Abstract
Objective:
Super-harmonic contrast-enhanced ultrasound imaging, also called acoustic angiography, has previously been used for imaging of microvasculature. This approach excites microbubble contrast agents near their resonance frequency and receives echoes at non-overlapping super-harmonic bandwidths. No integrated system currently exists could fully support this application. To fulfill this need, an integrated dual-channel transmit/receive system for super-harmonic imaging was designed, built and characterized experimentally.
Method:
The system was uniquely design for super-harmonic imaging and high-resolution B-mode imaging. A complete ultrasound system including a pulse generator, a data acquisition unit and a signal processing unit were integrated into a single package. The system was controlled by a field programmable gate array, on which multiple user-defined modes were implemented. A 6 MHz, 35 MHz dual-frequency dual-element intravascular ultrasound transducer was designed and used for imaging.
Result:
The system successfully obtained high resolution B-mode images of coronary artery ex vivo with 45 dB dynamic range. The system was capable of acquiring in vitro super-harmonic images of a vasa vasorum mimicking phantom with 30 dB contrast. It could detect a contrast agent filled tissue mimicking tube of 200 μm diameter.
Conclusion:
For the first time, high-resolution B-mode images and super-harmonic images were obtained in an intravascular phantom, made possible by the dedicated integrated system proposed. The system greatly reduced the cost and complexity of the super-harmonic imaging intended for preclinical study.
Significant:
The system showed promise for high-contrast intravascular microvascular imaging, which may have significant importance in assessment of the vasa vasorum associated with atherosclerotic plaques.
Index Terms—: Ultrasonic imaging, Biomedical electronics, Ultrasonic transducer, Intravascular ultrasound, Field-programmable gate arrays (FPGA), Acoustic angiography, Microbubble contrast agent
I. Introduction
ATHEROSCLEROTIC heart disease, which is also known as ischemic heart disease, is the number one cause of death globally [1, 2]. Atherosclerosis is a condition where the blood vessels progressively narrow and harden because of the formation of plaques, which are developed from the invasion of white blood cells and accumulation of lipid substances [3]. Plaques can be generally classified into two categories: stable or unstable based on their future propensity to rupture. While both kinds of plaques restrict normal blood flow, vulnerable plaque rupture has been hypothesized to be a primary reason of acute myocardial infraction, and leads to higher morbidity rate [3, 4]. It has been demonstrated that the advancing of plaque from stable to vulnerable in large arteries is related to the proliferation of the vasa vasorum, a supporting vessel network that supplies blood to the outer, diffusion-limited layers of the vessel wall [5–7]. Ruptured/vulnerable plaques show a higher degree of vasa vasorum proliferation, while the stable ones exhibit a lower degree [8, 9]. Compared to other criteria, the presence of vasa vasorum would provide information concerning plaque functionality which may be used in conjunction with morphological assessment for improved diagnosis of lethal plaques [10, 11]. By studying the plaque histology and its direct relation to clinical outcome, Hellings et al. have found that intra-plaque hemorrhage and vasa vasorum proliferation have strong prognostic value for systemic complications of cardiovascular diseases [12].
Ultrasound imaging is widely used in atherosclerosis screening and diagnosis [13]. When information regarding the vessel wall thickness, plaque size, morphology and composition is needed, catheter-based intravascular ultrasound (IVUS) is preferred compared to transcutaneous ultrasound imaging [14–16]. Typical IVUS utilizes miniature transducers with center frequencies between 10 to 40 MHz in order to produce images with sufficient resolution to analyze the morphology of the plaque burden as well as the vessel wall. However, traditional B-mode IVUS lacks the ability to detect vasa vasorum unaided. Many studies have focused on developing new IVUS technology for atherosclerosis imaging, such as super-high frequencies (>40 MHz), multi-frequency detection of thin fibrous caps, second harmonic contrast detection, and combining ultrasound with other imaging modalities as optical coherence tomography (OCT) or photoacoustic (PA) [17–21]. Among these methods, contrast-enhanced ultrasound has demonstrated its feasibility for vasa vasorum imaging in atherosclerotic lesions [22].
Contrast-enhanced ultrasound is a method of visualizing weakly scattering structures such as blood by perfusing the space with micron-sized contrast medium such as lipid encapsulated microbubbles [23]. These microbubble contrast agents (MCAs) are more compressible than surrounding blood or tissue and thus significantly enhance the acoustic backscatter from blood. Furthermore, MCAs respond very differently to ultrasound than tissue, a property which enables the ability to separate signals from bubbles and tissue [24]. Not only do microbubbles reflect the fundamental transmitted frequency, but they can also generate broadband harmonics at several multiples (10+) of the transmitted frequency when excited using moderate rarefractional pressures [25]. By rejecting frequency bands occupied by tissue reflection, the backscatter from tissue can be largely suppressed while retaining the higher-order nonlinear super-harmonics from MCAs (typically (> 3rd order).
Bouakaz et al. used a handheld phased array of interleaved elements (transmitting at 900 kHz and receiving at 2.8 MHz) to show that contrast specific imaging using higher order harmonics produced images having lower signal to noise ratios (SNR), but higher contrast to tissue ratios (CTR) [26]. Since then, several groups have studied the translation of contrast specific imaging to the IVUS platform. Goertz et al. developed a dual frequency IVUS probe to investigate contrast imaging using either 2nd harmonic (T20/R40 MHz) or subharmonic (T40/R20 MHz) approaches [27, 28], which could benefit from pulse-inversion for increased contrast sensitivity [29]. Radial modulation imaging of contrast agents has been performed using a commercial IVUS transducer where image quality using the approach could be maximized by optimizing the phase lag between modulation and interrogation pulses [30]. Maresca et al. have shown that chirp reversal is a viable method of imaging the ultra-harmonics, harmonics between the fundamental and second, produced by microbubbles for bandwidth limited transducers [31]. Each of these methods have translated a new contrast imaging approach to IVUS, but none as of yet have utilized the higher order harmonics for increased CTR that Bouakaz initially reported with their handheld transcutaneous transducer.
Using only higher-order super-harmonics (> 3rd order) to form images has been studied using specialized transducers having widely separated center frequencies. This method is commonly referred to as ‘acoustic angiography’ because it enables vessel imaging similar to that achieved with x-ray angiography. It has been characterized with high sensitivity to contrast agents as well as great resolution of vasculature in vivo, and shows promise for vasa vasorum and other microvascular structure detection [32–34].
Higher-order super-harmonic contrast-enhanced imaging (for simplicity: referred to as super-harmonic imaging) is only feasibly with transducers designed to cover large bandwidths. Dual-element, dual-frequency transducers are uniquely qualified to satisfy this requirement and are used in this imaging approach, because of the difficulty in making a single transducer having a fractional bandwidth to accommodate 4th or higher-order harmonics (ƒBW-6dB = 120%) [35]. Dual-frequency transducers with wideband separation between transmitting and receiving elements have been previously developed for super-harmonic imaging for both transcutaneous applications [33, 36] as well as a smaller design used for IVUS [35]. Previous IVUS transducers used a low-frequency (6 MHz) element for transmission and a high frequency (35 MHz) element for reception. In order to produce detectable nonlinear signals from MCAs, adequate peak negative pressure needs to be delivered at the focus of the receiving element. Since the transmission efficiency between transducers will likely vary, a robust imaging system should be equipped with a transmit pulser capable of transmitting pulses with varying pulse length and voltage amplitude. In addition to contrast-enhanced imaging, traditional B-mode images should be acquired using the high frequency elements in order to provide anatomical reference. Previous studies collected images using a modified commercial system (Vevo 770, VisualSonics, Toronto, Canada) and required an external power amplifier and function generator for producing the excitatory signal, which adds to the cost and complexity of the approach [33]. Currently, there are no imaging systems capable accommodating the unique design requirements imposed by this method of super-harmonic imaging.
In order to facilitate translation of super-harmonic imaging to pre-clinical and clinical applications, an integrated imaging system for the application is needed. A few research imaging systems have been developed for high frequency IVUS or dual-channel application [37–39], however, none of the lab-made or commercial imaging systems are fully capable for super-harmonic imaging. The development of a dual-channel transmit/receive system for super-harmonic imaging is reported in this paper. The system employs a field-programmable gate array (FPGA) as the central processing unit, making it reprogrammable in order to configure for multiple functions. The system is capable of doing user-defined pulse/burst generation, data acquisition, signal processing and PCIe signal communication. The layout of the imaging system is small and fits on a single 16×20 cm PCB board, making it portable and convenient for pre-clinical studies. A preliminary version of this work has been reported [40].
II. Methodology
A. Design Considerations
To obtain super-harmonic and B-mode imaging, two identical and interchangeable transmit/receive channels were included to work with dual-frequency, dual-element IVUS transducers. For super-harmonic imaging, a lower frequency transducer was used to transmit the excitation pulse, and a high frequency transducer was used to receive the echoes. For B-mode imaging, only the high frequency transducer was used for both transmit and receive.
On the receiving side, low distortion, low noise amplifiers were used to improve the dynamic range and to suppress baseline noise with a target SNR of 50 dB. Variable gain was implemented since the amplitude of super–harmonic signals can vary depending upon receive element sensitivity. The designed bandwidth of the receiving channel was from 1 MHz to 60 MHz, which covered the fundamental (6 MHz) to the 10th harmonic. The electrical impedance of the receiving channel was matched to 50 Ω at the transducer connection in order to efficiently operate the high frequency element of dual-frequency transducers.
When operating in pulse-echo mode for B-mode imaging, the transmitter should be able to provide a broadband pulse with an adjustable center frequency (e.g. 35 MHz). However, when operating in super-harmonic mode, the transmit pulser should also be able to generate a multiple cycle burst at a much lower center frequency in order to produce contrast specific images with higher SNR and dynamic range [41]. Considering there will be variance among transducer transmission efficiency, the pulse was designed to output high voltages and these voltages were made to be adjustable.
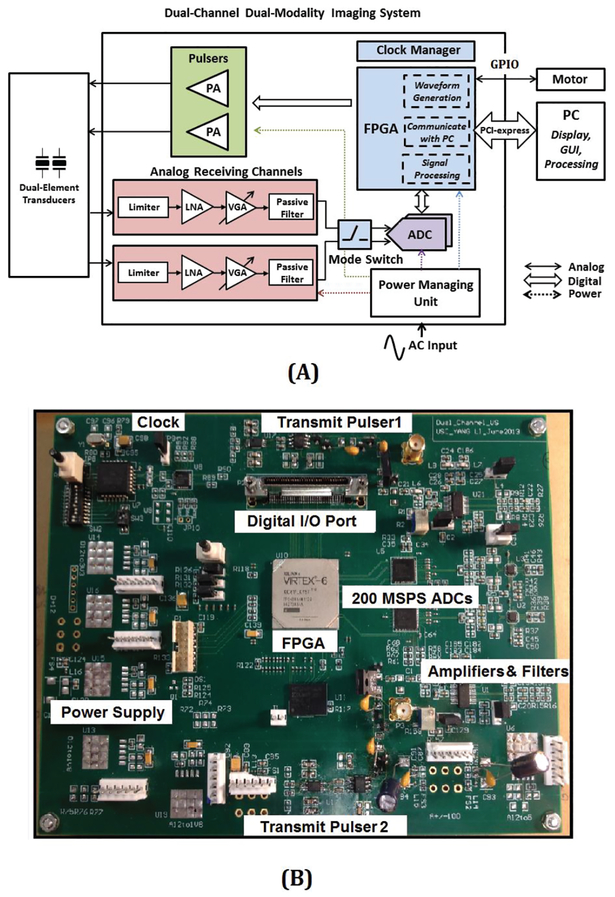
The block diagram of the integrated dual-frequency dual-channel imaging system is shown in Fig. 1(A). The system was controlled by a FPGA. Bipolar pulses or single frequency burst (pulse train) waveform could be generated inside the FPGA, which then passed to the transmit pulser. A digital Transmit/Receive (T/R) switch controlled the system to work in either dual-element mode for super-harmonic imaging or B-mode. After amplified and filtered by the analog receiving circuit, the returned echo signal was digitized by a 12-bit 200 MHz analog-to-digital converter (ADC). Separate AC-to-DC power supplies were used to provide the analog/digital working voltage (1 V, 2.5 V, 3.3 V and 5 V) and the pulse generator voltage (up to 200 Vpp). To synchronize with external motor, general purpose I/O pins were provided. The system communicated to the PC through a PCI-express digital I/O card. A summary of the important device parameters are listed in Table I. The system was fabricated based on a small-form-factor 6-layer printed circuit board (16×20 cm, 400 g), depicted in Fig. 1(B) Two SMA connectors were used for transducer connection.
Fig. 1.
(A) Block diagram of the integrated dual-frequency super-harmonic imaging system. Different color shows different power domain: blue: digital supply; green: high voltage for pulser; red, analog supply; purple, low-noise analog DC supply for ADCs. (B) Photograph of the system.
TABLE I.
Devices Used In The Proposed System
| Function Block | Device | Parameters | Manufacture Specification |
|---|---|---|---|
| Pulse Generator | TC6320 MOSFET Paira | Max Voltage | 200 Vpp |
| Rise/Falling Time | 15 ns | ||
| Amplifier | AD8331 Low-noise Amplifierb | Gain | −4.5 to 55 dB |
| −6dB Bandwidth | 100 MHz | ||
| Analog-to-Digital Converter | AD9230–210b | Sampling | 210 MHz |
| Resolution | 12 bits | ||
| Input Range | 1.25 V | ||
| FPGA | Virtex-6 XC6VLX75Tc | Number of IOs | 240 |
| Number of Logic Slices | 11640 | ||
| Data Transfer | PCIe Digital I/O Card PXIe6537Bd | Sampling Rate | 50 MSPS |
| Bus Width | 40 bits/Sample | ||
| DC-DC Converter | LT3083, LT3633e | Total Power Consumption | 3 W,12 V |
Supertex Inc., Sunnyvale, CA.
Analog Devices, Norwood, MA.
Xilinx, San Jose, CA.
National Instrument, Austin, TX.
Linear Technology, Milpitas, CA.
B. Specific Design Consideration
1). FPGA Logic
A Virtex-6 FPGA served as the central controller of the proposed system, working at 200 MHz clock rate. It received commands from the PC, controlled the pulse generator waveform and transmitted the data to the PC for display and further processing. The PC communicated to the FPGA through a PCI-express 40-channel digital I/O card, with 50 MHz clock rate, which could be programmed using Labview software (National Instrument, Austin, TX).
The digital logic diagram of the FPGA is shown in Fig. 2(A). To collect one frame of data, an Acq_Start signal was sent to the FPGA from PC through the digital I/O, together with several bits of control signals. Major user-defined control signals are listed in Table II. The mode switch was implemented on the FPGA, which controlled the on-and-off of the analog amplifier, and selected the channel for echo signal receiving.
Fig. 2.
(A) FPGA logic block diagrams. Dashed lines show the clock signal. (B) Logic timing of one frame data acquisition. Pulses number 1 and 3 are bipolar pulses for B-mode imaging; Pulses number 2 and 4 are multi-cycle pulse-train for super-harmonics imaging.
TABLE II.
Major User Defined Parameters Of The Proposed System
| Parameter | Selection | Control Signal(s) |
|---|---|---|
| Imaging Depth | 2048/4096/8192 sample points/line | Pre-loaded in FPGA |
| Number of Lines in a Frame | N:1–10000 | #of_Lines[0–7] |
| Number Pulses Repetition in Each Line | 1–2000 | #of_Pulses[0–6] |
| Transmit Pulse Polarity | 0° or 180° | Control [0] |
| Transmit Pulse Frequency | Any Integer Division of the Clock Frequency | Control [1–7] |
| Stimulation Mode | Pulse Echo Mode/Dual-element Mode | Mode |
| Receiving Channel | Chan1/Chan2 | Channel |
| Transfer Clock Speed | ≤50 MHz | Pre-loaded in FPGA |
Fig. 2(B) shows the timing diagram of image acquisition. At the rising edge of signal Pulse_Trigger, digital pulse/pulse-train were sent out through one pair of differential signal pins on the FPGA, which were then amplified by the pulse generator to get enough excitation voltage. We used single pulse excitation for B-mode imaging and multi-cycle burst for super-harmonic imaging, which were generated inside FPGA in the form of a differential digital wave. The digital pulse was then given to the transmit pulser. After echo signal was received, optional processing such as finite impulse response filtering, envelope detection was implemented on-board. Raw/processed data was stored into a first-in-first-out (FIFO) buffer. Once full, a FULL bit was generated, acknowledged the digital I/O card for data transferring of a frame of data. A Labview graphical user interface (GUI) was developed for control and data acquisition.
2). Analog Receiver Design
The echo signal from each transducer was passed into an individual analog receiving channel. To protect the receiver circuit, the signal first went through a custom passive limiter, which shunted the high amplitude transmit pulses to ground [42]. To provide enough gain for the low-amplitude super-harmonic signal, the received signals were then amplified by a low-noise variable-gain amplifier (AD8331), with overall adjustable gain of −4.5 dB to 54 dB. The −3 dB bandwidth of the amplifier spanned from 1 to 100 MHz when connected to a 50 Ω input impedance. The amplifier also converted the single-ended input to a differential output, which eliminated DC biasing. Signals were then low-pass filtered by a 3rd order Butterworth filter, with −6 dB cut-off frequency at 60 MHz. The signal was digitized by a 12-bit ADC AD9230 at a sampling rate of 200 MHz. After conversion, the differential digital signals were routed to the FPGA.
3). Pulse Generator
In order to drive the low-frequency transducer for super-harmonic imaging, a sinewave driving signal was the ideal choice, since this waveform produced minimum high-frequency harmonic leakage that could potentially contaminant the super-harmonic signal. For B-mode imaging, a broad-band pulse with center frequency near that of the high-frequency transducer was needed. To meet both the specifications, an adjustable frequency (6–35 MHz), high-voltage-gain power amplifier and a high-speed digital-to-analog converter had to be used. In additional to that, an impedance matching circuit to get rid of the shape distortion at variable driving frequency would also be necessary. These would increase the complexity and size of the circuit in a great extent.
We utilized an alternative solution, which was an on-and-off type high-voltage square wave generator. TC6320 from Supertex Inc. was used, in which a pair of high-speed PMOS and NMOS controlled the output of the positive and negative phase of the wave. The chip was able to provide square wave of 40–50 MHz with a maximum adjustable voltage output of 200 Vpp, which was the most compact and cost-effective solution we could find that meet the design specification. Two MOSFET drivers (ISL55110, Intersil Corp., Milpitas, CA) were used to amplify the digital pulses generated by the FPGA to 20 Vpp before entering the TC6320. The transmitted waveforms were passed through two stages of expanders using PMDB7000 high-speed, high-breakdown voltage diode (NXP Semiconductors, Eindhoven, Netherlands) before being sent to the transducer.
The transmit pulse generator was able to produce high voltage bipolar pulses for B-mode imaging, and burst (square wave train) for super-harmonic imaging. The center frequency of was controlled by the FPGA. Voltage output was adjusted by controlling the DC supply voltage to the MOSFET.
C. Transducer Design
For super-harmonic imaging, it was essential for the focus of the transmitting element to overlap the focus of the receiving element. In order to make the transducer small enough to fit into the intravascular catheter while keeping the T/R beams aligned, a stacked layer design was selected for this application [43]. According to the dimension of the transducer, the natural focus points of the 6 MHz and 35 MHz elements were about 1.8 and 1.7 mm, both of which were at the axial center of the transducer. The 35 MHz −3 dB beam was 100% covered within the 6 MHz −3 dB beam.
A photograph of the transducer is shown in Fig. 3(A). The transducer was designed as multiple layers with the high frequency layer in front of the low frequency layer, Fig. 3(B). The low frequency excitation wave was generated by the low frequency element (3 mm × 0.6 mm × 0.3 mm), and the wave could pass through the high frequency element to propagate into surrounding tissue. The high frequency super-harmonic wave generated by microbubbles was detected using the high frequency reception layer (0.5 mm × 0.6 mm × 0.065 mm), which was in front of the low frequency element. An acoustic filter layer was interleaved between the two piezoelectric layers. The acoustic filter layer functions as a matching layer for the low frequency wave, which did not block but enhanced the wave transmission efficiency. Meanwhile, the acoustic filter layer reflected the high frequency wave so that no aliasing echoes would occur [44, 45]. The whole transducer (4 mm × 0.6 mm × 0.5 mm) was mounted on a 20 Gauge hypodermic needle to mimic the housing for intravascular applications. The transducer was coated with a 15 μm thick parylene layer which functioned as both a passivation and matching layer of the high frequency element. A common ground was shared between the transmission and reception elements, while the low frequency excitation and the high frequency reception signals were connected individually. A detailed fabrication procedure was reported previously, and is summarized in Table III [35].
Fig. 3.
(A) Photograph of the transducer. The inset shows the front look of the acoustic stack. (B) Acoustic stack of the dual-layer transducer. (C)Experiment setup for vasa vasorum phantom imaging. (D) Microbubble size distribution.
TABLE III.
Transducer Used In The Current Study
| Parameter | Dual-Frequency Dual-Element IVUS Transducer | |
|---|---|---|
| Low Frequency Element | High Frequency Element | |
| Center Frequency | 6 MHz | 35 MHz |
| Active Material | PMN-PT | PMT-PT |
| Thickness | 300 μm | 65 μm |
| Matching Material | Al2O3/epoxy | Parylene |
| Backing Material | Silver epoxy | N/A |
| Aperture Size | 3×0.6 mm | 0.5×0.6 mm |
Two dual-element transducers were used in the study. One had 50% −6 dB fractional bandwidth for the 6 MHz component, which was primarily used for tests and imaging if not specify in the following context; the other had 20% −6 dB fractional bandwidth for the 6 MHz component. Both transducers had 50% −6 dB fractional bandwidth for the 35 MHz component.
D. Imaging Experimental Setup
Ex vivo IVUS imaging of heathy rabbit coronary arteries was conducted to demonstrate the B-mode imaging capability in addition to in vitro phantom imaging. These artery samples were donated by Dr. Tzung Hsiai from the University of California, Los Angeles, after the rabbit was sacrificed for an unrelated study. A 15 mm long aorta sample was removed from the animal. 5 mm of the sample was buried in a gelatin substrate for stabilization, and the rest of sample was submerged in formalin. During imaging experiment, formalin was replaced by water, and the transducer was placed inside the vessel longitudinally and rotated by a custom stepper motor. The system was operated in the pulse-echo mode, with the high frequency element of the dual-frequency IVUS transducer connected to one of the two channels. The pulse generator provided a single cycle, bipolar pulse at 60 Vpp and 33 MHz center frequency.
For super-harmonic in vitro imaging, both the high frequency element and the low-frequency element were connected to the system. The transducer was attached to the motor and placed in a 4 mm diameter channel cut through a tissue mimicking phantom. The phantom was prepared using a 0.075 g/mL concentration of 275 bloom gelatin with a 0.032 g/mL concentration of graphite scatters. The speed of sound was measured to be 1532 m/s using a pulse transmission approach. The acoustic attenuation coefficient was expressed by the following equation:
| (1) |
with units of dB/cm for α0 and MHz for f. In 4.5–7.5 MHz range, the phantom attenuation was α0=1.399, n=0.9439 (R2=0.952); in 15–30 MHz range, α0=0.1228 and n=1.4172 (R2=0.982). To mimic the size of vasa vasorum, an acoustically transparent tube with a 200 μm inner diameter was inserted through the phantom. The tube was transverse to the transducer imaging plane, as shown in Fig. 3(C). Additional details about the phantom formulation have been reported previously [46].
Polydisperse lipid-shelled microbubbles were made in the Dayton lab and were formulated as described previously [47]. Lipid solutions were formulated using 1,2-distearoyl-sn-glycero-3-phosophocholine (DSPC-Powder, Avanti Polar Lipids, Alabster, AL) and 1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-methoxy (polyethylene-glycol)-2000 (DSPE-PEG2000) in a 9 to 1 molar solution of propylene glycol, glycerol, and phosphate buffered saline (15%, 5%, and 80%, respectively). Lipids were agitated in the presence of decafluorobutane in 3 mL sealed glass vials using a commercial agitator (VialMix Shaker, Bristol-Myers-Squibb, New York, NY). Concentrations and sizes of microbubbles were measured optically using an Accusizer 780A (Particle Sizing Systems, Santa Barbara, CA) to measure microbubbles within a range from 0.5 to 6.0 μm. Microbubbles were diluted to a concentration of 8×107 microbubble/mL and then were pumped through the tube at a mean velocity of 4.4 cm/s while imaging. The microbubble population had a mean volume-weighted diameter of 1.24 μm. For comparison with other relevant studies, the size distribution of this microbubble population would approximate that of Definity™ (Bristol-Myers Squibb Medical Imaging, North Billericia, MA) that has been decanted for 1 hour prior to use [48]. A microbubble population having a smaller mean diameter (<2 μm) has been reported to provide increased nonlinear response at frequencies relevant to this study (>10 MHz) [49, 28]. The microbubble size distribution is shown in Fig. 3D.
Raw RF data was acquired in the current study. Data was post-processed in MATLAB (Mathworks, Natick, MA) using a 25–40 MHz 6th-order band-pass Butterworth digital filter. Envelop detection was applied for displaying using the Hilbert transform.
III. System Calibration And Imaging Result
A. Analog Receiver Characterization
The impulse response of the system was measured using the following methods. A pulse generated by Panametrics 5900PR (Olympus Corporation, Tokyo, Japan) was delivered through the transducer SMA connection port after being attenuated by 40 dB. The pulse waveform and the received signal collected by the proposed system are shown in Fig. 4. From the time domain waveform, it was evident that the baseline noise measured was removed by the analog receiving circuit. The frequency spectrum showed the analog receiver had a −6 dB cut-off frequency at 52 MHz, which was lower than the designed frequency of 60 MHz. The reason for the mismatch between the design and actual frequency was that a few passive components were not inserted at the input stage of the preamplifier in order to match the actual transducer impedance.
Fig. 4.
Impulse response of the receiver: (A) Input pulse voltage collected by the 2 GHz digital oscilloscope (dashed black) and the received signal (scaled to 1, blue) collected by the system. (B) The spectrum of the input pulse (dashed black), of the received signal (blue) and of analog filter simulation (red).
ADC SNR was 55.9 dB, which was defined by:
| (2) |
where Asignal was the full scale of ADC (1.25 V) and Anoise was measured by powering on the system without providing a signal input (open circuit). The noise level of the amplifier was tested by measuring the minimum detectable signal, where a sine wave was generated by the function generator and attenuated by a series of RF attenuators before being sent to the receiving channel of the system. At 35 MHz, the minimum detectable signal was 65 μV. The gain at 35 MHz was 42.9 dB.
B. Pulser Characterization
The output of the transmit pulse generator was measured by a digital oscilloscope (TDS 5052, Tektronix, OR). Fig. 5(A) and (C) show a pulse centered at 30 MHz and its spectrum together with the pulse echo signal collected from the 35 MHz element transducer by the proposed system. The −6 dB bandwidth of the pulse covered from 8 MHz to 50 MHz. Center frequency of the high frequency element was 35.6 MHz, −6 dB fractional bandwidth was 47%. For the low-frequency element, the actual center frequency was measured to be 5.8 MHz and −6 dB fractional bandwidth was 48%.
Fig. 5.
(A) Broad-band pulse and spectrum measured by oscilloscope. (B) 6 MHz pulse train and spectrum measured by oscilloscope. (C) Pulse echo signal of the 35 MHz element collected by the proposed system. (D) Acoustic pressure output of the 6 MHz element measured by the hydrophone, shown in hydrophone voltage.
The pulser could generate burst signal for super-harmonic imaging. As shown in Fig. 5(B), the spectrum of a 3-cycle square wave burst had strong odd harmonics, which were 10 dB weaker every 2 octaves. The acoustic output of the 6 MHz element driven by the burst was also measured, using a needle hydrophone (HNA-0400, Onda Corp., Sunnyvale, CA) placed axially 3 mm away from the transducer, a little farther than the near field of the transducer for both frequencies. Shown in Fig. 5(D), the odd-order-harmonic signal originated from the transmit pulse train was suppressed by the transducer, but there was still a 22 MHz high frequency component in addition to the 6 MHz baseband. It was important that the excitation acoustic wave should have weak high frequency leakage, because tissue scattering of the high frequency would impair the distinction between signal originating from contrast versus tissue. To suppress the 22 MHz component a high-pass filter with 25 MHz corner frequency was applied during image post-processing.
Fig. 6 shows the relationship between the supply voltage and low-frequency transducer acoustic output. The slope, or the transmit pressure to voltage ratio, was 19.37 kPa/V for a 2-cycle burst case, and 14.46 kPa/V for single cycle pulse. Above 140 Vpp, both 1-cycle and 2-cycle bursts could produce peak negative pressures over 1 MPa, which was sufficient to produce broad-band transient responses from MCAs for higher order super-harmonic imaging [34]. In practice, we limit the driving voltage under 150 Vpp for the durability of the transducer.
Fig. 6.
Acoustic pressure collected using a calibrated hydrophone. The transmitting element was excited using a 6 MHz 1-cycle or 2-cycle pulse.
C. Imaging Result
Fig. 7 shows the ex vivo B-mode image of rabbit coronary artery collected using the high frequency (35 MHz) element of the IVUS transducer and the proposed system. A dynamic range of 45 dB was achieved, with the front and back layer of artery were clearly visible in the image. The image was formed by acquiring 2000 lines for a single revolution. In addition to filtering and enveloped detection, a clutter filter was applied to the dataset to reduce stationary echoes.
Fig. 7.
Ex vivo B-mode image of the rabbit coronary artery obtained by 35 MHz transducer element.
Super-harmonic imaging was performed in the tissue mimicking phantom. The IVUS transducer was placed inside the lumen of phantom. In actual IVUS imaging, the position of the transducer was relatively arbitrary in the lumen, resulting in an imaging range of 1 mm to 10 mm (human artery diameter) to cover the vascularization in both plaques and the vasa vasorum inside the vessel wall. Thus, in the in vitro experiment, we chose the imaging plane where the vasa vasorum mimicking tube was 6 mm away from the transducer as an approximately median value of the imaging range. For B-mode imaging, water or MCAs were injected continuously into the vasa vasorum mimicking tube. The result with MCAs injected is given in Fig. 8(A), where the approximate location of the tube is circled, which can hardly be distinguished from the tissue. B-mode imaging with water injected was very similar to Fig. 8(A), result not shown. Super-harmonic image was acquired using the same setup, with the pulse generator providing a 3-cycle transmit burst at 120 Vpp. As shown in Fig. 8(B), the MCAs inside the tube that were invisible in B-mode were visualized using super-harmonic, while the fundamental frequency containing mostly tissue mimicking phantom backscatter was successfully suppressed. A dynamic range of 30 dB was achieved, which was a 10 dB improvement over previously reported results [35]. Stronger ring-down from the transmit wave can be seen near the center of the image. The image was formed by acquiring 400 lines for a single revolution.
Fig. 8.
Phantom super-harmonic imaging result. (A) B-mode image, when MCAs were injected into the micro tube. The approximated location of tube is circled with red. (B) Super-harmonic image obtained when MCAs were injected continuously into the micro-tube.
Fig. 9(A) shows a zoom-in view of MCAs inside the tube, re-drawn from Fig. 8(B). A scale bar (red) is drawn in the axial direction and has a length corresponding to 200 μm. The size of the tube in the image was approximately 300 μm from −6 dB measurements.
Fig. 9.
Expanded view of super-harmonic image. (A) is redraw from Fig. 8(B). (B) was collected when water was injected into the vessel mimicking tube. (C) and (D) were collected using a different transducer with narrower bandwidth. (D) was collected where the micro-tube ran parallel to the imaging plane. 200 μm along the axial direction is indicated by the red bars.
The vessel mimicking tube was not transparent under super-harmonic mode, probably due to the aging and dehydration of the tube material. Fig. 9(B) shows the super-harmonic image when water instead of MCAs were injected into the tube. The 200 μm lumen of the tube could be seen. There were three major ways to distinguish the MCA signal from the tube signal. First, the super-harmonics from MCAs covered an area that was wider than the tube diameter and had a vague layered structure, while the signal from tube had a clear layered structure representing the tube cross-section; second, the signal from MCAs was non-uniform across different frames, while that from the tube was uniform; third, the signal from the tube could be suppressed or eliminated when a small 2–3° tilt angle was applied to the sample, while the transducer was still able to rotate inside the lumen, but MCA signal was not affected by the tilt angle.
More super-harmonic images were collected using another dual-frequency transducer, whose 6 MHz element had 20% fractional bandwidth, 28% narrower than the one used for Fig. 8. Fig. 9(C) shows the cross-section view collected of the vasa vasorum mimicking tube, where the front and back surface of the tube cannot be seen. In Fig. 9(D), another imaging plane was chosen, where the tube ran nearly parallel to the imaging plane, so the longitudinal cross section of the tube could be imaged. The sizes of micro-tube in these images were about 500 μm.
IV. Discussion
The capability of the system for super-harmonic contrast-enhanced imaging has been demonstrated. In the phantom vasa vasorum study, a dynamic range of 30 dB was achieved. High frequency B-mode imaging was also acquired by the system. Some of the key design features for this integrated system that directly impacted the quality of the images obtained are discussed below.
A. Selection of Transmit and Receive Frequency
Although it was possible to excite the microbubbles above its resonance frequency and make harmonic images using a single transducer [27, 28], the nonlinear behavior of microbubbles were more pronounced when properly excited near their resonant frequency [50]. As a result, a transmit frequency was selected near the bubble’s resonance frequency, where less pressure might be used for contrast specific imaging, which was ideal since nonlinear signal generation from tissue scales dramatically with transmitted pressure [51].
On the receiving side, higher receiving frequency provided better rejection of the tissue backscatter signal. However, a trade-off had to be made considering the stronger attenuation effect of the high-frequency component and its impact on detectable signal strength. Higher-order harmonic imaging studies have been conducted to optimize which pressures and frequency combinations yielded optimal contrast to tissue ratios, generally noting that lower transmission frequencies and 3rd to 6th-order receiving frequencies produce higher dynamic range and SNR [41]. For the current study, 6 MHz transmitting and 35 MHz receiving was optimal considering both signal strength and backscattering rejection.
B. Resolution
The resolution of B-mode image can be estimated by measuring the −6 dB width of the point spread function, which was 30 μm axially and 110 μm laterally, measured by the proposed system and the high-frequency transducer element (fractional bandwidth 47%), results not shown. For super-harmonic contrast-enhanced imaging, however, because of the flow of MCAs, it was hard to identify a static point target and to define the resolution. As shown in Fig. 9, the size of the vasa vasorum mimicking tube in the image is larger than its actual size when the transmit transducer with narrower bandwidth was used, which might be explained by ring-down in the excitation waveform and axial aliasing. This implied that the super-harmonic imaging axial resolution was related to both transmit pulse-width and receive element resolution, which was in agreement with the findings in the literature [41]. Thus, in order to improve resolution, transmit and receive transducer with higher bandwidth need to be used, as well as shorter transmit driving signal.
According to the discussion above, imaging resolution can be improved by reducing the number of cycles in the transmit pulses. However, a certain threshold of negative peak pressure had to be reached, in order to excite detectable super-harmonics. Using the current transducer, we could not get any super-harmonic signal using 1-cyle 200 Vpp (maximum voltage) pulse. This could be explained by the damping introduced by the transducer. As shown in Fig. 5(D), when driving by a multi-cycle pulse train, the transmitting negative pressure ramped up and peaked at the 2nd pulse. If the time constant of the transducer could be reduced, presumably shorter pulse can be used to reach the transmit pressure threshold and thus resolution can be improved.
C. Dynamic Range
The absolute strength of the super-harmonic signal generated from the MCAs nonlinear oscillation was determined by the non-linear oscillation signal amplitude, which was directly related to the acoustic power delivered to the focus of the receive element. As the driving voltage to the transducer went up, the energy delivered to this region increased linearly (see Fig. 6). Other studies have reported that increasing the transmitted pressure used to excite microbubbles improved dynamic range until a threshold pressure was reached where it was likely that microbubble fragmentation occurred and limited further increases in dynamic range at higher pressures [52]. The loss of echogenicity from microbubbles fundamentally limited the maximum dynamic range obtainable.
The second factor that reduced the dynamic range of the system was the higher-order harmonic components that were emitted from the transmitting element. As shown in Fig. 5(D), there was a 22 MHz component generated by the 6 MHz transducer when driven by a 3-cycle burst. This harmonic component was closed the −6 dB bandwidth of the receiving element, meaning tissue could reflect higher order harmonics and reduce the dynamic range of the system. This high frequency component from the low-frequency transducer might be originated from the 19.5 MHz harmonic from the driving waveform, as shown from Fig. 5(B). To get rid of the harmonic, a passive harmonic reduction circuit reported in literature can be used, which can suppress the 3rd-order harmonic by more than 10 dB [53]. Using sinewave generator would minimize the harmonic, but the size and cost-effectiveness would be compromised as mentioned above.
A final factor that could be controlled to increase the dynamic range of the system would be the noise level. In the proposed system, a SNR of 56 dB was achieved. However, there was still room to improve the sensitivity and noise tolerance of the onboard amplifier and ADC converter circuits.
D. Future Improvement
Since only a single element was used for receiving in both imaging modes, it was possible to further reduce the system size by combining the two receiving channels into one using proper multiplexing. Another improvement that can be implemented was real-time imaging function, which would be important for intravascular in vivo imaging, considering the motion of blood vessel in normal heart cycle. By utilizing a data buffering system such as a double data rate type three synchronous dynamic random-access memory (DDR3 - SDRAM), current hardware structure and communication solution was capable to support real-time imaging.
Although only images from the IVUS setup were shown in the current study, a system such as this could be extended into other imaging applications such as transcutaneous operations using larger hand-held transducers.
V. Conclusion
An integrated, compact, dual-channel imaging system for super-harmonic contrast-enhanced ultrasound was developed. The system supported acquisition of high-frequency B-mode imaging, as well as high-order super-harmonic contrast-enhanced imaging. The measured SNR of the system was 56 dB with adjustable gain. When connected to a dual-element, dual-frequency (6 MHz, 35 MHz) intravascular transducer, the system was able to achieve 45 dB of dynamic range for ex vivo coronary artery imaging. It could also detect contrast agents within a 200 μm vasa vasorum mimicking vessel embedded in an in vitro phantom with a dynamic range of 30 dB, which was a 10 dB improvement over previously reported results. It was the first time that both high-resolution B-mode images and contrast-enhanced images were collected using intravascular transducer, which showed the capability of the system for intravascular vasa vasorum detection. Additional experiments for vasa vasorum imaging need to be carried out to investigate the capability of the system for preclinical study.
Acknowledgement
The authors would like to acknowledge Prof. Tzung Hsiai and his lab from UCLA for his gracious donation of animal tissue. The authors would like to acknowledge Prof. Ming Qian for his support on transducer fabrication and experiment.
This work was supported by the NIH grant R01EB015508 and P41-EB2002182.
Contributor Information
Yang Li, Department of Biomedical Engineering, University of Southern California, Los Angeles, CA, USA..
Jianguo Ma, Department of Mechanical and Aerospace Engineering, North Carolina State University, Raleigh, NC, USA..
K. Heath Martin, Joint Department of Biomedical Engineering, University of North Carolina/North Carolina State University, Chapel Hill, NC, USA..
Mingyue Yu, Department of Biomedical Engineering, University of Southern California, Los Angeles, CA, USA..
Teng Ma, Department of Biomedical Engineering, University of Southern California, Los Angeles, CA, USA..
Paul A. Dayton, Joint Department of Biomedical Engineering, University of North Carolina/North Carolina State University, Chapel Hill, NC, USA.
Xiaoning Jiang, Department of Mechanical and Aerospace Engineering, North Carolina State University, Raleigh, NC, USA..
K. Kirk Shung, Department of Biomedical Engineering, University of Southern California, Los Angeles, CA, USA..
Qifa Zhou, Department of Biomedical Engineering, University of Southern California, Los Angeles, CA, USA..
References
- [1].Alwan A, Global status report on noncommunicable diseases 2010: World Health Organization, 2011. [Google Scholar]
- [2].Olsson O, “Vertebral Angiography in the Diagnosis of Acoustic Nerve Tumours,” Acta Radiologica, vol. 39, pp. 265–272, 1953. [DOI] [PubMed] [Google Scholar]
- [3].Naghavi M, Libby P, Falk E, Casscells SW, Litovsky S, Rumberger J, et al. , “From vulnerable plaque to vulnerable patient a call for new definitions and risk assessment strategies: part I,” Circulation, vol. 108, pp. 1664–1672, 2003. [DOI] [PubMed] [Google Scholar]
- [4].Muller JE, Tofler GH, and Stone PH, “Circadian variation and triggers of onset of acute cardiovascular disease,” Circulation, vol. 79, pp. 733–43, April 1989. [DOI] [PubMed] [Google Scholar]
- [5].Moreno PR, Purushothaman KR, Fuster V, Echeverri D, Truszczynska H, Sharma SK, et al. , “Plaque neovascularization is increased in ruptured atherosclerotic lesions of human aorta: implications for plaque vulnerability,” Circulation, vol. 110, pp. 2032–8, October 5 2004. [DOI] [PubMed] [Google Scholar]
- [6].Moreno PR, Purushothaman KR, Sirol M, Levy AP, and Fuster V, “Neovascularization in human atherosclerosis,” Circulation, vol. 113, pp. 2245–52, May 9 2006. [DOI] [PubMed] [Google Scholar]
- [7].Heistad DD, Marcus ML, Larsen GE, and Armstrong ML, “Role of vasa vasorum in nourishment of the aortic wall,” Am J Physiol, vol. 240, pp. H781–7, May 1981. [DOI] [PubMed] [Google Scholar]
- [8].Falk E, Shah PK, and Fuster V, “Coronary Plaque Disruption,” Circulation, vol. 92, pp. 657–671, August 1 1995. [DOI] [PubMed] [Google Scholar]
- [9].Kockx MM, Cromheeke KM, Knaapen MW, Bosmans JM, De Meyer GR, Herman AG, et al. , “Phagocytosis and macrophage activation associated with hemorrhagic microvessels in human atherosclerosis,” Arterioscler Thromb Vasc Biol, vol. 23, pp. 440–6, March 1 2003. [DOI] [PubMed] [Google Scholar]
- [10].Gossl M, Malyar NM, Rosol M, Beighley PE, and Ritman EL, “Impact of coronary vasa vasorum functional structure on coronary vessel wall perfusion distribution,” Am J Physiol Heart Circ Physiol, vol. 285, pp. H2019–26, November 2003. [DOI] [PubMed] [Google Scholar]
- [11].Doyle B and Caplice N, “Plaque neovascularization and antiangiogenic therapy for atherosclerosis,” J Am Coll Cardiol, vol. 49, pp. 2073–80, May 29 2007. [DOI] [PubMed] [Google Scholar]
- [12].Hellings WE, Peeters W, Moll FL, Piers SR, van Setten J, van der Spek PJ, et al. , “Composition of carotid atherosclerotic plaque is associated with cardiovascular outcome: a prognostic study,” Circulation, vol. 121, pp. 1941–50, May 4 2010. [DOI] [PubMed] [Google Scholar]
- [13].Olin JW, Kaufman JA, Bluemke DA, Bonow RO, Gerhard MD, Jaff MR, et al. , “Atherosclerotic Vascular Disease Conference: Writing Group IV: imaging,” Circulation, vol. 109, pp. 2626–33, June 1 2004. [DOI] [PubMed] [Google Scholar]
- [14].Yock PG and Fitzgerald PJ, “Intravascular ultrasound: state of the art and future directions,” Am J Cardiol, vol. 81, pp. 27E–32E, April 9 1998. [DOI] [PubMed] [Google Scholar]
- [15].Nishimura RA, Edwards WD, Warnes CA, Reeder GS, Holmes DR Jr., Tajik AJ, et al. , “Intravascular ultrasound imaging: in vitro validation and pathologic correlation,” J Am Coll Cardiol, vol. 16, pp. 145–54, July 1990. [DOI] [PubMed] [Google Scholar]
- [16].Nair A, Kuban BD, Tuzcu EM, Schoenhagen P, Nissen SE, and Vince DG, “Coronary plaque classification with intravascular ultrasound radiofrequency data analysis,” Circulation, vol. 106, pp. 2200–6, October 22 2002. [DOI] [PubMed] [Google Scholar]
- [17].Li X, Ma T, Tian J, Han P, Zhou Q, and Shung KK, “Micromachined PIN-PMN-PT crystal composite transducer for high-frequency intravascular ultrasound (IVUS) imaging,” IEEE Trans Ultrason Ferroelectr Freq Control, vol. 61, pp. 1171–8, July 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Ma T, Yu MY, Chen ZY, Fei CL, Shung KK, and Zhou Q, “Multi-Frequency Intravascular Ultrasound (IVUS) Imaging,” Ieee Transactions on Ultrasonics Ferroelectrics and Frequency Control, vol. 62, pp. 97–107, January 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [19].Goertz DE, Frijlink ME, Tempel D, van Damme LC, Krams R, Schaar JA, et al. , “Contrast harmonic intravascular ultrasound: a feasibility study for vasa vasorum imaging,” Invest Radiol, vol. 41, pp. 631–8, August 2006. [DOI] [PubMed] [Google Scholar]
- [20].Li J, Li X, Mohar D, Raney A, Jing J, Zhang J, et al. , “Integrated IVUS-OCT for real-time imaging of coronary atherosclerosis,” JACC Cardiovasc Imaging, vol. 7, pp. 101–3, January 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Sethuraman S, Aglyamov SR, Amirian JH, Smalling RW, and Emelianov SY, “Intravascular photoacoustic imaging using an IVUS imaging catheter,” IEEE Trans Ultrason Ferroelectr Freq Control, vol. 54, pp. 978–86, May 2007. [DOI] [PubMed] [Google Scholar]
- [22].Granada JF and Feinstein SB, “Imaging of the vasa vasorum,” Nat Clin Pract Cardiovasc Med, vol. 5 Suppl 2, pp. S18–25, August 2008. [DOI] [PubMed] [Google Scholar]
- [23].Ferrara K, Pollard R, and Borden M, “Ultrasound microbubble contrast agents: fundamentals and application to gene and drug delivery,” Annu Rev Biomed Eng, vol. 9, pp. 415–47, 2007. [DOI] [PubMed] [Google Scholar]
- [24].Shi WT and Forsberg F, “Ultrasonic characterization of the nonlinear properties of contrast microbubbles,” Ultrasound Med Biol, vol. 26, pp. 93–104, January 2000. [DOI] [PubMed] [Google Scholar]
- [25].Kruse DE and Ferrara KW, “A new imaging strategy using wideband transient response of ultrasound contrast agents,” IEEE Trans Ultrason Ferroelectr Freq Control, vol. 52, pp. 1320–9, August 2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [26].Bouakaz A, Frigstad S, Ten Cate FJ, and de Jong N, “Super harmonic imaging: a new imaging technique for improved contrast detection,” Ultrasound Med Biol, vol. 28, pp. 59–68, January 2002. [DOI] [PubMed] [Google Scholar]
- [27].Goertz DE, Frijlink ME, de Jong N, and van der Steen AF, “Nonlinear intravascular ultrasound contrast imaging,” Ultrasound Med Biol, vol. 32, pp. 491–502, April 2006. [DOI] [PubMed] [Google Scholar]
- [28].Goertz DE, Frijlink ME, Tempel D, Bhagwandas V, Gisolf A, Krams R, et al. , “Subharmonic contrast intravascular ultrasound for vasa vasorum imaging,” Ultrasound Med Biol, vol. 33, pp. 1859–72, December 2007. [DOI] [PubMed] [Google Scholar]
- [29].Simpson DH, Chin CT, and Burns PN, “Pulse inversion Doppler: a new method for detecting nonlinear echoes from microbubble contrast agents,” IEEE Trans Ultrason Ferroelectr Freq Control, vol. 46, pp. 372–82, 1999. [DOI] [PubMed] [Google Scholar]
- [30].Yu FT, Villanueva FS, and Chen X, “Radial modulation contrast imaging using a 20-MHz single-element intravascular ultrasound catheter,” IEEE Trans Ultrason Ferroelectr Freq Control, vol. 61, pp. 779–91, May 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [31].Maresca D, Renaud G, van Soest G, Li X, Zhou Q, Shung KK, et al. , “Contrast-enhanced intravascular ultrasound pulse sequences for bandwidth-limited transducers,” Ultrasound Med Biol, vol. 39, pp. 706–13, April 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [32].Shelton SE, Lee YZ, Lee M, Cherin E, Foster FS, Aylward SR, et al. , “Quantification of Microvascular Tortuosity during Tumor Evolution Using Acoustic Angiography,” Ultrasound Med Biol, vol. 41, pp. 1896–904, July 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [33].Gessner RC, Frederick CB, Foster FS, and Dayton PA, “Acoustic angiography: a new imaging modality for assessing microvasculature architecture,” Int J Biomed Imaging, vol. 2013, p. 936593, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [34].Gessner RC, “Acoustic angiography: A new imaging platform for high resolution mapping of microvasculature and tumor assessment,” Ph.D. dissertation, Dept. Biomed. Eng., University of North Carolina, Chapel Hill, NC, 2013. [Google Scholar]
- [35].Ma J, Martin KH, Dayton PA, and Jiang X, “A Preliminary Engineering Design of Intravascular Dual-Frequency Transducers for Contrast-Enhanced Acoustic Angiography and Molecular Imaging,” Ieee Transactions on Ultrasonics Ferroelectrics and Frequency Control, vol. 61, pp. 870–880, May 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [36].Martin KH, Lindsey BD, Ma J, Lee M, Li S, Foster FS, et al. , “Dual-frequency piezoelectric transducers for contrast enhanced ultrasound imaging,” Sensors (Basel), vol. 14, pp. 20825–42, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [37].Ricci S, Boni E, Guidi F, Morganti T, and Tortoli P, “A programmable real-time system for development and test of new ultrasound investigation methods,” IEEE Trans Ultrason Ferroelectr Freq Control, vol. 53, pp. 1813–9, October 2006. [DOI] [PubMed] [Google Scholar]
- [38].Lewandowski M and Nowicki A, “High frequency coded imaging system with RF,” IEEE Trans Ultrason Ferroelectr Freq Control, vol. 55, pp. 1878–82, August 2008. [DOI] [PubMed] [Google Scholar]
- [39].Qiu WB, Chen Y, Li X, Yu YY, Cheng WF, Tsang FK, et al. , “An Open System for Intravascular Ultrasound Imaging,” Ieee Transactions on Ultrasonics Ferroelectrics and Frequency Control, vol. 59, pp. 2201–2209, October 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [40].Li Y, Ma J, Martin KH, Choi H, Dayton PA, Jiang X, et al. , “A configurable dual-frequency transmit/receive system for acoustic angiography imaging,” in Ultrasonics Symposium (IUS), 2014 IEEE International, 2014, pp. 731–733. [Google Scholar]
- [41].Lindsey BD, Rojas JD, Martin KH, Shelton SE, and Dayton PA, “Acoustic characterization of contrast-to-tissue ratio and axial resolution for dual-frequency contrast-specific acoustic angiography imaging,” IEEE Trans Ultrason Ferroelectr Freq Control, vol. 61, pp. 1668–87, October 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [42].Choi H, Kim MG, Cummins TM, Hwang JY, and Shung KK, “Power MOSFET-Diode-Based Limiter for High-Frequency Ultrasound Systems,” Ultrason Imaging, vol. 36, pp. 317–30, October 2014. [DOI] [PubMed] [Google Scholar]
- [43].Ma J, Martin KH, Li Y, Dayton PA, Shung KK, Zhou Q, et al. , “Design factors of intravascular dual frequency transducers for super-harmonic contrast imaging and acoustic angiography,” Phys Med Biol, vol. 60, pp. 3441–57, May 7 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [44].Ma J, Steer MB, and Jiang X, “An acoustic filter based on layered structure,” Applied Physics Letters, vol. 106, March 16 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [45].Ma J, Li S, Wang Z, and Jiang X, “Anti-matching design for wave isolation in dual frequency transducer for intravascular super-harmonic imaging,” in ASME 2014 International Mechanical Engineering Congress and Exposition, 2014, pp. V003T03A084–V003T03A084. [Google Scholar]
- [46].Madsen EL, Zagzebski JA, Banjavie RA, and Jutila RE, “Tissue mimicking materials for ultrasound phantoms,” Med Phys, vol. 5, pp. 391–4, Sep-Oct 1978. [DOI] [PubMed] [Google Scholar]
- [47].Streeter JE, Gessner R, Miles I, and Dayton PA, “Improving sensitivity in ultrasound molecular imaging by tailoring contrast agent size distribution: in vivo studies,” Mol Imaging, vol. 9, pp. 87–95, April 2010. [PMC free article] [PubMed] [Google Scholar]
- [48].Goertz DE, de Jong N, and van der Steen AF, “Attenuation and size distribution measurements of Definity and manipulated Definity populations,” Ultrasound Med Biol, vol. 33, pp. 1376–88, September 2007. [DOI] [PubMed] [Google Scholar]
- [49].Goertz DE, Frijlink M, Bouakaz A, Chin C, de Jong N, and van der Steen A, “The effects of bubble size on nonlinear scattering from microbubbles,” in Ultrasonics, 2003 IEEE Symposium on, 2003, pp. 1503–1506. [Google Scholar]
- [50].de Jong N, Cornet R, and Lancee CT, “Higher Harmonics of Vibrating Gas-Filled Microspheres. 1. Simulations,” Ultrasonics, vol. 32, pp. 447–453, November 1994. [Google Scholar]
- [51].Duck FA, “Nonlinear acoustics in diagnostic ultrasound,” Ultrasound Med Biol, vol. 28, pp. 1–18, January 2002. [DOI] [PubMed] [Google Scholar]
- [52].Gessner RC, Lukacs M, Lee M, Cherin E, Foster FS, and Dayton PA, “High-resolution, high-contrast ultrasound imaging using a prototype dual-frequency transducer: in vitro and in vivo studies,” IEEE Trans Ultrason Ferroelectr Freq Control, vol. 57, pp. 1772–81, August 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [53].Choi H, Jung H, Chen R, and Shung KK, “Harmonic distortion reduction technique of the power amplifier for very high frequency ultrasonic transducer applications,” in Ultrasonics Symposium (IUS), 2013 IEEE International, 2013, pp. 1564–1566. [Google Scholar]