Abstract
Acute kidney injury (AKI) is a frequent complication in hospitalised patients and is diagnosed by urinary output and serum creatinine. Serum creatinine is an indirect marker for renal glomerular filtration, but lacks specificity for damage to kidney tissue and the relatively late response to injury precludes early recognition of AKI. Timely diagnosis of kidney injury using biomarkers that provide information about the aetiology of kidney injury is an unmet clinical need. To overcome the suboptimal performance of serum creatinine, injury biomarkers have been proposed that predict AKI in diverse clinical settings. The clinical performance of these markers is considered moderate due to the lack of specificity for kidney tissue or the underlying injury mechanisms, poor test specificity and confounding by interventions or comorbidities. Hence, it is not unequivocally beneficial to implement current kidney injury biomarkers in the clinical laboratory for diagnostic purposes. In this article we review biomarkers that might fulfil AKI-related unmet clinical needs in the academic hospital setting.
Introduction: Timely Diagnosis of Kidney Injury is an Unmet Clinical Need
AKI is a frequent clinical complication that contributes significantly to patient morbidity and mortality in the hospitalised population.1 Up to 20–30% of patients who undergo elective cardiac surgery develop AKI within two days of intervention.2,3 About 5–6% of all critically ill patients admitted to an intensive care unit (ICU) develop AKI.1,4 Particularly, AKI is diagnosed in 40–50% of the septic patients at the ICU.4,5 In a tertiary care centre, AKI occurs in various clinical settings as patients are generally exposed to multiple risk factors for development of AKI, such as nephrotoxic drugs and/or major surgery.6 Therefore, in our tertiary care setting (with expertise in oncology, cardio-thoracic surgery and organ transplantation) timely diagnosis of AKI is of special interest.
Clinically, AKI is recognised by reduced urine output (e.g. urine output <0.5 mL/kg/h for 6 h) and an increase in serum creatinine values (e.g. increase ≥26.5 μmol/L within 48 h).7 Early diagnosis of hospital-acquired AKI is of significance because sustained injury may result in irreversible loss of function and/or chronic kidney disease (CKD) with the risk of end-stage renal disease (ESRD) and need of renal replacement therapy (RRT) (either dialysis or kidney transplantation).8
In contrast to serum creatinine, urinary kidney injury markers may provide insight in the location, severity and aetiology of injury.9 Several biomarkers for kidney injury have been evaluated in clinical trials in past years, such as kidney injury molecule-1 (KIM-1), neutrophil gelatinase-associated lipocalin (NGAL), interleukin-18 (IL-18), liver-type fatty acid-binding protein (L-FABP), N-acetyl-β-D-glucosaminidase (NAG), tissue inhibitor of metalloproteinase-2 (TIMP-2) and insulin-like growth factor-binding protein 7 (IGFBP7). These markers have been proposed as promising predictors of AKI occurrence to overcome the delayed response of serum creatinine. In addition, in the clinical setting, sequential monitoring of injury markers may help to identify patients with hospital-acquired AKI in order to prevent further or irreversible kidney damage.
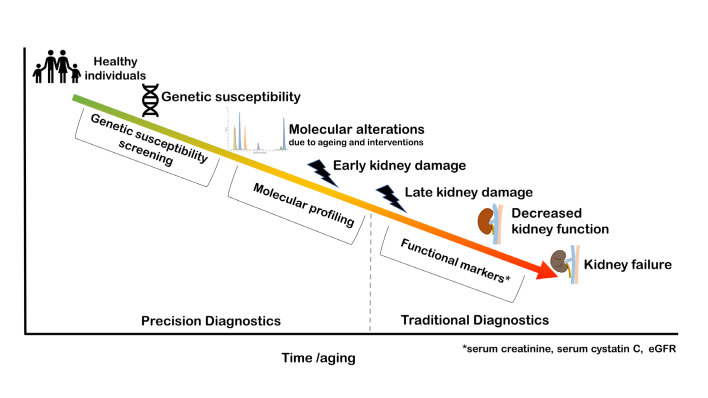
As demonstrated in the conceptual Figure 1, an ideal biomarker recognises molecular alterations and early kidney injury during ageing. Theoretically, genetic screening would provide a measure of susceptibility for developing kidney injury and disease, while molecular alterations revealed in proteomic or metabolomic studies may have the capability to recognise early damage and provide insight in the pathophysiology of early stage kidney disease. Together, this would allow for biology-driven precision diagnostics, which is a paradigm shift compared to the currently practiced traditional medicine, based on average patient populations.
Figure 1.
Conceptual illustration of precision diagnostics and traditional diagnostics in recognition of kidney injury and disease. Individuals may have genetic susceptibility for kidney injury development during life. Molecular alterations may occur during aging and in response to interventions and these may be recognised by molecular profiling via omics (e.g. metabolomics, peptidomics and proteomics). Precision diagnostics includes the early and precise recognition of genetic susceptibility and molecular alterations. Traditional diagnostics typically recognises late stage kidney damage and decreased kidney function through functional markers, such as serum creatinine, cystatin C and eGFR.
A perfect medical test for kidney injury combines high clinical sensitivity with specificity in detecting kidney injury while discriminating kidney injury from other clinical conditions that affect urinary output. In addition, the biomarkers kinetics in urine and the timing of biomarker testing should fit its intended use. Ideally, biomarker concentrations follow the clinical course of the patient, in a concentration-response manner, enabling non-invasive patient monitoring of kidney injury. For biomarker application in an academic hospital, a medical test should have additional value in clinical decision making aiming to improve patient outcome. Biomarker testing should also be feasible and affordable in medical laboratories of care centres. For test implementation into the medical laboratory, the test needs to be compliant with clinical chemistry standards, such as ISO15189:2012. The Test Evaluation Working Group of the European Federation of Clinical Chemistry and Laboratory Medicine (EFLM) has presented a framework for guidance in test evaluation, taking into account the proposed purpose and role of a test in care pathways.10,11
To date, AKI biomarker research has been focused on biomarker assessment for prediction of AKI in specific patient populations in clinical studies. Less attention has been paid to the clinical utility of AKI biomarkers in the setting of an academic hospital. In this non-exhaustive review, we describe the pathophysiology of AKI, biological characteristics of most clinically assessed AKI biomarkers12 and requirements for kidney injury biomarker implementation in the clinical laboratory. We focus only on AKI biomarkers present in easily accessible urine. Several reviews cover the multifactorial pathophysiology of AKI,13–15 so only the major concepts are summarised here to allow understanding of the role of potential biomarkers in AKI. Although it has our interest, investigation of the response of different biomarkers in different AKI aetiologies is beyond the scope of this review. For scientific literature collection, MEDLINE was searched using PubMed up to October 2018. Systematic evaluation and meta-analysis of the clinical performance of urine AKI biomarkers is beyond the scope of this review and can be found elsewhere.16–23
Pathophysiology of Kidney Injury
Aetiology
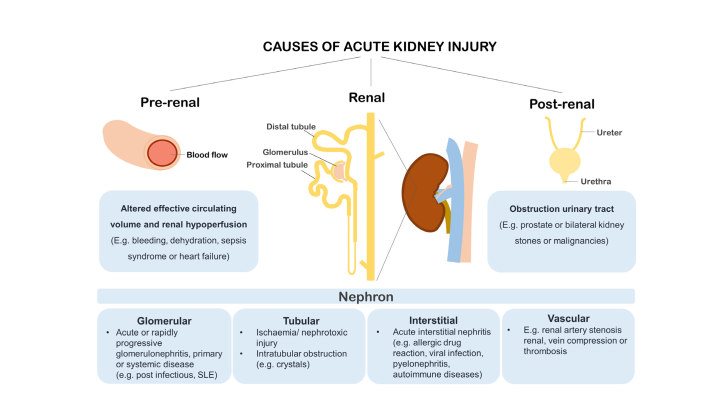
AKI is a clinical syndrome characterised by a rapid decline in kidney function that can have pre-renal, renal and post-renal causes (Figure 2). Commonly, AKI is induced by a change in systemic blood flow resulting in reduced kidney perfusion and glomerular filtration rate.24 Typical conditions that result in altered renal perfusion are sepsis syndrome, exposure to vasoconstrictive agents or nephrotoxic agents or procedure-related ischaemia and reperfusion. Rapid kidney function decline may initially be caused by haemodynamic changes, but kidney function can be restored by feedback via the renin-angiotensin system (RAS). In physiological conditions RAS regulates blood flow, however, abrupt and prolonged disruption of renal perfusion results in ischaemic conditions and AKI with or without structural tissue damage.24 Alternatively, direct injury to renal cells may be caused by inflammation or cytotoxicity. Here, we first address alterations in renal perfusion, followed by AKI caused by direct cellular injury.
Figure 2.
Clinical paradigm of AKI with pre-renal, renal or post-renal causes. AKI with a pre-renal cause, due to altered effective circulating volume, such as bleeding, dehydration and/or heart failure leading to reduced renal perfusion, is most prevalent in the hospitalised setting. AKI with renal origin may be a result from damage to glomerular, tubular, interstitial or vascular nephron compartments. Reduced urinary output in AKI may be caused by post-renal events such as obstruction of the urinary tract by an enlarged prostate, bilateral kidney stones or encasement by malignancy.
Sepsis is the most prevalent cause of AKI as it occurs in almost half of the patients admitted to the ICU.4 Compared to septic patients without kidney complications, those with AKI experience have longer duration of hospitalisation and a higher mortality rate.5 Greater abnormalities in haemodynamic parameters, such as mean heart rates and central venous pressure can be observed in patients with AKI.5 While the haemodynamic alterations dominate AKI in sepsis, inflammatory mechanisms are also involved in sepsis-induced AKI.25 In sepsis, leukocytes pass the glomerulus, release pro-inflammatory cytokines and may contribute to renal tubular epithelial cell injury.26 AKI as a complication after elective cardiac surgery is associated with low-flow and low pressure non-pulsatile perfusion, often resulting in renal hypoperfusion. The cardio-pulmonary bypass time and cross-clamp time during surgery, which can be considered as ischaemia time, are valuable parameters associated with AKI.27,28 Likewise, once blood flow is restored after surgery, reperfusion injury may occur.24 During ischaemia/reperfusion injury (IRI), cellular processes including inflammation and oxidative stress may contribute to the development of AKI.24
Hospital acquired AKI can also be induced by medication or diagnostic agents, especially when agents are administered in a higher dose and intravenously. Medication may have direct cytotoxic effects on renal cells but may also affect renal perfusion. For example, renal perfusion can be reduced by afferent vasodilation by non-steroidal anti-inflammatory drugs (NSAIDs). Particularly in critical haemodynamic conditions and/or in combination with angiotensin-converting-enzyme inhibitors (ACEi) that impair efferent vasoconstriction, renal perfusion can be strongly reduced.29 Other examples are the calcineurin inhibitors tacrolimus and cyclosporine, which are widely prescribed immunosuppressive agents for patients with a kidney allograft. Calcineurin inhibitors induce vasoconstriction by stimulation of the RAS and increase blood pressure in a dose-response dependent manner.30,31 Although calcineurin inhibitors initially induce functional changes with reversible nephrotoxicity,32 long-term exposure results in permanent nephron damage.33–35
Besides the effects through alterations in renal perfusion, medication may also have direct cytotoxic effects on renal cells. For example, chemotherapy including cisplatin administered by infusion typically results in rapid kidney function decline. Cisplatin is primarily cleared by the kidneys resulting in relatively toxic concentrations in the proximal tubular epithelial cells.36 The cellular uptake of cisplatin is facilitated by the presence of kidney-specific organic cation transporters (OCTs)37 that secrete cationic drugs in the epithelial lining of the proximal tubules, causing DNA damage resulting in apoptosis or necrosis.36 Other nephrotoxic agents with direct toxic effects are for example aminoglycoside antibiotics, such as streptomycin and gentamicin. Aminoglycosides are partially cleared by the kidneys, accumulate in the tubules and induce cell death by both apoptosis and necrosis of tubular epithelial cells.38,39
Biological Pathways in Kidney Injury
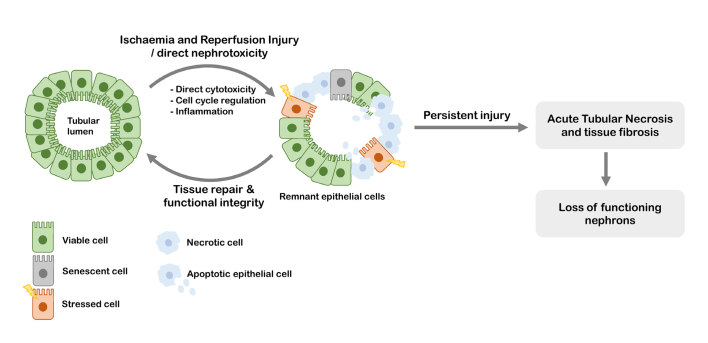
There is ample literature describing molecular biological pathways involved in the onset and progression of kidney injury,13,14,40–42 however, because of the multi-factorial nature of the disease, the molecular changes in the cell that occur during kidney injury are not completely understood. Typically, however, cellular stress is caused either by sepsis, ischaemia or cellular toxicity in the proximal tubular cells, the tubular cells of Henle’s loop or the distal tubular cells. Proximal tubule cells have a high metabolic rate and as such require large amounts of oxygen for the production of ATP.13 It is suggested that the altered renal perfusion results in decreased renal ATP concentrations, which in turn results in cellular stress and loss of structure. Irreversible damage to the kidneys may occur through different processes: cells may die either through apoptosis or necrosis,43 or lose their function by becoming senescent (Figure 3).44
Figure 3.
The epithelial linings in the kidney tubules are prone to ischaemic and reperfusion injury as well as direct nephrotoxicity. These conditions induce inflammation and alterations in the cell cycle, which may result in cell damage, apoptosis or necrotic cell death. Remnant viable epithelial cells can proliferate and thereby enable tissue repair. When repair mechanisms are insufficient, persistent injury results in acute tubular necrosis with tubular atrophy and interstitial fibrosis.
Initially, intra- and extra-cellular stress stimuli induce renal cell cycle arrest. In response to cell injury, cell cycle arrest is a protection mechanism against further cell damage and uncontrolled proliferation, as repair mechanisms can restore cell damage before cell growth (G2-phase) and division (M-phase). Cell cycle arrest is mediated by cyclin-dependent kinases, such as p21, p16 and p53, and is induced by intracellular (e.g. DNA damage) and/or extra-cellular (e.g. via integrin signaling) stimuli. Cell senescence, a state in which cells can no longer proliferate but remain metabolically active, occurs after p21-mediated cell cycle arrest.45 With respect to AKI, ischaemia-induced kidney injury is associated with elevated expression of p21, cell growth arrest and senescence.13,45 As senescent cells are metabolically active, they produce pro-inflammatory cytokines and extra cellular matrix (ECM) degrading enzymes, such as matrix metalloproteinases.46
While moderate cellular stress often results in senescence, severe stress may result in apoptosis or necrosis. Apoptosis may be induced through the intrinsic pathway, in which cellular stress leads to mitochondrial membrane permeability, resulting in release of apoptogenic factors, which eventually activate caspase 9.13 Alternatively, the extrinsic pathway may be activated by ligation of death receptors which activate caspase 8.47 Both pathways have been implicated in AKI; e.g. in a mouse model of sepsis-induced AKI, apoptosis was shown to be mediated by TNF-α and its death receptor TNFR148 and in humans it was shown that two key apoptogenic factors Bax and Bak are involved in ischaemia-induced AKI.47,49
Histological evidence for necrosis is typically found in proximal tubules, but minor events may also be observed in the distal tubules. Necrosis is characterised by swelling of the cells secondary to Na+ and Ca2+ influx, followed by the breakdown of the plasma membrane, resulting in the release of immunogenic damage-associated molecular patterns (DAMPs).50 DAMPs often induce an inflammatory response, which intensifies both tissue injury as well as tissue regeneration.
Inflammation is a key process that contributes to kidney injury but also enables tissue repair after ischaemic conditions. Injured cells activate the innate immune system as DAMPs derived from necrotic cells further induce local inflammation in the kidneys.51 Leukocytes, especially macrophages, infiltrate the tubule by passing the glomerulus and release pro-inflammatory cytokines. In inflammatory conditions, the proliferation of remnant tubular epithelial cells is a reaction mechanism for tissue repair which may be mediated by macrophages.13,52 However, during prolonged ischaemia or exposure to nephrotoxic compounds, this repair mechanism may be insufficient and ultimately results in tissue fibrosis. Inflammation and cell cycle arrest are both characteristics of kidney fibrosis, the process of ECM deposition and scar tissue formation, that contributes to irreversible nephron function loss.53
So far, AKI biomarkers have been thought to fall roughly into three categories: classical biomarkers for kidney function, such as serum creatinine and urinary output, which form the basis for the current Kidney Disease Improving Global Outcome (KDIGO), Acute Kidney Injury Network (AKIN) and Risk, Injury, Failure, Loss, End-Stage (RIFLE) criteria; inflammation-related biomarkers (e.g. KIM-1 and IL-18) and biomarkers that might reflect cell cycle arrest (e.g. IGFBP7 and TIMP-2).
AKI Diagnosis, Current Markers and Tests
AKI Diagnosis Based on Serum Creatinine and Urinary Output
Currently, diagnosis and classification of AKI is typically performed according to consensus criteria based on serum creatinine and/or urinary output, e.g. according to the KDIGO, AKIN or the RIFLE criteria.7,54,55 The KDIGO criteria define several conditions that affect the kidney: AKI, CKD and Acute Kidney Diseases and Disorders (AKD). Whereas AKI is defined based on serum creatinine increase (within 2–7 days) and urinary output, AKD and CKD are based on glomerular filtration rate (GFR) <60 mL/min/1.73 m2 for <3 months or >3 months, respectively. The term AKD also includes patients with a decrease in GFR by ≥35% or an increase in serum creatinine by >50% for <3 months.
Urinary output and serum creatinine, as measures of kidney injury, have a couple of disadvantages. Urinary output and serum creatinine are functional markers, which, due to the functional renal reserve (up to ~20–40% in healthy kidneys), only alters upon significant kidney function loss.56 Typically, this occurs in a late phase of kidney injury, and urinary output and serum creatinine therefore does not allow for early recognition of AKI in stages where damage could still be halted or reversed.57 Another disadvantage of the use of serum creatinine and urinary output, is that they only roughly estimate glomerular filtration function, but do not provide insight in the type of kidney injury, such as injury with pre/post renal cause or intrinsic organ damage (Figure 2). Besides, precise measurement of urinary output may not be feasible in clinical practice, although a decrease in urinary output may be noticed. Serum creatinine may also be affected by muscle composition and turnover, diet, drug interactions, volume of distribution and physical activity, which are typically altered during hospital stay.57–60
Alternatively to serum creatinine, serum/plasma cystatin C is a commonly used marker for the GFR. Cystatin C (16 kDa) is an extracellular cystatin protease inhibitor widely expressed through the human body and consistently produced.61–65 This small protein is freely filtered at the glomerulus and is cleared by reabsorption in the proximal tubule and excretion in urine. In the clinical laboratory, serum cystatin C is typically determined when serum creatinine measurement is likely to be confounded, for example by extremes in dietary protein intake, muscle mass or assay interferences.63,64,66 Cystatin C in serum has been shown to be less affected by gender, muscle mass and diet and was shown to be more sensitive for kidney function monitoring.67–69 While cystatin C in serum/plasma has been used as a functional marker for glomerular filtration, cystatin C in urine has is also described as a measure for tubular dysfunction in AKI.70–72 Next to the automated routine assays available for serum cystatin C, multiple validated immunoassay based methods for determination of urinary cystatin C have been developed for routine chemical analysers.61,71,73
Inflammation Markers
As previously described, inflammation is one of the characteristics of cellular response in AKI. Therefore, it may be expected that proteins involved in the inflammatory response could provide markers that indicate AKI at an earlier stage. Indeed, several inflammation related markers for AKI have recently been described. KIM-1 (39 kDa) is a transmembrane glycoprotein which is most well-known for its immune regulatory function in response to viral infection.74 KIM-1 is up-regulated in renal cells after injury and plays a role in phagocytosis of apoptotic bodies and necrotic debris.75,76 By immunohistochemistry staining, KIM-1 was found on the brush border of tubular epithelial cells and not on glomeruli in renal allograft biopsies.77,78 In urine, levels of KIM-1 were increased in patients with ischaemia induced acute tubular necrosis, a pathological phenotype underlying AKI diagnosed by histologic examination of kidney tissue, compared to patients with CKD.79 KIM-1 was found to be a promising biomarker for AKI in a prospective study including patients who underwent coronary artery bypass grafting (CABG), as urinary excretion of KIM-1 was predictive for development of AKI 12–24 h after the intervention (AUC ROC = 0.70–0.73).27
Another well-described inflammation marker is NGAL (23 kDa).80–82 NGAL secreted in urine can be in free form (monomer, dimer or trimer) or complexed with metalloproteinase-9 (MMP9).83,84 The main functions of NGAL are the regulation of the availability of Fe3+ and the inhibition of bacterial growth.81 In line with its bacteriostatic function, NGAL is mainly expressed in tissues regularly exposed to pathogens, such as the trachea, lungs and colon.81,82 In urinary tract infections, increased expression and excretion of NGAL in the urinary tract have been found as mechanisms for bacterial clearance by acidifying urine to limit bacterial growth.85 With respect to kidney injury, elevated expression of NGAL was found in renal tubular cells during inflammation81 and ischaemic injury.81,86 In murine models and cultured human proximal tubule cells, NGAL mRNA was up-regulated in cells that were undergoing proliferation and regeneration in ischaemic conditions.86 In the kidney, NGAL is localised in the cytoplasm of renal tubular cells in the cortex as well as the renal medulla.77,87 The up-regulated NGAL in tubular injury is believed to have a protective role in ischaemic conditions, which includes the inhibition of apoptosis and the stimulation of cell proliferation.14,88,89 With respect to its clinical evaluation, NGAL was considered a promising biomarker for AKI in patients who underwent cardiac surgery with cardiopulmonary bypass. Specifically, urinary NGAL levels 2 h after cardiopulmonary bypass were predictive for AKI (sensitivity = 82%, specificity = 90%, cut-off = 100 ng/mL NGAL).90
IL-18 (22 kDa) is a well-known pro-inflammatory cytokine, which is secreted by macrophages upon cleavage by caspase-1. Elevated urinary levels of IL-18 are associated with occurrence of AKI.20,91,92 In a study including patients who underwent cardiac surgery, urinary IL-18 was increased after 4–6 h after cardiopulmonary bypass and the performance in diagnosing AKI defined by the area under the ROC was 61–75%, depending on the timing of IL-18 measurement.93 Moreover, IL-18 was found associated with acute tubular necrosis, and not with eGFR defined CKD.92
Cell Cycle Arrest Markers
Cell cycle arrest is probably one of the first processes activated upon cellular stress in the kidney. Therefore, markers that could indicate cell cycle arrest of kidney tubule cells would likely provide early and specific markers for AKI. Indeed, recently two proteins that are associated with cell cycle arrest have been identified as promising markers for the detection of AKI. Proteins IGFBP7 (29 kDa) and TIMP-2 (24 kDa) are secreted proteins widely expressed through the human body,94 including in the proximal and distal tubule in the renal cortex.95 Both TIMP-2 and IGFBP7 are multifunctional proteins involved in various biological pathways.96 Among other functions related to the inhibition of matrix metalloproteinases (MMPs), TIMP-2 binds to α3β1 integrin inducing synthesis of cyclin-dependent kinase inhibitor p27 resulting in G1 cell growth arrest,96,97 while IGFBP7 induces p53 mediated cell cycle arrest and apoptosis.98,99 Urinary TIMP-2 and IGFBP7 measured directly after kidney transplantation have been shown to be predictive for delayed graft function.100 Interestingly, TIMP-2 and IGFBP7 have both been described in scientific literature as biomarkers for pathological conditions other than kidney injury, such as various malignancies.101–103
The product of TIMP-2 and IGFBP7 concentrations, [TIMP-2]*[IGFBP7], was introduced as a biomarker for AKI development in septic patients and patients who undergo cardiac surgery, in the point-of-care NephroCheck™ test (Astute Medical, San Diego).104 [TIMP-2]*[IGFBP7] has been described as a promising biomarker, since it performed better in the prediction of AKI in the ICU than other biomarkers, including NGAL, KIM-1, IL-18 and L-FABP (Sapphire study). In a follow-up study, combined data from the so-called Sapphire and Topaz multi-centre studies confirmed that urinary [TIMP-2]*[IGFBP7] was predictive for the development of moderate to severe AKI in patients who underwent major surgery.105
Other Biomarkers for Kidney Injury
Next to the inflammatory and cell cycle arrest markers, other mechanisms have been suggested for biomarkers L-FABP and NAG in kidney injury. L-FABP (14–15 kDa) was introduced as a marker for CKD rather than a marker for AKI.106 Urinary concentrations of L-FABP correlate with the degree of proteinuria and it has been proposed that L-FABP could be used for monitoring progression of glomerular disease.106 L-FABP is a protein highly expressed by hepatocytes in the liver and binds fatty-acids and cholesterol. In the kidney, L-FABP is expressed in the cytoplasmic regions in the proximal tubule and in the tubular lumen.107 Although the role of L-FABP in kidney injury remains to be unravelled, an anti-inflammatory and protective role of L-FABP in ischaemia has been suggested in which L-FABP mediates transport of accumulated cytotoxic levels of intracellular lipid peroxidation products to the urinary lumen in conditions of ischaemia.107
NAG (~130–150 kDa) is a widely expressed lysosomal enzyme responsible for the breakdown of glycoproteins.108 In the rat kidney, NAG is localised in both the proximal and distal tubule.109 Abnormal urinary NAG concentrations, have been found in patients with renal disease, with ischaemia-reperfusion injury, hypertension or after exposure to nephrotoxic agents.108,110,111 In addition, NAG has also been proposed as a marker for tubular dysfunction in type 2 diabetes.112,113 Interestingly, in patients with type 2 diabetes, urinary NAG is correlated to urinary albumin levels.112
Mechanism Underlying Increased Urinary Concentrations of Protein Biomarkers in AKI
For all discussed biomarkers, elevated urinary concentrations were associated with acute or chronic kidney injury. Yet, the molecular mechanism for elevated excretion of such biomarker proteins via urine is still unclear. For example, TIMP-2 and IGFBP7 are expressed in tubular cells, in specific distal and proximal compartments of the human nephron, and increased in situ expression and secretion of these markers has been proposed as the mechanism behind elevated urinary excretion of these proteins in ischaemic conditions.95 Recently, the underlying biological mechanism of increased urinary levels of TIMP-2 and IGFBP7 in kidney injury was studied in detail using mouse models of agent-induced and ischaemia-induced AKI.114 While urinary levels of TIMP-2 and IGFBP7 were increased in conditions of AKI, no increased expression of TIMP-2 and IGFBP7 was found in tubular cells in the renal cortex. It was emphasised that elevated urinary levels of TIMP-2 and IGFBP7 in AKI are a result of decreased glomerular permselectivity and increased tubular leakage, by either impaired reabsorption of glomerular filtered proteins or increased extracellular release by tubular cells.114 The proposed impaired reabsorption of proteins is in agreement with previously observed high concentrations of urinary NGAL and cystatin C in proteinuria, as a result of competition for reabsorption in the proximal tubules.115 Decreased permselectivity of the glomerular apparatus may be a result of direct damage to the glomerular basement membrane or increased intraglomerular hydraulic pressure by renal perfusion.116
The mechanisms underlying increased excretion of TIMP-2 and IGFBP7 in urine –decreased glomerular permselectivity and proximal tubular leakage– are likely to apply also to other kidney injury biomarkers. The low molecular weight proteins KIM-1, NGAL, IL-18, L-FABP, TIMP-2 and IGFBP7 are not specifically expressed in kidney tissue and are likely to pass the glomerular slit membrane, especially in conditions of decreased permselectivity of the glomerular apparatus. Consequently, KIM-1, NGAL, IL-18, L-FABP, TIMP-2 and IGFBP7 may be prone to confounding by inflammatory and cell cycle related processes being systemic or originating from different tissues in the human body.
Characteristics of AKI Biomarker Tests towards Translation to the Clinical Laboratory
Desirable Test Characteristics for Timely Diagnosis of Kidney Injury
Translation of promising biomarkers into actual medical tests is a difficult trajectory in which many markers fail. Examples of successful biomarker translation to the medical laboratory include cardiac troponin T and troponin I, which are injury markers specifically expressed in heart tissue, that indicate heart muscle damage. Nowadays, the high-sensitivity cardiac troponin test is a cornerstone test for the diagnosis of non-ST segment elevation myocardial infarction (NSTEMI) in patients with symptoms of acute coronary syndrome.117
Recently, a framework for medical test evaluation has been outlined by the European Federation of Clinical Chemistry and Laboratory Medicine (EFLM).10 Specifically, it addresses the importance of having a well-defined clinical need as starting point, followed by predefining the analytical and clinical performance needed for the intended use of the test. Analytical performance should be compliant with the intended use of the test, which could be early diagnosis (e.g. recognition of AKI after major surgery), patient monitoring (e.g. during periods of exposure to nephrotoxic compounds) or prognosis (e.g. AKI related outcomes such as RRT). For the purpose of timely diagnosis of kidney injury in the ICU or after major surgery, high clinical and analytical sensitivity and specificity to detect minor changes in concentration are required. According to the EFLM Test Evaluation framework,10 clinical performance specifications should be predefined and selected based on the intended use and benefit-harm ratio for patients. For the early detection of AKI after cardio-thoracic surgery, high sensitivity is required at the expense of lower specificity. One could imagine that if AKI biomarkers would be used in therapy monitoring, e.g. in patients receiving nephrotoxic agents at an oncology department, high specificity of a biomarker test is required for clinical decision making (dose adjustments, therapy switch etc.).
For biomarker based AKI risk prediction, a biomarker must have additional value to risk prediction based on clinical factors, including co-morbidities such as pre-existing CKD, proteinuria, diabetes and medication.118 The perfect biomarker or biomarker panel could discriminate between stages of kidney injury and is specific for kidney tissue (e.g. glomerular, tubular, vascular or interstitial topography, Figure 2). Furthermore, an optimal injury marker should have discriminating performance 2–6 h after the intervention to guide actionable AKI prevention strategies. Once the clinical unmet need is defined, and the potential role of the test is identified, the desired analytical and clinical performance, the clinical effectiveness, cost-effectiveness and the broader impact of the new medical test should be considered.119 In the next paragraphs, kidney injury biomarkers NGAL, KIM-1, IL-18, L-FABP, NAG, TIMP-2 and IGFBP7 will be assessed in relation to these Test Evaluation components.
The Clinical Performance of Urinary Kidney Injury Biomarkers has been Studied for Timely AKI Diagnosis
The strength of association between the kidney injury biomarkers and the clinical outcome AKI is typically assessed by the ability to predict kidney injury by means of the discriminating performance (test sensitivity and specificity, areas under the ROC curves and likelihood ratios). The performance of AKI biomarkers in clinical studies have predominantly been assessed for timely diagnosis of AKI in the ICU or after cardio-thoracic surgery. Typically, the predictive performance of AKI biomarkers is evaluated against the filtration marker serum creatinine defined occurrence of AKI as the primary endpoint. Patients are either classified in the AKI group or non-AKI group based on (minor) changes in serum creatinine with respect to an available baseline value, following the KDIGO criteria. However, serum creatinine is an inappropriate gold standard for AKI and therefore may be inadequate as the primary endpoint.116 Alternative endpoints for AKI biomarker evaluation are, for example, AKI severity and long-term renal function. In addition, non-creatinine endpoints, such as the need for RRT or mortality, may be used.120,121 In a recent meta-analysis, urine biomarkers NGAL, IL-18, cystatin C and [TIMP-2]*[IGFBP7] were found to be predictive for RRT in AKI.19
KIM-1, NGAL, IL-18, L-FABP and [TIMP-2]*[IGFBP7] have been extensively evaluated for their clinical performance in predicting AKI before serum creatinine rise. Test characteristics, including test sensitivity and specificity with a concentration cut-off, have been summarised in systematic reviews and meta-analyses.16–18,20,23,122–124 So far, a meta-analysis of test characteristics in predicting AKI is not available for NAG.
In Table 1, pooled test parameters sensitivity and specificity derived from meta-analyses are summarised, to give an indication of the diagnostic value of the kidney injury biomarkers. KIM-1 has the best characteristics by means of both test sensitivity (74%) and test specificity (86%). Overall, test specificity of NGAL (47%) and TIMP-2*IGFBP7 (57%) is poor and in individual studies, the concentration cut-offs have been set in a manner so that test sensitivity (88% and 84%) outweighs the test specificity. Various cutoff concentrations have been used in the assessed individual studies, which hampers direct interpretation of the pooled test sensitivity and specificity, which is, for example, the case for IL-18.20 In meta-analyses evaluating the clinical performance of [TIMP-2]*[IGFBP7] (NephroCheck™), the heterogeneity in (i) the cut-off points among the included studies, (ii) studied populations, (iii) timing of sampling were the main limitations for examining the diagnostic value.17,122,123 In case of the NephroCheck™, the cut-off value of 0.3 (unit = (ng/mL)2/1000, AKIRisk™) score results in a high rate of false positives and thus poor specificity (~0.50) and positive predictive value (PPV) (~0.20). Therefore, a cut-off value of 2 may be used to improve test specificity (~90%).105,125–128
Table 1.
The availability of information with respect to analytical test specifications, clinical performance, clinical effectives and cost-effectiveness of kidney injury biomarkers in scientific literature. Pooled test sensitivity and specificity from meta-analyses are given as indication for the clinical performance.
| Biomarker | Analytical specifications | Clinical performance | Clinical effectivity | Cost-effectiveness | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Avail. | Total CVA | Ref. | Avail. | Sensitivity (%) | Specificity (%) | Ref. | Avail. | Ref. | Avail. | |
| NGAL | + | 1.2–5.2% | 151, 153–155 | + | 88 | 47 | 18 | − | − | |
| [TIMP-2]x [IGFBP7] | + | 9.1–18% | 177 | + | 84 | 57 | 123 | + | 141,142 | +/−‡ |
| KIM-1 | +/− | * | + | 74 | 86 | 124 | − | − | ||
| IL-18 | +/− | * | + | 51 | 74 | 20 | − | − | ||
| L-FABP | +/− | * | + | 75 | 78 | 23 | − | − | ||
| NAG | +/− | * | +/−† | 80 | 65 | 110 | − | − | ||
Data are available (+), insufficient (+/−) or not available (−) CVA = Analytical variation
Research-use only immunoassays; no diagnostic tests with analytical specifications available;
Values derived from a single study (no meta-analysis);
Limited information available with respect to cost-effectiveness for use in a hospital population.
The diagnostic performance of a test is dependent on the timing of urine sample collection for biomarker measurement.129 The optimal timing of biomarker measurement depends on the intended use and the concentration profile over time. For early diagnosis of AKI in the ICU, rapid response in urine is desired, while for patient monitoring slow biomarker kinetics may be preferred. Although there is discrepancy in biomarker kinetics among studies, urinary NGAL typically peaks rapidly after major surgery (~2 h), TIMP-2, IGFB7, IL-18 and L-FABP have a delayed response (~6 h) and KIM-1 has a late optimum response (~12 h).9,130 Consequently, the timing of urine collection is critical for interpretation and serial sampling may be desired.
Since biomarkers in urine have high within-subject variation, one concentration cut-off as medical decision point together with individual test results can be inconclusive (diagnostic uncertainty/so-called ‘grey zone’).131 Alternatively, serial sampling may support biomarker interpretation and here the reference change values (RCVs) can be used in combination with conventional population reference intervals.132,133 The RCV is a fold change that indicates the biomarker change that is likely to be of clinical significance. If the change in test results of two or more measurements is lower than the RCV, the observed biomarker concentration increase or decrease may be due to biological and analytical variation, and the result is likely to be noise only. Examples of RCVs for NGAL and TIMP-2 are shown in Table 2. Serial biomarker measurements and the RCV can support clinical biomarker interpretation and this may be of interest for the purpose of biomarker guided patient monitoring of graft function or monitoring of kidney injury during therapy with nephrotoxic agents. Nevertheless, one may consider that serial measurements may not be feasible in a clinical setting and there may be lack of time for serial urine collection for the purpose of early diagnosis of AKI.
Table 2.
Performance specifications of biomarkers NGAL and TIMP-2 calculated from previously reported analytical and biological variation data in urine.
| Literature values | Calculated values | ||||||
|---|---|---|---|---|---|---|---|
| CVI (%) | CVG (%) | CVA (%) | Ref. | Index of individuality (CVI/CVG) | TEa (%)* | RCV (%)** | |
| TIMP-2 | 67.7 | 119.7 | 6.0 | 150 | 0.57 | 90 | 188 |
| NGAL | 86.3 | 196.2 | 5.2 | 150 | 0.44 | 125 | 240 |
CVI = within-subject variation, CVG = between-subject variation, CVA = analytical variation, TEa = total allowable error, RCV = reference change value interval.
TEa < k * 0.5 CVI + 0.25(CVI2 + CVG2)1/2; With k = 1.65 (inner 90%, desired TE) and α = 0.05,
RCV = z*21/2(CVA2 + CVI2)1/2, with z = 1.96 (95% probability that the change between results can be explained by biological and analytical variation only)
In summary, the clinical performance of AKI biomarkers is moderate probably due to several major reasons. First, the predictive performance of biomarkers is typically evaluated based on serum creatinine defined AKI, which can be considered as an inappropriate gold standard for AKI.134 Second, the current biomarkers lack specificity for kidney tissue or the underlying injury mechanisms and are prone to confounding by medical interventions and comorbidities. Third, the sample collection, storage and the assays used in the diverse clinical studies have not been standardised. Finally, the timing of (serial) sample collection seems to be critical for optimal predictive performance and is variable among studies.
Clinical Effectiveness: Improve Health Outcomes that are Relevant to the Individual Patient
Timely diagnosis of kidney injury, before serum creatinine rises, is believed to prevent further kidney damage and the incidence of severe AKI and the need of RRT. Therefore, the ultimate improvement of health outcome to the individual patient is the avoidance of irreversible kidney damage and development of ESRD. Other aimed beneficial outcomes of early AKI biomarkers are the shortened ICU and overall hospital stay.
The treatment of already developed AKI remains challenging and the effects of pharmacological interventions have been shown to be limited and inconclusive.135–138 Therefore, AKI management has been focused on the prevention of AKI development. Strategies for the prevention of AKI include: haemodynamic stabilisation (e.g. vascular fluid loading, administration of vasopressor agents or inotropic drugs), minimisation of cardiopulmonary bypass and the dose reduction or avoidance of nephrotoxic agents.139,140
The efficacy of patient management of individuals with high risk for developing AKI has been studied in two prospective studies. In these studies, high-risk AKI patients were identified by the NephroCheck™ biomarker analysis ([TIMP-2]*[IGFBP7] >0.3). The selected high-risk AKI patients were included in either the intervention or control group by randomisation and the observed effects are unrelated to biomarker testing.141,142 The effects of strict patient monitoring – consisting of optimisation of haemodynamic fluid status, avoidance or stopping nephrotoxic agents and preventing hyperglycaemia – on the incidence of AKI in high-risk patients at the ICU were investigated.142 Here, strict monitoring in the intervention group did not affect the incidence of moderate/severe AKI within the first day of ICU admission (primary endpoint) compared to the control group that received standard care.142 In contrast, in a study including high-risk AKI patients who underwent cardiac surgery, strict patient monitoring (including avoidance of nephrotoxic compounds, discontinuation of ACE inhibitors or angiotensin II receptor blockers for 48 h after surgery, haemodynamic monitoring and preventing hyperglycaemia) resulted in a reduced AKI incidence (55.1% intervention group vs. 71.7% control group).141 However, ICU stay, hospital stay or RRT requirement during a follow-up of 90 days was not affected.141 It would be interesting to test the clinical effect (patient outcome and hospital costs) for such an intervention by including patients based on the biomarker results.
Desirable Analytical Performance Specifications
The analytical specifications, including imprecision, inaccuracy and total allowable error, can be based on three tiers: (i) clinical outcome; (ii) biological variation of the measurand;143 and (iii) state-of-the-art/highest analytical performance technically achievable (criteria from the EFLM conferences).144–146 When biological variance data from literature is used to set analytical specifications, the Biological Variation Data Critical Appraisal Checklist (BIVAC) should be used for evaluation of the methodological quality of the study that obtained the biological variance data.147
To our knowledge, no studies have set the analytical performance specifications of biomarker assays on direct or indirect clinical outcome kidney injury today. However, the biological variance of AKI biomarkers in urine in apparently healthy populations and patient populations has been reported.148,149 In a study with apparently healthy subjects, the within-subject variation was substantial (CVI >60%) for urinary NGAL, KIM-1 and IL-18 (N = 9, study not fully BIVAC compliant).148 Moreover, the within-subject variation in patients with CKD (N = 80) was 68% for TIMP-2, 72% for KIM-1, 95% for IL-18 and 66% for albumin in urine (N = 80, values not normalised, BIVAC-compliant study).150 Typically, the within-subject variation of analytes in urine is higher compared to plasma. For example, the within-subject variation of NGAL in urine is 86% compared to 16% in plasma.150 For urinary NGAL, an immunoassay suitable for routine chemistry analysers is available for diagnostic use and the within-day and between-day variance has been studied in more detail.151,152
If the analytical specifications of biomarkers for early detection of kidney injury would be set based on available estimates of within-subject biologic variation (CVI) and the between-subject biologic variation (CVG) – according to Westgard guidelines – the total allowable error (TEa) will be large for kidney injury biomarkers measured in urine. For example, the TEa for urinary TIMP-2 and NGAL measurement, can be calculated based on available biological variation (CVI = 68% and CVG = 120% for TIMP-2 and CVI = 86% and CVG = 196% for NGAL) from a BIVAC compliant study (Table 2).147,150 The TEa obtained of 90% for TIMP-2 and 125% for NGAL is large compared to common clinical chemical laboratory standards (Table 2).
We suggest that the state-of the-art analytical performance tier may be more appropriate for setting the analytical specifications of urinary biomarkers for prediction of AKI. In clinical studies, AKI biomarkers have been measured using research use only ELISA kits, the NephroCheck™ point-of-care test (POCT) for [TIMP-2]*[IGFBP7] or NGAL immunoassays suitable for chemical analysers. For example, state-of-the art analytical performance of NGAL immunoassays for chemical analysers is CVA = 1.2–5.2%.153–155 A novel technique that has recently been introduced in the clinical laboratory for protein quantitation is mass spectrometry (MS). MS is a highly specific technique with multiplexing capability and analysis of individual proteoforms, that has advantages over immunoassays.156,157 Previously, we developed and showed long-term analytical imprecision of 5–6% using a multiplex MS-based test for the absolute quantification of apolipoproteins.158–160 Likewise, a panel of low abundant urine AKI biomarkers could potentially be quantified using a similarly precise, MS-based test.
The use of absolute biomarker concentrations has several issues to be discussed. Reported biomarker concentrations in clinical studies may not be interchangeable, as the diverse immunoassays available have not been harmonised and standardised. Secondly, variance in urinary flow rate contributes to the large biological variance of absolute urinary biomarker concentrations. This variance may be reduced by normalisation of absolute concentrations for urine dilution, using urinary output, urinary creatinine or osmolality.150,161–163 Normalisation of results introduces an extra variable, having its particular disadvantages. For example, oliguria is an easily recognisable parameter for normalisation, but standardised measurement of urinary output may not be feasible in clinical practice. Normalisation of urinary biomarkers by urinary creatinine has become more or less common practice in literature. Normalisation by urinary creatinine assumes constant excretion per volume urine, however urine creatinine excretion is variable and depends on previously described factors, such as muscle mass and diet.57 Hence, urinary creatinine could be considered as an imperfect parameter for normalisation. Osmolality may be an alternative parameter for normalisation and has been used in analysis of metabolites in urine, but its use in urinary protein analysis in the clinical chemistry field remains limited.161,164,165
Cost-effectiveness and Broader Impact
New in vitro diagnostic tests need to guide decision making, be cost-effective and have additional value to patient management for implementation in routine patient care pathways.11,166,167 Moreover, tests need to substitute other laboratory measurements or have significant additional value in the clinical management of kidney injury having incremental benefits over existing clinical pathways.10 The estimated assay costs for kidney injury testing are between $18 and $25 for NGAL, cystatin C, KIM-1, IL-18, L-FABP and $85 for [TIMP-2]*[IGFBP7] using one cartridge for the Nephrocheck™ POCT (cost of reagents only).168
The assessment of cost-effectiveness and broader impact of kidney injury biomarker guided patient management is required,169 but is currently lacking. However, the appropriate use of the Nephrocheck™ has been outlined and it was emphasised that the NephrocheckTM can be used on admission to ICU or in critically ill patients and may be used additionally to other diagnostic tests for kidney injury, such as furosemide testing.170 For the use of the Nephrocheck™ as an add-on test to serum creatinine, the clinical effectiveness needs to have evident additional value to potentially be cost-effective. However, the NephrocheckTM (cut-off 0.3) in the ICU or after major surgery has low specificity. Therefore, most patients (>50%) will be identified as high risk patients for AKI, but a large percentage of positively tested patients (~85%) will not develop AKI (positive predictive value, PPV = 15%).105,171 These numbers indicate a large burden of unnecessary and costly interventions, which should be weighed against the clinical benefits of the test. Specifically, false positive identification of patients with so-called high-risk for AKI may result in unnecessary avoidance of nephrotoxic agents, undermining optimal therapeutic strategies in patient care. If kidney injury testing, timely diagnosis of AKI and prevention treatment lead to reduced severity of AKI and reduced incidence of RRT, then the cost-benefit of kidney injury tests may be favourable.
Recap and Perspectives
AKI is recognised by reduced urinary output and increased levels of serum creatinine, which are both late stage functional markers. There is an unmet clinical need for biomarkers that detect AKI in an early stage and that have the capability to provide insight in the pathophysiology of kidney injury, such as differentiation between AKI with pre-renal cause or renal injury, such as acute tubular necrosis.
Urinary levels of KIM-1, NGAL, IL-18, L-FABP, NAG, TIMP-2 and IGFBP7 have been proposed as biomarkers for AKI in clinical studies and have gained interest for adaptation to clinical use in patient care.172–174 For adoption of biomarkers as part of routine care, improved clinical outcomes due to biomarker guided patient management need to be evaluated in the setting of the biomarkers’ intended use. AKI occurs in various clinical settings that need to be considered to find the verified intended use of a biomarker. Likewise, comorbidities and diverse AKI aetiologies need to be covered in well-designed biomarker-stratified clinical trials. The translation of AKI biomarkers in ‘donation after cardiac death’ kidney taking into account these essentials, is therefore extremely complex and expensive. In addition, a new AKI biomarker test needs to be compared to current practice and we here reflect that a gold standard test for AKI is lacking.
Notwithstanding the promising introduction of these individual proteins as molecular markers of pathological processes relevant to kidney injury, their specificity for kidney injury and their clinical performance seems to be insufficient for clinical implementation. To enhance clinical sensitivity and specificity, combinations of AKI biomarkers could be considered. Indeed, the combination of TIMP-2 and IGFBP7 has been included in a diagnostic test (Nephrocheck™) for the early detection of AKI in patients with acute cardiovascular compromise admitted to the ICU. The clinical performance of this test was superior to the individual markers KIM-1 and NGAL and was less affected by comorbidities.104,175 In-house, we have studied urinary TIMP-2 and IGFBP7 as ischaemia biomarkers in donation after cardiac death (DCD) kidney transplant recipients and concluded that TIMP-2, but not IGFBP7, was a promising biomarker for functionally defined delayed graft function.176
A biomarker panel including multiple kidney injury markers reflecting different pathological processes could increase clinical specificity and potentially even topographically localise pathology along the nephron. A combination panel of contemporary and emerging biomarkers could fulfil the predefined clinical performance specifications needed for early diagnosis of AKI.
Acknowledgement
The Dutch Foundation of Clinical Chemistry provided a Noyons Stipendium (2015) and Roche Diagnostics provided an independent research grant.
Footnotes
Competing Interests: None declared.
References
- 1.Lafrance JP, Miller DR. Acute kidney injury associates with increased long-term mortality. J Am Soc Nephrol. 2010;21:345–52. doi: 10.1681/ASN.2009060636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hu J, Chen R, Liu S, Yu X, Zou J, Ding X. Global Incidence and Outcomes of Adult Patients With Acute Kidney Injury After Cardiac Surgery: A Systematic Review and Meta-Analysis. J Cardiothorac Vasc Anesth. 2016;30:82–9. doi: 10.1053/j.jvca.2015.06.017. [DOI] [PubMed] [Google Scholar]
- 3.Hobson CE, Yavas S, Segal MS, Schold JD, Tribble CG, Layon AJ, et al. Acute kidney injury is associated with increased long-term mortality after cardiothoracic surgery. Circulation. 2009;119:2444–53. doi: 10.1161/CIRCULATIONAHA.108.800011. [DOI] [PubMed] [Google Scholar]
- 4.Uchino S, Kellum JA, Bellomo R, Doig GS, Morimatsu H, Morgera S, et al. Beginning and Ending Supportive Therapy for the Kidney (BEST Kidney) Investigators. Acute renal failure in critically ill patients: a multinational, multicenter study. JAMA. 2005;294:813–8. doi: 10.1001/jama.294.7.813. [DOI] [PubMed] [Google Scholar]
- 5.Bagshaw SM, Uchino S, Bellomo R, Morimatsu H, Morgera S, Schetz M, et al. Beginning and Ending Supportive Therapy for the Kidney (BEST Kidney) Investigators. Septic acute kidney injury in critically ill patients: clinical characteristics and outcomes. Clin J Am Soc Nephrol. 2007;2:431–9. doi: 10.2215/CJN.03681106. [DOI] [PubMed] [Google Scholar]
- 6.Roberts G, Phillips D, McCarthy R, Bolusani H, Mizen P, Hassan M, et al. Acute kidney injury risk assessment at the hospital front door: what is the best measure of risk? Clin Kidney J. 2015;8:673–80. doi: 10.1093/ckj/sfv080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kellum JA, Lameire N, Aspelin P, Barsoum RS, Burdmann EA, Goldstein SL, et al. Kidney disease: Improving global outcomes (KDIGO) acute kidney injury work group. KDIGO clinical practice guideline for acute kidney injury. Kidney Int Suppl. 2012;2:1–138. [Google Scholar]
- 8.Srisawat N, Kellum JA. Acute kidney injury: definition, epidemiology, and outcome. Curr Opin Crit Care. 2011;17:548–55. doi: 10.1097/MCC.0b013e32834cd349. [DOI] [PubMed] [Google Scholar]
- 9.Alge JL, Arthur JM. Biomarkers of AKI: a review of mechanistic relevance and potential therapeutic implications. Clin J Am Soc Nephrol. 2015;10:147–55. doi: 10.2215/CJN.12191213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Horvath AR, Lord SJ, StJohn A, Sandberg S, Cobbaert CM, Lorenz S, et al. Test Evaluation Working Group of the European Federation of Clinical Chemistry Laboratory Medicine. From biomarkers to medical tests: the changing landscape of test evaluation. Clin Chim Acta. 2014;427:49–57. doi: 10.1016/j.cca.2013.09.018. [DOI] [PubMed] [Google Scholar]
- 11.Monaghan PJ, Lord SJ, St John A, Sandberg S, Cobbaert CM, Lennartz L, et al. Test Evaluation Working Group of the European Federation of Clinical Chemistry and Laboratory Medicine. Biomarker development targeting unmet clinical needs. Clin Chim Acta. 2016;460:211–9. doi: 10.1016/j.cca.2016.06.037. [DOI] [PubMed] [Google Scholar]
- 12.Codorniu A, Lemasle L, Legrand M, Blet A, Mebazaa A, Gayat E. Methods used to assess the performance of biomarkers for the diagnosis of acute kidney injury: a systematic review and meta-analysis. Biomarkers. 2018;23:766–72. doi: 10.1080/1354750X.2018.1493616. [DOI] [PubMed] [Google Scholar]
- 13.Basile DP, Anderson MD, Sutton TA. Pathophysiology of acute kidney injury. Compr Physiol. 2012;2:1303–53. doi: 10.1002/cphy.c110041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bonventre JV, Yang L. Cellular pathophysiology of ischemic acute kidney injury. J Clin Invest. 2011;121:4210–21. doi: 10.1172/JCI45161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Makris K, Spanou L. Acute Kidney Injury: Definition, Pathophysiology and Clinical Phenotypes. Clin Biochem Rev. 2016;37:85–98. [PMC free article] [PubMed] [Google Scholar]
- 16.Haase M, Bellomo R, Devarajan P, Schlattmann P, Haase-Fielitz A NGAL Meta-analysis Investigator Group. Accuracy of neutrophil gelatinase-associated lipocalin (NGAL) in diagnosis and prognosis in acute kidney injury: a systematic review and meta-analysis. Am J Kidney Dis. 2009;54:1012–24. doi: 10.1053/j.ajkd.2009.07.020. [DOI] [PubMed] [Google Scholar]
- 17.Jia HM, Huang LF, Zheng Y, Li WX. Prognostic value of cell cycle arrest biomarkers in patients at high risk for acute kidney injury: A systematic review and meta-analysis. Nephrology (Carlton) 2017;22:831–7. doi: 10.1111/nep.13095. [DOI] [PubMed] [Google Scholar]
- 18.Kim S, Kim HJ, Ahn HS, Song JY, Um TH, Cho CR, et al. Is plasma neutrophil gelatinase-associated lipocalin a predictive biomarker for acute kidney injury in sepsis patients? A systematic review and meta-analysis. J Crit Care. 2016;33:213–23. doi: 10.1016/j.jcrc.2016.02.014. [DOI] [PubMed] [Google Scholar]
- 19.Klein SJ, Brandtner AK, Lehner GF, Ulmer H, Bagshaw SM, Wiedermann CJ, et al. Biomarkers for prediction of renal replacement therapy in acute kidney injury: a systematic review and meta-analysis. Intensive Care Med. 2018;44:323–36. doi: 10.1007/s00134-018-5126-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lin X, Yuan J, Zhao Y, Zha Y. Urine interleukin-18 in prediction of acute kidney injury: a systemic review and meta-analysis. J Nephrol. 2015;28:7–16. doi: 10.1007/s40620-014-0113-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Coca SG, Yalavarthy R, Concato J, Parikh CR. Biomarkers for the diagnosis and risk stratification of acute kidney injury: a systematic review. Kidney Int. 2008;73:1008–1. doi: 10.1038/sj.ki.5002729. [DOI] [PubMed] [Google Scholar]
- 22.Huang Y, Don-Wauchope AC. The clinical utility of kidney injury molecule 1 in the prediction, diagnosis and prognosis of acute kidney injury: a systematic review. Inflamm Allergy Drug Targets. 2011;10:260–7. doi: 10.2174/187152811796117735. [DOI] [PubMed] [Google Scholar]
- 23.Susantitaphong P, Siribamrungwong M, Doi K, Noiri E, Terrin N, Jaber BL. Performance of urinary liver-type fatty acid-binding protein in acute kidney injury: a meta-analysis. Am J Kidney Dis. 2013;61:430–9. doi: 10.1053/j.ajkd.2012.10.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wang Y, Bellomo R. Cardiac surgery-associated acute kidney injury: risk factors, pathophysiology and treatment. Nat Rev Nephrol. 2017;13:697–711. doi: 10.1038/nrneph.2017.119. [DOI] [PubMed] [Google Scholar]
- 25.Prowle JR, Bellomo R. Sepsis-associated acute kidney injury: macrohemodynamic and microhemodynamic alterations in the renal circulation. Semin Nephrol. 2015;35:64–74. doi: 10.1016/j.semnephrol.2015.01.007. [DOI] [PubMed] [Google Scholar]
- 26.Zarbock A, Gomez H, Kellum JA. Sepsis-induced acute kidney injury revisited: pathophysiology, prevention and future therapies. Curr Opin Crit Care. 2014;20:588–95. doi: 10.1097/MCC.0000000000000153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Elmedany SM, Naga SS, Elsharkawy R, Mahrous RS, Elnaggar AI. Novel urinary biomarkers and the early detection of acute kidney injury after open cardiac surgeries. J Crit Care. 2017;40:171–7. doi: 10.1016/j.jcrc.2017.03.029. [DOI] [PubMed] [Google Scholar]
- 28.Gist KM, Goldstein SL, Wrona J, Alten JA, Basu RK, Cooper DS, et al. Kinetics of the cell cycle arrest biomarkers (TIMP-2*IGFBP-7) for prediction of acute kidney injury in infants after cardiac surgery. Pediatr Nephrol. 2017;32:1611–9. doi: 10.1007/s00467-017-3655-y. [DOI] [PubMed] [Google Scholar]
- 29.Murray BM, Paller MS, Ferris TF. Effect of cyclosporine administration on renal hemodynamics in conscious rats. Kidney Int. 1985;28:767–74. doi: 10.1038/ki.1985.196. [DOI] [PubMed] [Google Scholar]
- 30.Robert N, Wong GW, Wright JM. Effect of cyclosporine on blood pressure. Cochrane Database Syst Rev. 2010;1:CD007893. doi: 10.1002/14651858.CD007893.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Naesens M, Kuypers DR, Sarwal M. Calcineurin inhibitor nephrotoxicity. Clin J Am Soc Nephrol. 2009;4:481–508. doi: 10.2215/CJN.04800908. [DOI] [PubMed] [Google Scholar]
- 32.Klintmalm GB, Iwatsuki S, Starzl TE. Nephrotoxicity of cyclosporin A in liver and kidney transplant patients. Lancet. 1981;1:470–1. doi: 10.1016/s0140-6736(81)91851-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Myers BD, Ross J, Newton L, Luetscher J, Perlroth M. Cyclosporine-associated chronic nephropathy. N Engl J Med. 1984;311:699–705. doi: 10.1056/NEJM198409133111103. [DOI] [PubMed] [Google Scholar]
- 34.Klintmalm G, Bohman SO, Sundelin B, Wilczek H. Interstitial fibrosis in renal allografts after 12 to 46 months of cyclosporin treatment: beneficial effect of low doses in early post-transplantation period. Lancet. 1984;2:950–4. doi: 10.1016/s0140-6736(84)91166-8. [DOI] [PubMed] [Google Scholar]
- 35.Farnsworth A, Hall BM, Duggin GG, Horvath JS, Tiller DJ. Interstitial fibrosis in renal allografts in patients treated with cyclosporin. Lancet. 1984;2:1470–1. doi: 10.1016/s0140-6736(84)91661-1. [DOI] [PubMed] [Google Scholar]
- 36.Miller RP, Tadagavadi RK, Ramesh G, Reeves WB. Mechanisms of Cisplatin nephrotoxicity. Toxins (Basel) 2010;2:2490–518. doi: 10.3390/toxins2112490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Urakami Y, Okuda M, Masuda S, Saito H, Inui KI. Functional characteristics and membrane localization of rat multispecific organic cation transporters, OCT1 and OCT2, mediating tubular secretion of cationic drugs. J Pharmacol Exp Ther. 1998;287:800–5. [PubMed] [Google Scholar]
- 38.Lopez-Novoa JM, Quiros Y, Vicente L, Morales AI, Lopez-Hernandez FJ. New insights into the mechanism of aminoglycoside nephrotoxicity: an integrative point of view. Kidney Int. 2011;79:33–45. doi: 10.1038/ki.2010.337. [DOI] [PubMed] [Google Scholar]
- 39.El Mouedden M, Laurent G, Mingeot-Leclercq MP, Taper HS, Cumps J, Tulkens PM. Apoptosis in renal proximal tubules of rats treated with low doses of aminoglycosides. Antimicrob Agents Chemother. 2000;44:665–75. doi: 10.1128/aac.44.3.665-675.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Agarwal A, Dong Z, Harris R, Murray P, Parikh SM, Rosner MH, et al. Acute Dialysis Quality Initiative XIII Working Group. Cellular and Molecular Mechanisms of AKI. J Am Soc Nephrol. 2016;27:1288–99. doi: 10.1681/ASN.2015070740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Takaori K, Yanagita M. Insights into the Mechanisms of the Acute Kidney Injury-to-Chronic Kidney Disease Continuum. Nephron. 2016;134:172–6. doi: 10.1159/000448081. [DOI] [PubMed] [Google Scholar]
- 42.Havasi A, Borkan SC. Apoptosis and acute kidney injury. Kidney Int. 2011;80:29–40. doi: 10.1038/ki.2011.120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Linkermann A, Chen G, Dong G, Kunzendorf U, Krautwald S, Dong Z. Regulated cell death in AKI. J Am Soc Nephrol. 2014;25:2689–701. doi: 10.1681/ASN.2014030262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Andrade L, Rodrigues CE, Gomes SA, Noronha IL. Acute Kidney Injury as a Condition of Renal Senescence. Cell Transplant. 2018;27:739–53. doi: 10.1177/0963689717743512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Andrade L, Rodrigues CE, Gomes SA, Noronha IL. Acute Kidney Injury as a Condition of Renal Senescence. Cell Transplant. 2018;27:739–53. doi: 10.1177/0963689717743512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Jun JI, Lau LF. Cellular senescence controls fibrosis in wound healing. Aging (Albany NY) 2010;2:627–31. doi: 10.18632/aging.100201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ashkenazi A, Dixit VM. Death receptors: signaling and modulation. Science. 1998;281:1305–8. doi: 10.1126/science.281.5381.1305. [DOI] [PubMed] [Google Scholar]
- 48.Cunningham PN, Dyanov HM, Park P, Wang J, Newell KA, Quigg RJ. Acute renal failure in endotoxemia is caused by TNF acting directly on TNF receptor-1 in kidney. J Immunol. 2002;168:5817–23. doi: 10.4049/jimmunol.168.11.5817. [DOI] [PubMed] [Google Scholar]
- 49.Castaneda MP, Swiatecka-Urban A, Mitsnefes MM, Feuerstein D, Kaskel FJ, Tellis V, et al. Activation of mitochondrial apoptotic pathways in human renal allografts after ischemiareperfusion injury. Transplantation. 2003;76:50–4. doi: 10.1097/01.TP.0000069835.95442.9F. [DOI] [PubMed] [Google Scholar]
- 50.Sarhan M, von Mässenhausen A, Hugo C, Oberbauer R, Linkermann A. Immunological consequences of kidney cell death. Cell Death Dis. 2018;9:114. doi: 10.1038/s41419-017-0057-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Rabb H, Griffin MD, McKay DB, Swaminathan S, Pickkers P, Rosner MH, et al. Acute Dialysis Quality Initiative Consensus XIII Work Group. Inflammation in AKI: Current Understanding, Key Questions, and Knowledge Gaps. J Am Soc Nephrol. 2016;27:371–9. doi: 10.1681/ASN.2015030261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Lee S, Huen S, Nishio H, Nishio S, Lee HK, Choi BS, et al. Distinct macrophage phenotypes contribute to kidney injury and repair. J Am Soc Nephrol. 2011;22:317–26. doi: 10.1681/ASN.2009060615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Zhou D, Liu Y. Renal fibrosis in 2015: Understanding the mechanisms of kidney fibrosis. Nat Rev Nephrol. 2016;12:68–70. doi: 10.1038/nrneph.2015.215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Bellomo R, Ronco C, Kellum JA, Mehta RL, Palevsky P Acute Dialysis Quality Initiative workgroup. Acute renal failure - definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care. 2004;8:R204–12. doi: 10.1186/cc2872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Mehta RL, Kellum JA, Shah SV, Molitoris BA, Ronco C, Warnock DG, et al. Acute Kidney Injury Network. Acute Kidney Injury Network: report of an initiative to improve outcomes in acute kidney injury. Crit Care. 2007;11:R31. doi: 10.1186/cc5713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Barai S, Gambhir S, Prasad N, Sharma RK, Ora M. Functional renal reserve capacity in different stages of chronic kidney disease. Nephrology (Carlton) 2010;15:350–3. doi: 10.1111/j.1440-1797.2010.01291.x. [DOI] [PubMed] [Google Scholar]
- 57.Makris K, Spanou L. Acute Kidney Injury: Diagnostic Approaches and Controversies. Clin Biochem Rev. 2016;37:153–75. [PMC free article] [PubMed] [Google Scholar]
- 58.Baxmann AC, Ahmed MS, Marques NC, Menon VB, Pereira AB, Kirsztajn GM, et al. Influence of muscle mass and physical activity on serum and urinary creatinine and serum cystatin C. Clin J Am Soc Nephrol. 2008;3:348–54. doi: 10.2215/CJN.02870707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Bilancio G, Lombardi C, Pisot R, De Santo NG, Cavallo P, Cirillo M. Effects of bed-rest on urea and creatinine: correlation with changes in fat-free mass. PLoS One. 2014;9:e108805. doi: 10.1371/journal.pone.0108805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Ducharme MP, Smythe M, Strohs G. Drug-induced alterations in serum creatinine concentrations. Ann Pharmacother. 1993;27:622–33. doi: 10.1177/106002809302700518. [DOI] [PubMed] [Google Scholar]
- 61.Herget-Rosenthal S, Feldkamp T, Volbracht L, Kribben A. Measurement of urinary cystatin C by particle-enhanced nephelometric immunoassay: precision, interferences, stability and reference range. Ann Clin Biochem. 2004;41:111–8. doi: 10.1258/000456304322879980. [DOI] [PubMed] [Google Scholar]
- 62.Uchida K, Gotoh A. Measurement of cystatin-C and creatinine in urine. Clin Chim Acta. 2002;323:121–8. doi: 10.1016/s0009-8981(02)00177-8. [DOI] [PubMed] [Google Scholar]
- 63.Patel SS, Molnar MZ, Tayek JA, Ix JH, Noori N, Benner D, et al. Serum creatinine as a marker of muscle mass in chronic kidney disease: results of a cross-sectional study and review of literature. J Cachexia Sarcopenia Muscle. 2013;4:19–29. doi: 10.1007/s13539-012-0079-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Tangri N, Stevens LA, Schmid CH, Zhang YL, Beck GJ, Greene T, et al. Changes in dietary protein intake has no effect on serum cystatin C levels independent of the glomerular filtration rate. Kidney Int. 2011;79:471–7. doi: 10.1038/ki.2010.431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Abrahamson M, Olafsson I, Palsdottir A, Ulvsbäck M, Lundwall A, Jensson O, et al. Structure and expression of the human cystatin C gene. Biochem J. 1990;268:287–94. doi: 10.1042/bj2680287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Grubb A, Horio M, Hansson LO, Björk J, Nyman U, Flodin M, et al. Generation of a new cystatin C-based estimating equation for glomerular filtration rate by use of 7 assays standardized to the international calibrator. Clin Chem. 2014;60:974–86. doi: 10.1373/clinchem.2013.220707. [DOI] [PubMed] [Google Scholar]
- 67.Kyhse-Andersen J, Schmidt C, Nordin G, Andersson B, Nilsson-Ehle P, Lindström V, et al. Serum cystatin C, determined by a rapid, automated particle-enhanced turbidimetric method, is a better marker than serum creatinine for glomerular filtration rate. Clin Chem. 1994;40:1921–6. [PubMed] [Google Scholar]
- 68.Newman DJ, Thakkar H, Edwards RG, Wilkie M, White T, Grubb AO, et al. Serum cystatin C measured by automated immunoassay: a more sensitive marker of changes in GFR than serum creatinine. Kidney Int. 1995;47:312–8. doi: 10.1038/ki.1995.40. [DOI] [PubMed] [Google Scholar]
- 69.Dharnidharka VR, Kwon C, Stevens G. Serum cystatin C is superior to serum creatinine as a marker of kidney function: a meta-analysis. Am J Kidney Dis. 2002;40:221–6. doi: 10.1053/ajkd.2002.34487. [DOI] [PubMed] [Google Scholar]
- 70.Koyner JL, Bennett MR, Worcester EM, Ma Q, Raman J, Jeevanandam V, et al. Urinary cystatin C as an early biomarker of acute kidney injury following adult cardiothoracic surgery. Kidney Int. 2008;74:1059–69. doi: 10.1038/ki.2008.341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Conti M, Moutereau S, Zater M, Lallali K, Durrbach A, Manivet P, et al. Urinary cystatin C as a specific marker of tubular dysfunction. Clin Chem Lab Med. 2006;44:288–91. doi: 10.1515/CCLM.2006.050. [DOI] [PubMed] [Google Scholar]
- 72.Koyner JL, Vaidya VS, Bennett MR, Ma Q, Worcester E, Akhter SA, et al. Urinary biomarkers in the clinical prognosis and early detection of acute kidney injury. Clin J Am Soc Nephrol. 2010;5:2154–65. doi: 10.2215/CJN.00740110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Makris K, Nikolaki E, Nanopoulos K, Pirgakis KM, Maltezos CK. Measurement of cystatin C in human urine by particle-enhanced turbidimetric immunoassay on an automated biochemistry analyzer. Clin Biochem. 2013;46:1128–30. doi: 10.1016/j.clinbiochem.2013.05.072. [DOI] [PubMed] [Google Scholar]
- 74.Kachko A, Costafreda MI, Zubkova I, Jacques J, Takeda K, Wells F, et al. Determinants in the Ig Variable Domain of Human HAVCR1 (TIM-1) Are Required To Enhance Hepatitis C Virus Entry. J Virol. 2018;92:92. doi: 10.1128/JVI.01742-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Ichimura T, Asseldonk EJ, Humphreys BD, Gunaratnam L, Duffield JS, Bonventre JV. Kidney injury molecule-1 is a phosphatidylserine receptor that confers a phagocytic phenotype on epithelial cells. J Clin Invest. 2008;118:1657–68. doi: 10.1172/JCI34487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Ichimura T, Bonventre JV, Bailly V, Wei H, Hession CA, Cate RL, et al. Kidney injury molecule-1 (KIM-1), a putative epithelial cell adhesion molecule containing a novel immunoglobulin domain, is up-regulated in renal cells after injury. J Biol Chem. 1998;273:4135–42. doi: 10.1074/jbc.273.7.4135. [DOI] [PubMed] [Google Scholar]
- 77.Bank JR, van der Pol P, Vreeken D, Monge-Chaubo C, Bajema IM, Schlagwein N, et al. Kidney injury molecule-1 staining in renal allograft biopsies 10 days after transplantation is inversely correlated with functioning proximal tubular epithelial cells. Nephrol Dial Transplant. 2017;32:2132–41. doi: 10.1093/ndt/gfx286. [DOI] [PubMed] [Google Scholar]
- 78.van Timmeren MM, van den Heuvel MC, Bailly V, Bakker SJ, van Goor H, Stegeman CA. Tubular kidney injury molecule-1 (KIM-1) in human renal disease. J Pathol. 2007;212:209–17. doi: 10.1002/path.2175. [DOI] [PubMed] [Google Scholar]
- 79.Han WK, Bailly V, Abichandani R, Thadhani R, Bonventre JV. Kidney Injury Molecule-1 (KIM-1): a novel biomarker for human renal proximal tubule injury. Kidney Int. 2002;62:237–44. doi: 10.1046/j.1523-1755.2002.00433.x. [DOI] [PubMed] [Google Scholar]
- 80.Kjeldsen L, Cowland JB, Borregaard N. Human neutrophil gelatinase-associated lipocalin and homologous proteins in rat and mouse. Biochim Biophys Acta. 2000;1482:272–83. doi: 10.1016/s0167-4838(00)00152-7. [DOI] [PubMed] [Google Scholar]
- 81.Chakraborty S, Kaur S, Guha S, Batra SK. The multifaceted roles of neutrophil gelatinase associated lipocalin (NGAL) in inflammation and cancer. Biochim Biophys Acta. 2012;1826:129–69. doi: 10.1016/j.bbcan.2012.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Cowland JB, Borregaard N. Molecular characterization and pattern of tissue expression of the gene for neutrophil gelatinase-associated lipocalin from humans. Genomics. 1997;45:17–23. doi: 10.1006/geno.1997.4896. [DOI] [PubMed] [Google Scholar]
- 83.Kjeldsen L, Johnsen AH, Sengeløv H, Borregaard N. Isolation and primary structure of NGAL, a novel protein associated with human neutrophil gelatinase. J Biol Chem. 1993;268:10425–32. [PubMed] [Google Scholar]
- 84.Yan L, Borregaard N, Kjeldsen L, Moses MA. The high molecular weight urinary matrix metalloproteinase (MMP) activity is a complex of gelatinase B/MMP-9 and neutrophil gelatinase-associated lipocalin (NGAL). Modulation of MMP-9 activity by NGAL. J Biol Chem. 2001;276:37258–65. doi: 10.1074/jbc.M106089200. [DOI] [PubMed] [Google Scholar]
- 85.Paragas N, Kulkarni R, Werth M, Schmidt-Ott KM, Forster C, Deng R, et al. α-Intercalated cells defend the urinary system from bacterial infection. J Clin Invest. 2014;124:2963–76. doi: 10.1172/JCI71630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Mishra J, Ma Q, Prada A, Mitsnefes M, Zahedi K, Yang J, et al. Identification of neutrophil gelatinase-associated lipocalin as a novel early urinary biomarker for ischemic renal injury. J Am Soc Nephrol. 2003;14:2534–43. doi: 10.1097/01.asn.0000088027.54400.c6. [DOI] [PubMed] [Google Scholar]
- 87.Mori K, Lee HT, Rapoport D, Drexler IR, Foster K, Yang J, et al. Endocytic delivery of lipocalin-siderophore-iron complex rescues the kidney from ischemia-reperfusion injury. J Clin Invest. 2005;115:610–21. doi: 10.1172/JCI23056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Han M, Li Y, Liu M, Li Y, Cong B. Renal neutrophil gelatinase associated lipocalin expression in lipopolysaccharide-induced acute kidney injury in the rat. BMC Nephrol. 2012;13:25. doi: 10.1186/1471-2369-13-25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Zhang YL, Qiao SK, Wang RY, Guo XN. NGAL attenuates renal ischemia/reperfusion injury through autophagy activation and apoptosis inhibition in rats. Chem Biol Interact. 2018;289:40–6. doi: 10.1016/j.cbi.2018.04.018. [DOI] [PubMed] [Google Scholar]
- 90.Bennett M, Dent CL, Ma Q, Dastrala S, Grenier F, Workman R, et al. Urine NGAL predicts severity of acute kidney injury after cardiac surgery: a prospective study. Clin J Am Soc Nephrol. 2008;3:665–73. doi: 10.2215/CJN.04010907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Parikh CR, Abraham E, Ancukiewicz M, Edelstein CL. Urine IL-18 is an early diagnostic marker for acute kidney injury and predicts mortality in the intensive care unit. J Am Soc Nephrol. 2005;16:3046–52. doi: 10.1681/ASN.2005030236. [DOI] [PubMed] [Google Scholar]
- 92.Parikh CR, Jani A, Melnikov VY, Faubel S, Edelstein CL. Urinary interleukin-18 is a marker of human acute tubular necrosis. Am J Kidney Dis. 2004;43:405–14. doi: 10.1053/j.ajkd.2003.10.040. [DOI] [PubMed] [Google Scholar]
- 93.Parikh CR, Mishra J, Thiessen-Philbrook H, Dursun B, Ma Q, Kelly C, et al. Urinary IL-18 is an early predictive biomarker of acute kidney injury after cardiac surgery. Kidney Int. 2006;70:199–203. doi: 10.1038/sj.ki.5001527. [DOI] [PubMed] [Google Scholar]
- 94.Fagerberg L, Hallström BM, Oksvold P, Kampf C, Djureinovic D, Odeberg J, et al. Analysis of the human tissue-specific expression by genome-wide integration of transcriptomics and antibody-based proteomics. Mol Cell Proteomics. 2014;13:397–406. doi: 10.1074/mcp.M113.035600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Emlet DR, Pastor-Soler N, Marciszyn A, Wen X, Gomez H, Humphries WH, 4th, et al. Insulin-like growth factor binding protein 7 and tissue inhibitor of metalloproteinases-2: differential expression and secretion in human kidney tubule cells. Am J Physiol Renal Physiol. 2017;312:F284–96. doi: 10.1152/ajprenal.00271.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Brew K, Nagase H. The tissue inhibitors of metalloproteinases (TIMPs): an ancient family with structural and functional diversity. Biochim Biophys Acta. 2010;1803:55–71. doi: 10.1016/j.bbamcr.2010.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Seo DW, Li H, Qu CK, Oh J, Kim YS, Diaz T, et al. Shp-1 mediates the antiproliferative activity of tissue inhibitor of metalloproteinase-2 in human microvascular endothelial cells. J Biol Chem. 2006;281:3711–21. doi: 10.1074/jbc.M509932200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Wajapeyee N, Serra RW, Zhu X, Mahalingam M, Green MR. Oncogenic BRAF induces senescence and apoptosis through pathways mediated by the secreted protein IGFBP7. Cell. 2008;132:363–74. doi: 10.1016/j.cell.2007.12.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Verhagen HJ, de Leeuw DC, Roemer MG, Denkers F, Pouwels W, Rutten A, et al. IGFBP7 induces apoptosis of acute myeloid leukemia cells and synergizes with chemotherapy in suppression of leukemia cell survival. Cell Death Dis. 2014;5:e1300. doi: 10.1038/cddis.2014.268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Yang J, Lim SY, Kim MG, Jung CW, Cho WY, Jo SK. Urinary Tissue Inhibitor of Metalloproteinase and Insulin-like Growth Factor-7 as Early Biomarkers of Delayed Graft Function After Kidney Transplantation. Transplant Proc. 2017;49:2050–4. doi: 10.1016/j.transproceed.2017.09.023. [DOI] [PubMed] [Google Scholar]
- 101.Ławicki S, Zajkowska M, Głażewska EK, Będkowska GE, Szmitkowski M. Plasma levels and diagnostic utility of VEGF, MMP-2 and TIMP-2 in the diagnostics of breast cancer patients. Biomarkers. 2017;22:157–64. doi: 10.1080/1354750X.2016.1252955. [DOI] [PubMed] [Google Scholar]
- 102.Gültekin GD, Çabuk B, Vural Ç, Ceylan S. Matrix metalloproteinase-9 and tissue inhibitor of matrix metalloproteinase-2: Prognostic biological markers in invasive prolactinomas. J Clin Neurosci. 2015;22:1282–7. doi: 10.1016/j.jocn.2015.02.021. [DOI] [PubMed] [Google Scholar]
- 103.Benassi MS, Pazzaglia L, Novello C, Quattrini I, Pollino S, Magagnoli G, et al. Tissue and serum IGFBP7 protein as biomarker in high-grade soft tissue sarcoma. Am J Cancer Res. 2015;5:3446–54. [PMC free article] [PubMed] [Google Scholar]
- 104.Kashani K, Al-Khafaji A, Ardiles T, Artigas A, Bagshaw SM, Bell M, et al. Discovery and validation of cell cycle arrest biomarkers in human acute kidney injury. Crit Care. 2013;17:R25. doi: 10.1186/cc12503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Gunnerson KJ, Shaw AD, Chawla LS, Bihorac A, Al-Khafaji A, Kashani K, et al. Sapphire Topaz investigators. TIMP2•IGFBP7 biomarker panel accurately predicts acute kidney injury in high-risk surgical patients. J Trauma Acute Care Surg. 2016;80:243–9. doi: 10.1097/TA.0000000000000912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Kamijo A, Kimura K, Sugaya T, Yamanouchi M, Hikawa A, Hirano N, et al. Urinary fatty acid-binding protein as a new clinical marker of the progression of chronic renal disease. J Lab Clin Med. 2004;143:23–30. doi: 10.1016/j.lab.2003.08.001. [DOI] [PubMed] [Google Scholar]
- 107.Yamamoto T, Noiri E, Ono Y, Doi K, Negishi K, Kamijo A, et al. Renal L-type fatty acid—binding protein in acute ischemic injury. J Am Soc Nephrol. 2007;18:2894–902. doi: 10.1681/ASN.2007010097. [DOI] [PubMed] [Google Scholar]
- 108.Price RG. The role of NAG (N-acetyl-beta-D-glucosaminidase) in the diagnosis of kidney disease including the monitoring of nephrotoxicity. Clin Nephrol. 1992;38(Suppl 1):S14–9. [PubMed] [Google Scholar]
- 109.Le Hir M, Dubach UC, Schmidt U. Quantitative distribution of lysosomal hydrolases in the rat nephron. Histochemistry. 1979;63:245–51. doi: 10.1007/BF00644546. [DOI] [PubMed] [Google Scholar]
- 110.Vaidya VS, Waikar SS, Ferguson MA, Collings FB, Sunderland K, Gioules C, et al. Urinary biomarkers for sensitive and specific detection of acute kidney injury in humans. Clin Transl Sci. 2008;1:200–8. doi: 10.1111/j.1752-8062.2008.00053.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Katagiri D, Doi K, Honda K, Negishi K, Fujita T, Hisagi M, et al. Combination of two urinary biomarkers predicts acute kidney injury after adult cardiac surgery. Ann Thorac Surg. 2012;93:577–83. doi: 10.1016/j.athoracsur.2011.10.048. [DOI] [PubMed] [Google Scholar]
- 112.Bouvet BR, Paparella CV, Arriaga SM, Monje AL, Amarilla AM, Almará AM. Evaluation of urinary N-acetyl-beta-D-glucosaminidase as a marker of early renal damage in patients with type 2 diabetes mellitus. Arq Bras Endocrinol Metabol. 2014;58:798–801. doi: 10.1590/0004-2730000003010. [DOI] [PubMed] [Google Scholar]
- 113.Sheira G, Noreldin N, Tamer A, Saad M. Urinary biomarker N-acetyl-β-D-glucosaminidase can predict severity of renal damage in diabetic nephropathy. J Diabetes Metab Disord. 2015;14:4. doi: 10.1186/s40200-015-0133-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Johnson AC, Zager RA. Mechanisms Underlying Increased TIMP2 and IGFBP7 Urinary Excretion in Experimental AKI. J Am Soc Nephrol. 2018;29:2157–67. doi: 10.1681/ASN.2018030265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Nejat M, Hill JV, Pickering JW, Edelstein CL, Devarajan P, Endre ZH. Albuminuria increases cystatin C excretion: implications for urinary biomarkers. Nephrol Dial Transplant. 2012;27(Suppl 3):iii96–103. doi: 10.1093/ndt/gfr222. [DOI] [PubMed] [Google Scholar]
- 116.Christensen EI, Gburek J. Protein reabsorption in renal proximal tubule-function and dysfunction in kidney pathophysiology. Pediatr Nephrol. 2004;19:714–21. doi: 10.1007/s00467-004-1494-0. [DOI] [PubMed] [Google Scholar]
- 117.Wu AH, Christenson RH, Greene DN, Jaffe AS, Kavsak PA, Ordonez-Llanos J, et al. Clinical Laboratory Practice Recommendations for the Use of Cardiac Troponin in Acute Coronary Syndrome: Expert Opinion from the Academy of the American Association for Clinical Chemistry and the Task Force on Clinical Applications of Cardiac Bio-Markers of the International Federation of Clinical Chemistry and Laboratory Medicine. Clin Chem. 2018;64:645–55. doi: 10.1373/clinchem.2017.277186. [DOI] [PubMed] [Google Scholar]
- 118.Hsu CY, Ordoñez JD, Chertow GM, Fan D, McCulloch CE, Go AS. The risk of acute renal failure in patients with chronic kidney disease. Kidney Int. 2008;74:101–7. doi: 10.1038/ki.2008.107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Monaghan PJ, Robinson S, Rajdl D, Bossuyt PM, Sandberg S, St John A, et al. Practical guide for identifying unmet clinical needs for biomarkers. EJIFCC. 2018;29:129–37. [PMC free article] [PubMed] [Google Scholar]
- 120.Billings FT, 4th, Shaw AD. Clinical trial endpoints in acute kidney injury. Nephron Clin Pract. 2014;127:89–93. doi: 10.1159/000363725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Palevsky PM, Molitoris BA, Okusa MD, Levin A, Waikar SS, Wald R, et al. Design of clinical trials in acute kidney injury: report from an NIDDK workshop on trial methodology. Clin J Am Soc Nephrol. 2012;7:844–50. doi: 10.2215/CJN.12791211. [DOI] [PubMed] [Google Scholar]
- 122.Liu C, Lu X, Mao Z, Kang H, Liu H, Pan L, et al. The diagnostic accuracy of urinary [TIMP-2]·[IGFBP7] for acute kidney injury in adults: A PRISMA-compliant meta-analysis. Medicine (Baltimore) 2017;96:e7484. doi: 10.1097/MD.0000000000007484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Su Y, Gong Z, Wu Y, Tian Y, Liao X. Diagnostic Value of Urine Tissue Inhibitor of Metalloproteinase-2 and Insulin-Like Growth Factor-Binding Protein 7 for Acute Kidney Injury: A Meta-Analysis. PLoS One. 2017;12:e0170214. doi: 10.1371/journal.pone.0170214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Shao X, Tian L, Xu W, Zhang Z, Wang C, Qi C, et al. Diagnostic value of urinary kidney injury molecule 1 for acute kidney injury: a meta-analysis. PLoS One. 2014;9:e84131. doi: 10.1371/journal.pone.0084131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Hoste EA, McCullough PA, Kashani K, Chawla LS, Joannidis M, Shaw AD, et al. Sapphire Investigators. Derivation and validation of cutoffs for clinical use of cell cycle arrest biomarkers. Nephrol Dial Transplant. 2014;29:2054–61. doi: 10.1093/ndt/gfu292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Bihorac A, Chawla LS, Shaw AD, Al-Khafaji A, Davison DL, Demuth GE, et al. Validation of cell-cycle arrest biomarkers for acute kidney injury using clinical adjudication. Am J Respir Crit Care Med. 2014;189:932–9. doi: 10.1164/rccm.201401-0077OC. [DOI] [PubMed] [Google Scholar]
- 127.Kimmel M, Shi J, Latus J, Wasser C, Kitterer D, Braun N, et al. Association of Renal Stress/Damage and Filtration Biomarkers with Subsequent AKI during Hospitalization among Patients Presenting to the Emergency Department. Clin J Am Soc Nephrol. 2016;11:938–46. doi: 10.2215/CJN.10551015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Kimmel M, Shi J, Wasser C, Biegger D, Alscher MD, Schanz MB. Urinary [TIMP-2]·[IGFBP7] - Novel Biomarkers to Predict Acute Kidney Injury. Am J Nephrol. 2016;43:375–82. doi: 10.1159/000446451. [DOI] [PubMed] [Google Scholar]
- 129.Endre ZH, Pickering JW, Walker RJ, Devarajan P, Edelstein CL, Bonventre JV, et al. Improved performance of urinary biomarkers of acute kidney injury in the critically ill by stratification for injury duration and baseline renal function. Kidney Int. 2011;79:1119–30. doi: 10.1038/ki.2010.555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Dong L, Ma Q, Bennett M, Devarajan P. Urinary biomarkers of cell cycle arrest are delayed predictors of acute kidney injury after pediatric cardiopulmonary bypass. Pediatr Nephrol. 2017;32:2351–60. doi: 10.1007/s00467-017-3748-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Finge T, Bertran S, Roger C, Candela D, Pereira B, Scott C, et al. Interest of Urinary [TIMP-2] × [IGFBP-7] for Predicting the Occurrence of Acute Kidney Injury After Cardiac Surgery: A Gray Zone Approach. Anesth Analg. 2017;125:762–9. doi: 10.1213/ANE.0000000000002116. [DOI] [PubMed] [Google Scholar]
- 132.Fraser CG. Reference change values. Clin Chem Lab Med. 2011;50:807–12. doi: 10.1515/CCLM.2011.733. [DOI] [PubMed] [Google Scholar]
- 133.Siest G, Henny J, Gräsbeck R, Wilding P, Petitclerc C, Queraltó JM, et al. The theory of reference values: an unfinished symphony. Clin Chem Lab Med. 2013;51:47–64. doi: 10.1515/cclm-2012-0682. [DOI] [PubMed] [Google Scholar]
- 134.Waikar SS, Betensky RA, Emerson SC, Bonventre JV. Imperfect gold standards for kidney injury biomarker evaluation. J Am Soc Nephrol. 2012;23:13–21. doi: 10.1681/ASN.2010111124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Bove T, Zangrillo A, Guarracino F, Alvaro G, Persi B, Maglioni E, et al. Effect of fenoldopam on use of renal replacement therapy among patients with acute kidney injury after cardiac surgery: a randomized clinical trial. JAMA. 2014;312:2244–53. doi: 10.1001/jama.2014.13573. [DOI] [PubMed] [Google Scholar]
- 136.Nadim MK, Forni LG, Bihorac A, Hobson C, Koyner JL, Shaw A, et al. Cardiac and Vascular Surgery-Associated Acute Kidney Injury: The 20th International Consensus Conference of the ADQI (Acute Disease Quality Initiative) Group. J Am Heart Assoc. 2018;7 doi: 10.1161/JAHA.118.008834. pii: e008834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Dardashti A, Ederoth P, Algotsson L, Brondén B, Grins E, Larsson M, et al. Erythropoietin and protection of renal function in cardiac surgery (the EPRICS Trial) Anesthesiology. 2014;121:582–90. doi: 10.1097/ALN.0000000000000321. [DOI] [PubMed] [Google Scholar]
- 138.Zacharias M, Mugawar M, Herbison GP, Walker RJ, Hovhannisyan K, Sivalingam P, et al. Interventions for protecting renal function in the perioperative period. Cochrane Database Syst Rev. 2013;9:CD003590. doi: 10.1002/14651858.CD003590.pub4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Ichai C, Vinsonneau C, Souweine B, Armando F, Canet E, Clec’h C, et al. Société française d’anesthésie et de réanimation (Sfar); Société de réanimation de langue française (SRLF); Groupe francophone de réanimation et urgences pédiatriques (GFRUP); Société française de néphrologie (SFN) Acute kidney injury in the perioperative period and in intensive care units (excluding renal replacement therapies) Ann Intensive Care. 2016;6:48. doi: 10.1186/s13613-016-0145-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Coleman MD, Shaefi S, Sladen RN. Preventing acute kidney injury after cardiac surgery. Curr Opin Anaesthesiol. 2011;24:70–6. doi: 10.1097/ACO.0b013e3283422ebc. [DOI] [PubMed] [Google Scholar]
- 141.Meersch M, Schmidt C, Hoffmeier A, Van Aken H, Wempe C, Gerss J, et al. Prevention of cardiac surgery-associated AKI by implementing the KDIGO guidelines in high risk patients identified by biomarkers: the PrevAKI randomized controlled trial. Intensive Care Med. 2017;43:1551–61. doi: 10.1007/s00134-016-4670-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Schanz M, Wasser C, Allgaeuer S, Schricker S, Dippon J, Alscher MD, et al. Urinary [TIMP-2]·[IGFBP7]-guided randomized controlled intervention trial to prevent acute kidney injury in the emergency department. Nephrol Dial Transplant. 2018 doi: 10.1093/ndt/gfy186. (In press) [DOI] [PubMed] [Google Scholar]
- 143.Westgard QC. Desirable Biological Variation Database specifications. [Accessed 14 April 2019]. http://www.westgard.com/biodatabase1.com.
- 144.Kallner A, McQueen M, Heuck C. The Stockholm Consensus Conference on quality specifications in laboratory medicine, 25–26 April 1999. Scand J Clin Lab Invest. 1999;59:475–6. doi: 10.1080/00365519950185175. [DOI] [PubMed] [Google Scholar]
- 145.Sandberg S, Fraser CG, Horvath AR, Jansen R, Jones G, Oosterhuis W, et al. Defining analytical performance specifications: Consensus Statement from the 1st Strategic Conference of the European Federation of Clinical Chemistry and Laboratory Medicine. Clin Chem Lab Med. 2015;53:833–5. doi: 10.1515/cclm-2015-0067. [DOI] [PubMed] [Google Scholar]
- 146.Horvath AR, Bossuyt PM, Sandberg S, John AS, Monaghan PJ, Verhagen-Kamerbeek WD, et al. Test Evaluation Working Group of the European Federation of Clinical Chemistry and Laboratory Medicine. Setting analytical performance specifications based on outcome studies - is it possible? Clin Chem Lab Med. 2015;53:841–8. doi: 10.1515/cclm-2015-0214. [DOI] [PubMed] [Google Scholar]
- 147.Aarsand AK, Røraas T, Fernandez-Calle P, Ricos C, Díaz-Garzón J, Jonker N, et al. European Federation of Clinical Chemistry and Laboratory Medicine Working Group on Biological Variation and Task and Finish Group for the Biological Variation Database. The Biological Variation Data Critical Appraisal Checklist: A Standard for Evaluating Studies on Biological Variation. Clin Chem. 2018;64:501–14. doi: 10.1373/clinchem.2017.281808. [DOI] [PubMed] [Google Scholar]
- 148.Zhang X, Gibson B, Jr, Mori R, Snow-Lisy D, Yamaguchi Y, Campbell SC, et al. Analytical and biological validation of a multiplex immunoassay for acute kidney injury biomarkers. Clin Chim Acta. 2013;415:88–93. doi: 10.1016/j.cca.2012.09.022. [DOI] [PubMed] [Google Scholar]
- 149.Pennemans V, Rigo JM, Faes C, Reynders C, Penders J, Swennen Q. Establishment of reference values for novel urinary biomarkers for renal damage in the healthy population: are age and gender an issue? Clin Chem Lab Med. 2013;51:1795–802. doi: 10.1515/cclm-2013-0157. [DOI] [PubMed] [Google Scholar]
- 150.Carter JL, Parker CT, Stevens PE, Eaglestone G, Knight S, Farmer CK, et al. Biological Variation of Plasma and Urinary Markers of Acute Kidney Injury in Patients with Chronic Kidney Disease. Clin Chem. 2016;62:876–83. doi: 10.1373/clinchem.2015.250993. [DOI] [PubMed] [Google Scholar]
- 151.Grenier FC, Ali S, Syed H, Workman R, Martens F, Liao M, et al. Evaluation of the ARCHITECT urine NGAL assay: assay performance, specimen handling requirements and biological variability. Clin Biochem. 2010;43:615–20. doi: 10.1016/j.clinbiochem.2009.12.008. [DOI] [PubMed] [Google Scholar]
- 152.Delanaye P, Rozet E, Krzesinski JM, Cavalier E. Urinary NGAL measurement: biological variation and ratio to creatinine. Clin Chim Acta. 2011;412:390. doi: 10.1016/j.cca.2010.10.011. [DOI] [PubMed] [Google Scholar]
- 153.Cruz DN, Virzì GM, Brocca A, Ronco C, Giavarina D. A comparison of three commercial platforms for urinary NGAL in critically ill adults. Clin Chem Lab Med. 2016;54:353–62. doi: 10.1515/cclm-2015-0464. [DOI] [PubMed] [Google Scholar]
- 154.Lippi G, Aloe R, Storelli A, Cervellin G, Trenti T. Evaluation of NGAL Test™, a fully-automated neutrophil gelatinase-associated lipocalin (NGAL) immunoassay on Beckman Coulter AU 5822. Clin Chem Lab Med. 2011;50:1581–4. doi: 10.1515/cclm.2011.839. [DOI] [PubMed] [Google Scholar]
- 155.Makris K, Stefani D, Makri E, Panagou I, Lagiou M, Sarli A, et al. Evaluation of a particle enhanced turbidimetric assay for the measurement of neutrophil gelatinase-associated lipocalin in plasma and urine on Architect-8000: Analytical performance and establishment of reference values. Clin Biochem. 2015;48:1291–7. doi: 10.1016/j.clinbiochem.2015.08.003. [DOI] [PubMed] [Google Scholar]
- 156.Ruhaak LR, Romijn FP, Smit NP, van der Laarse A, Pieterse MM, de Maat MP, et al. Detecting molecular forms of antithrombin by LC-MRM-MS: defining the measurands. Clin Chem Lab Med. 2018;56:1704–14. doi: 10.1515/cclm-2017-1111. [DOI] [PubMed] [Google Scholar]
- 157.Hoofnagle AN, Wener MH. The fundamental flaws of immunoassays and potential solutions using tandem mass spectrometry. J Immunol Methods. 2009;347:3–11. doi: 10.1016/j.jim.2009.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.van den Broek I, Romijn FP, Nouta J, van der Laarse A, Drijfhout JW, Smit NP, et al. Automated Multiplex LC-MS/MS Assay for Quantifying Serum Apolipoproteins A-I, B, C-I, C-II, C-III, and E with Qualitative Apolipoprotein E Phenotyping. Clin Chem. 2016;62:188–97. doi: 10.1373/clinchem.2015.246702. [DOI] [PubMed] [Google Scholar]
- 159.Ruhaak LR, Smit NP, Suchiman HE, Pieterse MM, Romijn FP, Beekman M, et al. MS-based proteomics: a metrological sound and robust alternative for apolipoprotein E phenotyping in a multiplexed test. Clin Chem Lab Med. 2019;57:e102–4. doi: 10.1515/cclm-2018-0782. [DOI] [PubMed] [Google Scholar]
- 160.Ruhaak LR, Smit NP, Romijn FP, Pieterse MM, van der Laarse A, van der Burgt YE, et al. Robust and Accurate 2-Year Performance of a Quantitative Mass Spectrometry-Based Apolipoprotein Test in a Clinical Chemistry Laboratory. Clin Chem. 2018;64:747–9. doi: 10.1373/clinchem.2017.285098. [DOI] [PubMed] [Google Scholar]
- 161.Hahn RG, Zdolsek J. Nephrocheck® results should be corrected for dilution. Acta Anaesthesiol Scand. 2017;61:261–2. doi: 10.1111/aas.12836. [DOI] [PubMed] [Google Scholar]
- 162.Middleton DR, Watts MJ, Lark RM, Milne CJ, Polya DA. Assessing urinary flow rate, creatinine, osmolality and other hydration adjustment methods for urinary biomonitoring using NHANES arsenic, iodine, lead and cadmium data. Environ Health. 2016;15:68. doi: 10.1186/s12940-016-0152-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Helmersson-Karlqvist J, Arnlöv J, Larsson A. Day-to-day variation of urinary NGAL and rational for creatinine correction. Clin Biochem. 2013;46:70–2. doi: 10.1016/j.clinbiochem.2012.09.022. [DOI] [PubMed] [Google Scholar]
- 164.Chetwynd AJ, Abdul-Sada A, Holt SG, Hill EM. Use of a pre-analysis osmolality normalisation method to correct for variable urine concentrations and for improved metabolomic analyses. J Chromatogr A. 2016;1431:103–10. doi: 10.1016/j.chroma.2015.12.056. [DOI] [PubMed] [Google Scholar]
- 165.Vogl FC, Mehrl S, Heizinger L, Schlecht I, Zacharias HU, Ellmann L, et al. GCKD Study Investigators. Evaluation of dilution and normalization strategies to correct for urinary output in HPLC-HRTOFMS metabolomics. Anal Bioanal Chem. 2016;408:8483–93. doi: 10.1007/s00216-016-9974-1. [DOI] [PubMed] [Google Scholar]
- 166.Rohr UP, Binder C, Dieterle T, Giusti F, Messina CG, Toerien E, et al. The Value of In Vitro Diagnostic Testing in Medical Practice: A Status Report. PLoS One. 2016;11:e0149856. doi: 10.1371/journal.pone.0149856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Sikaris KA. Enhancing the Clinical Value of Medical Laboratory Testing. Clin Biochem Rev. 2017;38:107–14. [PMC free article] [PubMed] [Google Scholar]
- 168.Binnenmars SH, Hijmans RS, Navis G, de Borst MH. Biomarkers of Renal Function: Towards Clinical Actionability. Clin Pharmacol Ther. 2017;102:481–92. doi: 10.1002/cpt.765. [DOI] [PubMed] [Google Scholar]
- 169.Horvath AR, Lord SJ, StJohn A, Sandberg S, Cobbaert CM, Lorenz S, et al. Test Evaluation Working Group of the European Federation of Clinical Chemistry Laboratory Medicine. From biomarkers to medical tests: the changing landscape of test evaluation. Clin Chim Acta. 2014;427:49–57. doi: 10.1016/j.cca.2013.09.018. [DOI] [PubMed] [Google Scholar]
- 170.Vijayan A, Faubel S, Askenazi DJ, Cerda J, Fissell WH, Heung M, et al. American Society of Nephrology Acute Kidney Injury Advisory Group. Clinical Use of the Urine Biomarker [TIMP-2] × [IGFBP7] for Acute Kidney Injury Risk Assessment. Am J Kidney Dis. 2016;68:19–28. doi: 10.1053/j.ajkd.2015.12.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.U.S. Food and Drug Administration. Evaluation of Automatic Class III Designation for NephroCheck® Test System Decision Summary. [Accessed 4 April 2019]. http://www.accessdata.fda.gov/cdrh_docs/reviews/den130031.pdf.
- 172.Kashani K, Cheungpasitporn W, Ronco C. Biomarkers of acute kidney injury: the pathway from discovery to clinical adoption. Clin Chem Lab Med. 2017;55:1074–89. doi: 10.1515/cclm-2016-0973. [DOI] [PubMed] [Google Scholar]
- 173.Fan W, Ankawi G, Zhang J, Digvijay K, Giavarina D, Yin Y, et al. Current understanding and future directions in the application of TIMP-2 and IGFBP7 in AKI clinical practice. Clin Chem Lab Med. 2019;57:567–76. doi: 10.1515/cclm-2018-0776. [DOI] [PubMed] [Google Scholar]
- 174.Gameiro J, Agapito Fonseca J, Jorge S, Lopes JA. Acute Kidney Injury Definition and Diagnosis: A Narrative Review. J Clin Med. 2018;7:7. doi: 10.3390/jcm7100307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Heung M, Ortega LM, Chawla LS, Wunderink RG, Self WH, Koyner JL, et al. Sapphire and Topaz Investigators. Common chronic conditions do not affect performance of cell cycle arrest biomarkers for risk stratification of acute kidney injury. Nephrol Dial Transplant. 2016;31:1633–40. doi: 10.1093/ndt/gfw241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Bank JR, Ruhaak R, Soonawala D, Mayboroda O, Romijn FP, van Kooten C, et al. Urinary TIMP-2 predicts the presence and duration of delayed graft function in donation after circulatory death kidney transplant recipients. Transplantation. 2019;103:1014–23. doi: 10.1097/TP.0000000000002472. [DOI] [PubMed] [Google Scholar]
- 177.Uettwiller-Geiger DL, Vijayendran R, Kellum JA, Fitzgerald RL. Analytical characteristics of a biomarker-based risk assessment test for acute kidney injury (AKI) Clin Chim Acta. 2016;455:93–8. doi: 10.1016/j.cca.2016.01.012. [DOI] [PubMed] [Google Scholar]