Abstract
Background
There are a number of ways of monitoring blood glucose in women with diabetes during pregnancy, with self‐monitoring of blood glucose (SMBG) recommended as a key component of the management plan. No existing systematic reviews consider the benefits/effectiveness of different techniques of blood glucose monitoring on maternal and infant outcomes among pregnant women with pre‐existing diabetes. The effectiveness of the various monitoring techniques is unclear. This review is an update of a review that was first published in 2014 and subsequently updated in 2017.
Objectives
To compare techniques of blood glucose monitoring and their impact on maternal and infant outcomes among pregnant women with pre‐existing diabetes.
Search methods
For this update, we searched Cochrane Pregnancy and Childbirth’s Trials Register, ClinicalTrials.gov, the WHO International Clinical Trials Registry Platform (ICTRP) (1 November 2018), and reference lists of retrieved studies.
Selection criteria
Randomised controlled trials (RCTs) and quasi‐RCTs comparing techniques of blood glucose monitoring including SMBG, continuous glucose monitoring (CGM), automated telemedicine monitoring or clinic monitoring among pregnant women with pre‐existing diabetes mellitus (type 1 or type 2). Trials investigating timing and frequency of monitoring were also eligible for inclusion. RCTs using a cluster‐randomised design were eligible for inclusion but none were identified.
Data collection and analysis
Two review authors independently assessed study eligibility, extracted data and assessed the risk of bias of included studies. Data were checked for accuracy. The quality of the evidence was assessed using the GRADE approach.
Main results
This review update includes a total of 12 trials (944) women (type 1 diabetes: 660 women; type 2 diabetes: 113 women; type 1 or type 2 (unspecified): 171 women. The trials took place in Europe, the USA and Canada. Three of the 12 included studies are at low risk of bias, eight studies are at moderate risk of bias, and one study is at high risk of bias. Four trials reported that they were provided with the continuous glucose monitors free of charge or at a reduced cost by the manufacturer.
Continuous glucose monitoring (CGM) versus intermittent glucose monitoring, (four studies, 609 women)
CGM may reduce hypertensive disorders of pregnancy (pre‐eclampsia and pregnancy‐induced hypertension) (risk ratio (RR) 0.58, 95% confidence interval (CI) 0.39 to 0.85; 2 studies, 384 women; low‐quality evidence), although it should be noted that only two of the four relevant studies reported data for this composite outcome. Conversely, this did not translate into a clear reduction for pre‐eclampsia (RR 0.65, 95% CI 0.39 to 1.08; 4 studies, 609 women, moderate‐quality evidence). There was also no clear reduction in caesarean section (average RR 0.94, 95% CI 0.75 to 1.18; 3 studies, 427 women; I2 = 41%; moderate‐quality evidence) or large‐for‐gestational age (average RR 0.84, 95% CI 0.57 to 1.26; 3 studies, 421 women; I2 = 70%; low‐quality evidence) with CGM. There was not enough evidence to assess perinatal mortality (RR 0.82, 95% CI 0.05 to 12.61, 71 infants, 1 study; low‐quality evidence), or mortality or morbidity composite (RR 0.80, 95% CI 0.61 to 1.06; 1 study, 200 women) as the evidence was based on single studies of low quality. CGM appears to reduce neonatal hypoglycaemia (RR 0.66, 95% CI 0.48 to 0.93; 3 studies, 428 infants). Neurosensory disability was not reported.
Other methods of glucose monitoring
For the following five comparisons, self‐monitoring versus a different type of self‐monitoring (two studies, 43 women); self‐monitoring at home versus hospitalisation (one study, 100 women), pre‐prandial versus post‐prandial glucose monitoring (one study, 61 women), automated telemedicine monitoring versus conventional system (three studies, 84 women), and constant CGM versus intermittent CGM (one study, 25 women), it is uncertain whether any of the interventions has any impact on any of our GRADE outcomes (hypertensive disorders of pregnancy, caesarean section, large‐for‐gestational age) because the quality of the evidence was found to be very low. This was due to evidence largely being derived from single trials, with design limitations and limitations with imprecision (wide CIs, small sample sizes, and few events). There was not enough evidence to assess perinatal mortality and neonatal mortality and morbidity composite. Other important outcomes, such as neurosensory disability, were not reported in any of these comparisons.
Authors' conclusions
Two new studies (406 women) have been incorporated to one of the comparisons for this update. Although the evidence suggests that CGM in comparison to intermittent glucose monitoring may reduce hypertensive disorders of pregnancy, this did not translate into a clear reduction for pre‐eclampsia, and so this result should be viewed with caution. There was no evidence of a difference for other primary outcomes for this comparison. The evidence base for the effectiveness of other monitoring techniques analysed in the other five comparisons is weak and based on mainly single studies with very low‐quality evidence. Additional evidence from large well‐designed randomised trials is required to inform choices of other glucose monitoring techniques and to confirm the effectiveness of CGM.
Plain language summary
Methods for monitoring blood glucose in pregnant women with diabetes to improve outcomes
What is the issue and why is this important?
If a mother already has diabetes when she becomes pregnant, she and her baby are at higher risk of various problems. Women with existing diabetes that is not well‐controlled at conception and in the first three months of pregnancy are at increased risk of miscarriage, having a baby with developmental problems or stillbirth. The baby is also at increased risk of developing diabetes in childhood. Problems for mothers include developing high blood pressure and associated ill‐health, early births, large babies, difficult births and the need for caesarean section. During labour the baby is at increased risk of a shoulder becoming stuck (shoulder dystocia) and of bleeding in the brain (intracranial haemorrhage). After birth the baby is more likely to have low blood sugar levels (hypoglycaemia), jaundice and breathing problems. This means they are more likely to be admitted to intensive care. During pregnancy, the mother will have her blood glucose (sugar) levels monitored so appropriate steps can be taken to control her blood sugar.
Several methods of monitoring blood glucose are used, including regular testing at antenatal clinics and self‐monitoring by women at home. The timing varies, such as monitoring before meals versus monitoring after meals, and how often levels are measured. For continuous glucose monitoring (CGM), technologies are used to transfer information directly from the woman to her clinician and include telemedicine (telephone and video systems, information technology) and digital technologies (mobile phones, tablets). The aim of these methods is to provide a more accurate measure of blood sugar levels so that they can be more effectively controlled, in order to reduce potential problems.
What evidence did we find?
This is an update of a review first published in 2014, updated in 2017. We searched for evidence from randomised controlled studies in November 2018. We identified 12 studies involving 944 women (type 1 diabetes: 660 women; type 2 diabetes: 113 women; in two trials (171 women) there was a mix of type 1 and type 2 diabetes. The trials were from Europe, USA and Canada.
There were six comparisons. These were: continuous versus intermittent monitoring of blood glucose (four studies, 609 women); two different ways of self‐monitoring (two studies, 43 women); self‐monitoring at home versus hospitalisation to control blood glucose levels (one study, 100 women); blood glucose monitoring before a meal (pre‐prandial) versus blood glucose monitoring after a meal (post‐prandial) (one study, 61 women); automated telemedicine monitoring versus conventional care (three studies, 84 women); and constant continuous monitoring versus intermittent continuous monitoring (one study, 25 women),
Continuous versus intermittent monitoring may reduce overall high blood pressure problems during pregnancy (two studies, 384 women, low‐quality evidence). However, it should be noted that only two of four relevant studies reported data for this outcome. There was more evidence on high blood pressure and protein in their urine (pre‐eclampsia), which showed no clear difference (four studies, 609 women). We also found no difference in the number of women having a caesarean section (three studies, 427 women; moderate‐quality evidence). There was not enough evidence to assess infant deaths or the combined outcome of infant deaths and ill‐health as these outcomes were based on single studies. Four studies received some support from commercial partners.
The other comparisons of different ways of monitoring blood glucose levels were based on very small studies or single studies with very low‐quality evidence that did not show any clear differences in outcomes.
What does this mean?
Although the evidence from randomised controlled studies suggests that continuous monitoring of blood glucose levels may be more effective in reducing high blood pressure problems during pregnancy, only two studies reported on this. There was no clear reduction for pre‐eclampsia based on evidence from four studies. For other methods of glucose monitoring, the review showed that there is not enough evidence to say with any certainty which monitoring method for blood glucose is best. More research is needed to find out which other monitoring method is best at reducing the risk of complications for pregnant women with pre‐existing diabetes and to confirm the effectiveness of continuous glucose monitoring.
Summary of findings
Background
Description of the condition
Types of diabetes
There are three main types of diabetes mellitus: type 1, type 2 and gestational diabetes mellitus (GDM). Type 1 or insulin‐dependent diabetes results from the body’s failure to produce sufficient insulin and accounts for a minority of the total burden of diabetes in a population. Type 2 diabetes results from a failure of the body to utilise insulin, causing high blood sugar levels. Type 2 diabetes alone constitutes about 85% to 95% of all diabetes globally (IDF 2010). Type 2 diabetes is a serious and growing global health problem that has evolved in association with rapid cultural and social changes, ageing populations, increasing urbanisation, dietary changes, reduced physical activity and other unhealthy lifestyle and behavioural patterns (WHO 1994). In GDM, women who were not previously diabetic develop carbohydrate intolerance resulting in hyperglycaemia (high blood sugar levels) with first onset or detection occurring during pregnancy (HAPO 2002). GDM develops in one in 25 pregnancies worldwide and it is associated with the increasing incidence of type 2 diabetes post‐pregnancy. There are also women who are diagnosed with type 1 or type 2 diabetes before they get pregnant and so have pre‐existing diabetes. This review focuses on women with pre‐existing diabetes.
Prevalence of diabetes
Diabetes mellitus is found in every population in the world and it is estimated that 6.6% of the global population in the age group of 20 to 79 years old had diabetes in 2010. By 2030, it is estimated that 7.8% of the adult population will have diabetes (IDF 2010).
Diabetes mellitus complicates about 2% to 3% of all pregnancies. Approximately 90% of diabetes in pregnancy is accounted for by GDM. Pre‐existing type 1 and type 2 diabetes account for the remaining 10% of diabetes during pregnancy (Moore 2010). This review considers only the care of pregnant women with pre‐existing diabetes. A separate Cochrane Review on GDM has been published (Raman 2017).
Complications of diabetes in pregnancy: for mother and baby
Women with diabetes of any kind are at increased risk of morbidity and mortality during pregnancy. Pregnancy outcomes for women with pre‐existing diabetes and their infants are poor compared to those for women who do not have diabetes (NICE 2008; NICE 2015). The risks to both women and infants include fetal macrosomia (large baby), preterm birth, birth trauma (to mother and infant), induction of labour or caesarean section, miscarriage, congenital malformation, stillbirth, transient neonatal morbidity, neonatal death, obesity or diabetes, or both developing later in the baby’s life (Gonzalez‐Gonzalez 2008; Kitzmiller 2008; NICE 2008; NICE 2015).
Women with diabetes have an increased risk of an early miscarriage and are at increased risk of having a baby with malformations. Both of these risks are associated with less than optimal glycaemic control around the time of conception and in the first trimester. The risks appear to be approximately equivalent for women with type 1 and type 2 diabetes. The increased rate of spontaneous miscarriages and fetal malformation appears to be low when glycaemic control is moderately raised, and higher with increasingly poor glycaemic control (IDF 2010; Jensen 2009). Women with diabetes should be encouraged to obtain the best possible glycaemic control before conception (Kitzmiller 2010). Women with uncontrolled glycaemic levels should be discouraged from becoming pregnant until their blood glucose control can be improved.
Macrosomia, defined as infant birthweight greater than 4.5 kg, remains the commonest complication of pregnancy in women with diabetes (IDF 2010; Kitzmiller 2008; NICE 2008; NICE 2015). Macrosomia occurs in 27% to 62% of infants of diabetic mothers compared with 10% of non‐diabetic mothers (Gabbe 2003). Nationwide studies from the Netherlands, the UK, and Denmark estimate that the risk of a mother with type 1 diabetes giving birth to a baby who is large‐for‐gestational age, or has macrosomia ranges from 48.8% to 62.5% (Kitzmiller 2008). Recent data confirm that women with type 2 diabetes have an equally high risk of giving birth to an infant with macrosomia (ACOG 2005; ADA 2004; Roland 2005). For mothers with diabetes, macrosomia leads to an increased risk of perineal lacerations, complications in labour, and delivery by caesarean section (Slocum 2004). There are increased risks for the infants of intracranial haemorrhage, shoulder dystocia, neonatal hypoglycaemia, jaundice, and respiratory distress (Thomas 2006), as well as the longer‐term health risks of insulin resistance, obesity and type 2 diabetes (McElduff 2005). Overt diabetes is an undisputed factor for preterm birth (Sibai 2000).
Fetal hyperglycaemia causes fetal hypoxia and acidosis, which may explain the excess stillbirth rates observed in women with poorly controlled diabetes (Kitzmiller 2008). Infants with macrosomia due to poor maternal glycaemic control and fetal hyperinsulinaemia are more likely to develop obesity and glucose intolerance later in life (Fetita 2006; Kitzmiller 2008). Long‐term (five to 15 years) follow‐up studies of infants of mothers with diabetes suggest that poor glycaemic control during pregnancy has a negative influence on intellectual and psychomotor development (Kitzmiller 2008). Both findings highlight the prolonged effects on offspring of intrauterine exposure to diabetes (Fetita 2006; Kitzmiller 2008).
Glycaemic control prior to conception and in early pregnancy
The increased risks in women with diabetes of an early miscarriage and of having a baby with malformations are associated with suboptimal glycaemic control before or around the time of conception, and in the first trimester. Guidelines recommend that women should achieve the best possible glycaemic control before conception: women who improve their glycaemic control before conception have a reduced rate of fetal malformation (Fuhrmann 1983; IDF 2010; NICE 2008; NICE 2015).
Maternal hyperglycaemia during the first few weeks of pregnancy is strongly associated with increased spontaneous abortions and major congenital malformations (Kitzmiller 1996; Ray 2001). After 12 weeks’ gestation, hyperglycaemia induces fetal hyperinsulinaemia, accelerated growth, and excess adiposity in animal models and in women with diabetes (Gabbe 2003). These risks appear to be approximately equivalent for women with type 1 and type 2 diabetes. The increased rate of spontaneous miscarriages appears to be low when the HbA1c is modestly raised, and higher with increasingly poor glycaemic control (Mills 1988; Rosenn 1991). The same pattern is also found with respect to the rate of fetal malformations (Greene 1989; Suhonen 2000).
Description of the intervention
Techniques of blood glucose monitoring
Glucose readings supply trend information that helps to identify and prevent unwanted periods of hypo‐ and hyperglycaemia that are associated with adverse outcomes for both mother and baby. Women with type 1 and type 2 diabetes are advised to self‐monitor their blood glucose throughout pregnancy (IDF 2010).
Techniques of blood glucose monitoring to be considered in this review include self‐monitoring of blood glucose (SMBG), continuous glucose monitoring (CGM) and clinic monitoring (for which timing and frequency of monitoring are also considered).
Self‐monitoring of blood glucose (SMBG): a glucose meter (glucometer), with or without memory, can be used to measure capillary glucose. Conventional intensified glucose monitoring is defined as three to four blood glucose measurements per day (ADA 2011). Post‐prandial glucose during pregnancy has been identified as the best predictor of neonatal macrosomia (de Veciana 1995; Moses 1999). Therefore, SMBG protocols for women with type 1 or type 2 diabetes during pregnancy stress the importance of measuring blood glucose after meals (Jovanovič 2009), while for non‐pregnant women with diabetes, pre‐prandial values are recommended (ADA 2011; NICE 2008; NICE 2015).
Continuous glucose monitoring (CGM): the continuous glucose monitors currently available measure blood glucose either with minimal invasiveness through continuous measurement of interstitial fluid (ISF) or with the non‐invasive method of applying electromagnetic radiation through the skin to blood vessels in the body. The technologies for bringing a sensor into contact with ISF include inserting an indwelling sensor subcutaneously (into the abdominal wall or arm) to measure ISF in situ or harvesting this fluid by various mechanisms that compromise the skin barrier and delivering the fluid to an external sensor (Choleau 2002). After a warm‐up period of up to two hours and a device‐specific calibration process, each device’s sensor provides a blood glucose reading every one to 10 minutes for up to 72 hours with the minimally invasive technology and up to three months with the non‐invasive technology. CGM can provide up to 288 measurements a day (Murphy 2007).
Clinic monitoring refers to routine glucose monitoring during antenatal visits either using capillary or whole blood.
Timing and frequency of glucose monitoring
Post‐prandial glucose monitoring has been shown to be able to improve glycaemic control and thus reduce the risk of neonatal hypoglycaemia, macrosomia and caesarean delivery (de Veciana 1995), as well as to reduce the incidence of pre‐eclampsia and neonatal triceps skinfold thickness (Manderson 2003). Post‐prandial glucose values were most strongly associated with increased birthweight in the studies in which both pre‐ and post‐meal glucose were measured (Mello 2000).
Pregnant women with diabetes mellitus are advised to test fasting and one‐hour post‐prandial blood glucose levels after every meal during pregnancy and those taking insulin are encouraged to test their blood glucose before going to bed at night (NICE 2008; NICE 2015). The American Diabetes Association also recommends SMBG before and after meals and occasionally at night time, to provide optimal results in pregnancy (Kitzmiller 2008).
The optimal frequency and timing of home glucose testing during pregnancy is unknown. In reality the frequency of glucose monitoring will depend on women's compliance, with few managing to carry out high numbers of tests daily (Kerssen 2006).
Educational approaches incorporating additional glucose testing after meals to improve glycaemic control in late gestation have shown potential to reduce birthweight (Howorka 2001).
Glycaemic control during pregnancy among women with pre‐existing diabetes
Pregnancy profoundly affects the management of diabetes (Gabbe 2003; Jovanovic 2006). Pregnancy is associated with changes in insulin sensitivity, which may lead to changes in plasma glucose levels. Hormonal changes during pregnancy cause a progressive increase in insulin resistance, necessitating intensive medical nutrition therapy and frequently adjusted insulin administration throughout the pregnancy. The control of hyperglycaemia in pregnant women with pre‐existing diabetes is essential in order to avoid the above mentioned adverse maternal and infant outcomes (Kitzmiller 2008). Macrosomia and other neonatal complications are minimised with intensified glycaemic control (Kerssen 2007; Kitzmiller 2008; Suhonen 2000).
If it is safely achievable, women with diabetes should aim to keep fasting blood glucose between 3.5 mmol/L and 5.9 mmol/L and one‐hour post‐prandial blood glucose below 7.8 mmol/L during pregnancy (NICE 2008; NICE 2015); HbA1c should be kept below 6.0% (ADA 2011). Excellent glycaemic control throughout the pregnancy is associated with the lowest risk for both maternal, fetal and neonatal complications (Kitzmiller 2008). On the other hand, the targets of glycaemic control for non‐pregnant women with type 1 or type 2 diabetes are less stringent, i.e. fasting blood glucose to be 3.9 mmol/L to 7.2 mmol/L and HbA1c less than 7.0% (ADA 2011).
How the intervention might work
Maternal glucose levels in women with pre‐existing diabetes directly influence those of the fetus. Fetal metabolic complications may give rise to macrosomia, congenital malformation, stillbirth and increased perinatal mortality (IDF 2010; Kapoor 2007; Kitzmiller 2008; NICE 2008; NICE 2015). Blood glucose monitoring allows adjustment of insulin dosage in relation to meal size and type, physical activity, stress and time of the day for women with pre‐existing diabetes during pregnancy (Davidson 2005). This will limit the maternal risk of hypoglycaemic episodes while avoiding prolonged periods of hyperglycaemia. However, the frequency and timing of glucose monitoring will also influence maternal and fetal outcomes.
Why it is important to do this review
Self‐monitoring of blood glucose is recommended as a key component of diabetes therapy during pregnancy and is included in the management plan (IDF 2010; Kitzmiller 2008; NICE 2008; ). No existing systematic reviews consider the benefits of various techniques of blood glucose monitoring on maternal and infant outcomes among pregnant women with pre‐existing diabetes. The effectiveness of the various monitoring techniques is unclear. This systematic review aims to generate information to guide pregnant women with pre‐existing diabetes and their clinicians in their choice of monitoring techniques in order to optimise maternal and infant outcomes. All trials that evaluate any techniques of blood glucose monitoring among pregnant women with pre‐existing diabetes will be considered. This Cochrane Review is an update of a review that was first published in 2014 (Moy 2014) and subsequently updated in 2017 (Moy 2017).
Objectives
To compare techniques of blood glucose monitoring and their impact on maternal and infant outcomes among pregnant women with pre‐existing diabetes.
Methods
Criteria for considering studies for this review
Types of studies
We included randomised controlled trials and quasi‐randomised trials. Cluster‐randomised trials were eligible for inclusion but none were identified. Trials using a cross‐over design were not eligible for inclusion. Abstracts were eligible for inclusion if sufficient information was provided to judge the quality and potential for bias of these trials.
Types of participants
Pregnant women with pre‐existing diabetes mellitus (type 1 or type 2). Women with gestational diabetes mellitus (GDM) were excluded.
Types of interventions
Techniques of blood glucose monitoring including continuous glucose monitoring (CGM), self‐monitoring of blood glucose (SMBG) or clinic monitoring. We also considered the timing and frequency of monitoring.
The following comparisons were considered in this update.
Continuous glucose monitoring (CGM) versus intermittent glucose monitoring (i.e. CGM versus standard care)
Self‐monitoring of blood glucose (SMBG) versus different type of SMBG
SMBG at home versus hospitalisation
Pre‐prandial versus post‐prandial glucose monitoring
Automated telemedicine monitoring versus conventional system
Constant CGM versus intermittent CGM (e.g. use of a glucose monitor 24 hours per day versus use of a monitor 14 days per month)
Types of outcome measures
For this update, we used the Cochrane Pregnancy and Childbirth core outcome set for reviews of diabetes in pregnancy, developed by the Cochrane Pregnancy and Childbirth Australasian satellite.
Primary outcomes
Mother
Hypertensive disorders of pregnancy (including pre‐eclampsia, pregnancy‐induced hypertension, eclampsia)
Caesarean section
Neonatal/infant
Large‐for‐gestational age
Perinatal mortality (stillbirth and neonatal mortality)
Mortality or morbidity composite (e.g. pregnancy loss (miscarriage, stillbirth, and neonatal death); birth injury; neonatal glycaemia; hyperbilirubinaemia; respiratory distress; and high level neonatal care of more than 24 hours)
Neurosensory disability
Secondary outcomes
Mother
Pre‐eclampsia
Pregnancy‐induced hypertension
Eclampsia
Induction of labour
Perineal trauma
Placental abruption
Postpartum haemorrhage
Postpartum infection
Weight gain during pregnancy
Adherence to the intervention
Behaviour changes associated with the intervention
Relevant biomarker changes associated with the intervention (e.g. adiponectin, free fatty acids, triglycerides, high‐density lipoproteins, low‐density lipoproteins, insulin)
Sense of well‐being and quality of life
Views of the intervention
Breastfeeding (e.g. at discharge, six weeks postpartum)
Use of additional pharmacotherapy
Glycaemic control during/end of treatment (as defined by trialists) (e.g. HbA1c, fructosamine, fasting blood glucose, post‐prandial blood glucose)
Maternal hypoglycaemia
Maternal mortality
Miscarriage
Instrumental vaginal birth*
Long‐term maternal outcomes
Postnatal depression
Postnatal weight retention or return to pre‐pregnancy weight
Body mass index (BMI)
GDM in a subsequent pregnancy
Type 1 diabetes
Impaired glucose tolerance
Cardiovascular health (as defined by trialists, including blood pressure, hypertension, cardiovascular disease, metabolic syndrome)
Neonatal/infant
Stillbirth
Neonatal mortality
Gestational age at birth
Preterm birth (less than 37 weeks' gestation and less than 34 weeks' gestation)
Apgar score (less than seven at five minutes)
Macrosomia
Small‐for‐gestational age
Birthweight and z‐score
Head circumference and z‐score
Length and z‐score
Ponderal index
Adiposity (e.g. BMI, skinfold thickness)
Shoulder dystocia
Bone fracture
Nerve palsy
Respiratory distress syndrome
Hypoglycaemia (variously defined)
Hyperbilirubinaemia
Neonatal hypocalcaemia
Polycythaemia
Relevant biomarker changes associated with the intervention (e.g. cord c peptide, cord insulin)
Major and minor anomalies
Later infant and childhood secondary outcomes
Weight and z‐scores
Height and z‐scores
Head circumference and z‐scores
Adiposity (e.g. as measured by BMI, skinfold thickness)
Blood pressure
Type 1 diabetes
Type 2 diabetes
Impaired glucose tolerance
Dyslipidaemia or metabolic syndrome
Educational achievement
Child in adulthood
Weight
Height
Adiposity (e.g. as measured by BMI, skinfold thickness)
Cardiovascular health (as defined by trialists, including blood pressure, hypertension, cardiovascular disease, metabolic syndrome)
Type 1 diabetes
Type 2 diabetes
Impaired glucose tolerance
Dyslipidaemia or metabolic syndrome
Employment, education and social status/achievement
Health service use
Number of hospital or health professional visits (e.g. midwife, obstetrician, physician, dietician, diabetic nurse)
Number of antenatal visits or admissions
Length of antenatal stay
Neonatal intensive care unit admission
Neonatal intensive care unit length of stay greater than 24 hours*
Length of postnatal stay (mother)
Length of postnatal stay (baby)
Costs to families associated with the management provided
Costs associated with the intervention
Cost of maternal care
Cost of offspring care
Not pre‐specified
Birth trauma (shoulder dystocia, bone fracture, nerve palsy) (not pre‐specified as a composite)
Neonatal glucose at age one hour
Transient tachypnoea
Diabetic ketoacidosis
Feeding difficulties
*Outcomes not pre‐specified in our protocol ‐ see Differences between protocol and review.
Search methods for identification of studies
The following methods section of this review is based on a standard template used by Cochrane Pregnancy and Childbirth.
Electronic searches
For this update, we searched Cochrane Pregnancy and Childbirth’s Trials Register by contacting their Information Specialist (1 November 2018).
The Register is a database containing over 25,000 reports of controlled trials in the field of pregnancy and childbirth. It represents over 30 years of searching. For full current search methods used to populate Pregnancy and Childbirth’s Trials Register including the detailed search strategies for CENTRAL, MEDLINE, Embase and CINAHL; the list of handsearched journals and conference proceedings, and the list of journals reviewed via the current awareness service, please follow this link.
Briefly, Cochrane Pregnancy and Childbirth’s Trials Register is maintained by their Information Specialist and contains trials identified from:
monthly searches of the Cochrane Central Register of Controlled Trials (CENTRAL);
weekly searches of MEDLINE (Ovid);
weekly searches of Embase (Ovid);
monthly searches of CINAHL (EBSCO);
handsearches of 30 journals and the proceedings of major conferences;
weekly current awareness alerts for a further 44 journals plus monthly BioMed Central email alerts.
Search results are screened by two people and the full text of all relevant trial reports identified through the searching activities described above is reviewed. Based on the intervention described, each trial report is assigned a number that corresponds to a specific Pregnancy and Childbirth review topic (or topics), and is then added to the Register. The Information Specialist searches the Register for each review using this topic number rather than keywords. This results in a more specific search set that has been fully accounted for in the relevant review sections (Included studies; Excluded studies; Ongoing studies).
In addition, we searched ClinicalTrials.gov and the WHO International Clinical Trials Registry Platform (ICTRP) for unpublished, planned and ongoing trial reports (1 November 2018 using the search methods detailed in Appendix 1.
Searching other resources
We contacted the author of Feig 2017 for additional information (19 March 2019), no reply received to date (26 April 2019).
We searched the reference lists of retrieved studies.
We did not apply any language or date restrictions.
Data collection and analysis
For methods used in the previous version of this review, seeMoy 2017.
For this update, the following methods were used for assessing the seven reports that were identified as a result of the updated search.
The following methods section of this review is based on a standard template used by Cochrane Pregnancy and Childbirth.
Selection of studies
Two review authors independently assessed for inclusion all the potential studies identified as a result of the search strategy. We resolved any disagreement through discussion or, if required, we consulted a third review author.
Data extraction and management
We designed a form to extract data. For eligible studies, two review authors extracted the data using the agreed form. We resolved discrepancies through discussion or, if required, we consulted a third review author. Data were entered into Review Manager software (RevMan 2014) and checked for accuracy.
When information regarding any of the above was unclear, we planned to contact authors of the original reports to provide further details.
Assessment of risk of bias in included studies
Two review authors independently assessed risk of bias for each study using the criteria outlined in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011). Any disagreement was resolved by discussion or by involving a third assessor.
(1) Random sequence generation (checking for possible selection bias)
We described for each included study the method used to generate the allocation sequence in sufficient detail to allow an assessment of whether it should produce comparable groups.
We assessed the method as:
low risk of bias (any truly random process, e.g. random number table; computer random number generator);
high risk of bias (any non‐random process, e.g. odd or even date of birth; hospital or clinic record number);
unclear risk of bias.
(2) Allocation concealment (checking for possible selection bias)
We described for each included study the method used to conceal allocation to interventions prior to assignment and assessed whether intervention allocation could have been foreseen in advance of, or during recruitment, or changed after assignment.
We assessed the methods as:
low risk of bias (e.g. telephone or central randomisation; consecutively numbered sealed opaque envelopes);
high risk of bias (open random allocation; unsealed or non‐opaque envelopes, alternation; date of birth);
unclear risk of bias.
(3.1) Blinding of participants and personnel (checking for possible performance bias)
We described for each included study the methods used, if any, to blind study participants and personnel from knowledge of which intervention a participant received. We considered that studies were at low risk of bias if they were blinded, or if we judged that the lack of blinding was unlikely to affect results. We assessed blinding separately for different outcomes or classes of outcomes.
We assessed the methods as:
low, high or unclear risk of bias for participants;
low, high or unclear risk of bias for personnel.
(3.2) Blinding of outcome assessment (checking for possible detection bias)
We described for each included study the methods used, if any, to blind outcome assessors from knowledge of which intervention a participant received. We assessed blinding separately for different outcomes or classes of outcomes.
We assessed methods used to blind outcome assessment as:
low, high or unclear risk of bias.
(4) Incomplete outcome data (checking for possible attrition bias due to the amount, nature and handling of incomplete outcome data)
We described for each included study, and for each outcome or class of outcomes, the completeness of data including attrition and exclusions from the analysis. We stated whether attrition and exclusions were reported and the numbers included in the analysis at each stage (compared with the total randomised participants), reasons for attrition or exclusion where reported, and whether missing data were balanced across groups or were related to outcomes. Where sufficient information was reported, or could be supplied by the trial authors, we planned to re‐include missing data in the analyses which we undertook.
We assessed methods as:
low risk of bias (e.g. no missing outcome data; missing outcome data balanced across groups);
high risk of bias (e.g. numbers or reasons for missing data imbalanced across groups; ‘as treated’ analysis done with substantial departure of intervention received from that assigned at randomisation);
unclear risk of bias.
(5) Selective reporting (checking for reporting bias)
We described for each included study how we investigated the possibility of selective outcome reporting bias and what we found.
We assessed the methods as:
low risk of bias (where it is clear that all of the study’s pre‐specified outcomes and all expected outcomes of interest to the review have been reported);
high risk of bias (where not all the study’s pre‐specified outcomes have been reported; one or more reported primary outcomes were not pre‐specified; outcomes of interest are reported incompletely and so cannot be used; study fails to include results of a key outcome that would have been expected to have been reported);
unclear risk of bias.
(6) Other bias (checking for bias due to problems not covered by (1) to (5) above)
We described for each included study any important concerns we had about other possible sources of bias.
(7) Overall risk of bias
We made explicit judgements about whether studies were at high risk of bias, according to the criteria given in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011). With reference to (1) to (6) above, we planned to assess the likely magnitude and direction of the bias and whether we considered it is likely to impact on the findings. In future updates, we will explore the impact of the level of bias through undertaking sensitivity analyses ‐ seeSensitivity analysis.
Assessment of the quality of the evidence using the GRADE approach
For this update we assessed the quality of the evidence using the GRADE approach as outlined in the GRADE handbook in order to assess the quality of the body of evidence relating to the following outcomes for all comparisons.
Hypertensive disorders of pregnancy (including pre‐eclampsia, pregnancy‐induced hypertension, eclampsia)
Caesarean section
Large‐for‐gestational age
Perinatal mortality (stillbirth and neonatal mortality)
We used the GRADEpro Guideline Development Tool to import data from Review Manager 5.3 (RevMan 2014) in order to create ’Summary of findings’ tables. A summary of the intervention effect and a measure of quality for each of the above outcomes was produced using the GRADE approach. The GRADE approach uses five considerations (study limitations, consistency of effect, imprecision, indirectness and publication bias) to assess the quality of the body of evidence for each outcome. The evidence can be downgraded from 'high quality' by one level for serious (or by two levels for very serious) limitations, depending on assessments for risk of bias, indirectness of evidence, serious inconsistency, imprecision of effect estimates or potential publication bias.
Measures of treatment effect
Dichotomous data
For dichotomous data, we presented results as summary risk ratio with 95% confidence intervals.
Continuous data
We used the mean difference if outcomes were measured in the same way between trials. In future updates, if appropriate, we will use the standardised mean difference to combine trials that measure the same outcome, but use different methods.
Unit of analysis issues
Trials with more than two intervention groups
Had we included trials with more than two techniques of glucose monitoring, we planned to analyse them according to the methods described in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011); the relevant pair of interventions would have been selected and the others excluded.
Cluster‐randomised trials
We did not identify any cluster‐randomised trials for inclusion. However, in future updates, if we identify any cluster‐randomised trials we will include them in the analyses along with individually‐randomised trials. We will adjust their sample sizes using the methods described in the Cochrane Handbook for Systematic Reviews of Interventions using an estimate of the intracluster correlation co‐efficient (ICC) derived from the trial (if possible), from a similar trial or from a study of a similar population. If we use ICCs from other sources, we will report this and conduct sensitivity analyses to investigate the effect of variation in the ICC. If we identify both cluster‐randomised trials and individually‐randomised trials, we plan to synthesise the relevant information. We will consider it reasonable to combine the results from both if there is little heterogeneity between the study designs and the interaction between the effect of intervention and the choice of randomisation unit is considered to be unlikely. We will also acknowledge heterogeneity in the randomisation unit and perform a sensitivity analysis to investigate the effects of the randomisation unit.
Dealing with missing data
For included studies, levels of attrition were noted. In future updates, if more eligible studies are included, the impact of including studies with high levels of missing data in the overall assessment of treatment effect will be explored by using sensitivity analysis.
For all outcomes, analyses were carried out, as far as possible, on an intention‐to‐treat basis, i.e. we attempted to include all participants randomised to each group in the analyses. The denominator for each outcome in each trial was the number randomised minus any participants whose outcomes were known to be missing.
Assessment of heterogeneity
We assessed statistical heterogeneity in each meta‐analysis using the Tau², I² and Chi² statistics. We regarded heterogeneity as substantial if an I² was greater than 30% and either a Tau² was greater than zero, or there was a low P value (less than 0.10) in the Chi² test for heterogeneity. Had we identified substantial heterogeneity (above 30%), we planned to explore it by pre‐specified subgroup analysis.
Assessment of reporting biases
In future updates, if there are 10 or more studies in the meta‐analysis we will investigate reporting biases (such as publication bias) using funnel plots. We will assess funnel plot asymmetry visually. If asymmetry is suggested by a visual assessment, we will perform exploratory analyses to investigate it.
Data synthesis
We carried out statistical analysis using the Review Manager software (RevMan 2014). We used fixed‐effect meta‐analysis for combining data where it was reasonable to assume that studies were estimating the same underlying treatment effect: i.e. where trials were examining the same intervention, and the trials’ populations and methods were judged sufficiently similar.
If there was clinical heterogeneity sufficient to expect that the underlying treatment effects differed between trials, or if substantial statistical heterogeneity was detected, we used random‐effects meta‐analysis to produce an overall summary, if an average treatment effect across trials was considered clinically meaningful. The random‐effects summary was treated as the average of the range of possible treatment effects and we discuss the clinical implications of treatment effects differing between trials. If the average treatment effect was not clinically meaningful, we did not combine trials. Where we used random‐effects analyses, the results are presented as the average treatment effect with 95% confidence intervals, and the estimates of Tau² and I².
Subgroup analysis and investigation of heterogeneity
Had we identified substantial heterogeneity, we planned to investigate it using subgroup analyses and to consider whether an overall summary was meaningful, and if it was, to use a random‐effects analysis to produce it.
We planned to restrict subgroup analyses to primary outcomes for the following subgroups:
types of diabetes mellitus (type 1 versus type 2 diabetes);
glycaemic control prior to pregnancy (pre‐pregnancy HbA1c within target range versus pre‐pregnancy HbA1c out of target range).
However, we did not carry out any subgroup analysis as there were too few trials included in any one comparison. Data for outcomes in included trials were also not reported separately by type of diabetes. Pre‐pregnancy glycaemic control for all women was comparable at baseline. These analyses will be conducted in future updates of the review, if more data become available.
Sensitivity analysis
We planned to undertake sensitivity analysis to explore differences between fixed‐effect or random‐effects analyses for outcomes with statistical heterogeneity.
We also planned to undertake sensitivity analysis to assess the effect on pooled results of studies considered to have a high risk of bias.
However, due to the scarcity of data in any one comparison, no sensitivity analyses were conducted. If more data become available, the planned sensitivity analyses will be carried out in future updates.
Results
Description of studies
See Characteristics of included studies; Characteristics of excluded studies.
Results of the search
See: Figure 1.
1.
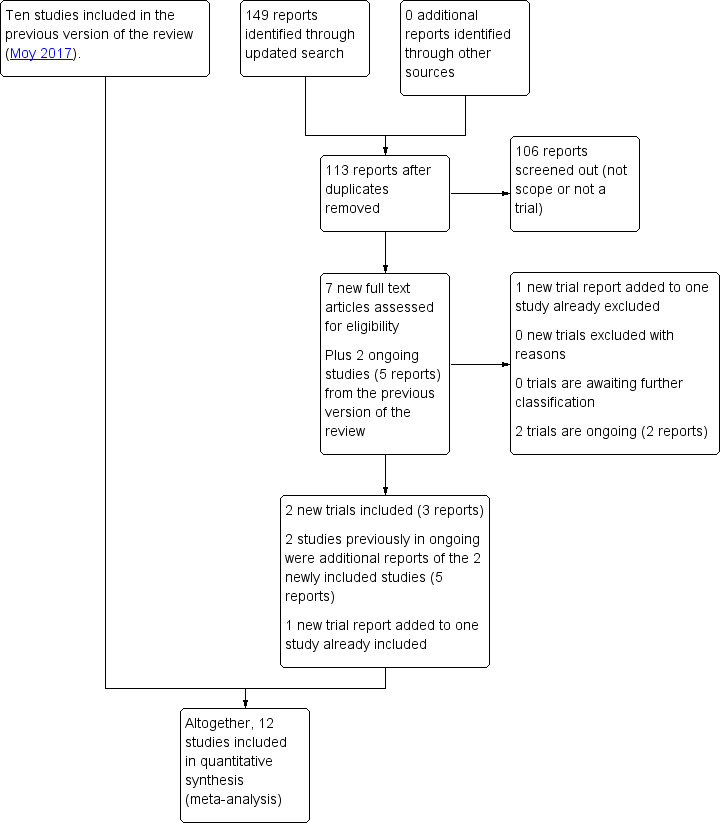
Study flow diagram 2018
For this 2018 update, we identified 149 trial reports to assess and 113 in total after duplicates had been removed. One‐hundred and six reports were screened out because they did not meet the scope for this review or were not randomised controlled trials. We then assessed seven trial reports for inclusion. We also reassessed the two ongoing studies listed in the previous version of the review (five reports).
We included two new trials (Feig 2017; Voormolen 2018) (three reports), added one trial report to an already included study (di Biase 1997), and added one trial report to an already excluded study (Bartholomew 2011). No new studies were excluded in this update. Two trials (two reports) are ongoing (Link 2018; Logan 2011), see Ongoing studies. The two studies previously listed in ongoing were additional reports of the newly included studies (five reports).
Included studies
Altogether, this review now comprises 12 included studies (944 women), all of which contributed data. Three of the 12 included trials were from the UK (Manderson 2003; Murphy 2008; Stubbs 1980), two were from Italy (Dalfrà 2009; di Biase 1997), and one each was from Sweden (Hanson 1984), Denmark (Secher 2013), Macedonia (Petrovski 2011), Poland (Wojcicki 2001), the US (Varner 1983), Canada (Feig 2017) and the Netherlands (Voormolen 2018).
For full details, see Characteristics of included studies.
Methods
All included studies were parallel group randomised controlled trials and involved two arms. The randomisation method was not always well described and in one study the allocation process was not truly random, and so was assessed as being 'quasi‐randomised' (Dalfrà 2009). All of the studies were described as being 'open‐label' and therefore not blinded. Two studies were multi‐centre trials: one was based in Canada and involved 31 hospitals in Canada, England, Scotland, Spain, Italy, Ireland and the USA (Feig 2017); and one involved 22 hospitals, university, teaching and non‐teaching hospitals in the Netherlands and one university hospital in Belgium (Voormolen 2018). The remaining trials were single centre (Dalfrà 2009; di Biase 1997; Hanson 1984; Manderson 2003; Murphy 2008; Petrovski 2011; Secher 2013; Stubbs 1980; Varner 1983; Wojcicki 2001).
Trial dates
Trial dates were not reported in the study reports for six trials (Dalfrà 2009; di Biase 1997; Manderson 2003; Petrovski 2011; Stubbs 1980; Wojcicki 2001).
For the remaining studies, trials dates were reported as follows: 25 March 2013 to 22 March 2016 (Feig 2017); 1 October 1979 to 1 October 1982 (Hanson 1984); September 2003 to 2006 (Murphy 2008); 15 February 2009 to 15 February 2011 (Secher 2013); 1 February 1980 to 16 September 1981 (Varner 1983); and July 2011 to September 2015 (Voormolen 2018).
Participants
The trials included in this review involved a total of 944 women; 660 with type 1 diabetes, 113 with type 2 diabetes, and 171 women with either type 1 or type 2 diabetes (data not reported separately).
Hanson 1984, Murphy 2008 and Secher 2013 included women with pre‐existing type 1 and type 2 diabetes (however, only Secher 2013 presented the results separately for type 1 and type 2 diabetes). Only women with pre‐existing type 1 diabetes were eligible to participate in di Biase 1997Feig 2017, Manderson 2003,Petrovski 2011,Stubbs 1980,Varner 1983, and Wojcicki 2001. In one trial (Feig 2017), they ran two trials in parallel for pregnant women and for women planning a pregnancy with type 1 diabetes. However the results for most outcomes were reported separately and so we have included the data for the pregnant women in this review. Women with pre‐existing type 1 diabetes and gestational diabetes participated in Dalfrà 2009, however the results are presented separately so only data for women with type 1 diabetes are included in this review. Women with pre‐existing type 1 and type 2 diabetes and gestational diabetes participated in Voormolen 2018, however the results are presented separately for some of the outcomes, so only data for women with type 1 and type 2 diabetes are included in this review. The ethnicity of the women was not mentioned in all trials. As these trials originated from the European countries and the USA, it is assumed that majority of the women were white or Caucasians.
Interventions and comparisons
Continuous glucose monitoring (CGM) was compared with intermittent glucose monitoring in trials by Feig 2017, Murphy 2008, Secher 2013 and Voormolen 2018. Stubbs 1980 and Varner 1983 compared self‐monitoring of blood glucose (SMBG) at home with standard care. In Stubbs 1980 the SMBG group measured blood glucose with a glucometer seven times a day, twice weekly and the standard care group (non‐meter group) checked urine glucose four times daily, with random blood glucose measured at fortnightly clinic visits. In Varner 1983, the SMBG group carried out daily home glucose monitoring four times daily and the standard care group carried out weekly venipuncture three times daily, measured on one day weekly. Hanson 1984 compared self‐monitoring blood glucose at home from the 32nd week until the 36th week of gestation, with weekly hospital visits, and hospitalisation during the 37th week to delivery with a group who were hospitalised from 32nd week until delivery. Manderson 2003 compared timing of glucose monitoring, i.e. pre‐prandial versus post‐prandial. Pre‐prandial refers to measurement of blood glucose before meals while post‐prandial refers to blood glucose measured two hours after a meal. Automated telemedicine monitoring versus conventional system were compared in studies by Dalfrà 2009, di Biase 1997 and Wojcicki 2001. Automated telemedicine monitoring refers to automated transmission of blood glucose values via telephone or Internet to the physicians, which allows immediate attention from the physicians. Petrovski 2011 compared constant CGM with intermittent CGM. CGM refers to glucose measured in subcutaneous tissues every 10 seconds and an average value is stored every five minutes, providing up to 288 measurements per day.
Outcomes
The primary outcome composite outcome, hypertensive disorders of pregnancy was reported by Feig 2017 (including pre‐eclampsia, pregnancy‐induced hypertension and worsening chronic hypertension), and by Voormolen 2018 (pre‐eclampsia and pregnancy‐induced hypertension); caesarean section was reported by Dalfrà 2009; Feig 2017; Hanson 1984; Manderson 2003; Murphy 2008; Petrovski 2011; Secher 2013; Varner 1983; large‐for‐gestational age was reported by Feig 2017; Manderson 2003; Murphy 2008;Secher 2013, (defined as birthweight 90th centile or above); perinatal mortality was reported by Hanson 1984; Manderson 2003; Murphy 2008;Varner 1983); neonatal mortality or morbidity composite was reported by Dalfrà 2009; Feig 2017; Varner 1983; and neurosensory disability was not reported by any trials.
Secondary maternal outcomes reported by the included studies were pre‐eclampsia (Feig 2017; Hanson 1984; Manderson 2003; Murphy 2008;Secher 2013; Voormolen 2018), pregnancy‐induced hypertension (Feig 2017; Hanson 1984; Voormolen 2018), placental abruption (Hanson 1984), weight gain during pregnancy (Feig 2017; Dalfrà 2009; Manderson 2003;Petrovski 2011), behaviour changes associated with the intervention (Feig 2017 (using hypoglycaemia fear survey (HFS II) behaviour subscale which measures two distinct aspects of behavioural avoidance to prevent hypoglycaemia), sense of well‐being and quality of life (Feig 2017 (using three different questionnaires (blood glucose monitoring system rating questionnaire (BGMSRQ), problem areas in diabetes (PAID), short‐form‐12)), use of additional pharmacotherapy (use of additional insulin therapy: Dalfrà 2009; insulin dose: di Biase 1997; Manderson 2003;Petrovski 2011), glycaemic control during/end of treatment (HbA1c) (Dalfrà 2009; di Biase 1997; Feig 2017; Manderson 2003; Murphy 2008; Petrovski 2011; Varner 1983;Wojcicki 2001), maternal hypoglycaemia (Feig 2017; Petrovski 2011) and miscarriage (Feig 2017; Murphy 2008; Secher 2013;Varner 1983).
Secondary perinatal/neonatal outcomes reported by the included studies were stillbirth (reported by Feig 2017; Manderson 2003), neonatal mortality (Murphy 2008; Varner 1983; Voormolen 2018), gestational age at birth (Dalfrà 2009; di Biase 1997; Manderson 2003; Murphy 2008; Varner 1983; Wojcicki 2001), preterm birth less than 37 weeks' gestation (Feig 2017; Hanson 1984; Manderson 2003; Murphy 2008; Petrovski 2011; Secher 2013), preterm birth less than 34 weeks' gestation (Feig 2017;) macrosomia (Feig 2017; Dalfrà 2009; Feig 2017; Manderson 2003; Petrovski 2011; Voormolen 2018: defined as birthweight greater than 4 kg in four studies and birthweight above 90th centile in two studies), small‐for‐gestational age (Feig 2017; Murphy 2008: defined as birthweight 10th centile or below), birthweight (Feig 2017; Dalfrà 2009; Manderson 2003; Murphy 2008; Stubbs 1980; Varner 1983), head circumference (Feig 2017), length (Feig 2017), adiposity (sum of four skin folds (triceps, subscapular, biceps, flank: Feig 2017 ) and (triceps skinfold thickness and subscapular skinfold thickness: Manderson 2003), shoulder dystocia (Feig 2017), respiratory distress syndrome (Feig 2017, Hanson 1984; Manderson 2003; Varner 1983), hypoglycaemia (Feig 2017; Hanson 1984; Manderson 2003; Murphy 2008; Petrovski 2011; Secher 2013; Varner 1983), hyperbilirubinaemia (Feig 2017; Hanson 1984; Manderson 2003; Varner 1983), neonatal hypocalcaemia (Varner 1983), polycythaemia (Varner 1983), relevant biomarker changes associated with the intervention (neonatal cord vein c‐peptide: Feig 2017; Varner 1983, (cord IGF‐1: Manderson 2003), and major anomalies (Feig 2017; Hanson 1984; Murphy 2008).
The only secondary health service use outcomes reported were antenatal hospital admission (Feig 2017; Hanson 1984), neonatal intensive care (NICU) admissions (Manderson 2003; Murphy 2008) and NICU length of admission > 24 hours (Feig 2017).
Outcomes that were not pre‐specified, but are reported in this review are maternal diabetic ketoacidosis (Feig 2017; Petrovski 2011), birth trauma (shoulder dystocia, bone fracture and nerve palsy, pre‐specified as individual outcomes but reported as a composite: Feig 2017; Manderson 2003), neonatal glucose at age one hour (Manderson 2003), transient tachypnoea (Manderson 2003), and feeding difficulties (Hanson 1984). Instrumental vaginal birth was reported in one trial (Voormolen 2018), but the data were not presented separately for women with pre‐existing diabetes and women with GDM. No other trial reported on instrumental vaginal birth.
Secondary maternal outcomes not reported by any of the included studies were: induction of labour, perineal trauma, postpartum haemorrhage, postpartum infection, adherence to the intervention, relevant biomarker changes associated with the intervention (e.g. adiponectin, free fatty acids, triglycerides, high‐density lipoproteins, low‐density lipoproteins, insulin), views of the intervention, maternal mortality.
Secondary perinatal/neonatal outcomes not reported by any of the included studies were: Apgar score (less than seven at five minutes), head circumference and z‐score, length and z‐score, ponderal index, adiposity measured by body mass index (BMI), and minor anomalies.
Health service use outcomes not reported by any of the included studies were: health service use, number of hospital or health professional visits (e.g. midwife, obstetrician, physician, dietician, diabetic nurse), number of antenatal visits, length of antenatal stay, length of postnatal stay (mother), length of postnatal stay (baby), costs to families associated with the management provided, costs associated with the intervention, cost of maternal care, and cost of offspring care.
No studies reported long‐term maternal outcomes (postnatal depression, postnatal weight retention or return to pre‐pregnancy weight, BMI, impaired glucose tolerance, cardiovascular health (as defined by trialists, including blood pressure, hypertension, cardiovascular disease, metabolic syndrome)), later infant or childhood outcomes (weight and z‐scores, height and z‐scores, head circumference and z‐scores, adiposity (e.g. as measured by BMI, skinfold thickness), blood pressure, type 1 diabetes, type 2 diabetes, impaired glucose tolerance, dyslipidaemia or metabolic syndrome, educational achievement), or child in adulthood outcomes (weight, height, adiposity (e.g. as measured by BMI, skinfold thickness), cardiovascular health (as defined by trialists, including blood pressure, hypertension, cardiovascular disease, metabolic syndrome), type 1 diabetes, type 2 diabetes, impaired glucose tolerance, dyslipidaemia or metabolic syndrome, employment, education and social status/achievement).
Some outcomes were reported in a form that could not be used in this review. Hanson 1984 reported the median antenatal hospital stay and neonatal hospital stay, but did not report the standard deviation of blood glucose values, and only reported HbA1c graphically. Manderson 2003 reported the median and interquartile range for cord insulin and length of stay in neonatal unit, and Secher 2013 reported weight gain in pregnancy, HbA1c, plasma glucose, gestational age at birth, and birthweight as median and range. Where results were reported as medians, we felt it was unlikely that the results were normally distributed, and excluded them from meta‐analyses. Percentage of maternal hypoglycaemic episodes was reported by Wojcicki 2001, however the total of all blood glucose data was not available, therefore the frequency was not estimable. Feig 2017 reported the median and interquartile range for the following outcomes, weight gain during pregnancy, postnatal weight retention or return to pre‐pregnancy weight, gestational age at birth and length of antenatal stay. Voormolen 2018 reported on many of the outcomes of this review (see Characteristics of included studies), but did not report these separately for pre‐gestational and gestational diabetes (we have written to authors requesting separate data for the pre‐gestational diabetes group of women).
Sources of trial funding
Sources of trial funding were not reported in two trials (Dalfrà 2009; di Biase 1997).
In Feig 2017, the trial was funded by the Juvenile Diabetes Research Foundation (JDRF) and grants under the JDRF Canadian Clinical Trial Network, a public‐private partnership. Metronic supplied the CGM sensors and CGM systems at reduced cost. In Hanson 1984, the source of funding was reported as being Expressens Perinatal forskningsfond, AIImanna Barnbordshusets Minnesfond, Svenska Diabetesstiftelsen, Nordisk Insulinfond, Swedish Medical Research Council (Project No. 3787), and Tielman's Fund for Pediatric Research. The Department of Health and Social Sevices, Northern lreland, the Northern Ireland Mother and Baby Appeal, the Metabolic Unit Research Fund, Royal Victoria Hospital Belfast, the Royal Maternity Hospital, and the Irish Perinatal Society funded the trial by Manderson 2003. Murphy 2008 was an investigator‐initiated study funded by the Ipswich Diabetes Centre Charity Research Fund. The study equipment (six CGMS Gold monitors and 300 sensors) was donated free of charge by Medtronic UK. The research was sponsored by Ipswich Hospital NHS Trust and was independent of all the study funders (Murphy 2008). The Macedonion Ministry of Health and the Health Care Fund of Macedonia funded Petrovski 2011. In Secher 2013, one of the authors received financial support from the European Foundation of the Study of Diabetes and LifeScan, Rigshopitalet's Research Foundation, the Capital Region of Denmark, the Medical Facuty Foundation of Copenhagen University, Aase and Ejnar Danielsen Foundation, and Master Joiner Sophus Jacobsen and his wife Astrid Jacobsens' Foundation. Stubbs 1980 was funded by the Medical Research Council Project Grant and the British Diabetic Association. Varner 1983 was funded by a Research Fellowship from the Iowa Affiliate of the American Diabetes Association. Voormolen 2018 was funded by ZonMw, The Dutch Organization for Health Research and Development .Continuous glucose monitors were purchased at a discounted price from Medtronic® and were reported as having no role in the study design, data collection, data analysis, data interpretation, or writing of the report. Wojcicki 2001 was supported by grants from the Polish State Committee for Scientific Research, the Bayer Diagnostic Division Warsaw, and the Polish Cellular Telephony Centertel.
Trial authors' declarations of interest
Trial authors' declarations of interest were not reported in Dalfrà 2009; di Biase 1997; Manderson 2003; Stubbs 1980; Varner 1983; Wojcicki 2001.
In Murphy 2008, two trial authors received honorariums for speaking at research symposiums sponsored by Medtronic in 2004 and 2005. In Feig 2017, eight authors report grants from the Juvenile Diabetes Research Foundation during the conduct of the study. Two authors report personal fees from Novo Nordisk, Roche and Medtronic, outside the submitted work. One author reports personal fees from Abbott Diabetes Care and Medtronic (MiniMed Academia), outside the submitted work. One author sits on the Medtronic European Scientific Advisory Board. All remaining authors declare no competing interests. The authors declared that they had no competing financial interests in Petrovski 2011 and in Secher 2013, other than those reported under 'funding' interests. In Voormolen 2018, one of the trial authors received a research grant from ZonMW (the Netherlands Organization for Health Research and Development) and a second author received research grants from Abbott, Dexcom, Medtronic and Sensonics, and also received personal fees from Roche Diabetes Care and Sensonics. A third author is supported by an NHMRC Practitioner Fellowshop (GNT1082548) and reports consultancy for ObsEVa, Merck and Guerbet. All other authors declare no support from any organization or conflict of interest.
See the Characteristics of included studies table for more details.
Excluded studies
No new trials were excluded in this update (2019), but one trial report was identified relating to an already excluded study (Bartholomew 2011).
Bartholomew 2011 was excluded as it is a cross‐over trial. Two trial registrations (NCT01630759; Walker 1999) were excluded; the former was a trial on women with gestational diabetes mellitus (GDM) while the latter was a clinical trial registration containing insufficient evidence to assess. We contacted the author, but there were no available data or published reports. Temple 2006 was excluded as it was an abstract on an observational study of eight pregnant women with type 1 diabetes using continuous glucose monitoring system (CGMS).
See the Characteristics of excluded studies table for more details.
Risk of bias in included studies
Three of the 12 included studies were at low risk of bias (Feig 2017; Murphy 2008; Secher 2013), eight studies were at moderate risk of bias (di Biase 1997; Hanson 1984; Manderson 2003; Petrovski 2011; Stubbs 1980; Varner 1983; Voormolen 2018; Wojcicki 2001), and one study was at high risk of bias (Dalfrà 2009). See Figure 2 and Figure 3.
2.

'Risk of bias' summary: review authors' judgements about each risk of bias item for each included study.
3.
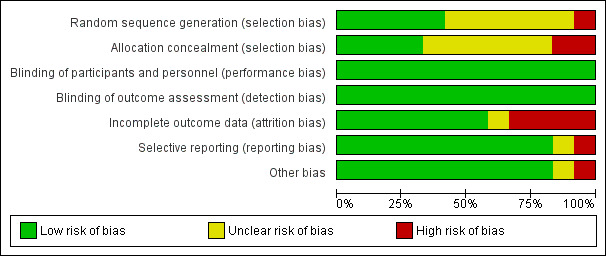
'Risk of bias' graph: review authors' judgements about each risk of bias item presented as percentages across all included studies.
Allocation
Random sequence generation
Five studies (Feig 2017; Murphy 2008; Secher 2013; Varner 1983; Voormolen 2018) described the random sequence generation using computer‐generated random numbers or a random number sequence (low risk of bias). Six trials (di Biase 1997; Hanson 1984; Manderson 2003; Petrovski 2011; Stubbs 1980; Wojcicki 2001) did not report how the sequence was generated (unclear risk of bias). One study was quasi‐randomised, allocating women to alternating groups (Dalfrà 2009) (high risk of bias).
Allocation concealment
Adequate and secure concealment of allocation was described in four trials (low risk of bias) (Feig 2017; Manderson 2003; Murphy 2008; Secher 2013); in one trial the randomisation schedule was created remotely by a programme manager, encrypted, and maintained in a secure database, with no access from the research team (Feig 2017), sealed envelopes were used in two of the trials (Manderson 2003; Murphy 2008), while the fourth (Secher 2013) used an automated telephone allocation service (Paravox) provided by an independent organisation. There was no concealment of allocation in Wojcicki 2001 and Dalfrà 2009 (high risk of bias). The other trials only mentioned the participants were randomly allocated into intervention or control groups without describing if there was any concealment of allocation (unclear risk of bias).
Blinding
Blinding of participants and personnel
As different techniques or timing of glucose monitoring were compared, blinding of neither participants nor assessors was feasible. However, since outcome measures were objective it is unlikely that lack of blinding introduced a risk of bias and so all studies were assessed as being at low risk.
Blinding of outcome assessors
As different techniques or timing of glucose monitoring were compared, blinding of neither participants nor assessors was feasible. However, since outcome measures were objective it is unlikely that lack of blinding introduced a risk of bias so all studies were assessed as being at low risk.
Incomplete outcome data
Four trials had high risk of bias for incomplete outcome data. Reasons given for attrition were women not completing the questionnaire (Dalfrà 2009), severe drug addiction, spontaneous abortions and death of mother (Hanson 1984), no results for analysis participants (Manderson 2003) and spontaneous miscarriage (Varner 1983). In other included studies, all women were accounted for in the analysis, or rates of attrition were described (low risk of bias). di Biase 1997and Wojcicki 2001 reported all outcome data. Four trials reported using intention‐to‐treat analysis (Murphy 2008; Petrovski 2011; Secher 2013; Stubbs 1980). One trial was assessed as being at unclear risk of bias (Voormolen 2018), because there were a high number of women refused to continue using CGM after the first or second time.
Selective reporting
It was unclear if there was any selective reporting in one trial (Dalfrà 2009),10 trials reported all expected outcome data (di Biase 1997; Feig 2017; Hanson 1984; Manderson 2003; Murphy 2008; Petrovski 2011; Secher 2013; Stubbs 1980; Varner 1983; Wojcicki 2001) (low risk of bias). One trial (Voormolen 2018) was assessed as being at high risk of bias because there were a number of maternal outcomes that were described in the methods of the full report and the protocol, but were not reported in the results section.
Other potential sources of bias
There were no other obvious potential sources of bias with the exception of Dalfrà 2009 and Voormolen 2018. Dalfrà 2009 did not use an intention‐to‐treat analysis, and there was no sample size calculation, or information on whether groups were comparable at baseline (high risk of bias). In Voormolen 2018, we had some concerns over missing outcomes and were unsure of the impact of this, (unclear risk of bias).
Effects of interventions
See: Table 1; Table 2; Table 3; Table 4; Table 5; Table 6
Summary of findings for the main comparison. Continuous glucose monitoring compared to intermittent glucose monitoring for women with pre‐existing diabetes.
| Continuous glucose monitoring compared to intermittent glucose monitoring for women with pre‐existing diabetes | ||||||
|
Patient or population: women with pre‐existing diabetes
Setting: 1 study in a hospital centre for pregnant women with diabetes in Denmark, 1 study in two secondary care multi‐disciplinary obstetric diabetic clinics in the UK, 1 multi‐centre study in 31 hospital and diabetic clinics in Canada, England, Scotland, Spain, Italy, Ireland and the USA, and 1 multi‐centre study in 22 hospital outpatient obstetric and endocrinology clinics (university, teaching and non‐teaching in the Netherlands and 1 university hospital in Belgium). Intervention: continuous glucose monitoring Comparison: intermittent glucose monitoring | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | Comments | |
| Risk with intermittent self‐glucose monitoring | Risk with continuous glucose monitoring | |||||
| Hypertensive disorders of pregnancy (including pre‐eclampsia, pregnancy‐induced hypertension, eclampsia) | Study population | RR 0.58 (0.39 to 0.85) | 384 (2 RCTs) | ⊕⊕⊝⊝ LOW 1 2 | ||
| 292 per 1000 | 170 per 1000 (114 to 248) | |||||
| Caesarean section | Study population | RR 0.94 (0.75 to 1.18) | 427 (3 RCTs) | ⊕⊕⊕⊝ MODERATE3 | ||
| 600 per 1000 | 564 per 1000 (450 to 708) | |||||
| Large‐for‐gestational age | Study population | RR 0.84 (0.57 to 1.26) | 421 (3 RCTs) | ⊕⊕⊝⊝ LOW 4 5 | ||
| 546 per 1000 | 459 per 1000 (311 to 688) | |||||
| Perinatal mortality (stillbirth and neonatal mortality) | Study population | RR 0.82 (0.05 to 12.61) | 71 (1 RCT) | ⊕⊕⊝⊝ LOW6 | ||
| 31 per 1000 | 26 per 1000 (2 to 394) | |||||
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: Confidence interval; RCT: randomised controlled trial; RR: Risk ratio | ||||||
| GRADE Working Group grades of evidence High certainty: We are very confident that the true effect lies close to that of the estimate of the effect Moderate certainty: We are moderately confident in the effect estimate. The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different Low certainty: Our confidence in the effect estimate is limited: The true effect may be substantially different from the estimate of the effect Very low certainty: We have very little confidence in the effect estimate: The true effect is likely to be substantially different from the estimate of effect | ||||||
1 We downgraded (1) level for serious limitations in study design due to unclear risk of allocation concealment and high risk for selective outcome reporting
2 We downgraded (1) level for serious indirectness due to the two studies reporting this composite outcome in different ways: Voormolen 2018 reported a composite of pregnancy‐induced hypertension and pre‐eclampsia for women with type 1 diabetes and type 2 diabetes for; and Feig 2017 reporting a composite of worsening chronic, gestational and pre‐eclampsia for women with type 1 diabetes
3 We downgraded (1) level for serious inconsistency due to evidence of statistical heterogeneity I2 = 41%
4 We downgraded (1) level for serious imprecision due to wide CI crossing the line of no effect
5 We downgraded (1) level for serious inconsistency due to evidence of statistical heterogeneity I2 = 70%
6 We downgrade (2) levels for very serious imprecision due to evidence derived from a single study, with a small number of events, wide CI crossing the line of no effect
Summary of findings 2. Self‐monitoring compared to a different type of self‐monitoring for women with pre‐existing diabetes.
| Self‐monitoring compared to standard care for women with pre‐existing diabetes | ||||||
| Patient or population: women with pre‐existing diabetes Setting: 1 study in a high‐risk obstetric clinic at University hospital in the USA Intervention: self‐monitoring Comparison: standard care | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Risk with standard care | Risk with self‐monitoring | |||||
| Hypertensive disorders of pregnancy (including pre‐eclampsia, pregnancy‐induced hypertension, eclampsia) | Study population | (0 studies) | The included study did not report this outcome. | |||
| Caesarean section | Study population | RR 0.78 (0.40 to 1.49) | 28 (1 RCT) | ⊕⊝⊝⊝ VERY LOW 1 2 | ||
| 643 per 1000 | 501 per 1000 (257 to 958) | |||||
| Large‐for‐gestational age | Study population | (0 studies) | The included study did not report this outcome. | |||
| Perinatal mortality (stillbirth and neonatal mortality) | Study population | RR 3.00 (0.13 to 67.91) | 28 (1 RCT) | ⊕⊝⊝⊝ VERY LOW 1 2 | There were no events in the standard care group and so anticipated absolute effects could not be calculated. | |
| 0 per 1000 | 0 per 1000 (0 to 0) | |||||
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: Confidence interval; RCT: randomised controlled trial; RR: Risk ratio | ||||||
| GRADE Working Group grades of evidence High quality: We are very confident that the true effect lies close to that of the estimate of the effect Moderate quality: We are moderately confident in the effect estimate: The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different Low quality: Our confidence in the effect estimate is limited: The true effect may be substantially different from the estimate of the effect Very low quality: We have very little confidence in the effect estimate: The true effect is likely to be substantially different from the estimate of effect | ||||||
1 We downgraded (1) level for serious limitations in design limitations due unclear allocation concealment and high risk for attrition
2 We downgraded (2) levels for very serious imprecision due to wide CI crossing the line of no effect, few events and small sample size
Summary of findings 3. Self‐monitoring at home compared to hospitalisation for women with pre‐existing diabetes.
| Self‐monitoring compared to hospitalisation for women with pre‐existing diabetes | ||||||
| Patient or population: women with pre‐existing diabetes Setting: 1 study in Sweden with monitoring at home or in hospital Intervention: self‐monitoring Comparison: hospitalisation | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Risk with hospitalisation | Risk with self‐monitoring | |||||
| Hypertensive disorders of pregnancy (including pre‐eclampsia, pregnancy‐induced hypertension, eclampsia) | Study population | RR 1.19 (0.41 to 3.51) |
100 (1 RCT) |
⊕⊝⊝⊝ VERY LOW1 2 | ||
| 109 per 1000 | 129 per 1000 (45 to 381) |
|||||
| Caesarean section | Study population | RR 0.96 (0.65 to 1.44) | 100 (1 RCT) | ⊕⊝⊝⊝ VERY LOW 1 2 | ||
| 500 per 1000 | 480 per 1000 (325 to 720) | |||||
| Large‐for‐gestational age | Study population | (0 studies) | The included study did not report this outcome. | |||
| Perinatal mortality (stillbirth and neonatal mortality) | Study population | RR 0.85 (0.05 to 13.24) | 100 (1 RCT) | ⊕⊝⊝⊝ VERY LOW 1 2 | ||
| 22 per 1000 | 18 per 1000 (1 to 288) | |||||
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: Confidence interval; RCT: randomised controlled trial; RR: Risk ratio | ||||||
| GRADE Working Group grades of evidence High quality: We are very confident that the true effect lies close to that of the estimate of the effect Moderate quality: We are moderately confident in the effect estimate: The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different Low quality: Our confidence in the effect estimate is limited: The true effect may be substantially different from the estimate of the effect Very low quality: We have very little confidence in the effect estimate: The true effect is likely to be substantially different from the estimate of effect | ||||||
1 We downgraded (1) level for serious limitations in study design due to unclear randomisation, allocation concealment and high risk for attrition
2 We downgraded (2) levels for very serious imprecision due to wide CI crossing the line of no effect, few events and small sample size
Summary of findings 4. Pre‐prandial compared to post‐prandial glucose monitoring for women with pre‐existing diabetes.
| Pre‐prandial compared to post‐prandial glucose monitoring for women with pre‐existing diabetes | ||||||
|
Patient or population: women with pre‐existing diabetes
Setting: 1 study in a joint metabolic and antenatal clinic in Belfast Intervention: pre‐prandial Comparison: post‐prandial glucose monitoring | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Risk with post‐prandial glucose monitoring | Risk with pre‐prandial | |||||
| Hypertensive disorders of pregnancy (including pre‐eclampsia, pregnancy‐induced hypertension, eclampsia) | Study population | (0 studies) | The included study did not report this composite outcome. | |||
| Caesarean section | Study population | RR 1.45 (0.92 to 2.28) | 61 (1 RCT) | ⊕⊝⊝⊝ VERY LOW 1 2 | ||
| 467 per 1000 | 677 per 1000 (429 to 1000) | |||||
| Large‐for‐gestational age | Study population | RR 1.16 (0.73 to 1.85) | 61 (1 RCT) | ⊕⊝⊝⊝ VERY LOW 1 2 | ||
| 500 per 1000 | 580 per 1000 (365 to 925) | |||||
| Perinatal mortality (stillbirth and neonatal mortality) | Study population | RR 2.91 (0.12 to 68.66) | 61 (1 RCT) | ⊕⊝⊝⊝ VERY LOW 1 2 | There were no events in the standard care group and so anticipated absolute effects could not be calculated. | |
| 0 per 1000 | 0 per 1000 (0 to 0) | |||||
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: Confidence interval; RCT: randomised controlled trial; RR: Risk ratio | ||||||
| GRADE Working Group grades of evidence High quality: We are very confident that the true effect lies close to that of the estimate of the effect Moderate quality: We are moderately confident in the effect estimate: The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different Low quality: Our confidence in the effect estimate is limited: The true effect may be substantially different from the estimate of the effect Very low quality: We have very little confidence in the effect estimate: The true effect is likely to be substantially different from the estimate of effect | ||||||
1 We downgraded (1) level for serious limitations in study design due to unclear methods of randomisation and high risk of attrition
2 We downgrade (2) levels for very serious limitations in imprecision due to wide CI crossing the line of no effect, few events and small sample size
Summary of findings 5. Automated telemedicine monitoring compared to conventional for women with pre‐existing diabetes.
| Automated telemedicine monitoring compared to conventional for women with pre‐existing diabetes | ||||||
| Patient or population: women with pre‐existing diabetes Setting: 2 studies in antenatal diabetic clinics in Italy, 1 study in gastroenterology and metabolic diseases clinic in Poland Intervention: automated telemedicine monitoring Comparison: conventional monitoring | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Risk with conventional monitoring | Risk with automated telemedicine monitoring | |||||
| Hypertensive disorders of pregnancy (including pre‐eclampsia, pregnancy‐induced hypertension, eclampsia) | Study population | (0 studies) | The included studies did not report this composite outcome. | |||
| Caesarean section | Study population | RR 0.96 (0.62 to 1.48) | 32 (1 RCT) | ⊕⊝⊝⊝ VERY LOW 1 2 | ||
| 733 per 1000 | 704 per 1000 (455 to 1000) | |||||
| Large‐for‐gestational age | Study population | (0 studies) | The included studies did not report this outcome. | |||
| Perinatal mortality (stillbirth and neonatal mortality) | Study population | (0 studies) | The included studies did not report this outcome. | |||
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: Confidence interval; RCT: randomised controlled trial; RR: Risk ratio | ||||||
| GRADE Working Group grades of evidence High quality: We are very confident that the true effect lies close to that of the estimate of the effect Moderate quality: We are moderately confident in the effect estimate: The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different Low quality: Our confidence in the effect estimate is limited: The true effect may be substantially different from the estimate of the effect Very low quality: We have very little confidence in the effect estimate: The true effect is likely to be substantially different from the estimate of effect | ||||||
1 We downgraded (2) levels for very serious design limitations due to high risk for randomisation, allocation concealment, attrition and other bias
2 We downgraded (2) levels for very serious imprecision due to wide CI crossing the line of no effect, few events and small sample size
Summary of findings 6. Constant CGM compared to Intermittent CGM for women with pre‐existing diabetes.
| Constant CGM compared to Intermittent CGM for women with pre‐existing diabetes | ||||||
| Patient or population: women with pre‐existing diabetes Setting: 1 study in University clinic of endocrinology, diabetes and metabolic disorders in Macedonia Intervention: constant CGM Comparison: intermittent CGM | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Risk with Intermittent CGM | Risk with constant CGM | |||||
| Hypertensive disorders of pregnancy (including pre‐eclampsia, pregnancy‐induced hypertension, eclampsia) | Study population | (0 studies) | The included study did not report this outcome. | |||
| Caesarean section | Study population | RR 0.77 (0.33 to 1.79) | 25 (1 RCT) | ⊕⊝⊝⊝ VERY LOW 1 2 | ||
| 538 per 1000 | 415 per 1000 (178 to 964) | |||||
| Large‐for‐gestational age | Study population | (0 studies) | The included study did not report this outcome. | |||
| Perinatal mortality (stillbirth and neonatal mortality) | Study population | (0 studies) | The included study did not report this outcome. | |||
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: Confidence interval; RCT: randomised controlled trial; RR: Risk ratio | ||||||
| GRADE Working Group grades of evidence High quality: We are very confident that the true effect lies close to that of the estimate of the effect Moderate quality: We are moderately confident in the effect estimate: The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different Low quality: Our confidence in the effect estimate is limited: The true effect may be substantially different from the estimate of the effect Very low quality: We have very little confidence in the effect estimate: The true effect is likely to be substantially different from the estimate of effect | ||||||
1 We downgraded (1) level for serious limitations in design due to unclear randomisation and allocation concealment
2 We downgraded (2) levels for very serious limitations in imprecision due to wide CI crossing the line of no effect, few events and small sample size
As there were various methods of glucose monitoring being implemented in the included trials, we structured the review using the following comparisons.
Continuous glucose monitoring (CGM) versus intermittent glucose monitoring
Self‐monitoring versus different types of self‐monitoring
Self‐monitoring at home versus hospitalisation
Pre‐prandial versus post‐prandial glucose monitoring
Automated telemedicine monitoring versus conventional system
Constant CGM versus intermittent CGM
Comparison 1 ‐ Continuous glucose monitoring (CGM) versus intermittent glucose monitoring
See Table 1.
Four studies compared CGM versus intermittent blood glucose monitoring (Feig 2017; Murphy 2008; Secher 2013; Voormolen 2018). The total number of women was 609, 384 type 1 diabetes (T1DM) and 191 with type 2 diabetes (T2DM). Feig 2017 contributed the largest number of women in this comparison (n = 215), all T1DM.
Feig 2017 used a CGM system to measure blood glucose. Women were trained to use the study devices and instructed to use them daily by local diabetes or antenatal clinic teams. CGM users were advised to verify the accuracy of their CGM measurements using their capillary glucose meter before insulin dose adjustment (n = 108). Women in the control group continued their usual method of capillary glucose monitoring (n = 107). Women in both groups were advised to test capillary blood glucose levels at least seven times daily and given written instructions for how to use capillary or CGM measures for insulin dose adjustment. Feig 2017 randomised both pregnant (n = 215) and women planning pregnancy (n = 110), but reported the results separately for the two cohorts. We have only included the data for the pregnant women in this review.
Voormolen 2018 randomised 300 pregnant women with T1DM (n = 109) and T2DM (n = 82), or with gestational diabetes (n = 109). We have only included the data for the women with T1DM and T2DM. However, many of our review outcomes were not reported separately, but mixed with the gestational diabetes cohort and so we have been unable to include all of the data. The CGM group had continuous glucose monitoring in addition to standard care. Women allocated to CGM were instructed to use the device for five to seven days every six weeks and glucose profiles were obtained retrospectively, directly after each use and evaluated by the local endocrinologist. Self‐monitoring of blood glucose (SMBG) was required for calibration of CGM. Readings from the CGM were uploaded to a web‐based program (n = 147 all women, 90 with T1DM and T2DM). Standard treatment consisted of self‐monitoring of blood glucose only (n = 153 all women, 97 with T1DM and T2DM). Women in both intervention and control groups performed SMBG (four to eight times/day: at least fasting, after every meal, at bedtime and, preferably before every meal).
Murphy 2008 used the CGM, which measured glucose in subcutaneous tissues every 10 seconds and an average value is stored every five minutes, providing up to 288 measurements per day (n = 38). The women were required to wear the CGM for seven days at intervals of four to six weeks. They were also advised to measure blood glucose at least seven times a day. The intermittent monitoring of glucose levels was the standard care in which women were advised to monitor glucose at least seven times a day (n = 33).
In Secher 2013, real time CGM for six days at pregnancy visits during eight, 12, 21, 27 and 33 weeks, in addition to routine pregnancy care was implemented on 79 women and intermittent monitoring with self‐monitored plasma glucose measurements of seven times daily was implemented on 75 women.
Primary outcomes
Continuous glucose monitoring may reduce the composite outcome of hypertensive disorders of pregnancy (risk ratio (RR) 0.58, 95% confidence interval (CI) 0.39 to 0.84; 2 studies, 384 women; low‐quality evidence,Analysis 1.1), although this did not translate into a clear reduction for pre‐eclampsia, see secondary outcomes below.
1.1. Analysis.
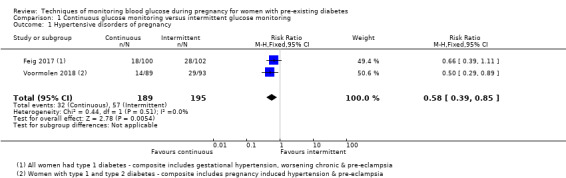
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 1 Hypertensive disorders of pregnancy.
Due to moderate heterogeneity, we used random‐effects analysis for caesarean section and there was no clear reduction in rate of caesarean section (average RR 0.94, 95% CI 0.75 to 1.18; 3 studies, 427 women; I2 = 41%; moderate‐quality evidence, Analysis 1.2), or large‐for‐gestational age (average RR 0.84, 95% CI 0.57 to 1.26; 3 studies, 421 women; I2 = 70%; low‐quality evidence, Analysis 1.3). There was not enough evidence to assess perinatal mortality (RR 0.82, 95% CI 0.05 to 12.61, 71 infants, 1 study, Analysis 1.4, low‐quality evidence) or the composite of neonatal mortality or morbidity (RR 0.80, 95% CI 0.61 to 1.06; 1 study, 200 women) as the evidence was based on single studies of low‐quality.
1.2. Analysis.
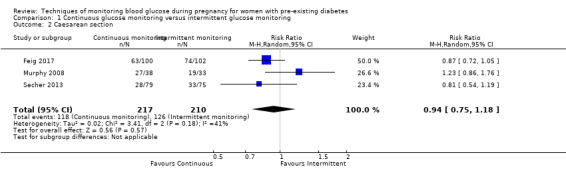
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 2 Caesarean section.
1.3. Analysis.
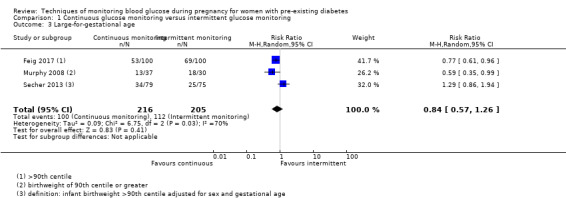
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 3 Large‐for‐gestational age.
1.4. Analysis.
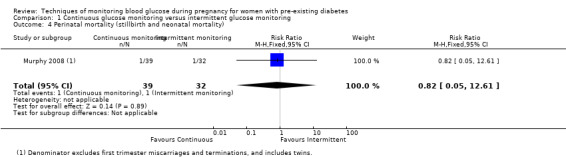
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 4 Perinatal mortality (stillbirth and neonatal mortality).
Neurosensory disability This outcome was not reported.
Secondary outcomes
There was no clear reduction in pre‐eclampsia (RR 0.65, 95% CI 0.39 to 1.08; 4 studies, 609 women; moderate‐quality evidence,Analysis 1.6) or to pregnancy‐induced hypertension (RR 0.67, 95% CI 0.38 to 1.16; 2 studies, 384 women; low‐quality evidence,Analysis 1.7) with CGM. There was little difference between groups in behaviour changes associated with the intervention as measured using the Hypoglycaemia Fear Survey (HFS II) (Lamb 2017) behaviour subscale at 34 weeks' gestation (mean difference (MD) 1.00, 95% CI ‐1.06 to 3.06; 1 study, 214 women, Analysis 1.8). The 15 items in HFS behaviour subscale measures behaviours aimed at avoiding hypoglycaemia and its possible negative consequences, with higher scores indicating higher fear of hypoglycaemia, with scores ranging from zero to 60.
1.8. Analysis.
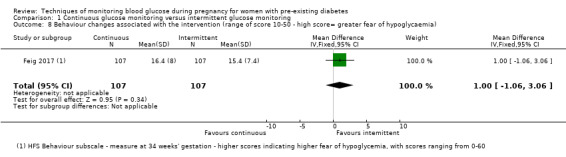
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 8 Behaviour changes associated with the intervention (range of score 10‐50 ‐ high score= greater fear of hypoglycaemia).
There was also little difference in sense of well‐being and quality of life as measured using the Short‐Form‐12 (SF‐12) and Problem Areas in Diabetes (PAID) (Venkataraman 2015) which were measured at 34 weeks' gestation (total score) (MD ‐0.70, 95% CI ‐2.50 to 1.10; 1 study, 214 women, Analysis 1.9) (MD 0.80, 95% CI ‐3.06 to 4.66; 1 study, 214 women, Analysis 1.10). The Short‐Form‐12 (SF‐12) is a health‐related quality‐of‐life questionnaire. It consists of 12 questions that measure eight domains assessing physical and mental health. Physical health domains include General Health (GH), Physical Functioning (PF), Role Physical (RP), and Body Pain (BP). Mental health domains include Vitality (VT), Social Functioning (SF), Role Emotional (RE), and Mental Health (MH). The instrument has been validated across a number of chronic diseases and conditions. Physical and Mental Health Composite Scales combine the 12 items, with a high score of 50 equating to good mental and physical health (Huo 2018). The Problem Areas in Diabetes (PAID) instrument was developed to measure emotional distress in people with diabetes. It is a 20‐item scale consisting of emotional problems commonly reported in type 1 and type 2 diabetes mellitus, and has been found to be a valid and reliable scale in Western populations (Venkataraman 2015), with scores ranging from zero to 100, with higher scores reflecting greater emotional distress. The CGM group scored slightly higher in their Blood Glucose Monitoring System Rating Questionnaire (BGMSRQ) (Peyrot 2009) total score at 34 weeks' gestation (MD 4.30, 95% CI 0.73 to 7.87; 1 study, 214 women, Analysis 1.11). This questionnaire aims to measure different aspects about the method of blood glucose monitoring: measures relate to convenience, interference, burden, control, overall satisfaction, desire to switch monitoring system, willingness to recommend current monitoring system, and comparison of current and prior blood glucose monitoring system.
1.9. Analysis.
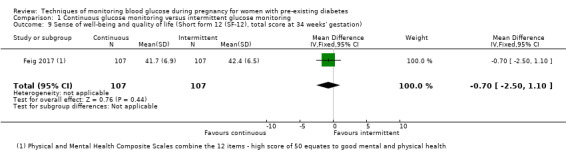
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 9 Sense of well‐being and quality of life (Short form 12 (SF‐12), total score at 34 weeks' gestation).
1.10. Analysis.
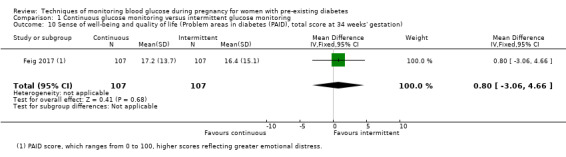
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 10 Sense of well‐being and quality of life (Problem areas in diabetes (PAID), total score at 34 weeks' gestation).
1.11. Analysis.
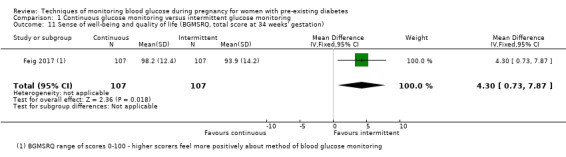
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 11 Sense of well‐being and quality of life (BGMSRQ, total score at 34 weeks' gestation).
CGM may make little or no difference to maternal glycaemic control as indicated by HbA1c levels (glycated haemoglobin): the mean HbA1c level in the continuous monitoring group was 0.37% lower, 0.78% lower to 0.04% higher (MD ‐0.37 %, 95% CI ‐0.78 to 0.04; 2 studies, 258 women; I2 = 81%, Analysis 1.12) and although more women in the continuous monitoring group achieved HbA1c levels less than or equal to 6.5% (48 mmol/mol) at 34 weeks (RR 1.27, 95% CI 1.00 to 1.62; 1 study, 187 women, Analysis 1.13), the results are based on a single study.
1.12. Analysis.
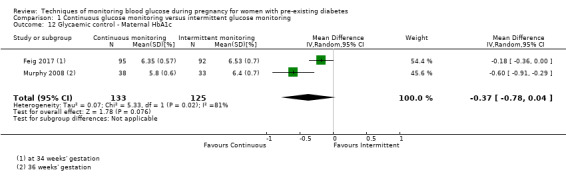
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 12 Glycaemic control ‐ Maternal HbA1c.
1.13. Analysis.
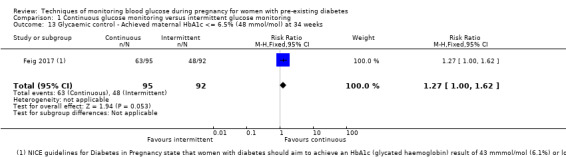
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 13 Glycaemic control ‐ Achieved maternal HbA1c <= 6.5% (48 mmol/mol) at 34 weeks.
There was no clear difference between groups for the following outcomes.
Maternal hypoglycaemia (severe) (RR 0.92, 95% CI 0.43 to 1.95; 1 study,154 women, Analysis 1.14)
Miscarriage (RR 1.24, 95% CI 0.47 to 3.26; 3 studies, 439 women, Analysis 1.15)
Stillbirth (RR 0.34, 95% CI 0.01 to 8.17; 1 study, 211 infants, Analysis 1.16)
Neonatal mortality (RR 0.92, 95% CI 0.13 to 6.37; 2 studies, 256 infants, Analysis 1.17)
Gestational age at birth (weeks) (MD 0.10 weeks, 95% CI ‐0.57 to 0.77; 1 study, 68 women, Analysis 1.18)
Preterm birth < 37 weeks (RR 0.96, 95% CI 0.72 to 1.29; 3 studies, 430 women, Analysis 1.19)
Preterm birth < 34 weeks (RR 0.46, 95% CI 0.17 to 1.28; 1 study, 211 women, Analysis 1.20)
Macrosomia (average RR 0.84, 95% CI 0.61 to 1.17; 3 studies, 451 women, I2 = 34%, Analysis 1.21)
Birthweight (kg) (MD ‐0.13 kg, 95% CI ‐0.38 to 0.12; 2 studies, 267 infants, I2 = 49%, Analysis 1.22)
Small‐for‐gestational age (RR 2.40, 95% CI 0.55 to 10.51; 2 studies, 269 infants, Analysis 1.23)
Head circumference (cm) (MD ‐0.20, 95% CI ‐0.79 to 0.39; 1 study, 160 infants, Analysis 1.24)
Length (crown‐heel length cm) (MD ‐0.20, 95% CI ‐0.79 to 0.39; 1 study, 160 infants, Analysis 1.25)
Adipositiy (sum of four skin folds mm) (MD ‐0.20, 95% CI ‐1.98 to 1.58; 1 study, 160 infants, Analysis 1.26)
Shoulder dystocia (RR 3.00, 95% CI 0.12 to 72.77; 1 study, 200 infants, Analysis 1.27)
Respiratory distress syndrome (RR 1.00, 95% CI 0.41 to 2.41; 1 study, 200 infants, Analysis 1.28)
Neonatal hypoglycaemia (RR 0.66, 95% CI 0.48 to 0.93; 3 studies, 428 infants, Analysis 1.29)
Neonatal hyperbilirubinaemia (RR 0.81, 95% CI 0.52 to 1.26; 1 study, 200 infants, Analysis 1.30)
Relevant biomarkers associated with the intervention (cord blood c‐peptide levels > 566 pmol/L) (RR 0.95, 95% CI 0.68 to 1.33; 1 study, 200 infants, Analysis 1.31)
Relevant biomarkers associated with the intervention (cord blood c‐peptide levels > 2725 pmol/L) (RR 1.00, 95% CI 0.33 to 3.00; 1 study, 200 infants, Analysis 1.32)
Major and minor anomalies (RR 0.71, 95% CI 0.16 to 3.13; 2 studies, 285 infants, Analysis 1.33)
Number of hospital admissions (mother) (RR 1.25, 95% CI 0.84 to 1.85; 1 study, 207 women, Analysis 1.34)
Neonatal intensive care unit admissions (average RR 0.76, 95% CI 0.42 to 1.35; 2 studies, 274 infants, Analysis 1.35)
Birth trauma (shoulder dystocia, bone fracture, nerve palsy) (RR 5.00, 95% CI 0.24 to 102.85; 1 study, 200 infants, Analysis 1.37)
Diabetic ketoacidosis (RR 1.01, 95% CI 0.14 to 7.03; 1 study, 207 women, Analysis 1.38)
1.14. Analysis.
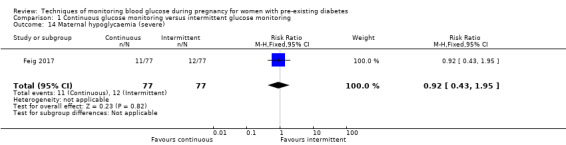
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 14 Maternal hypoglycaemia (severe).
1.15. Analysis.
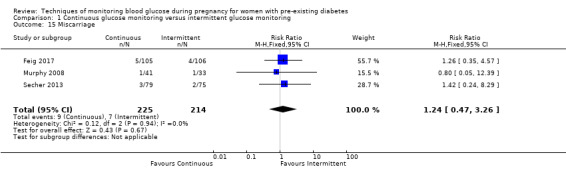
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 15 Miscarriage.
1.16. Analysis.
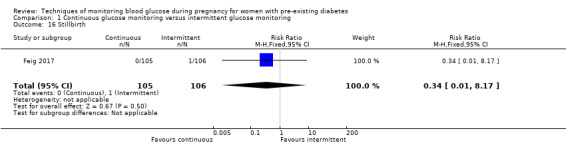
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 16 Stillbirth.
1.17. Analysis.
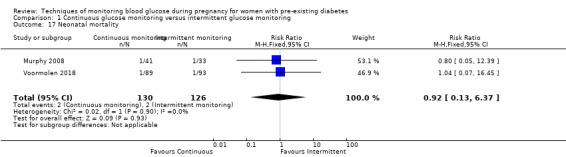
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 17 Neonatal mortality.
1.18. Analysis.
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 18 Gestational age at birth.
1.19. Analysis.
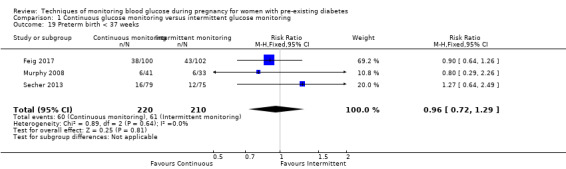
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 19 Preterm birth < 37 weeks.
1.20. Analysis.
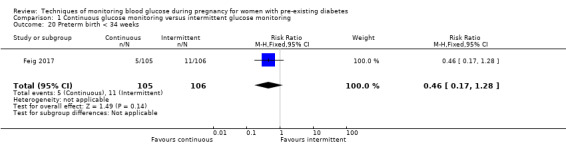
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 20 Preterm birth < 34 weeks.
1.21. Analysis.
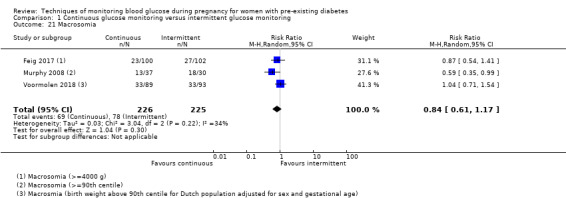
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 21 Macrosomia.
1.22. Analysis.
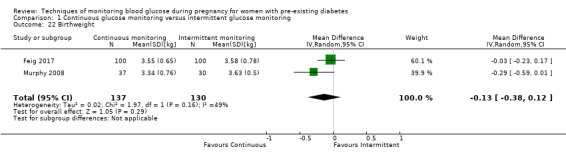
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 22 Birthweight.
1.23. Analysis.
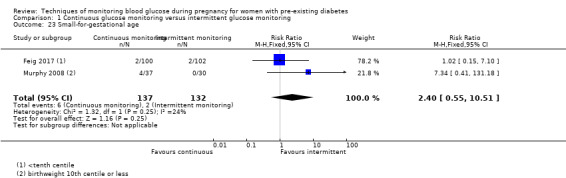
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 23 Small‐for‐gestational age.
1.24. Analysis.
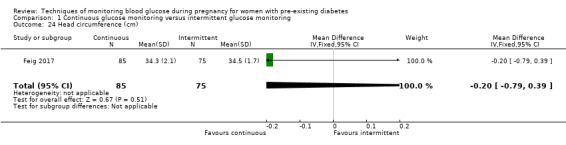
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 24 Head circumference (cm).
1.25. Analysis.
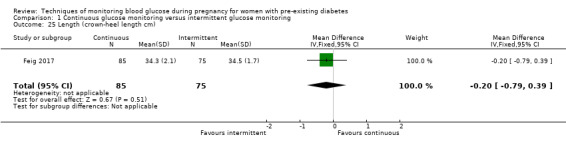
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 25 Length (crown‐heel length cm).
1.26. Analysis.
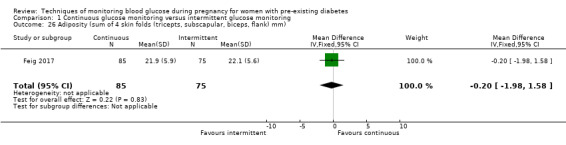
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 26 Adiposity (sum of 4 skin folds (tricepts, subscapular, biceps, flank) mm).
1.27. Analysis.
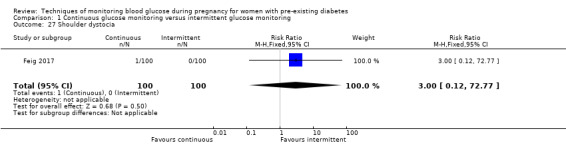
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 27 Shoulder dystocia.
1.28. Analysis.
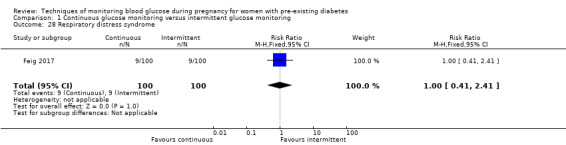
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 28 Respiratory distress syndrome.
1.29. Analysis.
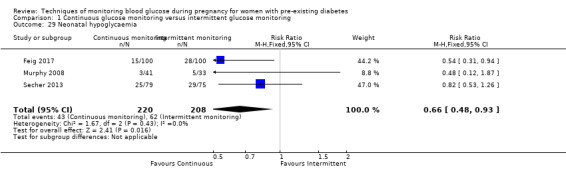
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 29 Neonatal hypoglycaemia.
1.30. Analysis.
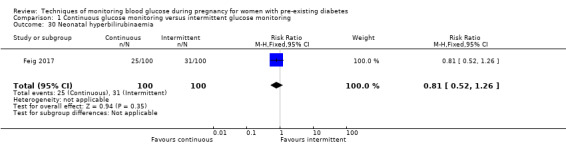
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 30 Neonatal hyperbilirubinaemia.
1.31. Analysis.
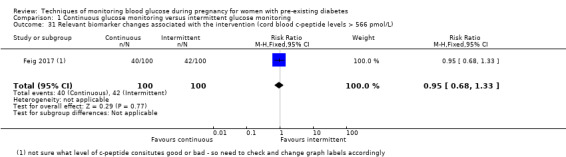
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 31 Relevant biomarker changes associated with the intervention (cord blood c‐peptide levels > 566 pmol/L).
1.32. Analysis.
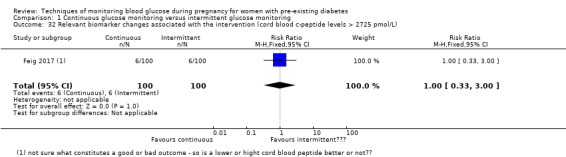
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 32 Relevant biomarker changes associated with the intervention (cord blood c‐peptide levels > 2725 pmol/L).
1.33. Analysis.
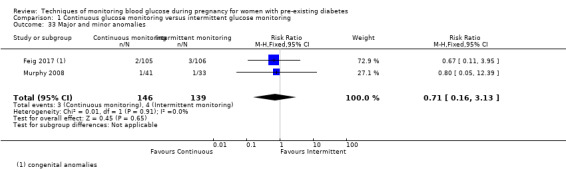
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 33 Major and minor anomalies.
1.34. Analysis.
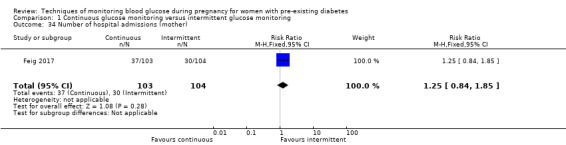
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 34 Number of hospital admissions (mother).
1.35. Analysis.
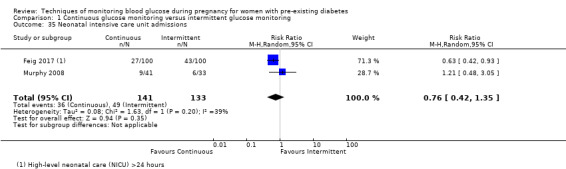
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 35 Neonatal intensive care unit admissions.
1.37. Analysis.
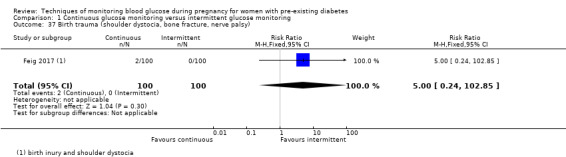
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 37 Birth trauma (shoulder dystocia, bone fracture, nerve palsy).
1.38. Analysis.
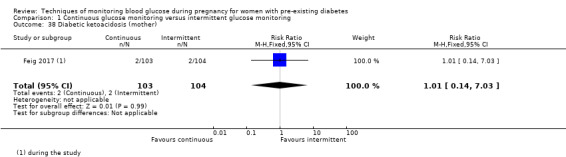
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 38 Diabetic ketoacidosis (mother).
It may reduce neonatal hypoglycaemia (RR 0.66, 95% CI 0.48 to 0.93; 3 studies, 428 infants, Analysis 1.29) and NICU admission of more than 24 hours (RR 0.63, 95% CI 0.42 to 0.93; 1 study, 200 infants, Analysis 1.36).
1.36. Analysis.
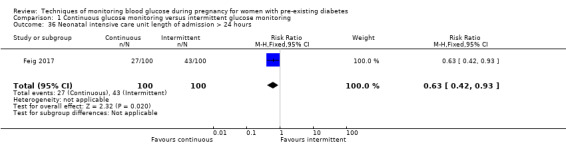
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 36 Neonatal intensive care unit length of admission > 24 hours.
None of the studies reported on our remaining secondary outcomes.
Comparison 2 ‐ Self‐monitoring versus a different type of self‐monitoring
See Table 2.
Two trials (Stubbs 1980; Varner 1983) compared self‐monitoring with a different type of self‐monitoring (standard care). In one trial (Stubbs 1980), a total of 13 pregnant women with T1DM were randomly allocated into self‐monitoring of blood glucose (SMBG) at home, seven times a day, twice per week. Another six women were allocated to standard care (urine check four times daily) and random blood glucose testing measured fortnightly during clinic visits.
In the other trial (Varner 1983), 30 T1DM women were assigned to self‐monitoring (n = 15) and standard care (n = 15). The self‐monitoring group carried out daily home glucose monitoring four times daily and the standard care group carried out weekly venipuncture three times daily, measured on one day weekly. One woman in each group had a first trimester spontaneous miscarriage, so results are presented for the remaining 28 women and infants. The self‐monitoring group was required to monitor fasting plus two‐hour post‐prandial morning, afternoon and evening glucose daily, while the standard care group were measured one day per week.
Primary outcomes
It is uncertain whether self‐monitoring reduces the risk of caesarean section (risk ratio (RR) 0.78, 95% confidence interval (CI) 0.40 to 1.49, 1 study, 28 women, Analysis 2.1, very low‐quality evidence) (Varner 1983). Varner 1983 also reported perinatal mortality and it was too small to show any differences between groups (perinatal mortality: RR 3.00, 95% CI 0.13 to 67.91, 1 study, 28 infants, very low‐quality evidence, Analysis 2.2).
2.1. Analysis.
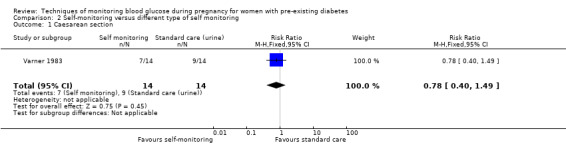
Comparison 2 Self‐monitoring versus different type of self monitoring, Outcome 1 Caesarean section.
2.2. Analysis.
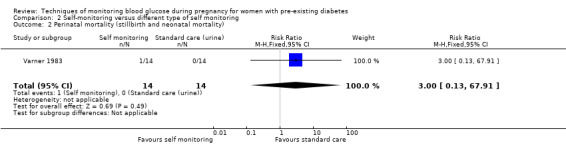
Comparison 2 Self‐monitoring versus different type of self monitoring, Outcome 2 Perinatal mortality (stillbirth and neonatal mortality).
Hypertensive disorders of pregnancy, large‐for‐gestational age, neonatal mortality and morbidity composite and neurosensory disability were not reported in either study.
Secondary outcomes
It is uncertain whether self‐monitoring makes any difference in maternal glycaemic control for post‐prandial blood glucose (MD ‐0.70 mmol/L, 95% CI ‐2.15 to 0.75; 1 study, 13 women, Analysis 2.3), or HbA1c (MD ‐0.10 %, 95% CI ‐1.93 to 1.73, 1 study, 28 women, Analysis 2.4). There were too few women to show any differences in miscarriage (RR 1.00, 95% CI 0.07 to 14.55, 1 study, 30 women, Analysis 2.5), neonatal mortality (RR 3.00, 95% CI 0.13 to 67.91, 1 study, 28 women, Analysis 2.6), or respiratory distress syndrome (RR 3.00, 95% CI 0.13 to 67.91, 1 study, 28 infants, Analysis 2.9). It is uncertain whether there are any differences in gestational age between self‐monitoring groups (MD 0.40 weeks, 95% CI ‐1.65 to 2.45, 1 study, 28 infants, Analysis 2.7), and or in infant birthweight (MD ‐0.18 kg, 95% CI ‐0.49 to 0.13, 2 studies, 41 infants, Analysis 2.8) due to limitations in small sample sizes.
2.3. Analysis.
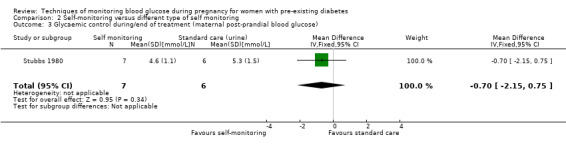
Comparison 2 Self‐monitoring versus different type of self monitoring, Outcome 3 Glycaemic control during/end of treatment (maternal post‐prandial blood glucose).
2.4. Analysis.
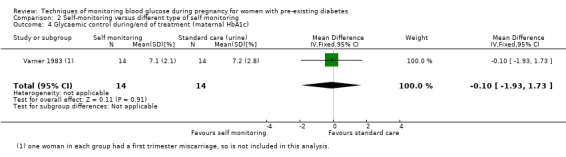
Comparison 2 Self‐monitoring versus different type of self monitoring, Outcome 4 Glycaemic control during/end of treatment (maternal HbA1c).
2.5. Analysis.
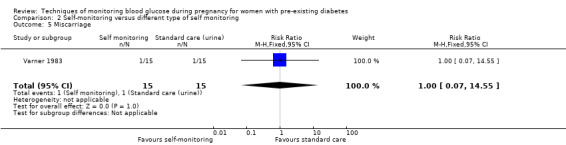
Comparison 2 Self‐monitoring versus different type of self monitoring, Outcome 5 Miscarriage.
2.6. Analysis.

Comparison 2 Self‐monitoring versus different type of self monitoring, Outcome 6 Neonatal mortality.
2.9. Analysis.
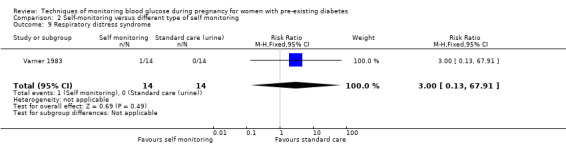
Comparison 2 Self‐monitoring versus different type of self monitoring, Outcome 9 Respiratory distress syndrome.
2.7. Analysis.
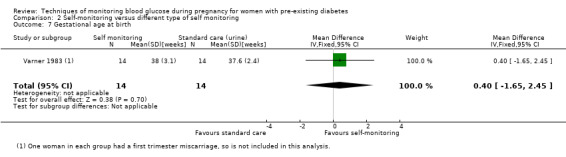
Comparison 2 Self‐monitoring versus different type of self monitoring, Outcome 7 Gestational age at birth.
2.8. Analysis.
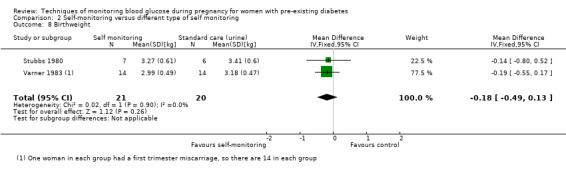
Comparison 2 Self‐monitoring versus different type of self monitoring, Outcome 8 Birthweight.
Again it is uncertain whether there are any differences for neonatal hypoglycaemia (RR 0.57, 95% CI 0.21 to 1.52, 1 study, 28 infants, Analysis 2.10), neonatal jaundice (hyperbilirubinaemia) (RR 0.56, 95% CI 0.25 to 1.24, 1 study, 28 infants, Analysis 2.11), hypocalcaemia (RR 1.00, 95% CI 0.07 to 14.45, 1 study, 28 infants, Analysis 2.12), polycythaemia (RR 0.33, 95% CI 0.01 to 7.55, 1 study, 28 infants, Analysis 2.13) and neonatal cord vein C‐peptide (MD 0.13 ng/nl, 95% CI ‐0.50 to 0.76, 1 study, 28 infants, Analysis 2.14).
2.10. Analysis.
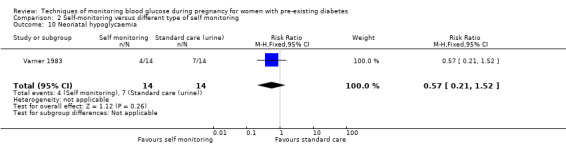
Comparison 2 Self‐monitoring versus different type of self monitoring, Outcome 10 Neonatal hypoglycaemia.
2.11. Analysis.
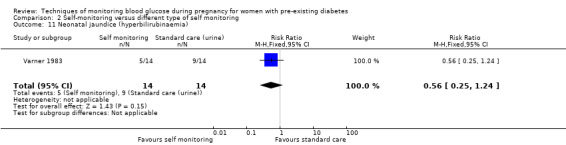
Comparison 2 Self‐monitoring versus different type of self monitoring, Outcome 11 Neonatal jaundice (hyperbilirubinaemia).
2.12. Analysis.
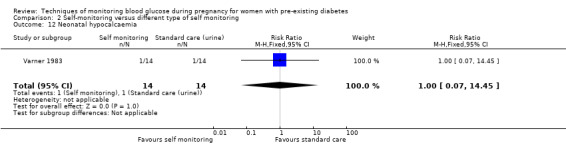
Comparison 2 Self‐monitoring versus different type of self monitoring, Outcome 12 Neonatal hypocalcaemia.
2.13. Analysis.
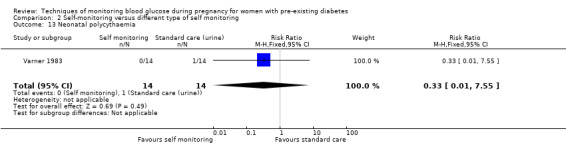
Comparison 2 Self‐monitoring versus different type of self monitoring, Outcome 13 Neonatal polycythaemia.
2.14. Analysis.
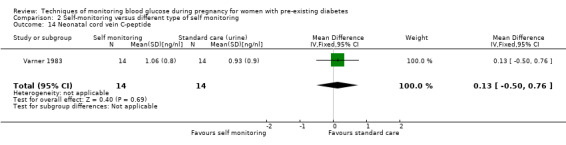
Comparison 2 Self‐monitoring versus different type of self monitoring, Outcome 14 Neonatal cord vein C‐peptide.
None of the studies reported on our remaining secondary outcomes.
Comparison 3 ‐ Self‐monitoring at home versus hospitalisation
See Table 3.
Only one study compared home self‐monitoring with hospitalisation (Hanson 1984). In this study, a total of 100 pregnant women with T1DM and T2DM were randomised. The home self‐monitoring group had 54 women while the hospital group had 46 women. The women from the home group self‐monitored their blood glucose from the 32nd until 36th week of gestation and then were hospitalised during the 37th week until delivery; the hospital group women were hospitalised from 32nd week until delivery. Blood glucose was monitored four times daily (7 AM, 9.30 AM, 3 PM and 7 PM) in both groups.
Primary outcomes
This study of 100 women did not report on the composite outcome, hypertensive disorders of pregnancy. It reported pre‐eclampsia and hypertension in pregnancy, but as separate outcomes.
The results were uncertain for caesarean section (RR 0.96, 95% CI 0.65 to 1.44, Analysis 3.2, very low‐quality evidence), and the sample size was too small to assess perinatal mortality (RR 0.85, 95% CI 0.05 to 13.24, Analysis 3.3, very low‐quality evidence).
3.2. Analysis.
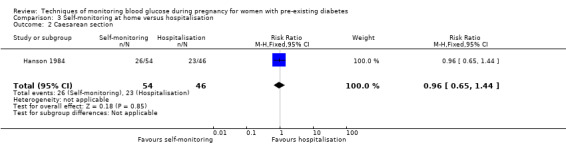
Comparison 3 Self‐monitoring at home versus hospitalisation, Outcome 2 Caesarean section.
3.3. Analysis.
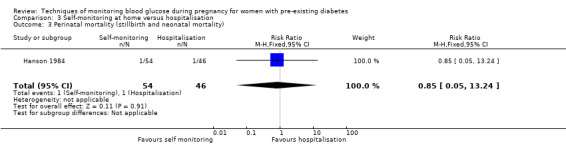
Comparison 3 Self‐monitoring at home versus hospitalisation, Outcome 3 Perinatal mortality (stillbirth and neonatal mortality).
Large‐for‐gestational age, mortality or morbidity composite, and neurosensory disability were not reported.
Secondary outcomes
It is uncertain whether there is any difference between self‐monitoring and hospitalisation were shown in the reported secondary outcomes: placental abruption (RR 1.70, 95% CI 0.16 to 18.19, Analysis 3.6); preterm birth < 37 weeks (RR 0.85, 95% CI 0.45 to 1.60, Analysis 3.7); respiratory distress syndrome (RR 2.56, 95% CI 0.28 to 23.74, Analysis 3.8); neonatal hypoglycaemia (RR 1.01, 95% CI 0.50 to 2.03, Analysis 3.9); neonatal jaundice (hyperbilirubinaemia) (RR 2.27, 95% CI 0.64 to 8.07, Analysis 3.10); major anomalies (RR 0.27, 95% CI 0.03 to 2.54, Analysis 3.11).
3.6. Analysis.
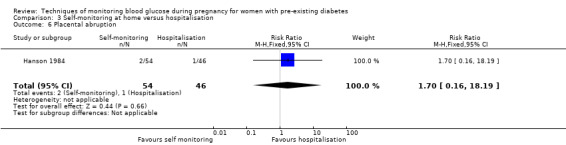
Comparison 3 Self‐monitoring at home versus hospitalisation, Outcome 6 Placental abruption.
3.7. Analysis.
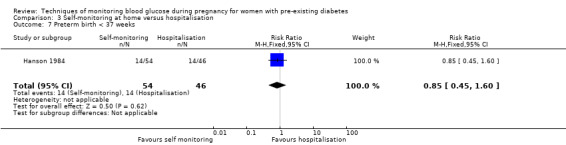
Comparison 3 Self‐monitoring at home versus hospitalisation, Outcome 7 Preterm birth < 37 weeks.
3.8. Analysis.
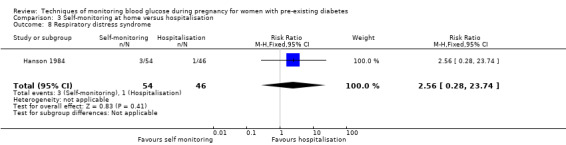
Comparison 3 Self‐monitoring at home versus hospitalisation, Outcome 8 Respiratory distress syndrome.
3.9. Analysis.
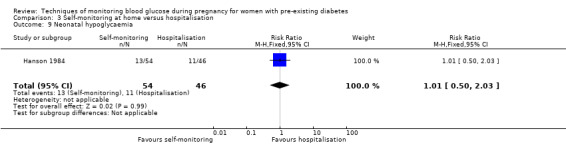
Comparison 3 Self‐monitoring at home versus hospitalisation, Outcome 9 Neonatal hypoglycaemia.
3.10. Analysis.
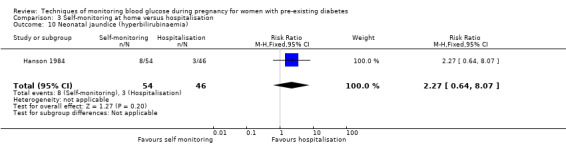
Comparison 3 Self‐monitoring at home versus hospitalisation, Outcome 10 Neonatal jaundice (hyperbilirubinaemia).
3.11. Analysis.
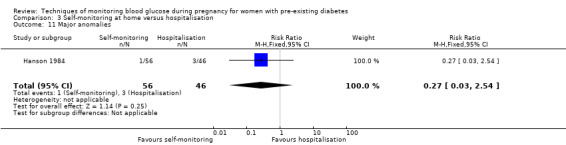
Comparison 3 Self‐monitoring at home versus hospitalisation, Outcome 11 Major anomalies.
As would be expected from the nature of the intervention, a lower proportion of women in the self‐monitoring group had antenatal hospital admission (RR 0.19, 95% CI 0.11 to 0.33, Analysis 3.12).
3.12. Analysis.
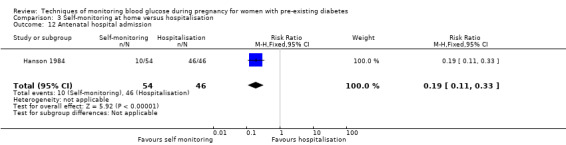
Comparison 3 Self‐monitoring at home versus hospitalisation, Outcome 12 Antenatal hospital admission.
Maternal glycaemic control was reported, however only mean blood glucose was given without standard deviations, and HbA1c was only presented graphically, so we were not able to include these data in the analyses. The mean blood glucose values during the study period were 6.0 mmol/L for the hospital group and 5.9 mmol/L for the home group.
Outcomes that were not pre‐specified
There were no differences between self‐monitoring and hospitalisation in terms of feeding difficulties (RR 0.85, 95% CI 0.41 to 1.78, Analysis 3.13).
3.13. Analysis.
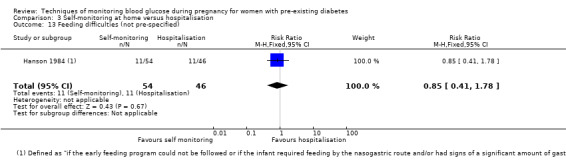
Comparison 3 Self‐monitoring at home versus hospitalisation, Outcome 13 Feeding difficulties (not pre‐specified).
None of the studies reported on our remaining secondary outcomes.
Comparison 4 ‐ Pre‐prandial versus post‐prandial glucose monitoring
See Table 4.
Only one trial compared pre‐prandial and post‐prandial glucose monitoring (Manderson 2003). Sixty‐one T1DM women were randomly assigned to pre‐prandial (n = 31) or post‐prandial (n = 30) blood glucose monitoring. The pre‐prandial group monitored their blood glucose before breakfast and pre‐prandially for each meal. The post‐prandial group monitored blood glucose after breakfast and one hour after the commencement of each meal.
Primary outcomes
In one study of 61 women (61 infants), it is uncertain whether there is any difference between pre‐prandial and post‐prandial glucose monitoring for caesarean section (RR 1.45, 95% CI 0.92 to 2.28, Analysis 4.1, very low‐quality evidence), large‐for‐gestational age (RR 1.16, 95% CI 0.73 to 1.85; Analysis 4.2, very low‐quality evidence) and perinatal mortality (RR 2.91, 95% CI 0.12 to 68.66, Analysis 4.3, very low‐quality evidence).
4.1. Analysis.
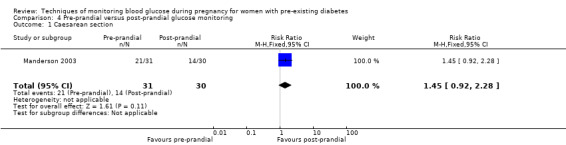
Comparison 4 Pre‐prandial versus post‐prandial glucose monitoring, Outcome 1 Caesarean section.
4.2. Analysis.
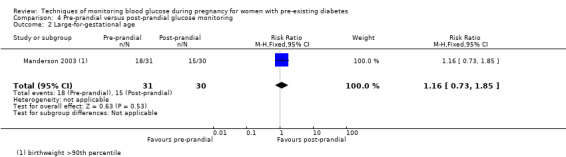
Comparison 4 Pre‐prandial versus post‐prandial glucose monitoring, Outcome 2 Large‐for‐gestational age.
4.3. Analysis.
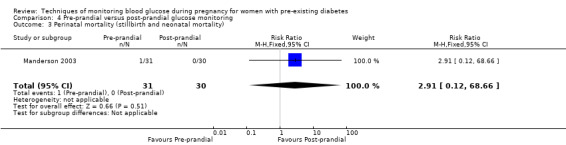
Comparison 4 Pre‐prandial versus post‐prandial glucose monitoring, Outcome 3 Perinatal mortality (stillbirth and neonatal mortality).
The study did not report the composite outcomes, hypertensive disorders of pregnancy (including pre‐eclampsia, pregnancy‐induced hypertension, eclampsia), mortality or morbidity composite, or neurosensory disability.
Secondary outcomes
It is uncertain whether there are any differences between pre‐prandial and post‐prandial glucose monitoring for the following outcomes: pre‐eclampsia (RR 6.43, 95% CI 0.82 to 50.11, Analysis 4.4); weight gain in pregnancy (MD ‐0.90 kg, 95% CI ‐3.86 to 2.06, Analysis 4.5); use of additional pharmacotherapy shown by insulin dose in units/day and units/kg (MD ‐17.40 units/day, 95% CI ‐43.41 to 8.61, Analysis 4.6; MD ‐0.20 units/kg, 95% CI ‐0.45 to 0.05, Analysis 4.7); glycaemic control shown by mean HbA1c (MD 0.30 %, 95% CI ‐0.08 to 0.68, Analysis 4.8); stillbirth (RR 2.91, 95% CI 0.12 to 68.66, Analysis 4.9); gestational age at birth (MD 0.20 weeks, 95% CI ‐0.84 to 1.24, Analysis 4.10); preterm birth < 37 weeks (RR 1.33, 95% CI 0.62 to 2.84, Analysis 4.11); macrosomia (RR 2.18, 95% CI 0.75 to 6.32, Analysis 4.12), birthweight (MD 0.24 kg, 95% CI ‐0.10 to 0.58, Analysis 4.13); subscapular skinfold thickness (adiposity) (MD 0.60 mm, 95% CI ‐0.18 to 1.38, Analysis 4.14); birth trauma (shoulder dystocia, bone fracture, nerve palsy) (RR 0.48, 95% CI 0.05 to 5.06, Analysis 4.16); respiratory distress syndrome (RR 0.97, 95% CI 0.06 to 14.78, Analysis 4.17); neonatal hypoglycaemia (RR 1.09, 95% CI 0.48 to 2.45, Analysis 4.18); neonatal jaundice (hyperbilirubinaemia) (RR 1.16, 95% CI 0.40 to 3.40, Analysis 4.19); cord IGF‐1 (MD 1.30 μg/L, 95% CI ‐0.70 to 3.30, Analysis 4.20); neonatal glucose at age one hour (not pre‐specified) (MD ‐0.20, 95% CI ‐0.88 to 0.48, Analysis 4.21); transient tachypnoea (not pre‐specified) (RR 2.58, 95% CI 0.76 to 8.81, Analysis 4.22); and neonatal intensive care admissions (RR 1.04, 95% CI 0.62 to 1.74, Analysis 4.23).
4.4. Analysis.
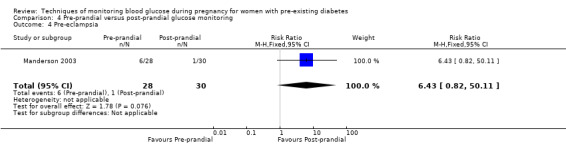
Comparison 4 Pre‐prandial versus post‐prandial glucose monitoring, Outcome 4 Pre‐eclampsia.
4.5. Analysis.
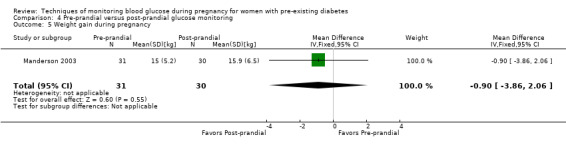
Comparison 4 Pre‐prandial versus post‐prandial glucose monitoring, Outcome 5 Weight gain during pregnancy.
4.6. Analysis.
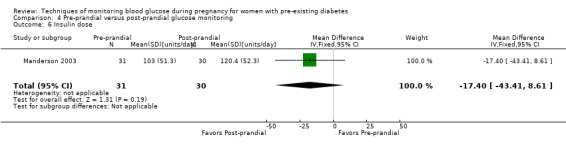
Comparison 4 Pre‐prandial versus post‐prandial glucose monitoring, Outcome 6 Insulin dose.
4.7. Analysis.
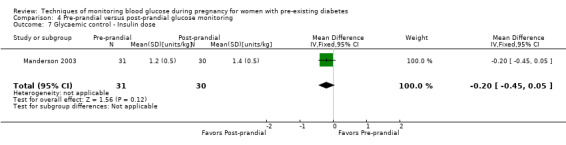
Comparison 4 Pre‐prandial versus post‐prandial glucose monitoring, Outcome 7 Glycaemic control ‐ Insulin dose.
4.8. Analysis.
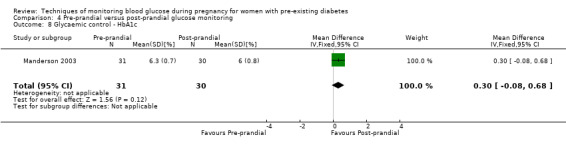
Comparison 4 Pre‐prandial versus post‐prandial glucose monitoring, Outcome 8 Glycaemic control ‐ HbA1c.
4.9. Analysis.
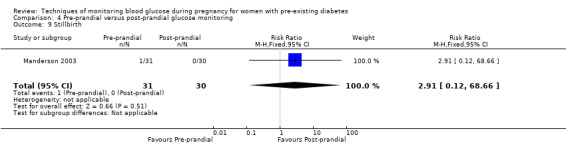
Comparison 4 Pre‐prandial versus post‐prandial glucose monitoring, Outcome 9 Stillbirth.
4.10. Analysis.
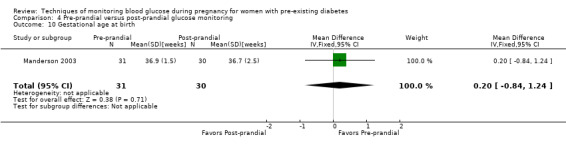
Comparison 4 Pre‐prandial versus post‐prandial glucose monitoring, Outcome 10 Gestational age at birth.
4.11. Analysis.
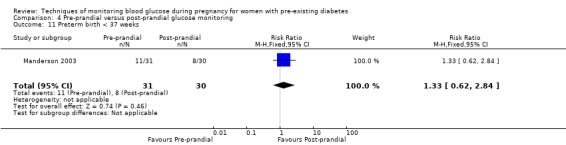
Comparison 4 Pre‐prandial versus post‐prandial glucose monitoring, Outcome 11 Preterm birth < 37 weeks.
4.12. Analysis.
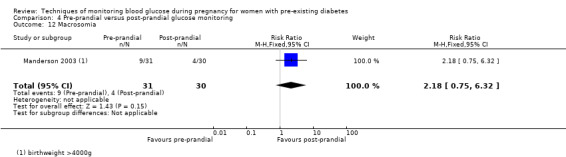
Comparison 4 Pre‐prandial versus post‐prandial glucose monitoring, Outcome 12 Macrosomia.
4.13. Analysis.
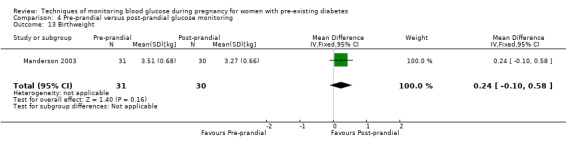
Comparison 4 Pre‐prandial versus post‐prandial glucose monitoring, Outcome 13 Birthweight.
4.14. Analysis.
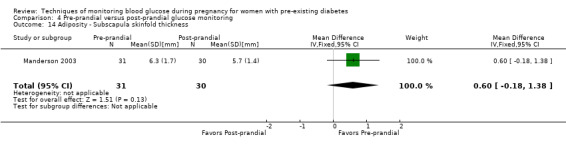
Comparison 4 Pre‐prandial versus post‐prandial glucose monitoring, Outcome 14 Adiposity ‐ Subscapula skinfold thickness.
4.16. Analysis.
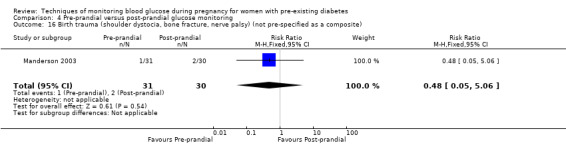
Comparison 4 Pre‐prandial versus post‐prandial glucose monitoring, Outcome 16 Birth trauma (shoulder dystocia, bone fracture, nerve palsy) (not pre‐specified as a composite).
4.17. Analysis.
Comparison 4 Pre‐prandial versus post‐prandial glucose monitoring, Outcome 17 Respiratory distress syndrome.
4.18. Analysis.
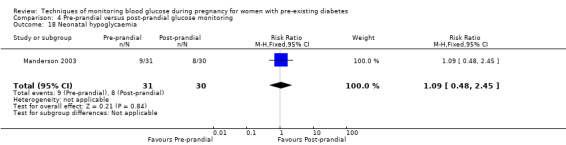
Comparison 4 Pre‐prandial versus post‐prandial glucose monitoring, Outcome 18 Neonatal hypoglycaemia.
4.19. Analysis.
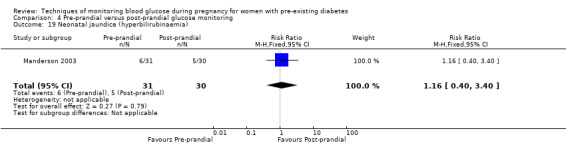
Comparison 4 Pre‐prandial versus post‐prandial glucose monitoring, Outcome 19 Neonatal jaundice (hyperbilirubinaemia).
4.20. Analysis.
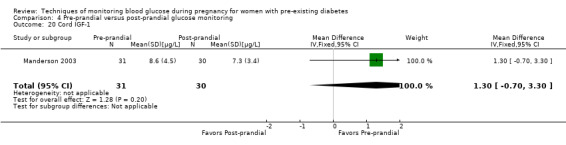
Comparison 4 Pre‐prandial versus post‐prandial glucose monitoring, Outcome 20 Cord IGF‐1.
4.21. Analysis.
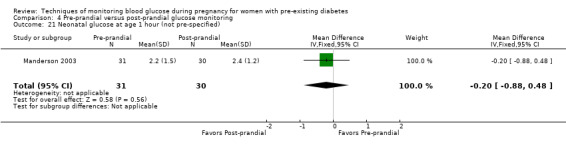
Comparison 4 Pre‐prandial versus post‐prandial glucose monitoring, Outcome 21 Neonatal glucose at age 1 hour (not pre‐specified).
4.22. Analysis.
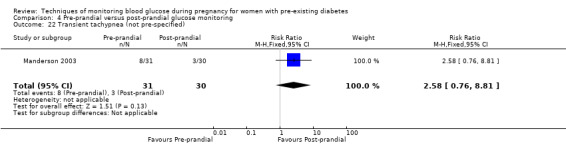
Comparison 4 Pre‐prandial versus post‐prandial glucose monitoring, Outcome 22 Transient tachypnea (not pre‐specified).
4.23. Analysis.
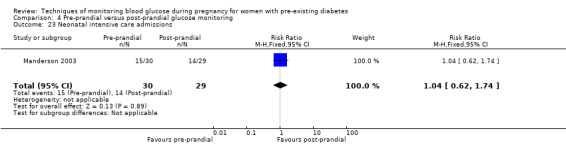
Comparison 4 Pre‐prandial versus post‐prandial glucose monitoring, Outcome 23 Neonatal intensive care admissions.
Infants in the pre‐prandial monitoring group had higher triceps skinfold thickness (adiposity) (MD 0.60 mm, 95% CI 0.04 to 1.16, Analysis 4.15), although the difference is small and should be considered in the context of no clear difference in large‐for‐gestational age, birthweight, macrosomia, and subscapular skinfold thickness.
4.15. Analysis.
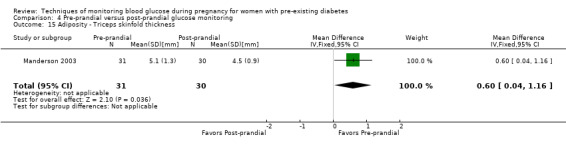
Comparison 4 Pre‐prandial versus post‐prandial glucose monitoring, Outcome 15 Adiposity ‐ Triceps skinfold thickness.
None of the studies reported on our remaining secondary outcomes.
Comparison 5 ‐ Automated telemedicine monitoring versus conventional system
See Table 5.
Three studies (Dalfrà 2009; di Biase 1997; Wojcicki 2001) compared automated telemedicine monitoring versus conventional system. Dalfrà 2009 included both pregnant women with T1DM (n = 32, data included in this review) and women with gestational diabetes (n = 203, data excluded from this review). Women in the telemedicine group were asked to submit their blood glucose data every week, and had a medical examination at the diabetes clinic once a month, while women in the control group had a medical examination every two weeks. di Biase 1997 (n = 20) and Wojcicki 2001 (n = 32) recruited T1DM women. di Biase 1997 used a DIANET system, which was an automated monitoring system using a telemedicine system with patient unit, diabetes workstation and the communication link to send all data to the diabetologist. The intermittent monitoring was conventional monitoring where the women were instructed to perform three or more tests of blood glucose per day using BM20‐800 strips with the results checked during routine clinic visits. Wojcicki 2001 used a telematic management system with the a glucometer connected to a modem interface where the blood glucose measurements could be transmitted to the central clinical control unit. The conventional group would only have their measurements examined during the routine clinical examinations every three weeks. All women (in both groups) were encouraged to measure their blood glucose at least six times per day.
Primary outcomes
Again, it is uncertain from one study (Dalfrà 2009) whether there is any difference between automated telemedicine monitoring and conventional monitoring for caesarean section (RR 0.96, 95% CI 0.62 to 1.48, 1 study, 32 women, Analysis 5.1, very low‐quality evidence) and the composite of neonatal mortality or morbidity (RR 1.18, 95% CI 0.53 to 2.62, 1 study, 32 infants, Analysis 5.2).
5.1. Analysis.
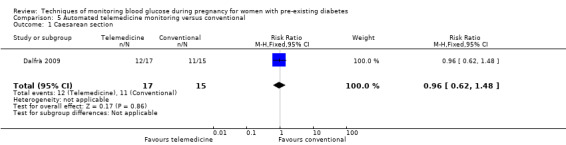
Comparison 5 Automated telemedicine monitoring versus conventional, Outcome 1 Caesarean section.
5.2. Analysis.
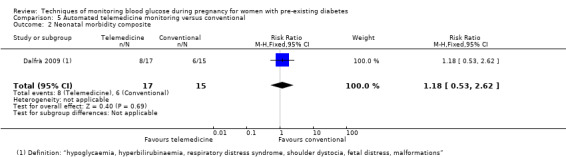
Comparison 5 Automated telemedicine monitoring versus conventional, Outcome 2 Neonatal morbidity composite.
di Biase 1997 and Wojcicki 2001 did not report these primary outcomes, and none of the studies contributing data to this comparison reported hypertensive disorders of pregnancy, large‐for‐gestational age, perinatal mortality (stillbirth and neonatal mortality), and neurosensory disability.
Secondary outcomes
In one study of 20 women (di Biase 1997), women in the automated telemedicine group had a higher mean insulin requirement at the end of the study (MD 18.40 units/day, 95% CI 12.88 to 23.92, Analysis 5.5). The women in the automated telemedicine group also had lower mean maternal fasting blood glucose before breakfast and before lunch at the end of the study (before breakfast: MD ‐1.00 mmol/L, 95% CI ‐1.22 to ‐0.78, Analysis 5.6; before lunch: MD ‐1.10 mmol/L, 95% CI ‐1.32 to ‐0.88, Analysis 5.7). There was high heterogeneity between studies for maternal HbA1c (random‐effects MD ‐0.17 %, 95% CI ‐0.82 to 0.48, 3 studies, 82 women, Tau² = 0.27, I² = 82%, Analysis 5.8) and maternal post‐prandial blood glucose (random‐effects MD ‐0.80 mmol/L, 95% CI ‐1.67 to 0.08, 3 studies, 50 women,Tau² = 0.35, I² = 86%, Analysis 5.9). Post hoc sensitivity analyses show that this was due to measurements from di Biase 1997. This study showed differences between groups in HbA1c and post‐prandial blood glucose, however the other two studies did not. It seems likely that the higher insulin doses given to women in the automated telemedicine group resulted in lower blood glucose measures.
5.5. Analysis.
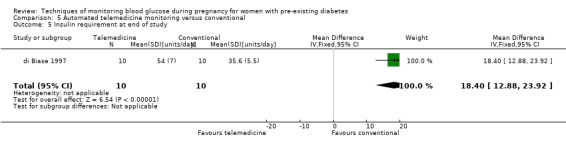
Comparison 5 Automated telemedicine monitoring versus conventional, Outcome 5 Insulin requirement at end of study.
5.6. Analysis.
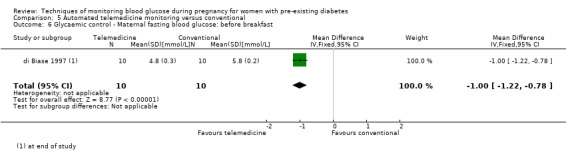
Comparison 5 Automated telemedicine monitoring versus conventional, Outcome 6 Glycaemic control ‐ Maternal fasting blood glucose: before breakfast.
5.7. Analysis.
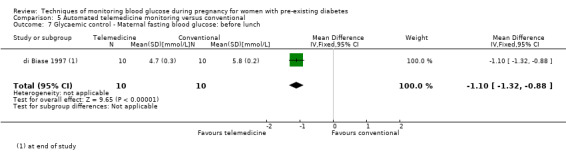
Comparison 5 Automated telemedicine monitoring versus conventional, Outcome 7 Glycaemic control ‐ Maternal fasting blood glucose: before lunch.
5.8. Analysis.
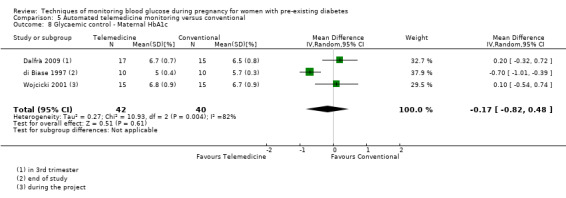
Comparison 5 Automated telemedicine monitoring versus conventional, Outcome 8 Glycaemic control ‐ Maternal HbA1c.
5.9. Analysis.
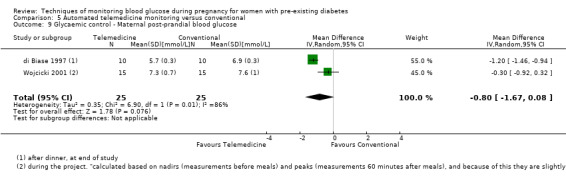
Comparison 5 Automated telemedicine monitoring versus conventional, Outcome 9 Glycaemic control ‐ Maternal post‐prandial blood glucose.
It was uncertain whether there was any difference between groups for: weight gain in pregnancy (MD ‐0.70, 95% CI ‐4.95 to 3.55, 1 study, 32 women, Analysis 5.10); use of additional insulin therapy (RR 1.00, 95% CI 0.89 to 1.12, 1 study, 32 women, Analysis 5.4); gestational age (MD 0.24 weeks, 95% CI ‐0.39 to 0.88, 3 studies, 84 women,Analysis 5.3); macrosomia (RR 1.18, 95% CI 0.31 to 4.43, 1 study, 32 infants, Analysis 5.11); or birthweight (MD ‐0.16 kg, 95% CI ‐0.64 to 0.32, 1 study, 32 infants, Analysis 5.12).
5.10. Analysis.
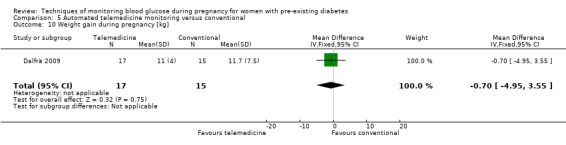
Comparison 5 Automated telemedicine monitoring versus conventional, Outcome 10 Weight gain during pregnancy [kg].
5.4. Analysis.
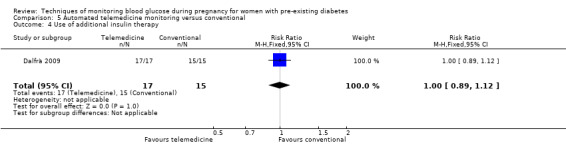
Comparison 5 Automated telemedicine monitoring versus conventional, Outcome 4 Use of additional insulin therapy.
5.3. Analysis.
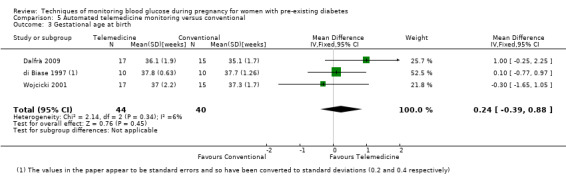
Comparison 5 Automated telemedicine monitoring versus conventional, Outcome 3 Gestational age at birth.
5.11. Analysis.
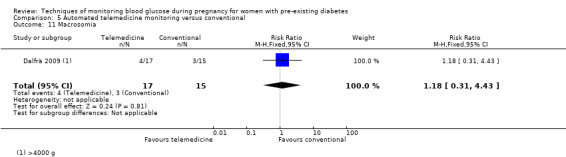
Comparison 5 Automated telemedicine monitoring versus conventional, Outcome 11 Macrosomia.
5.12. Analysis.
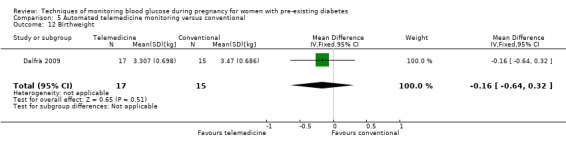
Comparison 5 Automated telemedicine monitoring versus conventional, Outcome 12 Birthweight.
The percentage of maternal hypoglycaemic episodes was reported by Wojcicki 2001, however, the total of all blood glucose data were not available, therefore the frequency was not estimable.
None of the studies reported on our remaining secondary outcomes.
Comparison 6 ‐ Constant CGM versus intermittent CGM
See Table 6.
Only one study compared constant CGM and intermittent CGM (Petrovski 2011). Twenty‐five T1DM women were randomised into constant CGM (n = 12) and intermittent CGM (n = 13) groups. The women in the constant CGM group wore the glucose sensor 24 hours per day while the intermittent CGM group wore the glucose sensor 14 days per month. The women in the intermittent CGM group measured blood glucose at least six times daily when not using the glucose sensor.
Primary outcomes
It is uncertain whether constant CGM makes any difference to rates of caesarean section (RR 0.77, 95% CI 0.33 to 1.79, 1 study, 25 women, very low‐quality evidence, Analysis 6.1). Other primary outcomes were not reported (hypertensive disorders of pregnancy, large‐for‐gestational age, perinatal mortality (stillbirth and neonatal mortality), mortality or morbidity composite, and neurosensory disability).
6.1. Analysis.
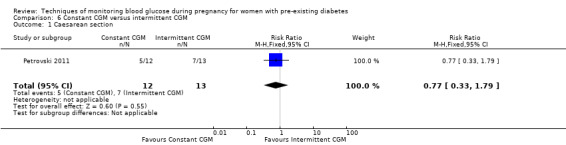
Comparison 6 Constant CGM versus intermittent CGM, Outcome 1 Caesarean section.
Secondary outcomes
Constant CGM makes little or no difference to weight gain in pregnancy (MD 0.50 kg, 95% CI ‐1.82 to 2.82, 1 study, 25 women, Analysis 6.2), insulin dosage (third trimester: MD ‐0.03, 95% CI ‐1.30 to 1.24, 1 study, 25 women, Analysis 6.3); maternal blood glucose (first trimester: MD ‐0.50 mmol/L, 95% CI ‐2.70 to 1.70, 1 study, 25 women, Analysis 6.4; third trimester: MD ‐0.14 mmol/L, 95% CI ‐2.00 to 1.72, 1 study, 25 women, Analysis 6.5); maternal HbA1c (first trimester: MD ‐0.30 %, 95% CI ‐1.13 to 0.53, 1 study, 25 women, Analysis 6.6; third trimester: MD ‐0.09 %, 95% CI ‐0.69 to 0.51, 1 study, 25 women, Analysis 6.7), maternal hypoglycaemia (RR 0.54, 95% CI 0.06 to 5.24, 1 study, 25 women, Analysis 6.8), diabetic ketoacidosis (not pre‐specified) (RR 0.36, 95% CI 0.02 to 8.05, 1 study, 25 women, Analysis 6.9), preterm birth < 37 weeks (RR 1.08, 95% CI 0.08 to 15.46, 1 study, 25 infants, Analysis 6.10), and macrosomia (RR 1.08, 95% CI 0.08 to 15.46, 1 study, 25 infants, Analysis 6.11). There were no events for neonatal hypoglycaemia (1 study, 25 infants Analysis 6.12).
6.2. Analysis.
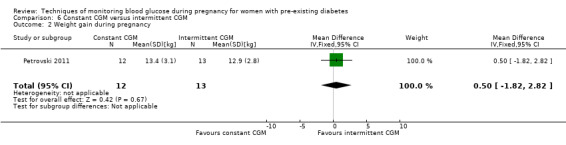
Comparison 6 Constant CGM versus intermittent CGM, Outcome 2 Weight gain during pregnancy.
6.3. Analysis.
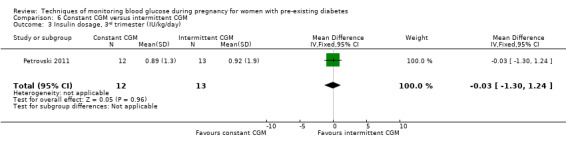
Comparison 6 Constant CGM versus intermittent CGM, Outcome 3 Insulin dosage, 3rd trimester (IU/kg/day).
6.4. Analysis.
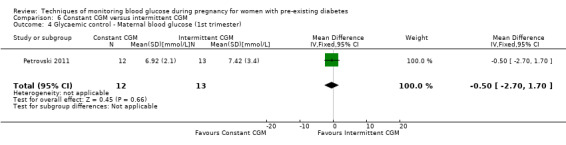
Comparison 6 Constant CGM versus intermittent CGM, Outcome 4 Glycaemic control ‐ Maternal blood glucose (1st trimester).
6.5. Analysis.
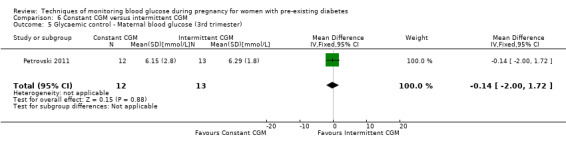
Comparison 6 Constant CGM versus intermittent CGM, Outcome 5 Glycaemic control ‐ Maternal blood glucose (3rd trimester).
6.6. Analysis.
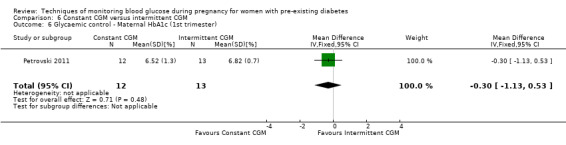
Comparison 6 Constant CGM versus intermittent CGM, Outcome 6 Glycaemic control ‐ Maternal HbA1c (1st trimester).
6.7. Analysis.
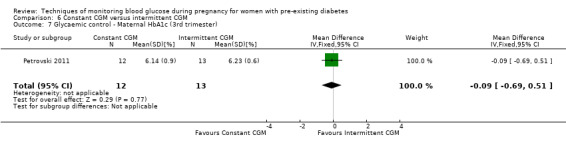
Comparison 6 Constant CGM versus intermittent CGM, Outcome 7 Glycaemic control ‐ Maternal HbA1c (3rd trimester).
6.8. Analysis.
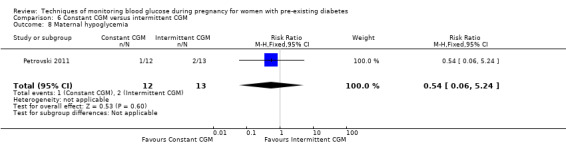
Comparison 6 Constant CGM versus intermittent CGM, Outcome 8 Maternal hypoglycemia.
6.9. Analysis.
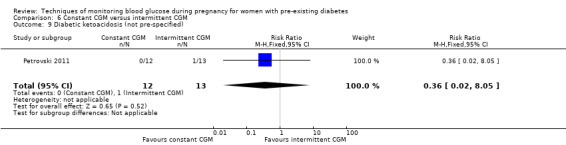
Comparison 6 Constant CGM versus intermittent CGM, Outcome 9 Diabetic ketoacidosis (not pre‐specified).
6.10. Analysis.
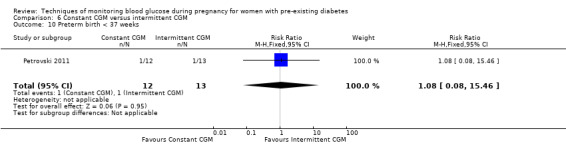
Comparison 6 Constant CGM versus intermittent CGM, Outcome 10 Preterm birth < 37 weeks.
6.11. Analysis.
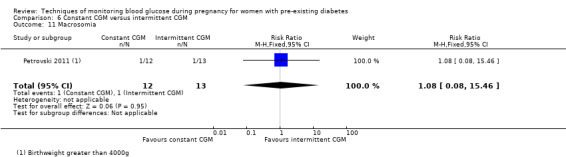
Comparison 6 Constant CGM versus intermittent CGM, Outcome 11 Macrosomia.
6.12. Analysis.
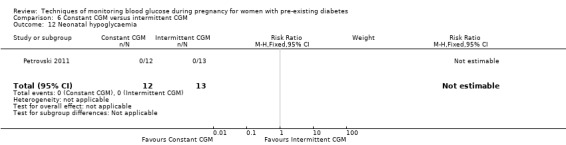
Comparison 6 Constant CGM versus intermittent CGM, Outcome 12 Neonatal hypoglycaemia.
None of the studies reported on our remaining secondary outcomes.
Discussion
Summary of main results
The objective of this review was to assess the various techniques of glucose monitoring among pregnant women with pre‐existing type 1 and type 2 diabetes and their impact on maternal and infant outcomes. We included 12 trials comparing six different pairs of glucose monitoring techniques: continuous glucose monitoring (CGM) versus intermittent glucose monitoring (Feig 2017; Murphy 2008; Secher 2013; Voormolen 2018), self‐monitoring versus a different type of self‐monitoring (Stubbs 1980; Varner 1983), self‐monitoring versus hospitalisation (Hanson 1984), pre‐prandial versus post‐prandial glucose monitoring (Manderson 2003), automated telemedicine monitoring versus conventional (Dalfrà 2009; di Biase 1997; Wojcicki 2001), and constant CGM versus intermittent CGM (Petrovski 2011). This review update includes a total of 12 trials (944) women (type 1 diabetes: 660 women; type 2 diabetes: 113 women; type 1 or type 2 (unspecified): 171 women. All trials originated from European countries, the USA and Canada.
With the addition of two new studies (406 women) to one of the comparisons (comparison 1 ‐ continuous glucose monitoring (CGM) versus intermittent glucose monitoring), the evidence suggests that CGM may reduce hypertensive disorders of pregnancy, though it should be noted that only two of the four relevant studies reported data for this composite outcome. Conversely, this did not translate into a clear reduction for pre‐eclampsia. There was no clear reduction in caesarean section or large‐for‐gestational age with CGM. There was not enough evidence to assess perinatal mortality or mortality or morbidity composite as the evidence was based on single studies of low quality. CGM appears to reduce neonatal hypoglycaemia. Neurosensory disability was not reported.
For the remaining five comparisons: self‐monitoring versus a different type of self‐monitoring (two studies, 43 women); self‐monitoring at home versus hospitalisation (one study, 100 women); pre‐prandial versus post‐prandial glucose monitoring (one study, 61 women); automated telemedicine monitoring versus conventional system (three studies, 84 women); and constant CGM versus intermittent CGM (one study, 25 women), it is uncertain whether any of the interventions has any impact on any of our GRADE outcomes (hypertensive disorders of pregnancy, caesarean section, large‐for‐gestational age) because the quality of the evidence was found to be very low. There was not enough evidence to assess perinatal mortality and neonatal mortality and morbidity composite. Other important outcomes, such as neurosensory disability, were not reported in any of these comparisons.
Outcomes relating to cost were not reported by any of the studies. Resource use (antenatal hospital admissions, neonatal intensive care admissions and neonatal intensive care unit length of admission > 24 hours) was reported only by single trials in three out of the six comparisons and so pooling of data was not possible.
Overall completeness and applicability of evidence
With the addition of two new studies (406 women) to one of the comparisons examining CGM versus intermittent monitoring, there was evidence to suggest that CGM may have an impact on hypertensive disorders of pregnancy, although there was no clear reduction for other outcomes associated with hypertension, such as pre‐eclampsia. The pooling of the data for hypertension disorders needs to be viewed with caution. Only two of the four relevant studies reported on this composite outcome of 'hypertensive disorders of pregnancy' including pre‐eclampsia, pregnancy‐induced hypertension, eclampsia, and they report it slightly differently (Feig 2017; Voormolen 2018). Voormolen 2018 reports a composite of pre‐eclampsia and pregnancy‐induced hypertension for women with both type 1 and type 2 diabetes and Feig 2017 reports a composite of worsening chronic, gestational hypertension and pre‐eclampsia for women with only type 1 diabetes. The ‘worsening chronic’ could be omitted from the analysis to make the two randomised controlled trials (RCTs) more comparable for pooling, although this makes only a small difference to the overall results (17/100 CGM and 27/102 versus 18/100 and 28/102). The real issue is that Murphy 2008 and Secher 2013 cannot be included in the analysis for this composite outcome because they both only report on pre‐eclampsia and not on pregnancy‐induced hypertension. The results for pre‐eclampsia therefore must be considered the most robust result in terms of complete reporting as all four RCTs report on it. The evidence base for the effectiveness of other monitoring techniques analysed in the other five comparisons is weak and based on mainly single studies with very low‐quality evidence and cannot be said to justify overall completeness of evidence.
All the included trials were conducted in Western countries ‐ Europe, the USA and Canada ‐ and it can be assumed that a majority of the women were Caucasian. Most of the pregnant women in the included studies had type 1 diabetes (n = 792) with much fewer having type 2 diabetes (n = 152). There were six pairs of intervention techniques in the included trials. There was difficulty in pooling the results due to this variation. The review's primary outcome, neurosensory disability, was not reported in any of the trials. The only secondary health service use outcomes reported were antenatal hospital admission, neonatal intensive care admissions and neonatal intensive care unit length of admission > 24 hours. Many of the reviews secondary maternal and perinatal/neonatal outcomes were not reported: induction of labour, perineal trauma, postpartum haemorrhage, postpartum infection, adherence to the intervention, maternal mortality, Apgar score (less than seven at five minutes). No studies reported long‐term maternal or infant outcomes and patient‐reported outcomes such as behaviour changes associated with the intervention and sense of well‐being and quality of life.
Quality of the evidence
Three of the 12 included studies were at low risk of bias (Feig 2017; Murphy 2008; Secher 2013), eight studies were at moderate risk of bias (di Biase 1997; Hanson 1984; Manderson 2003; Petrovski 2011; Stubbs 1980; Varner 1983; Voormolen 2018; Wojcicki 2001), and one study was at high risk of bias (Dalfrà 2009). Five trials (Feig 2017; Murphy 2008; Secher 2013; Varner 1983; Voormolen 2018) described the random sequence generation while adequate and secure concealment of allocation was described in four trials (Feig 2017; Manderson 2003; Murphy 2008; Secher 2013). It was unclear if there was any selective reporting in one trial (Dalfrà 2009), while 10 studies reported all expected outcome data (di Biase 1997; Feig 2017; Hanson 1984; Manderson 2003; Murphy 2008; Petrovski 2011; Secher 2013; Stubbs 1980; Varner 1983; Wojcicki 2001. In one trial, some outcomes reported in the protocol and methods of the trial report did not appear to have been adequately reported in the full trial report (Voormolen 2018) and was considered to be at high risk for this domain. Most of the trials had small numbers of women; six trials (Dalfrà 2009; Di Biase 1997; Petrovski 2011; Stubbs 1980; Varner 1983; Wojcicki 2001) only had a range of 13 to 32 participants. Any potential bias is likely to have been overshadowed by the small number and size of trials with their different intervention techniques of monitoring and reported outcomes. The trials are too small to show differences in important outcomes such as macrosomia, preterm birth, miscarriage or death of baby.
All the reported GRADE outcomes for comparisons 2, 3, 4, 5 and 6 were assessed as being very low‐quality evidence (Table 2; Table 3; Table 4; Table 5; Table 6). We downgraded most outcomes in these tables once for serious concerns due to limitations in design and twice for very serious imprecision (wide confidence intervals (CIs) crossing the line of no effect, small sample sizes, and few events).
Comparison 1 included more data than the other comparisons (four studies, 609 women), from studies assessed as being at lower risk of bias (Table 1). Consequently, we graded caesarean section as moderate‐quality evidence with downgrading one level for serious inconsistency due to evidence of statistical heterogeneity. We graded hypertensive disorders of pregnancy as low‐quality evidence with downgrading two levels for serious limitations in study design and serious indirectness due to two studies reporting the composite outcome in different ways. We graded large‐for‐gestational as low quality with downgrading two levels due to serious imprecision (wide CI crossing line of no effect) and serious inconsistency (statistical heterogeneity). We graded perinatal mortality (stillbirth and neonatal mortality) as low quality with downgrading two levels for very serious imprecision due to evidence derived from a single study, with a small number of events and wide CI crossing the line of no effect.
GRADE outcomes were often not reported. Caesarean section was the only GRADE outcome reported by studies in every comparison. The composite outcome of hypertensive disorders of pregnancy was only reported by studies in comparison 1. Large‐for‐gestational age was only reported by studies in comparisons 1 and 4. Perinatal mortality (stillbirth and neonatal mortality) was reported by studies in comparisons 1, 2, 3 and 4.
Potential biases in the review process
We attempted to minimise bias during the review process by having two people assess the eligibility of studies, assess risk of bias and extract data. GRADE quality assessments were also checked by a second person. We attempted to be as inclusive as possible in our search. However, we cannot rule out the possibility that we have missed relevant studies that were not published or are still ongoing. In addition, the proposed subgroup and sensitivity analyses could not be performed.
Agreements and disagreements with other studies or reviews
With the addition of two new studies (406 women) to one of the comparisons (comparison 1 ‐ continuous glucose monitoring (CGM) versus intermittent glucose monitoring), the evidence suggests that CGM may reduce hypertensive disorders of pregnancy, though as already stated, only two of the four relevant studies reported data for this composite outcome. Conversely, this did not translate into a clear reduction for pre‐eclampsia. CGM probably makes little or no difference to caesarean section, but may reduce neonatal hypoglycaemia. The findings from our review are on the whole very similar to another Cochrane Review (Raman 2017), which examined different methods and settings for glucose monitoring in gestational diabetes, and observed no clear differences between the CGM and self‐monitoring groups for any of their maternal or infant outcomes (caesarean section, large‐for‐gestational age, perinatal deaths). Hypertensive disorders of pregnancy were not reported by either study for their CGM comparison. The results for other comparisons were similar to findings from our review for telemedicine and self‐monitoring, in that there was insufficient evidence to show any clear effect for any of the outcomes examined (Raman 2017). Other reviews also found limited evidence for the effectiveness of real‐time CGM use in children, adults and patients with poorly controlled diabetes (Ghandi 2011; Langendam 2012; Pickup 2011). However, these reviews indicated that higher compliance of wearing the CGM device improves glycosylated haemoglobin A1c level (HbA1c) to a larger extent, and this is in line with our finding of a possible improvement in glycaemic control for women using CGM.
There were no available reviews on self‐monitoring of blood glucose (SMBG) among pregnant women with pre‐existing diabetes and so the findings of this review cannot be compared with any other. This review's findings are not altogether consistent with the findings of others that considered methods for blood glucose monitoring techniques amongst other diabetic populations. SMBG has been found to be effective for patients with type 1 diabetes (DCCT 1993) and patients with type 2 diabetes who are using insulin (Karter 2001). One Cochrane Review (Malanda 2012), concluded that SMBG in newly diagnosed patients with type 2 diabetes who are not using insulin is beneficial in lowering HbA1c. However, when the duration of diabetes is over one year, the overall glycaemic effects of SMBG are small at short term and subside after one year.
Women with type 1 and type 2 diabetes are advised to self‐monitor their blood glucose throughout pregnancy (IDF 2010). The control of hyperglycaemia in pregnant women with pre‐existing diabetes can reduce adverse maternal and infant outcomes (Kitzmiller 2008). A Cochrane Review has reported that pregnant women with type 1 or type 2 diabetes with tight to moderate glycaemic control had significantly lower risks for pre‐eclampsia, caesarean section and macrosomia (Middleton 2016). However, the evidence base for the relative effectiveness of monitoring techniques is inconclusive.
Other than the above mentioned studies or reviews, we are not aware of any other published reviews on techniques of glucose monitoring among pregnant women with pre‐existing diabetes.
Authors' conclusions
Implications for practice.
Two new studies (406 women) have been incorporated to one of the comparisons for this update. Although the evidence suggests that continuous glucose monitoring (CGM) in comparison to intermittent glucose monitoring may reduce hypertensive disorders of pregnancy, this did not translate into a clear reduction for pre‐eclampsia, and so this result should be viewed with caution. There was no evidence of a difference for other primary outcomes for this comparison. The evidence base for the effectiveness of other monitoring techniques analysed in the other five comparisons is weak and based on mainly single studies with very low‐quality evidence. The body of evidence from randomised trials assessing the effects of different techniques of monitoring blood glucose for women with pre‐existing diabetes is therefore incomplete. More trials are needed to confirm the potential benefits of CGM and the effects of other techniques of monitoring in order to inform practice.
Implications for research.
More research is needed to identify the most effective techniques of blood glucose monitoring in pregnant women and to confirm the effectiveness of CGM. The current evidence is limited by the small number of randomised controlled trials (RCTs) for most of the comparisons assessed, small sample sizes, and the variable methodological quality of the RCTs. More evidence is needed to assess the effects of different techniques of monitoring blood glucose for women with pre‐existing diabetes on outcomes for mothers and their children, including use and costs of health care, long‐term outcomes and patient‐reported outcomes. Future RCTs may consider collecting and reporting on the standard outcomes suggested in this review.
What's new
| Date | Event | Description |
|---|---|---|
| 3 June 2019 | Amended | We have edited the numbers of participants to clarify the number of women with type 1 or type 2 diabetes. We have also clarified that in two trials (171 women) the data were not presented separately for type 1 or type 2 diabetes. |
History
Protocol first published: Issue 2, 2012 Review first published: Issue 4, 2014
| Date | Event | Description |
|---|---|---|
| 1 November 2018 | New citation required but conclusions have not changed | The overall conclusions have not changed. There are data from two new studies, which adds some data to one of the six comparisons: 'Continuous glucose monitoring (CGM) versus intermittent glucose monitoring'. There is now evidence to suggest that CGM may reduce hypertensive disorders of pregnancy, although the results were less clear for the single outcome of pre‐eclampsia. CGM may reduce neonatal hypoglycaemia. |
| 1 November 2018 | New search has been performed | Search updated and six studies were assessed for eligibility. Two studies have been included (Feig 2017; Voormolen 2018), two are ongoing (Link 2018; Logan 2011), one is an additional study report of an already included study (di Biase 1997), and one is an additional report of an already excluded study (Bartholomew 2011). |
| 30 November 2016 | New citation required but conclusions have not changed | One new trial added and the conclusions remain unchanged. |
| 30 November 2016 | New search has been performed | Search updated, seven trial reports identified. One new trial added for this update from ongoing studies (Dalfrà 2009). The review now includes 10 trials. 'Risk of bias' assessments for blinding have been updated to include assessments of both performance and detection bias. 'Summary of findings' tables have been incorporated. |
Acknowledgements
We would like to thank Dr Prathap Tharyan's motivation and assistance in the completion of the previous version of this review.
We would like to thank Helen West for her contribution as an author to the previous version of this review (Moy 2017) and to Nancy Medley for helping with data checking and GRADE assessments (2019).
As part of the pre‐publication editorial process, this review has been commented on by two peers (an editor and referee who are external to the editorial team), two members of Cochrane Pregnancy and Childbirth's international panel of consumers and the Group's Statistical Adviser. The authors are grateful to the following peer reviewer for her time and comments: Dr Michelle Downie, Endocrinologist, Southland Hospital, Invercargill, New Zealand.
This project was supported by the National Institute for Health Research, via Cochrane Infrastructure funding to Cochrane Pregnancy and Childbirth. The views and opinions expressed therein are those of the authors and do not necessarily reflect those of their employers, the Systematic Reviews Programme, NIHR, NHS or the Department of Health.
Appendices
Appendix 1. Search methods for ClinicalTrials.gov and ICTRP
Each line was run separately
ICTRP
diabetes AND pregnancy AND monitoring
diabetes AND pregnant AND monitoring
diabetic AND pregnancy AND glucose
diabetic AND pregnant AND glucose
ClinicalTrials.gov
Advanced search
pregnancy | Interventional Studies | Diabetes | Glucose Control | Studies with Female Participants
pregnant | Interventional Studies | Diabetes | monitoring | Studies with Female Participants
pregnancy | Interventional Studies | Diabetes | blood glucose | Studies with Female Participants
Data and analyses
Comparison 1. Continuous glucose monitoring versus intermittent glucose monitoring.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Hypertensive disorders of pregnancy | 2 | 384 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.58 [0.39, 0.85] |
| 2 Caesarean section | 3 | 427 | Risk Ratio (M‐H, Random, 95% CI) | 0.94 [0.75, 1.18] |
| 3 Large‐for‐gestational age | 3 | 421 | Risk Ratio (M‐H, Random, 95% CI) | 0.84 [0.57, 1.26] |
| 4 Perinatal mortality (stillbirth and neonatal mortality) | 1 | 71 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.82 [0.05, 12.61] |
| 5 Mortality or morbidity composite (pregnancy loss (miscarriage, stillbirth, and neonatal death); birth injury; neonatal glycaemia; hyperbilirubinaemia; respiratory distress; and high level neonatal care of more than 24 hours) | 1 | 200 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.80 [0.61, 1.06] |
| 6 Pre‐eclampsia | 4 | 609 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.65 [0.39, 1.08] |
| 7 Pregnancy‐induced hypertension | 2 | 384 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.67 [0.38, 1.16] |
| 8 Behaviour changes associated with the intervention (range of score 10‐50 ‐ high score= greater fear of hypoglycaemia) | 1 | 214 | Mean Difference (IV, Fixed, 95% CI) | 1.00 [‐1.06, 3.06] |
| 9 Sense of well‐being and quality of life (Short form 12 (SF‐12), total score at 34 weeks' gestation) | 1 | 214 | Mean Difference (IV, Fixed, 95% CI) | ‐0.70 [‐2.50, 1.10] |
| 10 Sense of well‐being and quality of life (Problem areas in diabetes (PAID), total score at 34 weeks' gestation) | 1 | 214 | Mean Difference (IV, Fixed, 95% CI) | 0.80 [‐3.06, 4.66] |
| 11 Sense of well‐being and quality of life (BGMSRQ, total score at 34 weeks' gestation) | 1 | 214 | Mean Difference (IV, Fixed, 95% CI) | 4.30 [0.73, 7.87] |
| 12 Glycaemic control ‐ Maternal HbA1c | 2 | 258 | Mean Difference (IV, Random, 95% CI) | ‐0.37 [‐0.78, 0.04] |
| 13 Glycaemic control ‐ Achieved maternal HbA1c <= 6.5% (48 mmol/mol) at 34 weeks | 1 | 187 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.27 [1.00, 1.62] |
| 14 Maternal hypoglycaemia (severe) | 1 | 154 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.92 [0.43, 1.95] |
| 15 Miscarriage | 3 | 439 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.24 [0.47, 3.26] |
| 16 Stillbirth | 1 | 211 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.34 [0.01, 8.17] |
| 17 Neonatal mortality | 2 | 256 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.92 [0.13, 6.37] |
| 18 Gestational age at birth | 1 | 68 | Mean Difference (IV, Fixed, 95% CI) | 0.10 [‐0.57, 0.77] |
| 19 Preterm birth < 37 weeks | 3 | 430 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.96 [0.72, 1.29] |
| 20 Preterm birth < 34 weeks | 1 | 211 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.46 [0.17, 1.28] |
| 21 Macrosomia | 3 | 451 | Risk Ratio (M‐H, Random, 95% CI) | 0.84 [0.61, 1.17] |
| 22 Birthweight | 2 | 267 | Mean Difference (IV, Random, 95% CI) | ‐0.13 [‐0.38, 0.12] |
| 23 Small‐for‐gestational age | 2 | 269 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.40 [0.55, 10.51] |
| 24 Head circumference (cm) | 1 | 160 | Mean Difference (IV, Fixed, 95% CI) | ‐0.20 [‐0.79, 0.39] |
| 25 Length (crown‐heel length cm) | 1 | 160 | Mean Difference (IV, Fixed, 95% CI) | ‐0.20 [‐0.79, 0.39] |
| 26 Adiposity (sum of 4 skin folds (tricepts, subscapular, biceps, flank) mm) | 1 | 160 | Mean Difference (IV, Fixed, 95% CI) | ‐0.20 [‐1.98, 1.58] |
| 27 Shoulder dystocia | 1 | 200 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.0 [0.12, 72.77] |
| 28 Respiratory distress syndrome | 1 | 200 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.0 [0.41, 2.41] |
| 29 Neonatal hypoglycaemia | 3 | 428 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.66 [0.48, 0.93] |
| 30 Neonatal hyperbilirubinaemia | 1 | 200 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.81 [0.52, 1.26] |
| 31 Relevant biomarker changes associated with the intervention (cord blood c‐peptide levels > 566 pmol/L) | 1 | 200 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.95 [0.68, 1.33] |
| 32 Relevant biomarker changes associated with the intervention (cord blood c‐peptide levels > 2725 pmol/L) | 1 | 200 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.0 [0.33, 3.00] |
| 33 Major and minor anomalies | 2 | 285 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.71 [0.16, 3.13] |
| 34 Number of hospital admissions (mother) | 1 | 207 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.25 [0.84, 1.85] |
| 35 Neonatal intensive care unit admissions | 2 | 274 | Risk Ratio (M‐H, Random, 95% CI) | 0.76 [0.42, 1.35] |
| 36 Neonatal intensive care unit length of admission > 24 hours | 1 | 200 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.63 [0.42, 0.93] |
| 37 Birth trauma (shoulder dystocia, bone fracture, nerve palsy) | 1 | 200 | Risk Ratio (M‐H, Fixed, 95% CI) | 5.0 [0.24, 102.85] |
| 38 Diabetic ketoacidosis (mother) | 1 | 207 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.01 [0.14, 7.03] |
1.5. Analysis.
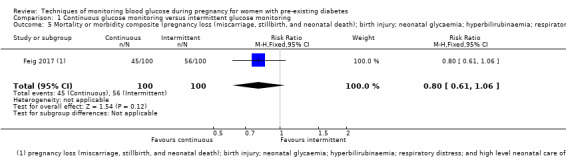
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 5 Mortality or morbidity composite (pregnancy loss (miscarriage, stillbirth, and neonatal death); birth injury; neonatal glycaemia; hyperbilirubinaemia; respiratory distress; and high level neonatal care of more than 24 hours).
1.6. Analysis.
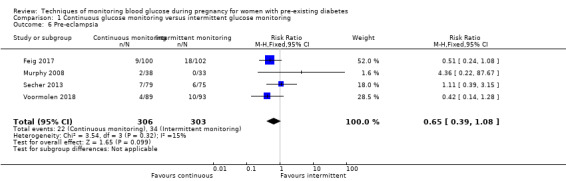
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 6 Pre‐eclampsia.
1.7. Analysis.
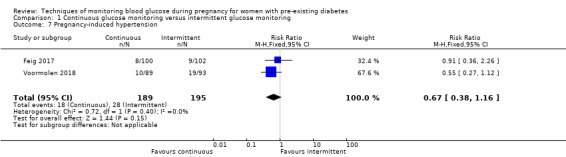
Comparison 1 Continuous glucose monitoring versus intermittent glucose monitoring, Outcome 7 Pregnancy‐induced hypertension.
Comparison 2. Self‐monitoring versus different type of self monitoring.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Caesarean section | 1 | 28 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.78 [0.40, 1.49] |
| 2 Perinatal mortality (stillbirth and neonatal mortality) | 1 | 28 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.0 [0.13, 67.91] |
| 3 Glycaemic control during/end of treatment (maternal post‐prandial blood glucose) | 1 | 13 | Mean Difference (IV, Fixed, 95% CI) | ‐0.70 [‐2.15, 0.75] |
| 4 Glycaemic control during/end of treatment (maternal HbA1c) | 1 | 28 | Mean Difference (IV, Fixed, 95% CI) | ‐0.10 [‐1.93, 1.73] |
| 5 Miscarriage | 1 | 30 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.0 [0.07, 14.55] |
| 6 Neonatal mortality | 1 | 28 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.0 [0.13, 67.91] |
| 7 Gestational age at birth | 1 | 28 | Mean Difference (IV, Fixed, 95% CI) | 0.40 [‐1.65, 2.45] |
| 8 Birthweight | 2 | 41 | Mean Difference (IV, Fixed, 95% CI) | ‐0.18 [‐0.49, 0.13] |
| 9 Respiratory distress syndrome | 1 | 28 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.0 [0.13, 67.91] |
| 10 Neonatal hypoglycaemia | 1 | 28 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.57 [0.21, 1.52] |
| 11 Neonatal jaundice (hyperbilirubinaemia) | 1 | 28 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.56 [0.25, 1.24] |
| 12 Neonatal hypocalcaemia | 1 | 28 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.0 [0.07, 14.45] |
| 13 Neonatal polycythaemia | 1 | 28 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.33 [0.01, 7.55] |
| 14 Neonatal cord vein C‐peptide | 1 | 28 | Mean Difference (IV, Fixed, 95% CI) | 0.13 [‐0.50, 0.76] |
Comparison 3. Self‐monitoring at home versus hospitalisation.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Hypertensive disorders of pregnancy | 1 | 100 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.19 [0.41, 3.51] |
| 2 Caesarean section | 1 | 100 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.96 [0.65, 1.44] |
| 3 Perinatal mortality (stillbirth and neonatal mortality) | 1 | 100 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.85 [0.05, 13.24] |
| 4 Pre‐eclampsia | 1 | 100 | Risk Ratio (M‐H, Fixed, 95% CI) | 4.26 [0.52, 35.16] |
| 5 Pregnancy‐induced hypertension | 1 | 100 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.43 [0.08, 2.22] |
| 6 Placental abruption | 1 | 100 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.70 [0.16, 18.19] |
| 7 Preterm birth < 37 weeks | 1 | 100 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.85 [0.45, 1.60] |
| 8 Respiratory distress syndrome | 1 | 100 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.56 [0.28, 23.74] |
| 9 Neonatal hypoglycaemia | 1 | 100 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.01 [0.50, 2.03] |
| 10 Neonatal jaundice (hyperbilirubinaemia) | 1 | 100 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.27 [0.64, 8.07] |
| 11 Major anomalies | 1 | 102 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.27 [0.03, 2.54] |
| 12 Antenatal hospital admission | 1 | 100 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.19 [0.11, 0.33] |
| 13 Feeding difficulties (not pre‐specified) | 1 | 100 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.85 [0.41, 1.78] |
3.1. Analysis.
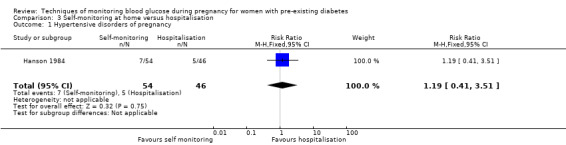
Comparison 3 Self‐monitoring at home versus hospitalisation, Outcome 1 Hypertensive disorders of pregnancy.
3.4. Analysis.
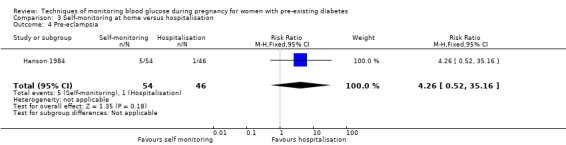
Comparison 3 Self‐monitoring at home versus hospitalisation, Outcome 4 Pre‐eclampsia.
3.5. Analysis.
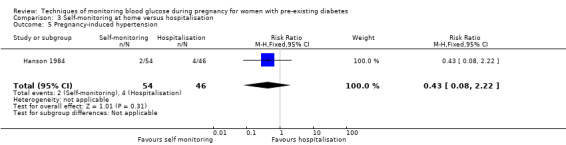
Comparison 3 Self‐monitoring at home versus hospitalisation, Outcome 5 Pregnancy‐induced hypertension.
Comparison 4. Pre‐prandial versus post‐prandial glucose monitoring.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Caesarean section | 1 | 61 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.45 [0.92, 2.28] |
| 2 Large‐for‐gestational age | 1 | 61 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.16 [0.73, 1.85] |
| 3 Perinatal mortality (stillbirth and neonatal mortality) | 1 | 61 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.91 [0.12, 68.66] |
| 4 Pre‐eclampsia | 1 | 58 | Risk Ratio (M‐H, Fixed, 95% CI) | 6.43 [0.82, 50.11] |
| 5 Weight gain during pregnancy | 1 | 61 | Mean Difference (IV, Fixed, 95% CI) | ‐0.90 [‐3.86, 2.06] |
| 6 Insulin dose | 1 | 61 | Mean Difference (IV, Fixed, 95% CI) | ‐17.40 [‐43.41, 8.61] |
| 7 Glycaemic control ‐ Insulin dose | 1 | 61 | Mean Difference (IV, Fixed, 95% CI) | ‐0.20 [‐0.45, 0.05] |
| 8 Glycaemic control ‐ HbA1c | 1 | 61 | Mean Difference (IV, Fixed, 95% CI) | 0.30 [‐0.08, 0.68] |
| 9 Stillbirth | 1 | 61 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.91 [0.12, 68.66] |
| 10 Gestational age at birth | 1 | 61 | Mean Difference (IV, Fixed, 95% CI) | 0.20 [‐0.84, 1.24] |
| 11 Preterm birth < 37 weeks | 1 | 61 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.33 [0.62, 2.84] |
| 12 Macrosomia | 1 | 61 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.18 [0.75, 6.32] |
| 13 Birthweight | 1 | 61 | Mean Difference (IV, Fixed, 95% CI) | 0.24 [‐0.10, 0.58] |
| 14 Adiposity ‐ Subscapula skinfold thickness | 1 | 61 | Mean Difference (IV, Fixed, 95% CI) | 0.60 [‐0.18, 1.38] |
| 15 Adiposity ‐ Triceps skinfold thickness | 1 | 61 | Mean Difference (IV, Fixed, 95% CI) | 0.60 [0.04, 1.16] |
| 16 Birth trauma (shoulder dystocia, bone fracture, nerve palsy) (not pre‐specified as a composite) | 1 | 61 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.48 [0.05, 5.06] |
| 17 Respiratory distress syndrome | 1 | 61 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.97 [0.06, 14.78] |
| 18 Neonatal hypoglycaemia | 1 | 61 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.09 [0.48, 2.45] |
| 19 Neonatal jaundice (hyperbilirubinaemia) | 1 | 61 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.16 [0.40, 3.40] |
| 20 Cord IGF‐1 | 1 | 61 | Mean Difference (IV, Fixed, 95% CI) | 1.30 [‐0.70, 3.30] |
| 21 Neonatal glucose at age 1 hour (not pre‐specified) | 1 | 61 | Mean Difference (IV, Fixed, 95% CI) | ‐0.20 [‐0.88, 0.48] |
| 22 Transient tachypnea (not pre‐specified) | 1 | 61 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.58 [0.76, 8.81] |
| 23 Neonatal intensive care admissions | 1 | 59 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.04 [0.62, 1.74] |
Comparison 5. Automated telemedicine monitoring versus conventional.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Caesarean section | 1 | 32 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.96 [0.62, 1.48] |
| 2 Neonatal morbidity composite | 1 | 32 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.18 [0.53, 2.62] |
| 3 Gestational age at birth | 3 | 84 | Mean Difference (IV, Fixed, 95% CI) | 0.24 [‐0.39, 0.88] |
| 4 Use of additional insulin therapy | 1 | 32 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.0 [0.89, 1.12] |
| 5 Insulin requirement at end of study | 1 | 20 | Mean Difference (IV, Fixed, 95% CI) | 18.4 [12.88, 23.92] |
| 6 Glycaemic control ‐ Maternal fasting blood glucose: before breakfast | 1 | 20 | Mean Difference (IV, Fixed, 95% CI) | ‐1.0 [‐1.22, ‐0.78] |
| 7 Glycaemic control ‐ Maternal fasting blood glucose: before lunch | 1 | 20 | Mean Difference (IV, Fixed, 95% CI) | ‐1.10 [‐1.32, ‐0.88] |
| 8 Glycaemic control ‐ Maternal HbA1c | 3 | 82 | Mean Difference (IV, Random, 95% CI) | ‐0.17 [‐0.82, 0.48] |
| 9 Glycaemic control ‐ Maternal post‐prandial blood glucose | 2 | 50 | Mean Difference (IV, Random, 95% CI) | ‐0.80 [‐1.67, 0.08] |
| 10 Weight gain during pregnancy [kg] | 1 | 32 | Mean Difference (IV, Fixed, 95% CI) | ‐0.70 [‐4.95, 3.55] |
| 11 Macrosomia | 1 | 32 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.18 [0.31, 4.43] |
| 12 Birthweight | 1 | 32 | Mean Difference (IV, Fixed, 95% CI) | ‐0.16 [‐0.64, 0.32] |
Comparison 6. Constant CGM versus intermittent CGM.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Caesarean section | 1 | 25 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.77 [0.33, 1.79] |
| 2 Weight gain during pregnancy | 1 | 25 | Mean Difference (IV, Fixed, 95% CI) | 0.5 [‐1.82, 2.82] |
| 3 Insulin dosage, 3rd trimester (IU/kg/day) | 1 | 25 | Mean Difference (IV, Fixed, 95% CI) | ‐0.03 [‐1.30, 1.24] |
| 4 Glycaemic control ‐ Maternal blood glucose (1st trimester) | 1 | 25 | Mean Difference (IV, Fixed, 95% CI) | ‐0.5 [‐2.70, 1.70] |
| 5 Glycaemic control ‐ Maternal blood glucose (3rd trimester) | 1 | 25 | Mean Difference (IV, Fixed, 95% CI) | ‐0.14 [‐2.00, 1.72] |
| 6 Glycaemic control ‐ Maternal HbA1c (1st trimester) | 1 | 25 | Mean Difference (IV, Fixed, 95% CI) | ‐0.30 [‐1.13, 0.53] |
| 7 Glycaemic control ‐ Maternal HbA1c (3rd trimester) | 1 | 25 | Mean Difference (IV, Fixed, 95% CI) | ‐0.09 [‐0.69, 0.51] |
| 8 Maternal hypoglycemia | 1 | 25 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.54 [0.06, 5.24] |
| 9 Diabetic ketoacidosis (not pre‐specified) | 1 | 25 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.36 [0.02, 8.05] |
| 10 Preterm birth < 37 weeks | 1 | 25 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.08 [0.08, 15.46] |
| 11 Macrosomia | 1 | 25 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.08 [0.08, 15.46] |
| 12 Neonatal hypoglycaemia | 1 | 25 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Dalfrà 2009.
| Methods | Quasi‐randomised trial. Women were sequentially assigned to telemedicine and control groups (not randomised). Period of study: not reported. |
|
| Participants | 88 women with gestational diabetes in the telemedicine group and 115 in the control group;
17 women with type 1 diabetes in the telemedicine group and 15 in the control group. Inclusion criteria: pregnant women with type 1 diabetes (enrolled in the study at their first visit after conception. Women with gestational diabetes included after a week from the diagnosis of gestational diabetes. Exclusion criteria: not described. |
|
| Interventions |
Intervention: automated telemedicine monitoring. Control: conventional system. |
|
| Outcomes |
Outcomes used in this review
|
|
| Notes |
Setting: 12 diabetes clinics. Country: Italy. Funding: not mentioned. Declarations of interest: not reported. Comments: data for women with gestational diabetes and type 1 diabetes are presented separately. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | High risk | Quote: “Women were sequentially assigned to two groups: one patient was followed up using the telemedicine approach and the next using the conventional approach (usual care).” |
| Allocation concealment (selection bias) | High risk | No attempt was made to conceal allocation. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | No attempt was made to blind women or personnel. Women were aware of whether they were being monitored using telemedicine or usual care. However, the outcomes were measured objectively and would not have been influenced by blinding or not blinding. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | No blinding of outcome assessment. However, all outcomes were objectively measured. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | 4/36 women with type 1 diabetes and 37/240 women with gestational diabetes were excluded because they did not complete questionnaires at the end of the study. It is unclear whether these were women with type 1 diabetes or gestational diabetes. |
| Selective reporting (reporting bias) | Unclear risk | This study was assessed from a published report, without the study protocol. The main outcomes were reported separately for type 1 diabetes and GDM, however some outcomes were not reported separately or were only reported in the text. |
| Other bias | High risk | The study did not use an intention‐to‐treat analysis. There is no sample size calculation, or information on whether groups were comparable at baseline. Women with type 1 diabetes only make up a small part of the whole study (32 out of 235 women). |
di Biase 1997.
| Methods | Randomised, parallel‐group, open‐label, 2‐armed, active controlled trial. Period of study: not mentioned. |
|
| Participants |
Number randomised: 20. Eligible were type 1 diabetes mellitus (IDDM) pregnant patients attending the Diabetes Unit specialising in the treatment of diabetes in pregnancy during the period of study. Inclusion criteria: type 1 DM pregnant patients. Exclusion criteria: not mentioned in the text. |
|
| Interventions |
Intervention: DIANET system ‐ continuous automated monitoring system using a telemedicine system ‐ patient unit, diabetes workstation and the communication link (n = 10). Control: conventional monitoring ‐ performed 3 or more tests of blood glucose per day using BM20‐800 strips (n = 10). |
|
| Outcomes |
Outcomes used in this review
|
|
| Notes |
Setting: Diabetes Unit specialising in the treatment of diabetes in pregnancy. Country: Italy. Funding: not mentioned. Declarations of interest: not reported. Comments
|
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Quote from report ‐ "Patients were consecutively chosen by 1 of the investigators. Stratified block randomisation was used to divide patients into 2 groups at baseline." The patients were randomly assigned to a control or DIANET group. Comment: methods of sequence allocation not stated. |
| Allocation concealment (selection bias) | Unclear risk | Comment: not mentioned. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Comment: no blinding of participants and personnel. However, this may not affect the results as all outcomes were objectively measured. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Comment: no blinding of outcome assessment. However, all outcomes were objectively measured. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: reported results of all participants (n = 20). |
| Selective reporting (reporting bias) | Low risk | As reported in the article all outcomes listed have been mentioned. |
| Other bias | Low risk | No obvious risk to other bias. |
Feig 2017.
| Methods | Multi‐centre, parallel randomised controlled trial – open‐label. They ran 2 trials in parallel for pregnant participants and for participants planning a pregnancy. Period of study: 25 March 2013 ‐ 22 March 2016. We report the results for pregnant participants. |
|
| Participants |
Number randomised: 325 (215 pregnant, 110 planning pregnancy). Inclusion criteria: women aged 18‐40 years, with type 1 diabetes for a minimum of 12 months, receiving intensive insulin therapy via multiple daily injections or an insulin pump, who were pregnant or planning pregnancy. Exclusion criteria: regular CGM users and women with severe nephropathy or medical conditions such as psychiatric illness requiring hospitalisation. Women using automatic insulin delivery options, such as low glucose suspend pumps, were not excluded. |
|
| Interventions |
Intervention: real‐time CGM in addition to capillary glucose monitoring. CGM system was aGuardian REAL‐Time or MiniMed Minilink system, both Medtronic, Northridge CA. Participants were trained to use the study devices and instructed to use them daily by local diabetes or antenatal clinic teams. CGM users were advised to verify the accuracy of CGM measurements using their capillary glucose meter before insulin dose adjustment, as per the regulatory labelling instructions (n = 108). Control: standard ‐ capillary glucose monitoring alone (home glucose monitoring). Participants in the control group continued their usual method of capillary glucose monitoring. Participants in both groups were advised to test capillary blood glucose levels at least 7 times daily (before and 1‐2 hours after meals and before bed) and given written instructions for how to use capillary or CGM measures for insulin dose adjustment, customised for methods of insulin delivery. Both groups had the same target glucose range of 3.5 to 7.8 mmol/L and same target HbA1c levels of no higher than 6.5% (48 mmol/mol) during pregnancy (n = 107). |
|
| Outcomes |
Outcomes used in this review
|
|
| Notes |
Setting: 31 hospitals in Canada, England, Scotland, Spain, Italy, Ireland and the USA. Country: Canada. Funding: the trial was funded by Juvenile Diabetes Research Foundation (JDRF) grants #17‐2011‐533, and grants under the JDRF Canadian Clinical Trial Network, a public‐private partnership including JDRF and FedDev Ontario and supported by JDRF #80‐2010‐585. Metronic supplied the CGM sensors and CGM systems at reduced cost. HRM conducts independent research supported by the National Institute for Health Research (Career Development Fellowship, CDF‐2013‐06‐035), and is supported by Tommy’s charity. The funders had no role in the trial design, data collection, data analysis, or data interpretation. Declarations of interest: 8 authors report grants from the Juvenile Diabetes Research Foundation during the conduct of the study. Two authors report personal fees from Novo Nordisk, Roche and Medtronic, outside the submitted work. 1 author reports personal fees from Abbott Diabetes Care and Medtronic (MiniMed Academia), outside the submitted work. 1 author sits on the Medtronic European Scientific Advisory Board. All remaining authors declare no completing interests. Comments
|
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Treatments allocated by a web‐based system using a computer‐generated randomisation list with permuted block sizes and stratified by method of insulin delivery (pump or multiple injections) and baseline HbA1c (< 7.5% vs ≥ 7.5% or 58 mmol/mol for the pregnancy trial). |
| Allocation concealment (selection bias) | Low risk | Randomisaton schedule created by a programming manager, encrypted, and maintained in a secure database – the co‐ordinating team and investigators had no access. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Used masked sensors – for control group – so suggests blinding and also HbA1c measures done at a central laboratory and were unavailable to participants and healthcare teams during the trial. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | All HbA1c measures done at a central laboratory. Samples were shipped after delivery and collection of cord blood and were unavailable to participants and healthcare teams during the trial. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Authors report that about 20% data were missing, lost to follow‐up. For primary outcome – change in HbA1c – 82% (89/108) and 79% (84/107) included in analysis from CGM and home monitoring groups. Also stated that "For the primary outcome, the patterns of change were similar between analyses of imputed and available HbA1c data". There is a study flow diagram and missing data appear balanced across groups. |
| Selective reporting (reporting bias) | Low risk | Cross‐checked the protocol with main published report and methods section – also checked Appendices in supplementary file – most of the outcomes appear to have been reported. |
| Other bias | Low risk | Groups balanced at baseline – differences noted in smoking, automated insulin delivery option, hypertension, severe hypoglycaemia in past year or during early pregnancy pre‐randomisation – but report states "any minor imbalances in baseline characteristics between CGM and control group participants were within the expected bounds for random allocation". |
Hanson 1984.
| Methods | Randomised, parallel‐group, open‐label, 2‐armed, active controlled trial. Period of study: 1 October 1979 ‐ 1 October 1982. |
|
| Participants |
Number randomised: 100. Eligible were type 1 diabetes mellitus (IDDM) and type 2 diabetes mellitus (NIDDM) pregnant patients attending the from 5 hospitals in Stockholm during the period of study. Inclusion criteria: patients with a diagnosis of diabetes, either insulin‐dependent or non‐insulin‐dependent prior to pregnancy. Exclusion criteria: not mentioned in text. |
|
| Interventions |
Intervention: patients self‐monitored their blood glucose at home from the 32nd week until the 36th week of gestation. Weekly hospital visit from 32‐36 weeks and then hospitalised during the 37th week until delivery (n = 54). Control: patients were hospitalised from 32nd week until delivery (n = 46). |
|
| Outcomes |
Outcomes used in this review
|
|
| Notes |
Setting: 5 hospitals in Stockholm. Country: Sweden. Funding: Expressens Perinatal forskningsfond, AIImanna Barnbordshusets Minnesfond, Svenska Diabetesstiftelsen, Nordisk Insulinfond, Swedish Medical Research Council (Project No. 3787), and Tielman's Fund for Pediatric Research. Declarations of interest: not reported. Comments
|
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: not mentioned. |
| Allocation concealment (selection bias) | Unclear risk | Comment: not mentioned. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Comment: no blinding of participants and personnel. However, this may not affect the results as all outcomes were objectively measured. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Comment: no blinding of outcome assessment. Objective measurements used. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Comment: 1 excluded for severe drug addiction, 8 spontaneous abortions and 1 mother died. |
| Selective reporting (reporting bias) | Low risk | No obvious risk to selective reporting. |
| Other bias | Low risk | No obvious risk to other bias. |
Manderson 2003.
| Methods | Randomised, parallel‐group, open‐label, 2‐armed, active controlled trial. Period of study: not mentioned. |
|
| Participants |
Number randomised: 61 Eligible were type 1 diabetes mellitus (IDDM) pregnant patients attending or referred to the Regional Joint Metabolic/Antenatal Clinic at the Royal Maternity Hospital, Belfast during the period of study. Inclusion criteria: type 1 DM pregnant women at 16 weeks' gestation. Exclusion criteria: women without results due to reasons such as: stillbirth, abortions, major congenital abnormalities. |
|
| Interventions |
Intervention: pre‐prandial glucose monitoring (n = 31). Control: post‐prandial glucose monitoring (n = 30). |
|
| Outcomes |
Outcomes used in this review
|
|
| Notes |
Setting: Regional Joint Metabolic/Antenatal Clinic at the Royal Maternity Hospital, Belfast. Country: UK. Funding: Department of Health and Social Sevices, Northern lreland, the Northern Ireland Mother and Baby Appeal, the Metabolic Unit Research Fund, Royal Victoria Hospital, Belfast, the Royal Maternity Hospital, Royal Victoria Hospital, Belfast, and the Irish Perinatal Society. Declarations of interest: not reported. Comments
|
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Quote ‐ "Women were randomly assigned at 16 weeks' gestation to 1 of 2 blood glucose monitoring protocols". Comment: method not mentioned. |
| Allocation concealment (selection bias) | Low risk | Quote ‐ "allocations were via a sealed envelope system, which the patient selected from a box at the clinic visit". |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Comment: no blinding of participants and personnel. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Comment: no blinding of outcome assessment. However, all outcomes were objectively measured. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Quote ‐ "74 patients were recruited. 13 were excluded because they did not have results for analysis. This left 61 diabetic women (31 pre‐prandial and 30 post‐prandial monitoring) with results suitable for analysis". |
| Selective reporting (reporting bias) | Low risk | No obvious risk to selective reporting. |
| Other bias | Low risk | No obvious risk to other bias. |
Murphy 2008.
| Methods | Randomised, parallel‐group, open‐label, 2‐armed, active controlled trial. Period of study: September 2003 to 2006. |
|
| Participants |
Number randomised: 71. Eligible were type 1 (IDDM) and type 2 (NIDDM) diabetes mellitus pregnant patients attending 2 secondary care diabetic antenatal clinics in the UK during the period of study. Inclusion criteria
Exclusion criteria
|
|
| Interventions | Intervention: continuous glucose monitor which measured glucose in subcutaneous tissues every 10 seconds and an average value is stored every 5 minutes, providing up to 288 measurements per day (n = 38). The participants were required to wear the CGMS for 7 days at intervals of 4‐6 weeks. They were also advised to measure blood glucose at least 7 times a day. Control: intermittent self‐monitoring of glucose levels (n = 33), at least 7 times a day (standard care). |
|
| Outcomes |
Outcomes used in this review
|
|
| Notes |
Setting: secondary care diabetic antenatal clinics. Country: UK. Funding: this was an investigator initiated study funded by the Ipswich Diabetes Centre Charity Research Fund. HRM also received salary support from Diabetes UK. The study equipment (6 x CGMS Gold monitors and 300 sensors) was donated free of charge by Medtronic UK. The research was sponsored by Ipswich Hospital NHS Trust and was independent of all the study funders. Declarations of interest: 2 trial authors received honorariums for speaking at research symposiums sponsored by Medtronic in 2004 and 2005. Comments
|
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote ‐ "The study statistician used computer generated randomised numbers in blocks of 20". |
| Allocation concealment (selection bias) | Low risk | Quote ‐ "Concealed in sealed envelopes. Research nurses trained in accordance with good clinical practice guidelines provided the women with their group allocation". |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Comment: no blinding of participants and personnel. However, this may not affect the results as all outcomes were objectively measured. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Comment: no blinding of outcome assessment. However, all outcomes were objectively measured. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: intention‐to‐treat analysis was applied. |
| Selective reporting (reporting bias) | Low risk | All expected outcomes appear to have been reported. |
| Other bias | Low risk | No obvious risk to other bias. |
Petrovski 2011.
| Methods | Randomised, parallel‐group, open‐label, 2‐armed, active controlled trial. Period of study: not mentioned. |
|
| Participants |
Number randomised: 25. Eligible were type 1 diabetes mellitus (IDDM) pregnant patients attending the University Clinic of Endocrinology, Diabetes and Metabolic Disorders in Skopje during the period of study. Inclusion criteria
Exclusion criteria
|
|
| Interventions | Intervention: constant CGM ‐ 24 hours/day (n = 12). Control: intermittent CGM ‐ 14 days per month (n = 13), measured blood glucose at least 6 times a day every second week (when not using the CGM). |
|
| Outcomes |
Outcomes used in this review
|
|
| Notes |
Setting: University Clinic of Endocrinology, Diabetes and Metabolic Disorders in Skopje. Country: Macedonia. Funding: Macedonion Ministry of Health and the Health Care Fund of Macedonia. Declarations of interest: the authors declared that they had no competing financial interests. Comments
|
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Quote ‐ "Patients were randomised into 2 groups". Comment: method not mentioned. |
| Allocation concealment (selection bias) | Unclear risk | Not mentioned. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Comment: no blinding of participants and personnel. However, this may not affect the results as all outcomes were objectively measured. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Comment: no blinding of outcome assessment. However, all outcomes were objectively measured. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Intention‐to‐treat analysis. |
| Selective reporting (reporting bias) | Low risk | All expected outcomes reported. |
| Other bias | Low risk | No obvious risk to other bias. |
Secher 2013.
| Methods | Randomised, parallel‐group, open‐label, 2‐armed, active controlled trial. Period of study: 15 February 2009 to 15 February 2011. |
|
| Participants |
Number randomised: 154. Eligible were 123 type 1 (IDDM) and 31 type 2 (NIDDM) pregnant patients referred to the Centre for Pregnant Women with Diabetes, Rigshospitalet, before 14 completed gestational weeks. Inclusion criteria
Exclusion criteria
|
|
| Interventions |
Intervention: real time CGM for 6 days at pregnancy visits during 8, 12, 21, 27 and 33 weeks, in addition to routine pregnancy care. Control: routine pregnancy care with self‐monitored plasma glucose measurements of 7 times daily. |
|
| Outcomes |
Outcomes used in this review
|
|
| Notes |
Setting: Centre for Pregnant women with Diabetes, Rigshospitalet. Country: Denmark. Funding: 1 of the authors received financial support from the European Foundation of the Study of Diabetes and LifeScan, Rigshopitalet's Research Foundation, the Capital Region of Denmark, the Medical Facuty Foundation of Copenhagen Univeristy, Aase and Ejnar Danielsen Foundation, and Master Joiner Sophus Jacobsen and his wife Astrid Jacobsens' Foundation. 1 author holds stocks in Novo Nordisk. 1 author received financial support from Novo Nordisk Foundation. The real‐time CGM monitors and links were supplied, and glucose sensors were offered at a reduced price by Medtronic. Declarations of interest: the authors declared no other potential conflicts of interest, other than those reported under 'funding'. interests. Comments
|
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote ‐ "a computer generated randomization program was used". |
| Allocation concealment (selection bias) | Low risk | Quote ‐ "..treatment allocation was properly concealed using automated telephone allocation service (Paravox) provided by an independent organization". |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Comment: no blinding of participants and personnel. However, this may not affect the results as all outcomes were objectively measured. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Comment: no blinding of outcome assessment. However, all outcomes were objectively measured. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Quote ‐ "Intention‐to‐treat analysis was carried out". |
| Selective reporting (reporting bias) | Low risk | All expected outcomes reported. |
| Other bias | Low risk | No obvious risk to other bias. |
Stubbs 1980.
| Methods | Randomised, parallel‐group, open‐label, 2‐armed, active controlled trial. Period of study: not mentioned. |
|
| Participants |
Number randomised: 13. Eligible were type 1 (IDDM) diabetes mellitus pregnant patients attending King College's Hospital. Inclusion criteria: type 1 DM pregnant women at 30‐31 weeks' gestation. Exclusion criteria: not mentioned. |
|
| Interventions |
Intervention: 1) glucometer group (n = 7) measured blood glucose at home ‐ 7 times a day, twice weekly (before and after each main meal and before bedtime). Control: non‐meter group (n = 6) ‐ checked urine glucose 4 times daily, random blood glucose measured at the fortnightly clinic visits. |
|
| Outcomes |
Outcomes used in this review
|
|
| Notes |
Setting: King's College hospital. Country: UK. Funding: Medical Research Council Project Grant and the British Diabetic Association. Declarations of interest: not reported. Comments
|
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Comment: not mentioned. |
| Allocation concealment (selection bias) | Unclear risk | Comment: not mentioned. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Comment: no blinding of participants and personnel. However, this may not affect the results as all outcomes were objectively measured. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Comment: no blinding of outcome assessment. However, all outcomes were objectively measured. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Comment: iIntention‐to‐treat. |
| Selective reporting (reporting bias) | Low risk | All expected outcomes reported. |
| Other bias | Low risk | No obvious risk to other bias. |
Varner 1983.
| Methods | Randomised, parallel‐group, open‐label, 2‐armed, active controlled trial. Period of study: 1 February 1980 to 16 September 1981. |
|
| Participants |
Number randomised: 30. Eligible were type 1 diabetes mellitus (IDDM) pregnant patients attending the High Risk Obstetric Clinic at the University of Iowa Hospitals and Clinics during the period of study. Inclusion criteria: less than 20 weeks' gestation. Exclusion criteria: not mentioned. |
|
| Interventions | Intervention: daily home glucose monitoring (n = 15) ‐ fasting, 2‐hour post‐prandial morning, afternoon and evening glucose values were measured daily. Control: weekly venipuncture (n = 15) ‐ fasting, 2 hours after breakfast, and 2 hours after lunch glucose levels measured on 1 day each week. |
|
| Outcomes |
Outcomes used in this review
|
|
| Notes |
Setting: High Risk Obstetric Clinic at the University of Iowa Hospitals and Clinics, Iowa. Country: USA. Funding: Research Fellowship from the Iowa Affiliate of the American Diabetes Association. Declarations of interest: not reported. Comments
|
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote ‐ "Patients were assigned to control and experimental groups using a random number sequence". |
| Allocation concealment (selection bias) | Unclear risk | Comment: not mentioned. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Comment: no blinding of participants and personnel. However, this may not affect the results as all outcomes were objectively measured. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Comment: no blinding of outcome assessment. However, all outcomes were objectively measured. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | 2 patients from each group had a first trimester spontaneous miscarriage and were excluded (2 out of 30 = 7%). |
| Selective reporting (reporting bias) | Low risk | All expected outcomes reported. |
| Other bias | Low risk | No obvious risk to other bias. |
Voormolen 2018.
| Methods | Nationwide multicentre, open‐label, parallel, pragmatic randomised controlled trial Period of study: July 2011 to September 2015 |
|
| Participants |
Number randomised: 300 pregnant women type 1 (n = 109), type 2 (n = 82), or with gestational diabetes (n = 109). Inclusion criteria: pregnant women with pre‐existing DM, at gestational age of before 16 weeks, or had GDM requiring insulin therapy before 30 weeks gestational age. Exclusion criteria: women with multiple pregnancies, under 18 years of age, or who had severe medical or psychological comorbidity |
|
| Interventions |
Intervention: CGM:iPro2 (Medtronic, Northridge, California) ‐ CGM in addition to standard care – self‐monitoring. Women allocated to CGM were instructed to use the device for 5‐7 days every 6 weeks and glucose profiles were obtained retrospectively, directly after each use and evaluated by the local endocrinologist. SMBG is required for calibration of CGM. Readings from the CGM are uploaded to a web‐based program; (n = 147 all women, 50 with T1DM, 40 T2DM). Control: standard treatment ‐ self‐monitoring of blood glucose only (n = 153 all women, 97 with type 2 diabetes). All participants in both intervention and control groups performed SMBG (4‐8 times/day: at least fasting, after every meal, at bedtime and, preferably before every meal). |
|
| Outcomes |
Outcomes used in this review
* outcome not reported separately for pre‐gestational and gestational diabetes |
|
| Notes |
Setting: 22 hospitals (university, teaching and non‐teaching in the Netherlands and 1 university hospital in Belgium. Country: the Netherlands. Funding: the trial was funded by ZonMw, The Dutch Organization for Health Research and Development 80‐82310‐97‐11157. The funder had no role in the study design, data collection, data analysis, data interpretation or writing of the report. Continuous glucose monitors were purchased at a discounted price from Medtronic® and they had no role in the study design, data collection, data analysis, data interpretation, or writing of the report. Declarations of interest: 1 of the trial authors received a research grant from ZonMW (the Netherlands Organization for Health Research and Development) and a second author received research grants from Abbott, Dexcom, Medtronic and Sensonics, and also received personal fees from Roche Diabetes Care and Sensonics. A third author is supported by an NHMRC Practitioner Fellowshop (GNT1082548) and reports consultancy for ObsEVa, Merck and Guerbet. All other authors declare no support from any organization or conflict of interest. Comments
|
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Web‐based computerised programme using 1:1 randomisation, stratified according to type of diabetes |
| Allocation concealment (selection bias) | Unclear risk | Not clearly described |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | No blinding – but outcome measures mainly objective, so unlikely to be affected by lack of blinding |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | No blinding – but outcome measures mainly objective, so unlikely to be affected by lack of blinding |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | High number of patients refused continued use of the CGM after the first or second time – a total of 66% of participants used CGM according to study protocol. However, there was a clear flow of participants in trial profile figure and reasons for dropouts, withdrawal provided. Conducted analyses on intention‐to‐treat and per‐protocol basis; 4 drop‐outs from CGM group and 6 from standard group so primary analyses were carried out according to the intention‐to‐treat principle – 143/147 included from the (CGM group) and 147/153 (standard group in the intention‐to‐treat analysis). Quite a high drop out rate – 95 women in the CGM group and 144 from the control group were included in the per‐protocol analyses. 48 discontinued intervention and 3 discontinued protocol for standard group leaving: 95/147 (66%) intervention group and 144/153 (98%) standard group |
| Selective reporting (reporting bias) | High risk |
Maternal outcomes not reported
Neonatal outcomes not reported
The above were outcomes in the methods of the full report – but were not presented in the results section. The protocol also reported the following outcomes that were not reported in the results: mode of delivery, perinatal death, glucose variability, costs and resource utilisation |
| Other bias | Unclear risk | Baseline characteristics – groups were similar. Data not presented separately for pre‐gestational diabetes (type 1 and type 2 diabetes) and GDM patients for most of the outcomes. |
Wojcicki 2001.
| Methods | Randomised, parallel‐group, open‐label, 2‐armed, active controlled trial. Period of study: not mentioned. |
|
| Participants |
Number randomised: 32. Eligible were type 1 diabetes mellitus (IDDM) pregnant patients attending the Clinic of Gastroenterology and Metabolic Diseases of the Medical Academy in Warsaw during the period of study. Inclusion criteria
Exclusion criteria
|
|
| Interventions |
Intervention: Telematic Management System (Central Clinical Unit and Patients' Teletransmission Modules) (n = 15) ‐ daily transfer of glycaemic data to diabetologist, at least 6 blood glucose measurements daily. Control: Standard care without Telematic Management System (n = 15), 6 blood glucose measurement daily and routine clinic visit every 3 weeks. |
|
| Outcomes |
Outcomes used in this review
|
|
| Notes |
Setting: Clinic of Gastroenterology and Metabolic Diseases of the Medical Academy in Warsaw. Country: Poland. Funding: supported by grants from the Polish State Committee for Scientific Research, the Bayer Diagnostic Division Warsaw, and the Polish Cellular Telephony Centertel. Declarations of interest: not reported. Comments
|
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Randomisation stated but method of sequence generation not clear. Quote: "Before randomization written consent was taken........". |
| Allocation concealment (selection bias) | High risk | Not possible as the same diabetologist was seeing both groups and knew to which group the participant belonged (control group could access the diabetologist by phone any time). |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Comment: no blinding of participants and personnel. However, this may not affect the results as all outcomes were objectively measured. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Comment: no blinding of participants and personnel. However, all outcomes were objectively measured. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | All participants accounted for and all data reported. |
| Selective reporting (reporting bias) | Low risk | No obvious risk to selective reporting. |
| Other bias | Low risk | No obvious risk to other bias. |
BMI: body mass index CGM: continuous glucose monitoring CGMS: continuous glucose monitoring system DM: diabetes mellitus GDM: gestational diabetes mellitus IDDM: insulin‐dependent diabetes mellitus IGF‐1: insulin‐like growth factor‐1 IQR: interquartile range NIDDM: non insulin‐dependent diabetes mellitus
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Bartholomew 2011 | Cross‐over trial. Included women with GDM AND pre‐existing type 2 diabetes: results are not presented separately. |
| NCT01630759 | Clinical trial registration ‐ for gestational diabetics only ‐ started in January 2012, expected to complete by April 2013. |
| Temple 2006 | Abstract of an observational study of 8 type 1 diabetic pregnant women using CGMS. |
| Walker 1999 | Clinical trial registration ‐ contacted author, no published data or report available. |
CGMS: continuous glucose monitoring system GDM: gestational diabetes mellitus
Characteristics of ongoing studies [ordered by study ID]
Link 2018.
| Trial name or title | Link H, NCT03504592. Utilizing mHealth to improve diabetes in an obstetric population. https://clinicaltrials.gov/ct2/show/NCT03504592 (first received 20 April 2018). |
| Methods | Randomised, parallel single‐centre trial, open‐label ‐ USA |
| Participants | Inclusion criteria: 18 to 60 years, pregnant, English speaking, diagnosed with diabetes during pregnancy or with known pre‐existing diabetes, have a smart phone |
| Interventions | MHealth technology ‐ participants to record blood glucose values with the assistance of a smart phone device compared to traditional care method of clinic |
| Outcomes | Primary: completeness and accuracy of blood glucose record Secondary: patient satisfaction, glucose values at goal, % change in HbA1C, clinic visits, unscheduled healthcare access episodes |
| Starting date | 1 May 2018 |
| Contact information | Heather Link, email: heather_link@urmc.rochester.edu |
| Notes | Estimated study completion ‐ June 2019 |
Logan 2011.
| Trial name or title | Managing diabetes during pregnancy in the wireless age: a RCT of glucose telemonitoring. https://clinicaltrials.gov/ct2/show/NCT01474525 (first received 18 November 2011). |
| Methods | Randomised, parallel single‐centre trial, open‐label ‐ Canada |
| Participants | Inclusion criteria: pregnant, diagnosed with gestational diabetes or type 2 diabetes, comfortable with instructions in English and be able to express themselves using simple phrases in English |
| Interventions | Home blood glucose telemonitoring system (system designed to send the measured blood glucose values directly to a hospital server, values recorded by glucometer are sent to a Blackberry cell phone, which services as the platform for data‐transmission to the central server) compared to usual care |
| Outcomes | Primary: mean blood glucose, based on the highest post‐prandial blood glucose reading each day, by trimester Secondary outcomes: mean fasting and post‐prandial blood glucose by trimester, percentage of values within recommended guidelines, adherence, onset of labour and mode of delivery Fetal outcomes: gestational age at delivery, birthweight, percentage of macrosomia, large‐for‐gestational age, small‐for‐gestational age, Apgar at 1 and 5 minutes Perinatal complications: premature, NICU admission, jaundice, shoulder dystocia, hypoglycaemia Provider usage: number of log‐ins onto the system, average amount of time spent on the system per week |
| Starting date | January 2010 |
| Contact information | Alexander G Logan, email: logan@lunenfeld.ca |
| Notes | Estimated study completion ‐ March 2012 |
HbA1C: haemoglobin A1C (glycated haemoglobin) NICU: neonatal intensive care unit RCT: randomised controlled trial
Differences between protocol and review
For the 2017 update, in order to improve consistency across reviews, we used the Cochrane Pregnancy and Childbirth core outcome set for reviews of diabetes in pregnancy, developed by the Cochrane Pregnancy and Childbirth Australasian satellite.
The outcomes specified in the last version of the review have been expanded to incorporate the core outcome set, but were as follows.
Primary outcomes
Maternal
Glycaemic control (HbA1c, fructosamine, fasting blood glucose, post‐prandial blood glucose)
Infant
Birthweight
Macrosomia greater than 4.5 kg
Secondary outcomes
Maternal
Frequency of hypoglycaemia
Antenatal hospital stay (percentage requiring admission, length of stay)
Induction of labour
Caesarean section rates
Miscarriage
Infant
Gestational age (at birth) or preterm birth less than 37/less than 34 weeks
Frequency of neonatal hypoglycaemia
Shoulder dystocia
Major and minor anomalies
Neonatal intensive care admissions
Death of baby including stillbirth/neonatal death
to the following.
The following outcomes were not pre‐specified in our protocol.
Birth trauma (shoulder dystocia, bone fracture, nerve palsy) (not pre‐specified as a composite)
Neonatal glucose at age one hour
Transient tachypnoea
Diabetic ketoacidosis
Feeding difficulties
We have added 'Summary of findings' tables and an assessment of the quality of the evidence using the GRADE approach.
For this update (2019)
We added in a search of ClinicalTrials.gov and the WHO International Clinical Trials Registry Platform (ICTRP) for unpublished, planned and ongoing trial reports. We have also included two new outcomes (instrumental delivery, and neonatal intensive care unit length of stay greater than 24 hours). We now include information about trial dates, sources of trial funding, and trial authors' declarations of interest.
We have two new secondary outcomes to the review:
Instrumental vaginal birth
Neonatal intensive care unit length of stay greater than 24 hours
In response to referee feedback, the following changes have been made.
GRADE outcomes have now been limited to primary outcomes of the review, so that Glycaemic control during/end of treatment (as defined by trialists) (e.g. HbA1c, fructosamine, fasting blood glucose, post‐prandial blood glucose) and preterm birth (less that 37 weeks' gestation and less than 34 weeks' gestation) have been removed from the 'Summary of findings' tables.
We have added pre‐eclampsia, pregnancy‐induced hypertension and eclampsia as secondary outcomes. They form part of the composite outcome 'hypertensive disorders of pregnancy' but had not been listed separately as individual secondary outcomes.
We have re‐ordered the comparisons so that comparison 5 is now comparison 1.
Contributions of authors
Foong Ming Moy (FMM), the contact person, is the guarantor of the review. Leanne Jones drafted the review update, extracted additional data, assessed study quality, undertook data entry and analysis in Review Manager, and prepared the 'Summary of findings' table for comparison 5. Three review authors (FMM, Amita Ray and Brian S Buckely) commented on drafts of the review update.
Sources of support
Internal sources
University of Malaya, Malaysia.
(LVJ) Cochrane Pregnancy and Childbirth Group, Department of Women's and Children's Health, The University of Liverpool, Liverpool, UK.
External sources
-
High Impact Research Grant (E000010‐20001), Malaysia.
Funding in providing the Cochrane Library data base and support in retrieving full text articles
-
NIHR Cochrane Programme Grant Project: 13/89/05 – Pregnancy and childbirth systematic reviews to support clinical guidelines, UK.
2017 update
-
National Institute of Health Research (NIHR) UK, UK.
NIHR Cochrane Reviews of NICE Priority: Project Ref NIHR 128651 (2019 update)
Declarations of interest
Foong Ming Moy: none known.
Amita Ray: none known.
Brian Buckley: none known.
Leanne Jones: is employed by the University of Liverpool as an Associate Editor/Deputy Managing Editor with Cochrane Pregnancy and Childbirth. Her employment is supported by the National Institute for Health Research, via Cochrane Infrastructure funding to Cochrane Pregnancy and Childbirth. She had no involvement with the editorial processes for this review update. Cochrane Pregnancy and Childbirth received an award from UK NIHR Cochrane Reviews of NICE Priority: Project Ref NIHR 128651, to update this review.
Edited (no change to conclusions)
References
References to studies included in this review
Dalfrà 2009 {published data only}
- Dalfrà MG, Nicolucci A, Lapolla A, TISG. The effect of telemedicine on outcome and quality of life in pregnant women with diabetes. Journal of Telemedicine & Telecare 2009;15(5):238‐42. [DOI] [PubMed] [Google Scholar]
di Biase 1997 {published data only}
- Fallucca F, DiBiase N, Sabbatini A, Borrello E, Sciullo E, Napoli A. Telemedicine in the treatment of diabetic pregnancy. Practical Diabetes International. 1996;13(4):115‐8. [Google Scholar]
- Biase N, Napoli A, Sabbatini A, Borrello E, Buongiorno AM, Fallucca F. Telemedicine in the treatment of diabetic pregnancy. Annali dell Istituto Superiore di Sanita 1997;33(3):347‐51. [PubMed] [Google Scholar]
Feig 2017 {published data only}
- Farrell A, Mergler S, Mason D, Sanchez J, Feig DS, Asztalos E. The use of logs and forms for the tracking of RT‐CGM devices in the CONCEPTT Trial. Clinical Trials 2013;10:S80. [Google Scholar]
- Feig DS, Asztalos E, Corcoy R, Leiva A, Donovan L, Hod M, et al. CONCEPTT: Continuous Glucose Monitoring in Women with Type 1 Diabetes in Pregnancy Trial: A multi‐center, multi‐national, randomized controlled trial ‐ Study protocol. BMC Pregnancy and Childbirth 2016;16(1):167. [PUBMED: 27430714] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feig DS, Donovan LE, Corcoy R, Murphy KE, Amiel SA, Hunt KF, et al. Continuous glucose monitoring in pregnant women with type 1 diabetes (conceptt): a multicentre international randomised controlled trial. Lancet 2017;390(10110):2347‐59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- NCT01788527. Continuous glucose monitoring in women with type 1 diabetes in pregnancy trial (CONCEPTT). clinicaltrials.gov/show/NCT01788527 Date first received: 19 December 2012.
Hanson 1984 {published data only}
- Hanson U, Persson B, Enochsson E, Lennerhagen P, Lindgren F, Lundstrom V, et al. Self‐monitoring of blood glucose by diabetic women during the third trimester of pregnancy. American Journal of Obstetrics and Gynecology 1984;150:817‐21. [DOI] [PubMed] [Google Scholar]
Manderson 2003 {published data only}
- Manderson J, Ennis C, Patterson C, Hadden D, Traub A. Pre‐eclampsia in type 1 diabetic pregnancy: preprandial versus postprandial capillary blood glucose monitoring. Hypertension in Pregnancy 2002;21(Suppl 1):142. [Google Scholar]
- Manderson JG, Patterson CC, Hadden DR, Traub AI, Ennis C, McCance DR. Preprandial versus postprandial blood glucose monitoring in type 1 diabetic pregnancy: a randomized controlled clinical trial. American Journal of Obstetrics and Gynecology 2003;189:507‐12. [DOI] [PubMed] [Google Scholar]
Murphy 2008 {published data only}
- ISRCTN84461581. A randomised controlled trial to evaluate the role of the continuous glucose monitoring system (CGMS) in pregnancies complicated by pre‐existing diabetes. isrctn.com/ISRCTN84461581 Date first received: 30 September 2005.
- Murphy HR, Rayman G, Duffield K, Lewis KS, Kelly S, Johal B, et al. Changes in the glycemic profiles of women with type 1 and type 2 diabetes during pregnancy. Diabetes Care 2007;30(11):2785‐91. [DOI] [PubMed] [Google Scholar]
- Murphy HR, Rayman G, Lewis K, Kelly S, Johal B, Duffield K, et al. Effectiveness of continuous glucose monitoring in pregnant women with diabetes: randomised clinical trial. BMJ 2008;337:a1680. [DOI] [PMC free article] [PubMed] [Google Scholar]
Petrovski 2011 {published data only}
- Petrovski G, Dimitrovski C, Bogoev M, Milenkovic T, Ahmeti I, Bitovska I. Is there a difference in pregnancy and glycemic outcome in patients with type 1 diabetes on insulin pump with constant or intermittent glucose monitoring? A pilot study. Diabetes Technology and Therapeutics 2011;13(11):1109‐13. [DOI] [PubMed] [Google Scholar]
Secher 2013 {published data only}
- Cordua S, Secher AL, Ringholm L, Damm P, Mathiesen ER. Real‐time continuous glucose monitoring during labour and delivery in women with type 1 diabetes ‐ observations from a randomized controlled trial. Diabetic Medicine 2013;30(11):1374‐81. [DOI] [PubMed] [Google Scholar]
- NCT00994357. The effect of real‐time continuous glucose monitoring on severe complications to pregnancy in women with diabetes: a randomised controlled study. clinicaltrials.gov/show/NCT00994357 Date first received: 13 October 2009.
- Secher AL, Mathiesen ER, Andersen HU, Peter D, Lene R. Severe hypoglycemia in pregnant women with type 2 diabetes‐ a relevant clinical problem. Diabetes Research and Clinical Practice 2013;102(2):e17‐8. [DOI] [PubMed] [Google Scholar]
- Secher AL, Ringholm L, Andersen HU, Damm P, Mathiesen ER. The effect of real‐time continuous glucose monitoring in diabetic pregnancy ‐ a randomised controlled trial. Diabetologia 2012;55(Suppl 1):S40. [Google Scholar]
- Secher AL, Ringholm L, Andersen HU, Damm P, Mathiesen ER. The effect of real‐time continuous glucose monitoring in pregnant women with diabetes: a randomized controlled trial. Diabetes Care 2013;36:1877‐83. [DOI] [PMC free article] [PubMed] [Google Scholar]
Stubbs 1980 {published data only}
- Stubbs SM, Alberti KG, Brudenell JM, Pyke DA, Watkins PJ, Stubbs WA. Management of the pregnant diabetic: home or hospital, with or without glucose meters. Lancet 1980;1:1122‐4. [DOI] [PubMed] [Google Scholar]
Varner 1983 {published data only}
- Varner MW. Efficacy of home glucose monitoring in diabetic pregnancy. American Journal of Medicine 1983;75:592‐6. [DOI] [PubMed] [Google Scholar]
Voormolen 2018 {published data only}
- Evers I. Effectiveness of continuous glucose monitoring during diabetic pregnancy (GlucoMOMS trial); a randomised controlled trial. Diabetes Technology and Therapeutics 2016;18:A13‐A14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Voormolen DN, DeVries JH, Franx A, Mol BW, Evers IM. Effectiveness of continuous glucose monitoring during diabetic pregnancy (GlucoMOMS trial); a randomised controlled trial. BMC Pregnancy and Childbirth 2012;12:164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Voormolen DN, DeVries JH, Sanson RM, Heringa MP, Valk HW, Kok M, et al. Continuous glucose monitoring during diabetic pregnancy (GlucoMOMS): a multicentre randomized controlled trial. Diabetes, Obesity & Metabolism 2018;20(8):1894‐902. [DOI] [PubMed] [Google Scholar]
- Voormolen DN, Heringa MP, Naaktgeboren CA, Franx A, DeVries JH, Kok M, et al. Efficacy of continuous glucose monitoring in diabetic pregnancy, the glucomoms trial. American Journal of Obstetrics and Gynecology 2017;216(1):S288, Abstract no: 488. [Google Scholar]
Wojcicki 2001 {published data only}
- Ladyzynski P, Wojcicki JM. Home telecare during intensive insulin treatment‐‐metabolic control does not improve as much as expected. Journal of Telemedicine and Telecare 2007;13(1):44‐7. [DOI] [PubMed] [Google Scholar]
- Wojcicki JM, Ladyzynski P, Krzymien J, Jozwicka E, Blachowicz J, Janczewska E, et al. What we can really expect from telemedicine in intensive diabetes treatment: results from 3‐year study on type 1 pregnant diabetic women. Diabetes Technology & Therapeutics 2001;3(4):581‐9. [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
Bartholomew 2011 {published data only}
- Bartholomew LM, Soules K, Church K, Shaha S, Burlingame J, Graham G, et al. Managing diabetes in pregnancy using cell phone/internet technology. Clinical Diabetes 2015;33(4):169‐74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bartholomew ML, Church K, Graham G, Burlingame J, Zalud I, Sauvage L, et al. Managing diabetes in pregnancy using cell phone/internet technology. American Journal of Obstetrics and Gynecology 2011;204(1 Suppl):S113‐S114. [Google Scholar]
- NCT01907516. Managing diabetes in pregnancy using cell phone/internet technology. clinicaltrials.gov/show/NCT01907516 Date first received: 22 July 2013.
NCT01630759 {published data only}
- NCT01630759. Remote monitoring of diabetes in pregnancy: a feasibility study for a randomised controlled trial. clinicaltrials.gov/show/NCT01630759 Date first received: 22 June 2012.
Temple 2006 {published data only}
- Temple RC, Duffield K, Lewis K, Murphy HR. Glycaemic control during pregnancy in women with long duration type 1 diabetes: lessons learn using continuous glucose monitoring systems. Diabetologia 2006;49(Suppl 1):S78. [Google Scholar]
Walker 1999 {published data only}
- Walker JD. Blood glucose monitoring strategies in diabetic: an audit of achievement of glycaemic goals and outcome of pregnancy. National Research Register (www.nrr.nhs.uk) 1999.
References to ongoing studies
Link 2018 {published data only}
- Link H, NCT03504592. Utilizing mHealth to improve diabetes in an obstetric population. https://clinicaltrials.gov/ct2/show/NCT03504592 (first received 20 April 2018).
Logan 2011 {published data only}
- Logan AG, NCT01474525. Managing diabetes during pregnancy in the wireless age: a RCT of glucose telemonitoring. https://clinicaltrials.gov/ct2/show/NCT01474525 (first received 18 November 2011).
Additional references
ACOG 2005
- ACOG Committee on Practice Bulletins, authors. Pregestational diabetes mellitus: ACOG Clinical Management Guidelines for Obstetrician‐Gynecologists #60. Obstetrics & Gynecology 2005;105:675‐85. [DOI] [PubMed] [Google Scholar]
ADA 2004
- ADA. Preconception care of women with diabetes (Position Statement). Diabetes Care 2004;27:S76‐S78. [DOI] [PubMed] [Google Scholar]
ADA 2011
- American Diabetes Association. Standards of Medical Care in Diabetes—2011. Diabetes Care 2011;34(Suppl 1):S11‐S61. [DOI] [PMC free article] [PubMed] [Google Scholar]
Choleau 2002
- Choleau C, Klein JC, Reach G, Aussedat B, Demaria‐Pesce V, Wilson GS, et al. Calibration of a subcutaneous amperometric glucose sensor. Part 1. Effect of measurement uncertainties on the determination of sensor sensitivity and background current. Biosensors and Bioelectronics 2002;17(8):641–6. [DOI] [PubMed] [Google Scholar]
Davidson 2005
- Davidson J. Strategies for improving glycemic control: effective use of glucose monitoring. American Journal of Medicine 2005;118(9 Suppl 1):27–32. [DOI] [PubMed] [Google Scholar]
DCCT 1993
- The Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long‐term complications in insulin‐dependent diabetes mellitus. New England Journal of Medicine 1993;329(14):977‐86. [DOI] [PubMed] [Google Scholar]
de Veciana 1995
- Veciana M, Major CA, Morgan MA, Asrat T, Toohey JS, Lien JM, et al. Postprandial versus preprandial blood glucose monitoring in women with gestational diabetes mellitus requiring insulin therapy. New England Journal of Medicine 1995;333(19):1239‐41. [DOI] [PubMed] [Google Scholar]
Fetita 2006
- Fetita LS, Sobngwi E, Serradas P, Calvo F, Gautier JF. Consequences of fetal exposure to maternal diabetes in offspring. Journal of Clinical Endocrinology and Metabolism 2006;91:3714‐24. [DOI] [PubMed] [Google Scholar]
Fuhrmann 1983
- Fuhrmann K, Reiher H, Semmler K, Fischer F, Fischer M, Glöckner E. Prevention of congenital malformations in infants of insulin‐dependent diabetic mothers. Diabetes Care 1983;6(3):219‐23. [DOI] [PubMed] [Google Scholar]
Gabbe 2003
- Gabbe SG, Graves CR. Management of diabetes mellitus complicating pregnancy. Obstetrics and Gynecology 2003;102:857‐68. [DOI] [PubMed] [Google Scholar]
Ghandi 2011
- Ghandi GY, Kovalaske M, Kudva Y, Walsh K, Elamin MB, Beers M, et al. Efficacy of continuous glucose monitoring in improving glycaemic control and reducing hypoglycemia: a systematic review and meta analysis of randomized trials. Journal of Diabetes Science and Technology 2011;5(4):952–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
Gonzalez‐Gonzalez 2008
- Gonzalez‐Gonzalez NL, Ramirez O, Mozas J, Melchor J, Armas H, Garcia‐Hernandez JA, et al. Factors influencing pregnancy outcome in women with type 2 versus type 1 diabetes mellitus. Acta Obstetricia et Gynecologica Scandinavica 2008;87:43‐9. [DOI] [PubMed] [Google Scholar]
Greene 1989
- Greene MF, Hare JW, Cloherty JP, Benacerraf BR, Soeldner JS. First‐trimester hemoglobin A1 and risk for major malformation and spontaneous abortion in diabetic pregnancy. Teratology 1989;39(3):225‐31. [DOI] [PubMed] [Google Scholar]
HAPO 2002
- HAPO Study Cooperative Research Group. The Hyperglycemia and Adverse Pregnancy Outcome (HAPO) Study. International Journal of Gynecology and Obstetrics 2002;78:69‐77. [DOI] [PubMed] [Google Scholar]
Higgins 2011
- Higgins JP, Green S, editors. Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011. Available from www.cochrane‐handbook.org.
Howorka 2001
- Howorka K, Pumprla J, Gabriel M, Feiks A, Schlusche C, Nowotny C, et al. Normalization of pregnancy outcome in pregestational diabetes through functional insulin treatment and modular out‐patient education adapted for pregnancy. Diabetic Medicine 2001;18(12):965‐72. [DOI] [PubMed] [Google Scholar]
Huo 2018
- Huo T, Guo Y, Shenkman E, Muller K. Assessing the reliability of the short form 12 (SF‐12) health survey in adults with mental health conditions: a report from the wellness incentive and navigation (WIN) study. Health and Quality of Life Outcomes 2018;16(34):1‐9. [DOI: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
IDF 2010
- IDF. The Diabetes Atlas. Brussels: International Diabetes Federation, 2010. [Google Scholar]
Jensen 2009
- Jensen DM, Korsholm L, Ovesen P, Beck‐Nielsen H, Moelsted‐Pedersen L, Westergaard JG, et al. Periconceptional A1C and risk of serious adverse pregnancy outcome in 933 women with type 1 diabetes. Diabetes Care 2009;32:1046‐8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Jovanovic 2006
- Jovanovic L, Nakai Y. Successful pregnancy in women with type 1 diabetes: from preconception through postpartum care. Endocrinology and Metabolism Clinics of North America 2006;35:79‐97, vi. [DOI] [PubMed] [Google Scholar]
Jovanovič 2009
- Jovanovič L. Medical Management of Pregnancy Complicated by Diabetes. 4th Edition. Alexandria, VA: American Diabetes Association, 2009. [Google Scholar]
Kapoor 2007
- Kapoor N, Sankaran S, Hyer S, Shehata H. Diabetes in pregnancy: a review of current evidence. Current Opinion in Obstetrics and Gynecology 2007;19:586‐90. [DOI] [PubMed] [Google Scholar]
Karter 2001
- Karter AJ, Ackerson LM, Darbinian JA, D'Agostino RB Jr, Ferrara A, Liu J, et al. Self‐monitoring of blood glucose levels and glycemic control: the Northern California Kaiser Permanente Diabetes registry. American Journal of Medicine 2001;111(1):1‐9. [DOI] [PubMed] [Google Scholar]
Kerssen 2006
- Kerssen A, Valk HW, Visser GH. Do HbA1c levels and the self‐monitoring of blood glucose levels adequately reflect glycaemic control during pregnancy in women with type 1 diabetes mellitus?. Diabetologia 2006;49(1):25‐8. [DOI] [PubMed] [Google Scholar]
Kerssen 2007
- Kerssen A, Valk HW, Visser GH. Increased second trimester maternal glucose levels are related to extremely large‐for‐gestational‐age infants in women with type 1 diabetes. Diabetes Care 2007;30:1069‐74. [DOI] [PubMed] [Google Scholar]
Kitzmiller 1996
- Kitzmiller JL, Buchanan TA, Kjos S, Combs CA, Ratner RE. Pre‐conception care of diabetes, congenital malformations and spontaneous abortions (ADATechnical Review). Diabetes Care 1996;19:514–41. [DOI] [PubMed] [Google Scholar]
Kitzmiller 2008
- Kitzmiller JL, Block JM, Brown FM, Catalano PM, Conway DL, Coustan DR, et al. Managing preexisting diabetes for pregnancy: summary of evidence and consensus recommendations for care. Diabetes Care 2008;31:1060‐79. [DOI] [PMC free article] [PubMed] [Google Scholar]
Kitzmiller 2010
- Kitzmiller JL, Wallerstein R, Correa A, Kwan S. Preconception care for women with diabetes and prevention of major congenital malformations. Birth Defects Research. Part A, Clinical and Molecular Teratology 2010;88(10):791‐803. [DOI] [PubMed] [Google Scholar]
Lamb 2017
- Lam AY, Xin X, Tan WB, Gardner DS, Goh SY. Psychometric validation of the Hypoglycemia Fear Survey‐II (HFS‐II) in Singapore. BMJ Open Diabetes Research and Care 2017;5:e000329. [DOI: 10.1136/bmjdrc-2016-000329] [DOI] [PMC free article] [PubMed] [Google Scholar]
Langendam 2012
- Langendam M, Luijf YM, Hooft L, DeVries JH, Mudde AH, Scholten RJ. Continuous glucose monitoring systems for type 1 diabetes mellitus. Cochrane Database of Systematic Reviews 2012, Issue 1. [DOI: 10.1002/14651858.CD008101.pub2; CD008101] [DOI] [PMC free article] [PubMed] [Google Scholar]
Malanda 2012
- Malanda UL, Welschen LM, Riphagen II, Dekker JM, Nijpels G, Bot SD. Self‐monitoring of blood glucose in patients with type 2 diabetes mellitus who are not using insulin. Cochrane Database of Systematic Reviews 2012, Issue 1. [DOI: 10.1002/14651858.CD005060.pub3; CD005060] [DOI] [PMC free article] [PubMed] [Google Scholar]
McElduff 2005
- McElduff A, Cheung NW, McIntyre HD, Lagstrom JA, Oats JJ, Ross GP, et al. The Australasian Diabetes in Pregnancy Society consensus guidelines for the management of type 1 and type 2 diabetes in relation to pregnancy (Position Statement). Medical Journal of Australia 2005;183:373‐7. [PubMed] [Google Scholar]
Mello 2000
- Mello G, Parretti E, Mecacci F, LaTorre P, Cioni R, Cianciulli D, et al. What degree of maternal metabolic control in women with type 1 diabetes is associated with normal body size and proportions in full‐term infants?. Diabetes Care 2000;23:1494‐8. [DOI] [PubMed] [Google Scholar]
Middleton 2016
- Middleton P, Crowther CA, Simmonds L. Different intensities of glycaemic control for pregnant women with pre‐existing diabetes. Cochrane Database of Systematic Reviews 2016, Issue 5. [DOI: 10.1002/14651858.CD008540.pub4] [DOI] [PMC free article] [PubMed] [Google Scholar]
Mills 1988
- Mills JL, Simpson JL, Driscoll SG, Jovanovic‐Peterson L, Allen M, Aarons JH, et al. Incidence of spontaneous abortion among normal women and insulin‐dependent diabetic women whose pregnancies were identified within 21 days of conception. New England Journal of Medicine 1988;319(25):1617‐23. [DOI] [PubMed] [Google Scholar]
Moore 2010
- Moore TR. Fetal exposure to gestational diabetes contributes to subsequent adult metabolic syndrome. American Journal of Obstetrics and Gynecology 2010;202:643‐9. [DOI] [PubMed] [Google Scholar]
Moses 1999
- Moses RG, Lucas EM, Knights S. Gestational diabetes mellitus. At what time should the postprandial glucose level be monitored?. Australian and New Zealand Journal Of Obstetrics and Gynaecology 1999;39(4):457‐60. [DOI] [PubMed] [Google Scholar]
Murphy 2007
- Murphy HR, Rayman G, Duffield K, Lewis KS, Kelly S, Johal B, et al. Changes in the glycemic profiles of women with type 1 and type 2 diabetes during pregnancy. Diabetes Care 2007;30(11):2785‐91. [DOI] [PubMed] [Google Scholar]
NICE 2008
- NICE. Diabetes in Pregnancy. Clinical Guideline 63. RCOG Press, 2008. [Google Scholar]
NICE 2015
- National Institute for Health and Care Excellence. Diabetes in pregnancy: management from preconception to the postnatal period [NG3]. Available at: https://www.nice.org.uk/guidance/ng3 [Accessed 19 March 2019] 2015. [PubMed]
Peyrot 2009
- Peyrot M, Rubin RR. Patient‐reported outcomes for an integrated real‐time continuous glucose monitoring/insulin pump system mark. Diabetes Technology and Therapeutics 2009;11(1):57‐62. [DOI] [PubMed] [Google Scholar]
Pickup 2011
- Pickup JC, Freeman SC, Sutton AJ. Glycaemic control in type 1 diabetes during real time continuous glucose monitoring compared with self monitoring of blood glucose: meta‐analysis of randomised controlled trials using individual patient data. BMJ 2011;343:d3805. [DOI] [PMC free article] [PubMed] [Google Scholar]
Raman 2017
- Raman P, Shepherd E, Dowswell T, Middleton P, Crowther CA. Different methods and settings for glucose monitoring for gestational diabetes during pregnancy. Cochrane Database of Systematic Reviews 2017, Issue 10. [DOI: 10.1002/14651858.CD011069.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Ray 2001
- Ray JG, O’Brien TE, Chan WS. Preconception care and the risk of congenital anomalies in the offspring of women with diabetes mellitus: a meta‐analysis. Quarterly Journal of Medicine 2001;94:435‐44. [DOI] [PubMed] [Google Scholar]
RevMan 2014 [Computer program]
- The Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager (RevMan). Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Roland 2005
- Roland JM, Murphy HR, Ball V, Northcote‐Wright J, Temple RC. The pregnancies of women with type 2 diabetes: poor outcomes but opportunities for improvement. Diabetic Medicine 2005;22:1774‐7. [DOI] [PubMed] [Google Scholar]
Rosenn 1991
- Rosenn B, Miodovnik M, Combs CA, Khoury J, Siddiqi TA. Pre‐conception management of insulin‐dependent diabetes: improvement of pregnancy outcome. Obstetrics & Gynecology 1991;77(6):846‐9. [PubMed] [Google Scholar]
Sibai 2000
- Sibai BM, Caritis SN, Hauth JC, MacPherson C, Dorsten JP, Klebanoff M, et al. Preterm delivery in women with pregestational diabetes mellitus or chronic hypertension relative to women with uncomplicated pregnancies. The National institute of Child health and Human Development Maternal‐ Fetal Medicine Units Network. American Journal of Obstetrics and Gynecology 2000;183(6):1520‐4. [DOI] [PubMed] [Google Scholar]
Slocum 2004
- Slocum J, Barcio L, Darany J, Friedley K, Homko C, Mills JJ, et al. Preconception to postpartum: management of pregnancy complicated by diabetes. Diabetes Educator 2004;30(5):740, 742‐4, 747‐53. [DOI] [PubMed] [Google Scholar]
Suhonen 2000
- Suhonen L, Hiilesmaa V, Teramo K. Glycaemic control during early pregnancy and fetal malformations in women with type I diabetes mellitus. Diabetologia 2000;43:79‐82. [DOI] [PubMed] [Google Scholar]
Thomas 2006
- Thomas AM. Pregnancy with preexisting diabetes. In: Mensing C, Cypress M, Halstensen C, McLaughlin S, Walker EA editor(s). Art and Science of Diabetes Self‐Management Education. A Desk Reference for Healthcare Professionals. Chicago: American Association of Diabetic Educators, 2006:233‐57. [Google Scholar]
Venkataraman 2015
- Venkataraman K, Tan LS, Bautista DC, Griva K, Zuniga YL, Amir M, et al. Psychometric Properties of the Problem Areas in Diabetes (PAID) Instrument in Singapore. PLOS One 2015;10(9):e0136759. [DOI] [PMC free article] [PubMed] [Google Scholar]
WHO 1994
- WHO. Prevention of diabetes mellitus. Report of a WHO Study Group. Geneva, 1994; Vol. 844:55‐9. [PubMed]
References to other published versions of this review
Moy 2012
- Moy FM, Ray A, Buckley BS. Techniques of monitoring blood glucose during pregnancy for women with pre‐existing diabetes. Cochrane Database of Systematic Reviews 2012, Issue 2. [DOI: 10.1002/14651858.CD009613] [DOI] [PubMed] [Google Scholar]
Moy 2014
- Moy FM, Ray A, Buckley BS. Techniques of monitoring blood glucose during pregnancy for women with pre‐existing diabetes. Cochrane Database of Systematic Reviews 2014, Issue 4. [DOI: 10.1002/14651858.CD009613.pub2] [DOI] [PubMed] [Google Scholar]
Moy 2017
- Moy FM, Ray A, Buckley BS, West HM. Techniques of monitoring blood glucose during pregnancy for women with pre‐existing diabetes. Cochrane Database of Systematic Reviews 2017, Issue 6. [DOI: 10.1002/14651858.CD009613.pub3] [DOI] [PMC free article] [PubMed] [Google Scholar]


