Abstract
Background
Parecoxib was the first COX‐2 available for parenteral administration, and may, given intravenously or intramuscularly, offer advantages over oral medication when patients have nausea and vomiting or are unable to swallow, such as in the immediate postoperative period.
Objectives
Assess the efficacy of single dose intravenous or intramuscular parecoxib in acute postoperative pain, the requirement for rescue medication, and any associated adverse events.
Search methods
We searched Cochrane CENTRAL, MEDLINE, EMBASE in November 2008.
Selection criteria
Randomised, double‐blind, placebo‐controlled clinical trials of parecoxib compared with placebo for relief of acute postoperative pain in adults.
Data collection and analysis
Two review authors independently assessed trial quality and extracted data. The area under the “pain relief versus time” curve was used to derive the proportion of participants with parecoxib and placebo experiencing at least 50% pain relief over 6 hours, using validated equations. The number‐needed‐to‐treat‐to‐benefit (NNT) was calculated using 95% confidence intervals (CI). The proportion of participants using rescue analgesia over a specified time period, and time to use of rescue analgesia, were sought as additional measures of efficacy. Information on adverse events and withdrawals were also collected.
Main results
Seven studies (1446 participants) were included. There was no significant difference between doses, or between intravenous and intramuscular administration for 50% pain relief over 6 hours: NNTs compared with placebo were 3.1 (2.4 to 4.5), 2.4 (2.1 to 2.8), and 1.8 (1.5 to 2.3) for 10, 20, and 40 mg parecoxib respectively. Fewer participants required rescue medication over 24 hours with parecoxib than placebo: parecoxib 40 mg was significantly better than parecoxib 20 mg (NNTs to prevent use of rescue medication 7.5 (5.3 to 12.8) and 3.3 (2.6 to 4.5) respectively; P < 0.0007). Median time to use of rescue medication was 3.1 hours, 6.9 hours and 10.6 hours with parecoxib 10 mg, 20 mg and 40 mg respectively, and 1.5 hours with placebo. Adverse events were generally mild to moderate, rarely led to withdrawal, and did not differ in frequency between groups. No serious adverse events were reported with parecoxib or placebo.
Authors' conclusions
A single dose of parecoxib 20 mg or 40 mg provided effective analgesia for 50 to 60% of those treated compared to about 15% with placebo, and was well tolerated. Duration of analgesia was longer, and significantly fewer participants required rescue medication over 24 hours with the higher dose.
Plain language summary
Parecoxib delivered intramuscularly or intravenously (injected in to the muscle or the vein) for acute postoperative pain in adults
The most common route for administration of postoperative analgesia is by mouth, but some patients are unable to swallow, feel nauseated, or vomit in the immediate postoperative period, and in these patients intravenous or intramuscular administration may be preferred. This review assessed seven studies of parecoxib, an injectable COX‐2 inhibitor, for acute postoperative pain relief. Single doses of 20 mg or 40 mg provided effective pain relief in 50 to 60% of treated individuals, compared with 15% treated with placebo. Duration of pain relief was longer with the higher dose (10.6 hours for 40 mg versus 6.9 hours for 20 mg), and significantly fewer individuals on the higher dose required rescue medication over 24 hours (66% versus 81%). Adverse events were generally mild to moderate in severity and were reported by just over half of treated individuals in both parecoxib and placebo groups.
Background
Acute pain occurs as a result of tissue damage, commonly accidentally due to an injury or as a result of surgery. Acute postoperative pain is a manifestation of inflammation due to tissue injury. The management of postoperative pain and inflammation is a critical component of patient care.
This is one of a series of reviews that aim to present evidence for relative analgesic efficacy through indirect comparisons with placebo, in very similar trials performed in a standard manner, with very similar outcomes, and over the same duration. Such relative analgesic efficacy does not in itself determine choice of drug for any situation or patient, but guides policy‐making at the local level.
Recent reviews include lumiracoxib (Roy 2007), paracetamol (Toms 2008), and celecoxib (Derry 2008), and the series will include updates of existing reviews like ibuprofen (Collins 1999) and aspirin (Oldman 1999), in addition to new reviews such as ketoprofen and dexketoprofen (Barden 2008), lornoxicam (Hall 2008), diflunisal (Moore 2008).
Single dose trials in acute pain are commonly short in duration, rarely lasting longer than 12 hours. The number of participants are small, allowing no reliable conclusions to be drawn about safety. To show that the analgesic is working it is necessary to use placebo (McQuay 2005). There are clear ethical considerations in doing this. These ethical considerations are answered by using acute pain situations where the pain is expected to go away, and by providing additional analgesia, commonly called rescue analgesia, if the pain has not diminished after about an hour. This is reasonable, because not all participants given an analgesic will have significant pain relief. Approximately 18% of participants given placebo will have significant pain relief (Moore 2006), and up to 50% may have inadequate analgesia with active medicines. The use of additional or rescue analgesia is hence important for all participants in the trials.
Clinical trials measuring the efficacy of analgesics in acute pain have been standardised over many years. Trials have to be randomised and double blind. Typically, in the first few hours or days after an operation, patients develop pain that is moderate to severe in intensity, and will then be given the test analgesic or placebo. Pain is measured using standard pain intensity scales immediately before the intervention, and then using pain intensity and pain relief scales over the following 4 to 6 hours for shorter acting drugs, and up to 12 or 24 hours for longer acting drugs. Pain relief of half the maximum possible pain relief or better (at least 50% pain relief) is typically regarded as a clinically useful outcome in this setting. For patients given rescue medication it is usual for no additional pain measurements to be made, and for all subsequent measures to be recorded as initial pain intensity or baseline (zero) pain relief (baseline observation carried forward). This process ensures that analgesia from the rescue medication is not wrongly ascribed to the test intervention. In some trials the last observation is carried forward, which gives an inflated response for the test intervention compared to placebo, but the effect has been shown to be negligible over 4 to 6 hours (Moore 2005). Patients often remain in the hospital or clinic for at least the first 6 hours following the intervention, with measurements supervised, although they may then be allowed home to make their own measurements in trials of longer duration.
Clinicians prescribe non‐steroidal anti‐inflammatory drugs (NSAIDs) on a routine basis for a range of mild‐to‐moderate pain. NSAIDs are the most commonly prescribed analgesic medications worldwide, and their efficacy for treating acute pain has been well demonstrated (Moore 2003). They reversibly inhibit cyclooxygenase (prostaglandin endoperoxide synthase), the enzyme mediating production of prostaglandins and thromboxane A2 (FitzGerald 2001). Prostaglandins mediate a variety of physiological functions such as maintenance of the gastric mucosal barrier, regulation of renal blood flow, and regulation of endothelial tone. They also play an important role in inflammatory and nociceptive processes. However, relatively little is known about the mechanism of action of this class of compounds aside from their ability to inhibit cyclooxygenase‐dependent prostanoid formation (Hawkey 1999). Since NSAIDs do not depress respiration and do not impair gastro‐intestinal motility, as do opioids (BNF 2002), they are clinically useful for treating pain after minor surgery and day surgery, and have an opiate‐sparing effect after more major surgery (Grahame‐Smith 2002).
Cyclooxygenase (COX) activity has been found to be associated with at least two distinct isoenzymes: COX‐1 and COX‐2. COX‐1 was hypothesized to be involved in the maintenance of physiologic functions such as gastric protection and haemostasis; COX‐2 was thought to be involved in pathophysiologic processes such as inflammation, pain and fever. These hypotheses led to the development of the selective COX‐2 inhibitors, such as celecoxib, rofecoxib and etoricoxib. These agents have analgesic efficacy comparable with conventional NSAIDs. In addition, they have no anti‐platelet activity at therapeutic doses, and therefore may be associated with reduced gastrointestinal adverse effects compared with conventional NSAIDs such as ibuprofen. Concerns about cardiovascular safety in long term use have led to the withdrawal of rofecoxib, and in some countries lumiracoxib.
The most common route for postoperative analgesia is oral, but when patients are unable to swallow, for instance perioperatively, parenteral administration (e.g., intramuscular, intravenous, intrathecal) may be preferred. Some NSAIDs can be administered intramuscularly (e.g., diclofenac, ketoprofen and ketorolac) but this route can be painful in itself. Diclofenac and ketorolac can be administered intravenously, but ketorolac is contraindicated for patients receiving heparin and those who might be susceptible to bleeding from gastrointestinal ulcers or who have renal impairment.
Parecoxib
Parecoxib was the first COX‐2 to be administered parenterally. It is a prodrug (the parent drug is inactive) that is rapidly hydrolysed in vivo to its active form, valdecoxib. Clinical trials have indicated that parecoxib is effective in treating postoperative pain resulting from oral surgery, orthopaedic surgery and abdominal hysterectomy pain. Other studies have demonstrated no significant effects on platelet function or upper gastrointestinal mucosa (Graff 2007; Harris 2004; Noveck 2001; Stoltz 2002). As a result, parecoxib sodium has been approved in European countries for the treatment of postoperative pain. In the UK, for example, parecoxib 20 mg or 40 mg powder (and solvent or solution for injection) is licensed for the short‐term treatment of postoperative pain.
In 2002 concerns were raised about the potential for serious adverse effects from parecoxib because of reactions experienced by some patients to valdecoxib, the active metabolite of parecoxib sodium. These effects included anaphylaxis, angioedema, and serious skin reactions such as toxic epidermal necrolysis (EMEA 2002), and led to withdrawal of valdecoxib in 2005. As a result, parecoxib is contraindicated in patients who have a history of sensitivity to sulphonamides (a type of antibiotic used to treat infections) because of the risk of severe adverse effects. A recent review of prescription event monitoring has shown that the incidence of serious skin reactions with any coxib is very low (crude rate 0.008%; Layton 2006).
A previously published systematic review (Barden 2003) assessed the evidence for the effectiveness of parecoxib for treating postoperative pain from four randomised controlled trials (RCTs) (620 participants) and concluded that parecoxib is an effective analgesic in the postoperative setting. Since then, new studies have been published, and it is hoped that they will provide additional data for more robust estimates of the benefits and harms of parecoxib.
Objectives
To assess the efficacy and adverse effects of single dose parecoxib in studies of acute postoperative pain using methods that permit comparison with other analgesics evaluated in standardised trials using almost identical methods and outcomes.
Methods
Criteria for considering studies for this review
Types of studies
Studies were included if they were double blind trials of single dose parecoxib compared with placebo for the treatment of moderate to severe postoperative pain in adults with at least ten participants randomly allocated to each treatment group. Multiple dose studies were included if appropriate data from the first dose was available. Cross‐over studies were included provided data from the first arm was presented separately. No language restriction was applied to the search for studies.
The following were excluded:
review articles, case reports, and clinical observations;
studies of experimental pain;
studies where pain relief is assessed only by clinicians, nurses or carers (i.e. not patient‐reported);
studies of less than four hours duration or studies that fail to present data over four to 6 hours post‐dose.
For postpartum pain, studies were included if the pain investigated was due to episiotomy or Caesarean section irrespective of the presence of uterine cramps; studies investigating pain due to uterine cramps alone were excluded.
Types of participants
Studies of adult participants (at least 15 years) with established postoperative pain of moderate to severe intensity following day surgery or in‐patient surgery were included. For studies using a visual analogue scale (VAS), pain of at least moderate intensity was equated to greater than 30 mm (Collins 1997).
Types of interventions
Parecoxib or matched placebo administered as a single parenteral dose for postoperative pain.
Types of outcome measures
Data was collected on the following outcomes:
participant characteristics;
patient reported pain at baseline (physician, nurse or carer reported pain will not be included in the analysis);
patient reported pain relief expressed at least hourly over four to 6 hours using validated pain scales (pain intensity and pain relief in the form of VAS or categorical scales, or both);
patient reported global evaluation of treatment (PGE);
time to use of rescue medication;
number of participants using rescue medication;
number of participants with one or more adverse events;
number of participants with serious adverse events;
number of withdrawals (all cause, adverse event).
Search methods for identification of studies
Electronic searches
To identify studies for inclusion in this review, the following electronic databases were searched:
Cochrane CENTRAL;
MEDLINE via Ovid;
EMBASE via Ovid;
Oxford Pain Relief Database (Jadad 1996a).
Please see Appendix 1 for the MEDLINE search strategy, Appendix 2 for the EMBASE search strategy and Appendix 3 for the Cochrane CENTRAL search strategy.
Searching other resources
Additional studies were sought from the reference lists of retrieved articles, textbooks and reviews. The manufacturing pharmaceutical company (Pharmacia) was not contacted for unpublished trial data.
Language
No language restriction was applied.
Data collection and analysis
QUOROM guidelines were followed (Moher 1999).
Selection of studies
Two review authors independently assessed and agreed the search results for studies that were included in the review.
Quality assessment
Two review authors independently assessed the included studies for quality using a five‐point scale (Jadad 1996b) that considers randomisation, blinding, study withdrawals and dropouts.
The scale used is as follows:
Is the study randomised? If yes give one point.
Is the randomisation procedure reported and is it appropriate? If yes add one point, if no deduct one point.
Is the study double blind? If yes then add one point.
Is the double blind method reported and is it appropriate? If yes add 1 point, if no deduct one point.
Are the reasons for patient withdrawals and dropouts described? If yes add one point.
The results are described in the 'Methodological quality of included studies' section below, and 'Characteristics of included studies' table.
Data management
Data was extracted by two review authors and recorded on a standard data extraction form. Data suitable for pooling was entered into RevMan 5.
Data analysis
For efficacy analyses we used the number of participants in each treatment group who were randomised, received medication, and provided at least one post‐baseline assessment. For safety analyses we used number of participants who received study medication in each treatment group. Analyses were planned for different doses. Sensitivity analyses were planned for the primary outcome to determine the effect of the pain model (dental versus other postoperative pain) and the quality score (two versus three or more). A minimum of two studies and 200 participants were required for any analysis (Moore 1998). Where only one active treatment arm in a study contributed to a particular comparison, the whole placebo group was used for analysis. When more than one active treatment arm contributed to a comparison, the placebo group was divided equally between the contributing arms for the analysis.
Primary outcome: number of participants with at least 50% pain relief
For each study, the mean TOTPAR, SPID, VAS TOTPAR or VAS SPID (Appendix 4) values for active and placebo were converted to %maxTOTPAR or %maxSPID by division into the calculated maximum value (Cooper 1991). The proportion of participants in each treatment group who achieved at least 50%maxTOTPAR were calculated using verified equations (Moore 1996; Moore 1997a; Moore 1997b). These proportions were then converted into the number of participants achieving at least 50%maxTOTPAR by multiplying by the total number of participants in the treatment group. Information on the number of participants with at least 50%maxTOTPAR for active and placebo was then used to calculate relative benefit/relative risk (RR), and number needed to treat to benefit (NNT).
Pain measures accepted for the calculation of TOTPAR or SPID were:
five‐point categorical pain relief (PR) scales with comparable wording to "none, slight, moderate, good or complete";
four‐point categorical pain intensity (PI) scales with comparable wording to "none, mild, moderate, severe";
visual analogue scales (VAS) for pain relief;
VAS for pain intensity.
If none of these measures were available, numbers of participants reporting "very good or excellent" on a five‐point categorical global scale with the wording "poor, fair, good, very good, excellent" were taken as those achieving at least 50% pain relief (Collins 2001).
Further details of the scales and derived outcomes are in the glossary (Appendix 4).
Secondary outcomes:
1. Use of rescue medication
Numbers of participants requiring rescue medication were used to calculate RR and numbers needed to treat to prevent (NNTp) use of rescue medication for treatment and placebo groups. Median (or mean) time to use of rescue medication was used to calculate the weighted mean of the median (or mean) for the outcome. Weighting was by number of participants.
2. Adverse events
Numbers of participants reporting adverse events for each treatment group were used to calculate RR and numbers needed to treat to harm (NNH) estimates for:
any adverse event;
any serious adverse event (as reported in the study);
withdrawal due to an adverse event.
3. Withdrawals
Withdrawals for reasons other than lack of efficacy (participants using rescue medication ‐ see above) and adverse events were noted, as were exclusions from analysis where data were presented.
Relative benefit or risk estimates were calculated with 95% confidence intervals (CI) using a fixed‐effect model (Morris 1995). NNT, NNTp and NNH with 95% CI were calculated using the pooled number of events by the method of Cook and Sackett (Cook 1995). A statistically significant difference from control was assumed when the 95% CI of the relative benefit did not include the number one.
Homogeneity of studies was assessed visually (L'Abbé 1987). The z test (Tramèr 1997) was used to determine if there was a significant difference between NNTs for different doses of active treatment, or between groups in the sensitivity analyses.
Results
Description of studies
Seven studies, with 1446 participants in total, fulfilled the entry criteria (Barton 2002; Bikhazi 2004; Daniels 2001; Malan 2005; Mehlisch 2003; Mehlisch 2004; Rasmussen 2002). Details of included and excluded studies are in the corresponding "Characteristics of studies" tables.
Three studies contained two relevant active treatment arms, (Barton 2002; Bikhazi 2004; Rasmussen 2002), one study contained four treatment arms (Daniels 2001), one study contained five treatment arms (Mehlisch 2004) and one contained seven treatment arms (Mehlisch 2003).
Six of the studies included a treatment arm using parecoxib 20 mg, with a total of 591 participants in the comparison with placebo (Barton 2002; Bikhazi 2004; Daniels 2001; Mehlisch 2003; Mehlisch 2004; Rasmussen 2002). Parecoxib was administered by the intravenous route in five of these studies, with a total of 414 participants, (Barton 2002; Bikhazi 2004; Daniels 2001; Mehlisch 2003; Rasmussen 2002) and by the intramuscular route in two studies, with a total of 177 participants (Daniels 2001; Mehlisch 2004).
Five of the studies included a treatment arm using parecoxib 40 mg, with a total of 519 participants in the comparison with placebo (Barton 2002; Bikhazi 2004; Daniels 2001; Malan 2005; Rasmussen 2002). Parecoxib was administered by the intravenous route in four of these studies, with a total of 311 participants, (Barton 2002; Bikhazi 2004; Daniels 2001; Rasmussen 2002) and by the intramuscular route in two studies, with a total of 208 participants (Daniels 2001; Malan 2005).
Two studies (Mehlisch 2003; Mehlisch 2004) used doses of 1 mg, with a total of 202 participants in the comparison with placebo; 2 mg with a total of 201 participants; 5 mg with a total of 202 participants; and 10 mg with a total of 200 participants. One of these studies (Mehlisch 2003) also used doses of 50 mg, with a total of 101 participants, and 100 mg, with a total of 101 participants in comparisons with placebo.
Three studies enrolled participants with dental pain following surgical extraction of at least two impacted third molars (Daniels 2001; Mehlisch 2003; Mehlisch 2004), three studies enrolled participants with pain following gynaecologic laparotomy surgery (Barton 2002; Bikhazi 2004; Malan 2005), and one study enrolled participants with pain following unilateral total knee replacement surgery (Rasmussen 2002).
There is an over‐representation of women in the included studies. Three of the seven studies were in gynecological patients, and the other four studies all included more women than men.
Study duration was 24 hours in six of the studies (Barton 2002; Bikhazi 2004; Daniels 2001; Mehlisch 2003; Mehlisch 2004; Rasmussen 2002) and 12 hours in one study (Malan 2005).
One study (Bikhazi 2004) included a multiple dose phase, but reported results for the first dose separately for some relevant outcomes. All other studies used only single doses.
Risk of bias in included studies
Each of the seven studies was scored for methodological quality. Two studies were given a quality score of five (Daniels 2001; Mehlisch 2004) and five studies a score of four (Barton 2002; Bikhazi 2004; Malan 2005; Mehlisch 2003; Rasmussen 2002). Full details can be found in the 'Characteristics of included studies' table.
Effects of interventions
Details of study efficacy outcomes (analgesia and use of rescue medication) are in Table 1, and details of adverse events and withdrawals are in Table 2. Summary tables are provided throughout the text.
1. Summary of Outcomes: analgesia and use of rescue medication.
| Analgesia | Rescue medication | |||||
| Study ID | Treatment | PR | Number with 50% PR | PGE: very good or excellent | Time to use (h) | % using |
| Barton 2002 | (1) Parecoxib 20 mg IV, n = 39 (2) Parecoxib 40 mg IV, n = 38 (3) Ketorolac 30 mg IV, n = 41 (4) Morphine 4 mg IV, n = 42 (5) Placebo, n = 42 |
TOTPAR 6: (1) 10.9 (2) 11.4 (5) 4.2 |
(1) 18/38 (2) 20/38 (5) 5/39 |
No usable data | (1) 6.2 (2) 6.5 (5) 1.8 |
at 24 h: (1) 92 (2) 87 (5) 97 |
| Bikhazi 2004 | (1) Parecoxib 20 mg IV, n = 40 (2) Parecoxib 40 mg IV, n = 41 (3) Ketorolac 30 mg IV, n = 42 (4) Morphine 4 mg IV, n = 40 (5) Placebo, n = 45 |
TOTPAR 6: (1) 12.0 (2) 15.4 (5) 8.0 |
(1) 21/38 (2) 30/41 (5) 14/44 |
No usable data | (1) 6.1 (2) 6.3 (5) 2.8 |
at 6 h: (1) 37 (2) 32 (5) 71 |
| Daniels 2001 | (1) Parecoxib 20 mg IM, n = 51 (2) Parecoxib 20 mg IV, n = 50 (3) Parecoxib 40 mg IM, n = 50 (4) Parecoxib 40 mg IV, n = 51 (5) Ketorolac 60 mg IM, n = 51 (6) Placebo, n = 51 |
TOTPAR 6: (1) 12.6 (2) 12.4 (3) 15.1 (4) 13.8 (6) 2.7 |
(1) 30/51 (2) 29/50 (3) 36/50 (4) 33/51 (6) 2/51 |
No usable data | (1) 9.3 (2) 7.1 (3) 21.7 (4) 15.7 (6) 1.1 |
at 24 h: (1) 76 (2) 80 (3) 50 (4) 53 (6) 94 |
| Malan 2005 | (1) Parecoxib 40 mg IM, n = 62 (2) Morphine 6 mg IM, n = 70 (3) Morphine 12 mg IM, n = 62 (4) Placebo, n = 70 |
TOTPAR 6: (1) 13.3 (4) 5.8 |
(1) 39/62 (4) 14/70 |
No usable data | (1) 5.9 (4) 2.2 |
at 12 h: (1) 79 (4) 99 at 6 h: (1) 49 (4) 90 |
| Mehlisch 2003 | (1) Parecoxib 1 mg IV, n = 51 (2) Parecoxib 2 mg IV, n = 51 (3) Parecoxib 5 mg IV, n = 51 (4) Parecoxib 10 mg IV, n = 51 (5) Parecoxib 20 mg IV, n = 51 (6) Parecoxib 50 mg IV, n = 51 (7) Parecoxib 100 mg IV, n = 51 (8) Ketorolac 30 mg IV, n = 50 (9) Placebo, n = 50 |
TOTPAR 6: (1) 3.8 (2) 3.4 (3) 8.7 (4) 8.7 (5) 12.3 (6) 12.6 (7) 13 (9) 1.8 |
(1) 5/51 (2) 4/51 (3) 19/51 (4) 19/51 (5) 28/50 (6) 30/51 (7) 31/51 (9) 0/50 |
No usable data | (1) 1.1 (2) 1.1 (3) 3.1 (4) 2.2 (5) 8.0 (6) 10.6 (7) 13.5 (9) 1.0 |
at 24 h: (1) 98 (2) 96 (3) 80 (4) 90 (5) 80 (6) 57 (7) 55 (9) 98 |
| Mehlisch 2004 | (1) Parecoxib 1 mg IM, n = 51 (2) Parecoxib 2 mg IM, n = 50 (3) Parecoxib 5 mg IM, n = 51 (4) Parecoxib 10 mg IM, n = 50 (5) Parecoxib 20 mg IM, n = 50 (6) Ketorolac 30 mg IM, n = 51 (7) Placebo, n = 50 |
TOTPAR 6: (1) 4.67 (2) 6.37 (3) 6.11 (4) 7.61 (5) 12.6 (7) 2.93 |
(1) 7/51 (2) 12/50 (3) 11/51 (4) 15/49 (5) 29/50 (7) 2/50 |
No usable data | (1) 1.53 (2) 1.57 (3) 1.53 (4) 4.07 (5) 7.68 (7) 1.05 |
at 24 h: (1) 84 (2) 84 (3) 84 (4) 92 (5) 66 (7) 86 |
| Rasmussen 2002 | (1) Parecoxib 20 mg IV, n = 43 (2) Parecoxib 40 mg IV, n = 42 (3) Ketorolac 30 mg IV, n = 42 (4) Morphine 4 mg IV, n = 42 (5) Placebo, n = 39 |
TOTPAR 6: (1) 7.89 (2) 10.81 (5) 5.19 |
(1) 14/43 (2) 17/36 (5) 6/37 |
No usable data | (1) 3.15 (2) 5.17 (5) 1.8 |
at 24 h: (1) 95 (2) 86 (5) 100 |
IM ‐ intramuscular; IV ‐ intravenous
2. Summary of Outcomes: adverse events and withdrawals.
| Adverse events | Withdrawals | ||||
| Study ID | Treatment | Any | Serious | Adverse event | Other |
| Barton 2002 | (1) Parecoxib 20 mg IV, n = 39 (2) Parecoxib 40 mg IV, n = 38 (3) Ketorolac 30 mg IV, n = 41 (4) Morphine 4 mg IV, n = 42 (5) Placebo, n = 42 |
(1) 34/39 (2) 32/38 (5) 31/42 |
None related to study medication but 2 in (4) | (1) 5/39 (2) 4/38 (5) 2/42 |
4 pts excluded from efficacy analysis: 3 pts in (5) before completing 1 hr assessment and 1 pt in (1) due to protocol violation |
| Bikhazi 2004 | (1) Parecoxib 20 mg IV, n = 40 (2) Parecoxib 40 mg IV, n = 41 (3) Ketorolac 30 mg IV, n = 42 (4) Morphine 4 mg IV, n = 40 (5) Placebo, n = 45 |
No single dose data | None related to study medication | No single dose data | 5 pts excluded from efficacy analysis: 1 had inadequate baseline pain, 4 used rescue med within 1 h |
| Daniels 2001 | (1) Parecoxib 20 mg IM, n = 51 (2) Parecoxib 20 mg IV, n = 50 (3) Parecoxib 40 mg IM, n = 50 (4) Parecoxib 40 mg IV, n = 51 (5) Ketorolac 60 mg IM, n = 51 (6) Placebo, n = 51 |
(1) 14/51 (2) 21/50 (3) 17/50 (4) 20/51 (6) 18/51 None related to dose or route of study medication |
None reported | None | 1 pt in (6) excluded due to non‐compliance |
| Malan 2005 | (1) Parecoxib 40 mg IM, n = 62 (2) Morphine 6 mg IM, n = 70 (3) Morphine 12 mg IM, n = 62 (4) Placebo, n = 70 |
(1) 36/62 (4) 41/70 Related to study medication (1) 19/62 (4) 28/70 |
None | (1) 1/62 | 14 pts excluded from efficacy analysis: protocol violation, failed inclusion and exclusion criteria, used rescue med within 1 h, missing consecutIVe PR scores |
| Mehlisch 2003 | (1) Parecoxib 1 mg IV, n = 51 (2) Parecoxib 2 mg IV, n = 51 (3) Parecoxib 5 mg IV, n = 51 (4) Parecoxib 10 mg IV, n = 51 (5) Parecoxib 20 mg IV, n = 51 (6) Parecoxib 50 mg IV, n = 51 (7) Parecoxib 100 mg IV, n = 51 (8) Ketorolac 30 mg IV, n = 50 (9) Placebo, n = 50 |
(1) 30/51 (2) 32/51 (3) 28/51 (4) 25/51 (5) 25/51 (6) 25/51 (7) 28/51 (9) 27/50 |
None | None | 3 pts excluded from efficacy analysis, 1 in (5) and 2 in (8), due to infiltration of study medication into surrounding tissue during administration |
| Mehlisch 2004 | (1) Parecoxib 1 mg IM, n = 51 (2) Parecoxib 2 mg IM, n = 50 (3) Parecoxib 5 mg IM, n = 51 (4) Parecoxib 10 mg IM, n = 50 (5) Parecoxib 20 mg IM, n = 50 (6) Ketorolac 30 mg IM, n = 51 (7) Placebo, n = 50 |
(1) 25/51 (2) 21/50 (3) 30/51 (4) 28/50 (5) 25/50 (7) 26/50 |
None | None | 2 pts excluded: 1 in (4) and 1 in (6), due to protocol violation |
| Rasmussen 2002 | (1) Parecoxib 20 mg IV, n = 43 (2) Parecoxib 40 mg IV, n = 42 (3) Ketorolac 30 mg IV, n = 42 (4) Morphine 4 mg IV, n = 42 (5) Placebo, n = 39 |
(1) 31/43 (2) 25/42 (5) 24/39 |
None related to study medication | (2) 1/42 | 10 pts excluded: 5 due to non‐compliance after administration of study medication; 4 due to protocol violation; 1 due to administration of the wrong dose of parecoxib |
IM ‐ intramuscular; IV ‐ intravenous; pt ‐ participant
Number of participants achieving at least 50% pain relief
The number of participants studies for parecoxib 1, 2 and 5 mg is low and results should therefore be interpreted with caution.
Parecoxib 1 mg versus placebo
Two studies provided data at this dose (Mehlisch 2003; Mehlisch 2004), in which 102 participants were treated with parecoxib 1 mg and 100 with placebo (please see Table 1 and Summary of results A).
The proportion of participants experiencing at least 50% pain relief over 6 hours with parecoxib 1 mg was 12% (12/102).
The proportion of participants experiencing at least 50% pain relief over 6 hours with placebo was 2% (2/100).
The relative benefit of parecoxib 1 mg compared with placebo was 4.9 (1.3 to 18).
The NNT for at least 50% pain relief over 6 hours was 10 (6.0 to 37).
Parecoxib 2 mg versus placebo
Two studies provided data at this dose (Mehlisch 2003; Mehlisch 2004), in which 101 participants were treated with parecoxib 2 mg and 100 with placebo (please see Table 1 and Summary of results A).
The proportion of participants experiencing at least 50% pain relief over 6 hours with parecoxib 2 mg was 16% (16/101).
The proportion of participants experiencing at least 50% pain relief over 6 hours with placebo was 2% (2/100).
The relative benefit of parecoxib 2 mg compared with placebo was 6.6 (1.8 to 24).
The NNT for at least 50% pain relief over 6 hours was 7.2 (4.6 to 17).
Parecoxib 5 mg versus placebo
Two studies provided data at this dose (Mehlisch 2003; Mehlisch 2004), in which 102 participants were treated with parecoxib 5 mg and 100 with placebo (please see Table 1 and Summary of results A).
The proportion of participants experiencing at least 50% pain relief over 6 hours with parecoxib 5 mg was 16% (16/101).
The proportion of participants experiencing at least 50% pain relief over 6 hours with placebo was 2% (2/100).
The relative benefit of parecoxib 5 mg compared with placebo was 12 (3.4 to 42).
The NNT for at least 50% pain relief over 6 hours was 3.7 (2.7 to 5.6).
Parecoxib 20 mg versus placebo
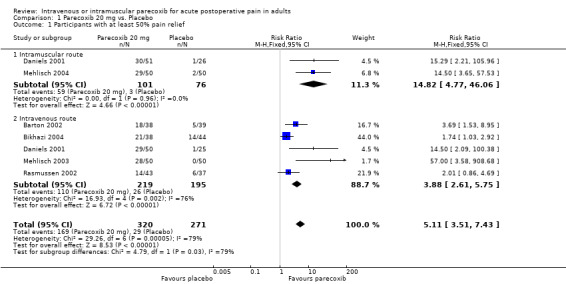
Six studies provided data at this dose (Barton 2002; Bikhazi 2004; Daniels 2001; Mehlisch 2003; Mehlisch 2004; Rasmussen 2002), in which 320 participants were treated with parecoxib 20 mg and 271 with placebo (please see Table 1; Figure 1; and Summary of results A).
The proportion of participants experiencing at least 50% pain relief over 6 hours with parecoxib 20 mg was 53% (169/320).
The proportion of participants experiencing at least 50% pain relief at 6 hours with placebo was 11% (29/271).
The relative benefit of parecoxib 20 mg compared with placebo was 5.1 (3.5 to 7.4).
The NNT for at least 50% pain relief over 6 hours was 2.4 (2.1 to 2.8). For every two participants treated with parecoxib 20 mg, one would experience at least 50% pain relief who would not have done so with placebo.
1.
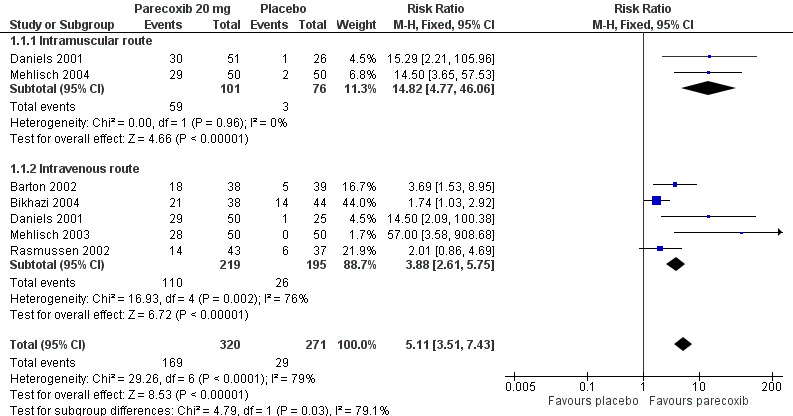
Forest plot of comparison: 2 Parecoxib 20 mg vs. Placebo, outcome: 2.1 Participants with at least 50% pain relief.
Parecoxib 40 mg versus placebo
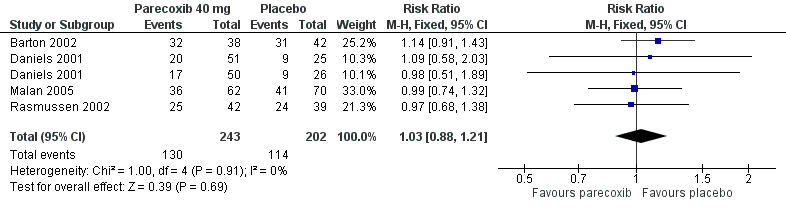
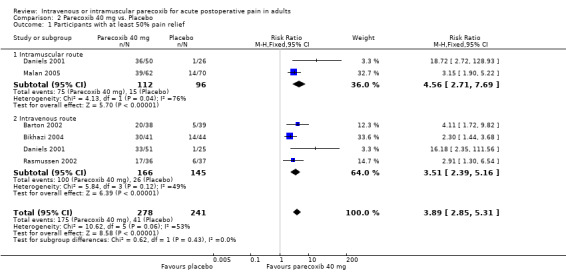
Five studies provided data at this dose (Barton 2002; Bikhazi 2004; Daniels 2001; Malan 2005; Rasmussen 2002), in which 278 participants were treated with parecoxib 40 mg and 241 with placebo (please see Table 1; Figure 2; and Summary of results A).
The proportion of participants experiencing at least 50% pain relief over 6 hours with parecoxib 40 mg was 63% (175/278).
The proportion of participants experiencing at least 50% pain relief over 6 hours with placebo was 17% (41/241).
The relative benefit of parecoxib 40 mg compared with placebo was 3.9 (2.9 to 5.3).
The NNT for at least 50% pain relief over 6 hours was 2.2 (1.9 to 2.6). For every two participants treated with 40 mg parecoxib, one would experience at least 50% pain relief who would not have done so with placebo.
2.
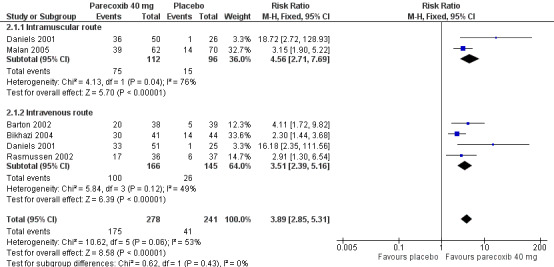
Forest plot of comparison: 3 Parecoxib 40 mg vs. Placebo, outcome: 3.1 Participants with at least 50% pain relief.
Parecoxib 50 mg and 100 mg were used in only one study (Mehlisch 2003), in which only 51 participants were treated with each dose, and are not analysed further.
Although there was a trend for dose response, no significant difference between parecoxib 20 mg and parecoxib 40 mg was demonstrated for the outcome of at least 50% pain relief over 6 hours.
| Summary of results A: participants with at least 50% pain relief over 6 hours | ||||||
| Dose | Number of studies | Number of participants | 50% PR parecoxib (%) | 50% PR placebo (%) | RB (95% CI) | NNT (95% CI) |
| 1 mg | 2 | 202 | 12 | 3 | 4.9 (1.3 to 18) | 10 (5.9 to 37) |
| 2 mg | 2 | 201 | 16 | 3 | 6.6 (1.8 to 24) | 7.2 (4.6 to 17) |
| 5 mg | 2 | 202 | 30 | 3 | 12.0 (3.4 to 42) | 3.7 (2.7 to 5.6) |
| 10 mg | 2 | 200 | 35 | 3 | 14.0 (3.9 to 49) | 3.1 (2.4 to 4.5) |
| 20 mg | 6 | 591 | 53 | 11 | 5.1 (3.5 to 7.4) | 2.4 (2.1 to 2.8) |
| 40 mg | 5 | 519 | 63 | 17 | 3.9 (2.9 to 5.3) | 2.2 (1.9 to 2.6) |
Sensitivity analysis
Sensitivity analyses were carried out to investigate the effect of various study characteristics on the primary efficacy outcome (please see Summary of results B).
Pain model: dental versus other surgery
Efficacy of pain relief in dental versus other surgical pain models were analysed. There were insufficient data to analyse by individual dose.
Three studies with 504 participants used parecoxib (20 to 50 mg) in dental pain (Daniels 2001; Mehlisch 2003; Mehlisch 2004). The relative benefit for at least 50% pain relief over 6 hours was 18.5 (7.8 to 43.9) and the NNT was 1.7 (1.6 to 1.9). Four studies (Barton 2002; Bikhazi 2004; Malan 2005; Rasmussen 2002) with 486 participants used parecoxib (20 to 40 mg) in other surgical pain, with a relative benefit of 2.6 (1.9 to 3.5) and an NNT of 3.0 (2.4 to 4.0).
The efficacy of parecoxib (20 to 50 mg) tended to be better in dental trials than for other surgical procedures (z = 4.86, P < 0.00006).
Study quality
All included studies had a quality score of at least three out of five and therefore sensitivity analysis was not carried out.
Route of administration
Efficacy of intravenous versus intramuscular administration was analysed for 20 mg and 40 mg doses.
Five studies with 414 participants gave parecoxib 20 mg intravenously (Barton 2002; Bikhazi 2004; Daniels 2001; Mehlisch 2003; Rasmussen 2002). The relative benefit for at least 50% pain relief over 6 hours was 2.9 (2.6 to 5.8) and the NNT was 2.7 (2.2 to 3.5). Two studies with 177 participants gave parecoxib 20 mg intramuscularly (Daniels 2001; Mehlisch 2004), with a RR of 15 (4.8 to 46) and an NNT of 1.8 (1.5 to 2.3).
Four studies with 311 participants gave parecoxib 40 mg intravenously (Barton 2002; Bikhazi 2004; Daniels 2001; Rasmussen 2002). The relative benefit for at least 50% pain relief over 6 hours was 3.5 (2.4 to 5.2) and the NNT was 2.4 (1.9 to 3.1). Two studies with 208 participants gave parecoxib 40 mg intramuscularly (Daniels 2001; Malan 2005), with a RR of 4.6 (2.7 to 7.7) and an NNT of 2.0 (1.6 to 2.5).
NNTs were lower (better) for the intramuscular than the intravenous route, but the 95% CIs were overlapping, indicating no statistically significant differences between these routes of administration in these studies.
| Summary of results B: sensitivity analyses of the primary outcome | |||||
| Pain model | Studies | Participants | Parecoxib (%) | Placebo (%) | NNT (95 % CI) |
| Dental (20 to 50 mg) | 3 | 504 | 61 | 3 | 1.7 (1.6 to 1.9) |
| Other surgery (20 to 40 mg) | 4 | 486 | 54 | 21 | 3.0 (2.4 to 4.0) |
| Route of administration | Studies | Participants | Parecoxib (%) | Placebo (%) | NNT (95% CI) |
| Intramuscular route (20 mg) | 2 | 177 | 58 | 4 | 1.8 (1.5 to 2.3) |
| Intravenous route (20 mg) | 5 | 414 | 50 | 13 | 2.7 (2.2 to 3.5) |
| Intramuscular route (40 mg) | 2 | 208 | 67 | 16 | 2.0 (1.6 to 2.5) |
| Intravenous route (40 mg) | 4 | 311 | 60 | 18 | 2.4 (1.9 to 3.1) |
Use of rescue medication
Number of participants using rescue medication in 24 hours
Five of the seven studies reported the numbers of participants using rescue medication in 24 hours (Barton 2002; Daniels 2001; Mehlisch 2003; Mehlisch 2004; Rasmussen 2002) (please see Table 1). The other two studies reported the numbers of participants using rescue medication at 6 hours (Bikhazi 2004) and 12 hours (Malan 2005) and are not included in the analysis (please see Summary of results C).
For parecoxib 10 mg there was no significant difference in the proportion of participants using rescue medication over 24 hours with parecoxib (91%) and placebo (92%)
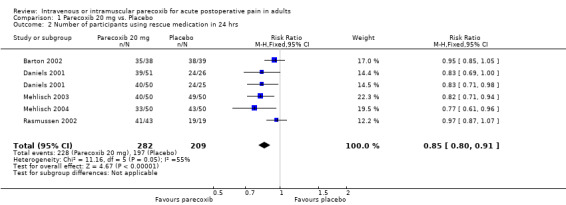
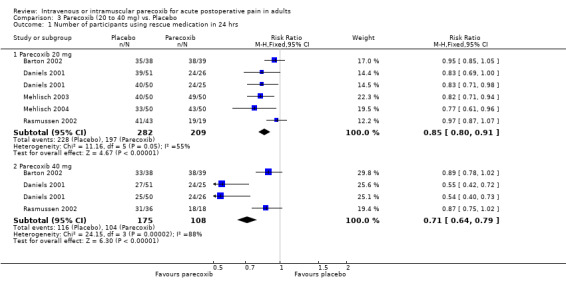
For parecoxib 20 mg, the proportion of participants using rescue medication over 24 hours was 81% for participants treated with parecoxib compared with 94% for those given placebo. This gives an NNTp of 7.5 (5.3 to 12.8)
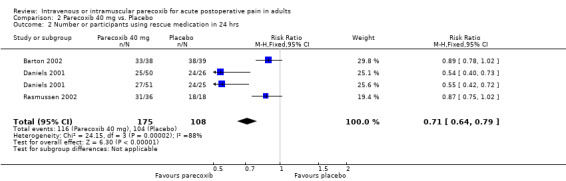
For parecoxib 40 mg, the proportion of participants using rescue medication over 24 hours was 66% for participants treated with parecoxib compared with 96% for those given placebo. This gives an NNTp of 3.3 (2.6 to 4.5).
Significantly fewer participants used rescue medication with parecoxib 40 mg than parecoxib 20 mg (z = 3.38, P < 0.0007) (please see Figure 3).
3.
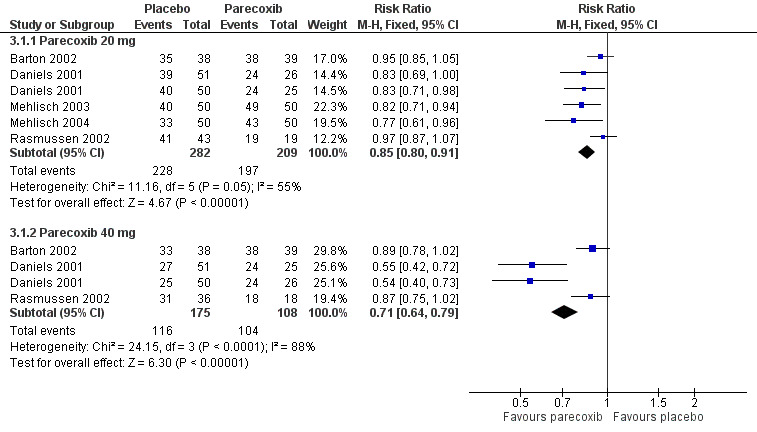
Forest plot of comparison: 4 Parecoxib (20 to 40 mg) vs. Placebo, outcome: 4.1 Number of participants using rescue medication in 24 h.
| Summary of results C: number using rescue medication over 24 hours | |||||
| Dose | Studies | Participants | Parecoxib (%) | Placebo (%) | NNTp |
| 10 mg | 2 | 200 | 91 | 92 | not calculated |
| 20 mg | 5 | 491 | 81 | 94 | 7.5 (5.3 to 12.8) |
| 40 mg | 3 | 283 | 66 | 96 | 3.3 (2.6 to 4.5) |
Time to use of rescue medication
All studies reported the median time to use of rescue medication which varied between 2.15 and 21.7 hours for active treatment (doses 10 to 100 mg) and 1.0 and 2.8 hours for placebo (please see Table 1 and Summary of results D).
For parecoxib 10 mg (Mehlisch 2003; Mehlisch 2004), the weighted mean of the median time to use of rescue medication was 3.1 hours for parecoxib compared with 1.0 hours for placebo.
For parecoxib 20 mg (Barton 2002; Bikhazi 2004; Daniels 2001; Mehlisch 2003, Mehlisch 2004Rasmussen 2002), the weighted mean of the median time to use of rescue medication was 6.9 hours for parecoxib compared with 1.6 hours for placebo.
For parecoxib 40 mg (Barton 2002; Daniels 2001; Malan 2005; Rasmussen 2002), the weighted mean of the median time to use of rescue medication was 10.6 hours for parecoxib compared with 2.0 hours for placebo.
| Summary of results D: weighted mean of median time to use of rescue medication | ||||
| Dose | Studies | Participants | Parecoxib (h) | Placebo (h) |
| 10 mg | 2 | 200 | 3.1 | 1.0 |
| 20 mg | 6 | 591 | 6.9 | 1.6 |
| 40 mg | 5 | 519 | 10.6 | 2.0 |
Adverse events
(Please see Table 2; Figure 4; Figure 5; and Summary of results E).
4.
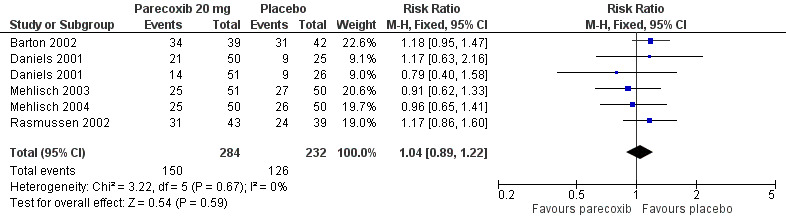
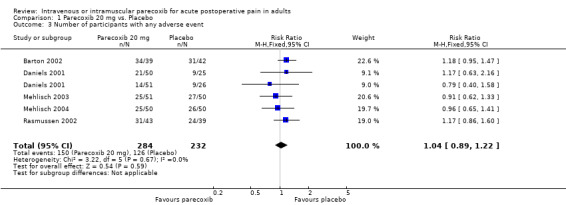
Forest plot of comparison: 2 Parecoxib 20 mg vs. Placebo, outcome: 2.3 Number or participants with any adverse event.
5.
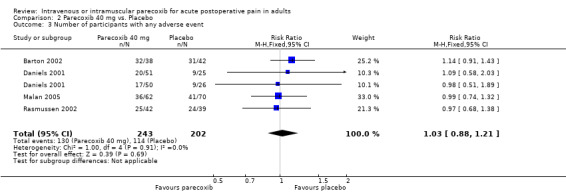
Forest plot of comparison: 3 Parecoxib 40 mg vs. Placebo, outcome: 3.3 Number of participants with any adverse event.
All seven included studies reported numbers of participants with any adverse event. The adverse event data was collected during the 24 hour treatment period and up to the post‐treatment visit at five to nine days for two studies (Mehlisch 2003; Mehlisch 2004), during the 24 hour treatment period only for three studies (Barton 2002; Daniels 2001; Rasmussen 2002) and during the 12 hour treatment period for one study (Malan 2005). One study reported numbers of participants with any adverse event but did not provide single dose data (Bikhazi 2004).
There was no significant difference between parecoxib (all doses) and placebo, with adverse events occurring at similar rates of around 54% in both groups, so NNHs were not calculated. Adverse events were generally described as mild to moderate in severity. No dose response was demonstrated.
One study reported two serious adverse events (Barton 2002). However, these occurred in participants in a treatment arm not relevant to the review (intravenous morphine).
| Summary of results E: participants with one or more adverse events | |||||
| Dose | Studies | Participants | Parecoxib (%) | Placebo (%) | NNH (95% CI) any AE |
| All | 6 | 1337 | 53 | 55 | not calculated |
| 20 mg | 5 | 516 | 53 | 54 | not calculated |
| 40 mg | 4 | 445 | 53 | 56 | not calculated |
Withdrawals
Participants who took rescue medication were classified as withdrawals due to lack of efficacy, and are reported under 'use of rescue medication' above.
Data on other withdrawals were generally poorly reported, probably because these were single dose studies where withdrawals for reasons other than lack of efficacy are uncommon. Some studies reported participants who had invalid data due to inadequate baseline pain, protocol violation, or use of rescue medication within the first hour, as withdrawals or exclusions. Whether these should be included in the intention‐to‐treat population is arguable. Attrition due to invalid data is unlikely to affect results.
A total of 13 participants were reported as withdrawing due to adverse events: five after parecoxib 20 mg, four after parecoxib 40 mg and two after placebo, mostly due to headache (Barton 2002); one due to an unspecified adverse event after parecoxib 40 mg (Malan 2005); and one after parecoxib 40 mg due to anxiety (Rasmussen 2002) (please see Table 2).
Discussion
This review included a total of 1446 participants. In total 320 participants were treated with a single dose of parecoxib 20 mg, and 278 with parecoxib 40 mg. This is around twice the number compared to a previous systematic review (Barden 2003), giving a more robust (Moore 1998), but almost identical result at these doses. In addition, the new studies provide limited amounts of data for lower doses.
The primary measure of efficacy was the proportion of participants achieving at least 50% pain relief over 6 hours. All doses of parecoxib showed a significant difference from placebo with NNT values of 10.3 (6.0 to 37.1) for 1 mg, 7.2 (4.6 to 16.7) for 2 mg, 3.7 (2.7 to 5.6) for 5 mg, 3.1 (2.4 to 4.5) for 10 mg, 2.4 (2.1 to 2.8) for 20 mg, and 2.2 (1.9 to 2.6) for 40 mg. The numbers of participants in each treatment group for 1 mg to 10 mg were low and the CIs are wide so data from these groups should be interpreted cautiously. There was no significant difference in the relative benefit or NNT for 50% pain relief over 6 hours between 20 mg and 40 mg, although a dose response trend was observed.
Because the same methods of analyses have been used, it is possible to compare the NNT for at least 50% pain relief over 6 hours for a single dose of parenteral parecoxib with that of a single dose of other analgesics. There are no data available for intravenous administration of other NSAIDs so comparisons with oral or intramuscular analgesics are made.
Analgesics with comparable efficacy to parecoxib (20 mg to 50 mg) include rofecoxib 50 mg (NNT 2.2 (1.9 to 2.4)) (Barden 2005), celecoxib 400 mg (NNT 2.5 (2.2 to 2.9) (Derry 2008), diclofenac 100 mg (NNT 2.3 (2.0 to 2.5)) (Derry 2009b), and naproxen 500 mg (NNT 2.7 (2.3 to 3.3)) (Derry 2009a).
Analgesics with lower efficacy include morphine 10 mg (IM) (NNT 2.9 (2.6 to 3.6)), ketorolac 30 mg (IM) (NNT 3.4 (2.5 to 4.9)) (Smith 2000), aspirin 600/650 mg (NNT 4.4 (4.0 to 4.9) (Oldman 1999) and paracetamol 600/650 mg (NNT 4.6 (3.9 to 5.5)) (Toms 2008).
Analgesics with superior efficacy include etoricoxib 180/240 mg (NNT 1.5 (1.3 to 1.7) and etoricoxib 120 mg (NNT 1.9 (1.7 to 2.1)) (Clarke 2009).
A current listing of reviews of analgesics in the single dose postoperative pain model can be found at www.medicine.ox.ac.uk/bandolier.
Significantly fewer participants required rescue medication with parecoxib than with placebo, and although no significant difference between 20 and 40 mg parecoxib was demonstrated for the primary outcome, significantly fewer participants required rescue medication with parecoxib 40 mg than with parecoxib 20 mg. Seven to eight individuals would need to be treated with parecoxib 20 mg, and three with parecoxib 40 mg to prevent one form needing rescue medication over 24 hours, who would have done so if treated with placebo. The median time to use of rescue medication varied greatly between studies, particularly for the active treatment arms, but was generally longer for parecoxib than placebo, and for parecoxib 40 mg than parecoxib 20 mg. The weighted mean of the median time to use of rescue medication was 1.5 hours for placebo, 3.1 hours for parecoxib 10 mg, 6.9 hours for parecoxib 20 mg and 10.6 hours for parecoxib 40 mg.
Longer duration of action is desirable in an analgesic, particularly in a postoperative setting where the patient may experience postoperative nausea, or be dependent on a third party to respond to a request for rescue medication, or both. Duration of pain relief and requirement of rescue medication information have only recently been recognised as important outcomes (Moore 2005), and a fuller evaluation of the importance of these outcomes will depend on more data being collected from other, ongoing, systematic reviews. For the studies included in this review, differences in use of rescue medication allowed discrimination between doses that was not seen with the primary outcome.
Assessment of adverse events is limited in single dose studies as the size and duration of the trials permits only the simplest analysis, as has been emphasised previously (Edwards 1999). There were insufficient data in these studies to compare individual adverse events. There was no significant difference between parecoxib and placebo for the numbers of participants experiencing any adverse event in the hours immediately following a single dose of the study medication, with rates of around 54% in both groups. Combining results was potentially hampered by the different periods over which the data was collected. Most adverse events were reported as mild to moderate in intensity, and were most likely to be related to the anaesthetic or surgical procedure (e.g. nausea, vomiting and somnolence). It is important to recognise that adverse event analysis after single dose administration will not reflect possible adverse events occurring with use of drugs for longer periods of time. In addition, the relatively small number of participants, even when all the trials were combined, is insufficient to detect rare but serious adverse events. An increase in serious adverse events (wound infection, cerebro‐ and cardiovascular events, and renal dysfunction) has been reported in patients undergoing cardiac surgery who received parecoxib for three days, followed by valdecoxib for 10 to 14 days (Nussmeier 2005; Ott 2003). No such events were seen in these single dose studies.
Importantly, none of the studies reported any clinically significant changes in clinical laboratory findings, physical examination or vital signs in any of the patients during the study period.
The sensitivity analysis did not demonstrate any significant effect of route of administration of parecoxib on relative benefit or NNT. A significantly increased efficacy of parecoxib, using pooled doses, in dental studies compared to studies involving other postsurgical pain was demonstrated. Differences between dental and other postsurgical pain have been noted before (Barden 2004), where consistently lower placebo responses in the dental pain model do not effect the NNT as a measurement of efficacy. In all three dental studies in this analysis, the placebo response rate was very low (2% to 4%), and this result may be due to random chance in the relatively small number (151) of placebo‐treated participants. The over‐representation of women, particularly in the non‐dental studies may have influenced the results, but this is thought to be unlikely as clinically meaningful differences between men and women in response to NSAIDs have not been demonstrated previously (Barden 2002).
Authors' conclusions
Implications for practice.
Parecoxib is an effective analgesic in postoperative pain with a low incidence of adverse events when given as a single dose. At a dose of 20 mg to 40 mg it provided effective analgesia for 50 to 60% of patients with moderate to severe postoperative pain following various types of surgery. For every two participants treated with parecoxib 20 mg or 40 mg, one would experience at least 50% pain relief who would not have done so with placebo. Associated adverse events were generally mild to moderate in intensity.
Implications for research.
Additional data is required to confirm the results of this review and provide a more robust estimate of efficacy, particularly for the lower doses. Further data for the lower doses will help determine the minimum effective dose and establish a dose response for parecoxib. In clinical practice the aim should always be to use the lowest dose possible to achieve the desired benefit with minimal risk of adverse events. More data for the intravenous and intramuscular routes will help to decide which route of administration is most effective for parecoxib in the clinical setting, while more data from different types of surgery may help determine whether there are clinically important differences between them.
What's new
| Date | Event | Description |
|---|---|---|
| 29 May 2019 | Amended | Contact details updated. |
| 2 July 2015 | Review declared as stable | This review will no longer be updated. The authors and editors are confident that further research will not change the conclusions. |
History
Protocol first published: Issue 2, 2004 Review first published: Issue 2, 2009
| Date | Event | Description |
|---|---|---|
| 15 September 2011 | Review declared as stable | The authors scanned the literature in August 2011 and are confident that there will be no change to the conclusions of this review and therefore a need to update it until at least 2015. |
| 11 May 2011 | Amended | Contact details updated. |
| 24 September 2010 | Amended | Contact details updated. |
| 9 November 2009 | Amended | Contact details updated. |
| 13 May 2009 | Amended | Contact details updated. |
| 20 June 2008 | New citation required and minor changes | New authors have taken over this title and brought the protocol up to date |
Notes
The original published protocol was withdrawn from publication on the 8th February 2007 due to contact loss of the author who intended to write the full review ‐ Jose Gomez‐Leon. The protocol had minor revisions in June 2008 to update it and bring it in line with a series of reviews of single dose analgesics in acute postoperative pain.
Acknowledgements
Jose Gomez‐Leon wrote the original protocol.
Appendices
Appendix 1. MEDLINE search strategy (via Ovid)
1. parecoxib [single term MESH]
2. parecoxib OR dynastat
3. OR/1‐2
4. PAIN, POSTOPERATIVE [single term MeSH]
5. ((postoperative adj4 pain$) or (post‐operative adj4 pain$) or post‐operative‐pain$ or (post$ NEAR pain$) or (postoperative adj4 analgesi$) or (post‐operative adj4 analgesi$) or ("post‐operative analgesi$")) [in title, abstract or keywords]
6. ((post‐surgical adj4 pain$) or (“post surgical” adj4 pain$) or (post‐surgery adj4 pain$))[in title, abstract or keywords]
7. ((“pain‐relief after surg$”) or (“pain following surg$”) or (“pain control after”)) [in title, abstract or keywords]
8. ((“post surg$” or post‐surg$) AND (pain$ or discomfort)) [in title, abstract or keywords]
9. ((pain$ adj4 “after surg$”) or (pain$ adj4 “after operat$”) or (pain$ adj4 “follow$ operat$”) or (pain$ adj4 “follow$ surg$”))[in title, abstract or keywords]
10. ((analgesi$ adj4 “after surg$”) or (analgesi$ adj4 “after operat$”) or (analgesi$ adj4 “follow$ operat$”) or (analgesi$ adj4 “follow$ surg$”))
11. OR/4‐10
12. randomized controlled trial.pt.
13. controlled clinical trial.pt.
14. randomized.ab.
15. placebo.ab.
16. drug therapy.fs.
17. randomly.ab.
18. trial.ab.
19. groups.ab.
20. OR/12‐19
21. humans.sh.
22. 20 AND 21
23. 3 AND 11 AND 22
Appendix 2. EMBASE search strategy (via Ovid)
1. parecoxib [single term MESH]
2. parecoxib OR dynastat
3. OR/1‐2
4. PAIN, POSTOPERATIVE [single term MeSH]
5. ((postoperative adj4 pain$) or (post‐operative adj4 pain$) or post‐operative‐pain$ or (post$ NEAR pain$) or (postoperative adj4 analgesi$) or (post‐operative adj4 analgesi$) or ("post‐operative analgesi$"))
6. ((post‐surgical adj4 pain$) or (“post surgical” adj4 pain$) or (post‐surgery adj4 pain$))
7. ((“pain‐relief after surg$”) or (“pain following surg$”) or (“pain control after”))
8. ((“post surg$” or post‐surg$) AND (pain$ or discomfort))
9. ((pain$ adj4 “after surg$”) or (pain$ adj4 “after operat$”) or (pain$ adj4 “follow$ operat$”) or (pain$ adj4 “follow$ surg$”))
10. ((analgesi$ adj4 “after surg$”) or (analgesi$ adj4 “after operat$”) or (analgesi$ adj4 “follow$ operat$”) or (analgesi$ adj4 “follow$ surg$”))
11. OR/4‐10
12. clinical trials.sh
13. controlled clinical trials.sh
14. randomized controlled trial.sh
15. double‐blind procedure.sh
16. (clin$ adj25 trial$)
17. ((doubl$ or trebl$ or tripl$) adj25 (blind$ or mask$))
18. placebo$
19. random$
20. OR/12‐19
21. 3 AND 11 AND 20
Appendix 3. Cochrane CENTRAL search strategy
1. parecoxib [single term MESH]
2. parecoxib OR dynastat [ti, ab, kw]
3. OR/1‐2
4. PAIN, POSTOPERATIVE [single term MeSH]
5. ((postoperative adj4 pain$) or (post‐operative adj4 pain$) or post‐operative‐pain$ or (post$ NEAR pain$) or (postoperative adj4 analgesi$) or (post‐operative adj4 analgesi$) or ("post‐operative analgesi$")) [ti, ab, kw]
6. ((post‐surgical adj4 pain$) or (“post surgical” adj4 pain$) or (post‐surgery adj4 pain$)) [ti, ab, kw]
7. ((“pain‐relief after surg$”) or (“pain following surg$”) or (“pain control after”)) [ti, ab, kw]
8. ((“post surg$” or post‐surg$) AND (pain$ or discomfort)) [ti, ab, kw]
9. ((pain$ adj4 “after surg$”) or (pain$ adj4 “after operat$”) or (pain$ adj4 “follow$ operat$”) or (pain$ adj4 “follow$ surg$”)) [ti, ab, kw]
10. ((analgesi$ adj4 “after surg$”) or (analgesi$ adj4 “after operat$”) or (analgesi$ adj4 “follow$ operat$”) or (analgesi$ adj4 “follow$ surg$”)) [ti, ab, kw]
11. OR/4‐10
12. clinical trials [exp MESH term]
13. controlled clinical trials [exp MESH term]
14. randomized controlled trial [exp MESH term]
15. double‐blind procedure [single term MESH]
16. (clin$ adj25 trial$) [ti, ab, kw]
17. ((doubl$ or trebl$ or tripl$) adj25 (blind$ or mask$)) [ti, ab, kw]
18. placebo$ [ti, ab, kw]
19. random$ [ti, ab, kw]
20. OR/12‐19
21. 3 AND 11 AND 20
Appendix 4. Glossary
Categorical rating scale
The commonest is the five category scale (none, slight, moderate, good or lots, and complete). For analysis numbers are given to the verbal categories (for pain intensity, none=0, mild=1, moderate=2 and severe=3, and for relief none=0, slight=1, moderate=2, good or lots=3 and complete=4). Data from different subjects is then combined to produce means (rarely medians) and measures of dispersion (usually standard errors of means). The validity of converting categories into numerical scores was checked by comparison with concurrent visual analogue scale measurements. Good correlation was found, especially between pain relief scales using cross‐modality matching techniques. Results are usually reported as continuous data, mean or median pain relief or intensity. Few studies present results as discrete data, giving the number of participants who report a certain level of pain intensity or relief at any given assessment point. The main advantages of the categorical scales are that they are quick and simple. The small number of descriptors may force the scorer to choose a particular category when none describes the pain satisfactorily.
VAS
Visual analogue scale: For pain intensity, lines with left end labelled "no pain" and right end labelled "worst pain imaginable", and for pain relief, lines with left end labelled "no relief of pain" and right end labelled "complete relief of pain", seem to overcome this limitation. Patients mark the line at the point which corresponds to their pain. The scores are obtained by measuring the distance between the no relief end and the patient's mark, usually in millimetres. The main advantages of VAS are that they are simple and quick to score, avoid imprecise descriptive terms and provide many points from which to choose. More concentration and coordination are needed, which can be difficult post‐operatively or with neurological disorders.
TOTPAR
Total pain relief (TOTPAR) is calculated as the sum of pain relief scores over a period of time. If a patient had complete pain relief immediately after taking an analgesic, and maintained that level of pain relief for 6 hours, they would have a 6‐hour TOTPAR of the maximum of 24. Differences between pain relief values at the start and end of a measurement period are dealt with by the composite trapezoidal rule. This is a simple method that approximately calculates the definite integral of the area under the pain relief curve by calculating the sum of the areas of several trapezoids that together closely approximate to the area under the curve.
SPID
Summed pain intensity difference (SPID) is calculated as the sum of the differences between the pain scores over a period of time. Differences between pain intensity values at the start and end of a measurement period are dealt with by the trapezoidal rule.
VAS TOTPAR and VAS SPID are visual analogue versions of TOTPAR and SPID.
See “Measuring pain” in Bandolier’s Little Book of Pain, Oxford University Press, Oxford. 2003; pp 7‐13 (Moore 2003).
Data and analyses
Comparison 1. Parecoxib 20 mg vs. Placebo.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Participants with at least 50% pain relief | 6 | 591 | Risk Ratio (M‐H, Fixed, 95% CI) | 5.11 [3.51, 7.43] |
| 1.1 Intramuscular route | 2 | 177 | Risk Ratio (M‐H, Fixed, 95% CI) | 14.82 [4.77, 46.06] |
| 1.2 Intravenous route | 5 | 414 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.88 [2.61, 5.75] |
| 2 Number of participants using rescue medication in 24 hrs | 5 | 491 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.85 [0.80, 0.91] |
| 3 Number of participants with any adverse event | 5 | 516 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.04 [0.89, 1.22] |
1.1. Analysis.
Comparison 1 Parecoxib 20 mg vs. Placebo, Outcome 1 Participants with at least 50% pain relief.
1.2. Analysis.
Comparison 1 Parecoxib 20 mg vs. Placebo, Outcome 2 Number of participants using rescue medication in 24 hrs.
1.3. Analysis.
Comparison 1 Parecoxib 20 mg vs. Placebo, Outcome 3 Number of participants with any adverse event.
Comparison 2. Parecoxib 40 mg vs. Placebo.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Participants with at least 50% pain relief | 5 | 519 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.89 [2.85, 5.31] |
| 1.1 Intramuscular route | 2 | 208 | Risk Ratio (M‐H, Fixed, 95% CI) | 4.56 [2.71, 7.69] |
| 1.2 Intravenous route | 4 | 311 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.51 [2.39, 5.16] |
| 2 Number or participants using rescue medication in 24 hrs | 3 | 283 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.71 [0.64, 0.79] |
| 3 Number of participants with any adverse event | 4 | 445 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.03 [0.88, 1.21] |
2.1. Analysis.
Comparison 2 Parecoxib 40 mg vs. Placebo, Outcome 1 Participants with at least 50% pain relief.
2.2. Analysis.
Comparison 2 Parecoxib 40 mg vs. Placebo, Outcome 2 Number or participants using rescue medication in 24 hrs.
2.3. Analysis.
Comparison 2 Parecoxib 40 mg vs. Placebo, Outcome 3 Number of participants with any adverse event.
Comparison 3. Parecoxib (20 to 40 mg) vs. Placebo.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Number of participants using rescue medication in 24 hrs | 5 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 1.1 Parecoxib 20 mg | 5 | 491 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.85 [0.80, 0.91] |
| 1.2 Parecoxib 40 mg | 3 | 283 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.71 [0.64, 0.79] |
3.1. Analysis.
Comparison 3 Parecoxib (20 to 40 mg) vs. Placebo, Outcome 1 Number of participants using rescue medication in 24 hrs.
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Barton 2002.
| Methods | RCT, DB single intravenous dose, 5 parallel groups Medication administered when baseline pain reached a moderate to severe intensity Pain assessment at 15, 30, 45, 60, 90, 120 mins then hourly up to 8 h, and at 12, 16 and 24 h |
|
| Participants | Elective gynaecological surgery Mean age 42 years N = 202 All F |
|
| Interventions | Parecoxib 20 mg IV, n = 39 Parecoxib 40 mg IV, n = 38 Ketorolac 30 mg IV, n = 41 Morphine 4 mg IV, n = 42 Placebo, n = 42 |
|
| Outcomes | PI: std 4 point scale and VAS PR: std 5 point scale PGE: non std scale Time to use of rescue medication Number of patients using rescue medication |
|
| Notes | Oxford Quality Score: R2, DB1, W1 Patients asked to refrain from rescue medication for 1 h |
|
Bikhazi 2004.
| Methods | RCT, DB single and multiple intravenous dose, 5 parallel groups Medication administered when baseline pain reached a moderate to severe intensity Pain assessment at 15, 30, 45, 60, 90, 120 mins then hourly up to 8 h, and at 10, 12, 16 and 24 h |
|
| Participants | Surgical hysterectomy Mean age 43 years N = 208 All F |
|
| Interventions | Parecoxib 20 mg IV, n = 40 Parecoxib 40 mg IV, n = 41 Ketorolac 30 mg IV, n = 42 Morphine 4 mg IV, n = 40 Placebo, n = 45 |
|
| Outcomes | PI: std 4 point scale and VAS PR: std 5 point scale PGE: non std scale Time to use of rescue medication Number of patients using rescue medication |
|
| Notes | Oxford Quality Score: R2, DB1, W1 Patients asked to refrain from rescue medication for 1 h |
|
Daniels 2001.
| Methods | RCT, DB single intravenous or intramuscular dose, 6 parallel groups Medication administered when baseline pain reached a moderate to severe intensity Pain assessment at 15, 30, 45, 60, 90, 120 mins then hourly up to 8 h, and at 10, 12, 16 and 24 h |
|
| Participants | Impacted third molar extraction Mean age 22 years N = 304 M = 117, F = 187 |
|
| Interventions | Parecoxib 20 mg IM, n = 51 Parecoxib 20 mg IV, n = 50 Parecoxib 40 mg IM, n = 50 Parecoxib 40 mg IV, n = 51 Ketorolac 60 mg IM, n = 51 Placebo, n = 51 |
|
| Outcomes | PI: std 4 point scale and VAS PR: std 5 point scale PGE: non std scale Time to use of rescue medication Number of patients using rescue medication |
|
| Notes | Oxford Quality Score: R2, DB2, W1 Patients asked to refrain from rescue medication for 1 h |
|
Malan 2005.
| Methods | RCT, DB single intramuscular dose, 4 parallel groups Medication administered when baseline pain reached a moderate to severe intensity Pain assessment at 0, 30, 60 mins then hourly to 8 h, and at 10 and 12 h |
|
| Participants | Gynaecologic laparotomy surgery Mean age 44 years N = 264 All F |
|
| Interventions | Parecoxib 40 mg IM, n = 62 Morphine 6 mg IM, n = 70 Morphine 12 mg IM, n = 62 Placebo, n = 70 |
|
| Outcomes | PI: std 4 point scale and VAS PR: std 5 point scale PGE: non std scale Time to use of rescue medication Number of patients using rescue medication |
|
| Notes | Oxford Quality Score: R2, DB1, W1 | |
Mehlisch 2003.
| Methods | RCT, DB, DD single intravenous dose, 9 parallel groups Medication administered when baseline pain reached a moderate to severe intensity Pain assessment at 15, 30, 45, 60, 90, 120 mins then hourly to 12 h, and at 16 and 24 h |
|
| Participants | Impacted third molar extraction Mean age 23 years N = 457 M = 141, F = 316 |
|
| Interventions | Parecoxib 1 mg IV, n = 51 Parecoxib 2 mg IV, n = 51 Parecoxib 5 mg IV, n = 51 Parecoxib 10 mg IV, n = 51 Parecoxib 20 mg IV, n = 51 Parecoxib 50 mg IV, n = 51 Parecoxib 100 mg IV, n = 51 Ketorolac 30 mg IV, n = 50 Placebo, n = 50 |
|
| Outcomes | PI: std 4 point scale and VAS PR: std 5 point scale PGE: non std scale Time to use of rescue medication Number of patients using rescue medication |
|
| Notes | Oxford Quality Score: R1, DB2, W1 | |
Mehlisch 2004.
| Methods | RCT, DB single intramuscular dose Medication administered when baseline pain reached a moderate to severe intensity Pain assessment at 5, 10, 15, 20, 30, 45, 60, 90, 120 mins then hourly to 12 h, and at 16 and 24 h |
|
| Participants | Impacted third molar extraction Mean age 23 years N = 353 M = 157, F = 196 |
|
| Interventions | Parecoxib 1 mg IM, n = 51 Parecoxib 2 mg IM, n = 50 Parecoxib 5 mg IM, n = 51 Parecoxib 10 mg IM, n = 50 Parecoxib 20 mg IM, n = 50 Ketorolac 30 mg IM, n = 51 Placebo, n = 50 |
|
| Outcomes | PI: std 4 point scale and VAS PR: std 5 point scale PGE: non std scale Time to use of rescue medication Number of patients using rescue medication |
|
| Notes | Oxford Quality Score: R2, DB2, W1 Patients asked to refrain from rescue medication for 1 h |
|
Rasmussen 2002.
| Methods | RCT, DB single intravenous dose, 5 parallel groups Medication administered when baseline pain reached a moderate to severe intensity Pain assessment at 0, 15, 30, 45, 60, 90, 120 mins then hourly up to 8 h, and at 10, 12, 16 and 24 h |
|
| Participants | Unilateral knee replacement surgery Mean age 65 years N = 208 M = 73, F = 135 |
|
| Interventions | Parecoxib 20 mg IV, n = 43 Parecoxib 40 mg IV, n = 42 Ketorolac 30 mg IV, n = 42 Morphine 4 mg IV, n = 42 Placebo, n = 39 |
|
| Outcomes | PI: std 4 point scale and VAS PR: std 5 point scale PGE: non std scale Time to use of rescue medication |
|
| Notes | Oxford Quality Score: R2, DB1, W1 | |
DB ‐ double blind; F ‐ female; IM ‐ intramuscular; IV ‐ intravenous; PGE ‐ patient global evaluation; M ‐ male; N ‐ total number of participants in study; n ‐ number of participants in treatment arm; PI ‐ pain intensity; PR ‐ pain relief; R ‐ randomised; RCT ‐ randomised controlled trial; std ‐ standard; VAS ‐ visual analogue scale; W ‐ withdrawal
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Hubbard 2003 | Intervention was administered immediately after surgery, before anaesthetic wore off. Therefore inadequate baseline pain. No single dose data. |
| Jirarattanaphochai 2008 | Intervention administered preoperatively. Therefore inadequate baseline pain. No single dose data. |
| Nussimeier 2006 | No single dose data. |
| Puolakka 2006 | Intervention was administered immediately after surgery, before anaesthetic wore off. Therefore inadequate baseline pain. Participants had access to PCA. |
| Snabes 2007 | No single dose data. Includes participants with pain assessed as < 45 on a 100‐mm visual analog scale. Therefore includes participants with inadequate baseline pain. |
| Tang 2002 | No single dose data. Participants had access to PCA. |
| Viscusi 2008 | Loading dose given without assessment of baseline pain. |
Characteristics of studies awaiting assessment [ordered by study ID]
Apfelbaum 2008.
| Methods | |
| Participants | |
| Interventions | |
| Outcomes | |
| Notes | Paper requested. |
Differences between protocol and review
The title has been changed to indicate that the review is limited to adults and to include the intramuscular route of administration.
Contributions of authors
RL and SD were involved with searching, data extraction, quality scoring, analysis and writing. RAM was involved in analysis and writing. HJM acted as arbitrator and was involved in writing. All review authors contributed to revising and re‐publishing the protocol.
Sources of support
Internal sources
Pain Research Funds, UK.
External sources
NHS Cochrane Collaboration Programme Grant Scheme, UK.
European Union Biomed 2 Grant no. BMH4 CT95 0172, UK.
Declarations of interest
RAM, HJM and SD have received research support from charities, government and industry sources at various times, but no such support was received for this work. RAM and HJM have consulted for various pharmaceutical companies. RAM, and HJM have received lecture fees from pharmaceutical companies related to analgesics and other healthcare interventions.
Stable (no update expected for reasons given in 'What's new')
References
References to studies included in this review
Barton 2002 {published data only}
- Barton SF, Langeland FF, Snabes MC, LeComte D, Kuss ME, Dhadda SS, et al. Efficacy and safety of intravenous parecoxib sodium in relieving acute postoperative pain following gynecologic laparotomy surgery. Anesthesiology 2002;97(2):306‐14. [DOI] [PubMed] [Google Scholar]
Bikhazi 2004 {published data only}
- Bikhazi GB, Snabes MC, Bajwa ZH, Davis DJ, LeComte D, Traylor L, et al. A clinical trial demonstrates the analgesic activity of intravenous parecoxib sodium compared with ketorolac or morphine after gynecologic surgery with laparotomy. American Journal of Obstetrics and Gynecology 2004;191(4):1183‐91. [DOI: 10.1016/j.ajog.2004.05.006] [DOI] [PubMed] [Google Scholar]
Daniels 2001 {published data only}
- Daniels SE, Grossman EH, Kuss ME, Talwalker S, Hubbard RC. A double‐blind, randomized comparison of intramuscularly and intravenously administered parecoxib sodium versus ketorolac and placebo in a post‐oral surgery pain model. Clinical Therapeutics 2001;23(7):1018‐31. [DOI: 10.1016/S0149-2918(01)80088-6] [DOI] [PubMed] [Google Scholar]
Malan 2005 {published data only}
- Malan TP Jr, Gordon S, Hubbard R, Snabes M. The cyclooxygenase‐2‐specific inhibitor parecoxib sodium is as effective as 12 mg of morphine administered intramuscularly for treating pain after gynecologic laparotomy surgery. Anesthesia and Analgesia 2005;100(2):454‐60. [DOI: 10.1213/01.ANE.0000143355.52418.CF] [DOI] [PubMed] [Google Scholar]
Mehlisch 2003 {published data only}
- Mehlisch DR, Desjardins PJ, Daniels S, Hubbard RC. Single doses of parecoxib sodium intravenously are as effective as ketorolac in reducing pain after oral surgery. Journal of Oral and Maxillofacial Surgery 2003;61(9):1030‐7. [DOI: 10.1016/S0278-2391(03)00315-X] [DOI] [PubMed] [Google Scholar]
Mehlisch 2004 {published data only}
- Mehlisch DR, Desjardins PJ, Daniels S, Hubbard RC. The analgesic efficacy of intramuscular parecoxib sodium in postoperative dental pain. The Journal of the American Dental Association 2004;135(11):1578‐90. [DOI] [PubMed] [Google Scholar]
Rasmussen 2002 {published data only}
- Rasmussen GL, Steckner K, Hogue C, Torri S, Hubbard RC. Intravenous parecoxib sodium for acute pain after orthopedic knee surgery. The American Journal of Orthopedics 2002;31(6):336‐43. [PubMed] [Google Scholar]
References to studies excluded from this review
Hubbard 2003 {published data only}
- Hubbard RC, Naumann TM, Traylor L, Dhadda S. Parecoxib sodium has opioid‐sparing effects in patients undergoing total knee arthroplasty under spinal anaesthesia. British Journal of Anaesthesia 2003;90(2):166‐72. [DOI] [PubMed] [Google Scholar]
Jirarattanaphochai 2008 {published data only}
- Jirarattanaphochai K, Thienthong S, Sriraj W, Jung S, Pulnitiporn A, Lertsinudom S, et al. Effect of parecoxib on postoperative pain after lumbar spine surgery: a bicenter, randomized, double‐blinded, placebo‐controlled trial. Spine 2008;33(2):132‐9. [DOI] [PubMed] [Google Scholar]
Nussimeier 2006 {published data only}
- Nussmeier NA, Whelton AA, Brown MT, Joshi GP, Langford RM, Singla NK, et al. Safety and efficacy of the cyclooxygenase‐2 inhibitors parecoxib and valdecoxib after noncardiac surgery. Anesthesiology 2006;104(3):518‐26. [DOI] [PubMed] [Google Scholar]
Puolakka 2006 {published data only}
- Puolakka PA, Puura AI, Pirhonen RA, Ranta AU, Autio V, et al. Lack of analgesic effect of parecoxib following laparoscopic cholecystectomy. Acta Anaesthesiologica Scandinavica 2006;50(8):1027‐32. [DOI] [PubMed] [Google Scholar]
Snabes 2007 {published data only}
- Snabes MC, Jakimiuk AJ, Kotarski J, Katz TK, Brown MT, Verburg KM. Parecoxib sodium administered over several days reduces pain after gynecologic surgery via laparotomy. Journal of Clinical Anesthesia 2007;19(6):448‐55. [DOI] [PubMed] [Google Scholar]
Tang 2002 {published data only}
- Tang J, Li S, White PF, Chen X, Wender RH, Quon R, et al. Effect of parecoxib, a novel intravenous cyclooxygenase type‐2 inhibitor, on the postoperative opioid requirement and quality of pain control. Anesthesiology 2002;96(6):1305‐9. [DOI] [PubMed] [Google Scholar]
Viscusi 2008 {published data only}
- Viscusi ER, Gimbel JS, Halder AM, Snabes M, Brown MT, Verburg KM. A multiple‐day regimen of parecoxib sodium 20 mg twice daily provides pain relief after total hip arthroplasty. Anesthesia and Analgesia 2008;107(2):652‐60. [DOI] [PubMed] [Google Scholar]
References to studies awaiting assessment
Apfelbaum 2008 {published data only}
- Apfelbaum JL, Desjardins PJ, Brown MT, Verburg KM. Multiple‐day efficacy of parecoxib sodium treatment in postoperative bunionectomy pain. The Clinical Journal of Pain 2008;24(9):784‐92. [DOI] [PubMed] [Google Scholar]
Additional references
Barden 2002
- Barden J, Edwards JE, Moore RA, McQuay HJ. Ibuprofen 400 mg is effective in women, and women are well represented in trials. BMC Anesthesiology 2002;2:6. [DOI] [PMC free article] [PubMed] [Google Scholar]
Barden 2004
- Barden J, Edwards JE, McQuay HJ, Moore RA. Pain and analgesic response after third molar extraction and other postsurgical pain. Pain 2004;107(1‐2):86‐90. [DOI] [PubMed] [Google Scholar]
Barden 2005
- Barden J, Edwards J, Moore RA, McQuay HJ. Single dose oral rofecoxib for postoperative pain. Cochrane Database of Systematic Reviews 2005, Issue 1. [DOI: 10.1002/14651858.CD004604.pub2] [DOI] [PubMed] [Google Scholar]
Barden 2008
- Barden J, Derry S, McQuay HJ, Moore RA. Single dose oral ketoprofen and dexketoprofen for acute postoperative pain in adults. Cochrane Database of Systematic Reviews 2008, Issue 4. [DOI: 10.1002/14651858.CD007355] [DOI] [PMC free article] [PubMed] [Google Scholar]
Clarke 2009
- Clarke R, Derry S, Moore RA, McQuay HJ. Single dose oral etoricoxib for acute postoperative pain in adults. Cochrane Database of Systematic Reviews 2009, Issue 2. [DOI: 10.1002/14651858.CD004309] [DOI] [PubMed] [Google Scholar]
Collins 1997
- Collins SL, Moore RA, McQuay HJ. The visual analogue pain intensity scale: what is moderate pain in millimetres?. Pain 1997;72:95‐7. [DOI] [PubMed] [Google Scholar]
Collins 1999
- Collins SL, Moore RA, McQuay HJ, Wiffen PJ, Edwards JE. Single dose oral ibuprofen and diclofenac for postoperative pain. Cochrane Database of Systematic Reviews 1999, Issue 1. [DOI: 10.1002/14651858.CD001548] [DOI] [PubMed] [Google Scholar]
Collins 2001
- Collins SL, Edwards J, Moore RA, Smith LA, McQuay HJ. Seeking a simple measure of analgesia for mega‐trials: is a single global assessment good enough?. Pain 2001;91:189‐94. [DOI] [PubMed] [Google Scholar]
Cook 1995
- Cook RJ, Sackett DL. The number needed to treat: a clinically useful measure of treatment effect. BMJ 1995;310:452‐4. [DOI] [PMC free article] [PubMed] [Google Scholar]
Cooper 1991
- Cooper SA. Single‐dose analgesic studies: the upside and downside of assay sensitivity. In: Max MB, Portenoy RK, Laska EM editor(s). The Design of Analgesic ClinicalTrials. Advances in Pain Research and Therapy. Vol. 18, New York: Raven Press, 1991:117‐24. [Google Scholar]
Derry 2008
- Derry S, Barden J, McQuay HJ, Moore RA. Single dose oral celecoxib for acute postoperative pain. Cochrane Database of Systematic Reviews 2008, Issue 3. [DOI: 10.1002/14651858.CD004233.pub2] [DOI] [PubMed] [Google Scholar]
Derry 2009a
- Derry C, Derry S, Moore RA, McQuay HJ. Single dose oral naproxen and naproxen sodium for acute postoperative pain in adults. Cochrane Database of Systematic Reviews 2009, Issue 1. [DOI: 10.1002/14651858.CD004234.pub3] [DOI] [PMC free article] [PubMed] [Google Scholar]
Derry 2009b
- Derry P, Derry S, Moore RA, McQuay HJ. Single dose oral diclofenac for acute postoperative pain in adults. Cochrane Database of Systematic Reviews 2009, Issue 2. [DOI: 10.1002/14651858.CD004768] [DOI] [PMC free article] [PubMed] [Google Scholar]
Edwards 1999
- Edwards JE, McQuay HJ, Moore RA, Collins SL. Reporting of adverse effects in clinical trials should be improved: lessons from acute postoperative pain. Journal of Pain and Symptom Management 1999;18(6):427‐37. [DOI] [PubMed] [Google Scholar]
EMEA 2002
- The European Agency for the Evaluation of Medicinal Products. EMEA public statement on parecoxib sodium (Dynastat/Rayzon/Xapit): risk of serious hypersensitivity and skin reactions. EMEA (25175/02), 2002. [Google Scholar]
FitzGerald 2001
- FitzGerald GA, Patrono C. The coxibs, selective inhibitors of cyclooxygenase‐2. New England Journal of Medicine 2001;345(6):433‐42. [DOI] [PubMed] [Google Scholar]
Graff 2007
- Graff J, Arabmotlagh M, Cheung R, Geisslinger G, Harder S. Effects of parecoxib and dipyrone on platelet aggregation in patients undergoing meniscectomy: a double‐blind, randomized, parallel‐group study. Clinical Therapeutics 2007;29(3):438‐47. [DOI] [PubMed] [Google Scholar]
Grahame‐Smith 2002
- Grahame‐Smith DG, Aronson JK. Oxford textbook of clinical pharmacology and drug therapy. 3rd Edition. Oxford: Oxford University Press, 2002. [ISBN: 13: 978‐0‐19‐263234‐0] [Google Scholar]
Hall 2008
- Hall PE, Derry S, Moore RA, McQuay HJ. Single dose oral lornoxicam for acute postoperative pain in adults. Cochrane Database of Systematic Reviews 2008, Issue 4. [DOI: 10.1002/14651858.CD007441] [DOI] [PMC free article] [PubMed] [Google Scholar]
Harris 2004
- Harris SI, Stoltz RR, LeComte D, Hubbard RC. Parecoxib sodium demonstrates gastrointestinal safety comparable to placebo in healthy subjects. Journal of Clinical Gastroenterology 2004;38(7):575‐80. [DOI] [PubMed] [Google Scholar]
Hawkey 1999
- Hawkey CJ. Cox‐2 inhibitors. Lancet 1999;353(9149):307‐14. [DOI] [PubMed] [Google Scholar]
Jadad 1996a
- Jadad A, Carroll D, Moore RA, McQuay HJ. Developing a database of published reports of randomised clinical trials in pain research. Pain 1996;66:239‐46. [DOI] [PubMed] [Google Scholar]
Jadad 1996b
- Jadad AR, Moore RA, Carroll D, Jenkinson C, Reynolds DJ, Gavaghan DJ, McQuay HJ. Assessing the quality of reports of randomized clinical trials: is blinding necessary?. Controlled Clinical Trials 1996;17:1‐12. [DOI] [PubMed] [Google Scholar]
L'Abbé 1987
- L'Abbé KA, Detsky AS, O'Rourke K. Meta‐analysis in clinical research. Annals of Internal Medicine 1987;107:224‐33. [DOI] [PubMed] [Google Scholar]
Layton 2006
- Layton D, Marshall V, Boshier A, Friedmann P, Shakir SA. Serious skin reactions and selective COX‐2 inhibitors: a case series from prescription‐event monitoring in England. Drug Safety 2006;29(8):687‐96. [DOI] [PubMed] [Google Scholar]
McQuay 2005
- McQuay HJ, Moore RA. Placebo. Postgraduate Medical Journal 2005;81:155‐60. [DOI] [PMC free article] [PubMed] [Google Scholar]
Moher 1999
- Moher D, Cook DJ, Eastwood S, Olkin I, Rennie D, Stroup DF. Improving the quality of reports of meta‐analyses of randomised controlled trials: the QUOROM statement. Quality of Reporting of Meta‐analyses. Lancet 1999;354(9193):1896‐900. [DOI: 10.1016/S0140-6736(99)04149-5] [DOI] [PubMed] [Google Scholar]
Moore 1996
- Moore A, McQuay H, Gavaghan D. Deriving dichotomous outcome measures from continuous data in randomised controlled trials of analgesics. Pain 1996;66(2‐3):229‐37. [DOI] [PubMed] [Google Scholar]
Moore 1997a
- Moore A, McQuay H, Gavaghan D. Deriving dichotomous outcome measures from continuous data in randomised controlled trials of analgesics: Verification from independent data. Pain 1997;69(1‐2):127‐30. [DOI] [PubMed] [Google Scholar]
Moore 1997b
- Moore A, Moore O, McQuay H, Gavaghan D. Deriving dichotomous outcome measures from continuous data in randomised controlled trials of analgesics: Use of pain intensity and visual analogue scales. Pain 1997;69(3):311‐5. [DOI] [PubMed] [Google Scholar]
Moore 1998
- Moore RA, Gavaghan D, Tramer M, Collins SL, McQuay HJ. Size is everything ‐ large amounts of information are needed to overcome random effects in estimating direction and magnitude of treatment effects. Pain 1998;73(3):209‐16. [DOI] [PubMed] [Google Scholar]
Moore 2003
- Moore RA, Edwards J, Barden J, McQuay HJ. Bandolier's Little Book of Pain. Oxford: Oxford University Press, 2003. [Google Scholar]
Moore 2005
- Moore RA, Edwards JE, McQuay HJ. Acute pain: individual patient meta‐analysis shows the impact of different ways of analysing and presenting results. Pain 2005;116(3):322‐31. [DOI] [PubMed] [Google Scholar]
Moore 2006
- Moore A, McQuay H. Bandolier's Little Book of Making Sense of the Medical Evidence. Oxford: Oxford University Press, 2006. [Google Scholar]
Moore 2008
- Moore RA, McQuay HJ, Wasey J, Derry S. Single dose oral diflunisal for acute postoperative pain in adults. Cochrane Database of Systematic Reviews 2008, Issue 4. [DOI: 10.1002/14651858.CD007440] [DOI] [PMC free article] [PubMed] [Google Scholar]
Morris 1995
- Morris JA, Gardner MJ. Calculating confidence intervals for relative risk, odds ratio and standardised ratios and rates. In: Gardner MJ, Altman DG editor(s). Statistics With Confidence. London: British Medical Journal, 1995:50‐63. [DOI] [PMC free article] [PubMed] [Google Scholar]
Noveck 2001
- Noveck RJ, Laurent A, Kuss M, Talwalker S, Hubbard RC. Parecoxib sodium does not impair platelet function in healthy elderly and non‐elderly individuals: Two randomised, controlled trials. Clinical Drug Investigation 2001;21(7):465‐76. [Google Scholar]
Nussmeier 2005
- Nussmeier NA, Whelton AA, Brown MT, Langford RM, Hoeft A, Parlow JL, et al. Complications of the COX‐2 inhibitors parecoxib and valdecoxib after cardiac surgery. New England Journal of Medicine 2005;352(11):1081‐91. [DOI] [PubMed] [Google Scholar]
Oldman 1999
- Oldman A, Smith LA, Collins S, Carroll D, Wiffen PJ, McQuay HJ, Rees J, Moore A. Single dose oral aspirin for acute pain. Cochrane Database of Systematic Reviews 1999, Issue 4. [DOI: 10.1002/14651858.CD002067] [DOI] [PubMed] [Google Scholar]
Ott 2003
- Ott E, Nussmeier NA, Duke PC, Feneck RO, Alston RP, Snabes MC, et al. Efficacy and safety of the cyclooxygenase 2 inhibitors parecoxib and valdecoxib in patients undergoing coronary artery bypass surgery. Journal of Thoracic and Cardiovascular Surgery 2003;125(6):1481‐92. [DOI] [PubMed] [Google Scholar]
Roy 2007
- Roy YM, Derry S, Moore RA. Single dose oral lumiracoxib for postoperative pain. Cochrane Database of Systematic Reviews 2007, Issue 4. [DOI: 10.1002/14651858.CD006865] [DOI] [PMC free article] [PubMed] [Google Scholar]
Smith 2000
- Smith LA, Carroll D, Edwards JE, Moore RA, McQuay HJ. Single‐dose ketorolac and pethidine in acute postoperative pain: systematic review with meta‐analysis. British Journal of Anaesthesia 2000;84(1):48‐58. [DOI] [PubMed] [Google Scholar]
Stoltz 2002
- Stoltz RR, Harris SI, Kuss ME, LeComte D, Talwalker S, Dhadda S, et al. Upper GI mucosal effects of parecoxib sodium in healthy elderly subjects. American Journal of Gastroenterology 2002;97(1):65‐71. [DOI] [PubMed] [Google Scholar]
Toms 2008
- Toms L, McQuay HJ, Derry S, Moore RA. Single dose oral paracetamol (acetaminophen) for postoperative pain in adults. Cochrane Database of Systematic Reviews 2008, Issue 4. [DOI: 10.1002/14651858.CD004602.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Tramèr 1997
- Tramèr MR, Reynolds DJM, Moore RA, McQuay HJ. Impact of covert duplicate publication on meta‐analysis: a case study. BMJ 1997;315:635‐9. [DOI] [PMC free article] [PubMed] [Google Scholar]
References to other published versions of this review
Barden 2003
- Barden J, Edwards JE, McQuay HJ, Moore RA. Oral valdecoxib and injected parecoxib for acute postoperative pain: a quantitative systematic review. BMC Anesthesiology 2003;3(1):1. [DOI] [PMC free article] [PubMed] [Google Scholar]


