Key Points
Trib1 controls both eosinophil lineage commitment and identity.
Trib1 integrates IL-5 and C/EBPα signals to promote eosinophil lineage fidelity by suppressing the neutrophil program.
Abstract
Eosinophils and neutrophils are critical for host defense, yet gaps in understanding how granulocytes differentiate from hematopoietic stem cells (HSCs) into mature effectors remain. The pseudokinase tribbles homolog 1 (Trib1) is an important regulator of granulocytes; knockout mice lack eosinophils and have increased neutrophils. However, how Trib1 regulates cellular identity and function during eosinophilopoiesis is not understood. Trib1 expression markedly increases with eosinophil-lineage commitment in eosinophil progenitors (EoPs), downstream of the granulocyte/macrophage progenitor (GMP). Using hematopoietic- and eosinophil-lineage–specific Trib1 deletion, we found that Trib1 regulates both granulocyte precursor lineage commitment and mature eosinophil identity. Conditional Trib1 deletion in HSCs reduced the size of the EoP pool and increased neutrophils, whereas deletion following eosinophil lineage commitment blunted the decrease in EoPs without increasing neutrophils. In both modes of deletion, Trib1-deficient mice expanded a stable population of Ly6G+ eosinophils with neutrophilic characteristics and functions, and had increased CCAAT/enhancer binding protein α (C/EBPα) p42. Using an ex vivo differentiation assay, we found that interleukin 5 (IL-5) supports the generation of Ly6G+ eosinophils from Trib1-deficient cells, but is not sufficient to restore normal eosinophil differentiation and development. Furthermore, we demonstrated that Trib1 loss blunted eosinophil migration and altered chemokine receptor expression, both in vivo and ex vivo. Finally, we showed that Trib1 controls eosinophil identity by modulating C/EBPα. Together, our findings provide new insights into early events in myelopoiesis, whereby Trib1 functions at 2 distinct stages to guide eosinophil lineage commitment from the GMP and suppress the neutrophil program, promoting eosinophil terminal identity and maintaining lineage fidelity.
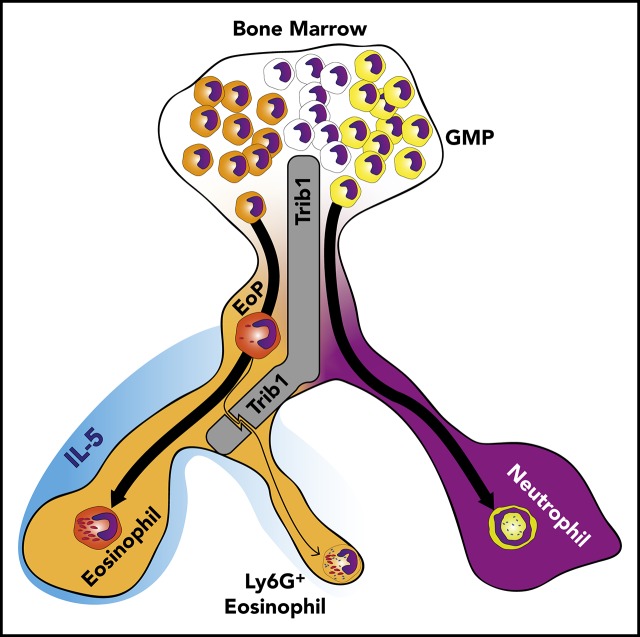
Visual Abstract
Introduction
Hematopoietic cells depend on a finely balanced network of signaling pathways to progress from multipotent progenitors to terminal effectors and maintain cellular identity. Eosinophils and neutrophils are vital for host defense yet contribute to the pathogenesis of many atopic and inflammatory conditions.1,2 These subsets develop from the granulocyte/macrophage progenitor (GMP),3 and eosinophils subsequently arise from a downstream committed interleukin 5 (IL-5) receptor α-positive (IL-5Rα+) eosinophil progenitor (EoP) in the bone marrow (BM).4 However, how eosinophil lineage commitment is regulated is not well understood.
Recent work implicates the tribbles pseudokinase family in myelopoiesis. Tribbles proteins primarily act as adaptors to promote protein degradation and/or sequestration.5-7 There are 3 mammalian tribbles homologs (Trib1-3) that are defined by a central serine/threonine kinase–like domain and C-terminal sequences that bind the E3 ubiquitin ligase COP1.8-12 Mice with germline deletion of Trib1 lack “M2” macrophages and eosinophils, and have more neutrophils, a phenotype that is influenced by the failure of Trib1 to promote CCAAT/enhancer binding protein α (C/EBPα) protein degradation.13 Myelopoiesis is unaffected in mice lacking Trib2 or Trib3.13,14
Although previous work revealed alterations in myeloid populations with Trib1 loss, the identity of the factors involved in lineage choice during granulopoiesis and terminal granulocyte identity are not well established. Using hematopoietic- and eosinophil-lineage–specific Trib1 deletion, we found that Trib1 regulates both granulocyte precursor lineage commitment and mature cell identity. Conditional Trib1 deletion in hematopoietic stem cells (HSCs) reduced the size of the EoP pool whereas deletion following eosinophil lineage commitment blunted the decrease in EoPs. In both modes of deletion, Trib1-deficient mice expanded a stable population of Ly6G+ eosinophils that failed to repress neutrophilic characteristics and functions. These cells had increased C/EBPα p42, and C/EPBα knockdown partly restored normal eosinophil development in the absence of Trib1 in vivo. We further demonstrated that Trib1 suppressed the neutrophil gene program in lineage-committed eosinophil precursors in response to IL-5. Additionally, IL-5 was unable to restore normal eosinophil differentiation or expansion from Trib1-deficient progenitors. We found that loss of Trib1 blunted eosinophil migration as Trib1-deficient eosinophils were only partly mobilized in response to type 2 lung inflammation or to eotaxin ex vivo. Together, our findings provide new insights into early steps in granulocyte development, where Trib1 acts at 2 distinct stages to control eosinophil lineage commitment from the GMP and suppress the neutrophil program in response to IL-5, promoting eosinophil terminal identity and lineage fidelity.
Materials and methods
Mice
Conditional Trib1 mice (cTrib1; C57BL/6-Trib1tm1. mrl; Taconic)15 were bred with VavCre+ (Tg[Vav1-cre]1Graf) mice16 to generate Trib1ΔHSC mice. EoCre+ mice (Epxtm1.1[cre]Jlee),17 a gift from James Lee (Mayo Clinic, Scottsdale, AZ), were bred with cTrib1 mice to generate Trib1ΔEos mice, and then with Rosa26-YFP reporter mice (Gt[ROSA]26Sortm1[EYFP]Cos; The Jackson Laboratory) to generate Trib1ΔEos-YFP mice. All Trib1+/+ (wild-type [WT]) mice used were either VavCre+ or EoCre+, as appropriate, with the WT cTrib1 allele. All mice were on the C57BL/6 background and analyzed between 5 and 12 weeks of age. Animals were housed in a specific pathogen-free facility at the University of Pennsylvania. Experiments were performed according to the guidelines from the National Institutes of Health with approved protocols from the University of Pennsylvania Animal Care and Use Committee.
Flow cytometry and cell sorting
A complete list of antibodies is provided in supplemental Table 1 (available on the Blood Web site). For complete methods, see supplemental Methods. For GMP and common myeloid progenitor (CMP) sorting, the lineage panel included Sca1, CD3ε, CD19, B220, NK1.1, Ter-119, CD127, CD11b, and Gr-1. The EoP lineage panel mirrored that for GMP/CMP sorting, except CD11b was excluded and Sca1 was gated out separately. Cells were analyzed on an LSR II or LSR Fortessa flow cytometer (BD) and data were analyzed with FlowJo software v.9.7 (TreeStar). Cells were sorted on a FACSAria II (BD) using a 70-μm nozzle at 70 psi. Gating strategies for mature cells (supplemental Figure 1A-B) and progenitors (supplemental Figure 1C-D) are provided. The frequency of live cells is shown on flow cytometry plots.
Ex vivo eosinophil culture
Eosinophils were generated ex vivo as previously described.18 Briefly, either whole BM or sorted GMPs were seeded at 1 × 106 cells per milliliter in RPMI 1640 media (Corning) supplemented with 15% fetal bovine serum (HyClone), 1% penicillin/streptomycin (Gibco), 2 mM l-glutamine (Gibco), 1 mM sodium pyruvate (Gibco), 10 mM N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid (Gibco), and 50 μM 2-mercaptoethanol (Sigma-Aldrich), in the presence of 100 ng/mL recombinant human FMS-like tyrosine kinase 3 ligand (PeproTech) and 100 ng/mL recombinant murine stem cell factor (PeproTech) and cultured for 4 days. On day 4, nonadherent cells were counted and resuspended at 1 × 106 cells per milliliter in the above RPMI 1640 media supplemented with 10 ng/mL recombinant murine IL-5 (PeproTech). The media was changed on day 8, and every 2 days thereafter, and cells were resuspended at 1 × 106 cells per milliliter with fresh IL-5. On days 10 to 14, cells were counted and processed for RNA or flow cytometry.
Phagocytosis assay
See supplemental Methods.
Transwell chemotaxis assay
Cultured eosinophils or whole BM were isolated and resuspended in RPMI 1640 medium with no phenol red (Gibco), supplemented with 0.5% low endotoxin bovine serum albumin (Sigma-Aldrich). A total of 1 × 106 cells were loaded in the upper chamber of a 6.5-mm transwell insert with a 5.0-μm pore polycarbonate membrane (Corning). The lower chamber contained media alone or media with keratinocyte-derived cytokine (KC)/CXCL1 (50 ng/mL; PeproTech) or eotaxin/CCL11 (500 ng/mL; BioLegend). Cells were allowed to migrate for 2 hours at 37°C, then collected, counted, and analyzed by flow cytometry. Whole BM was used as a control for cell migration.
Cytospins and light and electron microscopy
See supplemental Methods.
BM transplantation and C/EPBα shRNA knockdown
For mixed chimeras, CD45.1+ BM was mixed 1:1 with CD45.2+ Trib1+/+ or Trib1ΔHSC BM and 2 × 106 cells were injected via the tail vein into lethally irradiated C57BL/6.SJL mice (Charles River Laboratories). Mice were analyzed at 12 to 20 weeks posttransplant. For C/EPBα short hairpin RNA (shRNA) knockdown, cells were processed and transduced as described.14 Briefly, BM was collected from Trib1+/+ or Trib1ΔHSC mice 4 days after IV administration of 5-fluorouracil (250 mg/kg). Cells were transduced with lentivirus and 1 × 106 cells were injected IV into lethally irradiated recipients. Mice were analyzed at 9 weeks posttransplant.
Constructs and retroviruses
Production of high-titer virus was performed as described.14 Briefly, lentiviral pLKO.1 shRNA constructs19 were cotransfected into 293T cells (ATCC) with pMDL (gag-pol), pRSV-Rev, and pHIT123 (envelope). shRNA constructs expressed green fluorescent protein (GFP).
Papain treatment
Mice anesthetized with isoflurane (Phoenix) received 30 μL of intranasal phosphate-buffered saline (PBS) or 30 μg of papain (Millipore) daily for 5 days and were euthanized 24 hours later. Lungs were harvested following retrograde flushing through the heart with PBS, and were digested with collagenase D (Roche) and DNaseI (Sigma-Aldrich) for 45 minutes at 37°C. Lung digests were passed through a 70-μm filter and processed for flow cytometry.
Immunoblotting
See supplemental Methods for detailed immunoblotting methods.
qPCR
See supplemental Methods for detailed quantitative polymerase chain reaction (qPCR) methods.
Results
Trib1 regulates eosinophil lineage commitment from the GMP
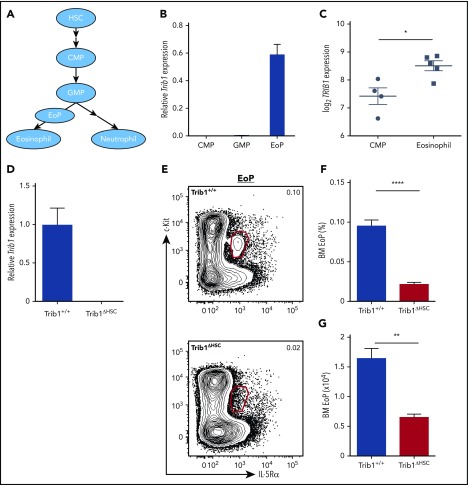
To understand Trib1 function in eosinophil commitment, we determined its expression in myeloid progenitors. During murine eosinophilopoiesis, EoPs arise from GMPs4 (Figure 1A). We measured Trib1 messenger RNA expression in CMPs, GMPs, and EoPs, and found that Trib1 is more highly expressed in EoPs compared with CMPs or GMPs (Figure 1B). TRIB1 is also upregulated during human eosinophil differentiation (Figure 1C),20 in which eosinophils arise from the CMP.21 To investigate how Trib1 modulates granulocyte development, we generated mice expressing VavCre16 and a conditional Trib1 allele15 to delete Trib1 in all postembryonic hematopoietic cells (Trib1ΔHSC). Trib1 deletion was validated by qPCR in the BM compared with mice expressing VavCre alone (Trib1+/+) (Figure 1D). Similar to Trib1 germline knockout mice,13 Trib1ΔHSC mice had increased neutrophils and markedly decreased eosinophils in the spleen, blood, lung, and colon (supplemental Figure 2A-I; data not shown).
Figure 1.
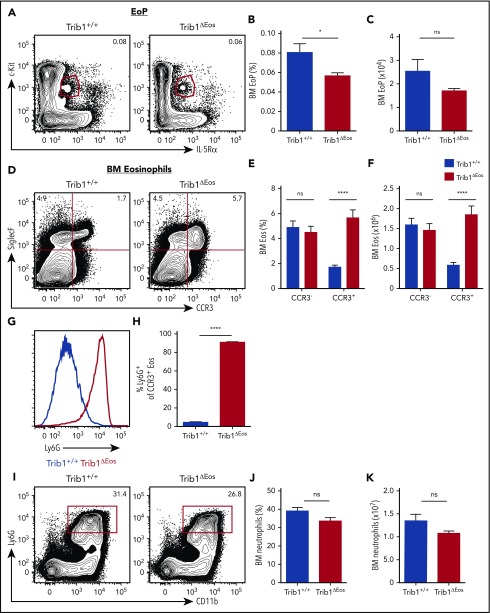
Trib1 expression increases with eosinophil commitment and is required for EoP homeostasis. (A) Model of eosinophil and neutrophil development from the HSC including the EoP. (B) Trib1 expression in sorted CMP, GMP, and EoP from WT C57BL/6 mice relative to 18s, normalized to Trib1+/+ neutrophils; representative of 3 experiments. (C) Relative expression of TRIB1 in human CMP and eosinophils (obtained from the differentiation map data set20) using BloodSpot.65 (D) qPCR from Trib1+/+ and Trib1ΔHSC whole BM assayed for Trib1 expression relative to 18s, normalized to Trib1+/+ BM; representative of 3 experiments. (E) Representative plots of BM EoP, gated on live, lineage−CD34+ cells. Frequency of live cells (F) and absolute number (G) of BM EoP; n = 4 mice per group, representative of 2 experiments. *P = .0131; **P = .0013; ****P < .0001; unpaired Student t test. Frequencies and error bars are mean ± standard error of the mean (SEM) of live cells.
Our conditional Trib1 deletion model provided the opportunity to investigate the effect of Trib1 deletion on the dynamics of eosinophil differentiation and identity, which was not previously explored. Analysis of different myeloid developmental subsets in Trib1ΔHSC mice showed that the CMP and GMP populations were unperturbed (supplemental Figure 2J-L); whereas the EoP population was significantly decreased (Figure 1E-G). These data not only show that Trib1 expression is induced following GMP differentiation, but also that this increased expression functionally impacts eosinophil commitment in an early progenitor.
Trib1ΔHSC mice expand a population of Ly6G+ eosinophils in the BM
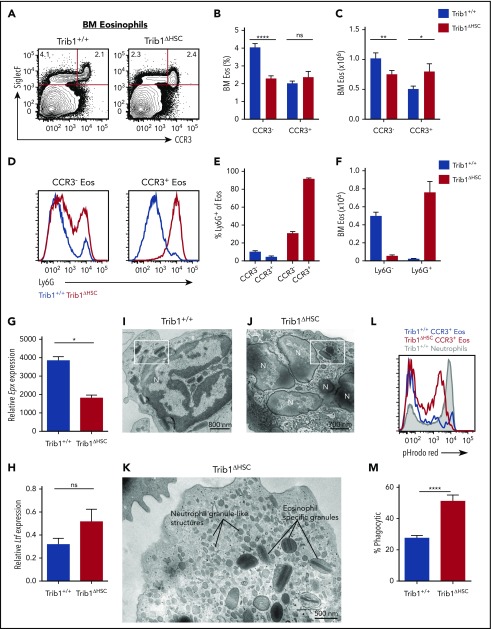
Given that the EoP defect occurred with Trib1 loss, we next examined terminal eosinophil maturation. As eosinophils mature in the BM, they gain expression of CCR3.22 Both CCR3− and CCR3+ eosinophils were present in the BM of Trib1ΔHSC mice (Figure 2A-C), and the number of CCR3+ eosinophils was increased in Trib1ΔHSC BM, accompanied by a decrease in the frequency and number of CCR3− eosinophils (Figure 2A-C). Strikingly, nearly all CCR3+ eosinophils in Trib1ΔHSC BM expressed surface Ly6G, a neutrophil-specific antigen23 (Figure 2D-E). A higher frequency of CCR3+ cells expressed Ly6G compared with CCR3− cells (Figure 2D-E). The Ly6G+SiglecF+CCR3+ cells found in Trib1ΔHSC mice were similar in number to Ly6G− eosinophils in Trib1+/+ BM (Figure 2F). Trib1ΔHSCLy6G+ eosinophils had reduced expression of the eosinophil granule protein gene eosinophil peroxidase (Epx; Figure 2G), but maintained high side scatter, as well as surface expression of Ly6C, F4/80, and IL-5Rα (CD125) (supplemental Figure 3A-B). Additionally, CCR3 and CXCR2 expression were unchanged (supplemental Figure 3C), whereas CXCR4 expression was increased (supplemental Figure 3C-D). In contrast to Trib1+/+CCR3+ eosinophils, Trib1ΔHSC eosinophils trended toward increased expression of the neutrophil secondary granule protein gene lactoferrin (Ltf; Figure 2H), and had hypersegmented nuclear architecture (supplemental Figure 3E-F). These cells were also present in chimeras engrafted with Trib1+/+ and Trib1ΔHSC BM (supplemental Figure 3G), establishing the cell-intrinsic nature of this population.
Figure 2.
Ly6G+ eosinophils are expanded in Trib1-deficient BM. (A) Representative plots of eosinophils in the BM from Trib1+/+ and Trib1ΔHSC mice, gated on live, CD11b+ cells. Frequency of live cells (B) and absolute number of eosinophils (C) in the BM from Trib1+/+ and Trib1ΔHSC mice; n = 24 mice per group, pooled from 7 experiments. (D) Representative histogram of Ly6G expression by SiglecF+CCR3− (left) and SiglecF+CCR3+ (right) eosinophils, Trib1+/+ (blue), Trib1ΔHSC (red). (E) Percentage of CCR3− and CCR3+ eosinophils expressing Ly6G. (F) Absolute number of BM CCR3+ eosinophils by Ly6G expression; n = 11-12 mice per group pooled from 4 experiments. (G) Epx and (H) Ltf expression in sorted BM CCR3+ eosinophils (SiglecF+CCR3+F4/80+CD11b+) relative to 18s and normalized to Trib1+/+ neutrophils; n = 2 mice per group, representative of 4 experiments. *P < .0232; **P < .0084; ****P < .0001; unpaired Student t test. (I-J) Representative electron micrographs of sorted BM CCR3+ eosinophils show typical eosinophil-specific granules (boxed areas in panels I and J) with a centrally located crystalloid electron dense core in both Trib1+/+ (I) and Trib1ΔHSC (J) eosinophils. (K) The cytoplasm of Trib1-deficient eosinophils also contains numerous round and smaller structures similar to neutrophil-specific granules. Representative of 2 experiments with a total of 75 electron micrographs evaluated. (L) Phagocytosis assay using pHrodo red-labeled Escherichia coli bioparticles with whole BM gated on Trib1+/+ neutrophils (filled gray), Trib1+/+ CCR3+ eosinophils (blue), and Trib1ΔHSC CCR3+ eosinophils (red). (M) Quantification of pHrodo red expression by CCR3+ BM eosinophils; n = 7 mice per group, pooled from 3 experiments. *P < .0232; **P < .0084; ****P < .0001; unpaired Student t test. Frequencies and error bars are mean ± SEM of live cells. N, nucleus; ns, not significant.
For a detailed morphological characterization of Trib1ΔHSC eosinophils, cells were prepared for conventional transmission electron microscopy.24 Both Trib1+/+ and Trib1ΔHSC eosinophils showed a typical cytoplasmic population of large, specific granules with a crystalloid electron-dense core and an outer electron-lucent matrix, delimited by a membrane (Figure 2I-J). This unique granule morphology is specific to eosinophils.25 The cytoplasm of Trib1ΔHSC eosinophils also showed numerous smaller, less dense, round or elongated structures, which resembled neutrophil-specific granules (Figure 2K); thus, Ly6G+ eosinophils from Trib1ΔHSC mice contain granules typical of both eosinophils and neutrophils. Consistent with these neutrophil-like characteristics, Trib1ΔHSC eosinophils were more phagocytic than their WT counterparts (Figure 2L-M). Together, these data suggest that Trib1 controls terminal eosinophil identity by repressing aspects of the neutrophil program.
Deletion of Trib1 following eosinophil lineage commitment results in a stable population of BM eosinophils that does not transition to other lineages
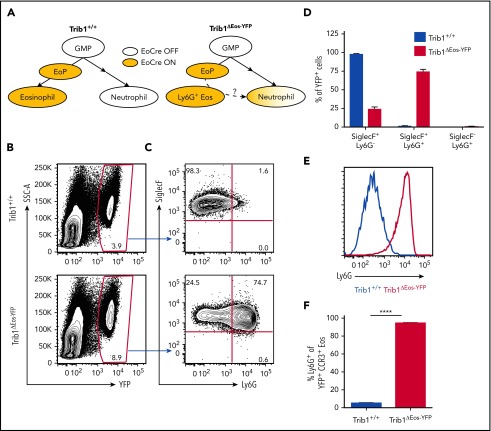
We observed an expansion of Ly6G+ eosinophils with characteristics of both eosinophils and neutrophils in the BM of Trib1ΔHSC mice, suggesting that these cells may be a granulocyte lineage intermediate that is capable of transitioning to either the neutrophil or eosinophil lineage. To test their developmental potential, we used a fate tracking strategy to follow the effects of Trib1 loss after eosinophil lineage commitment. Using mice expressing Cre under the control of the Epx promoter (EoCre),17 we generated mice lacking Trib1 in EoPs and eosinophils (Trib1ΔEos). We verified Trib1 deletion in BM-derived eosinophils from Trib1ΔEos mice compared with mice expressing EoCre alone (Trib1+/+; supplemental Figure 4A). We next crossed Trib1ΔEos mice to a YFP reporter mouse,26 resulting in YFP expression in EoCre-expressing cells (Trib1ΔEos-YFP; Figure 3A). Nearly all YFP-expressing Trib1ΔEos-YFP BM cells were SiglecF+ eosinophils, the majority of which expressed Ly6G, whereas YFP+ neutrophils (SiglecF−Ly6G+) or other cells were absent (Figure 3B-F). Some eosinophils from Trib1ΔEos-YFP BM had lower SiglecF expression, but resembled eosinophils by side scatter and F4/80 expression (data not shown). These data confirm that Trib1-deficient Ly6G+ eosinophils develop from eosinophil-committed cells and do not transition to other lineages.
Figure 3.
Trib1-deficient Ly6G+ eosinophils are a stable population that does not transition to other lineages. (A) Model of fate-tracking EoCre-mediated deletion of Trib1, and hypothesis of a full transition of Ly6G+ eosinophils from Trib1ΔEos BM to neutrophils. (B) Representative plots of BM from Trib1+/+ or Trib1ΔEos-YFP mice, gated on live cells. (C) Representative plots of YFP+ BM cells gated on live, YFP+ cells. (D) Distribution of YFP+ cells by SiglecF and Ly6G expression, as a fraction of YFP+ BM cells; n = 3 mice per group, representative of 4 experiments. (E) Representative histogram of Ly6G expression on YFP+SiglecF+CCR3+ cells, Trib1+/+ (blue), Trib1ΔEos-YFP (red). (F) Frequency of Ly6G expression on YFP+SiglecF+CCR3+ cells; n = 11 mice per group, pooled from 4 experiments. ****P < .0001; unpaired Student t test. Frequencies and error bars are mean ± SEM of live cells. SSC-A, side scatter area.
To determine when in development Trib1 modulates eosinophil identity, we further characterized the Trib1ΔEos mice. In contrast to Trib1ΔHSC mice, we observed a much smaller reduction in EoP numbers in Trib1ΔEos BM (Figure 4A-C). Additionally, there was an expansion of CCR3+ eosinophils in Trib1ΔEos BM (Figure 4D-F), however, few eosinophils were detected in the periphery (supplemental Figure 4B-F). The majority of Trib1ΔEos BM eosinophils expressed Ly6G (Figure 4G-H), similar to Trib1ΔHSC mice. In a notable difference from the Trib1ΔHSC mice, which show a neutrophil expansion (supplemental Figure 2A-I), the frequency and number of neutrophils were unchanged in the spleen, blood, and BM of the Trib1ΔEos mice (Figure 4I-K; supplemental Figure 4B-F). Together, these findings reveal that Trib1 regulates 2 distinct stages of eosinophil development: lineage commitment and terminal differentiation.
Figure 4.
Eosinophil-specific deletion of Trib1 results in Ly6G+ eosinophil development with improved preservation of EoPs. (A) Representative plots of BM EoP, gated on live, lineage−CD34+ cells from Trib1+/+ and Trib1ΔEos mice. Frequency of live cells (B) and absolute numbers (C) of BM EoP from Trib1+/+ and Trib1ΔEos mice; n = 6 mice per group, pooled from 2 experiments. (D) Representative plots of BM eosinophils from Trib1+/+ and Trib1ΔEos mice gated on live, CD11b+ cells. Frequency of live cells (E) and absolute numbers (F) of BM eosinophils by CCR3 expression. (G) Representative histogram of Ly6G expression, gated on live, CD11b+SiglecF+CCR3+, Trib1+/+ (blue), Trib1ΔHSC (red). (H) Frequency of SiglecF+CCR3+ cells expressing Ly6G; n = 11 mice per group, pooled from 4 experiments. (I) Representative plots of BM neutrophils from Trib1+/+ and Trib1ΔEos mice, gated on live, SiglecF− cells. (J) Frequency of live cells and (K) absolute numbers of BM neutrophils; n = 11 mice per group, pooled from 4 experiments. *P = .0289; ****P < .0001; unpaired Student t test. Frequencies and error bars are mean ± SEM of live cells.
Trib1 represses the neutrophil program in developing eosinophils ex vivo
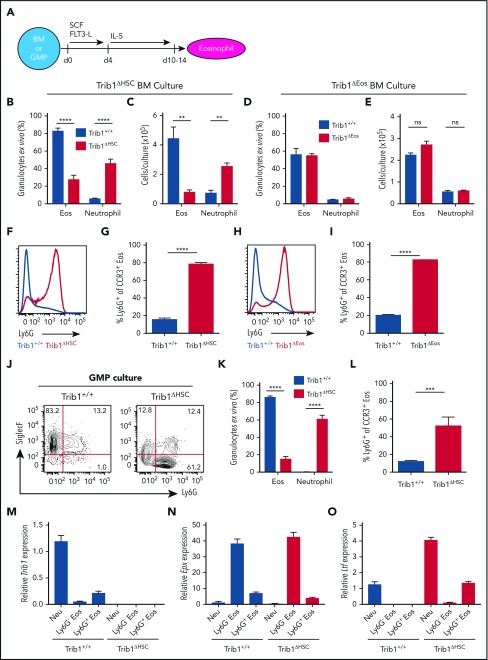
The cytokine IL-5 is sufficient to drive eosinophil development ex vivo.27,28 To assess the ability of exogenous IL-5 to restore normal eosinophil development from Trib1-null progenitors, we used a previously described ex vivo culture system18 (Figure 5A). After culture with IL-5, Trib1+/+ BM yielded a large population of eosinophils and, consistent with previous data, these cultures also yielded ∼20% Ly6G+ eosinophils.29 Trib1ΔHSC BM yielded far fewer eosinophils than Trib1+/+ BM at day 10 of culture (Figure 5B-C; supplemental Figure 5A); by day 14 of culture, eosinophils were nearly absent (data not shown). In contrast, cultures of Trib1ΔEos BM, in which Trib1 is deleted post–lineage commitment, showed no difference in eosinophil output compared with Trib1+/+ cells (Figure 5D-E; supplemental Figure 5B). Regardless of deletion timing, the majority of eosinophils produced by either Trib1ΔHSC or Trib1ΔEos BM were Ly6G+ (Figure 5F-5I; supplemental Figure 5A-B). In addition, neutrophils were the predominant population in the Trib1ΔHSC cultures (Figure 5B-C; supplemental Figure 5A), but were not increased in the Trib1ΔEos BM cultures compared with Trib1+/+ cultures (Figure 5D-E; supplemental Figure 5B). To determine whether lineage-restricted progenitors gave rise to Ly6G+ eosinophils, we sorted and cultured GMPs from Trib1+/+ and Trib1ΔHSC mice. Trib1 expression increased from undetectable levels to detectable levels by day 4 of culture, and continued to increase post–IL-5 addition (supplemental Figure 5C). After 10 days of culture, most of the eosinophils generated from the Trib1+/+ GMPs were Ly6G−, whereas the relative frequency of Ly6G+ eosinophils increased in the Trib1ΔHSC GMP cultures (Figure 5J-K). The enrichment was especially prominent in the more mature CCR3+ eosinophil population (Figure 5L), similar to both the in vivo (Figure 2D-E) and whole BM culture data (Figure 5F-I). Moreover, neutrophils were the dominant population in the Trib1ΔHSC GMP cultures, but nearly absent in the Trib1+/+ cultures (Figure 5J-K). Together, these data further reinforce the dual role of Trib1 in granulocyte development. Trib1 acts prior to eosinophil lineage commitment to increase eosinophilic potential, whereas Trib1 acts both before or following lineage commitment to repress the expansion of Ly6G+ eosinophils.
Figure 5.
Trib1 represses the neutrophil (Neu) program ex vivo. (A) Schematic of ex vivo eosinophil differentiation assay. Frequency of live cells (B) and day 10 cell output (C) using Trib1+/+ or Trib1ΔHSC BM. Frequency: n = 5 mice per group, pooled from 3 experiments; cell output: n = 3 wells per group, representative of 4 experiments. Frequency of live (D) cells and day 10 cell output (E) using Trib1+/+ or Trib1ΔEos BM. Frequency: n = 3 mice per group, representative of 6 experiments; cell output: n = 3 wells per group, representative of 5 experiments. (F) Representative histogram of Ly6G expression on SiglecF+CCR3+ eosinophils from Trib1+/+ (blue) or Trib1ΔHSC (red) IL-5 cultures. (G) Frequency of Ly6G expression on SiglecF+CCR3+ cells at day 10; n = 3 mice per group, representative of 4 experiments. (H) Representative histogram of Ly6G expression on SiglecF+CCR3+ eosinophils from Trib1+/+ (blue) or Trib1ΔEos (red) IL-5 cultures. (I) Frequency of Ly6G expression on SiglecF+CCR3+ cells; n = 3 mice per group, representative of 6 experiments. Eosinophils gated CD11b+SiglecF+CCR3+; neutrophils gated CD11b+Ly6G+SiglecF−. (J) Representative plots of day 10 IL-5 cultures of sorted GMPs from Trib1+/+ and Trib1ΔHSC mice, gated on live, CD11b+ cells. (K) Quantification of Ly6G expression on SiglecF+CCR3+ cells at day 10 of IL-5 GMP culture; n = 3-5 wells per genotype, representative of 2 experiments. (L) Quantification of granulocyte output at day 10 IL-5 culture of sorted GMPs; n = 3-5 wells per genotype, representative of 2 experiments. qPCR analysis of sorted neutrophils (CD11b+Ly6G+SiglecF−), and Ly6G− and Ly6G+ eosinophils (CD11b+SiglecF+CCR3+) from day 10 IL-5 culture of Trib1+/+ and Trib1ΔHSC BM for Trib1 (M), Epx (N), and Ltf (O), relative to 18s, normalized to Trib1+/+ neutrophils; n = 3, representative of 2 experiments. **P < .0084; ***P = .0002; ****P < .0001; unpaired Student t test. Frequencies and error bars are mean ± SEM of live cells. SCF, stem cell factor.
We next sorted eosinophils and neutrophils from day 10 IL-5 cultures of Trib1+/+ and Trib1ΔHSC BM to measure differences in gene expression. Trib1 expression was highest in Trib1+/+ neutrophils and absent in Trib1ΔHSC cells (Figure 5M). Epx expression was highest in Ly6G− eosinophils derived from either Trib1+/+ or Trib1ΔHSC BM and lower in Ly6G+ eosinophils derived from either Trib1+/+ or Trib1ΔHSC BM (Figure 5N). Ltf was expressed in neutrophils but not eosinophils (Ly6G+ or Ly6G−) from Trib1+/+ BM. In contrast, Ltf expression was increased in both eosinophils and neutrophils derived from Trib1ΔHSC BM (Figure 5O). These data suggest that Trib1 inhibits neutrophil gene expression in order to promote eosinophil development. In the absence of Trib1, developing progenitors are unable to extinguish or are biased toward the neutrophil program, beginning at or prior to the GMP stage.
To assay the functional capability of cells derived from the IL-5 culture system, we performed transwell assays to measure cell migration to eotaxin (CCL11) or KC (CXCL1), which attracts eosinophils30,31 or neutrophils,32,33 respectively (supplemental Figure 5D,G). Eosinophils derived from cultured Trib1+/+ BM migrated to eotaxin with greater efficiency than to KC, as expected (supplemental Figure 5E-F). Ly6G+ eosinophils derived from Trib1ΔHSC- and Trib1ΔEos-cultured cells also migrated to eotaxin, albeit with reduced efficiency, and minimally migrated to KC (supplemental Figure 5E-F). The Trib1ΔHSC-derived neutrophils also migrated to KC (supplemental Figure 5H), indicating that these cells functionally resemble bona fide neutrophils. As few neutrophils were produced from Trib1+/+ IL-5 cultures, we were unable to analyze their migration. Thus, both the eosinophils and neutrophils derived from Trib1-deficient cultures migrated to their respective attractants/chemokines, indicating that these are functionally competent granulocytes.
Trib1-deficient eosinophils show impaired mobilization in response to lung inflammation
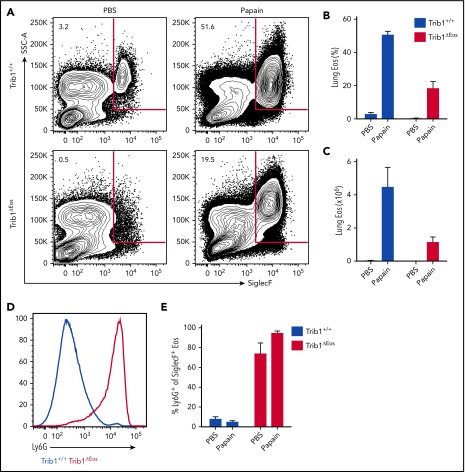
The Ly6G+ eosinophils present in Trib1ΔHSC and Trib1ΔEos mice are predominantly restricted to the BM and do not transition to other lineages (Figures 3 and 4). To determine whether these cells can be mobilized, Trib1+/+ and Trib1ΔEos mice were treated intranasally with papain, a protease allergen that stimulates eosinophil recruitment to the lung.34-36 Papain-treated Trib1ΔEos mice mobilized eosinophils to the lung and distal sites; however, the magnitude of mobilization was decreased (Figure 6A-C; supplemental Figure 6). Nearly all of the eosinophils recruited to the lungs of Trib1ΔEos mice post–papain treatment were Ly6G+ (Figure 6D-E). These data show that the Ly6G+ eosinophils generated by Trib1-deficient BM are bona fide eosinophils, as they are mobilized in response to type 2 inflammation; however, Trib1 is required for optimal migration.
Figure 6.
Partial rescue of Trib1-deficient eosinophil mobilization in response to type 2 lung inflammation. Mice were treated for 5 days with intranasal PBS or 30 μg of papain. (A) Representative plots of lung leukocytes gated on live, CD45+CD11bhiCD11c−. Frequency of live cells (B) and absolute number (C) of lung eosinophils gated on live, CD45+CD11bhiCD11c−SiglecF+ cells. (D) Representative histogram of Ly6G expression by lung eosinophils gated live, CD45+CD11bhiCD11c−SiglecF+, Trib1+/+ (blue), Trib1ΔEos (red). (E) Fraction of lung eosinophils expressing Ly6G; n = 3 mice per group, representative of 2 experiments. Frequencies and error bars are mean ± SEM of live cells.
Trib1 modulates granulocyte identity in part through regulation of C/EBPα
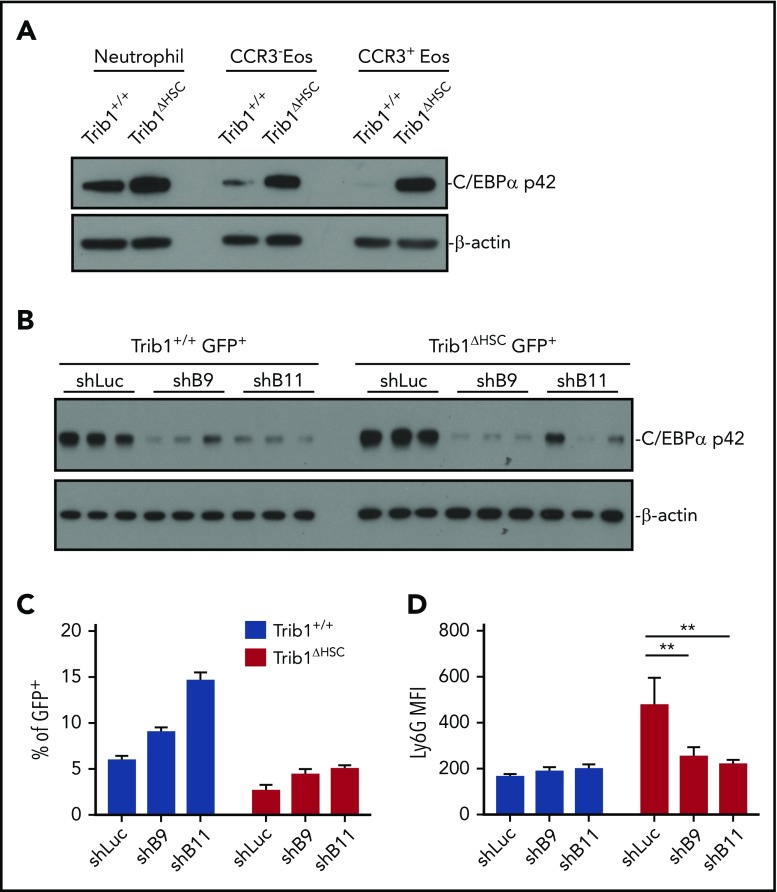
C/EBPα is a key regulator of granulocyte development19,37-39 and is a target of Trib1-mediated protein degradation.13,40,41 We hypothesized that Trib1 modulates C/EBPα protein levels in order to regulate granulocyte development. We measured the expression of C/EBPα protein in sorted BM neutrophils and CCR3− and CCR3+ eosinophils from Trib1+/+ and Trib1ΔHSC mice. Strikingly, C/EBPα p42 protein expression was increased in all 3 populations (Figure 7A). We also measured expression of GATA1 and C/EBPε, 2 key transcription factors in eosinophil development,42-46 in CCR3+ eosinophils from Trib1+/+ or Trib1ΔHSC mice. GATA1 expression was not affected, whereas C/EBPε, a target of C/EPBα during granulopoiesis,19 was increased in the absence of Trib1 (supplemental Figure 7A).
Figure 7.
Trib1 regulates C/EPBα levels to control eosinophil identity. (A) C/EBPα immunoblot analysis of sorted neutrophils (CD11b+Ly6G+SiglecF−F4/80−), and CCR3− and CCR3+ eosinophils (CD11b+SiglecF+F4/80+) from the BM of Trib1+/+ and Trib1ΔHSC mice; β-actin is the loading control. Representative of 2 experiments. (B) C/EBPα immunoblot analysis of sorted GFP+ BM cells from mice transplanted with shRNA-transduced BM cells, 9 weeks after transplantation with β-actin as a loading control. shLuc control targets firefly luciferase; shB9 and shB11 target Cebpa. Sorted GFP+ cells from 3 representative mice are shown. Representative of 2 experiments. (C) Percentage of GFP+ cells expressing control or C/EBPα shRNAs B9 or B11 cells expressing SiglecF with surface expression of SiglecF. Representative of 2 experiments. (D) Mean fluorescence intensity (MFI) of Ly6G expression on BM eosinophils (GFP+CD11b+SiglecF+) expressing either control or C/EBPα shRNAs B9 or B11; n = 4-5 mice per group, representative of 2 experiments. **P < .0052, 2-way analysis of variance with multiple comparisons. Frequencies and error bars are mean ± SEM of GFP+ cells.
To determine whether C/EBPα upregulation was responsible for altered granulocyte development in the absence of Trib1, we knocked down C/EBPα in hematopoietic progenitors from Trib1+/+ or Trib1ΔHSC mice using 2 previously validated shRNAs,19 transplanted the cells into recipients, and assessed BM eosinophils by flow cytometry after 9 weeks (Figure 7B-D; supplemental Figure 7B). We also sorted GFP+ BM cells to assess knockdown efficiency (Figure 7B). We observed decreased Ly6G expression on SiglecF+ eosinophils in the BM of recipients reconstituted with Trib1ΔHSC progenitors transduced with C/EBPα shRNAs B9 and B11 (Figure 7D). Together, these findings indicate that Trib1 modulates C/EBPα levels to promote eosinophil cell identity.
Discussion
Differentiation of hematopoietic cells from multipotent progenitors to terminally differentiated cells requires early progenitors to establish a unique cellular identity through activating and repressing specific gene sets. Previous work using germline Trib1 knockout mice demonstrated that eosinophil numbers were suppressed and neutrophil numbers were enhanced in the absence of Trib1,13 yet at what developmental stage Trib1 functioned was unknown. We found that Trib1 is expressed in EoPs, but is undetectable in CMPs and GMPs, with similar dynamics in human eosinophil development, suggesting a role for Trib1 at the earliest stages of eosinophil development. We show that Trib1 acts at 2 distinct points during eosinophil development, as revealed through stage-specific deletion studies. Trib1 loss in HSCs leads to a selective decrease in EoPs and a concomitant increase in neutrophils, suggesting that blocking early eosinophil commitment shunts myeloid progenitors toward neutrophils. In contrast, Trib1 loss in EoPs using EoCre does not impact eosinophil fidelity.
We observe a marked expansion of a Ly6G+ eosinophil population in Trib1-deficient BM, in contrast to WT mice, where Ly6G is primarily restricted to neutrophils.23 Ly6G+ eosinophils express aspects of the neutrophil program (Figure 2D-M), which is partly mediated by C/EBPα (Figure 7C-D); this phenotype is also observed in eosinophils generated ex vivo from Trib1-deficient progenitors. IL-5 alone is not sufficient to restore normal eosinophil differentiation in the absence of Trib1, but supports the generation of Ly6G+ eosinophils from both the Trib1ΔHSC and Trib1ΔEos cells ex vivo (Figure 5F-I). Additionally, IL-5 cannot drive eosinophil expansion of Trib1ΔHSC cells ex vivo, possibly due to a reduction in EoPs. This is in contrast to the slight expansion of CCR3+ eosinophils we observe in vivo in Trib1ΔHSC BM (Figure 2C). This suggests that there are additional factors that support this population in vivo and/or their altered CXCR4 expression (supplemental Figure 3C-D) contributes to BM retention. Nevertheless, these data suggest that Trib1 is required to both specify the eosinophil lineage and guide its proper differentiation.
Ly6G+ eosinophils are also present in WT BM, albeit at a much lower frequency, and are predominantly found among CCR3− eosinophils (Figure 2D-F). In contrast, in Trib1-deficient mice, Ly6G expression tracked with CCR3 acquisition, with more Trib1-deficient CCR3+ eosinophils expressing Ly6G compared with CCR3− eosinophils (Figure 2D-E). Additionally, a small population of Ly6G+ eosinophils was identified in WT murine lungs following fungal allergen challenge, as well as in ex vivo eosinophil cultures of WT BM.29 Our studies provide new insights into the characteristics and maintenance of this population by Trib1. We hypothesize that without Trib1, aspects of the neutrophil gene program are active, which stabilizes the existing Ly6G+ CCR3− and/or CCR3+ eosinophil populations. The Trib1-deficient Ly6G+ eosinophils phenotypically and functionally resemble eosinophils, yet also fail to repress neutrophilic characteristics, including expression of Ly6G, increased phagocytosis, and neutrophil-type granules. Increased phagocytosis and nuclear hypersegmentation are observed in certain activated eosinophil subsets,47-49 possibly suggesting a functional relevance for Trib1 in these cells.
Our Trib1 knockout studies show that there are multiple alterations in granulocyte output from Trib1-deficient progenitors depending on the timing of deletion, suggesting that Trib1 exerts important effects at different stages of development. Our data show that GMPs lacking Trib1 produce both eosinophils and neutrophils when cultured with IL-5, whereas WT GMPs primarily generate eosinophils (Figure 5J-K), suggesting that Trib1 modulates lineage priming of myeloid progenitors. We do not observe a neutrophil expansion in vivo when Trib1 is specifically deleted in eosinophils, further suggesting that Trib1 regulates early lineage commitment independently of terminal identity. A recent report identified a SiglecF+IL-5Rα− progenitor population that yielded neutrophils.50 It is unknown whether Trib1 modulates this population; however, our data suggest that Trib1 may be required to balance the neutrophil and eosinophil lineage programs in these developing progenitors. Although we do not observe Trib1 expression in the bulk GMP population, recent work identified Trib1 expression in individual CMP and GMP cells.51 Furthermore, recent reports show that individual myeloid progenitors may be “precommitted” to a specific lineage.51-53 Future studies are required to determine whether Trib1 influences eosinophil lineage priming in CMPs or GMPs, prior to the EoP stage, and to delineate the precise origin of these cells.
As reported, cells lacking Trib1 have increased amounts of C/EBPα13 (Figure 7A). The precise regulation of C/EBPα is critical in myeloid development,19,37-39,54,55 and neutrophils express higher levels of C/EBPα than eosinophils (Figure 7A). Furthermore, knockdown of C/EBPα partly normalized eosinophil differentiation in the absence of Trib1 (Figure 7B-D), suggesting that Trib1 controls eosinophil identity by regulating C/EPBα levels. Our observation of increased C/EBPε expression in Trib1-deficient eosinophils (supplemental Figure 7A) is consistent with the ability of C/EPBα to upregulate C/EBPε during granulopoiesis, possibly altering granule development.19 These findings suggest that Trib1 normally represses the neutrophil gene program in developing eosinophils, partly by decreasing C/EBPα protein expression, in order to promote eosinophil development. Importantly, our data suggest that precise regulation of C/EPBα by Trib1 is required for proper granulocyte development.
Although our papain challenge and transwell migration studies indicate that functional eosinophils are present in Trib1-deficient mice, eosinophil migration was altered (Figure 6; supplemental Figure 5E-F). Of particular interest is our finding that CXCR4 is increased on Trib1-deficient BM eosinophils (supplemental Figure 3C-D). We hypothesize that increased CXCR4 expression on Trib1-deficient BM eosinophils limits egress of these cells from the BM.56,57 This expands BM eosinophils (Figures 2A-C and 4D-F), reduces peripheral eosinophil numbers (supplemental Figure 2D-I) and decreases migration (Figure 6). Previous studies in mutant mice defined defects in eosinophil production due to alterations in transcription factors or granule proteins, or changes in eosinophil recruitment due to loss of CCR3 or eotaxin.44,58-64 Our current study shows that Trib1 also influences eosinophil production and recruitment. How Trib1 signals integrate with these other pathways remains to be defined.
In summary, our studies reveal Trib1 as a key regulator of eosinophil development and homeostasis. We find that Trib1 plays roles both at the earliest stages of eosinophil commitment, as well as in eosinophil identity and function, by influencing the response to cytokine signals and precisely tuning levels of C/EBPα. These findings clarify long-standing questions in granulopoiesis regarding regulation of cell lineage choice and identity, and provide a path forward in the study of eosinophil and neutrophil development.
Supplementary Material
The online version of this article contains a data supplement.
Acknowledgments
The authors thank the University of Pennsylvania Flow Cytometry and Cell Sorting Core facility for assistance with cell sorting; James Lee (Mayo Clinic) for the gift of the EoCre mice; Daniel Rader (University of Pennsylvania) for the gift of the cTrib1 mice; Walter Mowel, Dana Bellissimo, Maximilian Wengyn, Jorge Gutierrez, Roshni Kailar, Anne DeHart, and Jake Soifer for technical assistance; and the entire Pear laboratory, Jorge Henao-Mejia, and G. Scott Worthen for thoughtful discussions.
This work was supported by the National Institutes of Health, National Institute of Allergy and Infectious Diseases grant R01AI047833 (W.S.P.), National Heart, Lung, and Blood Institute grants T32HL743937 and F30HL136127 (E.A.M.), and National Cancer Institute grants F31CA189661 (K.S.R.), T32CA009140 (K.S.R.), and T32CA009140 (S.J.S.); American Cancer Society grant PF-15-065-01-TBG (S.J.S.); Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq; Brazil-311083/2014-5) and Fundação de Amparo a Pesquisa do Estado de Minas Gerais (Brazil-CBB-APQ-03647-16) (R.C.N.M.); and grants from the Samuel Waxman Cancer Research Foundation and Alex’s Lemonade Stand Foundation (W.S.P.). The University of Pennsylvania Flow Cytometry and Cell Sorting Core facility was supported by National Institutes of Health, National Cancer Institute grant P30-CA016520.
Footnotes
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Authorship
Contribution: E.A.M. and S.J.S. conceived, designed, and performed experiments and wrote the manuscript; K.S.R. and L.X. performed experiments; G.B.W. scored cytospins; R.C.N.M. imaged and analyzed electron microscopy samples; and W.S.P. conceived and designed experiments and wrote the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Warren S. Pear, Perelman School of Medicine, University of Pennsylvania, 556 BRB II/III, 421 Curie Blvd, Philadelphia, PA 19104; e-mail: wpear@pennmedicine.upenn.edu.
REFERENCES
- 1.Rosenberg HF, Dyer KD, Foster PS. Eosinophils: changing perspectives in health and disease. Nat Rev Immunol. 2013;13(1):9-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Amulic B, Cazalet C, Hayes GL, Metzler KD, Zychlinsky A. Neutrophil function: from mechanisms to disease. Annu Rev Immunol. 2012;30(1):459-489. [DOI] [PubMed] [Google Scholar]
- 3.Akashi K, Traver D, Miyamoto T, Weissman IL. A clonogenic common myeloid progenitor that gives rise to all myeloid lineages. Nature. 2000;404(6774):193-197. [DOI] [PubMed] [Google Scholar]
- 4.Iwasaki H, Mizuno S, Mayfield R, et al. . Identification of eosinophil lineage-committed progenitors in the murine bone marrow. J Exp Med. 2005;201(12):1891-1897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Grosshans J, Wieschaus E. A genetic link between morphogenesis and cell division during formation of the ventral furrow in Drosophila. Cell. 2000;101(5):523-531. [DOI] [PubMed] [Google Scholar]
- 6.Mata J, Curado S, Ephrussi A, Rørth P. Tribbles coordinates mitosis and morphogenesis in Drosophila by regulating string/CDC25 proteolysis. Cell. 2000;101(5):511-522. [DOI] [PubMed] [Google Scholar]
- 7.Rørth P, Szabo K, Texido G. The level of C/EBP protein is critical for cell migration during Drosophila oogenesis and is tightly controlled by regulated degradation. Mol Cell. 2000;6(1):23-30. [DOI] [PubMed] [Google Scholar]
- 8.Stein SJ, Mack EA, Rome KS, Pear WS. Tribbles in normal and malignant haematopoiesis. Biochem Soc Trans. 2015;43(5):1112-1115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Eyers PA, Keeshan K, Kannan N. Tribbles in the 21st century: the evolving roles of tribbles pseudokinases in biology and disease. Trends Cell Biol. 2017;27(4):284-298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lohan F, Keeshan K. The functionally diverse roles of tribbles. Biochem Soc Trans. 2013;41(4):1096-1100. [DOI] [PubMed] [Google Scholar]
- 11.Johnston J, Basatvat S, Ilyas Z, Francis S, Kiss-Toth E. Tribbles in inflammation. Biochem Soc Trans. 2015;43(5):1069-1074. [DOI] [PubMed] [Google Scholar]
- 12.Nakamura T. The role of Trib1 in myeloid leukaemogenesis and differentiation. Biochem Soc Trans. 2015;43(5):1104-1107. [DOI] [PubMed] [Google Scholar]
- 13.Satoh T, Kidoya H, Naito H, et al. . Critical role of Trib1 in differentiation of tissue-resident M2-like macrophages. Nature. 2013;495(7442):524-528. [DOI] [PubMed] [Google Scholar]
- 14.Stein SJ, Mack EA, Rome KS, et al. . Trib2 suppresses tumor initiation in Notch-driven T-ALL. PLoS One. 2016;11(5):e0155408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bauer RC, Sasaki M, Cohen DM, et al. . Tribbles-1 regulates hepatic lipogenesis through posttranscriptional regulation of C/EBPα. J Clin Invest. 2015;125(10):3809-3818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Georgiades P, Ogilvy S, Duval H, et al. . VavCre transgenic mice: a tool for mutagenesis in hematopoietic and endothelial lineages. Genesis. 2002;34(4):251-256. [DOI] [PubMed] [Google Scholar]
- 17.Doyle AD, Jacobsen EA, Ochkur SI, et al. . Homologous recombination into the eosinophil peroxidase locus generates a strain of mice expressing Cre recombinase exclusively in eosinophils. J Leukoc Biol. 2013;94(1):17-24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Dyer KD, Moser JM, Czapiga M, Siegel SJ, Percopo CM, Rosenberg HF. Functionally competent eosinophils differentiated ex vivo in high purity from normal mouse bone marrow. J Immunol. 2008;181(6):4004-4009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ma O, Hong S, Guo H, Ghiaur G, Friedman AD. Granulopoiesis requires increased C/EBPα compared to monopoiesis, correlated with elevated Cebpa in immature G-CSF receptor versus M-CSF receptor expressing cells. PLoS One. 2014;9(4):e95784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Novershtern N, Subramanian A, Lawton LN, et al. . Densely interconnected transcriptional circuits control cell states in human hematopoiesis. Cell. 2011;144(2):296-309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Mori Y, Iwasaki H, Kohno K, et al. . Identification of the human eosinophil lineage-committed progenitor: revision of phenotypic definition of the human common myeloid progenitor. J Exp Med. 2009;206(1):183-193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Voehringer D, van Rooijen N, Locksley RM. Eosinophils develop in distinct stages and are recruited to peripheral sites by alternatively activated macrophages. J Leukoc Biol. 2007;81(6):1434-1444. [DOI] [PubMed] [Google Scholar]
- 23.Daley JM, Thomay AA, Connolly MD, Reichner JS, Albina JE. Use of Ly6G-specific monoclonal antibody to deplete neutrophils in mice. J Leukoc Biol. 2008;83(1):64-70. [DOI] [PubMed] [Google Scholar]
- 24.Melo RC, Perez SA, Spencer LA, Dvorak AM, Weller PF. Intragranular vesiculotubular compartments are involved in piecemeal degranulation by activated human eosinophils. Traffic. 2005;6(10):866-879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Melo RCN, Weller PF. Contemporary understanding of the secretory granules in human eosinophils. J Leukoc Biol. 2018;104(1):85-93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Srinivas S, Watanabe T, Lin CS, et al. . Cre reporter strains produced by targeted insertion of EYFP and ECFP into the ROSA26 locus. BMC Dev Biol. 2001;1(1):4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Yamaguchi Y, Suda T, Suda J, et al. . Purified interleukin 5 supports the terminal differentiation and proliferation of murine eosinophilic precursors. J Exp Med. 1988;167(1):43-56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Fulkerson PC, Schollaert KL, Bouffi C, Rothenberg ME. IL-5 triggers a cooperative cytokine network that promotes eosinophil precursor maturation. J Immunol. 2014;193(8):4043-4052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Percopo CM, Brenner TA, Ma M, et al. . SiglecF+Gr1hi eosinophils are a distinct subpopulation within the lungs of allergen-challenged mice. J Leukoc Biol. 2017;101(1):321-328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Rothenberg ME, Luster AD, Lilly CM, Drazen JM, Leder P. Constitutive and allergen-induced expression of eotaxin mRNA in the guinea pig lung. J Exp Med. 1995;181(3):1211-1216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Jose PJ, Griffiths-Johnson DA, Collins PD, et al. . Eotaxin: a potent eosinophil chemoattractant cytokine detected in a guinea pig model of allergic airways inflammation. J Exp Med. 1994;179(3):881-887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Moser B, Clark-Lewis I, Zwahlen R, Baggiolini M. Neutrophil-activating properties of the melanoma growth-stimulatory activity. J Exp Med. 1990;171(5):1797-1802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Bozic CR, Kolakowski LF Jr, Gerard NP, et al. . Expression and biologic characterization of the murine chemokine KC. J Immunol. 1995;154(11):6048-6057. [PubMed] [Google Scholar]
- 34.Novey HS, Marchioli LE, Sokol WN, Wells ID. Papain-induced asthma--physiological and immunological features. J Allergy Clin Immunol. 1979;63(2):98-103. [DOI] [PubMed] [Google Scholar]
- 35.Halim TY, Krauss RH, Sun AC, Takei F. Lung natural helper cells are a critical source of Th2 cell-type cytokines in protease allergen-induced airway inflammation. Immunity. 2012;36(3):451-463. [DOI] [PubMed] [Google Scholar]
- 36.Oboki K, Ohno T, Kajiwara N, et al. . IL-33 is a crucial amplifier of innate rather than acquired immunity. Proc Natl Acad Sci USA. 2010;107(43):18581-18586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Zhang DE, Zhang P, Wang ND, Hetherington CJ, Darlington GJ, Tenen DG. Absence of granulocyte colony-stimulating factor signaling and neutrophil development in CCAAT enhancer binding protein alpha-deficient mice. Proc Natl Acad Sci USA. 1997;94(2):569-574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Zhang P, Iwasaki-Arai J, Iwasaki H, et al. . Enhancement of hematopoietic stem cell repopulating capacity and self-renewal in the absence of the transcription factor C/EBP alpha. Immunity. 2004;21(6):853-863. [DOI] [PubMed] [Google Scholar]
- 39.Radomska HS, Huettner CS, Zhang P, Cheng T, Scadden DT, Tenen DG. CCAAT/enhancer binding protein alpha is a regulatory switch sufficient for induction of granulocytic development from bipotential myeloid progenitors. Mol Cell Biol. 1998;18(7):4301-4314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Dedhia PH, Keeshan K, Uljon S, et al. . Differential ability of Tribbles family members to promote degradation of C/EBPalpha and induce acute myelogenous leukemia. Blood. 2010;116(8):1321-1328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Murphy JM, Nakatani Y, Jamieson SA, Dai W, Lucet IS, Mace PD. Molecular mechanism of CCAAT-enhancer binding protein recruitment by the TRIB1 pseudokinase. Structure. 2015;23(11):2111-2121. [DOI] [PubMed] [Google Scholar]
- 42.Kulessa H, Frampton J, Graf T. GATA-1 reprograms avian myelomonocytic cell lines into eosinophils, thromboblasts, and erythroblasts. Genes Dev. 1995;9(10):1250-1262. [DOI] [PubMed] [Google Scholar]
- 43.Hirasawa R, Shimizu R, Takahashi S, et al. . Essential and instructive roles of GATA factors in eosinophil development. J Exp Med. 2002;195(11):1379-1386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Yu C, Cantor AB, Yang H, et al. . Targeted deletion of a high-affinity GATA-binding site in the GATA-1 promoter leads to selective loss of the eosinophil lineage in vivo. J Exp Med. 2002;195(11):1387-1395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Yamanaka R, Barlow C, Lekstrom-Himes J, et al. . Impaired granulopoiesis, myelodysplasia, and early lethality in CCAAT/enhancer binding protein epsilon-deficient mice. Proc Natl Acad Sci USA. 1997;94(24):13187-13192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Lekstrom-Himes JA, Dorman SE, Kopar P, Holland SM, Gallin JI. Neutrophil-specific granule deficiency results from a novel mutation with loss of function of the transcription factor CCAAT/enhancer binding protein epsilon. J Exp Med. 1999;189(11):1847-1852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hatano Y, Taniuchi S, Masuda M, et al. . Phagocytosis of heat-killed Staphylococcus aureus by eosinophils: comparison with neutrophils. APMIS. 2009;117(2):115-123. [DOI] [PubMed] [Google Scholar]
- 48.Mesnil C, Raulier S, Paulissen G, et al. . Lung-resident eosinophils represent a distinct regulatory eosinophil subset. J Clin Invest. 2016;126(9):3279-3295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Abdala Valencia H, Loffredo LF, Misharin AV, Berdnikovs S. Phenotypic plasticity and targeting of Siglec-F(high) CD11c(low) eosinophils to the airway in a murine model of asthma. Allergy. 2016;71(2):267-271. [DOI] [PubMed] [Google Scholar]
- 50.Bolden JE, Lucas EC, Zhou G, et al. . Identification of a Siglec-F+ granulocyte-macrophage progenitor. J Leukoc Biol. 2018;104(1):123-133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Olsson A, Venkatasubramanian M, Chaudhri VK, et al. . Single-cell analysis of mixed-lineage states leading to a binary cell fate choice. Nature. 2016;537(7622):698-702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Drissen R, Buza-Vidas N, Woll P, et al. . Distinct myeloid progenitor-differentiation pathways identified through single-cell RNA sequencing. Nat Immunol. 2016;17(6):666-676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Paul F, Arkin Y, Giladi A, et al. . Transcriptional heterogeneity and lineage commitment in myeloid progenitors [published correction appears in Cell. 2016;164(1-2):325]. Cell. 2015;163(7):1663-1677. [DOI] [PubMed] [Google Scholar]
- 54.Iwama A, Osawa M, Hirasawa R, et al. . Reciprocal roles for CCAAT/enhancer binding protein (C/EBP) and PU.1 transcription factors in Langerhans cell commitment. J Exp Med. 2002;195(5):547-558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Iwasaki H, Mizuno S, Arinobu Y, et al. . The order of expression of transcription factors directs hierarchical specification of hematopoietic lineages. Genes Dev. 2006;20(21):3010-3021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Martin C, Burdon PC, Bridger G, Gutierrez-Ramos JC, Williams TJ, Rankin SM. Chemokines acting via CXCR2 and CXCR4 control the release of neutrophils from the bone marrow and their return following senescence. Immunity. 2003;19(4):583-593. [DOI] [PubMed] [Google Scholar]
- 57.Devi S, Wang Y, Chew WK, et al. . Neutrophil mobilization via plerixafor-mediated CXCR4 inhibition arises from lung demargination and blockade of neutrophil homing to the bone marrow. J Exp Med. 2013;210(11):2321-2336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Lee JJ, Dimina D, Macias MP, et al. . Defining a link with asthma in mice congenitally deficient in eosinophils. Science. 2004;305(5691):1773-1776. [DOI] [PubMed] [Google Scholar]
- 59.Bettigole SE, Lis R, Adoro S, et al. . The transcription factor XBP1 is selectively required for eosinophil differentiation. Nat Immunol. 2015;16(8):829-837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Doyle AD, Jacobsen EA, Ochkur SI, et al. . Expression of the secondary granule proteins major basic protein 1 (MBP-1) and eosinophil peroxidase (EPX) is required for eosinophilopoiesis in mice. Blood. 2013;122(5):781-790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Rothenberg ME, MacLean JA, Pearlman E, Luster AD, Leder P. Targeted disruption of the chemokine eotaxin partially reduces antigen-induced tissue eosinophilia. J Exp Med. 1997;185(4):785-790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Pope SM, Fulkerson PC, Blanchard C, et al. . Identification of a cooperative mechanism involving interleukin-13 and eotaxin-2 in experimental allergic lung inflammation. J Biol Chem. 2005;280(14):13952-13961. [DOI] [PubMed] [Google Scholar]
- 63.Pope SM, Zimmermann N, Stringer KF, Karow ML, Rothenberg ME. The eotaxin chemokines and CCR3 are fundamental regulators of allergen-induced pulmonary eosinophilia. J Immunol. 2005;175(8):5341-5350. [DOI] [PubMed] [Google Scholar]
- 64.Humbles AA, Lu B, Friend DS, et al. . The murine CCR3 receptor regulates both the role of eosinophils and mast cells in allergen-induced airway inflammation and hyperresponsiveness. Proc Natl Acad Sci USA. 2002;99(3):1479-1484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Bagger FO, Sasivarevic D, Sohi SH, et al. . BloodSpot: a database of gene expression profiles and transcriptional programs for healthy and malignant haematopoiesis. Nucleic Acids Res. 2016;44(D1):D917-D924. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.