Abstract
Kidney disease is a global public health problem, affecting over 750 million persons worldwide. The burden of kidney disease varies substantially across the world, as does its detection and treatment. In many settings, rates of kidney disease and the provision of its care are defined by socio-economic, cultural, and political factors leading to significant disparities. World Kidney Day 2019 offers an opportunity to raise awareness of kidney disease and highlight disparities in its burden and current state of global capacity for prevention and management. Here, we highlight that many countries still lack access to basic diagnostics, a trained nephrology workforce, universal access to primary health care, and renal replacement therapies. We point to the need for strengthening basic infrastructure for kidney care services for early detection and management of acute kidney injury and chronic kidney disease across all countries and advocate for more pragmatic approaches to providing renal replacement therapies. Achieving universal health coverage worldwide by 2030 is one of the World Health Organization's Sustainable Development Goals. While universal health coverage may not include all elements of kidney care in all countries, understanding what is feasible and important for a country or region with a focus on reducing the burden and consequences of kidney disease would be an important step towards achieving kidney health equity.
Keywords: Acute Kidney Injury, End Stage Renal Disease, Global Health, Health Equity, Social Determinants of Health
Resumo
A doença renal é um problema de saúde pública global, afetando mais de 750 milhões de pessoas em todo o mundo. O ônus da doença renal varia substancialmente em todo o mundo, assim como sua detecção e tratamento. Em muitos contextos, as taxas de doença renal e a provisão de seus cuidados são definidas por fatores socioeconômicos, culturais e políticos que levam a disparidades significativas. O Dia Mundial do Rim 2019 oferece uma oportunidade para aumentar a conscientização sobre doenças renais e destacar as disparidades em seu impacto e estado atual da capacidade global de prevenção e tratamento. Aqui, destacamos que muitos países ainda carecem de acesso a diagnósticos básicos, uma força de trabalho treinada em nefrologia, acesso universal à atenção primária à saúde e terapias de substituição renal. Apontamos para a necessidade de fortalecer a infra-estrutura básica para serviços de cuidados renais para detecção e tratamento precoce de lesão renal aguda e doença renal crônica em todos os países e defender abordagens mais pragmáticas para o fornecimento de terapias de substituição renal. Alcançar a cobertura universal de saúde em todo o mundo até 2030 é um dos Objetivos de Desenvolvimento Sustentável da Organização Mundial da Saúde. Embora a cobertura universal de saúde não inclua todos os elementos do tratamento renal em todos os países, entender o que é viável e importante para um país ou região com foco na redução do impacto e das consequências da doença renal seria um passo importante para alcançar a equidade na saúde renal.
Palavras-chave: Lesão Renal Aguda, Doença renal em estágio final, Saúde global, Equidade em Saúde, Determinantes Sociais da Saúde
INTRODUCTION
Kidney disease is a global public health problem, affecting over 750 million persons worldwide.1 , 2 The burden of kidney disease varies substantially across the world, as does its detection and treatment. While the magnitude and impact of kidney disease is better defined in developed countries, emerging evidence suggests developing countries have similar or even greater kidney disease burden.3
In many settings, rates of kidney disease and the provision of its care are defined by socioeconomic, cultural, and political factors leading to significant disparities in disease burden, even in developed countries.4 - 6 These disparities exist across the spectrum of kidney disease - from preventive efforts to curb development of acute kidney injury (AKI) or chronic kidney disease (CKD), to screening for kidney disease among persons at high risk, to access to subspecialty care and treatment of kidney failure with renal replacement therapy (RRT). World Kidney Day 2019 offers an opportunity to raise awareness of kidney disease and highlight disparities in its burden and current state of global capacity for prevention and management. In this editorial, we highlight these disparities and emphasize the role of public policies and organizational structures in addressing them. We outline opportunities to improve our understanding of disparities in kidney disease, how best they can be addressed, and how to streamline efforts towards achieving kidney health equity across the globe.
BURDEN OF KIDNEY DISEASE
Availability of data reflecting the full burden of kidney disease varies substantially due to limited or inconsistent data collection and surveillance practices worldwide (Table 1).7 While several countries have national data collection systems, particularly for end stage renal disease (ESRD) (e.g. United States Renal Data System,8 Latin American Dialysis and Renal Transplant Registry,9 and Australia and New Zealand Dialysis and Transplant Registry10), high quality data on non-dialysis CKD is limited,11 and often the quality of ESRD data is quite variable across settings. This is of particular concern in low-income countries. For example, a meta-analysis of 90 studies on CKD burden conducted across Africa showed very few (only 3%) with robust data.12 The provision of adequate resources and workforce to establish and maintain surveillance systems (screening programs and registries) is essential and requires substantial investment.13 Incorporating kidney disease surveillance parameters in existing chronic disease prevention programs might enhance global efforts towards high quality information on kidney disease burden and attendant consequences.14
Table 1. World Bank Country Group Chronic Kidney Disease Gaps.
| CKD+ Care | Low-Income Countries (%) | Lower-Middle-Income Countries (%) | Upper Middle-Income Countries (%) | High-Income Countries (%) |
|---|---|---|---|---|
| Governmental recognition of CKD as a health priority | 59 | 50 | 17 | 29 |
| Government funds all aspects of CKD care | 13 | 21 | 40 | 53 |
| Availability of CKD management and referral guidelines (international, national, or regional) | 46 | 73 | 83 | 97 |
| Existence of current CKD detection programs | 6 | 24 | 24 | 32 |
| Availability of dialysis registries | 24 | 48 | 72 | 89 |
| Availability of academic centers for renal clinical trial management | 12 | 34 | 62 | 63 |
+CKD: chronic kidney disease.
Source:7
In addition to a need for functional surveillance systems, the global importance of kidney disease (including AKI and CKD) is yet to be widely acknowledged, making it a neglected disease on the global policy agenda. For instance, the World Health Organization (WHO) Global Action Plan for the Prevention and Control of Non-Communicable Diseases (NCDs) (2013)15 focuses on cardiovascular diseases, cancer, chronic respiratory diseases, and diabetes, but not kidney disease despite advocacy efforts by relevant stakeholders such as the International Society of Nephrology (ISN) and the International Federation of Kidney Foundations through activities like World Kidney Day. This is quite concerning as estimates from the Global Burden of Disease study in 2015 showed that around 1.2 million people were known to have died of CKD,16 and over 2 million people died in 2010 because they had no access to dialysis. It is estimated that another 1.7 million die from AKI on an annual basis.17 , 18 It is possible, therefore, that kidney disease may contribute to more deaths than the four main NCDs targeted by the current NCD Action Plan.
Risk factors for kidney disease
Data over the recent decades have linked a host of genetic, environmental, socio-demographic and clinical factors to risk of kidney disease. The population burden of kidney disease is known to correlate with socially-defined factors in most societies across the world. This is better documented in high-income countries, where racial/ethnic minority groups and people of low socioeconomic status carry a high burden of disease. Extensive data has demonstrated that racial and ethnic minorities (e.g. African-Americans in the United States, Aboriginal groups in Canada and Australia, Indo-Asians in the United Kingdom, and others) suffer disproportionally from advanced and progressive kidney disease.19 - 21 The associations of socioeconomic status and risk of progressive CKD and eventual kidney failure have also been well described with lower socioeconomic status individuals bearing the greatest burden.22 , 23
Recent works have associated apolipoprotein L1 (APOL1) risk variants24 , 25 with increased kidney disease burden among persons with African ancestry. In Central America and Southeastern Mexico, Mesoamerican nephropathy [also referred to as CKD of unknown causes (CKDu)] has emerged as an important etiology of kidney disease. While multiple exposures have been studied for their potential role in CKDu, recurrent dehydration and heat stress are common denominators in most cases.26 Other, perhaps more readily modifiable, risk factors for kidney disease and CKD progression that disproportionally impact socially disadvantaged groups have also been identified including disparate rates and poor control of clinical risk factors such as diabetes and hypertension, as well as lifestyle behaviours.
Diabetes is the leading cause of advanced kidney disease worldwide.27 In 2016, 1 in 11 adults worldwide had diabetes and more than 80% were living in low- and middle-income countries28 where resources for optimal care are limited. Hypertension is also estimated to affect 1 billion individuals worldwide,29 and is the second leading attributed cause of CKD.27 Hypertension control is important for slowing CKD progression as well as decreasing mortality risk among persons with or without CKD. Hypertension is present in over 90% of individuals with advanced kidney disease,8 , 27 yet racial/ethnic minorities and low-income persons with CKD living in high-income countries have poorer blood pressure control than their more socially advantaged counterparts.30
Lifestyle behaviours, including dietary patterns, are strongly influenced by socioeconomic status. In recent years, several healthful dietary patterns have been associated with favorable CKD outcomes.31 Low-income individuals often face barriers to healthful eating which may increase their risk of kidney disease.32 - 34 People of low socioeconomic status often experience food insecurity (limited access to affordable nutritious foods), which is a risk factor for CKD35 and progression to kidney failure.36 In low-income countries, food insecurity may lead to undernutrition and starvation,37 , 38 which has implications for both the individual, and in the case of women of child-bearing age, could lead to their children having low birth weight and related sequelae, including CKD.39 Rates of undernourishment are as high as 35% or more in countries such as Haiti, Namibia, and Zambia.40 However, in high-income countries, food insecurity is associated with overnutrition, and food insecure persons have increased risk of overweight and obesity.41 , 42 Further, food insecurity has been associated with several diet-related conditions, including diabetes and hypertension.43 , 44
Acute kidney injury
AKI is an under-detected condition that is estimated to occur in 8-16% of hospital admissions45 and is now well-established as a risk factor for CKD.46 Disparities in AKI risk are also common, following a similar pattern to those observed in CKD.47 AKI related to nephrotoxins, alternative (traditional) medicines, infectious agents, as well as hospitalizations and related procedures are more pronounced in low- and lower-middle-income countries and contribute to increased risk of mortality and CKD in those settings.48 Importantly, the majority (85% of more than 13 million) of annual AKI cases worldwide are experienced in low- and lower-middle-income countries leading to 1.4 million deaths.49
HEALTH POLICIES AND FINANCING OF KIDNEY DISEASE CARE
Due to the complex and costly nature of kidney disease care, its provision is tightly linked with the public policies and financial status of individual countries. For example, gross domestic product is correlated with lower dialysis-to-transplantation ratios, suggesting greater rates of kidney transplantation in more financially solvent nations.2 In several high-income countries, universal health care is provided by the government and includes CKD and ESRD care. In others, such as the United States, ESRD care is publicly financed for citizens, however, optimal treatment of CKD and its risk factors may not be accessible for persons lacking health insurance, and regular care of undocumented immigrants with kidney disease is not covered.50 In low- and lower-middle-income countries, neither CKD nor ESRD care may be publicly financed and CKD prevention efforts are often limited. In several such countries, collaborations between public and private sectors have emerged to provide funding for RRT. For example, in Karachi, Pakistan, a program of dialysis and kidney transplantation through joint community and government funding has existed for more than 25 years.51
In many settings, individuals with advanced CKD who have no or limited public or private sector funding for care shoulder substantial financial burden. A systematic review of 260 studies including patients from 30 countries identified significant challenges including: fragmented care of indeterminate duration, reliance on emergency care, and fear of catastrophic life events because of diminished financial capacity to withstand them.52 Another study, conducted in Mexico, found that patients and families were burdened with having to navigate multiple health and social care structures, negotiate treatments and costs, finance their health care, and manage health information.53 Challenges may be even greater for families of children with ESRD, as many regions lack qualified paediatric care centers.54
ORGANIZATION AND STRUCTURES FOR KIDNEY DISEASE CARE
The lack of recognition and therefore absence of a global action plan for kidney disease partly explains the substantial variation in structures and capacity for kidney care around the globe. This has resulted in variations in government priorities, healthcare budgets, care structures, and human resource availability.55 Effective and sustainable advocacy efforts are needed at global, regional, and national levels to get kidney disease recognized and placed on the global policy agenda.
In 2017, the ISN collected data on country-level capacity for kidney care delivery using a survey, the Global Kidney Health Atlas (GKHA),56 which aligned with the WHO's building blocks of a health system.57 The GKHA highlights limited awareness of kidney disease and its consequences and persistent inequities in resources required to tackle the burden of kidney disease across the globe. For example, CKD was recognized as a health care priority by government in only 36% of countries that participated in this survey. The priority was inversely related to income level: CKD was a health care priority in more than half of low- and lower-middle-income countries, but in less than 30% of upper-middle- and high-income countries.
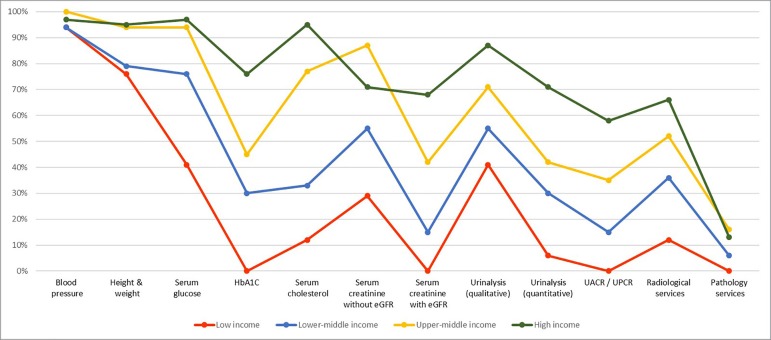
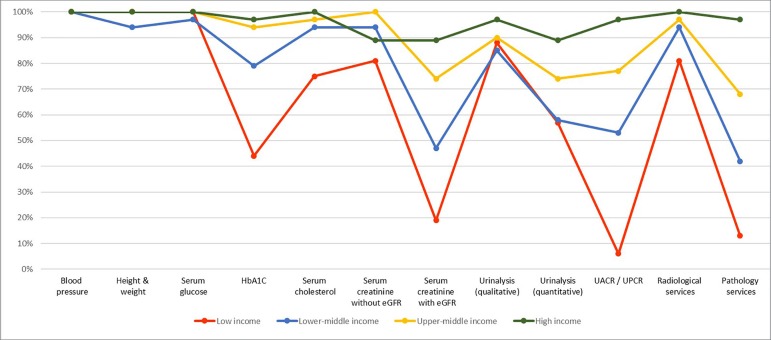
On capacity and resources for kidney care, many countries still lack access to basic diagnostics, a trained nephrology workforce, universal access to primary health care, and RRT technologies. Low- and lower-middle-income countries, especially in Africa, had limited services for the diagnosis, management, and monitoring of CKD at the primary care level, with only 12% having serum creatinine measurement including estimated glomerular filtration rate. Twenty-nine percent of low-income countries had access to qualitative urinalysis using urine test strips; however, no low-income country had access to urine albumin-to-creatinine ratio or urine protein-to-creatinine ratio measurements at the primary care level. Across all world countries, availability of services at the secondary/tertiary care level was considerably higher than at the primary care level (Figures 1 and 2).7 , 58
Figure 1. Health Care Services for Identification and Management of Chronic Kidney Disease by Country Income Level: Primary Care.
HbA1C: Glycated hemoglobin; eGFR: estimated glomerular filtration rate; UACR: urine albumin-to-creatinine ratio; UPCR: urine protein-to-creatinine ratio. Primary care = Basic health facilities at community levels (clinics, dispensaries, small local hospitals).
Figure 2. Health Care Services for Identification and Management of Chronic Kidney Disease by Country Income Level: Secondary/Specialty Care.
HbA1C: Glycated hemoglobin; eGFR: estimated glomerular filtration rate; UACR: urine albumin-to-creatinine ratio; UPCR: urine protein-to-creatinine ratio. Secondary care/Specialty care = Health facilities at a level higher than primary care (clinics, hospitals, academic centers).
Renal replacement therapies
The distribution of RRT technologies varied widely. On the surface, all countries reported having chronic hemodialysis services, and more than 90% of countries reported having acute hemodialysis. However, access to and distribution of RRT across countries and regions was highly inequitable, often requiring prohibitive out-of-pocket expenditure, particularly in low-income regions. For instance, more than 90% of upper-middle- and high-income countries reported having chronic peritoneal dialysis services, whereas these services were available in 64% and 35% of low- and lower-middle-income countries, respectively. In comparison, acute peritoneal dialysis had the lowest availability across all countries. More than 90% of upper-middle- and high-income countries reported having kidney transplant services, with more than 85% of these countries reporting both living and deceased donors as the organ source. As expected, low-income countries had the lowest availability of kidney transplant services, with only 12% reporting availability, and live donors as the only source.
Workforce for kidney care
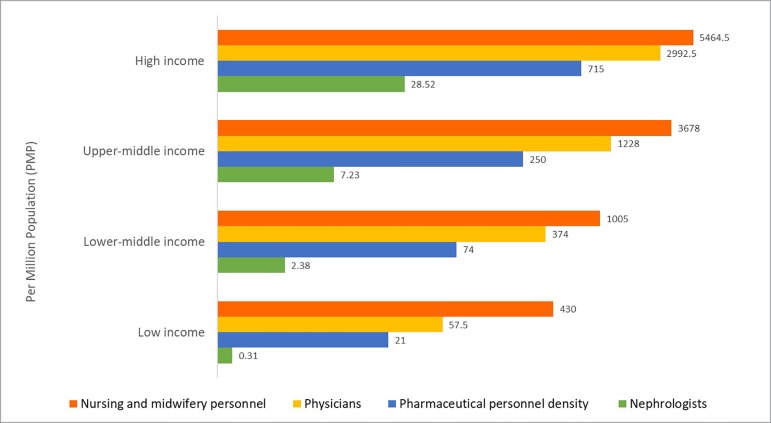
Considerable international variation was also noted in the distribution of the kidney care workforce, particularly nephrologists. The lowest density (<5 nephrologists per million population) was very common in low-income countries, whereas the highest density (>15 nephrologists per million population) was reported mainly in high-income countries (Figure 3).7 , 59 , 60 Most countries reported nephrologists as primarily responsible for both CKD and AKI care. Primary care physicians (PCPs) had more responsibility for CKD care than for AKI care, as 64% of countries reported PCPs primarily responsible for CKD and 35% for AKI. Intensive care specialists were primarily responsible for AKI in 75% of countries, likely because AKI is typically treated in hospitals. However, only 45% of low-income countries reported that intensive care specialists were primarily responsible for AKI, compared to 90% of high-income countries; this discrepancy may be due to a general shortage of intensive care specialists in low-income countries.
Figure 3. Nephrologist Availability (density per million population) compared to Physician, Nursing, and Pharmaceutical Personnel Availability by Country Income Level*.
* Logarithmic scale used for x-axis [log(x+1)] due to the large range in provider's density
Pharmaceutical personnel include: pharmacists, pharmaceutical assistants, pharmaceutical technicians. Nursing and midwifery personnel include: professional nurses, professional midwives, auxiliary nurses, auxiliary midwives, enrolled nurses, enrolled midwives and related occupations such as dental nurses.
Source: Nephrologists: Global Kidney Health Atlas (2016); Physician, nursing and pharmaceutical personnel: WHO Global Health Observatory Indicator (most recent available year)7,59 , http://apps.who.int/gho/data/node.main-amro.HWF?lang=en60
The appropriate number of nephrologists in a country depends on many factors, including need, priority, and resources, and as such there is no global standard with respect to nephrologist density. Regardless, the demonstrated low density in low-income countries calls for concern as nephrologists are essential to provide leadership in kidney disease care, and a lack of nephrologists may result in adverse consequences for policy and practice. But quite encouragingly, the number of nephrologists and nephropathologists is rising in low- and lower-middle-income countries, in part thanks to fellowship programs supported by international nephrology organizations.61 , 62 It is important to note that the role of a nephrologist may differ depending on how the health care system is structured. The density statistic merely represents the number of nephrologists per million population and provides no indication of the adequacy to meet the needs of the population or quality of care, which depends on volume of patients with kidney disease and other workforce support (for example, availability of multidisciplinary teams).
For other care providers essential for kidney care, there were international variations in distribution (availability and adequacy). Overall, provider shortages were highest for renal pathologists (86% of countries reported a shortage), vascular access coordinators (81%), and dietitians (78%), and the shortages were more common in low-income countries. Few countries (35%) reported a shortage in laboratory technicians. This information highlights significant inter- and intra-regional variability in the current capacity for kidney care across the world. Important gaps in awareness, services, workforce, and capacity for optimal care delivery were identified in many countries and regions.7 The findings have implications for policy development towards establishment of robust kidney care programs, particularly for low- and lower-middle-income countries.63 This has therefore provided a baseline understanding of where countries and regions stand with respect to several domains of the health system, thus allowing the monitoring of progress through the implementation of various strategies at achieving equitable and quality care for the many patients with kidney disease across the globe.
How could this information be used to mitigate existing barriers to kidney care? First, basic infrastructure for services must be strengthened at the primary care level for early detection and management of AKI and CKD across all countries.63 Second, whilst optimal kidney care should obviously emphasize prevention to reduce adverse consequences of kidney disease at the population level, countries (particularly low- and lower-middle-income countries) should be supported at the same time to adopt more pragmatic approaches in providing RRT. For example, acute peritoneal dialysis could be an attractive modality for AKI, as this is as effective as hemodialysis, requires far less infrastructure, and can be performed with solutions and catheters adapted to local resources.64 , 65 Third, kidney transplantation should be encouraged through increased awareness among the public and political leaders across countries, as this is the clinically optimal modality of RRT and it is also cost-effective, provided that costs of the surgery and long-term medication and follow-up are made sustainable through public (and/or private) funding.66 Currently, most kidney transplants are conducted in high-income countries in part due to lack of resources and knowledge in low and lower-middle-income countries as well as cultural practices and absence of legal frameworks governing organ donation.66
CONCLUSION
Socially disadvantaged individuals experience a disproportionate burden of kidney disease worldwide. The provision and delivery of kidney care varies widely across the world. Achieving universal health coverage worldwide by 2030 is one of the WHO Sustainable Development Goals.67 While universal health coverage may not include all elements of kidney care in all countries (as this is usually a function of political, economic, and cultural factors), understanding what is feasible and important for a country or region with a focus on reducing the burden and consequences of kidney disease would be an important step towards achieving kidney health equity.
ACKNOWLEDGMENTS
The authors thank the Global Kidney Health Atlas team, M. Lunney, and M.A. Osman.
Footnotes
This article was published in Kidney International, volume 95, pages 242-248, Copyright World Kidney Day 2019, Steering Committee (2019).
REFERENCES
- 1.GBD 2015 DALYs and HALE Collaborators Global, regional, and national disability-adjusted life-years (DALYs) for 315 diseases and injuries and healthy life expectancy (HALE), 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet. 2016;388:1603–1658. doi: 10.1016/S0140-6736(16)31460-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bikbov B, Perico N, Remuzzi G, GBD Genitourinary Diseases Expert Group Disparities in Chronic Kidney Disease Prevalence among Males and Females in 195 Countries: Analysis of the Global Burden of Disease 2016 Study. Nephron. 2018;139:313–318. doi: 10.1159/000489897. [DOI] [PubMed] [Google Scholar]
- 3.Hill NR, Fatoba ST, Oke JL, Hirst JA, O'Callaghan CA, Lasserson DS, et al. Global Prevalence of Chronic Kidney Disease - A Systematic Review and Meta-Analysis. PLoS One. 2016;11:e0158765. doi: 10.1371/journal.pone.0158765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Crews DC, Pfaff T, Powe NR. Socioeconomic factors and racial disparities in kidney disease outcomes. Semin Nephrol. 2013;33:468–475. doi: 10.1016/j.semnephrol.2013.07.008. [DOI] [PubMed] [Google Scholar]
- 5.Nicholas SB, Kalantar-Zadeh K, Norris KC. Socioeconomic disparities in chronic kidney disease. Adv Chronic Kidney Dis. 2015;22:6–15. doi: 10.1053/j.ackd.2014.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Crews DC, Liu Y, Boulware LE. Disparities in the burden, outcomes, and care of chronic kidney disease. Curr Opin Nephrol Hypertens. 2014;23:298–305. doi: 10.1097/01.mnh.0000444822.25991.f6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bello AK, Levin A, Tonelli M, Okpechi IG, Feehally J, Harris D, et al. Assessment of Global Kidney Health Care Status. JAMA. 2017;317:1864–1881. doi: 10.1001/jama.2017.4046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.United States Renal Data System 2017 USRDS annual data report: Epidemiology of kidney disease in the United States. 2017. [2018 Aug 22]. [Internet] Available from: https://www.usrds.org/adr.aspx. [DOI] [PMC free article] [PubMed]
- 9.Cusumano AM, Gonzalez Bedat MC, García-García G, Maury Fernandez S, Lugon JR, Poblete Badal H, et al. Latin American Dialysis and Renal Transplant Registry: 2008 report (data 2006) Clin Nephrol. 2010;74:3–8. [PubMed] [Google Scholar]
- 10.ANZDATA Registry 40th Report, Chapter 2: Prevalence of End Stage Kidney Disease. 2018. [2018 Aug 22]. [Internet] Available from: http://www.anzdata.org.au.
- 11.Centers for Disease Control and Prevention - CDC Chronic Kidney Disease Surveillance System-United States. 2018. [2018 Aug 22]. [Internet] Available from: https://nccd.cdc.gov/ckd/default.aspx.
- 12.Stanifer JW, Jing B, Tolan S, Helmke N, Mukerjee R, Naicker S, et al. The epidemiology of chronic kidney disease in sub-Saharan Africa: a systematic review and meta-analysis. Lancet Glob Health. 2014;2:e174–e181. doi: 10.1016/S2214-109X(14)70002-6. [DOI] [PubMed] [Google Scholar]
- 13.Davids MR, Eastwood JB, Selwood NH, Arogundade FA, Ashuntantang G, Benghanem Gharbi M, et al. A renal registry for Africa: first steps. Clin Kidney J. 2016;9:162–167. doi: 10.1093/ckj/sfv122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Levin A, Tonelli M, Bonventre J, Coresh J, Donner JA, Fogo AB, et al. ISN Global Kidney Health Summit participants Global kidney health 2017 and beyond: a roadmap for closing gaps in care, research, and policy. Lancet. 2017;390:1888–1917. doi: 10.1016/S0140-6736(17)30788-2. [DOI] [PubMed] [Google Scholar]
- 15.World Health Organization - WHO Global Action Plan for the prevention and control of noncommunicable diseases. 2013-2020. 2013. [2018 Aug 22]. [Internet] Available from: https://www.who.int/nmh/events/ncd_action_plan/en/
- 16.GBD 2015 Mortality and Causes of Death Collaborators Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980-2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet. 2016;388:1459–1544. doi: 10.1016/S0140-6736(16)31012-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Liyanage T, Ninomiya T, Jha V, Neal B, Patrice HM, Okpechi I, et al. Worldwide access to treatment for end-stage kidney disease: a systematic review. Lancet. 2015;385:1975–1982. doi: 10.1016/S0140-6736(14)61601-9. [DOI] [PubMed] [Google Scholar]
- 18.Mehta RL, Cerdá J, Burdmann EA, Tonelli M, García-García G, Jha V, et al. International Society of Nephrology's 0by25 initiative for acute kidney injury (zero preventable deaths by 2025): a human rights case for nephrology. Lancet. 2015;385:2616–2643. doi: 10.1016/S0140-6736(15)60126-X. [DOI] [PubMed] [Google Scholar]
- 19.Samuel SM, Palacios-Derflingher L, Tonelli M, Manns B, Crowshoe L, Ahmed SB, et al. Association between First Nations ethnicity and progression to kidney failure by presence and severity of albuminuria. CMAJ. 2014;186:E86–E94. doi: 10.1503/cmaj.130776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Nicholas SB, Kalantar-Zadeh K, Norris KC. Racial disparities in kidney disease outcomes. Semin Nephrol. 2013;33:409–415. doi: 10.1016/j.semnephrol.2013.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.van den Beukel TO, de Goeij MC, Dekker FW, Siegert CE, Halbesma N, PREPARE Study Group Differences in progression to ESRD between black and white patients receiving predialysis care in a universal health care system. Clin J Am Soc Nephrol. 2013;8:1540–1547. doi: 10.2215/CJN.10761012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Crews DC, Gutiérrez OM, Fedewa SA, Luthi JC, Shoham D, Judd SE, et al. Low income, community poverty and risk of end stage renal disease. BMC Nephrol. 2014;15:192–192. doi: 10.1186/1471-2369-15-192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Garrity BH, Kramer H, Vellanki K, Leehey D, Brown J, Shoham DA. Time trends in the association of ESRD incidence with area-level poverty in the US population. Hemodial Int. 2016;20:78–83. doi: 10.1111/hdi.12325. [DOI] [PubMed] [Google Scholar]
- 24.Parsa A, Kao WH, Xie D, Astor BC, Li M, Hsu CY, et al. AASK Study InvestigatorsCRIC Study Investigators APOL1 risk variants, race, and progression of chronic kidney disease. N Engl J Med. 2013;369:2183–2196. doi: 10.1056/NEJMoa1310345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Peralta CA, Bibbins-Domingo K, Vittinghoff E, Lin F, Fornage M, Kopp JB, et al. APOL1 Genotype and Race Differences in Incident Albuminuria and Renal Function Decline. J Am Soc Nephrol. 2016;27:887–893. doi: 10.1681/ASN.2015020124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Correa-Rotter R. Mesoamerican Nephropathy or Chronic Kidney Disease of Unknown Origin. In: García-García G, Agodoa L, Norris K, editors. Chronic Kidney Disease in Disadvantaged Populations. San Diego: Academic Press; 2017. pp. 221–228. [Google Scholar]
- 27.Kidney Disease: Improving Global Outcomes (KDIGO) KDIGO 2012 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney Int Suppl. 2013;3:1–150. doi: 10.1038/ki.2013.243. [DOI] [PubMed] [Google Scholar]
- 28.Chan JC, Gregg EW, Sargent J, Horton R. Reducing global diabetes burden by implementing solutions and identifying gaps: a Lancet Commission. Lancet. 2016;387:1494–1495. doi: 10.1016/S0140-6736(16)30165-9. [DOI] [PubMed] [Google Scholar]
- 29.Kearney PM, Whelton M, Reynolds K, Muntner P, Whelton PK, He J. Global burden of hypertension: analysis of worldwide data. Lancet. 2005;365:217–223. doi: 10.1016/S0140-6736(05)17741-1. [DOI] [PubMed] [Google Scholar]
- 30.Plantinga LC, Miller 3rd ER, Stevens LA, Saran R, Messer K, Flowers N, et al. Centers for Disease Control and Prevention Chronic Kidney Disease Surveillance Team Blood pressure control among persons without and with chronic kidney disease: US trends and risk factors 1999-2006. Hypertension. 2009;54:47–56. doi: 10.1161/HYPERTENSIONAHA.109.129841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Banerjee T, Liu Y, Crews DC. Dietary Patterns and CKD Progression. Blood Purif. 2016;41:117–122. doi: 10.1159/000441072. [DOI] [PubMed] [Google Scholar]
- 32.Johnson AE, Boulware LE, Anderson CA, Chit-ua-aree T, Kahan K, Boyér LL, et al. Perceived barriers and facilitators of using dietary modification for CKD prevention among African Americans of low socioeconomic status: a qualitative study. BMC Nephrol. 2014;15:194–194. doi: 10.1186/1471-2369-15-194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Crews DC, Kuczmarski MF, Miller 3rd ER, Zonderman AB, Evans MK, Powe NR. Dietary habits, poverty, and chronic kidney disease in an urban population. J Ren Nutr. 2015;25:103–110. doi: 10.1053/j.jrn.2014.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Suarez JJ, Isakova T, Anderson CA, Boulware LE, Wolf M, Scialla JJ. Food Access, Chronic Kidney Disease, and Hypertension in the U.S. Am J Prev Med. 2015;49:912–920. doi: 10.1016/j.amepre.2015.07.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Crews DC, Kuczmarski MF, Grubbs V, Hedgeman E, Shahinian VB, Evans MK, et al. Centers for Disease Control and Prevention Chronic Kidney Disease Surveillance Team Effect of food insecurity on chronic kidney disease in lower-income Americans. Am J Nephrol. 2014;39:27–35. doi: 10.1159/000357595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Banerjee T, Crews DC, Wesson DE, Dharmarajan S, Saran R, Ríos Burrows N, et al. CDC CKD Surveillance Team Food Insecurity, CKD, and Subsequent ESRD in US Adults. Am J Kidney Dis. 2017;70:38–47. doi: 10.1053/j.ajkd.2016.10.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Pasricha SR, Biggs BA. Undernutrition among children in South and South-East Asia. J Paediatr Child Health. 2010;46:497–503. doi: 10.1111/j.1440-1754.2010.01839.x. [DOI] [PubMed] [Google Scholar]
- 38.Tanumihardjo SA, Anderson C, Kaufer-Horwitz M, Bode L, Emenaker NJ, Haqq AM, et al. Poverty, obesity, and malnutrition: an international perspective recognizing the paradox. J Am Diet Assoc. 2007;107:1966–1972. doi: 10.1016/j.jada.2007.08.007. [DOI] [PubMed] [Google Scholar]
- 39.Piccoli GB, Alrukhaimi M, Liu ZH, Zakharova E, Levin A, World Kidney Day Steering Committee Women and kidney disease: reflections on World Kidney Day 2018. Kidney Int. 2018;93:278–283. doi: 10.1016/j.kint.2017.11.008. [DOI] [PubMed] [Google Scholar]
- 40.Food and Agriculture Organization of the United Nations The FAO Hunger Map 2015. 2015. [2018 Aug 22]. [Internet] Available from: http://www.fao.org/3/a-i4674e.pdf.
- 41.Shariff ZM, Khor GL. Obesity and household food insecurity: evidence from a sample of rural households in Malaysia. Eur J Clin Nutr. 2005;59:1049–1058. doi: 10.1038/sj.ejcn.1602210. [DOI] [PubMed] [Google Scholar]
- 42.Popkin BM. Contemporary nutritional transition: determinants of diet and its impact on body composition. Proc Nutr Soc. 2011;70:82–91. doi: 10.1017/S0029665110003903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Seligman HK, Laraia BA, Kushel MB. Food insecurity is associated with chronic disease among low-income NHANES participants. J Nutr. 2010;140:304–310. doi: 10.3945/jn.109.112573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Castillo DC, Ramsey NL, Yu SS, Ricks M, Courville AB, Sumner AE. Inconsistent Access to Food and Cardiometabolic Disease: The Effect of Food Insecurity. Curr Cardiovasc Risk Rep. 2012;6:245–250. doi: 10.1007/s12170-012-0236-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Sawhney S, Marks A, Fluck N, Levin A, Prescott G, Black C. Intermediate and Long-term Outcomes of Survivors of Acute Kidney Injury Episodes: A Large Population-Based Cohort Study. Am J Kidney Dis. 2017;69:18–28. doi: 10.1053/j.ajkd.2016.05.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Heung M, Steffick DE, Zivin K, Gillespie BW, Banerjee T, Hsu CY, et al. Centers for Disease Control and Prevention CKD Surveillance Team Acute Kidney Injury Recovery Pattern and Subsequent Risk of CKD: An Analysis of Veterans Health Administration Data. Am J Kidney Dis. 2016;67:742–752. doi: 10.1053/j.ajkd.2015.10.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Grams ME, Matsushita K, Sang Y, Estrella MM, Foster MC, Tin A, et al. Explaining the racial difference in AKI incidence. J Am Soc Nephrol. 2014;25:1834–1841. doi: 10.1681/ASN.2013080867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Luyckx VA, Tuttle KR, Garcia-Garcia G, Gharbi MB, Heerspink HJL, Johnson DW, et al. Reducing major risk factors for chronic kidney disease. Kidney Int Suppl. 2017;7:71–87. doi: 10.1016/j.kisu.2017.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lewington AJ, Cerda J, Mehta RL. Raising awareness of acute kidney injury: a global perspective of a silent killer. Kidney Int. 2013;84:457–467. doi: 10.1038/ki.2013.153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Cervantes L, Tuot D, Raghavan R, Linas S, Zoucha J, Sweeney L, et al. Association of Emergency-Only vs Standard Hemodialysis With Mortality and Health Care Use Among Undocumented Immigrants With End-stage Renal Disease. JAMA Intern Med. 2018;178:188–195. doi: 10.1001/jamainternmed.2017.7039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Rizvi SA, Naqvi SA, Zafar MN, Akhtar SF. A kidney transplantation model in a low-resource country: an experience from Pakistan. Kidney Int Suppl. 2013;3:236–240. doi: 10.1038/kisup.2013.22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Roberti J, Cummings A, Myall M, Harvey J, Lippiett K, Hunt K, et al. Work of being an adult patient with chronic kidney disease: a systematic review of qualitative studies. BMJ Open. 2018;8:e023507. doi: 10.1136/bmjopen-2018-023507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Kierans C, Padilla-Altamira C, Garcia-Garcia G, Ibarra-Hernandez M, Mercado FJ. When health systems are barriers to health care: challenges faced by uninsured Mexican kidney patients. PLoS One. 2013;8:e54380. doi: 10.1371/journal.pone.0054380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Harambat J, Ekulu PM. Inequalities in access to paediatric ESRD care: a global health challenge. Pediatr Nephrol. 2016;31:353–358. doi: 10.1007/s00467-015-3263-7. [DOI] [PubMed] [Google Scholar]
- 55.Murray CJ, Frenk J. A framework for assessing the performance of health systems. Bull World Health Organ. 2000;78:717–731. [PMC free article] [PubMed] [Google Scholar]
- 56.Bello AK, Levin A, Tonelli M, Okpechi IG, Feehally J, Harris D, et al. Global Kidney Health Atlas: A report by the International Society of Nephrology on the current state of organization and structures for kidney care across the globe. Brussels: International Society of Nephrology. 2017. [2018 Jul 24]. [Internet] Available from: https://www.theisn.org/images/ISN_advocacy/GKHAtlas_Linked_Compressed1.pdf.
- 57.World Health Organization - WHO Monitoring the building blocks of health systems: a handbook of indicators and their measurement strategies. 2010. [2018 Aug 22]. [Internet] Available from: http://www.who.int/healthinfo/systems/WHO_MBHSS_2010_full_web.pdf.
- 58.Htay H, Alrukhaimi M, Ashuntantang GE, Bello AK, Bellorin-Font E, Benghanem Gharbi M, et al. Global access of patients with kidney disease to health technologies and medications: findings from the Global Kidney Health Atlas project. Kidney Int Suppl. 2018;8:64–73. doi: 10.1016/j.kisu.2017.10.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Osman MA, Alrukhaimi M, Ashuntantang GE, Bellorin-Font E, Benghanem Gharbi M, Braam B, et al. Global nephrology workforce: gaps and opportunities toward a sustainable kidney care system. Kidney Int Suppl. 2018;8:52–63. doi: 10.1016/j.kisu.2017.10.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.World Health Organization WHO Global Health Observatory indicator views. 2016. [2018 Jul 24]. [Internet] Available from: http://apps.who.int/gho/data/node.imr#ndx-P.
- 61.Feehally J. The International Society of Nephrology (ISN): Roles & challenges in Africa and other resource-limited communities. Clin Nephrol. 2016;86:3–7. doi: 10.5414/CNP86S101. [DOI] [PubMed] [Google Scholar]
- 62.Harris DC, Dupuis S, Couser WG, Feehally J. Training nephrologists from developing countries: does it have a positive impact? Kidney Int Suppl. 2012;2:275–278. doi: 10.1038/kisup.2012.32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Couser WG, Remuzzi G, Mendis S, Tonelli M. The contribution of chronic kidney disease to the global burden of major noncommunicable diseases. Kidney Int. 2011;80:1258–1270. doi: 10.1038/ki.2011.368. [DOI] [PubMed] [Google Scholar]
- 64.Cullis B, Abdelraheem M, Abrahams G, Balbi A, Cruz DN, Frishberg Y, et al. Peritoneal dialysis for acute kidney injury. Perit Dial Int. 2014;34:494–517. doi: 10.3747/pdi.2013.00222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Chionh CY, Soni SS, Finkelstein FO, Ronco C, Cruz DN. Use of peritoneal dialysis in AKI: a systematic review. Clin J Am Soc Nephrol. 2013;8:1649–1660. doi: 10.2215/CJN.01540213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Muralidharan A, White S. The need for kidney transplantation in low- and middle-income countries in 2012: an epidemiological perspective. Transplantation. 2015;99:476–481. doi: 10.1097/TP.0000000000000657. [DOI] [PubMed] [Google Scholar]
- 67.World Health Organization - WHO Universal health coverage (UHC) - fact sheet. 2017. [2017 Jul 24]. [Internet] Available from: http://www.who.int/news-room/fact-sheets/detail/universal-health-coverage-(uhc)