Abstract
Background
Papillary thyroid microcarcinoma (PTMC) generally is a cancer with excellent prognosis, but the term “cancer” sounds severe and harsh, which can elicit emotional and physical responses from patients. To eliminate the word “cancer,” the term noninvasive follicular thyroid neoplasm with papillary-like nuclear features (NIFTP) was introduced. However, not all PTMCs can be classified as NIFTP. Sometimes, very aggressive PTMC cases might be observed. Some authors suggest that one of the risk factors for poor prognosis is lymph node metastasis. The aim of the study was to evaluate some clinicopathological features of PTMC as the risk factors for lymph node metastasis.
Material and methods
We performed a retrospective chart review and selected 177 patients with PTMC. To analyze the cases with potentially aggressive behavior, we enrolled PTMC patients with lymph node metastases (pN1, central, and/or lateral) and evaluated some of their clinicopathological features.
Results
The logistic regression analysis results demonstrated significantly higher rates of multifocal or bilateral tumor occurrence in the PTMC patients with pN1 than in the patients with pN0 (P < 0.0001 for both). In addition, the occurrence of thyroid tumors with sizes above 0.5 cm was a significant risk factor for lymph node metastasis (P < 0.0001). The results of the ROC analyses showed that the presence of multifocal or bilateral tumors and tumor sizes above 0.5 cm were significant predictors of lymph node metastasis (P < 0.0001 for all).
Conclusions
Multifocal and bilateral PTMC tumors with diameters above 0.5 cm should be treated aggressively as “true cancer” and might benefit from lymph node dissection. Unifocal PTMC tumors with diameters equal to or below 0.5 cm may be treated less aggressively.
Keywords: Papillary thyroid microcarcinoma, Aggressiveness, Metastasis
Introduction
Papillary thyroid microcarcinoma (PTMC) is a thyroid cancer with small tumors that are 1.0 cm maximum in diameter [1]. The term “PTMC” was introduced for the first time by the World Health Organization (WHO) in 1989 [1]. The majority of PTMCs are clinically silent and sometimes might be found from histopathological examinations after a thyroidectomy performed due to benign thyroid diseases [2]. Additionally, PTMCs are found in autopsies of individuals who died of non-thyroid-related diseases. PTMC is also the most common type of malignant thyroid tumor with an increasing prevalence, but also with an excellent prognosis. Therefore, the term “carcinoma” is thought by many authors to be too severe and harsh since it can elicit emotional and physical responses from the patients [3, 4]. On the other hand, some very aggressive PTMC cases might be observed [5]. Choi et al. stated that no definite biological or clinical parameters currently exist to distinguish low-risk indolent PTMC from potentially aggressive PTMC [6]. According to some authors, one of the risk factors for aggressive PTMCs is lymph node metastasis [7–18]. These cases may benefit mostly from lymph node dissection. Sometimes, we diagnose palpable metastatic neck lymph nodes as the first sign of PTMC; these tumors are known as occult papillary thyroid microcarcinomas.
To promote not aggressive surgical approach and save patients’ psychological distress of cancer diagnosis, a new name for indolent PTMC such as noninvasive follicular thyroid neoplasm with papillary-like nuclear features (NIFTP) was introduced [19]. However, the term NIFTP in histological diagnosis is strictly stringent [20]. For example, the tumor has to present encapsulation, purely follicular architecture, the presence of nuclear features of papillary thyroid cancer (PTC), and absence of capsular and vascular invasions. They strictly formulated the inclusion and exclusion criteria because not every PTMC can be classified as NIFTP.
It is suggested that the increasing prevalence of PTMCs with excellent prognoses is caused by the increased use of ultrasound examinations of the thyroid. The management of these “clinically silent thyroid incidentalomas” is still controversial [3]. However, the most common subsequent clinical evaluation is ultrasound-guided fine needle aspiration biopsy (UG-FNAB). If the diagnostic result of this procedure is estimated as category V or VI according to The Bethesda System for Reporting Thyroid Cytopathology (TBSRTC) and PTMC is highly suspected, then the first clinical dilemma appears. The main question is to what extent surgery should be performed. Which of the procedures, hemithyroidectomy or thyroidectomy, is the better therapeutic option? The next clinical dilemma regards lymph node dissection. The problem is more straightforward if we have enlarged lymph nodes of the neck (measuring more than 1.0 cm in the short axis diameter), and subsequently, metastases are microscopically confirmed. In such situations, therapeutic lymph node dissection is obviously recommended. A different situation is when the neck lymph nodes are not pathologically enlarged. This clinical status is often highlighted because some authors say that microscopic neck lymph node metastases are present in over 60–80% of PTMC patients [9, 21]. Therefore, the question is if prophylactic node dissection should be taken into consideration.
Regarding the increasing rate of PTMC occurrences with often indolent clinical behavior, various options for disease management have emerged. However, so far, the most common treatment that is recommended by many authors is surgery. Regardless of the PTMC’s “clinically silent behavior,” researchers recommend either thyroidectomy or minimum hemithyroidectomy with isthmectomy. The second management option recommended by other researchers is active surveillance with surgical treatment when tumor progression occurs [22, 23].
After considering all the mentioned dilemmas, a fundamental question appears: which PTMC cases should be treated as “true cancer” and which as “precancer”? To answer this question, we evaluated some clinicopathological features of PTMCs in stage pN1 to select tumors that should be treated aggressively as “true cancers,” and then, we evaluated some clinicopathological features of PTMCs in stage pN0, which may be treated less aggressively as “precancers.”
Materials and methods
We performed retrospective chart reviews of 4716 patients who were admitted and surgically treated in one center between 2008 and 2017. Among the patients, 434 (9.2%) had thyroid malignancies; finally, we selected 177 (3.75%) patients with papillary thyroid cancers, which were described as the tumors with 1.0 cm maximum in diameter on the basis of histopathological examinations and thereby were classified as papillary thyroid microcarcinomas (PTMCs). All of the patients were staged in accordance with TNM staging criteria (tumor-node-metastasis) proposed by AJCC 8th Edition [24]. Preoperative thyroid ultrasonography, UG-FNAB, and cytological examinations were performed in all cases. After surgery, the final histopathological classification was performed according to World Health Organization Guidelines. The surgical tissue specimens were fixed in 10% buffered formalin and diagnosed histopathologically at the Department of Pathomorphology, Wroclaw Medical University. Representative blocks were selected. Because of the fact that papillary thyroid carcinoma can be multifocal, the adjacent and opposite lobe was sampled and all pale areas were processed. Lesions with a diameter maximum of 1.0 cm or less were processed in their entirety; however, a minimum of 5–8 blocks were taken from each lesion. Serial sectioning and careful cutting of the representative tissue sample was done. A routine method of specimen processing was performed. Sections were cut in 4-μm thickness on which conventional hematoxylin and eosin (H&E) staining sections were prepared. H&E sections were evaluated by two experienced thyroid lesion pathologists to confirm the diagnosis, features of the tumor, and the extent of the malignant process. Patients were diagnosed as PTMC, if the largest tumor diameter was equal or less 1 cm. To analyze the cases with potentially aggressive behavior, we enrolled PTMC patients with lymph node metastases (pN1, central, and/or lateral) and evaluated some of their clinicopathological features. Of the 177 patients with PTMC, 116 (65.5%) were diagnosed with thyroid malignancies before surgery, 63 (35.6%) subjects presented with multifocal tumors, and 15 (8.5%) presented with bilateral tumors (Table 1). The PTMC patients were divided into two groups according to the histopathological diagnosis of their lymph node metastases: the pN0 group (n = 115)—without metastasis, and the pN1 group (n = 62)—with lymph node metastasis.
Table 1.
Backgrounds and clinicopathological characteristics of PTMC patients (n = 177)
| Parameters | N (%) or mean ± SD |
|---|---|
| Gender: | |
| Male | 23 (13.0) |
| Female | 154 (87.0) |
| Age (years) | 48.3 + 14.1 |
| Age | |
| < 55 years | 109 (61.6) |
| ≥ 55 years | 68 (38.4) |
| Diagnosis of malignancy | |
| Before surgery | 116 (65.5) |
| After surgery | 61 (34.5) |
| Diagnosed as multifocal | |
| No | 114 (64.4) |
| Yes | 63 (35.6) |
| Diagnosed as bilateral | |
| No | 162 (91.5) |
| Yes | 15 (8.5) |
| Tumor size | |
| < 0.5 cm | 81 (45.8) |
| ≥ 0.5 cm | 96 (54.2) |
| Lymph node metastasis | |
| No | 115 (65.0) |
| Yes | 62 (35.0) |
| Type of surgery | |
| Total | 126 (71.2) |
| No-total | 51 (28.8) |
| Reoperation | |
| No | 131 (74.0) |
| Yes | 46 (26.0) |
PTMC papillary thyroid microcarcinoma, SD standard deviation
Statistical analysis
Descriptive data were presented as numbers of observations and percentages or as averages and standard deviations (± SD). Student’s t tests for independent samples or chi-square tests were applied for group comparisons. The Kaplan-Meier method and log-rank test were performed to compare the disease-free survival distribution of N0 and N1 patients. Multivariable logistic regression analysis was used to confirm the selected factors that were associated with an increased risk of lymph node metastases. The diagnostic potential of each independent variable was determined by receiver operating characteristic (ROC) analysis and was expressed in terms of the area under the ROC curve (AUC). The accuracy, sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), likelihood ratio of positive results (LR(+)), likelihood ratio of negative results (LR(−)), and Youden Index were also calculated. All calculated P values were two-sided, and P < 0.05 was considered significant. Statistical analyses were performed using Statistica 13.1 (TIBCO Software Inc., CA, USA).
Results
To date, none of the 177 patients in our series who underwent surgery and subsequent observation died of thyroid cancer. By questionnaire, we confirmed that 5 patients died of not PTMC-related disease.
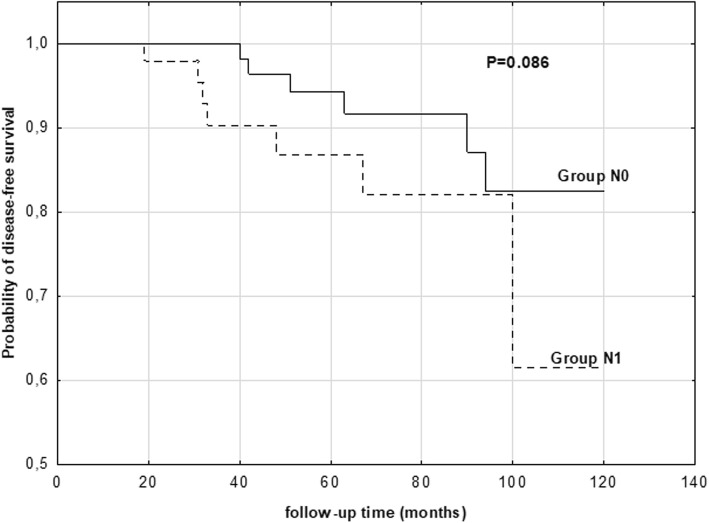
We created the model of lymph nodes status as a predictor of disease-free survival. The probabilities of disease-free survival in the PTMC patients with N0 or N1 lymph node metastasis at the term of observation are showed on Fig. 1. The proportion of disease-free survival was highest in N0 patients (83%) than in N1 patients (62%). These differences were not significant; however, the trend was observed (83% vs. 62%), P = 0.086.
Fig. 1.
Relationship between lymph node metastasis and the probability of disease-free survival of PTMC patients
Multivariable logistic regression analysis was performed for the selection of clinical and pathological predictors of lymph node metastasis (Table 2). We demonstrated significantly higher rates of multifocal or bilateral tumor occurrence in the PTMC patients in the pN1 group than in the patients in the pN0 group (P < 0.0001 for both) (Table 2). In addition, the occurrence of thyroid tumors above 0.5 cm in size was a significant risk factor for lymph node metastasis (P < 0.0001), whereas age, gender, and capsular invasion were not found to be significant predictors of lymph node metastasis (P > 0.05).
Table 2.
Multiple logistic regression analysis of clinical and pathological factors that can be predictors of lymph node metastasis (pN0/pN1, 0/1) in the patients with PTMC. Results were also confirmed by chi-square test
| Variables | Patients with pN0 (n = 115) | Patients with pN1 (n = 62) | P value (χ2 test) | Logistic regression analysis | ||
|---|---|---|---|---|---|---|
| OR | ± 95% CI | P value (Wald test) | ||||
| N (%) | N (%) | |||||
| Gender | 0.065 | 0.93 | 0.42–2.06 | 0.858 | ||
| Male | 11 (9.6) | 12 (19.3) | ||||
| Female | 104 (90.4) | 50 (80.7) | ||||
| Age | 0.303 | 0.96 | 0.51–1.80 | 0.890 | ||
| < 55 years | 74 (64.4) | 35 (56.5) | ||||
| ≥ 55 years | 41 (35.6) | 27 (43.5) | ||||
| Diagnosed as multifocal | < 0.0001* | 9.40 | 4.6–19.3 | < 0.0001* | ||
| No | 94 (81.7) | 20 (32.3) | ||||
| Yes | 21 (18.3) | 42 (67.7) | ||||
| Diagnosed as bilateral | < 0.0001* | 2.90 | 0.0–12.8 | < 0.0001* | ||
| No | 115 (100.0) | 47 (75.8) | ||||
| Yes | 0 (0.0) | 15 (24.2) | ||||
| Tumor size | < 0.0001* | 65.80 | 15.1–287.3 | < 0.0001* | ||
| < 0.5 cm | 79 (68.7) | 2 (3.2) | ||||
| ≥ 0.5 cm | 36 (31.3) | 60 (96.8) | ||||
| Capsular invasion | 0.047 | 2.54 | 0.98–6.56 | 0.052 | ||
| No | 106 (92.2) | 51 (82.3) | ||||
| Yes | 9 (7.8) | 11 (17.7) | ||||
PTMC papillary thyroid microcarcinoma, CI coefficient interval, *statistically significant
The diagnostic potential of three selected factors was evaluated in terms of their capability to differentiate PTMC patients with lymph node metastasis from PTMC patients with pN0. The results of the ROC analysis showed that multifocal or bilateral tumors and tumor sizes above 0.5 cm were significant predictors of lymph node metastasis (P < 0.0001 for all) (Table 3). The presence of tumors above 0.5 cm in size and multifocal tumors were found to be the best predictors of lymph node metastasis in PTMC (Fig. 2).
Table 3.
Diagnostic potential of multifocal and bilateral tumors presence and tumors size above 5 mm as indicators of lymph node metastasis in PTMC patients
| ROC analysis | Presence of multifocal tumors | Presence of bilateral tumors | Presence of tumor ≥ 0.5 cm |
|---|---|---|---|
| AUC (± 95%CI) | 0.747 (0.668–0.827) | 0.621 (0.529–0.713) | 0.827 (0.767–0.888) |
| P value | < 0.0001 | < 0.0001 | < 0.0001 |
| SE | 0.041 | 0.047 | 0.031 |
| Sensitivity | 0.677 | 0.242 | 0.968 |
| Specificity | 0.817 | 1.000 | 0.687 |
| Accuracy | 0.768 | 0.734 | 0.785 |
| LR (+) | 0.667 | 1.000 | 0.625 |
| LR (−) | 0.825 | 0.710 | 0.975 |
| PPV | 0.183 | 0.000 | 0.313 |
| NPV | 0.323 | 0.758 | 0.032 |
| Youden Index | 0.495 | 0.242 | 0.665 |
PTMC papillary thyroid microcarcinoma
Fig. 2.
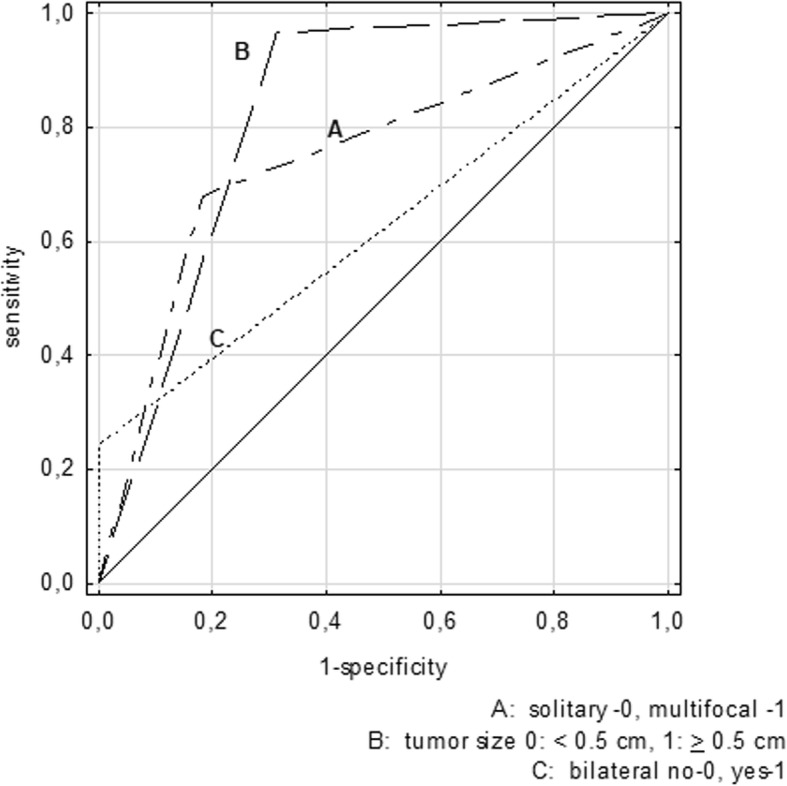
Comparison of receiver operating curves (ROC) for the presence of multifocal or bilateral tumors or tumor size above 0.5 cm. The performance of a marker unable to discriminate between pN0 and pN1 patients, whose area under the ROC curve (AUC) is equal to 0.5 was marked with a constant line
Discussion
In light of the recent evidence regarding the safety of nonsurgical management of patients with PTMC, the term “precancer” to describe tumors that are often indolent tumor may be introduced. Even the American Thyroid Association (ATA) guidelines allow for an active surveillance management approach instead of immediate surgery in patients with low-risk PTMC [25, 26]. Recently, we have observed that clinicians for some select individuals who are diagnosed with PTMC offer active surveillance instead of surgical treatment [27, 28]. According to the NCCN Guidelines Version 1.2019 rather, minimum lobectomy is recommended; however, some criteria like no prior radiation exposure, no distant metastases, no cervical lymph node metastases, and no extrathyroidal extension must be presented [29].
Currently, the term NIFTP is commonly used and is suggested for many well-differentiated thyroid tumors [19]. The main goals of introducing the term NIFTP were firstly, deleting the word “carcinoma” describing an indolent form of PTMC and secondly, to prevent overdiagnosis and overtreatment [20]. In our study, after accurate reanalysis of all PTMC specimens by two experienced pathologists, who performed histopathological examinations, according to their suggestions, we did not decide to name these tumors NIFTP, because of some pathologic characteristics such as capsular invasions or the presence of true papillae defined as exclusion criteria [19].
With increasing awareness of the overdiagnosis with overtreatment phenomena of PTMCs along with the recent clinical observations and ATA guideline recommendations, it seems very useful to introduce data about PTMCs that may be treated as a “precancer.” This information may help endocrinologists, radiologists, and surgeons to understand the real clinical and pathological nature of PTMC cases. This is why some authors suggest changing the PTMC terminology, which in their opinion, may replace patients’ perceptions of their cancer diagnosis [4]. On the other hand, very aggressive PTMC might be sometimes observed [3]. One of the most unfavorable clinicopathological features of aggressive PTMCs is lymph node metastasis [7, 8]. Jiang et al. suggested that the presence of central or lateral lymph node metastases in papillary thyroid cancer (PTC) cases affects the prognosis and treatment of the individuals [8]. Moreover, these metastases increase the rate of regional recurrence and mortality, especially in elderly patients [30]. Some authors suggest that almost 80% of PTMC patients develop lymph node micrometastases, which are usually diagnosed in postsurgical histopathology [21]. However, lymph node metastases are diagnosed in only 30% of patients with PTC during clinical examinations [21]. It was also presented in Xue’s review that the accuracy of preoperative ultrasonography for the diagnosis of lymph node metastases is not very high and can be as high as 48.3% [26]. It is very important to assess the clinicopathological features that could be significantly specific for lymph node metastasis and that are usually undetected during routine physical examinations. Making an accurate selection of PTMC cases that could be treated as “precancer” requires high sensitivity in examinations to even exclude cases of lymph node micrometastases. These excluded cases should be treated more aggressively as they are considered “true cancers.” Xue et al. revealed that the sensitivity of ultrasound ranged from 22.6 to 55% in predicting central lymph node metastases, which means that almost half of the patients with metastases were incorrectly diagnosed [26].
Generally, in Poland, the diagnoses and treatment experiences widely vary for patients with PTMC. The majority of these tumors are asymptomatic and are often discovered by diagnostic imaging examinations ordered due to issues that are unrelated to thyroid pathology. This suspicion of PTMC starts a diagnostic and treatment cascade, which almost always leads to surgery. These medical procedures often result in physical and emotional side effects in patients diagnosed with PTMC [4]. Considering the rate of overdiagnosis and to avoid stressful and uncomfortable situations, we should inform the patients about the potential consequences of thyroid testing and its possible final diagnosis. However, if we suspect PTMC after thyroid diagnostic tests, we should discuss all clinical aspects of the discovered tumor with the patient. One very useful and informative message is that clinicians often describe PTMC as a “small and slow-growing tumor.” We also should inform the patients that currently, more and more researchers offer patients the option of active surveillance instead of surgery. According to some authors’ results, even lymph node metastases are not a crucial problem [22, 23]. These authors conclude that the rate of occurrence of lymph node metastases in individuals with PTMC who are with active surveillance is comparable to the rate observed in patients with PTMC who underwent thyroidectomies immediately after diagnosis. Moreover, the authors also estimated that the outcomes of thyroid surgery are the same regardless of whether surgical treatment is undertaken immediately after PTMC diagnosis or after the first observation of tumor progression. Oda et al. state that delayed surgery in patients with PTMC, who initially followed active surveillance, is safe, and the results are comparable to individuals who underwent immediate surgical management [27]. Currently, we can observe that some authors recommend active surveillance for PTMC instead of surgical treatment. Next, we should discuss the current ATA guideline recommendations that allow performing a hemithyroidectomy instead of a total thyroid resection in the majority of PTMC cases. However, we observe that most of the patients, even after discussions with accurate and precise clinical information, choose surgical treatment options. Some patients prefer less invasive surgical procedures, such as a hemithyroidectomy, to avoid taking levothyroxine supplements for the rest of their life. However, these individuals should also be informed that all surgical options, even partial thyroid resection, will require thyroid hormone supplements [31, 32].
As of now, some basic fundamental questions still remain: “What we can do to avoid stressful and difficult clinical situations that elicit emotional and physical responses from patients?” and “What should be the proper management of PTMC?” Some authors controversially suggest abandoning fine needle aspiration biopsies of all thyroid nodules equal to or below 1.0 cm in diameter. Such an approach should reduce the number of unnecessary diagnostic procedures for PTMC [29]. Additionally, the authors of this study believe that the main focus of PTMC concerns is overdiagnosis rather than overtreatment. Neck ultrasound examinations are widely and easily accessible, which often cause unsuspected diagnoses of the thyroid nodule. Subsequently, if we have even one unfavorable ultrasound feature to describe a newly diagnosed incidentaloma, almost every clinician will recommend a UG-FNAB [33]. Some authors suggest that involving patients in the shared decision-making process about whether to perform a UG-FNAB might be an effective strategy in reducing the number of unnecessary diagnostic procedures associated with incidental PTMCs [34, 35].
Some authors have suggested that metastases frequently affect both the central and ipsilateral neck lymph nodes, even in patients with PTMC [9]. Other authors added that skip lesions, i.e., the involvement of the lateral compartment without central lymph nodes, are rare, but their incidence is similar to that of larger PTCs [36]. However, other authors say that patients with multifocal PTMC tumors are more likely to have lymph node metastases [9]. Gur et al. analyzed all types of PTC (including PTMC) and estimated that the risk of local recurrence, lymph node metastasis, and distant metastasis are increased in multifocal PTC patients, so they concluded that multifocality is a poor prognostic factor for PTC [37]. In our study, we demonstrated significantly higher rates of multifocal or bilateral tumor occurrence in PTMC patients with lymph node metastases than in patients without metastases (pN1 vs. pN0). Some authors have assessed that the location of the primary PTMC tumor did not predict the pattern of lymph node metastases, which is similar to observations of PTCs [38]. In our study, we did not conduct analyses between tumor locations and lymph node metastasis patterns. Wada et al. showed that the size of the primary PTMC tumor slightly influenced the frequency of lymph node metastasis. The frequency was 55.7% for tumors that were 0.5 cm in diameter and 73.7% for those that were > 0.5 and 1.0 cm in diameter. The authors did not observe an effect on nodal recurrence [9]. In our study, we noticed that tumor sizes above 0.5 cm were a significant risk factor for lymph node metastasis. The presence of tumors above 0.5 cm in size and multifocal lesions were found to be the best predictors of PTMC lymph node metastasis. Because of the results of our study, which showed that multifocal or bilateral tumors and tumor size above 0.5 cm were significant predictors of lymph node metastasis, it may implicate some additional pathologist’s procedures. In some individual cases, i.e., with lymph node metastases and larger tumors (≥ 0.5 cm), the whole resected thyroid tissue should be pathologically examined to exclude multi- and bilaterality of PTMC.
There are several limitations in this study. Firstly, it is limited by its retrospective design. Secondly, the study is also limited by the number of multifocal and bilateral PTMC. However, multifocal and bilateral PTMC is not very often observed. The next limitation of our study is its single center analysis. Thus, multicenter and large cohorts study should be performed to identify the larger number of multifocal and bilateral PTMC.
Conclusions
Multifocal or bilateral PTMC tumors with diameters above 0.5 cm should be treated aggressively as “true cancer.” Because lymph node metastases are observed more commonly in such cases, thyroidectomy and selected central/lateral lymph node dissection are recommended. These cases might benefit mostly from this procedure. Unifocal PTMC tumors with diameters equal to or below 0.5 cm in some individuals may be treated less aggressively; a hemithyroidectomy with isthmectomy is an acceptable procedure.
Our study helps clinicians to more safely and effectively select patients for whom aggressive treatment is recommended or is not necessary. Additionally, for some select individuals with PTMCs that are stage N0, their diagnoses could be explained as a “precancer” for emotional comfort.
Acknowledgements
The authors are grateful to all the staff at the study center who contributed to this study.
Abbreviations
- ATA
American Thyroid Association
- PTC
Papillary thyroid cancer
- PTMC
Papillary thyroid microcarcinoma
- TBSRTC
The Bethesda System for Reporting Thyroid Cytopathology
- UG-FNAB
Ultrasound-guided fine needle aspiration biopsy
Authors’ contributions
KK contributed to the conceptualization of the study. KK, BW, ZF, DP, and ŁN contributed to obtaining the data of the study. KK and DD contributed to the formal analysis of the study. KK contributed to the investigation of the study. KK and DD contributed to the methodology of the study. KK contributed to the project administration of the study. KK, BW, ZF, DP, and ŁN contributed to the resources of the study. KK and JR contributed to the supervision of the study. KK and DD contributed to the validation of the study. KK and DD contributed to the writing of the original draft of the study. KK contributed to the writing, review, and editing of the manuscript. All authors read and approved the final manuscript.
Funding
This work did not receive funding from the public or private sectors.
Availability of data and materials
Not applicable.
Ethics approval and consent to participate
All research was carried out in compliance with the Helsinki Declaration. This study was approved by the Ethics Committee of Wroclaw Medical University (number KB-783/2017). The data were analyzed anonymously and retrospectively on the basis of medical records. The authors did not have access to any identifying patient information and did not have any direct access to the study participants.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Krzysztof Kaliszewski, Phone: +48 71 733 30 00, Email: krzysztofkali@wp.pl.
Dorota Diakowska, Email: dorota.diakowska@umed.wroc.pl.
Beata Wojtczak, Email: dorota.diakowska@umed.wroc.pl.
Zdzisław Forkasiewicz, Email: zetfor@interia.pl.
Dominika Pupka, Email: dominika.pupka@gmail.com.
Łukasz Nowak, Email: lllukasz.nowak@gmail.com.
Jerzy Rudnicki, Email: jrudnicki@op.pl.
References
- 1.Hedinger C, Williams ED, Sobin LH. The WHO histological classification of thyroid tumors: a commentary on the second edition. Cancer. 1989;63:908–911. doi: 10.1002/1097-0142(19890301)63:5<908::AID-CNCR2820630520>3.0.CO;2-I. [DOI] [PubMed] [Google Scholar]
- 2.Kaliszewski K, Strutyńska-Karpińska M, Zubkiewicz-Kucharska A, et al. Should the prevalence of incidental thyroid cancer determine the extent of surgery in multinodular goiter? PLoS One. 2016;11:e0168654. doi: 10.1371/journal.pone.0168654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lewiński A, Adamczewski Z. Papillary thyroid carcinoma: a cancer with an extremely diverse genetic background and prognosis. Pol Arch Intern Med. 2017;127:388–389. doi: 10.20452/pamw.4058. [DOI] [PubMed] [Google Scholar]
- 4.Nickel B, Brito JP, Moynihan R, Barratt A, Jordan S, McCaffery K. Patients’ experiences of diagnosis and management of papillary thyroid microcarcinoma: a qualitative study. BMC Cancer. 2018;18:242. doi: 10.1186/s12885-018-4152-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hong YR, Yan CX, Mo GQ, et al. Conventional US, elastography, and contrast enhanced US features of papillary thyroid microcarcinoma predict central compartment lymph node metastases. Sci Rep. 2015;5:7748. doi: 10.1038/srep07748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Choi JB, Lee WK, Lee SG, et al. Long-term oncologic outcomes of papillary thyroid microcarcinoma according to the presence of clinically apparent lymph node metastasis: a large retrospective analysis of 5,348 patients. Cancer Manag Res. 2018;10:2883–2891. doi: 10.2147/CMAR.S173853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kim JY, Jung EJ, Park T, et al. Impact of tumor size on subclinical central lymph node metastasis in papillary thyroid microcarcinoma depends on age. World J Surg Oncol. 2015;13:88. doi: 10.1186/s12957-015-0478-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Jiang W, Wei HY, Zhang HY, Zhuo QL. Value of contrast-enhanced ultrasound combined with elastography in evaluating cervical lymph node metastasis in papillary thyroid carcinoma. World J Clin Casus. 2019;7:49–57. doi: 10.12998/wjcc.v7.i1.49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wada N, Duh QY, Sugino K, et al. Lymph node metastasis from 259 papillary thyroid microcarcinomas: frequency, pattern of occurrence and recurrence, and optimal strategy for neck dissection. Ann Surg. 2003;237:399–407. doi: 10.1097/01.SLA.0000055273.58908.19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Noguchi S, Yamashita H, Uchino S, Watanabe S. Papillary microcarcinoma. World J Surg. 2008;32:747–753. doi: 10.1007/s00268-007-9453-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ito Y, Tomoda C, Uruno T, et al. Clinical significance of metastasis to the central compartment from papillary microcarcinoma of the thyroid. World J Surg. 2006;30:91–99. doi: 10.1007/s00268-005-0113-y. [DOI] [PubMed] [Google Scholar]
- 12.Liu LS, Liang J, Li JH, et al. The incidence and risk factors for central lymph node metastasis in cN0 papillary thyroid microcarcinoma: a meta-analysis. Eur Arch Otorhinolaryngol. 2017;274:1327–1338. doi: 10.1007/s00405-016-4302-0. [DOI] [PubMed] [Google Scholar]
- 13.Sakorafas GH, Sampanis D, Safioleas M. Cervical lymph node dissection in papillary thyroid cancer: current trends, persisting controversies, and unclarified uncertainties. Surg Oncol. 2010;19:e57–e70. doi: 10.1016/j.suronc.2009.04.002. [DOI] [PubMed] [Google Scholar]
- 14.White ML, Doherty GM. Level VI lymph node dissection for papillary thyroid cancer. Minerva Chir. 2007;62:383–393. [PubMed] [Google Scholar]
- 15.Sadowski BM, Snyder SK, Lairmore TC. Routine bilateral central lymph node clearance for papillary thyroid cancer. Surgery. 2009;146:696–703. doi: 10.1016/j.surg.2009.06.046. [DOI] [PubMed] [Google Scholar]
- 16.Moo TA, McGill J, Allendorf J, Lee J, Fahey T, Zarnegar R. Impact of prophylactic central neck lymph node dissection on early recurrence in papillary thyroid carcinoma. World J Surg. 2010;34:1187–1191. doi: 10.1007/s00268-010-0418-3. [DOI] [PubMed] [Google Scholar]
- 17.Chang YW, Kim HS, Kim HY, Lee JB, Bae JW, Son GS. Should central lymph node dissection be considered for all papillary thyroid microcarcinoma? Asian J Surg. 2016;39:197–201. doi: 10.1016/j.asjsur.2015.02.006. [DOI] [PubMed] [Google Scholar]
- 18.Zhi J, Zhao J, Gao M, et al. Impact of major different variants of papillary thyroid microcarcinoma on the clinicopathological characteristics: the study of 1041 cases. Int J Clin Oncol. 2018;23:59–65. doi: 10.1007/s10147-017-1170-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hung YP, Barletta JA. A user’s guide to non-invasive follicular thyroid neoplasm with papillary-like nuclear features (NIFTP) Histopathology. 2018;72:53–69. doi: 10.1111/his.13363. [DOI] [PubMed] [Google Scholar]
- 20.Amendoeira I, Maia T, Sobrinho-Simoes M. Non-invasive follicular thyroid neoplasm with papillary-like nuclear features (NIFTP): impact on the reclassification of thyroid nodules. Endocr Relat Cancer. 2018;25:R247–R258. doi: 10.1530/ERC-17-0513. [DOI] [PubMed] [Google Scholar]
- 21.Sancho JJ, Lennard TW, Paunovic I, Triponez F, Sitges-Serra A. Prophylactic central neck dissection in papillary thyroid cancer: a consensus report of the European Society of Endocrine Surgeons (ESES) Langenbeck’s Arch Surg. 2014;399:155–163. doi: 10.1007/s00423-013-1152-8. [DOI] [PubMed] [Google Scholar]
- 22.Ito Y, Miyauchi A, Kihara M, Higashiyama T, Kobayashi K, Miya A. Patient age is significantly related to the progression of papillary microcarcinoma of the thyroid under observation. Thyroid. 2014;24:27–34. doi: 10.1089/thy.2013.0367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sugitani I, Fujimoto Y, Yamada K. Association between serum thyrotropin concentration and growth of asymptomatic papillary thyroid microcarcinoma. World J Surg. 2014;38:673–678. doi: 10.1007/s00268-013-2335-8. [DOI] [PubMed] [Google Scholar]
- 24.Perrier ND, Brierley JD, Tuttle RM. Differentiated and anaplastic thyroid carcinoma: major changes in the American Joint Committee on Cancer eighth edition cancer staging manual. CA Cancer J Clin. 2017;68:55–63. doi: 10.3322/caac.21439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Haugen BR, Alexander EK, Bible KC, et al. 2015 American Thyroid Association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: the American Thyroid Association guidelines task force on thyroid nodules and differentiated thyroid cancer. Thyroid. 2016;26:1–133. doi: 10.1089/thy.2015.0020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Xue S, Wang P, Hurst ZA, Chang YS, Chen G. Active surveillance for papillary thyroid microcarcinoma: challenges and prospects. Front Endocrinol (Lausanne) 2018;9:736. doi: 10.3389/fendo.2018.00736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Oda H, Miyauchi A, Ito Y, et al. Incidences of unfavorable events in the management of low-risk papillary microcarcinoma of the thyroid by active surveillance versus immediate surgery. Thyroid. 2016;26:150–155. doi: 10.1089/thy.2015.0313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Miyauchi A. Clinical trials of active surveillance of papillary microcarcinoma of the thyroid. World J Surg. 2016;40:516–522. doi: 10.1007/s00268-015-3392-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.NCCN Clinical Practice Guidelines in Oncology, 2019. Available in: https://www.nccn.org/professionals/physician_gls/pdf/thyroid.pdf
- 30.Zaydfudim V, Feurer ID, Griffin MR, Phay JE. The impact of lymph node involvement on survival in patients with papillary and follicular thyroid carcinoma. Surgery. 2008;144:1070–1077. doi: 10.1016/j.surg.2008.08.034. [DOI] [PubMed] [Google Scholar]
- 31.Park HK, Kim DW, Ha TK, et al. Factors associated with postoperative hypothyroidism after lobectomy in papillary thyroid microcarcinoma patients. Endocr Res. 2015;40:49–53. doi: 10.3109/07435800.2014.933975. [DOI] [PubMed] [Google Scholar]
- 32.Lee DY, Seok J, Jeong WJ, Ahn SH. Prediction of thyroid hormone supplementation after thyroid lobectomy. J Surg Res. 2015;193:273–278. doi: 10.1016/j.jss.2014.07.003. [DOI] [PubMed] [Google Scholar]
- 33.Woliński K, Szkudlarek M, Szczepanek-Parulska E, Ruchała M. Usefulness of different ultrasound features of malignancy in predicting the type of thyroid lesions: a meta-analysis of prospective studies. Pol Arch Med Wewn. 2014;124:97–104. doi: 10.20452/pamw.2132. [DOI] [PubMed] [Google Scholar]
- 34.Stacey D, Legare F, Col NF, et al. Decision aids for people facing health treatment or screening decisions. Cochrane Database Syst Rev. 2014;1:CD001431. doi: 10.1002/14651858.CD001431.pub4. [DOI] [PubMed] [Google Scholar]
- 35.Hoffmann TC, Legare F, Simmons MB, et al. Shared decision making: what do clinicians need to know and why should they bother? Med J Aust. 2014;201:35–39. doi: 10.5694/mja14.00002. [DOI] [PubMed] [Google Scholar]
- 36.Sivanandan R, Soo KC. Pattern of cervical lymph node metastases from papillary carcinoma of the thyroid. Br J Surg. 2001;88:1241–1244. doi: 10.1046/j.0007-1323.2001.01843.x. [DOI] [PubMed] [Google Scholar]
- 37.Gur EO, Karaisli S, Haciyanli S, et al. Multifocality related factors in papillary thyroid carcinoma. Asian J Surg. 2019;42:297–302. doi: 10.1016/j.asjsur.2018.05.004. [DOI] [PubMed] [Google Scholar]
- 38.Mirallie E, Visset J, Sagan C, et al. Localization of cervical node metastasis of papillary thyroid carcinoma. World J Surg. 1999;23:970–973. doi: 10.1007/s002689900609. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.