Abstract
Carotenoids are natural pigments with substantial applications in nutraceutical, pharmaceutical, and food industries. In this study, optimization of the fermentation process for enhancement of β-carotene and biomass production by Exiguobacterium acetylicum S01 was achieved by employing statistical designs including the Placket-Burman design (PBD) and response surface methodology (RSM). Among the seven variables investigated by two levels in PBD, glucose, peptone, pH and temperature were indicated as crucial variables (p < 0.0001) for β-carotene and biomass productivity. Response surface methodology was further applied to evaluate the optimal concentrations of these four variables for maximum β-carotene and biomass productivity. The optimized medium contained glucose 1.4 g/L, peptone 26.5 g/L, pH 8.5, and temperature 30 °C, respectively. A significant increase in β-carotene (40.32 ± 2.55 mg/L) and biomass (2.19 ± 0.10 g/L) productivities in E. acetylicum S01 were achieved by using RSM, which was 3.47-fold and 2.36-fold higher in the optimized medium compared to the un-optimized medium. Further, the optimum fermentation condition in the 5-L bioreactor was achieved a maximal β-carotene yield of 107.22 ± 5.78 mg/L within 96 h. Moreover, the expression levels of carotenoid biosynthetic genes (phytoene desaturase (CrtI) and phytoene synthase (CrtB)) were up-regulated (2.89-fold and 3.71-fold) in E. acetylicum under the optimized medium conditions. Overall, these results suggest that E. acetylicum S01 can be used as a promising microorganism for the commercial production of β-carotene.
Keywords: Biotechnology, Food science, Microbiology, Food technology
1. Introduction
Carotenoids are yellow to red colored pigments synthesized through the terpenoid biosynthetic pathway. They are most plentiful in microorganisms and plants, which provide several biological functions such as in photoprotection, as light harvesting molecules, and membrane stabilizers (Britton et al., 2004). The other important role of certain carotenoids is to provide precursors of vitamin A, scavenging free radicals and enhancing in vitro antibody production (Vilchez et al., 2011). In particular, evidence from epidemiological studies has shown that a high dietary intake of β-carotene and other carotenoids prevent the development of colorectal cancers, age-related macular degeneration, regulation of lipid metabolism and also reduce cardiovascular diseases through their roles as antioxidants (Vilchez et al., 2011; Pal et al., 2012). Moreover, a few carotenoids are commonly used commercially as nutritional supplements (lutein), pharmaceutical (β-carotene), food colorants (canthaxanthin) and food supplements (astaxanthin) for humans as well as aquatic animals (Baron et al., 2008). Recently, the global market for carotenoids is expanding and was US$1.5 billion in 2017 (McWilliams, 2018). The demand for carotenoids is currently provided through chemical synthesis. However, the utility of these carotenoids in food and pharmaceutical applications is questioned because of their toxicity (Misawa, 2011).
In this perspective, the commercial production of microbial carotenoids using microorganisms has received interest owing to its high efficiency to produce diverse the nature of metabolites during fermentation (Venil et al., 2013). A number of investigations became available over the last few decades on carotenoids produced by microorganisms. These include bacterial strains belonging to Bacillus, Exiguobacterium, Corynebacterium and Rhodococcus, the microalgae Haematococcus and Dunaliella, the fungus Blakeslea trispora and yeasts, among others of the genera Rhodotorula, Sporobolomyces, and Phaffia (Venil et al., 2013; Johnson and Schroeder, 1996; Nelis and DeLeenheer, 1991). Hitherto, few carotenoids in the genus Exiguobacterium have been reported (e.g., β-carotene produced by Exiguobacterium profundum PHM11 (Patel et al., 2018), xanthophylls, produced by Exiguobacterium aurantiacum FH (Sasidharan et al., 2013), and astaxanthin, produced by Exiguobacterium sp. (Shatila et al., 2013). They all have great commercial potential for food and biopharmaceutical applications.
Currently, remarkable achievements were made in the optimization of medium composition and the optimal range of media turned out to be a critical factor accountable for the commercial success of the fermentation process. Several reports demonstrated that the synthesis of carotenoids is critically driven by media composition, pH, and temperature (Hamidi et al., 2014). In addition, the optimization of different variables for the production of desired metabolites are commonly species-specific and all the components in complex media are often not essential for the microbial synthesis of carotenoids (Johnson and Schroeder, 1996; Nelis and De-Leenheer, 1991). In this regard, fermentation media and physical factors should be upgraded in the fermentation technology for economic carotenoid production. Medium optimization by the conventional “one-factor-at-a-time” approach involves changing one factor while fixing the others at a specific level. The non-statistical “one-factor-at-a-time” method is laborious and excessively time-consuming and also fails to determine the interactions among the variables being studied (Das et al., 2015). These drawbacks of a non-statistical optimization process could be overcome by employing statistical tools. The Plackett-Burman design (PBD) is the most commonly applied method to identify the crucial factors among a large number of factors. Only the most effective factors with positive significance can be selected for further optimization. Others with less significance or high negative effect on response value may be omitted in further optimizations (Das et al., 2015). Response surface methodology coupled with the central composite rotatable design (RSM-CCRD) has been extensively used to study the interaction between factors and also identifying optimal conditions of factors for a desirable response (Plackett and Burman, 1946). Further, RSM fits the experimental response(s) to a quadratic function, assesses the relationships between the response(s) and independent variables, and defines the effect of the independent variables, alone or in combination, in the processes. The methodology has been utilized successfully to optimize the composition of microbiological media, improve fermentation processes and for developing desired products (Liong and Shah, 2005). Nevertheless, studying the optimization of fermentation conditions and medium composition in the production of carotenoids appears as a promising way to enhance the commercial production of desired metabolites. However, the optimization of β-carotene production by Exiguobacterium acetylicum S01 using statistical design has not yet been studied extensively but requires further detailed exploration. We have previously reported the isolation and functional characterization of pigmented probiotic strain E. acetylicum S01 (Jinendiran et al., 2017). Hence, the present study aimed to investigate the effects of medium components (MCs) and essential physical factors (PFs) on β-carotene and biomass productivity and additionally to investigate a suitable medium for maximizing the β-carotene production by E. acetylicum S01 based on a sequential statistical experimental design approach.
2. Materials and methods
2.1. Microorganism and fermentation conditions
The potential probiotic bacterium E. acetylicum S01 (accession number KU058431.1) was isolated from a soil sample collected from the Sathaiyar river basin in the Madurai region, Tamil Nadu, India as described previously (Jinendiran et al., 2017). It was cultured and maintained on nutrient agar (HiMedia, AS061) slant at 4 °C and sub-cultured monthly. Submerged fermentation was performed in nutrient broth with the following composition: peptone 10 g/L, beef extract 10 g/L, starch 10 g/L, sodium chloride 5 g/L and initial pH was adjusted to 7.0. Briefly, 1% of seed culture was inoculated from an 18 h log phase culture into 250 mL Erlenmeyer flask comprising 50 mL culture medium and incubated on a rotary incubator shaker (120 rpm) at 37 °C for 96 h.
2.2. Screening of MCs and PFs by using classical OFAT experiments
The conventional one-factor-at-a-time (OFAT) optimization approach was applied to evaluate the effects of MCs (sucrose, glucose, and glycerol) and PFs (pH and temperature) on β-carotene and biomass production (BP) by E. acetylicum S01. The starch was replaced with sucrose, glucose, and glycerol at a concentration of 0.5% in the fermentation medium as a sole carbon source. Similarly, the beef extract was replaced with yeast extract, soya peptone, and meat peptone, at a concentration of 1% in the culture medium as a sole nitrogen source. Likewise, sodium chloride was replaced with magnesium chloride; ferric citrate, sodium sulphate, and calcium chloride were used at 0.85%. In order to optimize the effect of different temperatures from 25, 35, 40, 45 and 50 °C and pH values from 6, 7, 8, 9, and 10 1N NaOH was used. The amounts of substrates contributing to a maximum production of β-carotene and biomass were further optimized concordant to the statistical experimental design depicted below. All experiments were expressed in three biological repeats and mean values were used for the analysis.
2.3. Statistical optimization by Plackett–Burman and RSM-CCRD
For identifying the significant factors for β-carotene and BP production, different medium components (peptone, beef extract, glucose, MgCl2, NaCl) and physical factors (pH, temperature) were assessed using Plackett–Burman experimental design. A total number of runs to be obtained according to PB are k +1 where k is the number of factors. For each variable was evaluated at two levels: low (-1) and high (+1), and the centre level (0) was detailed in Table 1. The effect of each factor on each response was observed using the following equation (Eq. 1).
| (1) |
Table 1.
Level of media components as well as important parameters, their codes, and levels involved in PBD design criterion.
| Code | Factor (units) | -1 | 0 | +1 |
|---|---|---|---|---|
| XA | NaCl (g/L) | 1 | 5 | 9 |
| XB | Beef extract (g/L) | 2 | 10 | 18 |
| XC | Peptone (g/L) | 2 | 10 | 18 |
| XD | MgCl2 (g/L) | 1 | 5 | 9 |
| XE | Glucose (g/L) | 2 | 10 | 18 |
| XF | pH | 4 | 7 | 10 |
| XG | Temperatures (o C) | 25 | 32.5 | 40 |
Where, Y is the response, β0 represents the model intercept, βi is the linear coefficient determination and Xi is the independent variable. The regression analysis, determining the variables with confidence levels above 95% (p < 0.05) were considered as influencing β-carotene and BP production significantly.
RSM was applied to optimize the screened variables for enhanced β-carotene and biomass production by applying the central composite rotatable design (CCRD). A full factorial central composite rotatable design of 24 = 16 plus 6 centre points and (2×4 = 8) star point conducting to a total of 30 experimental runs were performed. The variables were used at five experimental levels (-α, -1, 0, +1 and +α) each as shown in Table 2. The interrelationship among the coded values and actual values are described below equation:
| (2) |
Table 2.
Coded values of independent variables.
| Independent variables | Code levels |
||||
|---|---|---|---|---|---|
| -2.38 (-α) | -1 | 0 | +1 | +2.38 (+α) | |
| Glucose (g/L) | 1 | 1.5 | 2 | 2.5 | 3.0 |
| Peptone (g/L) | 1.0 | 9.5 | 18 | 26.5 | 35 |
| pH | 7.0 | 8.5 | 10 | 11.5 | 13 |
| Temperature (°C) | 20 | 30 | 40 | 50 | 60 |
Where, Xi, the dimensionless value of independent variables, xi, the real value of independent variables, xcp, the real value of independent variables at the centre point, and Δxi is the step change of the real value of the variable representing to a variation of a unit for dimensionless value of the variables. The β-carotene and BP production were taken as the responses. The relationship of the independent variables, their interactions and the responses were computed by the second-order polynomial model was expressed in the quadratic equation:
| (3) |
Where, Y is the predicted response, β0 is intercept term, βi is linear coefficient, βii is quadratic coefficient and βij is interactive coefficient and Xi and Xj represent the independent variables in the form of coded values. All statistical analyses were performed employing Design Expert Software program (Version 10, State-Ease Inc., Minneapolis, Minnesota, USA). The three-dimensional response surface plots were plotted by varying the concentrations of two variables and keeping the concentrations of other variables at zero level. Optimization of β-carotene and BP production was further confirmed by validating the responses obtained from the optimized medium S01 and an un-optimized medium S01.
2.4. Production of β-carotene in a bioreactor
In order to validate the optimized fermentation medium for β-carotene production at large-scale, the experiments were performed in a 5.0 L (3.5-L working volume) stirred tank bioreactor (Bioflow 310, New Brunswick Scientific, USA). Fermentation was carried out using an optimized medium with the initial pH of 8.5 after sterilizing the medium in situ. After sterilization, 1% of the log phase seed culture was aseptically transferred into a sterile medium and the bioreactor was kept at 30 °C. The pH was maintained constant at 8.5 and the impeller speed was maintained at 250 rpm. The dissolved oxygen content of the fermented broth was assessed using a dissolved oxygen electrode (Metter-Toledo International, Switzerland). After 96 h incubation, the fermented culture was removed for analysis of the β-carotene yield.
2.5. Determination of biomass and β-carotene productivity
The fermentation flasks were removed at suitable time intervals and the β-carotene content was determined following the procedure described by Davis (1949). After cultivation, the cells from 50 mL of culture broth were harvested by centrifugation at 6500×g for 10 min and washed twice in phosphate buffer saline (1X PBS, pH 7.2). In order to extract β-carotene, obtained biomass of E. acetylicum S01 was ground by mortar and pestle using methanol: acetone (7:3, v/v). The organic layer was then collected by centrifugation at 6500×g for 10 min. The extracted carotenoid fraction was filtered, evaporated in the dark and dissolved in methanol. An absorbance of β-carotene was measured at 450 nm by a spectrophotometer (UV-1800 series, Shimadzu, Japan) and the obtained OD was estimated by its original standard. A standard graph of β-carotene was plotted for its concentration varied from 100 to 500 μg/mL. The biomass productivity was determined using the below-mentioned formula (Naziri et al., 2014).
| Biomass productivity (g/L) = [Final biomass DCW (g)–Initial biomass DCW (g)] | (4) |
2.6. RNA isolation and gene expression analysis
In order to evaluate the carotenoid biosynthetic gene expression, E. acetylicum S01 was grown in basal media (un-optimized) and production media (RSM optimized) on a rotary incubator shaker (120 rpm) at 30 °C for 96 h. Total RNA was extracted from the early stationary phase bacterial culture using a TRIzol reagent (TaKara, Japan) following the manufacturer's protocol. Isolated total RNA was treated with DNAase and quantified using a NanoDrop-1000 (Thermo Fisher Scientific, USA). One microgram of DNAase treated RNA was used for cDNA synthesis using a RevertAid cDNA synthesis kit (Thermo Fisher Scientific, USA) according to the manufacturer's protocol. Primers were designed from published coding sequences (CDS) for carotenoid biosynthesis genes (phytoene desaturase (CrtI) and phytoene synthase (CrtB) in E. acetylicum DSM 20416 using the Invitrogen primer designing tool at standard parameters. Primer sequences used in this study were shown in Table 3. PCR reactions were carried out in an Applied Biosystems (ABI) Veriti Thermal Cycler (ABI, USA) under the following conditions: initial denaturation at 94 °C for 5 min; followed by 30 cycles of denaturation at 94 °C for 1 min, annealing at 57 °C for 40 sec (CrtB), 62 °C for 40 sec (CrtI) and 60 °C for 35 sec followed by elongation at 72 °C for 1 min. Pyrroline-5-carboxylate reductase gene (ProC) served as a housekeeping gene in order to normalize the relative gene expression levels. The sqRT–PCR analysis was carried out in three biological repeats in each experimental triplicate of each gene.
Table 3.
List of primers used for sqRT-PCR amplification.
| Gene | Genebank accession number | Amplicon size (bp) | Anneal | Primer |
|---|---|---|---|---|
| CrtB | NZ_JNIR01000001.1 | 283 | 57 °C–40s | FWD: 5′- TCGATGATACCGTCGATGAA -3′ |
| REV: 5′- GCAAGGATGGGTAACAGCAT -3′ | ||||
| CrtI | NZ_JNIR01000001.1 | 244 | 62 °C–40s | FWD: 5′- AAATTATGATGCGCGAGTCC -3′ |
| REV: 5′- CCGTTGAACAAATCGTCCTT -3′ | ||||
| ProC | CP001615.1 | 228 | 60 °C–35S | FWD: 5′- GCCGACATCGTCTTTGCATC -3′ |
| REV: 5′- GACACCGTTACTCGTCTCGG -3′ |
CrtB: phytoene synthase; CrtI: phytoene desaturase; ProC: pyrroline-5-carboxylate reductase; pb: basepair.
2.7. Statistical analysis
Data are expressed as mean ± standard deviation (SD). All results were subjected to a one-way analysis of variance (ANOVA). Multiple comparisons were performed with Tukey's test to analyze the difference between the means of each group. The graph pad prism statistical software (Windows-based) was used for the statistical analysis of the data (San Diego, CA, USA). The significance levels were accepted if the null hypothesis was rejected at p < 0.05.
3. Results
3.1. Medium optimization using the conventional OFAT method
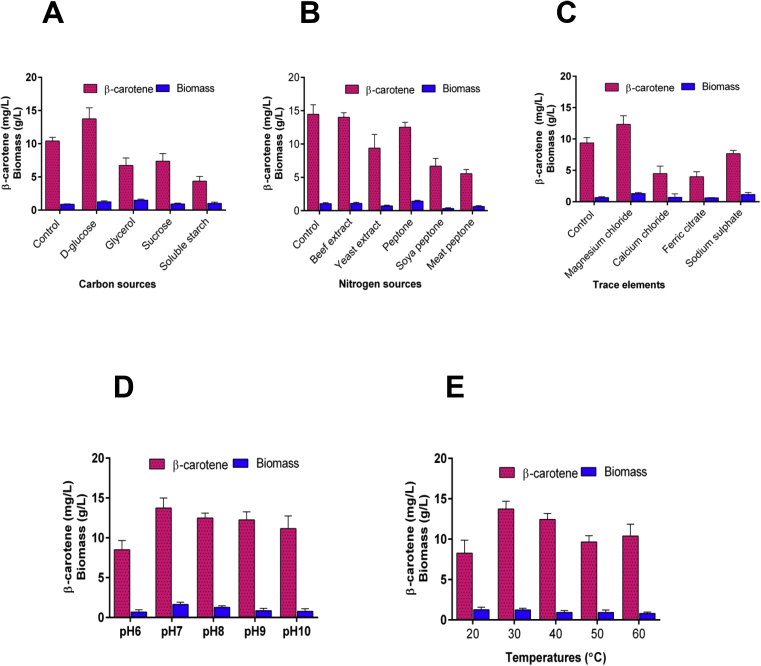
The OFAT optimization approach was efficiently used to evaluate the effects of various factors, including carbon, nitrogen sources, trace elements, and physical factors on biomass and β-carotene production in E. acetylicum S01. The results revealed that glucose (13.76 ± 2.22 mg/L) gave a maximum yield of β-carotene production (Fig. 1A) compared to sucrose (7.39 ± 1.12 mg/L). In addition, glycerol and starch were incorporated in the basal medium as a sole carbon source. Glycerol did not have a major influence on β-carotene production, whereas, it provides a significantly higher BP. The influence of various nitrogen sources on biomass and β-carotene productivities were depicted in Fig. 1B. Among the nitrogen sources, beef extract gave maximum β-carotene production (14.02 ± 0.59 mg/L) and BP (1.10 ± 0.11 g/L); whereas β-carotene production (9.38 ± 1.83 mg/L) and BP (0.71 ± 0.14 mg/L) were less in yeast extract (Fig. 1B). In addition, soya peptone and meat peptone affected overall productivities less compared to the control. MgCl2 (5 g/L) provided a significantly higher β-carotene production (12.34 ± 1.31 mg/L), whereas β-carotene production (7.67 ± 0.44 g/L) was less in sodium sulphate at 7.5 g/L (Fig. 1C). Additionally, ferric citrate and calcium chloride had neither a major effect on the production of β-carotene nor on biomass (Fig. 1C). A pH of 7 allowed maximum β-carotene production (13.74 ± 1.15 mg/L), whereas a pH of 6 and 10 were not favourable for its BP (Fig. 1D). Similarly, a temperature of 30 °C was considerably enhanced by the production of β-carotene (14.74 ± 1.02 mg/L), whereas a temperature of 50 °C decreased BP (Fig. 1E).
Fig. 1.
Effects of different medium components and physical factors on biomass (g/L) and β-carotene (mg/L) productivities in E. acetylicum S01. Data are shown as mean ± SD (n = 3). Bar graphs showing the effects of carbon source (A), nitrogen source (B), trace element (C), pH (D), and temperature (E).
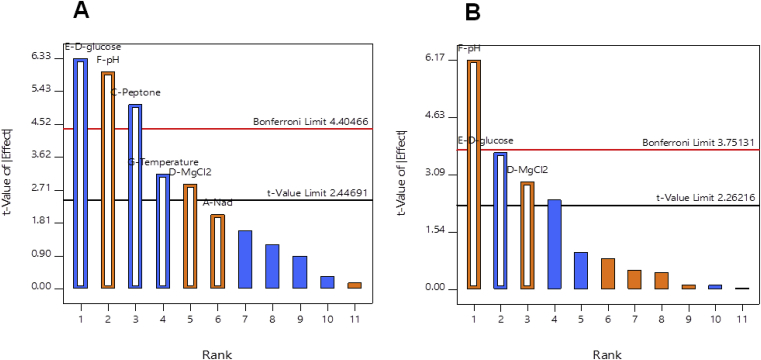
3.2. Optimization of fermentation medium using Placket-Burman design
Plackett-Burman analysis was used to identify which variables provided significant effects on β-carotene and biomass productivity. The Placket-Burman design matrix and results were shown in Table 4. The effects of the variables on significant responses were shown in Fig. 2. The PB model showed significant responses with a p-value of 0.0004 (β-carotene) and 0.0022 (BP). The regression analysis and the ANOVA for the PBD were shown in Table 5. The coefficient of determination (R2) of 0.9612 (BP) and 0.8824 (β-carotene) demonstrated that 96.12% and 88.24% of the variability in the responses were explained by the model. The ANOVA results revealed that glucose (p = 0.0014), pH (p = 0.0019), peptone (p = 0.0039), temperature (p = 0.0253), MgCl2 (p = 0.0346) and NaCl (p = 0.0962) had low p-values in BP (Table 5). In particular, the pH showed a significant impact (p = 0.0003) on β-carotene production than provided by glucose (p = 0.0062), and MgCl2 (p = 0.0202), respectively, as shown in Table 5. The following first-order polynomial model was demonstrated by regression:
| BP = - 0.700 + [0.137 * X1] – [0.339 * X3] + [0.1931 * X4] – [0.424 * X5] + [0.400 * X6] – [0.211 * X7]. | (5) |
| β-carotene = + 0.470 + [0.149 * X4] - [0.190 * X5] + [0.320 * X6] | (6) |
Where, X1 = NaCl, X3 = peptone, X4 = MgCl2, X5 = glucose, X6 = pH and X7 = temperature are variables. The other factors without significant influence (p > 0.05) on both responses were not included in Eqs. (5) and (6). The regression coefficient of the selected factors with a significant effect on BP and β-carotene yield (Eqs. (5) and (6)) indicated that the four variables, including glucose, peptone, pH, and temperature had critical effects on overall productivity. Accordingly, based on the results from PBD experiment, most crucial variables (glucose, peptone, pH and temperature) with positive effect were further investigated by a CCRD experiment to find the optimal range of the variables and their interactive effect(s).
Table 4.
Plackett–Burman design matrix of independent variables and their corresponding experimental and predicted yields of biomass (g/L) and β-carotene (mg/L) productivities in E. acetylicum S01.
| Runs | Factors |
Biomass (g/L) |
β-carotene (mg/L) |
||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| A | B | C | D | E | F | G | Experimentala | Predicted | Experimentala | Predicted | |
| 1 | 9 | 18 | 2 | 9 | 18 | 10 | 25 | 1.89 ± 0.18 | 1.94 ± 1.20 | 4.40 ± 0.17 | 6.88 ± 3.68 |
| 2 | 1 | 18 | 18 | 1 | 18 | 10 | 40 | 0.03 ± 0.01 | 0.03 ± 0.02 | 2.67 ± 0.39 | 3.66 ± 1.96 |
| 3 | 9 | 2 | 18 | 9 | 2 | 10 | 40 | 1.31 ± 0.82 | 1.08 ± 0.67 | 19.96 ± 1.08 | 18.61 ± 8.35 |
| 4 | 1 | 18 | 2 | 9 | 18 | 4 | 40 | 0.04 ± 0.01 | 0.61 ± 0.38 | 2.16 ± 0.28 | 1.67 ± 0.89 |
| 5 | 1 | 2 | 18 | 1 | 18 | 10 | 25 | 0.4 ± 0.01 | 0.08 ± 0.05 | 2.63 ± 0.17 | 3.66 ± 1.96 |
| 6 | 1 | 2 | 2 | 9 | 2 | 10 | 40 | 1.18 ± 0.91 | 2.76 ± 1.70 | 10.42 ± 0.09 | 15.61 ± 8.35 |
| 7 | 9 | 2 | 2 | 1 | 18 | 4 | 40 | 0.03 ± 0.09 | 0.04 ± 0.02 | 0.42 ± 0.07 | 0.89 ± 0.47 |
| 8 | 9 | 18 | 2 | 1 | 2 | 10 | 25 | 3.32 ± 0.43 | 5.65 ± 3.48 | 10.29 ± 1.56 | 8.31 ± 4.44 |
| 9 | 9 | 18 | 18 | 1 | 2 | 4 | 40 | 0.04 ± 0.01 | 0.07 ± 0.04 | 1.43 ± 0.35 | 2.02 ± 1.08 |
| 10 | 1 | 18 | 18 | 9 | 2 | 4 | 25 | 0.16 ± 0.18 | 0.24 ± 0.14 | 1.92 ± 0.78 | 3.79 ± 2.03 |
| 11 | 9 | 2 | 18 | 9 | 18 | 4 | 25 | 0.04 ± 0.17 | 0.06 ± 0.03 | 1.65 ± 0.26 | 1.67 ± 0.89 |
| 12 | 1 | 2 | 2 | 1 | 2 | 4 | 25 | 0.64 ± 0.08 | 0.47 ± 0.29 | 1.88 ± 0.65 | 2.02 ± 1.08 |
| 13 | 5 | 10 | 10 | 5 | 10 | 7 | 32.5 | 0.68 ± 0.03 | 0.27 ± 0.16 | 11.75 ± 0.46 | 3.73 ± 1.99 |
All media components (g/L): A – NaCl; B – beef extract; C – peptone; D – magnesium chloride; E –glucose; F – pH; G – temperature (oC).
Mean of values of three biological repeats and the data represents the mean ± SD (n = 3).
Fig. 2.
Pareto chart analysis: the effect of media component on biomass (A) and β-carotene (B) productivities were represented by t-value.
Table 5.
Regression analysis and analysis of variance (ANOVA) for the experimental results of the Plackett–Burman design first-order model response biomass and β-carotene.
|
Response to biomass productivity (g/L) | |||||||
|
Factora |
Coefficient estimation |
DFb |
Standard error |
Sum of squares |
F value |
Pro < F |
Con, level (%) |
| Model | -0.700 | 6 | 0.067 | 6.68 | 20.64 | 0.0022 | 99.9 |
| A | 0.137 | 1 | 0.067 | 0.225 | 4.18 | 0.0962 | 99.8 |
| C | -0.339 | 1 | 0.067 | 1.39 | 25.67 | 0.0039 | 99.9 |
| D | 0.193 | 1 | 0.067 | 0.447 | 8.29 | 0.0346 | 99.9 |
| E | -0.424 | 1 | 0.067 | 2.17 | 40.12 | 0.0014 | 99.9 |
| F | 0.400 | 1 | 0.067 | 1.92 | 35.64 | 0.0019 | 99.9 |
| G |
-0.211 |
1 |
0.067 |
0.536 |
9.93 |
0.0253 |
99.5 |
|
Response to β-carotene productivity (mg/L) | |||||||
|
Factora |
Coefficient estimation |
DFb |
Standard error |
Sum of squares |
F-value |
Pro < F |
Con, level (%) |
| Model | 0.470 | 3 | 0.051 | 1.93 | 20.01 | 0.0004 | 99.9 |
| D | 0.149 | 1 | 0.051 | 0.269 | 8.35 | 0.0202 | 99.9 |
| E | -0.190 | 1 | 0.051 | 0.436 | 13.55 | 0.0062 | 99.9 |
| F | 0.320 | 1 | 0.051 | 1.23 | 38.13 | 0.0003 | 99.9 |
A, NaCl; C, peptone; D, MgCl2; E, glucose; F, pH; G, temperature.
Degree of freedom, R2 = 0.9612 (Biomass), R2 = 0.8824 (β-carotene).
3.3. Optimization of the fermentation medium by RSM
The RSM was used to optimize medium compositions (glucose and peptone) and physical factors (pH and temperature) for enhancing biomass and β-carotene production using a central composite rotatable design (CCRD). The experimental design and respective yields of each response are shown in Table 6. Our experimental results were close to the values predicted by the model for biomass and β-carotene productivity (Table 6). Adequacy and fitness of biomass and β-carotene productivity were determined by ANOVA and the regression coefficients linear and quadratic interaction of all variable, along with p-values at <95% (p < 0.05) confidence levels were shown in Tables 7 and 8. The model was highly significant with a very low p-value (Prob < F) (0.0001). The p-values were used to identify the significance of each coefficient and the pattern of mutual interactions between the test variables. The estimated coefficient and the corresponding p-values demonstrate that among the tested variables used in the study, B (peptone), C (pH), D (temperature), B × C (peptone × pH), B × D (peptone × temperature), C × D (pH × temperature) provided significant model terms. pH and temperature showed a significant effect (p < 0.0001) on β-carotene production, followed by peptone (p < 0.01). A corresponding second-order response model (Eqs. (7) and (8)) was repressed by the following regression analysis:
| BP = – 0.1415 + [0.0692 * A] – [0.0333 * B] – [0.1195 * C] – [0.4368 * D] – [0.0583 * AB] – [0.0100 * AC] – [-0.0481* AD] + [+0.1347 * BC] – [-0.2099 * BD] – [-0.1020 * CD] + [+0.0109 * A2] – [-0.2117 * B2] – [-0.1590 * C2] – [-0.1585 * D2] | (7) |
| β-carotene = + 0.813 + [0.016 * A] + [0.064 * B] – [0.158 * C] – [0.334 * D] + [0.023 * AB] – [0.016 * AC] + [0.008 * AD] – [0.013 * BC] – [0.104* BD] + [0.104* CD] – [0.014 * A2] – [0.003 * B2] – [0. 023* C2] – [0.112 * D2] | (8) |
Table 6.
Central composite rotatable design matrix of independent variables and their corresponding experimental and predicated yields of biomass and β-carotene productivities.
| Runs | Media concentration (g/L) |
Biomass (g/L) |
β-carotene (mg/L) |
|||||
|---|---|---|---|---|---|---|---|---|
| A | B | C | D | Experimentala | Predicted | Experimentala | Predicted | |
| 1 | 1.5 | 9.5 | 8.5 | 30 | 0.78 ± 0.02 | 0.65 ± 0.25 | 13.34 ± 0.10 | 12.38 ± 3.59 |
| 2 | 2.5 | 9.5 | 8.5 | 30 | 0.77 ± 0.02 | 0.71 ± 0.32 | 15.46 ± 0.18 | 12.44 ± 3.61 |
| 3 | 1.5 | 26.5 | 8.5 | 30 | 0.96 ± 0.04 | 0.88 ± 0.31 | 24.99 ± 0.018 | 25.82 ± 2.49 |
| 4 | 2.5 | 26.5 | 8.5 | 30 | 1.34 ± 0.14 | 1.11 ± 0.50 | 39.75 ± 1.15 | 36.11 ± 3.32 |
| 5 | 1.5 | 9.5 | 11.5 | 30 | 0.1 ± 0.09 | 0.15 ± 0.06 | 4.84 ± 0.28 | 4.25 ± 1.13 |
| 6 | 2.5 | 9.5 | 11.5 | 30 | 0.06 ± 0.04 | 0.10 ± 0.04 | 2.66 ± 0.18 | 3.91 ± 1.04 |
| 7 | 1.5 | 26.5 | 11.5 | 30 | 0.84 ± 0.48 | 1.44 ± 0.65 | 7.17 ± 0.41 | 7.26 ± 1.94 |
| 8 | 2.5 | 26.5 | 11.5 | 30 | 1.37 ± 0.08 | 1.17 ± 0.53 | 8.99 ± 0.32 | 8.33 ± 2.23 |
| 9 | 1.5 | 9.5 | 8.5 | 50 | 0.18 ± 0.07 | 0.29 ± 0.13 | 2.75 ± 0 | 2.96 ± 0.79 |
| 10 | 2.5 | 9.5 | 8.5 | 50 | 0.5 ± 0.35 | 0.56 ± 0.25 | 2.72 ± 0.7 | 2.93 ± 0.78 |
| 11 | 1.5 | 26.5 | 8.5 | 50 | 0.02 ± 0 | 0.02 ± 0.01 | 2.5 ± 0.54 | 1.72 ± 0.46 |
| 12 | 2.5 | 26.5 | 8.5 | 50 | 0.06 ± 0.05 | 0.05 ± 0.02 | 2.79 ± 0.50 | 2.12 ± 0.56 |
| 13 | 1.5 | 9.5 | 11.5 | 50 | 0.6 ± 0.03 | 0.07 ± 0.03 | 1.62 ± 0.28 | 1.87 ± 0.50 |
| 14 | 2.5 | 9.5 | 11.5 | 50 | 0.05 ± 0.03 | 0.07 ± 0.03 | 1.95 ± 0.43 | 1.99 ± 0.53 |
| 15 | 1.5 | 26.5 | 11.5 | 50 | 0.06 ± 0.03 | 0.04 ± 0.01 | 0.90 ± 0.16 | 1.34 ± 0.35 |
| 16 | 2.5 | 26.5 | 11.5 | 50 | 0.04 ± 0.02 | 0.05 ± 0.02 | 1.54 ± 0.16 | 1.78 ± 0.47 |
| 17 | 1 | 18 | 10 | 40 | 0.68 ± 0.12 | 0.62 ± 0.28 | 4.39 ± 0.01 | 5 ± 1.34 |
| 18 | 3 | 18 | 10 | 40 | 1.29 ± 0.17 | 0.98 ± 0.44 | 5.90 ± 0.05 | 5.69 ± 1.52 |
| 19 | 2 | 1 | 10 | 40 | 0.24 ± 0.06 | 0.10 ± 0.04 | 5.03 ± 0.75 | 5.31 ± 1.42 |
| 20 | 2 | 35 | 10 | 40 | 0.06 ± 0.08 | 0.09 ± 0.04 | 6.31 ± 0.32 | 6.56 ± 1.75 |
| 21 | 2 | 18 | 7 | 40 | 0.38 ± 0.64 | 0.31 ± 0.14 | 6.04 ± 0.10 | 9.27 ± 2.48 |
| 22 | 2 | 18 | 13 | 40 | 0.1 ± 0.01 | 0.08 ± 0.03 | 3.65 ± 0.20 | 2.62 ± 0.70 |
| 23 | 2 | 18 | 10 | 20 | 1.92 ± 0.12 | 1.08 ± 0.48 | 7.54 ± 0.16 | 9.65 ± 2.58 |
| 24 | 2 | 18 | 10 | 60 | 0.02 ± 0 | 0.02 ± 0.01 | 0.56 ± 0.02 | 0.48 ± 0.12 |
| 25 | 2 | 18 | 10 | 40 | 0.66 ± 0.07 | 0.80 ± 0.36 | 6.61 ± 0.18 | 6.75 ± 1.80 |
| 26 | 2 | 18 | 10 | 40 | 0.68 ± 0.08 | 0.80 ± 0.36 | 6.23 ± 0.40 | 6.75 ± 1.80 |
| 27 | 2 | 18 | 10 | 40 | 0.78 ± 0.04 | 0.80 ± 0.36 | 6.42 ± 0.12 | 6.75 ± 1.80 |
| 28 | 2 | 18 | 10 | 40 | 0.77 ± 0.08 | 0.80 ± 0.36 | 6.42 ± 0.63 | 6.75 ± 1.80 |
| 29 | 2 | 18 | 10 | 40 | 0.71 ± 0.09 | 0.80 ± 0.36 | 6.80 ± 0.03 | 6.75 ± 1.80 |
| 30 | 2 | 18 | 10 | 40 | 0.77 ± 0.37 | 0.80 ± 0.36 | 6.60 ± 0.04 | 6.75 ± 1.80 |
Mean of three replicate measurements; data are shown mean ± SD (n = 3). A, glucose; B, Peptone, C, pH and C, Temperature.
Table 7.
Analysis of variance (ANOVA) for the experimental results in the central composite rotatable design quadratic model, response biomass (g/L).
| Factora | Coefficient estimation | DFb | Standard error | Sum of squares | F value | Pro < F | Con, level (%) |
|---|---|---|---|---|---|---|---|
| Model | -0.141 | 14 | 0.081 | 8.51 | 15.17 | <0.0001 | 99.9 |
| A | 0.069 | 1 | 0.040 | 0.114 | 2.87 | 0.1110 | 99.8 |
| B | -0.033 | 1 | 0.040 | 0.026 | 0.664 | 0.4276 | 99.6 |
| C | -0.119 | 1 | 0.040 | 0.342 | 8.56 | 0.0104 | 99.9 |
| D | -0.436 | 1 | 0.040 | 4.58 | 114.30 | <0.0001 | 99.9 |
| AB | -0.058 | 1 | 0.050 | 0.054 | 1.36 | 0.2619 | 99.7 |
| AC | 0.001 | 1 | 0.050 | 0.001 | 0.039 | 0.8449 | 99.2 |
| AD | -0.048 | 1 | 0.050 | 0.037 | 0.924 | 0.3516 | 99.6 |
| BC | 0.134 | 1 | 0.050 | 0.290 | 7.24 | 0.0167 | 99.9 |
| BD | -0.209 | 1 | 0.050 | 0.705 | 17.60 | 0.0008 | 99.9 |
| CD | -0.102 | 1 | 0.050 | 0.166 | 4.16 | 0.0594 | 99.9 |
| A2 | 0.010 | 1 | 0.038 | 0.003 | 0.082 | 0.7785 | 99.3 |
| B2 | -0.211 | 1 | 0.038 | 1.23 | 30.69 | <0.0001 | 99.9 |
| C2 | -0.159 | 1 | 0.038 | 0.693 | 17.32 | 0.0008 | 99.9 |
| D2 | -0.158 | 1 | 0.038 | 0.688 | 17.20 | 0.0009 | 99.9 |
A, glucose; B, Peptone; C, pH; D, Temperatures.
Degree of freedom, R2 = 0.9340.
Table 8.
Analysis of variance (ANOVA) for the experimental results in the central composite rotatable design quadratic model, response β-carotene (mg/L).
| Factora | Coefficient estimation | DFb | Standard error | Sum of squares | F value | Pro < F | Con, level (%) |
|---|---|---|---|---|---|---|---|
| Model | 0.813 | 14 | 0.052 | 4.12 | 17.71 | <0.0001 | 99.9 |
| A | 0.016 | 1 | 0.026 | 0.006 | 0.380 | 0.546 | 99.4 |
| B | 0.064 | 1 | 0.026 | 0.099 | 5.98 | 0.027 | 99.6 |
| C | -0.158 | 1 | 0.026 | 0.605 | 36.43 | <0.0001 | 99.9 |
| D | -0.334 | 1 | 0.026 | 2.68 | 161.67 | <0.0001 | 99.9 |
| AB | 0.023 | 1 | 0.032 | 0.008 | 0.510 | 0.485 | 99.7 |
| AC | -0.016 | 1 | 0.032 | 0.004 | 0.275 | 0.607 | 99.3 |
| AD | 0.008 | 1 | 0.032 | 0.001 | 0.075 | 0.787 | 99.4 |
| BC | -0.013 | 1 | 0.032 | 0.003 | 0.181 | 0.676 | 99.6 |
| BD | -0.104 | 1 | 0.032 | 0.175 | 10.58 | 0.005 | 99.4 |
| CD | 0.104 | 1 | 0.032 | 0.174 | 10.51 | 0.005 | 99.6 |
| A2 | -0.104 | 1 | 0.024 | 0.005 | 0.347 | 0.564 | 99.7 |
| B2 | -0.003 | 1 | 0.024 | 0.003 | 0.020 | 0.888 | 99.4 |
| C2 | -0.023 | 1 | 0.024 | 0.014 | 0.887 | 0.361 | 99.8 |
| D2 | -0.112 | 1 | 0.024 | 0.347 | 20.91 | 0.0004 | 99.9 |
A, glucose; B, Peptone; C, pH; D, Temperature.
Degree of freedom, R2 = 0.9429.
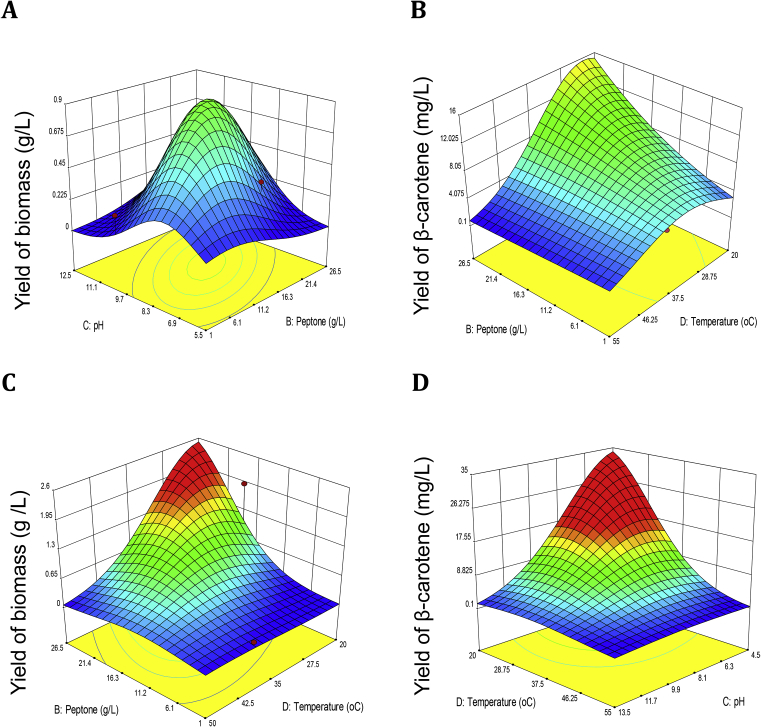
The fit of the model was also shown by the coefficient of determination R2 0.9340 and 0.9459, suggesting that 93.40% and 94.49% variability, respectively, in the responses were explained by the model. Consequently, the 3D response surface plot exhibited the interaction of pH vs. peptone (Fig. 3A), peptone vs. temperature (Fig. 3B, C) and temperature vs. pH (Fig. 3D), respectively. Based on these results, an optimized fermentation medium was composed of (g/L): glucose 1.4 g/L, peptone 26.6 g/L, and were set at pH 8.5 and 30 °C temperature, respectively. The maximum predicted response for biomass and β-carotene production was obtained and experimentally confirmed. The maximum production of biomass and β-carotene achieved experimentally using the optimized medium was 39.75 ± 1.15 mg/L and 1.92 ± 0.12 g/L, whereas the predicted values were 36.11 ± 3.32 mg/L and 1.08 ± 0.48 g/L, based on the results of the RSM regression study.
Fig. 3.
Three-dimensional (3-D) response surface plots representing the interactions of the two factors with biomass (g/L) and β-carotene (mg/L) productivities in E. acetylicum S01 has grown in the RSM-CCRD optimized medium. The 3-D surface plots of pH and peptone (A), peptone and temperature (B and C), and temperature and pH (E).
3.4. Model validation
In order to validate optimized medium suggested by RSM-CCRD, the experimental run 4 for maximum β-carotene production was scaled up to 1.5 L submerged shake flask cultures, using optimized fermentation medium containing an optimum concentration of glucose 1.4 g/L, peptone 26.6 g/L, pH 8.5 and temperature 30 °C, respectively. The β-carotene and biomass productivity were raised to 40.32 ± 2.55 mg/L (3.47-fold) and 2.19 ± 0.10 (2.36-fold) compared to an un-optimized medium (Table 9).
Table 9.
Validation of RSM optimized media.
| Responses | Before medium optimization | RSM optimized media | After optimization fold increase |
|---|---|---|---|
| Biomass g/L | 0.67 ± 0.088 | 2.19 ± 0.10 | 2.36-fold |
| Β-carotene mg/L | 11.02 ± 0.15 | 40.32 ± 2.55 | 3.47-fold |
3.5. Production of β-carotene at bioreactor scale
In order to confirm the possibility of using an optimized fermentation medium for β-carotene production at larger bioreactor scale, fermentation of E. acetylicum S01 was carried out using a 5-L bioreactor. In this study, an optimized medium supported the productivity of β-carotene at 107.22 ± 5.78 mg/L (3.09 ± 1.26 fold) than un-optimized medium condition. This indicates that the optimum culture conditions obtained by the RSM experimental design in submerged fermentations were supportive for the enhanced production of β-carotene in a bioreactor.
3.6. Expression levels of carotenoid biosynthetic genes in E. acetylicum S01
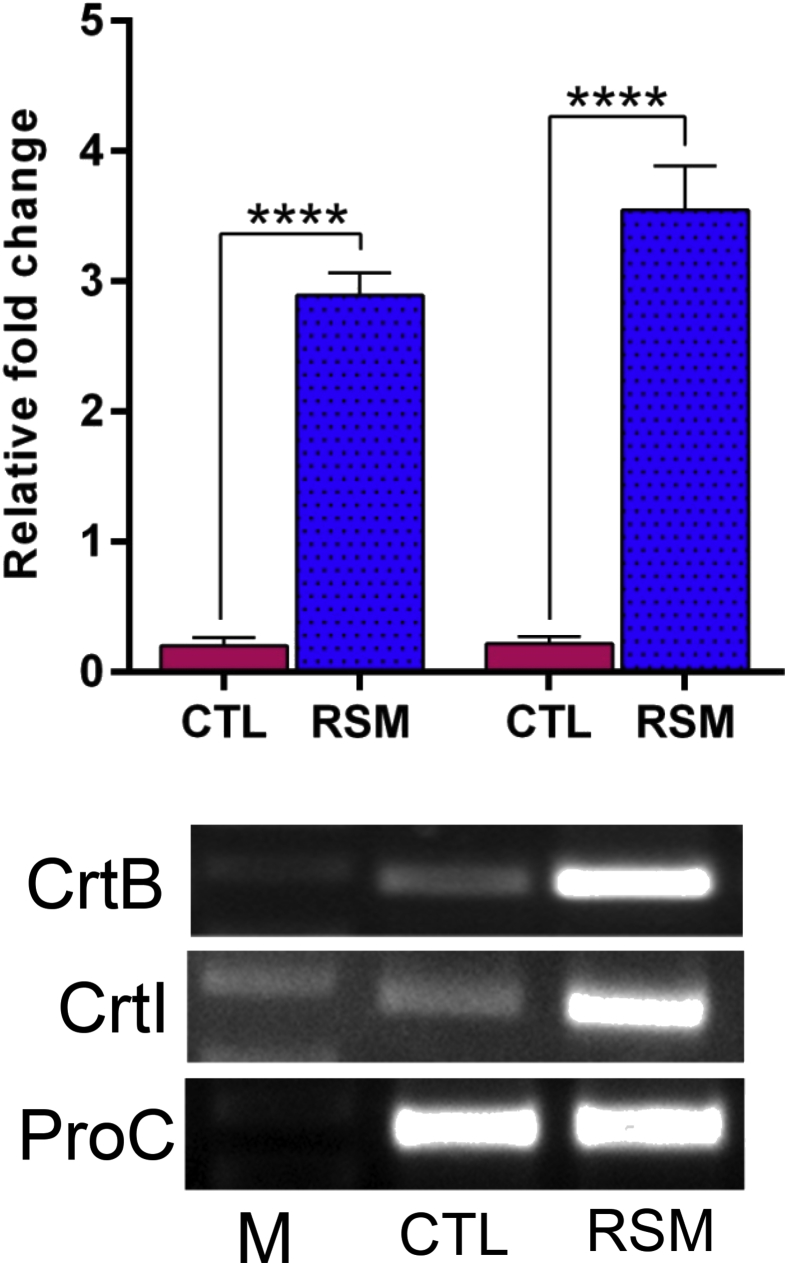
An expression level of genes in carotenoid biosynthesis under different culture conditions during an early stationary phase indicates the role of enzymes at particular culture conditions. The expression levels CrtI and CrtB were significantly (p = 0.0001) up-regulated, 2.89-fold and 3.71-fold in the RSM optimized condition compared with the un-optimized conditions (Fig. 4).
Fig. 4.
Gene expression level of carotenoid biosynthetic genes for the early stationary phase cultures of E. acetylicum S01 cultivated in un-optimized medium (control) and RSM optimized medium conditions for 96 h. Data represent the mean of the three independent biological repeats (n = 3). Bar graphs showing CrtI and CrtB gene expression in un-optimized medium (control) and RSM optimized medium conditions. ****p < 0.0001 indicates a significant difference between the un-optimized medium in response to RSM optimized medium conditions. Non-adjusted images provided in the supplementary material. M - DNA ladder (0.25 kb–10 kb), CTL - Un-optimized medium, RSM - RSM Optimized medium.
4. Discussion
Carotenoids are tetraterpenoid pigments with eight isoprene units and polyene chain conjugate conjugated double bonds. They have been used several therapeutic applications that include anti-inflammatory, anti-diabetic, and anticancer agents (Fiedor and Burda, 2014; Vilchez et al., 2011). Among microbial carotenoids, β-carotene is a vital antioxidant, peruser of vitamin A, and marketed food pigment, and various non-microbial and microbial organisms have a high potential for industrial applications. For β-carotene production by the green alga Dunaliella sp. are needed various stressful conditions such as intensive light, O2 and CO2 during the day and night cycle, as well as high salt concentrations (Ben-Amotz, 1999). Species belongings to the genera Blakeslea sp. are also has producing high β-carotene yields with several studies on the industrial production (Bhosale and Gadre, 2001; Choudhari and Rekha, 2008). Moreover, the production of β-carotene by mutants of the yeast R. glutinis and X. dendrorhous was also reported in previous studies (Pi et al., 2018).
Bacteria have the ability to grow commonly faster than fungi and microalgae. However, there are only a few bacteria that are able to synthesize β-carotene. Johnson and co-workers have reported a few bacterial strains that are capable to produce β-carotene and other carotenoids (Venil et al., 2013). Among the bacterial genera, Exiguobacterium sp. is well-known for the production of β-carotene, astaxanthin, and xanthophylls, respectively (Patel et al., 2018; Sasidharan et al., 2013; Shatila et al., 2013). In the present study, the one-factor-at-a-time optimization results suggested that the adequate concentration of glucose in the fermentation medium would facilitate the enhancement of β-carotene production by E. acetylicum S01, for the first time. A plausible explanation for the significant improvement in the β-carotene production that the strain utilizes the glucose can easily be absorbed in the biosynthetic pathway of β-carotene. Similarly, Sowmya and Sachindra (2015) supported our result that in the absence of any sugar in the production medium the carotenoids yield was low 115 ± 10.4 whereas it was increased in the presence of glucose (166.1 ± 13.33) by Flavobacteriacea sp. Choudhari and Singhal (2008) found increasing β-carotene (99 ± 2 mg/L) production by the Blakeslea trispora strain in the presence of glucose. Previously, Khodaiyan et al. (2007) reported that glucose was an important carbon source for canthaxanthin production in D. natronolimnaea HS-1. It has been documented that medium components contain a mixture of essential amino acids that are capable of enhancing the growth and carotenoid production by microbes (Sowmya and Sachindra, 2015; Wang et al., 2012). Among the nitrogen sources evaluated in this study, beef extract and peptone was found to be supportive of the production of biomass and β-carotene in E. acetylicum S01. Similarly, Wang and co-workers also found a β-carotene yield of 2.45 μg/mL by S. marcescens RB3 at optimal cultivation conditions in the presence of 2% (w/v) peptone and 0.3% (w/v) beef extract, respectively (Wang et al., 2012). Sun et al. (2004) studied the production of carotenoids from P. rhodozyma and found that an addition of 1% (w/v) peptone and beef extract in the medium resulted in maximum carotenoid production. Taken together, both carbon and nitrogen sources in the fermentation medium can improve the growth and β-carotene production of E. acetylicum S01 through the biosynthesis of metabolic pathways.
The present study also investigated plausible the effects of various trace elements in the production of β-carotene. Magnesium chloride increased the β-carotene yield compared to other elements. It could be clarified by hypothesizing a plausible activation and inhibition mechanism by tested metal ions on unique carotenogenic enzymes (Buzzini and Martini, 2005; Gerhard, 2001). Magnesium plays a critical role as a co-factor of different enzymes. Our finding is supported by an earlier study, where divalent cation salts (Mg2+) were reported to show the maximum influence on carotenoid production from R. glutinis by stimulating enzymes necessary for the biosynthesis of carotenoids (Bhosale and Gadre, 2001). Our results suggest that Mg2+ is necessary for enhanced growth and β-carotene production by E. acetylicum S01.
An earlier study showed enhanced β-carotene production at strong alkaline conditions could only be achieved by the pellet establishment with reduced mass transfer limitations (Seon-Won et al., 1996). In the present study, both biomass and β-carotene productivity were increased at different pH conditions. A plausible explanation of the significant increase in the β-carotene and biomass production by E. acetylicum S01 under different pH conditions could be that neutral pH is essential for normal growth and alkaline pH is important for carotenoid biosynthesis under stress conditions. A previous study showed that optimal physical factors (pH 7 and 30 °C) are important for the production of zeaxanthin and was achieved by a sponge-inhabiting bacterium Sphingomonas sp. (Thawornwiriyanun et al., 2012). Shatila and collaborators reported a total cellular pigment of 534.51 μg/g dry cell weight when E. aurantiacum FH was grown in LB medium (pH 7) at 30 °C under shake flask culture (Shatila et al., 2013). Korumilli and Susmita (2014) reported that B. clausii yielded a maximum pigment production of 107 ± 1.2 mg/3g dry biomass at optimal culture conditions set at pH 7 and a temperature of 35 °C. The optimum pH 7.5 was achieved 1.8-fold higher biomass (2.54 g/L) and carotenoid production (2.25 mg/L) by Paracoccus sp strain LL1 (Kumar et al., 2018). Collectively, these results suggest that both pH and temperature are the most significant factors, which may affect cell growth and carotenoid biosynthetic pathways.
Statistical optimization of the fermentation medium is a comprehensive and important tool for planning experiments; building models and evaluating the effects of different variables and their optimal concentration of particular variables are responsible for the commercial success of the fermentation process. In this study, we observed that statistical experimental design could be an important tool in optimizing the fermentation medium for enhanced β-carotene and biomass production by E. acetylicum S01. Among the seven variables tested by two level PBD, glucose, peptone, pH and temperature were identified as the most significant variables (confidence levels of above 95%) for biomass and β-carotene productivity. The optimal levels of these four variables were further verified by employing the CCRD of RSM. We observed that enhanced β-carotene (3.47-fold) and biomass (2.36-fold) production by E. acetylicum S01 was achieved under optimized fermentation medium condition compared to the un-optimized medium. Similarly, RSM has been successfully employed in Halorubrum sp. TBZ126 at optimal conditions that include a pH range between 7.51 and 7.94 and temperatures between 31 °C and 32 °C. Such conditions resulted in an 18.33 % and 20.55 % increase in biomass and total carotenoid yields, respectively (Hamidi et al., 2014). Choudhari and Singhal (2008) reported a maximum -carotene yield of 139 ± 1 mg/L when B. trispora was grown in RSM optimized medium in submerged fermentation conditions. Abdelhafez and co-works reported a maximum β-carotene yield of 2.24 mg/L when Serratia marcescens was grown in RSM optimized medium in shake flask culture (Abdelhafez et al., 2016). Valduga et al. (2009) reported that the enhanced total carotenoid production of 1,019 μg/L, when Sporidiobolus salmonicolor was grown in RSM optimized fermentation medium in submerged culture. To the best of our knowledge, this is the first communication reporting enhanced β-carotene and biomass production by E. acetylicum S01 using an RSM-CCRD approach. However, the results of the validation experiments demonstrated that this optimum fermentation medium led to a maximum enhancement in β-carotene production from the probiotic strain E. acetylicum S01 by bioreactor cultivation. Overall, these results suggest that optimum fermentation conditions for β-carotene and biomass production by E. acetylicum S01 could be developed for industrial applications as a commercial β-carotene producing microorganism.
Despite a number of investigations have been reported for the carotenoid biosynthetic pathway and the genes involved, very little is known about the regulation of the expression of carotenoid biosynthetic genes (Cunningham and Gantt, 1998). In the present study, attempts were made to compare the changes in carotenoid production with an expression of carotenoid biosynthetic genes under optimized and un-optimized conditions in E. acetylicum S01. We observed that the enhanced yield of β-carotene productivity and the expression of carotenoid biosynthetic genes (CrtI and CrtB) were correspondingly higher in E. acetylicum S01 under optimized medium conditions. The possible explanation for the up-regulation of carotenoid biosynthetic genes in E. acetylicum S01 in response to RSM optimized medium conditions, which may due to the stimulation of the carotenoid biosynthesis pathway. Only a few reports are available to depict the induction of biosynthetic genes due to the fermentation or stress conditions in the nutrients. In agreement with our results from an earlier study, the expression levels of carotenogenic genes (CrtI and CrtB) were differentially expressed in X. dendrorhous grown on fermentable carbon sources (Wozniak et al., 2011). Our results supported by the previous study the up-regulation of phytoene desaturase (pd) and phytoene synthase (ps) in E. profundum PHM11 under stress conditions (Patel et al., 2018). However, pH and temperature are the most important environmental factors affecting the growth and development of bacteria. Although it was found that appropriate high temperatures favour more β-carotene production (Lee et al., 2004; Vadali et al., 2005; Kim et al., 2009; Araya-Garay et al., 2012), when the strain S01 was grown at 30 °C the β-carotene and biomass productivity were higher than at either 40 or 20 °C. A plausible explanation of the significant up-regulation of both genes in E. acetylicum S01 under optimized medium conditions may be alkaline pH 8.5 which is important for carotenoid biosynthesis under stress conditions. While in un-optimized medium conditions pH 7.0 is neutral, it is essential for normal growth. Phytoene desaturase, the first enzyme involved in phytoene conversion to colored carotenoids, catalyzes a rate-limiting step in carotenoid biosynthesis (Chamovitz et al., 1993). The catalytic functions of bacterial phytoene desaturase are diverse, which can lead to low lycopene concentrations because of its poor catalytic specificity. In addition, high phytoene desaturase concentrations or a low phytoene supply favour the formation of lycopene to β-carotene via β-lycopene cyclase (Araya-Garay et al., 2012; Stickforth and Sandmann, 2007). The β-carotene yield of optimized fermentation medium condition was the highest. This is probably owing to the fact that the expression of CrtI and CrtB genes, which increase its translation efficiency and the final substrate conversion rate to β-carotene. At the same time, β-carotene is only synthesized from FPP after successful cluster gene expression of CrtE, CrtB and CrtI, which means that balanced gene expression is needed to avoid excessive accumulation of intermediate products that can inhibit cell growth. Further investigations are needed to determine the regulatory mechanisms of carotenoid biosynthetic gene expression.
5. Conclusion
In conclusion, we report, for the first time, the enhanced level of β-carotene (3.47-fold) and biomass (2.36-fold) productivity and the up-regulation of carotenoid biosynthetic genes (CrtI and CrtB) in E. acetylicum S01 under RSM optimized medium conditions. This result suggests that a statistical analysis had the advantage of identifying the most significant medium components and their optimal levels, and thus was useful for operating the fermentation process towards the accumulation of wanted metabolite.
Declarations
Author contribution statement
Jinendiran Sekar: Conceived and designed the experiments; Performed the experiments; Analyzed and interpreted the data; Wrote the paper.
B.S. Dileep Kumar, Hans-Uwe Dahms, Charli Deepak Arulanandam: Analyzed and interpreted the data; Wrote the paper.
Natesan Sivakumar: Conceived and designed the experiments; Analyzed and interpreted the data; Contributed reagents, materials, analysis tools or data; Wrote the paper.
Funding statement
This work was supported by the DST-Science and Engineering Research Board, DST-SERB (Ref. SB/YS/LS-05/2014), Government of India, for providing financial support. DST-PURSE Phase-II and UGC-SAP, MKU for providing instrumentation facilities.
Competing interest statement
The authors declare no conflict of interest.
Additional information
No additional information is available for this paper.
Appendix A. Supplementary data
The following is the supplementary data related to this article:
References
- Abdelhafez A.A., Husseiny S.M., Ali A.A., Sanad H.M. Optimization of β-carotene production from agro-industrial by-products by Serratia marcescens ATCC 27117 using Plackett–Burman design and central composite design. Ann. Agric. Sci. 2016;61:87–96. [Google Scholar]
- Araya-Garay J.M., Feijoosiota L., Rosadossantos F., Veiga-Crespo P., Villa T.G. Construction of new Pichia pastoris X-33 strains for production of lycopene and β-carotene. Appl. Microbiol. Biotechnol. 2012;93:2483–2492. doi: 10.1007/s00253-011-3764-7. [DOI] [PubMed] [Google Scholar]
- Baron M., Davies S., Alexander L., Snellgrove D., Sloman K.A. The effect of dietary pigments on the coloration and behaviour of flame-red dwarf gourami, Colisa lalia. Anim. Behav. 2008;75:1041–1051. [Google Scholar]
- Ben-Amotz A. Dunaliella β-carotene: from science to commerce. In: Seckbach J., editor. Enigmatic Microorganisms and Life in Extreme Environments. Kluwer; Dordrecht: 1999. pp. 399–410. [Google Scholar]
- Bhosale P.B., Gadre R.V. Production of β-carotene by a mutant of Rhodotorula glutinis. Appl. Microbiol. Biotechnol. 2001;55:423–427. doi: 10.1007/s002530000570. [DOI] [PubMed] [Google Scholar]
- Britton G., Liaaen-Jensen S., Pfander H. Birkhauser Verlag; Basel. Switzerland: 2004. Carotenoids Handbook. [Google Scholar]
- Buzzini P., Martini A. Optimization of carotenoid production by Rhodotorulagraminis DBVPG 7021 as a function of trace element concentration by means of response surface analysis. Enzym. Microb. Technol. 2005;36:687–692. [Google Scholar]
- Chamovitz D., Sandmann G., Hirschberg J. Molecular and biochemical characterization of herbicide-resistant mutants of cyanobacteria reveals that phytoene desaturation is a rate-limiting step in carotenoid biosynthesis. J. Biol. Chem. 1993;268:17348–17353. [PubMed] [Google Scholar]
- Choudhari S., Singhal R. Media optimization for the production of β-carotene by Blakeslea trispora A statistical approach. Biores. Technol. 2008;99:722–730. doi: 10.1016/j.biortech.2007.01.044. [DOI] [PubMed] [Google Scholar]
- Choudhari S., Rekha S. Media optimization for the production of β-carotene by Blakeslea trispora: a statistical approach. Bioresour. Technol. 2008;99:722–730. doi: 10.1016/j.biortech.2007.01.044. [DOI] [PubMed] [Google Scholar]
- Cunningham F.X., Gantt E. Genes and enzymes of carotenoids biosynthesis in plants. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1998;49:557–558. doi: 10.1146/annurev.arplant.49.1.557. [DOI] [PubMed] [Google Scholar]
- Das S., Bhattacharya A., Haldar S., Ganguly A., Gu S., Tinga Y.P., Chatterjee P.K. Optimization of enzymatic saccharification of water hyacinth biomass for bio-ethanol: comparison between artificial neural network and response surface methodology. Sustain. Mater. Tech. 2015;3:17–28. [Google Scholar]
- Davis W.B. Preparation of lycopene from tomato paste for use as a spectrophotometric standard. Anal. Chem. 1949;21:1226–1228. [Google Scholar]
- Fiedor J., Burda K. Potential role of carotenoids as antioxidants in human health and disease. Nutrients. 2014;6:466–488. doi: 10.3390/nu6020466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerhard S. Carotenoid biosynthesis and biotechnological application. Arch. Biochem. Biophys. 2001;385:4–12. doi: 10.1006/abbi.2000.2170. [DOI] [PubMed] [Google Scholar]
- Hamidi M., Abdin M.Z., Nazemyieh H., Hejazi M.A., Hejazi M.S. Optimization of total carotenoid production by Halorubrum Sp. TBZ126 using response surface methodology. J. Microb. Biochem. Technol. 2014;6:286–294. [Google Scholar]
- Jinendiran S., Boopathi S., Sivakumar N., Selvakumar G. Functional characterization of probiotic potential of novel pigmented bacterial strains for aquaculture applications. Pro. Antimicrob. Prot. 2017:1–12. doi: 10.1007/s12602-017-9353-z. [DOI] [PubMed] [Google Scholar]
- Johnson E.A., Schroeder W.A. Microbial carotenoids. Adv. Biochem. Eng. Biotechnol. 1996;53:119–178. doi: 10.1007/BFb0102327. [DOI] [PubMed] [Google Scholar]
- Khodaiyan Faramarz, Razavi S.H., Emam-Djomeh Z., Mousavi S.M.A., Hejazi M.A. Effect of culture conditions on canthaxanthin production by Dietzia natronolimnaea HS-1. J. Microbiol. Biotechnol. 2007;17:195–201. [PubMed] [Google Scholar]
- Kim S.W., Kim J.B., Ryu J.M., Jung J.K., Kim J.H. High-level production of lycopene in metabolically engineered E. coli. Process. Biochem. 2009;44:899–905. [Google Scholar]
- Korumilli T., Susmita M. Carotenoid production by Bacillus clausii using rice powder as the sole substrate: pigment analyses and optimization of key production parameters. J. Biochem. Technol. 2014;5:788–794. [Google Scholar]
- Kumar P., Jun H.B., Kim B.S. Co-production of polyhydroxyalkanoates and carotenoids through bioconversion of glycerol by Paracoccus sp. strain LL1. Int. J. Biol. Macromol. 2018;107:2552–2558. doi: 10.1016/j.ijbiomac.2017.10.147. [DOI] [PubMed] [Google Scholar]
- Lee P.C., Mijts B.N., Schmidtdannert C. Investigation of factors influencing production of the monocyclic carotenoid torulene in metabolically engineered Escherichia coli. Appl. Microbiol. Biotechnol. 2004;65:538–546. doi: 10.1007/s00253-004-1619-1. [DOI] [PubMed] [Google Scholar]
- Liong M.T., Shah N.P. Optimization of cholesterol removal by probiotics in the presence of prebiotics by using a response surface method. Appl. Environ. Microbiol. 2005;71:1745–1753. doi: 10.1128/AEM.71.4.1745-1753.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McWilliams A. 2018. Food Colors Market by Type (Natural, Synthetic, Nature-Identical), Application (Beverages, Processed Food, Bakery & Confectionery Products, Oils & Fats, Dairy Products, Meat, Poultry, Seafood), Form, Solubility, and Region - Global Forecast to 2023.https://www.bccresearch.com/market-research/food-and-beverage/the-global-market-for-carotenoids-fod025f.html [Google Scholar]
- Misawa N. Pathway engineering for functional isoprenoids. Curr. Opin. Biotechnol. 2011;22:627–633. doi: 10.1016/j.copbio.2011.01.002. [DOI] [PubMed] [Google Scholar]
- Naziri D., Hamidi M., Hassanzadeh S., Tarhriz V., Zanjani B.M., Nazemyieh H., Hejazi M.A., Hejazi M.S. Analysis of carotenoid production by Halorubrum sp. TBZ126; an extremely halophilic archeon from urmia lake. Adv. Pharmaceut. Bull. 2014;4:61–67. doi: 10.5681/apb.2014.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelis H.J., DeLeenheer A.P. Microbial sources of carotenoid pigments used in food and feeds. J. Appl. Bacteriol. 1991;70:181–191. [Google Scholar]
- Pal D., Banerjee S., Ghosh A.K. Dietary-induced cancer prevention: an expanding research arena of emerging diet related to health care system. J. Adv. Pharm. Technol. Res. 2012;3:16–24. doi: 10.4103/2231-4040.93561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patel V.K., Sharma A., Singh S., Chakdar H., Kalra A., Saxena A.K., Pandiyan K. Halotolerant Exiguobacterium profundum PHM11 tolerate salinity by accumulating L-Proline and fine-tuning gene expression profiles of related metabolic pathways. Front. Microbiol. 2018;9:423. doi: 10.3389/fmicb.2018.00423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pi H.W., Anandharaj M., Kao Y.Y., Lin Y.J., Chang J.J., Li W.H. Engineering the oleaginous red yeast Rhodotorula glutinis for simultaneous β-carotene and cellulase production. Sci. Rep. 2018;8:10850. doi: 10.1038/s41598-018-29194-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plackett R.L., Burman J.P. The design of optimum multifactorial experiments. Biometrika. 1946;33:305–325. [Google Scholar]
- Sasidharan P., Raja R., Karthik C., Sharma R., Indra-Arulselvi P. Isolation and characterization of yellow pigment producing Exiguobacterium sp. J. Biochem. Technol. 2013;4:632–635. [Google Scholar]
- Seon-Won K., Weon-Tack S., Young-Hoon P. Increased β-carotene synthesis in Blakeslea trispora under strong alkaline condition. Biotechnol. Lett. 1996;18:1287–1290. [Google Scholar]
- Shatila F., Yusef H., Holail H. Pigment production by Exiguobacterium aurantiacum FH, a novel Lebanese strain. Int. J. Curr. Microbiol. App. Sc. 2013;2:176–191. [Google Scholar]
- Sowmya R., Sachindra N.M. Carotenoid production by Formosa sp. KMW, a marine bacterium of Flavobacteriaceae family: influence of culture conditions and nutrient composition. Biocat. Agricult. Biotechnol. 2015;4:559–567. [Google Scholar]
- Stickforth P., Sandmann G. Kinetic variations determine the product pattern of phytoene desaturase from Rubrivivax gelatinosus. Arch. Biochem. Biophys. 2007;461:235–241. doi: 10.1016/j.abb.2007.02.035. 2007. [DOI] [PubMed] [Google Scholar]
- Sun N., Lee S., Song K.B. Characterization of a carotenoid hyper-producing yeast mutant isolated by low dose gamma irradiation. Int. J. Food Microbiol. 2004;94:263–267. doi: 10.1016/S0168-1605(03)00311-8. [DOI] [PubMed] [Google Scholar]
- Thawornwiriyanun P., Tanasupawat S., Dechsakulwatana C., Techkarnjanaruk Somkiet, Worapot Identification of newly zeaxanthin-producing bacteria isolated from sponges in the Gulf of Thailand and their zeaxanthin production. Appl. Biochem. Biotechnol. 2012;167:2357–2368. doi: 10.1007/s12010-012-9760-2. [DOI] [PubMed] [Google Scholar]
- Vadali R.V., Fu Y., Bennett G.N., San K.Y. Enhanced lycopene productivity by manipulation of carbon flow to isopentenyl diphosphate in Escherichia coli. Biotechnol. Prog. 2005;21:1558–1561. doi: 10.1021/bp050124l. [DOI] [PubMed] [Google Scholar]
- Valduga E., Valerio A., Treichel H., Junior A.F., Luccio M.D. Optimization of the Production of total carotenoids by Sporidiobolus salmonicolor (CBS 2636) using response surface technique. Food Bioproc. Technol. 2009;2:415–421. [Google Scholar]
- Venil C.K., Zakaria Z.A., Ahmada W.A. Bacterial pigments and their applications. Process. Biochem. 2013;48:1065–1079. [Google Scholar]
- Vilchez Carlos, Forjan Eduardo, Cuaresma Maria, Bedmar Francisco, Garbayo I., Vega J.M. Marine Carotenoids: biological functions and commercial applications. Mar. Drugs. 2011;9:319–333. doi: 10.3390/md9030319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang B., Lin L., Lu L., Chen W. Optimization of β-carotene production by a newly isolated Serratia marcescens strain. Electron. J. Biotechnol. 2012;15:1–8. [Google Scholar]
- Wozniak A., Lozano C., Barahona S., Niklitschek M., Marcoleta A., Alcaino J. Differential carotenoid production and gene expression in Xanthophyllomyces dendrorhous grown in a non-fermentable carbon source FEMS. Yeast. Res. 2011;11:252–262. doi: 10.1111/j.1567-1364.2010.00711.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.