Lung adenocarcinoma is a non–small-cell lung cancer and one of the most invasive forms of lung cancers associated with poor prognosis. It is characterized by distinct cellular and molecular features such as malignant epithelia comprised of cuboidal to low columnar cells resembling club cells or type II pneumocytes (1). Diagnosis and treatment of lung adenocarcinoma largely rely on establishing molecular biomarker profiles through histological examination as well as genomic analysis for unique mutations that contribute to invasiveness. A large variety of driver mutations have been established in lung adenocarcinoma (e.g., EGFR, KRAS, ALK, HER2, BRAF, ROS, and MET) that are responsible for the majority of diagnosed adenocarcinoma cases (1–3). Several successful therapies targeting these mutations have been approved by the U.S. Food and Drug Administration and shown to be effective, but ∼30% of adenocarcinoma tumors have unknown drivers. Although we have made good progress in understanding and targeting the molecular mechanisms of lung tumor metastasis, there remains a need to identify additional molecular targets in lung adenocarcinoma.
In this issue of the Journal, Tsoyi and colleagues (pp. 659–666) report that syndecan-2 (SDC2), but not SDC1, is highly overexpressed in human lung adenocarcinoma (4). SDC1 and SDC2 are proteoglycans, key components of the extracellular matrix (ECM) that provide structural support for the lung and play crucial roles in normal and diseased lungs (5–9). The proteoglycans are a family of charged molecules that contain a core protein and one or more covalently attached glycosaminoglycan chains. Syndecans are cell-surface heparan sulfate proteoglycans that not only provide support for the ECM but also bind growth factors that guide morphogen gradients during development and act as coreceptors for receptor tyrosine kinases and receptors for atherogenic lipoproteins and cell activation (8). Overexpression of syndecans has been shown to enhance tumor cell invasiveness and proliferation, but this is dependent on cancer origin and type (10, 11).
Recently, SDC2 has been receiving increased attention for its relatedness to the pathogenesis of various cancers, including lung cancer (12). In a study reported in this issue of the Journal, Tsoyi and colleagues analyzed lung adenocarcinoma samples in comparison with normal lung samples, and found significantly elevated SDC2 mRNA and protein by immunohistochemistry (4). Their results indicate that SDC2 is mainly expressed in tumor-affected epithelium and associated macrophages. Because there are several types of lung adenocarcinomas, the authors conducted a retrospective analysis of deidentified lung samples of squamous, adenosquamous, small-cell, and papillary adenocarcinoma. They found no significant differences in SDC2 expression among these lung cancer types as assessed by lung cancer tissue microarrays. It would be interesting and clinically relevant to determine whether increased SDC2 mRNA levels correlate with a specific adenocarcinoma subtype or stage of tumor progression in a prospective study.
To demonstrate that SDC2 plays a role in adenocarcinoma tumorigenicity, the authors investigated the invasive properties of the lung adenocarcinoma cell lines A549 and NCI-H23 in vitro in response to silencing or overexpression of SDC2. Using Matrigel invasion assays, they demonstrated that shRNA-induced silencing of SDC2 expression decreased the invasiveness of A549 and NCI-H23 cells, whereas overexpression of SDC2 enhanced the invasive properties of A549 cells in an MMP9-dependent fashion. Identifying a link between SDC2 and MMP9 is logical considering that MMP9 has been associated with poor prognosis in lung cancer (13). In future studies, however, a detailed clinicopathological correlation of SDC2 and MMP9 in well-phenotyped human lung cancer samples or even primary cells from different human lung adenocarcinoma subtypes would make the results more clinically relevant and impactful.
Tsoyi and colleagues further uncover a novel mechanism through which SDC2 potentiates lung adenocarcinoma cell invasiveness by activating syntenin-1 and NF-κB signaling (4). Because it is known that SDC2 can bind syntenin-1 (14), they propose that syntenin-1 acts as a mediator of the SDC2 stimulatory effect on MMP9 production. They substantiate their hypothesis by demonstrating that syntenin-1 overexpression restores MMP9 production and the invasive capacity of SDC2-silenced A549 cells. Based on the knowledge that NF-κB regulates many nuclear expressed genes, including MMPs, and that NF-κB activation can be mediated by syntenin-1 (15, 16), the authors also propose a mechanism of SDC2-induced and syntenin-1–dependent MMP9 stimulation via NF-κB. Using similar gain- and loss-of-function experiments in A549 cells, they demonstrate that SDC2 regulates NF-κB cytosol-to-nuclear translocation and its binding to the MMP9 promoter. They also show that xenografts of SDC2-silenced A549 cells in severe combined immunodeficient mice have decreased tumor size in response to SDC2 downregulation. The key in vitro experiments were performed in another NSCLC cell line, NCI-H23, in addition to A549 cells. Future studies using well-phenotyped human lung cancer samples or primary cells from different human lung adenocarcinoma subtypes and cell-specific knockouts of SDC2 would advance the mechanistic findings of this study.
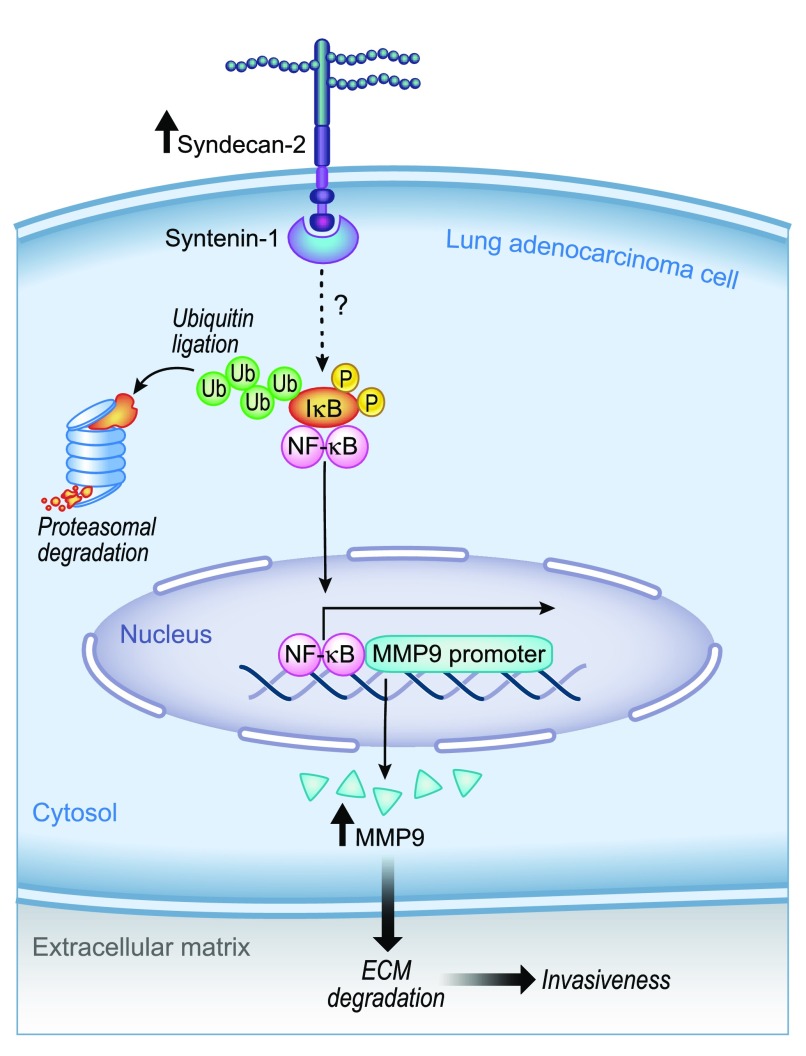
In conclusion, Tsoyi and colleagues propose an elegant mechanism whereby SDC2 potentiates tumor invasiveness in lung adenocarcinoma. This is schematically represented in Figure 1, which shows that upregulation of SDC2 in lung adenocarcinoma leads to ECM degradation and invasiveness mediated through MMP9 upregulation, which is controlled through syntenin-1–NF-κB signaling. The authors also highlight the potential of SDC2 as a novel target for lung adenocarcinoma therapies. Although it is likely that inhibition of this cell-surface heparan sulfate proteoglycan or activation of the intracellular syntenin-1 will be difficult to achieve with small-molecule inhibitors, the rapid expansion of inhaled RNA–based technologies may provide exciting new therapeutic options for targeting the syndecan-2 pathway (17).
Figure 1.
Schematic representation of a potential mechanism of cell invasiveness potentiated by syndecan-2 in lung adenocarcinoma. ECM = extracellular matrix; MMP9 = metalloproteinase 9; P = phosphor; Ub = ubiquitin. Illustration by Jacqueline Schaffer.
Footnotes
Author disclosures are available with the text of this article at www.atsjournals.org.
References
- 1.Kuhn E, Morbini P, Cancellieri A, Damiani S, Cavazza A, Comin CE. Adenocarcinoma classification: patterns and prognosis. Pathologica. 2018;110:5–11. [PubMed] [Google Scholar]
- 2.Liang H, Liu X, Wang M. Immunotherapy combined with epidermal growth factor receptor-tyrosine kinase inhibitors in non-small-cell lung cancer treatment. Onco Targets Ther. 2018;11:6189–6196. doi: 10.2147/OTT.S178497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wang J, Wang Y, Tong M, Pan H, Li D. Research progress of the clinicopathologic features of lung adenosquamous carcinoma. Onco Targets Ther. 2018;11:7011–7017. doi: 10.2147/OTT.S179904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tsoyi K, Osorio JC, Chu SG, Fernandez IE, Poli De Frias S, Sholl L, et al. Lung adenocarcinoma syndecan-2 potentiates cell invasiveness Am J Respir Cell Mol Biol 201960659–666.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Frevert CW, Sannes PL. Matrix proteoglycans as effector molecules for epithelial cell function. Eur Respir Rev. 2005;14:137. [Google Scholar]
- 6.Nikitovic D, Berdiaki A, Spyridaki I, Krasanakis T, Tsatsakis A, Tzanakakis GN. Proteoglycans-biomarkers and targets in cancer therapy. Front Endocrinol (Lausanne) 2018;9:69. doi: 10.3389/fendo.2018.00069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Merrilees MJ, Hankin EJ, Black JL, Beaumont B. Matrix proteoglycans and remodelling of interstitial lung tissue in lymphangioleiomyomatosis. J Pathol. 2004;203:653–660. doi: 10.1002/path.1577. [DOI] [PubMed] [Google Scholar]
- 8.Iozzo RV, Schaefer L. Proteoglycan form and function: a comprehensive nomenclature of proteoglycans. Matrix Biol. 2015;42:11–55. doi: 10.1016/j.matbio.2015.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Iozzo RV, Sanderson RD. Proteoglycans in cancer biology, tumour microenvironment and angiogenesis. J Cell Mol Med. 2011;15:1013–1031. doi: 10.1111/j.1582-4934.2010.01236.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Choi Y, Chung H, Jung H, Couchman JR, Oh E-S. Syndecans as cell surface receptors: unique structure equates with functional diversity. Matrix Biol. 2011;30:93–99. doi: 10.1016/j.matbio.2010.10.006. [DOI] [PubMed] [Google Scholar]
- 11.Munesue S, Kusano Y, Oguri K, Itano N, Yoshitomi Y, Nakanishi H, et al. The role of syndecan-2 in regulation of actin-cytoskeletal organization of Lewis lung carcinoma-derived metastatic clones. Biochem J. 2002;363:201–209. doi: 10.1042/0264-6021:3630201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Mytilinaiou M, Nikitovic D, Berdiaki A, Kostouras A, Papoutsidakis A, Tsatsakis AM, et al. Emerging roles of syndecan 2 in epithelial and mesenchymal cancer progression. IUBMB Life. 2017;69:824–833. doi: 10.1002/iub.1678. [DOI] [PubMed] [Google Scholar]
- 13.Hiratsuka S, Nakamura K, Iwai S, Murakami M, Itoh T, Kijima H, et al. MMP9 induction by vascular endothelial growth factor receptor-1 is involved in lung-specific metastasis. Cancer Cell. 2002;2:289–300. doi: 10.1016/s1535-6108(02)00153-8. [DOI] [PubMed] [Google Scholar]
- 14.Beekman JM, Coffer PJ. The ins and outs of syntenin, a multifunctional intracellular adaptor protein. J Cell Sci. 2008;121:1349–1355. doi: 10.1242/jcs.026401. [DOI] [PubMed] [Google Scholar]
- 15.Boukerche H, Aissaoui H, Prévost C, Hirbec H, Das SK, Su Z-Z, et al. Src kinase activation is mandatory for MDA-9/syntenin-mediated activation of nuclear factor-κB. Oncogene. 2010;29:3054–3066. doi: 10.1038/onc.2010.65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kegelman TP, Das SK, Hu B, Bacolod MD, Fuller CE, Menezes ME, et al. MDA-9/syntenin is a key regulator of glioma pathogenesis. Neuro Oncol. 2014;16:50–61. doi: 10.1093/neuonc/not157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.MacLeod AR, Crooke ST. RNA therapeutics in oncology: advances, challenges, and future directions. J Clin Pharmacol. 2017;57:S43–S59. doi: 10.1002/jcph.957. [DOI] [PubMed] [Google Scholar]