Abstract
Dendranthema indicum var. aromaticum plant has been widely used as herbal medicine in China, however, the material basis responsible for the therapeutic benefits remains largely unclear. This study aimed to provide an optimized method for extracting and characterizing phenolic compounds in D. indicum var. aromaticum flower. Firstly, an ultrasound-assisted method combined with central composite circumscribed (CCC) design was applied to optimize phenolic compound extraction. Ethanol-acetic acid (70%:2%, v/v) was selected as solvent, and the optimal extraction condition was: extraction temperature, 57 °C; solid/liquid ratio, 1:30 g/mL; extraction time, 20 min. Secondly, an effective and economic HPLC-PDA-ESI-MSn method was established and validated for phenolic compound characterization and quantification. As a result, 14 phenolic compounds were identified, including 8 phenolic acids and 6 flavonoids, and for the first time, oleuropein derivatives, chrysoeriol, and tricin are reported in D. indicum var. aromaticum flower. The content of phenolics identified by HPLC-MSn was 6.42 ± 0.32 mg/g DW. The optimized method for extraction and characterization of phenolic compounds has significant meaning to future pharmaceutical and medicinal research on D. indicum var. aromaticum, and the results in this study can provide references for herbal research.
Subject terms: Chemical biology, Secondary metabolism, Mass spectrometry
Introduction
Dendranthema indicum var. aromaticum is a new varietas of Dendranthema, growing in sunny area with an altitude of more than 2000 meters in Shen Nongjia area of Hubei province, China. The whole plant of the new varietas emits strong aroma, and the folk usually dry the leaves and petals to use as sachet. What’s more, D. indicum var. aromaticum plant is widely used as Chinese herbal medicine to prevent cold, treat headache, enteritis, constipation, coronary heart disease and hypertension.
Researchers reported that D. indicum var. aromaticum essential oil has strong anti-microbial and anti-oxidant activities1. D. indicum var. aromaticum is a good source of phenolics. Some flavonoids were isolated from this plant, including luteolin, apigenin, and acacetin2. Luteolin was reported to display excellent anti-oxidant, anti-inflammatory, and anti-allergic activities3. In various human cancer cell lines, cell cycle arrest and apoptosis were induced by adding acacetin into cell culture medium4,5. Many in vivo and in vitro investigations have revealed that intake of some phenolic compounds especially certain flavonoids contributes to the prevention of hypertension6,7. All these studies provide theoretical basis for the therapeutic benefits of D. indicum var. aromaticum plant, however, the material basis being responsible remains largely unclear. To date, research on D. indicum var. aromaticum mainly focused on chemical constituent of essential oil, but very little on its phenolics composition. To better explore values of D. indicum var. aromaticum in scientific research and in medicinal resource development, it makes sense to figure out the phenolic profile in D. indicum var. aromaticum.
As far as we know, currently there is no systematic method of extracting and analyzing phenolic compounds in D. indicum var. aromaticum flower. Therefore, the first objective of the present study was to optimize a high-efficiency method to extract phenolic compounds from D. indicum var. aromaticum flower. Ultrasound-assisted extraction, a commonly used technique of bioactive substances extraction from food products8 and plant materials9–11 was adopted in this study, in addition, response surface methodology was used to optimize the combination of various extraction conditions. To analyze phenolic compounds, a good HPLC analytical method must balance resolution, time cost and solvent cost. Therefore, the second objective of this study was to establish an effective and economic HPLC-PDA-ESI-MSn analytical method to characterize phenolics composition in D. indicum var. aromaticum flower.
Results and Discussion
Optimization of phenolic compounds extraction
Extraction methods of phenolic compounds in plant material include soxhlet, microwave-assisted extraction, supercritical fluid extraction, ultrasound-assisted extraction, etc.12–14. With broad literature retrieval, ultrasound-assisted extraction was found to be a simple and effective method of phenoilcs extraction8,10,15, which does not require complicated equipment or technique and could greatly increase the extraction efficiency by strengthening the fragmentation process and assisting the release, diffusion, and dissolution of the components inside cells11,16. Furthermore, ultrasound-assisted extraction is more moderate and more secure to operate for possible future large-scale extraction. Therefore, ultrasound-assisted extraction was chosen in this study.
Aqueous methanol, ethanol, and acetone were usually used for phenolic compounds extraction from botanical materials, especially from herbs10,15. Acid, for instance, hydrochloric acid, formic acid, and acetic acid was often used to acidify the extraction environment for better efficiency15,17. Considering the possible future industrial application of the phenolic extraction, relatively low toxic solvents ethanol and acetic acid were chosen in this study. Figure 1 shows the effect of different proportion of ethanol and acetic acid combinations on total phenolic (TP) content. In general, the extraction effect was increased by adding more acetic acid at lower concentration of ethanol (30% or 50%) but not at higher concentration of ethanol (70%). The highest TP content was obtained from ethanol-acetic acid (70%:2%, v/v) solvent, therefore, ethanol-acetic acid (70%:2%, v/v) was selected to be the extracting solvent in the following studies.
Figure 1.
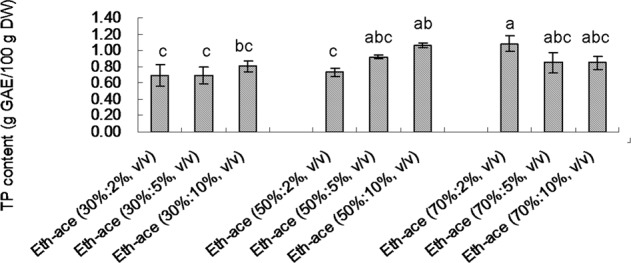
Effect of different proportion of ethanol and acetic acid combinations on TP content. Means of each two treatments were compared using least significant difference (LSD) statistic method. Lowercase letters a, b, and c were used to mark significance of difference (p < 0.05). Same letters between treatments mean insignificant difference. Different letters between treatments mean significant difference.
To set up an appropriate CCC model, the ideal value range of independent variables should cover the inflection point of each independent variable. Therefore, the effect of each single factor on TP content was tested to obtain an approximate range for the CCC model construction, and the results were shown in Fig. 2. The increase of TP content was observed over the extraction time range (20∼30 min) (Fig. 2A), solid/liquid ratio range (1:10∼1:20 g/mL) (Fig. 2B), and extraction temperature range (30∼50 °C) (Fig. 2C). Moderate higher temperature and longer extraction time could enhance the solubility of phenolic compounds and accelerate the whole extraction process, however, degradation should always be considered when extraction time and temperature exceed an appropriate range. The inflection point of each variable was selected as central point of the CCC design: extraction time, 30 min; solid/liquid ratio, 1:20 g/mL; extraction temperature, 50 °C.
Figure 2.
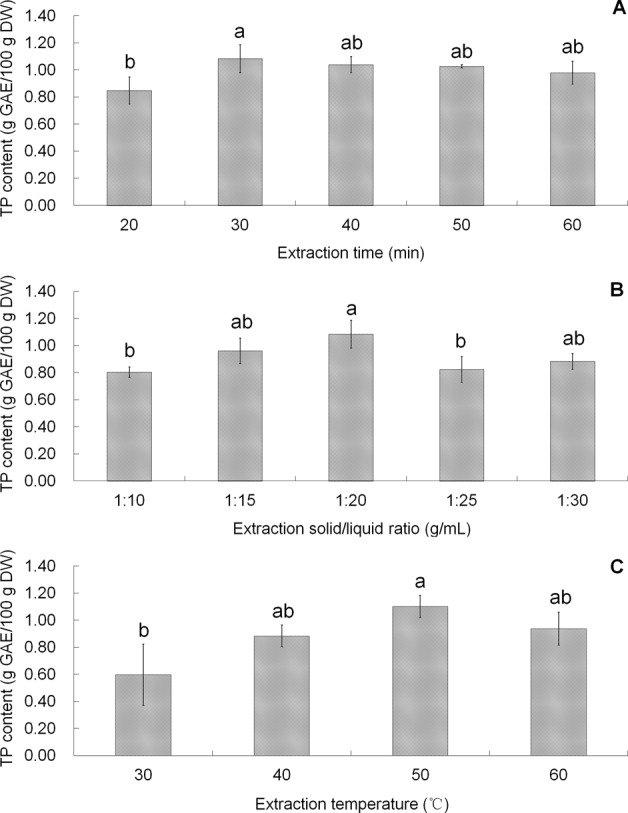
Effect of single factors (A) extraction time, (B) solid/liquid ratio, and (C) temperature on TP content. Means of each two treatments were compared using least significant difference (LSD) statistic method. Lowercase letters a and b were used to mark significance of difference (p < 0.05). Same letters between treatments mean insignificant difference. Different letters between treatments mean significant difference.
Extraction conditions and the corresponding results under CCC design were shown in Table 1. Statistical analysis revealed that quadratic model significantly fitted to the CCC design (p = 0.0002, R2 = 0.9211, statistic results were not shown). As seen in Fig. 3A, at certain extraction time, TP content rose with the increase of solid/liquid ratio. Further, relatively higher TP content was obtained when solid/liquid ratio tending to 1:30 with extraction time tending to 20 min simultaneously. As shown in Fig. 3B, TP content increased following temperature increasing at certain extraction time, and the rising trend was more obvious in 35 to 40 min extraction time range. In Fig. 3C, TP content went up following the rise of solid/liquid ratio under certain temperature. The more the solid/liquid ratio was close to 1:30 g/mL, the higher TP content was obtained in the temperature range of 50∼60 °C.
Table 1.
Central composite circumscribed (CCC) design with five levels and three variables for phenolic compound extraction in D. indicum var. aromaticum flower and observed responsesa.
| Run order | Independent variables | TP content (g GAEb/100 g DW) | |||||
|---|---|---|---|---|---|---|---|
| Coded | Uncoded | ||||||
| Time (min) | Solid/liquid ratio (g/mL) | Temperature (°C) | Time (min) | Solid/liquid ratio (g/mL) | Temperature (°C) | ||
| 1 | −1 | −1 | −1 | 20 | 1:10 | 40 | 0.71 |
| 2 | 1 | −1 | −1 | 40 | 1:10 | 40 | 0.93 |
| 3 | −1 | 1 | −1 | 20 | 1:30 | 40 | 1.16 |
| 4 | 1 | 1 | −1 | 40 | 1:30 | 40 | 1.04 |
| 5 | −1 | −1 | 1 | 20 | 1:10 | 60 | 0.84 |
| 6 | 1 | −1 | 1 | 40 | 1:10 | 60 | 1.03 |
| 7 | −1 | 1 | 1 | 20 | 1:30 | 60 | 1.27 |
| 8 | 1 | 1 | 1 | 40 | 1:30 | 60 | 1.21 |
| 9 | −1.68 | 0 | 0 | 13 | 1:20 | 50 | 1.11 |
| 10 | 1.68 | 0 | 0 | 47 | 1:20 | 50 | 1.28 |
| 11 | 0 | −1.68 | 0 | 30 | 1:03 | 50 | 0.68 |
| 12 | 0 | 1.68 | 0 | 30 | 1:37 | 50 | 1.23 |
| 13 | 0 | 0 | −1.68 | 30 | 1:20 | 33 | 0.89 |
| 14 | 0 | 0 | 1.68 | 30 | 1:20 | 67 | 1.13 |
| 15 | 0 | 0 | 0 | 30 | 1:20 | 50 | 1.02 |
| 16 | 0 | 0 | 0 | 30 | 1:20 | 50 | 1.20 |
| 17 | 0 | 0 | 0 | 30 | 1:20 | 50 | 1.07 |
| 18 | 0 | 0 | 0 | 30 | 1:20 | 50 | 1.26 |
| 19 | 0 | 0 | 0 | 30 | 1:20 | 50 | 1.15 |
| 20 | 0 | 0 | 0 | 30 | 1:20 | 50 | 1.07 |
aExperimental results for TP content are mean value of triplicates.
bGallic acid equivalent.
Figure 3.
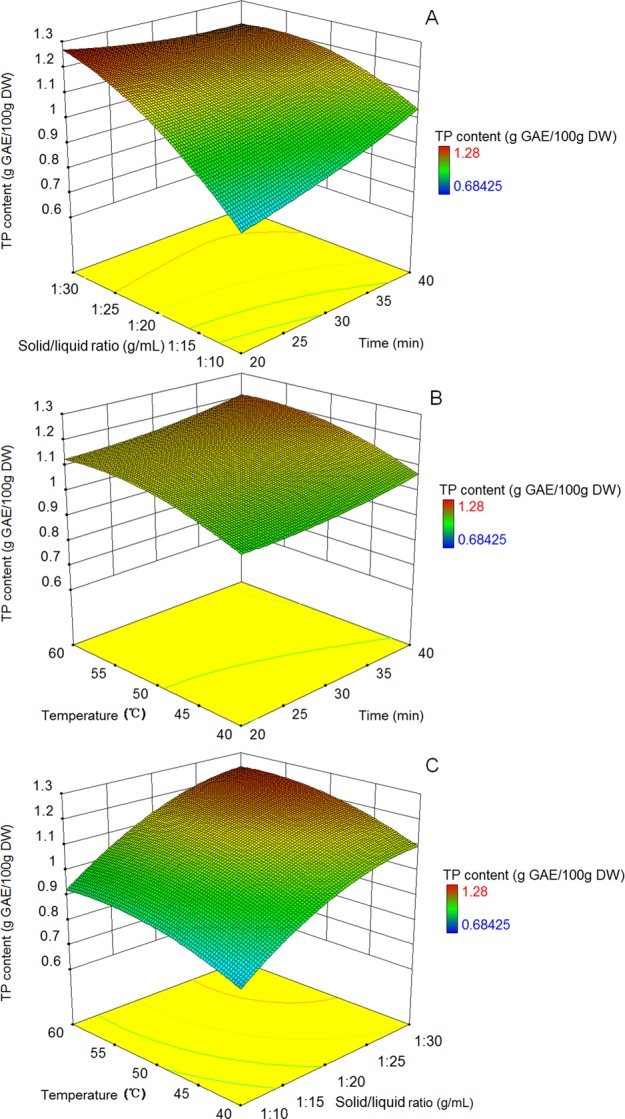
Interaction effect (A) between extraction time and solid/liquid ratio, (B) between extraction time and temperature, (C) between extraction solid/liquid ratio and temperature on TP content obtained from CCC design.
The optimal ultrasound-assisted phenolic extraction condition obtained from the CCC model was shown in Table 2: extraction temperature, 57 °C; solid/liquid ratio, 1:30 g/mL; extraction time, 20 min. The model predicted a maximum response of 1.29 g GAE/100 g DW under optimal condition. TP content of 1.27 ± 0.08 g GAE/100 g DW obtained from real experiments validated the CCC model (Table 2). Mircea. et al. obtained 0.08∼0.15 g GAE/100 g DW phenolics from methanolic extract of D. indicum (L.) Des Moul (another variety of D. indicum) flower through ultrasound-assisted extraction18. The TP content we got in this study was about 10 times higher than that of D. indicum (L.) Des Moul, which reveals the potential of developing D. indicum var. aromaticum as a source of phenolics.
Table 2.
Optimal condition and TP content obtained from prediction and real experiment under optimal condition.
| Optimal condition | TP content (g GAEa/100 g DW) | |||
|---|---|---|---|---|
| Time (min) | Solid/liquid ratio (g/mL) | Temperature (°C) | Predicted | Experimentalb |
| 20 | 1:30 | 57 | 1.29 | 1.27 ± 0.08 |
aGallic acid equivalent.
bMean ± SD (n = 3).
Optimization of HPLC conditions
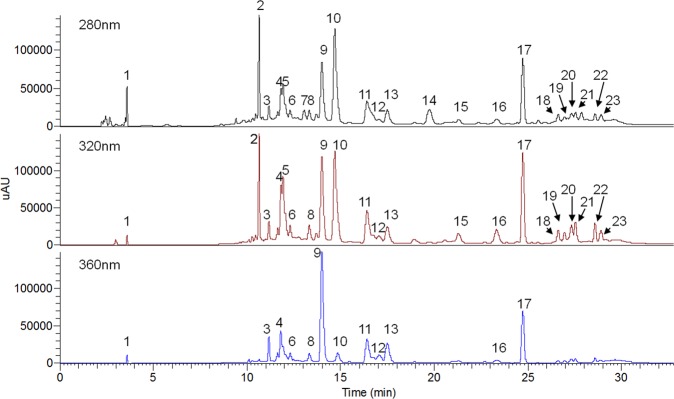
To analyze phenolic compounds by HPLC, acid was usually added in mobile phase9,19. Adding adequate amount of acid into mobile phase is beneficial to achieve complete separation, to lighten peak trailing, and to improve resolution of the compounds20. In this study, mobile phase B was fixed as acetonitrile, and mobile phase A was tested by various concentrations of formic acid (0.1%, 0.5%, and 1% v/v). It was found that using 0.1% or 0.5% formic acid did not result in the separation result as satisfactory as using 1% formic acid. Therefore, 1% formic acid was chosen in the following studies. Two 5 μm C18 columns (250 * 4.6 mm and 150 * 4.6 mm) were tested. By comparing with the longer column, using the shorter one could decrease flow rate from 1 mL/min to 0.6 mL/min and greatly shorten the elution time without affecting the separation result much. At last, various gradient methods were tried until a satisfactory chromatogram was obtained. Eventually, an effective and economic HPLC method was established. Figure 4 shows the HPLC-PDA chromatograms of phenolic compounds in D. indicum var. aromaticum flower.
Figure 4.
HPLC-PDA chromatograms of phenolic profile in D. indicum var. aromaticum flower under 280 nm, 320 nm, and 360 nm wavelength.
Identification of phenolic compounds
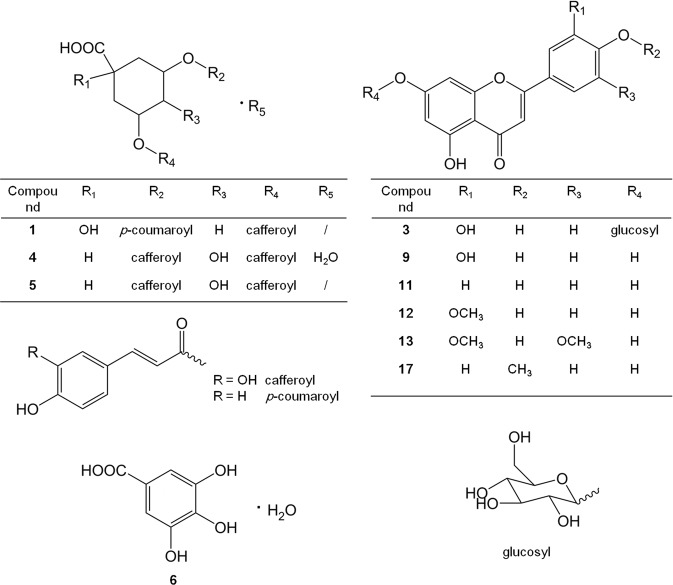
In order to identify phenolic compounds, the external standard retention time and UV spectra from literatures were used to assess the target peaks preliminary, and the molecular structures were deduced based on MS and corresponding MSn fragment signals. The identified phenolic compounds were classified into hydroxybenzonic acid group, hydroxycinnamic acid group, and flavonoid group in this study. Besides, several fatty acids were identified together with the phenolic compounds. A summary of the MSn fragmentations of all compounds were shown (Table 3), and definable structures of identified phenolic compounds were drawn (Fig. 5).
Table 3.
HPLC-MSn information on phenolic compounds and hydroxy fatty acids identification in D. indicum var. aromaticum flower.
| Peak No. | Rt (min) | UV λmax (nm) | [M-H]− | MSn m/z (% base peak) | Identification |
|---|---|---|---|---|---|
| 1 | 3.59 | 260 | 499 | MS2[499]: 481 (30), 353 (10), 191 (100), 173 (60) | 3-O- Caffeoyl-5-O-p- coumaroylquinic acid |
| 2 | 10.64 | 230, 300 | 685 | MS2[685]: 539 (100), 523 (60), 665 (35); MS3[539]: 377 (100) | Coumaroyloleuropein |
| 3 | 11.17 | 250, 340 | 447 | MS2[447]: 285 (100) | Luteolin-O-glucoside |
| 4 | 11.81 | 250, 330 | 533 | MS2[533]: 353 (100), 335 (5), 191 (8), 173 (2); MS3[353]: 309 (1), 191 (100), 179 (16), 173 (6), 135 (4) | 3,5-Dicaffeoylquinic acid monohydrate |
| 5 | 11.92 | 240, 330 | 515 | MS2[515]: 353 (100), 335 (5), 191 (4); MS3[353]: 191 (100), 179 (40), 173 (10), 135 (7) | 3,5-Dicaffeoylquinic acid |
| 6 | 12.30 | 250, 270, 320 | 187 | MS2[187]: 169 (6), 125 (100) | Gallic acid monohydrate |
| 7 | 13.05 | 250, 290 | 173 | MS2[173]: 146 (8), 131 (100), 127 (15) | Shikimic acid isomer |
| 8 | 13.32 | 250, 270, 320 | 569 | MS2[569]: 551 (2), 525 (100), 459 (4), 417 (20), 391 (10); MS3[525]: 507 (16), 482 (18), 427 (10), 379 (28), 235 (15), 193 (100) | Methyoxyoleuropein isomer |
| 9 | 13.99 | 250, 350 | 285 | MS2[285]: 241 (35), 217 (20), 199 (25), 175 (25) | Luteolin |
| 10 | 14.68 | 300 | 453 | MS2[453]: 435 (2), 411 (6), 393 (100); MS3[393]: 363 (20), 249 (30), 231 (100), 205 (35); MS4[231]: 187 (100) | Prenyl-dimethoxy- caffeoyl-p-coumaric acid |
| 11 | 16.41 | 270, 330 | 269 | MS2[269]: 225 (40), 201 (12), 149 (10) | Apigenin |
| 12 | 17.05 | 250, 330 | 299 | MS2[299]: 284 (100); MS3[284]: 256 (100), 227 (10), 212 (5) | Chrysoeriol |
| 13 | 17.49 | 250, 270, 340 | 329 | MS2[329]: 314 (100); MS3[314]: 299 (100), 285 (10) | Tricin |
| 14 | 19.74 | 260 | 659 | MS2[659]: 615 (100), 591 (1), 573 (1), 505 (1), 265 (2); MS3[615]: 573 (100) | Unknown |
| 15 | 21.30 | 240, 310 | 293 | MS2[293]: 275 (100), 265 (25), 231 (45), 205 (40), 249 (90), 193 (28), 163 (20) | Monohydroxy- octadecaditrienoic acid |
| 16 | 23.33 | 250, 310 | /a | /a | Unknown |
| 17 | 24.73 | 270, 330 | 283 | MS2[283]: 269 (100) | Acacetin |
| 18 | 26.61 | 270, 330, 350 | /a | /a | Unknown |
| 19 | 26.96 | 270, 310 | 313 | MS2[313]: 295 (100), 277 (45), 215 (10), 183 (18) | Dihydroxy- octadecenoic acid |
| 20 | 27.33 | 270, 320, 350 | 309 | MS2[309]: 291 (50), 265 (10), 247 (100) | Dihydroxy- octadecatrienoic acid I |
| 21 | 27.54 | 270, 320 | 309 | MS2[309]: 291 (100), 265 (32), 247 (30) | Dihydroxy- octadecatrienoic acid II |
| 22 | 28.58 | 310 | 295 | MS2[295]: 277 (100), 265 (30) | Monohydroxy- octadecadienoic acid |
| 23 | 28.91 | 270, 320, 350 | 297 | MS2[297]: 279 (100), 251 (65), 223 (20) | Monohydroxy- octadecenoic acid |
aNo data available.
Figure 5.
Definable chemical structures of identified phenolic compounds in D. indicum var. aromaticum flower.
Hydroxycinnamic acid group
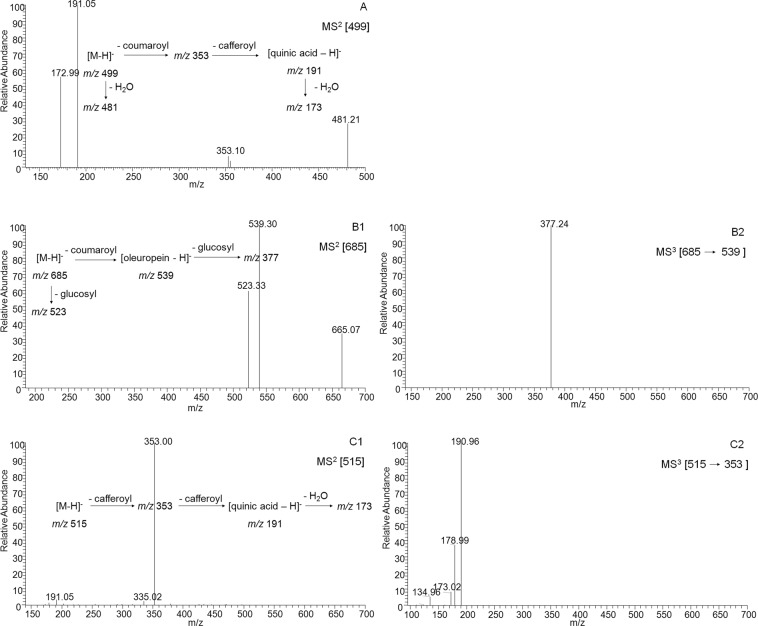
Compound 1 gave [M-H]− ion at m/z 499, and the parent ion produced fragment ions at m/z 353 ([M-H-146]−) and 191([M-H-146-162]−) in MS2 spectrum, illustrating the losses of a coumaroyl moiety (146 Da) and a caffeoyl moiety (162 Da). In addition, fragment ion at m/z 191 revealed the existence of quinic acid moiety. Refering to previous report on ion fragments intensity characters of different esterification position of quinic acid structure21, compound 1 was identified as 3-O- caffeoyl-5-O-p- coumaroylquinic acid (Fig. 6A).
Figure 6.
MSn spectra and major fragmentations for the typical hydroxycinnamic acid derivatives in D. indicum var. aromaticum flower. (A) Compound 1 (3-O-caffeoyl-5-O-p- coumaroylquinic acid); (B) compound 2 (coumaroyloleuropein); (C) compound 5 (3,5-dicaffeoylquinic acid).
Compound 2 was identified as coumaroyloleuropein. As illustrated in Fig. 6B, firstly, the presence of its base ion at m/z 539 in MS2 with next base ion at m/z 377 in MS3 indicated the cleavage of a glucosyl from oleuropein22,23. Secondly, base ion at m/z 539 generated from parent ion at m/z 685 revealed the loss of a coumaroyl moiety, therefore, a structure of coumaroyloleuropein was deduced. Oleuropein is a bitter phenolic compound which mainly exists in green olives, olive leaves, and argan oil24,25. It is for the first time to report the presence of oleuropein derivative in D. indicum var. aromaticum flower.
For compound 8, its parent ion at m/z 569 was 30 Da higher than that of oleuropein at m/z 539, which means that compound 8 might be methoxyoleuropein, but its MSn spectrum did not give fragment ions as m/z 539 or 377 as expected according to literature reports22,23. Compound 8 was tentatively named as methoxyoleuropein isomer.
Spectrum of compound 5 showed parent ion at m/z 515 in MS and fragment ions at m/z 353 ([M-H-162]−) and 191([M-H-162-162]−) in MS2, indicating that two caffeoyl moieties (162 Da) cleaved from a quinic acid moiety. According to its characteristic ion fragment intensity pattern, compound 5 was deduced as 3,5-dicaffeoylquinic acid (Fig. 6C)26. Compound 4 gave almost the same ion fragment pattern as compound 5, except that its parent ions at m/z 533 was 18 Da higher than that of compound 5 ([M-H]− at m/z 515), therefore, it was deduced as 3,5-dicaffeoylquinic acid monohydrate.
Compound 10 was tentatively deduced to be prenyl-dimethoxy-caffeoyl-p-coumaric acid. The MS2 base fragment ion at m/z 393 [M-H-60]− could be produced by the losses of two methoxy groups from [M-H]− ion at m/z 453. Further, fragment ion at m/z 231 in MS3 would be produced by the cleavage of a caffeoyl moiety from the MS2 fragment ion at m/z 393, in addition, the fragment ion at m/z 231 in MS3 proved a prenylcoumaric acid moiety. Considering ion at m/z 393 in MS2, cleavage of two methyl groups from the preneyl moiety could produce the ion at m/z 363 in MS3.
Hydroxybenzoic acid group
Compound 6 exhibited a fragment peak [M-H-H2O]− at m/z 169 with a base ion peak [M-H-H2O-CO2]− at m/z 125, corresponding to MS signals produced by gallic acid standard. Furthermore, parent ion of compound 6 at m/z 187 (18 Da higher than that of gallic acid) indicated the peak was gallic acid monohydrate19.
Compound 7 had the same parent ion with shikimic acid at m/z 173, but its UV absorbance and MS2 fragments did not fit to previous report on shikimic acid27. The obtained information of MS fragments of compound 7 is insufficient to deduce its precise structure, so compound 7 was called shikimic acid isomer tentatively.
Flavonoid group
There were totally six flavonoids identified in D. indicum var. aromaticum flower in this study, and all of them are flavones and flavone derivatives.
Through comparison of retention time, UV spectrum, and MSn patterns with external standards, luteolin, apigenin, and acacetin were identified for compounds 9, 11, and 17, respectively. UV spectrum of compound 3 was pretty similar as luteolin, and the cleavage of a glucose from luteolin molecule could produce MS2 ion at m/z 285 ([M-H-162]−) from the parent ion at m/z 447. Thus, compound 3 was deduced as luteolin-O-glucoside.
The identical MS patterns of compound 12 ([M-H]− 299; MS2[299]: 284 (100); MS3[284]: 256 (100)) and of compound 13 ([M-H]− 329; MS2[329]: 314 (100); MS3[314]: 299 (100)) have been reported as chrysoeriol (3′-methoxy derivative of luteolin) and tricin in previous studies19,28. Chrysoeriol exists in many species of Artemisia genus29, and tricin occurs in rice bran and other grass specie such as wheat, maize, and barley30, both of these two flavones are for the first time to be reported in D. indicum var. aromaticum flower.
Other compounds
Several hydroxy fatty acids were identified together with phenolic compounds. Compounds 15, 19, 20, 21, 22, and 23 were characterized to be hydroxy fatty acids, according to the phenomenon that water molecules cleaved from the aliphatic moiety consecutively in their MS2 spectra31,32. These peaks show up at end of the chromatogram, and most of them have longer retention time than the phenolic compounds except monohydroxy-octadecaditrienoic (compound 15) which comes earlier than peak of acacetin (compound 17) in the chromatogram (Fig. 4). Hydroxy fatty acids were found to have many biological activities, for instance, cytotoxicity and anti-inflammatory activity33, which might contribute to the therapeutic effects of D. indicum var. aromaticum plant, too.
Method validation and phenolic compounds quantification
In this study, parameters of linearity, sensitivity, accuracy and precision were used for HPLC method validation. Results for method validation were summarized in Table 4. The correlation coefficients of the calibration curves were all over 0.99, which reflected that the detected concentration values were highly coincident with the real values. The method sensitivity was confirmed to be adequate as LOD and LOQ values were lower than 0.036 μg/mL and 0.109 μg/mL. Recovery rates of apigenin, acacetin, and luteolin obtained here varied from 99.50% to 102.19%, and the RSD values were lower than 1.84% and 2.70% for intra-day and inter-day experiments, respectively, indicating high accuracy and good precision of the analytical method in this study.
Table 4.
Results for method validation.
| No. | Standard | Linear regression curve | R2 | LOD (μg/mL) | LOQ (μg/mL) | Recovery rate (%)a | RSD (%)b | |
|---|---|---|---|---|---|---|---|---|
| Intra-day | Inter-day | |||||||
| 1 | Gallic acid | Y = 18817X − 10155 | 0.9996 | 0.014 | 0.042 | /c | /c | /c |
| 2 | Caffeic acid | Y = 43686X − 19547 | 0.9998 | 0.010 | 0.030 | /c | /c | /c |
| 3 | Apigenin | Y = 29655X + 6320.6 | 0.9999 | 0.013 | 0.040 | 99.50 ± 2.69 | 1.16 | 2.33 |
| 4 | Acacetin | Y = 26463X + 9248.4 | 0.9999 | 0.013 | 0.041 | 102.19 ± 3.10 | 0.68 | 1.54 |
| 5 | Luteolin | Y = 24041X + 44178 | 0.9989 | 0.036 | 0.109 | 100.47 ± 2.96 | 1.84 | 2.70 |
aRecovery rate (%) = [(measured amount after spiking − measured amount before spiking)/actual amount spiked] * 100.
bRSD (%) = (SD/Mean) * 100, where RSD means relative standard deviation and SD means standard deviation.
cNot analyzed.
As shown in Table 5, content of phenolics in D. indicum var. aromaticum flower identified by HPLC-MSn was 6.42 ± 0.32 mg/g DW, consisting of 3.63 ± 0.17 mg/g DW of flavonoids and 2.79 ± 0.15 mg/g DW of phenolic acids. Among all the phenolic compounds in D. indicum var. aromaticum flower, luteolin (1.61 ± 0.11 mg/g DW) took up the highest proportion, around 25% of the TP content, which was found to be similar to the luteolin content (0.5∼2.1 mg/g DW) in flower of D. indicum (original variety of D. indicum var. aromaticum) reported in previous study34.
Table 5.
Content of phenolic compounds in D. indicum var. aromaticum flower.
| Peak No. | Compound | Group | Peak Area proportion (% total area) | Contenta (mg/g DW) |
|---|---|---|---|---|
| 1 | 3-O-Caffeoyl-5-O-p- coumaroylquinic acid | Hydroxycinnamic acid | 2.21 | 0.25 ± 0.01 |
| 2 | Coumaroyloleuropein | Hydroxycinnamic acid | 9.69 | 0.42 ± 0.03 |
| 3 | Luteolin-O-glucoside | Flavonoid | 2.49 | 0.16 ± 0.01 |
| 4 | 3,5-Dicaffeoylquinic acid monohydrate | Hydroxycinnamic acid | 4.34 | 0.20 ± 0.00 |
| 5 | 3,5-Dicaffeoylquinic acid | Hydroxycinnamic acid | 5.43 | 0.24 ± 0.00 |
| 6 | Gallic acid monohydrate | Hydroxybenzoic acid | 2.80 | 0.31 ± 0.02 |
| 7 | Shikimic acid isomer | Hydroxybenzoic acid | 3.07 | 0.34 ± 0.02 |
| 8 | Methyoxyoleuropein isomer | Hydroxycinnamic acid | 2.97 | 0.14 ± 0.01 |
| 9 | Luteolin | Flavonoid | 19.75 | 1.61 ± 0.11 |
| 10 | Prenyl-dimethoxy-caffeoyl-p- coumaric acid | Hydroxycinnamic acid | 20.88 | 0.89 ± 0.06 |
| 11 | Apigenin | Flavonoid | 9.12 | 0.57 ± 0.02 |
| 12 | Chrysoeriol | Flavonoid | 2.23 | 0.13 ± 0.01 |
| 13 | Tricin | Flavonoid | 3.95 | 0.24 ± 0.00 |
| 17 | Acacetin | Flavonoid | 11.09 | 0.92 ± 0.02 |
| Phenolic acids content | 2.79 ± 0.15 | |||
| Flavonoids content | 3.63 ± 0.17 | |||
| Content of phenolics identified by HPLC-MSn | 6.42 ± 0.32 |
aMean ± SD (n = 3).
Gong. et al. have isolated 20 mg acacetin from 1000 g dry flower of D. indicum var. aromaticum by absolute ethanol cold extraction and column chromatographyl35. Comparing with the acacetin content in this study (0.92 ± 0.02 mg/g DW, 0.92‰), the low yield of acacetin (0.02‰) in Gong’s work could be mainly caused by low extraction efficiency and high waste ratio during the isolation procedure. Previous studies stated that change of climate conditions (temperature, irradiation, rainfall, ect.) between years and different harvest periods could influence chemical composition and component content in plant material36,37. There were two phenolic compounds acacetin-7-O-β-D-glucopy ranoside and apignein-7-O-β-D-glucopy ranoside isolated from D. indicum var. aromaticum flower in Lu’s research by column chromatography and semi-preparative HPLC in 20092 but not found in this study. Even though plant materials were harvested from the same location, certain degree of difference of chemical profile would exist between different material batches, which should be considered and accepted in research work.
Conclusions
The phenolic compound composition of D. indicum var. aromaticum flower was studied extensively for the first time. Firstly, ethanol-acetic acid (70%:2%, v/v) was selected as extraction solvent. Ultrasound-assisted extraction method was optimized by CCC design, and under optimal condition (extraction temperature, 57 °C; solid/liquid ratio, 1:30 g/mL; extraction time, 20 min), 1.27 ± 0.08 g GAE/100 g DW TP content was obtained from D. indicum var. aromaticum flower. Secondly, an effective and economic HPLC-PDA-ESI-MSn method was established, and the analytical method was validated by sensitivity, accuracy, and precision. As a result, 14 phenolic compounds were identified and quantified, including 8 phenolic acids and 6 flavonoids. For the first time, oleuropein derivatives, chrysoeriol, and tricin are reported in D. indicum var. aromaticum flower. In summary, the optimized method for extracting and characterizing phenolic compounds in D. indicum var. aromaticum flower has significant meaning for future pharmaceutical and medicinal research on D. indicum var. aromaticum plant, and the results in this work would provide references for future herbal research.
Material and Methods
Chemicals and materials
Eight standards were used in this work: gallic acid, caffeic acid, luteolin were purchased from Adamas Reagent (Shanghai, China); acacetin was purchased from TCI (Tokyo, Japan); apigenin was purchased from WAKO Pure Chemical Industries (Osaka, Japan); protocatechuic acid, ferulic acid, and hesperidin were purchased from Sigma-Aldrich (MO, USA). Acetonitrile and formic acid in HPLC grade were purchased from Fisher Chemical (Geel, Belgium). Ethanol and acetic acid in AR grade were purchased from Sinopharm Chemical Reagent Co., Ltd (Shanghai, China). Folin-Ciocalteu and Na2CO3 were purchased from Sigma-Aldrich (MO, USA).
Flower of D. indicum var. aromaticum was harvested in Shen Nongjia area of Hubei province, China. The plant species was identified by Prof. Keli Chen, Hubei University of Chinese Medicine, and authenticated by Herbarium, Kunming Institute of Botany, Chinese Academy of Sciences. The certificate of plant material authentication was provided as Supplementary. Plant material was harvested freshly, and spread at a shady, cool and well-ventilated place to dry naturally. Dry material was kept in −80 °C freezer before using.
Sample extraction
Dry flowers were milled to a fine and uniform particle size by a YB-500A grander (Shanghai Lijian Machinery Co., Ltd, Shanghai, China). Half gram of the powder was mixed with certain volume of solvent, and followed with an ultrasound bath extraction (KQ-600DB, 40 kHz, Kunshan Ultrasonic Instruments Co., Ltd, China). The ultrasound working power was set at 360 W. External water circulated from a water bath to keep the extraction temperature stable. Centrifugation of 10,000 g was operated at 4 °C for 10 min after extraction, using an Allegra X-30R centrifuge (Bechman Coulter, Inc., Califonia, USA). Supernatant was used for TP content measurement or HPLC-MS analysis. All extraction experiments were operated in triplicate.
Selection of extraction solvent
Different concentrations of ethanol (30%, 50%, and 70%) and acetic acid (2%, 5%, and 10%) were mixed in orthogonal design. Half gram of weighed samples were ultrasound extracted with 10 mL various solvents at 40 °C for 30 min. Supernatant was collected after centrifugation as described above.
Selection of the range of extraction time
Half gram of weighed samples were ultrasound extracted with 10 mL ethanol-acetic acid (70%:2%, v/v) (i.e. Vethanol:Vacetic acid:Vwater = 70:2:28) solvent at 40 °C for different time (20, 30, 40, 50, and 60 min). Supernatant was collected after centrifugation as described above.
Selection of the range of solid/liquid ratio
Half gram of weighed samples were ultrasound extracted with ethanol-acetic acid (70%:2%, v/v) under different solid/liquid ratio (g/mL) (1:10, 1:15, 1:20, 1:25, and 1:30) at 40 °C for 30 min. Supernatant was collected after centrifugation as described above.
Selection of the range of extraction temperature
Half gram of weighed samples were ultrasound extracted with 10 mL ethanol-acetic acid (70%:2%, v/v) at different temperature (30, 40, 50, and 60 °C) for 30 min. Supernatant was collected after centrifugation as described above.
CCC design
Experiment was designed according to CCC model with five levels (−1.68, −1, 0, 1, and 1.68) and three variables (extraction temperature, solid/liquid ratio, and extraction time) to achieve the best variable combination for TP extraction. The CCC design consists of six central points, eight factorial points, and six axial points, generating 20 sets of experiments. Both coded and uncoded form of independent variables were shown in Table 1.
Determination of TP content
Folin-Ciocalteu method reported in Cicco’s study38 was adopted to measure the TP content, with slight modifications. In brief, 150 μL of appropriately diluted extract, 150 μL of Folin-Ciocalteu (50%, v/v), and 1.2 mL of Na2CO3 (5%, m/v) were mixed. After two hours’ incubation at room temperature, absorbance under 760 nm was measured with a blank (150 μL of extraction solvent instead of the extract) using a nucleic acid/protein analyzer (Beckman Coulter, DU 730, CA, USA). Calibration curve was established using gallic acid standard. Results were recorded as gallic acid equivalent (g GAE/100 g DW). Absorbance experiments were operated in triplicate.
HPLC-PDA and HPLC-ESI-MSn conditions
Phenolic compounds profile in D. indicum var. aromaticum flower was seperated by HPLC-PDA in an Ultimate 3000 system (Thermo Fisher Scientific, Waltham, MA, USA). Phenolic compounds were firstly eluted by gradient program consisting of mobile phase A (0.1%, 0.5%, or 1% of formic acid) and mobile phase B (acetonitrile), and then 1% formic acid was chosen for further analysis. An Agilent C18 (5 μm, 250 * 4.6 mm) column and a Phenomenex Luna C18 (5 μm, 150 * 4.6 mm) column were tested secondly. The shorter column was chosen based on a comprehensive comparison of separation effect, elution time, and solvent cost. In addition, several gradient programs were tested until satisfactory separation results were achieved. Finally, a gradient elution program was modified by using mobile phase A (1% formic acid) and mobile phase B (acetonitrile) at 0.6 mL/min flow rate with 5 μL of injection volume. The gradient program was as follows: 5% B at 0–3 min; 5–40% B at 3–8 min; 40% B at 8–15 min; 40–50% B at 15–20 min; 50–95% B at 20–25 min; 95-5% B at 25–30 min; 5% B at 30–33 min. Column temperature was 30 °C. Wavelengths of 280 nm, 320 nm, and 360 nm were set for recording chromatograms, according to literature reports15,39,40.
HPLC-ESI-MSn (Thermo Fisher Scientific, Waltham, MA, USA) was used to identify and characterize phenolic compounds. The same HPLC condition described above was used. Selected compounds were analyzed by MS2, MS3, till MS4 as needed. Negative mode of ionization was performed, and a mass range of m/z 50∼700 was covered for full scan. Collision gas was ultrahigh pure helium (He). The ionization parameters were set as follows:, 40 units/min of sheath gas (N2); 2 units/min of auxiliary gas (N2); 4.50 kV of spray voltage; 300 °C of capillary temperature; −1.00 V of capillary voltage; −8.77 V of tube lens offset voltage.
Method validation and phenolic compounds quantification
Five calibration curves were established with different concentrations for different standards: 0.5, 5, 25, and 50 μg/mL for gallic acid (at 280 nm wavelength), caffeic acid (at 320 nm wavelength), and apigenin (at 320 nm wavelength); 5, 25, 50, and 100 μg/mL for acacetin (at 320 nm wavelength); 5, 25, 50, and 150 μg/mL for luteolin (at 360 nm wavelength). According to signal to noise ratios of 3 and 10, limit of detection (LOD) and limit of quantification (LOQ) were calculated, respectively, to check the method sensitivity.
To confirm method accuracy, apigenin, acacetin, and luteolin standards were used for recovery rate test. Three levels of each standard were spiked to 0.7 mL of sample extract: apigenin (10, 20, and 30 μg/mL), acacetin (15, 30, and 45 μg/mL), luteolin (25, 50, and 75 μg/mL).
To determine intra-day and inter-day precision, spiked samples with standards were analyzed at five different time points in one day and at the same time point for five consecutive days.
Since not all standards for each compound were commercially available, only apigenin, acacetin, and luteolin were quantified directly with their authentic standards, while other compounds were quantified by internal standards with similar structure and properties using relative response factor (RRF). The internal standard protocatechuic acid was used to quantify 3-O-caffeoyl-5-O-p-coumaroylquinic acid, gallic acid monohydrate, and shikimic acid isomer. The internal standard ferulic acid was used to quantify coumaroyloleuropein, 3,5-dicaffeoylquinic acid monohydrate, 3,5-dicaffeoylquinic acid, methyoxyoleuropein isomer, and prenyl-dimethoxy-caffeoyl-p-coumaric acid. The internal standard hesperidin was used to quantify tricin, luteolin-O-glucoside and chrysoeriol.
Fixed concentration (20 μg/mL) of internal standards were mixed with the sample extract and with the known compounds (25 μg/mL) mentioned above. Quantification of the unknown compounds was carried out according to the following formula:
where RRF = (Ak/Ai)/(Ck/Ci); Ak means peak area of known phenolic compound; Ai means peak area of internal standard; Ck means concentration of known phenolic compound; Ci means concentration of internal standard; A means peak area of unknown phenolic compound; R means recovery rate.
Statistical analysis
For the single factor extraction tests, ANOVA was adopted and the TP content of different extraction treatments were analyzed under Tukey’s test by Minitab 17 (Minitab Inc., State College, PA, USA). Data were expressed as mean ± SD (n = 3). Statistic of the CCC design was performed using Design Expert 10 (Stat Ease Inc., Minneapolis, USA). ANOVA was adopted for analyzing model main effect and interaction between variables. P < 0.05 was considered as significant.
Acknowledgements
This study was financially supported by China Tobacco Hubei Industrial Co. Ltd (Project No. 2017B025XL02), Guangxi Science and Technology Project (GuiKeAD17195081), and Bagui Scholar Program of Guangxi Province (2016). The authors gratefully acknowledge Guangfeng Luo, Hubei Xinye Tobacco Science and Technology Development Co. LTD, Wuhan, China for the language polishing.
Author Contributions
L.Z. wrote the manuscript. Z.Y., C.L. and Y.Z. provided technical support and helpful discussion. L.Z. and L.R. conducted the experiments. G.X. and Y.L. designed the study. Z.Y., C.L. and Y.L. participated in writing and modifying the manuscript. All authors approved the manuscript.
Data Availability
The datasets generated and analyzed in the current study are included in the main text and the raw data are available from the corresponding author.
Competing Interests
The authors declare no competing interests.
Footnotes
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Yi Liu, Email: yiliuchem@whu.edu.cn.
Chao Li, Email: lichao@hbtobacco.cn.
References
- 1.Kun LZWNZ, Yunzhui SWLWX. Studies on Antimicrobial and Antioxidation in Vitro of Essential Oil from D. indicum Var. Aromaticum [J] Journal of China Three Gorges University (Natural Sciences) 2006;2:022. [Google Scholar]
- 2.Lu J, et al. Studies on chemical constituents of Dendranthema indicum var. aromaticum. Zhong yao cai= Zhongyaocai= Journal of Chinese medicinal materials. 2009;32:53–55. [PubMed] [Google Scholar]
- 3.Seelinger G, Merfort I, Schempp CM. Anti-Oxidant, Anti-inflammatory and Anti-Allergic Activities of Luteolin. Planta Med. 2008;74:1667–1677. doi: 10.1055/s-0028-1088314. [DOI] [PubMed] [Google Scholar]
- 4.Shim HY, et al. Acacetin-induced apoptosis of human breast cancer MCF-7 cells involves caspase cascade, mitochondria-mediated death signaling and SAPK/JNK1/2-c-Jun activation. Mol Cells. 2007;24:95–104. [PubMed] [Google Scholar]
- 5.Hsu YL, Kuo PL, Liu CF, Lin CC. Acacetin-induced cell cycle arrest and apoptosis in human non-small cell lung cancer A549 cells. Cancer Lett. 2004;212:53–60. doi: 10.1016/j.canlet.2004.02.019. [DOI] [PubMed] [Google Scholar]
- 6.Cassidy A, et al. Habitual intake of flavonoid subclasses and incident hypertension in adults. Am J Clin Nutr. 2011;93:338–347. doi: 10.3945/ajcn.110.006783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kwon YI, Apostolidis E, Kim YC, Shetty K. Health benefits of traditional corn, beans, and pumpkin: In vitro studies for hyperglycemia and hypertension management. J Med Food. 2007;10:266–275. doi: 10.1089/jmf.2006.234. [DOI] [PubMed] [Google Scholar]
- 8.Tao Y, Sun DW. Enhancement of Food Processes by Ultrasound: A Review. Crit Rev. Food Sci. 2015;55:570–594. doi: 10.1080/10408398.2012.667849. [DOI] [PubMed] [Google Scholar]
- 9.Lv, Y. Triterpenes and phenolic compounds in apple fruit (Malus domestica Borkh.). Vol. 2016 (2016).
- 10.Wang J, Sun BG, Cao YP, Tian YA, Li XH. Optimisation of ultrasound-assisted extraction of phenolic compounds from wheat bran. Food Chem. 2008;106:804–810. doi: 10.1016/j.foodchem.2007.06.062. [DOI] [Google Scholar]
- 11.Toma M, Vinatoru M, Paniwnyk L, Mason TJ. Investigation of the effects of ultrasound on vegetal tissues during solvent extraction. Ultrason Sonochem. 2001;8:137–142. doi: 10.1016/S1350-4177(00)00033-X. [DOI] [PubMed] [Google Scholar]
- 12.Al-Owaisi M, Al-Hadiwi N, Khan SA. GC-MS analysis,determination of total phenolics,flavonoid content and free radical scavenging activities of various crude extracts of Moringa peregrina(Forssk.) Fiori leaves. Asian Pacific Journal of Tropical Biomedicine. 2014;4:964–970. doi: 10.12980/APJTB.4.201414B295. [DOI] [Google Scholar]
- 13.Kothari V, Gupta A, Naraniwal M. Comparative study of various methods for extraction of antioxidant and antibacterial compounds from plant seeds. Journal of Natural Remedies. 2012;12:162–173. [Google Scholar]
- 14.Kala HK, Mehta R, Sen KK, Tandey R, Mandal V. Critical analysis of research trends and issues in microwave assisted extraction of phenolics: Have we really done enough. TrAC Trends in Analytical Chemistry. 2016;85:140–152. doi: 10.1016/j.trac.2016.09.007. [DOI] [Google Scholar]
- 15.Vagiri M, Ekholm A, Andersson SC, Johansson E, Rumpunen K. An Optimized Method for Analysis of Phenolic Compounds in Buds, Leaves, and Fruits of Black Currant (Ribes nigrum L.) J Agr Food Chem. 2012;60:10501–10510. doi: 10.1021/jf303398z. [DOI] [PubMed] [Google Scholar]
- 16.Wang T, Liang H, Yuan Q. Optimization of ultrasonic-stimulated solvent extraction of sinigrin from Indian mustard seed (Brassica Juncea L.) using response surface methodology. Phytochemical analysis: PCA. 2011;22:205–213. doi: 10.1002/pca.1266. [DOI] [PubMed] [Google Scholar]
- 17.Tabart J, Kevers C, Evers DL, Dommes J. Ascorbic acid, phenolic acid, flavonoid, and carotenoid profiles of selected extracts from Ribes nigrum. J Agr Food Chem. 2011;59:4763–4770. doi: 10.1021/jf104445c. [DOI] [PubMed] [Google Scholar]
- 18.Mircea CC, Cioancă O, Draghia L, Hăncianu M. Morphological Characteristics, Phenolic and Terpenoid Profiles in Garden Chrysanthemum Grown in Different Nutritional Conditions. Notulae Botanicae Horti Agrobotanici Cluj-Napoca. 2015;43:371–379. [Google Scholar]
- 19.Kang JG, Price WE, Ashton J, Tapsell LC, Johnson S. Identification and characterization of phenolic compounds in hydromethanolic extracts of sorghum wholegrains by LC-ESI-MSn. Food Chem. 2016;211:215–226. doi: 10.1016/j.foodchem.2016.05.052. [DOI] [PubMed] [Google Scholar]
- 20.Dalluge JJ, Nelson BC, Thomas JB, Sander LC. Selection of column and gradient elution system for the separation of catechins in green tea using high-performance liquid chromatography. J Chromatogr A. 1998;793:265–274. doi: 10.1016/S0021-9673(97)00906-0. [DOI] [PubMed] [Google Scholar]
- 21.Clifford MN, Marks S, Knight S, Kuhnert N. Characterization by LC-MSn of four new classes of p-coumaric acid-containing diacyl chlorogenic acids in green coffee beans. J Agr Food Chem. 2006;54:4095–4101. doi: 10.1021/jf060536p. [DOI] [PubMed] [Google Scholar]
- 22.Savarese M, Demarco E, Sacchi R. Characterization of phenolic extracts from olives (Olea europaea cv. Pisciottana) by electrospray ionization mass spectrometry. Food Chem. 2007;105:761–770. doi: 10.1016/j.foodchem.2007.01.037. [DOI] [Google Scholar]
- 23.Ryan D, et al. Liquid chromatography with electrospray ionisation mass spectrometric detection of phenolic compounds from Olea europaea. J Chromatogr A. 1999;855:529–537. doi: 10.1016/S0021-9673(99)00719-0. [DOI] [PubMed] [Google Scholar]
- 24.Rigacci, S. & Stefani, M. Nutraceutical Properties of Olive Oil Polyphenols. An Itinerary from Cultured Cells through Animal Models to Humans. Int J Mol Sci17 (2016). [DOI] [PMC free article] [PubMed]
- 25.Charrouf Z, Guillaume D. Phenols and polyphenols from Argania spinosa. Am J Food Technol. 2007;2:679–683. doi: 10.3923/ajft.2007.679.683. [DOI] [Google Scholar]
- 26.Clifford MN, Knight S, Kuhnert N. Discriminating between the six isomers of dicaffeoylquinic acid by LC-MSn. J Agr Food Chem. 2005;53:3821–3832. doi: 10.1021/jf050046h. [DOI] [PubMed] [Google Scholar]
- 27.Bylund D, Norstrom SH, Essen SA, Lundstrom US. Analysis of low molecular mass organic acids in natural waters by ion exclusion chromatography tandem mass spectrometry. J Chromatogr A. 2007;1176:89–93. doi: 10.1016/j.chroma.2007.10.064. [DOI] [PubMed] [Google Scholar]
- 28.Friscic M, Bucar F, Pilepic KH. LC-PDA-ESI-MSn analysis of phenolic and iridoid compounds from Globularia spp. J Mass Spectrom. 2016;51:1211–1236. doi: 10.1002/jms.3844. [DOI] [PubMed] [Google Scholar]
- 29.Avula B, et al. Quantitative Determination of Flavonoids by Column High-Performance Liquid Chromatography with Mass Spectrometry and Ultraviolet Absorption Detection in Artemisia afra and Comparative Studies with Various Species of Artemisia Plants. J Aoac Int. 2009;92:633–644. [PubMed] [Google Scholar]
- 30.Jiao JJ, et al. Separation and purification of tricin from an antioxidant product derived from bamboo leaves. J Agr Food Chem. 2007;55:10086–10092. doi: 10.1021/jf0716533. [DOI] [PubMed] [Google Scholar]
- 31.Llorent-Martínez EJ, Spínola V, Gouveia S, Castilho PC. HPLC-ESI-MSn characterization of phenolic compounds, terpenoid saponins, and other minor compounds in Bituminaria bituminosa. Industrial Crops and Products. 2015;69:80–90. doi: 10.1016/j.indcrop.2015.02.014. [DOI] [Google Scholar]
- 32.Martin-Arjol I, Bassas-Galia M, Bermudo E, Garcia F, Manresa A. Identification of oxylipins with antifungal activity by LC–MS/MS from the supernatant of Pseudomonas 42A2. Chemistry and physics of lipids. 2010;163:341–346. doi: 10.1016/j.chemphyslip.2010.02.003. [DOI] [PubMed] [Google Scholar]
- 33.Spínola V, Pinto J, Castilho PC. Identification and quantification of phenolic compounds of selected fruits from Madeira Island by HPLC-DAD–ESI-MSn and screening for their antioxidant activity. Food Chem. 2015;173:14–30. doi: 10.1016/j.foodchem.2014.09.163. [DOI] [PubMed] [Google Scholar]
- 34.He DX, Zhang W, Qin MJ. Content analysis of flavonoids in Dendranthema indicum flower from different locations by HPLC. Journal of Plant Resources & Environment. 2009;18:91–93. [Google Scholar]
- 35.Gong, F., Wang, G., Xiao, Y. & Wu, X. Studies on the chemical constituents of the flowers of dendranthema indicum (l.) des monl var. aromaticum q. h. liu et s. f. zhang. Natural Product Research & Development (1994).
- 36.Andersson SC, Rumpunen K, Johansson E, Olsson ME. Carotenoid content and composition in rose hips (Rosa spp.) during ripening, determination of suitable maturity marker and implications for health promoting food products. Food Chem. 2011;128:689–696. doi: 10.1016/j.foodchem.2011.03.088. [DOI] [Google Scholar]
- 37.Zhong, L. J. Anti-cancer effects of bioactive compounds from rose hip fruit in human breast cancer cell lines. Doctor thesis, Sveriges lantbruksuniv. (2017).
- 38.Cicco N, Lanorte MT, Paraggio M, Viggiano M, Lattanzio V. A reproducible, rapid and inexpensive Folin-Ciocalteu micro-method in determining phenolics of plant methanol extracts. Microchem J. 2009;91:107–110. doi: 10.1016/j.microc.2008.08.011. [DOI] [Google Scholar]
- 39.Zeng Z, et al. Synchronous determination with double-wavelength by RP-HPLC-UV and optimization of ultrasound-assisted extraction of phenolic acids from Caragana species using response surface methodology. Journal of Pharmaceutical & Biomedical Analysis. 2017;140:182. doi: 10.1016/j.jpba.2017.03.017. [DOI] [PubMed] [Google Scholar]
- 40.Aneta W, Jan O, Piotr L. Polyphenolic compounds and antioxidant activity of new and old apple varieties. Journal of Agricultural & Food Chemistry. 2008;56:6520–6530. doi: 10.1021/jf800510j. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated and analyzed in the current study are included in the main text and the raw data are available from the corresponding author.