ABSTRACT
In 2012, Middle East respiratory syndrome coronavirus (MERS-CoV) emerged. To date, more than 2300 cases have been reported, with an approximate case fatality rate of 35%. Epidemiological investigations identified dromedary camels as the source of MERS-CoV zoonotic transmission and evidence of MERS-CoV circulation has been observed throughout the original range of distribution. Other new-world camelids, alpacas and llamas, are also susceptible to MERS-CoV infection. Currently, it is unknown whether Bactrian camels are susceptible to infection. The distribution of Bactrian camels overlaps partly with that of the dromedary camel in west and central Asia. The receptor for MERS-CoV, DPP4, of the Bactrian camel was 98.3% identical to the dromedary camel DPP4, and 100% identical for the 14 residues which interact with the MERS-CoV spike receptor. Upon intranasal inoculation with 107 plaque-forming units of MERS-CoV, animals developed a transient, primarily upper respiratory tract infection. Clinical signs of the MERS-CoV infection were benign, but shedding of large quantities of MERS-CoV from the URT was observed. These data are similar to infections reported with dromedary camel infections and indicate that Bactrians are susceptible to MERS-CoV and given their overlapping range are at risk of introduction and establishment of MERS-CoV within the Bactrian camel populations.
KEYWORDS: MERS-CoV, Bactrian camel, dromedary camel, virus shedding, natural reservoir
Introduction
Middle East respiratory syndrome coronavirus (MERS-CoV) circulates in dromedary camels and is responsible for severe respiratory disease in humans [1–9]. Experimental infections and field observations indicate that infected dromedaries shed large quantities of virus via nasal secretions and that signs of clinical disease are limited to rhinorrhea and a mild elevation in body temperature [10–12]. Infectious virus was detected in nasal swabs collected during the first week from experimentally infected dromedaries and RNA was detected for 35 days post-infection (dpi) [10,11,13,14]. In addition, productive infections after experimental inoculation with MERS-CoV of new world camelids, llama's and alpaca, have been reported suggesting a broad host tropism of MERS-CoV for camelids [13,15,16].
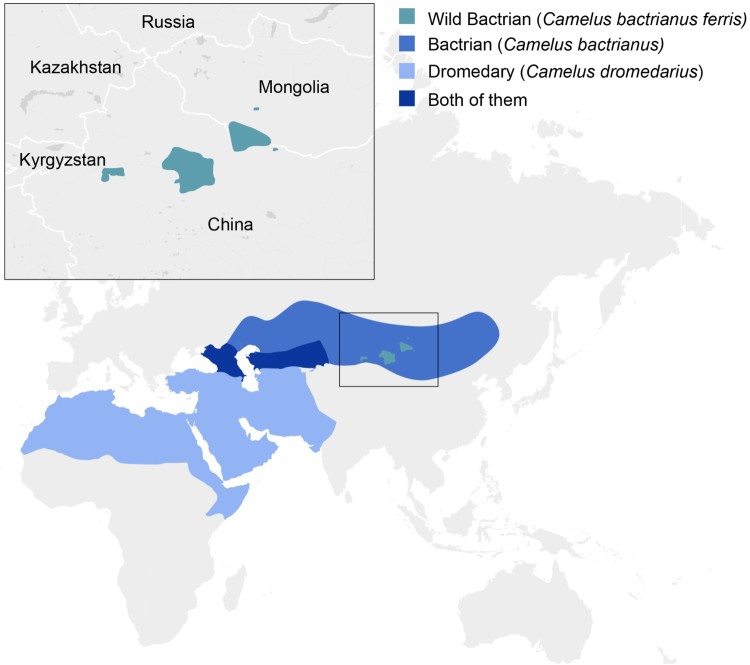
Old world camelids include dromedary and Bactrian camels. Bactrian camels consist of two subspecies: the critically endangered wild Bactrian camels (Camelus bactrianus ferus), and domesticated Bactrian camels (Camelus bactrianus bactrianus). They are distributed over Western, Central and South Asia, including an area that overlaps with the distribution of dromedary camels (Figure 1A) [17,18]. A 2014 survey of 30 Bactrian camels from 12 herds in southern Mongolia failed to detect circulation of MERS-CoV either by qRT-PCR on nasal swabs or serology, indicating either that the virus was not circulating in these herds or that these animals are not susceptible to infection [19]. Similarly, a 2015 survey of 190 Bactrian camels from 10 herds in the West Inner Mongolia Autonomous Region of China failed to detect circulation of MERS-CoV by qRT-PCR or serology [20]. Sero-surveillance conducted in Kazakhstan did not detected any evidence for circulation of MERS-CoV in 455 sampled dromedary camels and 95 Bactrian camels [21]. In order to determine the susceptibility of Bactrian camels to infection with MERS-CoV and the potential risk for introduction of MERS-CoV in areas with Bactrian camels, we inoculated two Bactrian camels and monitored them for nasal shedding and seroconversion and compared the response to experimental historic data from dromedary camels.
Figure 1.
Geographical distribution of Bactrian camels. Geographical distribution of dromedary camels, Bactrian camels and wild Bactrian camels, including the area in which dromedary camels and Bactrian camels are co-localized.
Materials and methods
Ethics Statement: All experiments were approved by the Colorado State University Institutional Animal Care and Use Committee. Work with infectious MERS-CoV strains under ABSL3 conditions was approved by the Institutional Biosafety Committee (IBC). Inactivation and removal of samples form high containment was preformed according to IBC-approved protocols.
Study design
Two intact adult male domesticated Bactrian camels were obtained through private sale and housed in an Animal Biosafety Level 3 facility for the entirety of the experiment. They had access to food and water ad libitum and were observed at least once daily. Each camel was sedated with xylazine and inoculated intranasally with MERS-CoV (strain HCoV-EMC/2012) diluted in phosphate-buffered saline for a total dose of 107 plaque-forming units (PFU) delivered in 3 mL per nare. Nasal swabs were collected from both camels daily 1–5 days post inoculation (dpi), and from Bactrian camel 1 on day 7, 14, 21 and 28. Serum samples were collected from both animals on day 0, and from Bactrian camel 1 on 7, 14, 21, and 28 dpi. Bactrian camel 2 was euthanized on 5 dpi and Bactrian camel 1 on 28 dpi; Bactrian camel 2 was necropsied and tissues processed for virus titration and histopathology.
RNA extraction and quantitative PCR
RNA was extracted from swabs, fecal samples and serum samples using the QiaAmp Viral RNA kit (Qiagen) according to the manufacturer's instructions. For detection of viral RNA, 5 μl of RNA was used in a one-step real-time RT–PCR upE assay using the Rotor-GeneTM probe kit (Qiagen) according to manufacturer's instructions [22]. Standard dilutions of a titered virus stock were run in parallel, to calculate TCID50 equivalents in the samples.
Virus titration and serology
Shedding and tissue distribution of MERS-CoV was evaluated by virus titration on Vero cells as described previously [10,13]. In short, swab samples in viral transport medium and homogenized tissues (∼10% w/v) were titrated for MERS-CoV virus by plaque assay. Briefly, ten-fold serial dilutions of samples were prepared in BA-1 medium containing 100 mg gentamicin, 200,000 U penicillin G, 100 mg streptomycin and 5 mg amphotericin/L, and 0.1 ml volumes were inoculated onto confluent monolayers of VeroE6 cells grown in six-well cell culture plates in duplicate. The inoculated cells were incubated for 45 min at 37°C in 5% CO2 in air and then overlaid with 2 ml/well of MEM without phenol red and containing 0.5% agarose, 2% fetal bovine serum and antibiotics as described above. Two days after the initial overlay, a second overlay was added which was identical to the first except for inclusion of neutral red (33 mg/L). Plaques were counted on days 1 and 3 after the second overlay and virus titers expressed as plaque-forming units (pfu) per ml.
Plaque reduction neutralization assay
Neutralizing antibodies were detected using a plaque reduction neutralization assay (PRNT) using 90% plaque reduction as previously described [10,13]. In short, camel sera were heat inactivated for 30 min at 56°C and two-fold dilutions beginning at 1:5 prepared in BA-1 medium. These samples were mixed with an equal volume of MERS-CoV to obtain a virus concentration of 100 pfu/0.1 ml and an initial serum dilution of 1:10. The virus-serum mixtures were incubated at 37°C for 60 min, then inoculated onto VeroE6 cells as described for plaque assay. Plaque reduction neutralization assay (PRNT) titers were calculated as the reciprocal of the highest dilution that resulted in >90% neutralization of virus relative to no serum control samples.
Histopathology and immunohistochemistry
Tissues were collected from Bactrian camel 2 and fixed for >7 days in 10% neutral-buffered formalin and embedded in paraffin using standard techniques. Histopathologic evaluation was performed on slides stained with hematoxylin and eosin. Other slides were deparaffinized and immunostained using a rabbit polyclonal antiserum to MERS-CoV (EMC/2012) as primary antibody. Slides were evaluated by a board-certified veterinary pathologist.
Results
Dipeptidyl peptidase 4 (DPP4) analysis
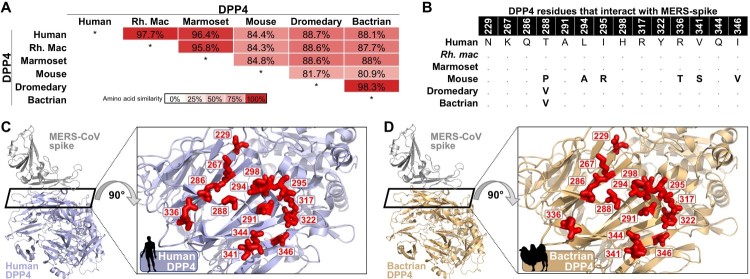
The host receptor of MERS-CoV, DPP4, was compared between Bactrian camels and species known to be susceptible (human, non-human primates and dromedary camels) and not susceptible (mice) [23]. Bactrian DPP4 was 98.3% identical to the dromedary camel DPP4 on the amino acid level, and only varied from human DPP4 by one amino acid in the residues that specifically interact with the MERS-CoV spike (Figure 1B and C). A modelled co-structure of MERS-CoV spike and Bactrian camel DPP4 showed that the interfacing amino acids were oriented similarly to the known co-crystal structure of MERS-CoV spike and human DPP4. The 100% identity of the 14 amino acid residues between dromedary camel and Bactrian camel DPP4 in the interaction regions indicates that MERS-CoV RBD should bind to Bactrian camel DPP4. This suggests that the MERS-CoV spike would be able to utilize Bactrian camel DPP4 (Figure 2D and E).
Figure 2.
Bactrian camel dipeptidyl peptidase 4 (DPP4) analyses. (A) DPP4 amino acid sequence identity matrix. White indicates no similarity, dark red indicates 100% similarity. (B) DPP4 species amino acid variation within the 14 described contact points with MERS-CoV spike. (C) Co-structure of MERS-CoV spike interaction with human DPP4 (PDB: 4L72). Inset shows a top-down view of the 14 spike-contact points on DPP4 (coloured in red). (D) Co-structure of MERS-CoV spike interaction with the predicted structure of Bactrian camel DPP4 (Swissmodel). Inset shows a top-down view of the 14 spike-contact points on DPP4 (coloured in red).
Clinical disease
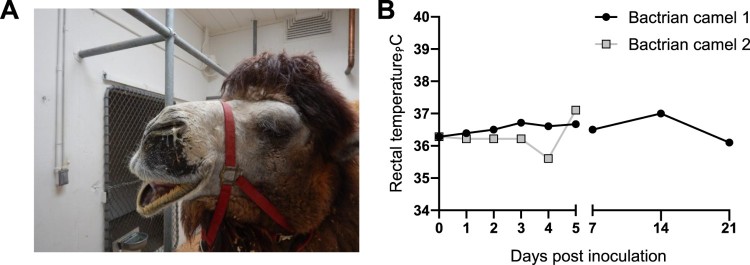
Two mature Bactrian camels were inoculated with 107 TCID50 of a human isolate of MERS CoV (strain HCoV-EMC/2012) [1] via the intranasal route. Bactrian camel 1 developed noticeable bilateral discharge beginning on 5 days dpi that continued through 7 dpi. Nasal discharge was observed in Bactrian camel 2 on 3 dpi after the animal became agitated due to handling; coughing and bilateral discharge was observed on 4 dpi, and the animal had bilateral discharge at necropsy on 5 dpi (Figure 3A). Body temperature for both animals remained within normal limits following virus inoculation and for the duration of the study (Figure 3B).
Figure 3.
Clinical signs in Bactrian camels inoculated with Middle East respiratory syndrome coronavirus (MERS-CoV). (A) Nasal discharge observed in Bactrian camel 2; both Bactrian camels displayed nasal discharge during the experiment (B) Rectal temperature during the experiment. Rectal temperatures are indicated for each camel by lines with geometric shapes.
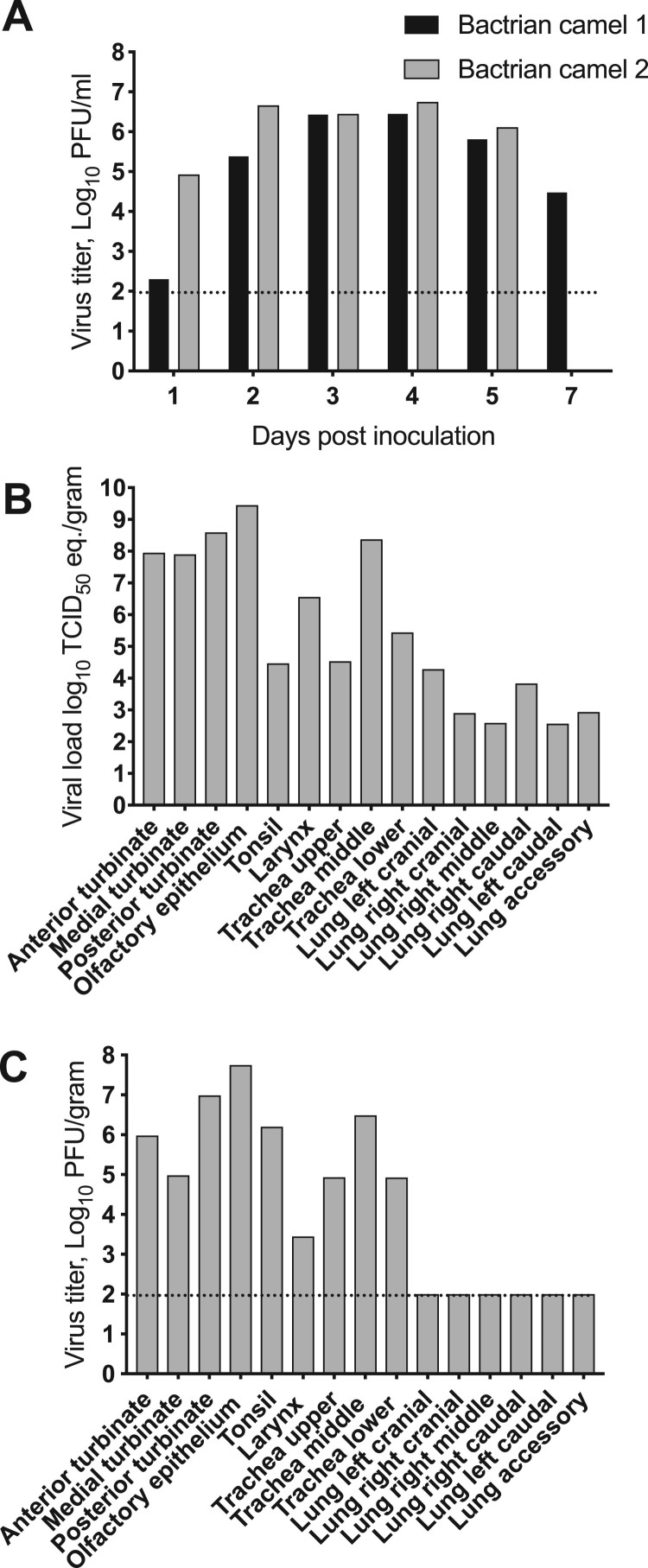
Viral shedding and tissue distribution of MERS-CoV
Infectious virus was detected in nasal swab samples beginning on 1 dpi and continued through 5 dpi for both animals. Bactrian camel 2 had detectable infectious virus on 7 dpi, but not on 14, 21, or 28 dpi (Figure 4A). Virus shedding reached its peak at 4 dpi, with 106.5 and 106.8 PFU/ml for Bactrian camel 1 and Bactrian 2 respectively. We compared the shedding of the two Bactrian camels with that of two dromedary camels inoculated in a previous experiment by area under curve analyses (AUC) (3), with Bactrian camels (AUC 23.85, 21.61–26.09 95% CI) and dromedary camels (AUC 23.18, 21.92–24.45 95% CI) being virtually identical.
Figure 4.
MERS-CoV shedding and tissue distribution in Bactrian camels. (A) Virus shedding from the upper respiratory tract in Bactrian camels inoculated with MERS-CoV determined by plaque assay. (B,C) Replication of MERS-CoV in the upper respiratory tract of Bactrian camels determined by qRT-PCR and plaque assay. The dotted line indicates the detection limits of the assays.
Tissue distribution was only evaluated in Bactrian camel 2 on 5 dpi. MERS-CoV RNA was detected in primarily in the upper respiratory tract (nasal turbinates, olfactory epithelium, tonsil, larynx and trachea) and limited amounts in the lower respiratory tract (lungs) (Figure 4B). Infectious virus was detected in nasal turbinates, olfactory epithelium, tonsil, larynx and trachea suggesting an upper respiratory tract infection (Figure 4C). No virus replication was observed in any of the other tissues analysed: lungs, heart, spleen, kidney, bladder, duodenum, colon, jejunum or lymph nodes (retropharyngeal, mediastinal, mesenteric, tracheobronchial and prescapular).
Histopathology and immunohistochemistry
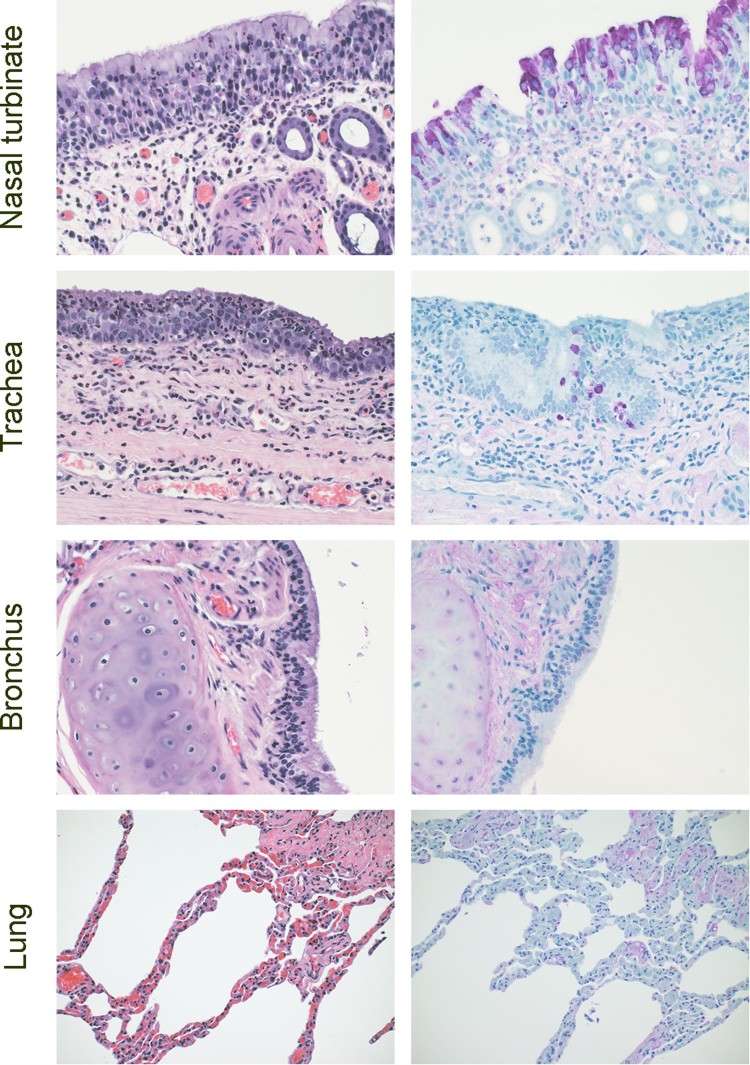
Tissues were collected from Bactrian camel 2 on 5 dpi and evaluated for pathology and the presence of viral antigens by immunohistochemistry. The observed lesions in the upper respiratory tract of the camels were characterized as mild to moderate subacute sinusitis, with epithelial necrosis and lympocytic submucosal inflammation in the nasal turbinates and subacute tracheitis with epithelial necrosis and lympocytic submucosal inflammation and squamous metaplasia. Viral antigen was detected within the epithelial cells of the nasal turbinates (primarily neuroeptithelium) and trachea (columnar epithelium) (Figure 5).
Figure 5.
Histopathology in Bactrian camels infected with MERS-CoV. Histopathologic changes at 5 days post inoculation in camel 2 inoculated with MERS-CoV. Tissues were collected and stained with hematoxylin and eosin (left panel). AntiMERS-CoV immunohistochemical results (right panel) are visible as a dark-purple stain. Viral antigen was detected within the epithelial cells of the nasal turbinates (primarily neuroeptithelium) and trachea (columnar epithelium). Original magnification ×400.
Serology
Both camels were seronegative at the time of infection and only Bactrian camel 1 was monitored for seroconversion as Bactrian camel 2 was euthanized on 5 dpi. Neutralizing antibodies were first detected in sera from Bactrian camel 1 on 14 dpi; titers were 20, 20, and 40 on days 14, 21, and 28, respectively (Table 1).
Table 1. Bactrian camel neutralizing antibody titer against MERS-CoV as determined by plaque reduction neutralization test.
| Day | Bactrian camel 1 | Bactrian camel 2 |
|---|---|---|
| 0 | <10 | <10 |
| 7 | <10 | NA |
| 14 | 20 | NA |
| 21 | 20 | NA |
| 28 | 40 | NA |
Discussion
Dromedary camels play a prominent role in the circulation, maintenance, and the zoonotic transmission of MERS-CoV [2,3,24]. In the present study we demonstrated that Bactrian camels are also susceptible to infection with MERS-CoV and, like dromedary camels [10,13], shed large quantities of infectious virus in nasal secretions. Moreover, the virus shedding kinetics of MERS-CoV in Bactrian camels was virtually identical to previous experimental studies in dromedary camels. Similar to infections reported in both naturally and experimentally infected dromedaries, infected Bactrian camels displayed only minor clinical disease characterized by mild to moderate quantities of nasal discharge.
The limitation of this study was use of only two animals and of those, only one was monitored for seroconversion. Nonetheless, the susceptibility and the magnitude and pattern of nasal virus shedding was nearly identical in both animals and clearly demonstrated that Bactrian camels are susceptible to MERS-CoV. The susceptibility of the Bactrian camel to MERS-CoV suggests a general pattern of susceptibility in new and old-world camelids. Experimental infections in camelids have resulted in productive infection and shedding of MERS-CoV [10,14–16], in contrast to other livestock species such as goat, sheep, pigs and horses where either no or very limited infection was observed [16,25,26]. Interestingly, the MERS-CoV amounts in the upper respiratory tract and the shedding from the upper respiratory tract in the new world camelids, llama and alpaca, are generally 2 log lower compared to those of the old world camelids, dromedary and Bactrian camel [10,13–16]. In addition, upon experimental MERS-CoV transmission studies with alpaca, although transmission between infected animal and contact animal occurred, the contact animal had 2 log lower virus shedding than the inoculated animals. This potentially suggests a lower ability of MERS-CoV for sustained transmission in this species [15].
Despite the current lack of field evidence of MERS-CoV infection in Bactrian camels, this study demonstrates that Bactrian camels can be readily infected and shed large quantities of virus in nasal secretions. If MERS-CoV were to be introduced into populations of Bactrian camels, we would expect that a potential endemic and sustained pattern of infection may result and they could act as a reservoir, similar to dromedaries, potentially exposing associated human communities to infection.
Funding Statement
This study was supported by the Division of Intramural Research of the NIAID.
RAB and VJM contributed equally to this article.
Acknowledgements
We thank Paul Gordy, Angela Bosco-Lauth, Rick Brandes, Greg Harding, and Joel Artver for their assistance with animal husbandry, Ryan Kissinger and Anita Mora for assistance with the graphics. All animal work described was approved by the Institutional Animal Care and Use Committee at Colorado State University and was performed in compliance with recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institute of Health.
Disclosure statement
No potential conflict of interest was reported by the authors.
ORCID
Vincent J. Munsterhttp://orcid.org/0000-0002-2288-3196
References
- 1.Zaki AM, van Boheemen S, Bestebroer TM, et al. Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia. N Engl J Med. 2012;367:1814–1820. doi: 10.1056/NEJMoa1211721. [DOI] [PubMed] [Google Scholar]
- 2.Azhar EI, et al. Evidence for camel-to-human transmission of MERS coronavirus. N Engl J Med. 2014;370:2499–2505. doi: 10.1056/NEJMoa1401505. [DOI] [PubMed] [Google Scholar]
- 3.Conzade R, et al. Reported direct and Indirect contact with dromedary camels among laboratory-confirmed MERS-CoV cases. Viruses. 2018;10; doi: 10.3390/v10080425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Haagmans BL, et al. Middle East respiratory syndrome coronavirus in dromedary camels: an outbreak investigation. Lancet Infect Dis. 2014;14:140–145. doi: 10.1016/s1473-3099(13)70690-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hemida MG, et al. MERS coronavirus in dromedary camel Herd, Saudi Arabia. Emerging Infect Dis. 2014;20; doi: 10.3201/eid2007.140571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Memish ZA, et al. Human infection with MERS coronavirus after exposure to infected camels, Saudi Arabia, 2013. Emerging Infect Dis. 2014;20:1012–1015. doi: 10.3201/eid2006.140402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Meyer B, et al. Antibodies against MERS coronavirus in dromedary camels, United Arab Emirates, 2003 and 2013. Emerging Infect Dis. 2014;20:552–559. doi: 10.3201/eid2004.131746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Muller MA, et al. MERS coronavirus neutralizing antibodies in camels, Eastern Africa, 1983-1997. Emerging Infect Dis. 2014;20:2093–2095. doi: 10.3201/eid2012.141026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.de Wit E, van Doremalen N, Falzarano D, et al. SARS and MERS: recent insights into emerging coronaviruses. Nat Rev Microbiol. 2016;14:523–534. doi: 10.1038/nrmicro.2016.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Adney DR, et al. Replication and shedding of MERS-CoV in upper respiratory tract of inoculated dromedary camels. Emerg Infect Dis. 2014;20:1999–2005. doi: 10.3201/eid2012.141280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Khalafalla AI, et al. MERS-CoV in upper respiratory tract and lungs of dromedary camels, Saudi Arabia, 2013-2014. Emerging Infect Dis. 2015;21:1153–1158. doi: 10.3201/eid2107.150070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.van Doremalen N, et al. High Prevalence of Middle East respiratory coronavirus in Young dromedary camels in Jordan. Vector Borne Zoonotic Dis. 2017;17:155–159. doi: 10.1089/vbz.2016.2062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Adney DR, et al. Efficacy of an adjuvanted Middle East respiratory syndrome coronavirus spike Protein Vaccine in dromedary camels and alpacas. Viruses. 2019;11; doi: 10.3390/v11030212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Haagmans BL, et al. An orthopoxvirus-based vaccine reduces virus excretion after MERS-CoV infection in dromedary camels. Science. 2016;351:77–81. doi: 10.1126/science.aad1283. [DOI] [PubMed] [Google Scholar]
- 15.Adney DR, Bielefeldt-Ohmann H, Hartwig AE, et al. Infection, replication, and transmission of Middle East respiratory syndrome coronavirus in alpacas. Emerging Infect Dis. 2016;22:1031–1037. doi: 10.3201/2206.160192 doi: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Vergara-Alert J, et al. Livestock susceptibility to infection with Middle East respiratory syndrome coronavirus. Emerging Infect Dis. 2017;23:232–240. doi: 10.3201/eid2302.161239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mammal Species of the World A taxonomic and geographic reference. https://www://www.departments.bucknell.edu/biology/resources/msw3/browse.asp?id=14200112 (2005).
- 18.Imamura K, et al. The distribution of the Two Domestic camel species in Kazakhstan caused by the demand of industrial stockbreeding. J Arid Land Stud. 2017;26:233–236. doi: 10.14976/jals.26.4_233. [DOI] [Google Scholar]
- 19.Chan SM, et al. Absence of MERS-coronavirus in Bactrian camels, southern Mongolia, November 2014. Emerging Infect Dis. 2015;21:1269–1271. doi: 10.3201/eid2107.150178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Liu R, et al. Absence of Middle East respiratory syndrome coronavirus in Bactrian camels in the West Inner Mongolia Autonomous Region of China: surveillance study results from July 2015. Emerg Microbes Infect. 2015;4:e73, doi: 10.1038/emi.2015.73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Miguel E, et al. Absence of Middle East respiratory syndrome coronavirus in Camelids, Kazakhstan, 2015. Emerging Infect Dis. 2016;22:555–557. doi: 10.3201/eid2203.151284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Corman V, et al. Detection of a novel human coronavirus by real-time reverse-transcription polymerase chain reaction. Euro Surveill. 2012;17:1–9. [DOI] [PubMed] [Google Scholar]
- 23.van Doremalen N, et al. Host species restriction of Middle East respiratory syndrome coronavirus through its receptor, dipeptidyl peptidase 4. J Virol. 2014;88:9220–9232. doi: 10.1128/jvi.00676-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Aly M, Elrobh M, Alzayer M, et al. Occurrence of the Middle East respiratory syndrome coronavirus (MERS-CoV) across the Gulf Corporation Council countries: Four years update. PloS one. 2017;12:e0183850), doi: 10.1371/journal.pone.0183850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.de Wit E, et al. Domestic Pig Unlikely reservoir for MERS-CoV. Emerging Infect Dis. 2017;23:985–988. doi: 10.3201/eid2306.170096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Adney DR, et al. Inoculation of Goats, sheep, and horses with MERS-CoV does Not result in productive viral shedding. Viruses. 2016;8; doi: 10.3390/v8080230. [DOI] [PMC free article] [PubMed] [Google Scholar]