Graphical abstract
Keywords: Curcumol, Interstitial cystitis, Bioinformatics, Biomarkers, PTK2, protein-protein interaction
Highlights
-
•
A PPI network showing protein interaction was produced.
-
•
3 top biotargets of curcumol against IC were identified.
-
•
Human IC sections showed increased PTK2, p-PTK2Tyr397 expressions.
-
•
Curcumol-treated IC mice benefited reduced PTK2, p-PTK2Tyr397 expressions.
-
•
PTK2 may be a potential biomarker for screening and treating IC.
Abstract
This study was designed to reveal the predictive targets and biological mechanisms of curcumol against interstitial cystitis (IC). By use of available databases and bioinformatic assays, pathogenetic targets of IC and functional targets of curcumol were identified respectively. A network of functional protein-protein interaction (PPI) was produced before screening the main predictive targets, biological processes and signaling pathways of curcumol against IC. In bioinformatic findings, the data of ingenuity pathway analysis (IPA) delineated that curcumol exerted anti-IC benefits through regulating multipronged signaling pathways, including tyrosine protein kinase-2 (PTK2) pathway. Further, optimal 18 biotargets of curcumol against IC were harvested through differential expression analysis. And the predictive targets of receptor tyrosine-protein kinase erbB-2 (ERBB2), epidermal growth factor receptor (EGFR) and PTK2 were the most important molecules. In further validated experiments, PTK2 and phosphorylation PTK2 (p-PTK2) were representatively selected for testing by human and animal IC samples. As results, increased immunoreactive proteins of tumor necrosis factor alpha (TNF-α), PTK2 and p-PTK2Tyr397 in human IC sections were observed, accompanied with altered urinary parameters. Interestingly, curcumol-treated IC mice showed that intracellular expressions of PTK2, p-PTK2Tyr397 in bladder samples were reduced, accompanied with lowered blood inflammatory cytokines of interleukin 6 (IL-6), TNF-α. In conclusion, the current bioinformatic data and preliminary findings unravel that the predominant targets of curcumol against IC may be the potential biological markers for screening and treating IC, such as PTK2 molecule.
Introduction
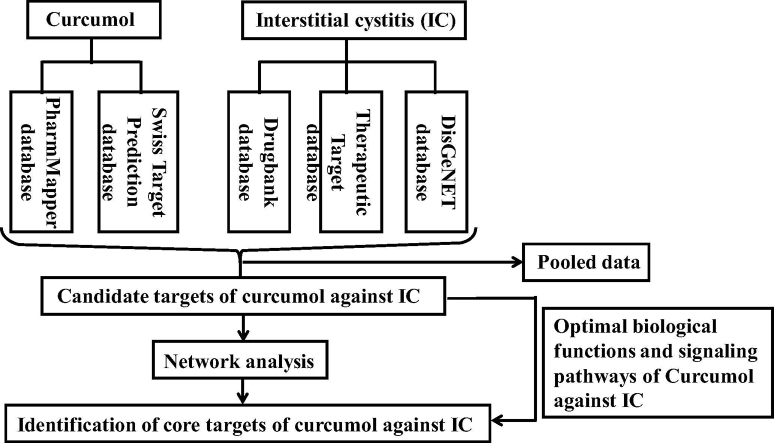
Interstitial cystitis (IC), a urinary tract infection, refers to the chronic inflammation and stress that disrupt the upper urinary tract functions. Pathological syndromes of IC may have urinary frequency, pain with urination, and dysuria, hematuria, hemorrhage [1], [2]. The common cause of infection is Escherichia coli. In addition, drug-induced hemorrhagic cystitis is another inflammation of the bladder [3], [4]. The main clinical therapy against IC is antibiotic medication by the time- and dose-dependent manners, such as nitrofurantoin, trimethoprim (uncomplicated case), phenazopyridine (complicated case) [5]. However, a long-term treatment of antibiotics may induce drug resistance over time. Therefore, further development of candidate medication to treat IC is needed. Curcumol, isolated from Rhizoma Curcumaeis, is a bioactive component with potent pharmacological activities. And it is characterized with potential anti-inflammatory, anti-virus, anti-microbial, and anti-cancer effects [6], [7]. Increasing evidences has suggested that curcumol plays a potent inhibitory effect on the proliferation of human bladder cancer cells [8]. However, the pharmacological study of curcumol against IC is limited presently. In addition to literature analysis, a predictive tool of network pharmacology can optimize and propose the main functional targets and molecular mechanisms of bioactive component against disease [9]. Therefore, the current study used network pharmacology-analyzed bioinformatic data to uncover the main predictive targets, and correlative biological processes and signaling pathways of curcumol against IC. In parallel, the samples of clinical IC and curcumol-treated rats were collected and established to characterize the pathological and pharmacological biotargets, respectively. Together, the graphical abstract of this study design was demonstrated visibly in Fig. 1.
Fig. 1.
This study used bioinformatic assays to predict the main biotargets and molecular pathways of curcumol against IC, followed by experimental validation.
Experimental
Identification of candidate targets of curcumol against IC
All curcumol-associated functional targets were collected from the databases of PharmMapper (http://lilab.ecust.edu.cn/pharmmapper/submit_file.php) and Swiss Target Prediction (http://www.swisstargetprediction.ch/index.php). In addition, pathogenetic and therapeutic targets of IC were produced from the databases of DisGeNET (http://www.disgenet.org/web/DisGeNET/menu/search?0), Drugbank (https://www.drugbank.ca/), Therapeutic Target Database (https://db.idrblab.org/ttd/), respectively. Further, the curcumol-pharmacological targets were pooled with cystitis-pathologic targets before picking up the predictive targets of curcumol against IC.
Construction of PPI network and verification of main targets of curcumol against IC
The pooled targets of curcumol against IC were projected into FunRich_3.1.3 software (http://www.funrich.org/) to establish the target-functional proteins. And a PPI network of predictive targets was constructed. In addition, the identifiable data were further imported to Cytoscape (v3.6.1) (https://cytoscape.org/). The network analyzer setting was used to visualize the network targets of curcumol against IC based on topological parameters. The optimal targets were identified according to the maximum degree values.
Confirmation of biological processes and molecular pathways of curcumol against IC
The Database for Annotation, Visualization and Integrated Discovery (DAVID) database (https://david.ncifcrf.gov/home.jsp) was used to extract the available biological functions related to core targets. These data were further introduced in the Omicshare Cloud Platform (http://www.omicshare.com/tools/Home/Soft/gogsea) to visualize the biological processes and signaling pathways from the core targets of curcumin anti-IC. And a p-value was used to plot advanced bubble diagrams of biological processes and signaling pathways.
Human designs
Adult male patients (n = 3) were diagnostically determined as IC through the biochemical, pathological, and medical imaging tests at Department of Urology Surgery. The serological data and clinical imaging were collected for further analyses. Additionally, IC samples of all these cases were surgically isolated during precancerous screening, followed by immunohistochemical and immunofluorescence stains. All principles of this human study were conducted following the guidelines issued by Declaration of Helsinki [10].
Animal designs
Adult female Kunming mice were obtained from the Experimental Animal Unit of Guilin Medical University (Guilin, China). The mice were maintained in animal house under the controlled conditions of 22 ± 1 °C, around 60 ± 5% humidity, 12 altered hours in cycled light/dark. The mice were allowed to have filtered water and rodent chow freely. The present study was authorized through Institutional Animal Care and Use Committee of the Guilin Medical University. After acclimatization for at least one week, the anticancer drug-induced IC mice by cyclophosphamide (CPS) was established as previously described [11]. Briefly, the fresh-prepared CPS solution were given to mice via oral gavage with 50 mg/kg in body weight every 3 days, followed by curcumol treatments (40 and 80 mg/kg; w/w) for consecutive 12 days. At the end of experiment, the serum and bladder samples from all mice were isolated for biochemical tests, and stains used for immunohistochemistry and immunofluorescence. For more details, all immunostaining procedures were referenced in previous description [12].
Statistical analysis
The blood results were expressed as mean ± standard deviation (SD). The mice data were assayed through statistical product and service solutions (SPSS) 19.0 software (IL, USA). Multiple comparisons were calculated by using the analysis of variance (ANOVA) with Tukey's post hoc test. The statistical significance was considered as P < 0.05.
Results
Candidate targets and identification of core targets
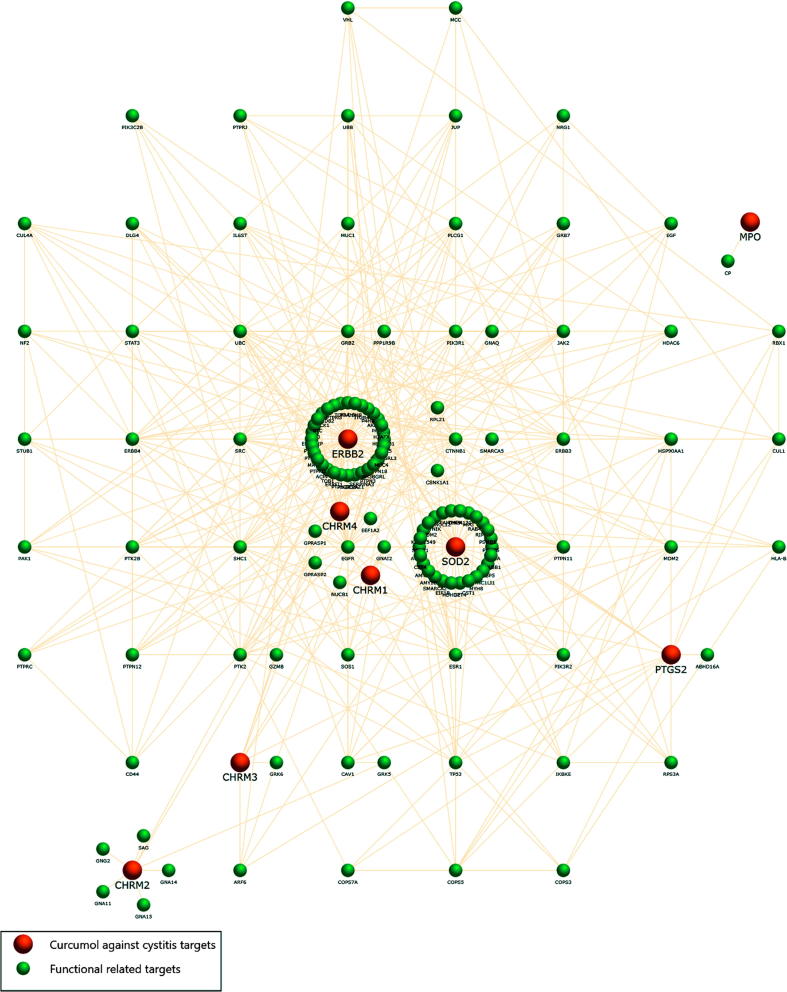
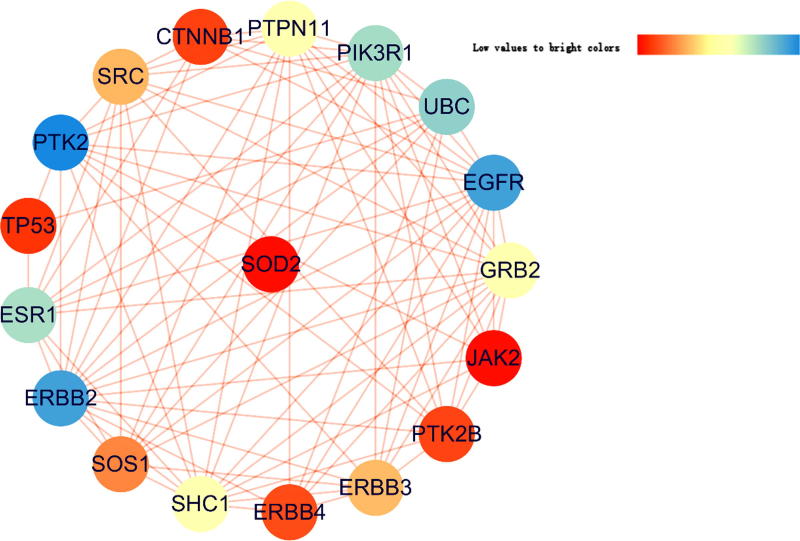
After bioinformatic assays, all 64 IC-pathogenic targets and 314 curcumol-therapeutic targets were screened. For further validation, 8 predictive targets were identified ultimately. A PPI network of curcumol against IC was constructed through assaying a function-related protein-protein interaction in these 8 functional targets. As shown in PPI network analysis, the hub network had 137 nodes and 449 edges with interactions and associations (Fig. 2). In topology parameters, the shorter path length and the larger median value indicated the more important targets. After calculation, 18 core targets were optimally obtained. According to the resultant visualization, the predictive targets of ERBB2, EGFR and PTK2 were the most important (Fig. 3).
Fig. 2.
A PPI network was used to construct the targets of curcumol against IC (Red nodes) and functional related targets (Green nodes) before the main targets were identified and collected.
Fig. 3.
After being analyzed, the main targets of curcumol against IC were identified and correlated. As a result, 3 top targets were representatively found as ERBB2, EGFR and PTK2 prior to further validation.
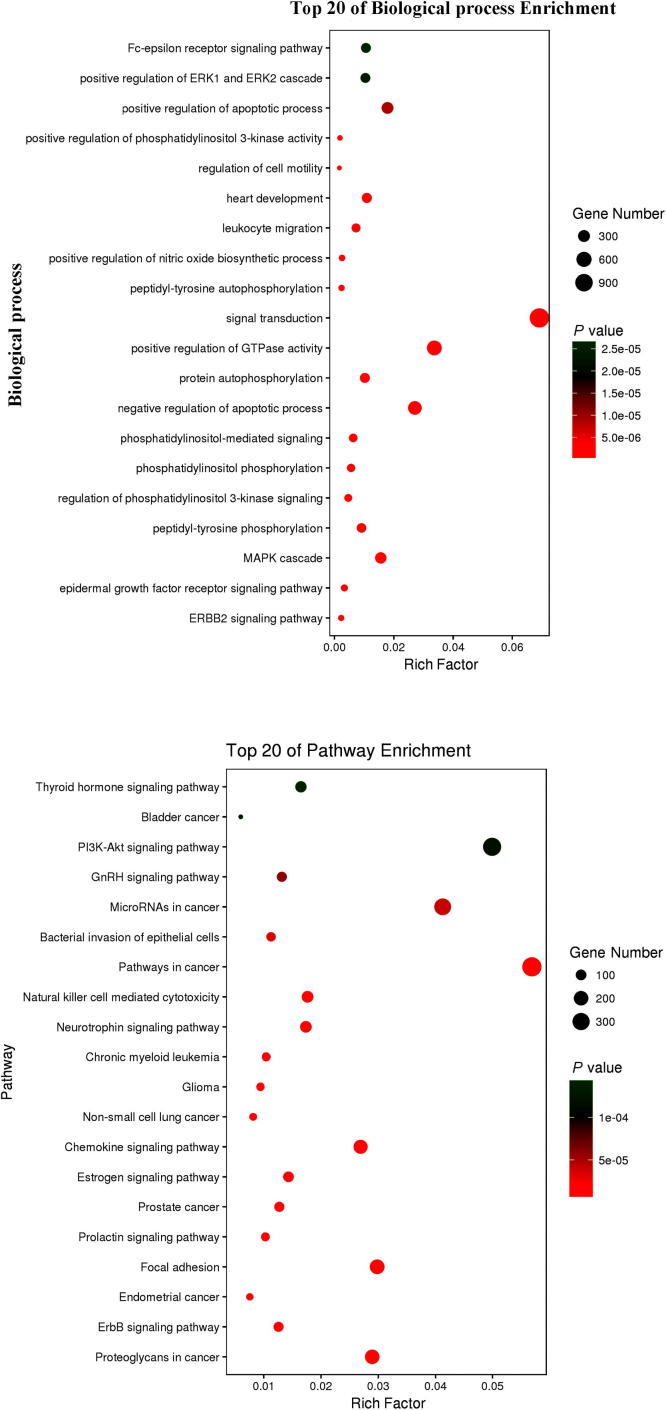
Biological function and signaling pathways of curcumol against IC
As results, the top 20 biological function enrichments were obtained through DAVID database and omicshare cloud platform. As shown in Fig. 4, the results showed that the biological processes from the predictive targets of curcumol against IC were principally linked to regulating the ERBB2 signaling pathway, epidermal growth factor receptor (EGFR) signaling pathway, MAPK cascade, peptidyl-tyrosine phosphorylation, regulation of phosphatidylinositol 3-kinase signaling phosphatidylinositol phosphorylation (PI3-K), phosphatidylinositol-mediated signaling, negative regulation of apoptotic process, protein autophosphorylation, positive regulation of GTPase activity, signal transduction, peptidyl-tyrosine autophosphorylation, positive regulation of nitric oxide biosynthetic process, leukocyte migration, heart development, regulation of cell motility, positive regulation of phosphatidylinositol 3-kinase activity, positive regulation of apoptotic process, positive regulation of ERK1 and ERK2 cascade, Fc-epsilon receptor signaling pathway. In addition, the signaling pathways were chiefly involved in the Proteoglycans in cancer, ErbB signaling pathway, Endometrial cancer, Focal adhesion, Prolactin signaling pathway, Prostate cancer, Estrogen signaling pathway, Chemokine signaling pathway, Non-small cell lung cancer, Glioma, Chronic myeloid leukemia, Neurotrophin signaling pathway, Natural killer cell mediated cytotoxicity, Pathways in cancer, Bacterial invasion of epithelial cells, MicroRNAs in cancer, GnRH signaling pathway, PI3K-Akt signaling pathway, Bladder cancer, Thyroid hormone signaling pathway, respectively.
Fig. 4.
After data analyses in DAVID tool, top 20 biological processes and signaling pathways of curcumol against IC were showed respectively prior to further bioinformatic discussion.
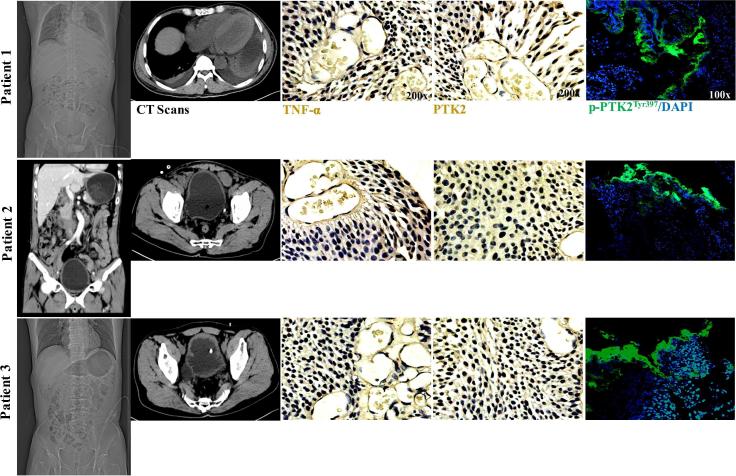
Case reports of patients with IC
To reveal the clinical and histopathological characteristics of IC, human data and samples were used to assay. In urinary examination, these IC patients showed positive urinary protein, bacterial infection, and visible epithelial cell shedding (Table 1). As shown in clinical computed tomography (CT) screening, chronic cystitis with urinary tract infection was observed. In addition, histopathological stains resulted in elevated intracellular expression of TNF-α in these IC sections. In order to characterize the predictive target of PTK2 in human samples, histopathological and immunofluorescent stains were conducted for further validation. As a result, the IC sections showed increased expressions of PTK2, p-PTK2Tyr397 in bladder tissues (Fig. 5).
Table 1.
Clinical parameters of urinary tests in IC patients.
| Clinical indicators | Case data | Clinical standards |
|---|---|---|
| UP | Positive | Negative |
| pH | 6.5 ± 0.4 | 4.5–8.0 |
| RBC | 18702.1 ± 12366.94 | 0–16.1 |
| WBC | 88.9 ± 61.14 | 0–9.2 |
| EC | 10.63 ± 7.67 | 0–8.7 |
| CAST | 1.06 ± 1.01 | 0–2.25 |
| BACT | 9.87 ± 7.49 | 0–50 |
| X.TAL | 0 ± 0 | 0–10 |
| YLC | 45.2 ± 78.29 | 0–10 |
| SRC | 8.5 ± 8.86 | 0–3 |
| P.CAST | 0.46 ± 0.43 | 0–1 |
| Cond. | 12.77 ± 1.81 | 5–38 |
| Total | 155246 ± 99709.97 | 0–40,000 |
Abbreviations: UP, urinary protein; RBC, red blood cell; WBC, white blood cell; EC, epithelial cells; CAST, cast; BACT, bacteria; X.TAL, crystallization; YLC, yeast cell; SRC, small round epithelial cells; P.CAST, pathological cast; Cond, conductivity; TOTAL, total count.
Fig. 5.
Clinical characteristics of patients with IC. As results, medical CT scans showed urocystic lesions with chronic cystitis, urinary tract infection. Further, immunohistochemical staining showed visible expression of cellular TNF-α in IC sections. And the immunofluorescent stains displayed increased expressions of PTK2, p-PTK2Tyr397 in IC sections.
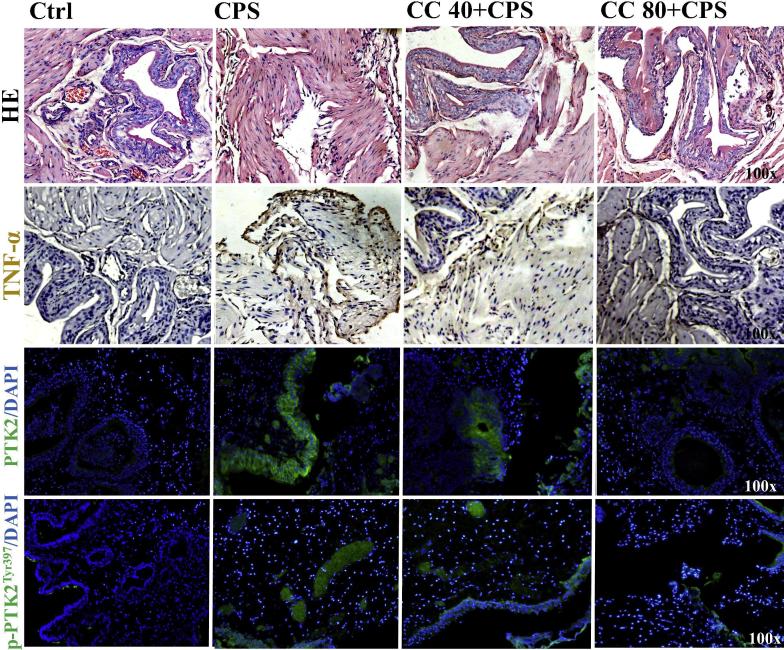
Experimental study of PTK2 targets in curcumol-treated mice
To further validate the predictive PTK2 targets of curcumol against IC in vivo, well-established CPS-induced IC in mice were used for experimental assays. As results, CPS-lesioned mice showed reduced body weights, and elevated contents of lactate dehydrogenase (LDH), IL-6, TNF-α in sera (P < 0.05). Interestingly, high dose of curcumol treatment beneficially increased body mass, and lowered blood contents of LD, IL-6, TNF-α (P < 0.05) (Table 2). In histopathological observation with hematoxylin-eosin (HE) stains, CPS-lesioned mice exhibited morphological changes, inflammatory infiltration, and necrotic cells in bladder tissues. However, curcumol-treated mice showed reduced CPS-induced cytotoxicity in bladder cells, such as decreased necrotic cells and inflammatory lesions. Followed by immunohistochemical staining, CPS-lesioned bladders in mice resulted in increased intracellular expression of TNF-α, while curcumol-treated mice showed reduced endogenous TNF-α expression in bladder sections.
Table 2.
Fundamental data of curcumol-treated CPS-lesioned mice.
| Parameters | Control | CPS | CC40 + CPS | CC80 + CPS |
|---|---|---|---|---|
| Body mass (day 1) | 26.4 ± 0.8 | 26.5 ± 0.7 | 26.7 ± 0.8 | 26.4 ± 0.7 |
| Body mass (day 6) | 29.6 ± 0.9 | 24.2 ± 1.1a | 25.3 ± 0.9 | 27.6 ± 0.8 |
| Body mass (day 12) | 31.7 ± 1.5 | 21.8 ± 1.3a | 27.7 ± 0.9 | 28.8 ± 0.7b |
| LDH (mmol/L) | 12.13 ± 0.45 | 48.69 ± 1.59a | 33.66 ± 3.95 | 23.70 ± 2.96b |
| TNF-α (pg/mL) | 17.72 ± 1.62 | 65.84 ± 7.59a | 41.05 ± 4.28 | 31.21 ± 3.11b |
| IL6 (pg/mL) | 11.65 ± 1.06 | 46.47 ± 4.50a | 28.52 ± 4.48 | 21.18 ± 2.12b |
Abbreviations: CPS, cyclophosphamide; LDH, lactate dehydrogenase; TNF-α, tumor necrosis factor α; IL-6, interleukin 6; CC40, 80, curcumol 40, 80 mg/kg. Note: when
P < 0.05 compared to control group.
P < 0.05 compared to CPS group.
In addition, immunofluorescence staining was used to check intracellular expressions of PTK2, p-PTK2Tyr397. As results, CPS-lesioned mice showed elevated PTK2, p-PTK2Tyr397 expressions in bladder sections. After cotreatment with curcumol, the cellular expressions of PTK2, p-PTK2Tyr397 were down-regulated in comparison with those in CPS-lesioned controls (Fig. 6).
Fig. 6.
Anti-IC activities of curcumol on CPS-lesioned mice. As showed in HE stains, CPS-lesioned mice resulted in morphological changes in bladder tissues. And increased expression of intracellular TNF-α in the bladder were observed in CPS-lesioned mice by using immunohistochemical stain. Interestingly, curcumol-treated mice showed bladder morphological improvement, and reduced TNF-α expression. As showed in immunofluorescence staining, CPS-lesioned mice resulted in elevated PTK2, p-PTK2Tyr397 expressions in bladder sections. However, down-regulated expressions of TNF-α, PTK2, p-PTK2Tyr397 in the bladder cells of curcumol-treated mice were observed, accompanied with ameliorative histomorphology in bladder tissues.
Discussion
In this study, a bioactive component-target-disease network was constructed by using the network pharmacology-based bioinformatics. And resultant 18 core targets were identified through further analysis. According to the bioinformatic results, predictive ErbB2, EGFR and PTK2 were the most important targets of curcumol against IC. Tyrosine protein kinases (PTKs) are a group of functional enzymes that can catalyze the phosphorylation of substrate tyrosine residues, and these molecules play the key roles in cellular signaling pathways [13], [14]. It is believed that the parathyroid hormones (PTHs) signaling is mainly involved in cell growth and proliferation through modulating the PTKs-MAPK pathway [15]. Mitogen-activated protein kinase (MAPK) functions as a “bridge” connecting extracellular stimuli and intracellular gene expression. It functionally regulates tumor growth and metastasis, as well as biologically participates in cell proliferation, differentiation and apoptosis through a series of effectors’ activation and inactivation [16], [17]. The biomolecules of ErbB2, EGFR and PTK2 belong to PTK family. ErbB2 refers to one of the important oncogenes in the EGFR family, and it is a transmembrane protein with tyrosine kinase activity [18], [19]. EGFR is a receptor protein that distributes on the membrane of epithelial cells. When combined with ligands, EGFR activates the downstream signaling molecules and induces the biological effects, such as cell proliferation, differentiation and migration. In addition, EGFR plays a promotive role in transformation of malignant cells [20], [21]. PTK2, known as focal adhesion kinase (FAK), is a non-receptor protein tyrosine kinase located in the cytoplasm. Functionally, the activation of PTK2 is to promote the formation of tumor, and it also has other biological activities of regulating cellular apoptosis and stress [22], [23]. Recently, some studies have found that PTK2 is positively expressed in bladder cancer cells, and the over-expression of PTK2 facilitates the neoplastic cells to escape from apoptotic signals, gradually promoting tumor growth, metastasis [24], [25]. Interestingly, these functional molecular pathways from literature review were similarly proposed in this study. However, the experimental investigation of PTK2 in interstitial cystitis is not yet conducted. Based on bioinformatic findings, the current study predicted the core targets of curcumol against IC through a network pharmacology method. As a result, a key biotarget of PTK2 was representatively selected for further validation in human samples. In addition to positive detection of TNF-α expression in IC sections, intravesical PTK2, p-PTK2Tyr397 were markedly up-regulated through immunostaining. The activated PTK2 were positive correlation with cystitis conditions in urinary tests. These clinicopathological findings indicated that intracellular over-expression of PTK2 might be a key pathological promoter in development of IC. Further, PTK2 might be a potential therapeutic target for IC.
To further validate experimental study in vivo, CPS-induced IC mice were used to evaluate the pharmacological activities of curcumol against IC. As results, curcumol-treated mice showed reduced inflammatory cytokines (IL-6, TNF-α) in sera and suppressed intracellular cytotoxicity, characterized with down-regulated expressions of PTK2, p-PTK2Tyr397 in bladder cells. Therefore, this study can be reasoned that the anti-IC activities of curcumol might benefit from the anti-inflammatory effects through suppressing intravesical PTK2 and phosphorylation bioactivities.
Conclusions
Collectively, these bioinformatic and experimental findings demonstrate that predictive targets of curcumol against IC may be mainly linked to inhibition of inflammation-associated pathways and inactivation of intracellular PTK2 activity in bladder cells. It highlights that bioinformatic method based on network pharmacology may predict the biological markers for IC screening and treating. Interestingly, PTK2 may be a potential biomarker for curcumol treating IC.
Conflict of interest
The authors have declared no conflict of interest.
Acknowledgments
This study was supported by the National Natural Science Foundation of Guangxi Province (No. 2017GXNSFBA198049, 2018GXNSFAA281153), and the National Natural Science Foundation of China (No. 81660091).
Footnotes
Peer review under responsibility of Cairo University.
References
- 1.Wang A., Nizran P., Malone M.A., Riley T. Urinary tract infections. Prim Care. 2013;40:687–706. doi: 10.1016/j.pop.2013.06.005. [DOI] [PubMed] [Google Scholar]
- 2.Lane DR, Takhar SS. Diagnosis and management of urinary tract infection and pyelonephritis 2011;29:539–52. [DOI] [PubMed]
- 3.Sabetkish S., Sabetkish N., Kajbafzadeh A.M. Early detection of deep wound infection in bladder exstrophy and hypospadias using a novel intervention. J Wound Care. 2018;27:686–691. doi: 10.12968/jowc.2018.27.10.686. [DOI] [PubMed] [Google Scholar]
- 4.Johnston D., Schurtz E., Tourville E., Jones T., Boemer A., Giel D. Risk factors associated with severity and outcomes in pediatric patients with hemorrhagic cystitis. J Urol. 2016;195:1312–1317. doi: 10.1016/j.juro.2015.11.035. [DOI] [PubMed] [Google Scholar]
- 5.Ogawa T., Ishizuka O., Ueda T., Tyagi P., Chancellor M.B., Yoshimura N. Current and emerging drugs for interstitial cystitis/bladder pain syndrome (IC/BPS) Expert Opin Emerg Drugs. 2015;20:555–570. doi: 10.1517/14728214.2015.1105216. [DOI] [PubMed] [Google Scholar]
- 6.Lu J.J., Dang Y.Y., Huang M. Anti-cancer properties of terpenoids isolated from Rhizoma Curcumae – a review. J Ethnopharmacol. 2012;143:406–411. doi: 10.1016/j.jep.2012.07.009. [DOI] [PubMed] [Google Scholar]
- 7.Chen X., Zong C., Gao Y., Xu W.S., Chen X.P., Wang Y.T. Curcumol exhibits anti-inflammatory properties by interfering with the JNK-mediated AP-1 pathway in lipopolysaccharide-activated RAW264.7 cells. Eur J Pharmacol. 2014;723:339–345. doi: 10.1016/j.ejphar.2013.11.007. [DOI] [PubMed] [Google Scholar]
- 8.Zhou L., Wei E., Zhou B., Bi G., Gao L., Zhang T. Anti-proliferative benefit of curcumol on human bladder cancer cells via inactivating EZH2 effector. Biomed Pharmacother. 2018;104:798–805. doi: 10.1016/j.biopha.2018.05.101. [DOI] [PubMed] [Google Scholar]
- 9.Su M., Guo C., Liu M., Liang X., Yang B. Therapeutic targets of vitamin C on liver injury and associated biological mechanisms: a study of network pharmacology. Int Immunopharmacol. 2019;66:383–387. doi: 10.1016/j.intimp.2018.11.048. [DOI] [PubMed] [Google Scholar]
- 10.Guo C., Pan Q., Su M., Li R. Clinical immunophenotype of nasopharyngeal neuroendocrine carcinoma with metastatic liver cancer. Clin Chim Acta. 2017;471:283–285. doi: 10.1016/j.cca.2017.06.016. [DOI] [PubMed] [Google Scholar]
- 11.Ge B., Yang D., Wu X., Zhu J., Wei W., Yang B. Cytoprotective effects of glycyrrhetinic acid liposome against cyclophosphamide-induced cystitis through inhibiting inflammatory stress. Int Immunopharmacol. 2018;54:139–144. doi: 10.1016/j.intimp.2017.11.010. [DOI] [PubMed] [Google Scholar]
- 12.Zhou R., Xu X., Liu M., Wu X., Li R. Immunophenotypes of ductal epithelial cells in advanced pancreatic ductal adenocarcinoma. Digestion. 2018;1:1–5. doi: 10.1159/000492861. [DOI] [PubMed] [Google Scholar]
- 13.Verma S., Sharma S. Protein tyrosine phosphatase as potential therapeutic target in various disorders. Curr Mol Pharmacol. 2018;11:191–202. doi: 10.2174/1874467211666180226161846. [DOI] [PubMed] [Google Scholar]
- 14.Elson A. Stepping out of the shadows: oncogenic and tumor-promoting protein tyrosine phosphatases. Int J Biochem Cell Biol. 2018;96:135–147. doi: 10.1016/j.biocel.2017.09.013. [DOI] [PubMed] [Google Scholar]
- 15.Kim H.J., Lin D., Lee H.J., Li M., Liebler D.C. Quantitative profiling of protein tyrosine kinases in human cancer cell lines by multiplexed parallel reaction monitoring assays. Mol Cell Proteomics: MCP. 2016;15:682–691. doi: 10.1074/mcp.O115.056713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jiang M., Niu C., Cao J., Ni D.A., Chu Z. In silico-prediction of protein-protein interactions network about MAPKs and PP2Cs reveals a novel docking site variants in Brachypodium distachyon. Sci Rep. 2018;8:15083. doi: 10.1038/s41598-018-33428-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kim E.K., Choi E.J. Compromised MAPK signaling in human diseases: an update. Arch Toxicol. 2015;89(6):867–882. doi: 10.1007/s00204-015-1472-2. [DOI] [PubMed] [Google Scholar]
- 18.Hervent A.S., De Keulenaer G.W. Molecular mechanisms of cardiotoxicity induced by ErbB receptor inhibitor cancer therapeutics. Int J Mol Sci. 2012;13:12268–12286. doi: 10.3390/ijms131012268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Vermeulen Z., Segers V.F., De Keulenaer G.W. ErbB2 signaling at the crossing between heart failure and cancer. Basic Res Cardiol. 2016;111(6):60. doi: 10.1007/s00395-016-0576-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Herbst R.S. Review of epidermal growth factor receptor biology. Int J Radiat Oncol Biol Phys. 2004;59:21–26. doi: 10.1016/j.ijrobp.2003.11.041. [DOI] [PubMed] [Google Scholar]
- 21.Singh D., Attri B.K., Gill R.K., Bariwal J. Review on EGFR inhibitors: critical updates. Mini Rev Med Chem. 2016;16:1134–1166. doi: 10.2174/1389557516666160321114917. [DOI] [PubMed] [Google Scholar]
- 22.Chan K.T., Cortesio C.L., Huttenlocher A. FAK alters invadopodia and focal adhesion composition and dynamics to regulate breast cancer invasion. J Cell Biol. 2009;185:357–370. doi: 10.1083/jcb.200809110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mitra S.K., Schlaepfer D.D. Integrin-regulated FAK-Src signaling in normal and cancer cells. Curr Opin Cell Biol. 2006;18:516–523. doi: 10.1016/j.ceb.2006.08.011. [DOI] [PubMed] [Google Scholar]
- 24.Sulzmaier F.J., Jean C., Schlaepfer D.D. FAK in cancer: mechanistic findings and clinical applications. Nat Rev Cancer. 2014;14:598–610. doi: 10.1038/nrc3792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zhu X., Bao Y., Guo Y., Yang W. Proline-rich protein tyrosine kinase 2 in inflammation and cancer. Cancers (Basel) 2018;10(5) doi: 10.3390/cancers10050139. [DOI] [PMC free article] [PubMed] [Google Scholar]