oncol 2017;35(suppl):1. doi: 10.1200/JCO.2017.35.5_suppl.128034063 [DOI] [Google Scholar]
- 230.Okwuosa TM, Barac A. Burgeoning cardio-oncology programs: challenges and opportunities for early career cardiologists/faculty directors. J Am Coll Cardiol 2015;66:1193–1197. doi: 10.1016/j.jacc.2015.07.033 [DOI] [PubMed] [Google Scholar]
- 231.US Food and Drug Administration. FDA Public Workshop: cardiovascular toxicity assessment in oncology trials https://www.fda.gov/Drugs/NewsEvents/ucm513031.htm. Accessed December 27, 2018.
- 232.National Institutes of Health. https://grants.nih.gov/grants/guide/pa-files/PA-16-035.html. Accessed December 27, 2018.
- 233.Denlinger CS, Sanft T, Baker KS, Baxi S, Broderick G, Demark-Wahnefried W, Friedman DL, Goldman M, Hudson M, Khakpour N, King A, Koura D, Kvale E, Lally RM, Langbaum TS, Melisko M, Montoya JG, Mooney K, Moslehi JJ, O’Connor T, Overholser L, Paskett ED, Peppercorn J, Rodriguez MA, Ruddy KJ, Silverman P, Smith S, Syrjala KL, Tevaarwerk A, Urba SG, Wakabayashi MT, Zee P, Freedman-Cass DA, McMillian NR. Survivorship, version 2.2017, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw 2017;15:1140–1163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Creager MA, Gornik HL, Gray BH, Hamburg NM, Iobst WF, Mohler ER 3rd, White CJ. COCATS 4 Task Force 9: training in vascular medicine. J Am Coll Cardiol 2015;65:1832–1843. doi: 10.1016/j.jacc.2015.03.025 [DOI] [PubMed] [Google Scholar]
- 235.Cortes JE, Kantarjian H, Shah NP, Bixby D, Mauro MJ, Flinn I, O’Hare T, Hu S, Narasimhan NI, Rivera VM, Clackson T, Turner CD, Haluska FG, Druker BJ, Deininger MW, Talpaz M. Ponatinib in refractory Philadelphia chromosome-positive leukemias. N Engl J Med 2012;367:2075–2088. doi: 10.1056/NEJMoa1205127 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Cortes JE, Kim DW, Pinilla-Ibarz J, le Coutre P, Paquette R, Chuah C, Nicolini FE, Apperley JF, Khoury HJ, Talpaz M, DiPersio J, DeAngelo DJ, Abruzzese E, Rea D, Baccarani M, Müller MC, Gambacorti-Passerini C, Wong S, Lustgarten S, Rivera VM, Clackson T, Turner CD, Haluska FG, Guilhot F, Deininger MW, Hochhaus A, Hughes T, Goldman JM, Shah NP, Kantarjian H; PACE Investigators. A phase 2 trial of ponatinib in Philadelphia chromosome-positive leukemias. N Engl J Med 2013;369:1783–1796. doi: 10.1056/NEJMoa1306494 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Groarke JD, Cheng S, Moslehi J. Cancer-drug discovery and cardiovascular surveillance. N Engl J Med 2013;369:1779–1781. doi: 10.1056/NEJMp1313140 [DOI] [PubMed] [Google Scholar]
- 238.Ferri N, Siegl P, Corsini A, Herrmann J, Lerman A, Benghozi R. Drug attrition during pre-clinical and clinical development: understanding and managing drug-induced cardiotoxicity. Pharmacol Ther 2013;138:470–484. doi: 10.1016/j.pharmthera.2013.03.005 [DOI] [PubMed] [Google Scholar]


