Abstract
Arrhythmias coexist in patients with heart failure (HF) and LV dysfunction. Tachycardias, AF and premature ventricular contractions (PVCs) are known to trigger a reversible dilated cardiomyopathy (CM) referred as arrhythmia-induced CM (AiCM). It remains unclear why some patients are more prone to develop AiCM despite similar arrhythmia burdens. The challenge is to determine whether arrhythmias are fully, partially or at all responsible for an observed LV dysfunction. AiCM should be suspected in patients with mean HR above 100 bpm, AF and/or PVCs burden equal or greater than 10%. Reversal of cardiomyopathy by elimination of the arrhythmia confirms AiCM. Therapeutic choice depends on the culprit arrhythmia, patient comorbidities and preferences. Following recovery of LV function, patients require continued follow up if an abnormal myocardial substrate is present. Appropriate diagnosis and treatment of AiCM is likely to improve quality of life, clinical outcomes and reduce hospital admission and health care spending.
Keywords: Supraventricular tachycardia, Arrhythmia, Premature ventricular contractions, Cardiomyopathy, Left ventricular dysfunction, Heart failure
Condensed Abstract
Persistent tachycardia, atrial fibrillation and frequent ventricular ectopy can trigger a reversible form of dilated cardiomyopathy, referred as arrhythmia-induced cardiomyopathy (AiCM). Clinical presentation may vary from asymptomatic to severe heart failure. Clinicians should have a high suspicion especially in patients without another obvious etiology. Ambulatory ECG monitors are key to screen and properly diagnose AiCM. Arrhythmia suppression will not only reverse LV dysfunction with its associated morbidity and mortality, but most importantly improve patient’s health, quality of life and long-term prognosis.
Introduction
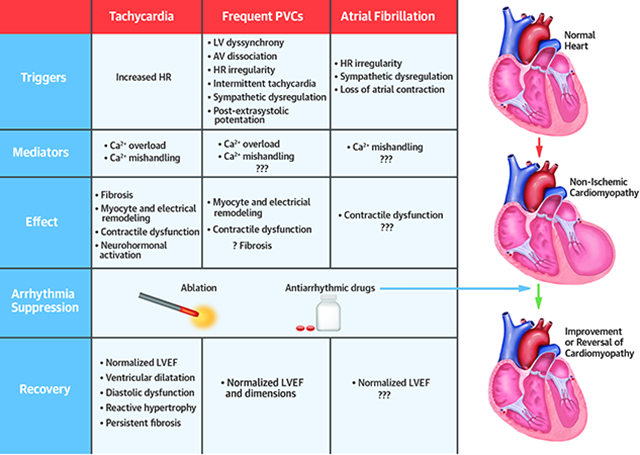
Arrhythmias have been long considered part of the clinical presentation of heart failure (HF) and cardiomyopathy (CM). Yet, supraventricular or ventricular tachyarrhythmias alone can result or trigger a reversible non-ischemic CM. Most recently, atrial fibrillation (AF) despite adequate rate control, as well as, premature ventricular contractions (PVCs) have been recognized as a unique etiology of non-ischemic dilated cardiomyopathy (1,2,3). Since the recognition of PVC-Cardiomyopathy. Thus, a more inclusive term of Arrhythmia-induced CM (AiCM) has emerged to include both, tachycardia-induced CM (T-CM), AF-induced CM (AF-CM) and PVC-Cardiomyopathy (PVC-CM) (Central Illustration). However, this term does not include recently recognized CM due to conduction abnormalities / dyssynchrony, such as chronic RV pacing, left bundle branch block and pre-excitation (1,4).
Central Illustration. Arrhythmia-induced Cardiomyopathies: Possible Triggers, Mediators, Effect, and Recovery.
While T-CM was first described in a patient with atrial fibrillation at the beginning of the 20th century, PVC-CM was only recognized nine decades later in 1998 (6,7). Significant skepticism remained on the cause-relationship between arrhythmias and cardiomyopathies until experimental animal models developed in 1962 and 2011 (8,9), respectively, proved that these sustained arrhythmias could result in LV dysfunction in structurally normal hearts. This review article presents an update of the current understanding of AiCM.
Tachycardia-induced Cardiomyopathy (T-CM)
Definition and prevalence
T-CM refers to the presence of a reversible LV dysfunction solely due to increase in ventricular rates, regardless of tachycardia origin. The risk of developing T-CM depends not only on the type but also duration and rate of tachycardia. The overall prevalence and incidence of T-CM is unclear and likely underestimated.
A study reported T-CM in 2.7% patients referred for radiofrequency ablation (RFA), however it also included patients referred for PVC ablation (10). T-CM has been reported in 10% of patients with atrial tachycardia (AT) (11), and as high as 37% in patients with incessant AT. Moreover, permanent junctional reciprocating tachycardia (PJRT) appears to have the highest association with T-CM (20–50%) as it frequently presents as an incessant supraventricular tachycardia (SVT) (12). Despite atrial fibrillation (AF) being the most prevalent arrhythmia, there is no clear data on the prevalence of T-CM in this population. Only a single study reported T-CM in 4% of patients referred for pulmonary vein isolation (13), however, this data is confounded by selection and referral biases. Children are also prone to T-CM resulting most frequently from AT (59%), PJRT (23%) and ventricular tachycardia (7%) (12).
Causes
T-CM can manifest in the setting of either an incessant or paroxysmal tachycardia and it should be suspected if no other cause of LV dysfunction is identified. A frequent challenge is to identify a superimposed T-CM when tachycardia worsens a known CM. T-CM has been reported to present weeks or months to years after the onset of tachycardia (14).
Supraventricular arrhythmias are the most common etiology, namely AF and atrial flutter with rapid ventricular response. Although rare, other arrhythmias such as incessant or very frequent paroxysmal AT, persistent AV reciprocating tachycardia (AVRT) and AV nodal reentrant tachycardia (AVNRT), sustained sinus tachycardia, frequent ventricular tachycardias (idiopathic, bundle branch and fascicular) and pacemaker mediated tachycardia can also be responsible (11,14). In general, it is suspected that faster tachycardia rates and ventricular arrhythmias cause a more severe T-CM although no studies exist to support this hypothesis (1).
Pathophysiology & Mechanism
Animal models have been key to understand the pathophysiology and mechanism of T-CM. Similar to humans, animals exposed to persistent tachycardia using a continuous rapid atrial or ventricular pacing develop heart failure symptoms, LV systolic dysfunction and dilatation, decrease in LV dP/dtmax and myocardial blood flow and increase in LV wall stress and end-diastolic pressure and volume (9,15,16). Dilatation tends to be biventricular with mild thinning or no associated hypertrophy or change in heart mass (9,15). The progression of these physiological changes include a decrease in systemic blood pressure and increase in LV and pulmonary artery pressure, which plateaus at 1 week, while cardiac output, ejection fraction and volumes continue to deteriorate the following 4 weeks with development of symptomatic HF within 2–3 weeks (9).
T-CM is characterized by structural and functional myocardial changes (Table 1, Central Illustration). Similar to human studies, T-CM models have also demonstrated electrical remodeling and abnormal Ca homeostasis thought to be responsible for impaired excitation-contraction coupling and diastolic dysfunction (9,16–18). Only total Ca cycling, Ca-channel inhibition and basal ATPase activity have demonstrated a statistical correlation with decrease in LV ejection fraction (LVEF) (17).
Table 1.
Summary of abnormalities found in tachycardia and PVC-Cardiomyopathies based in human and animal models.
| Feature | T-CM | PVC- CM | |
|---|---|---|---|
| Tissue remodeling (8,9,16,18,19,59) | Myocardial blood flow | Decreased | ?? |
| Fibrosis | Mild | Absent or mild / ↑ collagen type I | |
| Extracellular matrix and myocyte basal membrane | Disarray | ?? | |
| Myocyte misalignment | Present | ?? | |
| Inflammation | Macrophage -dominated ↑↑ MHC- class II; ↑ MMP-9) ↑ D68 macrophages |
Absent | |
| Myocyte remodeling (8,16,18,64) | Myocyte loss | Present | ?? |
| Apoptosis | Increased | Unchanged | |
| T-tubules | Depletion | Depletion | |
| Sarcomere | Loss and change in sarcomere | ?? | |
| Mitochondria | Abnormal size, architecture and function ↑ number at intercalated disc ↑ MPC1 |
Unchanged Normal oxidative phosphorylation | |
| ATP and Na-K ATPase | Reduced | ?? | |
| β -adrenergic receptors | ↓ number of β-receptors ↓ G stimulator protein density & adenylate cyclase activity ↑ G inhibitory protein density |
?? | |
| Oxidative stress | Increased | ?? | |
| Glucose metabolism | Impaired | ?? | |
| Electrical remodeling (8,16,17,24,59,63,64) | Action potential duration | Prolonged | Prolonged (heterogeneity) |
| VERP | Increased | Increased | |
| Ion Currents | ↓ ICa | ↓ ICa and Cav1.2 ↓ Ito and Kv4.3 ↓ IKr |
|
| Ca2+ Transient / SR release | Decreased | Decreased | |
| SR Ca2+-uptake (SERCA2a) | Decreased | Decreased | |
| CaMKII-alpha | Increased | Increased | |
| SR Ca2+ store | Decreased | Unchanged | |
| SR Ca2+ leak | Decreased | Unchanged | |
| Na+/Ca2+ exchanger (NCX) - Cai extrusion | Increased | Unclear (contradictory data) | |
| Dyad remodeling | ?? | ↓ JPH-2 & BIN-1 |
Note: MHC-class II, major histocompatibility complex Class II; MMP-9, anti-matrix metallopeptidase; MPC1, mitochondrial pyruvate carrier; VERP, Ventricular effective refractory period; Cav1.2, L-Type Ca2+ channel pore-forming subunit; ICa, L-type Ca2+ current; Ito, Potassium transient outward current; IKr, rapid delay potassium current; SR, sarcoplasmic reticulum; SERCA2a, Sarcoplasmic/endoplasmic reticulum Ca2+ ATPase-2a; JPH-2, junctophilin-2 (a dyad scaffolding protein); BIN-1, a key protein involved in Cav1.2 targeting to T-tubules; CaMKII-alpha, Ca2+/calmodulin-dependent protein kinase II; ??, unknown.
Cessation of tachy-pacing results in normalization of right atrial and arterial pressure with significant recovery of LVEF and cardiac output by 48 hours, and full normalization after 1 to 2 weeks (9). However, a week after resolution of tachycardia, LV mass increases by 26% and LV remains dilated and myocytes continue to demonstrate contractile dysfunction (15). Moreover, only Ca cycling (sum of Ca uptake and Ca release), Ca-uptake and CK activity significantly normalized (17) 4 weeks after cessation of tachy-pacing. Importantly, some changes such as fibrosis appear to persist despite elimination of the tachycardia and normalization of LV function (16,19).
Clinical presentation, diagnosis and imaging features
Clinical studies have found a variable time from onset of arrhythmia symptoms to development of T-CM, ranging from 3 to 120 days with an overall LVEF of 32% (14). Regardless of tachyarrhythmia, heart failure symptoms will manifest earlier at higher tachycardia rates (1,9,16), such as patients with persistent atrial flutter or tachycardia with 2:1 AV conduction with rates greater than 150 bpm. A recent clinical study found a more severe LV dysfunction (LVEF 29.3 ± 6.6%) in T-CM when compared to dilated and inflammatory CM (32.1 ± 10.2% and 41.9 ± 12.9%, respectively; p <0.001) (18).
Major reported symptoms include palpitations (29%), HF class III-IV (47%) and syncope/presyncope (12%), while the remaining may have no symptoms (10). Sudden cardiac death is uncommon but has been reported in up to 8–12% despite treatment and resolution of cardiomyopathy (14,20).
T-CM should be suspected in patients with LV dysfunction and a prior or persistent or frequent paroxysmal tachycardia without obvious etiology (Table 2). A superimposed T-CM should be considered despite underlying secondary CM (ischemic, infiltrative or toxic/drug-related) if the tachycardia is present. Thus, an ambulatory ECG monitor for at least a 2-week period is key to confirm or exclude T-CM.
Table 2.
Reversible and irreversible causes of cardiomyopathy
| Reversible | Irreversible |
|---|---|
| Transient ischemia / post-cardiac arrest | Extensive / multiple myocardial infarctions |
| Subacute valvular heart disease | Hypertrophic cardiomyopathy |
| Uncontrolled hypertension | Cardiac sarcoidosis |
| LBBB - cardiomyopathy | End-stage valvular heart disease |
| Pacing-induced cardiomyopathy | Infectious (e.g. Chagas disease) |
| Drug or alcohol abuse | |
| Endocrine (severe hypothyroidism) | |
| PVC-induced Cardiomyopathy | |
| Stress-induced Cardiomyopathy | |
| Peripartum Cardiomyopathy | |
| Inflammatory / Infectious (e.g. myocarditis, sepsis) |
Echocardiogram or cardiac magnetic resonance can assist in excluding other etiologies. T-CM is characterized by a dilated CM (increased LV end-diastolic dimension and area) with moderate to severe biventricular systolic dysfunction and normal LV septal and posterior wall thickness (lack of hypertrophy). Mitral insufficiency may be present due to LV and mitral annular dilatation with lack of leaflet coaptation (16).
Neurohormonal markers such as brain natriuretic peptide (BNP) and pro-BNP are commonly elevated depending on the degree of heart failure and CM (14,21). Moreover, a sudden drop of pro-BNP within a week of elimination of tachycardia is supportive of T-CM (21). However, the final diagnosis of T-CM can be only confirmed after recovery or improvement of LV systolic function within 1 −6 months after elimination of the tachyarrhythmia.
Treatment
A major feature of T-CM is its reversibility once tachycardia is eliminated. Thus, the mainstay treatment consists on suppression of tachycardia based on the culprit arrhythmia (Table 3) with antiarrhythmics (AADs) and/or RFA. Nevertheless, initial treatment of T-CM, should include initiation and optimization of medical therapy for heart failure and LV systolic dysfunction (beta blockers, angiotensin-converting enzyme inhibitors or angiotensin receptor blockers, diuretics and aldosterone blockers) to optimize reverse remodeling.
Table 3.
Treatment options of T-CM based on tachy-arrhythmia.
| Arrhythmia | Treatment |
|---|---|
| Sinus tachycardia / Thyrotoxicosis | BB + treatment of underlying disease |
| Atrial fibrillation with RVR | Rhythm (PVI +/− AADs) vs. Rate control vs. AVJ |
| Atrial flutter with RVR | Radiofrequency ablation |
| Atrial Tachycardia | Radiofrequency ablation vs. AADs |
| AVRT / AVNRT | Radiofrequency ablation |
| Rapid Atrial/Ventricular pacing (pacemaker mediated tachycardia) | Reprogram pacemaker |
| Sustained Ventricular Tachycardia | Radiofrequency ablation +/− AADs |
Note: BB, beta-blockers; PVI, pulmonary vein isolation; AADs, antiarrhythmic drugs; AVJ, atrioventricular nodal ablation
Elimination of tachyarrhythmia not only resolves LV function within 4–12 weeks, but also improves heart failure symptoms by at least one NYHA class in most patients (9,14,16). Multivariate analysis demonstrated that age, tachycardia rate and baseline LVEF and LVEDD were predictors of recovery in a pediatric population (12). Unfortunately, the recovery of T-CM is not always complete. Histopathologic abnormalities, diastolic dysfunction and ventricular dilatation with a hypertrophic response may persist despite normalization of LVEF (15,16,18,19).
In the presence of superimposed T-CM, full reversibility of LV function is unlikely after treatment of tachyarrhythmia. Nevertheless, treatment should not be discouraged as it may have small yet significant benefits.
Tachycardia recurrence in prior history of T-CM
Studies have documented that recurrence of T-CM and heart failure symptoms upon arrhythmia recurrence sooner and with at least the same severity compared to initial presentation (14,20). It is speculated that persistence of underlying histopathological abnormalities from initial presentation are likely responsible for a more rapid and severe presentation if arrhythmia recurs (16,19). Yet, LV dysfunction recovers to prior or normal levels with elimination of new or recurrent tachycardia. Thus, a permanent treatment such as ablation therapy should be especially considered in arrhythmias with a high success or cure rate such as atrial flutter, AVNRT, AVRT and AT.
AF-induced Cardiomyopathy (AF-CM)
AF is frequently considered as a primary cause for T-CM. Limited evidence suggests that ventricular rate during AF does not predict reversibility of CM (22). This has raised the question of whether AF duration and/or irregularity predicts LV dysfunction rather than ventricular rate. More recently, AF ablation trials in HF have questioned current recommendations that rate control alone is appropriate in patients with paroxysmal / persistent AF and associated CM / HF (3,23). This is in light of the significant improvement of LV function and HF symptoms after AF ablation when compared to medical therapy, supporting the premise that AF alone can lead to CM despite appropriate rate control.
AF-CM is defined as LV systolic dysfunction in patients with paroxysmal or persistent atrial fibrillation despite appropriate rate control. Thus, an ambulatory Holter monitor is key to rule out poor rate control and T-CM. Despite AF being the most prevalent arrhythmia, the prevalence and factors that predispose or prevent AF-CM are unknown. A frequent clinical challenge is to recognize whether the AF is due to HF and cardiomyopathy or vice versa.
The mechanism of AF-CM is unknown (Central Figure). It is believed that AF-CM is triggered in part due to 1) HR irregularity with calcium mishandling (24) and 2) loss of atrial contraction/ emptying associated with sympathetic activation contributing to limited ventricular filling and increased filling pressures, functional mitral regurgitation and diastolic dysfunction (2,25). Unfortunately, no AF-CM animal models exist to better understand causality, risk factors and/or mechanism involved in its pathogenesis.
AF-CM is a diagnosis of exclusion and should be primarily suspected in patients with non-ischemic CM and persistent AF that do not improve after appropriate medical therapy and rate control. Due to the overlap with T-CM and lack of animal models, it is unclear how the time course, clinical, laboratory or imaging features differ between AF-CM and T-CM. A final AF-CM diagnosis can only be corroborated if LV systolic function improves or normalizes after elimination of AF.
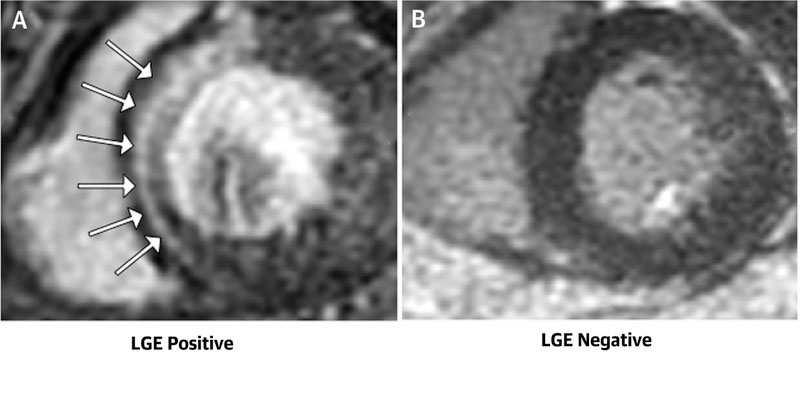
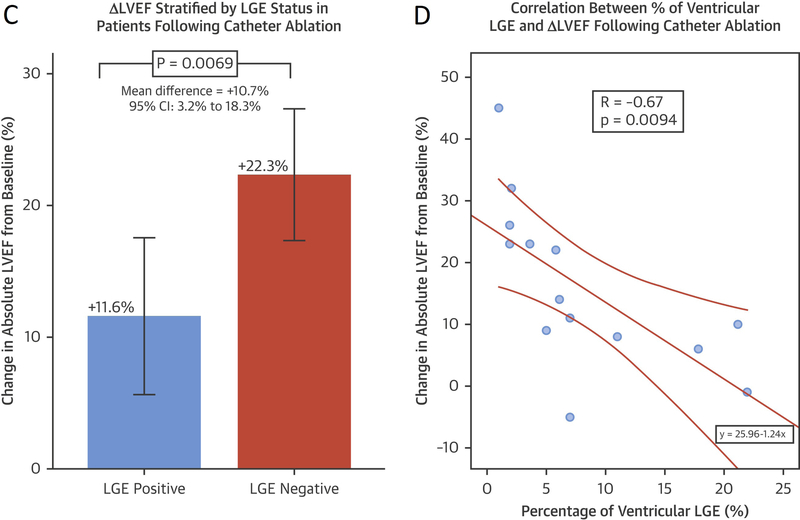
Restoration of sinus rhythm should be considered if AF-CM is suspected. AF ablation has been reported to achieve sinus rhythm from 50–88% in both paroxysmal and persistent AF patients with HF and CM (2,3,23,26). While complication rate of AF ablation is low (2–3%), a second ablation is frequently required. In contrast, AADs have an overall 30–50% success rate to maintain sinus rhythm with frequent discontinuation due to side effects (2,26).Further evidence suggests that AF ablation maybe superior to AADs in AF-CM (2). Landmark AF trials with AADs have failed to demonstrate outcome benefits including HF admissions in patients with and without HF or CM (2,27,28). This is in contrast to randomized clinical studies comparing AF ablation as a rhythm control versus rate control strategy, reporting an 8–18% absolute increase in LVEF in 60–70% in patients with AF and CM randomized to ablation (2,13,23,29). The only trial comparing rhythm control strategies (ablation vs. amiodarone) in patients with AF and CM demonstrated ablation to be superior by improving freedom of AF (70 vs. 34%), quality of life, HF admissions (31 vs. 57%) and mortality (8 vs. 18%) after 2-year follow-up (26). Lastly, the CAMERA-MRI study supports the use of cardiac MRI with late-gadolinium enhancement as a screening tool to predict CM reversibility before ablation. The absence of ventricular scar or scar burden <10% predicted reversibility of AF-CM (Figure 1) (23).
Figure 1.
Representative examples of patients with AF with late gadolinium enhancement (LGE) in a patient without (A) and with AF-CM (B). Top panels demonstrate the presence (A) and absence (B) of scar in cardiac MR with late-gadolinium enhancement (LGE) in a patient without and with AF-CM, respectively. Panel C demonstrate change in LVEF from baseline stratified by the presence or absence of scar, while Panel D demonstrates correlation between % of scar (LGE) and change in LVEF from baseline after AF ablation (permission obtained Prabhu S, et al. The CAMERA-MRI Study. J Am Coll Cardiol 2017;70:1949–1961) (23).
PVC-Cardiomyopathy
Due to its unique features and lack of tachycardia, PVC-CM is now recognized as a distinct clinical entity by the most recent 2016 AHA Scientific Statement of dilated cardiomyopathies (1). PVC-CM is defined as the development of LV dysfunction caused solely by frequent PVCs. Moreover, superimposed PVC-CM can be defined as worsening of LVEF by at least 10% due to frequent PVCs in a previously known CM. Even though, frequent PVCs are commonly referred as PVC burden greater than 5%, a PVC burden ≥ 10% is often considered high and significant to trigger PVC-CM.
Epidemiology of PVCs and PVC-cardiomyopathy
The incidence of PVCs in a 10-second 12-lead ECG is estimated between 1% to 4% of patients without heart disease (30,31). However, the prevalence of PVCs is significantly higher during an ambulatory ECG recordings, (40% and 75% of participants on 24- to 48-hour ambulatory Holter monitoring) (31). This can be explained by a significant variability of PVC frequency with time (32).
Prevalence of PVCs is also age-dependent with <1% in children younger than 11 years and near 70% in subjects 75 years and older (30,31). PVCs are more frequently associated with post-myocardial infarction, coronary heart disease and dilated CM and HF (30,34). Furthermore, almost half of patients with HF class II and III had frequent PVCs (>1,000 PVCs/day) (34).
Clinical studies have found high PVC burden associated with LV dysfunction, increased risk of systolic heart failure (HR 1.48–1.8) and mortality (HR 1.31) (35–41) even after adjusting for age and other ECG abnormalities (30). Surprisingly, a six-fold increased risk (HR 6.5) of systolic HF has been reported in subjects <65 years with PVCs without other cardiovascular risk factors (42) and a higher mortality risk for those with HR >100 bpm (30). Lastly, a significant increased risk of incident stroke (HR 1.71, CI 1.14–2.59) has been reported on a secondary analysis of the Atherosclerosis Risk in Communities (ARIC) study, which the investigators attribute to a possible atrioventricular remodeling (43).
Prevalence of PVC-CM has been reported at 7% amongst patients with frequent PVC burden greater than 10% (44). Nevertheless, PVC-CM is likely underestimated (31). Clinical studies have reported a diagnosis of PVC-CM in 9–30% of patients referred for RFA of PVC (35,38,45–47). Similarly, a secondary analysis of the CHF-STAT study (48) (>10 PVCs/hour and LVEF <40%) demonstrated an estimated rate of PVC-CM of 40% in all patients with CM and up to 66% in non-ischemic CM (49). This data supports frequent PVCs as a significant and modifiable risk factor for systolic HF and increased mortality.
Acute effects of PVCs and potential triggers of PVC-CM
PVCs have acute intrinsic effects that are innate to their ectopic and premature origin, including heart rate irregularity and post-extrasystolic potentiation, LV dyssynchrony, atrioventricular (AV) dyssynchrony and increased heart rate (Table 4) (41,50–54). Furthermore, it is unclear if and how these triggers by altering hemodynamics and autonomic nervous system (ANS) contribute to the development of PVC-CM (55,56).
Table 4.
Acute effects and potential triggers of PVC-CM.
| Intrinsic PVC effects / Triggers |
|---|
| Post-extrasystolic potentiation |
| Increase in contractility that follows either an atrial or ventricular extrasystole, associated with Ca2+ overload (54). |
| Has an inverse relationship to PVC coupling interval (prematurity): shorter PVC coupling intervals (early PVCs) have a greater intracellular Ca2+ and post-extrasystolic potentiation (54). |
| Increase in O2 consumption with little change in cardiac output despite reduction LVEDP (mean 13mmHg) and increase in coronary flow (54). |
| LV dyssynchrony |
| Uncoordinated contraction of the LV segments. |
| Cause disruption and progression of dyssynergic LV wall motion resulting in LV dysfunction (41,85,100). |
| Directly proportional to the PVC coupling interval: longer PVC coupling intervals (late PVCs) demonstrate a greater LV dyssynchrony (52). |
| Tachycardia |
| Mean heart rate in ambulatory ECG Holters in PVC-CM is frequently normal, likely due to compensatory pauses. |
| Clinical and animal studies have not found any difference in mean heart rate between PVC-CM and other etiologies(41,50,65). |
| AV dyssynchrony |
| Occurs when atrial contraction takes place against a closed AV valve during PVC, demonstrated as a prominent “a” wave in the pulmonary capillary wedge pressure (PCWP) tracing. |
| PVC-PCWP augmentation (defined as prominent a wave > 15mmHg) was associated with a lower LVEF (52±9 vs. 62±10%, p<0.01) and shorter coupling interval (432±41 vs. 522±54ms, p<0.0001) compared to those without PVC-PCWP augmentation despite having similar PVC burden (53). |
| Autonomic Nervous System (ANS) |
| PVCs alone have shown to acutely increase sympathetic nerve activity not only at a cardiac level but also peripherally (56) |
| PVCs elicit a greater neuronal response than other stimuli such as aortic or inferior vena cava occlusion (55) |
| PVCs with variable prematurity had the greatest neuronal response (convergent, sympathetic and parasympathetic input) when compared to early and late PVCs (55) |
Post-extrasystolic potentiation together with HR irregularity, LV dyssynchrony, AV dissociation and alteration in ANS are possible triggers of PVC-CM. However, differences in cellular and electrical remodeling between PVC- and T-CM (Table 1) reinforce the argument that tachycardia is unlikely to be the sole trigger for PVC-CM.
The role of post-extrasystolic potentiation and HR irregularity could be addressed by evaluating the chronic effects of frequent PACs, which lack LV dyssynchrony. While, Pacchia et.al. (57) demonstrated that CM could be induced by atrial bigeminy, this has not been observed clinically and in other animal models (58,59)(60). These findings suggest that heart rate irregularity and post-extrasystolic potentiation play a limited role, if any, on the pathophysiology of PVC-CM.
Finally, Gerstenfeld et.al. demonstrated in a bigeminy PVC animal model that LV dysfunction is more pronounced in PVCs from LV epicardium as they demonstrate a higher degree of dyssynchrony when compared to endocardial RV free wall (59). This is also consistent with clinical studies where epicardial PVCs and QRS >150ms were predictors of PVC-CM (61,62).
Potential mechanism(s) of PVC-CM
In contrast to T-CM, the cellular mechanism of PVC-CM has not been extensively studied. Yet, it is clear that there are distinct differences in histopathological and cellular features compared to other HF models including T-CM (Central Figure, Table 1) (8,9,15,16,18,19,59,63,64).
The primary cause of contractile dysfunction in PVC-CM appears to be disorders of the calcium-induced calcium release mechanism itself, with alterations of dyad (L-type Ca channel and Ryanodine receptor) function proposed as a potential mechanism. Similar to other cardiomyopathies, this PVC-CM model has revealed electrophysiological remodeling (Table 1). Histopathological abnormalities are distinct without evidence of increased inflammation or apoptosis and minimal or no fibrosis. Mitochondrial studies have demonstrated no changes in oxidative phosphorylation (8). These findings are supported clinically by the lack of scar on cardiac MRI of patients with PVC-CM (38). These findings further confirm a primary functional abnormality as a primary mechanism of this reversible CM (8,38,59,63,64). Whether all the cellular and molecular changes are in response to the CM rather than the cause of the CM remains unclear.
Predictors of PVC-CM
Clinical evaluation of underlying effects of frequent PVCs have limitations due to a variability of PVC features (origin, prematurity, frequency, QRS width), presence of confounding patient comorbidities and small sample population.
PVC burden has been shown to be a major predictor of PVC-CM (OR 1.25 per each percent increase in PVC burden; CI 1.1–1.42) (35,45,62,65–67). Two main studies have shown that PVC burden greater than 16% and 24% best identifies patients with a diagnosis of PVC-CM (sensitivity and specificity of 79–100 and 78–87%, respectively) (35,44). While these and other studies suggest that a PVCs burden of at least 10% is required to induce PVC-CM (35,44,68–70), other studies question this minimal PVC thresholds since they have shown improvement in LV function with PVC burden as low as 6–8% (48,51,66,71–73). The length of ambulatory ECG monitoring has important implications, since increasing the duration from a 24-hr to a 7-day ambulatory Holter monitor can doubled the number of patients who reach the 10% threshold (33).
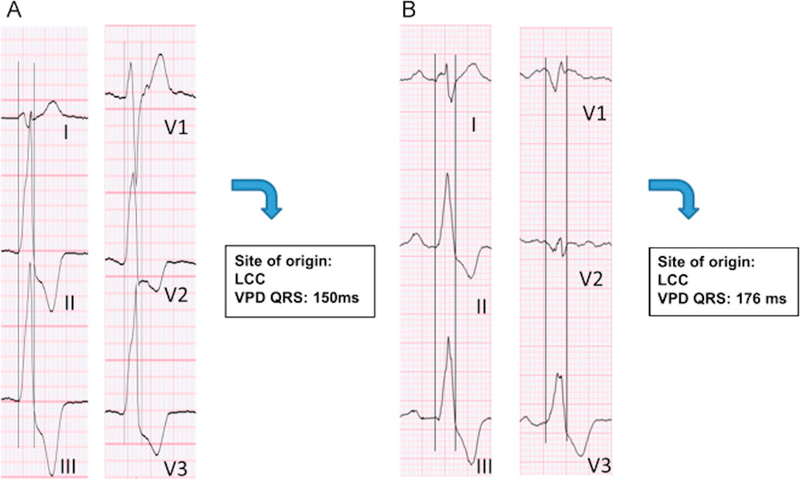
Nevertheless, some patients do not develop a CM even with high PVC burden. Thus, it is likely that other patient’s characteristics and/or PVC features play a role in the pathophysiology of PVC-CM. Some other PVC features have been found to be independent predictors for PVC-CM such as male gender (74), lack of symptoms (adjusted OR 13.1; CI 4.1–37) or duration of palpitations >30 months, variability of PVC coupling interval (OR 1.04; CI 1.03–1.07(39)), QRS duration of PVC >150ms, and epicardial origin (37,44,45,61,62,65,67,74,75) (Figure 2). Other less frequently reported independent predictors are BMI >30kg/m2 (OR 3.03; CI 1.2–7.7) (39), less variability in circadian PVC distribution (OR 16.3; CI 1.7–155) (76) and presence of retrograde P wave (OR 2.79; CI 1.08–7.19) (67). AV dyssynchrony during PVCs could potentially be a predictor but yet remains to be studied (53,67). Except for PVC burden, most predictors have been variably reported reflecting the heterogeneity of different populations. Although further validation is required, a PVC-CM index, including PVC burden, PVC-QRS width and epicardial origin, has been developed in an attempt to identify patients with high probability of PVC-CM (62).
Figure 2.
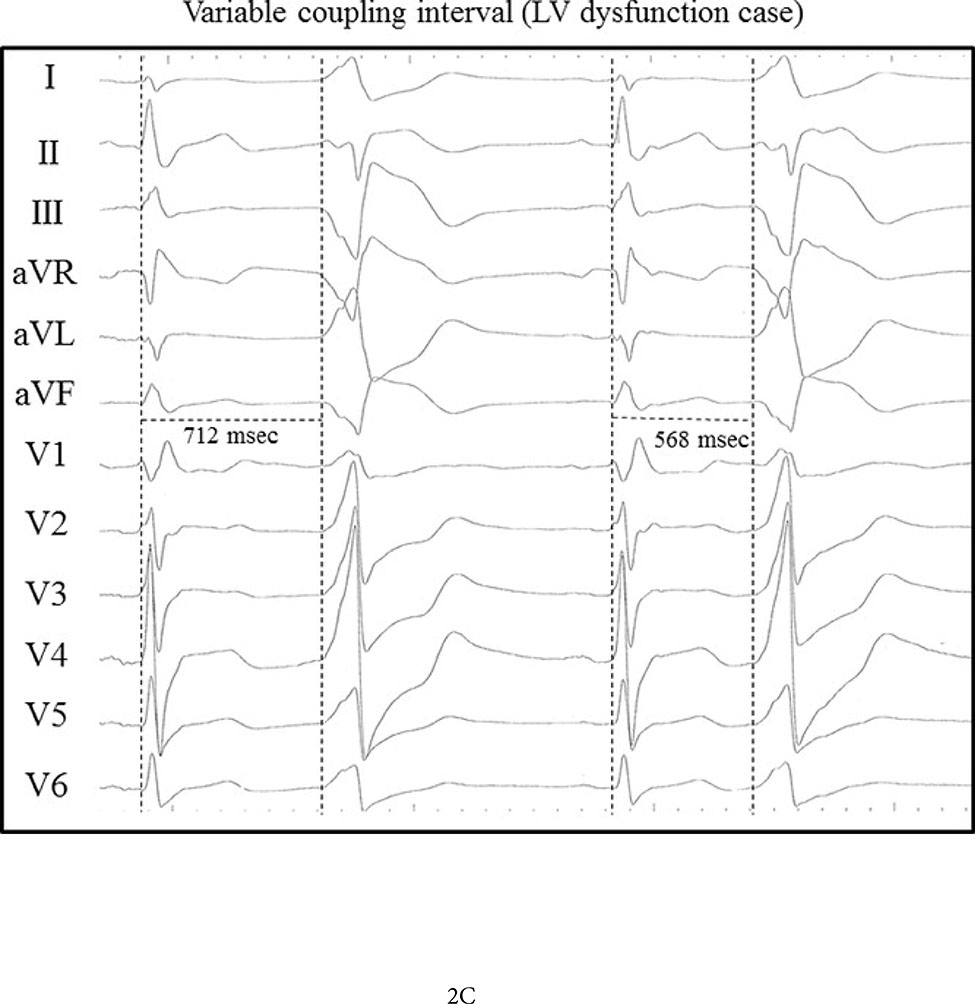
Representative cases of patients similar high PVC burden from left coronary cusp without (A) and with (B) associated CM (LVEF 40%). PVC-CM has a wider PVC QRS duration (172ms) when compared to preserved LV function (150ms) (permission obtained Carballeira Pol L, et al. Heart Rhythm 2014; 11:299–306) (75). (C) Representative case of PVC-CM with dispersion of CI of 144ms (cutoff of CI dispersion >99ms best identified patients with and without PVC-CM) (permission obtained Kawamura M, et al. J Cardiovasc Electrophysiol 2014;25:756–62) (39).
Even though, short PVC coupling intervals have been associated with idiopathic ventricular fibrillation (77), most studies have not found a clear relationship between PVC coupling interval and CM (37). While some studies have reported that interpolated PVCs or coupling interval <450ms may be predictors to develop PVC-CM (68,78), others reported that PVC coupling interval variability (dispersion) is not only associated with a higher risk of PVC-CM but also cardiovascular mortality (39,79,80). A potential explanation (demonstrated in animal data) is the large cardiac neuronal disturbance demonstrated in PVCs with variable coupling interval, beyond what is seen in short or long coupling intervals (55). Animal studies are currently underway to investigate the effects of PVC coupling interval on LVEF.
PVC locations, other than epicardial origin has not shown to be predictor of PVC-CM (45,68). One of the largest series have found that PVC-CM have a PVC origin from coronary sinus (epicardial) in 24%, RV outflow in 21%, LV outflow tract in 28%, mitral annulus (endocardial) in 7%, RV/LV septum in 5% and RV/LV apex in 4% (65).
Lastly, genetic predisposition could explain why some patients are prone to develop PVC-CM while others do not despite similar PVC burden. For instance, R222Q missense variant of the Nav1.5 subunit of sodium channel, resulting in a greater and earlier sodium current, has been attributed to a rate-dependent ectopy of Purkinje and associated with a CM reversible upon treatment with amiodarone or flecainide (81).
Clinical presentation, diagnosis and imaging features
The time course for the development of PVC-CM is unclear, but it is estimated to occur within months up to several years (62,67,75). While animal studies with persistent high PVC burden (33–50%) develop CM within 4 weeks (8,50), human studies are not consistent in part due to the unclear onset and variability of PVCs.
PVC-CM may have a wide range of presentations, from asymptomatic or vague symptomatology to heart failure and even syncope. It is unclear why some patients have symptoms related to PVCs while others do not, but a PVC coupling interval ratio <0.5 (PVC CI ratio: PVC coupling interval/Sinus coupling interval) has been proposed as an important marker of symptoms (82). A careful history and pertinent testing should be obtained to rule out other causes of cardiomyopathy (Table 2), while physical examination is frequently normal with the exception of irregular heart sounds and mild signs of heart failure.
PVC-CM is a diagnosis of exclusion, to be suspected in patients with frequent PVCs greater than 10%, especially in non-ischemic CM. A challenge is to identify when PVCs are the etiology of a CM or just “innocent bystanders” in patients with CM. Even if PVCs are the result of CM, these PVCs if frequent may contribute to and further worsen CM and HF symptoms, referred to as “superimposed’ PVC-CM (73,83). In selected cases, echocardiographic and PVC features can help identify these patients (Table 5) (1).
Table 5.
Clinical and PVC features to identify PVC-Cardiomyopathy.
| CM resulting in PVCs | PVCs causing CM | |
|---|---|---|
| Patient characteristics | Older with known heart disease | Healthy otherwise |
| Comorbidities | CAD, myocarditis, RV dysplasia | No prior cardiac hx |
| Echocardiogram | Segmental hypokinesis, LVEF < 25% | Global hypokinesis, LVEF 37 ± 10% |
| Cardiac MRI (late-gadolinium enhancement) | Significant scar | Absence or minimal scar burden (≤ 9gm) |
| PVC Frequency | < 5,000 / 24 hrs (< 5%) | ≥ 10,000 / 24 hours (>10%) |
| PVC pattern | Multifocal | Monomorphic |
| QRS morphology | Non-specific | RVOT / LVOT / Epicardial |
| Response to PVC suppression | No change in LV function | Improvement of LV function |
Even though the ECG is important, a prolonged ambulatory ECG monitor is essential to improve the diagnostic yield of high PVC burden. As noted by Loring et.al. (33), a minimum of 6 days is required to detect an individual’s maximum PVC burden. In contrast, a 24-hour Holter identified only 53% of patients with a PVC burden greater than 10%, which would likely miss almost half (47%) of patients with potential PVC-CM diagnosis (33).
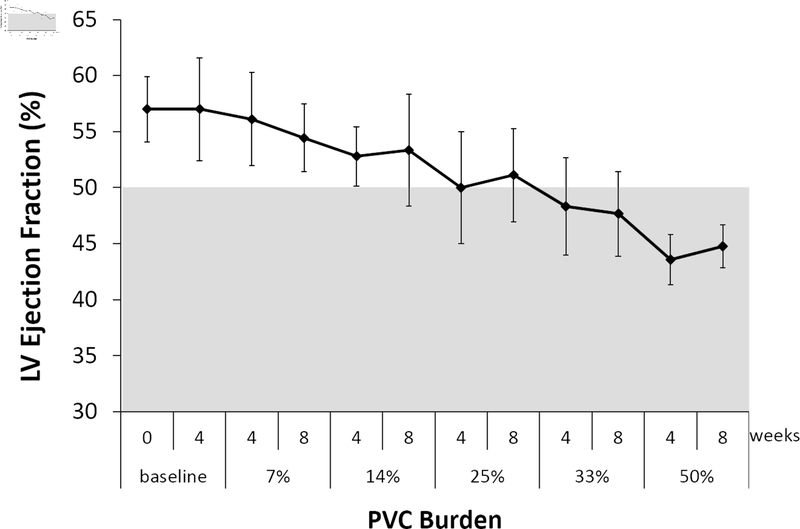
PVC-CM is characterized by mild to moderate LV systolic dysfunction, LV dilatation, mild MR and LA enlargement, which resolved within 2–12 weeks after elimination of PVCs (8,41,50). Cardiac imaging is key to identify LV dysfunction and suspect the diagnosis of PVC-CM in patients with high PVC burden (≥10%) (Table 5). Cardiac MR with late-gadolinium enhancement has the advantage of identifying scar and quantify scar burden, which in turn potentially predicts response to PVC suppression (84). Few clinical and animal studies have also demonstrated diastolic dysfunction after 12-weeks of chronic ventricular bigeminy (50,85). Interestingly, a subclinical form of PVC-CM (LVEF ≥50%) has been reported using speckle tracking by a decrease in radial, circumferential and longitudinal strain that can reverse after RFA (86,87). This is further supported by translational studies demonstrating a mild and linear decrease of LV systolic function with 7, 14 and 25% PVC burden (Figure 3) (50).
Figure 3.
Progression of LV ejection fraction in a large animal PVC model after 4 and 8weeks of a progressive incremental PVC burden starting from 0% (baseline) to 7%, 14%, 24%, 33%, and 50%. (p <0.0001, 1-way ANOVA). Even though PVC-CM is not seen until PVC burden of 33%, a decline in LVEF can be noted at lower PVC burdens (Permission obtained Tan AY, et al. Heart Rhythm 2016;13:755–61) (50).
Most recently, myocarditis has been implicated as a potential trigger for frequent PVCs and CM (88), while elevated hs-CRP has been reported to be an independent predictor of PVCs in a Chinese population study (89). Thus, it may be clinically challenging to determine a causal versus bystander role of PVCs in an inflammatory process.
Treatment
Currently, a PVC suppression strategy with RFA or AADs is a widely accepted intervention to treat a CM that might be caused or exacerbated by frequent PVCs (1). However, the treatment of frequent PVCs (≥ 10% burden) without LV dysfunction (LVEF ≥50%), symptoms or idiopathic ventricular fibrillation is less clear. Due to the lack of data and potential risk of developing PVC-CM, these patients require close monitoring every 6–12 months or more closely if heart failure symptoms develop. Echocardiogram should be repeated to confirm a normal LV function, while a prolonged ambulatory ECG monitor should be considered to reassess PVC burden. Even though spontaneous resolution of frequent PVCs has not been evaluated, data from the CHF-STAT trial demonstrated that 12% of patients on placebo had spontaneous and significant decrease in PVC burden at 6 months (49).
PVC suppression is considered successful if burden is decreased by >80% of baseline PVCs as it likely represents a true effect of treatment rather than spontaneous PVC variability (90). However, this criterion was based on 24-hour Holter data and it is unclear if this is different nowadays with extended 2- or even 4-week ambulatory monitors. Current therapies, RFA and AADs, have similar long-term PVC suppression success rate between 70 to 80% (45,48,62,68,70,91). Successful RFA may be limited in patients with PVCs originating from papillary muscle, epicardium or nearby critical structures such as coronary arteries and conduction system (40,45,68). Thus, antiarrhythmic therapy may be necessary in about 5–15% of patients after RFA (68). PVC suppression strategies (RFA or AADs) carries an overall low risk. While the complication rates of RFA have been reported between 5–8%, antiarrhythmics have a discontinuation rate of near 10% due to short- and long-term side effects (45,68,84,91,92) in addition to potentially decreased efficacy overtime (93).
Randomized clinical studies of AADs have only been performed prior to the recognition of PVC-CM as a unique entity. While the CAST trial demonstrated an increase mortality with class IC agents in patients with frequent ectopy after MI, the GESICA, CAMIAT and CHF-STAT trials demonstrated at least a trend towards a decrease in mortality after MI and in non-ischemic CM with the use of amiodarone (48,94). No randomized-prospective studies has compared the efficacy and outcomes between RFA and AAD therapy. A contemporary retrospective study has shown that PVC reduction was greater with RFA than antiarrhythmics (mean reduction: RFA −15.5 ± 1.3% vs. AADs −4.8 ± o.8, p<0.001) (68). While RFA and antiarrhythmic drugs can successfully suppress high PVC burdens, a single retrospective study suggests that RFA may be more effective in patients with lower PVC burdens (68).
PVC suppression in PVC-CM has been shown to improve LV function, LV dilatation, mitral regurgitation and BNP levels (1,70). The mean improvement of LVEF after RFA in most studies is between 10 to 15% (Table 6) (45,65,68,70,91,92) even in superimposed PVC-CM. A recent multicenter retrospective study of 245 patients with non-ischemic CM and frequent PVCs (mean PVC burden 20 ± 13%) demonstrated improvement of LV function in 67% of patients after RFA (45). Similarly, a prospective study demonstrated a significant decrease in BNP levels while primary prophylaxis ICD implantation was avoided in 80% of all patients with PVC burden >13% due to significant improvement of LVEF after successful RFA (95). Interestingly, another study (40) found that 81% of patients (n=36) with abnormal baseline eGFR (<60mL/min) had significant improvement in renal function (eGFR 51 to 57mL/min) after RFA of PVCs.
Table 6.
Summary of studies of PVC-Cardiomyopathy.
| LV Ejection Fraction | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Author | # Pts | Baseline PVC Burden | PVC-CM rate | Suppression strategy | CM | Successful Suppression | Pre-PVC suppression | Post-PVC suppression | Follow-up (months) | Notes/ Outcome/Predictors (P) |
| Singh (1995) (48,49) | 336 | >10 PVC/hr | 40% | Amiodarone vs. Placebo (RC)* | I+NI | 72% (Amio)12% (placebo) | A: 24.9 ± 8% P: 25.7 ± 8% |
A: 33.7 ± 11% P: 29.2 ± 11% |
45 | * 15% pts in placebo had LVEF improvement (delta EF ≥10%) despite PVC suppression <80% |
| Duffee (1998) (6) | 5 | >20,000/day | -- | AADs | NI | 35% | 27 ± 10 % | 49 ± 17 % | 6 ± 3 | |
| Yargadala (2005) (71) | 8 | 17,541/day | -- | RFA | NI* | 87% | 39 ± 6 % | 62 +6 % | 3 | * included PVC from RVOT only |
| Bogun (2007) (36) | 18 | 37 ± 13% | 82% | RFA | NI | 80% | 34 ± 13 % | 59 ± 7 % | 46 | |
| Taieb (2007) (101) | 6 | 17,717/day | -- | RFA | NI | ? | 42 ± 2.5 % | 57 ± 3.7 % | 6 | |
| Sarrazin (2009) (73) | 12 | 22 ± 12% | 66% | RFA | I* | 100% | 38 ± 11 % | 51 ± 0.1 | 14 ± 13 | *included pts with remote hx of MI referred for IC & PVC ≥5%; NYHA improved after RFA |
| Baman (2010) (35) | 57 | 33 ± 13% | 81% | RFA | NI* | 84% | 35 ± 9% | 54 ± 10% | 48 | * excluded CAD only; Burden (P) |
| Hasdemir (2011) (44) | 17 | 29 ± 9% | -- | RFA | NI* | 75% | 38 ± 7% | 53 ± 7% | 4 | * excluded prior cardiac or ischemic disease |
| Mountantonakis (2011) (91) | 69 | 29 ± 13% | -- | RFA | NI | 78% | 35 ± 9% | 48 ± 10%* | 11 ± 6 | LVEF improvement correlated with ablation outcome and decline in PVC burden. |
| Lu (2012) (46) | 24 | 15 ± 6% | 58% | RFA | I+NI | ? | 32 ± 15% | 43 ± 14% | 8 | |
| Ban (2013) (67) | 28 | 26 ± 10% | 75% | RFA | NI | 91% | 44 ± 5% | 55 ± 6% | 19 ± 17 | Burden (P), retrograde P waves (P) |
| Yokokawa (2013) (47) | 87 | 26 ± 11% | 86% | RFA | NI | 86% | 39 ± 10% | 59 ± 4% | 6 – 45 | 32% with delayed recovery of LVEF (5–45 mo). Epicardial origin predicted delayed recovery. |
| Zhong (2014) (68) | 121 | 25% (RFA) 22% (AAD) | 32% | RFA vs. AAD (NR) | I+NI | 86% (RFA) 49% (AAD) | 42% | 55%* | 6. 3 ± 2.4 | * EF restored 47% (RFA) vs. 21% (AAD); PVC CI <450ms (P) |
| El Kadri (2015) (83) | 30 | 23 ± 8.8% | 50% | RFA | NI* | 60% | 34 ± 15% | 45 ± 17% | 30 ± 28 | *Included pts with scar or prior CM; NYHA improved after RFA |
| Panela (2015) (70) | 66 | 21 ± 12% | 26% | RFA | I+NI | 76% | 28 ± 4% | 42 ± 12% | 12 | Included only LVEF <35% referred for ICD |
| Latchamsetty (2015) (45) | 245 | 27 ± 13% | 67% | RFA | NI | 71% | 38 % | 50 % | 20 ± 22 | Burden (P), Male gender (P), epicardial origin (P) |
| Sadron Blaye- Felice (2016) (65) | 96 | 26 ± 12% | -- | RFA | NI* | 80% | 38 ± 10% | 50 ± 13% | 24 ± 21 | *39% had HD (ischemic, HTN< valvular) with high suspicious of PVC-CM; Burden (P), Epicardial origin (P) & lack of palpitations (P) |
| Hamon (2016) (62) | 58 | 23 ± 12% | 54% | RFA | I+NI+SHD | 91% | 38 ± 9% | 55 ± 9% | 22 ± 15% | Burden (P), Epicardial origin (P), PVC-QRS duration (P), SHD (P) |
| Lee (2018) (66) | 54 | 28% (19–44) | 61% | RFA | I+NI | 73% | 40% (30–46) | 52% (45–56) | 7 (median) | Burden (P), Male gender (P) |
Note: All studies were observational, except CHF-STAT study (48) which was a randomized control trial; PVC-CM rate refers to the percentage of patients with improvement of LV function (normalized or increased by ≥10–15%) after PVC suppression. I, Ischemic CM; NI, Non-ischemic CM; SHD, structural heart disease including valvular heart disease, ischemic, hypertensive and dilated CM
see column “notes”; P, predictors of PVC-CM only by multivariate analysis.
Successful ablation (OR 15.7; 1.4–180), myocardial scar mass <9gm (OR 0.9; 0.81 – 99) and reduction of mean PVC burden (OR 1.09; 1.01–1.16) have been shown to be independent predictors of response to RFA (84,91). This supports assessment of scar burden using cardiac MR with late-gadolinium enhancement to predict responders versus non-responders to PVC suppression (Figure 4). However, if significant PVC burden reduction (>20%) is achieved, the presence of myocardial scar seems to be less relevant to predict response (84). In contrast, PVC location does not appear to predict improvement of LVEF (92). Recently, post-extrasystolic potentiation assessed by invasive BP monitoring has been described as a predictor of LV function recovery after radiofrequency ablation (96).
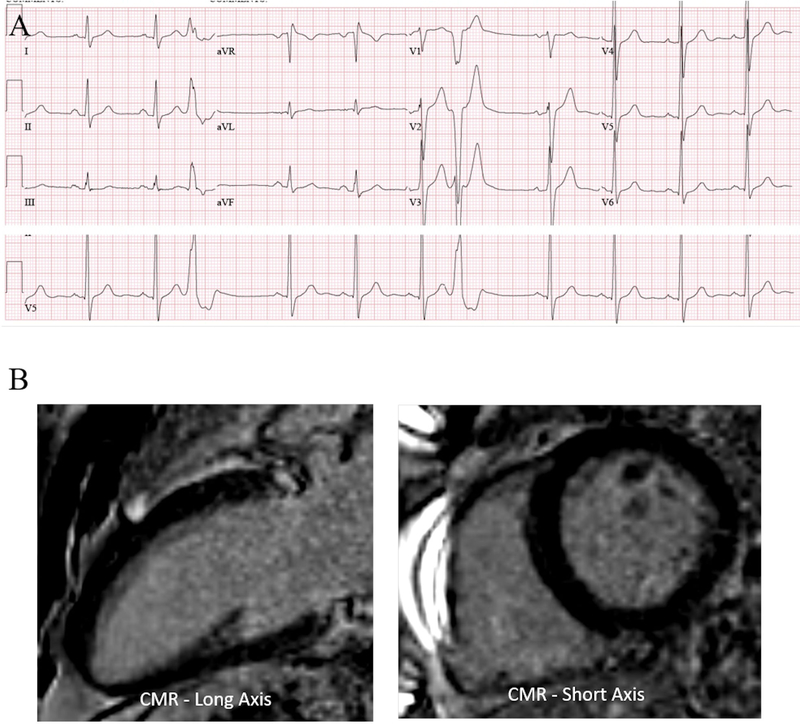
Figure 4. Representative PVC-cardiomyopathy.
A 53-year-old man with 21% PVC burden and LVEF of 40% underwent PVC ablation. Successful ablation was achieved at the mid-septal RVOT just above pulmonary valve with a PVC burden of 1.5% after RFA. (A) 12-lead ECG of representative PVC. (B) Baseline CMR demonstrate absence of scar with late-gadolinium enhancement. LVEF normalized to 55% after 3 months, diagnostic of PVC-CM.
Clinical studies have consistently demonstrated a significant increase in LV function after PVC suppression with antiarrhythmics, ranging from 10 – 13% (48,68). Because the CAST trial included patients with ischemic CM, the guidelines discourage the use of Class IC antiarrhythmics not only in ischemic but also in non-ischemic CM patients. Most recently, a small retrospective study (93) demonstrated that flecainde and propafenone (Class IC) can also improve LV function (LVEF from 37.4 ± 2 to 49 ± 1.9%), even without an 80% PVC suppression (PVC burden from 36.2 ± 3.5% to 10 ± 2.4%) without an increase in ventricular arrhythmias and/or death (93).
While current literature supports improvement of LV function and symptoms by elimination of PVCs, there is limited data that PVC suppression will subsequently modify the risk of cardiovascular events including heart failure and death (97,98). Over the past few years it has become clear that comparative effectiveness trials are needed to understand how to best treat patients with frequent PVCs and CM (95,97). Currently, a pilot multicenter study “Prospective Assessment of PVC Suppression in Cardiomyopathy: A pilot study” (PAPS: Pilot) is ongoing to better understand the prevalence of frequent PVCs and CM, prove feasibility of a large-scale randomized clinical trial (ClinicalTrials.gov Identifier: NCT03228823). Focused studies to understand the prevalence of PVC-CM are key to provide a better perspective of the magnitude of this clinical entity and potential impact in the HF population.
Clinical significance of arrhythmia-induced CM
Tachycardia, AF and PVCs are highly prevalent in patients with CM and HF, and they must be considered as potential cause of a HF and CM (Figure 5). While, it is unclear why some patients with high arrhythmia burden (frequent HR > 100bpm and/or PVC burden ≥ 10%) do not develop CM, these patients are at risk and should undergo close monitoring every 6–12 months or sooner if symptoms develop. The risk factors for developing PVC-CM include male gender, lack of symptoms, and PVCs QRS duration >150ms, epicardial origin and/or variable coupling interval, As any other CM, AiCM can lead to heart failure admissions and implantation of defibrillators and resynchronization devices (51,73,95,99). Thus, AiCM carries a significant financial burden if untreated, which makes diagnosis and treatment paramount to improve morbidity and potentially decrease health care costs. A better understanding of the mechanism of tachycardia and PVC induced CM could lead to novel therapies to prevent and improve outcomes especially when antiarrhythmics or RFA are not feasible or unsuccessful.
Figure 5. Proposed management of potential Arrhythmia-induced CM.
Footnotes: (~) Consider following the algorithm even if CAD is documented or worsening of prior CM is noted (superimposed AiCM). (^) Two-week ambulatory Holter is preferred as increases the diagnosis yield of high PVC burden (≥10%). (*) Consider cardiac MR to assess scar burden and predict response to PVC suppression. Short-term observation is reasonable for PVC-CM as 15% of cases may improve without PVC suppression strategy (49). (©) Continue close surveillance and HF med Rx in those with abnormal LV dimensions and presence of scar (cardiac MRI)
Summary / conclusions
Arrhythmia-induced CM, a reversible CM, has a significant variety of presentations from asymptomatic to severe heart failure symptoms. Clinicians should have a high index of suspicion of superimposed AiCM even in patients with an obvious etiology (Figure 5). T-CM should be strongly considered in patients with paroxysmal or persistent SVTs, primarily atrial fibrillation / flutter, atrial tachycardia and persistent junctional reciprocating tachycardia with heart rates above 100 bpm. AF-CM should be suspected in patients with non-ischemic CM and paroxysmal, persistent or permanent AF even with appropriate rate control, while, PVC-CM should be considered in patients with non-ischemic CM and PVC burden equal or greater than 10%. Appropriate diagnosis and treatment of AiCM will not only reverse LV dysfunction with its associated morbidity, mortality and healthcare spending, but most importantly improve quality of life and long-term prognosis. Future clinical studies are needed to compare standard treatment strategies and identify best long-term PVC suppression and prevention of recurrence of PVC-CM.
Highlights.
Tachycardias, atrial fibrillation, and premature ventricular contractions are known to trigger a reversible dilated cardiomyopathy.
Arrhythmia-induced cardiomyopathy should be highly suspected in patients without an obvious etiology.
Ambulatory ECG monitors are key to screen and properly diagnose arrhythmia-induced cardiomyopathy.
Reversal of cardiomyopathy by elimination of arrhythmia not only confirms the diagnosis but may significantly improve outcomes.
Acknowledgments
NIH Funding: 1R01HL139874–01 (PI: Huizar), 5R34HL138110–02 (PI: Huizar).
Abbreviations
- AADs
Antiarrhythmic drugs
- AiCM
Arrhythmia-induced cardiomyopathy
- AFiCM
Atrial fibrillation-induced cardiomyopathy
- ANS
Autonomic nervous system
- AT
Atrial tachycardia
- CM
Cardiomyopathy
- PJRT
Permanent Junctional reciprocating tachycardia
- PVC-CM
Premature ventricular contraction-cardiomyopathy
- RFA
Radiofrequency ablation
- T-CM
Tachycardia-induced cardiomyopathy
Footnotes
Disclosures: None relevant to this manuscript
Tweet: Clinicians should request Holters and suspect Arrhythmia-induced cardiomyopathy when LV dysfunction without obvious etiology is found. We present an update of this topic.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Bozkurt B, Colvin M, Cook J et al. Current Diagnostic and Treatment Strategies for Specific Dilated Cardiomyopathies: A Scientific Statement From the American Heart Association. Circulation 2016;134:e579–e646. [DOI] [PubMed] [Google Scholar]
- 2.Trulock KM, Narayan SM, Piccini JP. Rhythm control in heart failure patients with atrial fibrillation: contemporary challenges including the role of ablation. J Am Coll Cardiol 2014;64:710–21. [DOI] [PubMed] [Google Scholar]
- 3.Marrouche NF, Brachmann J, Andresen D et al. Catheter Ablation for Atrial Fibrillation with Heart Failure. N Engl J Med 2018;378:417–427. [DOI] [PubMed] [Google Scholar]
- 4.Fazio G, Mongiovi M, Sutera L, Novo G, Novo S, Pipitone S. Segmental dyskinesia in Wolff-Parkinson-White syndrome: a possible cause of dilatative cardiomyopathy. Int J Cardiol 2008;123:e31–4. [DOI] [PubMed] [Google Scholar]
- 5.Gossage AMBHJ. On auricular fibrillation. Quarterly Journal of Medicine 1913;6:435–440. [Google Scholar]
- 6.Duffee DF, Shen WK, Smith HC. Suppression of frequent premature ventricular contractions and improvement of left ventricular function in patients with presumed idiopathic dilated cardiomyopathy. Mayo Clin Proc 1998;73:430–3. [DOI] [PubMed] [Google Scholar]
- 7.Chugh SS, Shen WK, Luria DM, Smith HC. First evidence of premature ventricular complex-induced cardiomyopathy: a potentially reversible cause of heart failure. J Cardiovasc Electrophysiol 2000; 11: 328–9. [DOI] [PubMed] [Google Scholar]
- 8.Huizar JF, Kaszala K, Potfay J et al. Left ventricular systolic dysfunction induced by ventricular ectopy: a novel model for premature ventricular contraction-induced cardiomyopathy. Circ Arrhythm Electrophysiol 2011;4:543–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Shinbane JS, Wood MA, Jensen DN, Ellenbogen KA, Fitzpatrick AP, Scheinman MM. Tachycardia-induced cardiomyopathy: a review of animal models and clinical studies. J Am Coll Cardiol 1997;29:709–15. [DOI] [PubMed] [Google Scholar]
- 10.Donghua Z, Jian P, Zhongbo X et al. Reversal of cardiomyopathy in patients with congestive heart failure secondary to tachycardia. J Interv Card Electrophysiol 2013;36:27–32; discussion 32. [DOI] [PubMed] [Google Scholar]
- 11.Medi C, Kalman JM, Haqqani H et al. Tachycardia-mediated cardiomyopathy secondary to focal atrial tachycardia: long-term outcome after catheter ablation. J Am Coll Cardiol 2009;53:1791–7. [DOI] [PubMed] [Google Scholar]
- 12.Moore JP, Patel PA, Shannon KM et al. Predictors of myocardial recovery in pediatric tachycardia-induced cardiomyopathy. Heart Rhythm 2014;11: 1163–9. [DOI] [PubMed] [Google Scholar]
- 13.Gentlesk PJ, Sauer WH, Gerstenfeld EP et al. Reversal of left ventricular dysfunction following ablation of atrial fibrillation. J Cardiovasc Electrophysiol 2007;18:9–14. [DOI] [PubMed] [Google Scholar]
- 14.Watanabe H, Okamura K, Chinushi M et al. Clinical characteristics, treatment, and outcome of tachycardia induced cardiomyopathy. Int Heart J 2008;49:39–47. [DOI] [PubMed] [Google Scholar]
- 15.Spinale FG, Holzgrefe HH, Mukherjee R et al. LV and myocyte structure and function after early recovery from tachycardia-induced cardiomyopathy. Am J Physiol 1995;268:H836–47. [DOI] [PubMed] [Google Scholar]
- 16.Gupta S, Figueredo VM. Tachycardia mediated cardiomyopathy: pathophysiology, mechanisms, clinical features and management. Int J Cardiol 2014;172:40–6. [DOI] [PubMed] [Google Scholar]
- 17.O’Brien PJ, Moe GW, Nowack LM, Grima EA, Armstrong PW. Sarcoplasmic reticulum Ca-release channel and ATP-synthesis activities are early myocardial markers of heart failure produced by rapid ventricular pacing in dogs. Can J Physiol Pharmacol 1994;72:999–1006. [DOI] [PubMed] [Google Scholar]
- 18.Mueller KAL, Heinzmann D, Klingel K et al. Histopathological and Immunological Characteristics of Tachycardia-Induced Cardiomyopathy. J Am Coll Cardiol 2017;69:2160–2172. [DOI] [PubMed] [Google Scholar]
- 19.Ling LH, Kalman JM, Ellims AH et al. Diffuse ventricular fibrosis is a late outcome of tachycardia-mediated cardiomyopathy after successful ablation. Circ Arrhythm Electrophysiol 2013;6:697–704. [DOI] [PubMed] [Google Scholar]
- 20.Nerheim P, Birger-Botkin S, Piracha L, Olshansky B. Heart failure and sudden death in patients with tachycardia-induced cardiomyopathy and recurrent tachycardia. Circulation 2004;110:247–52. [DOI] [PubMed] [Google Scholar]
- 21.O’Brien PJ, Ianuzzo CD, Moe GW, Stopps TP, Armstrong PW. Rapid ventricular pacing of dogs to heart failure: biochemical and physiological studies. Can J Physiol Pharmacol 1990;68:34–9. [DOI] [PubMed] [Google Scholar]
- 22.Redfield MM, Kay GN, Jenkins LS, Mianulli M, Jensen DN, Ellenbogen KA. Tachycardia-related cardiomyopathy: a common cause of ventricular dysfunction in patients with atrial fibrillation referred for atrioventricular ablation. Mayo Clin Proc 2000;75:790–5. [DOI] [PubMed] [Google Scholar]
- 23.Prabhu S, Taylor AJ, Costello BT et al. Catheter Ablation Versus Medical Rate Control in Atrial Fibrillation and Systolic Dysfunction: The CAMERA-MRI Study. J Am Coll Cardiol 2017;70:1949–1961. [DOI] [PubMed] [Google Scholar]
- 24.Ling LH, Khammy O, Byrne M et al. Irregular rhythm adversely influences calcium handling in ventricular myocardium: implications for the interaction between heart failure and atrial fibrillation. Circ Heart Fail 2012;5:786–93. [DOI] [PubMed] [Google Scholar]
- 25.Cha YM, Redfield MM, Shen WK, Gersh BJ. Atrial fibrillation and ventricular dysfunction: a vicious electromechanical cycle. Circulation 2004;109:2839–43. [DOI] [PubMed] [Google Scholar]
- 26.Di Biase L, Mohanty P, Mohanty S et al. Ablation Versus Amiodarone for Treatment of Persistent Atrial Fibrillation in Patients With Congestive Heart Failure and an Implanted Device: Results From the AATAC Multicenter Randomized Trial. Circulation 2016;133:1637–44. [DOI] [PubMed] [Google Scholar]
- 27.Al-Khatib SM, Shaw LK, Lee KL, O’Connor C, Califf RM. Is rhythm control superior to rate control in patients with atrial fibrillation and congestive heart failure? Am J Cardiol 2004;94:797–800. [DOI] [PubMed] [Google Scholar]
- 28.Roy D, Talajic M, Nattel S et al. Rhythm control versus rate control for atrial fibrillation and heart failure. N Engl J Med 2008;358:2667–77. [DOI] [PubMed] [Google Scholar]
- 29.Chen MS, Marrouche NF, Khaykin Y et al. Pulmonary vein isolation for the treatment of atrial fibrillation in patients with impaired systolic function. J Am Coll Cardiol 2004;43:1004–9. [DOI] [PubMed] [Google Scholar]
- 30.Engel G, Cho S, Ghayoumi A et al. Prognostic significance of PVCs and resting heart rate. Ann Noninvasive Electrocardiol 2007;12:121–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lee GK, Klarich KW, Grogan M, Cha YM. Premature ventricular contraction-induced cardiomyopathy: a treatable condition. Circ Arrhythm Electrophysiol 2012;5:229–36. [DOI] [PubMed] [Google Scholar]
- 32.Schmidt G, Ulm K, Barthel P, Goedel-Meinen L, Jahns G, Baedeker W. Spontaneous variability of simple and complex ventricular premature contractions during long time intervals in patients with severe organic heart disease. Circulation 1988;78:296–301. [DOI] [PubMed] [Google Scholar]
- 33.Loring Z, Hanna P, Pellegrini CN. Longer Ambulatory ECG Monitoring Increases Identification of Clinically Significant Ectopy. Pacing Clin Electrophysiol 2016;39:592–7. [DOI] [PubMed] [Google Scholar]
- 34.Chen T, Koene R, Benditt DG, Lu F. Ventricular ectopy in patients with left ventricular dysfunction: should it be treated? J Card Fail 2013;19:40–9. [DOI] [PubMed] [Google Scholar]
- 35.Baman TS, Lange DC, Ilg KJ et al. Relationship between burden of premature ventricular complexes and left ventricular function. Heart Rhythm 2010;7:865–9. [DOI] [PubMed] [Google Scholar]
- 36.Bogun F, Crawford T, Reich S et al. Radiofrequency ablation of frequent, idiopathic premature ventricular complexes: comparison with a control group without intervention. Heart Rhythm 2007;4:863–7. [DOI] [PubMed] [Google Scholar]
- 37.Del Carpio Munoz F, Syed FF, Noheria A et al. Characteristics of premature ventricular complexes as correlates of reduced left ventricular systolic function: study of the burden, duration, coupling interval, morphology and site of origin of PVCs. J Cardiovasc Electrophysiol 2011;22:791–8. [DOI] [PubMed] [Google Scholar]
- 38.Hasdemir C, Yuksel A, Camli D et al. Late gadolinium enhancement CMR in patients with tachycardia-induced cardiomyopathy caused by idiopathic ventricular arrhythmias. Pacing Clin Electrophysiol 2012;35:465–70. [DOI] [PubMed] [Google Scholar]
- 39.Kawamura M, Badhwar N, Vedantham V et al. Coupling interval dispersion and body mass index are independent predictors of idiopathic premature ventricular complex-induced cardiomyopathy. J Cardiovasc Electrophysiol 2014;25:756–62. [DOI] [PubMed] [Google Scholar]
- 40.Maeda S, Chik WW, Liang JJ et al. Recovery of renal dysfunction after catheter ablation of outflow tract ventricular arrhythmias in patients with ventricular premature depolarization-mediated cardiomyopathy. J Interv Card Electrophysiol 2017;48:43–50. [DOI] [PubMed] [Google Scholar]
- 41.Takemoto M, Yoshimura H, Ohba Y et al. Radiofrequency catheter ablation of premature ventricular complexes from right ventricular outflow tract improves left ventricular dilation and clinical status in patients without structural heart disease. J Am Coll Cardiol 2005;45:1259–65. [DOI] [PubMed] [Google Scholar]
- 42.Agarwal V, Vittinghoff E, Whitman IR, Dewland TA, Dukes JW, Marcus GM. Relation Between Ventricular Premature Complexes and Incident Heart Failure. Am J Cardiol 2017;119:1238–1242. [DOI] [PubMed] [Google Scholar]
- 43.Agarwal SK, Heiss G, Rautaharju PM, Shahar E, Massing MW, Simpson RJ Jr. Premature ventricular complexes and the risk of incident stroke: the Atherosclerosis Risk In Communities (ARIC) Study. Stroke 2010;41:588–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Hasdemir C, Ulucan C, Yavuzgil O et al. Tachycardia-induced cardiomyopathy in patients with idiopathic ventricular arrhythmias: the incidence, clinical and electrophysiologic characteristics, and the predictors. J Cardiovasc Electrophysiol 2011;22:663–8. [DOI] [PubMed] [Google Scholar]
- 45.Latchamsetty R, Yokokawa M, Morady F et al. Multicenter Outcomes for Catheter Ablation of Idiopathic Premature Ventricular Complexes. JACC Clin Electrophysiol 2015;1:116–123. [DOI] [PubMed] [Google Scholar]
- 46.Lu F, Benditt DG, Yu J, Graf B. Effects of catheter ablation of “asymptomatic” frequent ventricular premature complexes in patients with reduced (<48%) left ventricular ejection fraction. Am J Cardiol 2012;110:852–6. [DOI] [PubMed] [Google Scholar]
- 47.Yokokawa M, Good E, Crawford T et al. Recovery from left ventricular dysfunction after ablation of frequent premature ventricular complexes. Heart Rhythm 2013;10:172–5. [DOI] [PubMed] [Google Scholar]
- 48.Singh SN, Fletcher RD, Fisher SG et al. Amiodarone in patients with congestive heart failure and asymptomatic ventricular arrhythmia. Survival Trial of Antiarrhythmic Therapy in Congestive Heart Failure. N Engl J Med 1995;333:77–82. [DOI] [PubMed] [Google Scholar]
- 49.Huizar JF, Fisher SG, Kaszala K et al. Amiodarone is an Effective Treatment of PVC-Cardiomyopathy in the Veteran Population (Abstract 14667). Circulation 2017;136. [Google Scholar]
- 50.Tan AY, Hu YL, Potfay J et al. Impact of ventricular ectopic burden in a premature ventricular contraction-induced cardiomyopathy animal model. Heart Rhythm 2016;13:755–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Sadron Blaye-Felice M, Hamon D, Sacher F et al. Reversal of left ventricular dysfunction after ablation of premature ventricular contractions related parameters, paradoxes and exceptions to the rule. Int J Cardiol 2016;222:31–6. [DOI] [PubMed] [Google Scholar]
- 52.Potfay J, Kaszala K, Tan AY et al. Abnormal Left Ventricular Mechanics of Ventricular Ectopic Beats: Insights Into Origin and Coupling Interval in Premature Ventricular Contraction-Induced Cardiomyopathy. Circ Arrhythm Electrophysiol 2015;8:1194–200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Kuroki K, Tada H, Seo Y et al. Prediction and mechanism of frequent ventricular premature contractions related to haemodynamic deterioration. Eur J Heart Fail 2012;14:1112–20. [DOI] [PubMed] [Google Scholar]
- 54.Cooper MW. Postextrasystolic potentiation. Do we really know what it means and how to use it? Circulation 1993;88:2962–71. [DOI] [PubMed] [Google Scholar]
- 55.Hamon D, Rajendran PS, Chui RW et al. Premature Ventricular Contraction Coupling Interval Variability Destabilizes Cardiac Neuronal and Electrophysiological Control: Insights From Simultaneous Cardioneural Mapping. Circ Arrhythm Electrophysiol 2017;10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Smith ML, Hamdan MH, Wasmund SL, Kneip CF, Joglar JA, Page RL. High-frequency ventricular ectopy can increase sympathetic neural activity in humans. Heart Rhythm 2010;7:497–503. [DOI] [PubMed] [Google Scholar]
- 57.Pacchia CF, Akoum NW, Wasmund S, Hamdan MH. Atrial bigeminy results in decreased left ventricular function: an insight into the mechanism of PVC-induced cardiomyopathy. Pacing Clin Electrophysiol 2012;35:1232–5. [DOI] [PubMed] [Google Scholar]
- 58.Kowgli NG, Jovin D, O’Quinn M et al. Neither Irregularity nor Tachycardia are Triggers of PVC-cardiomyopathy: Comparing Persistent Atrial and Ventricular Ectopy in an Animal Model. Heart Rhythm 2018;15:S594. [Google Scholar]
- 59.Walters TE, Rahmutula D, Szilagyi J et al. Left Ventricular Dyssynchrony Predicts the Cardiomyopathy Associated with Premature Ventricular Contractions (In Press). J Am Coll Cardiol 2018 [DOI] [PubMed] [Google Scholar]
- 60.Akyeampong D, Tan AY, kaszala K, Ellenbogen KA, Huizar JF. Premature Atrial Contractions Are Not associated With Left Ventricular Dysfunction (Abstract 14896). Circulation 2016;134. [Google Scholar]
- 61.Yokokawa M, Kim HM, Good E et al. Impact of QRS duration of frequent premature ventricular complexes on the development of cardiomyopathy. Heart Rhythm 2012;9:1460–4. [DOI] [PubMed] [Google Scholar]
- 62.Hamon D, Blaye-Felice MS, Bradfield JS et al. A New Combined Parameter to Predict Premature Ventricular Complexes Induced Cardiomyopathy: Impact and Recognition of Epicardial Origin. J Cardiovasc Electrophysiol 2016;27:709–17. [DOI] [PubMed] [Google Scholar]
- 63.Wang Y, Eltit JM, Kaszala K et al. Cellular Mechanism of Premature Ventricular Contraction-Induced Cardiomyopathy. Heart Rhythm 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Jiang M, Zhang M, Howren M et al. JPH-2 interacts with Cai-handling proteins and ion channels in dyads: Contribution to premature ventricular contraction-induced cardiomyopathy. Heart Rhythm 2016;13:743–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Sadron Blaye-Felice M, Hamon D, Sacher F et al. Premature ventricular contraction-induced cardiomyopathy: Related clinical and electrophysiologic parameters. Heart Rhythm 2016;13:103–10. [DOI] [PubMed] [Google Scholar]
- 66.Lee A, Denman R, Haqqani HM. Ventricular Ectopy in the Context of Left Ventricular Systolic Dysfunction: Risk Factors and Outcomes Following Catheter Ablation. Heart Lung Circ 2018. [DOI] [PubMed] [Google Scholar]
- 67.Ban JE, Park HC, Park JS et al. Electrocardiographic and electrophysiological characteristics of premature ventricular complexes associated with left ventricular dysfunction in patients without structural heart disease. Europace 2013;15:735–41. [DOI] [PubMed] [Google Scholar]
- 68.Zhong L, Lee YH, Huang XM et al. Relative efficacy of catheter ablation vs antiarrhythmic drugs in treating premature ventricular contractions: a single-center retrospective study. Heart Rhythm 2014;11:187–93. [DOI] [PubMed] [Google Scholar]
- 69.Penela D, Van Huls Van Taxis C, Van Huls Vans Taxis C et al. Neurohormonal, structural, and functional recovery pattern after premature ventricular complex ablation is independent of structural heart disease status in patients with depressed left ventricular ejection fraction: a prospective multicenter study. J Am Coll Cardiol 2013;62:1195–202. [DOI] [PubMed] [Google Scholar]
- 70.Penela D, Acosta J, Aguinaga L et al. Ablation of frequent PVC in patients meeting criteria for primary prevention ICD implant: Safety of withholding the implant. Heart Rhythm 2015;12:2434–42. [DOI] [PubMed] [Google Scholar]
- 71.Yarlagadda RK, Iwai S, Stein KM et al. Reversal of cardiomyopathy in patients with repetitive monomorphic ventricular ectopy originating from the right ventricular outflow tract. Circulation 2005;112:1092–7. [DOI] [PubMed] [Google Scholar]
- 72.Shanmugam N, Chua TP, Ward D. ‘Frequent’ ventricular bigeminy--a reversible cause of dilated cardiomyopathy. How frequent is ‘frequent’? Eur J Heart Fail 2006;8:869–73. [DOI] [PubMed] [Google Scholar]
- 73.Sarrazin JF, Labounty T, Kuhne M et al. Impact of radiofrequency ablation of frequent post-infarction premature ventricular complexes on left ventricular ejection fraction. Heart Rhythm 2009;6:1543–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Yokokawa M, Kim HM, Good E et al. Relation of symptoms and symptom duration to premature ventricular complex-induced cardiomyopathy. Heart Rhythm 2012;9:92–5. [DOI] [PubMed] [Google Scholar]
- 75.Carballeira Pol L, Deyell MW, Frankel DS et al. Ventricular premature depolarization QRS duration as a new marker of risk for the development of ventricular premature depolarization-induced cardiomyopathy. Heart Rhythm 2014; 11:299–306. [DOI] [PubMed] [Google Scholar]
- 76.Bas HD, Baser K, Hoyt J et al. Effect of circadian variability in frequency of premature ventricular complexes on left ventricular function. Heart Rhythm 2016;13:98–102. [DOI] [PubMed] [Google Scholar]
- 77.Knecht S, Sacher F, Wright M et al. Long-term follow-up of idiopathic ventricular fibrillation ablation: a multicenter study. J Am Coll Cardiol 2009;54:522–8. [DOI] [PubMed] [Google Scholar]
- 78.Olgun H, Yokokawa M, Baman T et al. The role of interpolation in PVC-induced cardiomyopathy. Heart Rhythm 2011;8:1046–9. [DOI] [PubMed] [Google Scholar]
- 79.Lee CH, Park KH, Nam JH et al. Increased variability of the coupling interval of premature ventricular contractions as a predictor of cardiac mortality in patients with left ventricular dysfunction. Circ J 2015;79:2360–6. [DOI] [PubMed] [Google Scholar]
- 80.Bradfield JS, Homsi M, Shivkumar K, Miller JM. Coupling interval variability differentiates ventricular ectopic complexes arising in the aortic sinus of valsalva and great cardiac vein from other sources: mechanistic and arrhythmic risk implications. J Am Coll Cardiol 2014;63:2151–2158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Mann SA, Castro ML, Ohanian M et al. R222Q SCN5A mutation is associated with reversible ventricular ectopy and dilated cardiomyopathy. J Am Coll Cardiol 2012;60:1566–73. [DOI] [PubMed] [Google Scholar]
- 82.Hwang JK, Park SJ, On YK, Kim JS, Park KM. Clinical Characteristics and Features of Frequent Idiopathic Ventricular Premature Complexes in the Korean Population. Korean Circ J 2015;45:391–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.El Kadri M, Yokokawa M, Labounty T et al. Effect of ablation of frequent premature ventricular complexes on left ventricular function in patients with nonischemic cardiomyopathy. Heart Rhythm 2015;12:706–13. [DOI] [PubMed] [Google Scholar]
- 84.Penela D, Martinez M, Fernandez-Armenta J et al. Influence of myocardial scar on the response to frequent premature ventricular complex ablation. Heart 2018. [DOI] [PubMed] [Google Scholar]
- 85.Topaloglu S, Aras D, Cagli K et al. Evaluation of left ventricular diastolic functions in patients with frequent premature ventricular contractions from right ventricular outflow tract. Heart Vessels 2007;22:328–34. [DOI] [PubMed] [Google Scholar]
- 86.Wijnmaalen AP, Delgado V, Schalij MJ et al. Beneficial effects of catheter ablation on left ventricular and right ventricular function in patients with frequent premature ventricular contractions and preserved ejection fraction. Heart 2010;96:1275–80. [DOI] [PubMed] [Google Scholar]
- 87.Yao J, Xu J, Yong YH, Cao KJ, Chen SL, Xu D. Evaluation of global and regional left ventricular systolic function in patients with frequent isolated premature ventricular complexes from the right ventricular outflow tract. Chin Med J (Engl) 2012;125:214–20. [PubMed] [Google Scholar]
- 88.Tung R, Bauer B, Schelbert H et al. Incidence of abnormal positron emission tomography in patients with unexplained cardiomyopathy and ventricular arrhythmias: The potential role of occult inflammation in arrhythmogenesis. Heart Rhythm 2015;12:2488–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Chen Y, Wu S, Li W et al. Higher High-Sensitivity C Reactive Protein is Associated with Future Premature Ventricular Contraction: a Community Based Prospective Cohort Study. Scientific Reports 2018;8:1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Anastasiou-Nana MI, Menlove RL, Nanas JN, Anderson JL. Changes in spontaneous variability of ventricular ectopic activity as a function of time in patients with chronic arrhythmias. Circulation 1988;78:286–95. [DOI] [PubMed] [Google Scholar]
- 91.Mountantonakis SE, Frankel DS, Gerstenfeld EP et al. Reversal of outflow tract ventricular premature depolarization-induced cardiomyopathy with ablation: effect of residual arrhythmia burden and preexisting cardiomyopathy on outcome. Heart Rhythm 2011;8:1608–14. [DOI] [PubMed] [Google Scholar]
- 92.Zang M, Zhang T, Mao J, Zhou S, He B. Beneficial effects of catheter ablation of frequent premature ventricular complexes on left ventricular function. Heart 2014;100:787–93. [DOI] [PubMed] [Google Scholar]
- 93.Hyman MC, Mustin D, Supple G et al. Class IC antiarrhythmic drugs for suspected premature ventricular contraction-induced cardiomyopathy. Heart Rhythm 2018;15:159–163. [DOI] [PubMed] [Google Scholar]
- 94.Naccarelli GV, Wolbrette DL, Patel HM, Luck JC. Amiodarone: clinical trials. Curr Opin Cardiol 2000;15:64–72. [DOI] [PubMed] [Google Scholar]
- 95.Penela D, Acosta J, Aguinaga L et al. Ablation of frequent PVC in patients meeting criteria for primary prevention ICD implant: Safety of withholding the implant. Heart Rhythm 2015;12:2434–42. [DOI] [PubMed] [Google Scholar]
- 96.Krishnan B, Sankar A, Anand I et al. Post-Extrasystolic Potentiation as a Predictor of Recovery of Left Ventricular Dysfunction After Radiofrequency Catheter Ablation. JACC Clin Electrophysiol 2017;3:1283–1291. [DOI] [PubMed] [Google Scholar]
- 97.Dukes JW, Dewland TA, Vittinghoff E et al. Ventricular Ectopy as a Predictor of Heart Failure and Death. J Am Coll Cardiol 2015;66:101–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Noda T, Shimizu W, Taguchi A et al. Malignant entity of idiopathic ventricular fibrillation and polymorphic ventricular tachycardia initiated by premature extrasystoles originating from the right ventricular outflow tract. J Am Coll Cardiol 2005;46:1288–94. [DOI] [PubMed] [Google Scholar]
- 99.Meinertz T, Hofmann T, Kasper W et al. Significance of ventricular arrhythmias in idiopathic dilated cardiomyopathy. Am J Cardiol 1984;53:902–7. [DOI] [PubMed] [Google Scholar]
- 100.Kirk JA, Kass DA. Electromechanical dyssynchrony and resynchronization of the failing heart. Circ Res 2013;113:765–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Taieb JM, Maury P, Shah D et al. Reversal of dilated cardiomyopathy by the elimination of frequent left or right premature ventricular contractions. J Interv Card Electrophysiol 2007;20:9–13. [DOI] [PubMed] [Google Scholar]